A Novel Phenylpyrrolidine Derivative: Synthesis and Effect on Cognitive Functions in Rats with Experimental Ishemic Stroke
Abstract
:1. Introduction
2. Results and Discussion
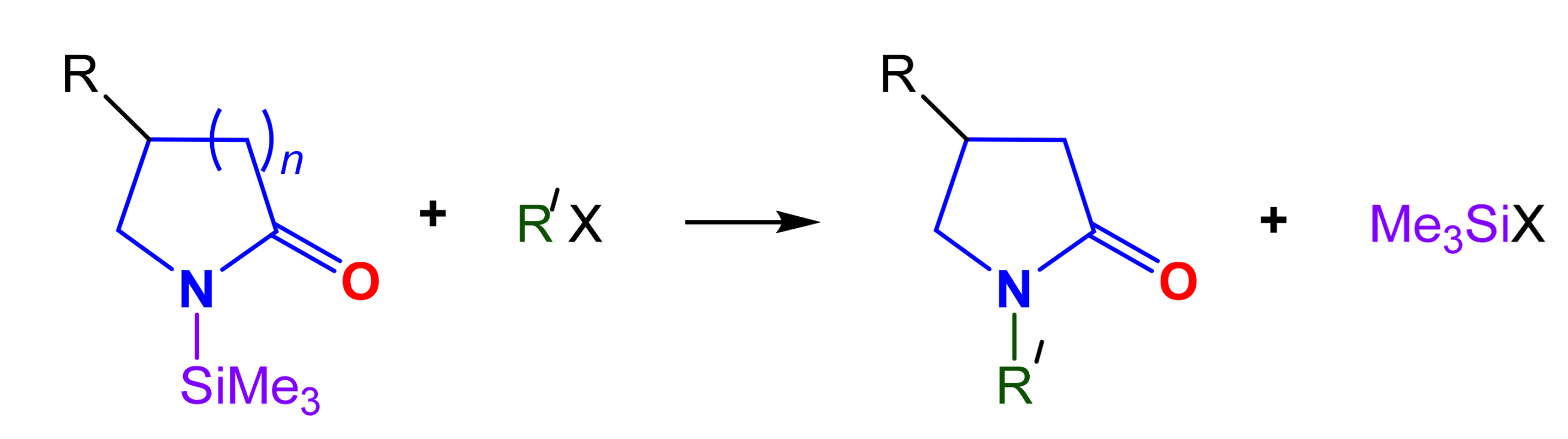
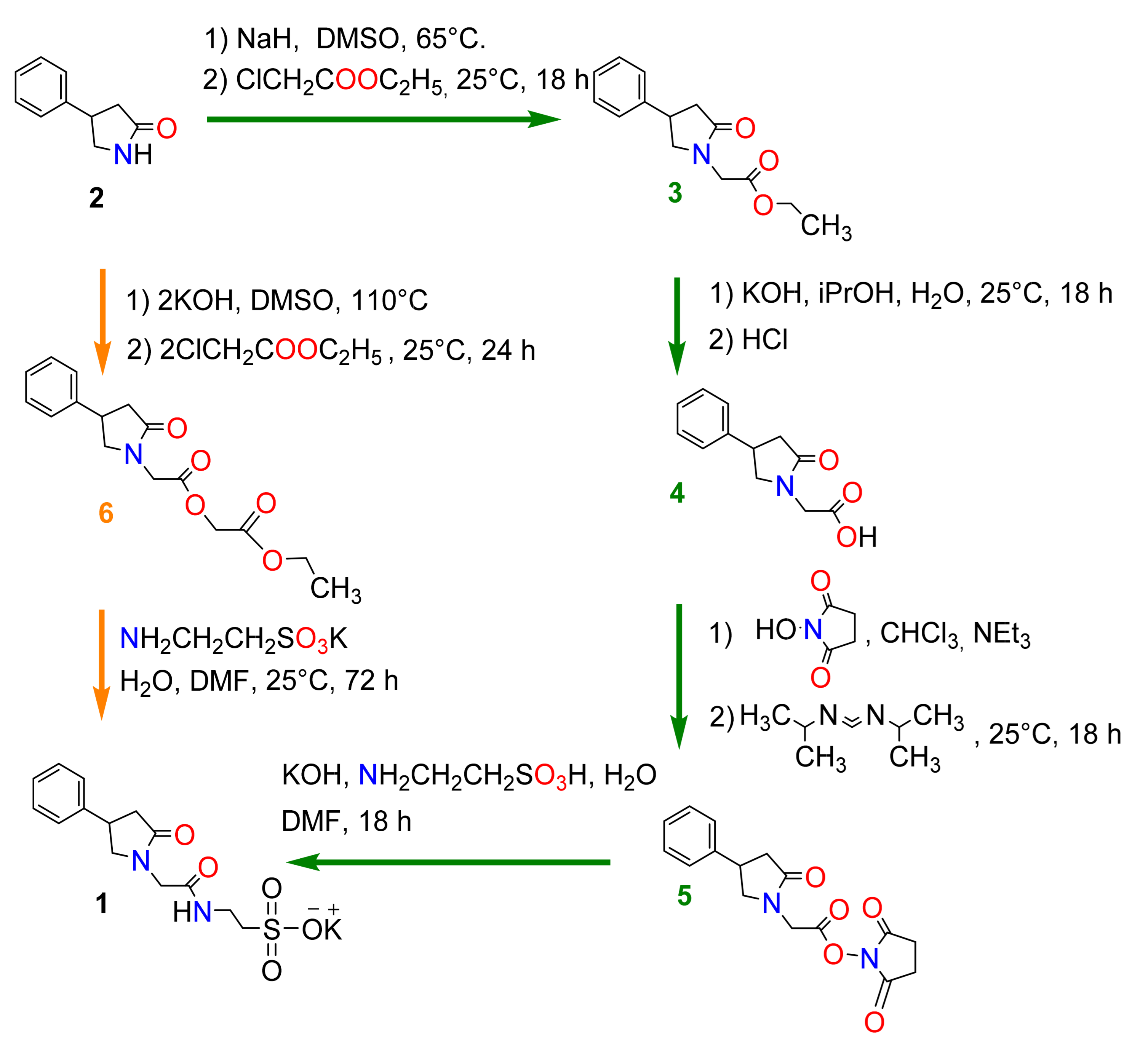
2.1. Chemistry
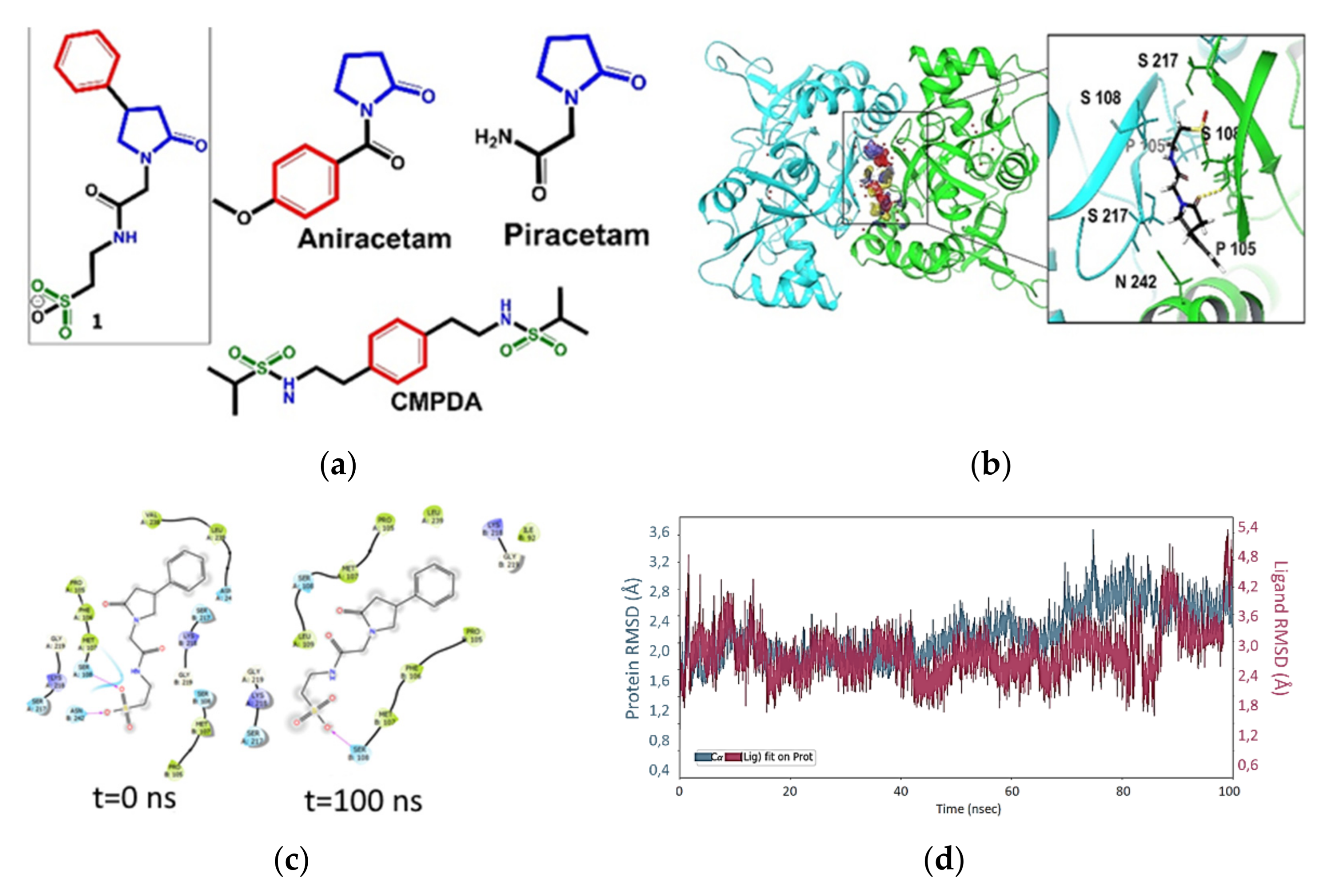
2.2. The Structure-Activity Relationship (SAR)
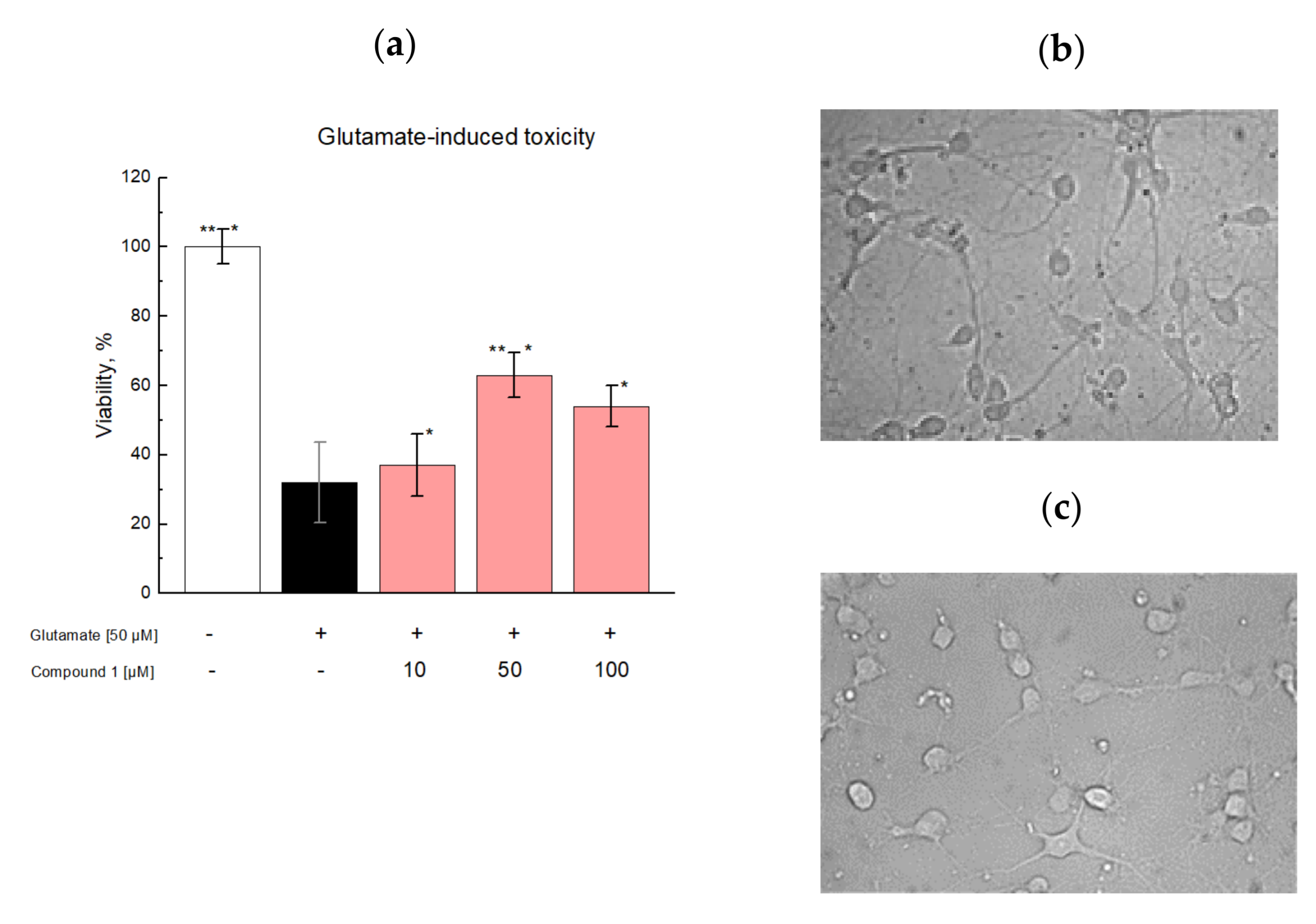
2.3. In Vitro Studies
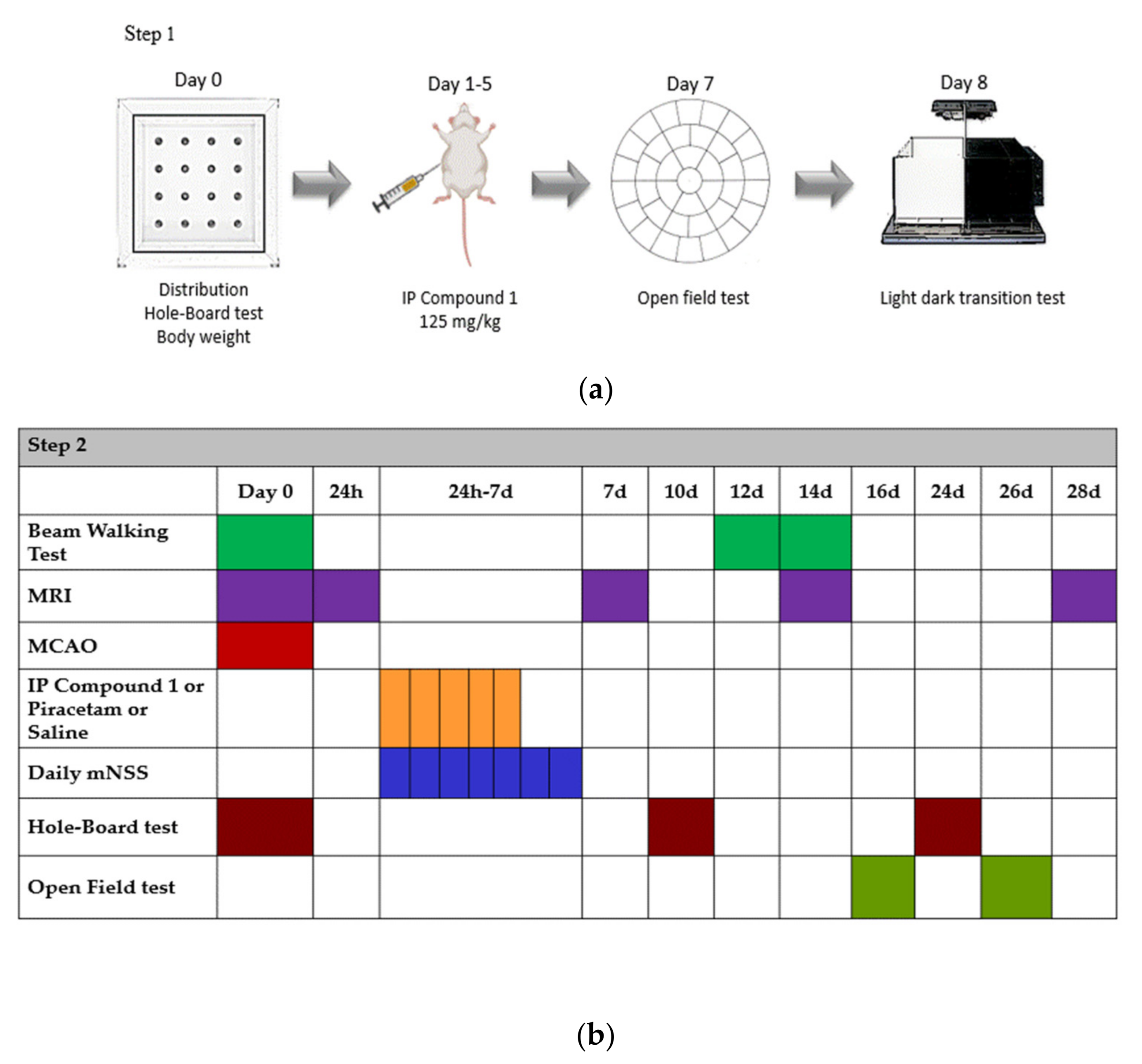
2.4. In Vivo Studies
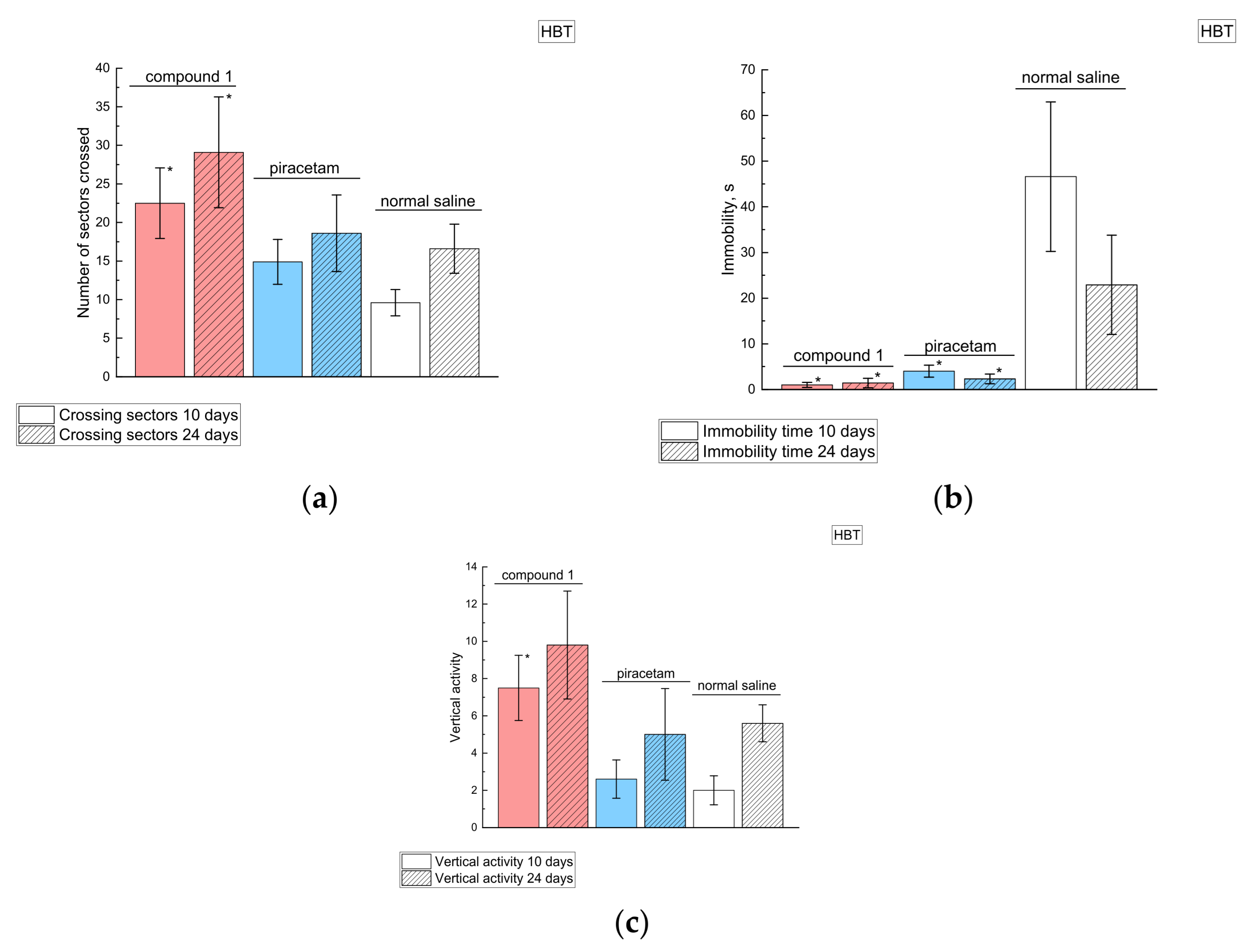
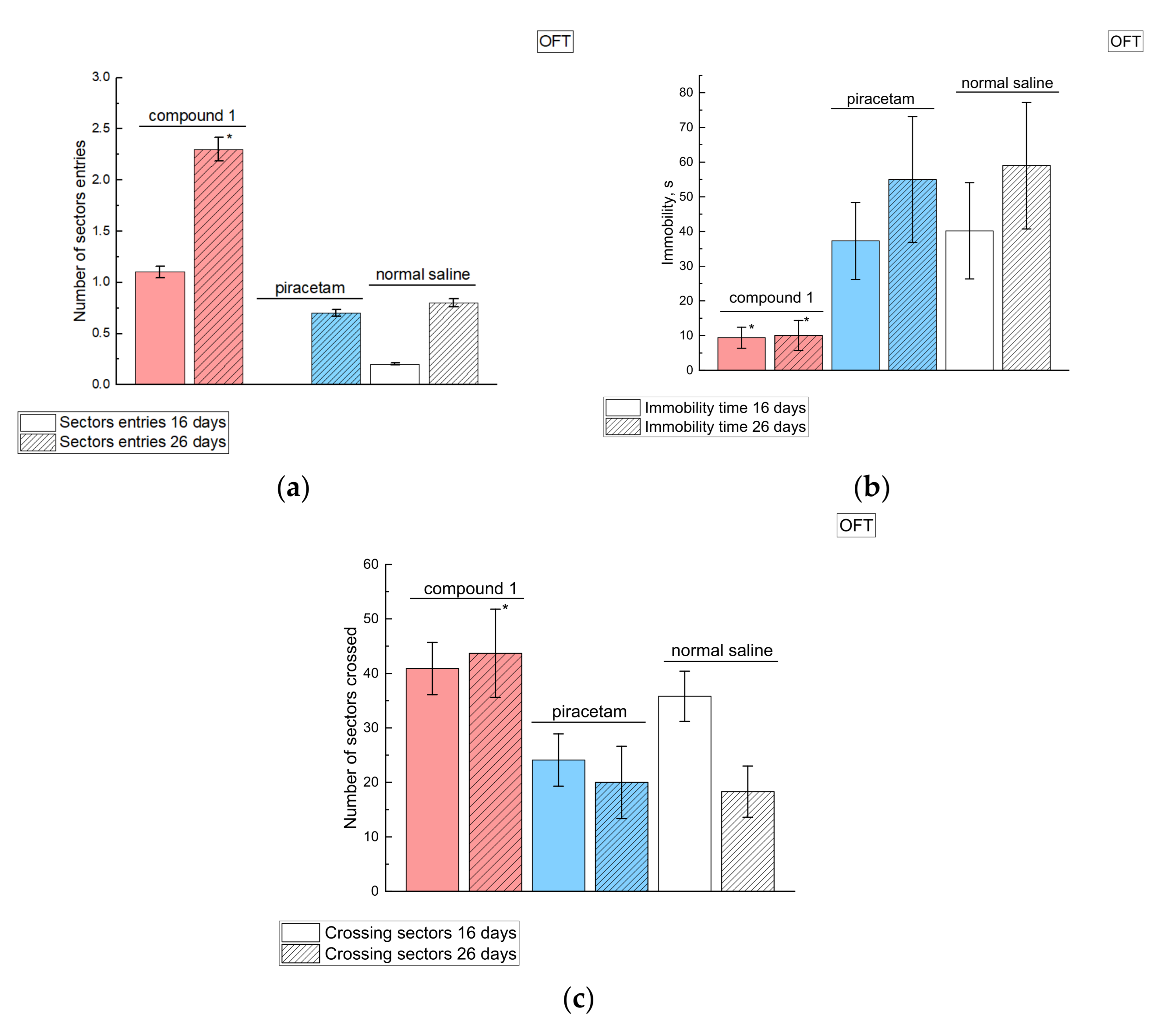
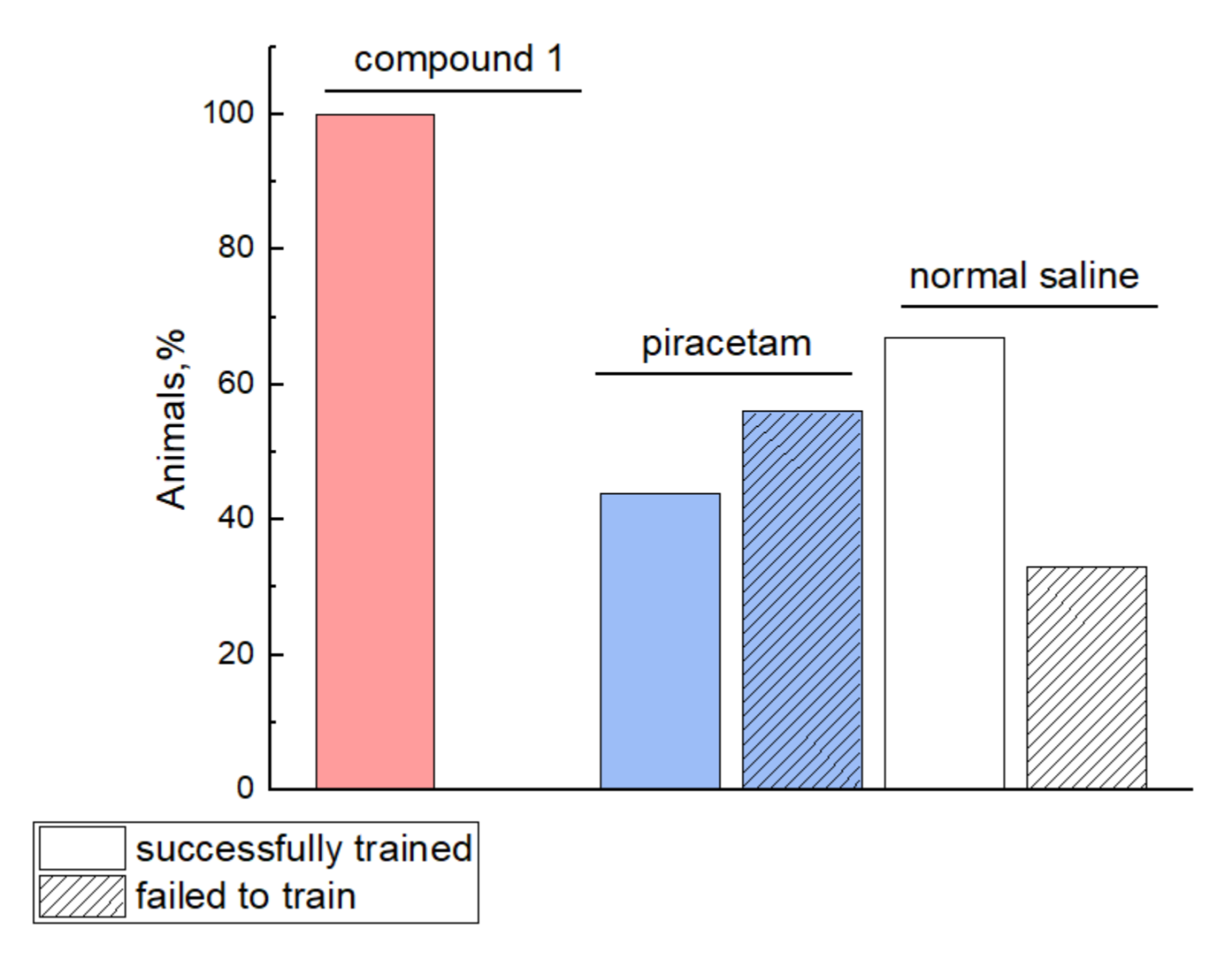
2.4.1. Behavioral Effects of Compound 1 in Intact Animals
2.4.2. Effects of Compound 1 in Rats with MCAO Model
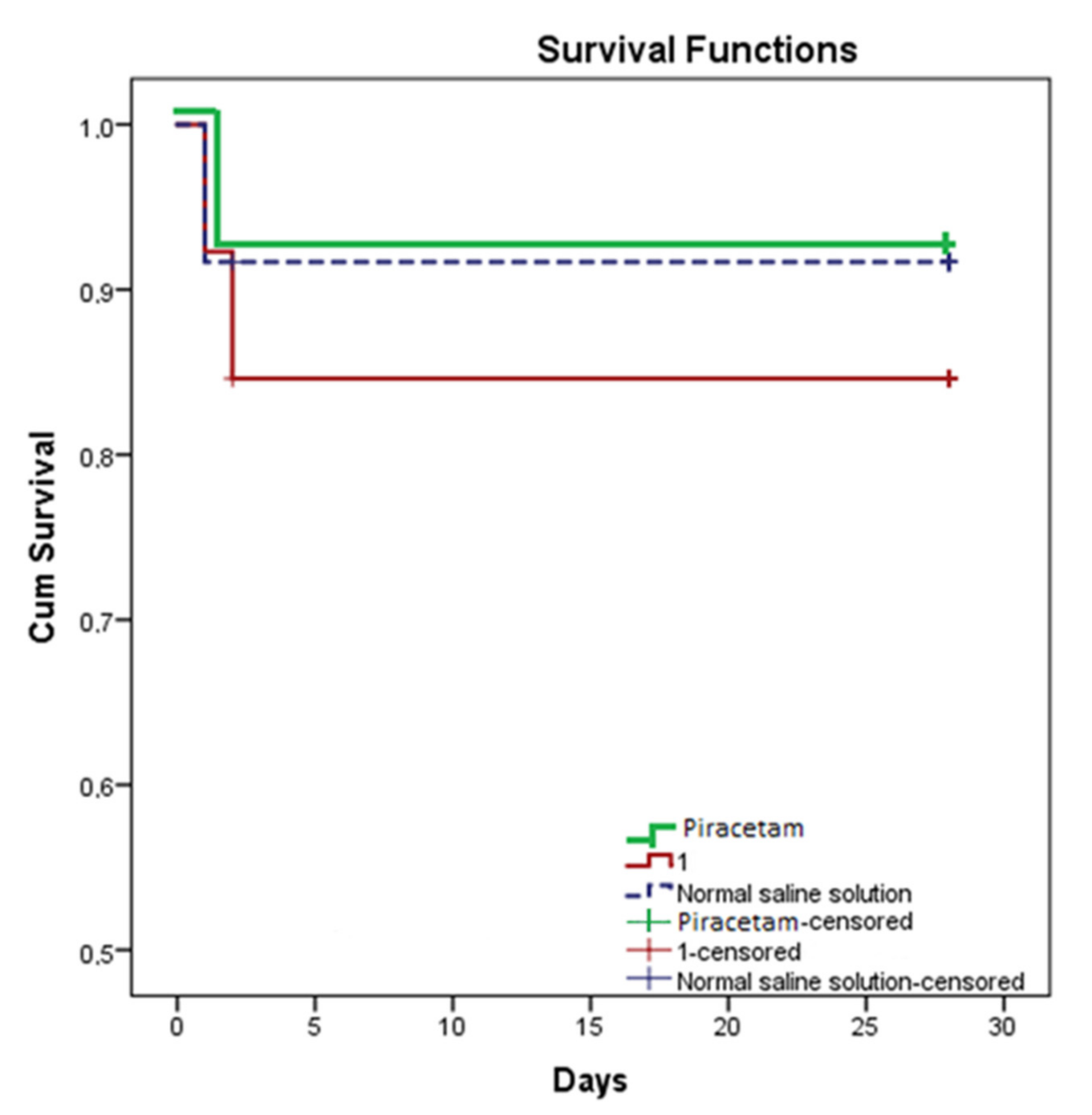
Survival Rate
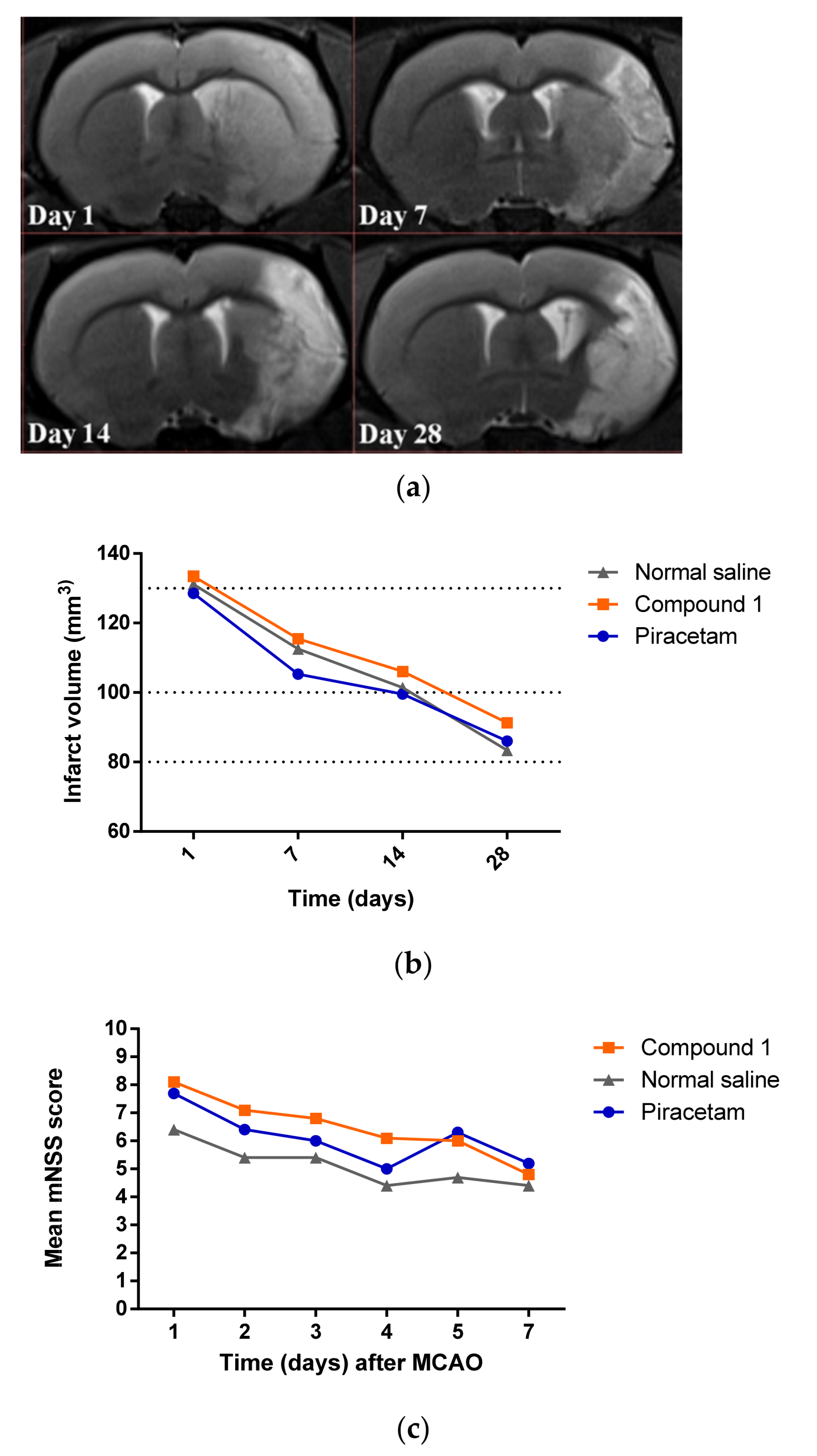
Cerebral Infarct Volume
Neurological Deficit
2.5. The Blood–Brain Barrier Permeability of Compound 1
2.6. Molecular Docking and Molecular Dynamic
3. Materials and Methods
3.1. Chemistry
3.2. In Silico Studies
3.2.1. The Structure–Activity Relationship (SAR)
3.2.2. Calculation Details
3.2.3. Protein and Ligand Preparation
3.2.4. Active Site Analysis
3.2.5. Molecular Docking and Dynamics
3.2.6. Calculation Method of BBB Permeability
3.3. In Vitro Studies
3.4. Analysis of Compound 1 in Rat’s Plasma and Brain by HPLC-MS/MS
3.4.1. Preparation of Working Solutions
3.4.2. HPLC-MS Conditions
3.5. Animals
3.5.1. Transient Middle Cerebral Artery Occlusion
3.5.2. MRI Measurements
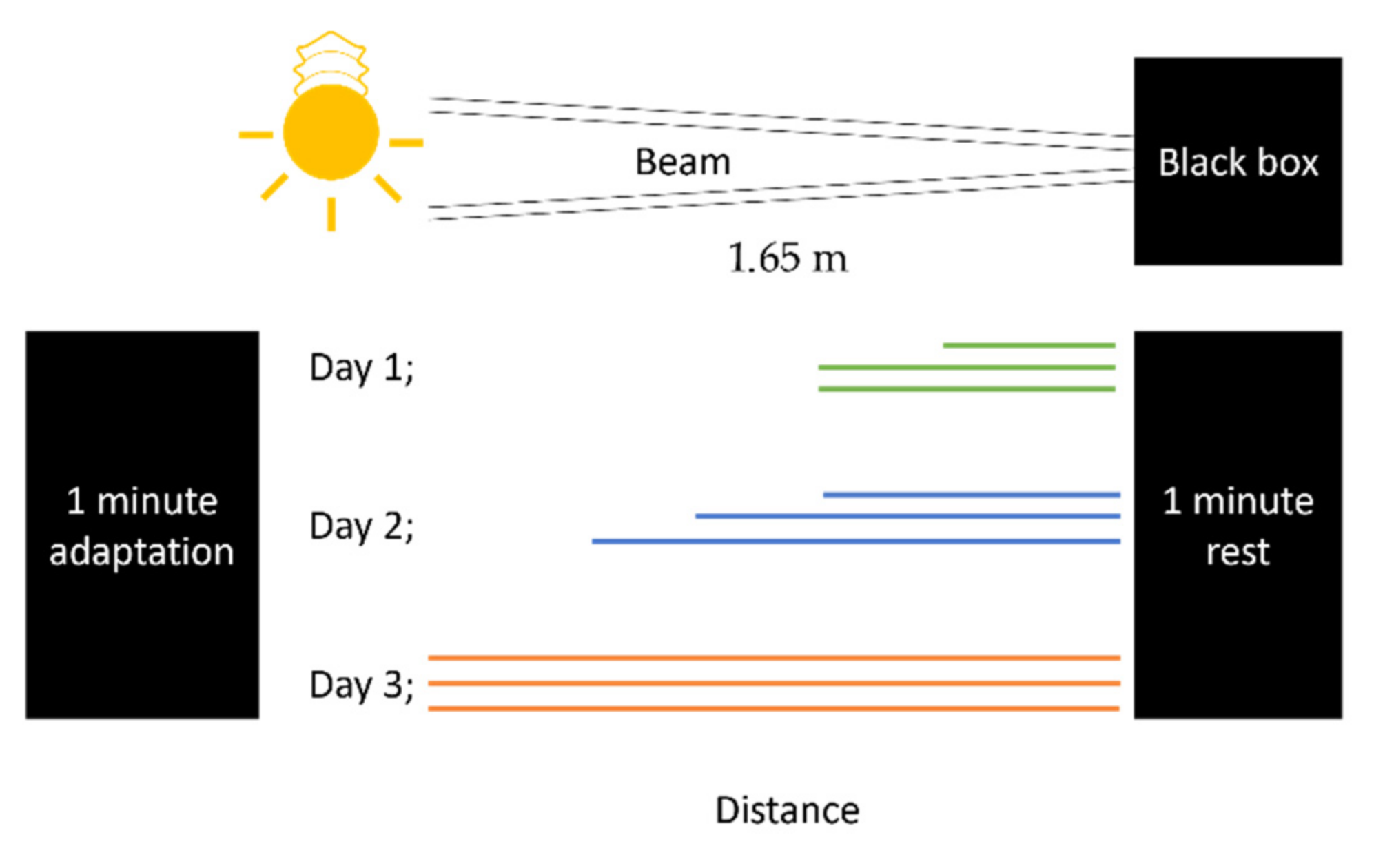
3.5.3. Behavioral Tests
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Deuschl, G.; Beghi, E.; Fazekas, F.; Varga, T.; Christoforidi, K.A.; Sipido, E.; Bassetti, C.L.; Vos, T.; Feigin, V.L. The burden of neurological diseases in Europe: An analysis for the Global Burden of Disease Study 2017. Lancet Public Health 2020, 5, e551–e567. [Google Scholar] [CrossRef]
- Cramer, S.C. Drugs to Enhance Motor Recovery After Stroke. Stroke 2015, 46, 2998–3005. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Powers, W.J.; Rabinstein, A.A.; Ackerson, T.; Adeoye, O.M.; Bambakidis, N.C.; Becker, K.; Biller, J.; Brown, M.; Demaerschalk, B.M.; Hoh, B.; et al. Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke 2019, 50, e344–e418. [Google Scholar] [PubMed]
- Malykh, A.G.; Sadaie, M.R. Piracetam and piracetam-like drugs: From basic science to novel clinical applications to CNS disorders. Drugs 2010, 70, 287–312. [Google Scholar] [CrossRef]
- Genton, P.; Van Vleymen, B. Piracetam and levetiracetam: Close structural similarities but different pharmacological and clinical profiles. Epileptic Disord. 2000, 2, 99–105. [Google Scholar]
- Shipov, A.G.; Kramarova, E.P.; Negrebetsky, V.V.; Pogozhikh, S.A.; Yu, I.B. Synthesis, Molecular and Crystal Structure of PHE Notropyl. Bull. RSMU 2006, 1, 56–61. [Google Scholar]
- Filimonov, D.A.; Druzhilovskiy, D.S.; Lagunin, A.A.; Gloriozova, T.A.; Rudik, A.V.; Dmitriev, A.V.; Pogodin, P.V.; Poroikov, V.V. Computer-aided Prediction of Biological Activity Spectra for Chemical Compounds: Opportunities and Limitations. Biomed. Chem. Res. Methods 2018, 1, e00004. [Google Scholar] [CrossRef] [Green Version]
- Filimonov, D.A.; Lagunin, A.A.; Gloriozova, T.A.; Rudik, A.V.; Druzhilovskii, D.S.; Pogodin, P.V.; Poroikov, V.V. Prediction of the Biological Activity Spectra of Organic Compounds Using the Pass Online Web Resource. Chem. Heterocycl. Compd. 2014, 50, 444–457. [Google Scholar] [CrossRef]
- Traynelis, S.F.; Wollmuth, L.P.; McBain, C.J.; Menniti, F.S.; Vance, K.M.; Ogden, K.K.; Hansen, K.B.; Yuan, H.; Myers, S.J.; Dingledine, R. Glutamate receptor ion channels: Structure, regulation, and function. Pharmacol. Rev. 2010, 62, 405–496. [Google Scholar] [CrossRef] [Green Version]
- Burd, I.; Welling, J.; Kannan, G.; Johnston, M.V. Chapter Five—Excitotoxicity as a Common Mechanism for Fetal Neuronal Injury with Hypoxia and Intrauterine Inflammation. In Advances in Pharmacology; Schwarcz, R., Ed.; Academic Press: Cambridge, MA, USA, 2016; Volume 76, pp. 85–101. [Google Scholar]
- Sestakova, N.; Puzserova, A.; Kluknavsky, M.; Bernatova, I. Determination of motor activity and anxiety-related behaviour in rodents: Methodological aspects and role of nitric oxide. Interdiscip. Toxicol. 2013, 6, 126–135. [Google Scholar] [CrossRef] [Green Version]
- Lu, M.; Chen, J.; Lu, D.; Yi, L.; Mahmood, A.; Chopp, M. Global test statistics for treatment effect of stroke and traumatic brain injury in rats with administration of bone marrow stromal cells. J. Neurosci. Methods 2003, 128, 183–190. [Google Scholar] [CrossRef]
- Encarnacion, A.; Horie, N.; Keren-Gill, H.; Bliss, T.M.; Steinberg, G.K.; Shamloo, M. Long-term behavioral assessment of function in an experimental model for ischemic stroke. J. Neurosci. Methods 2011, 196, 247–257. [Google Scholar] [CrossRef] [Green Version]
- Cai, W.; Stewart, R.; Mueller, C.; Li, Y.J.; Shen, W.D. Poststroke depression and risk of stroke recurrence and mortality: Protocol of a meta-analysis and systematic review. BMJ Open 2018, 8, e026316. [Google Scholar] [CrossRef]
- Li, W.; Xiao, W.-M.; Chen, Y.-K.; Qu, J.-F.; Liu, Y.-L.; Fang, X.-W.; Weng, H.-Y.; Luo, G.-P. Anxiety in Patients With Acute Ischemic Stroke: Risk Factors and Effects on Functional Status. Front. Psychiatry 2019, 10, 257. [Google Scholar] [CrossRef]
- Rohde, D.; Gaynor, E.; Large, M.; Mellon, L.; Hall, P.; Brewer, L.; Bennett, K.; Williams, D.; Dolan, E.; Callaly, E.; et al. The Impact of Cognitive Impairment on Poststroke Outcomes: A 5-Year Follow-Up. J. Geriatr. Psychiatry Neurol. 2019, 32, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Malik, V.A.; Di Benedetto, B. The Blood-Brain Barrier and the EphR/Ephrin System: Perspectives on a Link Between Neurovascular and Neuropsychiatric Disorders. Front. Mol. Neurosci. 2018, 11, 127. [Google Scholar] [CrossRef]
- Clark, D.E. In silico prediction of blood-brain barrier permeation. Drug. Discov. Today 2003, 8, 927–933. [Google Scholar] [CrossRef]
- Bennett, B.; Matagne, A.; Michel, P.; Leonard, M.; Cornet, M.; Meeus, M.-A.; Toublanc, N. Seletracetam (UCB 44212). Neurotherapeutics 2007, 4, 117–122. [Google Scholar] [CrossRef]
- Huang, L.; Shang, E.; Fan, W.; Li, X.; Li, B.; He, S.; Fu, Y.; Zhang, Y.; Li, Y.; Fang, W. S-oxiracetam protect against ischemic stroke via alleviating blood brain barrier dysfunction in rats. Eur. J. Pharm. Sci. 2017, 109, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Doheny, M.H.; O’Connell, M.T.; Patsalos, P.N. A high-performance liquid-chromatographic microanalytical procedure for the rapid estimation of piracetam in plasma or cerebrospinal fluid. J. Pharm. Pharmacol. 1996, 48, 514–516. [Google Scholar] [CrossRef] [PubMed]
- Winblad, B. Piracetam: A review of pharmacological properties and clinical uses. CNS Drug Rev. 2005, 11, 169–182. [Google Scholar] [CrossRef] [PubMed]
- Harmsen, B.; Robeyns, K.; Wouters, J.; Leyssens, T. A Study of Fasoracetam’s Solid State Forms: A Potential Anti-Alzheimer Pharmaceutical. J. Pharm. Sci. 2017, 106, 1317–1321. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Andjelkovic, A.V.; Zhu, L.; Yang, T.; Bennett, M.V.L.; Chen, J.; Keep, R.F.; Shi, Y. Blood-brain barrier dysfunction and recovery after ischemic stroke. Prog. Neurobiol. 2018, 163–164, 144–171. [Google Scholar] [CrossRef]
- Paliwal, P.; Dash, D.; Krishnamurthy, S. Pharmacokinetic Study of Piracetam in Focal Cerebral Ischemic Rats. Eur. J. Drug Metab. Pharmacokinet. 2018, 43, 205–213. [Google Scholar] [CrossRef]
- Black, M.D. Therapeutic potential of positive AMPA modulators and their relationship to AMPA receptor subunits. A review of preclinical data. Psychopharmacology 2005, 179, 154–163. [Google Scholar] [CrossRef] [PubMed]
- Thomas, W. AMPA Receptor Antagonists for the Treatment of Stroke. Curr. Drug Targets-CNS Neurol. Disord. 2005, 4, 153–159. [Google Scholar]
- Gitto, R.; De Luca, L.; Pagano, B.; Citraro, R.; De Sarro, G.; Costa, L.; Ciranna, L.; Chimirri, A. Synthesis and anticonvulsant evaluation of N-substituted isoquinoline AMPA receptor antagonists. Bioorganic Med. Chem. 2008, 16, 2379–2384. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, L.G.; Dos Santos, R.N.; Oliva, G.; Andricopulo, A.D. Molecular Docking and Structure-Based Drug Design Strategies. Molecules 2015, 20, 13384–13421. [Google Scholar] [CrossRef]
- Ingvar, M.; Ambros-Ingerson, J.; Davis, M.; Granger, R.; Kessler, M.; Rogers, G.A.; Schehr, R.S.; Lynch, G. Enhancement by an ampakine of memory encoding in humans. Exp. Neurol. 1997, 146, 553–559. [Google Scholar] [CrossRef] [Green Version]
- Michael, J.O.N.; David, B.; Dennis, M.Z.; Eric, S.N. AMPA Receptor Potentiators for the Treatment of CNS Disorders. Curr. Drug Targets-CNS Neurol. Disord. 2004, 3, 181–194. [Google Scholar]
- Martín-Martín, M.L.; Bartolomé-Nebreda, J.M.; Conde-Ceide, S.; Alonso de Diego, S.A.; López, S.; Martínez-Viturro, C.M.; Tong, H.M.; Lavreysen, H.; Macdonald, G.J.; Steckler, T.; et al. Discovery and SAR of novel series of imidazopyrimidinones and dihydroimidazopyrimidinones as positive allosteric modulators of the metabotropic glutamate receptor 5 (mGlu5). Bioorganic Med. Chem. Lett. 2015, 25, 1310–1317. [Google Scholar] [CrossRef] [Green Version]
- Froestl, W.; Muhs, A.; Pfeifer, A. Cognitive Enhancers (Nootropics). Part 1: Drugs Interacting with Receptors. J. Alzheimer’s Dis. 2012, 32, 793–887. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Timm, D.E.; Benveniste, M.; Weeks, A.M.; Nisenbaum, E.S.; Partin, K.M. Structural and Functional Analysis of Two New Positive Allosteric Modulators of GluA2 Desensitization and Deactivation. Mol. Pharmacol. 2011, 80, 267–280. [Google Scholar] [CrossRef] [Green Version]
- Jin, R.; Clark, S.; Weeks, A.M.; Dudman, J.T.; Gouaux, E.; Partin, K.M. Mechanism of Positive Allosteric Modulators Acting on AMPA Receptors. J. Neurosci. 2005, 25, 9027–9036. [Google Scholar] [CrossRef]
- Lagunin, A.; Zakharov, A.; Filimonov, D.; Poroikov, V. QSAR Modelling of Rat Acute Toxicity on the Basis of PASS Prediction. Mol. Inform. 2011, 30, 241–250. [Google Scholar] [CrossRef] [PubMed]
- Harpsøe, K.; Liljefors, T.; Balle, T. Prediction of the binding mode of biarylpropylsulfonamide allosteric AMPA receptor modulators based on docking, GRID molecular interaction fields and 3D-QSAR analysis. J. Mol. Graph. Model. 2008, 26, 874–883. [Google Scholar] [CrossRef]
- Lavrov, M.I.; Veremeeva, P.N.; Karlov, D.S.; Zamoyski, V.L.; Grigoriev, V.V.; Palyulin, V.A. Tricyclic derivatives of bispidine as AMPA receptor allosteric modulators. Mendeleev Commun. 2019, 29, 619–621. [Google Scholar] [CrossRef]
- Lavrov, M.I.; Karlov, D.S.; Voronina, T.A.; Grigoriev, V.V.; Ustyugov, A.A.; Bachurin, S.O.; Palyulin, V.A. Novel Positive Allosteric Modulators of AMPA Receptors Based on 3,7-Diazabicyclo [3.3.1]nonane Scaffold. Mol. Neurobiol. 2020, 57, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Announcing Schrödinger Software Release 2020-1|Schrödinger. Available online: https://www.schrodinger.com/user-announcement/announcing-schrodinger-software-release-2020-1/ (accessed on 19 August 2021).
- Leung, S.S.F.; Sindhikara, D.; Jacobson, M.P. Simple Predictive Models of Passive Membrane Permeability Incorporating Size-Dependent Membrane-Water Partition. J. Chem. Inf. Modeling 2016, 56, 924–929. [Google Scholar] [CrossRef] [PubMed]
- Berman, H.M.; Battistuz, T.; Bhat, T.N.; Bluhm, W.F.; Bourne, P.E.; Burkhardt, K.; Feng, Z.; Gilliland, G.L.; Iype, L.; Jain, S.; et al. The Protein Data Bank. Acta Cryst. D Biol. Crystallogr. 2002, 58 Pt 6, 899–907. [Google Scholar] [CrossRef]
- Shivakumar, D.; Harder, E.; Damm, W.; Friesner, R.A.; Sherman, W. Improving the Prediction of Absolute Solvation Free Energies Using the Next Generation OPLS Force Field. J. Chem. Theory Comput. 2012, 8, 2553–2558. [Google Scholar] [CrossRef] [PubMed]
- Hilgenberg, L.G.W.; Smith, M.A. Preparation of Dissociated Mouse Cortical Neuron Cultures. JoVE 2007, e562. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gubskiy, I.L.; Namestnikova, D.D.; Cherkashova, E.A.; Chekhonin, V.P.; Baklaushev, V.P.; Gubsky, L.V.; Yarygin, K.N. MRI Guiding of the Middle Cerebral Artery Occlusion in Rats Aimed to Improve Stroke Modeling. Transl. Stroke Res. 2018, 9, 417–425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rasband, W.S.; Image, J.U.S. National Institutes of Health: Bethesda, MD, USA, 1997–2018. Available online: https://imagej.nih.gov/ij/ (accessed on 19 August 2021).
- Cui, L.L.; Golubczyk, D.; Jolkkonen, J. Top 3 Behavioral Tests in Cell Therapy Studies After Stroke: Difficult to Stop a Moving Train. Stroke 2017, 48, 3165–3167. [Google Scholar] [CrossRef]
- Feeney, D.M.; Gonzalez, A.; Law, W.A. Amphetamine, haloperidol, and experience interact to affect rate of recovery after motor cortex injury. Science 1982, 217, 855–857. [Google Scholar] [CrossRef]
- Goldstein, L.B.; Davis, J.N. Beam-walking in rats: Studies towards developing an animal model of functional recovery after brain injury. J. Neurosci. Methods 1990, 31, 101–107. [Google Scholar] [CrossRef]
| Effect | Pa | Pi | Mechanism of Action | Pa | Pi |
|---|---|---|---|---|---|
| Nootropic | 0.705 | 0.040 | Neurotransmitter antagonist | 0.502 | 0.039 |
| Antianginal | 0.544 | 0.032 | |||
| Antihypoxic | 0.553 | 0.025 | |||
| Cytoprotective | 0.609 | 0.027 |
| Compound 1 | Normal Saline | Piracetam | |
|---|---|---|---|
| Crossings (n) | 53.7 ± 10.3 | 53.6 ± 14.2 | 65.4 ± 20.4 |
| Number of center sector crossings (center) (n) | 1.6 ± 0.6 * | 0.8 ± 0.9 | 1.4 ± 1.2 |
| Vertical activity (n) | 10.3 ± 4.3 * | 16.8 ± 6.1 | 15.9 ± 7.5 |
| Immobility duration (sec) | 12.8 ± 8.4* | 25.6 ± 13.8 | 16.0 ± 10.4 |
| Grooming time (sec) | 40.6 ± 33.2 | 37.1 ± 21.6 | 14.4 ± 9.4 |
| Compound 1 | Normal Saline | Piracetam | |
|---|---|---|---|
| Time spent in the light compartment (sec) | 16.5 ± 8.3 * | 9.8 ± 5.2 | 26.3 ± 13.4 * |
| Number of crossings (n) | 2.9 ± 1.2 * | 1.4 ± 0.8 | 3.7 ± 1.6 * |
| Number of peeking out events (n) | 5.2 ± 2.8 | 1.6 ± 1.4 | 4.8 ± 2.5 |
| Compound 1 | Normal Saline | Piracetam | |
|---|---|---|---|
| Left forelimb score | 42.2 ± 28.0 | 26.0 ± 24.2 | 32.8 ± 26.4 |
| Right forelimb score | 11.6 ± 13.8 | 16.6 ± 17.2 | 12.4 ± 16.0 |
| Left hindlimb score | 28.8 ± 15.3 | 28.3 ± 30.2 | 24.8 ± 22.2 |
| Right hindlimb score | 45.8 ± 30.4 | 35.7 ± 32.6 | 36.0 ± 30.4 |
| Ampakines | Log Pm 1 | PCaco, cm s−1 (QPPCaco) 2 | PMDCK, cm s−1 (QPPMDCK) 3 | QPlogBB 4 | Literature Report of BBB Permeability |
|---|---|---|---|---|---|
| Seletracetam | −4.4 | 279.0 | 1268.6 | −0.4 | Very good [19] |
| Oxiracetam | −4.7 | 56.0 | 82.73 | −1.0 | Low [20] |
| Piracetam | −4.5 | 165.0 | 261.5 | −0.5 | Very good [21,22] |
| Fasoracetam | −4.5 | 227.8 | 410.6 | −0.4 | Low (30%) [23] |
| Phenylpiracetam | −4.6 | 155.3 | 252.1 | −0.7 | Unknown |
| Compound 1 | −6.5 | 20.5 | 20.7 | −1.5 | Low |
| Rats | Cplasma mkg mL−1 | Cstriatum ng mg−1 | Ccortex ng/mg | Cplasma/Cstriatum | Cplasma/Ccortex |
|---|---|---|---|---|---|
| Intact animals | 16.1 ± 5.8 | 247.2 ± 42.6 | 255.1 ± 78.2 | 651.2 ± 52.0 | 630.9 ± 98.6 |
| Animals after MCAO | 13.8 ± 3.6 | 1533.6 ± 24.5 | 3633.8 ± 40.1 | 89.7 ± 9.4 | 37.9 ± 6.0 |
| ID Structure | Glide Score, kcal mol–1 | |LE| | IFD Score, kcal mol–1 | ΔGMM-GBSA, kcal mol–1 | H-bond Bridges |
|---|---|---|---|---|---|
| Aniracetam | −10.17 | 0.63 | −1114.25 | −42.28 | Water-SER-A-108; Water-SER-B-108 |
| Piracetam | −8.83 | 0.88 | −1115.15 | −25.19 | Water-SER-A-108 Water-ASN-B-242 Ser-A-217 |
| CMPDA | −10.69 | 0.43 | −1110.25 | n/d | Pro-A-105 Pro-C-105 |
| 1-R | −11.65 | 0.53 | −1118.45 | −34.42 | SER-A-108 |
| 1-S | −9.91 | 0.45 | −1116.58 | −19.48 | Water-SER-A-217 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borozdenko, D.A.; Ezdoglian, A.A.; Shmigol, T.A.; Gonchar, D.I.; Lyakhmun, D.N.; Tarasenko, D.V.; Golubev, Y.V.; Cherkashova, E.A.; Namestnikova, D.D.; Gubskiy, I.L.; et al. A Novel Phenylpyrrolidine Derivative: Synthesis and Effect on Cognitive Functions in Rats with Experimental Ishemic Stroke. Molecules 2021, 26, 6124. https://doi.org/10.3390/molecules26206124
Borozdenko DA, Ezdoglian AA, Shmigol TA, Gonchar DI, Lyakhmun DN, Tarasenko DV, Golubev YV, Cherkashova EA, Namestnikova DD, Gubskiy IL, et al. A Novel Phenylpyrrolidine Derivative: Synthesis and Effect on Cognitive Functions in Rats with Experimental Ishemic Stroke. Molecules. 2021; 26(20):6124. https://doi.org/10.3390/molecules26206124
Chicago/Turabian StyleBorozdenko, Denis A., Aiarpi A. Ezdoglian, Tatiana A. Shmigol, Darya I. Gonchar, Dmitri N. Lyakhmun, Dmitri V. Tarasenko, Yaroslav V. Golubev, Elvira A. Cherkashova, Daria D. Namestnikova, Ilya L. Gubskiy, and et al. 2021. "A Novel Phenylpyrrolidine Derivative: Synthesis and Effect on Cognitive Functions in Rats with Experimental Ishemic Stroke" Molecules 26, no. 20: 6124. https://doi.org/10.3390/molecules26206124
APA StyleBorozdenko, D. A., Ezdoglian, A. A., Shmigol, T. A., Gonchar, D. I., Lyakhmun, D. N., Tarasenko, D. V., Golubev, Y. V., Cherkashova, E. A., Namestnikova, D. D., Gubskiy, I. L., Lagunin, A. A., Gubsky, L. V., Chekhonin, V. P., Borisevich, S. S., Gureev, M. A., Shagina, A. D., Kiseleva, N. M., Negrebetsky, V. V., & Baukov, Y. I. (2021). A Novel Phenylpyrrolidine Derivative: Synthesis and Effect on Cognitive Functions in Rats with Experimental Ishemic Stroke. Molecules, 26(20), 6124. https://doi.org/10.3390/molecules26206124









