Proceedings of Chemistry, Pharmacology, Pharmacokinetics and Synthesis of Biflavonoids
Abstract
1. Introduction
2. Distribution of Biflavonoids
3. The Scaffold of Biflavonoids
4. Subtypes of Biflavonoids
4.1. C-C Type
4.2. C-Linear Fragment-C Type
4.3. Complex Biflavonoids
5. Pharmacology of Biflavonoids
5.1. Antioxidant
5.2. Anti-Inflammatory Properties
5.3. Antiviral Activities
5.4. Antibacterial and Antifungal Activities
5.5. Anti-Diabetic and Anti-Atherosclerosis
5.6. Alzheimer’s Disease and Parkinson’s Disease
5.7. Cytotoxic Activity and Antitumor Activities
5.8. Anti-Angiogenesis
5.9. Other
6. Pharmacokinetics
7. The Biosynthesis and Synthesis of Biflavonoids
7.1. The Biosynthesis of Biflavonoids
7.2. The Synthesis of Biflavonoids
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gontijo, V.S.; Dos Santos, M.H.; Viegas, C., Jr. Biological and Chemical Aspects of Natural Biflavonoids from Plants: A Brief Review. Mini Rev. Med. Chem. 2017, 17, 834–862. [Google Scholar] [CrossRef]
- Yu, S.; Yan, H.; Zhang, L.; Shan, M.; Chen, P.; Ding, A.; Li, S.F. A Review on the Phytochemistry, Pharmacology, and Pharmacokinetics of Amentoflavone, a Naturally-Occurring Biflavonoid. Molecules 2017, 22, 299. [Google Scholar] [CrossRef] [PubMed]
- Parveen, M.; Ilyas, M.; Mushfiq, M.; Busudan, O.A.; Muhaisen, H.M. A new biflavonoid from leaves of Garcinia nervosa. Nat. Prod. Res. 2004, 18, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Kamiya, K.; Satake, T. Chemical constituents of Baeckea frutescens leaves inhibit copper-induced low-density lipoprotein oxidation. Fitoterapia 2010, 81, 185–189. [Google Scholar] [CrossRef]
- Hu, J.-F.; Garo, E.; Hough, G.W.; Goering, M.G.; O’Neil-Johnson, M.; Eldridge, G.R. Acuminatanol, the first 2′,2‴-bis-dihydrobiflavonol from the aqueous extract of Trichoscypha acuminata. Tetrahedron Lett. 2007, 48, 5747–5749. [Google Scholar] [CrossRef]
- Nonaka, G.; Kawahara, O.; Nishioka, I. Tannins and Related Compounds. XV. A New Class of Dimeric Flavan-3-ol Gallates, Theasinensins A and B, and Proanthocyanidin Gallates from Green Tea Leaf. (1). Chem. Pharm. Bull. 1983, 31, 3906–3914. [Google Scholar] [CrossRef]
- Yang, X.; Kang-Ping, X.; Zhen-Xing, Z.; Gui-Shan, T. Advances in chemodiversity from Selaginella. Cent. South. Pharm. 2017, 15, 129–142. [Google Scholar]
- Yun-Yun, Y.; Lu, H.; Ping, W.; Guo-Zhu, S.; Tian-Tian, S.; Chang-Cai, B. Advances on chemical constituents and bioactivities of genus Stellera. China J. Chin. Mater. Med. 2015, 40, 4324–4332. [Google Scholar]
- Reddy, B.A.K.; Reddy, N.P.; Gunasekar, D.; Blond, A.; Bodo, B. Biflavonoids from Ochna lanceolata. Phytochem. Lett. 2008, 1, 27–30. [Google Scholar] [CrossRef]
- Adem, F.A.; Mbaveng, A.T.; Kuete, V.; Heydenreich, M.; Ndakala, A.; Irungu, B.; Yenesew, A.; Efferth, T. Cytotoxicity of isoflavones and biflavonoids from Ormocarpum kirkii towards multi-factorial drug resistant cancer. Phytomedicine 2019, 58, 152853. [Google Scholar] [CrossRef]
- Manga, S.S.E.; Tih, A.E.; Ghogomu, R.T.A.; Blond, B.B. Biflavonoid constituents of Campylospermum mannii. Biochem. Syst. Ecol. 2009, 37, 402–404. [Google Scholar] [CrossRef]
- Chen, L.Y.; Chen, I.S.; Peng, C.F. Structural elucidation and bioactivity of biflavonoids from the stems of Wikstroemia taiwanensis. Int. J. Mol. Sci. 2012, 13, 1029–1038. [Google Scholar] [CrossRef]
- Nyandat, E.; Hassanali, A.; Vicente, Y.D.; Multari, G.; Galeffi, C. The 7,7″-β-diglucoside of (2S,3R)-chamaejasmin from Ormocarpum kirkii. Phytochemistry 1990, 29, 2361–2364. [Google Scholar] [CrossRef]
- Kim, A.R.; Jin, Q.; Jin, H.G.; Ko, H.J.; Woo, E.R. Phenolic compounds with IL-6 inhibitory activity from Aster yomena. Arch. Pharm. Res. 2014, 37, 845–851. [Google Scholar] [CrossRef]
- Li, J.; Lu, L.Y.; Zeng, L.H.; Zhang, C.; Hu, J.L.; Li, X.R. Sikokianin D, a new C-3/C-3”-biflavanone from the roots of Wikstroemia indica. Molecules 2012, 17, 7792–7797. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.-J.; Foubert, K.; Dhooghe, L.; Lemière, F.; Maregesi, S.; Coleman, C.M.; Zou, Y.; Ferreira, D.; Apers, S.; Pieters, L. Rapid isolation and identification of minor natural products by LC–MS, LC–SPE–NMR and ECD: Isoflavanones, biflavanones and bisdihydrocoumarins from Ormocarpum kirkii. Phytochemistry 2012, 79, 121–128. [Google Scholar] [CrossRef]
- Dhooghe, L.; Maregesi, S.; Mincheva, I.; Ferreira, D.; Marais, J.P.J.; Lemière, F.; Matheeussen, A.; Cos, P.; Maes, L.; Vlietinck, A.; et al. Antiplasmodial activity of (I-3,II-3)-biflavonoids and other constituents from Ormocarpum kirkii. Phytochemistry 2010, 71, 785–791. [Google Scholar] [CrossRef]
- Li, X.-Q.; Rahman, K.; Zhu, J.-Y.; Zhang, H. Chemical Constituents and Pharmacological Activities of Stellera chamaejasme. Curr. Pharm. Des. 2018, 24, 2825–2838. [Google Scholar] [CrossRef]
- NISHIMUTA, S.; Taki, M.; Takaishi, S.; Iijima, Y.; Akiyama, T. Structures of 4-aryl-coumarin (neoflavone) dimers isolated from Pistacia chinensis BUNGE and their estrogen-like activity. Chem. Pharm. Bull. 2000, 48, 505–508. [Google Scholar] [CrossRef] [PubMed]
- Zhi, X.; Jian-hui, S. Research Progress of Biflavonoids. China J. Mod. Med. 2004, 14, 88–91. [Google Scholar]
- Chien, S.-C.; Liu, H.-K.; Kuo, Y.-H. Two New Compounds from the Leaves of Calocedrus microlepic var. formosana. Chem. Pharm. Bull. 2004, 52, 762–763. [Google Scholar] [CrossRef]
- Lee, C.-W.; Choi, H.-J.; Kim, H.-S.; Kim, D.-H.; Chang, I.-S.; Moon, H.T.; Lee, S.-Y.; Oh, W.K.; Woo, E.-R. Biflavonoids isolated from Selaginella tamariscina regulate the expression of matrix metalloproteinase in human skin fibroblasts. Bioorg. Med. Chem. 2008, 16, 732–738. [Google Scholar] [CrossRef] [PubMed]
- Kamil, M.; Ilyas, M.; Rahman, W.; Hasaka, N.; Okigawa, M.; Kawano, N. Taiwaniaflavone and its derivatives: A new series of biflavones from Taiwania cryptomerioides Hayata. J. Chem. Soc. Perkin Trans. 1 1981, 12, 553–559. [Google Scholar] [CrossRef]
- Sakasai, M.; Fukui, H.; Yamane, H.; Kyaw, A.N.; Tahara, S. A New Class of Biflavonoids: 2′-Hydroxy genistein Dimers from the Roots of White Lupin. Z. Nat. C 2000, 55, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Jiang, H.-X.; Huang, H.; Zhu, R.-L.; Jiang, B. Ring-B Linked Bidihydroflavonoids from Thuidium kanedae Sak. Chin. J. Chem. 2006, 24, 393–395. [Google Scholar] [CrossRef]
- Hamada, T. Studies on the medicinal plant in the “Sambutsu-cho” of Bungo Province possessed by the Kumamoto Clan (II); studies on the medicinal herbs. Yakushigaku Zasshi 1992, 27, 117–124. [Google Scholar]
- Wan, H.; Ge, L.; Li, J.; Zhang, K.; Wu, W.; Peng, S.; Zou, X.; Zhou, H.; Zhou, B.; Zeng, X. Effects of a novel biflavonoid of Lonicera japonica flower buds on modulating apoptosis under different oxidative conditions in hepatoma cells. Phytomedicine 2019, 57, 282–291. [Google Scholar] [CrossRef]
- Sum, T.H.; Sum, T.J.; Collins, S.; Galloway, W.; Twigg, D.G.; Hollfelder, F.; Spring, D.R. Divergent synthesis of biflavonoids yields novel inhibitors of the aggregation of amyloid beta (1-42). Org. Biomol. Chem. 2017, 15, 4554–4570. [Google Scholar] [CrossRef]
- Zheng, R.; Rui-jie, C.; Yan-ying, Y.; Shu-wen, C. Research Progresses on Chemical Constituents of Genus Daphne genus and Their Bioactivities. Food Sci. 2009, 30, 249–258. [Google Scholar]
- Duanrui, S.; Shouxun, Z. Non-alkaloid constituents from aerial parts of Stephania tetrandra. J. Jining Med. Coll. 1993, 2, 1–5. [Google Scholar]
- Pegnyemb, D.E.; Tih, R.G.; Sondengam, B.L.; Blond, A.; Bodo, B. Flavonoids of Ochna afzelii. Phytochemistry 2003, 64, 661–665. [Google Scholar] [CrossRef]
- Machado, M.B.; Lopes, L.M.X. Tetraflavonoid and biflavonoids from Aristolochia ridicula. Phytochemistry 2008, 69, 3095–3102. [Google Scholar] [CrossRef] [PubMed]
- Machado, M.B.; Lopes, L.M.X. Chalcone–flavone tetramer and biflavones from Aristolochia ridicula. Phytochemistry 2005, 66, 669–674. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, F.J.C.; Boralle, N.; Silva, D.H.S.; Lopes, L.M.X. Bi- and tetraflavonoids from Aristolochia ridicula. Phytochemistry 2000, 55, 823–832. [Google Scholar] [CrossRef]
- Lee, N.-Y.; Min, H.-Y.; Lee, J.; Nam, J.-W.; Lee, Y.-J.; Han, A.-R.; Wiryawan, A.; Suprapto, W.; Lee, S.K.; Seo, E.-K. Identification of a new cytotoxic biflavanone from Selaginella doederleinii. Chem. Pharm. Bull. 2008, 56, 1360–1361. [Google Scholar] [CrossRef]
- Park, S.Y.; Nguyen, P.H.; Kim, G.; Jang, S.N.; Lee, G.H.; Phuc, N.M.; Wu, Z.; Liu, K.H. Strong and Selective Inhibitory Effects of the Biflavonoid Selamariscina A against CYP2C8 and CYP2C9 Enzyme Activities in Human Liver Microsomes. Pharmaceutics 2020, 12, 343. [Google Scholar] [CrossRef]
- Lin, L.-C.; Kuo, Y.-C.; Chou, C.-J. Cytotoxic biflavonoids from Selaginella delicatula. J. Nat. Prod. 2000, 63, 627–630. [Google Scholar] [CrossRef] [PubMed]
- Kassem, M.E.S.; El-Desoky, S.K.; Sharaf, M. Biphenyl esters and biflavonoids from the fruits of Schinus terebenthefolus. Chem. Nat. Compd. 2004, 40, 447–450. [Google Scholar] [CrossRef]
- Chen, J.-J.; Duh, C.-Y.; Chen, J.-F. New cytotoxic biflavonoids from Selaginella delicatula. Planta Med. 2005, 71, 659–665. [Google Scholar] [CrossRef]
- Gu, S.; Xu, L.; Sun, N. Studies on chemical compositions of Podocarpus imbricatus. China J. Chin. Matera Med. 1995, 20, 105–106. [Google Scholar]
- Bahia, M.V.; Santos, J.B.D.; David, J.P.D.L.; David, J.M. Biflavonoids and other phenolics from Caesalpinia pyramidalis (Fabaceae). J. Braz. Chem. Soc. 2005, 16, 1402–1405. [Google Scholar] [CrossRef]
- Aguilar, M.I.; Romero, M.G.; Chávez, M.I.; King-Díaz, B.; Lotina-Hennsen, B. Biflavonoids Isolated from Selaginella lepidophylla Inhibit Photosynthesis in Spinach Chloroplasts. J. Agric. Food Chem. 2008, 56, 6994–7000. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Wang, N.; Fan, M.; Chen, H.; Liu, H.; Yao, X. A new biflavonoid from Selaginella uncinata. Asian J. Tradit. Med. 2007, 2, 92–97. [Google Scholar]
- Zheng, J.-X.; Wang, N.-L.; Liu, H.-W.; Chen, H.-F.; Li, M.-M.; Wu, L.-Y.; Fan, M.; Yao, X.-S. Four new biflavonoids from Selaginella uncinata and their anti-anoxic effect. J. Asian Nat. Prod. Res. 2008, 10, 945–952. [Google Scholar] [CrossRef] [PubMed]
- Cane, H.; Saidi, N.; Yahya, M.; Darusman, D.; Erlidawati, E.; Safrida, S.; Musman, M. Macrophylloflavone: A New Biflavonoid from Garcinia macrophylla Mart. (Clusiaceae) for Antibacterial, Antioxidant, and Anti-Type 2 Diabetes Mellitus Activities. Sci. World J. 2020, 2020, 2983129. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, A.; Kotoky, J.; Das, K.K.; Banerji, J.; Chakraborty, T. Abiesin, a biflavonoid of abies webbiana. Phytochemistry 1984, 23, 704–705. [Google Scholar] [CrossRef]
- Seeger, T.; Zinsmeister, H.D.; Geiger, H. The Biflavonoid Pattern of Rhytidiadelphus squarrosus (Hedw.) Warnst. Z. Nat. C 1990, 45, 583–586. [Google Scholar] [CrossRef]
- Anhut, S.; Seeger, T.; Zinsmeister, H.D.; Geiger, H. New Dihydrobiflavones from the Moss Plagiomnium cuspidatum. Z. Nat. C 1989, 44, 189–192. [Google Scholar] [CrossRef]
- Markham, K.R.; Andersen, Ø.M.; Viotto, E.S. Unique biflavonoid types from the moss Dicranoloma robustum. Phytochemistry 1988, 27, 1745–1749. [Google Scholar] [CrossRef]
- Rampendahl, C.; Seeger, T.; Geiger, H.; Zinsmeister, H.D. The biflavonoids of Plagiomnium undulatum. Phytochemistry 1996, 41, 1621–1624. [Google Scholar] [CrossRef]
- Matamela, T.; Green, I.R.; Mtunzi, F.M. A Novel Biflavonoid from Rhus leptodictya. Nat. Prod. Commun. 2016, 11, 1279–1280. [Google Scholar] [CrossRef]
- Tih, A.E.; Ghogomu, R.T.; Sondengam, B.L.; Caux, C.; Bodo, B. Minor biflavonoids from Lophira alata leaves. J. Nat. Prod. 2006, 69, 1206–1208. [Google Scholar] [CrossRef]
- Pieters, L.; Mbwambo, Z.H.; Kapingu, M.C.; Moshi, M.J.; Machumi, F.; Apers, S.; Cos, P.; Ferreira, D.; Marais, J.P.J.; Berghe, D.V.; et al. Antiparasitic Activity of Some Xanthones and Biflavonoids and Identification of a New Biflavanoid from the Root Bark of Garcinia livingstonei. Planta Med. 2006, 72, P_003. [Google Scholar] [CrossRef]
- Al-Shagdari, A.; Alarcon, A.B.; Cuesta-Rubio, O.; Piccinelli, A.L.; Rastrelli, L. Biflavonoids, main constituents from Garcinia bakeriana leaves. Nat. Prod. Commun. 2013, 8, 1237–1240. [Google Scholar] [CrossRef]
- Min, Y. Current Status of Research on Biflavonoids in Garcinia. Guangdong Pharm. 2004, 14, 5–8. [Google Scholar]
- Konoshima, M.; Ikeshiro, Y. Fukugiside, the first biflavonoid glycoside from garcinia spicata hook. f. Tetrahedron Lett. 1970, 11, 1717–1720. [Google Scholar] [CrossRef]
- Terashima, K.; Aqil, M.; Niwa, M. Garcinianin, a novel biflavonoid from the roots of garcinia kola. Heterocycles 1995, 41, 2245–2250. [Google Scholar]
- Osorio, E.; Londono, J.; Bastida, J. Low-density lipoprotein (LDL)-antioxidant biflavonoids from Garcinia madruno. Molecules 2013, 18, 6092–6100. [Google Scholar] [CrossRef] [PubMed]
- Saelee, A.; Phongpaichit, S.; Mahabusarakam, W. A new prenylated biflavonoid from the leaves of Garcinia dulcis. Nat. Prod. Res. 2015, 29, 1884–1888. [Google Scholar] [CrossRef]
- Ren, Y.; de Blanco, E.J.C.; Fuchs, J.R.; Soejarto, D.D.; Burdette, J.E.; Swanson, S.M.; Kinghorn, A.D. Potential Anticancer Agents Characterized from Selected Tropical Plants. J. Nat. Prod. 2019, 82, 657–679. [Google Scholar] [CrossRef]
- Joshi, B.S.; Kamat, V.N.; Viswanathan, N. The isolation and structure of two biflavones from Garcinia talboti. Phytochemistry 1970, 9, 881–888. [Google Scholar] [CrossRef]
- Kim, C.S.; Bae, M.; Oh, J.; Subedi, L.; Suh, W.S.; Choi, S.Z.; Son, M.W.; Kim, S.Y.; Choi, S.U.; Oh, D.C.; et al. Anti-Neurodegenerative Biflavonoid Glycosides from Impatiens balsamina. J. Nat. Prod. 2017, 80, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Bao-Min, F.; Yue-Hu, F.; Yong-Qi, W. Distribution of Biflavonoids in Six Species of Thymelaeaceae. J. Dalian Univ. 2003, 24, 95–98+112. [Google Scholar]
- Iwu, M.; Igboko, O. Flavonoids of Garcinia kola Seeds. J. Nat. Prod. 1982, 45, 650–651. [Google Scholar] [CrossRef]
- Cechinel Filho, V.; da Silva, K.L.; de Souza, M.M.; Oliveira, A.E.; Yunes, R.A.; Guimaraes, C.L.; Verdi, L.G.; Simionatto, E.L.; Delle Monache, F. I3-naringenin-II8--4′OMe-eriodictyol: A new potential analgesic agent isolated from Rheedia gardneriana leaves. Z. Nat. C J. Biosci. 2000, 55, 820–823. [Google Scholar]
- Konoshima, M.; Ikeshiro, Y.; Miyahara, S.; Yen, K.-Y. The constitution of biflavonoids from Garcinia plants. Tetrahedron Lett. 1970, 11, 4203–4206. [Google Scholar] [CrossRef]
- Stark, T.D.; Germann, D.; Balemba, O.B.; Wakamatsu, J.; Hofmann, T. New Highly in Vitro Antioxidative 3,8″-Linked Biflav(an)ones and Flavanone-C-glycosides from Garcinia buchananii Stem Bark. J. Agric. Food Chem. 2013, 61, 12572–12581. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.F.; Camara, C.A.; Agra, M.D.F.; Silva, T.M.S. Biflavonoids from the unripe fruits of Clusia paralicola and their antioxidant activity. Nat. Prod. Commun. 2012, 7, 1597–1600. [Google Scholar] [CrossRef]
- Kitanov, G.M. Biflavone, flavonol, and xanthone glycosides from Hypericum aucheri. Chem. Nat. Compd. 1988, 24, 390–391. [Google Scholar] [CrossRef]
- Terashima, K.; Kondo, Y.; Aqil, M.; Waziri, M.; Niwa, M. A study of biflavanones from the stems of garcinia kola (GUTTIFERAE). Heterocycles 1999, 50, 283–290. [Google Scholar]
- Loo, P.V.; Bruyn, A.D.; Verzele, M. On the liquid chromatography and identification of the flavonoids, present in the “sumach tannic acid” extracted fromRhus coriaria. Chromatographia 1988, 25, 15–20. [Google Scholar]
- Zhang, X.; Wang, G.; Huang, W.; Ye, W.; Li, Y. Biflavonoids from the Roots of Wikstroemia indica. Nat. Prod. Commun. 2011, 6, 1111–1114. [Google Scholar] [CrossRef]
- Ito, C.; Itoigawa, M.; Miyamoto, Y.; Rao, K.S.; Takayasu, J.; Okuda, Y.; Mukainaka, T.; Tokuda, H.; Nishino, H.; Furukawa, H. A New Biflavonoid from Calophyllum panciflorum with Antitumor-Promoting Activity. J. Nat. Prod. 1999, 62, 1668–1671. [Google Scholar] [CrossRef]
- Li, X.-C.; Joshi, A.S.; Tan, B.; ElSohly, H.N.; Walker, L.A.; Zjawiony, J.K.; Ferreira, D. Absolute configuration, conformation, and chiral properties of flavanone-(3→8″)-flavone biflavonoids from Rheedia acuminata. Tetrahedron 2002, 58, 8709–8717. [Google Scholar] [CrossRef]
- Babu, V.; Ali, S.M.; Sultana, S.; Ilyas, M. A biflavonoid from Garcinia nervosa. Phytochemistry 1988, 27, 3332–3335. [Google Scholar] [CrossRef]
- Ferrari, J.; Terreaux, C.; Kurtán, T.; Szikszai-Kiss, A.; Antus, S.; Msonthi, J.D.; Hostettmann, K. Isolation and On-Line LC/CD Analysis of 3,8”-Linked Biflavonoids from Gnidia involucrata. Helv. Chim. Acta 2003, 86, 2768–2778. [Google Scholar] [CrossRef]
- Niwa, M.; Terashima, K.; Ishida, T.; Furukawa, T.; Takaya, Y. Constituents of green and ripened fruit of Garcinia subelliptica. Heterocycles 2008, 75, 407–413. [Google Scholar] [CrossRef]
- Jingxian, P.; Huyi, Z.; Xianbin, Y.; Meifang, H. Biflavones from the testa of Ginkgo biloba L. J. Plant Resour. Environ. 1995, 4, 17–21. [Google Scholar]
- Wenli, M.; Jiao, W.; Haofu, D. Advances in studies on chemical constituents in plants of Cephalotaxus Sieb. et Zucc. and their pharmacological activities. Chin. Tradit. Herb. Drugs 2006, 37, 452–458. [Google Scholar]
- Chun, Y.; Jun-Song, W.; Ling-Yi, K. A new biflavone from needles of Taxus canadensis. China J. Chin. Mater. Med. 2016, 41, 443–445. [Google Scholar]
- Murthy, S.S.N. A biflavanone from Semecarpus anacardium. Phytochemistry 1983, 22, 2636–2638. [Google Scholar] [CrossRef]
- Zhang, R.R.; Lin, Z.X.; Lu, X.Y.; Xia, X.; Jiang, R.W.; Chen, Q.B. CGY-1, a biflavonoid isolated from cardiocrinum giganteum seeds, improves memory deficits by modulating the cholinergic system in scopolamine-treated mice. Biomed. Pharm. 2019, 111, 496–502. [Google Scholar] [CrossRef]
- Xia, X.H.; Zhang, Y.; Xi, Y.B.; Wang, G.H.; Yang, L.Q.; Xue, K.F. Advances in Studies on Chemical Constituents and Bioactivites Actions of Ginkgo Biloba, L. Chin. J. Exp. Tradit. Med. Formulae 2009, 15, 100–104. [Google Scholar]
- Li, M.; Li, B.; Xia, Z.M.; Tian, Y.; Zhang, D.; Rui, W.J.; Dong, J.X.; Xiao, F.J. Anticancer Effects of Five Biflavonoids from Ginkgo Biloba, L. Male Flowers In Vitro. Molecules 2019, 24, 1496. [Google Scholar] [CrossRef]
- Xiao, S.; Mu, Z.Q.; Cheng, C.R.; Ding, J. Three new biflavonoids from the branches and leaves of Cephalotaxus oliveri and their antioxidant activity. Nat. Prod. Res. 2019, 33, 321–327. [Google Scholar] [CrossRef]
- Liu, T.X.; Wang, S.H. Research Progress on Use of Cycas Revolute. J. MUC (Natural Sciences Edition) 2016, 25, 49–54. [Google Scholar]
- Das, B.; Mahender, G.; Koteswara Rao, Y.; Prabhakar, A.; Jagadeesh, B. Biflavonoids from Cycas beddomei. Chem. Pharm. Bull. 2005, 53, 135–136. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Huang, J.; Hua, H.; Sun, B.; Gao, H.; Wu, L. A new biflavone from the twigs and leaves of Taxus cuspidata Sieb et Zucc. Asian J. Tradit. Med. 2007, 2, 235–238. [Google Scholar]
- Sun, M.; Feng, X.; Yin, M.; Chen, Y.; Zhao, X.; Dong, Y. A biflavonoid from stems and leaves of Lonicera macranthoides. Chem. Nat. Compd. 2012, 48, 231–233. [Google Scholar] [CrossRef]
- Rao, N.S.P.; Row, L.R.; Brown, R.T. Phenolic constituents of Semecarpus anacardium. Phytochemistry 1973, 12, 671–681. [Google Scholar] [CrossRef]
- Murthy, S.S.; Rao, N.S.; Anjaneyulu, A.S.; Row, L.R. Confirmation of structures of semecarpus biflavanones A1 and A2. Planta Med. 1981, 43, 46–50. [Google Scholar] [CrossRef]
- Murthy, S.S.N. New biflavonoid from Semercarpus anacardium Linn. Chim. Acta Turc. Istanb. 1992, 20, 33. [Google Scholar]
- Thompson, R.S.; Jacques, D.; Haslam, E.; Tanner, R.J.N. Plant proanthocyanidins. Part I. Introduction; the isolation, structure, and distribution in nature of plant procyanidins. J. Chem. Soc. Perkin Trans. 1 1972, 1387–1399. [Google Scholar] [CrossRef]
- Ölschläger, C.; Regos, I.; Zeller, F.J.; Treutter, D. Identification of galloylated propelargonidins and procyanidins in buckwheat grain and quantification of rutin and flavanols from homostylous hybrids originating from F. esculentum×F. homotropicum. Phytochemistry 2008, 69, 1389–1397. [Google Scholar] [CrossRef]
- Kashiwada, Y.; Nonaka, G.-I.; Nishioka, I. Tannins and Related Compounds. XLVIII.: Rhubarb. (7). Isolation and Characterization of New Dimeric and Trimeric Procyanidins. Chem. Pharm. Bull. 1986, 34, 4083–4091. [Google Scholar] [CrossRef]
- Wang, J.-N.; Hano, Y.; Nomura, T.; Chen, Y.-J. Procyanidins from the seeds of Vitis amurensis. Phytochemistry 2000, 53, 1097–1102. [Google Scholar] [CrossRef]
- Nilanonta, C.; Isaka, M.; Kittakoop, P.; Palittapongarnpim, P.; Kamchonwongpaisan, S.; Pittayakhajonwut, D.; Tanticharoen, M.; Thebtaranonth, Y. Antimycobacterial and Antiplasmodial Cyclodepsipeptides from the Insect Pathogenic Fungus Paecilomyces tenuipes BCC 1614. Planta Med. 2000, 66, 756–758. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, F.; Nonaka, G.-i.; Nishioka, I. Tannins and Related Compounds. XC: 8-C-Ascorbyl (−)-Epigallocatechin 3-O-Gallate and Novel Dimeric Flavan -3-ols, Oolonghomobisflavans A and B, from Oolong Tea. (3). Chem. Pharm. Bull. 1989, 37, 3255–3263. [Google Scholar] [CrossRef]
- Danne, A.; Petereit, F.; Nahrstedt, A. Flavan-3-ols, prodelphinidins and further polyphenols from Cistus salvifolius. Phytochemistry 1994, 37, 533–538. [Google Scholar] [CrossRef]
- Nonaka, G.-I.; Muta, M.; Nishioka, I. Myricatin, a galloyl flavanonol sulfate and prodelphinidin gallates from Myrica rubra. Phytochemistry 1983, 22, 237–241. [Google Scholar] [CrossRef]
- Geiss, F.; Heinrich, M.; Hunkler, D.; Rimpler, H. Proanthocyanidins with (+)-epicatechin units from Byrsonima crassifolia bark. Phytochemistry 1995, 39, 635–643. [Google Scholar] [CrossRef]
- Viviers, P.M.; Young, D.A.; Botha, J.J.; Ferreira, D.; Roux, D.G.; Hull, W.E. Synthesis of condensed tannins. Part 6. The sequence of units, coupling positions and absolute configuration of the first linear [4,6:4,6]-triflavanoid with terminal 3,4-diol function. J. Chem. Soc. Perkin Trans. 1 1982, 13, 535–540. [Google Scholar] [CrossRef]
- Steenkamp, J.A.; Malan, J.C.S.; Roux, D.G.; Ferreira, D. Oligomeric flavanoids. Part 1. Novel dimeric profisetinidins from Colophospermum mopane. J. Chem. Soc. Perkin Trans. 1 1988, 6, 1325–1330. [Google Scholar] [CrossRef]
- Ferreira, D.; Cornelius du Preez, I.; Wijnmaalen, J.C.; Roux, D.G. Biflavanoid proguibourtinidin carboxylic acids and their biflavanoid homologues from Acacia luederitzii. Phytochemistry 1985, 24, 2415–2422. [Google Scholar] [CrossRef]
- Malan, E.; Swinny, E.; Ferreira, D.; Steynberg, P. The structure and synthesis of proguibourtinidins from Cassia abbreviata. Phytochemistry 1996, 41, 1209–1213. [Google Scholar] [CrossRef]
- Park, K.H.; Kim, S.K.; Choi, S.E.; Kwon, J.H.; Oh, M.H.; Lee, M.W. Three New Stereoisomers of Condensed Tannins from the Roots of Rosa multiflora. Chem. Pharm. Bull. 2010, 58, 1227–1231. [Google Scholar] [CrossRef][Green Version]
- Hemingway, R.W.; Foo, L.Y.; Porter, L.J. Linkage isomerism in trimeric and polymeric 2,3-cis-procyanidins. J. Chem. Soc. Perkin Trans. 1 1982, 13, 1209–1216. [Google Scholar] [CrossRef]
- Yeap Foo, L.; Karchesy, J.J. Procyanidin dimers and trimers from Douglas fir inner bark. Phytochemistry 1989, 28, 1743–1747. [Google Scholar] [CrossRef]
- Foo, L.Y.; Newman, R.; Waghorn, G.; McNabb, W.C.; Ulyatt, M.J. Proanthocyanidins from Lotus corniculatus. Phytochemistry 1996, 41, 617–624. [Google Scholar] [CrossRef]
- Malan, E.; Sireeparsad, A. The structure and synthesis of the first dimeric proteracacinidins from acacia galpinii. Phytochemistry 1995, 38, 237–239. [Google Scholar] [CrossRef]
- Bennie, L.; Coetzee, J.; Malan, E.; Ferreira, D. (4→6)-Coupled proteracacinidins and promelacacinidins from Acacia galpinii and Acacia caffra. Phytochemistry 2002, 60, 521–532. [Google Scholar] [CrossRef]
- Li, J.; Xu, P.-S.; Zou, Z.-X.; Zou, H.; Long, H.-P.; Tan, L.-H.; Liu, R.-H.; Wang, Y.-K.; Xu, K.-P.; Tan, G.-S. Three new compounds from the roots of Juglans mandshurica Maxim. Phytochem. Lett. 2017, 20, 40–44. [Google Scholar] [CrossRef]
- Lou, H.; Yuan, H.; Ma, B.; Ren, D.; Ji, M.; Oka, S. Polyphenols from peanut skins and their free radical-scavenging effects. Phytochemistry 2004, 65, 2391–2399. [Google Scholar] [CrossRef]
- Nonaka, G.; Nishioka, I.; Nagasawa, T.; Oura, H. Tannins and Related Compounds. I. Rhubarb (1). Chem. Pharm. Bull. 1981, 29, 2862–2870. [Google Scholar] [CrossRef]
- Bekker, M.; Bekker, R.; Brandt, V.E. Two flavonoid glycosides and a miscellaneous flavan from the bark of Guibourtia coleosperma. Phytochemistry 2006, 67, 818–823. [Google Scholar] [CrossRef]
- Cheng, H.-Y.; Yang, C.-M.; Lin, T.-C.; Shieh, D.-E.; Lin, C.-C. ent-Epiafzelechin-(4alpha-->8)-epiafzelechin extracted from Cassia javanica inhibits herpes simplex virus type 2 replication. J. Med. Microbiol. 2006, 55, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Hartisch, C.; Kolodziej, H. Galloylhamameloses and proanthocyanidins from Hamamelis virginiana. Phytochemistry 1996, 42, 191–198. [Google Scholar] [CrossRef]
- Nonaka, G.-I.; Miwa, N.; Nishioka, I. Stilbene glycoside gallates and proanthocyanidins from Polygonum multiflorum. Phytochemistry 1982, 21, 429–432. [Google Scholar] [CrossRef]
- Hsu, F.-L.; Nonaka, G.-I.; Nishioka, I. Acylated flavanols and procyanidins from Salix sieboldiana. Phytochemistry 1985, 24, 2089–2092. [Google Scholar] [CrossRef]
- Lokvam, J.; Coley, P.D.; Kursar, T.A. Cinnamoyl glucosides of catechin and dimeric procyanidins from young leaves of Inga umbellifera (Fabaceae). Phytochemistry 2004, 65, 351–358. [Google Scholar] [CrossRef]
- Zhang, B.; Nonaka, G.-I.; Nishioka, I. Potentillanin, a biflavanoid and a procyanidin glycoside from Potentilla viscosa. Phytochemistry 1988, 27, 3277–3280. [Google Scholar] [CrossRef]
- Ishimaru, K.; Nonaka, G.-I.; Nishioka, I. Flavan-3-ol and procyanidin glycosides from quercus miyagii. Phytochemistry 1987, 26, 1167–1170. [Google Scholar] [CrossRef]
- Tanaka, T.; Nonaka, G.-I.; Nishioka, I. 7-O-Galloyl-(+)-catechin and 3-O-galloylprocyanidin B-3 from Sanguisorba officinalis. Phytochemistry 1983, 22, 2575–2578. [Google Scholar] [CrossRef]
- Abe, I.; Seki, T.; Noguchi, H.; Kashiwada, Y. Galloyl Esters from Rhubarb are Potent Inhibitors of Squalene Epoxidase, a Key Enzyme in Cholesterol Biosynthesis. Planta Med. 2000, 66, 753–756. [Google Scholar] [CrossRef] [PubMed]
- Liimatainen, J.; Karonen, M.; Sinkkonen, J. Procyanidin xylosides from the bark of Betula pendula. Phytochemistry 2012, 76, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.J. Isolation of 3-Galloylprocyanidin B3, a Glucosyltransferase Inhibitor from the Korean Green Tea Leaves. J. Appl. Biol. Chem. 2000, 43, 273–276. [Google Scholar]
- Cong, H.J.; Zhang, S.W.; Zhang, C.; Huang, Y.J.; Xuan, L.J. A novel dimeric procyanidin glucoside from Polygonum aviculare. Chin. Chem. Lett. 2012, 23, 820–822. [Google Scholar] [CrossRef]
- Ozawa, T.; Hiroto, M.; Imagawa, H. Procyanidins from Sago Palm Pith. Agric. Biol. Chem. 1990, 54, 217–218. [Google Scholar]
- Reddy, K.R.S.; Srimannarayana, G.; Rao, N.V.S. Ein proanthocyanidin-dimenes aus cassia auriculata-blumen. Cheminform 1973, 4, 291. [Google Scholar]
- Ariga, T.; Asao, Y. Isolation, Identification and Organoleptic Astringency of Dimeric Proanthocyanidins Occurring in Azuki Beans. Agric. Biol. Chem. 1981, 45, 2709–2712. [Google Scholar]
- Middelkoop, T.B.; Labadie, R.P. The Action of Saraca asoca Roxb. de Wilde Bark on the PGH2 Synthetase Enzyme Complex of the Sheep Vesicular Gland. Z. Nat. C 1985, 40, 523–526. [Google Scholar] [CrossRef]
- Morimoto, S.; Nonaka, G.-I.; Chen, R.-F.; Nishioka, I. Tannins and Related Compounds. LXI: Isolation and Structures of Novel Bi- and Triflavanoids from the Leaves of Cassia fistula L. Chem. Pharm. Bull. 1988, 36, 39–47. [Google Scholar] [CrossRef]
- Weinges, K.; Göritz, K.; Nader, F. Zur Kenntnis der Proanthocyanidine, XI1) Konfigurationsbestimmung von C30H26O12-Procyanidinen und Strukturaufklärung eines neuen Procyanidins. Eur. J. Org. Chem. 1968, 715, 164–171. [Google Scholar]
- Kashiwada, Y.; Iizuka, H.; Yoshioka, K.; Chen, R.-F.; Nonaka, G.-i.; Nishioka, I. Tannins and Related Compounds. XCIII: Occurrence of Enantiomeric Proanthocyanidins in the Leguminosae Plants, Cassia fistula L. and C. javanica L. Chem. Pharm. Bull. 1990, 38, 888–893. [Google Scholar] [CrossRef]
- Nunes, D.S.; Haag, A.; Bestmann, H.-J. Two proanthocyanidins from the bark of Dalbergia monet̊ari. Phytochemistry 1989, 28, 2183–2186. [Google Scholar] [CrossRef]
- Messanga, B.B.; Ghogomu, R.; Sondengam, B.L.; Martin, M.-T.; Blond, A.; Brouard, J.-P.; Bodo, B. Calodenin C: A New Guibourtinidol-(4α→8)-afzelechin from Ochna calodendron. Planta Med. 1998, 64, 760–761. [Google Scholar] [CrossRef] [PubMed]
- Bicker, J.; Petereit, F.; Hensel, A. Proanthocyanidins and a phloroglucinol derivative from Rumex acetosa L. Fitoterapia 2009, 80, 483–495. [Google Scholar] [CrossRef] [PubMed]
- Monache, F.D.; Pomponi, M.; Marini-Bettolo, G.B.; D’Albuquerque, I.L.; de Lima, O.G. A methylated catechin and proanthocyanidins from the celastraceae. Phytochemistry 1976, 15, 573–574. [Google Scholar] [CrossRef]
- Nonaka, G.; Nishioka, I. Novel Biflavonoids, Chalcan-flavan Dimers from Gambir. Chem. Pharm. Bull. 1980, 28, 3145–3149. [Google Scholar] [CrossRef]
- Hsu, F.; Nonaka, G.; Nishioka, I. Tannins and Related Compounds. XXXI. Isolation and Characterization of Proanthocyanidins in Kandelia candel (L.) DRUCE. Chem. Pharm. Bull. 1985, 33, 3142–3152. [Google Scholar] [CrossRef]
- Achenbach, H.; Benirschke, G. Joannesialactone and other compounds from Joannesia princeps. Phytochemistry 1997, 45, 149–157. [Google Scholar] [CrossRef]
- Karioti, A.; Bilia, A.R.; Gabbiani, C.; Messori, L.; Skaltsa, H. Proanthocyanidin glycosides from the leaves of Quercus ilex L. (Fagaceae). Tetrahedron Lett. 2009, 50, 1771–1776. [Google Scholar] [CrossRef]
- Wu, B.; Wang, K.; Wu, X. A New Phenolic Diglycoside Produced in Response to Copper Toxicity and a New Flavan Dimer from the Leaves of Viburnum ichangense (Hemsl.) Rehd. Helv. Chim. Acta 2011, 94, 1677–1684. [Google Scholar] [CrossRef]
- Foo, L.Y.; Karchesy, J.J. Procyanidin polymers of Douglas fir bark: Structure from degradation with phloroglucinol. Phytochemistry 1989, 28, 3185–3190. [Google Scholar] [CrossRef]
- Qa’dan, F.; Petereit, F.; Mansoor, K.; Nahrstedt, A. Antioxidant oligomeric proanthocyanidins from Cistus salvifolius. Nat. Prod. Res. 2006, 20, 1216–1224. [Google Scholar] [CrossRef]
- Kusano, R.; Ogawa, S.; Matsuo, Y.; Tanaka, T.; Yazaki, Y.; Kouno, I. α-Amylase and Lipase Inhibitory Activity and Structural Characterization of Acacia Bark Proanthocyanidins. J. Nat. Prod. 2011, 74, 119–128. [Google Scholar] [CrossRef]
- Palazzo de Mello, J.; Petereit, F.; Nahrstedt, A. Prorobinetinidins from Stryphnodendron adstringens. Phytochemistry 1996, 42, 857–862. [Google Scholar] [CrossRef]
- Botha, J.J.; Ferreira, D.; Roux, D.G. Synthesis of condensed tannins. Part 4. A direct biomimetic approach to [4,6]-and [4,8]-biflavanoids. J. Chem. Soc. Perkin Trans. 1 1981, 12, 1235–1245. [Google Scholar] [CrossRef]
- Viviers, P.M.; Botha, J.J.; Ferreira, D.; Roux, D.G.; Saayman, H.M. Synthesis of condensed tannins. Part 7. Angular [4,6: 4,8]-prorobinetinidin triflavanoids from black wattle (‘Mimosa’) bark extract. J. Chem. Soc. Perkin Trans. 1 1983, 14, 17–22. [Google Scholar] [CrossRef]
- Palazzo de Mello, J.C.; Petereit, F.; Nahrstedt, A. A dimeric proanthocyanidin from Stryphnodendron adstringens. Phytochemistry 1999, 51, 1105–1107. [Google Scholar] [CrossRef]
- Makhmatkulov, A.B.; Kuliev, Z.A.; Vdovin, A.D.; Malikov, V.M. Proanthocyanidins ofPolygonum corarium. II. Chem. Nat. Compd. 1994, 30, 214–222. [Google Scholar] [CrossRef]
- Dawang, S.; Zuchun, Z.; Wong, H.; Lai, Y.F. Tannins and other phenolics from Myrica esculenta bark. Phytochemistry 1988, 27, 579–583. [Google Scholar] [CrossRef]
- Kwan Hu, K.; Kuliev, Z.A.; Vdovin, A.D.; Yagudaev, M.R.; Malikov, V.M. Structure of rhodisin and rhodisinoside. Chem. Nat. Compd. 1989, 25, 618–619. [Google Scholar] [CrossRef]
- Hussein, G.; Nakamura, N.; Meselhy, M.R.; Hattori, M. Phenolics from Maytenus senegalensis. Phytochemistry 1999, 50, 689–694. [Google Scholar] [CrossRef]
- Nonaka, G.-I.; Sakai, R.; Nishioka, I. Hydrolysable tannins and proanthocyanidins from green tea. Phytochemistry 1984, 23, 1753–1755. [Google Scholar] [CrossRef]
- Danne, A.; Petereit, F.; Nahrstedt, A. Proanthocyanidins from Cistus incanus. Phytochemistry 1993, 34, 1129–1133. [Google Scholar] [CrossRef]
- Schmidt, C.A.; Murillo, R.; Heinzmann, B.; Laufer, S.; Wray, V.; Merfort, I. Structural and Conformational Analysis of Proanthocyanidins from Parapiptadenia rigida and Their Wound-Healing Properties. J. Nat. Prod. 2011, 74, 1427–1436. [Google Scholar] [CrossRef]
- Zhang, Y.-J.; Tanaka, T.; Iwamoto, Y.; Yang, C.-R.; Kouno, I. Novel Norsesquiterpenoids from the Roots of Phyllanthus emblica. J. Nat. Prod. 2000, 63, 1507–1510. [Google Scholar] [CrossRef]
- Gupta, R.K.; Haslam, E. Plant proanthocyanidins. Part 7. Prodelphinidins from Pinus sylvestris. J. Chem. Soc. Perkin Trans. 1 1981, 12, 1148–1150. [Google Scholar] [CrossRef]
- Foo, L.Y.; Porter, L.J. Prodelphinidin polymers: Definition of structural units. J. Chem. Soc. Perkin Trans. 1 1978, 10, 1186–1190. [Google Scholar] [CrossRef]
- Hashimoto, F.; Nonaka, G.-i.; Nishioka, I. Tannins and Related Compounds. LXXVII: Novel Chalcan-flavan Dimers, Assamicains A, B and C, and a New Flavan-3-ol and Proanthocyanidins from the Fresh Leaves of Camella sinensis L. var. assamica KITAMURA. Chem. Pharm. Bull. 1989, 37, 77–85. [Google Scholar] [CrossRef]
- Weinges, K.; Schick, H. Dodecaacetylprodelphinidin B3 from the dried leaves of Ziziphus spina-christi. Phytochemistry 1995, 38, 505–507. [Google Scholar] [CrossRef]
- Sun, D.; Wong, H.; Foo, L.Y. Proanthocyanidin dimers and polymers from Quercus dentata. Phytochemistry 1987, 26, 1825–1829. [Google Scholar] [CrossRef]
- Krishnamoorthy, V.; Seshadri, T.R. A new proanthocyanidin from the stem bark of Myrica nagi thumb. Tetrahedron 1966, 22, 2367–2371. [Google Scholar] [CrossRef]
- Steynberg, J.P.; Steynberg, J.P.; Vincent Brandt, E.; Ferreira, D.; Hemingway, R.W. Oligomeric flavanoids. Part 26. Structure and synthesis of the first profisetinidins with epifisetinidol constituent units. J. Chem. Soc. Perkin Trans. 1 1997, 13, 1943–1950. [Google Scholar] [CrossRef]
- Malan, J.C.S.; Young, D.A.; Steenkamp, J.A.; Ferreira, D. Oligomeric flavanoids. Part 2. The first profisetinidins with dihydroflavonol constituent units. J. Chem. Soc. Perkin Trans. 1 1988, 9, 2567–2572. [Google Scholar] [CrossRef]
- Steynberg, J.P.; Burger, J.F.W.; Malan, J.C.S.; Cronjé, A.; Young, D.A.; Ferreira, D. Natural (−)-fisetinidol-(4,8)-(−)-epicatechin profisetinidins. Phytochemistry 1990, 29, 275–277. [Google Scholar] [CrossRef]
- Drewes, S.E.; Roux, D.G.; Eggers, S.H.; Feeney, J. Three diastereoisomeric 4,6-linked bileucofisetinidins from the heartwood of Acacia mearnsii. J. Chem. Soc. C Org. 1967, 1217–1227. [Google Scholar] [CrossRef]
- Hatano, T.; Yamashita, A.; Hashimoto, T.; Ito, H.; Kubo, N.; Yoshiyama, M.; Shimura, S.; Itoh, Y.; Okuda, T.; Yoshida, T. Flavan dimers with lipase inhibitory activity from Cassia nomame. Phytochemistry 1997, 46, 893–900. [Google Scholar] [CrossRef]
- Akhavan, M.; Shafaghat, A.; Salimi, F. Novel acetylated chalcone and biflavonoid glycosides from Trigonosciadium brachytaenium (Boiss.) Alava. Nat. Prod. Res. 2013, 27, 2111–2117. [Google Scholar] [CrossRef]
- Yadav, S.; Bhadoria, B.K. Two dimeric flavonoids from Baiihinia purpured. Indian J. Chem. Sect. B Org. Chem. Incl. Med. Chem. 2005, 44, 2604–2607. [Google Scholar]
- Felício, J.D.A.; Gonçalez, E.; Braggio, M.M.; Costantino, L.; Albasini, A.; Lins, A.P. Inhibition of lens aldose reductase by biflavones from Ouratea spectabilis. Planta Med. 1995, 61, 217–220. [Google Scholar] [CrossRef]
- Khan, N.U.; Ilyas, M.; Rahman, W.; Mashima, T.; Okigawa, M.; Kawano, N. Biflavones from the leaves of Araucaria bidwillii Hooker and Agathis alba foxworthy (araucariaceae). Tetrahedron 1972, 28, 5689–5695. [Google Scholar] [CrossRef]
- Ilyas, M.; Seligmann, O.; Wagner, H. Biflavones from the Leaves of Araucaria rulei F. Muell. and a Survey on Biflavanoids of the Araucaria Genus. Z. Nat. C 1977, 32, 206–209. [Google Scholar] [CrossRef]
- Ngo Mbing, J.; Enguehard-Gueiffier, C.; Atchadé, A.d.T.; Allouchi, H.; Gangoué-Piéboji, J.; Mbafor, J.T.; Tih, R.G.; Pothier, J.; Pegnyemb, D.E.; Gueiffier, A. Two biflavonoids from Ouratea nigroviolacea. Phytochemistry 2006, 67, 2666–2670. [Google Scholar] [CrossRef]
- Chen, F.-C.; Lin, Y.-M. Rhusflavanone, a new biflavanone from the seeds of wax-tree. J. Chem. Soc. Perkin Trans. 1 1976, 1, 98–101. [Google Scholar] [CrossRef]
- Ndongo, J.T.; Shaaban, M.; Mbing, J.N.; Bikobo, D.N.; Atchadé, A.d.T.; Pegnyemb, D.E.; Laatsch, H. Phenolic dimers and an indole alkaloid from Campylospermum flavum (Ochnaceae). Phytochemistry 2010, 71, 1872–1878. [Google Scholar] [CrossRef]
- Shrestha, S.; Park, J.H.; Lee, D.Y.; Cho, J.G.; Cho, S.; Yang, H.J.; Yong, H.I.; Yoon, M.S.; Han, D.S.; Baek, N.I. Rhus parviflora and its biflavonoid constituent, rhusflavone, induce sleep through the positive allosteric modulation of GABA(A)-benzodiazepine receptors. J. Ethnopharmacol. 2012, 142, 213–220. [Google Scholar] [CrossRef] [PubMed]
- D’Arc Felicio, J.; Rossi, M.H.; Park, H.R.; Gonçalez, E.; Braggio, M.M.; David, J.M.; Cordeiro, I. Biflavonoids from Ouratea multiflora. Fitoterapia 2001, 72, 453–455. [Google Scholar] [CrossRef]
- Moreira, I.C.; de Carvalho, M.G.; Bastos, A.B.F.O.; Braz-Filho, R. A flavone dimer from Ouratea hexasperma. Phytochemistry 1999, 51, 833–838. [Google Scholar] [CrossRef]
- Sharma, S.K.; Vasudeva, N.; Rathi, P.; Ali, M. Isolation and identification of a new phytosterol ester from tephrosia purpurea (linn.) pers. root. Int. J. Chem. Sci. 2008, 6, 1734–1741. [Google Scholar]
- Yan, X.X.; Pan, Z.H.; Cheng, L.; Ning, D.S.; Zu-Qiang, L.I.; Luo, L. Chemical constituents of Sabina squamata(1). Guihaia 2015, 35, 428–430. [Google Scholar]
- Ye, Y.; Guo, Y.; Luo, Y.T.; Wang, Y.F. Isolation and free radical scavenging activities of a novel biflavonoid from the shells of Camellia oleifera Abel. Fitoterapia 2012, 83, 1585–1589. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Lin, Y.; Ho, T.; Ueng, T. Synthesis of Hexa-O-methyl-8,8”-binaringenin. Cheminform 1975, 3, 833–836. [Google Scholar] [CrossRef]
- Ferraro, G.E.; Martino, V.S.; Coussio, J.D. 4′,4”-Dimethylcuppressuflavanone from Eupatorium subhastatum. J. Nat. Prod. 1988, 51, 586–587. [Google Scholar] [CrossRef]
- Miceli, N.; Trovato, A.; Dugo, P.; Cacciola, F.; Donato, P.; Marino, A.; Bellinghieri, V.; Barbera, T.M.L.; Güvenç, A.; Taviano, M.F. Comparative analysis of flavonoid profile, antioxidant and antimicrobial activity of the berries of Juniperus communis L. var. communis and Juniperus communis L. var. saxatilis Pall. from Turkey. J. Agric. Food Chem. 2009, 57, 6570–6577. [Google Scholar] [CrossRef] [PubMed]
- Ofman, D.J.; Markham, K.R.; Vilain, C.; Molloy, B.P.J. Flavonoid profiles of New Zealand kauri and other species of Agathis. Phytochemistry 1995, 38, 1223–1228. [Google Scholar] [CrossRef]
- Ilyas, M.; Usmani, J.N.; Bhatnagar, S.P.; Ilyas, M.; Rahman, W. WB1 and W11, the first optically active biflavones. Tetrahedron Lett. 1968, 9, 5515–5517. [Google Scholar] [CrossRef]
- Meselhy, M.R. Constituents from Moghat, the Roots of Glossostemon bruguieri (Desf.). Molecules 2003, 8, 614–621. [Google Scholar] [CrossRef]
- Chen, F.-C.; Lin, Y.-M.; Lin, Y.-C. Neorhusflavanone, a New Biflavanone from Wax-tree. Heterocycles 1978, 9, 663–668. [Google Scholar] [CrossRef]
- Adjapmoh, M.F.; Toze, F.A.; Songue, J.L.; Langat, M.K.; Kapche, G.D.; Hameed, A.; Lateef, M.; Shaiq, M.A.; Mbaze, L.M.; Wansi, J.D.; et al. A New Ceramide and Biflavonoid from the Leaves of Parinari hypochrysea (Chrysobalanaceae). Nat. Prod. Commun. 2016, 11, 615–620. [Google Scholar] [CrossRef] [PubMed]
- Jia, B.X.; Ren, F.X.; Jia, L.; Chen, X.Q.; Yang, J.; Wang, Q. Baeckein E, a new bioactive C-methylated biflavonoid from the roots of Baeckea frutescens. Nat. Prod. Res. 2013, 27, 2069–2075. [Google Scholar] [CrossRef]
- Okigawa, M.; Kawano, N.; Aqil, M.; Rahman, W. Ochnaflavone and its derivatives: A new series of diflavonyl ethers from Ochna squarrosa Linn. J. Chem. Soc. Perkin Trans. 1 1976, 5, 580–583. [Google Scholar] [CrossRef]
- Ma, J.L.; Li, N.; Li, X. One new biflavone glucoside from the leaves of Lonicera japonica Thunb. Chin. J. Med. Chem. 2009, 19, 63–64. [Google Scholar]
- Likhitwitayawuid, K.; Rungserichai, R.; Ruangrungsi, N.; Phadungcharoen, T. Flavonoids from Ochna integerrima. Phytochemistry 2001, 56, 353–357. [Google Scholar] [CrossRef]
- Jayakrishna, G.; Reddy, M.K.; Jayaprakasam, B.; Gunasekar, D.; Blond, A.; Bodo, B. A new biflavonoid from Ochna beddomei. J. Asian Nat. Prod. Res. 2003, 5, 83–87. [Google Scholar] [CrossRef]
- Pegnyemb, D.E.; Mbing, J.N.; de Theodore Atchade, A.; Tih, R.G.; Sondengam, B.L.; Blond, A.; Bodo, B. Antimicrobial biflavonoids from the aerial parts of Ouratea sulcata. Phytochemistry 2005, 66, 1922–1926. [Google Scholar] [CrossRef]
- Makhafola, T.J.; Samuel, B.B.; Elgorashi, E.E.; Eloff, J.N. Ochnaflavone and ochnaflavone 7-O-methyl ether two antibacterial biflavonoids from Ochna pretoriensis (Ochnaceae). Nat. Prod. Commun. 2012, 7, 1601–1604. [Google Scholar] [CrossRef]
- Reutrakul, V.; Ningnuek, N.; Pohmakotr, M.; Yoosook, C.; Napaswad, C.; Kasisit, J.; Santisuk, T.; Tuchinda, P. Anti HIV-1 Flavonoid Glycosides from Ochna integerrima. Planta Med. 2007, 73, 683–688. [Google Scholar] [CrossRef]
- Rao, K.V.; Sreeramulu, K.; Venkata Rao, C.; Gunasekar, D.; Martin, M.T.; Bodo, B. Two New Biflavonoids from Ochna obtusata. J. Nat. Prod. 1997, 60, 632–634. [Google Scholar] [CrossRef]
- Jayaprakasam, B.; Damu, A.G.; Rao, K.V.; Gunasekar, D.; Blond, A.; Bodo, B. 7-O-Methyltetrahydroochnaflavone, a New Biflavanone from Ochna beddomei. J. Nat. Prod. 2000, 63, 507–508. [Google Scholar] [CrossRef]
- Ariyasena, J.; Baek, S.-H.; Perry, N.B.; Weavers, R.T. Ether-Linked Biflavonoids from Quintinia acutifolia. J. Nat. Prod. 2004, 67, 693–696. [Google Scholar] [CrossRef]
- Mbukwa, E.; Chacha, M.; Majinda, R.R.T. Phytochemical Constituents of Vangueria Infausta: Their Radical Scavenging and Antimicrobial Activities. Arkivoc 2006, 2007, 104–112. [Google Scholar] [CrossRef]
- Wild, S.H.; Roglic, G.; Green, A.; Sicree, R.; King, H. Global Prevalence of Diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care 2004, 27, 1047–1053. [Google Scholar] [CrossRef] [PubMed]
- Sievers, H.; Burkhardt, G.; Becker, H.; Zinsmeister, H.D. Hypnogenols and other dihydroflavonols from the moss Hypnum cupressiforme. Phytochemistry 1992, 31, 3233–3237. [Google Scholar] [CrossRef]
- Ali, D.M.; Wong, K.C.; Lim, P.K. Flavonoids from Blumea balsamifera. Fitoterapia 2005, 76, 128–130. [Google Scholar] [CrossRef]
- Sabudak, T.; Demirkiran, O.; Ozturk, M.; Topcu, G. Phenolic compounds from Trifolium echinatum Bieb. and investigation of their tyrosinase inhibitory and antioxidant activities. Phytochemistry 2013, 96, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Tartaglione, L.; Gambuti, A.; De Cicco, P.; Ercolano, G.; Ianaro, A.; Taglialatela-Scafati, O.; Moio, L.; Forino, M. NMR-based phytochemical analysis of Vitis vinifera cv Falanghina leaves. Characterization of a previously undescribed biflavonoid with antiproliferative activity. Fitoterapia 2018, 125, 13–17. [Google Scholar] [CrossRef]
- Carini, J.P.; Kaiser, S.; Ortega, G.G.; Bassani, V.L. Development, optimisation and validation of a stability-indicating HPLC method of achyrobichalcone quantification using experimental designs. Phytochem. Anal. 2013, 24, 193–200. [Google Scholar] [CrossRef]
- Kumar, N.; Singh, B.; Bhandari, P.; Gupta, A.P.; Uniyal, S.K.; Kaul, V.K. Biflavonoids from Lonicera japonica. Phytochemistry 2005, 66, 2740–2744. [Google Scholar] [CrossRef] [PubMed]
- Bitchagno, G.T.; Tankeo, S.B.; Tsopmo, A.; Simo Mpetga, J.D.; Tchinda, A.T.; Fobofou, S.A.; Nkuete, A.H.; Wessjohann, L.A.; Kuete, V.; Tane, P. Ericoside, a new antibacterial biflavonoid from Erica mannii (Ericaceae). Fitoterapia 2016, 109, 206–211. [Google Scholar] [CrossRef]
- Nakazawa, K. Syntheses of Ring-substituted Flavonoids and Allied Compounds. XI. Synthesis of Hinokiflavone. Chem. Pharm. Bull. 1968, 16, 2503–2511. [Google Scholar] [CrossRef]
- Gadek, P.A.; Quinn, C.J. Biflavones of the subfamily cupressoideae, cupressaceae. Phytochemistry 1985, 24, 267–272. [Google Scholar] [CrossRef]
- Geiger, H.; de Groot-Pfleiderer, W. Die biflavone von Taxodium distichum. Phytochemistry 1973, 12, 465–466. [Google Scholar] [CrossRef]
- Markham, K.R.; Sheppard, C.; Geiger, H. 13C NMR studies of some naturally occurring amentoflavone and hinokiflavone biflavonoids. Phytochemistry 1987, 26, 3335–3337. [Google Scholar] [CrossRef]
- Gadek, P.A. Biflavonoids from the seed testa of cycadales. Phytochemistry 1982, 21, 889–890. [Google Scholar] [CrossRef]
- Miura, H.; Kawano, N. The Partial Demethylation of Flavones. IV. Formation of New Bisflavones, Hinokiflavone-7, 7"-dimethyl Ether and Neocryptomerin. Chem. Pharm. Bull. 1968, 16, 1838–1840. [Google Scholar] [CrossRef][Green Version]
- Miura, H.; Kawano, N.; Anthony, C.W., Jr. Cryptomerin A and B, Hinokiflavone Methyl Ethers from the Leaves of Cryptomeria japonica. Chem. Pharm. Bull. 1966, 14, 1404–1408. [Google Scholar] [CrossRef]
- Meurer-Grimes, B.; Yu, J. Chamaecyparin—A Rare Biflavone from Selaginella Species. Z. Nat. C 1999, 54, 1143–1144. [Google Scholar] [CrossRef]
- Swamy, R.C.; Kunert, O.; Schühly, W.; Bucar, F.; Ferreira, D.; Rani, V.S.; Kumar, B.R.; Appa Rao, A.V.N. Structurally Unique Biflavonoids from Selaginella chrysocaulos and Selaginella bryopteris. Chem. Biodivers. 2006, 3, 405–414. [Google Scholar] [CrossRef]
- Silva, G.L.; Chai, H.; Gupta, M.P.; Farnsworth, N.R.; Cordell, G.A.; Pezzuto, J.M.; Beecher, C.W.W.; Douglas Kinghorn, A. Cytotoxic biflavonoids from Selaginella willdenowii. Phytochemistry 1995, 40, 129–134. [Google Scholar] [CrossRef]
- Sobha Rani, M.; Venkata Rao, C.; Gunasekar, D.; Blond, A.; Bodo, B. A biflavonoid from Cycas beddomei. Phytochemistry 1998, 47, 319–321. [Google Scholar] [CrossRef]
- Jayaprakasam, B.; Damu, A.G.; Gunasekar, D.; Blond, A.; Bodo, B. A biflavanone from Cycas beddomei. Phytochemistry 2000, 53, 515–517. [Google Scholar] [CrossRef]
- Akongwi, M.; Tih, A.E.; Nyongbela, K.D.; Samje, M.; Ghogomu, R.T.; Bodo, B. Brevipedicelones D and E, Two C-O-C Flavonoid Dimmers from the Leaves of Garcinia brevipedicellata and Anti-onchocercal Activity. Nat. Prod. Bioprospect. 2019, 9, 61–68. [Google Scholar] [CrossRef]
- Dora, G.; Edwards, J.M. Taxonomic Status of Lanaria lanata and Isolation of a Novel Biflavone. J. Nat. Prod. 1991, 54, 796–801. [Google Scholar] [CrossRef]
- Weniger, B.; Vonthron-Senecheau, C.; Arango, G.J.; Kaiser, M.; Brun, R.; Anton, R. A bioactive biflavonoid from Campnosperma panamense. Fitoterapia 2004, 75, 764–767. [Google Scholar] [CrossRef] [PubMed]
- Velandia, J.R.; Carvalho, M.G.D.; Braz-Filho, R.; Werle, A.A. Biflavonoids and a glucopyranoside derivative from Ouratea semiserrata. Phytochem. Anal. 2002, 13, 283–292. [Google Scholar] [CrossRef]
- Daniel, J.F.d.S.; Carvalho, M.G.d.; Cardoso, R.d.S.; Agra, M.d.F.; Eberlin, M.N. Others flavonoids from Ouratea hexasperma (Ochnaceae). J. Braz. Chem. Soc. 2005, 16, 634–638. [Google Scholar] [CrossRef][Green Version]
- Mahjoub, M.A.; Ammar, S.; Mighri, Z. A new biflavonoid and an isobiflavonoid from Rhus tripartitum. Nat. Prod. Res. 2005, 19, 723–729. [Google Scholar] [CrossRef]
- Li, Q.; Gao, W.; Cao, J.; Bi, X.; Chen, G.; Zhang, X.; Xia, X.; Zhao, Y. New cytotoxic compounds from flowers of Lawsonia inermis L. Fitoterapia 2014, 94, 148–154. [Google Scholar] [CrossRef]
- Parsons, I.C.; Gray, A.I.; Waterman, P.G.; Hartley, T.G. New Triterpenes and Flavonoids from the Leaves of Bosistoa brassii. J. Nat. Prod. 1993, 56, 46–53. [Google Scholar] [CrossRef]
- Hatano, T.; Miyatake, H.; Natsume, M.; Osakabe, N.; Takizawa, T.; Ito, H.; Yoshida, T. Proanthocyanidin glycosides and related polyphenols from cacao liquor and their antioxidant effects. Phytochemistry 2002, 59, 749–758. [Google Scholar] [CrossRef]
- Lee, D.F.; Swinny, E.E.; Jones, G.P. NMR identification of ethyl-linked anthocyanin–flavanol pigments formed in model wine ferments. Tetrahedron Lett. 2004, 45, 1671–1674. [Google Scholar] [CrossRef]
- Abe, Y.; Sawada, A.; Momose, T.; Sasaki, N.; Kawahara, N.; Kamakura, H.; Goda, Y.; Ozeki, Y. Structure of an anthocyanin–anthocyanin dimer molecule in anthocyanin-producing cells of a carrot suspension culture. Tetrahedron Lett. 2008, 49, 7330–7333. [Google Scholar] [CrossRef]
- Wang, Q.; Han, N.; Wu, X.; Tai, W.; Dai, N.; Wu, R.; Wu, J.; Bao, B. A biflavonoid glycoside from Lomatogonium carinthiacum (Wulf) Reichb. Nat. Prod. Res. 2015, 29, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Iinuma, M.; Tosa, H.; Tanaka, T.; Ito, T.; Asai, F. Chemical Constituents of Guttiferaeous Plants and Their Bioactivities. Symp. Chem. Nat. Prod. 1996, 38, 409–414. [Google Scholar]
- Bai, H.; Li, W.; Koike, K.; Dou, D.; Pei, Y.; Chen, Y.; Nikaido, T. A novel biflavonoid from roots of Glycyrrhiza uralensis cultivated in China. Chem. Pharm. Bull. 2003, 51, 1095–1097. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chen, R.J.; Cao, S.W.; Ruan, Z. Isolation of chemical constituents from Daphne odora var. Margirmt by high-speed counter-current chromatography. Chem. Nat. Compd. 2009, 45, 534–535. [Google Scholar] [CrossRef]
- Liang, S.; Tian, J.-M.; Feng, Y.; Liu, X.-H.; Xiong, Z.; Zhang, W.-D. Flavonoids from Daphne aurantiaca and Their Inhibitory Activities against Nitric Oxide Production. Chem. Pharm. Bull. 2011, 59, 653–656. [Google Scholar] [CrossRef]
- Taniguchi, M.; Fujiwara, A.; Baba, K. Three flavonoids from Daphne odora. Phytochemistry 1997, 45, 183–188. [Google Scholar] [CrossRef]
- Huang, W.-H.; Zhou, G.-X.; Wang, G.-C.; Chung, H.-Y.; Ye, W.-C.; Li, Y.-L. A new biflavonoid with antiviral activity from the roots of Wikstroemia indica. J. Asian Nat. Prod. Res. 2012, 14, 401–406. [Google Scholar] [CrossRef]
- Zheng, W.-F.; Shi, F. Three biflavonoids from ethanol extract of the root of Daphne genkwa. Acta Pharm. Sin. 2005, 40, 438–442. [Google Scholar]
- Zhou, G.-X.; Jiang, R.-W.; Cheng, Y.; Ye, W.-C.; Shi, J.-G.; Gong, N.-B.; Lu, Y. Daphnogirins A and B, Two Biflavones from Daphne giraldii. Chem. Pharm. Bull. 2007, 55, 1287–1290. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Shen, L.; Wang, K. A new biflavonoid from Daphniphyllum angustifolium Hutch. Fitoterapia 2009, 80, 461–464. [Google Scholar] [CrossRef] [PubMed]
- Tao, H.; Wang, L.; Cui, Z.; Zhao, D.; Liu, Y. Dimeric proanthocyanidins from the roots of Ephedra sinica. Planta Med. 2008, 74, 1823–1825. [Google Scholar] [CrossRef]
- Bilia, A.R.; Morelli, I.; Hamburger, M.; Hostetmann, K. Flavans and A-type proanthocyanidins from Prunus prostrata. Phytochemistry 1996, 43, 887–892. [Google Scholar] [CrossRef]
- Kolodziej, H.; Sakar, M.K.; Burger, J.F.W.; Engelshowe, R.; Ferreira, D. A-type proanthocyanidins from Prunus spinosa. Phytochemistry 1991, 30, 2041–2047. [Google Scholar] [CrossRef]
- Porter, L.J.; Ma, Z.; Chan, B.G. Cacao procyanidins: Major flavanoids and identification of some minor metabolites. Phytochemistry 1991, 30, 1657–1663. [Google Scholar] [CrossRef]
- Baldé, A.M.; Pieters, L.A.; Wray, V.; Kolodziej, H.; Berghe, D.A.V.; Claeys, M.; Vlietinck, A.J. Dimeric and trimeric proanthocyanidins possessing a doubly linked structure from Pavetta owariensis. Phytochemistry 1991, 30, 4129–4135. [Google Scholar] [CrossRef]
- Baldé, A.M.; Pieters, L.A.; Gergely, A.; Kolodziej, H.; Claeys, M.; Vlietinck, A.J. A-type Proanthocyanidins from stem-bark of Pavetta owariensis. Phytochemistry 1991, 30, 337–342. [Google Scholar] [CrossRef]
- Kamiya, K.; Watanabe, C.; Endang, H.; Umar, M.; Satake, T. Studies on the Constituents of Bark of Parameria laevigata MOLDENKE. Chem. Pharm. Bull. 2001, 49, 551–557. [Google Scholar] [CrossRef] [PubMed]
- Vivas, N.; Glories, Y.; Pianet, I.; Barbe, B.; Laguerre, M. A complete structural and conformational investigation of procyanidin A2 dimer. Tetrahedron Lett. 1996, 37, 2015–2018. [Google Scholar] [CrossRef]
- De Bruyne, T.; Pieters, L.; Witvrouw, M.; De Clercq, E.; Vanden Berghe, D.; Vlietinck, A.J. Biological Evaluation of Proanthocyanidin Dimers and Related Polyphenols. J. Nat. Prod. 1999, 62, 954–958. [Google Scholar] [CrossRef] [PubMed]
- Su, B.-N.; Hwang, B.Y.; Chai, H.; Carcache-Blanco, E.J.; Kardono, L.B.S.; Afriastini, J.J.; Riswan, S.; Wild, R.; Laing, N.; Farnsworth, N.R.; et al. Activity-Guided Fractionation of the Leaves of Ormosia sumatrana Using a Proteasome Inhibition Assay. J. Nat. Prod. 2004, 67, 1911–1914. [Google Scholar] [CrossRef] [PubMed]
- Jia, B.X.; Zeng, X.L.; Ren, F.X.; Jia, L.; Chen, X.Q.; Yang, J.; Liu, H.M.; Wang, Q. Baeckeins F-I, four novel C-methylated biflavonoids from the roots of Baeckea frutescens and their anti-inflammatory activities. Food Chem. 2014, 155, 31–37. [Google Scholar] [CrossRef]
- Tih, R.G.; Sondengam, B.L.; Martin, M.T.; Bodo, B. Structure of lophirones B and C, biflavonoids from the bark of Lophira lanceolata. Phytochemistry 1989, 28, 1557–1559. [Google Scholar] [CrossRef]
- Pegnyemb, D.E.; Tih, R.G.; Sondengam, B.L.; Blond, A.; Bodo, B. Biflavonoids from Ochna afzelii. Phytochemistry 2001, 57, 579–582. [Google Scholar] [CrossRef]
- Messanga, B.; Tih, R.G.; Sondengam, B.-L.; Martin, M.-T.; Bodo, B. Biflavonoids from Ochna calodendron. Phytochemistry 1994, 35, 791–794. [Google Scholar] [CrossRef]
- Tih, R.G.; Sondengam, B.L.; Martin, M.T.; Bodo, B. Structure of the chalcone dimers lophirone F, and H from Lophira lanceolata stem bark. Phytochemistry 1990, 29, 2289–2293. [Google Scholar] [CrossRef]
- Anuradha, V.; Srinivas, P.V.; Ranga Rao, R.; Manjulatha, K.; Purohit, M.G.; Madhusudana Rao, J. Isolation and synthesis of analgesic and anti-inflammatory compounds from Ochna squarrosa L. Bioorg. Med. Chem. 2006, 14, 6820–6826. [Google Scholar] [CrossRef] [PubMed]
- Lingfang, P.; Lihe, L.; Liguo, Y.; Xueping, L.; Tao, C.; Zhaoyun, Z. A new biflavone from Dysosma versipellis. Acta Pharm. Sin. 2016, 51, 1281–1284. [Google Scholar]
- Young, D.A.; Ferreira, D.; Roux, D.G. Synthesis of condensed tannins. Part 10. ‘Dioxane-linked’ profisetinidins. J. Chem. Soc. Perkin Trans. 1 1983, 14, 2031–2035. [Google Scholar] [CrossRef]
- Kaewamatawong, R.; Likhitwitayawuid, K.; Ruangrungsi, N.; Takayama, H.; Kitajima, M.; Aimi, N. Novel Biflavonoids from the Stem Bark of Ochna integerrima. J. Nat. Prod. 2002, 65, 1027–1029. [Google Scholar] [CrossRef] [PubMed]
- Messanga, B.B.; Tih, R.G.; Kimbu, S.F.; Sondengam, B.L.; Martin, M.T.; Bodo, B. Calodenone, a New Isobiflavonoid from Ochna calodendron. J. Nat. Prod. 1992, 55, 245–248. [Google Scholar] [CrossRef]
- Pegnyemb, D.E.; Tih, R.G.; Sondengam, B.L.; Blond, A.; Bodo, B. Isolation and Structure Elucidation of a New Isobiflavonoid From Ochna afzelii. Pharm. Biol. 2003, 41, 92–95. [Google Scholar] [CrossRef][Green Version]
- Geiger, H.; Markham, K.R. Campylopusaurone, an auronoflavanone biflavonoid from the mosses campylopus clavatus and campylopus holomitrium. Phytochemistry 1992, 31. [Google Scholar] [CrossRef]
- Messi, B.B.; Ndjoko-Ioset, K.; Hertlein-Amslinger, B.; Lannang, A.M.; Nkengfack, A.E.; Wolfender, J.L.; Hostettmann, K.; Bringmann, G. Preussianone, a new flavanone-chromone biflavonoid from Garcinia preussii Engl. Molecules 2012, 17, 6114–6125. [Google Scholar] [CrossRef] [PubMed]
- Jia, C.; Han, T.; Xu, J.; Li, S.; Sun, Y.; Li, D.; Li, Z.; Hua, H. A new biflavonoid and a new triterpene from the leaves of Garcinia paucinervis and their biological activities. J. Nat. Med. 2017, 71, 642–649. [Google Scholar] [CrossRef]
- Baba, K.; Takeuchi, K.; Tabata, Y.; Taniguchi, M.; Kozawa, M. Chemical studies on the constituents of the thymelaeaceous plants. IV. Structure of a new spiro biflavonoid, genkwanol A, from the root of Daphne genkwa Sieb. et Zucc. Yakugaku Zasshi 1987, 107, 525–529. [Google Scholar] [CrossRef]
- Yang, B.-H.; Zhang, W.-D.; Liu, R.-H.; Tan, C.-H.; Li, T.-Z.; Zhang, C.; Xu, X.-K.; Su, J. Spiro-biflavonoids from Larix olgensis Henry var. koreana Nakai. Helv. Chim. Acta 2005, 88, 2892–2896. [Google Scholar] [CrossRef]
- Shen, Z.; Falshaw, C.P.; Haslam, E.; Begley, M.J. A novel spiro-biflavonoid from Larix gmelini. J. Chem. Soc. Chem. Commun. 1985, 16, 1135–1137. [Google Scholar] [CrossRef]
- Andrade, A.W.L.; Machado, K.D.C.; Machado, K.D.C.; Figueiredo, D.D.R.; David, J.M.; Islam, M.T.; Uddin, S.J.; Shilpi, J.A.; Costa, J.P. In vitro antioxidant properties of the biflavonoid agathisflavone. Chem. Cent. J. 2018, 12, 75. [Google Scholar] [CrossRef] [PubMed]
- Lixian, W.; Yuanyuan, Y.; Meng, S.; Qi, W.; Changsheng, D.; Xin’an, H.; Jianping, S. Chemical Constituents from Garcinia kola Seeds and Their Anti-Oxidant Activity. Tradit. Chin. Drug Res. Clin. Pharmacol. 2020, 31, 1133–1140. [Google Scholar]
- Jeong, E.J.; Hwang, L.; Lee, M.; Lee, K.Y.; Ahn, M.J.; Sung, S.H. Neuroprotective biflavonoids of Chamaecyparis obtusa leaves against glutamate-induced oxidative stress in HT22 hippocampal cells. Food Chem. Toxicol. 2014, 64, 397–402. [Google Scholar] [CrossRef]
- Park, H.; Kim, Y.H.; Chang, H.W.; Kim, H.P. Anti-inflammatory activity of the synthetic C-C biflavonoids. J. Pharm. Pharm. 2006, 58, 1661–1667. [Google Scholar] [CrossRef]
- Banerjee, T.; Valacchi, G.; Ziboh, V.A.; van der Vliet, A. Inhibition of TNFalpha-induced cyclooxygenase-2 expression by amentoflavone through suppression of NF-kappaB activation in A549 cells. Mol. Cell Biochem. 2002, 238, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Ye, T.; Long, T.; Peng, X. Ginkgetin exerts anti-inflammatory effects on cerebral ischemia/reperfusion-induced injury in a rat model via the TLR4/NF-kappaB signaling pathway. Biosci. Biotechnol. Biochem. 2019, 83, 675–683. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.P.; Park, H.; Son, K.H.; Chang, H.W.; Kang, S.S. Biochemical pharmacology of biflavonoids: Implications for anti-inflammatory action. Arch. Pharm. Res. 2008, 31, 265–273. [Google Scholar] [CrossRef]
- Coulerie, P.; Eydoux, C.; Hnawia, E.; Stuhl, L.; Maciuk, A.; Lebouvier, N.; Canard, B.; Figadere, B.; Guillemot, J.C.; Nour, M. Biflavonoids of Dacrydium balansae with potent inhibitory activity on dengue 2 NS5 polymerase. Planta Med. 2012, 78, 672–677. [Google Scholar] [CrossRef]
- Li, F.; Song, X.; Su, G.; Wang, Y.; Wang, Z.; Jia, J.; Qing, S.; Huang, L.; Wang, Y.; Zheng, K.; et al. Amentoflavone Inhibits HSV-1 and ACV-Resistant Strain Infection by Suppressing Viral Early Infection. Viruses 2019, 11, 466. [Google Scholar] [CrossRef]
- De Freitas, C.S.; Rocha, M.E.N.; Sacramento, C.Q.; Marttorelli, A.; Ferreira, A.C.; Rocha, N.; de Oliveira, A.C.; de Oliveira Gomes, A.M.; Dos Santos, P.S.; da Silva, E.O.; et al. Agathisflavone, a Biflavonoid from Anacardium occidentale L. Inhibits Influenza Virus Neuraminidase. Curr. Top. Med. Chem. 2020, 20, 111–120. [Google Scholar] [CrossRef]
- Tang, S.; Bremner, P.; Kortenkamp, A.; Schlage, C.; Gray, A.I.; Gibbons, S.; Heinrich, M. Biflavonoids with cytotoxic and antibacterial activity from Ochna macrocalyx. Planta Med. 2003, 69, 247–253. [Google Scholar] [CrossRef]
- Nandu, T.G.; Subramenium, G.A.; Shiburaj, S.; Viszwapriya, D.; Iyer, P.M.; Balamurugan, K.; Rameshkumar, K.B.; Karutha Pandian, S. Fukugiside, a biflavonoid from Garcinia travancorica inhibits biofilm formation of Streptococcus pyogenes and its associated virulence factors. J. Med. Microbiol. 2018, 67, 1391–1401. [Google Scholar] [CrossRef]
- Lee, J.; Choi, Y.; Woo, E.R.; Lee, D.G. Isocryptomerin, a novel membrane-active antifungal compound from Selaginella tamariscina. Biochem. Biophys. Res. Commun. 2009, 379, 676–680. [Google Scholar] [CrossRef]
- Lee, J.H. Involvement of T-cell immunoregulation by ochnaflavone in therapeutic effect on fungal arthritis due to Candida albicans. Arch. Pharm. Res. 2011, 34, 1209–1217. [Google Scholar] [CrossRef] [PubMed]
- Ramalingam, S.; Karuppiah, M.; Thiruppathi, M.; Palanivelu, S.; Panchanatham, S. Antioxidant potential of biflavonoid attenuates hyperglycemia by modulating the carbohydrate metabolic enzymes in high fat diet/streptozotocin induced diabetic rats. Redox Rep. 2020, 25, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.K.; Weng, Z.M.; Ge, G.B.; Li, H.L.; Ding, L.L.; Dai, Z.R.; Hou, X.D.; Leng, Y.H.; Yu, Y.; Hou, J. Biflavones from Ginkgo biloba as novel pancreatic lipase inhibitors: Inhibition potentials and mechanism. Int. J. Biol. Macromol. B 2018, 118, 2216–2223. [Google Scholar] [CrossRef] [PubMed]
- Xianming, W.; Aiqiong, L.; Lili, Z.; Jian, L. Study on wound healing mechanism of the foot ulcer in diabetic rats by isoginkgetin. J. Xiangnan Univ. (Med. Sci.) 2019, 21, 6–10. [Google Scholar]
- Zhou, Q.; Han, X.; Li, R.; Zhao, W.; Bai, B.; Yan, C.; Dong, X. Anti-atherosclerosis of oligomeric proanthocyanidins from Rhodiola rosea on rat model via hypolipemic, antioxidant, anti-inflammatory activities together with regulation of endothelial function. Phytomedicine 2018, 51, 171–180. [Google Scholar] [CrossRef]
- Tabares-Guevara, J.H.; Lara-Guzman, O.J.; Londono-Londono, J.A.; Sierra, J.A.; Leon-Varela, Y.M.; Alvarez-Quintero, R.M.; Osorio, E.J.; Ramirez-Pineda, J.R. Natural Biflavonoids Modulate Macrophage-Oxidized LDL Interaction In Vitro and Promote Atheroprotection In Vivo. Front. Immunol. 2017, 8, 923. [Google Scholar] [CrossRef]
- Uddin, M.S.; Kabir, M.T.; Tewari, D.; Mathew, B.; Aleya, L. Emerging signal regulating potential of small molecule biflavonoids to combat neuropathological insults of Alzheimer’s disease. Sci. Total Environ. 2020, 700, 134836. [Google Scholar] [CrossRef]
- Thapa, A.; Chi, E.Y. Biflavonoids as Potential Small Molecule Therapeutics for Alzheimer’s Disease. Adv. Exp. Med. Biol. 2015, 863, 55–77. [Google Scholar]
- Thapa, A.; Woo, E.R.; Chi, E.Y.; Sharoar, M.G.; Jin, H.G.; Shin, S.Y.; Park, I.S. Biflavonoids are superior to monoflavonoids in inhibiting amyloid-beta toxicity and fibrillogenesis via accumulation of nontoxic oligomer-like structures. Biochemistry 2011, 50, 2445–2455. [Google Scholar] [CrossRef] [PubMed]
- Sirimangkalakitti, N.; Juliawaty, L.D.; Hakim, E.H.; Waliana, I.; Saito, N.; Koyama, K.; Kinoshita, K. Naturally occurring biflavonoids with amyloid β aggregation inhibitory activity for development of anti-Alzheimer agents. Bioorg. Med. Chem. Lett. 2019, 29, 1994–1997. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.Y.; Kang, S.S.; Lee, S.K.; Han, B.H. Polyphenolic Biflavonoids Inhibit Amyloid-Beta Fibrillation and Disaggregate Preformed Amyloid-Beta Fibrils. Biomol. Ther. 2020, 28, 145–151. [Google Scholar] [CrossRef]
- Olajide, O.J.; Ugbosanmi, A.T.; Enaibe, B.U.; Ogunrinola, K.Y.; Lewu, S.F.; Asogwa, N.T.; Akapa, T.; Imam, A.; Ibrahim, A.; Gbadamosi, I.T.; et al. Cerebellar Molecular and Cellular Characterization in Rat Models of Alzheimer’s Disease: Neuroprotective Mechanisms of Garcinia Biflavonoid Complex. Ann. Neurosci. 2017, 24, 32–45. [Google Scholar] [CrossRef][Green Version]
- Cao, Q.; Qin, L.; Huang, F.; Wang, X.; Yang, L.; Shi, H.; Wu, H.; Zhang, B.; Chen, Z.; Wu, X. Amentoflavone protects dopaminergic neurons in MPTP-induced Parkinson’s disease model mice through PI3K/Akt and ERK signaling pathways. Toxicol. Appl. Pharm. 2017, 319, 80–90. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.Q.; Wang, M.Y.; Fu, X.R.; Peng, Y.; Gao, G.F.; Fan, Y.M.; Duan, X.L.; Zhao, B.L.; Chang, Y.Z.; Shi, Z.H. Neuroprotective effects of ginkgetin against neuroinjury in Parkinson’s disease model induced by MPTP via chelating iron. Free Radic. Res. 2015, 49, 1069–1080. [Google Scholar] [CrossRef]
- Galati, G.; O’Brien, P.J. Potential toxicity of flavonoids and other dietary phenolics: Significance for their chemopreventive and anticancer properties. Free Radic. Biol. Med. 2004, 37, 287–303. [Google Scholar] [CrossRef]
- Li, Y.Y.; Lu, X.Y.; Sun, J.L.; Wang, Q.Q.; Zhang, Y.D.; Zhang, J.B.; Fan, X.H. Potential hepatic and renal toxicity induced by the biflavonoids from Ginkgo biloba. Chin. J. Nat. Med. 2019, 17, 672–681. [Google Scholar] [CrossRef]
- Lin, Y.-M.; Chen, F.-C.; Lee, K.-H. Hinokiflavone, a Cytotoxic Principle from Rhus succedanea and the Cytotoxicity of the Related Biflavonoids. Planta Med. 1989, 55, 166–168. [Google Scholar] [CrossRef]
- Lopes Andrade, A.W.; Dias Ribeiro Figueiredo, D.; Torequl Islam, M.; Viana Nunes, A.M.; da Conceicao Machado, K.; da Conceicao Machado, K.; Uddin, S.J.; Ahmed Shilpi, J.; Rouf, R.; de Carvalho Melo-Cavalcante, A.A.; et al. Toxicological evaluation of the biflavonoid, agathisflavone in albino Swiss mice. Biomed. Pharmacother. 2019, 110, 68–73. [Google Scholar] [CrossRef]
- Yao, W.; Lin, Z.; Shi, P.; Chen, B.; Wang, G.; Huang, J.; Sui, Y.; Liu, Q.; Li, S.; Lin, X.; et al. Delicaflavone induces ROS-mediated apoptosis and inhibits PI3K/AKT/mTOR and Ras/MEK/Erk signaling pathways in colorectal cancer cells. Biochem. Pharmacol. 2020, 171, 113680. [Google Scholar] [CrossRef]
- Yao, W.; Lin, Z.; Wang, G.; Li, S.; Chen, B.; Sui, Y.; Huang, J.; Liu, Q.; Shi, P.; Lin, X.; et al. Delicaflavone induces apoptosis via mitochondrial pathway accompanying G2/M cycle arrest and inhibition of MAPK signaling cascades in cervical cancer HeLa cells. Phytomedicine 2019, 62, 152973. [Google Scholar] [CrossRef]
- Mu, W.; Cheng, X.; Zhang, X.; Liu, Y.; Lv, Q.; Liu, G.; Zhang, J.; Li, X. Hinokiflavone induces apoptosis via activating mitochondrial ROS/JNK/caspase pathway and inhibiting NF-kappaB activity in hepatocellular carcinoma. J. Cell Mol. Med. 2020, 24, 8151–8165. [Google Scholar] [CrossRef]
- Wada, S.; Hitomi, T.; Tokuda, H.; Tanaka, R. Anti-tumor-initiating effects of spiro-biflavonoids from Abies sachalinensis. Chem. Biodivers. 2010, 7, 2303–2308. [Google Scholar] [CrossRef]
- Li, P.; Yue, G.G.; Kwok, H.F.; Long, C.L.; Lau, C.B.; Kennelly, E.J. Using Ultra-Performance Liquid Chromatography Quadrupole Time of Flight Mass Spectrometry-Based Chemometrics for the Identification of Anti-angiogenic Biflavonoids from Edible Garcinia Species. J. Agric. Food Chem. 2017, 65, 8348–8355. [Google Scholar] [CrossRef] [PubMed]
- Tarallo, V.; Lepore, L.; Marcellini, M.; Dal Piaz, F.; Tudisco, L.; Ponticelli, S.; Lund, F.W.; Roepstorff, P.; Orlandi, A.; Pisano, C.; et al. The biflavonoid amentoflavone inhibits neovascularization preventing the activity of proangiogenic vascular endothelial growth factors. J. Biol. Chem. 2011, 286, 19641–19651. [Google Scholar] [CrossRef] [PubMed]
- Cabrini, D.A.; Patino, A.C.; Nunez, V.; Osorio, E. The biflavonoid morelloflavone inhibits the enzymatic and biological activities of a snake venom phospholipase A2. Chem. Biol. Interact. 2014, 220, 94–101. [Google Scholar]
- Antia, B.S.; Pansanit, A.; Ekpa, O.D.; Ekpe, U.J.; Mahidol, C.; Kittakoop, P. Alpha-glucosidase inhibitory, aromatase inhibitory, and antiplasmodial activities of a biflavonoid GB1 from Garcinia kola stem bark. Planta Med. 2010, 76, 276–277. [Google Scholar] [CrossRef]
- Wu, X.-N.; Yang, Y.; Zhang, H.-H.; Zhong, Y.-S.; Wu, F.; Yu, B.; Yu, C.-H. Robustaflavone-4′-dimethyl ether from Selaginella uncinata attenuated lipopolysaccharide-induced acute lung injury via inhibiting FLT3-mediated neutrophil activation. Int. Immunopharmacol. 2020, 82, 106338–106342. [Google Scholar] [CrossRef] [PubMed]
- Jalil, J.; Jantan, I.; Ghani, A.A.; Murad, S. Platelet-activating factor (PAF) antagonistic activity of a new biflavonoid from Garcinia nervosa var. pubescens King. Molecules 2012, 17, 10893–10901. [Google Scholar] [CrossRef]
- Qu, X.; Li, Q.; Zhang, X.; Wang, Z.; Wang, S.; Zhou, Z. Amentoflavone protects the hematopoietic system of mice against γ-irradiation %J Pharmaceutical Society of Korea. Arch. Pharm. Res. 2019, 42, 1021–1029. [Google Scholar] [CrossRef]
- Yamaguchi, L.F.; Kato, M.J.; Di Mascio, P. Biflavonoids from Araucaria angustifolia protect against DNA UV-induced damage. Phytochemistry 2009, 70, 615–620. [Google Scholar] [CrossRef]
- Campos, P.M.; Prudente, A.S.; Horinouchi, C.D.; Cechinel-Filho, V.; Favero, G.M.; Cabrini, D.A.; Otuki, M.F. Inhibitory effect of GB-2a (I3-naringenin-II8-eriodictyol) on melanogenesis. J. Ethnopharmacol. 2015, 174, 224–229. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, K.; Matlin, A.J.; Lowell, A.M.; Moore, M.J. The biflavonoid isoginkgetin is a general inhibitor of Pre-mRNA splicing. J. Biol. Chem. 2008, 283, 33147–33154. [Google Scholar] [CrossRef] [PubMed]
- Kwak, W.J.; Han, C.K.; Son, K.H.; Chang, H.W.; Kang, S.S.; Park, B.K.; Kim, H.P. Effects of Ginkgetin from Ginkgo biloba Leaves on cyclooxygenases and in vivo skin inflammation. Planta Med. 2002, 68, 316–321. [Google Scholar] [CrossRef]
- Lim, H.; Son, K.H.; Chang, H.W.; Kang, S.S.; Kim, H.P. Effects of anti-inflammatory biflavonoid, ginkgetin, on chronic skin inflammation. Biol. Pharm. Bull. 2006, 29, 1046–1049. [Google Scholar] [CrossRef]
- Kim, T.Y.; Park, N.J.; Jegal, J.; Choi, S.; Lee, S.W.; Hang, J.; Kim, S.N.; Yang, M.H. Chamaejasmine Isolated from Wikstroemia dolichantha Diels Suppresses 2,4-Dinitrofluoro-benzene-Induced Atopic Dermatitis in SKH-1 Hairless Mice. Biomolecules 2019, 9, 697. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.; Ren, Q.; Yang, C.; Zhang, T.; Li, J.; Wang, X.; Qu, X.; Zhang, X.; Zhou, Z.; Zhang, Z.; et al. Liquid chromatography-tandem mass spectrometry determination and pharmacokinetic analysis of amentoflavone and its conjugated metabolites in rats. J. Agric. Food Chem. 2015, 63, 1957–1966. [Google Scholar] [CrossRef] [PubMed]
- Yin, R.; Xiong, K.; Wen, S.; Wang, Y.; Xu, F. Development and validation of an LC-MS/MS method for the determination of hinokiflavone in rat plasma and its application to a pharmacokinetic study. Biomed. Chromatogr. 2017, 31, 3821–3840. [Google Scholar] [CrossRef] [PubMed]
- Shan, C.-X.; Guo, S.-C.; Yu, S.; Shan, M.-Q.; Li, S.F.Y.; Chai, C.; Cui, X.-B.; Zhang, L.; Ding, A.-W.; Wu, Q.-N. Simultaneous Determination of Quercitrin, Afzelin, Amentoflavone, Hinokiflavone in Rat Plasma by UFLC-MS-MS and Its Application to the Pharmacokinetics of Platycladus orientalis Leaves Extract. J. Chromatogr. Sci. 2018, 56, 895–902. [Google Scholar] [CrossRef] [PubMed]
- Alzand, K.I.; Mohamed, M.A. Flavonoids: Chemistry, Biochemistry and Antioxidant activity. J. Pharm. Res. 2012, 5, 4013–4020. [Google Scholar]
- Gomes-Copelanda, K.K.P.; Lédob, A.d.S.; Almeidac, F.T.C.d.; Moreirad, B.O.; Santosd, D.C.d.; Santosd, R.A.F.; Jorge Mauricio Davidd, J.P.D. Effect of elicitors in Poincianella pyramidalis callus culture in the biflavonoid biosynthesis. Ind. Crop. Prod. 2018, 126, 421–425. [Google Scholar] [CrossRef]
- Ying, X.; Ling-bo, Q.; Jin-wei, Y. Research Progress on the Extraction and Synthesis of Biflavonoid Compounds. J. Henan Univ. Technol. (Nat. Sci. Ed.) 2010, 31, 78–85. [Google Scholar]
- Ndoile, M.M.; van Heerden, F.R. Total synthesis of ochnaflavone. Beilstein J. Org. Chem. 2013, 9, 1346–1351. [Google Scholar] [CrossRef]
- Zhang, Y.; Lin, S.; Shi, A.; Yang, Y.; Tang, W. The synthetic research of (±)-2,3,2”,3”-Tetrahydroochnaflavone. Chin. J. Org. Chem. 2015, 35, 2114–2118. [Google Scholar] [CrossRef]
- Chen, J.; Chang, H.W.; Kim, H.P.; Park, H. Synthesis of phospholipase A2 inhibitory biflavonoids. Bioorganic Med. Chem. Lett. 2006, 16, 2373–2375. [Google Scholar] [CrossRef]
- Moon, T.C.; Quan, Z.; Kim, J.; Kim, H.P.; Kudo, I.; Murakami, M.; Park, H.; Chang, H.W. Inhibitory effect of synthetic C-C biflavones on various phospholipase A(2)s activity. Bioorg Med. Chem. 2007, 15, 7138–7143. [Google Scholar] [CrossRef]
- Lim, H.; Kim, S.B.; Park, H.; Chang, H.W.; Kim, H.P. New anti-inflammatory synthetic biflavonoid with C-C (6-6”) linkage: Differential effects on cyclooxygenase-2 and inducible nitric oxide synthase. Arch. Pharm. Res. 2009, 32, 1525–1531. [Google Scholar] [CrossRef] [PubMed]
- Yunchang, T. Synthesis of I3’,II8-apigenin Biflavone and Inhibitory Activity Evaluation as α-Glucosidase Inhibitors. Master’s Thesis, Tianjin University of Science and Technology, Tianjin, China, 2018. [Google Scholar]
- Ming, L. Synthesis of Wikstrol A/B and Morelloflavone. Master’s, Thesis, Tianjin University of Science and Technology, Tianjin, China, 2018. [Google Scholar]
- Zhang, Z.T.; Gao, R.L.; Zhuang, S.K. Synthesis of biflavones and their interaction with DNA. Acta Pharm. Sin. 2009, 44, 873–878. [Google Scholar]
- Ying, X.; Jinwei, Y.; Yongmei, X.; Pu, M.; Gonggong, H. The Process of Biflavonoids’s Synthesis by Acid Catalysis. In Proceedings of the Academic Annual Meeting of Henan Chemical Society and Celebration of its 70th Anniversary, Nanyang, China, 24 September 2010; p. 1. [Google Scholar]
- Ying, X.; Jin-wei, Y.; Ling-bo, Q. Synthesis and Reaction Mechanism of Biflavonoids. In Proceedings of the The 12th National Annual Meeting of Applied Chemistry of the Chinese Chemical Society, Zhengzhou, China, 17 October 2011; p. 2. [Google Scholar]
- Baron, V.; Mead, K.T. Synthesis of 3-benzylidene-dihydrofurochromen-2-ones: Promising intermediates for biflavonoid synthesis. Heterocycl. Commun. 2015, 21, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Ren, Q.X.; Zhou, Z.; Wang, S.Q. Preparation and analytical characterization of micronized amentoflavone by antisolvent freeze-drying method. Int. J. Pharm. Res. 2013, 40, 237–241. [Google Scholar]
- DeKosky, S.T.; Williamson, J.D.; Fitzpatrick, A.L.; Kronmal, R.A.; Ives, D.G.; Saxton, J.A.; Lopez, O.L.; Burke, G.; Carlson, M.C.; Fried, L.P.; et al. Ginkgo biloba for prevention of dementia: A randomized controlled trial. JAMA 2008, 300, 2253–2262. [Google Scholar] [CrossRef] [PubMed]
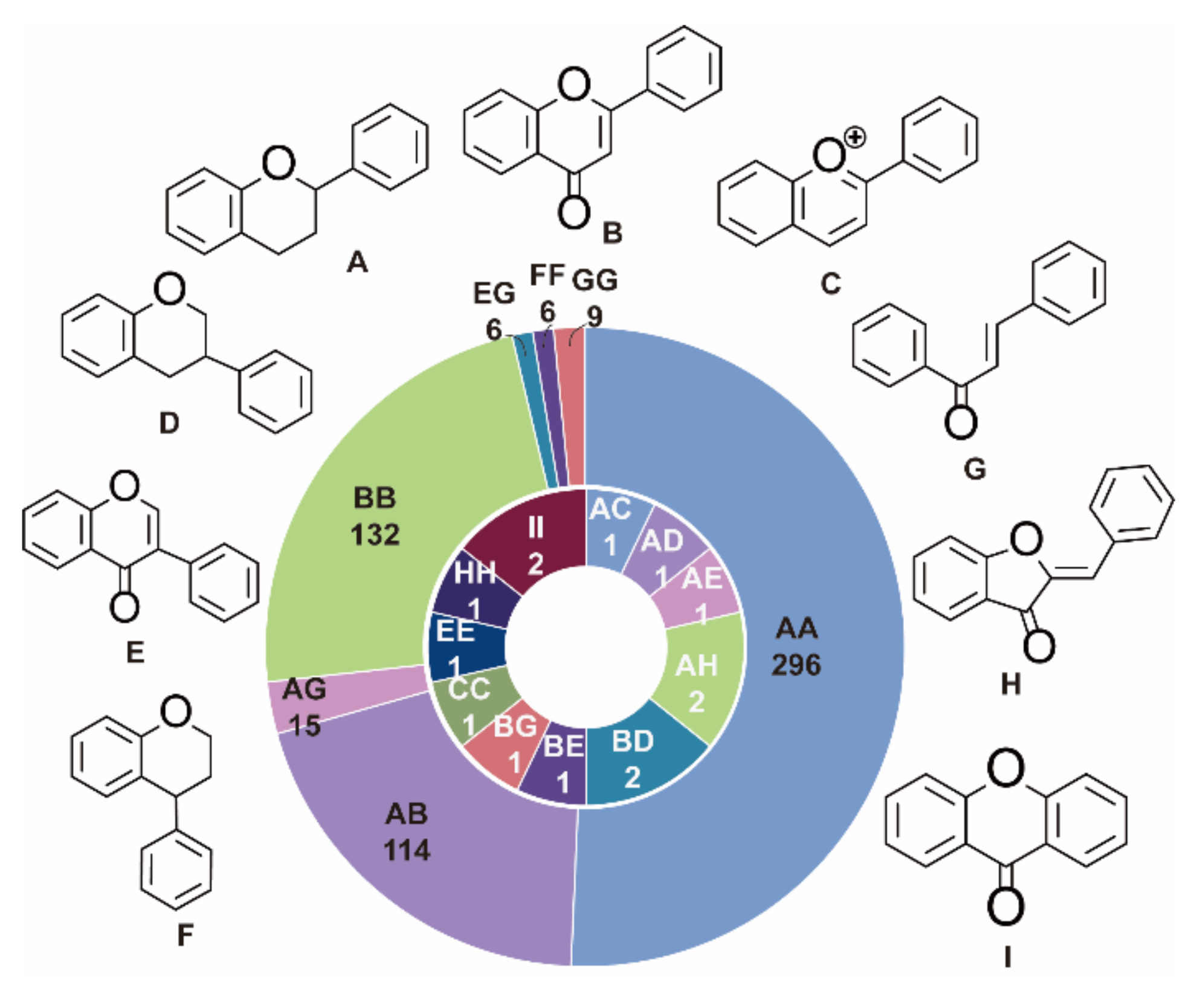
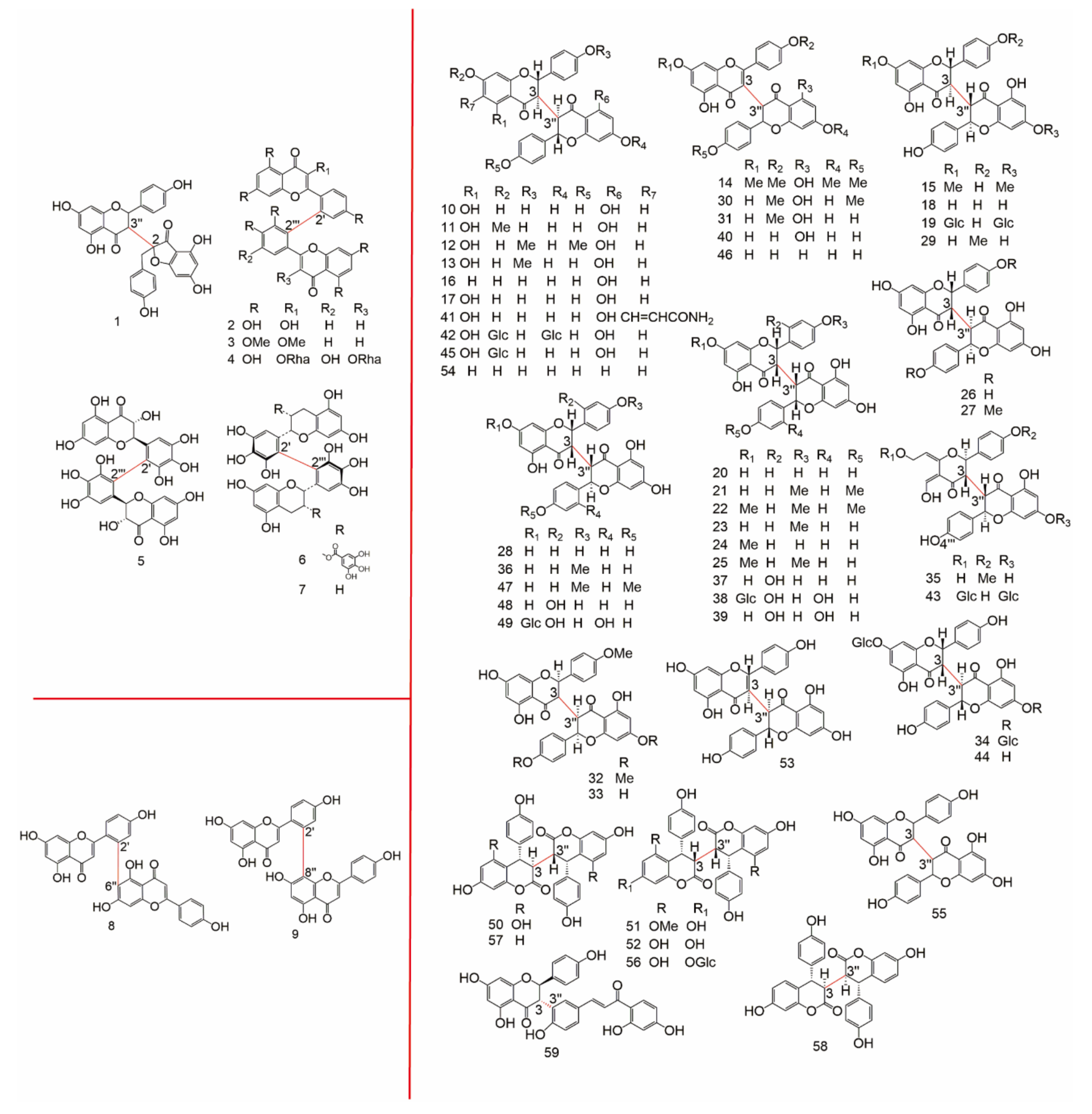
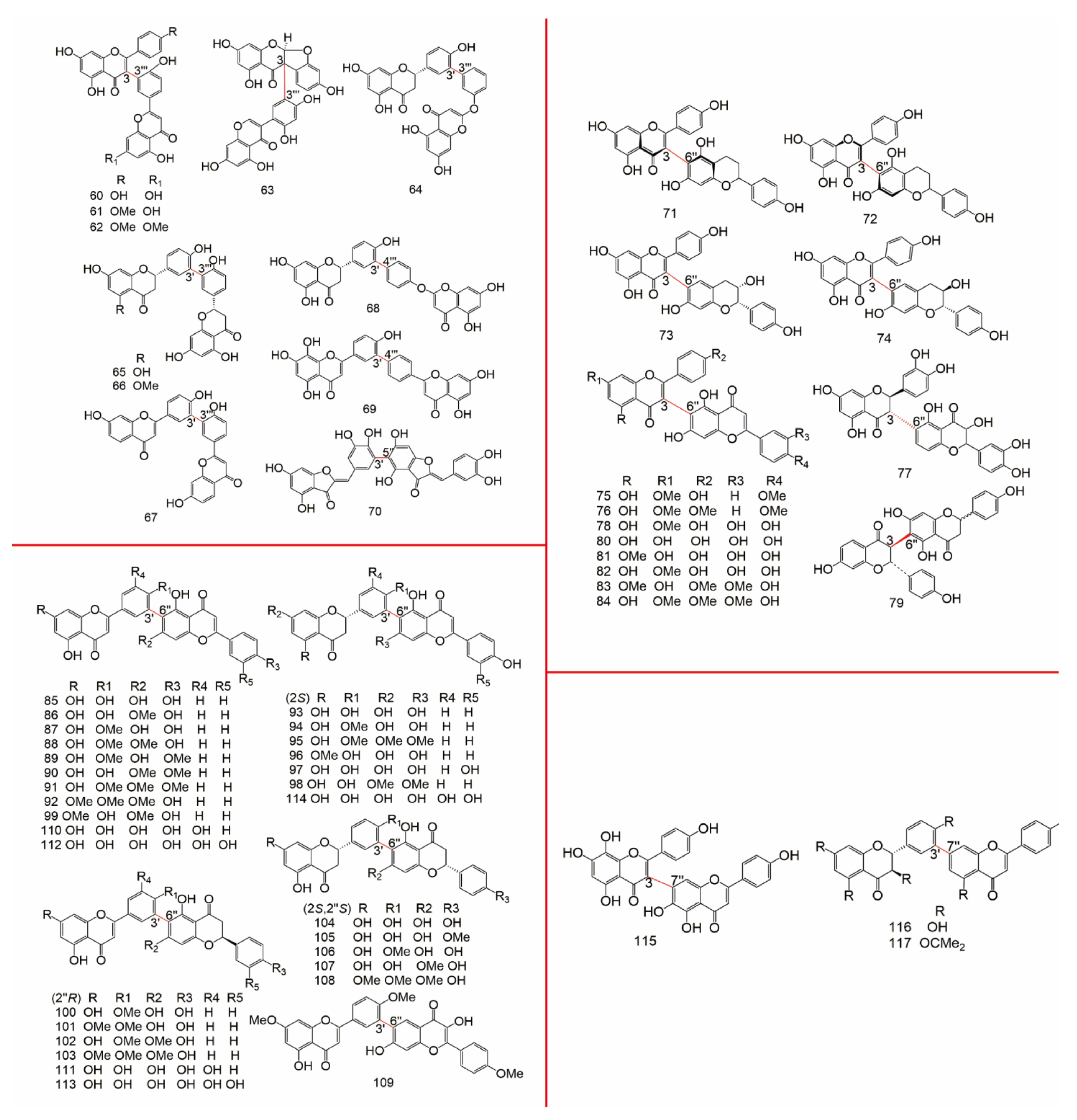
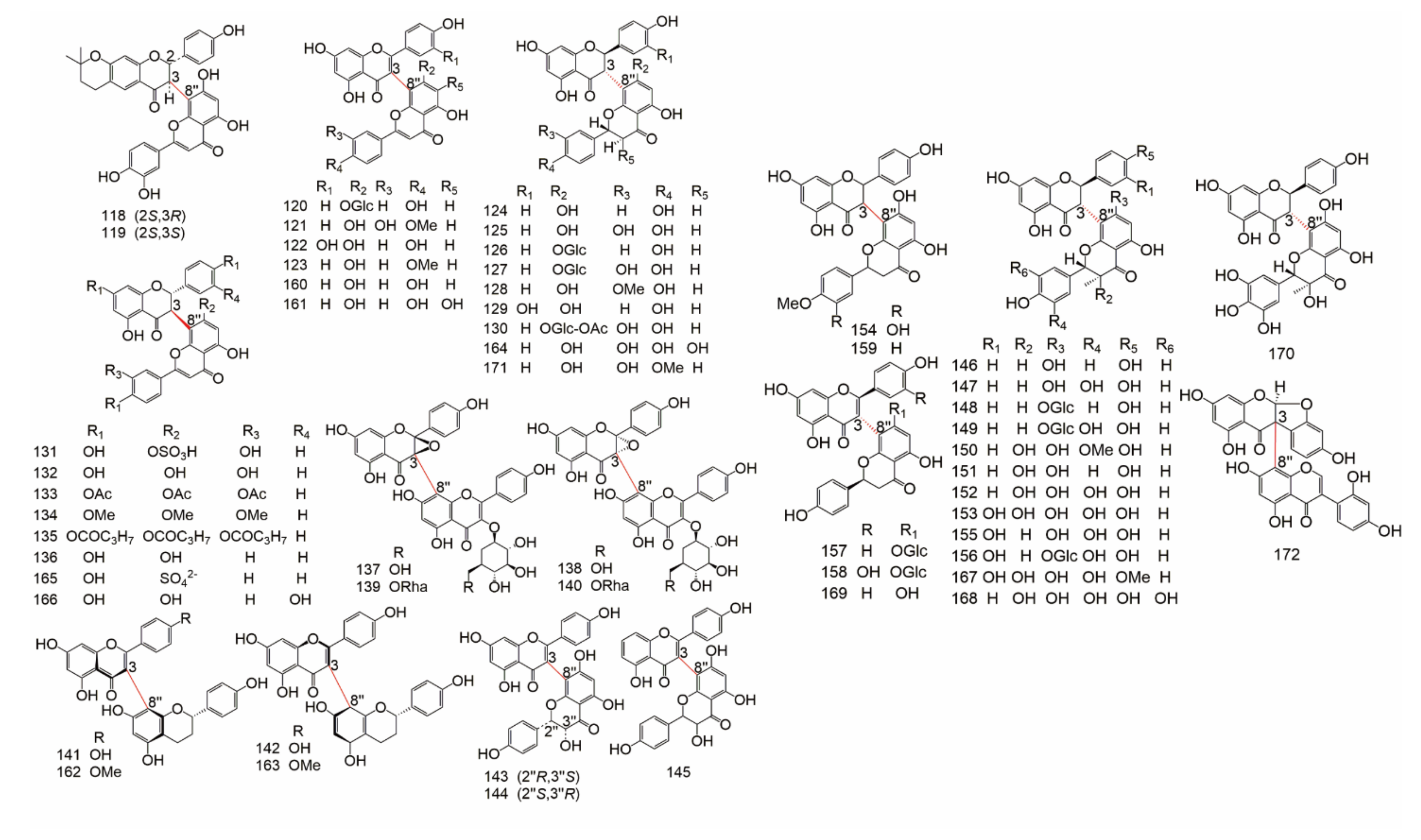
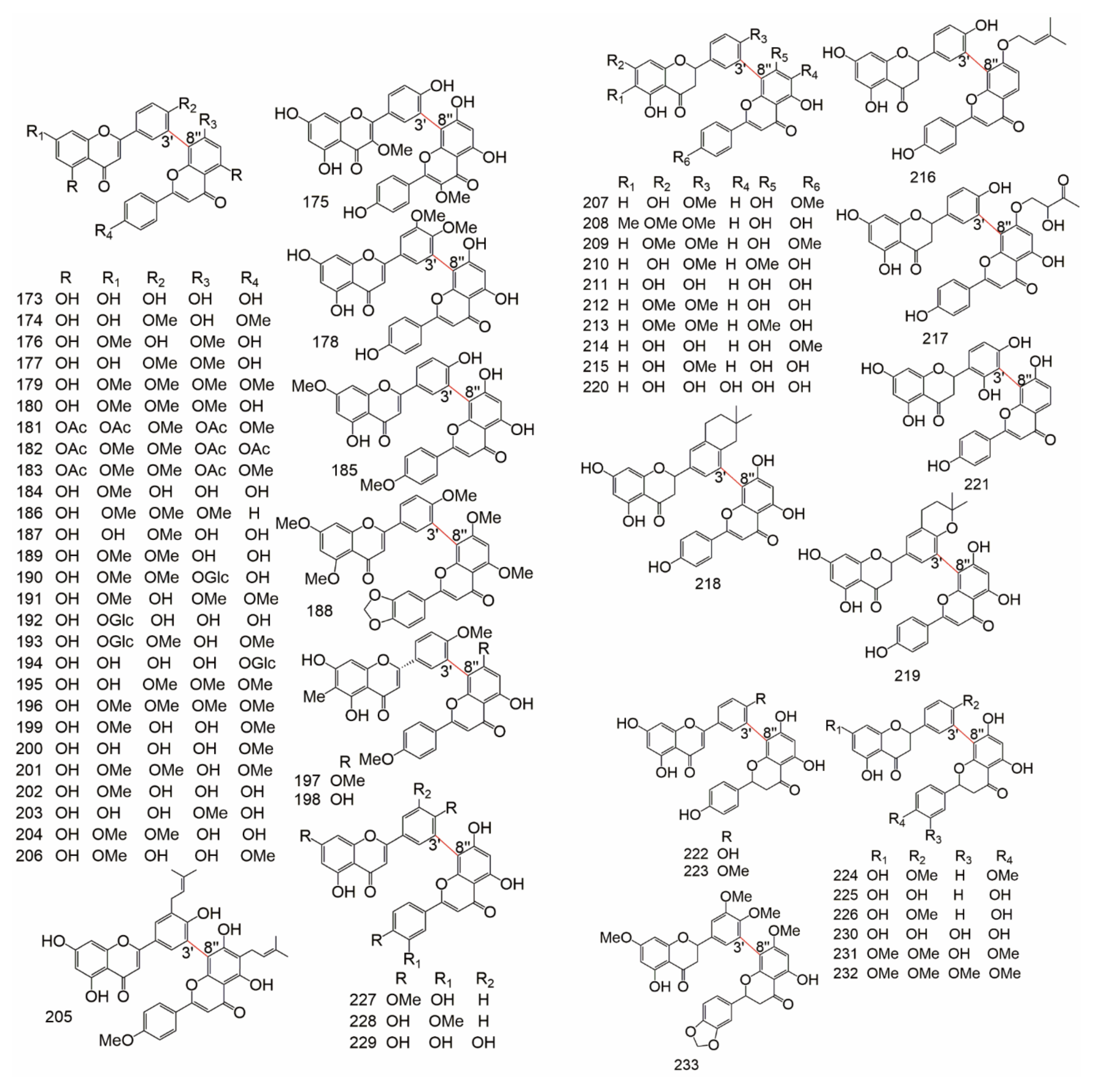
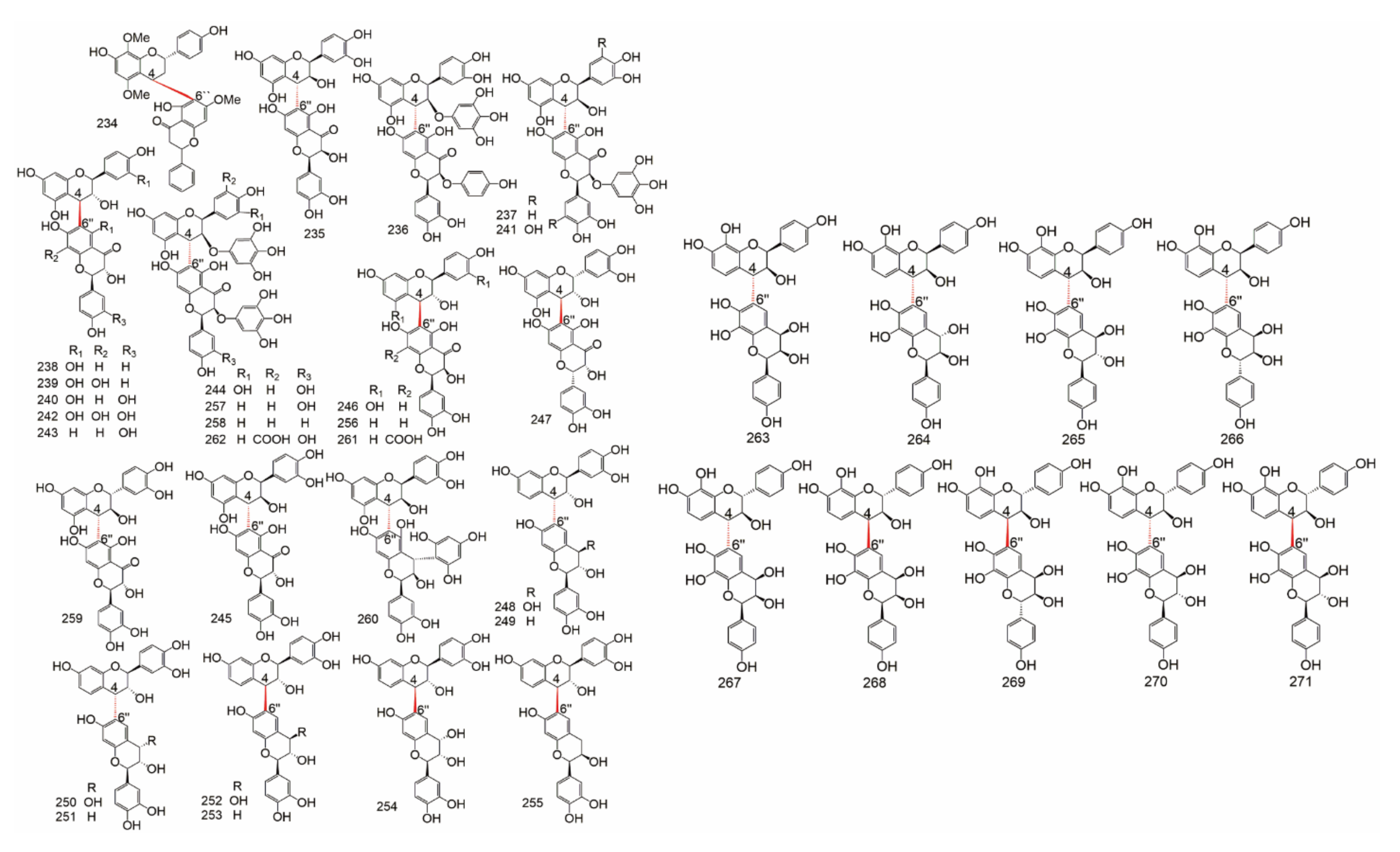
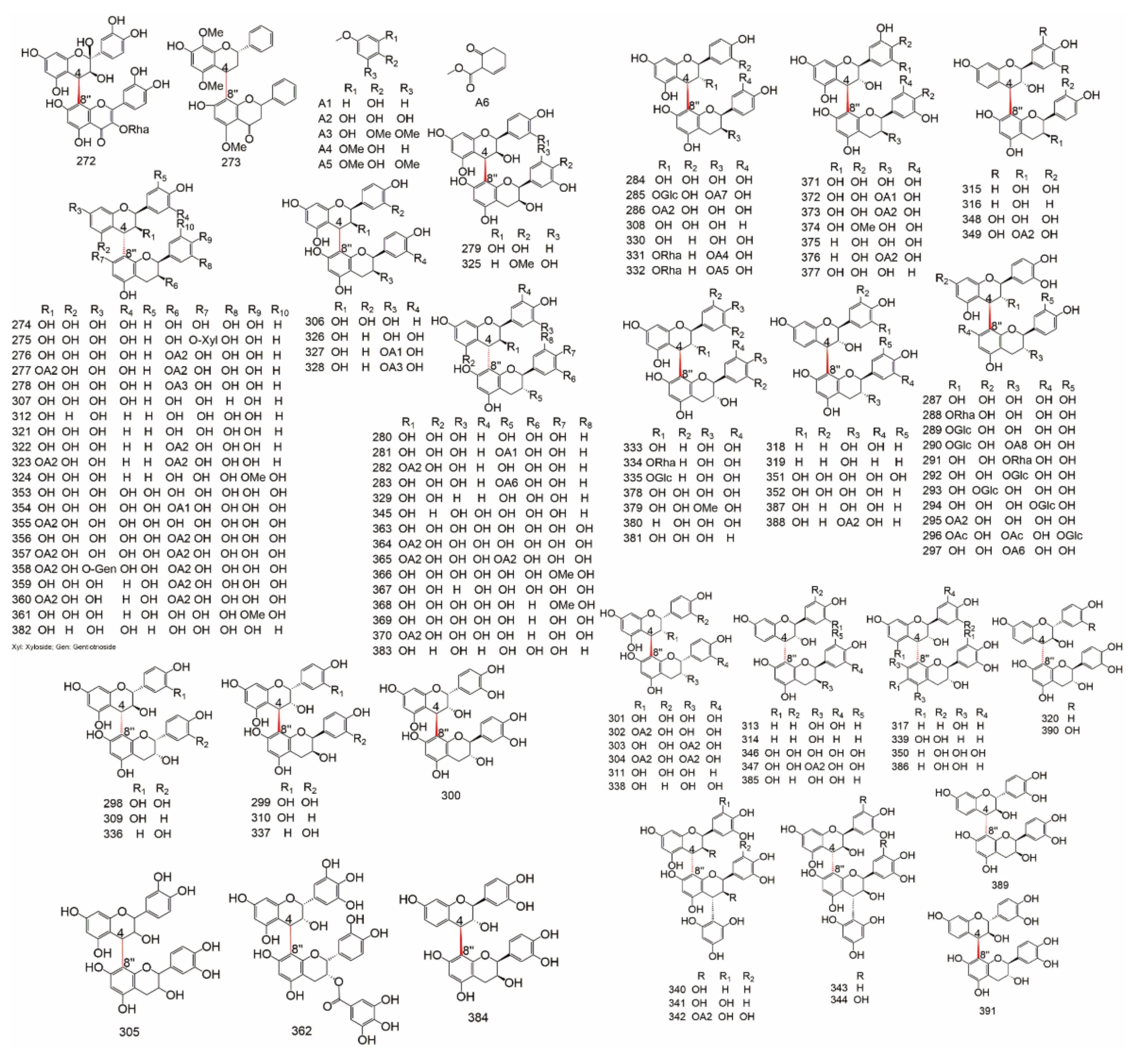


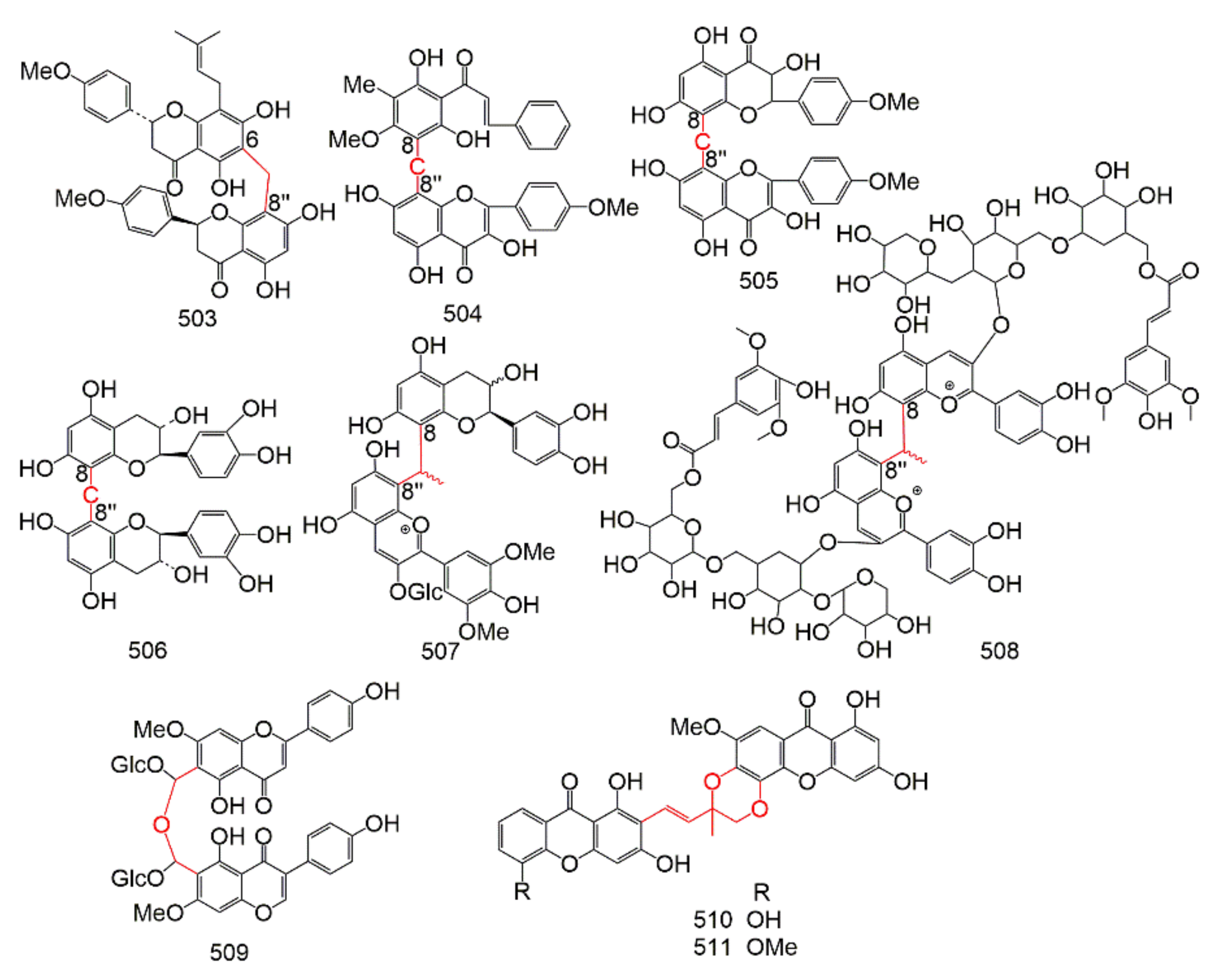

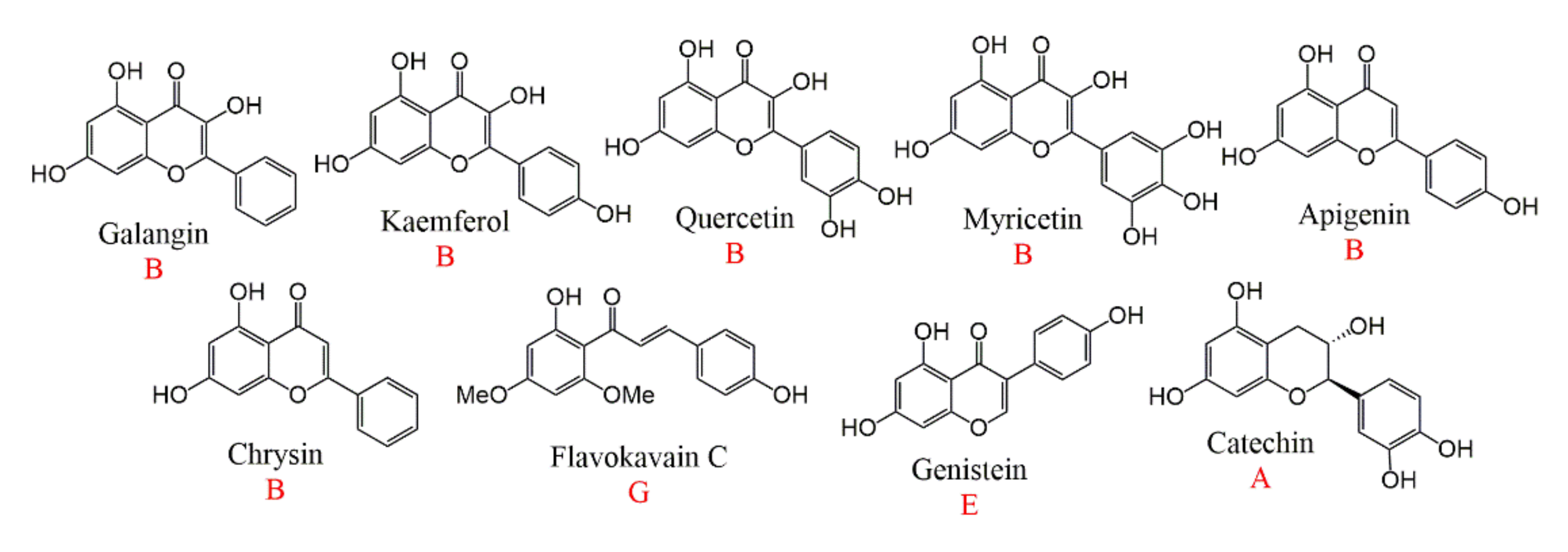
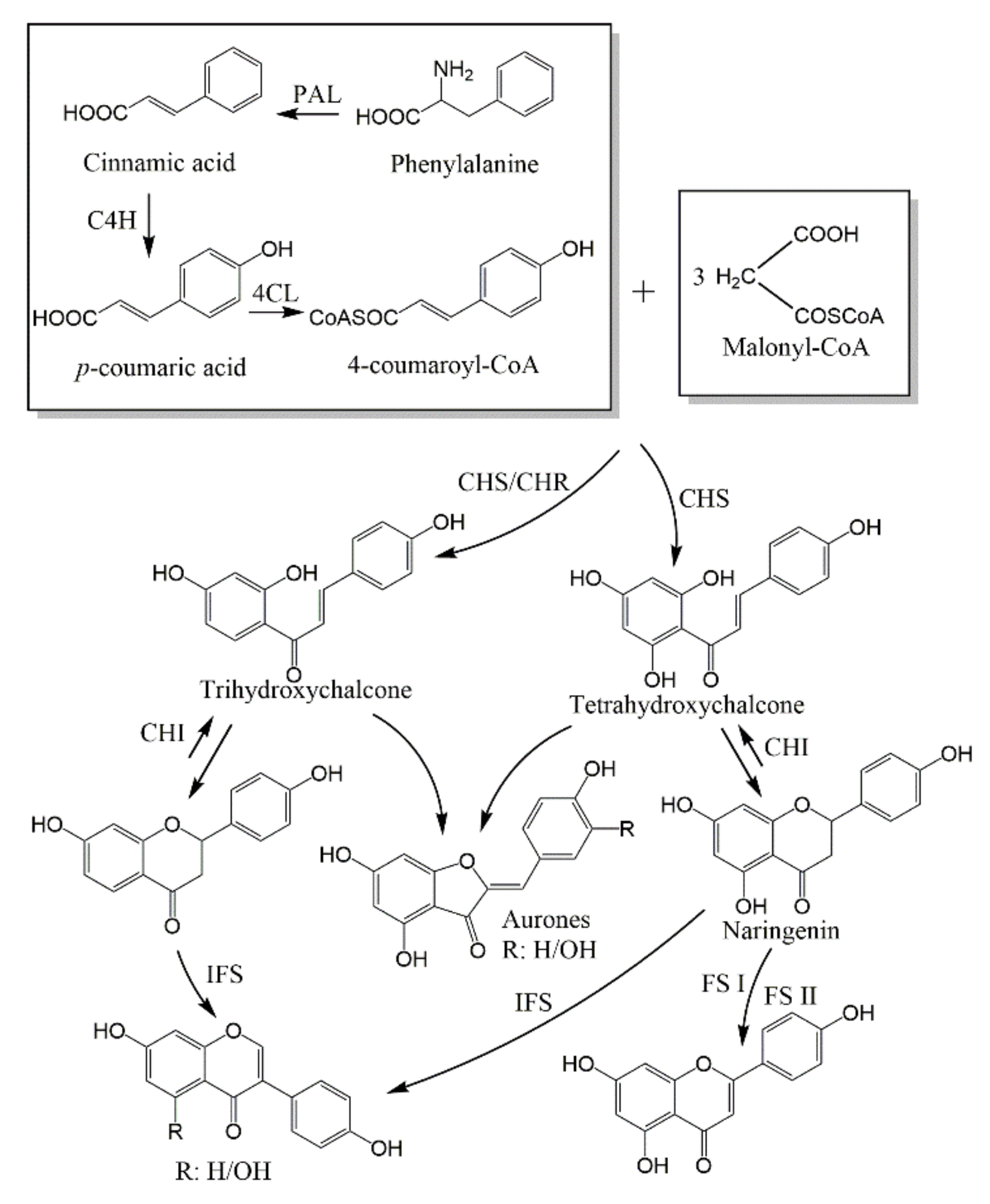



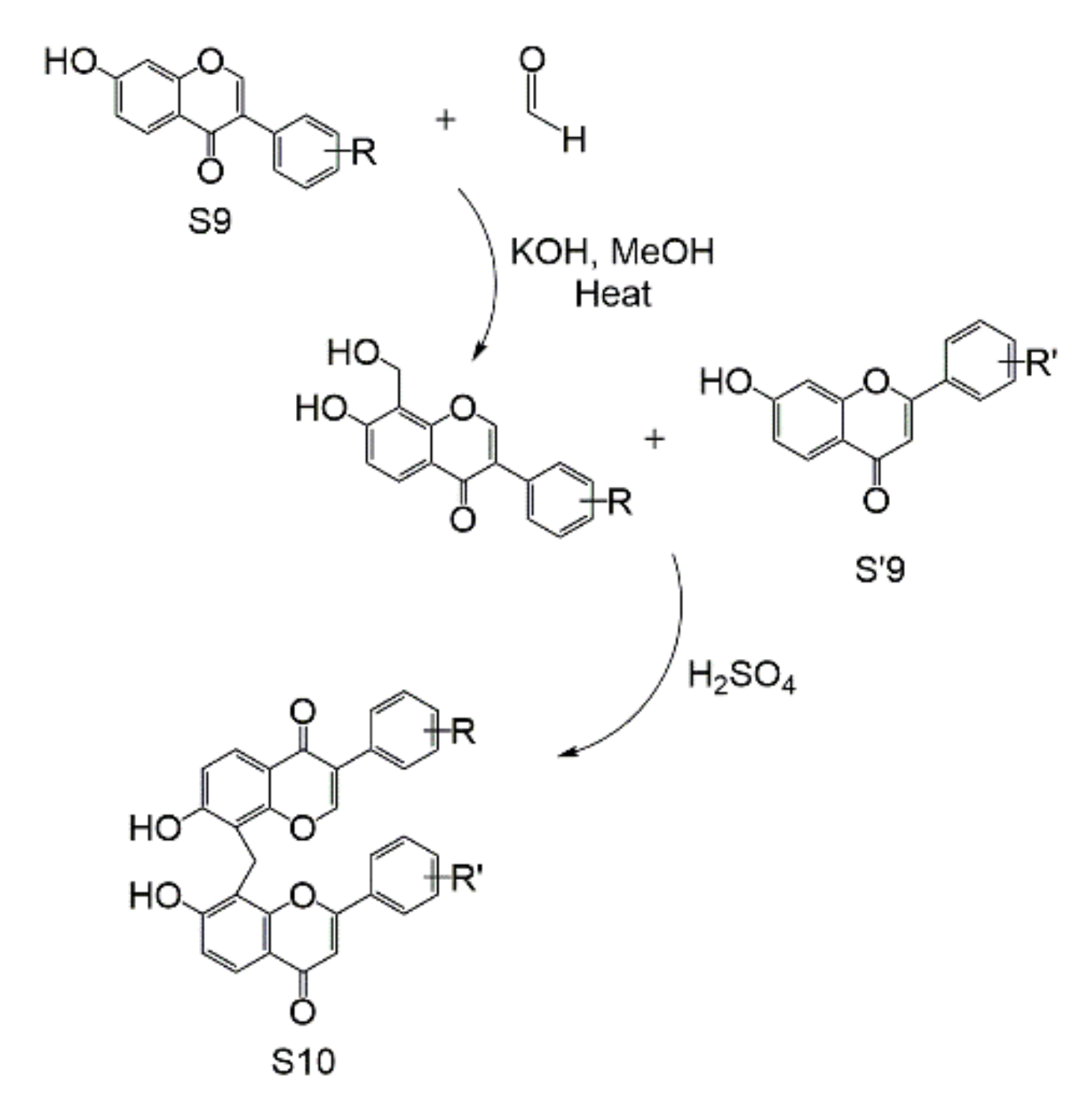
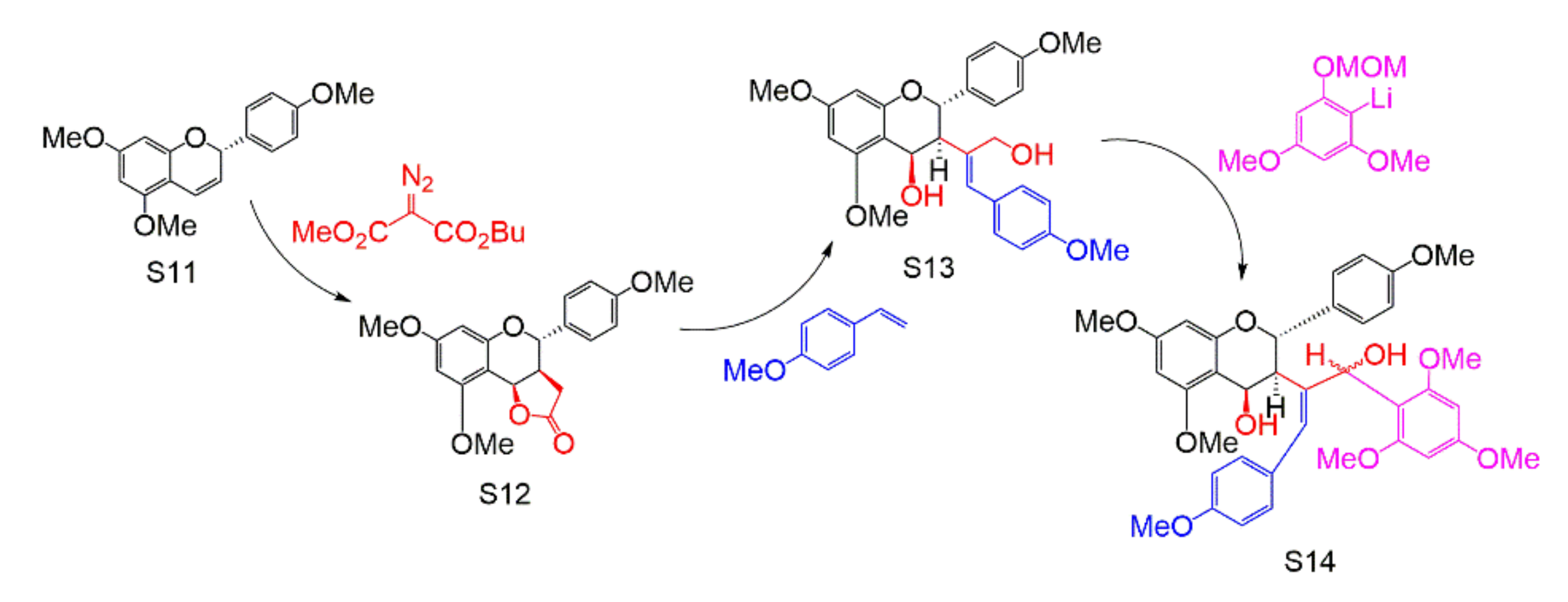
| Subtype | No. | Compounds Name | Monomer Type | Origin (Family *) | References |
|---|---|---|---|---|---|
| 2-3′′ | 1 | Linobiflavonoid | AH | Thym | [1] |
| 2′-2′′′ | 2 | I-3,II-3,I-5,II-5,I-7,II-7,I-4′,II-4′-octahydroxy[I-2′,II-2′]biflavone | BB | Clus | [3] |
| 3 | I-3,II-3,I-5,II-5,I-7,II-7,I-4′,II-4′-octamethoxy[I-2′,II-2′]biflavone | BB | Clus | [3] | |
| 4 | 3,3′′-di-O-α-L-rhamnopyranoside,2′,2′′′-bimyricetin | BB | Myrt | [4] | |
| 5 | Acuminatanol | AA | Anac | [5] | |
| 6 | Theasinensin A | AA | Thea | [6] | |
| 7 | Theasinensin B | AA | Thea | [6] | |
| 2′-6′′ | 8 | 2′,6′′-Biapignin | BB | Sela | [7] |
| 2′-8′′ | 9 | 2′,8′′-Biapignin | BB | Sela | [7] |
| 3-3′′ | 10 | Chamaejasmine | AA | Thym, Legu | [8] |
| 11 | 7-methoxychamaejasmin | AA | Thym | [8] | |
| 12 | Ruixianglangdu B | AA | Thym | [8] | |
| 13 | Isosikokianin | AA | Thym | [8] | |
| 14 | 7,4′,7′′,4′′-tetramethylisochamaejasmin | AA | Ochn | [9] | |
| 15 | 7,7′′-di-O-methylchamaejasmin | AA | Legu | [10] | |
| 16 | Campylospermone A | AA | Ochn | [10,11] | |
| 17 | Campylospermone B | AA | Ochn | [11] | |
| 18 | Isochamaejasmine | AA | Thym | [12] | |
| 19 | 7,7′′-di-O-glucosylisochamaejasmin | AA | Thym | [13] | |
| 20 | Neochamaejasmin A | AA | Thym | [8] | |
| 21 | Chamaejasmenin B | AA | Thym | [8] | |
| 22 | Chamaejasmenin C | AA | Thym | [8] | |
| 23 | Sikokianin A | AA | Thym | [8] | |
| 24 | 7-methoxyneochamaejasmin A | AA | Thym | [8] | |
| 25 | Chamaejasmenin D | AA | Thym | [8] | |
| 26 | Isoneochamaejasmin A | AA | Thym | [8] | |
| 27 | Isochamaejasmine B | AA | Thym | [8] | |
| 28 | Neochamaejasmin B | AA | Thym | [8] | |
| 29 | Sikokianin B | AA | Thym | [12] | |
| 30 | Chamaejasmenin A | AA | Thym | [8] | |
| 31 | Sikokianin C | AA | Thym | [8,12] | |
| 32 | Ruixianglangdu A | AA | Thym | [8] | |
| 33 | Isosikokianin A | AA | Thym | [8] | |
| 34 | Asteryomenin | AA | Aste | [14] | |
| 35 | Wikstaiwanone C | AA | Thym | [12] | |
| 36 | Sikokianin D | AA | Thym | [15] | |
| 37 | 2′′′-dehydroxy-2,2′′-bisteppogenin | AA | Thym | [1] | |
| 38 | 2,2′′-bisteppogenin 7-O-β-glucopyranoside | AA | Thym | [1] | |
| 39 | 2,2′′-bisteppogenin | AA | Thym | [1] | |
| 40 | Apigenil-(I-3,II-3)-naringenin | AA | Legu | [1] | |
| 41 | 6-aminoacryloylchamaejasmin | AA | Legu, Thym | [1,16] | |
| 42 | Ormocarpin | AA | Legu | [1,13,17] | |
| 43 | (−)-7,7′′-di-O-glucosylchamaejasmin | AA | Legu | [1,13] | |
| 44 | (−)-(2S, 3S, 2′′S, 3′′R)-7-O-glucosylchamaejasmin | AA | Legu | [1] | |
| 45 | (2S, 3R, 2′′S, 3′′R)-7-O-glucosylchamaejasmin | AA | Legu | [1,17] | |
| 46 | Campilospermone B | AA | Ochn | [1] | |
| 47 | Neochamaejasmin C | AA | Thym | [1] | |
| 48 | 7-methoxyneochamaejasmin B | AA | Thym | [1] | |
| 49 | 2′′′-dehydroxy-2,2′′-bisteppogenin 7-O-β-glucopyranoside | AA | Thym | [1] | |
| 50 | 3′′-epidiphysin | FF | Legu | [1] | |
| 51 | 5,5′′-di-O-methyldiphysin | FF | Legu | [1,17] | |
| 52 | Diphysin | FF | Legu | [1] | |
| 53 | 2,3-didehydro-(+)-chamaejasmin | AA | Thym | [16] | |
| 54 | 3,3′-biliquiritigenin | AA | Ochn, Legu | [17] | |
| 55 | Euchamaejasmin A | AA | Thym | [18] | |
| 56 | 7-O-β-D-glucopyranoside-diphysin | FF | Legu | [17] | |
| 57 | 3-Epimer, 5,5′-dideoxy-diphysin | FF | Anac | [19] | |
| 58 | 3,3′-diepimer, 5,5′-dideoxy-diphysin | FF | Anac | [19] | |
| 59 | 6′′′-hydroxylophirone | AG | Ochn | [20] |
| Subtype | No. | Compounds Name | Monomer Type | Origin (Family *) | References |
|---|---|---|---|---|---|
| 3-3′′′ | 60 | Taiwaniaflavone | BB | Cupr, Sela, Taxo | [21,22,23] |
| 61 | 7-O-methyltaiwaniaflavone | BB | Taxo | [23] | |
| 62 | 4′′′,7-di-O-methyltaiwaniaflavone | BB | Taxo | [23] | |
| 63 | Lupinabisone A | AB | Legu | [24] | |
| 3′-3′′′ | 64 | 2,3-dihydro-3′,3′′′-biapigenin | BB | Sela | [7] |
| 65 | 3′,3′′-binaringenin | BB | Sela | [7,25] | |
| 66 | Thuidinin | BB | Thui | [25] | |
| 67 | Kudzuisoflavone A | EE | Faba | [26] | |
| 3′-4′′′ | 68 | Chrysocauloflavone III | AB | Sela | [7] |
| 69 | Japoflavone D | BB | Capr | [27] | |
| 3′-5′′ | 70 | Aulacomniumbiaureusidin | HH | Aula | [28] |
| 3-6′′ | 71 | Daphnodorin K1 | AB | Thym | [29] |
| 72 | Daphnodorin K2 | AB | Thym | [29] | |
| 73 | Wikstaiwanone A | AB | Thym | [12] | |
| 74 | Wikstaiwanone B | AB | Thym | [12] | |
| 75 | Stephaflavone A | BB | Meni | [30] | |
| 76 | Stephaflavone B | BB | Meni | [30] | |
| 77 | Isomanniflavone | AA | Clus | [1] | |
| 78 | Ridiculuflavone | BB | Aris | [1] | |
| 79 | Afzelone B | AA | Ochn | [31] | |
| 80 | Ridiculuflavone A | BB | Aris | [32,33] | |
| 81 | Ridiculuflavone B | BB | Aris | [32,33] | |
| 82 | Ridiculuflavone D | BB | Aris | [32] | |
| 83 | Ridiculuflavone C | BB | Aris | [32] | |
| 84 | 4′′′,5,5′′,7′′-tetrahydroxy-3′′′,4′,7-trimethoxy-3,6′′-biflavone | BB | Aris | [32,34] | |
| 3′-6′′ | 85 | Robustaflavone | BB | Anac, Arau, Clus, Sela | [35,36] |
| 86 | 7′′-O-methylrobustaflavone | BB | Sela | [7] | |
| 87 | 4′-O-methylrobustaflavone | BB | Sela | [7,35,37] | |
| 88 | 7,4′-di-O-methylrobustaflavone | BB | Sela | [7,38] | |
| 89 | 4′,4′′′-di-O-methylrobustaflvone | BB | Sela | [7,39] | |
| 90 | 4′,7′′-di-O-methylrobustaflavone | BB | Sela | [7,37] | |
| 91 | 7,4′,4′′′-tri-O-methylrobustaflvone | BB | Sela | [7,39] | |
| 92 | Imbricataflavone A | AB | Podo, Sela | [7,35,40] | |
| 93 | Caesalflavone | AB | Legu, Sela | [7,41,42] | |
| 94 | Uncinatabiflavone D | AB | Sela | [7,43,44] | |
| 95 | 7,4′,7′′-tri-O-methyl-2,3-dihydrorobustaflavone | AB | Sela | [7,39] | |
| 96 | 5-O-methyl-2,3-dihydrorobustaflavone | AB | Sela | [7,42] | |
| 97 | Macrophylloflavone | AB | Sela | [45] | |
| 98 | 2,3-dihydrorobustaflavone 7,7′′-dimethyl ether | AB | Sela, Thym | [1] | |
| 99 | Imbricataflavone B | AB | Podo | [40] | |
| 100 | 4′-O-methyl-2′′,3′′-dihydrorobustaflavone | AB | Sela, Thym | [7,43] | |
| 101 | 7,4′-di-O-methyl-2′′,3′′-dihydrorobustaflvone | AB | Arau, Sela, Thym | [7,37] | |
| 102 | 4′,7′′-di-O-methyl-2′′,3′′-dihydrorobustaflavone | AB | Arau, Sela, Thym | [36] | |
| 103 | 7,4′,7′′-tri-O-methyl-2′′,3′′-dihydrorobustaflavone | AB | Sela, Thym | [7,37] | |
| 104 | Robustaflavanone | AA | Sela, Thym | [7,38] | |
| 105 | Uncinatabiflavone A | AA | Sela | [7,43,44] | |
| 106 | Uncinatabiflavone B | AA | Sela | [7,43,44] | |
| 107 | Uncinatabiflavone C | AA | Sela | [7,43,44] | |
| 108 | 7,4′,7′′-tri-O-methyl-2,3,2′′,3′′-tetrahydrorobustaflavone | AA | Sela | [7,35] | |
| 109 | Abiesin | BB | Pina | [46] | |
| 110 | 5′-hydroxyrobustaflavone | BB | Hylo | [47] | |
| 111 | 2′′,3′′-dihydro-5′′′-hydroxyrobustaflavone | AB | Mnia | [48] | |
| 112 | 5′,6′′-biluteolin | BB | Hylo, Dicr | [49] | |
| 113 | 2,3-dihydro-5′,6′′-biluteolin | AB | Dicr | [49] | |
| 114 | 2′′,3′′-dihydro-5′,6′′-biluteolin | AB | Mnia | [50] | |
| 3-7′′ | 115 | 5,5′′,6′′,7,8-pentahydroxy-2,2′′-bis(p-hydroxyphenyl)-4H,4′′H(3,7′′-bichromene)-4,4′′-dione | BB | Anac | [51] |
| 3′-7′′ | 116 | Lophirone M | AB | Ochn | [52] |
| 117 | Lophirone M hexa-acetate | AB | Ochn | [52] |
| No. | Compounds Name | Monomer Type | Origin (Family *) | References |
|---|---|---|---|---|
| 118 | Garciniaflavone E | AB | Clus | [1] |
| 119 | Garciniaflavone F | AB | Clus | [1] |
| 120 | Morelloflavone-7′′-O-β-D-glucosíde | BB | Clus | [1] |
| 121 | (+)-4′′′-O-methylmorelloflavone | AB | Clus | [1] |
| 122 | Biapigenin | BB | Clus | [1] |
| 123 | 4′′′-O-methyl-I3,II8-binaringenin | AA | Clus | [1,53] |
| 124 | Volkensiflavone | AB | Clus | [54] |
| 125 | Morelloflavone | AB | Clus | [55] |
| 126 | Spicataside | AB | Clus | [55] |
| 127 | Fukugiside | AB | Clus | [55] |
| 128 | 3′′′-O-methylfukugetin | AB | Clus | [1] |
| 129 | Garcinianin | AB | Clus | [55,56,57] |
| 130 | Madrunoudeaside | AB | Clus | [58] |
| 131 | Morelloflavone-7′-sulfate | AB | Clus | [59] |
| 132 | (2R,3S)-morelloflavone | AB | Mora | [60] |
| 133 | 7,4′,7′′,3′′′,4′′′-penta-O-acetylmorelloflavone | BB | Mora | [60] |
| 134 | 7,4′,7′′,3′′′,4′′′-penta-O-methylmorelloflavone | BB | Mora | [60] |
| 135 | 7,4′,7′′,3′′′,4′′′-penta-O-butanoylmorelloflavone | BB | Mora | [60] |
| 136 | Talbotaflavone | BB | Clus | [1,61] |
| 137 | Balsamiside A | AB | Bals | [62] |
| 138 | Balsamiside B | AB | Bals | [62] |
| 139 | Balsamiside C | AB | Bals | [62] |
| 140 | Balsamiside D | AB | Bals | [62] |
| 141 | Daphnodorin D1 | AB | Thym | [29] |
| 142 | Daphnodorin D2 | AB | Thym | [29] |
| 143 | Wikstrol A | AB | Thym | [63] |
| 144 | Wikstrol B | AB | Thym | [63] |
| 145 | II-3,I-5, II-5,II-7,I-4′,II-4′-hexahydroxy-(I-3,II-8)-flavonylflavanonol | BB | Clus | [1] |
| 146 | GB-1a | AA | Clus | [54,64] |
| 147 | GB-2a | AB | Clus | [65] |
| 148 | GB-1a-7′′-O-glycoside | AA | Clus | [55] |
| 149 | Xanthochymuside | AA | Clus | [55,66] |
| 150 | Kolaflavanone | AA | Clus | [55,64] |
| 151 | GB-1 | AA | Clus | [55,64] |
| 152 | GB-2 | AA | Clus | [55,64] |
| 153 | Manniflavanone | AA | Clus | [54,64] |
| 154 | GB-2a-II-4′-OMe | BB | Clus | [65] |
| 155 | Buchananiflavone | AA | Clus | [1,67] |
| 156 | Manniflavone-7′′-O-β-D-glucopyranoside | AB | Clus | [1] |
| 157 | (2R,3S,2′′R)3,8′′-binaringenin-7′′-O-β-glucoside | AA | Clus | [1,68] |
| 158 | (2R, 3S, 2′′R, 3′′R)GB1-7′′-O-β-glucoside | AA | Clus | [1,68] |
| 159 | Ent-naringenil-(I-3α,II-8)-4′-O-metilnaringenin | BB | Clus | [1,53] |
| 160 | 3,8′′-biapigenin | BB | Poly, Clus | [69,70] |
| 161 | Sumaflavone | BB | Anac | [7,71] |
| 162 | 4′-methoxydaphnodorin D1 | AB | Thym | [72] |
| 163 | 4′-methoxydaphnodorin D2 | AB | Thym | [72] |
| 164 | Pancibiflavonol | AB | Clus | [73] |
| 165 | Volkensiflavone 7-sulfate | AB | Clus | [74] |
| 166 | 8-(3′,4′,5,7-tetrahydroxyflavanon-3-yl)-4′,5,7-trihydroxyflavone | AB | Clus | [75] |
| 167 | GB-3 | AA | Clus | [64] |
| 168 | GB-4 | AA | Clus | [76] |
| 169 | GB-2b | AA | Clus | [64] |
| 170 | GB-4a | AA | Clus | [76] |
| 171 | 4′′′-O-methylfukugetin | AB | Clus | [77] |
| 172 | Lupinalbisone B | AB | Legu | [24] |
| No. | Compounds Name | Monomer Type | Origin (Family *) | References |
|---|---|---|---|---|
| 173 | Amentoflavone | BB | Anac, Capr, Clus, Cupr, Gink, Pina, Podo, Pter, Sela, Taxa, Taxo | [2] |
| 174 | Isoginkgetin | BB | Gink, Sela | [2,7] |
| 175 | 3′,8′′-biisokaempferide | BB | Pter, Vell | [1,2] |
| 176 | 7,7′′-di-O-methylamentoflavone | BB | Arau, Cupr, Podo, Sela, Taxo | [2,7] |
| 177 | 4′,7′′-di-O-methylamentoflavone | BB | Arau, Sela, Taxo | [2,7] |
| 178 | 5′-methoxybilobetin | BB | Gink, Mnia | [2] |
| 179 | 7,4,7′′,4′′′-tetra-O-methylamentoflavone | BB | Sela | [2,7] |
| 180 | 7,4′,7′′-tri-O-methylamentoflavone | BB | Arau, Ceph, Cupr, Taxa | [2] |
| 181 | Acetyl ginkgetin | BB | Gink | [78] |
| 182 | Acetyl isogenkgetin | BB | Gink | [78] |
| 183 | Acetyl sciadopitysin | BB | Gink | [78] |
| 184 | 6-C-methyl-7-O-methylamentoflavone | BB | Ceph | [79] |
| 185 | 3′′′-hydroxy-4′′′,7-dimethylamentoflavone | BB | Aris, Taxa | [80] |
| 186 | Anacarduflavone | BB | Anac | [81] |
| 187 | Bilcarobetin | BB | Gink, Sela | [2,7] |
| 188 | CGY-1 | BB | Lili | [82] |
| 189 | Ginkgetin | BB | Arau, Gink, Sela, Taxa, Taxo | [2,7] |
| 190 | Ginkgetin 7-O-D-glucopyranoside | BB | Gink | [83] |
| 191 | Heveaflavone | BB | Euph, Sela | [2,7] |
| 192 | 7-O-gluamentoflavone | BB | Cupr, Gink | [84] |
| 193 | Isoginkgetin 7-O-D-glucopyranoside | BB | Gink | [83] |
| 194 | Amentoflavone 7′′-O-β-D-glucopyranoside | BB | Cupr, Gink | [84] |
| 195 | Kayaflavone | BB | Podo, Sela, Taxa, Taxo | [2,7] |
| 196 | Oliveriflavone | BB | Taxa | [79] |
| 197 | Oliveriflavone B | BB | Taxa | [85] |
| 198 | Oliveriflavone C | BB | Taxa | [85] |
| 199 | Podocarduflavone B | BB | Podo | [2] |
| 200 | Podocarpusflavone A | BB | Podo, Sela | [2,7] |
| 201 | Sciadopitysin | BB | Cupr, Gink, Podo, Taxa, Taxo | [2] |
| 202 | Sequoiaflavone | BB | Sela, Taxo | [2,7] |
| 203 | Sotetsuflavone | BB | Cyca, Sela, Taxo | [2,7] |
| 204 | Taiwaniaflavone A | BB | Ceph | [2] |
| 205 | Dulcisbiflavonoid A | BB | Clus | [59] |
| 206 | Putraflavone | BB | Euph, Podo | [1] |
| 207 | (2S,2′′S)-2,3-di-hydroisoginkgetin | AB | Cyca | [86] |
| 208 | 2,3-dihydro-6-methylginkgetin | AB | Ceph | [1] |
| 209 | 2,3-dihydrosciadopitysin | AB | Gink, Taxo | [83] |
| 210 | 4′,7′′-di-O-methyl-2,3-dihydroamentoflavone | AB | Sela | [7] |
| 211 | (2S)-2,3-dihydroamentoflavone | AB | Sela | [7] |
| 212 | 7,4′-di-O-methyl-2,3-dihydroamentoflavone | AB | Cupr, Sela | [7] |
| 213 | 7,4′,7′′-tri-O-methyl-2,3-dihydroamentoflavone | AB | Arau, Sela, Taxa | [7] |
| 214 | 2,3-dihydro-4-O-methylamentoflavone | AB | Cyca, Sela | [87] |
| 215 | (2S)-4′-O-methyl-2,3-tetrahydroamentoflavone | AB | Cyca, Sela | [7] |
| 216 | Garciniaflavone A | AB | Clus | [1,2] |
| 217 | Garciniaflavone B | AB | Clus | [1,2] |
| 218 | Garciniaflavone C | AB | Clus | [1,2] |
| 219 | Garciniaflavone D | AB | Clus | [1,2] |
| 220 | 6′′-hydroxy-2,3-dihydroamentoflavone | AB | Sela | [7] |
| 221 | Selamariscina A | AB | Sela | [36] |
| 222 | 2′′,3′′-dihydroamentoflavone | AB | Anac, Cyca, Sela, Taxo | [7] |
| 223 | 4′-O-methyl-2′′,3′′-dihydroamentoflavone | AB | Cyca, Sela | [1,7] |
| 224 | (2S,2′′S)-2,3,2′′,3′′-tetrahydroisoginkgetin | AA | Arau, Cyca, Podo | [86] |
| 225 | (2S,2′′S)-2,3,2′′,3′′-tetrahydroamentoflavone | AA | Anac, Cyca, Sela | [7,86] |
| 226 | (2S,2′′S)-4′-O-methyl-2,3,2′′,3′′-tetrahydroamentoflavone | AA | Sela | [7] |
| 227 | Taxusbiflavone A | BB | Capr | [88] |
| 228 | 3′′′-methoxyamentoflavone | BB | Anac | [89] |
| 229 | 3′′′,5′-dihydroxyamentoflavone | BB | Dicr | [49] |
| 230 | (2S,2′′S)-3′,4′,4′′′,5,5′′,7′′-hexahydroxy-8,3′′′-biflavanone | AA | Anac | [81] |
| 231 | 3′,5,5′′-trihydroxy-4′,4′′′,7′′-trimethoxy-8,3′′′-biflavanone | AA | Anac | [90] |
| 232 | 5,5′′-dihydroxy-3′,4′,4′′′,7′′-tetramethoxy-8,3′′′-biflavanone | AA | Anac | [91] |
| 233 | Anacarduflavanone | AA | Anac | [92] |
| No. | Compounds Name | Monomer Type | Origin (Family *) | References |
|---|---|---|---|---|
| 234 | Sarcandrone D | AA | Chlo | [1] |
| 235 | Procyanidin B5 | AA | Dava, Malv, Pina, Rosa, Sapi | [93,94] |
| 236 | Epicatechin 3-O-gallate-(4β→6)-epicatechin 3-O-(4-hydroxybenzoate) | AA | Myro | [95] |
| 237 | 3′-O-galloylprocyanidin B5 | AA | Vita | [95,96] |
| 238 | 3,3′-di-O-galloylprocyanidin B5 | AA | Poly | [97] |
| 239 | Epigallocatechin 3-O-gallate-(4β→6)-epicatechin 3-O-gallate | AA | Thea | [98] |
| 240 | Epicatechin 3-O-gallate-(4β→6)-epigallocatechin 3-O-gallate | AA | Thea | [98] |
| 241 | Epigallocatechin-(4β→6)-epigallocatechin 3-O-gallate | AA | Cist | [99] |
| 242 | 3,3′-di-O-galloylprodelphinidin B5 | AA | Myri | [100] |
| 243 | Epiafzelechin 3-O-gallate-(4β→6)-epigallocatechin 3-O-gallate | AA | Thea | [98] |
| 244 | Procyanidin B6 | AA | Cupr, Eric, Rosa | [93] |
| 245 | Procyanidin B7 | AA | Cupr, Eric | [93] |
| 246 | Procyanidin B8 | AA | Eric, Rosa, Sali | [93] |
| 247 | Ent-epicatechin-(4α→6)-ent-epicatechin | AA | Malp | [101] |
| 248 | Fisetinidol-(4β→6)-fisetinidol-4β-ol | AA | Legu | [102] |
| 249 | Fisetinidol-(4β→6)-fisetinidol | AA | Legu | [103] |
| 250 | Fisetinidol-(4β→6)-fisetinidol-4α-ol | AA | Legu | [102] |
| 251 | Fisetinidol-(4β→6)-ent-epifisetinidol | AA | Legu | [103] |
| 252 | Fisetinidol-(4α→6)-fisetinidol-4β-ol | AA | Legu | [102] |
| 253 | Fisetinidol-(4α→6)-fisetinidol | AA | Legu | [103] |
| 254 | Fisetinidol-(4α→6)-fisetinidol-4α-ol | AA | Legu | [102] |
| 255 | Fisetinidol-(4α→6)-ent-epifisetinidol | AA | Legu | [103] |
| 256 | Globiflorin 3B1 | AA | Legu | [104] |
| 257 | Globiflorin 3B2 | AA | Legu | [104] |
| 258 | Guibourtinidol-(4α→6)-afzelechin | AA | Legu | [105] |
| 259 | ent-Guibourtinidol-(4β→6)-catechin | AA | Rosa | [106] |
| 260 | Epicatechin-(4β→6)-epicatechin-(4β→2)-phloroglucinol | AA | Legu, Pina, Rosa | [107,108,109] |
| 261 | Guibourtinidol-(4α→6)-epicatechin-8-carboxylic acid | AA | Legu | [104] |
| 262 | Guibourtinidol-(4α→6)-catechin-8-carboxylic acid | AA | Legu | [104] |
| 263 | Epioritin-(4β→6)-epioritin-4α-ol | AA | Legu | [110] |
| 264 | Epioritin-(4β→6)-epioritin-4β-ol | AA | Legu | [110] |
| 265 | Epioritin-(4β→6)-oritin-4α-ol | AA | Legu | [111] |
| 266 | Epioritin-(4β→6)-ent-oritin-4α-ol | AA | Legu | [110] |
| 267 | Ent-Oritin-(4β→6)-epioritin-4α-ol | AA | Legu | [110] |
| 268 | Ent-Oritin-(4α→6)-epioritin-4α-ol | AA | Legu | [111] |
| 269 | Ent-Oritin-(4α→6)-epioritin-4β-ol | AA | Legu | [111] |
| 270 | Ent-Oritin-(4β→6)-oritin-4α-ol | AA | Legu | [110] |
| 271 | Ent-Oritin-(4α→6)-oritin-4α-ol | AA | Legu | [111] |
| No. | Compounds Name | Monomer Type | Origin (Family *) | References |
|---|---|---|---|---|
| 272 | Juglbiflavone A | AB | Jugl | [112] |
| 273 | Sarcandrone | AA | Chlo | [1] |
| 274 | Procyanidin B2 | AA | Aizo, Rosa, Sapi | [94,113,114] |
| 275 | Procyanidin B2 7′-xyloside | AA | Legu | [115] |
| 276 | 3′-galloylprocyanidin B2 | AA | Poly,Vita | [6] |
| 277 | 3,3′-digalloylprocyanidin B2 | AA | Poly, Thea, Rosa | [6,114] |
| 278 | 3′-O-(3,4-di-O-methylgalloyl)procyanidin B2 | AA | Poly | [94] |
| 279 | Epicatechin-(4α→8)-epicatechin | AA | Rosa | [116] |
| 280 | Procyanidin B1 | AA | Legu | [114] |
| 281 | 3′-(4-hydroxybenzoyl)procyanidin B1 | AA | Hama | [117] |
| 282 | 3-galloylprocyanidin B1 | AA | Poly | [114,118] |
| 283 | 3′-O-(1-hydroxy-6-oxo-2-cyclohexene-1-carboxylate)procyanidin B1 | AA | Sali | [119] |
| 284 | Procyanidin B4 | AA | Rosa | [113] |
| 285 | 3-O-β-D-glucopyranoside, 3′-O-(6-O-E-cinnamoyl-β-D-glucopyranoside)Procyanidin B4 | AA | Legu | [120] |
| 286 | 3′-galloylprocyanidin B4 | AA | Euph | [6] |
| 287 | Procyanidin B3 | AA | Sali | [113,121] |
| 288 | 3-rhamnoside-procyanidin B3 | AA | Faga | [122] |
| 289 | 3-glucoside-procyanidin B3 | AA | Faga | [122] |
| 290 | 3-O-β-D-glucopyranoside, 3′-O-(2-O-E-cinnamoyl-β-D-glucopyranoside)procyanidin B3 | AA | Legu | [120] |
| 291 | Procyanidin B3 3′-rhamnoside | AA | Faga | [122] |
| 292 | Procyanidin B3 3′-O-glucoside | AA | Rosa | [123] |
| 293 | Procyanidin B3 7-glucoside | AA | Poly | [124] |
| 294 | Catechin-(4α→8)-catechin 7-xyloside | AA | Betu | [125] |
| 295 | 3-galloylprocyanidin B3 | AA | Rosa | [126] |
| 296 | 3,3′-di-Ac-3′′′-O-β-D-glucopyranoside procyanidin B3 | AA | Poly | [127] |
| 297 | 3′-O-(1-Hydroxy-6-oxo-2-cyclohexene-1-carboxylate)procyanidin B3 | AA | Sali | [119] |
| 298 | Epicatechin-(4β→8)-ent-epicatechin | AA | Arec | [128] |
| 299 | Ent-epicatechin-(4α→8)-epicatechin | AA | Arec | [128] |
| 300 | Ent-epicatechin-(4α→8)-catechin | AA | Arec | [128] |
| 301 | Ent-epicatechin-(4α→8)-ent-epicatechin | AA | Arec | [128] |
| 302 | 3-O-(3,4,5-trihydroxybenzoyl)ent-epicatechin-(4α→8)-ent-epicatechin | AA | Malp | [101] |
| 303 | 3′-O-(3,4,5-trihydroxybenzoyl)ent-epicatechin-(4α→8)-ent-epicatechin | AA | Malp | [101] |
| 304 | 3,3′-bis-O-(3,4,5-trihydroxybenzoyl)ent-epicatechin-(4α→8)-ent-epicatechin | AA | Malp | [101] |
| 305 | Auricassidin | AA | Legu | [129] |
| 306 | 3,3′,4′,5,7-pentahydroxyflavan-(4→8)-3,4′,5,7-tetrahydroxyflavan | AA | Legu | [130] |
| 307 | Epicatechin-(4β→8)-epiafzelechin | AA | Legu | [131,132] |
| 308 | Catechin-(4α→8)-epiafzelechin | AA | Legu | [133] |
| 309 | Epicatechin-(4β→8)-ent-epiafzelechin | AA | Legu | [134] |
| 310 | Ent-epicatechin-(4α→8)-epiafzelechin | AA | Legu | [134] |
| 311 | Ent-epicatechin-(4α→8)-ent-epiafzelechin | AA | Legu | [134] |
| 312 | Epiguibourtinidol-(4β→8)-epicatechin | AA | Legu | [135] |
| 313 | Guibourtinidol-(4β→8)-epicatechin | AA | Legu | [105] |
| 314 | Guibourtinidol-(4β→8)-epiafzelechin | AA | Legu | [105] |
| 315 | Guibourtinidol-(4α→8)-epicatechin | AA | Legu | [104] |
| 316 | Guibourtinidol-(4α→8)-epiafzelechin | AA | Legu | [105] |
| 317 | Guibourtinidol-(4β→8)-catechin | AA | Legu | [104] |
| 318 | Guibourtinidol-(4α→8)-catechin | AA | Legu | [104] |
| 319 | Calodenin C | AA | Legu | [136] |
| 320 | Ent-guibourtinidol-(4β→8)-epicatechin | AA | Legu | [105] |
| 321 | Epiafzelechin-(4β→8)-epicatechin | AA | Legu, Poly | [134,137] |
| 322 | 3′-O-(3,4,5-trihydroxybenzoyl)epiafzelechin-(4β→8)-epicatechin | AA | Poly | [137] |
| 323 | 3,3′-bis-O-(3,4,5-trihydroxybenzoyl)epiafzelechin-(4β→8)-epicatechin | AA | Poly | [137] |
| 324 | Ouratea proanthocyanidin A | AA | Cela, Ochn | [138] |
| 325 | Ouratea proanthocyanidin B | AA | Cela, Ochn | [138] |
| 326 | Epiafzelechin-(4ξ→8)-epicatechin | AA | Poly | [94] |
| 327 | 3′-O-(4-hydroxybenzoyl)epiafzelechin-(4ξ→8)-epicatechin | AA | Poly | [94] |
| 328 | 3′-O-(3-hydroxy-4,5-dimethoxybenzoyl)epiafzelechin-(4ξ→8)-epicatechin | AA | Poly | [94] |
| 329 | Gambiriin C | AA | Rubi | [139] |
| 330 | Afzelechin-(4α→8)-epicatechin | AA | Rhiz | [140] |
| 331 | 3′-O-(4-hydroxy-3-methoxybenzoyl), 3-O-α-L-rhamnopyranoside-afzelechin-(4α→8)-epicatechin | AA | Euph | [141] |
| No. | Compounds Name | Monomer Type | Origin (Family *) | References |
|---|---|---|---|---|
| 332 | 3′-O-(4-hydroxy-3,5-dimethoxybenzoyl), 3-O-α-L-rhamnopyranoside-afzelechin-(4α→8)-epicatechin | AA | Euph | [141] |
| 333 | afzelechin-(4α→8)-catechin | AA | Rhiz, Rosa | [121,140] |
| 334 | 3-O-α-L-rhamnopyranoside-afzelechin-(4α→8)-catechin | AA | Faga | [142] |
| 335 | 3-O-β-D-glucopyranoside-afzelechin-(4α→8)-catechin | AA | Faga | [142] |
| 336 | Epiafzelechin-(4β→8)-ent-epicatechin | AA | Legu | [132] |
| 337 | Ent-epiafzelechin-(4α→8)-epicatechin | AA | Legu | [132] |
| 338 | Ent-epiafzelechin-(4α→8)-ent-epicatechin | AA | Legu | [134] |
| 339 | Ichangol | AA | Adox | [143] |
| 340 | Epicatechin-(4β→8)-epicatechin-(4β→2)-phloroglucinol | AA | Legu, Pina | [109,144] |
| 341 | Epigallocatechin-(4β→8)-epicatechin-(4β→2)-phloroglucinol | AA | Legu | [109] |
| 342 | Epigallocatechin-(4β→8)-epigallocatechin-(4β→2)-phloroglucinol 3,3′-digallate | AA | Cist | [145] |
| 343 | Catechin-(4α→8)-epicatechin-(4β→2)-phloroglucinol | AA | Pina | [144] |
| 344 | Gallocatechin-(4α→8)-epigallocatechin-(4β→2)-phloroglucinol | AA | Cist | [145] |
| 345 | Epirobinetinidol-(4β→8)-catechin | AA | Legu | [146] |
| 346 | Robinetinidol-(4β→8)-epigallocatechin | AA | Mimo | [147] |
| 347 | Robinetinidol-(4β→8)-epigallocatechin-3′-gallate | AA | Mimo | [147] |
| 348 | Robinetinidol-(4α→8)-epigallocatechin | AA | Mimo | [147] |
| 349 | Robinetinidol-(4α→8)-epigallocatechin-3′-gallate | AA | Mimo | [147] |
| 350 | Robinetinidol-(4β→8)-catechin | AA | Legu | [148] |
| 351 | Robinetinidol-(4α→8)-gallocatechin | AA | Legu | [149] |
| 352 | Robinetinidol-(4α→8)-catechin | AA | Legu | [149] |
| 353 | Prodelphinidin B2 | AA | Phyl, Legu, Myri | [100] |
| 354 | 3′-O-(4-hydroxybenzoyl)prodelphinidin B2 | AA | Legu | [150] |
| 355 | 3-O-galloylprodelphinidin B2 | AA | Cist, Poly | [99,151] |
| 356 | 3′-galloylprodelphinidin B2 | AA | Cist, Myri, Thea | [6,99,100,152] |
| 357 | Rhodisin | AA | Myri, Cras | [100,153] |
| 358 | Rhodisinoside | AA | Cras | [153] |
| 359 | Epicatechin-(4β→8)-epigallocatechin-3-O-gallate | AA | Thea | [98] |
| 360 | Epicatechin-3-O-gallate-(4β→8)-epigallocatechin-3-O-gallate | AA | Thea | [98] |
| 361 | Epicatechin-(4β→8)-4′-O-methylepigallocatechin | AA | Cela | [154] |
| 362 | Epigallocatechin-(4β→8)-epicatechin-3-O-gallate | AA | Thea | [155] |
| 363 | Prodelphinidin B1 | AA | Cist, Legu | [150,156] |
| 364 | 3-galloylprodelphinidin B1 | AA | Cist, Hama | [117,156] |
| 365 | 3,3′-digalloylprodelphinidin B1 | AA | Myri | [100] |
| 366 | Epigallocatechin-(4β→8)-4′-O-methylgallocatechin | AA | Legu | [157] |
| 367 | Epicatechin-(4β→8)-gallocatechin | AA | Phyl | [158] |
| 368 | Epicatechin-(4β→8)-4′-O-methylgallocatechin | AA | Legu | [157] |
| 369 | Epigallocatechin-(4β→8)-catechin | AA | Legu, Pina | [117,159] |
| 370 | 3′′′-Deoxy, 3-O-(3,4,5-trihydroxybenzoyl)epigallocatechin-(4β→8)-catechin | AA | Hama | [117] |
| 371 | Prodelphinidin B4 | AA | Thea, Phyl, Gros | [160] |
| 372 | Gallocatechin-(4α→8)-epigallocatechin-3-O-(4-hydroxybenzoate) | AA | Mimo | [150] |
| 373 | 3′-galloylprodelphinidin B4 | AA | Thea | [98] |
| 374 | 4′′,4′′′-di-me ether-prodelphinidin B4 | AA | Legu | [150] |
| 375 | Catechin-(4α→8)-epigallocatechin | AA | Thea | [161] |
| 376 | Catechin-(4α→8)-epigallocatechin-3-O-gallate | AA | Thea | [161] |
| 377 | Gallocatechin-(4α→8)-epicatechin | AA | Thea | [161] |
| 378 | Prodelphinidin B3 | AA | Faga, Rham | [162,163] |
| 379 | 4′′,4′′′-di-O-methylprodelphinidin B3 | AA | Legu | [157] |
| 380 | Catechin-(4α→8)-gallocatechin | AA | Cist | [156] |
| 381 | Prodelphinidin C | AA | Hama, Myri, Faga, Sali | [117,160,163,164] |
| 382 | Epifisetinidol-(4β→8)-epicatechin | AA | Legu | [135] |
| 383 | Epifisetinidol-(4β→8)-catechin | AA | Legu | [165] |
| 384 | Fisetinidol-(4β→8)-epicatechin | AA | Legu | [166] |
| 385 | Fisetinidol-(4α→8)-epicatechin | AA | Legu | [166,167] |
| 386 | Fisetinidol-(4β→8)-catechin | AA | Legu | [166] |
| 387 | Fisetinidol-(4α→8)-catechin | AA | Legu | [168] |
| 388 | Fisetidinol-(4α→8)-3-O-galloylcatechin | AA | Legu | [166] |
| 389 | Ent-fisetinidol-(4β→8)-epicatechin | AA | Anac | [149] |
| 390 | Ent-fisetinidol-(4β→8)-catechin | AA | Anac, Legu | [149,169] |
| 391 | Ent-fisetinidol-(4α→8)-catechin | AA | Anac, Legu | [149] |
| Subtype | No. | Compounds Name | Monomer Type | Origin (Family *) | References |
|---|---|---|---|---|---|
| 4′-8′′ | 392 | 5,3′,5′′,4′′′-tetrahydroxy-3′′′,5′′′-dimethoxy-biflavone(4′→8′′)-7-O-(2-rhamnoside)rhamnoside | BB | Apia | [170] |
| 5-5′′ | 393 | 8,8′-bis(7,8-dihydroxy-2-C-methyl-2H-1-benzopyran-5-yl)-4,4′-dimethoxy-[5,5′-bi-6H-furo[3,2-h][1]benzopyran]-6,6′-dione | BB | Legu | [171] |
| 394 | 3-C-(6-deoxy-α-L-mannopyranosyl)-3′-C-α-d-glucopyranosyl-2,2′-bis(4-hydroxyphenyl)-7,7′-dimethyl-[5,5′-bi-4H-1-benzopyran]-4,4′-dione | BB | Legu | [171] | |
| 6-6′′ | 395 | Succedaneaflavanone | AA | Anac | [1] |
| 396 | 6,6′′-bigenkwanin | BB | Ochn | [172] | |
| 6-γ | 397 | 8-methylsocotrin-3′-methoxy-4′-ol | AG | Drac | [1] |
| 398 | 8-methylsocotrin-4′-methoxy-3′-ol | AG | Drac | [1] | |
| 399 | 8-methylsocotrin-3-methoxy-4-ol | AG | Drac | [1] | |
| 400 | 8-methylsocotrin-4-methoxy-3-ol | AG | Drac | [1] | |
| 401 | 8-methylsocotrin-4-ol | AG | Drac | [1] | |
| 6-8′′ | 402 | 6,8′′-bigenkwanin | BB | Arau, Ochn | [172,173] |
| 403 | Agathisflavone | BB | Anac, Ochn | [173] | |
| 404 | 7,7′′,4′′′-tri-O-methylagathisflavone | BB | Arau | [1,174] | |
| 405 | 7,4′′′-di-O-methylagathisflavone | BB | Arau | [1] | |
| 406 | Agathisflavone A | BB | Arau | [173,174] | |
| 407 | Ouratine A | BB | Ochn | [1,175] | |
| 408 | Agatisflavone | AB | Ochn | [1] | |
| 409 | 7,4′,7′′,4′′′-tetra-O-methylcupressuflavone | AB | Sela | [7] | |
| 410 | 7,4′,7′′-tri-O-methylcupressuflavone | AB | Arau | [1] | |
| 411 | 7,7′′-di-O-methylcupressuflavone | AB | Arau | [1] | |
| 412 | Rhusflavone | AB | Anac | [176] | |
| 413 | Lateriflavanone | AB | Clus | [1] | |
| 414 | 4′′′-O-methylagatisflavone | AB | Clus, Ochn | [54,177] | |
| 415 | Rhusflavanone | AA | Anac | [178] | |
| 416 | 6,8′′-binaringenin | AA | Clus | [178] | |
| 417 | 3′′′,4′,4′′′,5,5′′,7,7′′-hepta-me ether-3,3′′,3′′′,4′,4′′′, 5,5′′,7,7′′-nonahydroxy-6,8′′-biflavanone | AA | Ochn | [179] | |
| 418 | Ouratine B | BB | Ochn | [175] | |
| 419 | 4′′′-O-methylagathisflavone | BB | Ochn | [177] | |
| 420 | 7′′-O-methylagathisflavone | BB | Ochn | [180] | |
| 421 | Agathisflavone B | BB | Arau | [174] | |
| 7-7′′ | 422 | 4′-methoxy-7,7′′-biflavone | BB | Legu | [181] |
| 8-8′′ | 423 | Cupressuflavone | AA | Anac, Arau, Cupr | [182] |
| 424 | 3,3′′-dihydroxycupressuflavone | BB | Thea | [183] | |
| 425 | 4′-O-methylcupressuflavone | BB | Clus | [54] | |
| 426 | Mesuaferrone B | AB | Anac, Clus | [178,184] | |
| 427 | 4′,4′′′-di-O-methylcupressuflavanone | AA | Comp | [185] | |
| 428 | (R)4′-O-β-D-glucopyranoside-cupressuflavone | AA | Cupr | [186] | |
| 429 | (S)4′-O-β-D-glucopyranoside-cupressuflavone | AA | Cupr | [186] | |
| 430 | 7-me ether-cupressuflavone | BB | Arau | [173,187] | |
| 431 | 8,8′′-bigenkwanin | BB | Arau, Cupr | [186,187] | |
| 432 | W11 | BB | Arau, Phyl | [188] | |
| 433 | 4′,7,7′′-tri-O-methylcupressuflavone | BB | Arau | [187] | |
| 434 | WB1 | BB | Arau | [173,187] | |
| 435 | Moghatin | BB | Malv | [189] | |
| 436 | Neorhusflavanone | AA | Anac, Calo | [184,190] |
| Subtype | No. | Compounds Name | Monomer Type | Origin (Family *) | References |
|---|---|---|---|---|---|
| 3′-O-3′′′ | 437 | Sparinaritin | AB | Chry | [191] |
| 3-O-4′′ | 438 | Epioritin(4β-3)-epioritin-4β-ol | AA | Legu | [20] |
| 3-O-4′′′ | 439 | Delicaflavone | BB | Sela | [7] |
| 440 | 5,7,4′,5′′-tetrahydroxy-7′′-metroxy-[3-O-4′′′]-biflavone | BB | Sela | [7] | |
| 441 | Chrysocauloflavone I | AB | Sela | [7] | |
| 442 | Chrysocauloflavone II | AB | Sela | [7] | |
| 443 | Baeckein E | AB | Myrt | [192] | |
| 444 | Baeckein C | AB | Myrt | [192] | |
| 445 | Baeckein D | AB | Myrt | [192] | |
| 3′-O-4′′′ | 446 | Ochnaflavone | BB | Ochn | [7,193] |
| 447 | Ochnaflvone 7′′-O-β-D-glucopyranoside | BB | Ochn | [194] | |
| 448 | 2′′,3′′-dihydroochnaflavone | AB | Ochn | [7,195] | |
| 449 | 2,3-dihydro-4′,7,7′′-tri-O-methylochnaflavone | AB | Ochn | [7,196] | |
| 450 | Sulcatone A | AB | Ochn | [197] | |
| 451 | 4′-me ether-ochnaflavone | BB | Ochn | [193] | |
| 452 | 7-O-methylochnaflavone | BB | Ochn | [198] | |
| 453 | 7′′-O-methylochnaflavone | BB | Ochn | [199] | |
| 454 | 4′,7-di-O-methylochnaflavone | BB | Ochn | [193] | |
| 455 | 2,3-dihydroochnaflavone | AB | Ochn | [200] | |
| 456 | 2,3-dihydro-7-O-methylochnaflavone | AB | Ochn | [200] | |
| 457 | 2,3-dihydro-7′′-O-methylochnaflavone | AB | Ochn | [9] | |
| 458 | 2′′,3′′-dihydro-7′′-O-methylochnaflavone | AB | Ochn | [9] | |
| 459 | 2,2′′,3,3′′-tetrahydroochnaflavone | AA | Para | [201] | |
| 460 | 2,2′′,3,3′′-tetrahydro-7-O-methylochnaflavone | AA | Ochn, Para | [201] | |
| 461 | 2,2′′,3,3′′-tetrahydro-7,7′′-di-O-methylochnaflavone | AA | Para | [202] | |
| 462 | 3′′′-hydroxyochnaflavone | BB | Rubi | [203] | |
| 463 | 6,6′′-dimethylochnaflavone | BB | Sela | [1] | |
| 464 | 2,3-dihydro-6,6′′-dimethylochnaflavone | AB | Sela | [196] | |
| 465 | Hypnumbiflavanoid B | AA | Hypn, Ochn | [197,204,205] | |
| 3-O-7′′ | 466 | 3-O-7′′-biluteolin | BB | Aste | [206] |
| 3′-O-7′′ | 467 | Lophirone L | BB | Ochn | [52] |
| 468 | Lophirone penta-acetate | BB | Ochn | [52] | |
| 469 | 5,7,4′,5′′,3′′′,4′′′′-hexahydroxy-3′′-O-β-glucosyl-3′,7′′-O-biflavone | BB | Legu | [207] | |
| 470 | 5,7,4′,5′′,3′′′,4′′′′-hexahydroxy-3,3′′-di-O-β-glucosyl-3′,7′′-O-biflavone | BB | Vita | [208] | |
| 4-O-4′′ | 471 | Epimesquitol(4β-4)-epioritin-4β-ol | AA | Legu | [20] |
| 4′-O-4′′′ | 472 | Achyrobichalcone | GG | Aste | [209] |
| 473 | Loniflavone | BB | Capr | [1] | |
| 474 | 3′-O-methylloniflavone | BB | Capr | [210] | |
| 475 | Oniflavone | BB | Capr | [210] | |
| 476 | Ericoside | AB | Eric | [211] | |
| 4′-O-6′′ | 477 | Hinokiflavone | BB | Cupr, Psil, Sela, Taxo, Cyca | [212,213,214,215,216] |
| 478 | Isocryptomerin | BB | Cupr, Sela, Taxo | [7,213,214,215,217] | |
| 479 | Neocryptomerin | BB | Podo | [7] | |
| 480 | Cryptomerin B | BB | Taxo | [7,214,218] | |
| 481 | Chamaecyparin | BB | Cupr, Sela | [7,217,219] | |
| 482 | 2,3-dihydrohinokiflavone | AB | Cupr, Cyca | [7,215,216] | |
| 483 | (2S)-2,3-dihydroisocryptomerin | AB | Sela | [7,37] | |
| 484 | 2′′,3′′-dihydrohinokiflavone | AB | Sela | [7,220] | |
| 485 | 2′′,3′′-dihydroisocryptomerin | AB | Sela | [7,221] | |
| 486 | 7-O-methyl-2′′,3′′-dihydroisocryptomerin | AB | Sela | [221] | |
| 487 | Taiwaniaflavone B | AB | Capr | [7,79] | |
| 488 | (2S,2S’’)-2,3,2′′,3′′-tetrahydrohinokiflavone | AA | Cyca | [7,222] | |
| 489 | 7′′-O-methyl-2,3,2′′,3′′-tetrahydrohinokiflavone | AA | Sela | [7] | |
| 490 | 7,4′′-di-O-methyl-2,3,2′′,3′′-tetrahydrohinokiflavone | AA | Sela | [7] | |
| 491 | Oliveriflavone A | AA | Capr | [85] | |
| 492 | Cryptomerin A | BB | Taxo | [215,218] | |
| 493 | 2,2′′,3,3′′-tetrahydro-7,7′′-di-O-methylhinokiflavone | AA | Cyca | [223] | |
| 494 | 2,3-dihydrochamaecyparin | AB | Sela | [39] | |
| 4′-O-7′′ | 495 | Brevipedicelone E | BB | Clus | [224] |
| 4′-O-8′′ | 496 | Lanaroflavone | BB | Anac, Lana | [225,226] |
| 497 | 7-O-methyllanaroflavone | BB | Ochn | [227] | |
| 498 | 4′′′,7-di-O-methyllanaroflavone | BB | Ochn | [227] | |
| 499 | 7,7′′-di-O-methyllanaroflavone | BB | Ochn | [228] | |
| 5-O-5′′ | 500 | Potifulgene | AA | Rosa | [1] |
| 6-O-7′′ | 501 | Masazinoflavanone | AA | Anac | [229] |
| 7-O-7′′ | 502 | (myricetin-3-O-α-L-rhamnoside(C7I-O-C7II)myricetin-3-O-α-L-rhamnoside | BB | Legu | [230] |
| 6-C-8′′ | 503 | Bosistoabiflavanone | AA | Ruta | [231] |
| 8-C-8′′ | 504 | Ttrianguletin | BG | Adia | [20] |
| 505 | Pentagrametin | AB | Adia | [20] | |
| 506 | Di(8-catechinyl)methane | AA | Malv | [232] | |
| 507 | 3-O-β-D-glucopyranoside-malvidin 8-(8-ethylcatechin) | AC | Red wine | [233] | |
| 508 | 3,3′-bis-O-[β-d-xylopyranosyl-(1→2)-[4-hydroxy-3,5-dimethoxy-E-cinnamoyl-(→6)-β-d-glucopyranosyl-(1→6)]-β-d-galactopyranoside] | CC | Apia | [234] | |
| Others | 509 | Carinoside A | BE | Gent | [235] |
| 510 | Mesuferrol A | II | Clus | [236] | |
| 511 | Mesuferrol B | II | Clus | [236] |
| Subtype | No. | Compounds Name | Monomer Type | Origin (Family *) | References |
|---|---|---|---|---|---|
| C-C & C-C | 512 | Licobichalcone | GG | Legu | [237] |
| C-C & C-C-C | 513 | Selacyclicbifkavone A | AB | Sela | [7] |
| C-C & C-O-C | 514 | Licoagrodin | AD | Legu | [20] |
| 515 | Daphnodorin A | AG | Thym | [29,238] | |
| 516 | Daphnodorin B | AG | Thym | [29,238] | |
| 517 | Dihydrodaphnodorin B | AG | Thym | [239] | |
| 518 | Daphnodorin J | AG | Thym | [29,238,240] | |
| 519 | Daphnodorin E | AA | Thym | [29] | |
| 520 | Daphnodorin H | AA | Thym | [29] | |
| 521 | 4′-methoxydaphnodorin E | AA | Thym | [241] | |
| 522 | Daphnodorin F | AA | Thym | [29] | |
| 523 | Daphnodorin G | AA | Thym | [29] | |
| 524 | Lawsonia biflavone A | AB | Lyth | [230] | |
| 525 | 3′-O-methyldaphnodorin G | AA | Thym | [242] | |
| 526 | 4′′-O-methyldaphnodorin E | AA | Thym | [241] | |
| 527 | 3-O-methyldaphnodorin H | AA | Thym | [242] | |
| 528 | 3′-O-methyldaphnodorin H | AA | Thym | [242] | |
| 529 | Daphnogirin A | AA | Thym | [243] | |
| 530 | Daphnogirin B | AA | Thym | [243] | |
| 531 | Ephedrannin B | AB | Ephe, Daph | [244,245] | |
| 532 | Ephedrannin A | AB | Ephe, Daph, Vita | [244,245] | |
| 533 | Ent-epiafzelechin-(2α→7, 4α→8)-quercetin | AB | Rosa | [246] | |
| 534 | Proanthocyanidin A5′ | AA | Ephe, Rosa | [247,248] | |
| 535 | 3-O-α-L-arabinopyranosylproanthocyanidin A5′ | AA | Malv | [248] | |
| 536 | 3-O-β-D-galactopyranosylproanthocyanidin A5′ | AA | Malv | [248] | |
| 537 | Pavetannin A2 | AA | Ephe, Rosa, Rubi | [249] | |
| 538 | 3-O-α-L-arabinopyranosylpavetannin A2 | AA | Malv | [232] | |
| 539 | Ent-epicatechin-(2α→7,4α→8)-ent-catechin | AA | Rubi | [250] | |
| 540 | Ent-epicatechin-(2α→7,4α→8)-ent-epicatechin | AA | Ephe | [248] | |
| 541 | Proanthocyanidin A2 | AA | Sapi, Legu, Laur | [251,252,253] | |
| 542 | 3′-O-trans-cinnamoylproanthocyanidin A2 | AA | Legu | [254] | |
| 543 | Proanthocyanidin A1 | AA | Sapi, Legu, Laur | [253] | |
| 544 | Proanthocyanidin A4 | AA | Sapi, Rubi | [250] | |
| 545 | Baeckein F | AB | Myrt | [1] | |
| 546 | Baeckein G | AB | Myrt | [255] | |
| 547 | Baeckein H | AB | Myrt | [1] | |
| 548 | Baeckein I | AB | Myrt | [255] | |
| 549 | Lophirone C | GG | Ochn | [256,257] | |
| 550 | Dihydrolophirone C | GG | Ochn | [257] | |
| 551 | Isolophirone C | GG | Ochn | [257] | |
| 552 | Lophirone K | GG | Ochn | [258] | |
| 553 | Lophirone F | GG | Ochn | [259] | |
| 554 | Lophirone G | GG | Ochn | [259] | |
| 555 | Lophirone L‡ | GG | Ochn | [260] | |
| C-O-C & C-O-C | 556 | Dysoverine D | AB | Berb | [261] |
| 557 | Dysoverine F | AB | Berb | [261] | |
| 558 | Dysoverine A | AB | Berb | [261] | |
| 559 | (2R,2′R,3S,3′S,4α,4′α)-3′,4′,7-trihydroxyflavan-(3→O→4)(4→O→3)-3′,4′,7-trihydroxyflavan | AA | Legu | [262] |
| Subtype | No. | Compounds Name | Monomer Type | Origin (Family *) | References |
|---|---|---|---|---|---|
| Bicyclic type | 560 | Daphnodorin M | AG | Thym | [29] |
| 561 | Daphnodorin N | AG | Thym | [29] | |
| 562 | Stelleranol | AG | Thym | [1] | |
| 563 | Genkwanol B | AG | Thym | [1] | |
| 564 | Genkwanol C | AG | Thym | [1] | |
| Atom-shared type | 565 | Chamaechromone | EG | Thym | [63] |
| 566 | Mohsenone | EG | Thym | [63] | |
| 567 | Isomohsenone | EG | Thym | [63] | |
| 568 | Lophirone A | EG | Ochn | [263] | |
| 569 | Calodenone | EG | Ochn, Anac | [264] | |
| 570 | Afzelone D | EG | Ochn | [265] | |
| 571 | Campylopusaurone | AH | Clus | [266] | |
| 572 | Preussianone | AB | Clus | [267] | |
| 573 | Paucinervin K | AE | Clus | [268] | |
| 574 | Lancedatin A | BD | Legu | [20] | |
| 575 | Lancedatin B | BD | Legu | [20] | |
| Spirobiflavonoids | 576 | Absienol A | AA | Mora | [60] |
| 577 | Absienol B | AA | Pina | [1] | |
| 578 | Absienol C | AA | Mora | [60] | |
| 579 | Absienol D | AA | Pina | [1] | |
| 580 | Absienol E | AA | Mora | [60] | |
| 581 | Absienol F | AA | Pina | [1] | |
| 582 | Daphnodorin C | AA | Thym | [29] | |
| 583 | Daphnodorin I | AA | Thym | [29] | |
| 584 | Genkwanol A | AA | Thym | [269] | |
| 585 | 2′′-hydroxygenkwanol A | AA | Thym | [1] | |
| 586 | 4′-methylgenkwanol A | AA | Thym | [1] | |
| 587 | Olgensisinol A | AA | Pina | [270] | |
| 588 | Olgensisinol B | AA | Pina | [270] | |
| 589 | Olgensisinol C | AA | Pina | [270] | |
| 590 | Olgensisinol D | AA | Pina | [270] | |
| 591 | Vitisinol | AA | Pina | [270] | |
| 592 | Larixinol | AA | Pina | [271] |
| Name | Testline | Delivery Route | Doses(mg/kg) | Method | T1/2(h) | References |
|---|---|---|---|---|---|---|
| Amentoflavone | Rat plasma | i.p. | 10 | LC-MS/MS | 3.42 ± 1.45 | [320] |
| Amentoflavone | Rat plasma | i.v. | 10 | LC-MS/MS | 5.88 ± 1.78 | [320] |
| Amentoflavone | Rat plasma | p.o. | 300 | LC-MS/MS | 11.3 ± 3.61 | [320] |
| Amentoflavone | Rat plasma | p.o. | 4.31 | UFLC-MS/MS | 2.60 ± 1.34 | [322] |
| Hinokiflavone | Rat plasma | p.o. | 4.30 | UFLC-MS/MS | 2.11 ± 0.29 | [322] |
| Hinokiflavone | Rat plasma | i.v. | 1.0 | LC-MS/MS | 6.10 ± 1.86 | [321] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, X.; Yang, F.; Huang, X. Proceedings of Chemistry, Pharmacology, Pharmacokinetics and Synthesis of Biflavonoids. Molecules 2021, 26, 6088. https://doi.org/10.3390/molecules26196088
He X, Yang F, Huang X. Proceedings of Chemistry, Pharmacology, Pharmacokinetics and Synthesis of Biflavonoids. Molecules. 2021; 26(19):6088. https://doi.org/10.3390/molecules26196088
Chicago/Turabian StyleHe, Xinqian, Fan Yang, and Xin’an Huang. 2021. "Proceedings of Chemistry, Pharmacology, Pharmacokinetics and Synthesis of Biflavonoids" Molecules 26, no. 19: 6088. https://doi.org/10.3390/molecules26196088
APA StyleHe, X., Yang, F., & Huang, X. (2021). Proceedings of Chemistry, Pharmacology, Pharmacokinetics and Synthesis of Biflavonoids. Molecules, 26(19), 6088. https://doi.org/10.3390/molecules26196088






