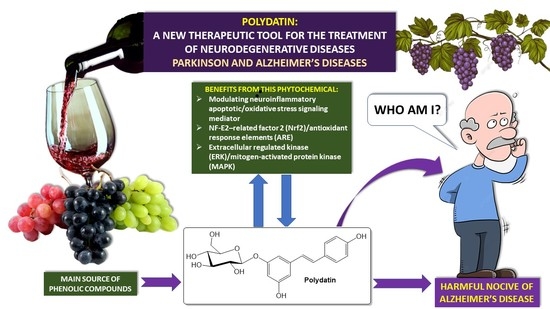
The Neuroprotective Role of Polydatin: Neuropharmacological Mechanisms, Molecular Targets, Therapeutic Potentials, and Clinical Perspective
Abstract
1. Introduction
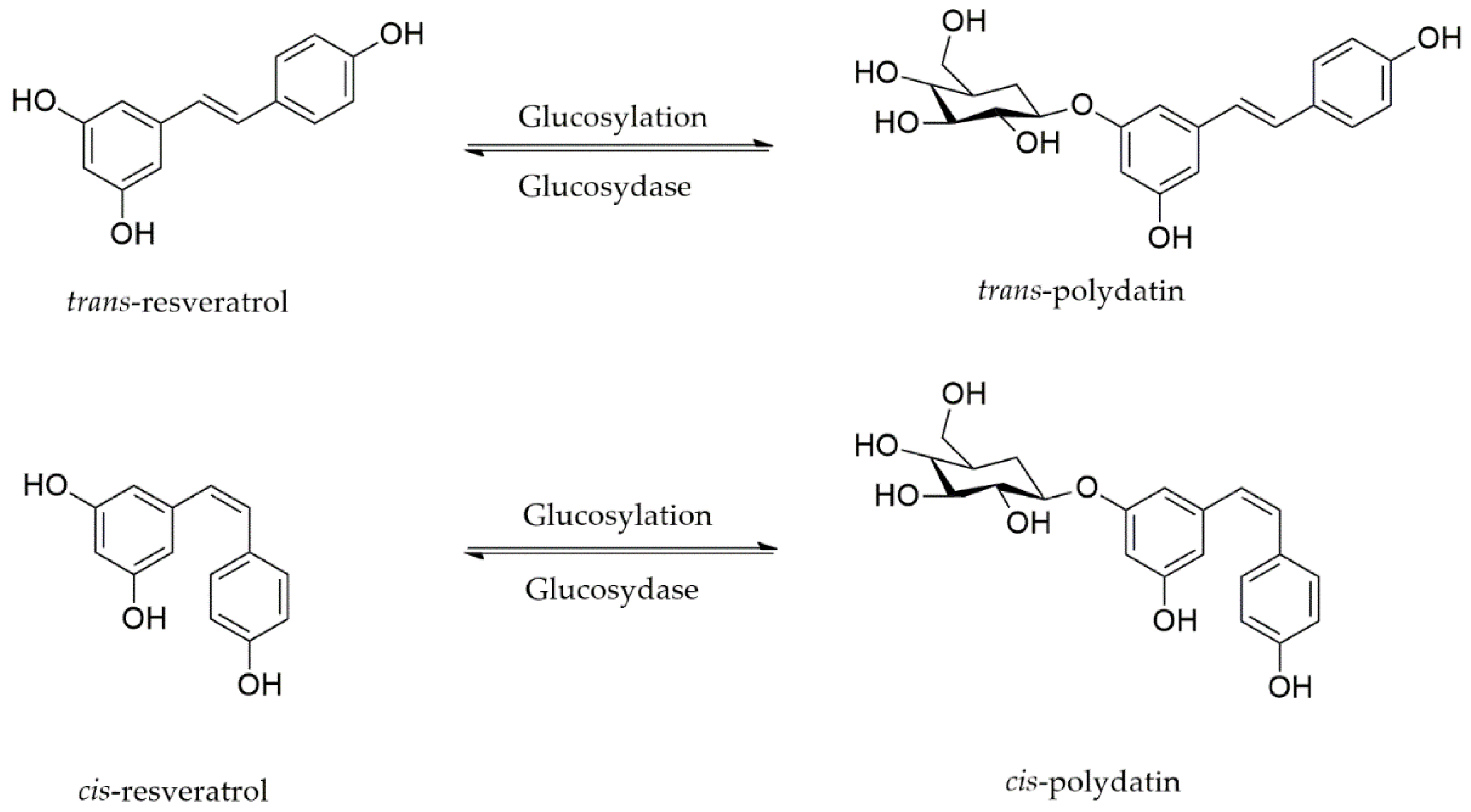
2. Polydatin: Chemical Structure, Sources, and Pharmacokinetic Properties
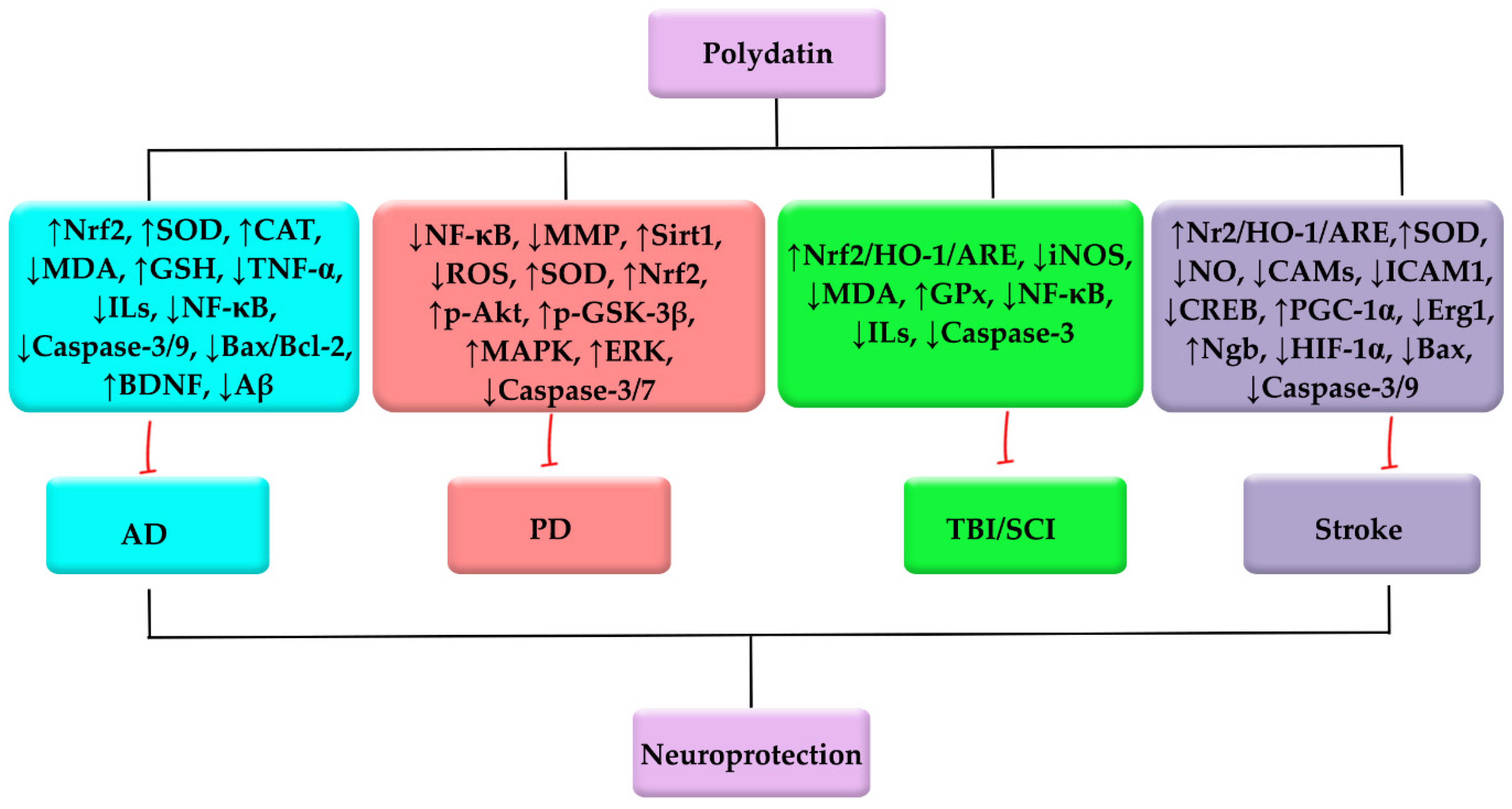
3. Polydatin against NDDs
3.1. Polydatin against AD, and Cognition/Memory Dysfunction
3.2. Polydatin against PD
3.3. Polydatin against Central Nervous System (Brain/Spinal Cord) Injuries
3.4. Polydatin against Stroke: As a Coupled Complication to NDDs
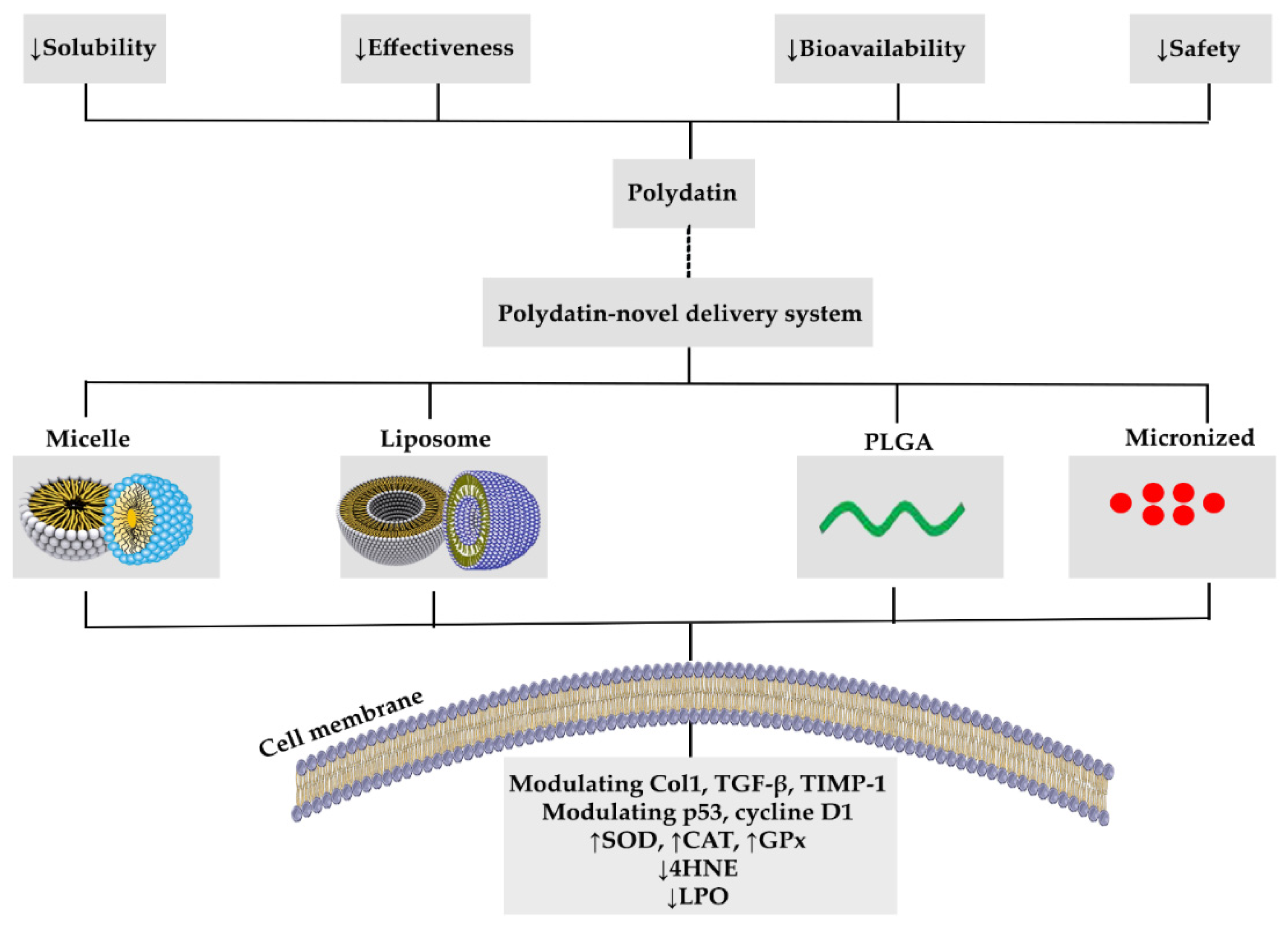
4. Polydatin Novel Delivery Systems: Nanoformulations, and Targeted Therapy
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AD | Alzheimer’s disease |
| Akt | Protein kinase B |
| Atg5 | Autophagy Related 5 |
| ATF6 | Activating transcription factor 6 (ATF6) |
| ATP | Adenosine triphosphate |
| Aβ | Amyloid beta |
| Bcl-2 | B-cell lymphoma 2 |
| ARE | Antioxidant response element |
| BDNF | Brain-derived neurotrophic factor |
| BMSCs | Bone marrow mesenchymal stem cell |
| CAMs | Cell adhesion molecules |
| CAT | Catalase |
| Cdk5 | Cyclin dependent kinase 5 |
| Col1 | Collagen type 1 |
| Cyt | Cytochrome |
| DM | Diabetes mellitus |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| Egr1 | Early growth response 1 |
| ERK | Extracellular-signal-regulated kinase |
| Gli1 | Homolog1 |
| GRP78 | Glucose-regulated protein |
| GPx | Glutathione peroxidase |
| GSH | Glutathione |
| GSK-3β | Glycogen synthase kinase-3β |
| GSSG | Glutathione disulfide |
| HEK-293T | Human embryonic kidney cells |
| HIBI | Hypoxic-ischemic brain injury |
| HIF-1α | Hypoxia-inducible factor 1-alpha |
| HO-1 | Heme oxygenase-1 |
| ICAM-1 | Intercellular adhesion molecule-1 |
| IL | Interleukin |
| iNOS | Inducible nitric oxide synthase |
| LPO | Lipid peroxidation |
| LPS | Lipopolysaccharides |
| MALAT1 | Metastasis associated lung adenocarcinoma transcript 1 |
| MAPK | Mitogen-activated protein kinase |
| MCAO | Middle cerebral artery occlusion |
| MDA | Malondialdehyde |
| MIF | Macrophage migration inhibitory factor |
| MMP | Mitochondrial membrane potential |
| MPTP | 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine |
| nAChRs | Nicotinic acetylcholine receptors |
| NDDs | Neurodegenerative diseases |
| mPTP | Mitochondrial permeability transition pore |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| Ngb | Neuroglobin |
| NO | Nitric oxide |
| Nrf2 | Nuclear factor E2-related factor 2 |
| NQO-1 | NAD(P)H Quinone Dehydrogenase 1 |
| OGD | Oxygen-glucose deprivation |
| Ptch1 | Patched-1 |
| PD | Parkinson’s disease |
| PD-MC | Polydatin-loaded micelle |
| PI3K | Phosphoinositide 3-kinases |
| PLGA | Poly (lactic-co-glycolic acid) |
| ROS | Reactive oxygen species |
| SCI | Spinal cord injury |
| SD | Sprague–Dawley |
| SGLT1 | Sodium-dependent glucose transporter |
| Sirt1 | Sirtuin 1 |
| SMNs | Spinal motor neurons |
| SOD | Superoxide dismutase |
| TBI | Traumatic brain injury |
| TGF-β | Transforming growth factor-beta |
| TIMP-1 | Tissue inhibitor of metalloproteinases 1 |
| TLR | Toll-like receptor |
| TNF-α | Tumor necrosis factor α |
| VCAM-1 | Vascular cell adhesion molecule-1 |
| 4-HNE | 4-Hydroxynonenal |
| 6-OHDA | 6-hydroxydopamine |
References
- Trapp, B.D.; Nave, K.-A. Multiple sclerosis: An immune or neurodegenerative disorder? Annu. Rev. Neurosci. 2008, 31, 247–269. [Google Scholar] [CrossRef]
- Heneka, M.T.; McManus, R.M.; Latz, E. Inflammasome signalling in brain function and neurodegenerative disease. Nat. Rev. Neurosci. 2018, 19, 610–621. [Google Scholar] [CrossRef]
- LaFerla, F.M.; Oddo, S. Alzheimer’s disease: Aβ, tau and synaptic dysfunction. Trends Mol. Med. 2005, 11, 170–176. [Google Scholar] [CrossRef]
- Mamik, M.K.; Power, C. Inflammasomes in neurological diseases: Emerging pathogenic and therapeutic concepts. Brain 2017, 140, 2273–2285. [Google Scholar] [CrossRef] [PubMed]
- Abbaszadeh, F.; Fakhri, S.; Khan, H. Targeting apoptosis and autophagy following spinal cord injury: Therapeutic ap-proaches to polyphenols and candidate phytochemicals. Pharmacol. Res. 2020, 160, 105069. [Google Scholar] [CrossRef] [PubMed]
- Floyd, R.A. Antioxidants, Oxidative Stress, and Degenerative Neurological Disorders. Proc. Soc. Exp. Boil. Med. 1999, 222, 236–245. [Google Scholar] [CrossRef]
- Degan, D.; Ornello, R.; Tiseo, C.; Carolei, A.; Sacco, S.; Pistoia, F. The Role of Inflammation in Neurological Disorders. Curr. Pharm. Des. 2018, 24, 1485–1501. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Chen, M.; Jiang, J. Mitochondrial dysfunction in neurodegenerative diseases and drug targets via apoptotic signaling. Mitochondrion 2019, 49, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Fakhri, S.; Abbaszadeh, F.; Dargahi, L.; Jorjani, M. Astaxanthin: A mechanistic review on its biological activities and health benefits. Pharmacol. Res. 2018, 136, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Fakhri, S.; Abbaszadeh, F.; Jorjani, M. On the therapeutic targets and pharmacological treatments for pain relief following spinal cord injury: A mechanistic review. Biomed. Pharmacother. 2021, 139, 111563. [Google Scholar] [CrossRef] [PubMed]
- Zarneshan, S.N.; Fakhri, S.; Farzaei, M.H.; Khan, H.; Saso, L. Astaxanthin targets PI3K/Akt signaling pathway toward potential therapeutic applications. Food. Chem. Toxicol. 2020, 145, 111714. [Google Scholar] [CrossRef]
- Gravandi, M.M.; Fakhri, S.; Zarneshan, S.N.; Yarmohammadi, A.; Khan, H. Flavonoids modulate AMPK/PGC-1α and inter-connected pathways toward potential neuroprotective activities. Metab. Brain Dis. 2021, 36, 1501–1521. [Google Scholar] [CrossRef] [PubMed]
- Fakhri, S.; Iranpanah, A.; Gravandi, M.M.; Moradi, S.Z.; Ranjbari, M.; Majnooni, M.B.; Echeverría, J.; Qi, Y.; Wang, M.; Liao, P.; et al. Natural products attenuate PI3K/Akt/mTOR signaling pathway: A promising strategy in regulating neurodegeneration. Phytomedicine 2021, 91, 153664. [Google Scholar] [CrossRef] [PubMed]
- Dvorakova, M.; Landa, P. Anti-inflammatory activity of natural stilbenoids: A review. Pharmacol. Res. 2017, 124, 126–145. [Google Scholar] [CrossRef] [PubMed]
- Xiao, K.; Zhang, H.-J.; Xuan, L.-J.; Zhang, J.; Xu, Y.-M.; Bai, D.-L. Stilbenoids: Chemistry and bioactivities. In Bioactive Natural Products (Part L); Attaur, R., Ed.; Elsevier: Amsterdam, The Netherlands, 2008; Volume 34, pp. 453–646. [Google Scholar]
- Henry, C.; Vitrac, X.; Decendit, A.; Ennamany, R.; Krisa, S.; Mérillon, J.-M. Cellular uptake and efflux of trans-piceid and its aglycone trans-resveratrol on the apical membrane of human intestinal Caco-2 cells. J. Agric. Food Chem. 2005, 53, 798–803. [Google Scholar] [CrossRef] [PubMed]
- Henry-Vitrac, C.; Desmoulière, A.; Girard, D.; Mérillon, J.-M.; Krisa, S. Transport, deglycosylation, and metabolism of trans-piceid by small intestinal epithelial cells. Eur. J. Nutr. 2006, 45, 376–382. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.-J.; Yu, H.-W.; Yang, Y.-Z.; Wu, W.-Y.; Chen, T.-Y.; Jia, K.-K.; Kang, L.-L.; Jiao, R.-Q.; Kong, L.-D. Polydatin prevents fructose-induced liver inflammation and lipid deposition through increasing miR-200a to regulate Keap1/Nrf2 pathway. Redox Biol. 2018, 18, 124–137. [Google Scholar] [CrossRef] [PubMed]
- Tang, K.S.; Tan, J.S. The protective mechanisms of polydatin in cerebral ischemia. Eur. J. Pharmacol. 2019, 842, 133–138. [Google Scholar] [CrossRef]
- Tang, K.S. Protective Effects of Polydatin Against Dementia-Related Disorders. Curr. Neuropharmacol. 2020, 19, 127–135. [Google Scholar] [CrossRef]
- Du, Q.-H.; Peng, C.; Zhang, H. Polydatin: A review of pharmacology and pharmacokinetics. Pharm. Biol. 2013, 51, 1347–1354. [Google Scholar] [CrossRef]
- Ribeiro de Lima, M.T.; Waffo-Téguo, P.; Teissedre, P.L.; Pujolas, A.; Vercauteren, J.; Cabanis, J.C.; Mérillon, J.M. Determination of stilbenes (trans-astringin, cis- and trans- piceid, and cis- and trans- resveratrol) in Portuguese wines. J. Agric. Food Chem. 1999, 47, 2666–2670. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Andres-Lacueva, C.; Lamuela-Raventós, R.M.; Berenguer, T.; Jakszyn, P.; Martínez, C.; Sánchez, M.J.; Navarro, C.; Chirlaque, M.D.; Tormo, M.-J.; et al. Concentrations of resveratrol and derivatives in foods and estimation of dietary intake in a Spanish population: European Prospective Investigation into Cancer and Nutrition (EPIC)-Spain cohort. Br. J. Nutr. 2008, 100, 188–196. [Google Scholar] [CrossRef]
- Jensen, J.S.; Wertz, C.F.; O’Neill, V.A. Preformulation Stability of trans-Resveratrol and trans-Resveratrol Glucoside (Piceid). J. Agric. Food Chem. 2010, 58, 1685–1690. [Google Scholar] [CrossRef]
- Galeano-Díaz, T.; Durán-Merás, I.; Airado-Rodríguez, D. Isocratic chromatography of resveratrol and piceid after previous generation of fluorescent photoproducts: Wine analysis without sample preparation. J. Sep. Sci. 2007, 30, 3110–3119. [Google Scholar] [CrossRef]
- Wang, H.-L.; Gao, J.-P.; Han, Y.-L.; Xu, X.; Wu, R.; Gao, Y.; Cui, X.-H. Comparative studies of polydatin and resveratrol on mutual transformation and antioxidative effect in vivo. Phytomedicine 2015, 22, 553–559. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Zhao, Y.; Chen, X.; Zheng, Y.; Wu, X.; Wang, R.; Li, T.; Yu, Q.; Jing, J.; Ma, L.; et al. Quantitative determination of trans-polydatin, a natural strong anti-oxidative compound, in rat plasma and cellular environment of a human colon ade-nocarcinoma cell line for pharmacokinetic studies. J. Chromatogr. B 2007, 855, 145–151. [Google Scholar] [CrossRef]
- Lv, G.; Lou, Z.; Chen, S.; Gu, H.; Shan, L. Pharmacokinetics and tissue distribution of 2,3,5,4′-tetrahydroxystilbene-2-O-β-d-glucoside from traditional Chinese medicine Polygonum multiflorum following oral ad-ministration to rats. J. Ethnopharmacol. 2011, 137, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Lanzilli, G.; Cottarelli, A.; Nicotera, G.; Guida, S.; Ravagnan, G.; Fuggetta, M.P. Anti-inflammatory Effect of Resveratrol and Polydatin by In Vitro IL-17 Modulation. Inflammation 2011, 35, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.-Q.; Xie, Y.-L.; Gui, S.-H.; Zhang, X.; Mo, Z.-Z.; Sun, C.-Y.; Li, C.-L.; Luo, D.-D.; Zhang, Z.-B.; Su, Z.-R.; et al. Polydatin attenuates D-galactose-induced liver and brain damage through its anti-oxidative, anti-inflammatory and anti-apoptotic effects in mice. Food Funct. 2016, 7, 4545–4555. [Google Scholar] [CrossRef] [PubMed]
- Jayalakshmi, P.; Devika, P. Assessment of in vitro antioxidant activity study of polydatin. J. Pharm. Phytochem. 2019, 8, 55–58. [Google Scholar]
- Zeng, Z.; Chen, Z.; Li, T.; Zhang, J.; Gao, Y.; Xu, S.; Cai, S.; Zhao, K.-S. Polydatin: A new therapeutic agent against multiorgan dysfunction. J. Surg. Res. 2015, 198, 192–199. [Google Scholar] [CrossRef]
- Liu, H.-B.; Meng, Q.-H.; Huang, C.; Wang, J.-B.; Liu, X.-W. Nephroprotective effects of polydatin against ischemia/reperfusion injury: A role for the PI3K/Akt signal pathway. Oxidative Med. Cell. Longev. 2015, 2015, 362158. [Google Scholar] [CrossRef]
- Lv, R.; Du, L.; Zhang, L.; Zhang, Z. Polydatin attenuates spinal cord injury in rats by inhibiting oxidative stress and microglia apoptosis via Nrf2/HO-1 pathway. Life Sci. 2019, 217, 119–127. [Google Scholar] [CrossRef]
- Jiang, K.-F.; Zhao, G.; Deng, G.-Z.; Wu, H.-C.; Yin, N.-N.; Chen, X.-Y.; Qiu, C.-W.; Peng, X.-L. Polydatin ameliorates Staphylo-coccus aureus-induced mastitis in mice via inhibiting TLR2-mediated activation of the p38 MAPK/NF-κB pathway. Acta Pharmacol. Sin. 2017, 38, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Starkov, A.A. The Role of Mitochondria in Reactive Oxygen Species Metabolism and Signaling. Ann. N. Y. Acad. Sci. 2008, 1147, 37–52. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Chen, T.; Lei, X.; Li, Y.; Dai, X.; Cao, Y.; Ding, Q.; Lei, X.; Li, T.; Lin, X. Neuroprotective effects of polydatin against mitochondrial-dependent apoptosis in the rat cerebral cortex following ischemia/reperfusion injury. Mol. Med. Rep. 2016, 14, 5481–5488. [Google Scholar] [CrossRef]
- Zeng, Z.; Yang, Y.; Dai, X.; Xu, S.; Li, T.; Zhang, Q.; Zhao, K.-S.; Chen, Z. Polydatin ameliorates injury to the small intestine induced by hemorrhagic shock via SIRT3 activation-mediated mitochondrial protection. Expert Opin. Ther. Targets 2016, 20, 645–652. [Google Scholar] [CrossRef]
- Mathys, H.; Davila-Velderrain, J.; Peng, Z.; Gao, F.; Mohammadi, S.; Young, J.Z.; Menon, M.; He, L.; Abdurrob, F.; Jiang, X.; et al. Single-cell transcriptomic analysis of Alzheimer’s disease. Nature 2019, 570, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Cheignon, C.; Tomas, M.; Bonnefont-Rousselot, D.; Faller, P.; Hureau, C.; Collin, F. Oxidative stress and the amyloid beta peptide in Alzheimer’s disease. Redox Boil. 2018, 14, 450–464. [Google Scholar] [CrossRef]
- Milanini, B.; Valcour, V. Differentiating HIV-Associated Neurocognitive Disorders From Alzheimer’s Disease: An Emerging Issue in Geriatric NeuroHIV. Curr. HIV/AIDS Rep. 2017, 14, 123–132. [Google Scholar] [CrossRef]
- Schneider, J.A.; Arvanitakis, Z.; Leurgans, S.E.; Bennett, D.A. The neuropathology of probable Alzheimer disease and mild cognitive impairment. Ann. Neurol. Off. J. Am. Neurol. Assoc. Child Neurol. Soc. 2009, 66, 200–208. [Google Scholar] [CrossRef]
- Fakhri, S.; Pesce, M.; Patruno, A.; Moradi, S.Z.; Iranpanah, A.; Farzaei, M.H.; Sobarzo-Sánchez, E. Attenuation of Nrf2/Keap1/ARE in Alzheimer’s disease by plant secondary metabolites: A mechanistic review. Molecules 2020, 25, 4926. [Google Scholar] [CrossRef] [PubMed]
- Li, R.-P.; Wang, Z.-Z.; Sun, M.-X.; Hou, X.-L.; Sun, Y.; Deng, Z.-F.; Xiao, K. Polydatin protects learning and memory impair-ments in a rat model of vascular dementia. Phytomedicine 2012, 19, 677–681. [Google Scholar] [CrossRef] [PubMed]
- Tong, Y.; Wang, K.; Sheng, S.; Cui, J. Polydatin ameliorates chemotherapy-induced cognitive impairment (chemobrain) by inhibiting oxidative stress, inflammatory response, and apoptosis in rats. Biosci. Biotechnol. Biochem. 2020, 84, 1201–1210. [Google Scholar] [CrossRef] [PubMed]
- Cauli, O. Oxidative Stress and Cognitive Alterations Induced by Cancer Chemotherapy Drugs: A Scoping Review. Antioxidants 2021, 10, 1116. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Qu, Y.; He, H.; Fan, X.; Qin, Y.; Mao, W.; Xu, L. Protective effect of polydatin on learning and memory impairments in neonatal rats with hypoxic-ischemic brain injury by up-regulating brain-derived neurotrophic factor. Mol. Med. Rep. 2014, 10, 3047–3051. [Google Scholar] [CrossRef][Green Version]
- Zhang, Y.; Li, S.; Wang, W.; Xu, C.; Liang, S.; Liu, M.; Hao, W.; Zhang, R. Beneficial effects of polydatin on learning and memory in rats with chronic ethanol exposure. Int. J. Clin. Exp. Pathol. 2015, 8, 11116–11123. [Google Scholar]
- Rivière, C.; Richard, T.; Quentin, L.; Krisa, S.; Mérillon, J.-M.; Monti, J.-P. Inhibitory activity of stilbenes on Alzheimer’s β-amyloid fibrils in vitro. Bioorg. Med. Chem. 2007, 15, 1160–1167. [Google Scholar] [CrossRef]
- Rivière, C.; Delaunay, J.-C.; Immel, F.; Cullin, C.; Monti, J.-P. The Polyphenol Piceid Destabilizes Preformed Amyloid Fibrils and Oligomers In Vitro: Hypothesis on Possible Molecular Mechanisms. Neurochem. Res. 2008, 34, 1120–1128. [Google Scholar] [CrossRef]
- Xiao, H.-T.; Qi, X.-L.; Liang, Y.; Lin, C.-Y.; Wang, X.; Guan, Z.-Z.; Hao, X.-Y. Membrane permeability-guided identification of neuroprotective components fromPolygonum cuspidatun. Pharm. Biol. 2013, 52, 356–361. [Google Scholar] [CrossRef]
- Xu, C.-Y.; Li, S.; Chen, L.; Hou, F.-J.; Zhng, R.-L. [Effect of polydatin on learning and memory and expression of NR2B in the prefrontal cortex of rats with chronic alcoholism]. Chin. J. Appl. Physiol. 2011, 27, 213–235. [Google Scholar]
- de Lau, L.M.L.; Breteler, M.M.B. Epidemiology of Parkinson’s disease. Lancet Neurol. 2006, 5, 525–535. [Google Scholar] [CrossRef]
- Shtilbans, A.; Henchcliffe, C. Biomarkers in Parkinson’s disease. Curr. Opin. Neurol. 2012, 25, 460–465. [Google Scholar] [CrossRef]
- Jiang, T.; Sun, Q.; Chen, S. Oxidative stress: A major pathogenesis and potential therapeutic target of antioxidative agents in Parkinson’s disease and Alzheimer’s disease. Prog. Neurobiol. 2016, 147, 1–19. [Google Scholar] [CrossRef]
- Roy, S. Synuclein and dopamine: The Bonnie and Clyde of Parkinson’s disease. Nat. Neurosci. 2017, 20, 1514–1515. [Google Scholar] [CrossRef]
- Bai, H.; Ding, Y.; Li, X.; Kong, D.; Xin, C.; Yang, X.; Zhang, C.; Rong, Z.; Yao, C.; Lu, S.; et al. Polydatin protects SH-SY5Y in models of Parkinson’s disease by promoting Atg5-mediated but parkin-independent autophagy. Neurochem. Int. 2020, 134, 104671. [Google Scholar] [CrossRef] [PubMed]
- Kang, L.; Liu, S.; Li, J.; Tian, Y.; Xue, Y.; Liu, X. Parkin and Nrf2 prevent oxidative stress-induced apoptosis in intervertebral endplate chondrocytes via inducing mitophagy and anti-oxidant defenses. Life Sci. 2020, 243, 117244. [Google Scholar] [CrossRef]
- Yun, S.P.; Kam, T.-I.; Panicker, N.; Kim, S.; Oh, Y.; Park, J.-S.; Kwon, S.-H.; Park, Y.J.; Karuppagounder, S.S.; Park, H.; et al. Block of A1 astrocyte conversion by microglia is neuroprotective in models of Parkinson’s disease. Nat. Med. 2018, 24, 931–938. [Google Scholar] [CrossRef] [PubMed]
- Kujawska, M.; Jodynis-Liebert, J. Polyphenols in Parkinson’s Disease: A Systematic Review of In Vivo Studies. Nutrients 2018, 10, 642. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhang, D.-Q.; Liao, Z.; Wang, B.; Gong, S.; Wang, C.; Zhang, M.-Z.; Wang, G.-H.; Cai, H.; Liao, F.-F.; et al. Anti-oxidant polydatin (piceid) protects against substantia nigral motor degeneration in multiple rodent models of Parkinson’s disease. Mol. Neurodegener. 2015, 10, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Liu, J.; Meng, T.; Li, Y.; He, D.; Ran, X.; Chen, G.; Guo, W.; Kan, X.; Fu, S.; et al. Polydatin prevents lipopolysaccharide (LPS)-induced Parkinson’s disease via regulation of the AKT/GSK3β-Nrf2/NF-κB signaling axis. Front. Immunol. 2018, 9, 2527. [Google Scholar] [CrossRef]
- Bachiller, S.; Jiménez-Ferrer, I.; Paulus, A.; Yang, Y.; Swanberg, M.; Deierborg, T.; Boza-Serrano, A. Microglia in neurological diseases: A road map to brain-disease dependent-inflammatory response. Front. Cell. Neurosci. 2018, 12, 488. [Google Scholar] [CrossRef]
- Hong, C.T.; Chau, K.-Y.; Schapira, A.H.V. Meclizine-induced enhanced glycolysis is neuroprotective in Parkinson disease cell models. Sci. Rep. 2016, 6, 25344. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Wang, S.; Shi, X.; Feng, X. Polydatin alleviates parkinsonism in MPTP-model mice by enhancing glycolysis in dopaminergic neurons. Neurochem. Int. 2020, 139, 104815. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Sun, L.; Chen, X.; Zhang, D. Oxidative stress, mitochondrial damage and neurodegenerative diseases. Neural Regen. Res. 2013, 8, 2003–2014. [Google Scholar] [CrossRef] [PubMed]
- Potdar, S.; Parmar, M.S.; Ray, S.D.; Cavanaugh, J.E. Protective effects of the resveratrol analog piceid in dopaminergic SH-SY5Y cells. Arch. Toxicol. 2018, 92, 669–677. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.R.; Shaikh, M.A.; Baloch, N.A.; Nazir, S.; Abrar, H.; Ulhaq, H.S.I. Neuroprotective Potential of Polydatin Against Motor Abnormalities and Dopaminergic Neuronal Loss in Rotenone Induced Parkinson Model. Int. J. Morphol. 2018, 36, 584–591. [Google Scholar] [CrossRef]
- Fakhri, S.; Tomas, M.; Capanoglu, E.; Hussain, Y.; Abbaszadeh, F.; Lu, B.; Hu, X.; Wu, J.; Zou, L.; Smeriglio, A. Antioxidant and anticancer potentials of edible flowers: Where do we stand? Crit. Rev. Food Sci. Nutr. 2021, 1–57. [Google Scholar] [CrossRef]
- Shi, K.; Zhang, J.; Dong, J.-F.; Shi, F.-D. Dissemination of brain inflammation in traumatic brain injury. Cell. Mol. Immunol. 2019, 16, 523–530. [Google Scholar] [CrossRef]
- Fakhri, S.; Piri, S.; Majnooni, M.B.; Farzaei, M.H.; Echeverría, J. Targeting neurological manifestations of coronaviruses by candidate phytochemicals: A mechanistic approach. Front. Pharmacol. 2020, 11, 621099. [Google Scholar] [CrossRef] [PubMed]
- Nouri, Z.; Fakhri, S.; Nouri, K.; Wallace, C.E.; Farzaei, M.H.; Bishayee, A. Targeting multiple signaling pathways in cancer: The rutin therapeutic approach. Cancers 2020, 12, 2276. [Google Scholar] [CrossRef]
- Zhan, J.; Li, X.; Luo, D.; Yan, W.; Hou, Y.; Hou, Y.; Chen, S.; Luan, J.; Zhang, Q.; Lin, D. Polydatin Attenuates OGD/R-Induced Neuronal Injury and Spinal Cord Ischemia/Reperfusion Injury by Protecting Mitochondrial Function via Nrf2/ARE Signaling Pathway. Oxidative Med. Cell. Longev. 2021, 2021, 6687212. [Google Scholar] [CrossRef] [PubMed]
- Zhan, J.; Li, X.; Luo, D.; Hou, Y.; Hou, Y.; Chen, S.; Xiao, Z.; Luan, J.; Lin, D. Polydatin promotes the neuronal differentiation of bone marrow mesenchymal stem cells in vitro and in vivo: Involvement of Nrf2 signalling pathway. J. Cell. Mol. Med. 2020, 24, 5317–5329. [Google Scholar] [CrossRef]
- Chen, M.; Hou, Y.; Lin, D. Polydatin Protects Bone Marrow Stem Cells against Oxidative Injury: Involvement of Nrf 2/ARE Pathways. Stem Cells Int. 2016, 2016, 9394150. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; He, S.; Cai, Q.; Li, F.; Wang, S.; Tao, K.; Xi, Y.; Qin, H.; Gao, G.; Feng, D. Polydatin alleviates traumatic brain injury: Role of inhibiting ferroptosis. Biochem. Biophys. Res. Commun. 2021, 556, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Prochazka, A. Targeted stimulation of the spinal cord to restore locomotor activity. Nat. Med. 2016, 22, 125–126. [Google Scholar] [CrossRef] [PubMed]
- Fakhri, S.; Kiani, A.; Jalili, C.; Abbaszadeh, F.; Piri, S.; Farzaei, M.H.; Rastegari-Pouyani, M.; Mohammadi-Noori, E.; Khan, H. Intrathecal Administration of Melatonin Ameliorates the Neuroinflammation-mediated Sensory and Motor Dysfunction in a rat Model of Compression Spinal Cord Injury. Curr. Mol. Pharmacol. 2020. [Google Scholar] [CrossRef]
- Gu, Z.; Li, L.; Li, Q.; Tan, H.; Zou, Z.; Chen, X.; Zhang, Z.; Zhou, Y.; Wei, D.; Liu, C.; et al. Polydatin alleviates severe traumatic brain injury induced acute lung injury by inhibiting S100B mediated NETs formation. Int. Immunopharmacol. 2021, 98, 107699. [Google Scholar] [CrossRef]
- Wolfe, C.D.A. The impact of stroke. Br. Med Bull. 2000, 56, 275–286. [Google Scholar] [CrossRef]
- Ruan, W.; Li, J.; Xu, Y.; Wang, Y.; Zhao, F.; Yang, X.; Jiang, H.; Zhang, L.; Saavedra, J.M.; Shi, L.; et al. MALAT1 up-regulator poly-datin protects brain microvascular integrity and ameliorates stroke through C/EBPβ/MALAT1/CREB/PGC-1α/PPARγ path-way. Cell. Mol. Neurobiol. 2019, 39, 265–286. [Google Scholar] [CrossRef] [PubMed]
- Shah, F.A.; Kury, L.A.; Li, T.; Zeb, A.; Koh, P.O.; Liu, F.; Zhou, Q.; Hussain, I.; Khan, A.U.; Jiang, Y.; et al. Polydatin attenuates neu-ronal loss via reducing neuroinflammation and oxidative stress in rat MCAO models. Front. Pharmacol. 2019, 10, 663. [Google Scholar] [CrossRef]
- Ji, H.; Zhang, X.; Du, Y.; Liu, H.; Li, S.; Li, L. Polydatin modulates inflammation by decreasing NF-κB activation and oxidative stress by increasing Gli1, Ptch1, SOD1 expression and ameliorates blood–brain barrier permeability for its neuroprotective effect in pMCAO rat brain. Brain Res. Bull. 2012, 87, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Bheereddy, P.; Yerra, V.G.; Kalvala, A.K.; Sherkhane, B.; Kumar, A. SIRT1 activation by polydatin alleviates oxidative damage and elevates mitochondrial biogenesis in experimental diabetic neuropathy. Cell Mol. Neurobiol. 2020, 1–15. [Google Scholar] [CrossRef]
- Cheng, Y.; Zhang, H.-T.; Sun, L.; Guo, S.; Ouyang, S.; Zhang, Y.; Xu, J. Involvement of cell adhesion molecules in polydatin protection of brain tissues from ischemia–reperfusion injury. Brain Res. 2006, 1110, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.Y.; Fang, X.Y.; Zhang, H. Effect of polydatin on expression of p53 and Notch1 in brain tissue of ischemic cerebrovascular disease. J. Biol. Regul. Homeost. Agents 2018, 32, 133–138. [Google Scholar] [PubMed]
- Liu, N.; Yu, Z.; Gao, X.; Yun, S.S.; Yuan, J.; Xun, Y.; Wang, T.; Yan, F.; Yuan, S.; Zhang, J.; et al. Establishment of cell-based neu-roglobin promoter reporter assay for neuroprotective compounds screening. CNS Neurol. Disord.-Drug Targets (Former. Curr. Drug Targets-CNS Neurol. Disord.) 2016, 15, 629–639. [Google Scholar]
- Xu, B.; Lin, H.-B.; Zhou, H.; Xu, J.-P. [Protective effect of polydatin on a PC12 cell model of oxygen-glucose deprivation]. Nan fang yi ke da xue xue bao J. South. Med Univ. 2010, 30, 1041–1043. [Google Scholar]
- Liu, N.; Yu, Z.; Xiang, S.; Zhao, S.; Tjärnlund-Wolf, A.; Xing, C.; Zhang, J.; Wang, X. Transcriptional regulation mechanisms of hypoxia-induced neuroglobin gene expression. Biochem. J. 2012, 443, 153–164. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhang, G.; Bie, X.; Liu, M.; Yang, J.; Wan, H.; Zhang, Y. Effect of polydatin on dynamic changes of excitatory amino acids in cerebrospinal fluid of cerebral hemorrhage rats. China J. Chin. Mater. Med. 2010, 35, 3038–3042. [Google Scholar]
- Guan, S.-Y.; Zhang, K.; Wang, X.-S.; Yang, L.; Feng, B.; Tian, D.-D.; Gao, M.-R.; Liu, S.-B.; Liu, A.; Zhao, M.-G. Anxiolytic effects of polydatin through the blockade of neuroinflammation in a chronic pain mouse model. Mol. Pain 2020, 16, 1744806919900717. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Yu, Z.; Li, Y.; Yuan, J.; Zhang, J.; Xiang, S.; Wang, X. Transcriptional regulation of mouse neuroglobin gene by cyclic AMP responsive element binding protein (CREB) in N2a cells. Neurosci. Lett. 2013, 534, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Tan, H.-P.; Liu, C.-Y.; Yu, L.-T.; Wei, D.-N.; Zhang, Z.-C.; Lu, K.; Zhao, K.-S.; Maegele, M.; Cai, D.-Z.; et al. Polydatin prevents the induction of secondary brain injury after traumatic brain injury by protecting neuronal mitochondria. Neural Regen. Res. 2019, 14, 1573–1582. [Google Scholar] [CrossRef]
- Lv, R.; Du, L.; Liu, X.; Zhou, F.; Zhang, Z.; Zhang, L. Polydatin alleviates traumatic spinal cord injury by reducing microglial inflammation via regulation of iNOS and NLRP3 inflammasome pathway. Int. Immunopharmacol. 2019, 70, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Qin, J.; Li, H.; Feng, X.; Lv, Y.; Yang, J. Effect of Polydatin on Neurological Function and the Nrf2 Pathway during Intracerebral Hemorrhage. J. Mol. Neurosci. 2020, 70, 1332–1337. [Google Scholar] [CrossRef] [PubMed]
- Pelaz, B.; Alexiou, C.; Alvarez-Puebla, R.A.; Alves, F.; Andrews, A.M.; Ashraf, S.; Balogh, L.P.; Ballerini, L.; Bestetti, A.; Brendel, C.; et al. Diverse Applications of Nanomedicine. ACS Nano 2017, 11, 2313–2381. [Google Scholar] [CrossRef] [PubMed]
- Yousef, A.I.; Shawki, H.H.; El-Shahawy, A.A.; El-Twab, S.M.A.; Abdel-Moneim, A.; Oishi, H. Polydatin mitigates pancreatic β-cell damage through its antioxidant activity. Biomed. Pharmacother. 2021, 133, 111027. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Moneim, A.; El-Shahawy, A.; Yousef, A.I.; El-Twab, S.M.A.; Elden, Z.E.; Taha, M. Novel polydatin-loaded chitosan nanoparticles for safe and efficient type 2 diabetes therapy: In silico, in vitro and in vivo approaches. Int. J. Biol. Macromol. 2020, 154, 1496–1504. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, F.; Galaly, S.R.; Mohamed, H.M.; Abdel-Moneim, A.; Abdul-Hamid, M. Ameliorative effect of polydatin and poly-datin-loaded chitosan nanoparticles against diabetes-induced pulmonary disorders in rats. J. Taibah Univ. Sci. 2021, 15, 37–49. [Google Scholar] [CrossRef]
- Fabris, S.; Momo, F.; Ravagnan, G.; Stevanato, R. Antioxidant properties of resveratrol and piceid on lipid peroxidation in micelles and monolamellar liposomes. Biophys. Chem. 2008, 135, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Gong, H.; Li, R.; Huang, J.; Cai, M.; Lan, T.; Huang, W.; Guo, Y.; Zhou, Z.; An, Y.; et al. Nanodrug with ROS and pH Dual-Sensitivity Ameliorates Liver Fibrosis via Multicellular Regulation. Adv. Sci. 2020, 7, 1903138. [Google Scholar] [CrossRef] [PubMed]
- Guan, Q.; Chen, W.; Hu, X.; Wang, X.; Li, L. Novel nanoliposomal delivery system for polydatin: Preparation, characterization, and in vivo evaluation. Drug Des. Dev. Ther. 2015, 9, 1805–1813. [Google Scholar] [CrossRef] [PubMed]
- Fakhri, S.; Abbaszadeh, F.; Jorjani, M.; Pourgholami, M.H. The effects of anticancer medicinal herbs on vascular endothelial growth factor based on pharmacological aspects: A review study. Nutr. Cancer 2021, 73, 1–15. [Google Scholar] [CrossRef]
- Xiao, J. Dietary Flavonoid Aglycones and Their Glycosides: Which Show Better Biological Significance? Crit. Rev. Food Sci. Nutr. 2017, 57, 1874–1905. [Google Scholar] [CrossRef] [PubMed]
- Vijayalakshmi, S.; Mariadoss, A.V.A.; Ramachandran, V.; Shalini, V.; Agilan, B.; Sangeetha, C.C.; Balupillai, A.; Kotakadi, V.S.; Karthikkumar, V.; Ernest, D. Polydatin Encapsulated Poly [Lactic-co-glycolic acid] Nanoformulation Counteract the 7,12-Dimethylbenz[a] Anthracene Mediated Experimental Carcinogenesis through the Inhibition of Cell Proliferation. Antioxidants 2019, 8, 375. [Google Scholar] [CrossRef] [PubMed]
- Cremon, C.; Stanghellini, V.; Barbaro, M.R.; Cogliandro, R.F.; Bellacosa, L.; Santos, J.; Vicario, M.; Pigrau, M.; Cotoner, C.A.; Lobo, B.; et al. Randomised clinical trial: The analgesic properties of dietary supplementation with palmitoylethanolamide and polydatin in irritable bowel syndrome. Aliment. Pharmacol. Ther. 2017, 45, 909–922. [Google Scholar] [CrossRef] [PubMed]
- Lagoa, R.; Silva, J.; Rodrigues, J.R.; Bishayee, A. Advances in phytochemical delivery systems for improved anticancer activity. Biotechnol. Adv. 2020, 38, 107382. [Google Scholar] [CrossRef]
- Kashyap, D.; Tuli, H.S.; Yerer, M.B.; Sharma, A.; Sak, K.; Srivastava, S.; Pandey, A.; Garg, V.K.; Sethi, G.; Bishayee, A. Natural product-based nanoformulations for cancer therapy: Opportunities and challenges. Semin. Cancer Biol. 2021, 69, 5–23. [Google Scholar] [CrossRef]
- Fakhri, S.; Moradi, S.Z.; Farzaei, M.H.; Bishayee, A. Modulation of dysregulated cancer metabolism by plant secondary me-tabolites: A mechanistic review. Semin. Cancer Biol. 2020, 74, 1–156. [Google Scholar] [CrossRef]
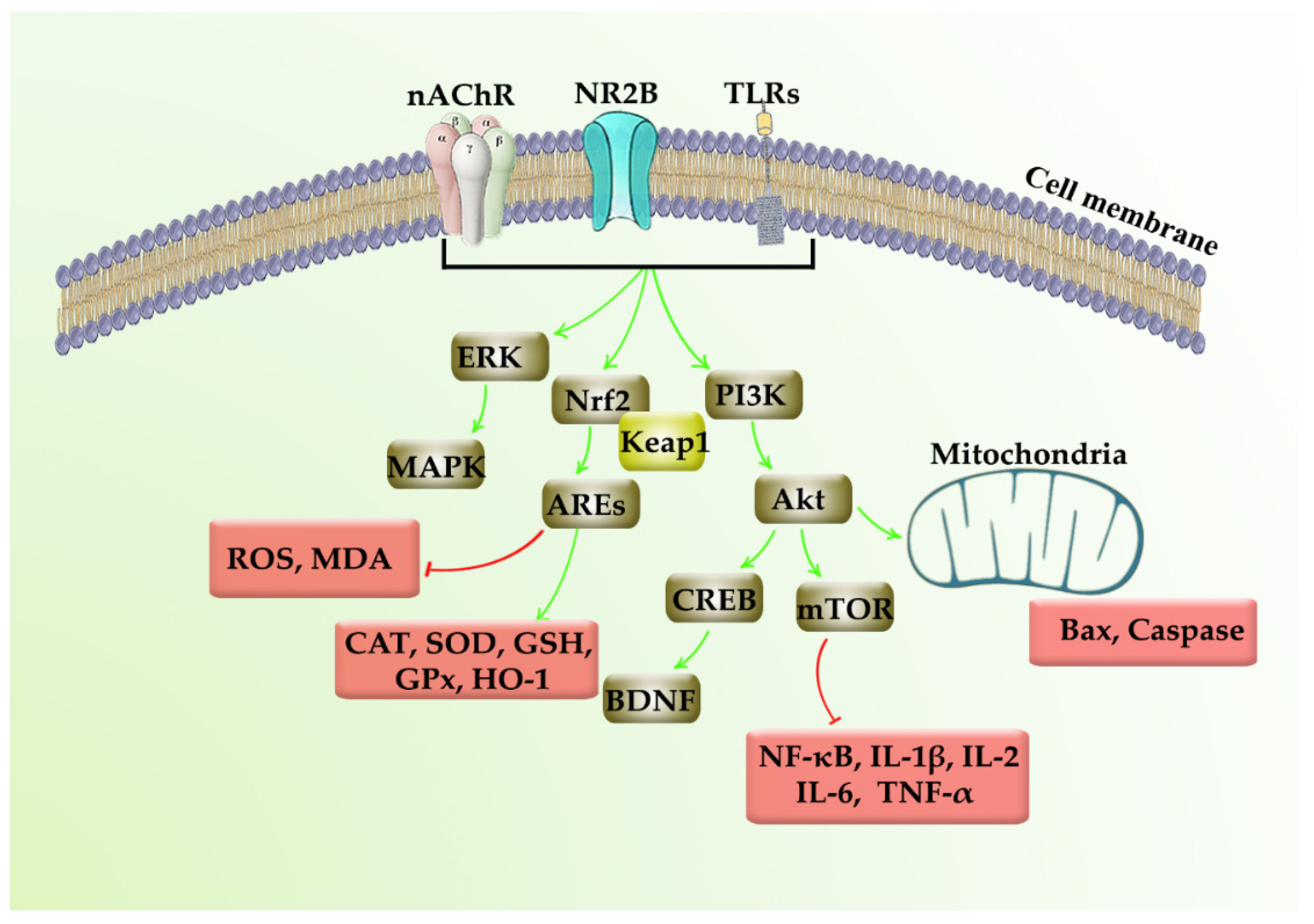
| NDDs | Methods | Models | Neuropharmacological Mechanisms | References |
|---|---|---|---|---|
| AD | Chronic cerebral hypoperfusion | in vivo: Sprague–Dawley rats | ↓MDA, ↑CAT, ↑SOD | [44] |
| Doxorubicin-induced cognitive impairment | in vivo: Sprague–Dawley rats | ↓Nrf2, ↑NF-κB, ↓caspase-3, ↓caspase-9 | [45,46] | |
| HIBI | in vivo: Sprague–Dawley rats | ↓Memory deficient, ↑BDNF | [47] | |
| Chronic ethanol exposure | in vivo: Sprague–Dawley rats | ↑Cell survival, ↓cdk5, ↓functional defects | [48] | |
| Polymerization of Aβ | in vitro: Aβ25–35 | ↓Aβ25–35 polymerization | [49,50] | |
| D-galactose-induced | in vitro: DPPH in vivo: Male Kunming mice | ↑Body weight, ↓MDA, ↑CAT, ↑SOD, ↑GSH, ↓IL-1β, ↓TNF-α, ↓IL-6, ↓ Bax/Bcl-2, ↓caspase-3 | [30] | |
| PD | Rotenone-induced | in vitro: Human neuroblastoma SH-SY5Y | ↓Mitochondrial dysfunction, Atg5-mediated autophagy, modulating MMP, ↑Sirt 1, ↓ROS | [57] |
| LPS-induced | in vitro: Microglial BV-2 cells in vivo: Wistar rats | ↑p-GSK-3β, ↑p-Akt, ↑Ser9, ↑Nrf2, ↓NF-κB | [62] | |
| MPTP-induced | in vivo: Adult male BALB/c mice | ↑Glycolysis, ↑ATP production, ↓motor dysfunction | [65] | |
| Rotenone-induced | in vivo: Sprague–Dawley rats | ↑ATP, ↑SOD, ↑thioredoxin, | [61] | |
| in vitro: Dopaminergic SH-SY5Y cells | ↑MAPK, ↓caspase-3, caspase-7, ↓LPO, ↑ERK1/2/5 | [67] | ||
| TBI/SCI | Brain injury | in vivo: Wistar albino male rats | ↓MDA, ↑antioxidant potential | [93] |
| SCI | in vitro: murine microglia BV2 cells in vivo: Sprague-Dawley rats | ↑Nrf2, ↑HO-1, ↓caspase-3, ↓Bax/Bcl-2 ratio | [34] | |
| Oxygen glucose deprivation/re-oxygenation-induced mitochondrial injury | in vivo: C57BL/6J mice in vitro: SMNs | ↑Intracellular calcium levels, ↑mPTP, ↓ROS, ↓apoptosis, ↑ATP, ↓Keap1, ↑Nrf2, ↑HO-1, ↑NQO-1 | [73] | |
| Neuronal differentiation of BMSCs | in vivo: C57BL/6 mice in vitro: Bone marrow mesenchymal stem cell (BMSC) | ↑Nrf2 | [74,75] | |
| Secondary damage of TBI | in vivo: TBI mouse model in vitro: Neuro2A cells | ↓GPx, ↑MDA, ↓accumulation of free Fe2+ | [76] | |
| SCI | in vivo: Sprague-Dawley rats in vitro: Murine microglia BV2 cells | ↓TNF-α, ↓IL-1β, ↓NO, ↓iNOS, ↓ IL-6, ↓NF-κB | [94] | |
| D-galactose-induced | in vivo: Male Kunming mice in vitro: DPPH | ↓TNF-α, ↓IL-1β, ↓IL-6, ↓ caspase-3, ↓Bax/Bcl-2 | [30] | |
| Stroke | MCAO | in vivo: Sprague–Dawley rats | ↑Nrf2, ↑HO-1, ↓ROS, ↓p38, ↑Gli1, ↑Ptch1, ↑SOD1 | [82,83] |
| Intracerebral hemorrhage | in vivo: Wistar rat | ↑Neurological function, ↑NO, ↑SOD, ↑MDA, ↑GSSG, ↑GSH, ↑Nrf2 | [95] | |
| MCAO | in vivo: Sprague–Dawley rats | ↑Bcl-2, ↓IL-1β, ↓TNF-α, ↓ IL-6, ↓Bax, ↓caspases-3/9 | [37] | |
| Ischemia–reperfusion injury | in vivo: Sprague–Dawley rats | ↓CAMs, ↓E-selectin, ↓L-selectin, ↓ICAM-1 | [85] | |
| OGD | in vitro: Human embryonic kidney cells (HEK-293T) in vivo: Sprague–Dawley rats | ↑MALAT1, ↑CREB, ↑PGC-1α | [81] | |
| MCAO | in vivo: Sprague–Dawley rats | ↓Edema, ↓apoptosis, p53/Notch1 modulation | [86] | |
| OGD | in vitro: PC12 cell | ↓CREB, ↓HIF-1α, ↓p56, ↓Egr1, ↑Ngb, ↓NO | [87,88,89] | |
| Hypoxia/ischemia and oxidative stress-induced injury | in vitro: N2a cells | ↓CREB, ↑BDNF, ↑Shh, ↑Ngb, ↓apoptosis | [19,92] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fakhri, S.; Gravandi, M.M.; Abdian, S.; Akkol, E.K.; Farzaei, M.H.; Sobarzo-Sánchez, E. The Neuroprotective Role of Polydatin: Neuropharmacological Mechanisms, Molecular Targets, Therapeutic Potentials, and Clinical Perspective. Molecules 2021, 26, 5985. https://doi.org/10.3390/molecules26195985
Fakhri S, Gravandi MM, Abdian S, Akkol EK, Farzaei MH, Sobarzo-Sánchez E. The Neuroprotective Role of Polydatin: Neuropharmacological Mechanisms, Molecular Targets, Therapeutic Potentials, and Clinical Perspective. Molecules. 2021; 26(19):5985. https://doi.org/10.3390/molecules26195985
Chicago/Turabian StyleFakhri, Sajad, Mohammad Mehdi Gravandi, Sadaf Abdian, Esra Küpeli Akkol, Mohammad Hosein Farzaei, and Eduardo Sobarzo-Sánchez. 2021. "The Neuroprotective Role of Polydatin: Neuropharmacological Mechanisms, Molecular Targets, Therapeutic Potentials, and Clinical Perspective" Molecules 26, no. 19: 5985. https://doi.org/10.3390/molecules26195985
APA StyleFakhri, S., Gravandi, M. M., Abdian, S., Akkol, E. K., Farzaei, M. H., & Sobarzo-Sánchez, E. (2021). The Neuroprotective Role of Polydatin: Neuropharmacological Mechanisms, Molecular Targets, Therapeutic Potentials, and Clinical Perspective. Molecules, 26(19), 5985. https://doi.org/10.3390/molecules26195985