Study of the Seasonal Variations of the Fatty Acid Profiles of Selected Macroalgae
Abstract
:1. Introduction
2. Results and Discussion
2.1. FA Content in the Macroalgae
2.1.1. Fucus spiralis
2.1.2. Bifurcaria bifurcata
2.1.3. Saccorhiza polyschides
2.1.4. Ulva lactuca
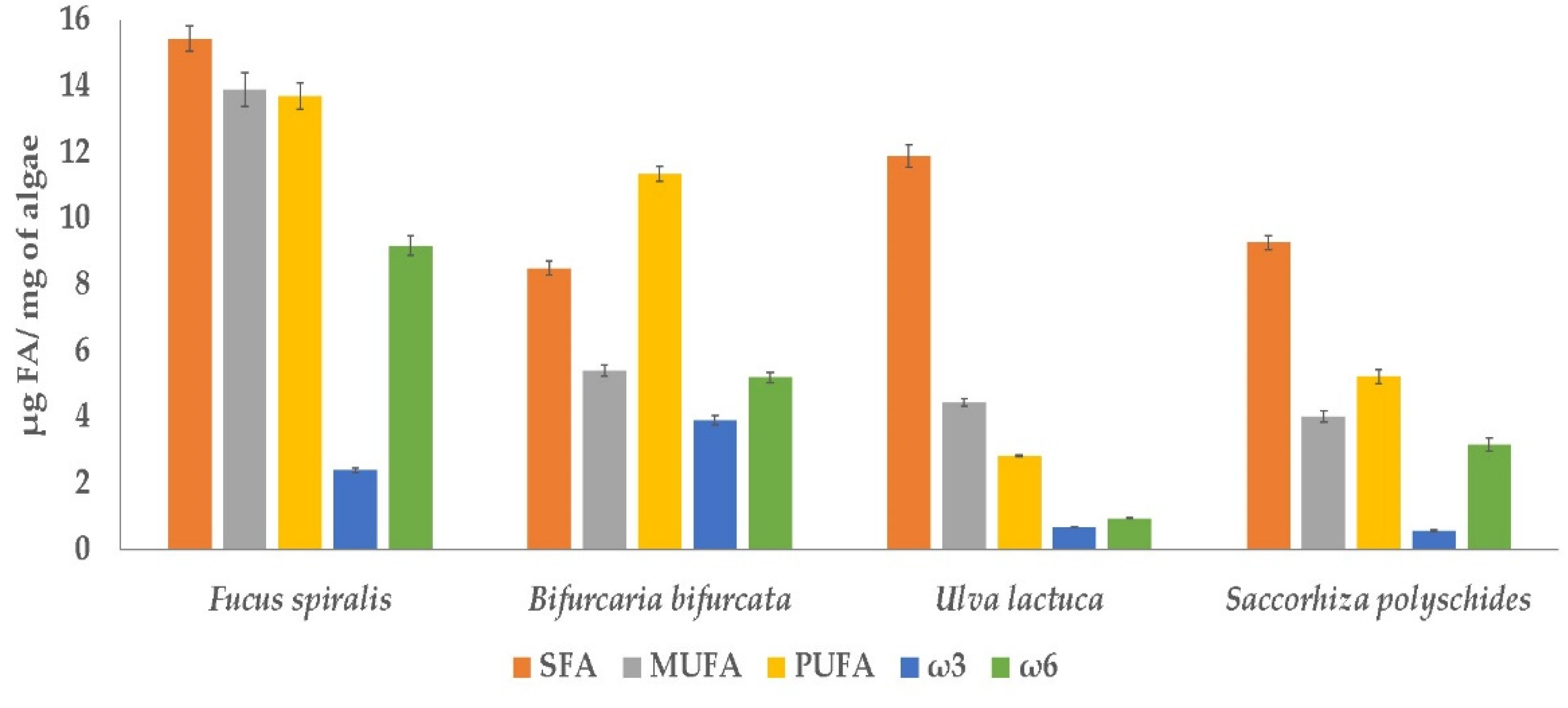
2.1.5. Comparison of the FA Profiles of the Different Macroalgae
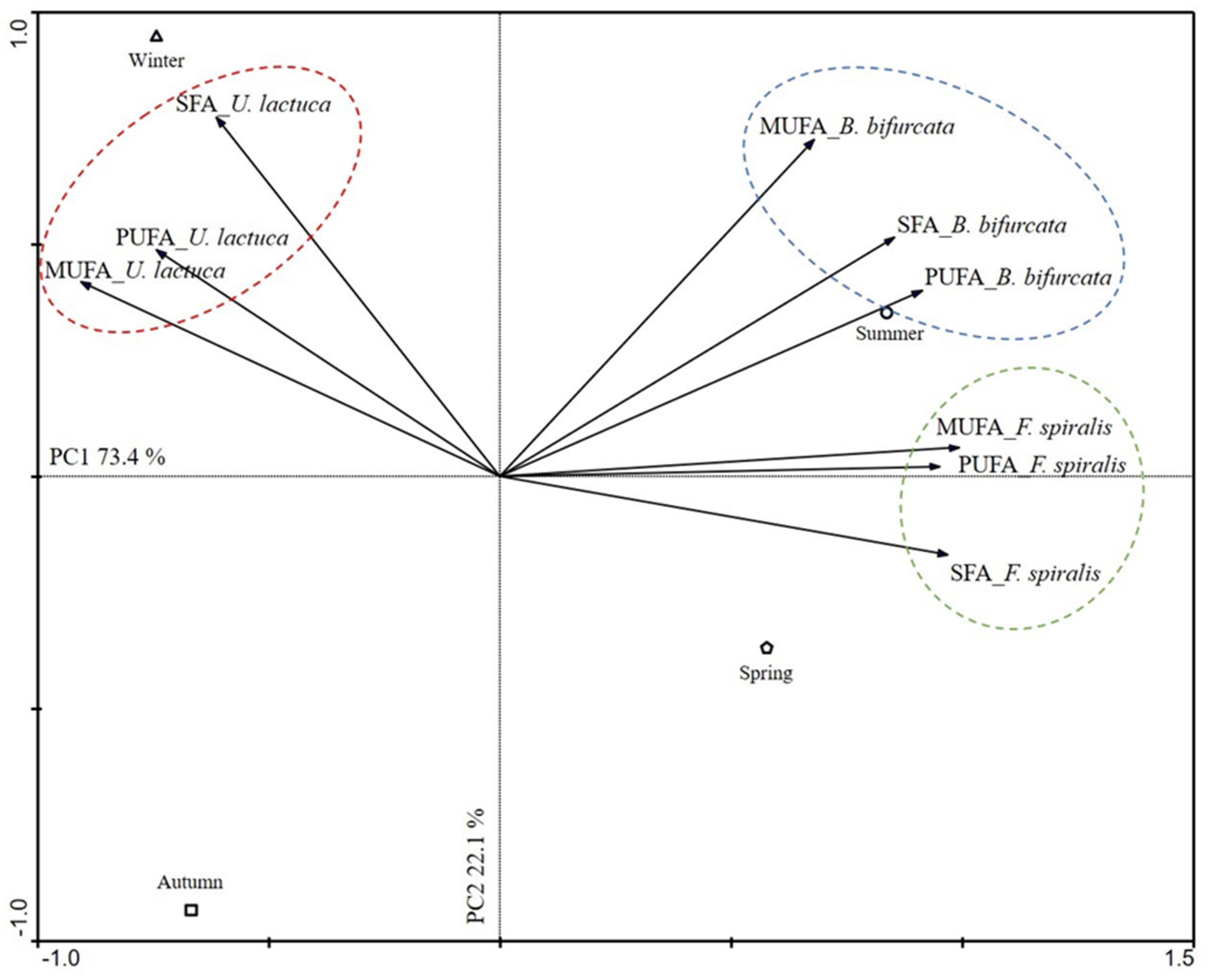
2.2. Seasonal Influence on the Production of FA
3. Materials and Methods
3.1. Reagents
3.2. Sample Collection and Preparation
3.3. Determination of the Fatty Acid Profile
3.4. Data Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Belc, N.; Smeu, I.; Macri, A.; Vallauri, D.; Flynn, K. Reformulating foods to meet current scientific knowledge about salt, sugar and fats. Trends Food Sci. Technol. 2019, 84, 25–28. [Google Scholar] [CrossRef]
- Estruch, R.; Vendrell, E.; Ruiz-León, A.M.; Casas, R.; Castro-Barquero, S.; Alvarez, X. Reformulation of pastry products to improve effects on health. Nutrients 2020, 12, 1709. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Dietetic Products, Nutrition and Allergies. Scientific opinion on dietary reference values for fats, including saturated fatty acids, polyunsaturated fatty acids, monounsaturated fatty acids, trans fatty acids, and cholesterol. EFSA J. 2010, 8, 1461. [Google Scholar] [CrossRef] [Green Version]
- López-Pedrouso, M.; Lorenzo, J.M.; Gullón, B.; Campagnol, P.C.B.; Franco, D. Novel strategy for developing healthy meat products replacing saturated fat with oleogels. Curr. Opin. Food Sci. 2021, 40, 40–45. [Google Scholar] [CrossRef]
- Sartaj, K.; Prasad, R. Production of omega-3 and omega-6 PUFA from food crops and fishes. In Nutraceutical Fatty Acids from Oleaginous Microalgae: A Human Health Perspective; Patel, A.K., Matsakas, L., Eds.; Scrivener Publishing LLC: Beverly, MA, USA, 2020; pp. 187–208. [Google Scholar]
- Visioli, F.; Poli, A. Fatty acids and cardiovascular risk. Evidence, lack of evidence, and diligence. Nutrients 2020, 12, 3782. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P.; Gene, O. An increase in the omega-6/omega-3 fatty acid ratio increases the risk for obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef] [Green Version]
- Harwood, J.L. Algae: Critical sources of very long-chain polyunsaturated fatty acids. Biomolecules 2019, 9, 708. [Google Scholar] [CrossRef] [Green Version]
- Colombo, S.M.; Rodgers, T.F.M.; Diamond, M.L.; Bazinet, R.P.; Arts, M.T. Projected declines in global DHA availability for human consumption as a result of global warming. AMBIO 2020, 49, 865–880. [Google Scholar] [CrossRef] [Green Version]
- Wells, M.L.; Potin, P.; Craigie, J.S.; Raven, J.A.; Merchant, S.S.; Helliwell, K.E.; Smith, A.G.; Camire, M.E.; Brawley, S.H. Algae as nutritional and functional food sources: Revisiting our understanding. J. Appl. Phycol. 2017, 29, 949–982. [Google Scholar] [CrossRef]
- Van Ginneken, V.J.T.; Helsper, J.P.F.G.; De Visser, W.; Van Keulen, H.; Brandenburg, W.A. Polyunsaturated fatty acids in various macroalgal species from north Atlantic and tropical seas. Lipids Health Dis. 2011, 10, 4–11. [Google Scholar] [CrossRef] [Green Version]
- Paiva, L.; Lima, E.; Neto, A.; Baptista, J. Seasonal variability of the biochemical composition and antioxidant properties of Fucus spiralis at two Azorean Islands. Mar. Drugs 2018, 16, 248. [Google Scholar] [CrossRef] [Green Version]
- Lopes, D.; Melo, T.; Rey, F.; Meneses, J.; Monteiro, F.L.; Helguero, L.A.; Abreu, M.H.; Lillebø, A.I.; Calado, R.; Domingues, M.R. Valuing bioactive lipids from green, red and brown macroalgae from aquaculture, to foster functionality and biotechnological applications. Molecules 2020, 25, 3883. [Google Scholar] [CrossRef]
- Susanto, E.; Fahmi, A.S.; Abe, M.; Hosokawa, M.; Miyashita, K. Lipids, fatty acids, and fucoxanthin content from temperate and tropical brown seaweeds. Aquat. Procedia 2016, 7, 66–75. [Google Scholar] [CrossRef]
- Barbosa, M.; Fernandes, F.; Pereira, D.M.; Azevedo, I.C.; Sousa-Pinto, I.; Andrade, P.B.; Valentão, P. Fatty acid patterns of the kelps Saccharina latissima, Saccorhiza polyschides and Laminaria ochroleuca: Influence of changing environmental conditions. Arab. J. Chem. 2020, 13, 45–58. [Google Scholar] [CrossRef]
- Seca, A.M.L.; Gouveia, V.L.M.; Carmo Barreto, M.; Silva, A.M.S.; Pinto, D.C.G.A. Comparative study by GC-MS and chemometrics on the chemical and nutritional profile of Fucus spiralis L. juvenile and mature life-cycle phases. J. Appl. Phycol. 2018, 30, 2539–2548. [Google Scholar] [CrossRef]
- Francisco, J.; Horta, A.; Pedrosa, R.; Afonso, C.; Cardoso, C.; Bandarra, N.M.; Gil, M.M. Bioaccessibility of antioxidants and fatty acids from Fucus spiralis. Foods 2020, 9, 440. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silva, G.; Pereira, R.B.; Valentão, P.; Andrade, P.B.; Sousa, C. Distinct fatty acid profile of ten brown macroalgae. Braz. J. Pharmacogn. 2013, 23, 608–613. [Google Scholar] [CrossRef] [Green Version]
- Fernandes, F.; Barbosa, M.; Pereira, D.M.; Sousa-Pinto, I.; Valentão, P.; Azevedo, I.C.; Andrade, P.B. Chemical profiling of edible seaweed (Ochrophyta) extracts and assessment of their in vitro effects on cell-free enzyme systems and on the viability of glutamate-injured SH-SY5Y cells. Food Chem. Toxicol. 2018, 116, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Grozdanic, N.; Kosanic, M.; Zdunic, G.; Savikin, K.; Etahiri, S.; Assobhei, O.; Benba, J.; Petovic, S.; Matic, I.Z.; Stanojkovic, T.P.; et al. Fucus spiralis extract and fractions: Anticancer and pharmacological potentials. J. Balk. Union Oncol. 2020, 25, 1219–1229. [Google Scholar]
- Peinado, I.; Girón, J.; Koutsidis, G.; Ames, J.M. Chemical composition, antioxidant activity and sensory evaluation of five different species of brown edible seaweeds. Food Res. Int. 2014, 66, 36–44. [Google Scholar] [CrossRef] [Green Version]
- Paiva, L.; Lima, E.; Patarra, R.F.; Neto, A.I.; Baptista, J. Edible Azorean macroalgae as source of rich nutrients with impact on human health. Food Chem. 2014, 164, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Patarra, R.F.; Leite, J.; Pereira, R.; Baptista, J.; Neto, A.I. Fatty acid composition of selected macrophytes. Nat. Prod. Res. 2013, 27, 665–669. [Google Scholar] [CrossRef]
- Niccolai, A.; Chini Zittelli, G.; Rodolfi, L.; Biondi, N.; Tredici, M.R. Microalgae of interest as food source: Biochemical composition and digestibility. Algal Res. 2019, 42, 101617. [Google Scholar] [CrossRef]
- Belattmania, Z.; Engelen, A.H.; Pereira, H.; Serrão, E.A.; Custódio, L.; Varela, J.C.; Zrid, R.; Reani, A.; Sabour, B. Fatty acid composition and nutraceutical perspectives of brown seaweeds from the Atlantic coast of Morocco. Int. Food Res. J. 2018, 25, 1520–1527. [Google Scholar]
- Lorenzo, J.M.; Agregán, R.; Munekata, P.E.S.; Franco, D.; Carballo, J.; Şahin, S.; Lacomba, R.; Barba, F.J. Proximate composition and nutritional value of three macroalgae: Ascophyllum nodosum, Fucus vesiculosus and Bifurcaria bifurcata. Mar. Drugs 2017, 15, 360. [Google Scholar] [CrossRef] [Green Version]
- Alves, C.; Pinteus, S.; Simões, T.; Horta, A.; Silva, J.; Tecelão, C.; Pedrosa, R. Bifurcaria bifurcata : A key macro-alga as a source of bioactive compounds and functional ingredients. Int. J. Food Sci. Technol. 2016, 51, 1638–1646. [Google Scholar] [CrossRef]
- Santos, S.A.O.; Trindade, S.S.; Oliveira, C.S.D.; Parreira, P.; Rosa, D.; Duarte, M.F.; Ferreira, I.; Cruz, M.T.; Rego, A.M.; Abreu, M.H.; et al. Lipophilic fraction of cultivated Bifurcaria bifurcata R. Ross: Detailed composition and in vitro prospection of current challenging bioactive properties. Mar. Drugs 2017, 15, 340. [Google Scholar] [CrossRef] [Green Version]
- Santos, F.; Monteiro, J.P.; Duarte, D.; Melo, T.; Lopes, D.; da Costa, E.; Domingues, M.R. Unraveling the lipidome and antioxidant activity of native Bifurcaria bifurcata and invasive Sargassum muticum seaweeds: A lipid perspective on how systemic intrusion may present an opportunity. Antioxidants 2020, 9, 642. [Google Scholar] [CrossRef]
- Rodrigues, D.; Freitas, A.C.; Pereira, L.; Rocha-Santos, T.A.P.; Vasconcelos, M.W.; Roriz, M.; Rodríguez-Alcalá, L.M.; Gomes, A.M.P.; Duarte, A.C. Chemical composition of red, brown and green macroalgae from Buarcos bay in Central West Coast of Portugal. Food Chem. 2015, 183, 197–207. [Google Scholar] [CrossRef]
- Schmid, M.; Stengel, D.B. Intra-thallus differentiation of fatty acid and pigment profiles in some temperate Fucales and Laminariales. J. Phycol. 2015, 51, 25–36. [Google Scholar] [CrossRef]
- Sánchez-Machado, D.I.; López-Cervantes, J.; López-Hernández, J.; Paseiro-Losada, P. Fatty acids, total lipid, protein and ash contents of processed edible seaweeds. Food Chem. 2004, 85, 439–444. [Google Scholar] [CrossRef]
- Garcia, J.; Palacios, V.; Roldán, A. Nutritional potential of four seaweed species collected in the Barbate Estuary (Gulf of Cadiz, Spain). J. Nutr. Food Sci. 2016, 06, 505. [Google Scholar] [CrossRef]
- Kostetsky, E.; Chopenko, N.; Barkina, M.; Velansky, P.; Sanina, N. Fatty acid composition and thermotropic behavior of glycolipids and other membrane lipids of Ulva lactuca (Chlorophyta) inhabiting different climatic zones. Mar. Drugs 2018, 16, 494. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCauley, J.I.; Meyer, B.J.; Winberg, P.C.; Skropeta, D. Parameters affecting the analytical profile of fatty acids in the macroalgal genus Ulva. Food Chem. 2016, 209, 332–340. [Google Scholar] [CrossRef]
- Schmid, M.; Guihéneuf, F.; Stengel, D.B. Fatty acid contents and profiles of 16 macroalgae collected from the Irish Coast at two seasons. J. Appl. Phycol. 2014, 26, 451–463. [Google Scholar] [CrossRef]
- Bikker, P.; van Krimpen, M.M.; van Wikselaar, P.; Houweling-Tan, B.; Scaccia, N.; van Hal, J.W.; Huijgen, W.J.J.; Cone, J.W.; López-Contreras, A.M. Biorefinery of the green seaweed Ulva lactuca to produce animal feed, chemicals and biofuels. J. Appl. Phycol. 2016, 28, 3511–3525. [Google Scholar] [CrossRef] [Green Version]
- Tabarsa, M.; Rezaei, M.; Ramezanpour, Z.; Waaland, J.R. Chemical compositions of the marine algae Gracilaria salicornia (Rhodophyta) and Ulva lactuca (Chlorophyta) as a potential food source. J. Sci. Food Agric. 2012, 92, 2500–2506. [Google Scholar] [CrossRef]
- Ortiz, J.; Romero, N.; Robert, P.; Araya, J.; Lopez-Hernández, J.; Bozzo, C.; Navarrete, E.; Osorio, A.; Rios, A. Dietary fiber, amino acid, fatty acid and tocopherol contents of the edible seaweeds Ulva lactuca and Durvillaea antarctica. Food Chem. 2006, 99, 98–104. [Google Scholar] [CrossRef]
- Cardoso, C.; Ripol, A.; Afonso, C.; Freire, M.; Varela, J.; Quental-Ferreira, H.; Pousão-Ferreira, P.; Bandarra, N. Fatty acid profiles of the main lipid classes of green seaweeds from fish pond aquaculture. Food Sci. Nutr. 2017, 5, 1186–1194. [Google Scholar] [CrossRef]
- Jayasinghe, G.D.T.M.; Jinadasa, B.K.K.K.; Chinthaka, S.D.M. Study on lipid content and fatty acid profile of four marine macro algae (seaweeds) collected from South East Coast of Sri Lanka. Asian J. Chem. Pharm. Sci. 2018, 3, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Sacks, F.M.; Lichtenstein, A.H.; Wu, J.H.Y.; Appel, L.J.; Creager, M.A.; Kris-Etherton, P.M.; Miller, M.; Rimm, E.B.; Rudel, L.L.; Robinson, J.G.; et al. Dietary Fats and Cardiovascular Disease: A Presidential Advisory From the American Heart Association. Circulation 2017, 136, e1–e23. [Google Scholar] [CrossRef]
- Khairy, H.M.; El-Shafay, S.M. Seasonal variations in the biochemical composition of some common seaweed species from the coast of Abu Qir Bay, Alexandria, Egypt. Oceanologia 2013, 55, 435–452. [Google Scholar] [CrossRef] [Green Version]
- Mohy El-Din, S.M. Temporal variation in chemical composition of Ulva lactuca and Corallina mediterranea. Int. J. Environ. Sci. Technol. 2019, 16, 5783–5796. [Google Scholar] [CrossRef]
- Fernández, A.; Grienke, U.; Soler-Vila, A.; Guihéneuf, F.; Stengel, D.B.; Tasdemir, D. Seasonal and geographical variations in the biochemical composition of the blue mussel (Mytilus edulis L.) from Ireland. Food Chem. 2015, 177, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Zar, J.H. Biostatistical Analysis, 5th ed.; Prentice Hall: Hoboken, NJ, USA, 2010; ISBN 0131008463. [Google Scholar]
- ter Braak, C.; Smilauer, P.N. CANOCO Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination (Version 4.5); Canoco: Ithaca, NY, USA, 2002. [Google Scholar]
- Bro, R.; Smilde, A.K. Analytical methods principal component analysis. Methods 2014, 6, 2812–2831. [Google Scholar] [CrossRef] [Green Version]
| F. spiralis | B. bifurcata | U. lactuca | S. polyschides | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Winter | Summer | Spring | Autumn | Winter | Summer | Spring | Autumn | Winter | Summer | Spring | Autumn | Summer | |
| C4:0 | 0.58 ± 0.00 | 0.58 ± 0.02 | 0.57 ± 0.03 | 0.58 ± 0.03 | 0.57 ± 0.01 | 0.57 ± 0.01 | 0.58 ± 0.03 | 0.57 ± 0.02 | 0.63 ± 0.01 a | 0.87 ± 0.04 b | 0.60 ± 0.02 a | 0.65 ± 0.03 a | 0.80 ± 0.01 |
| C6:0 | 0.53 ± 0.01 | 0.52 ± 0.01 | 0.52 ± 0.03 | 0.51 ± 0.01 | 0.72 ± 0.04 | 0.69 ± 0.02 | 0.70 ± 0.01 | 0.69 ± 0.00 | 0.60 ± 0.01 | 0.54 ± 0.01 | 0.58 ± 0.01 | 0.62 ± 0.01 | 0.51 ± 0.01 |
| C8:0 | nd | nd | nd | nd | nd | nd | nd | nd | 0.51 ± 0.01 ac | 0.64 ± 0.02 b | 0.49 ± 0.00 a | 0.53 ± 0.01 bc | 0.63 ± 0.01 |
| C10:0 | nd | nd | nd | nd | nd | nd | nd | nd | 0.73 ± 0.01 a | 0.55 ± 0.02 b | 0.54 ± 0.01 b | 0.67 ± 0.02 c | nd |
| C12:0 | 0.53 ± 0.00 | 0.50 ± 0.00 | 0.52 ± 0.00 | 0.53 ± 0.01 | nd | nd | nd | nd | 0.70 ± 0.02 a | 0.54 ± 0.00 bc | 0.53 ± 0.00 b | 0.65 ± 0.03 ac | 0.53 ± 0.01 |
| C13:0 | nd | nd | nd | nd | nd | nd | nd | nd | 0.46 ± 0.02 a | 0.31 ± 0.02 b | 0.31 ± 0.01 b | 0.45 ± 0.03 a | 0.31 ± 0.01 |
| C14:0 | 2.74 ± 0.03 | 4.01 ± 0.25 | 3.87 ± 0.27 | 2.93 ± 0.47 | 0.96 ± 0.04 a | 1.06 ± 0.03 b | 0.97 ± 0.04 a | 0.93 ± 0.03 a | 0.66 ± 0.00 a | 0.64 ± 0.01 ab | 0.53 ± 0.01 c | 0.60 ± 0.03 bc | 0.89 ± 0.04 |
| C15:0 | 0.30 ± 0.00 a | 0.33 ± 0.01 ab | 0.34 ± 0.01 b | 0.31 ± 0.02 a | 0.24 ± 0.01 | 0.25 ± 0.01 | 0.24 ± 0.00 | 0.22 ± 0.01 | 0.39 ± 0.01 c | 0.28 ± 0.01 d | 0.28 ± 0.00 d | 0.39 ± 0.01 c | 0.27 ± 0.01 |
| C16:0 | 4.38 ± 0.05 a | 5.92 ± 0.27 ab | 6.41 ± 0.33 b | 4.66 ± 0.80 a | 3.50 ± 0.25 ce | 3.93 ± 0.21 d | 3.72 ± 0.25 de | 3.31 ± 0.14 c | 6.20 ± 0.32 f | 5.58 ± 0.33 g | 5.58 ± 0.30 g | 5.32 ± 0.32 g | 3.63 ± 0.19 |
| C17:0 | 0.12 ± 0.00 a | 0.16 ± 0.00 b | 0.15 ± 0.01 b | 0.13 ± 0.00 a | 0.16 ± 0.01 cd | 0.18 ± 0.03 c | 0.15 ± 0.00 d | 0.15 ± 0.00 d | nd | nd | nd | nd | 0.12 ± 0.00 |
| C18:0 | 0.56 ± 0.01 a | 0.95 ± 0.02 b | 0.79 ± 0.03 c | 0.59 ± 0.03 a | 0.48 ± 0.01 | 0.49 ± 0.01 | 0.48 ± 0.00 | 0.47 ± 0.02 | 0.49 ± 0.00 | 0.49 ± 0.01 | 0.48 ± 0.01 | 0.48 ± 0.01 | 0.51 ± 0.01 |
| C20:0 | 1.97 ± 0.02 | 1.94 ± 0.09 | 2.31 ± 0.10 | 1.98 ± 0.34 | nd | nd | nd | nd | nd | nd | nd | nd | 0.87 ± 0.05 |
| C21:0 | nd | nd | nd | nd | nd | nd | nd | nd | 0.22 ± 0.00 | 0.22 ± 0.00 | 0.22 ± 0.00 | 0.22 ± 0.00 | 0.21 ± 0.00 |
| C22:0 | nd | nd | nd | nd | 0.69 ± 0.01 | 0.69 ± 0.04 | 0.69 ± 0.00 | 0.65 ± 0.05 | 0.91 ± 0.03 a | 0.83 ± 0.02 b | 0.86 ± 0.02 b | 0.86 ± 0.02 b | nd |
| C23:0 | nd | nd | nd | nd | 0.20 ± 0.00 | 0.19 ± 0.01 | 0.20 ± 0.01 | 0.19 ± 0.01 | nd | nd | nd | nd | nd |
| C24:0 | 0.46 ± 0.00 a | 0.53 ± 0.01 b | 0.50 ± 0.01 c | 0.47 ± 0.01 d | 0.48 ± 0.01 | 0.46 ± 0.02 | 0.46 ± 0.01 | 0.46 ± 0.02 | 0.44 ± 0.00 e | 0.42 ± 0.00 f | 0.42 ± 0.00 f | 0.43 ± 0.01 e | nd |
| ΣSFA | 12.16 ± 0.07 | 15.44 ± 0.38 | 15.99 ± 0.44 | 12.69 ± 0.99 | 8.00 ± 0.26 | 8.51 ± 0.22 | 8.18 ± 0.25 | 7.64 ± 0.16 | 12.94 ± 0.32 | 11.89 ± 0.33 | 11.41 ± 0.30 | 11.87 ± 0.33 | 9.28 ± 0.20 |
| C14:1 | 0.24 ± 0.00 a | 0.26 ± 0.00 b | 0.25 ± 0.00 c | 0.24 ± 0.00 a | 0.32 ± 0.02 | 0.36 ± 0.04 | 0.33 ± 0.01 | 0.30 ± 0.00 | 0.33 ± 0.01 d | 0.25 ± 0.02 e | 0.26 ± 0.00 de | 0.32 ± 0.00 d | 0.26 ± 0.01 |
| C15:1 | 0.30 ± 0.01 a | 0.27 ± 0.00 b | 0.29 ± 0.00 c | 0.29 ± 0.01 ac | 0.27 ± 0.00 | 0.27 ± 0.00 | 0.27 ± 0.00 | 0.27 ± 0.01 | 0.41 ± 0.00 g | 0.28 ± 0.00 h | 0.29 ± 0.00 gh | 0.35 ± 0.03 g | 0.28 ± 0.00 |
| C16:1 | 0.69 ± 0.01 a | 0.81 ± 0.02 ab | 0.87 ± 0.04 b | 0.68 ± 0.09 a | 0.89 ± 0.05 | 0.85 ± 0.05 | 0.85 ± 0.05 | 0.83 ± 0.02 | 0.99 ± 0.03 a | 0.90 ± 0.07 ab | 0.65 ± 0.02 c | 0.87 ± 0.04 b | 0.91 ± 0.05 |
| C17:1 | 0.25 ± 0.00 a | 0.28 ± 0.00 b | 0.28 ± 0.01 b | 0.25 ± 0.01 a | 0.22 ± 0.00 c | 0.25 ± 0.01 d | 0.23 ± 0.00 e | 0.21 ± 0.01 c | 0.24 ± 0.00 f | 0.22 ± 0.00 g | 0.23 ± 0.00 h | 0.24 ± 0.00 f | nd |
| C18:1ω9t | 0.22 ± 0.00 a | 0.24 ± 0.00 ab | 0.29 ± 0.01 b | 0.22 ± 0.00 a | 0.24 ± 0.00 c | 0.26 ± 0.01 d | 0.23 ± 0.00 c | 0.23 ± 0.01 c | 0.41 ± 0.01 e | 0.23 ± 0.00 f | 0.28 ± 0.00 g | 0.28 ± 0.00 g | nd |
| C18:1ω9c | 5.09 ± 0.05 a | 10.98 0.52 b | 8.71 0.75 c | 5.19 0.79 a | 2.33 0.17 | 2.40 0.14 | 2.27 0.18 | 2.22 0.10 | 0.53 ± 0.01 de | 0.78 ± 0.04 d | 0.81 ± 0.02 d | 0.51 ± 0.01 e | 1.79 0.16 |
| C18:1ω7 | 0.24 ± 0.00 a | 0.26 ± 0.00 b | 0.28 ± 0.01 c | 0.23 ± 0.00 a | 0.28 ± 0.00 | 0.28 ± 0.01 | 0.31 ± 0.03 | 0.30 ± 0.01 | 2.49 0.12 d | 1.13 ± 0.08 e | 1.26 ± 0.03 e | 1.92 0.11 f | 0.57 ± 0.03 |
| C20:1ω9 | nd | nd | nd | nd | nd | nd | nd | nd | 0.22 ± 0.00 a | 0.22 ± 0.01 b | 0.23 ± 0.00 c | 0.22 ± 0.00 d | nd |
| C24:1ω9 | 0.58 ± 0.02 | 0.52 ± 0.02 | 0.56 ± 0.02 | 0.57 ± 0.05 | 0.30 ± 0.01 | 0.33 ± 0.05 | 0.30 ± 0.01 | 0.30 ± 0.01 | 0.25 ± 0.01 a | 0.22 ± 0.00 b | 0.25 ± 0.01 a | 0.27 ± 0.02 a | nd |
| C22:1ω9 | 0.29 ± 0.00 ab | 0.28 ± 0.00 a | 0.31 ± 0.01 b | 0.27 ± 0.01 a | 0.41 ± 0.02 cd | 0.43 ± 0.01 d | 0.43 ± 0.01 c | 0.36 ± 0.01 e | 0.25 ± 0.00 f | 0.21 ± 0.00 g | 0.22 ± 0.00 h | 0.22 ± 0.00 i | 0.23 ± 0.01 |
| ΣMUFA | 7.90 ± 0.06 | 13.90 ± 0.52 | 11.84 ± 0.75 | 7.96 ± 0.79 | 5.26 ± 0.18 | 5.42 ± 0.17 | 5.23 ± 0.19 | 5.01 ± 0.10 | 6.12 ± 0.13 | 4.44 ± 0.11 | 4.48 ± 0.04 | 5.20 ± 0.13 | 4.03 ± 0.17 |
| C18:3ω3 (ALA) | nd | nd | nd | nd | 1.03 ± 0.07 a | 1.26 ± 0.10 b | 1.10 ± 0.07 c | 0.95 ± 0.05 ac | 0.74 ± 0.02 d | 0.34 ± 0.01 e | 0.43 ± 0.01 f | 0.47 ± 0.01 g | nd |
| C18:5ω3 | nd | nd | nd | nd | 0.17 ± 0.00 | 0.17 ± 0.01 | 0.17 ± 0.00 | 0.17 ± 0.00 | 0.21 ± 0.01 a | 0.19 ± 0.00 b | 0.21 ± 0.00 a | 0.20 ± 0.01 a | 0.16 ± 0.00 |
| C20:4ω3 | 0.85 ± 0.01 | 0.82 ± 0.01 | 0.88 ± 0.02 | 0.84 ± 0.03 | 0.74 ± 0.02 | 0.74 ± 0.05 | 0.72 ± 0.01 | 0.70 ± 0.02 | nd | nd | nd | nd | nd |
| C20:5ω3 (EPA) | 1.58 ± 0.01 | 1.58 ± 0.07 | 1.89 ± 0.09 | 1.40 ± 0.23 | 0.60 ± 0.02 a | 0.71 ± 0.02 b | 0.86 ± 0.05 c | 0.49 ± 0.01 d | nd | nd | nd | nd | 0.41 ± 0.02 |
| C21:5ω3 | nd | nd | nd | nd | 0.38 ± 0.01 | 0.39 ± 0.07 | 0.37 ± 0.02 | 0.34 ± 0.01 | nd | nd | nd | nd | nd |
| C22:5ω3 (DPA) | nd | nd | nd | nd | 0.39 ± 0.02 | 0.46 ± 0.07 | 0.40 ± 0.01 | 0.36 ± 0.01 | 0.22 ± 0.00 a | 0.16 ± 0.00 b | 0.17 ± 0.00 c | 0.18 ± 0.00 d | nd |
| C22:6ω3 (DHA) | nd | nd | nd | nd | 0.17 ± 0.01 | 0.18 ± 0.01 | 0.17 ± 0.00 | 0.16 ± 0.01 | nd | nd | nd | nd | nd |
| Σ ω3 | 2.43 ± 0.02 | 2.40 ± 0.07 | 2.77 ± 0.09 | 2.24 ± 0.23 | 3.49 ± 0.08 | 3.91 ± 0.15 | 3.79 ± 0.09 | 3.16 ± 0.05 | 1.16 ± 0.02 | 0.69 ± 0.01 | 0.81 ± 0.01 | 0.85 ± 0.02 | 0.57 ± 0.02 |
| C18:2ω6t (LA) | nd | 0.24 ± 0.00 | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd |
| C18:2ω6c (LA) | 1.74 ± 0.00 a | 2.19 ± 0.10 ab | 2.46 ± 0.15 b | 1.71 ± 0.29 a | 0.51 ± 0.02 c | 0.68 ± 0.03 d | 0.61 ± 0.03 e | 0.50 ± 0.02 c | 0.46 ± 0.02 f | 0.49 ± 0.02 f | 0.56 ± 0.01 g | 0.38 ± 0.01 h | 0.61 ± 0.05 |
| C18:3ω6 (GLA) | 0.31 ± 0.00 a | 0.36 ± 0.01 b | 0.35 ± 0.01 b | 0.30 ± 0.01 a | 0.26 ± 0.01 | 0.29 ± 0.03 | 0.25 ± 0.01 | 0.26 ± 0.01 | 0.22 ± 0.00 c | 0.22 ± 0.00 c | 0.23 ± 0.00 d | 0.22 ± 0.00 e | 0.24 ± 0.01 |
| C20:3ω6 | 0.34 ± 0.00 a | 0.45 ± 0.01 b | 0.45 ± 0.02 b | 0.34 ± 0.02 a | 0.45 ± 0.01 cd | 0.47 ± 0.01 c | 0.47 ± 0.02 c | 0.43 ± 0.02 d | nd | nd | nd | nd | 0.26 ± 0.01 |
| C20:4ω6 (AA) | 4.21 ± 0.04 a | 5.94 ± 0.28 b | 5.61 ± 0.35 ab | 4.22 ± 0.61 a | 2.63 ± 0.15 c | 3.10 ± 0.15 d | 2.94 ± 0.15 d | 2.45 ± 0.11 c | nd | nd | nd | nd | 2.07 ± 0.19 |
| C22:3ω6 | nd | nd | nd | nd | 0.33 ± 0.02 ab | 0.36 ± 0.02 c | 0.35 ± 0.01 ac | 0.31 ± 0.01 b | nd | 0.24 ± 0.01 a | 0.22 ± 0.00 b | 0.23 ± 0.01 ab | nd |
| C22:5ω6 | nd | nd | nd | nd | 0.27 ± 0.02 | 0.30 ± 0.03 | 0.28 ± 0.01 | 0.25 ± 0.00 | nd | nd | nd | nd | nd |
| Σ ω6 | 6.60 ± 0.04 | 9.18 ± 0.30 | 8.87 ± 0.38 | 6.56 ± 0.68 | 4.45 ± 0.15 | 5.21 ± 0.16 | 4.91 ± 0.16 | 4.20 ± 0.11 | 0.69 ± 0.02 | 0.95 ± 0.03 | 1.01 ± 0.01 | 0.83 ± 0.02 | 3.18 ± 0.20 |
| C16:2ω4 | 0.27 ± 0.01 a | 0.25 ± 0.00 b | 0.27 ± 0.00 a | 0.28 ± 0.01 c | 0.25 ± 0.00 | 0.25 ± 0.01 | 0.26 ± 0.00 | 0.25 ± 0.01 | 0.46 ± 0.01 d | 0.29 ± 0.00 e | 0.30 ± 0.00 e | 0.41 ± 0.03 de | 0.27 ± 0.01 |
| C18:4 * | 1.96 ± 0.01 | 1.53 ± 0.04 | 2.00 ± 0.07 | 1.88 ± 0.26 | 1.32 ± 0.06 a | 1.37 ± 0.06 a | 1.38 ± 0.04 a | 1.17 ± 0.03 b | 1.04 ± 0.02 c | 0.66 ± 0.01 d | 0.73 ± 0.00 e | 0.75 ± 0.01 e | 1.21 ± 0.04 |
| C20:2 | 0.34 ± 0.00 a | 0.35 ± 0.01 ab | 0.42 ± 0.02 b | 0.32 ± 0.02 a | 0.36 ± 0.01 | 0.35 ± 0.02 | 0.34 ± 0.02 | 0.34 ± 0.01 | 0.23 ± 0.00 | 0.22 ± 0.00 | 0.22 ± 0.01 | 0.22 ± 0.00 | nd |
| C22:2 | nd | nd | nd | nd | 0.23 ± 0.01 a | 0.28 ± 0.00 b | 0.24 ± 0.00 abc | 0.23 ± 0.00 c | nd | nd | nd | nd | nd |
| ΣPUFA | 11.59 ± 0.27 | 13.71 ± 0.40 | 14.32 ± 0.48 | 11.28 ± 0.81 | 10.11 ± 0.18 | 11.36 ± 0.23 | 10.92 ± 0.19 | 9.35 ± 0.13 | 3.57 ± 0.04 | 2.82 ± 0.04 | 3.07 ± 0.02 | 3.06 ± 0.03 | 5.23 ± 0.20 |
| PUFA/ SFA | 0.95 ± 0.05 | 0.89 ± 0.12 | 0.90 ± 0.17 | 0.89 ± 0.43 | 1.26 ± 0.27 | 1.34 ± 0.50 | 1.33 ± 0.26 | 1.22 ± 0.23 | 0.28 ± 0.03 | 0.24 ± 0.03 | 0.27 ± 0.03 | 0.26 ± 0.04 | 0.60 ± 0.11 |
| FA | 31.65 ± 0.28 | 43.05 ± 0.75 | 42.15 ± 0.99 | 31.92 ± 1.51 | 23.37 ± 0.36 | 25.29 ± 0.36 | 24.33 ± 0.37 | 22.00 ± 0.23 | 22.63 ± 0.35 | 19.15 ± 0.35 | 18.96 ± 0.30 | 20.13 ± 0.35 | 18.54 ± 0.33 |
| ω6 ω3 | 2.72 ± 0.06 | 3.83 ± 0.34 | 3.21 ± 0.37 | 2.93 ± 0.85 | 1.28 ± 0.21 | 1.33 ± 0.41 | 1.30 ± 0.19 | 1.33 ± 0.17 | 0.59 ± 0.05 | 1.38 ± 0.09 | 1.25 ± 0.04 | 0.98 ± 0.06 | 5.60 ± 0.80 |
| h/H | 2.74 | 2.78 | 2.54 | 2.54 | 3.45 | 3.37 | 3.45 | 3.39 | 1.41 | 1.17 | 1.24 | 1.39 | 2.05 |
| DHA/ EPA | nd | nd | nd | nd | 0.28 ± 0.01 | 0.25 ± 0.01 | 0.19 ± 0.01 | 0.32 ± 0.02 | nd | nd | nd | nd | nd |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pereira, T.; Horta, A.; Barroso, S.; Mendes, S.; Gil, M.M. Study of the Seasonal Variations of the Fatty Acid Profiles of Selected Macroalgae. Molecules 2021, 26, 5807. https://doi.org/10.3390/molecules26195807
Pereira T, Horta A, Barroso S, Mendes S, Gil MM. Study of the Seasonal Variations of the Fatty Acid Profiles of Selected Macroalgae. Molecules. 2021; 26(19):5807. https://doi.org/10.3390/molecules26195807
Chicago/Turabian StylePereira, Tatiana, André Horta, Sónia Barroso, Susana Mendes, and Maria M. Gil. 2021. "Study of the Seasonal Variations of the Fatty Acid Profiles of Selected Macroalgae" Molecules 26, no. 19: 5807. https://doi.org/10.3390/molecules26195807
APA StylePereira, T., Horta, A., Barroso, S., Mendes, S., & Gil, M. M. (2021). Study of the Seasonal Variations of the Fatty Acid Profiles of Selected Macroalgae. Molecules, 26(19), 5807. https://doi.org/10.3390/molecules26195807









