Novel CBG Derivatives Can Reduce Inflammation, Pain and Obesity
Abstract
1. Introduction
2. Results
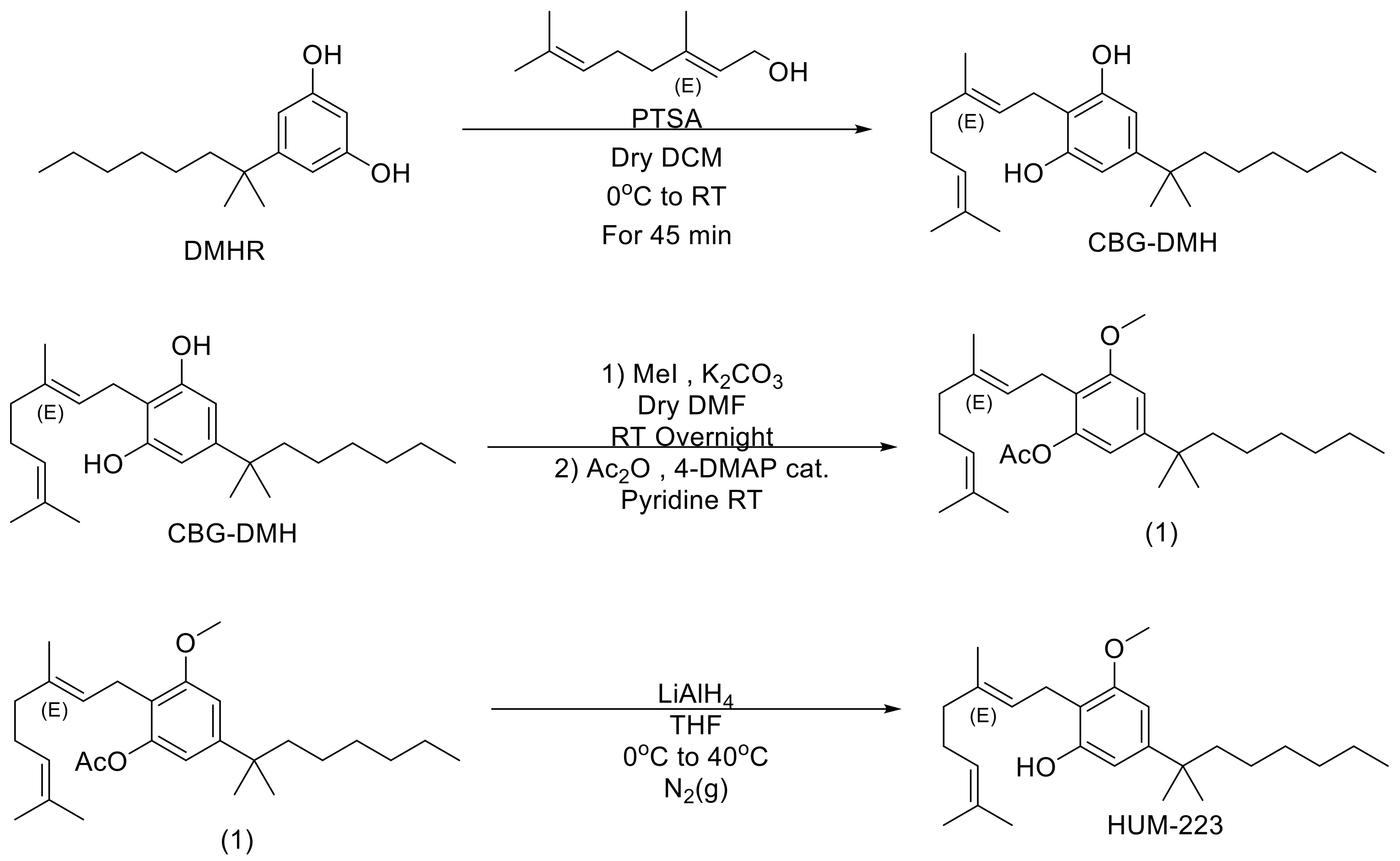
2.1. Chemistry Development and Synthesis of HUM-223, HUM-233 and HUM-234
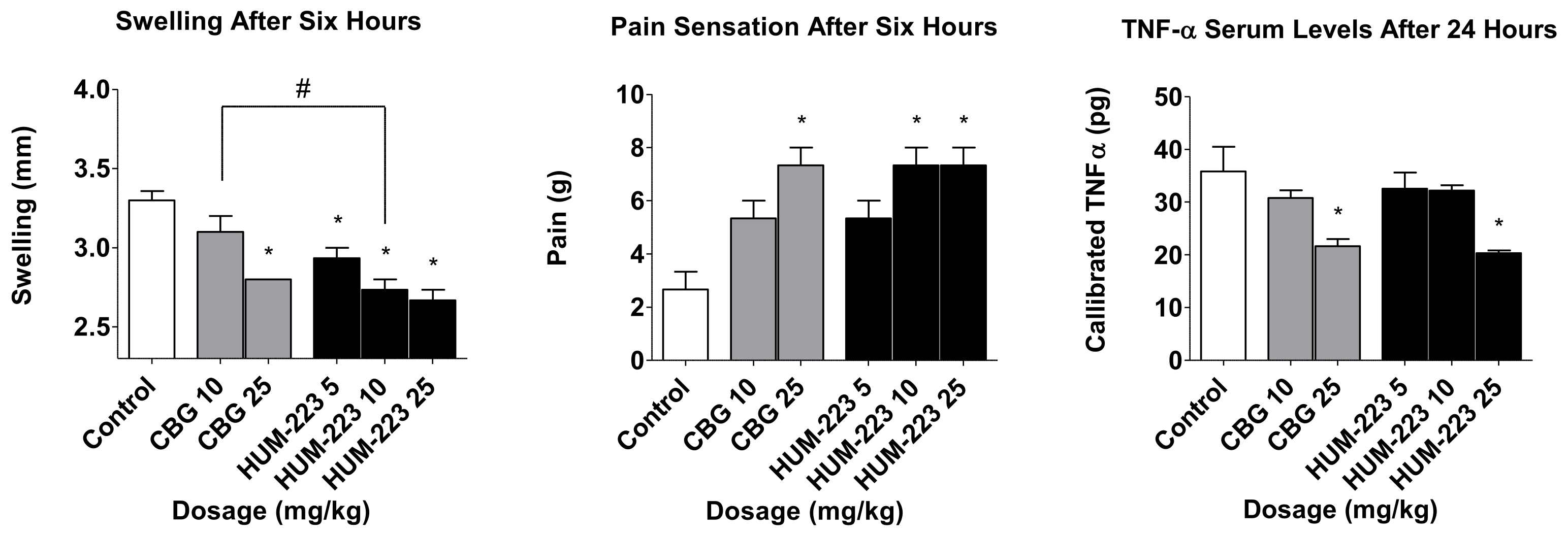
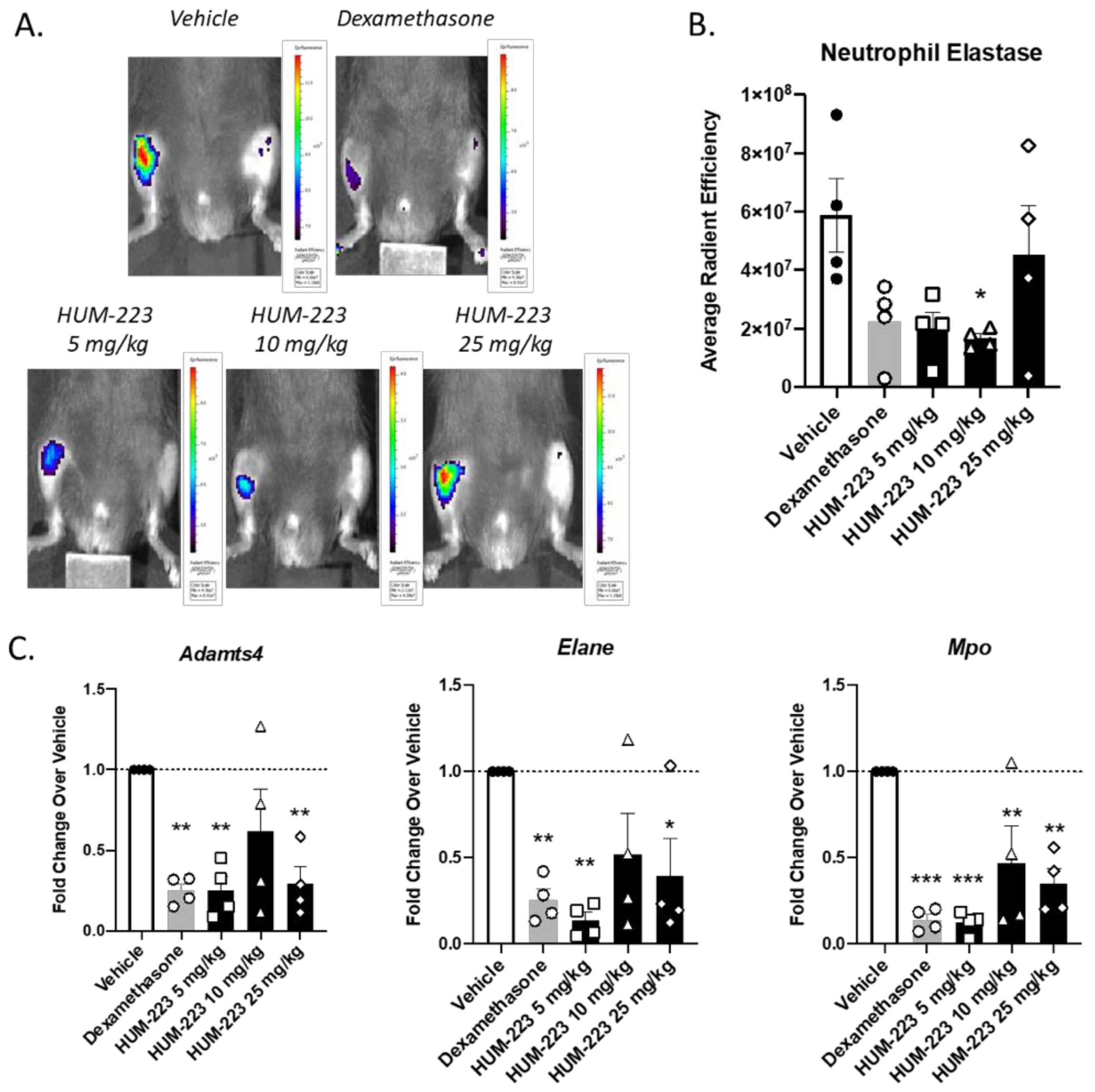
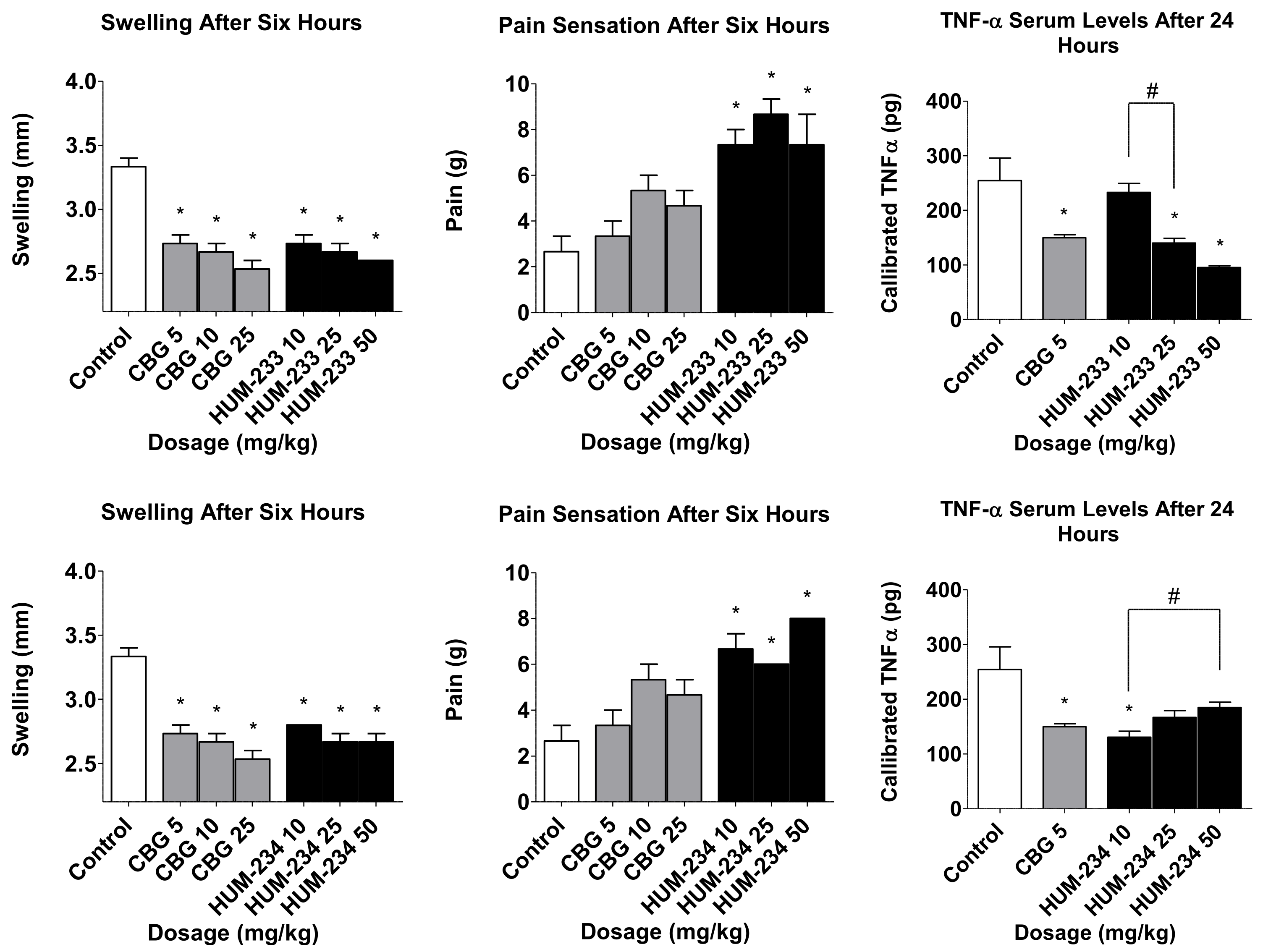
2.2. Evaluation of Anti-Inflammatory Activity
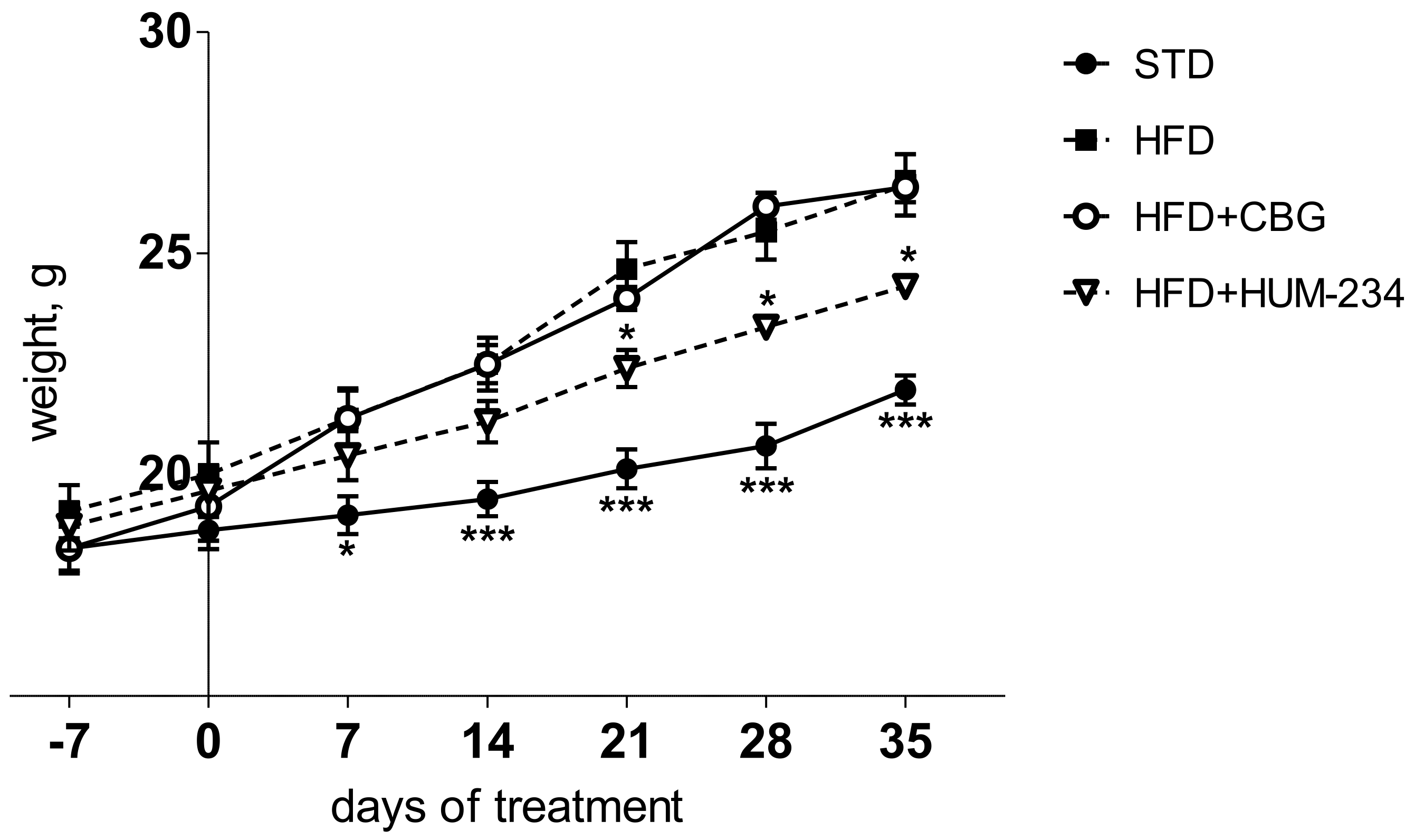
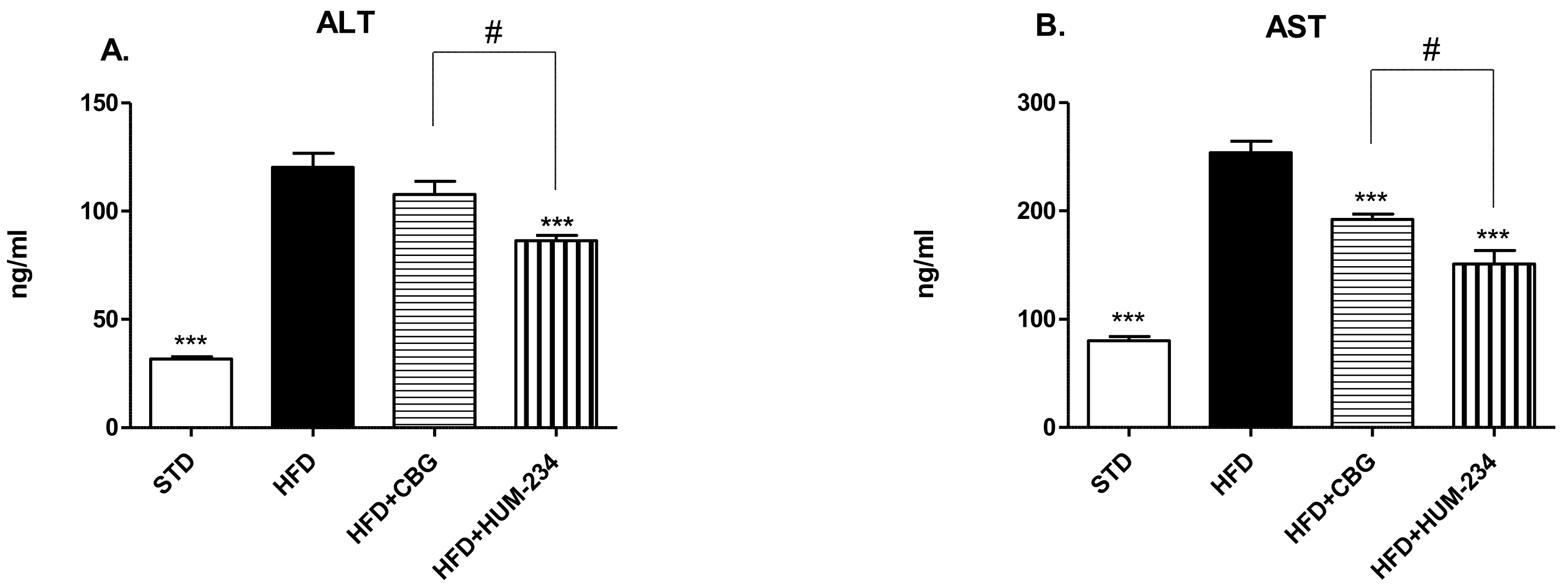
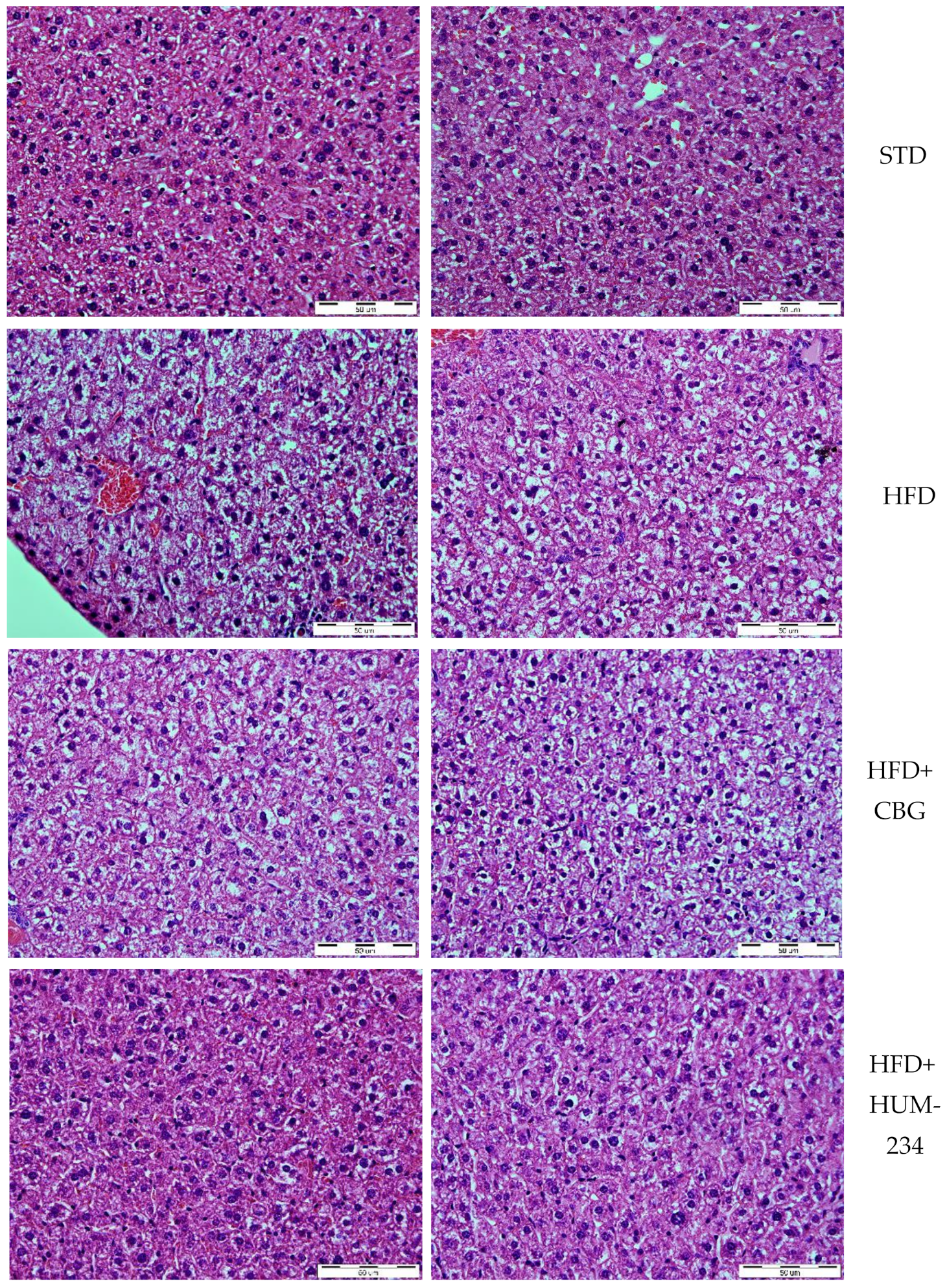
2.3. Evaluation of Effects on Weight Gain
3. Discussion
4. Materials and Methods
4.1. NMR Spectroscopy
4.2. Mass Spectrometry
4.3. Chemical Synthesis
4.3.1. 1,1-Dimethylheptyl Cannabigerol (CBG-DMH)
4.3.2. 1″,1″-Dimethylheptyl-monomethoxycannabigerol (HUM-223)
4.3.3. Monomethoxycannabigeroyl-3-morpholinopropraonate Maleate (HUM-233)
4.3.4. Monomethoxycannabigeroyl-3-morpholinoproprionate (HUM-234)
4.4. Biological Evaluation
4.4.1. Animals
4.4.2. Induction and Treatment of Paw Inflammation (Paw ZIA)
4.4.3. Evaluation of Edema
4.4.4. Pain Assay
4.4.5. Measurement of TNFα
4.4.6. Zymosan Induced Arthritis of the Knee
4.4.7. Diet-Induced Obesity
4.4.8. Liver Injury
4.4.9. Determination of ALT and AST Levels
4.5. Statistical Analyses
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Gaoni, Y.; Mechoulam, R. The structure and synthesis of cannabigerol, a new hashish constituent. Proc. Chem. Soc. 1964, 82. [Google Scholar] [CrossRef]
- Nachnani, R.; Raup-Konsavage, W.M.; Vrana, K.E. The Pharmacological Case for Cannabigerol. J. Pharmacol. Exp. Ther. 2021, 376, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Hill, K.P.; Palastro, M.D.; Johnson, B.; Ditre, J.W. Cannabis and Pain: A Clinical Review. Cannabis Cannabinoid Res. 2017, 2, 96–104. [Google Scholar] [CrossRef]
- Nichols, J.M.; Kaplan, B.L.F. Immune Responses Regulated by Cannabidiol. Cannabis Cannabinoid Res. 2020, 5, 12–31. [Google Scholar] [CrossRef]
- Gugliandolo, A.; Pollastro, F.; Grassi, G.; Bramanti, P.; Mazzon, E. In Vitro Model of Neuroinflammation: Efficacy of Cannabigerol, a Non-Psychoactive Cannabinoid. Int. J. Mol. Sci. 2018, 19, 1992. [Google Scholar] [CrossRef]
- Burgaz, S.; García, C.; Gómez-Cañas, M.; Muñoz, E.; Fernández-Ruiz, J. Development of An Oral Treatment with the PPAR-γ-Acting Cannabinoid VCE-003.2 against the Inflammation-Driven Neuronal Deterioration in Experimental Parkinson’s Disease. Molecules 2019, 24, 2702. [Google Scholar] [CrossRef] [PubMed]
- di Giacomo, V.; Chiavaroli, A.; Orlando, G.; Cataldi, A.; Rapino, M.; Di Valerio, V.; Leone, S.; Brunetti, L.; Menghini, L.; Recinella, L.; et al. Neuroprotective and Neuromodulatory Effects Induced by Cannabidiol and Cannabigerol in Rat Hypo-E22 cells and Isolated Hypothalamus. Antioxidants 2020, 9, 71. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Alonso, J.; Paraíso-Luna, J.; Navarrete, C.; del Río, C.; Cantarero, I.; Palomares, B.; Aguareles, J.; Fernández-Ruiz, J.; Bellido, M.L.; Pollastro, F.; et al. VCE-003.2, a novel cannabigerol derivative, enhances neuronal progenitor cell survival and alleviates symptomatology in murine models of Huntington’s disease. Sci. Rep. 2016, 6, 29789. [Google Scholar] [CrossRef]
- Borrelli, F.; Fasolino, I.; Romano, B.; Capasso, R.; Maiello, F.; Coppola, D.; Orlando, P.; Battista, G.; Pagano, E.; Di Marzo, V.; et al. Beneficial effect of the non-psychotropic plant cannabinoid cannabigerol on experimental inflammatory bowel disease. Biochem. Pharmacol. 2013, 85, 1306–1316. [Google Scholar] [CrossRef] [PubMed]
- Mammana, S.; Cavalli, E.; Gugliandolo, A.; Silvestro, S.; Pollastro, F.; Bramanti, P.; Mazzon, E. Could the Combination of Two Non-Psychotropic Cannabinoids Counteract Neuroinflammation? Effectiveness of Cannabidiol Associated with Cannabigerol. Medicina 2019, 55, 747. [Google Scholar] [CrossRef]
- Henley, D.; Lightman, S.; Carrell, R. Cortisol and CBG—Getting cortisol to the right place at the right time. Pharmacol. Ther. 2016, 166, 128–135. [Google Scholar] [CrossRef]
- Ruhaak, L.R.; Felth, J.; Karlsson, P.C.; Rafter, J.J.; Verpoorte, R.; Bohlin, L. Evaluation of the Cyclooxygenase Inhibiting Effects of Six Major Cannabinoids Isolated from Cannabis sativa. Biol. Pharm. Bull. 2011, 34, 774–778. [Google Scholar] [CrossRef]
- Bjerregaard, L.G.; Jensen, B.W.; Ängquist, L.; Osler, M.; Sørensen, T.I.A.; Baker, J.L. Change in Overweight from Childhood to Early Adulthood and Risk of Type 2 Diabetes. N. Engl. J. Med. 2018, 378, 1302–1312. [Google Scholar] [CrossRef] [PubMed]
- Brierley, D.I.; Samuels, J.; Duncan, M.; Whalley, B.J.; Williams, C.M. Cannabigerol is a novel, well-tolerated appetite stimulant in pre-satiated rats. Psychopharmacology 2016, 233, 3603–3613. [Google Scholar] [CrossRef]
- Cascio, M.; Gauson, L.; Stevenson, L.; Ross, R.; Pertwee, R. Evidence that the plant cannabinoid cannabigerol is a highly potent α2-adrenoceptor agonist and moderately potent 5HT1A receptor antagonist. Br. J. Pharmacol. 2010, 159, 129–141. [Google Scholar] [CrossRef]
- de Petrocellis, L.; Orlando, P.; Moriello, A.S.; Aviello, G.; Stott, C.; Izzo, A.A.; di Marzo, V. Cannabinoid actions at TRPV channels: Effects on TRPV3 and TRPV4 and their potential relevance to gastrointestinal inflammation. Acta Physiol. 2012, 204, 255–266. [Google Scholar] [CrossRef]
- Navarro, G.; Varani, K.; Reyes-Resina, I.; Sánchez de Medina, V.; Rivas-Santisteban, R.; Sánchez-Carnerero Callado, C.; Vincenzi, F.; Casano, S.; Ferreiro-Vera, C.; Canela, E.I.; et al. Cannabigerol Action at Cannabinoid CB1 and CB2 Receptors and at CB1–CB2 Heteroreceptor Complexes. Front. Pharmacol. 2018, 9, 632. [Google Scholar] [CrossRef]
- Greene, E.R.; Huang, S.; Serhan, C.N.; Panigrahy, D. Regulation of inflammation in cancer by eicosanoids. Prostagland. Other Lipid Mediat. 2011, 96, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Shachar, I.; Karin, N. The dual roles of inflammatory cytokines and chemokines in the regulation of autoimmune diseases and their clinical implications. J. Leukoc. Biol. 2013, 93, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Ertunc, M.E.; Hotamisligil, G.S. Lipid signaling and lipotoxicity in metaflammation: Indications for metabolic disease pathogenesis and treatment. J. Lipid Res. 2016, 57, 2099–2114. [Google Scholar] [CrossRef]
- Lackey, D.E.; Olefsky, J.M. Regulation of metabolism by the innate immune system. Nat. Rev. Endocrinol. 2015, 12, 15–28. [Google Scholar] [CrossRef]
- Yang, Y.; Lv, J.; Jiang, S.; Ma, Z.; Wang, D.; Hu, W.; Deng, C.; Fan, C.; Di, S.; Sun, Y.; et al. The emerging role of Toll-like receptor 4 in myocardial inflammation. Cell Death Dis. 2016, 7, e2234. [Google Scholar] [CrossRef]
- Kim, S.Y.; Solomon, D.H. Comparative safety of nonsteroidal anti-inflammatory drugs. Nat. Rev. Cardiol. 2011, 8, 193–195. [Google Scholar] [CrossRef][Green Version]
- Wehling, M. Non-steroidal anti-inflammatory drug use in chronic pain conditions with special emphasis on the elderly and patients with relevant comorbidities: Management and mitigation of risks and adverse effects. Eur. J. Clin. Pharmacol. 2014, 70, 1159–1172. [Google Scholar] [CrossRef] [PubMed]
- Heffler, E.; Madeira, L.N.G.; Ferrando, M.; Puggioni, F.; Racca, F.; Malvezzi, L.; Passalacqua, G.; Canonica, G.W. Inhaled Corticosteroids Safety and Adverse Effects in Patients with Asthma. J. Allergy Clin. Immunol. Pract. 2018, 6, 776–781. [Google Scholar] [CrossRef]
- Sarnes, E.; Crofford, L.; Watson, M.; Dennis, G.; Kan, H.; Bass, D. Incidence and US Costs of Corticosteroid-Associated Adverse Events: A Systematic Literature Review. Clin. Ther. 2011, 33, 1413–1432. [Google Scholar] [CrossRef] [PubMed]
- Krensky, A.M.; Vicenti, F.; Bennett, W.M. Immunosuppressants, tolerogens, and immunostimulants. In Goodman & Gilman’s the Pharmacological Basis of Therapeutics; Brunton, L.L., Lazo, J.S., Parker, K.L., Eds.; McGraw-Hill: New York, NY, USA, 2005; pp. 1405–1431. ISBN 0071422803. [Google Scholar]
- Bow, E.W.; Rimoldi, J.M. The Structure–Function Relationships of Classical Cannabinoids: CB1/CB2 Modulation. Perspect. Medicin. Chem. 2016, 8, PMC.S32171. [Google Scholar] [CrossRef]
- Mechoulam, R.; Kogan, N.; Gallily, R.; Breuer, A. Novel Cannabidiol Derivatives and Their Use as Anti-Inflammatory Agents. WO Patent 107879, 12 September 2008. [Google Scholar]
- Smyth, E.M.; Burke, A.; FitzGerald, G.A. Lipid-derived autacoids: Eicosanoids and platelet-activating factor. In Goodman & Gilman’s the Pharmacological Basis of Therapeutics; Brunton, L.L., Lazo, J.S., Parker, K.L., Eds.; McGraw-Hill: New York, NY, USA, 2005; pp. 653–670. ISBN 0071422803. [Google Scholar]
- Day, R.O.; Graham, G.G. The vascular effects of COX-2 selective inhibitors. Aust. Prescr. 2004, 27, 142–145. [Google Scholar] [CrossRef]
- Bally, M.; Dendukuri, N.; Rich, B.; Nadeau, L.; Helin-Salmivaara, A.; Garbe, E.; Brophy, J.M. Risk of acute myocardial infarction with NSAIDs in real world use: Bayesian meta-analysis of individual patient data. BMJ 2017, 357, j1909. [Google Scholar] [CrossRef]
- Lanas, A.; Chan, F.K.L. Peptic ulcer disease. Lancet 2017, 390, 613–624. [Google Scholar] [CrossRef]
- Brater, D.C.; Harris, C.; Redfern, J.S.; Gertz, B.J. Renal Effects of COX-2-Selective Inhibitors. Am. J. Nephrol. 2001, 21, 1–15. [Google Scholar] [CrossRef]
- Bleumink, G.S.; Feenstra, J.; Sturkenboom, M.C.J.M.; Stricker, B.H.C. Nonsteroidal Anti-Inflammatory Drugs and Heart Failure. Drugs 2003, 63, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Ahmet, A.; Ward, L.; Krishnamoorthy, P.; Mandelcorn, E.D.; Leigh, R.; Brown, J.P.; Cohen, A.; Kim, H. A practical guide to the monitoring and management of the complications of systemic corticosteroid therapy. Allergy Asthma Clin. Immunol. 2013, 9, 30. [Google Scholar] [CrossRef] [PubMed]
- Feldmann, M.; Maini, R.N. TNF defined as a therapeutic target for rheumatoid arthritis and other autoimmune diseases. Nat. Med. 2003, 9, 1245–1250. [Google Scholar] [CrossRef]
- Mechoulam, R.; Sumariwalla, P.F.; Feldmann, M.; Gallily, R. Cannabinoids in Models of Chronic Inflammatory Conditions. Phytochem. Rev. 2005, 4, 11–18. [Google Scholar] [CrossRef]
- Sumariwalla, P.F.; Gallily, R.; Tchilibon, S.; Fride, E.; Mechoulam, R.; Feldmann, M. A novel synthetic, nonpsychoactive cannabinoid acid (HU-320) with antiinflammatory properties in murine collagen-induced arthritis. Arthritis Rheum. 2004, 50, 985–998. [Google Scholar] [CrossRef]
- Pellati, F.; Borgonetti, V.; Brighenti, V.; Biagi, M.; Benvenuti, S.; Corsi, L. Cannabis sativa L. and Nonpsychoactive Cannabinoids: Their Chemistry and Role against Oxidative Stress, Inflammation, and Cancer. Biomed Res. Int. 2018, 2018, 1691428. [Google Scholar] [CrossRef]
- Malfait, A.M.; Gallily, R.; Sumariwalla, P.F.; Malik, A.S.; Andreakos, E.; Mechoulam, R.; Feldmann, M. The nonpsychoactive cannabis constituent cannabidiol is an oral anti-arthritic therapeutic in murine collagen-induced arthritis. Proc. Natl. Acad. Sci. USA 2000, 97, 9561–9566. [Google Scholar] [CrossRef]
- Gallily, R.; Yekhtin, Z.; Hanuš, L.O. Overcoming the Bell-Shaped Dose-Response of Cannabidiol by Using Cannabis Extract Enriched in Cannabidiol. Pharmacol. Pharm. 2015, 6, 75–85. [Google Scholar] [CrossRef]
- Carmona-Rivera, C.; Carlucci, P.M.; Goel, R.R.; James, E.; Brooks, S.R.; Rims, C.; Hoffmann, V.; Fox, D.A.; Buckner, J.H.; Kaplan, M.J. Neutrophil extracellular traps mediate articular cartilage damage and enhance cartilage component immunogenicity in rheumatoid arthritis. JCI Insight 2020, 5, e139388. [Google Scholar] [CrossRef]
- Rose, K.W.J.; Taye, N.; Karoulias, S.Z.; Hubmacher, D. Regulation of ADAMTS Proteases. Front. Mol. Biosci. 2021, 8, 701959. [Google Scholar] [CrossRef] [PubMed]
- Odobasic, D.; Yang, Y.; Muljadi, R.C.M.; O’Sullivan, K.M.; Kao, W.; Smith, M.; Morand, E.F.; Holdsworth, S.R. Endogenous Myeloperoxidase Is a Mediator of Joint Inflammation and Damage in Experimental Arthritis. Arthritis Rheumatol. 2014, 66, 907–917. [Google Scholar] [CrossRef] [PubMed]
- Gallily, R.; Breuer, A.; Mechoulm, R. Cyclohexenyl Compounds, Compositions Comprising Them and Uses Thereof. U.S. Patent 10,239,848, 26 March 2019. [Google Scholar]
- Meldrum, D.R.; Morris, M.A.; Gambone, J.C. Obesity pandemic: Causes, consequences, and solutions—But do we have the will? Fertil. Steril. 2017, 107, 833–839. [Google Scholar] [CrossRef]
- Teixeira, L.G.; Leonel, A.J.; Aguilar, E.C.; Batista, N.V.; Alves, A.C.; Coimbra, C.C.; Ferreira, A.V.M.; De Faria, A.M.C.; Cara, D.C.; Alvarez Leite, J.I. The combination of high-fat diet-induced obesity and chronic ulcerative colitis reciprocally exacerbates adipose tissue and colon inflammation. Lipids Health Dis. 2011, 10, 204. [Google Scholar] [CrossRef]
- Doll, S.; Paccaud, F.; Bovet, P.; Burnier, M.; Wietlisbach, V. Body mass index, abdominal adiposity and blood pressure: Consistency of their association across developing and developed countries. Int. J. Obes. 2002, 26, 48–57. [Google Scholar] [CrossRef]
- Latino-Martel, P.; Cottet, V.; Druesne-Pecollo, N.; Pierre, F.H.F.; Touillaud, M.; Touvier, M.; Vasson, M.-P.; Deschasaux, M.; Le Merdy, J.; Barrandon, E.; et al. Alcoholic beverages, obesity, physical activity and other nutritional factors, and cancer risk: A review of the evidence. Crit. Rev. Oncol. Hematol. 2016, 99, 308–323. [Google Scholar] [CrossRef]
- Suárez-Carmona, W.; Sánchez-Oliver, A.J.; González-Jurado, J.A. Pathophysiology of obesity: Current view. Rev. Chil. Nutr. 2017, 44, 226–233. [Google Scholar] [CrossRef]
- Lima, M.G.; Tardelli, V.S.; Brietzke, E.; Fidalgo, T.M. Cannabis and Inflammatory Mediators. Eur. Addict. Res. 2021, 27, 16–24. [Google Scholar] [CrossRef]
- Baek, S.H.; Yook, C.N.; Han, D.S. Boron-Trifluoride Etherate on Alumina-A Modified Lewis-Acid Reagent(V) A Convenient Single-Step Synthesis of Cannabinoids. Bull. Korean Chem. Soc. 1995, 16, 293–296. [Google Scholar]
- Vermeij, E.A.; Koenders, M.I.; Blom, A.B.; Arntz, O.J.; Bennink, M.B.; van den Berg, W.B.; van Lent, P.L.E.M.; van de Loo, F.A.J. In vivo molecular imaging of cathepsin and matrix metalloproteinase activity discriminates between arthritic and osteoarthritic processes in mice. Mol. Imaging 2014, 13, 1–10. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kogan, N.M.; Lavi, Y.; Topping, L.M.; Williams, R.O.; McCann, F.E.; Yekhtin, Z.; Feldmann, M.; Gallily, R.; Mechoulam, R. Novel CBG Derivatives Can Reduce Inflammation, Pain and Obesity. Molecules 2021, 26, 5601. https://doi.org/10.3390/molecules26185601
Kogan NM, Lavi Y, Topping LM, Williams RO, McCann FE, Yekhtin Z, Feldmann M, Gallily R, Mechoulam R. Novel CBG Derivatives Can Reduce Inflammation, Pain and Obesity. Molecules. 2021; 26(18):5601. https://doi.org/10.3390/molecules26185601
Chicago/Turabian StyleKogan, Natalya M., Yarden Lavi, Louise M. Topping, Richard. O. Williams, Fiona E. McCann, Zhanna Yekhtin, Marc Feldmann, Ruth Gallily, and Raphael Mechoulam. 2021. "Novel CBG Derivatives Can Reduce Inflammation, Pain and Obesity" Molecules 26, no. 18: 5601. https://doi.org/10.3390/molecules26185601
APA StyleKogan, N. M., Lavi, Y., Topping, L. M., Williams, R. O., McCann, F. E., Yekhtin, Z., Feldmann, M., Gallily, R., & Mechoulam, R. (2021). Novel CBG Derivatives Can Reduce Inflammation, Pain and Obesity. Molecules, 26(18), 5601. https://doi.org/10.3390/molecules26185601






