p53 Antibodies as a Diagnostic Marker for Cancer: A Meta-Analysis
Abstract
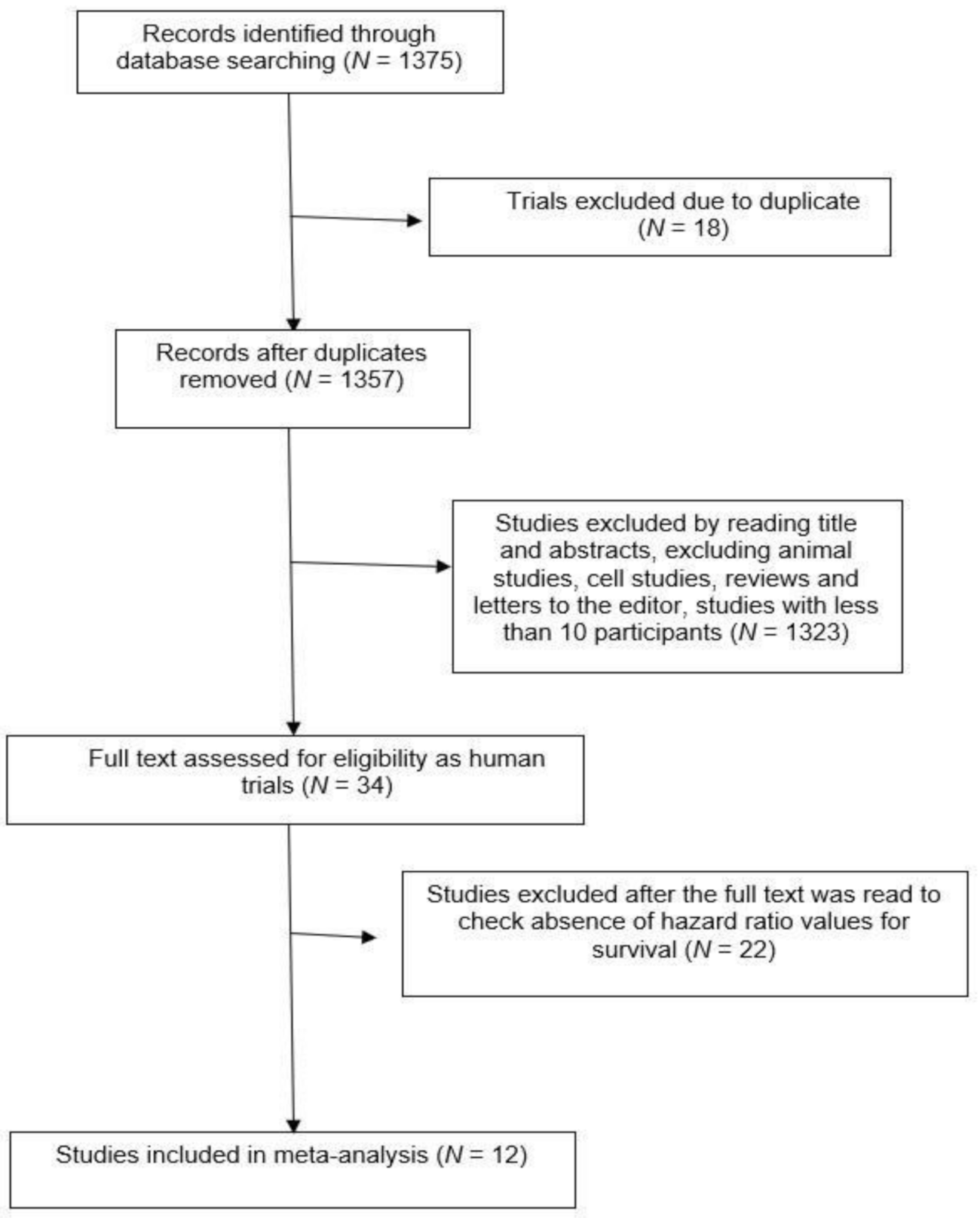
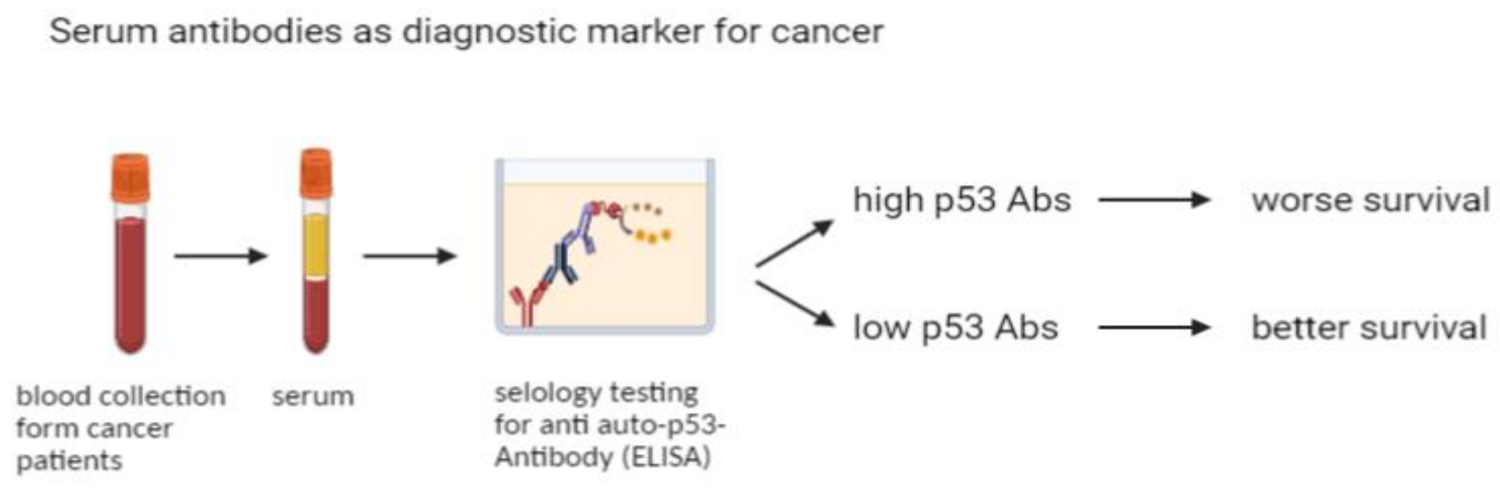
1. Introduction
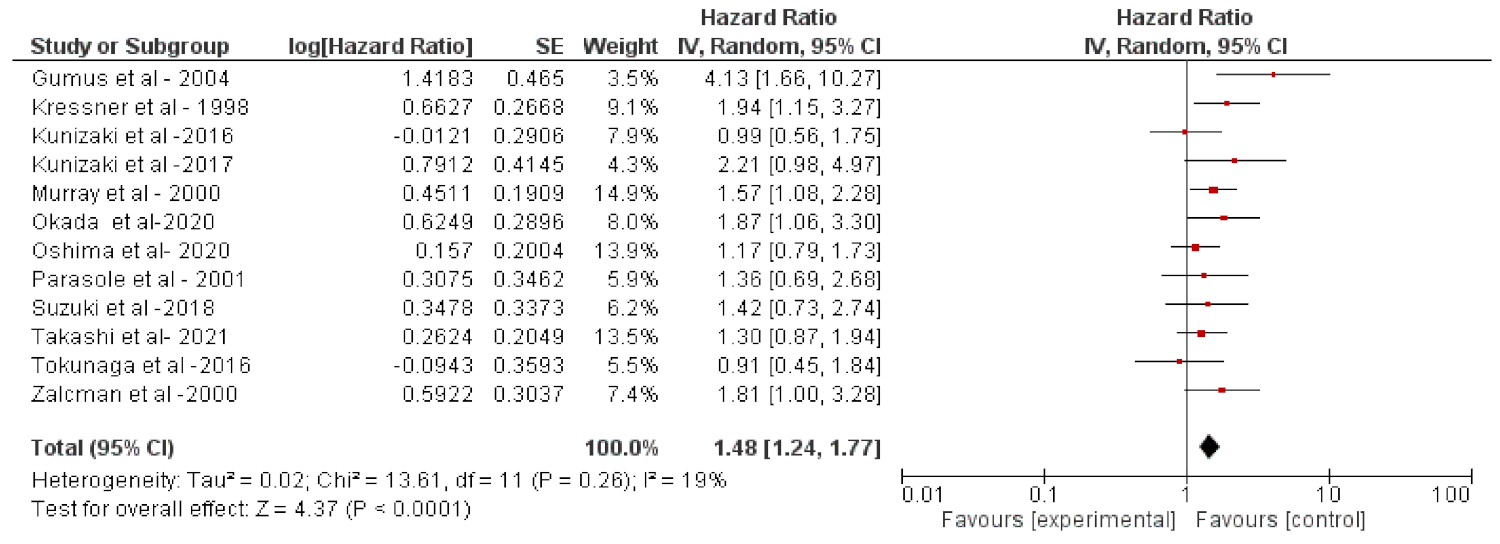
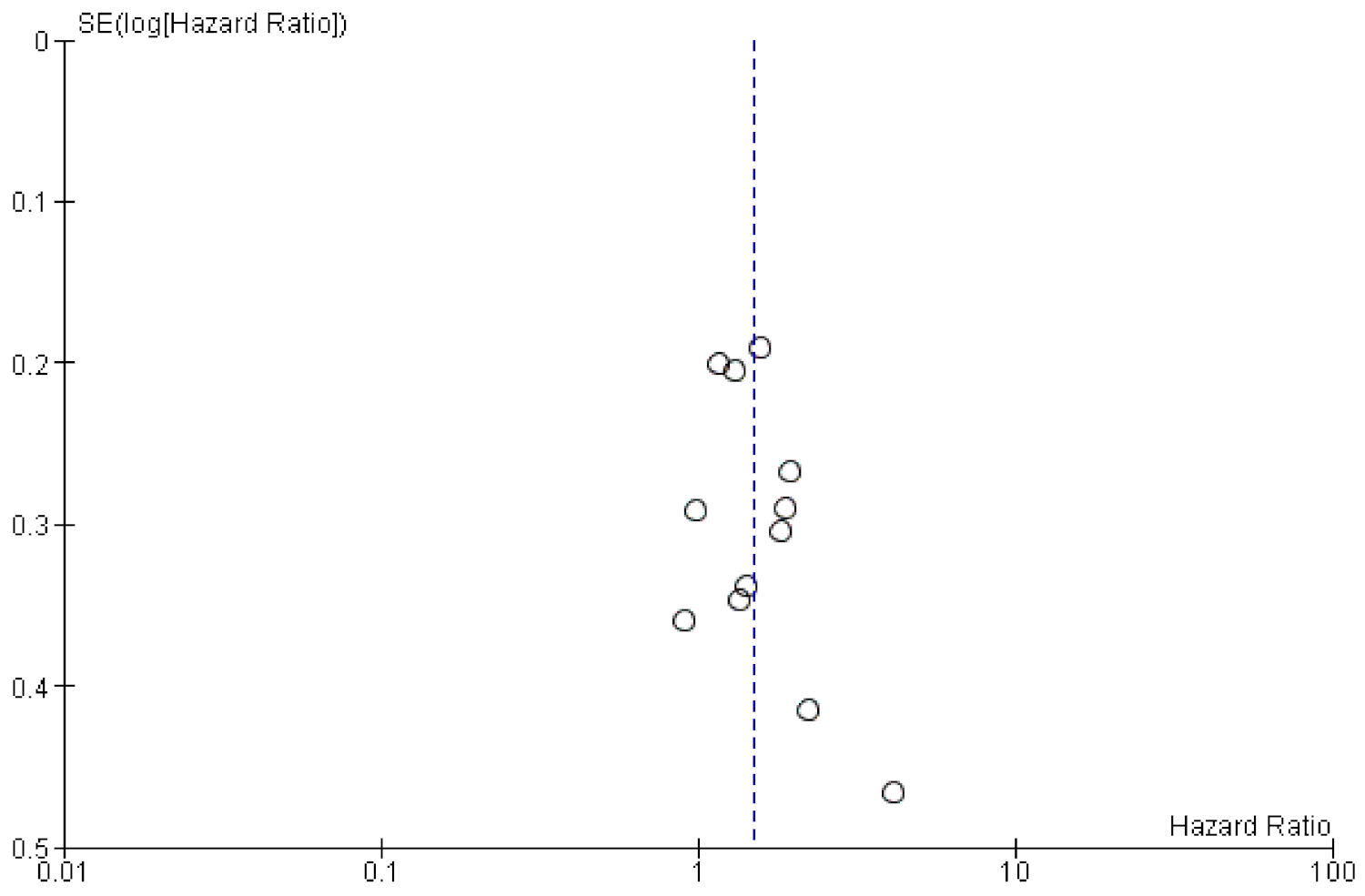
2. Results
3. Discussion
4. Materials and Methods
5. Conclusions
6. Competing Interests
Author Contributions
Funding
Institutional Review Board Statement and Ethics Approval
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Hollstein, M.; Sidransky, D.; Vogelstein, B.; Harris, C.C. p53 Mutations in Human Cancers. Science 1991, 253, 49–53. [Google Scholar] [CrossRef]
- Kandoth, C.; McLellan, M.D.; Vandin, F.; Ye, K.; Niu, B.; Lu, C.; Xie, M.; Zhang, Q.; McMichael, J.F.; Wyczalkowski, M.A.; et al. Mutational Landscape and Significance across 12 Major Cancer Types. Nature 2013, 502, 333–339. [Google Scholar] [CrossRef]
- Juven, T.; Barak, Y.; Zauberman, A.; George, D.L.; Oren, M. Wild Type p53 Can Mediate Sequence-Specific Transactivation of an Internal Promoter within the mdm2 Gene. Oncogene 1993, 8, 3411–3416. [Google Scholar]
- Wu, X.; Bayle, J.H.; Olson, D.; Levine, A.J. The p53-Mdm-2 Autoregulatory Feedback Loop. Genes Dev. 1993, 7, 1126–1132. [Google Scholar] [CrossRef] [PubMed]
- Momand, J.; Zambetti, G.P.; Olson, D.C.; George, D.; Levine, A.J. The Mdm-2 Oncogene Product Forms a Complex with the p53 Protein and Inhibits p53-Mediated Transactivation. Cell 1992, 69, 1237–1245. [Google Scholar] [CrossRef]
- Chen, J.; Wu, X.; Lin, J.; Levine, A.J. Mdm-2 Inhibits the G1 Arrest and Apoptosis Functions of the p53 Tumor Suppressor Protein. Mol. Cell. Biol. 1996, 16, 2445–2452. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.N.; Roe, A.E.; Donehower, L.A.; Bradley, A. Rescue of Embryonic Lethality in Mdm2-Deficient Mice by Absence of p53. Nature 1995, 378, 206–208. [Google Scholar] [CrossRef] [PubMed]
- Lukashchuk, N.; Vousden, K.H. Ubiquitination and Degradation of Mutant p53. Mol. Cell. Biol. 2007, 27, 8284–8295. [Google Scholar] [CrossRef]
- Sobhani, N.; D’Angelo, A.; Wang, X.; Young, K.H.; Generali, D.; Li, Y. Mutant p53 as an Antigen in Cancer Immunotherapy. Int. J. Mol. Sci. 2020, 21, 4087. [Google Scholar] [CrossRef]
- Gumus, E.; Erdamar, S.; Demirel, G.; Horasanli, K.; Kendirci, M.; Miroglu, C. Association of Positive Serum Anti-p53 Antibodies with Poor Prognosis in Bladder Cancer Patients. Int. J. Urol. 2004, 11, 1070–1077. [Google Scholar] [CrossRef]
- Kressner, U.; Glimelius, B.; Bergström, R.; Påhlman, L.; Larsson, A.; Lindmark, G. Increased Serum p53 Antibody Levels Indicate Poor Prognosis in Patients with Colorectal Cancer. Br. J. Cancer 1998, 77, 1848–1851. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kunizaki, M.; Sawai, T.; Takeshita, H.; Tominaga, T.; Hidaka, S.; To, K.; Miyazaki, T.; Hamamoto, R.; Nanashima, A.; Nagayasu, T. Clinical Value of Serum p53 Antibody in the Diagnosis and Prognosis of Colorectal Cancer. Anticancer Res. 2016, 36, 4171–4175. [Google Scholar] [PubMed]
- Kunizaki, M.; Fukuda, A.; Wakata, K.; Tominaga, T.; Nonaka, T.; Miyazaki, T.; Matsumoto, K.; Sumida, Y.; Hidaka, S.; Yasutake, T.; et al. Clinical Significance of Serum p53 Antibody in the Early Detection and Poor Prognosis of Gastric Cancer. Anticancer Res. 2017, 37, 1979–1984. [Google Scholar] [PubMed]
- Murray, P.V.; Soussi, T.; O’Brien, M.E.; Smith, I.E.; Brossault, S.; Norton, A.; Ashley, S.; Tavassoli, M. Serum p53 Antibodies: Predictors of Survival in Small-Cell Lung Cancer? Br. J. Cancer 2000, 83, 1418–1424. [Google Scholar] [CrossRef][Green Version]
- Parasole, R.; Izzo, F.; Perrone, F.; Pignata, S.; Galati, M.G.; Leonardi, E.; Castiglione, F.; Orlando, R.; Castello, G.; Esposito, G.; et al. Prognostic Value of Serum Biological Markers in Patients with Hepatocellular Carcinoma. Clin. Cancer Res. 2001, 7, 3504–3509. [Google Scholar]
- Tokunaga, R.; Sakamoto, Y.; Nakagawa, S.; Yoshida, N.; Baba, H. The Utility of Tumor Marker Combination, Including Serum P53 Antibody, in Colorectal Cancer Treatment. Surg. Today 2017, 47, 636–642. [Google Scholar] [CrossRef]
- Zalcman, G.; Trédaniel, J.; Schlichtholz, B.; Urban, T.; Milleron, B.; Lubin, R.; Meignin, V.; Couderc, L.J.; Hirsch, A.; Soussi, T. Prognostic Significance of Serum p53 Antibodies in Patients with Limited-Stage Small Cell Lung Cancer. Int. J. Cancer 2000, 89, 81–86. [Google Scholar] [CrossRef]
- Kunizaki, M.; Hamasaki, K.; Wakata, K.; Tobinaga, S.; Sumida, Y.; Hidaka, S.; Yasutake, T.; Miyazaki, T.; Matsumoto, K.; Yamasaki, T.; et al. Clinical Value of Serum p53 Antibody in the Diagnosis and Prognosis of Esophageal Squamous Cell Carcinoma. Anticancer Res. 2018, 38, 1807–1813. [Google Scholar]
- Takashi, S.; Satoshi, Y.; Akihiko, O.; Naoya, Y.; Yusuke, T.; Kentaro, M.; Yu, O.; Yasuaki, N.; Koichi, Y.; Takashi, F.; et al. Clinical Impact of Preoperative Serum p53 Antibody Titers in 1487 Patients with Surgically Treated Esophageal Squamous Cell Carcinoma: A Multi-Institutional Study. Esophagus 2021, 18, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Okada, R.; Otsuka, Y.; Wakabayashi, T.; Shinoda, M.; Aoki, T.; Murakami, M.; Arizumi, S.; Yamamoto, M.; Aramaki, O.; Takayama, T.; et al. Six Autoantibodies as Potential Serum Biomarkers of Hepatocellular Carcinoma: A Prospective Multicenter Study. Int. J. Cancer 2020, 147, 2578–2586. [Google Scholar] [CrossRef] [PubMed]
- Oshima, Y.; Suzuki, T.; Yajima, S.; Nanami, T.; Shiratori, F.; Funahashi, K.; Shimada, H. Serum p53 Antibody: Useful for Detecting Gastric Cancer but Not for Predicting Prognosis after Surgery. Surg. Today 2020, 50, 1402–1408. [Google Scholar] [CrossRef] [PubMed]
- Mattioni, M.; Soddu, S.; Prodosmo, A.; Visca, P.; Conti, S.; Alessandrini, G.; Facciolo, F.; Strigari, L. Prognostic Role of Serum p53 Antibodies in Lung Cancer. BMC Cancer 2015, 15, 148. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Yajima, S.; Ishioka, N.; Nanami, T.; Oshima, Y.; Washizawa, N.; Funahashi, K.; Otsuka, S.; Nemoto, T.; Shimada, H. Prognostic Significance of High Serum p53 Antibody Titers in Patients with Esophageal Squamous Cell Carcinoma. Esophagus 2018, 15, 294–300. [Google Scholar] [CrossRef]
- Mattioni, M.; Soddu, S.; Porrello, A.; D’Alessandro, R.; Spila, A.; Guadagni, F. Serum Anti-p53 Antibodies as a Useful Marker for Prognosis of Gastric Carcinoma. Int. J. Biol. Markers 2007, 22, 302–306. [Google Scholar] [CrossRef]
- Goodell, V.; Salazar, L.G.; Urban, N.; Drescher, C.W.; Gray, H.; Swensen, R.E.; McIntosh, M.W.; Disis, M.L. Antibody Immunity to the p53 Oncogenic Protein Is a Prognostic Indicator in Ovarian Cancer. J. Clin. Oncol. 2006, 24, 762–768. [Google Scholar] [CrossRef]
- Porzsolt, F.; Schmid, M.; Höher, D.; Muche, R.; Gaus, W.; Montenarh, M. Biologic Relevance of Auto-Anti Bodies against p53 in Patients with Metastatic Breast Cancer. Oncol. Res. Treatment 1994, 17, 402–408. [Google Scholar] [CrossRef]
- Mantel, N.; Haenszel, W. Statistical Aspects of the Analysis of Data from Retrospective Studies of Disease. J. Natl. Cancer Inst. 1959, 22, 719–748. [Google Scholar] [PubMed]
- DerSimonian, R.; Laird, N. Meta-Analysis in Clinical Trials Revisited. Contemp. Clin. Trials 2015, 45, 139–145. [Google Scholar] [CrossRef]
| Study Reference | Patients | Methods | Inclusion/Exclusion Criteria | Intervention | Follow-Up Time | Prognostic Value of s-p53-Abs | Type of Study |
|---|---|---|---|---|---|---|---|
| [10] | 76 patients with transitional urinary bladder cell carcinoma | S-p53-Abs ELISA. Antibodies for p53-wt 184 CRC patients | Inclusion: transitional cell urinary bladder cancer Exclusion: secondary organ cancer; immunodeficiency state; ages over 90; other urinary bladder tumors. | Surgery (TUR) Surgery + chemotherapy + radiotherapy (advanced stage) | 34 months | There was an association between the presence of s-p53-Abs and tumor p53 gene overexpression (p = 0.001). | Prospective |
| [11] | 184 CRC patients. Dukes’ stage: A (n = 31); B (n = 84); C (n = 41); D (n = 28) | S-p53-Abs ELISA. Antibodies for p53-wt 184 CRC patients | Inclusion: primary colon cancer | Routine Biopsy Surgery | 96 months | p53-Abs correlated with shorter survival (p = 0.02). | Retrospective |
| [12] | 170 CRC patients | S-p53Ab, CEA ELISA. Antibody for p53-wt | Inclusion: primary colon cancer Exclusion: previous radiotherapy or chemotherapy | Surgery (resected tumor specimen) | 93.6 months (median value) | Positivity for s-p53Ab in CRC did not correlate with overall survival. Kaplan–Meier analysis revealed significant differences between patients with elevated s-p53Ab and CEA and those with elevated levels of either one or neither of these factors (p < 0.001). | Retrospective |
| [13] | 208 GC patients | S-p53Ab Detected with anti-p53 detection kit MESACUP anti-p53 Test Antibody for p53-wt | Inclusion: Histologically confirmed GC Exclusion: previously chemotherapy, radiotherapy and those who died within 30 days after surgery | Surgery | 34 months | Did not observe any significant correlation between S-p53Ab in GC and overall survival (hazard ratio (HR) = 2.052; 95% confidence interval CI) = 0.891–4.726; p = 0.091). Conversely, Cox regression analysis revealed that a high level of CA19-9 was an independent prognostic factor for GC (hazard ratio (HR) = 3.864; 95% confidence interval (CI) = 1.248–11.959; p = 0.019). | Retrospective |
| [14] | 231 SCLC patients | S-p53-Abs ELISA. Antibodies for p53-wt | Inclusion: primary SCLC | Surgery Chemotherapy (227 out of 231 patients) | 3 months (at least) | High levels of p53-Abs correlated with worse survival prospects compared to patients with lower levels of the antibodies (p = 0.02). | Retrospective |
| [15] | 80 HCC patients | S-p53-Abs ELISA. Antibodies for p53-wt | Inclusion: Cytohistological of AFP level-based diagnosis of HCC | Percutaneous injection (21) Surgery (15) Radiofrequency interstitial ablation (10) Chemotherapy (4) TACE (8) Combinational treatment (5) No treatment (17) | 36 months | Anti-p53 was not useful as a prognostic factor. | Retrospective |
| [16] | 244 CRC patients | CEA, CA19-9, S-P53Ab Antibody for p53-wt | Inclusion: preoperative CEA, CA-19 and S-P53Ab. Primary tumor diagnosis | Surgery (colectomy plus lymph nodes dissection) Chemotherapy (in case of CRC recurrence) | 33.8 months (median) | S-P53Ab had no power to predict the prognosis (p = 0.786). Combined CEA and CA19-9 positivity was an exclusive independent prognostic factor (p = 0.034). | Retrospective |
| [17] | 97 SCLC patients | S-p53-Abs ELISA. Antibodies for p53-wt | Inclusion: newly and proven diagnosed lung cancer | Bronchial biopsy Chemotherapy (cisplatin, etoposide, doxorubicin, cyclophosphamide) Radiotherapy for those with brain metastasis | 18.1 months (median) | Patients with limited-stage SCLC and p53-Ab had a median survival time of 10 months, whereas limited-stage SCLC patients without p53-Ab had a 17-month median survival time (p = 0.014). | Prospective |
| [18] | 133 esophageal squamous cell carcinoma (ESCC) patients | S-p53Ab, SCC-Ag, CEA Antibody for p53-wt | Inclusion: histologically confirmed ESCC Exclusion: patients who died after 30 days after treatment and those who had preoperative radiotherapy | Surgery | 36 months (median) | S-p53Ab was detected in 39.1% (52 out of 133) of patients with ESCC, including 40.0% (20 out of 50) of patients with early-stage ESCC (p = 0.009) | Retrospective |
| [22] | 201 lung cancer patients | S-p53 antibodies by ELISA | Inclusion: Primary lung cancer | Surgery Chemotherapy (Stage IIIB and IV) Radiotherapy (if required) | 63 months | Patients with lower levels of p53Abs survived significantly longer than patients with higher levels of p53Abs (p = 0.049). | Retrospective |
| [19] | 1487 esophageal squamous cell carcinoma | S-p53 antibodies by ELISA | Inclusion: radical surgery with no neoadjuvant treatment | Esophagectomy | 42 months (median) | s-p53-Ab positive status was not significantly associated with poor overall survival | Retrospective |
| [20] | 160 hepatocellular carcinoma | Six hepatocellular carcinoma-associated antigens, including Sui1, p62, RalA, p53, NY-ESO-1, and c-myc antibodies by ELISA (TAA Panel) | Inclusion: histologically proven HCC Exclusion: coexisting or metachronous cancer within 5 disease-free years | Surgery | 60 months | The positivity for the TAA panel was independently associated with poor prognosis (p = 0.030) | Retrospective |
| [21] | 72 gastric cancers | S-p53 antibodies by ELISA | Inclusion: primary gastric cancer Exclusion: previous chemotherapy; coexisting cancer | Surgery | 32 months (median) | Overall survival was not associated with the antibodies | Retrospective |
| [23] | 105 esophageal squamous cell carcinoma | S-p53 antibodies by ELISA | Inclusion: primary esophageal squamous cell carcinoma Exclusion: metastatic disease; neoadjuvant therapy | Surgery | 35 months (median) | While seropositive patients did not demonstrate significant poor overall survival, high-titer patients demonstrated significant poor overall survival based on the multivariate analysis (p < 0.001). | Retrospective |
| Study Reference | Patients | Methods | Prognostic Value of s-p53-Abs | Type of Study | Inference |
|---|---|---|---|---|---|
| [24] | 111 gastric carcinoma patients | S-p53-Abs Levels of p53-mut were determined with a selective, quantitative ELISA kit | The survival time of serum-positive patients was significantly longer than that of patients with low/negative serum levels, with a survival rate of 41.2% and 14.9%, respectively, over 48 months (p < 0.05). | Retrospective | Significant correlation seen between levels of S-p53-mut Abs and patient survival rate |
| [25] | 104 ovarian cancer patients | S-p53-Abs ELISA. Antibodies against p53K132Q (c.394A > C). | Overall survival (OS) was significantly higher for patients with antibodies to mutant p53 when compared with patients without p53 antibodies (p = 0.01). | Retrospective | OS is significantly increased in advanced stage ovarian cancer patients with antibodies to p53 |
| [17] | 134 lung cancer patients | S-p53-Abs by Immunofluorescence. Antibodies against p53 R273H (c.818G > A) by ELISA. | Presence of anti-p53 autoantibodies is almost exclusively linked to the presence of malignant disease. | Retrospective | Presence of anti-p53 Abs had a significant correlation with shorter survival in NSCLC. |
| [26] | 50 BC patients | S-p53-Abs ELISA. Antibodies against p53R273H (c.818G > A). | s-p53-Abs were higher in BC patients with high risk vs. patients with low risk. The difference was not statistically significant (p = 0.15). | Retrospective | Presence of s-p53-Abs showed higher risk for BC patients. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sobhani, N.; Roviello, G.; D’Angelo, A.; Roudi, R.; Neeli, P.K.; Generali, D. p53 Antibodies as a Diagnostic Marker for Cancer: A Meta-Analysis. Molecules 2021, 26, 6215. https://doi.org/10.3390/molecules26206215
Sobhani N, Roviello G, D’Angelo A, Roudi R, Neeli PK, Generali D. p53 Antibodies as a Diagnostic Marker for Cancer: A Meta-Analysis. Molecules. 2021; 26(20):6215. https://doi.org/10.3390/molecules26206215
Chicago/Turabian StyleSobhani, Navid, Giandomenico Roviello, Alberto D’Angelo, Raheleh Roudi, Praveen Kumar Neeli, and Daniele Generali. 2021. "p53 Antibodies as a Diagnostic Marker for Cancer: A Meta-Analysis" Molecules 26, no. 20: 6215. https://doi.org/10.3390/molecules26206215
APA StyleSobhani, N., Roviello, G., D’Angelo, A., Roudi, R., Neeli, P. K., & Generali, D. (2021). p53 Antibodies as a Diagnostic Marker for Cancer: A Meta-Analysis. Molecules, 26(20), 6215. https://doi.org/10.3390/molecules26206215









