Anticancer Effects of the Corchorus olitorius Aqueous Extract and Its Bioactive Compounds on Human Cancer Cell Lines
Abstract
:1. Introduction
2. Results
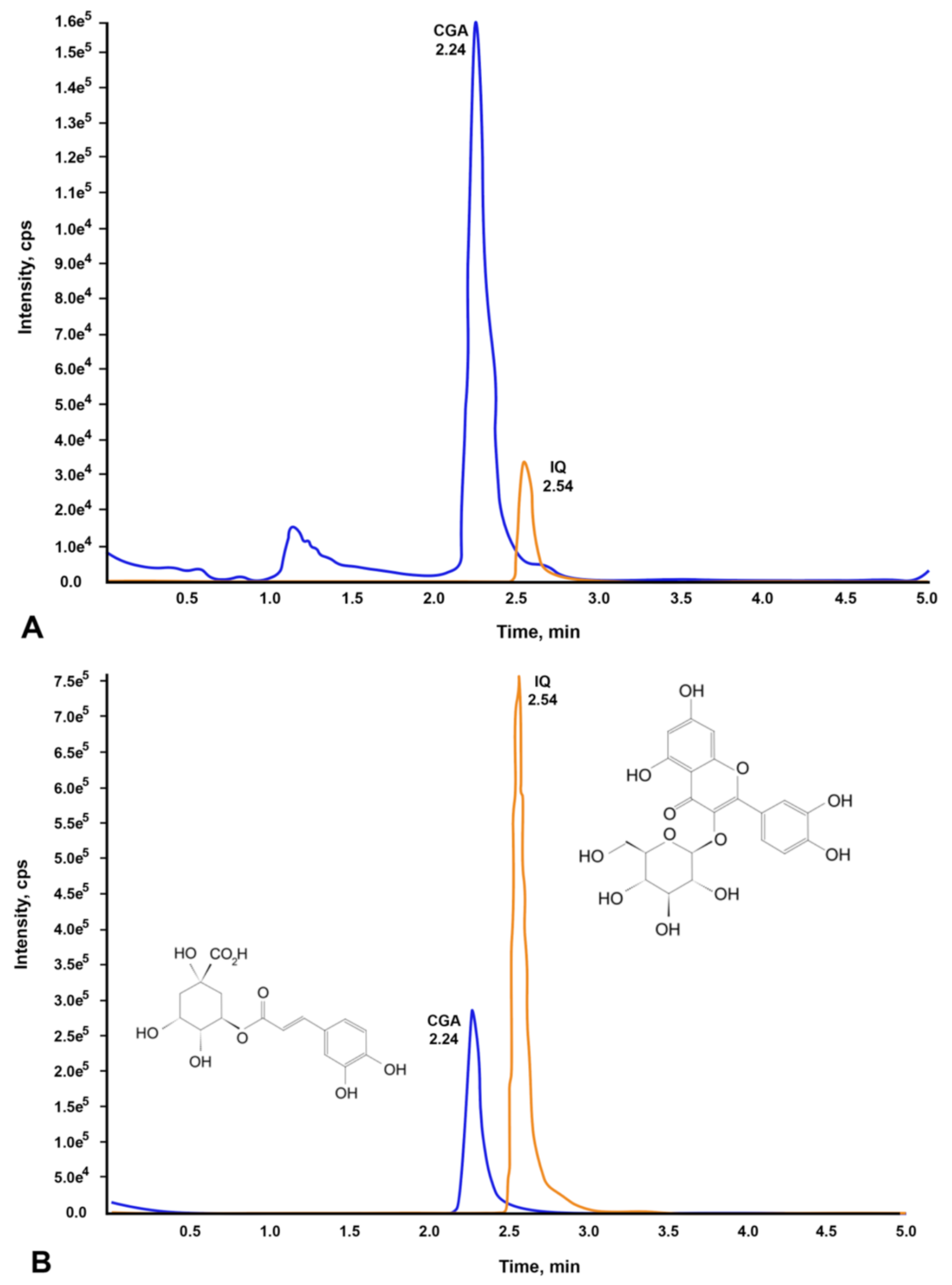
2.1. Identification and Quantification of Chlorogenic Acid and Isoquercetin
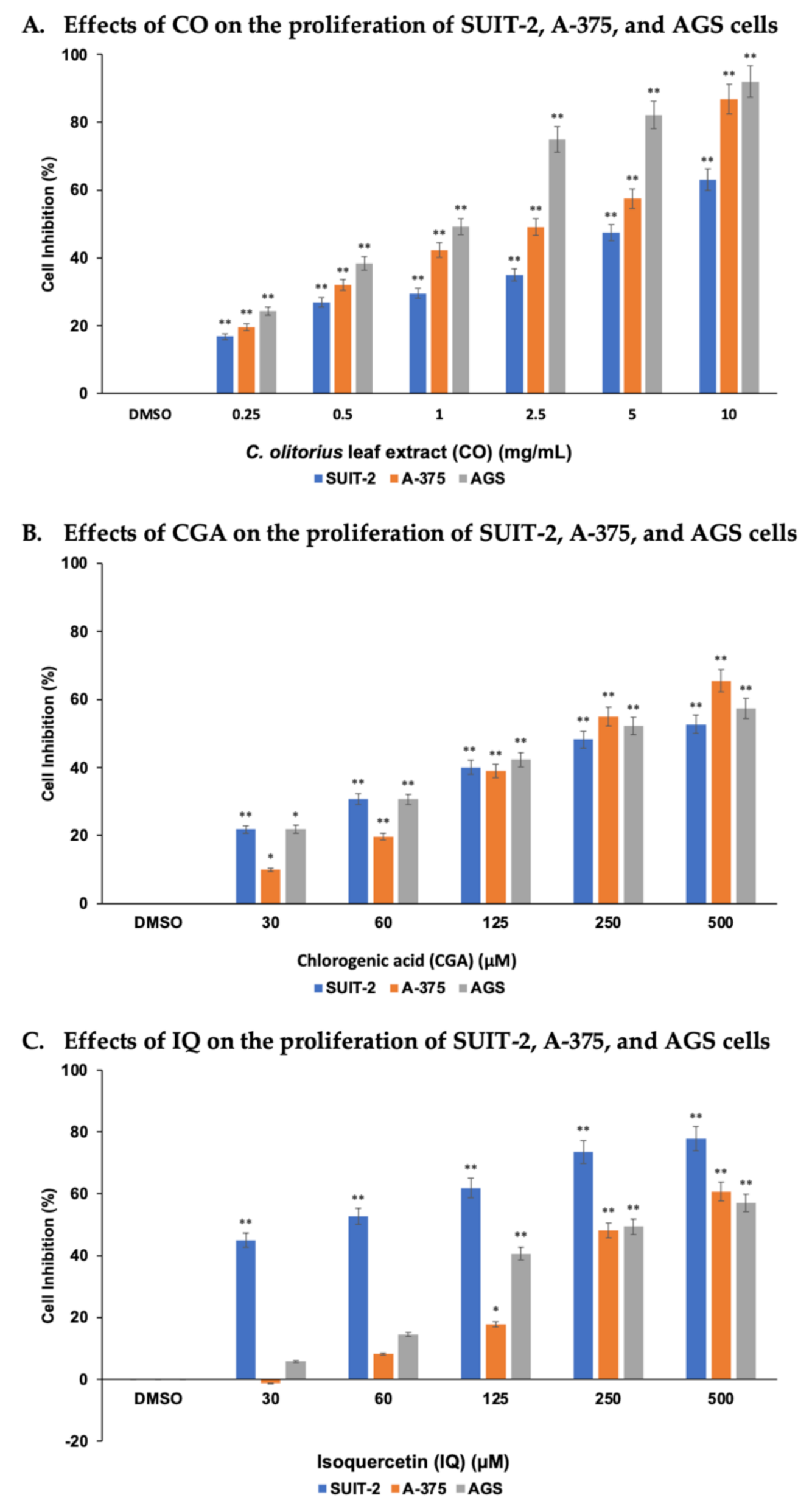
2.2. CO, CGA, and IQ Inhibited Human Cancer Cells’ Proliferation
2.3. CO, CGA, and IQ Inhibit Angiogenesis and Tumor Growth in Ovo
3. Discussion
4. Materials and Methods
4.1. Plant Collection and Authentication
4.2. Plant Preparation
4.3. Plant Extraction
4.4. Solid-Phase Extraction
4.5. LC-MS/MS Analysis
4.6. Cell Culture
4.7. 3-(4, 5-Dimethyl thiazolyl-2)-2, 5-Diphenyltetrazolium Bromide (MTT) Assay
4.8. Tumor Growth and Angiogenesis in the Chorioallantoic Membrane Model
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Jubb, A.M.; Oates, A.J.; Holden, S.; Koeppen, H. Predicting benefit from anti-angiogenic agents in malignancy. Nat. Rev. Cancer 2006, 6, 626–635. [Google Scholar] [CrossRef] [PubMed]
- Kerbel, R.; Folkman, J. Clinical translation of angiogenesis inhibitors. Nat. Rev. Cancer 2002, 2, 727–739. [Google Scholar] [CrossRef] [PubMed]
- Kunimasa, K.; Ohta, T.; Tani, H.; Kato, E.; Eguchi, R.; Kaji, K.; Ikeda, K.; Mori, H.; Mori, M.; Tatefuji, T.; et al. Resveratrol derivative-rich melinjo (Gnetum gnemon L.) seed extract suppresses multiple angiogenesis-related endothelial cell functions and tumor angiogenesis. Mol. Nutr. Food Res. 2011, 55, 1730–1734. [Google Scholar] [CrossRef]
- Lam, I.K.; Alex, D.; Wang, Y.-H.; Liu, P.; Liu, A.-L.; Du, G.-H.; Lee, S.M. In vitro and in vivo structure and activity relationship analysis of polymethoxylated flavonoids: Identifying sinensetin as a novel antiangiogenesis agent. Mol. Nutr. Food Res. 2012, 56, 945–956. [Google Scholar] [CrossRef]
- Piao, X.-M.; Gao, F.; Zhu, J.-X.; Wang, L.-J.; Zhao, X.; Li, X.; Sheng, M.-M.; Zhang, Y. Cucurbitacin B inhibits tumor angiogenesis by triggering the mitochondrial signaling pathway in endothelial cells. Int. J. Mol. Med. 2018, 42, 1018–1025. [Google Scholar] [CrossRef] [Green Version]
- Tabata, Y. Tissue regeneration based on growth factor release. Tissue Eng. 2003, 9, 5–15. [Google Scholar] [CrossRef]
- Zhu, L.; Niu, G.; Fang, X.; Chen, X. Preclinical molecular imaging of tumor angiogenesis. Q. J. Nucl. Med. Mol. Imaging 2010, 54, 291–308. [Google Scholar] [PubMed]
- Kerbel, R.S. Tumor angiogenesis. N. Engl. J. Med. 2008, 358, 2039–2049. [Google Scholar] [CrossRef] [Green Version]
- Verheul, H.; Voest, E.; Schlingemann, R.O. Are tumours angiogenesis-dependent? J. Pathol. 2004, 202, 5–13. [Google Scholar] [CrossRef]
- Shibuya, M. Structure and function of VEGF/VEGF-receptor system involved in angiogenesis. Cell Struct. Funct. 2001, 26, 25–35. [Google Scholar] [CrossRef] [Green Version]
- Ferrara, N. Vascular endothelial growth factor: Basic science and clinical progress. Endocr. Rev. 2004, 25, 581–611. [Google Scholar] [CrossRef]
- DOST—Department of Science and Technology. Harmonized National R&D Agenda (HNRDA) 2017–2022. Available online: http://dost.gov.ph/phocadownload/Downloads/Journals/Approved%20Harmonized%20National%20RD%20Agenda%20%202017-2022.pdf (accessed on 10 October 2018).
- Oboh, G.; Ademiluyi, A.O.; Akinyemi, A.; Henle, T.; Saliu, J.A.; Schwarzenbolz, U. Inhibitory effect of polyphenol-rich extracts of jute leaf (Corchorus olitorius) on key enzyme linked to type 2 diabetes (α-amylase and α-glucosidase) and hypertension (angiotensin I converting) in vitro. J. Funct. Foods 2012, 4, 450–458. [Google Scholar] [CrossRef]
- Zakaria, Z.; Sulaiman, M.; Arifah, A.; Jais, A.M.; Somchit, M.; Kirisnaven, K.; Punnitharr, D.; Safarul, M.; Fatimah, C.; Johari, R. The anti-inflammatory and antipyretic activities of corchorus olitorius in rats. J. Pharmacol. Toxicol. 2006, 1, 139–146. [Google Scholar] [CrossRef] [Green Version]
- Soladoye, M.O.; Chukwuma, E.C.; Feyisola, R.T. Ethnobotanical survey of plants used in the traditional treatment of female infertility in Southwestern Nigeria. Ethnobot. Res. Appl. 2014, 12, 81–90. [Google Scholar]
- Barku, V.Y.A.; Boye, A.; Quansah, N. Antioxidant and wound healing studies on the extracts of Corchorus olitorius leaf. World Essay J. 2013, 1, 67–73. [Google Scholar]
- Owoyele, B.; Oyewole, A.L.; Alimi, M.L.; Sanni, S.A.; Oyeleke, S.A. Anti-inflammatory and antipyretic properties of Corchorus olitorius aqueous root extract in Wistar rats. J. Basic Clin. Physiol. Pharmacol. 2015, 26, 363–368. [Google Scholar] [CrossRef]
- Abdel-Wahhab, M.A.; Ibrahim, M.I.M.; Pieters, R.; van der Walt, A.M.; Abdel-Aziem, S.H.; Bezuidenhout, C.C.; Giesy, J.P. Aqueous extract of Corchorus olitorius decreases cytotoxicity of aflatoxin B1and fumonisin B1in H4IIE-luc cells. Hepatoma Res. 2015, 1, 75–85. [Google Scholar] [CrossRef] [Green Version]
- Appleton, J. Evaluating the bioavailability of isoquercetin. Nat. Med. J. 2010, 2, 1–6. [Google Scholar]
- Yoshikawa, M.; Shimada, H.; Saka, M.; Yoshizumi, S.; Yamahara, J.; Matsuda, H. Medicinal foodstuffs. V. Moroheiya (1): Absolute stereostructures of corchoionosides A, B, and C, histamine inhibitors from the leaves of Vietnamese Corchorus olitorius L. (Tiliaceae). Chem. Pharm. Bull. 1997, 45, 464–469. [Google Scholar] [CrossRef] [Green Version]
- Nardini, M.; Cirillo, E.; Natella, F.; Scaccini, C. Absorption of phenolic acids in humans after coffee consumption. J. Agric. Food Chem. 2002, 50, 5735–5741. [Google Scholar] [CrossRef]
- Liang, N.; Kitts, D.D. Role of chlorogenic acids in controlling oxidative and inflammatory stress conditions. Nutrients 2015, 8, 16. [Google Scholar] [CrossRef] [Green Version]
- Jiang, Y.; Satoh, K.; Kusama, K.; Watanabe, S.; Sakagami, H. Interaction between chlorogenic acid and antioxidants. Anticancer Res. 2000, 20, 2473–2476. [Google Scholar] [PubMed]
- Yagasaki, K.; Miura, Y.; Okauchi, R.; Furuse, T. Inhibitory effects of chlorogenic acid and its related compounds on the invasion of hepatoma cells in culture. Cytotechnology 2000, 33, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Belkaid, A.; Currie, J.-C.; Desgagnés, J.; Annabi, B. The chemopreventive properties of chlorogenic acid reveal a potential new role for the microsomal glucose-6-phosphate translocase in brain tumor progression. Cancer Cell Int. 2006, 6, 7. [Google Scholar] [CrossRef] [Green Version]
- Noratto, G.; Porter, W.; Byrne, D.; Cisneros-Zevallos, L. Identifying peach and plum polyphenols with chemopreventive potential against estrogen-independent breast cancer cells. J. Agric. Food Chem. 2009, 57, 5219–5226. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-J.; Zhou, C.-Y.; Qiu, C.-H.; Lu, X.-M.; Wang, Y.-T. Chlorogenic acid induced apoptosis and inhibition of proliferation in human acute promyelocytic leukemia HL-60 cells. Mol. Med. Rep. 2013, 8, 1106–1110. [Google Scholar] [CrossRef] [Green Version]
- Nishimura, J.; Saegusa, Y.; Dewa, Y.; Jin, M.; Kawai, M.; Kemmochi, S.; Harada, T.; Hayashi, S.M.; Shibutani, M.; Mitsumori, K. Antioxidant enzymatically modified isoquercitrin or melatonin supplementation reduces oxidative stress-mediated hepato-cellular tumor promotion of oxfendazole in rats. Arch. Toxicol. 2010, 84, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Emura, K.; Yokomizo, A.; Toyoshi, T.; Moriwaki, M. Effect of enzymatically modified isoquercitrin in spontaneously hyper-tensive rats. J. Nutr. Sci. Vitaminol. 2007, 53, 68–74. [Google Scholar] [CrossRef] [Green Version]
- De Araújo, M.E.M.B.; Franco, Y.E.M.; Alberto, T.G.; Sobreiro, M.A.; Conrado, M.A.V.; Priolli, D.G.; Sawaya, A.; Ruiz, A.L.T.G.; de Carvalho, J.E.; de Carvalho, P. Enzymatic de-glycosylation of rutin improves its antioxidant and antiproliferative activities. Food Chem. 2013, 141, 266–273. [Google Scholar] [CrossRef] [Green Version]
- You, H.J.; Ahn, H.J.; Ji, G.E. Transformation of rutin to antiproliferative quercetin-3-glucoside by aspergillus niger. J. Agric. Food Chem. 2010, 58, 10886–10892. [Google Scholar] [CrossRef] [PubMed]
- Jones, D. Parallels of resistance between angiogenesis and lymphangiogenesis inhibition in cancer therapy. Cells 2020, 9, 762. [Google Scholar] [CrossRef] [Green Version]
- NCI—National Cancer Institute. Metastasis. Available online: https://www.cancer.gov/publications/dictionaries/cancer-terms/def/metastasis (accessed on 10 October 2018).
- Loumerem, M.; Alercia, A. Descriptors for jute (Corchorus olitorius L.). Genet. Resour. Crop Evol. 2016, 63, 1103–1111. [Google Scholar] [CrossRef]
- Handoussa, H.; Hanafi, R.; Eddiasty, I.; El-Gendy, M.; El Khatib, A.; Linscheid, M.; Mahran, L.; Ayoub, N. Anti-inflammatory and cytotoxic activities of dietary phenolics isolated from corchorus olitorius and vitis vinifera. J. Funct. Foods 2013, 5, 1204–1216. [Google Scholar] [CrossRef]
- Li, C.-J.; Huang, S.-Y.; Wu, M.-Y.; Chen, Y.-C.; Tsang, S.-F.; Chyuan, J.-H.; Hsu, H.-Y. Induction of apoptosis by ethanolic extract of corchorus olitorius leaf in human hepatocellular carcinoma (HepG2) cells via a mitochondria-dependent pathway. Molecules 2012, 17, 9348–9360. [Google Scholar] [CrossRef] [Green Version]
- Işeri, D.; Yurtcu, E.; Sahin, F.I.; Haberal, M. Corchorus olitorius(jute) extract induced cytotoxicity and genotoxicity on human multiple myeloma cells (ARH-77). Pharm. Biol. 2013, 51, 766–770. [Google Scholar] [CrossRef]
- Deka, S.J.; Gorai, S.; Manna, D.; Trivedi, V. Evidence of PKC binding and translocation to explain the anticancer mechanism of chlorogenic acid in breast cancer cells. Curr. Mol. Med. 2017, 17, 79–89. [Google Scholar] [CrossRef]
- Sarkate, A.; Banerjee, S.; Mir, J.I.; Roy, P.; Sircar, D. Antioxidant and cytotoxic activity of bioactive phenolic metabolites isolated from the yeast-extract treated cell culture of apple. Plant Cell Tissue Organ Cult. 2017, 130, 641–649. [Google Scholar] [CrossRef]
- Wang, X.; Liu, J.; Xie, Z.; Rao, J.; Xu, G.; Huang, K.; Li, W.; Yin, Z. Chlorogenic acid inhibits proliferation and induces apoptosis in A498 human kidney cancer cells via inactivating PI3K/Akt/mTOR signalling pathway. J. Pharm. Pharmacol. 2019, 71, 1100–1109. [Google Scholar] [CrossRef]
- Zhan, Y.; Li, R.; Feng, C.; Li, X.; Huang, S.; Wang, L.; Liu, Z.; Jiang, J.; Han, Y. Chlorogenic acid inhibits esophageal squamous cell carcinoma growth in vitro and in vivo by downregulating the expression of BMI1 and SOX. Biomed. Pharmacother. 2020, 121, 109602. [Google Scholar] [CrossRef] [PubMed]
- Yamagata, K.; Izawa, Y.; Onodera, D.; Tagami, M. Chlorogenic acid regulates apoptosis and stem cell marker-related gene expression in A549 human lung cancer cells. Mol. Cell. Biochem. 2018, 441, 9–19. [Google Scholar] [CrossRef]
- Weng, C.-J.; Yen, G.-C. Chemopreventive effects of dietary phytochemicals against cancer invasion and metastasis: Phenolic acids, monophenol, polyphenol, and their derivatives. Cancer Treat. Rev. 2012, 38, 76–87. [Google Scholar] [CrossRef] [PubMed]
- Hara, S.; Morita, R.; Ogawa, T.; Segawa, R.; Takimoto, N.; Suzuki, K.; Hamadate, N.; Hayashi, S.-M.; Odachi, A.; Ogiwara, I.; et al. Tumor suppression effects of bilberry extracts and enzymatically modified isoquercitrin in early preneoplastic liver cell lesions induced by piperonyl butoxide promotion in a two-stage rat hepatocarcinogenesis model. Exp. Toxicol. Pathol. 2014, 66, 225–234. [Google Scholar] [CrossRef]
- Chen, Q.; Li, P.; Xu, Y.; Li, Y.; Tang, B. Isoquercitrin inhibits the progression of pancreatic cancer in vivo and in vitro by regulating opioid receptors and the mitogen-activated protein kinase signalling pathway. Oncol. Rep. 2015, 33, 840–848. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, G.; Tang, B.; Tang, K.; Dong, X.; Deng, J.; Liao, L.; Liao, Z.; Yang, H.; He, S. Isoquercitrin inhibits the progression of liver cancer in vivo and in vitro via the MAPK signalling pathway. Oncol. Rep. 2014, 31, 2377–2384. [Google Scholar] [CrossRef] [Green Version]
- Siddiqui, I.A.; Malik, A.; Adhami, V.M.; Asim, M.; Hafeez, B.B.; Sarfaraz, S.; Mukhtar, H. Green tea polyphenol EGCG sensitizes human prostate carcinoma LNCaP cells to TRAIL-mediated apoptosis and synergistically inhibits biomarkers associated with angiogenesis and metastasis. Oncogene 2008, 27, 2055–2063. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sudha, T.; Yalcin, M.; Lin, H.-Y.; Elmetwally, A.M.; Nazeer, T.; Arumugam, T.; Phillips, P.; Mousa, S.A. Suppression of pancreatic cancer by sulfated non-anticoagulant low molecular weight heparin. Cancer Lett. 2014, 350, 25–33. [Google Scholar] [CrossRef] [Green Version]

| Treatment | IC50 Values | ||
|---|---|---|---|
| SUIT-2 | A-375 | AGS | |
| CO (mg/mL) | 6.47 ± 0.11 ## | 4.05 ± 0.09 ** | 2.54 ± 0.09 *** |
| CGA (µM) | 373.53 ± 11.29 ns | 306.64 ± 20.83 ns | 330.74 ± 13.19 ns |
| IQ (µM) | 144.45 ± 12.34 # | 369.29 ± 23.78 * | 355.97 ± 19.93 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tosoc, J.P.S.; Nuñeza, O.M.; Sudha, T.; Darwish, N.H.E.; Mousa, S.A. Anticancer Effects of the Corchorus olitorius Aqueous Extract and Its Bioactive Compounds on Human Cancer Cell Lines. Molecules 2021, 26, 6033. https://doi.org/10.3390/molecules26196033
Tosoc JPS, Nuñeza OM, Sudha T, Darwish NHE, Mousa SA. Anticancer Effects of the Corchorus olitorius Aqueous Extract and Its Bioactive Compounds on Human Cancer Cell Lines. Molecules. 2021; 26(19):6033. https://doi.org/10.3390/molecules26196033
Chicago/Turabian StyleTosoc, John Paul Sese, Olga Macas Nuñeza, Thangirala Sudha, Noureldien H. E. Darwish, and Shaker A. Mousa. 2021. "Anticancer Effects of the Corchorus olitorius Aqueous Extract and Its Bioactive Compounds on Human Cancer Cell Lines" Molecules 26, no. 19: 6033. https://doi.org/10.3390/molecules26196033
APA StyleTosoc, J. P. S., Nuñeza, O. M., Sudha, T., Darwish, N. H. E., & Mousa, S. A. (2021). Anticancer Effects of the Corchorus olitorius Aqueous Extract and Its Bioactive Compounds on Human Cancer Cell Lines. Molecules, 26(19), 6033. https://doi.org/10.3390/molecules26196033









