1. Introduction
In contrast to their bulk counterparts, two-dimensional materials, such as graphene, exhibit unique electronic properties and ultra-high specific surface areas. These unusual characteristics result in energy band structures sensitive to external perturbations and attract significant research interest [
1]. Recently, Kondo et al. successfully synthesised hydrogen boride (HB) nanosheets [
2]. The synthesis method involves the 3-day reaction of layered bulk magnesium diboride (MgB
2) powder with an ion-exchange resin in an organic solvent to promote the exfoliation of MgB
2. This is the first experimentally synthesised borophane (hydrogenated borophene), which is composed of sp
2-bonded boron, forming hexagonal boron networks with bridge hydrogens. The chemical composition of the material is HB, where the hydrogen/boron atomic ratio is 1/1. Previous experiments have shown that hydrogen boride sheets have a high H
2 content (8.5 wt %) [
2], unique electronic properties [
3,
4], solid acid catalytic properties [
5,
6], photoinduced hydrogen release capability [
7], and a unique ability to reduce metal ions to form nanocomposites [
8,
9,
10]. In addition, theoretical models and computer simulations demonstrate that borophane is a promising candidate for applications such as anode material, alcohol gas sensors, current limiters, and photodetectors [
11,
12,
13,
14].
At present, studies on the synthesis of hydrogen boride have been carried out only for a powder sample obtained from finely dispersed MgB
2 [
2,
3,
4,
5,
6,
7,
8,
9,
10]. However, hydrogen boride thin films deposited on substrates would offer better opportunities for the in-depth analysis of the physical properties and investigation of possible applications of this material. Thus far, there has been no research on well-defined thin films of hydrogen boride. The epitaxial growth of hydrides is quite a challenging objective, owing to their thermal and chemical instability in air and multiple charged states [
15,
16,
17].
Herein, we have developed a facile method for the fabrication of thin films of hydrogen boride using an ion-exchange treatment of deposited MgB2 films under ambient temperature and pressure. Firstly, oriented MgB2 films on SrTiO3 (STO) substrates were prepared by pulsed laser deposition (PLD). Afterwards, the deposited MgB2 films were treated with an ion-exchanger in an organic solvent to fabricate hydrogen boride films. Thin films wherein B-H bonds were introduced were successfully synthesised by using two types of ion-exchangers: proton exchange resin and formic acid. We also observed significant hydrogen release from these films under ultraviolet (UV) light irradiation.
2. Results and Discussions
To fabricate the single-phase MgB
2 film, a two-step PLD method was used as described in the experimental section.
Figure 1 shows the out-of-plane XRD patterns of the MgB
2 film deposited on the STO (100) substrate. As shown in the inset image, a black film was formed on the STO substrate. Only the
10-11 diffraction peak of the MgB
2 film was detected, except for the peaks associated with the substrate, indicating its strong orientation. Such oriented growth of MgB
2 is in agreement with previous reports [
18,
19,
20]. Although a strong
10-11 peak appeared, our in-plane XRD analysis could not detect the diffraction peaks because the film thickness was insufficient for in-plane diffraction. Consecutively, we were unable to investigate the lattice matching relationship in the plane, and the film was described as a single-oriented thin film. The thickness of the MgB
2 film varied between 50 nm and 270 nm (see
Supplementary Information, Figure S1). When the film was prepared by a one-step PLD method (MgB
2 deposition without Mg deposition), a peak of boron-rich magnesium boride (MgB
x) was observed in addition to the MgB
2 peak (
Figure S2). The most likely reason for this is the lack of Mg in the one-step PLD method due to the high volatility of Mg. The expected candidates of the produced MgB
x compound are listed in the
Supplementary Information (Table S1). When a different crystalline substrate, such as sapphire, was used in a two-step PLD process, no diffraction peaks of MgB
2 were observed (
Figure S3). These results indicate that a dense and oriented MgB
2 film can be deposited on the STO (100) substrate by a two-step PLD method.
Next, the oriented MgB
2 film was treated with ion-exchange resin in acetonitrile. MgB
2 is a layered compound in which positive magnesium ions with a valence number close to divalent are intercalated between negatively charged hexagonal boron layers, forming a honeycomb structure [
21,
22,
23]. In the present study, an ion-exchange resin was ground in an agate mortar to increase the reaction area for efficient ion-exchange in a reaction time equivalent to that of powder sample synthesis. During the ion-exchange treatment, Mg
2+ ions in MgB
2 are changed with protons. Therefore, we expect that negatively charged boron states (B
−) can remain even after the process, similar to previous powder sample experiments [
2]. HAXPES analysis was performed to characterise the valence states of the boron and magnesium species in the film.
Figure 2a shows the HAXPES spectra of the B 1s core-level before and after the ion-exchange treatment. As a result, the majority of the boron sites retained their negatively charged B
− state (peak at ~188.9 eV), even after the treatment. This indicates that oxidation does not occur during the ion-exchange, and impurities, such as boric acid, are not formed (B 1s of boric acid is known to appear at 193.2 eV [
2]). As shown in
Figure 2b, after the ion-exchange the Mg 2p core-level peak broadens, indicating that the surroundings of Mg in the thin film change significantly. X-ray diffraction data revealed a gradual decrease of the MgB
2 10-11 peaks after the treatment, suggesting that its crystallinity becomes worse after ion-exchange (see
Supplementary Information, Figures S4 and S5).
Since photo-emission spectroscopy analysis is known to have low sensitivity to hydrogen species, TOF-SIMS analysis of the thin MgB
2 films was performed.
Figure 3 shows the TOF-SIMS depth profile of the ionised hydrogen species (H
+) in the films. The red line demonstrates the H
+ species profile of the MgB
2 film. In this case, the observed H
+ species on the surface originate from adsorbed water and/or hydrocarbon impurities. The ion-exchanged films exhibit a greater amount of hydrogen species than that of the untreated film. It can be assumed that the resin would not be an efficient reagent because of the limited exchange process at the solid-to-solid interface between MgB
2 and the resin. Therefore, as an alternative ion-exchange method, liquid formic acid was used as a proton donor for more efficient ion-exchange. It is worth noting that the film treated with formic acid displayed a higher hydrogen content than the film treated with ion-exchange resin. The etching rate of the TOF-SIMS analysis was approximately 0.35–0.4 nm/s and was determined by the depth of the rastered area and sputtering time (
Figure S6). Considering the etching rate, the 200 s sputtering time corresponds to 70–80 nm etching depth. The depth distribution of the hydrogen species varied depending on the method. In the formic acid-treated thin films, hydrogen species were introduced at a depth of less than 100 nm from the surface.
We also investigated the TOF-SIMS depth profiles of the boron and magnesium species (
Figure S7). In the MgB
2 film before the ion-exchange treatment, enrichment in boron species was observed near the surface (
Figure S7a, red line) due to the volatilization of Mg in the outermost surface of the MgB
2 thin film during the PLD process, as discussed above. After the ion-exchange process with the resin, both the boron and magnesium species’ contents decreased. The decrease in the abundance of the magnesium species is due to leaching via the ion-exchange with protons inhomogeneously, while the lower boron species content can be explained by the exfoliation of boron-rich layers from the thin film. The decrease in the boron species signal was more significant compared to that of the magnesium species. The integrated values of the TOF-SIMS signal intensities from 0 to 20 s (corresponding to the 7–8 nm layer on the top surface) before and after the ion-exchange treatments were calculated and compared. Then, the relative values after the resin ion-exchange with respect to the values before the ion-exchange were 0.51 and 0.71 for the boron species and magnesium species, respectively.
TOF-SIMS analysis of the boron and magnesium species were also performed for thin films treated with formic acid (
Figure S8). Similar to the resin treatment, the content of both boron and magnesium species was reduced after the formic acid treatment. In this case, the decrease in the magnesium signal was more significant than that of the boron species. These results indicate that a long-term treatment with formic acid can induce a proton ion-exchange process rather than an exfoliation of the boron layers in the MgB
2 film. We also evaluated the TOF-SIMS signal of the carbon species after the sample was treated with formic acid (
Figure S9). The signal intensities of the carbon species were very low, even after the formic acid treatment. As shown in
Figure 3, the hydrogen species signal was stronger and detectable at a greater depth below the surface compared to the carbon species signal (
Figure S9). These results indicate that the dissociated protons from formic acid were successfully introduced into the MgB
2 film during the ion-exchange process.
Next, FT-IR measurements were conducted in an attempt to confirm the formation of B-H and/or B-H-B bonds in the ion-exchanged film as an additional spectroscopic evidence for the ion exchange of Mg
2+ by protons. In spite of using the highly sensitive ATR method, no signals characteristic of these bonds were detected, presumably because the film was too thin. Instead, the FT-IR spectrum of MgB
2 powder was investigated (
Figure S10), commercially available from Sigma-Aldrich and handled in the same manner as the thin films. Notably, after the ion-exchange treatment in formic acid, a peak appeared around 2500 cm
−1. According to a previous report, this signal corresponds to the terminal B-H stretching mode in hydrogen boride sheets [
4]. Since we followed an identical ion-exchange process for both the powder sample and the thin film, we assume that a similar B-H bond was formed in the film sample, as the existence of hydrogen species was also confirmed in the film by TOF-SIMS measurements.
We also compared the surface morphologies of the as-deposited and ion-exchanged films by SEM and AFM, as shown in
Figure 4. The SEM (
Figure 4a) and AFM (
Figure 4b) images of the as-deposited MgB
2 thin film (before the ion-exchange) reveal cross-shaped rectangular patterns. This implies that the MgB
2 film has an epitaxial relationship with the STO substrate, although we could not confirm this by in-plane XRD. The peak to valley (PTV) height difference in the AFM image was 50–100 nm (
Figure 4b,
Figure S11a). In contrast, the film treated with the ion-exchange resin exhibited a smoother surface compared to the as-deposited MgB
2 film, with a PTV height difference of less than 50 nm (
Figure 4c,d and
Figure S11b). This is likely because of the physical contact (mechanical abrasion) between the MgB
2 thin film and the resin during the ion-exchange. The thickness of the film after the ion-exchange treatment was less than 200 nm (
Figure S12). This might be one of the reasons for the lower H content and its narrower depth distribution observed in the TOF-SIMS profile. On the other hand, the film treated with formic acid for 2 days exhibited more pronounced crisscross patterns on the surface and a larger PTV height difference (50–200 nm), as shown in
Figure 4e,f and
Figure S11c. This is because the ion-exchange and exfoliation were more efficient when using formic acid, as compared to the use of ion-exchange resin. These results reveal that both the hydrogen content and the surface morphology of the thin films can vary significantly depending on the type of ion-exchange treatment. This also suggests that, by choosing an appropriate ion-exchange method and conditions, the morphology and hydrogen content of the films can be controlled for various device applications.
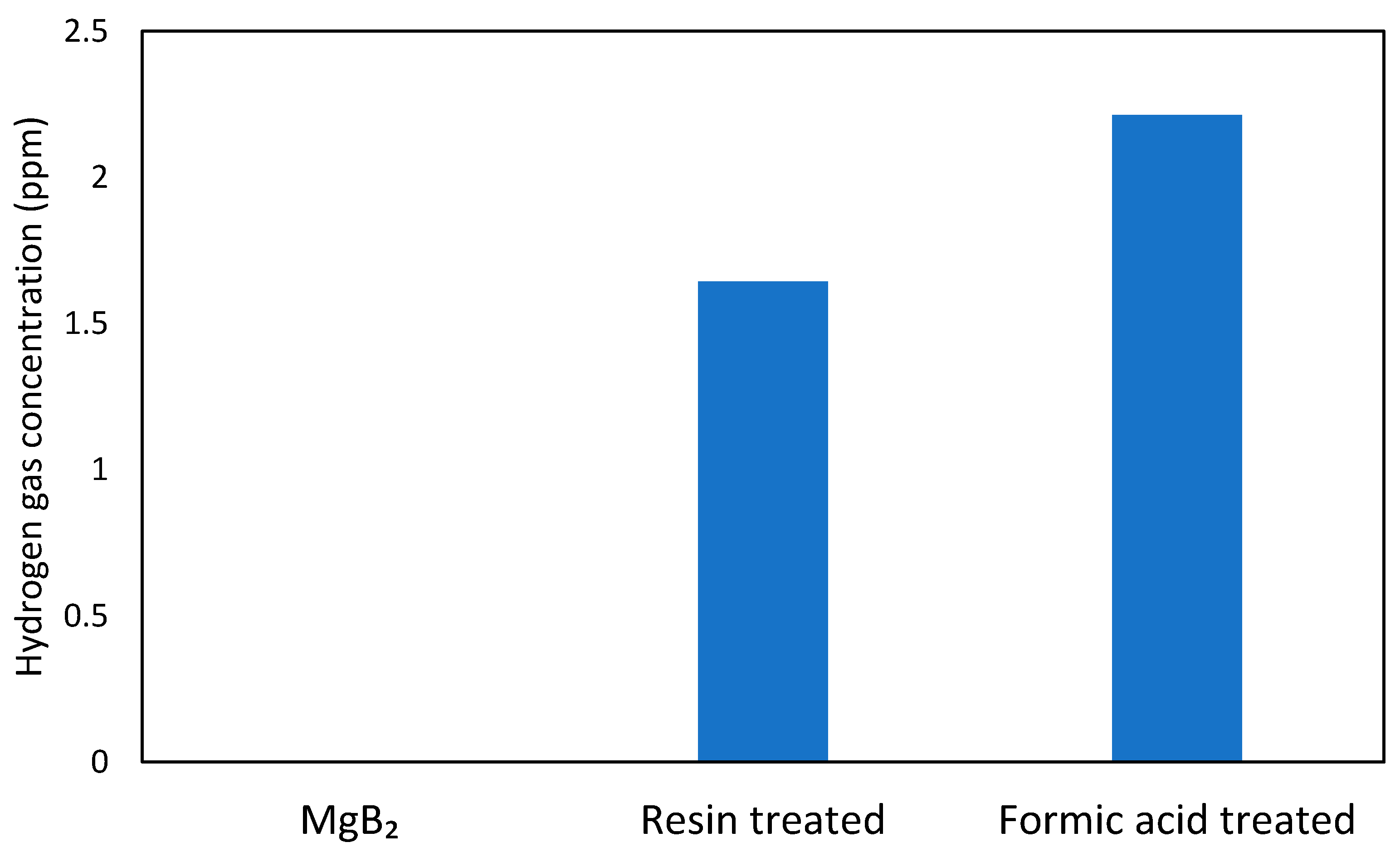
Previous experiments on powder systems have demonstrated that UV irradiation of the B-H bonds in hydrogen boride sheets can lead to hydrogen gas release [
7]. In this study, the hydrogen release was induced by photoirradiation, which causes the electron transition from the σ-bonding state (valence band) to the anti-bonding state of boron and hydrogen orbitals (conduction band) with a gap of 3.8 eV and causes the self-reduction of protons together with oxidation of boron in HB, even under mild ambient conditions. We anticipated that a similar mechanism might lead to hydrogen emission from the ion-exchanged thin films upon UV irradiation. As shown in
Figure 5, hydrogen molecules were detected from both types of ion-exchanged films upon UV light irradiation, while the as-deposited MgB
2 did not release hydrogen gas. The amount of hydrogen released from the formic acid treated film was larger than that released from the resin-treated film. These results agree well with the TOF-SIMS data, which showed a higher hydrogen species content in the film treated with formic acid compared to the other one. That strongly implies that B-H bonds were formed in the ion-exchanged film, as suggested by the FT-IR analysis, and hydrogen molecules can be liberated by an electron transition from the bonding state to the anti-bonding state of hydrogen and boron orbitals under UV light irradiation [
7].
Based on the experimental data, we can propose a reaction scheme for the ion-exchange process in MgB
2 films, as shown in
Figure 6. The oriented MgB
2 thin film reacts with ion-exchange resin or formic acid (left panel) causing H
+ to intercalate inside the film and Mg
2+ ions to be released from the film in the acetonitrile solution (middle panel), similar to a layered metal oxide case [
24]. In addition to the ion-exchange, a partial exfoliation of boron layers occurred at the outermost surface, leaving H
+ inside the thin film to form B-H bonds (right panel). In terms of the chemical composition, the ion-exchanged film is depicted as an inhomogeneous hybrid material with a top layer mainly consisting of HB and a bottom layer mainly consisting of MgB
2.
3. Materials and Methods
3.1. Materials and Equipment
SrTiO3 (100) single-crystal substrates were purchased from SHINKOSHA CO., LTD. Mg powder (212–600 μm, 99.9%, Wako, Osaka, Japan) and MgB2 powder (99%; Sigma-Aldrich, St. Louis, MO, USA) were purchased to use MgB2 thin film deposition. Acetonitrile (99.5% (JIS Special grade), FUJIFILM Wako Pure Chemical Industries Ltd., Osaka, Japan), a cation-exchange resin (15JS-HG⋅DRY, Organo Corp., Tokyo, Japan) and formic acid (Kanto Chemical Co., Inc., Tokyo, Japan) were purchased for ion-exchange treatment.
A pulsed laser ablation device (PLFD-221-1R, Freedom Ltd., Kawasaki, Japan) was used for PLD. X-ray diffractometer (Smartlab, Rigaku Corporation, Tokyo, Japan), laser microscope (LEXT OLS5100, Olympus Corporation, Tokyo, Japan), TOF-SIMS 5-100-AD (ION-TOF GmbH, Germany), FT/IR-6100 (JASCO, Co., Ltd., Tokyo, Japan), atomic force microscope (SPM-9700, Shimadzu Corp., Kyoto, Japan) and scanning electron microscope (JEM-2010F, JEOL, Ltd., Tokyo, Japan) were used for characterization of prepared films.
3.2. Preparation of MgB2 Thin Films
The MgB2 thin films were deposited on SrTiO3 (100) single-crystal substrates by PLD using the fourth harmonic wavelength (266 nm) of a neodymium: yttrium aluminium garnet (Nd: YAG) laser. The substrate size was 1.0 × 1.0 cm2. The thin film deposition was performed under argon partial pressure of 1.0 mTorr and substrate temperature of 700 °C. A high-purity single-phase MgB2 film was deposited via a two-step deposition using subsequently two targets: a metallic magnesium (Mg) target pellet for 10 min and an MgB2 pellet for 60 min for the same substrate. The former process was necessary to compensate for the Mg supply in the MgB2 film because of the high volatility of Mg. The Mg pellet was prepared by uniaxial pressing using Mg powder, while the MgB2 pellet was prepared using commercial powder.
3.3. Ion-Exchange Treatment of MgB2 Thin Films
Two different types of ion-exchange method were employed to treat the deposited MgB
2 thin films: the first method involved a 3-day reaction with a cation-exchange resin (400 mg) that was ground in an agate mortar in acetonitrile while referencing the previous report [
2]. As an alternative ion-exchange method, a solution of 3.6 mL of acetonitrile and 0.4 mL of formic acid as a proton donor was prepared according to the report by Kawamura et al. [
25], and MgB
2 thin films were immersed into the solution for either 3 h or 2 d. Both treatments were conducted at room temperature and atmospheric pressure in a glovebox (<1 ppm O
2) to prevent air exposure. The ion-exchanged film samples were washed with acetonitrile and dried under a nitrogen atmosphere.
3.4. Characterization of Prepared Films
X-ray diffraction (XRD) measurements were performed using a Rigaku Smartlab X-ray diffractometer (45 kV, 200 mA, radiation source: Cu(Kα1)). The film thickness was measured using a laser microscope. We evaluated the height difference between the MgB2 thin film and the bare STO substrate near the edge of the thin film.
Hard X-ray photoemission spectroscopy (HAXPES) measurements were performed at BL15XU of SPring-8 (Super Photon Ring 8 GeV, Hyōgo Prefecture, Japan). The excitation photon energy and total energy resolution were set to 5.95 keV and 240 meV, respectively. The measurements were taken at room temperature and the pressure of the analysis chamber of HAXPES was 1.1 × 10
−7 Pa. Details of HAXPES experiments at BL15XU were described elsewhere [
26].
Time-of-flight secondary ion mass spectroscopy (TOF-SIMS) depth profile analyses were performed using a TOF-SIMS 5-100-AD. The analysis parameters were as follows: primary ion source, Bi3++; energy, 60 keV; area, 100 × 100 μm2. The sputtering parameters were as follows: sputtering ion source, O2; energy, 2 keV; area, 300 × 300 μm2. To investigate the relationship between the actual depth and the sputtering time of the TOF-SIMS, the depth of the rastered area by ion beam was measured using a laser microscope and the sputtering rate was estimated.
Fourier-transform infrared spectroscopy (FT-IR) spectra were recorded using an FT/IR-6100 (MCT detector). The MgB2 film was too thin for the detection of the FT-IR signals of B-H and/or B-H-B bonds. Thus, an additional FT-IR analysis was conducted using ion-exchanged MgB2 powder by an attenuated total reflection (ATR) method using a diamond holder. For FT-IR analysis, MgB2 powder was treated in the same manner as the MgB2 film. Atomic force microscopy (AFM) images were obtained in dynamic imaging mode using an SPM-9700 with a silicon cantilever. Scanning electron microscope (SEM) images were obtained using a JEM-2010F (6.0 kV).
3.5. Photoinduced Hydrogen Gas Release
A film sample was placed in a quartz cell (3.5 mL with septum screw cap) under a nitrogen atmosphere without any solvent. Hydrogen production in the quartz cell under near-UV light irradiation was evaluated by gas chromatography (Tracera-GC-2010 Plus with a BID detector, Shimadzu, Co., Ltd., Japan). A film sample was placed under dark conditions for 1 h, followed by UV irradiation for 2 h. Hydrogen generation was determined by the difference in hydrogen amounts before and after the UV irradiation. UV irradiation was performed using a mercury-xenon (Hg-Xe) lamp with a 340 nm band-pass filter, similar to previous reports [
7]. As a control experiment, the MgB
2 film without an ion-exchange treatment was also evaluated in the same manner as the ion-exchanged films.