Preparation of Hop Estrogen-Active Material for Production of Food Supplements
Abstract
:1. Introduction
2. Results and Discussion
2.1. Analysis of Starting Hop Material
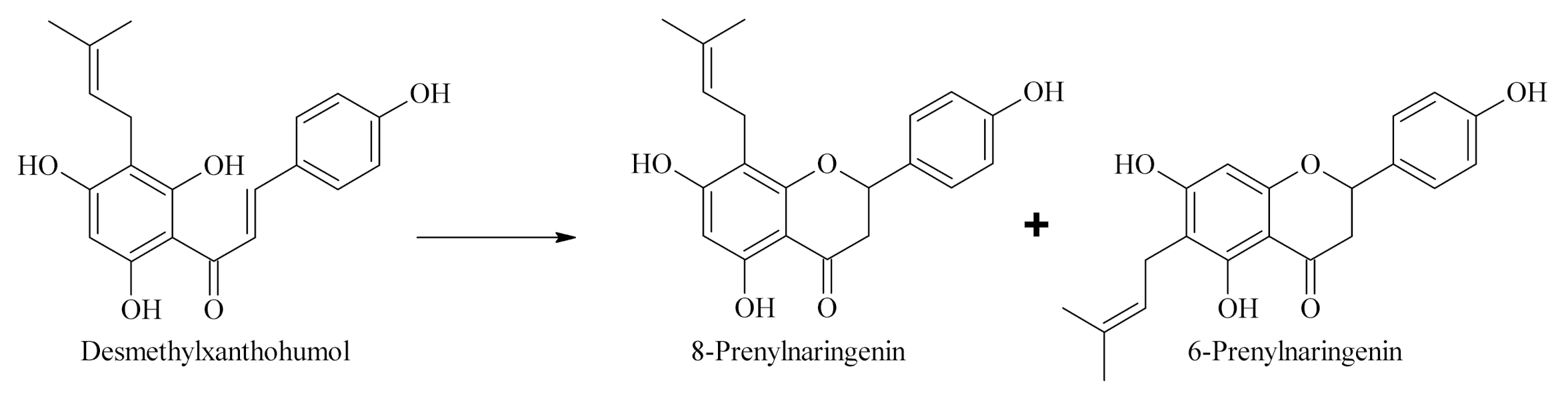
2.2. Determination of Appropriate Conditions for the Isomerization of Prenylflavonoids
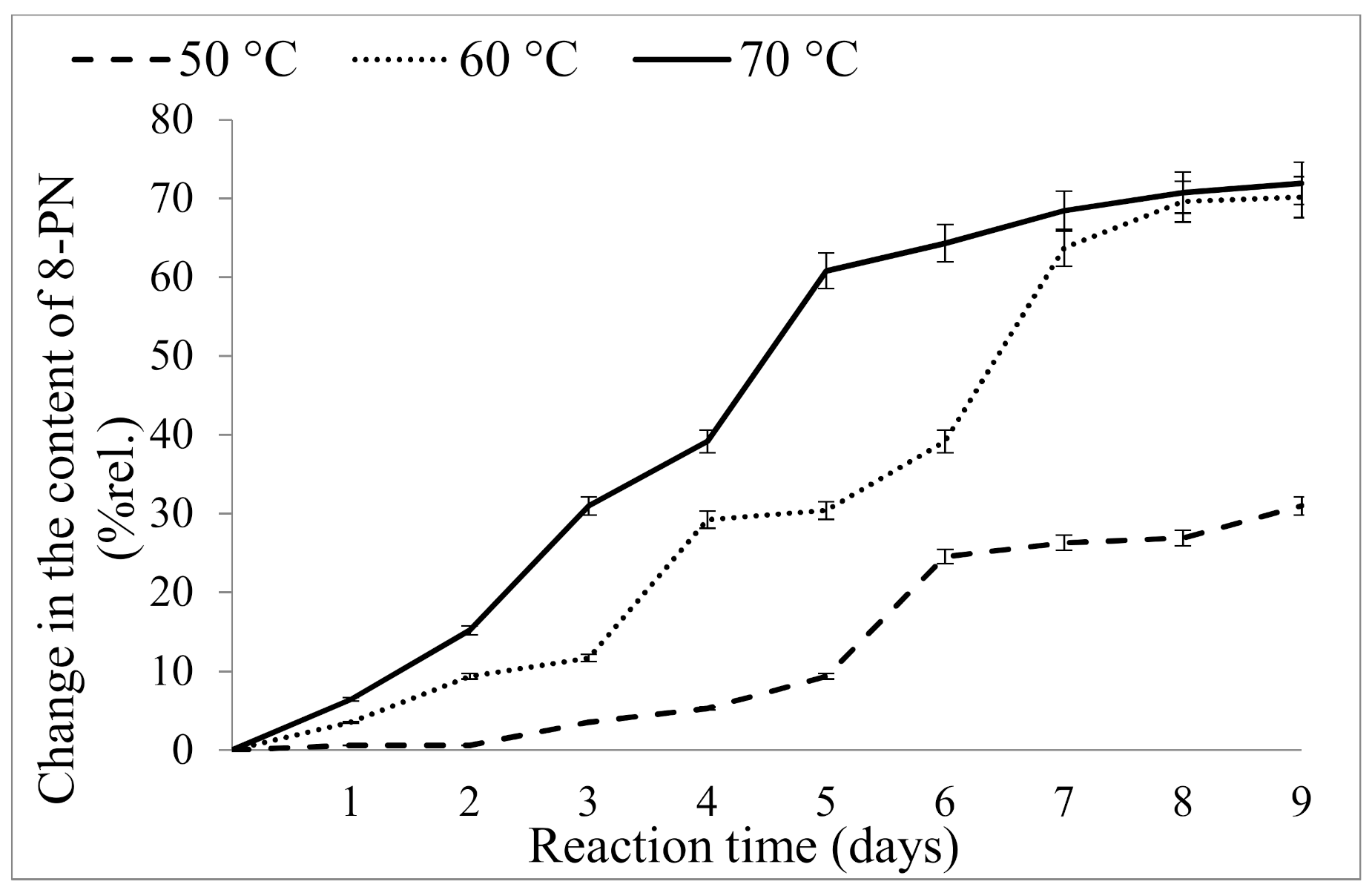
2.2.1. Influence of Temperature
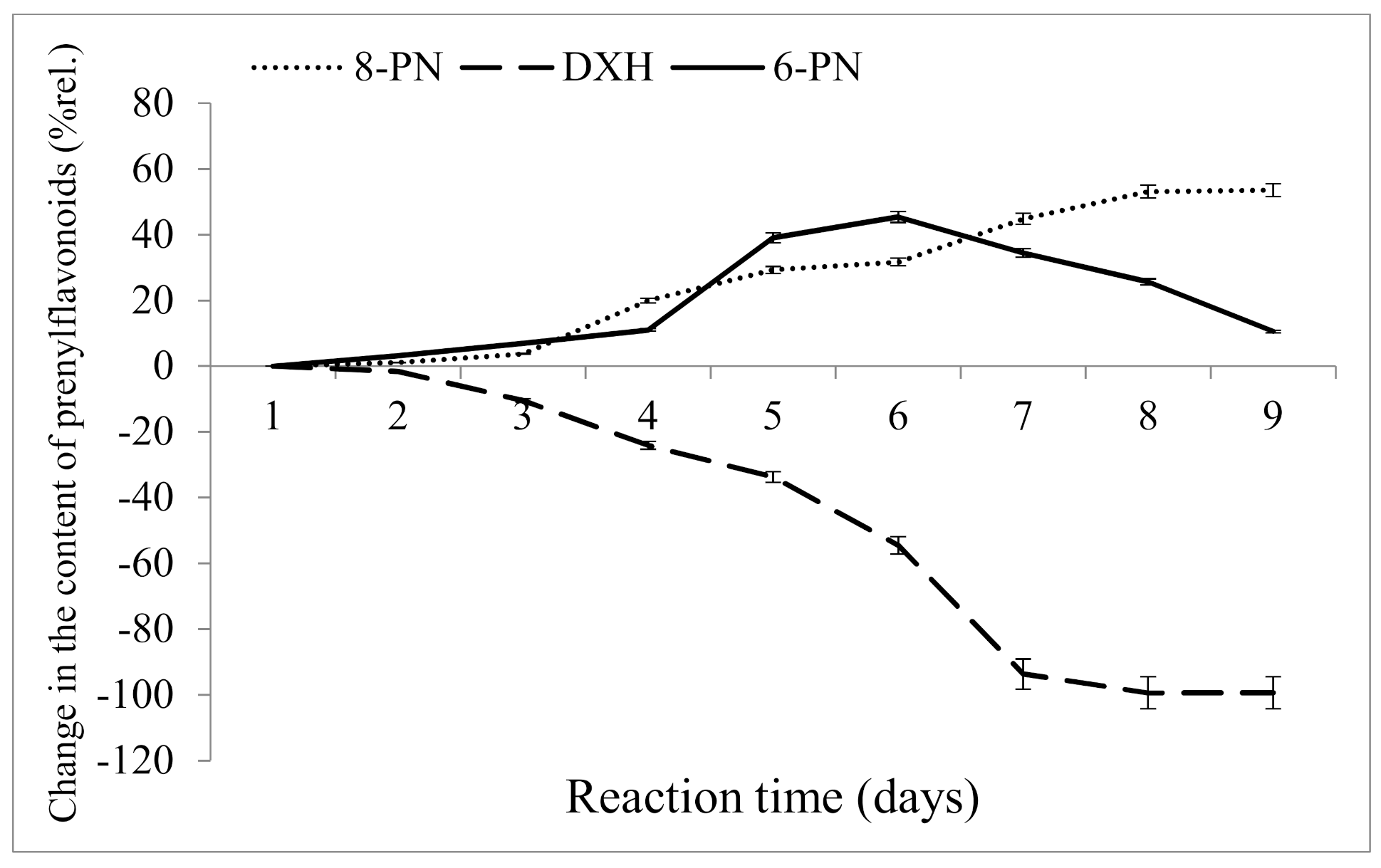
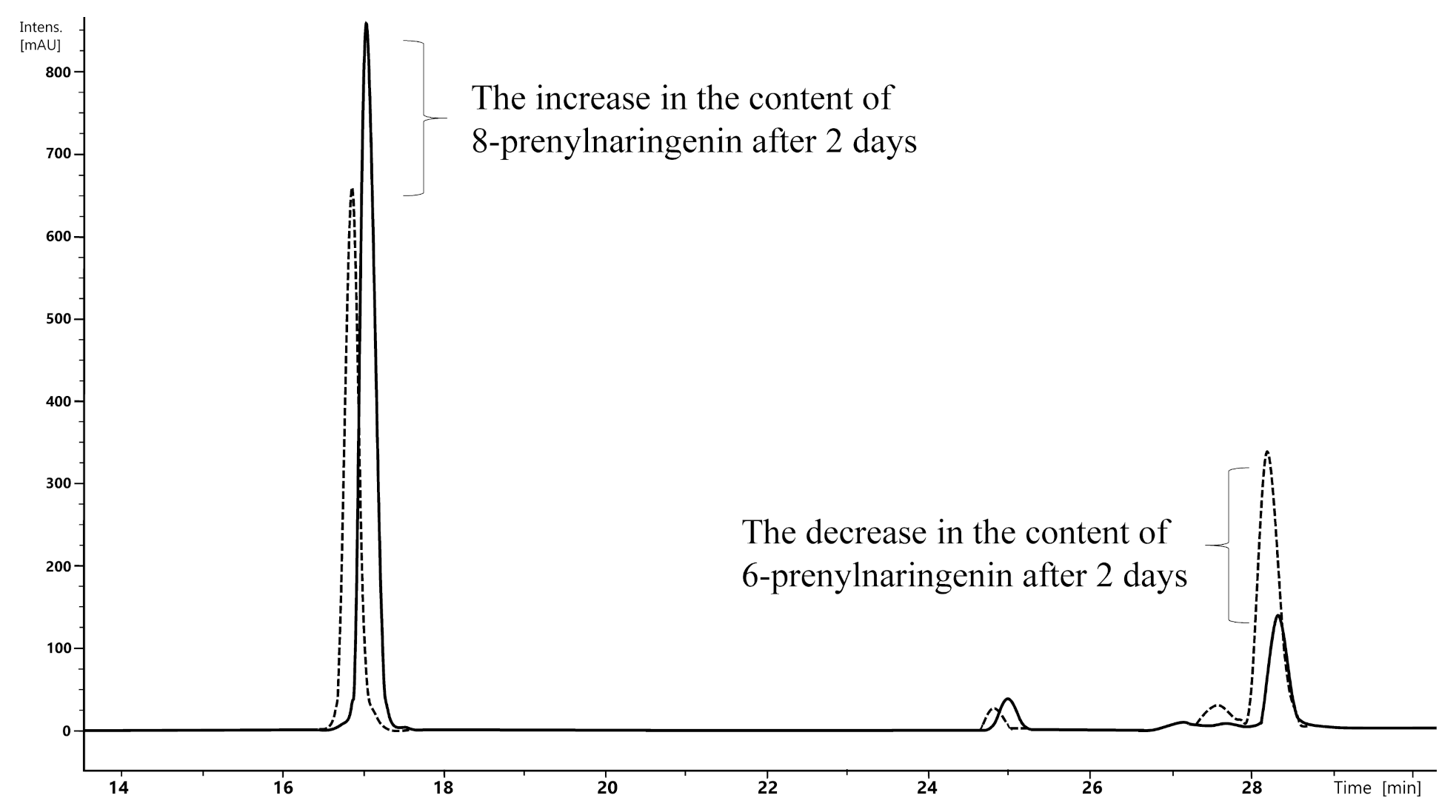
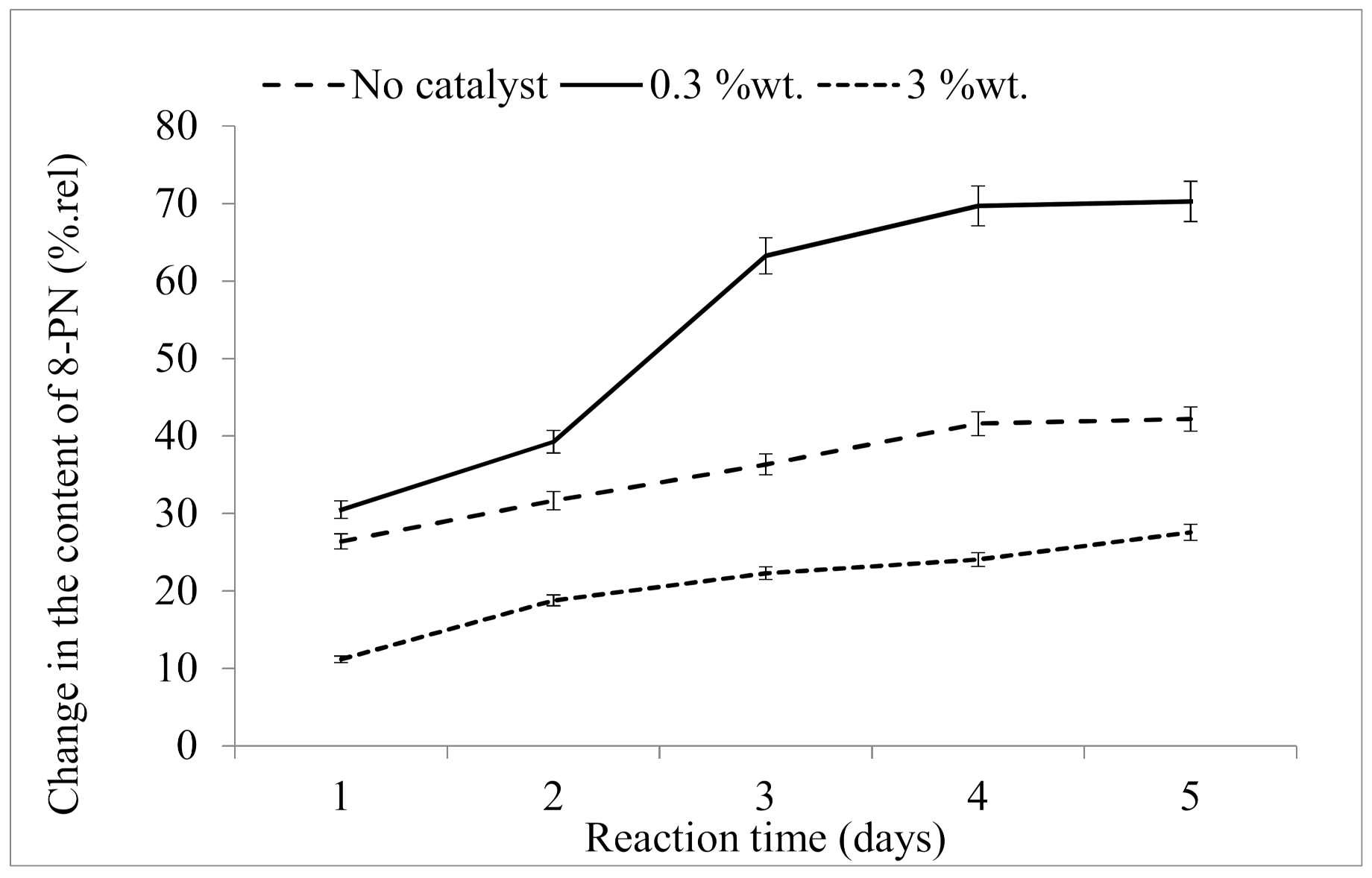
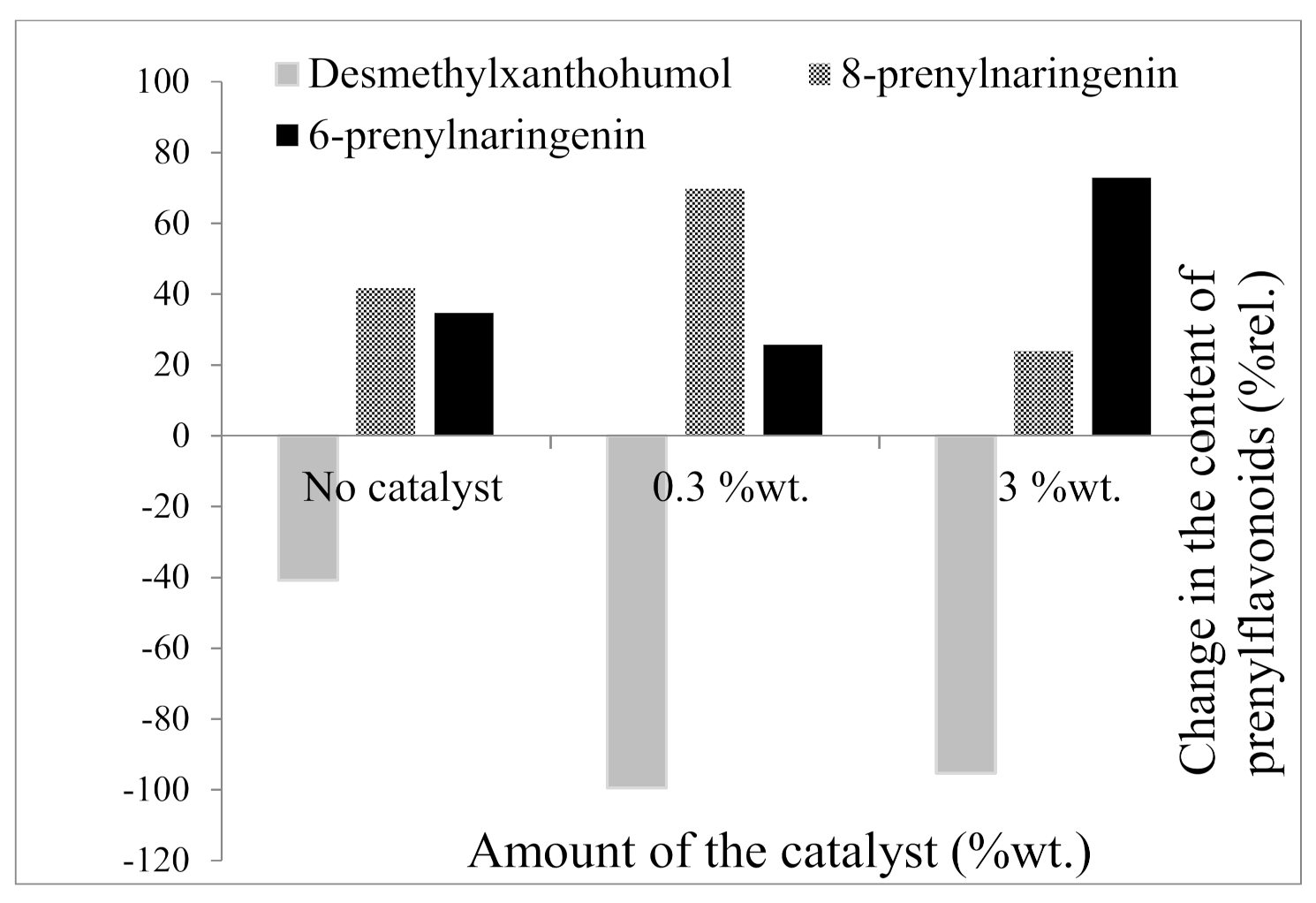
2.2.2. Influence of Reaction Time
2.2.3. Influence of Magnesium Ions
3. Materials and Methods
3.1. Chemicals
3.2. Hop Material
3.3. Isomerisation of Prenylflavonoids in Hop Material
3.4. Preparation of Samples and HPLC-PDA Analysis
3.5. HPLC-PDA Method of Analysis Validation
3.6. Statistics
4. Conclusions
- Isomerization for 8 days;
- A temperature of 60 °C;
- The use of a 0.3 %wt. magnesium oxide catalyst.
Author Contributions
Funding
Conflicts of Interest
References
- Karabín, M.; Hudcová, T.; Jelínek, L.; Dostálek, P. Biologically Active Compounds from Hops and Prospects for Their Use. Compr. Rev. Food Sci. Food Saf. 2016, 15, 542–567. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zanoli, P.; Zavatti, M. Pharmacognostic and pharmacological profile of Humulus lupulus L. J. Ethnopharmacol. 2008, 116, 383–396. [Google Scholar] [CrossRef]
- Almaguer, C.; Schonberger, C.; Gastl, M.; Arendt, E.K.; Becker, T. Humulus lupulus—A story that begs to be told. A review. J. Inst. Brew. 2014, 120, 289–314. [Google Scholar] [CrossRef]
- Krofta, K.; Vrabcova, S.; Mravcova, L.; Dostalek, P.; Karabin, M.; Jelinek, L.; Hudcova, T. Classification of Czech Hops According to their Contents of Prenylflavonoids. Kvasny Prum. 2015, 61, 62–68. [Google Scholar] [CrossRef]
- Hartkorn, A.; Hoffmann, F.; Ajamieh, H.; Vogel, S.; Heilmann, J.; Gerbes, A.L.; Vollmar, A.M.; Zahler, S. Antioxidant Effects of Xanthohumol and Functional Impact on Hepatic Ischemia-Reperfusion Injury. J. Nat. Prod. 2009, 72, 1741–1747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miranda, C.L.; Stevens, J.F.; Ivanov, V.; McCall, M.; Frei, B.; Deinzer, M.L.; Buhler, D.R. Antioxidant and prooxidant actions of prenylated and nonprenylated chalcones and flavanones in vitro. J. Agric. Food Chem. 2000, 48, 3876–3884. [Google Scholar] [CrossRef] [PubMed]
- Ambroz, M.; Lnenickova, K.; Matouskova, P.; Skalova, L.; Bousova, I. Antiproliferative Effects of Hop-derived Prenylflavonoids and Their Influence on the Efficacy of Oxaliplatine, 5-fluorouracil and Irinotecan in Human ColorectalC Cells. Nutrients 2019, 11, 879. [Google Scholar] [CrossRef] [Green Version]
- Hudcova, T.; Bryndova, J.; Fialova, K.; Fiala, J.; Karabin, M.; Jelinek, L.; Dostalek, P. Antiproliferative effects of prenylflavonoids from hops on human colon cancer cell lines. J. Inst. Brew. 2014, 120, 225–230. [Google Scholar] [CrossRef]
- Gerhauser, C.; Alt, A.; Heiss, E.; Gamal-Eldeen, A.; Klimo, K.; Knauft, J.; Neumann, I.; Scherf, H.R.; Frank, N.; Bartsch, H.; et al. Cancer chemopreventive activity of Xanthohumol, a natural product derived from hop. Mol. Cancer Ther. 2002, 1, 959–969. [Google Scholar]
- Araujo, J.R.; Goncalves, P.; Martel, F. Chemopreventive effect of dietary polyphenols in colorectal cancer cell lines. Nutr. Res. 2011, 31, 77–87. [Google Scholar] [CrossRef]
- Gerhauser, C. Broad spectrum antiinfective potential of xanthohumol from hop (Humulus lupulus L.) in comparison with activities of other hop constituents and xanthohumol metabolites. Mol. Nutr. Food Res. 2005, 49, 827–831. [Google Scholar] [CrossRef]
- Stompor, M.; Zarowska, B. Antimicrobial Activity of Xanthohumol and Its Selected Structural Analogues. Molecules 2016, 21, 608. [Google Scholar] [CrossRef]
- Peluso, M.R.; Miranda, C.L.; Hobbs, D.J.; Proteau, R.R.; Stevens, J.F. Xanthohumol and Related Prenylated Flavonoids Inhibit Inflammatory Cytokine Production in LPS-Activated THP-1 Monocytes: Structure-Activity Relationships and In Silico Binding to Myeloid Differentiation Protein-2 (MD-2). Planta Med. 2010, 76, 1536–1543. [Google Scholar] [CrossRef]
- Monteiro, R.; Calhau, C.; Silva, A.O.E.; Pinheiro-Silva, S.; Cuerreiro, S.; Gartner, F.; Azevedo, I.; Soares, R. Xanthohumol inhibits inflammatory factor production and angiogenesis in breast cancer xenografts. J. Cell. Biochem. 2008, 104, 1699–1707. [Google Scholar] [CrossRef] [PubMed]
- Stulikova, K.; Karabin, M.; Nespor, J.; Dostalek, P. Therapeutic Perspectives of 8-Prenylnaringenin, a Potent Phytoestrogen from Hops. Molecules 2018, 23, 660. [Google Scholar] [CrossRef] [Green Version]
- Milligan, S.R.; Kalita, J.C.; Heyerick, A.; Rong, H.; De Cooman, L.; De Keukeleire, D. Identification of a potent phytoestrogen in hops (Humulus lupulus L.) and beer. J. Clin. Endocrinol. Metab. 1999, 84, 2249–2252. [Google Scholar] [CrossRef] [PubMed]
- Chadwick, L.R.; Pauli, G.F.; Farnsworth, N.R. The pharmacognosy of Humulus lupulus L. (hops) with an emphasis on estrogenic properties. Phytomedicine 2006, 13, 119–131. [Google Scholar] [CrossRef] [Green Version]
- Koch, W.; Heim, G. Hormone in Hopfen und Bier. Brauwissenschaft 1953, 8, 152–153. [Google Scholar]
- Schaefer, O.; Humpel, M.; Fritzemeier, K.H.; Bohlmann, R.; Schleuning, W.D. 8-Prenyl naringenin is a potent ER alpha selective phytoestrogen present in hops and beer. J. Steroid Biochem. Mol. Biol. 2003, 84, 359–360. [Google Scholar] [CrossRef]
- Helle, J.; Kraker, K.; Bader, M.I.; Keiler, A.M.; Zierau, O.; Vollmer, G.; Welsh, J.; Kretzschmar, G. Assessment of the proliferative capacity of the flavanones 8-prenylnaringenin, 6-(1.1-dimethylallyl)naringenin and naringenin in MCF-7 cells and the rat mammary gland. Mol. Cell. Endocrinol. 2014, 392, 125–135. [Google Scholar] [CrossRef]
- Effenberger, K.E.; Westendorf, J. Hop-Derived Phytoestrogens Alter Osteoblastic Phenotype and Gene Expression. In Beer in Health and Disease Prevention; Preedy, V.R., Ed.; Elsevier Inc.: San Diego, CA, USA, 2009; pp. 735–745. [Google Scholar]
- van Duursen, M.B.M.; Smeets, E.E.J.W.; Rijk, J.C.W.; Nijmeijer, S.M.; van den Berg, M. Phytoestrogens in menopausal supplements induce ER-dependent cell proliferation and overcome breast cancer treatment in an in vitro breast cancer model. Toxicol. Appl. Pharmacol. 2013, 269, 132–140. [Google Scholar] [CrossRef]
- Monteiro, R.; Faria, A.; Azevedo, I.; Calhau, C. Modulation of breast cancer cell survival by aromatase inhibiting hop (Humulus lupulus L.) flavonoids. J. Steroid Biochem. Mol. Biol. 2007, 105, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Cos, P.; De Bruyne, T.; Apers, S.; Vanden Berghe, D.; Pieters, L.; Vlietinck, A.J. Phytoestrogens: Recent developments. Planta Med. 2003, 69, 589–599. [Google Scholar] [PubMed] [Green Version]
- Takahashi, T.A.; Johnson, K.M. Menopause. Med. Clin. N. Am. 2015, 99, 521–534. [Google Scholar] [CrossRef] [PubMed]
- Stuenkel, C.A.; Davis, S.R.; Gompel, A.; Lumsden, M.A.; Murad, M.H.; Pinkerton, J.V.; Santen, R.J. Treatment of Symptoms of the Menopause: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2015, 100, 3975–4011. [Google Scholar] [CrossRef] [PubMed]
- Lobo, R.A. Hormone-replacement therapy: Current thinking. Nat. Rev. Endocrinol. 2017, 13, 220–231. [Google Scholar] [CrossRef]
- Ming, L.G.; Lv, X.; Ma, X.N.; Ge, B.F.; Zhen, P.; Song, P.; Zhou, J.; Ma, H.P.; Xian, C.J.; Chen, K.M. The Prenyl Group Contributes to Activities of Phytoestrogen 8-Prenynaringenin in Enhancing Bone Formation and Inhibiting Bone Resorption In Vitro. Endocrinology 2013, 154, 1202–1214. [Google Scholar] [CrossRef] [Green Version]
- Tobe, H.; Muraki, Y.; Kitamura, K.; Komiyama, O.; Sato, Y.; Sugioka, T.; Maruyama, H.B.; Matsuda, E.; Nagai, M. Bone resorption inhibitors from hop extract. Biosci. Biotechnol. Biochem. 1997, 61, 158–159. [Google Scholar] [CrossRef]
- Humpel, M.; Isaksson, P.; Schaefer, O.; Kaufmann, U.; Ciana, P.; Maggi, A.; Schleuning, W.D. Tissue specificity of 8-prenylnaringenin: Protection from ovariectomy induced bone loss with minimal trophic effects on the uterus. J. Steroid Biochem. Mol. Biol. 2005, 97, 299–305. [Google Scholar] [CrossRef]
- Vázquez Loureiro, P.; Hernández Jiménez, I.; Sendón, R.; Rodriguez-Bernaldo de Quirós, A.; Barbosa-Pereira, L. Determination of Xanthohumol in Hops, Food Supplements and Beers by HPLC. Foods 2019, 8, 435. [Google Scholar] [CrossRef] [Green Version]
- Heyerick, A.; Vervarcke, S.; Depypere, H.; Bracke, M.; De Keukeleire, D. A first prospective, randomized, double-blind, placebo-controlled study on the use of a standardized hop extract to alleviate menopausal discomforts. Maturitas 2006, 54, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Milligan, S.R. Reproductive and Estrogenic Effects of 8-Prenylnaringenin in Hops. In Beer in Health and Disease Prevention; Preedy, V.R., Ed.; Elsevier Inc.: San Diego, CA, USA, 2009; pp. 711–723. [Google Scholar]
- Milligan, S.R.; Kalita, J.C.; Pocock, V.; Van de Kauter, V.; Stevens, J.F.; Deinzer, M.L.; Rong, H.; De Keukeleire, D. The endocrine activities of 8-prenylnaringenin and related hop (Humulus lupulus L.) flavonoids. J. Clin. Endocrinol. Metab. 2000, 85, 4912–4915. [Google Scholar] [CrossRef] [PubMed]
- Diller, R.A.; Riepl, H.M.; Rose, O.; Frias, C.; Henze, G.; Prokop, A. Desmethylxanthohumol from Hops, Chemistry and Biological Effects. In Beer in Health and Disease Prevention; Preedy, V.R., Ed.; Elsevier Inc.: San Diego, CA, USA, 2009; pp. 703–709. [Google Scholar]
- Stevens, J.F.; Taylor, A.W.; Clawson, J.E.; Deinzer, M.L. Fate of xanthohumol and related prenylflavonoids from hops to beer. J. Agric. Food Chem. 1999, 47, 2421–2428. [Google Scholar] [CrossRef]
- Nesvadba, V.; Krofta, K.; Polončíková, Z. Hop (Humulus lupulus L.) breeding aimed at high contents of desmethylxanthohumol (DMX). Agriculture (Poľnohospodárstvo) 2011, 57, 105–109. [Google Scholar] [CrossRef] [Green Version]
- Possemiers, S.; Verstraete, W.; Heyerick, A.; De Keukeleire, D. Enzymatic Demethylation of Flavonoids. WO2006099699A1, 28 September 2006. [Google Scholar]
- Possemiers, S.; Heyerick, A.; Robbens, V.; De Keukeleire, D.; Verstraete, W. Activation of proestrogens from hops (Humulus lupulus L.) by intestinal microbiota; Conversion of isoxanthohumol into 8-prenylnaringenin. J. Agric. Food Chem. 2005, 53, 6281–6288. [Google Scholar] [CrossRef] [Green Version]
- Fu, M.L.; Wang, W.; Chen, F.; Dong, Y.C.; Liu, X.J.; Ni, H.; Chen, Q.H. Production of 8-prenylnaringenin from isoxanthohumol through biotransformation by fungi cells. J. Agric. Food Chem. 2011, 59, 7419–7426. [Google Scholar] [CrossRef]
- Aniol, M.; Szymanska, K.; Zolnierczyk, A. An effcient synthesis of the phytoestrogen 8-prenylnaringenin from isoxanthohumol with magnesium iodide etherate. Tetrahedron 2008, 64, 9544–9547. [Google Scholar] [CrossRef]
- Urmann, C.; Riepl, H. Semi-Synthetic Approach Leading to 8-Prenylnaringenin and 6-Prenylnaringenin: Optimization of the Microwave-Assisted Demethylation of Xanthohumol Using Design of Experiments. Molecules 2020, 25, 4007. [Google Scholar] [CrossRef]
- Wilhelm, H.; Wessjohann, L.A. An efficient synthesis of the phytoestrogen 8-prenylnaringenin from xanthohumol by a novel demethylation process. Tetrahedron 2006, 62, 6961–6966. [Google Scholar] [CrossRef]
- Federal Institute of Occupational Safety and Health. Classification of Prokaryotes (Bacteria and Archaea) into Risk Groups. Available online: https://www.baua.de/EN/Service/Legislative-texts-and-technical-rules/Rules/TRBA/pdf/TRBA-466.pdf?__blob=publicationFile&v=2 (accessed on 26 November 2020).
- Rong, H.; Zhao, Y.; Lazou, K.; De Keukeleire, D.; Milligan, S.R.; Sandra, P. Quantitation of 8-prenylnaringenin, a novel phytoestrogen in hops (Humulus lupulus L.), hop products, and beers, by benchtop HPLC-MS using electrospray ionization. Chromatographia 2000, 51, 545–552. [Google Scholar] [CrossRef]
- Jelinek, L.; Karabin, M.; Kotlikova, B.; Hudcova, T.; Dostalek, P. Application of a hop by-product in brewing: Reduction in the level of haze-active prolamines and improved antioxidant properties of the beer. J. Inst. Brew. 2014, 120, 99–104. [Google Scholar] [CrossRef]
- Intelmann, D.; Batram, C.; Kuhn, C.; Haseleu, G.; Meyerhof, W.; Hofmann, T. Three TAS2R Bitter Taste Receptors Mediate the Psychophysical Responses to Bitter Compounds of Hops (Humulus lupulus L.) and Beer. Chemosens. Percept. 2009, 2, 118–132. [Google Scholar] [CrossRef]
- Krofta, K.; Patzak, J.; Nesvadba, V.; Mikyska, A.; Slaby, M.; Cejka, P. VITAL-The Czech hop hybrid variety-Part I. Kvasny Prum. 2013, 59, 2–13. [Google Scholar] [CrossRef]
- Kostrzewa, D.; Dobrzynska-Inger, A.; Roj, E.; Grzeda, K.; Kozlowski, K. Isomerization of hop extract alpha-acids. J. Inst. Brew. 2016, 122, 493–499. [Google Scholar] [CrossRef] [Green Version]
- Maes, F.; De Keukeleire, D.; Heyerick, A. Production of Hop Extracts Having Oestrogenic and Antiproliferative Bioactivity. US20110130447A1, 2 June 2011. [Google Scholar]
- Magalhaes, P.J.; Carvalho, D.O.; Cruz, J.M.; Guido, L.F.; Barros, A.A. Fundamentals and Health Benefits of Xanthohumol, a Natural Product Derived from Hops and Beer. Nat. Prod. Commun. 2009, 4, 591–610. [Google Scholar] [CrossRef] [Green Version]
- Karabin, M.; Jelinek, L.; Kincl, T.; Hudcova, T.; Kotlikova, B.; Dostalek, P. New approach to the production of xanthohumol-enriched beers. J. Inst. Brew. 2013, 119, 98–102. [Google Scholar] [CrossRef]
- Jaskula, B.; Kafarski, P.; Aerts, G.; De Cooman, L. A kinetic study on the isomerization of hop alpha-acids. J. Agric. Food Chem. 2008, 56, 6408–6415. [Google Scholar] [CrossRef]
- Burkhardt, R.J.; Wilson, R.J. Process for the Preparation of Isomerized Hop Pellets. US4946691A, 7 August 1990. [Google Scholar]
- American Society of Brewing Chemists. ASBC Methods of Analysis, Hops 14. α-Acids and β-Acids in Hops and Hop Extracts by HPLC (International Method). Available online: http://methods.asbcnet.org/methods/hops-14.pdf (accessed on 26 November 2020).
- Dostalek, P.; Hudcova, T.; Jelinek, L.; Fialova, K.; Karabin, M. Method for Preparing Hop Material with Increased Content 8-Prenylnaringenin. CZ 307451 B6 20180905, 5 September 2018. [Google Scholar]
| Prenylflavonoid | (mg/100 gDW) |
|---|---|
| Desmethylxanthohumol | 97.4 |
| 8-Prenylnaringenin | 17.1 |
| 6-Prenylnaringenin | 46.7 |
| Xanthohumol | 920.2 |
| Isoxanthohumol | 29.4 |
| α-Bitter acids | 1004 |
| β-Bitter acids | n.d |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karabín, M.; Haimannová, T.; Fialová, K.; Jelínek, L.; Dostálek, P. Preparation of Hop Estrogen-Active Material for Production of Food Supplements. Molecules 2021, 26, 6065. https://doi.org/10.3390/molecules26196065
Karabín M, Haimannová T, Fialová K, Jelínek L, Dostálek P. Preparation of Hop Estrogen-Active Material for Production of Food Supplements. Molecules. 2021; 26(19):6065. https://doi.org/10.3390/molecules26196065
Chicago/Turabian StyleKarabín, Marcel, Tereza Haimannová, Kristýna Fialová, Lukáš Jelínek, and Pavel Dostálek. 2021. "Preparation of Hop Estrogen-Active Material for Production of Food Supplements" Molecules 26, no. 19: 6065. https://doi.org/10.3390/molecules26196065
APA StyleKarabín, M., Haimannová, T., Fialová, K., Jelínek, L., & Dostálek, P. (2021). Preparation of Hop Estrogen-Active Material for Production of Food Supplements. Molecules, 26(19), 6065. https://doi.org/10.3390/molecules26196065







