A Review on Natural Fiber Bio-Composites, Surface Modifications and Applications
Abstract
1. Introduction
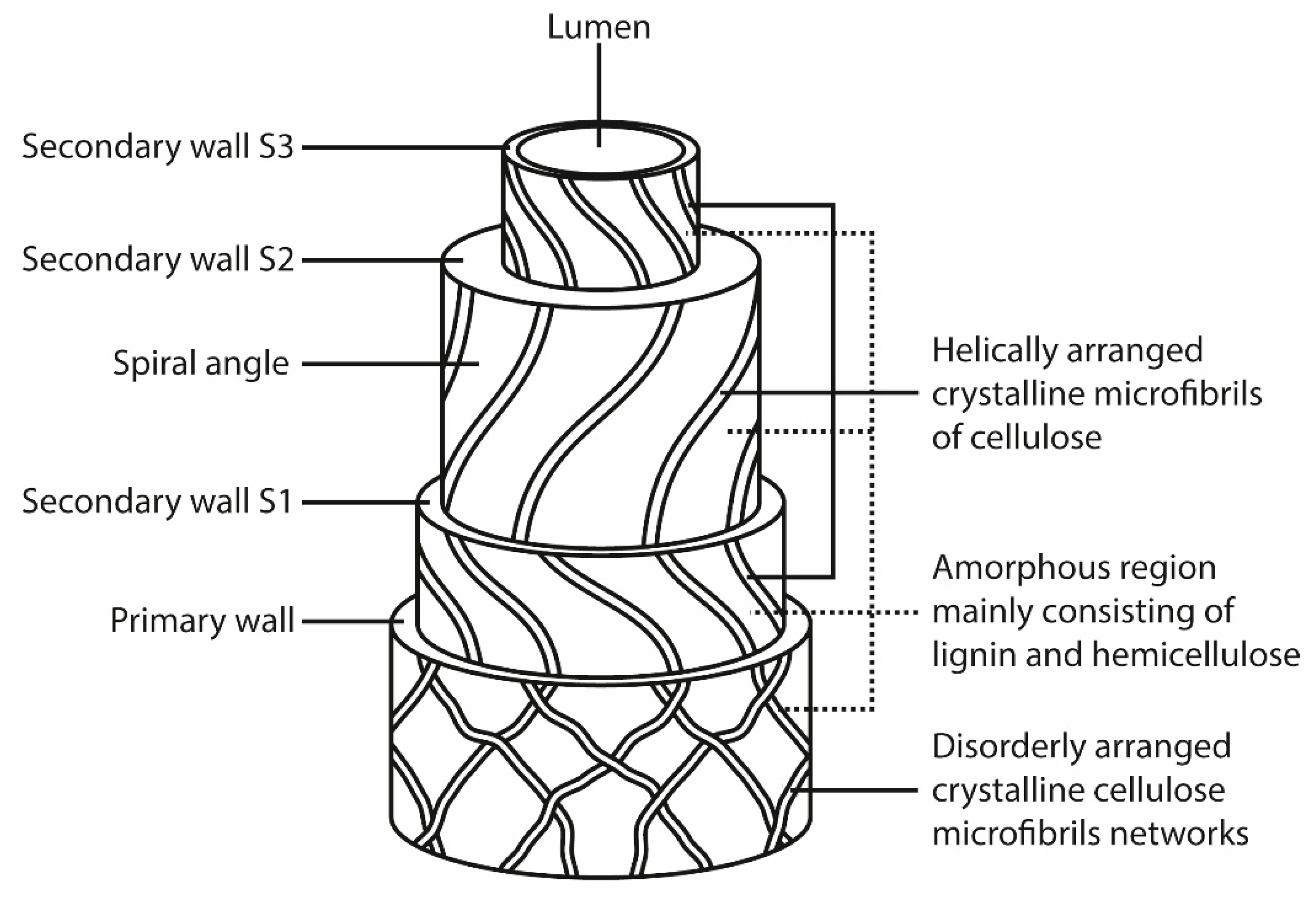
2. Lignocellulosic Fibers
2.1. Cellulose
2.2. Hemicellulose
3. Fiber Modification
3.1. Fiber/Matrix Adhesion
3.2. Reducing Moisture Absorption by Natural Fiber
3.3. Thermal Degradation and Flammability Properties
3.4. Mechanical Properties
4. Biodegradable Matrix Materials
4.1. Polybutylene Succinate (PBS)
4.2. Polylactic Acid (PLA)
4.3. Poly Hydroxyalkanote (PHA)
5. Processing Techniques of Bio-Composites
5.1. Compression Molding
5.2. Extrusion
6. Applications of Bio-Composites
6.1. Automobile Industry
6.2. Construction and Textile Industry
7. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Ojha, S.; Raghavendra, G.; Acharya, S.K. A Comparative Investigation of Bio Waste Filler (Wood Apple-Coconut) Reinforced Polymer Composites. Polym. Compos. 2014, 35, 180–185. [Google Scholar] [CrossRef]
- Molaba, T.P.; Chapple, S.; John, M.J. Aging Studies on Flame Retardant Treated Lignocellulosic Fibers. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Liu, W.; Drzal, L.T.; Mohanty, A.K.; Misra, M. Influence of Processing Methods and Fiber Length on Physical Properties of Kenaf Fiber Reinforced Soy Based Biocomposites. Compos. Part B Eng. 2007, 38, 352–359. [Google Scholar] [CrossRef]
- Wiley: Lignocellulosic Polymer Composites: Processing, Characterization, and Properties—Vijay Kumar Thakur. Available online: http://www.wiley.com/WileyCDA/WileyTitle/productCd-1118773578.html (accessed on 7 December 2017).
- Book Review: An Inconvenient Truth. Available online: http://www.webofcreation.org/religious-education/179-book-review-an-inconvenient-truth (accessed on 8 December 2017).
- Guna, V.; Ilangovan, M.; Ananthaprasad, M.G.; Reddy, N. Hybrid Biocomposites. Polym. Compos. 2018. [Google Scholar] [CrossRef]
- Banik, N.; Dey, V.; Sastry, G.R.K. An Overview of Lignin & Hemicellulose Effect upon Biodegradable Bamboo Fiber Composites Due to Moisture. Mater. Today Proc. 2017, 4, 3222–3232. [Google Scholar] [CrossRef]
- A Statistical Approach to Develop Biocomposites from Epoxy Resin, Poly(Furfuryl Alcohol), Poly(Propylene Carbonate), and Biochar—Mashouf Roudsari—2017—Journal of Applied Polymer Science—Wiley Online Library. Available online: http://onlinelibrary.wiley.com/doi/10.1002/app.45307/full (accessed on 7 December 2017).
- Biocomposites Based on Polylactic Acid and Olive Solid Waste Fillers: Effect of Two Compatibilization Approaches on the Physicochemical, Rheological and Mechanical Properties—Khemakhem—2016—Polymer Composites—Wiley Online Library. Available online: http://onlinelibrary.wiley.com/doi/10.1002/pc.24094/full (accessed on 7 December 2017).
- A Study of the Thermal and Water Diffusivity Properties of Cellulose Nanofibril Reinforced Starch/PVA Bionanocomposite Films—Das—2015—Advances in Polymer Technology—Wiley Online Library. Available online: http://onlinelibrary.wiley.com/doi/10.1002/adv.21552/full (accessed on 7 December 2017).
- A Sustainable and Resilient Approach through Biochar Addition in Wood Polymer Composites—ScienceDirect. Available online: http://www.sciencedirect.com/science/article/pii/S0048969715000789 (accessed on 8 December 2017).
- Adam, J.; Korneliusz, B.A.; Agnieszka, M. Dynamic Mechanical Thermal Analysis of Biocomposites Based on PLA and PHBV—A Comparative Study to PP Counterparts. J. Appl. Polym. Sci. 2013, 130, 3175–3183. [Google Scholar] [CrossRef]
- Effect of Nanoclay and Silica on Mechanical and Morphological Properties of Jute Cellulose Polyethylene Biocomposites—Rahman—2016—Journal of Vinyl and Additive Technology—Wiley Online Library. Available online: http://onlinelibrary.wiley.com/doi/10.1002/vnl.21554/full (accessed on 8 December 2017).
- Integration of Biobased Functionalized Feedstock and Plastisol in Epoxy Resin Matrix toward Developing Structural Jute Biocomposites with Enhanced Impact Strength and Moisture Resistance Properties—Bhosale—2014—Polymer Composites—Wiley Online Library. Available online: http://onlinelibrary.wiley.com/doi/10.1002/pc.23192/full (accessed on 8 December 2017).
- Ramzy, A.; Beermann, D.; Steuernagel, L.; Meiners, D.; Ziegmann, G. Developing a New Generation of Sisal Composite Fibres for Use in Industrial Applications. Compos. Part B Eng. 2014, 66, 287–298. [Google Scholar] [CrossRef]
- Biocomposites Market Research Report by Material Type, by Fiber Type, by Application—Global Forecast to 2025—Cumulative Impact of COVID-19. Available online: https://www.reportlinker.com/p05913586/Biocomposites-Market-Research-Report-by-Material-Type-by-Fiber-Type-by-Application-Global-Forecast-to-Cumulative-Impact-of-COVID-19.html?utm_source=GNW (accessed on 9 October 2020).
- An Investigation of Sound Absorption Coefficient on Sisal Fiber Poly Lactic Acid Bio-Composites—Jayamani—2015—Journal of Applied Polymer Science—Wiley Online Library. Available online: http://onlinelibrary.wiley.com/doi/10.1002/app.42470/full (accessed on 7 December 2017).
- A Review: Natural Fiber Composites Selection in View of Mechanical, Light Weight, and Economic Properties—Ahmad—2014—Macromolecular Materials and Engineering—Wiley Online Library. Available online: http://onlinelibrary.wiley.com/doi/10.1002/mame.201400089/full (accessed on 7 December 2017).
- Jatropha Deoiled Cake Filler-Reinforced Medium-Density Polyethylene Biocomposites: Effect of Filler Loading and Coupling Agent on the Mechanical, Dynamic Mechanical and Morphological Properties—Elshaarani—2013—Polymer Composites—Wiley Online Library. Available online: http://onlinelibrary.wiley.com/doi/10.1002/pc.22479/full (accessed on 8 December 2017).
- Jawaid, M.; Abdul Khalil, H.P.S. Cellulosic/Synthetic Fibre Reinforced Polymer Hybrid Composites: A Review. Carbohydr. Polym. 2011, 86, 1–18. [Google Scholar] [CrossRef]
- Soykeabkaew, N.; Arimoto, N.; Nishino, T.; Peijs, T. All-Cellulose Composites by Surface Selective Dissolution of Aligned Ligno-Cellulosic Fibres. Compos. Sci. Technol. 2008, 68, 2201–2207. [Google Scholar] [CrossRef]
- Darder, M.; Aranda, P.; Ruiz-Hitzky, E. Bionanocomposites: A New Concept of Ecological, Bioinspired, and Functional Hybrid Materials. Adv. Mater. 2007, 19, 1309–1319. [Google Scholar] [CrossRef]
- Faruk, O.; Bledzki, A.K.; Fink, H.-P.; Sain, M. Biocomposites Reinforced with Natural Fibers: 2000–2010. Prog. Polym. Sci. 2012, 37, 1552–1596. [Google Scholar] [CrossRef]
- Sharma, A.K.; Bhandari, R.; Aherwar, A.; Rimašauskienė, R. Matrix Materials Used in Composites: A Comprehensive Study. Mater. Today Proc. 2020, 21, 1559–1562. [Google Scholar] [CrossRef]
- Microfibrillated Cellulose—Its Barrier Properties and Applications in Cellulosic Materials: A Review—ScienceDirect. Available online: http://www.sciencedirect.com/science/article/pii/S014486171200447X (accessed on 8 December 2017).
- Satyanarayana, K.G.; Arizaga, G.G.; Wypych, F. Biodegradable Composites Based on Lignocellulosic Fibers—An Overview. Prog. Polym. Sci. 2009, 34, 982–1021. [Google Scholar] [CrossRef]
- Norgren, M.; Edlund, H. Lignin: Recent Advances and Emerging Applications. Curr. Opin. Colloid Interface Sci. 2014, 19, 409–416. [Google Scholar] [CrossRef]
- Kumar, S.; Mohanty, A.K.; Erickson, L.; Misra, M. Lignin and Its Applications with Polymers. J. Biobased Mater. Bioenergy 2009, 3, 1–24. [Google Scholar] [CrossRef]
- Li, S.; Xiao, M.; Zheng, A.; Xiao, H. Cellulose Microfibrils Grafted with PBA via Surface-Initiated Atom Transfer Radical Polymerization for Biocomposite Reinforcement. Biomacromolecules 2011, 12, 3305–3312. [Google Scholar] [CrossRef]
- Sajith, S.; Arumugam, V.; Dhakal, H.N. Comparison on Mechanical Properties of Lignocellulosic Flour Epoxy Composites Prepared by Using Coconut Shell, Rice Husk and Teakwood as Fillers. Polym. Test. 2017, 58, 60–69. [Google Scholar] [CrossRef]
- Reddy, N.; Yang, Y. Biofibers from Agricultural Byproducts for Industrial Applications. Trends Biotechnol. 2005, 23, 22–27. [Google Scholar] [CrossRef]
- Célino, A.; Freour, S.; Jacquemin, F.; Casari, P. The Hygroscopic Behavior of Plant Fibers: A Review. Front. Chem. 2014, 1. [Google Scholar] [CrossRef]
- Biocomposites from Abaca Strands and Polypropylene. Part I: Evaluation of the Tensile Properties—ScienceDirect. Available online: http://www.sciencedirect.com/science/article/pii/S0960852409009638 (accessed on 9 December 2017).
- Biocomposites Containing Cellulose Fibers Treated with Nanosized Elastomeric Latex for Enhancing Impact Strength—ScienceDirect. Available online: http://www.sciencedirect.com/science/article/pii/S0266353813000328 (accessed on 9 December 2017).
- Signori, F.; Pelagaggi, M.; Bronco, S.; Righetti, M.C. Amorphous/Crystal and Polymer/Filler Interphases in Biocomposites from Poly (Butylene Succinate). Thermochim. Acta 2012, 543, 74–81. [Google Scholar] [CrossRef]
- Biocomposites from Wheat Proteins and Fibers: Structure/Mechanical Properties Relationships—ScienceDirect. Available online: http://www.sciencedirect.com/science/article/pii/S0926669012004438 (accessed on 9 December 2017).
- Kellersztein, I.; Amir, E.; Dotan, A. Grafting of Wheat Straw Fibers with Poly (ε-Caprolactone) via Ring-Opening Polymerization for Poly(Lactic Acid) Reinforcement. Polym. Adv. Technol. 2016, 27, 657–664. [Google Scholar] [CrossRef]
- Cunha, M.; Berthet, M.-A.; Pereira, R.; Covas, J.A.; Vicente, A.A.; Hilliou, L. Development of Polyhydroxyalkanoate/Beer Spent Grain Fibers Composites for Film Blowing Applications. Polym. Compos. 2015, 36, 1859–1865. [Google Scholar] [CrossRef]
- García, A.V.; Santonja, M.R.; Sanahuja, A.B.; Selva, M.D.C.G. Characterization and Degradation Characteristics of Poly(ε-Caprolactone)-Based Composites Reinforced with Almond Skin Residues. Polym. Degrad. Stab. 2014, 108, 269–279. [Google Scholar] [CrossRef]
- Ku, H.; Wang, H.; Pattarachaiyakoop, N.; Trada, M. A Review on the Tensile Properties of Natural Fiber Reinforced Polymer Composites. Compos. Part B Eng. 2011, 42, 856–873. [Google Scholar] [CrossRef]
- Biocomposites Based on Poly(Butylene Succinate) and Curaua: Mechanical and Morphological Properties—ScienceDirect. Available online: https://www.sciencedirect.com/science/article/pii/S0142941815001415 (accessed on 11 December 2017).
- Synthesis, Characterization and Properties of Novel Linear Poly(Butylene Fumarate) Bearing Reactive Double Bonds—Documents. Available online: https://docslide.net/documents/synthesis-characterization-and-properties-of-novel-linear-polybutylene-fumarate.html (accessed on 11 December 2017).
- Zhao, Q.; Tao, J.; Yam, R.C.M.; Mok, A.C.K.; Li, R.K.Y.; Song, C. Biodegradation Behavior of Polycaprolactone/Rice Husk Ecocomposites in Simulated Soil Medium. Polym. Degrad. Stab. 2008, 93, 1571–1576. [Google Scholar] [CrossRef]
- All-Cellulose and All-Wood Composites by Partial Dissolution of Cotton Fabric and Wood in Ionic Liquid—ScienceDirect. Available online: http://www.sciencedirect.com/science/article/pii/S0144861713007480 (accessed on 11 December 2017).
- Cocca, M.; Avolio, R.; Gentile, G.; Di Pace, E.; Errico, M.E.; Avella, M. Amorphized Cellulose as Filler in Biocomposites Based on Poly(ɛ-Caprolactone). Carbohydr. Polym. 2015, 118, 170–182. [Google Scholar] [CrossRef]
- Mastali, M.; Abdollahnejad, Z.; Pacheco-Torgal, F. Carbon Dioxide Sequestration of Fly Ash Alkaline-Based Mortars Containing Recycled Aggregates and Reinforced by Hemp Fibres. Constr. Build. Mater. 2018, 160, 48–56. [Google Scholar] [CrossRef]
- Mechanical and Thermal Properties of Polypropylene Reinforced with Doum Fiber: Impact of Fibrillization|SpringerLink. Available online: https://link.springer.com/chapter/10.1007/978-3-319-46610-1_11 (accessed on 9 December 2017).
- Thomas, S.; Woh, Y.-K.; Wang, R.; Goh, K.L. Probing the Hydrophilicity of Coir Fibres: Analysis of the Mechanical Properties of Single Coir Fibres. Procedia Eng. 2017, 200, 206–212. [Google Scholar] [CrossRef]
- Arfaoui, M.A.; Dolez, P.I.; Dubé, M.; David, É. Development and Characterization of a Hydrophobic Treatment for Jute Fibres Based on Zinc Oxide Nanoparticles and a Fatty Acid. Appl. Surf. Sci. 2017, 397, 19–29. [Google Scholar] [CrossRef]
- Quiles-Carrillo, L.; Montanes, N.; Sammon, C.; Balart, R.; Torres-Giner, S. Compatibilization of Highly Sustainable Polylactide/Almond Shell Flour Composites by Reactive Extrusion with Maleinized Linseed Oil. Ind. Crops Prod. 2017. [Google Scholar] [CrossRef]
- de Oliveira, J.P.; Bruni, G.P.; Lima, K.O.; Halal, S.L.M.E.; da Rosa, G.S.; Dias, A.R.G.; Zavareze, E.D.R. Cellulose Fibers Extracted from Rice and Oat Husks and Their Application in Hydrogel. Food Chem. 2017, 221, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Montaño-Leyva, B.; Gontard, N.; Angellier-Coussy, H. Poly (3-Hydroxybutyrate-Co-Hydroxyvalerate) and Wheat Straw Fibers Biocomposites Produced by Co-Grinding: Processing and Mechanical Behavior. J. Compos. Mater. 2017, 51, 985–996. [Google Scholar] [CrossRef]
- Biocomposites From Switchgrass and Lignin Hybrid and Poly(Butylene Succinate) Bioplastic: Studies on Reactive Compatibilization and Performance Evaluation—Sahoo—2013—Macromolecular Materials and Engineering—Wiley Online Library. Available online: http://onlinelibrary.wiley.com/doi/10.1002/mame.201300038/abstract (accessed on 9 December 2017).
- Spinacé, M.A.; De Paoli, M.-A. Biocomposite of a Multilayer Film Scrap and Curauá Fibers: Preparation and Environmental Degradation. J. Thermoplast. Compos. Mater. 2017, 30, 225–240. [Google Scholar] [CrossRef]
- Hashim, M.Y.; Amin, A.M.; Marwah, O.M.F.; Othman, M.H.; Yunus, M.R.M.; Huat, N.C. The Effect of Alkali Treatment under Various Conditions on Physical Properties of Kenaf Fiber. J. Phys. Conf. Ser. 2017, 914, 012030. [Google Scholar] [CrossRef]
- Pereira, G.B.; Pereira, G.C.; Lima, M.; Jesus, B.; Silva, E.D.A.; Benini, K.C.C.; Bandeira, C.; Montoro, S.R. Featuring High Impact Polystyrene Composites Strengthened with Green Coconut Fiber Developed for Automotive Industry Application. J. Res. Updat. Polym. Sci. 2017, 6, 17–20. [Google Scholar]
- Inoue, K.; Bigeard, A.; Hirogaki, T.; Aoyama, E.; Ogawa, K.; Nobe, H. Fabrication of Complex-Shape Products From a Binder-Free Green Composite Using Bamboo Fibers and Powders Extracted with a Machining Center. Am. Soc. Mech. Eng. 2017, 58165, V004T05A023. [Google Scholar] [CrossRef]
- Mohammadkazemi, F.; Aguiar, R.; Cordeiro, N. Improvement of Bagasse Fiber–Cement Composites by Addition of Bacterial Nanocellulose: An Inverse Gas Chromatography Study. Cellulose 2017, 24, 1803–1814. [Google Scholar] [CrossRef]
- KOH Activated Carbon Derived from Biomass-Banana Fibers as an Efficient Negative Electrode in High Performance Asymmetric Supercapacitor—ScienceDirect. Available online: http://www.sciencedirect.com/science/article/pii/S2095495616300961 (accessed on 9 December 2017).
- Patil, N.V.; Rahman, M.M.; Netravali, A.N. “Green” Composites Using Bioresins from Agro-Wastes and Modified Sisal Fibers. Polym. Compos. 2019. [Google Scholar] [CrossRef]
- Low-Density Polyethylene/Sugarcane Fiber Composites from Recycled Polymer and Treated Fiber by Steam Explosion: Journal of Natural Fibers. Available online: http://www.tandfonline.com/doi/abs/10.1080/15440478.2017.1379044 (accessed on 9 December 2017).
- Gallos, A.; Paës, G.; Allais, F.; Beaugrand, J. Lignocellulosic Fibers: A Critical Review of the Extrusion Process for Enhancement of the Properties of Natural Fiber Composites. RSC Adv. 2017, 7, 34638–34654. [Google Scholar] [CrossRef]
- Fengel, D.; Wegener, G. Wood—Chemistry, Ultrastructure, Reactions; Walter de Gruyter: Berlin, Germany; New York, NY, USA, 1984; Volume 613, Available online: http://onlinelibrary.wiley.com/doi/10.1002/pol.1985.130231112/abstract (accessed on 15 December 2017).
- John, M.J.; Thomas, S. Biofibres and Biocomposites. Carbohydr. Polym. 2008, 71, 343–364. [Google Scholar] [CrossRef]
- Abdul Khalil, H.P.S.; Davoudpour, Y.; Islam, M.d.N.; Mustapha, A.; Sudesh, K.; Dungani, R.; Jawaid, M. Production and Modification of Nanofibrillated Cellulose Using Various Mechanical Processes: A Review. Carbohydr. Polym. 2014, 99, 649–665. [Google Scholar] [CrossRef] [PubMed]
- Kalia, S.; Dufresne, A.; Cherian, B.M.; Kaith, B.S.; Avérous, L.; Njuguna, J.; Nassiopoulos, E. Cellulose-Based Bio- and Nanocomposites: A Review. Available online: https://www.hindawi.com/journals/ijps/2011/837875/ (accessed on 15 December 2017).
- Khalil, H.A.; Bhat, A.H.; Yusra, A.I. Green Composites from Sustainable Cellulose Nanofibrils: A Review. Carbohydr. Polym. 2012, 87, 963–979. [Google Scholar] [CrossRef]
- Dufresne, A. Nanocellulose: A New Ageless Bionanomaterial. Mater. Today 2013, 16, 220–227. [Google Scholar] [CrossRef]
- Hansen, C.M.; Björkman, A. The Ultrastructure of Wood from a Solubility Parameter Point of View. Holzforsch. Int. J. Biol. Chem. Phys. Technol. Wood 2009, 52, 335–344. [Google Scholar] [CrossRef]
- Rowell, R.M.; Han, J.S.; Rowell, J.S. Characterization and Factors Effecting Fiber Properties. In Natural Polymers and Agrofibers Based Composites; Embrapa Instrumentacao Agropecu: Sao Carlos, Brazil, 2000; pp. 115–134. [Google Scholar]
- Yan, L.; Kasal, B.; Huang, L. A Review of Recent Research on the Use of Cellulosic Fibres, Their Fibre Fabric Reinforced Cementitious, Geo-Polymer and Polymer Composites in Civil Engineering. Compos. Part B Eng. 2016, 92, 94–132. [Google Scholar] [CrossRef]
- Dittenber, D.B.; GangaRao, H.V. Critical Review of Recent Publications on Use of Natural Composites in Infrastructure. Compos. Part Appl. Sci. Manuf. 2012, 43, 1419–1429. [Google Scholar] [CrossRef]
- John, M.J.; Anandjiwala, R.D. Recent Developments in Chemical Modification and Characterization of Natural Fiber-Reinforced Composites. Polym. Compos. 2008, 29, 187–207. [Google Scholar] [CrossRef]
- Feughelman, M. Mechanical Properties and Structure of Alpha-Keratin Fibres: Wool, Human Hair and Related Fibres; UNSW Press: Sydney, Australia, 1997. [Google Scholar]
- Fakirov, S.; Bhattacharyya, D. Handbook of Engineering Biopolymers: ’Homopolymers, Blends, and Composites; Bhattacharya, D., Ed.; Hanser Publications: Munich, Germany; Cincinnati, OH, USA, 2007; ISBN 978-1-56990-405-3. [Google Scholar]
- Badrinath, R.; Senthilvelan, T. Comparative Investigation on Mechanical Properties of Banana and Sisal Reinforced Polymer Based Composites. Procedia Mater. Sci. 2014, 5, 2263–2272. [Google Scholar] [CrossRef]
- Cherian, B.M.; Leão, A.L.; de Souza, S.F.; Thomas, S.; Pothan, L.A.; Kottaisamy, M. Isolation of Nanocellulose from Pineapple Leaf Fibres by Steam Explosion. Carbohydr. Polym. 2010, 81, 720–725. [Google Scholar] [CrossRef]
- Ramesh, M.; Palanikumar, K.; Reddy, K.H. Plant Fibre Based Bio-Composites: Sustainable and Renewable Green Materials. Renew. Sustain. Energy Rev. 2017, 79, 558–584. [Google Scholar] [CrossRef]
- Ramakrishna, G.; Sundararajan, T. Impact Strength of a Few Natural Fibre Reinforced Cement Mortar Slabs: A Comparative Study. Cem. Concr. Compos. 2005, 27, 547–553. [Google Scholar] [CrossRef]
- Naidu, A.L.; Raghuveer, D.; Suman, P. Studies on Characterization and Mechanical Behavior of Banana Peel Reinforced Epoxy Composites. Int. J. Sci. Eng. Res. 2013, 4, 844. [Google Scholar]
- Athijayamani, A.; Thiruchitrambalam, M.; Natarajan, U.; Pazhanivel, B. Effect of Moisture Absorption on the Mechanical Properties of Randomly Oriented Natural Fibers/Polyester Hybrid Composite. Mater. Sci. Eng. A 2009, 517, 344–353. [Google Scholar] [CrossRef]
- Kozłowski, R.; Władyka-Przybylak, M. Flammability and Fire Resistance of Composites Reinforced by Natural Fibers. Polym. Adv. Technol. 2008, 19, 446–453. [Google Scholar] [CrossRef]
- Manikandan, N.; Morshed, M.N.; Karthik, R.; Al Azad, S.; Deb, H.; Rumi, T.M.; Ahmed, M.R. Improvement of Mechanical Properties of Natural Fiber Reinforced Jute/Polyester Epoxy Composite through Meticulous Alkali Treatment. Am. J. Curr. Org. Chem. 2017, 3, 9–18. [Google Scholar]
- Joseph, S.; Oommen, Z.; Thomas, S. Environmental Durability of Banana-Fiber-Reinforced Phenol Formaldehyde Composites. J. Appl. Polym. Sci. 2006, 100, 2521–2531. [Google Scholar] [CrossRef]
- Popelka, A.; Novák, I.; Al-Maadeed, M.A.S.; Ouederni, M.; Krupa, I. Effect of Corona Treatment on Adhesion Enhancement of LLDPE. Surf. Coat. Technol. 2017, 335, 118–125. [Google Scholar] [CrossRef]
- Ragoubi, M.; Bienaimé, D.; Molina, S.; George, B.; Merlin, A. Impact of Corona Treated Hemp Fibres onto Mechanical Properties of Polypropylene Composites Made Thereof. Ind. Crops Prod. 2010, 31, 344–349. [Google Scholar] [CrossRef]
- Marais, S.; Gouanvé, F.; Bonnesoeur, A.; Grenet, J.; Poncin-Epaillard, F.; Morvan, C.; Métayer, M. Unsaturated Polyester Composites Reinforced with Flax Fibers: Effect of Cold Plasma and Autoclave Treatments on Mechanical and Permeation Properties. Compos. Part Appl. Sci. Manuf. 2005, 36, 975–986. [Google Scholar] [CrossRef]
- Seki, Y.; Sever, K.; Sarikanat, M.; Güleç, H.A.; Tavman, I.H. The Influence of Oxygen Plasma Treatment of Jute Fibers on Mechanical Properties of Jute Fiber Reinforced Thermoplastic Composites. In Proceedings of the 5th International Advanced Technologies Symposium (IATS’09), Karabuk, Turkey, 13–15 May 2009. [Google Scholar]
- Bilba, K.; Arsene, M.-A. Silane Treatment of Bagasse Fiber for Reinforcement of Cementitious Composites. Compos. Part Appl. Sci. Manuf. 2008, 39, 1488–1495. [Google Scholar] [CrossRef]
- Law, T.T.; Ishak, Z.A. Water Absorption and Dimensional Stability of Short Kenaf Fiber-Filled Polypropylene Composites Treated with Maleated Polypropylene. J. Appl. Polym. Sci. 2011, 120, 563–572. [Google Scholar] [CrossRef]
- Pommet, M.; Juntaro, J.; Heng, J.Y.; Mantalaris, A.; Lee, A.F.; Wilson, K.; Kalinka, G.; Shaffer, M.S.; Bismarck, A. Surface Modification of Natural Fibers Using Bacteria: Depositing Bacterial Cellulose onto Natural Fibers to Create Hierarchical Fiber Reinforced Nanocomposites. Biomacromolecules 2008, 9, 1643–1651. [Google Scholar] [CrossRef] [PubMed]
- Joseph, K.; Mattoso, L.H.C.; Toledo, R.D.; Thomas, S.; De Carvalho, L.H.; Pothen, L.; Kala, S.; James, B. Natural Fiber Reinforced Thermoplastic Composites. Nat. Polym. Agrofibers Compos. 2000, 159, 159–201. [Google Scholar]
- Mohanty, A.K.; Misra, M.; Drzal, L.T. Surface Modifications of Natural Fibers and Performance of the Resulting Biocomposites: An Overview. Compos. Interfaces 2001, 8, 313–343. [Google Scholar] [CrossRef]
- Valadez-Gonzalez, A.; Cervantes-Uc, J.M.; Olayo, R.; Herrera-Franco, P.J. Chemical Modification of Henequen Fibers with an Organosilane Coupling Agent. Compos. Part B Eng. 1999, 30, 321–331. [Google Scholar] [CrossRef]
- Valadez-Gonzalez, A.; Cervantes-Uc, J.M.; Olayo, R.; Herrera-Franco, P.J. Effect of Fiber Surface Treatment on the Fiber–Matrix Bond Strength of Natural Fiber Reinforced Composites. Compos. Part B Eng. 1999, 30, 309–320. [Google Scholar] [CrossRef]
- Mishra, S.; Mohanty, A.K.; Drzal, L.T.; Misra, M.; Parija, S.; Nayak, S.K.; Tripathy, S.S. Studies on Mechanical Performance of Biofibre/Glass Reinforced Polyester Hybrid Composites. Compos. Sci. Technol. 2003, 63, 1377–1385. [Google Scholar] [CrossRef]
- Kabir, M.M.; Wang, H.; Lau, K.T.; Cardona, F. Chemical Treatments on Plant-Based Natural Fibre Reinforced Polymer Composites: An Overview. Compos. Part B Eng. 2012, 43, 2883–2892. [Google Scholar] [CrossRef]
- Wang, B.; Panigrahi, S.; Crerar, W.; Tabil, L. Application of Pre-Treated Flax Fibers in Composites. In Proceedings of the CSAE SCGR, Montréal, QC, Canada, 6–9 July 2003. [Google Scholar]
- Kohli, D.; Garg, S.; Jana, A.K.; Maiti, M. Synthesis of Graft Copolymers for Green Composite Films and Optimization of Reaction Parameters Using Taguchi (L16) Orthogonal Array. Indian Chem. Eng. 2017, 59, 136–158. [Google Scholar] [CrossRef]
- Beyaz, K.; Charton, M.; Rouilly, A.; Vedrenne, E.; Vaca-Garcia, C.; Benaboura, A.; Thiebaud-Roux, S. Synthesis of Graft-Copolymers from Palm Cellulose and Solketal Acrylate and Their Characterization. Ind. Crops Prod. 2017, 97, 32–40. [Google Scholar] [CrossRef]
- Rahman, M.M.; Mallik, A.K.; Khan, M.A. Influences of Various Surface Pretreatments on the Mechanical and Degradable Properties of Photografted Oil Palm Fibers. J. Appl. Polym. Sci. 2007, 105, 3077–3086. [Google Scholar] [CrossRef]
- Kalia, S.; Kaith, B.S.; Kaur, I. Pretreatments of Natural Fibers and Their Application as Reinforcing Material in Polymer Composites—A Review. Polym. Eng. Sci. 2009, 49, 1253–1272. [Google Scholar] [CrossRef]
- Methacanon, P.; Weerawatsophon, U.; Sumransin, N.; Prahsarn, C.; Bergado, D.T. Properties and Potential Application of the Selected Natural Fibers as Limited Life Geotextiles. Carbohydr. Polym. 2010, 82, 1090–1096. [Google Scholar] [CrossRef]
- Zakaria, S.; Kok Poh, L. Polystyrene-Benzoylated EFB Reinforced Composites. Polym.-Plast. Technol. Eng. 2002, 41, 951–962. [Google Scholar] [CrossRef]
- Sgriccia, N.; Hawley, M.C.; Misra, M. Characterization of Natural Fiber Surfaces and Natural Fiber Composites. Compos. Part Appl. Sci. Manuf. 2008, 39, 1632–1637. [Google Scholar] [CrossRef]
- Hosseinaei, O.; Wang, S.; Enayati, A.A.; Rials, T.G. Effects of Hemicellulose Extraction on Properties of Wood Flour and Wood–Plastic Composites. Compos. Part Appl. Sci. Manuf. 2012, 43, 686–694. [Google Scholar] [CrossRef]
- Espert, A.; Vilaplana, F.; Karlsson, S. Comparison of Water Absorption in Natural Cellulosic Fibres from Wood and One-Year Crops in Polypropylene Composites and Its Influence on Their Mechanical Properties. Compos. Part Appl. Sci. Manuf. 2004, 35, 1267–1276. [Google Scholar] [CrossRef]
- Wang, Y.; Wei, Q.; Wang, S.; Chai, W.; Zhang, Y. Structural and Water Diffusion of Poly (Acryl Amide)/Poly (Vinyl Alcohol) Blend Films: Experiment and Molecular Dynamics Simulations. J. Mol. Graph. Model. 2017, 71, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Sain, M.; Cooper, P.A. Study of Moisture Absorption in Natural Fiber Plastic Composites. Compos. Sci. Technol. 2006, 66, 379–386. [Google Scholar] [CrossRef]
- Benard, P.; Kroener, E.; Vontobel, P.; Kaestner, A.; Carminati, A. Water Percolation through the Root-Soil Interface. Adv. Water Resour. 2016, 95, 190–198. [Google Scholar] [CrossRef]
- Gupta, A. Synthesis, Chemical Resistance, and Water Absorption of Bamboo Fiber Reinforced Epoxy Composites. Polym. Compos. 2016, 37, 141–145. [Google Scholar] [CrossRef]
- Dhakal, H.N.; Zhang, Z.Y.; Richardson, M.O.W. Effect of Water Absorption on the Mechanical Properties of Hemp Fibre Reinforced Unsaturated Polyester Composites. Compos. Sci. Technol. 2007, 67, 1674–1683. [Google Scholar] [CrossRef]
- Baltazar-y-Jimenez, A.; Awal, A.; Sain, M. Natural Composites: Fiber Modification. In Wiley Encyclopedia of Composites; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Pott, G.T.; Hueting, D.J.; Van Deursen, J.H. Reduction of Moisture Sensitivity in Wood and Natural Fibers for Polymer Composites. In Proceedings of the 3rd International Wood and Natural Fiber Composites Symposium, Kassel, Germany, 19–20 September 2000. [Google Scholar]
- Li, X.; Tabil, L.G.; Panigrahi, S. Chemical Treatments of Natural Fiber for Use in Natural Fiber-Reinforced Composites: A Review. J. Polym. Environ. 2007, 15, 25–33. [Google Scholar] [CrossRef]
- Lepetit, A.; Drolet, R.; Tolnai, B.; Zerrouki, R.; Montplaisir, D. Effect of Acetylation on the Properties of Microfibrillated Cellulose-LDPE Composites. J. Appl. Polym. Sci. 2017, 134, 44933. [Google Scholar] [CrossRef]
- Tserki, V.; Zafeiropoulos, N.E.; Simon, F.; Panayiotou, C. A Study of the Effect of Acetylation and Propionylation Surface Treatments on Natural Fibres. Compos. Part Appl. Sci. Manuf. 2005, 36, 1110–1118. [Google Scholar] [CrossRef]
- Khalil, H.A.; Ismail, H. Effect of Acetylation and Coupling Agent Treatments upon Biological Degradation of Plant Fibre Reinforced Polyester Composites. Polym. Test. 2000, 20, 65–75. [Google Scholar] [CrossRef]
- Kumar, D.R.; Mohanraj, P. Review on Natural Fiber in Various Pretreatment Conditions for Preparing Perfect Fiber. Asian J. Appl. Sci. Technol. AJAST 2017, 1, 66–78. [Google Scholar]
- Islam, M.N.; Ahmed, N.; Hossain, M.Y.; Rahman, A.; Sultana, A. Effect of PH on the Adsorption Kinetics of Cr (VI) on Sodium Chlorite Treated Coconut Coir. Bangladesh J. Sci. Ind. Res. 2016, 51, 95–100. [Google Scholar] [CrossRef][Green Version]
- Rambabu, N.; Panthapulakkal, S.; Sain, M.; Dalai, A.K. Production of Nanocellulose Fibers from Pinecone Biomass: Evaluation and Optimization of Chemical and Mechanical Treatment Conditions on Mechanical Properties of Nanocellulose Films. Ind. Crops Prod. 2016, 83, 746–754. [Google Scholar] [CrossRef]
- Kalaprasad, G.; Francis, B.; Thomas, S.; Kumar, C.R.; Pavithran, C.; Groeninckx, G.; Thomas, S. Effect of Fibre Length and Chemical Modifications on the Tensile Properties of Intimately Mixed Short Sisal/Glass Hybrid Fibre Reinforced Low Density Polyethylene Composites. Polym. Int. 2004, 53, 1624–1638. [Google Scholar] [CrossRef]
- Rajesh, M.; Pitchaimani, J. Mechanical Characterization of Natural Fiber Intra-Ply Fabric Polymer Composites: Influence of Chemical Modifications. J. Reinf. Plast. Compos. 2017, 36, 1651–1664. [Google Scholar] [CrossRef]
- Mengeloglu, F.; Karakus, K. Thermal Degradation, Mechanical Properties and Morphology of Wheat Straw Flour Filled Recycled Thermoplastic Composites. Sensors 2008, 8, 500–519. [Google Scholar] [CrossRef] [PubMed]
- Pandey, J.K.; Ahn, S.H.; Lee, C.S.; Mohanty, A.K.; Misra, M. Recent Advances in the Application of Natural Fiber Based Composites. Macromol. Mater. Eng. 2010, 295, 975–989. [Google Scholar] [CrossRef]
- Corrales, F.; Vilaseca, F.; Llop, M.; Girones, J.; Mendez, J.A.; Mutje, P. Chemical Modification of Jute Fibers for the Production of Green-Composites. J. Hazard. Mater. 2007, 144, 730–735. [Google Scholar] [CrossRef] [PubMed]
- Gulati, D.; Sain, M. Fungal-Modification of Natural Fibers: A Novel Method of Treating Natural Fibers for Composite Reinforcement. J. Polym. Environ. 2006, 14, 347–352. [Google Scholar] [CrossRef]
- Wang, W.; Sain, M.; Cooper, P.A. Hygrothermal Weathering of Rice Hull/HDPE Composites under Extreme Climatic Conditions. Polym. Degrad. Stab. 2005, 90, 540–545. [Google Scholar] [CrossRef]
- Hollaway, L.C. A Review of the Present and Future Utilisation of FRP Composites in the Civil Infrastructure with Reference to Their Important In-Service Properties. Constr. Build. Mater. 2010, 24, 2419–2445. [Google Scholar] [CrossRef]
- Stark, N.M.; White, R.H.; Mueller, S.A.; Osswald, T.A. Evaluation of Various Fire Retardants for Use in Wood Flour–Polyethylene Composites. Polym. Degrad. Stab. 2010, 95, 1903–1910. [Google Scholar] [CrossRef]
- Ngo, T.D.; Ton-That, M.T.; Hu, W. Innovative and Sustainable Approaches to Enhance Fire Resistance of Cellulosic Fibers for Green Polymer Composites. SAMPE J. 2013, 49, 31–37. [Google Scholar]
- Manfredi, L.B.; Rodríguez, E.; Wladyka-Przybylak, M.; Vazquez, A. Thermal Properties and Fire Resistance of Jute-Reinforced Composites. Compos. Interfaces 2010, 17, 663–675. [Google Scholar] [CrossRef]
- Azwa, Z.N.; Yousif, B.F. Characteristics of Kenaf Fibre/Epoxy Composites Subjected to Thermal Degradation. Polym. Degrad. Stab. 2013, 98, 2752–2759. [Google Scholar] [CrossRef]
- Azwa, Z.N.; Yousif, B.F.; Manalo, A.C.; Karunasena, W. A Review on the Degradability of Polymeric Composites Based on Natural Fibres. Mater. Des. 2013, 47, 424–442. [Google Scholar] [CrossRef]
- Manfredi, L.B.; Rodríguez, E.S.; Wladyka-Przybylak, M.; Vázquez, A. Thermal Degradation and Fire Resistance of Unsaturated Polyester, Modified Acrylic Resins and Their Composites with Natural Fibres. Polym. Degrad. Stab. 2006, 91, 255–261. [Google Scholar] [CrossRef]
- Suardana, N.P.G.; Ku, M.S.; Lim, J.K. Effects of Diammonium Phosphate on the Flammability and Mechanical Properties of Bio-Composites. Mater. Des. 2011, 32, 1990–1999. [Google Scholar] [CrossRef]
- Kozlowski, R.; Helwig, M. Progress in Fire Retardants for Lignocellulosic Materials. In Proceedings of the 6th Arab International Conference on Materials Science, Materials & Fire, Alexandria, Egypt, 22–25 March 1998; pp. 1–11. [Google Scholar]
- Garcia, M.; Hidalgo, J.; Garmendia, I.; García-Jaca, J. Wood–Plastics Composites with Better Fire Retardancy and Durability Performance. Compos. Part Appl. Sci. Manuf. 2009, 40, 1772–1776. [Google Scholar] [CrossRef]
- Hamid, M.R.Y.; Ab Ghani, M.H.; Ahmad, S. Effect of Antioxidants and Fire Retardants as Mineral Fillers on the Physical and Mechanical Properties of High Loading Hybrid Biocomposites Reinforced with Rice Husks and Sawdust. Ind. Crops Prod. 2012, 40, 96–102. [Google Scholar] [CrossRef]
- Vinod, B.; Anandajothi, M. Mechanical and Tribological Properties of Abaca-Roselle/Cardanol Formaldehyde Hybrid Composites. Mater. Res. Express 2020, 6, 125363. [Google Scholar] [CrossRef]
- Kencanawati, C.; Suardana, N.P.G.; Sugita, I.K.G.; Suyasa, I.W.B. Characteristics of Fiber Treatments on Tensile and Impact Strengths of Pine Resin/Areca Husk Fiber Biocomposites. Int. J. Civ. Eng. Technol. 2020, 11, 86–93. [Google Scholar]
- Sajna, V.P.; Mohanty, S.; Nayak, S.K. Influence of Nanoclay and Graft Copolymer on the Thermal and Flammability Properties of Poly (Lactic Acid)/Banana Fiber Biocomposites. J. Vinyl Addit. Technol. 2017, 23, E81–E91. [Google Scholar] [CrossRef]
- Fiore, V.; Scalici, T.; Valenza, A. Effect of Sodium Bicarbonate Treatment on Mechanical Properties of Flax-Reinforced Epoxy Composite Materials. J. Compos. Mater. 2018, 52, 1061–1072. [Google Scholar] [CrossRef]
- Dhakal, H.N.; Ismail, S.O.; Zhang, Z.; Barber, A.; Welsh, E.; Maigret, J.-E.; Beaugrand, J. Development of Sustainable Biodegradable Lignocellulosic Hemp Fiber/Polycaprolactone Biocomposites for Light Weight Applications. Compos. Part Appl. Sci. Manuf. 2018, 113, 350–358. [Google Scholar] [CrossRef]
- Lakshmanan, A.; Ghosh, R.K.; Dasgupta, S.; Chakraborty, S.; Ganguly, P.K. Optimization of Alkali Treatment Condition on Jute Fabric for the Development of Rigid Biocomposite. J. Ind. Text. 2018, 47, 640–655. [Google Scholar] [CrossRef]
- Abdellaoui, H.; Bensalah, H.; Raji, M.; Rodrigue, D.; Bouhfid, R. Laminated Epoxy Biocomposites Based on Clay and Jute Fibers. J. Bionic Eng. 2017, 14, 379–389. [Google Scholar] [CrossRef]
- Prakash, V.A.; Viswanthan, R. Fabrication and Characterization of Echinoidea Spike Particles and Kenaf Natural Fibre-Reinforced Azadirachta-Indica Blended Epoxy Multi-Hybrid Bio Composite. Compos. Part Appl. Sci. Manuf. 2019, 118, 317–326. [Google Scholar] [CrossRef]
- Debeli, D.K.; Qin, Z.; Guo, J. Study on the Pre-Treatment, Physical and Chemical Properties of Ramie Fibers Reinforced Poly (Lactic Acid)(PLA) Biocomposite. J. Nat. Fibers 2018, 15, 596–610. [Google Scholar] [CrossRef]
- Xie, Q.; Li, F.; Li, J.; Wang, L.; Li, Y.; Zhang, C.; Xu, J.; Chen, S. A New Biodegradable Sisal Fiber–Starch Packing Composite with Nest Structure. Carbohydr. Polym. 2018, 189, 56–64. [Google Scholar] [CrossRef]
- Rowell, R.M. Properties and Performance of Natural-Fibre Composites; Elsevier: Amsterdam, The Netherlands, 2008; pp. 4–36. [Google Scholar]
- Juarez, C.; Duran, A.; Valdez, P.; Fajardo, G. Performance of “Agave Lecheguilla” Natural Fiber in Portland Cement Composites Exposed to Severe Environment Conditions. Build. Environ. 2007, 42, 1151–1157. [Google Scholar] [CrossRef]
- Shalwan, A.; Yousif, B.F. In State of Art: Mechanical and Tribological Behaviour of Polymeric Composites Based on Natural Fibres. Mater. Des. 2013, 48, 14–24. [Google Scholar] [CrossRef]
- Saheb, D.N.; Jog, J.P. Natural Fiber Polymer Composites: A Review. Adv. Polym. Technol. 1999, 18, 351–363. [Google Scholar] [CrossRef]
- Sampathkumar, D.; Punyamurthy, R.; Bennehalli, B.; Venkateshappa, S.C. Effect of Esterification on Moisture Absorption of Single Areca Fiber. Int. J. Agric. Sci. 2012, 4, 227. [Google Scholar]
- Binoj, J.S.; Raj, R.E.; Sreenivasan, V.S.; Thusnavis, G.R. Morphological, Physical, Mechanical, Chemical and Thermal Characterization of Sustainable Indian Areca Fruit Husk Fibers (Areca catechu L.) as Potential Alternate for Hazardous Synthetic Fibers. J. Bionic Eng. 2016, 13, 156–165. [Google Scholar] [CrossRef]
- Stokke, D.D.; Wu, Q.; Han, G. Introduction to Wood and Natural Fiber Composites; John Wiley & Sons: Hoboken, NJ, USA, 2013. [Google Scholar]
- Yu, Y.; Wang, H.; Lu, F.; Tian, G.; Lin, J. Bamboo Fibers for Composite Applications: A Mechanical and Morphological Investigation. J. Mater. Sci. 2014, 49, 2559–2566. [Google Scholar] [CrossRef]
- Huda, M.S.; Drzal, L.T.; Mohanty, A.K.; Misra, M. Chopped Glass and Recycled Newspaper as Reinforcement Fibers in Injection Molded Poly (Lactic Acid)(PLA) Composites: A Comparative Study. Compos. Sci. Technol. 2006, 66, 1813–1824. [Google Scholar] [CrossRef]
- Zahn, H.; Föhles, J.; Nlenhaus, M.; Schwan, A.; Spel, M. Wool as a Biological Composite Structure. Ind. Eng. Chem. Prod. Res. Dev. 1980, 19, 496–501. [Google Scholar] [CrossRef]
- Kumar, R.; Obrai, S.; Sharma, A. Chemical Modifications of Natural Fiber for Composite Material. Chem. Sin. 2011, 2, 219–228. [Google Scholar]
- Mohanty, A.K.; Misra, M.; Hinrichsen, G. Biofibers, Biodegradable Polymers and Biocomposites: An Overview. Macromol. Mater. Eng. 2000, 276, 1–24. [Google Scholar] [CrossRef]
- Akil, H.; Omar, M.F.; Mazuki, A.A.M.; Safiee, S.; Ishak, Z.M.; Bakar, A.A. Kenaf Fiber Reinforced Composites: A Review. Mater. Des. 2011, 32, 4107–4121. [Google Scholar] [CrossRef]
- Wambua, P.; Ivens, J.; Verpoest, I. Natural Fibres: Can They Replace Glass in Fibre Reinforced Plastics? Compos. Sci. Technol. 2003, 63, 1259–1264. [Google Scholar] [CrossRef]
- Gurunathan, T.; Mohanty, S.; Nayak, S.K. A Review of the Recent Developments in Biocomposites Based on Natural Fibres and Their Application Perspectives. Compos. Part Appl. Sci. Manuf. 2015, 77, 1–25. [Google Scholar] [CrossRef]
- Summerscales, J.; Dissanayake, N.; Virk, A.; Hall, W. A Review of Bast Fibres and Their Composites. Part 2–Composites. Compos. Part Appl. Sci. Manuf. 2010, 41, 1336–1344. [Google Scholar] [CrossRef]
- Rowell, R.M.; Sanadi, A.R.; Caulfield, D.F.; Jacobson, R.E. Utilization of Natural Fibers in Plastic Composites: Problems and Opportunities. Lignocellul.-Plast. Compos. 1997, 13, 23–51. [Google Scholar]
- Joshy, M.K.; Mathew, L.; Joseph, R. Influence of Fiber Surface Modification on the Mechanical Performance of Isora-Polyester Composites. Int. J. Polym. Mater. 2008, 58, 2–20. [Google Scholar] [CrossRef]
- Narayan, R. Biobased and Biodegradable Polymer Materials: Principles, Concepts and Technology Exemplars. In Proceedings of the World Polymer Congress and 41st International Symposium on Macromolecules, MACRO-2006, Rio de Janeiro, Brazil, 16–21 July 2006. [Google Scholar]
- Netravali, A.N.; Chabba, S. Composites Get Greener. Mater. Today 2003, 6, 22–29. [Google Scholar] [CrossRef]
- Saad, G.R.; Seliger, H. Biodegradable Copolymers Based on Bacterial Poly ((R)-3-Hydroxybutyrate): Thermal and Mechanical Properties and Biodegradation Behaviour. Polym. Degrad. Stab. 2004, 83, 101–110. [Google Scholar] [CrossRef]
- Mohanty, A.K.; Misra, M.; Drzal, L.T. Sustainable Bio-Composites from Renewable Resources: Opportunities and Challenges in the Green Materials World. J. Polym. Environ. 2002, 10, 19–26. [Google Scholar] [CrossRef]
- Van de Velde, K.; Kiekens, P. Biopolymers: Overview of Several Properties and Consequences on Their Applications. Polym. Test. 2002, 21, 433–442. [Google Scholar] [CrossRef]
- Alavudeen, A.; Rajini, N.; Karthikeyan, S.; Thiruchitrambalam, M.; Venkateshwaren, N. Mechanical Properties of Banana/Kenaf Fiber-Reinforced Hybrid Polyester Composites: Effect of Woven Fabric and Random Orientation. Mater. Des. 1980–2015 2015, 66, 246–257. [Google Scholar] [CrossRef]
- Eagan, J.M.; Xu, J.; Di Girolamo, R.; Thurber, C.M.; Macosko, C.W.; LaPointe, A.M.; Bates, F.S.; Coates, G.W. Combining Polyethylene and Polypropylene: Enhanced Performance with PE/IPP Multiblock Polymers. Science 2017, 355, 814–816. [Google Scholar] [CrossRef]
- Carrillo-Escalante, H.J.; Alvarez-Castillo, A.; Valadez-Gonzalez, A.; Herrera-Franco, P.J. Effect of Fiber-Matrix Adhesion on the Fracture Behavior of a Carbon Fiber Reinforced Thermoplastic-Modified Epoxy Matrix. Carbon Lett. 2016, 19, 47–56. [Google Scholar] [CrossRef][Green Version]
- Isikgor, F.H.; Becer, C.R. Lignocellulosic Biomass: A Sustainable Platform for the Production of Bio-Based Chemicals and Polymers. Polym. Chem. 2015, 6, 4497–4559. [Google Scholar] [CrossRef]
- Jawaid, M.; Paridah, M.T.; Saba, N. 1.1 Biomass, Source and Its Compositions; Universiti Putra Malaysia: Serdang, Malaysia, 2017. [Google Scholar]
- Miller, S.A.; Landis, A.E.; Theis, T.L. Feature: Environmental Trade-Offs of Biobased Production; ACS Publications: Washington, DC, USA, 2007. [Google Scholar]
- Geyer, R. Chapter 2—Production, use, and fate of synthetic polymers. In Plastic Waste and Recycling; Letcher, T.M., Ed.; Academic Press: Cambridge, MA, USA, 2020; pp. 13–32. ISBN 978-0-12-817880-5. [Google Scholar]
- Xu, Y.; Xu, J.; Liu, D.; Guo, B.; Xie, X. Synthesis and Characterization of Biodegradable Poly (Butylene Succinate-Co-Propylene Succinate) s. J. Appl. Polym. Sci. 2008, 109, 1881–1889. [Google Scholar] [CrossRef]
- Hottle, T.A.; Bilec, M.M.; Landis, A.E. Sustainability Assessments of Bio-Based Polymers. Polym. Degrad. Stab. 2013, 98, 1898–1907. [Google Scholar] [CrossRef]
- Xu, J.; Guo, B.-H. Microbial succinic acid, its polymer poly (butylene succinate), and applications. In Plastics from Bacteria; Springer: Berlin/Heidelberg, Germany, 2010; pp. 347–388. [Google Scholar]
- Xu, J.; Guo, B.-H. Poly (Butylene Succinate) and Its Copolymers: Research, Development and Industrialization. Biotechnol. J. 2010, 5, 1149–1163. [Google Scholar] [CrossRef] [PubMed]
- Cukalovic, A.; Stevens, C.V. Feasibility of Production Methods for Succinic Acid Derivatives: A Marriage of Renewable Resources and Chemical Technology. Biofuels Bioprod. Biorefining 2008, 2, 505–529. [Google Scholar] [CrossRef]
- Zeikus, J.G.; Jain, M.K.; Elankovan, P. Biotechnology of Succinic Acid Production and Markets for Derived Industrial Products. Appl. Microbiol. Biotechnol. 1999, 51, 545–552. [Google Scholar] [CrossRef]
- Lee, P.C.; Lee, W.G.; Lee, S.Y.; Chang, H.N. Succinic Acid Production with Reduced By-Product Formation in the Fermentation of Anaerobiospirillum Succiniciproducens Using Glycerol as a Carbon Source. Biotechnol. Bioeng. 2001, 72, 41–48. [Google Scholar] [CrossRef]
- Biosuccinic Acid Ready for Take Off? Available online: https://www.chemistryworld.com/news/biosuccinic-acid-ready-for-take-off/3000559.article (accessed on 28 December 2017).
- Haas, T.; Jaeger, B.; Weber, R.; Mitchell, S.F.; King, C.F. New Diol Processes: 1,3-Propanediol and 1,4-Butanediol. Appl. Catal. Gen. 2005, 280, 83–88. [Google Scholar] [CrossRef]
- Tserki, V.; Matzinos, P.; Pavlidou, E.; Vachliotis, D.; Panayiotou, C. Biodegradable Aliphatic Polyesters. Part I. Properties and Biodegradation of Poly (Butylene Succinate-Co-Butylene Adipate). Polym. Degrad. Stab. 2006, 91, 367–376. [Google Scholar] [CrossRef]
- Takahashi, H.; Hayakawa, T.; Ueda, M. Convenient Synthesis of Poly (Butylene Succinate) Catalyzed by Distannoxane. Chem. Lett. 2000, 29, 684–685. [Google Scholar] [CrossRef]
- Papageorgiou, G.Z.; Bikiaris, D.N. Crystallization and Melting Behavior of Three Biodegradable Poly (Alkylene Succinates). A Comparative Study. Polymer 2005, 46, 12081–12092. [Google Scholar] [CrossRef]
- Deng, L.-M.; Wang, Y.-Z.; Yang, K.-K.; Wang, X.-L.; Zhou, Q.; Ding, S.-D. A New Biodegradable Copolyester Poly (Butylene Succinate-Co-Ethylene Succinate-Co-Ethylene Terephthalate). Acta Mater. 2004, 52, 5871–5878. [Google Scholar] [CrossRef]
- Nikolic, M.S.; Djonlagic, J. Synthesis and Characterization of Biodegradable Poly (Butylene Succinate-Co-Butylene Adipate) s. Polym. Degrad. Stab. 2001, 74, 263–270. [Google Scholar] [CrossRef]
- Han, Y.-K.; Kim, S.-R.; Kim, J. Preparation and Characterization of High Molecular Weight Poly (Butylene Succinate). Macromol. Res. 2002, 10, 108–114. [Google Scholar] [CrossRef]
- Li, F.; Xu, X.; Yu, J.; Cao, A. The Morphological Effects upon Enzymatic Degradation of Poly (Butylene Succinate-Co-Butylene Terephthalate) s (PBST). Polym. Degrad. Stab. 2007, 92, 1053–1060. [Google Scholar] [CrossRef]
- Bhatia, A.; Gupta, R.; Bhattacharya, S.; Choi, H. Compatibility of Biodegradable Poly (Lactic Acid)(PLA) and Poly (Butylene Succinate)(PBS) Blends for Packaging Application. Korea-Aust. Rheol. J. 2007, 19, 125–131. [Google Scholar]
- Cao, A.; Okamura, T.; Nakayama, K.; Inoue, Y.; Masuda, T. Studies on Syntheses and Physical Properties of Biodegradable Aliphatic Poly (Butylene Succinate-Co-Ethylene Succinate) s and Poly (Butylene Succinate-Co-Diethylene Glycol Succinate) s. Polym. Degrad. Stab. 2002, 78, 107–117. [Google Scholar] [CrossRef]
- Mathew, A.P.; Oksman, K.; Sain, M. Mechanical Properties of Biodegradable Composites from Poly Lactic Acid (PLA) and Microcrystalline Cellulose (MCC). J. Appl. Polym. Sci. 2005, 97, 2014–2025. [Google Scholar] [CrossRef]
- Tsuji, H. Poly (Lactic Acid). Bio-Based Plast. Mater. Appl. 2013, 171–239. [Google Scholar]
- Garlotta, D. A Literature Review of Poly (Lactic Acid). J. Polym. Environ. 2001, 9, 63–84. [Google Scholar] [CrossRef]
- Rasal, R.M.; Janorkar, A.V.; Hirt, D.E. Poly (Lactic Acid) Modifications. Prog. Polym. Sci. 2010, 35, 338–356. [Google Scholar] [CrossRef]
- Lunt, J.; Shafer, A.L. Polyactic Acid Polymers from Corn-Applications in the Textiles Industry; Cargill Dow Polymers LLC: Minnetonka, MN, USA. Available online: http://www.jimluntllc.com/pdfs/PolylacticAcidPolymersFromCorn.pdf (accessed on 1 December 2020).
- Gupta, B.; Revagade, N.; Hilborn, J. Poly (Lactic Acid) Fiber: An Overview. Prog. Polym. Sci. 2007, 32, 455–482. [Google Scholar] [CrossRef]
- Griffith, L.G. Polymeric Biomaterials. Acta Mater. 2000, 48, 263–277. [Google Scholar] [CrossRef]
- Linnemann, B.; Sri Harwoko, M.; Gries, T. FIBER TABLE-Fiber Table Polylactide Fibers (PLA). Chem. Fibers Int. 2003, 53, 426–433. [Google Scholar]
- Tsuji, H. Polyesters 3 (Biopolymers, Vol. 4); Wiley-VCH GmbH: Weinheim, Germany, 2002; pp. 129–177. [Google Scholar]
- Jamshidian, M.; Tehrany, E.A.; Imran, M.; Jacquot, M.; Desobry, S. Poly-Lactic Acid: Production, Applications, Nanocomposites, and Release Studies. Compr. Rev. Food Sci. Food Saf. 2010, 9, 552–571. [Google Scholar] [CrossRef]
- Lasprilla, A.J.; Martinez, G.A.; Lunelli, B.H.; Jardini, A.L.; Maciel Filho, R. Poly-Lactic Acid Synthesis for Application in Biomedical Devices—A Review. Biotechnol. Adv. 2012, 30, 321–328. [Google Scholar] [CrossRef]
- Richard, F.G.; Nwabunma, D. Poly (Lactic Acid) Synthesis, Structures, Properties, Processing, and Application; Wiley Series on Polymer Engineering and Technology; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2010. [Google Scholar]
- Kim, E.; Shin, E.W.; Yoo, I.-K.; Chung, J.S. Characteristics of Heterogeneous Titanium Alkoxide Catalysts for Ring-Opening Polymerization of Lactide to Produce Polylactide. J. Mol. Catal. Chem. 2009, 298, 36–39. [Google Scholar] [CrossRef]
- Löfgren, A.; Albertsson, A.-C.; Dubois, P.; Jérôme, R. Recent Advances in Ring-Opening Polymerization of Lactones and Related Compounds. J. Macromol. Sci. Part C Polym. Rev. 1995, 35, 379–418. [Google Scholar] [CrossRef]
- Auras, R.; Harte, B.; Selke, S. An Overview of Polylactides as Packaging Materials. Macromol. Biosci. 2004, 4, 835–864. [Google Scholar] [CrossRef]
- Malmgren, T.; Mays, J.; Pyda, M. Characterization of Poly (Lactic Acid) by Size Exclusion Chromatography, Differential Refractometry, Light Scattering and Thermal Analysis. J. Therm. Anal. Calorim. 2006, 83, 35–40. [Google Scholar] [CrossRef]
- Vink, E.T.; Rabago, K.R.; Glassner, D.A.; Gruber, P.R. Applications of Life Cycle Assessment to NatureWorksTM Polylactide (PLA) Production. Polym. Degrad. Stab. 2003, 80, 403–419. [Google Scholar] [CrossRef]
- Auras, R.A.; Harte, B.; Selke, S.; Hernandez, R. Mechanical, Physical, and Barrier Properties of Poly (Lactide) Films. J. Plast. Film Sheeting 2003, 19, 123–135. [Google Scholar] [CrossRef]
- Dorgan, J.R.; Lehermeier, H.; Mang, M. Thermal and Rheological Properties of Commercial-Grade Poly (Lactic Acid) s. J. Polym. Environ. 2000, 8, 1–9. [Google Scholar] [CrossRef]
- Lim, L.-T.; Auras, R.; Rubino, M. Processing Technologies for Poly (Lactic Acid). Prog. Polym. Sci. 2008, 33, 820–852. [Google Scholar] [CrossRef]
- Di Lorenzo, M.L. Crystallization Behavior of Poly (L-Lactic Acid). Eur. Polym. J. 2005, 41, 569–575. [Google Scholar] [CrossRef]
- Oyama, H.T.; Tanaka, Y.; Kadosaka, A. Rapid Controlled Hydrolytic Degradation of Poly (l-Lactic Acid) by Blending with Poly (Aspartic Acid-Co-l-Lactide). Polym. Degrad. Stab. 2009, 94, 1419–1426. [Google Scholar] [CrossRef]
- Gollwitzer, H.; Thomas, P.; Diehl, P.; Steinhauser, E.; Summer, B.; Barnstorf, S.; Gerdesmeyer, L.; Mittelmeier, W.; Stemberger, A. Biomechanical and Allergological Characteristics of a Biodegradable Poly (D, L-Lactic Acid) Coating for Orthopaedic Implants. J. Orthop. Res. 2005, 23, 802–809. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R.C.; Kiely, K.D.; Eppley, B.L. The Fate of Resorbable Poly-L-Lactic/Polyglycolic Acid (LactoSorb) Bone Fixation Devices in Orthognathic Surgery. J. Oral Maxillofac. Surg. 2001, 59, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Steuer, H.; Fadale, R.; Müller, E.; Müller, H.-W.; Planck, H.; Schlosshauer, B. Biohybride Nerve Guide for Regeneration: Degradable Polylactide Fibers Coated with Rat Schwann Cells. Neurosci. Lett. 1999, 277, 165–168. [Google Scholar] [CrossRef]
- Yang, F.; Murugan, R.; Wang, S.; Ramakrishna, S. Electrospinning of Nano/Micro Scale Poly (L-Lactic Acid) Aligned Fibers and Their Potential in Neural Tissue Engineering. Biomaterials 2005, 26, 2603–2610. [Google Scholar] [CrossRef]
- Kumbar, S.G.; Nukavarapu, S.P.; James, R.; Nair, L.S.; Laurencin, C.T. Electrospun Poly (Lactic Acid-Co-Glycolic Acid) Scaffolds for Skin Tissue Engineering. Biomaterials 2008, 29, 4100–4107. [Google Scholar] [CrossRef]
- Chen, G.-Q. A Microbial Polyhydroxyalkanoates (PHA) Based Bio-and Materials Industry. Chem. Soc. Rev. 2009, 38, 2434–2446. [Google Scholar] [CrossRef] [PubMed]
- Reddy, C.S.K.; Ghai, R.; Kalia, V. Polyhydroxyalkanoates: An Overview. Bioresour. Technol. 2003, 87, 137–146. [Google Scholar] [CrossRef]
- Philip, S.; Keshavarz, T.; Roy, I. Polyhydroxyalkanoates: Biodegradable Polymers with a Range of Applications. J. Chem. Technol. Biotechnol. 2007, 82, 233–247. [Google Scholar] [CrossRef]
- Verlinden, R.A.; Hill, D.J.; Kenward, M.A.; Williams, C.D.; Radecka, I. Bacterial Synthesis of Biodegradable Polyhydroxyalkanoates. J. Appl. Microbiol. 2007, 102, 1437–1449. [Google Scholar] [CrossRef]
- Chen, G.-Q.; Hajnal, I.; Wu, H.; Lv, L.; Ye, J. Engineering Biosynthesis Mechanisms for Diversifying Polyhydroxyalkanoates. Trends Biotechnol. 2015, 33, 565–574. [Google Scholar] [CrossRef]
- Khanna, S.; Srivastava, A.K. Recent Advances in Microbial Polyhydroxyalkanoates. Process Biochem. 2005, 40, 607–619. [Google Scholar] [CrossRef]
- Urtuvia, V.; Villegas, P.; González, M.; Seeger, M. Bacterial Production of the Biodegradable Plastics Polyhydroxyalkanoates. Int. J. Biol. Macromol. 2014, 70, 208–213. [Google Scholar] [CrossRef]
- Le Meur, S.; Zinn, M.; Egli, T.; Thöny-Meyer, L.; Ren, Q. Production of Medium-Chain-Length Polyhydroxyalkanoates by Sequential Feeding of Xylose and Octanoic Acid in Engineered Pseudomonas Putida KT2440. BMC Biotechnol. 2012, 12, 53. [Google Scholar] [CrossRef]
- Rocha, R.C.; da Silva, L.F.; Taciro, M.K.; Pradella, J.G. Production of Poly (3-Hydroxybutyrate-Co-3-Hydroxyvalerate) P (3HB-Co-3HV) with a Broad Range of 3HV Content at High Yields by Burkholderia Sacchari IPT 189. World J. Microbiol. Biotechnol. 2008, 24, 427–431. [Google Scholar] [CrossRef]
- Loo, C.-Y.; Lee, W.-H.; Tsuge, T.; Doi, Y.; Sudesh, K. Biosynthesis and Characterization of Poly (3-Hydroxybutyrate-Co-3-Hydroxyhexanoate) from Palm Oil Products in a Wautersia Eutropha Mutant. Biotechnol. Lett. 2005, 27, 1405–1410. [Google Scholar] [CrossRef]
- López-Cuellar, M.R.; Alba-Flores, J.; Rodríguez, J.G.; Pérez-Guevara, F. Production of Polyhydroxyalkanoates (PHAs) with Canola Oil as Carbon Source. Int. J. Biol. Macromol. 2011, 48, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Keshavarz, T.; Roy, I. Polyhydroxyalkanoates: Bioplastics with a Green Agenda. Curr. Opin. Microbiol. 2010, 13, 321–326. [Google Scholar] [CrossRef]
- Singh, M.; Kumar, P.; Ray, S.; Kalia, V.C. Challenges and Opportunities for Customizing Polyhydroxyalkanoates. Indian J. Microbiol. 2015, 55, 235–249. [Google Scholar] [CrossRef] [PubMed]
- Yamane, T.; Fukunaga, M.; Lee, Y.W. Increased PHB Productivity by High-Cell-Density Fed-Batch Culture of Alcaligenes Latus, a Growth-Associated PHB Producer. Biotechnol. Bioeng. 1996, 50, 197–202. [Google Scholar] [CrossRef]
- Pandian, S.R.; Deepak, V.; Kalishwaralal, K.; Rameshkumar, N.; Jeyaraj, M.; Gurunathan, S. Optimization and Fed-Batch Production of PHB Utilizing Dairy Waste and Sea Water as Nutrient Sources by Bacillus Megaterium SRKP-3. Bioresour. Technol. 2010, 101, 705–711. [Google Scholar] [CrossRef] [PubMed]
- Kulpreecha, S.; Boonruangthavorn, A.; Meksiriporn, B.; Thongchul, N. Inexpensive Fed-Batch Cultivation for High Poly (3-Hydroxybutyrate) Production by a New Isolate of Bacillus Megaterium. J. Biosci. Bioeng. 2009, 107, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Shahhosseini, S. Simulation and Optimisation of PHB Production in Fed-Batch Culture of Ralstonia Eutropha. Process Biochem. 2004, 39, 963–969. [Google Scholar] [CrossRef]
- Raza, Z.A.; Abid, S.; Banat, I.M. Polyhydroxyalkanoates: Characteristics, Production, Recent Developments and Applications. Int. Biodeterior. Biodegrad. 2018, 126, 45–56. [Google Scholar] [CrossRef]
- Jacquel, N.; Lo, C.-W.; Wei, Y.-H.; Wu, H.-S.; Wang, S.S. Isolation and Purification of Bacterial Poly (3-Hydroxyalkanoates). Biochem. Eng. J. 2008, 39, 15–27. [Google Scholar] [CrossRef]
- Ibrahim, M.H.; Steinbüchel, A. Poly (3-Hydroxybutyrate) Production from Glycerol by Zobellella Denitrificans MW1 via High-Cell-Density Fed-Batch Fermentation and Simplified Solvent Extraction. Appl. Environ. Microbiol. 2009, 75, 6222–6231. [Google Scholar] [CrossRef]
- Koller, M. Recent Advances in Biotechnology Volume, 1: Microbial Biopolyester Production, Performance and Processing Microbiology, Feedstocks, and Metabolism; Bentham Science Publishers: Oak Park, IL, USA, 2016; Volume 1. [Google Scholar]
- Hejazi, P.; Vasheghani-Farahani, E.; Yamini, Y. Supercritical Fluid Disruption of Ralstonia Eutropha for Poly (β-Hydroxybutyrate) Recovery. Biotechnol. Prog. 2003, 19, 1519–1523. [Google Scholar] [CrossRef]
- Kunasundari, B.; Sudesh, K. Isolation and Recovery of Microbial Polyhydroxyalkanoates. Express Polym. Lett. 2011, 5, 620–634. [Google Scholar] [CrossRef]
- Divyashree, M.S.; Shamala, T.R.; Rastogi, N.K. Isolation of Polyhydroxyalkanoate from Hydrolyzed Cells of Bacillus Flexus Using Aqueous Two-Phase System Containing Polyethylene Glycol and Phosphate. Biotechnol. Bioprocess Eng. 2009, 14, 482–489. [Google Scholar] [CrossRef]
- Porter, M.M.; Lee, S.; Tanadchangsaeng, N.; Jaremko, M.J.; Yu, J.; Meyers, M.; McKittrick, J. Porous hydroxyapatite-polyhydroxybutyrate composites fabricated by a novel method via centrifugation. In Mechanics of Biological Systems and Materials; Springer: Berlin/Heidelberg, Germany, 2013; Volume 5, pp. 63–71. [Google Scholar]
- Hong, H.; Dong, N.; Shi, J.; Chen, S.; Guo, C.; Hu, P.; Qi, H. Fabrication of a Novel Hybrid Heart Valve Leaflet for Tissue Engineering: An in Vitro Study. Artif. Organs 2009, 33, 554–558. [Google Scholar] [CrossRef]
- Valappil, S.P.; Boccaccini, A.R.; Bucke, C.; Roy, I. Polyhydroxyalkanoates in Gram-Positive Bacteria: Insights from the Genera Bacillus and Streptomyces. Antonie Van Leeuwenhoek 2007, 91, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Bugnicourt, E.; Cinelli, P.; Lazzeri, A.; Alvarez, V.A. Polyhydroxyalkanoate (PHA): Review of Synthesis, Characteristics, Processing and Potential Applications in Packaging. Express Polym. Lett. 2014, 8, 791–808. [Google Scholar] [CrossRef]
- Abid, S.; Raza, Z.A.; Rehman, A. Synthesis of Poly (3-Hydroxybutyrate) Nanospheres and Deposition Thereof into Porous Thin Film. Mater. Res. Express 2016, 3, 105042. [Google Scholar] [CrossRef]
- Bajpai, P.K.; Ahmad, F.; Chaudhary, V. Processing and Characterization of Bio-Composites. Handb. Ecomater. 2017, 1–18. [Google Scholar]
- Le Duc, A.; Vergnes, B.; Budtova, T. Polypropylene/Natural Fibres Composites: Analysis of Fibre Dimensions after Compounding and Observations of Fibre Rupture by Rheo-Optics. Compos. Part Appl. Sci. Manuf. 2011, 42, 1727–1737. [Google Scholar] [CrossRef]
- Faruk, O.; Bledzki, A.K.; Fink, H.-P.; Sain, M. Progress Report on Natural Fiber Reinforced Composites. Macromol. Mater. Eng. 2014, 299, 9–26. [Google Scholar] [CrossRef]
- Bledzki, A.K.; Jaszkiewicz, A.; Murr, M.; Sperber, V.E.; Lützendgrf, R.; Reußmann, T. Processing techniques for natural-and wood-fibre composites. In Properties and Performance of Natural-Fibre Composites; Elsevier: Amsterdam, The Netherlands, 2008; pp. 163–192. [Google Scholar]
- Bledzki, A.K.; Specht, K.; Cescutti, G.; Müssig, M. Comparison of Different Compounding Processes by an Analysis of Fibres Degradation. In Proceedings of the 3rd International Conference on Eco-Composites, Stockholm, Sweden, 20–21 June 2005; p. B3. [Google Scholar]
- Chaudhary, V.; Bajpai, P.K.; Maheshwari, S. Studies on Mechanical and Morphological Characterization of Developed Jute/Hemp/Flax Reinforced Hybrid Composites for Structural Applications. J. Nat. Fibers 2018, 15, 80–97. [Google Scholar] [CrossRef]
- Pickering, K.L.; Efendy, M.A.; Le, T.M. A Review of Recent Developments in Natural Fibre Composites and Their Mechanical Performance. Compos. Part Appl. Sci. Manuf. 2016, 83, 98–112. [Google Scholar] [CrossRef]
- Huda, M.S.; Drzal, L.T.; Ray, D.; Mohanty, A.K.; Mishra, M. Natural-fiber composites in the automotive sector. In Properties and Performance of Natural-Fibre Composites; Elsevier: Amsterdam, The Netherlands, 2008; pp. 221–268. [Google Scholar]
- Azaman, M.D.; Sapuan, S.M.; Sulaiman, S.; Zainudin, E.S.; Abdan, K. An Investigation of the Processability of Natural Fibre Reinforced Polymer Composites on Shallow and Flat Thin-Walled Parts by Injection Moulding Process. Mater. Des. 2013, 50, 451–456. [Google Scholar] [CrossRef]
- Leong, Y.W.; Thitithanasarn, S.; Yamada, K.; Hamada, H. Compression and injection molding techniques for natural fiber composites. In Natural Fibre Composites; Elsevier: Amsterdam, The Netherlands, 2014; pp. 216–232. [Google Scholar]
- Akampumuza, O.; Wambua, P.; Ahmed, A.; Li, W.; Qin, X.-H. Review of the Applications of Biocomposites in the Automotive Industry. Polym. Compos. 2017, 38, 2553–2569. [Google Scholar] [CrossRef]
- Alves, C.; Silva, A.J.; Reis, L.G.; Freitas, M.; Rodrigues, L.B.; Alves, D.E. Ecodesign of Automotive Components Making Use of Natural Jute Fiber Composites. J. Clean. Prod. 2010, 18, 313–327. [Google Scholar] [CrossRef]
- Ho, M.; Wang, H.; Lee, J.-H.; Ho, C.; Lau, K.; Leng, J.; Hui, D. Critical Factors on Manufacturing Processes of Natural Fibre Composites. Compos. Part B Eng. 2012, 43, 3549–3562. [Google Scholar] [CrossRef]
- Srinivas, K.; Naidu, A.L.; Raju Bahubalendruni, M. A Review on Chemical and Mechanical Properties of Natural Fiber Reinforced Polymer Composites. Int. J. Perform. Eng. 2017, 13, 189–200. [Google Scholar] [CrossRef]
- Mallick, P.K. Fiber-Reinforced Composites: Materials, Manufacturing, and Design; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Haag, K.; Padovani, J.; Fita, S.; Trouvé, J.-P.; Pineau, C.; Hawkins, S.; De Jong, H.; Deyholos, M.K.; Chabbert, B.; Müssig, J. Influence of Flax Fibre Variety and Year-to-Year Variability on Composite Properties. Ind. Crops Prod. 2017, 98, 1–9. [Google Scholar] [CrossRef]
- Du, Y.; Wu, T.; Yan, N.; Kortschot, M.T.; Farnood, R. Fabrication and Characterization of Fully Biodegradable Natural Fiber-Reinforced Poly (Lactic Acid) Composites. Compos. Part B Eng. 2014, 56, 717–723. [Google Scholar] [CrossRef]
- Chaitanya, S.; Singh, I. Processing of PLA/Sisal Fiber Biocomposites Using Direct-and Extrusion-Injection Molding. Mater. Manuf. Process. 2017, 32, 468–474. [Google Scholar] [CrossRef]
- Ranganathan, N.; Oksman, K.; Nayak, S.K.; Sain, M. Structure Property Relation of Hybrid Biocomposites Based on Jute, Viscose and Polypropylene: The Effect of the Fibre Content and the Length on the Fracture Toughness and the Fatigue Properties. Compos. Part Appl. Sci. Manuf. 2016, 83, 169–175. [Google Scholar] [CrossRef]
- Awal, A.; Rana, M.; Sain, M. Thermorheological and Mechanical Properties of Cellulose Reinforced PLA Bio-Composites. Mech. Mater. 2015, 80, 87–95. [Google Scholar] [CrossRef]
- Evans, W.J.; Isaac, D.H.; Suddell, B.C.; Crosky, A. Natural Fibres and Their Composites: A Global Perspective. In Proceedings of the Risø International Symposium on Materials Science, Roskilde, Denmark, 2–5 September 2002; pp. 1–14. [Google Scholar]
- Alkbir, M.F.M.; Sapuan, S.M.; Nuraini, A.A.; Ishak, M.R. Fibre Properties and Crashworthiness Parameters of Natural Fibre-Reinforced Composite Structure: A Literature Review. Compos. Struct. 2016, 148, 59–73. [Google Scholar] [CrossRef]
- Mohanty, A.K.; Seydibeyoglu, M.O.; Sahoo, S.; Misra, M. 4.08-Matching Crops for Selected Bioproducts A2-Moo-Young, Murray. In Comprehensive Biotechnology; Academic Press: Burlington, VT, USA, 2011. [Google Scholar]
- AL-Oqla, F.M.; Omari, M.A. Sustainable biocomposites: challenges, potential and barriers for development. In Green Biocomposites; Springer: Berlin/Heidelberg, Germany, 2017; pp. 13–29. [Google Scholar]
- Al-Oqla, F.M.; Sapuan, S.M. Polymer Selection Approach for Commonly and Uncommonly Used Natural Fibers under Uncertainty Environments. Jom 2015, 67, 2450–2463. [Google Scholar] [CrossRef]
- Mooney, B.P. The Second Green Revolution? Production of Plant-Based Biodegradable Plastics. Biochem. J. 2009, 418, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Andresen, C.; Demuth, C.; Lange, A.; Stoick, P.; Pruszko, R. Biobased Automobile Parts Investigation. A Report Developed for the USDA Office of Energy Policy and New Uses; Iowa State University: Ames, IA, USA, 2012. [Google Scholar]
- La Mantia, F.P.; Morreale, M. Green Composites: A Brief Review. Compos. Part Appl. Sci. Manuf. 2011, 42, 579–588. [Google Scholar] [CrossRef]
- Toyota Green Innovations. Available online: http://www.toyota.com (accessed on 5 January 2021).
- Mosallam, A.S.; Bayraktar, A.; Elmikawi, M.; Pul, S.; Adanur, S. Polymer Composites in Construction: An Overview; UC Irvine: Irvine, CA, USA, 2015. [Google Scholar]
- Kinnane, O.; Reilly, A.; Grimes, J.; Pavia, S.; Walker, R. Acoustic Absorption of Hemp-Lime Construction. Constr. Build. Mater. 2016, 122, 674–682. [Google Scholar] [CrossRef]
| Fiber | Cellulose (%) | Hemicellulose (%) | Lignin (%) | Pectin (%) | Wax (%) | Reference |
|---|---|---|---|---|---|---|
| Abaca | 56–63 | 21–25 | 7–12 | 0.8 | 3 | [23,71,72,73] |
| Alfa | 45.4 | 38.5 | 14.9 | - | 2 | [71,72] |
| Areca | 57.35–58.21 | 13–15.42 | 23–24 | - | 0.12 | [74] |
| Bagasse | 32–44 | 19–24 | 22 | 10 | - | [71,75] |
| Bamboo | 26–43 | 20.5 | 21–31 | - | - | [23,71] |
| Banana | 62–64 | 12.5 | 5–10 | 4 | - | [31,75,76,77] |
| Barley | 31–45 | 27–38 | 14–19 | - | 2–7 | [31] |
| Coir | 45.6 | 20 | 45 | 4 | - | [71,72,78] |
| Corn | 38–40 | 28 | 7–21 | - | 3.6–7 | [31] |
| Cotton | 82.7–90 | 4 | 0.75 | 6 | 0.6 | [71,72] |
| Curaua | 70.7–73.6 | 9.9 | 7.5–11.1 | - | - | [23,71,72,75] |
| Eucalyptus | 41.7 | 32.56 | 25.4 | 8.2 | 0.22 | [79] |
| Flax | 62–72.5 | 14.5–20.6 | 2.5 | 0.9 | - | [23,71,75] |
| Hemp | 81 | 14–22 | 4–13 | 0.9 | 0.8 | [23,71,80] |
| Henequen | 60–77.6 | 28 | 8–13.1 | - | 0.5 | [71,72] |
| Hibiscus | 28 | 25 | 22.7 | - | - | [81] |
| Isora | 74 | - | 23 | - | 1.1 | [71] |
| Jute | 59–71.5 | 12–20 | 9–13 | 0.2 | 0.5 | [23,71,82,83] |
| Kenaf | 53.5 | 21–33 | 17–21.5 | 2 | - | [71,78,84] |
| Phromium | 67 | 30 | 11 | - | - | [71] |
| Pineapple | 80.5 | 17.5 | 8.3–12.7 | 4 | - | [23,71] |
| Ramie | 72 | 5–16.7 | 0.6–0.8 | 2 | - | [71,80] |
| Rice husk | 28–36 | 23–28 | 12–14 | - | 14–20 | [81] |
| Sisal | 60–73 | 11.5–14 | 8–11 | 1.2 | - | [23,71,85] |
| Sorghum | 27 | 25 | 11 | - | - | [31] |
| Wheat | 33–38 | 26–32 | 17–19 | - | 6.8 | [79] |
| Composite | Fabrication Method | Key Findings and Mechanical Properties | Effect of Surface Treatments | References |
|---|---|---|---|---|
| Abaca–Roselle/Cardanol formaldehyde composite | Compression molding | Natural fibers improved thermal, wear resistance, and mechanical properties of the composite and improved the hardness, density, and tensile strength of the matrix material. Tensile and flexural properties improved due to the presence of carbon and silica. | Alkali treatment increased fiber/matrix adhesion due to the removal of impurities and increased mechanical properties. | [140] |
| Areca fibers/Pine resin composite | Solvent casting method | The tensile strength of the composite is affected by the adhesion of the fiber/matrix; 10 wt.% areca fibers and 90 wt.% pine resin exhibited better mechanical properties due to efficient stress transfer between fibers and matrix. | Alkali treatment increased fiber/matrix adhesion. Tensile strength increased by 25%, while impact strength increased up to 24% due to treatment. | [141] |
| Banana fibers/PLA/Nanoclay composite | Melt blending | Nanoclay and PLA improved composite stability, flame resistance, and thermal properties. Nanoclay formed a protective layer at the surface to prevent flame and acted as a thermal barrier to prevent degradation. | Silane treatment improved fiber/matrix adhesion by increasing the contact area of fibers. | [142] |
| Flax/epoxy composite | Vacuum infusion | Flax/epoxy composite is suspectable to water absorption due to high void content. | Sodium bicarbonate-treated fibers had less void content mainly due to the removal of impurities. With the increase in sodium bicarbonate concentration in fiber treatment, properties such as flexural, tensile strength and flexural moduli increased. | [143] |
| Hemp fibers/polycaprolactone bio-composite | Twin screw extrusion | Flexural, tensile and impact properties of composite are improved. With the increase in aspect ratio of hemp fiber, water absorption increased. Flexural strength increased by 169% and flexural modulus increased by 285% for the aspect ratio of 26. Hemp fibers increased the stiffness of the composite. | [144] | |
| Jute fibers/unsaturated polyester resin | Hand lay-up and compression molding | Jute fibers enhanced properties such as tensile, flexural strength, flexural modulus, and interlaminar shear strength. Untreated fibers lead to low density and low volume fraction. | Alkali-treated fibers showed an increase in tensile, flexural strength, flexural modulus, and interlaminar shear strength due to better fiber/matrix adhesion. Alkali treatment removes hemicellulose and increases interlocking points in fibers for better adhesion and stress transfer. | [145] |
| Jute fibers/clay/epoxy bio-composite | Compression molding | The addition of 15 wt.% clay improved mechanical properties due to uniform dispersion in a composite. Clay can agglomerate, which increases composite porosity and decreases fiber/matrix adhesion. | Alkali treatment improved fiber/matrix adhesion with increased cellulose after removing pectin, lignin, and other impurities. An increase in cellulose content leads to better interfacial adhesion. | [146] |
| Kenaf fibers/sea urchin spike filler/neem oil/epoxy composite | Hand lay-up | Neem oil made epoxy eco-friendly while sea urchin spike filler and kenaf fibers increased the toughness of the composite. The addition of neem oil leads to the formation of an interpolymer-penetrating network and ketone groups, which decreased hardness and overall tensile strength of the composite. | Amino silane-treated particles dispersed well in matrix material without agglomeration, which improved wear resistance and thermal degradation. Treated fiber formed a layer at the fiber/matrix interface, and high temperature was required to break this layer. Modified fibers increased the moisture resistance in the composite. | [147] |
| Ramie fibers/PLA composite | Hot compression molding | Low temperature and pressure in compression molding had led to poor fiber/matrix adhesion and wettability. | Alkali/silane-treated fibers composite had better tensile strength, modulus, and impact strength. Cellulose content increased due to the removal of impurities from fibers, which improved mechanical properties. Treated fibers had better stress transfer due to the formation of covalent bonds between fibers and matrix. | [148] |
| Sisal fibers/starch composite | Hot pressing | Compressive and tensile strength of the composite increased with the addition of sisal fibers. The addition of natural fibers increased the biodegradability properties of the composite. | Alkaline treatment increased fiber/matrix adhesion, which improved mechanical properties. | [149] |
| Fiber | Density (g/cm3) | Diameter (µm) | Micro-Fibrillar Angle (°) | Moisture Content (%) | Tensile Strength (MPa) | Elongation at Break (%) | References |
|---|---|---|---|---|---|---|---|
| Abaca | 1.5 | 10–30 | 20–25 | 5–10 | 400–980 | 3–10 | [23,75,154,155] |
| Areca | 0.7–0.8 | - | - | - | 147–322 | 10.2–13.15 | [156,157] |
| Bagasse | 1.25 | 10–34 | - | - | 222–290 | 1.1 | [23,75,158] |
| Bamboo | 0.6–1.11 | 240–330 | - | 9.16 | 140–800 | 1.40 | [23,72,159,160,161] |
| Banana | 1.35 | 50–250 | 11–12 | 10.71 | 529–914 | 3 | [80,161,162,163] |
| Coir | 1.2–1.5 | 100–450 | 30–49 | 8–11.36 | 175–180 | 30 | [18,40,74,75,159,164] |
| Cotton | 1.5–1.6 | 12–35 | - | 7.85–8.5 | 287–597 | 7–8 | [72,163,164,165,166,167] |
| Curaua | 1.4 | 170 | - | - | 500–1150 | 3.7–4.3 | [23,161] |
| Flax | 1.5 | 5–38 | 5–10 | 1.2–8 | 345–1035 | 2.7–3.2 | [23,161,167,168,169] |
| Hemp | 1.48 | - | 2–6.2 | 6.2–12 | 690 | 1.6 | [23,161,163,164,169] |
| Henequen | 1.2 | - | - | - | 430–570 | 3.7–5.9 | [71,72] |
| Isora | 1.2–1.3 | - | - | - | 500–600 | 5–6 | [71,170] |
| Jute | 1.3–1.5 | 20–200 | 8 | 12.5–13.7 | 200–773 | 1.5–1.8 | [23,75,164,168,171] |
| Kenaf | 1.4 | 70–250 | 2–6.2 | 6.2–12 | 930 | 1.5 | [23,165,163,165] |
| Nettle | 1.51 | 20–80 | - | 11–17 | 650 | 1.7 | [75,155,167] |
| Oil Palm | 0.7–1.55 | 150–500 | 42–46 | - | 80–248 | 3.2 | [23,75,165,172] |
| Palf | 0.8–1.6 | 20–80 | 14 | 11.8 | 180–1627 | 1.6–14.5 | [74,75,112] |
| Piassava | 1.4 | - | - | - | 134–143 | 7.8–21.9 | [74,75,173] |
| Pineapple | 0.8–1.6 | 8–41 | - | 10–13 | 170–1627 | 2.4 | [23,162,167,174] |
| Ramie | 1.5 | 50 | 69–83 | 220–938 | 2–3.8 | [23,163,164] | |
| Sisal | 1.5 | 50–300 | - | 11 | 511–635 | 3–7 | [23,163,164] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zwawi, M. A Review on Natural Fiber Bio-Composites, Surface Modifications and Applications. Molecules 2021, 26, 404. https://doi.org/10.3390/molecules26020404
Zwawi M. A Review on Natural Fiber Bio-Composites, Surface Modifications and Applications. Molecules. 2021; 26(2):404. https://doi.org/10.3390/molecules26020404
Chicago/Turabian StyleZwawi, Mohammed. 2021. "A Review on Natural Fiber Bio-Composites, Surface Modifications and Applications" Molecules 26, no. 2: 404. https://doi.org/10.3390/molecules26020404
APA StyleZwawi, M. (2021). A Review on Natural Fiber Bio-Composites, Surface Modifications and Applications. Molecules, 26(2), 404. https://doi.org/10.3390/molecules26020404






