Cordycepin Sensitizes Cholangiocarcinoma Cells to Be Killed by Natural Killer-92 (NK-92) Cells
Abstract
1. Introduction
2. Results
2.1. Cytotoxicity of Cordycepin in CCA Cell Lines KKU-213A, KKU-100, and KKU055
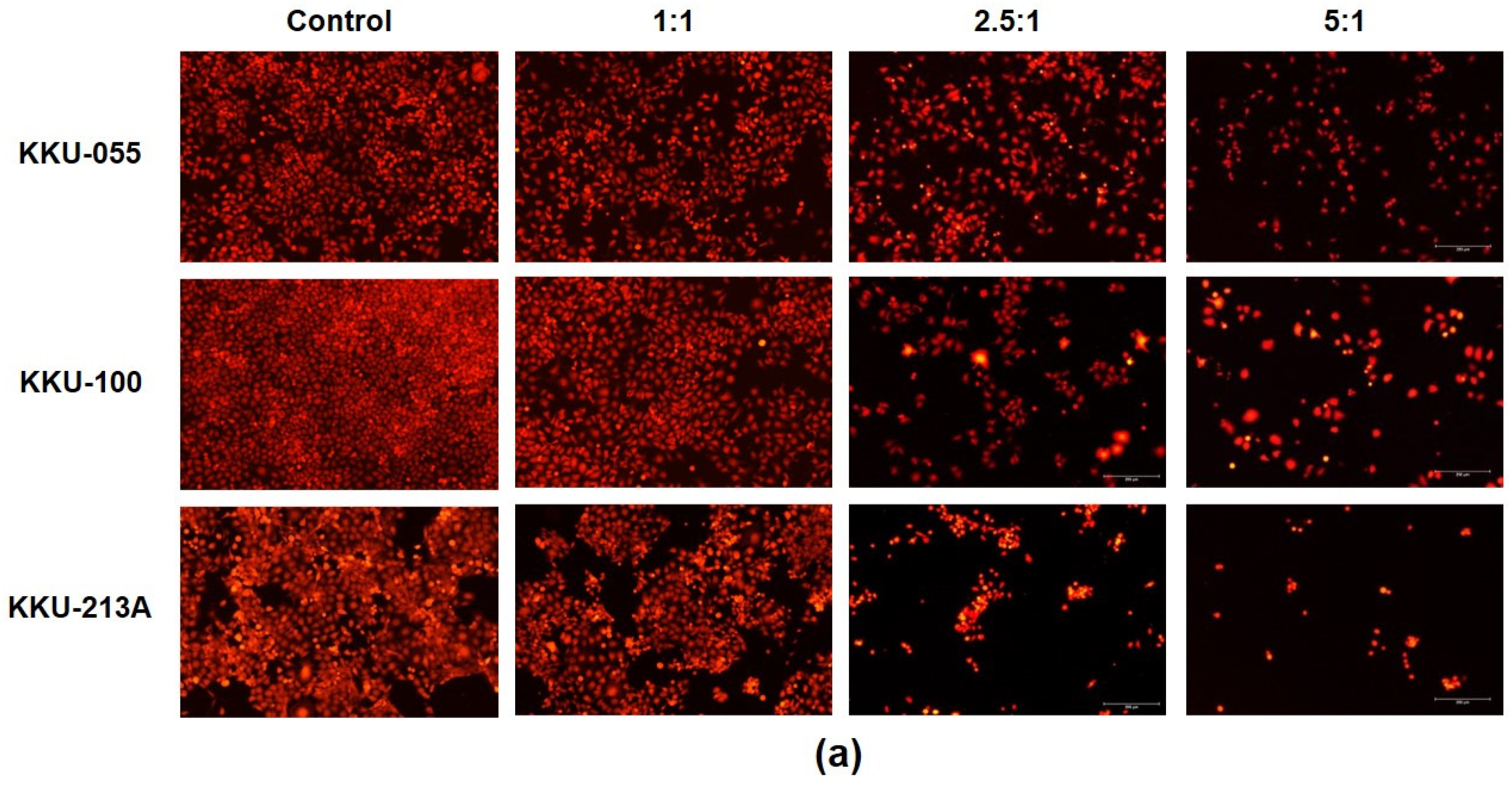
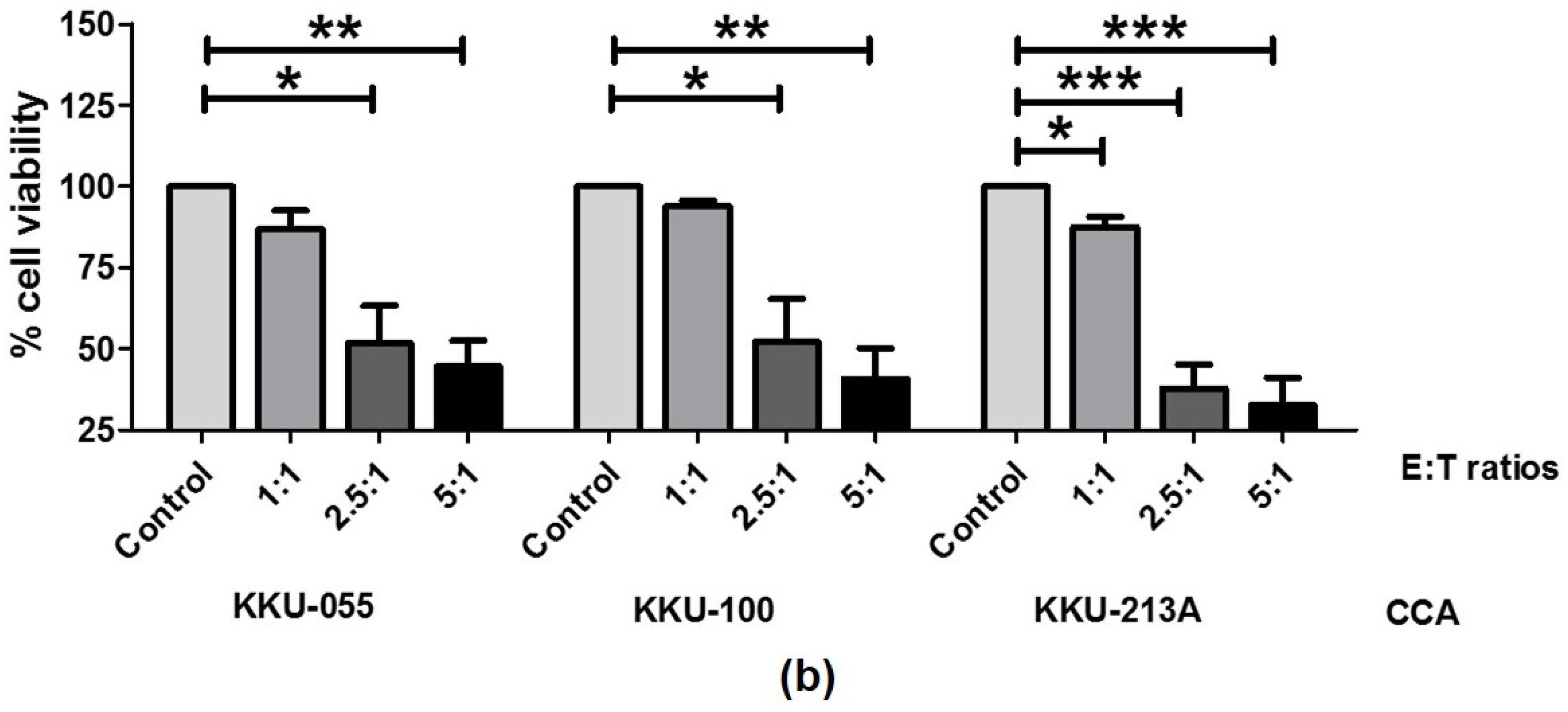
2.2. The Killing Ability of NK-92 to Eradicate CCA Cell Lines
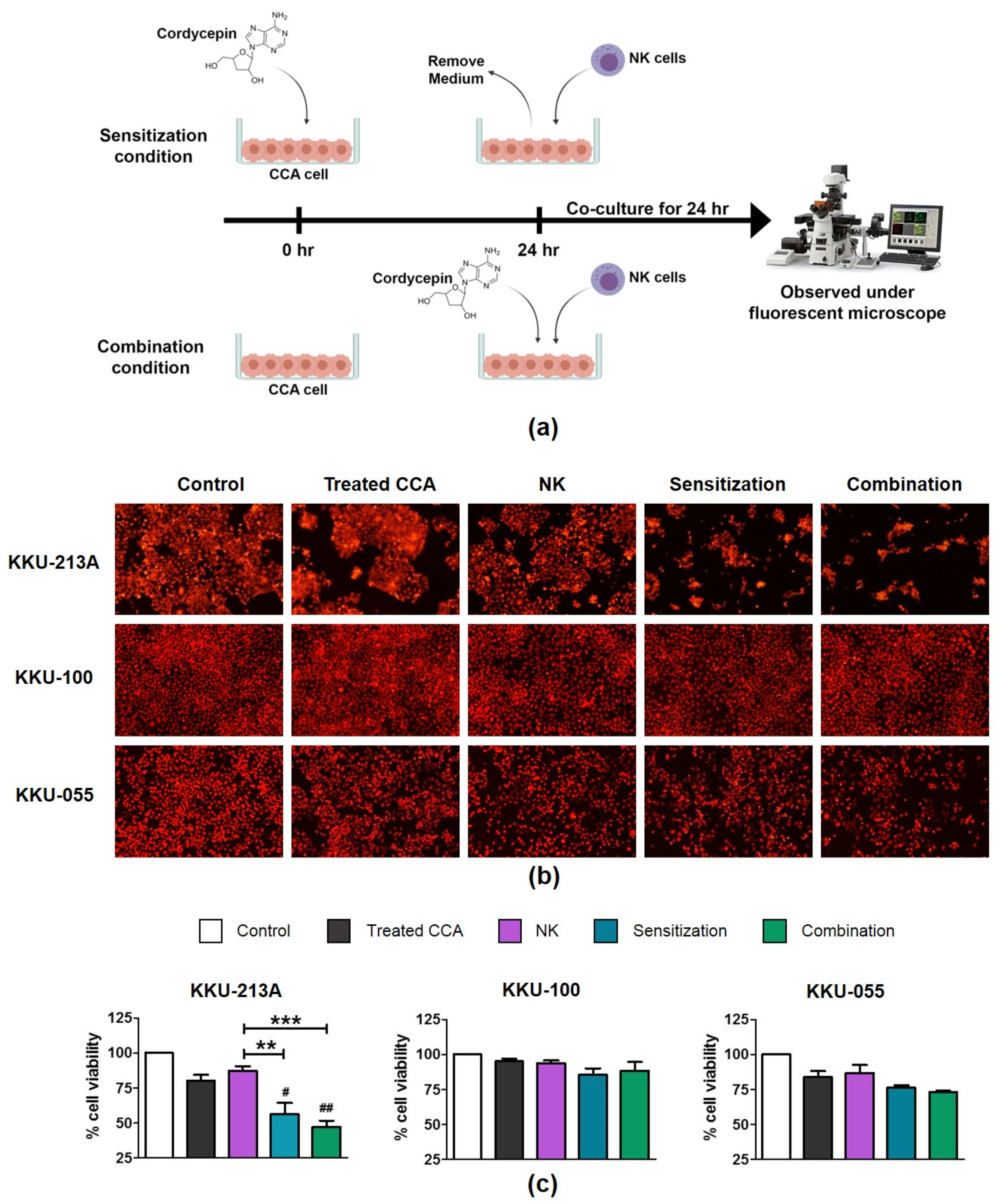
2.3. Treatment of Cordycepin Enhanced the NK-92 Killing Ability
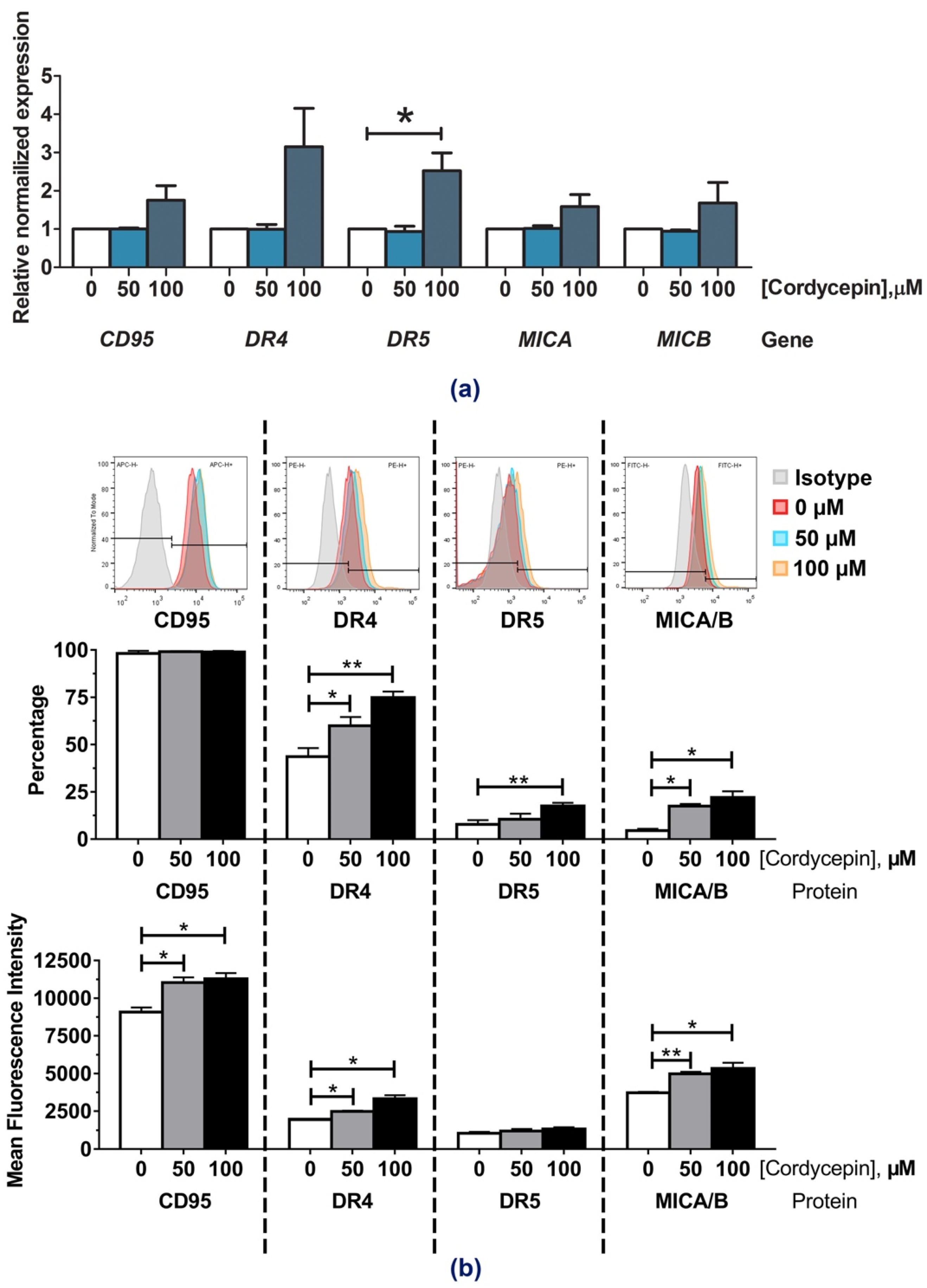
2.4. The Effects of Cordycepin on Modulating CD95, DR4, DR5, MICA/B Expression in CCA Cell Lines
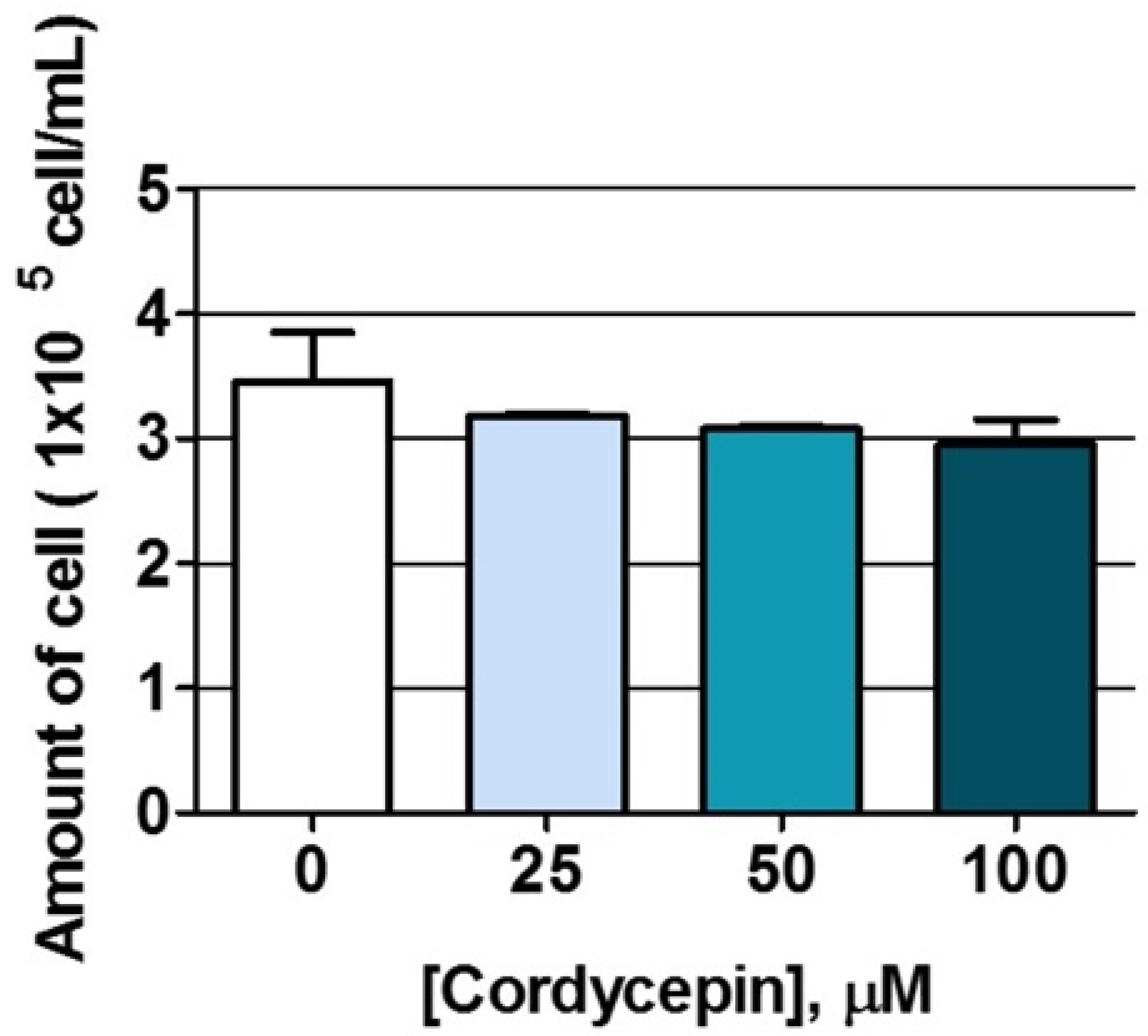
2.5. The Effects of Cordycepin on NK-92 Cell Proliferation
2.6. Cordyceps militaris Extract Combined with Cordycepin Enhanced Cytotoxicity of NK-92 to Eradicate CCA Cell Lines
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Cytotoxicity of Cordycepin on CCA Cell Lines
4.3. Killing Assay
4.4. Quantitative Real-Time Reverse Transcription PCR (qRT-PCR)
4.5. Trypan Blue Exclusion
4.6. Plant Extraction
4.7. High Performance Liquid Chromatography (HPLC) of the Extracts
4.8. The Cytotoxicity Assay of C. Militaris and I. Tenuipes Extracts on CCA Cell Lines
4.9. The Killing Assay of NK Cell Enhancing with C. Militaris and I. Tenuipes Extracts
4.10. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rizvi, S.; Khan, S.A.; Hallemeier, C.L.; Kelley, R.K.; Gores, G.J. Cholangiocarcinoma-evolving concepts and therapeutic strategies. Nat. Rev. Clin. Oncol. 2018, 15, 95–111. [Google Scholar] [CrossRef]
- Khan, S.A.; Tavolari, S.; Brandi, G. Cholangiocarcinoma: Epidemiology and risk factors. Liver Int. 2019, 39 (Suppl. S1), 19–31. [Google Scholar] [CrossRef]
- Alsaleh, M.; Leftley, Z.; Barbera, T.A.; Sithithaworn, P.; Khuntikeo, N.; Loilome, W.; Yongvanit, P.; Cox, I.J.; Chamodol, N.; Syms, R.R.; et al. Cholangiocarcinoma: A guide for the nonspecialist. Int. J. Gen. Med. 2019, 12, 13–23. [Google Scholar] [CrossRef]
- Kambakamba, P.; DeOliveira, M.L. Perihilar cholangiocarcinoma: Paradigms of surgical management. Am. J. Surg. 2014, 208, 563–570. [Google Scholar] [CrossRef]
- Butthongkomvong, K.; Sirachainan, E.; Jhankumpha, S.; Kumdang, S.; Sukhontharot, O.U. Treatment outcome of palliative chemotherapy in inoperable cholangiocarcinoma in Thailand. Asian Pac. J. Cancer Prev. 2013, 14, 3565–3568. [Google Scholar] [CrossRef][Green Version]
- Esfahani, K.; Roudaia, L.; Buhlaiga, N.; del Rincon, S.V.; Papneja, N.; Miller, W.H., Jr. A review of cancer immunotherapy: From the past, to the present, to the future. Curr. Oncol. 2020, 27 (Suppl. S2), S87–S97. [Google Scholar] [CrossRef]
- Feins, S.; Kong, W.; Williams, E.F.; Milone, M.C.; Fraietta, J.A. An introduction to chimeric antigen receptor (CAR) T-cell immunotherapy for human cancer. Am. J. Hematol. 2019, 94, S3–S9. [Google Scholar] [CrossRef]
- Rosenberg, J.; Huang, J. CD8+ T cells and NK cells: Parallel and complementary soldiers of immunotherapy. Curr. Opin. Chem. Eng. 2018, 19, 9–20. [Google Scholar] [CrossRef]
- Liu, S.; Galat, V.; Galat, Y.; Lee, Y.K.A.; Wainwright, D.; Wu, J. NK cell-based cancer immunotherapy: From basic biology to clinical development. J. Hematol. Oncol. 2021, 14, 7. [Google Scholar] [CrossRef]
- Boyiadzis, M.; Agha, M.; Redner, R.L.; Sehgal, A.; Im, A.; Hou, J.-Z.; Farah, R.; Dorritie, K.A.; Raptis, A.; Lim, S.H.; et al. Phase 1 clinical trial of adoptive immunotherapy using “off-the-shelf” activated natural killer cells in patients with refractory and relapsed acute myeloid leukemia. Cytotherapy 2017, 19, 1225–1232. [Google Scholar] [CrossRef]
- Zhang, J.; Zheng, H.; Diao, Y. Natural killer cells and current applications of chimeric antigen receptor-modified NK-92 cells in tumor immunotherapy. Int. J. Mol. Sci. 2019, 20, 317. [Google Scholar] [CrossRef]
- Williams, B.A.; Law, A.D.; Routy, B. A phase I trial of NK-92 cells for refractory hematological malignancies relapsing after autologous hematopoietic cell transplantation shows safety and evidence of efficacy. Oncotarget 2017, 8, 89256. [Google Scholar] [CrossRef]
- Beatty, G.L.; Gladney, W.L. Immune escape mechanisms as a guide for cancer immunotherapy. Clin. Cancer Res. 2015, 21, 687–692. [Google Scholar] [CrossRef]
- Khalil, D.N.; Smith, E.L.; Brentjens, R.J.; Wolchok, J.D. The future of cancer treatment: Immunomodulation, CARs and combination immunotherapy. Nat. Rev. Clin. Oncol. 2016, 13, 394. [Google Scholar] [CrossRef]
- Wen, C.C.; Chen, H.M.; Yang, N.S. Developing Phytocompounds from Medicinal Plants as Immunomodulators. Adv. Bot. Res. 2012, 62, 197–272. [Google Scholar]
- Lee, C.T.; Huang, K.S.; Shaw, J.F.; Chen, J.R.; Kuo, W.S.; Shen, G.; Grumezescu, A.M.; Holban, A.M.; Wang, Y.T.; Wang, J.S.; et al. Trends in the Immunomodulatory Effects of Cordyceps militaris: Total Extracts, Polysaccharides and Cordycepin. Front. Pharmacol. 2020, 11, 575704. [Google Scholar] [CrossRef]
- Di Sotto, A.; Vitalone, A.; di Giacomo, S. Plant-Derived Nutraceuticals and Immune System Modulation: An Evidence-Based Overview. Vaccines 2020, 8, 468. [Google Scholar] [CrossRef]
- Jung, S.J.; Jung, E.S.; Choi, E.K.; Sin, H.S.; Ha, K.C.; Chae, S.W. Immunomodulatory effects of a mycelium extract of Cordyceps (Paecilomyces hepiali; CBG-CS-2): A randomized and double-blind clinical trial. BMC Complement. Altern. Med. 2019, 19, 77. [Google Scholar] [CrossRef]
- Jin, Y.; Meng, X.; Qiu, Z.; Su, Y.; Yu, P.; Qu, P. Anti-tumor and anti-metastatic roles of cordycepin, one bioactive compound of Cordyceps militaris. Saudi J. Biol. Sci. 2018, 25, 991–995. [Google Scholar] [CrossRef]
- Kim, H.G.; Shrestha, B.; Lim, S.Y.; Yoon, D.H.; Chang, W.C.; Shin, D.J.; Han, S.K.; Park, S.M.; Park, J.H.; Park, H.I.; et al. Cordycepin inhibits lipopolysaccharide-induced inflammation by the suppression of NF-kappaB through Akt and p38 inhibition in RAW 264.7 macrophage cells. Eur. J. Pharmacol. 2006, 545, 192–199. [Google Scholar] [CrossRef]
- Shin, S.; Moon, S.; Park, Y.; Kwon, J.; Lee, S.; Lee, C.K.; Cho, K.; Ha, N.J.; Kim, K. Role of Cordycepin and Adenosine on the Phenotypic Switch of Macrophages via Induced Anti-inflammatory Cytokines. Immune Netw. 2009, 9, 255–264. [Google Scholar] [CrossRef]
- Jeong, M.H.; Lee, C.M.; Lee, S.W.; Seo, S.Y.; Seo, M.J.; Kang, B.W.; Jeong, Y.K.; Choi, Y.J.; Yang, K.M.; Jo, W.S. Cordycepin-enriched Cordyceps militaris induces immunomodulation and tumor growth delay in mouse-derived breast cancer. Oncol. Rep. 2013, 30, 1996–2002. [Google Scholar] [CrossRef]
- Sag, D.; Ayyildiz, Z.O.; Gunalp, S.; Wingender, G. The role of TRAIL/DRs in the modulation of immune cells and responses. Cancers 2019, 11, 1469. [Google Scholar] [CrossRef]
- Yoshimoto, Y.; Kawada, M.; Ikeda, D.; Ishizuka, M. Involvement of doxorubicin-induced Fas expression in the antitumor effect of doxorubicin on Lewis lung carcinoma in vivo. Int. Immunopharmacol. 2005, 5, 281–288. [Google Scholar] [CrossRef]
- Xing, S.; de Andrade, L.F. NKG2D and MICA/B shedding: A ‘tag game’between NK cells and malignant cells. Clin. Transl. Immunol. 2020, 9, e1230. [Google Scholar] [CrossRef]
- Sripa, B.; Seubwai, W.; Vaeteewoottacharn, K.; Sawanyawisuth, K.; Silsirivanit, A.; Kaewkong, W.; Muisuk, K.; Dana, P.; Phoomak, C.; Lert-Itthiporn, W.; et al. Functional and genetic characterization of three cell lines derived from a single tumor of an Opisthorchis viverrini-associated cholangiocarcinoma patient. Hum. Cell 2020, 33, 695–708. [Google Scholar] [CrossRef]
- Thanan, R.; Kaewlert, W.; Sakonsinsiri, C.; Chaiprasert, T.; Armartmuntree, N.; Muengsaen, D.; Techasen, A.; Klanrit, P.; Lert-Itthiporn, W.; Pinlaor, S.; et al. Opposing roles of FoxA1 and FoxA3 in intrahepatic cholangiocarcinoma progression. Int. J. Mol. Sci. 2020, 21, 1796. [Google Scholar] [CrossRef]
- Thonsri, U.; Seubwai, W.; Waraasawapati, S.; Wongkham, S.; Boonmars, T.; Cha’on, U.; Wongkham, C. Antitumor Effect of Shikonin, a PKM2 Inhibitor, in Cholangiocarcinoma Cell Lines. Anticancer Res. 2020, 40, 5115–5124. [Google Scholar] [CrossRef]
- Bedard, P.L.; Hansen, A.R.; Ratain, M.J.; Siu, L.L. Tumour heterogeneity in the clinic. Nature 2013, 501, 355–364. [Google Scholar] [CrossRef]
- Dubuisson, A.; Micheau, O. Antibodies and derivatives targeting DR4 and DR5 for cancer therapy. Antibodies 2017, 6, 16. [Google Scholar] [CrossRef]
- Shirley, S.; Morizot, A.; Micheau, O. Regulating TRAIL receptor-induced cell death at the membrane: A deadly discussion. Recent Pat. Anti-Cancer Drug Discov. 2011, 6, 311–323. [Google Scholar] [CrossRef]
- Peter, M.; Hadji, A.; Murmann, A.; Brockway, S.; Putzbach, W.; Pattanayak, A.; Ceppi, P. The role of CD95 and CD95 ligand in cancer. Cell Death Differ. 2015, 22, 549–559. [Google Scholar] [CrossRef]
- Zhao, Y.; Chen, N.; Yu, Y.; Zhou, L.; Niu, C.; Liu, Y.; Tian, H.; Lv, Z.; Han, F.; Cui, J. Prognostic value of MICA/B in cancers: A systematic review and meta-analysis. Oncotarget 2017, 8, 96384–96395. [Google Scholar] [CrossRef]
- Wong, Y.Y.; Moon, A.; Duffin, R.; Barthet-Barateig, A.; Meijer, H.A.; Clemens, M.J.; de Moor, C.H. Cordycepin inhibits protein synthesis and cell adhesion through effects on signal transduction. J. Biol. Chem. 2010, 285, 2610–2621. [Google Scholar] [CrossRef]
- Hawley, S.A.; Ross, F.A.; Russell, F.M.; Atrih, A.; Lamont, D.J.; Hardie, D.G. Mechanism of Activation of AMPK by Cordycepin. Cell Chem. Biol. 2020, 27, 214–222. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, Y.; Lu, J.; Wang, Y.; Wang, J.; Meng, Q.; Lee, R.J.; Wang, D.; Teng, L. Cordycepin, a natural antineoplastic agent, induces apoptosis of breast cancer cells via caspase-dependent pathways. Nat. Prod. Commun. 2016, 11, 63–68. [Google Scholar] [CrossRef]
- Lee, S.-J.; Kim, S.-K.; Choi, W.-S.; Kim, W.-J.; Moon, S.-K. Cordycepin causes p21WAF1-mediated G2/M cell-cycle arrest by regulating c-Jun N-terminal kinase activation in human bladder cancer cells. Arch. Biochem. Biophys. 2009, 490, 103–109. [Google Scholar] [CrossRef]
- Lee, S.-J.; Moon, G.-S.; Jung, K.-H.; Kim, W.-J.; Moon, S.-K. c-Jun N-terminal kinase 1 is required for cordycepin-mediated induction of G2/M cell-cycle arrest via p21WAF1 expression in human colon cancer cells. Food Chem. Toxicol. 2010, 48, 277–283. [Google Scholar] [CrossRef]
- Wu, W.-D.; Hu, Z.-M.; Shang, M.-J.; Zhao, D.-J.; Zhang, C.-W.; Hong, D.-F.; Huang, D.-S. Cordycepin down-regulates multiple drug resistant (MDR)/HIF-1α through regulating AMPK/mTORC1 signaling in GBC-SD gallbladder cancer cells. Int. J. Mol. Sci. 2014, 15, 12778–12790. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.H.; Jeong, J.-W.; Lee, J.H.; Kim, G.-Y.; Cheong, J.; Jeong, Y.K.; Yoo, Y.H.; Choi, Y.H. Cordycepin increases sensitivity of Hep3B human hepatocellular carcinoma cells to TRAIL-mediated apoptosis by inactivating the JNK signaling pathway. Oncol. Rep. 2013, 30, 1257–1264. [Google Scholar] [CrossRef]
- Wang, C.; Mao, Z.; Wang, L.; Zhang, F.; Wu, G.; Wang, D.; Shi, J. Cordycepin inhibits cell growth and induces apoptosis in human cholangiocarcinoma. Neoplasma 2017, 64, 834–839. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Yan, W.; Lv, Y.; Wang, X.; Jin, G.; Cui, M.; Lin, Z.; Ren, X. Cordycepin inhibits cancer cell proliferation and angiogenesis through a DEK interaction via ERK signaling in cholangiocarcinoma. J. Pharmacol. Exp. Ther. 2020, 373, 279–289. [Google Scholar]
- Hur, H. Chemical ingredients of Cordyceps militaris. Mycobiology 2008, 36, 233–235. [Google Scholar] [CrossRef] [PubMed]
- Thomadaki, H.; Tsiapalis, C.M.; Scorilas, A. The effect of the polyadenylation inhibitor cordycepin on human Molt-4 and Daudi leukaemia and lymphoma cell lines. Cancer Chemother. Pharmacol. 2008, 61, 703–711. [Google Scholar] [CrossRef]
- Tan, L.; Song, X.; Ren, Y.; Wang, M.; Guo, C.; Guo, D.; Gu, Y.; Li, Y.; Cao, Z.; Deng, Y. Anti-inflammatory effects of cordycepin: A review. Phytother. Res. 2021, 35, 1284–1297. [Google Scholar] [CrossRef]
- Yoon, S.Y.; Park, S.J.; Park, Y.J. The anticancer properties of cordycepin and their underlying mechanisms. Int. J. Mol. Sci. 2018, 19, 3027. [Google Scholar] [CrossRef]
- Lui, J.C.; Wong, J.W.; Suen, Y.-K.; Kwok, T.; Fung, K.; Kong, S. Cordycepin induced eryptosis in mouse erythrocytes through a Ca2+-dependent pathway without caspase-3 activation. Arch. Toxicol. 2007, 81, 859–865. [Google Scholar] [CrossRef]
- Lee, J.B.; Adrower, C.; Qin, C.; Fischer, P.M.; de Moor, C.H.; Gershkovich, P. Development of cordycepin formulations for preclinical and clinical studies. AAPS PharmSciTech 2017, 18, 3219–3226. [Google Scholar]
- Study of Cordycepin Plus Pentostatin in Patients with Refractory TdT-Positive Leukemia. Available online: https://clinicaltrials.gov/ct2/show/NCT00709215 (accessed on 22 September 2021).
- Chemotherapy with Cordycepin Plus Pentostatin in Treating Patients with Refractory Acute Lymphocytic or Chronic Myelogenous Leukemia. Available online: https://clinicaltrials.gov/ct2/show/NCT00003005 (accessed on 22 September 2021).
- Verbraecken, J.; van de Heyning, P.; de Backer, W.; van Gaal, L. Body surface area in normal-weight, overweight, and obese adults. A comparison study. Metabolism 2006, 55, 515–524. [Google Scholar]
- Sripa, B.; Leungwattanawanit, S.; Nitta, T.; Wongkham, C.; Bhudhisawasdi, V.; Puapairoj, A.; Sripa, C.; Miwa, M. Establishment and characterization of an opisthorchiasis-associated cholangiocarcinoma cell line (KKU-100). World J. Gastroenterol. 2005, 11, 3392. [Google Scholar] [CrossRef]

| Compound | CC50 of CCA Cell Lines (µM) | ||
|---|---|---|---|
| CCA-KKU-213A | CCA-KKU-100 | CCA-KKU-055 | |
| Cordycepin | 119.1 | >400 * | 135.8 |
| Extract | CC50 of CCA Cell Lines (µg/mL) | ||
|---|---|---|---|
| CCA-KKU-213A | CCA-KKU-100 | CCA-KKU-055 | |
| Cordyceps militaris | 85.48 | 122.7 | 88.88 |
| Isaria tenuipes | >500 * | >500 * | >500 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panwong, S.; Wathikthinnakon, M.; Kaewkod, T.; Sawasdee, N.; Tragoolpua, Y.; Yenchitsomanus, P.-t.; Panya, A. Cordycepin Sensitizes Cholangiocarcinoma Cells to Be Killed by Natural Killer-92 (NK-92) Cells. Molecules 2021, 26, 5973. https://doi.org/10.3390/molecules26195973
Panwong S, Wathikthinnakon M, Kaewkod T, Sawasdee N, Tragoolpua Y, Yenchitsomanus P-t, Panya A. Cordycepin Sensitizes Cholangiocarcinoma Cells to Be Killed by Natural Killer-92 (NK-92) Cells. Molecules. 2021; 26(19):5973. https://doi.org/10.3390/molecules26195973
Chicago/Turabian StylePanwong, Suthida, Methi Wathikthinnakon, Thida Kaewkod, Nunghathai Sawasdee, Yingmanee Tragoolpua, Pa-thai Yenchitsomanus, and Aussara Panya. 2021. "Cordycepin Sensitizes Cholangiocarcinoma Cells to Be Killed by Natural Killer-92 (NK-92) Cells" Molecules 26, no. 19: 5973. https://doi.org/10.3390/molecules26195973
APA StylePanwong, S., Wathikthinnakon, M., Kaewkod, T., Sawasdee, N., Tragoolpua, Y., Yenchitsomanus, P.-t., & Panya, A. (2021). Cordycepin Sensitizes Cholangiocarcinoma Cells to Be Killed by Natural Killer-92 (NK-92) Cells. Molecules, 26(19), 5973. https://doi.org/10.3390/molecules26195973







