Luminescent and Magnetic Tb-MOF Flakes Deposited on Silicon
Abstract
:1. Introduction
2. Results
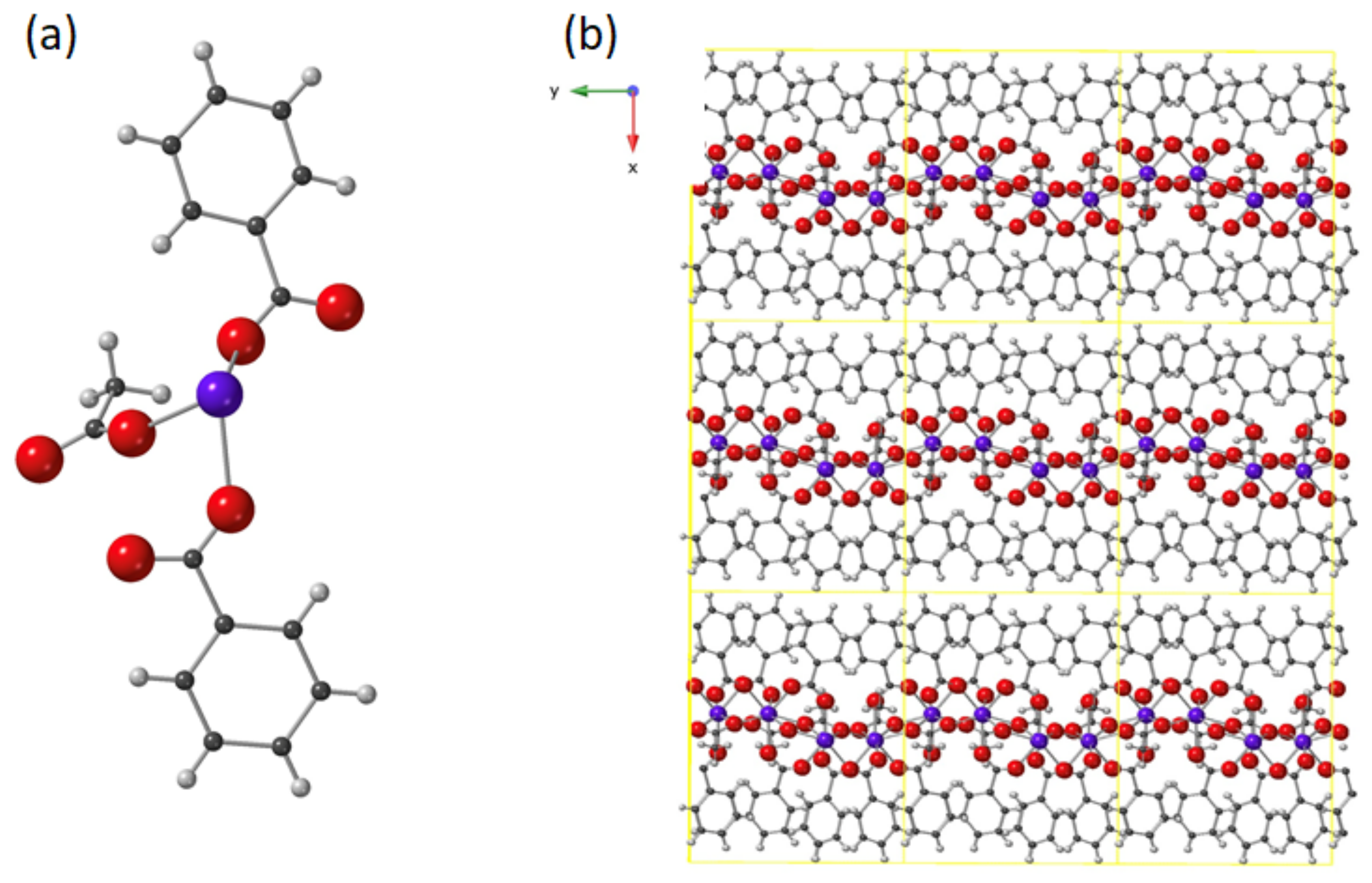
2.1. Synthesis and Crystal Structure
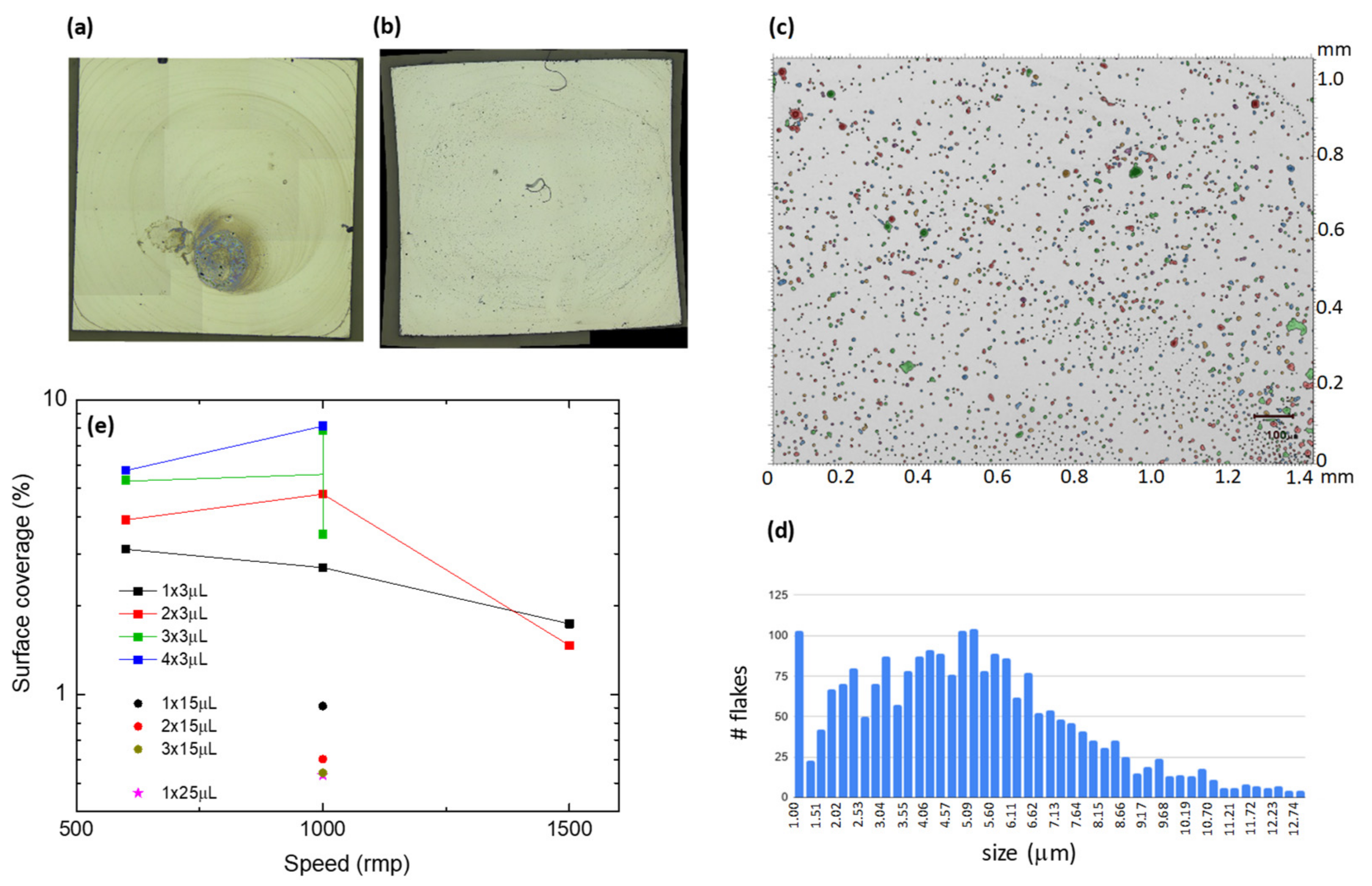
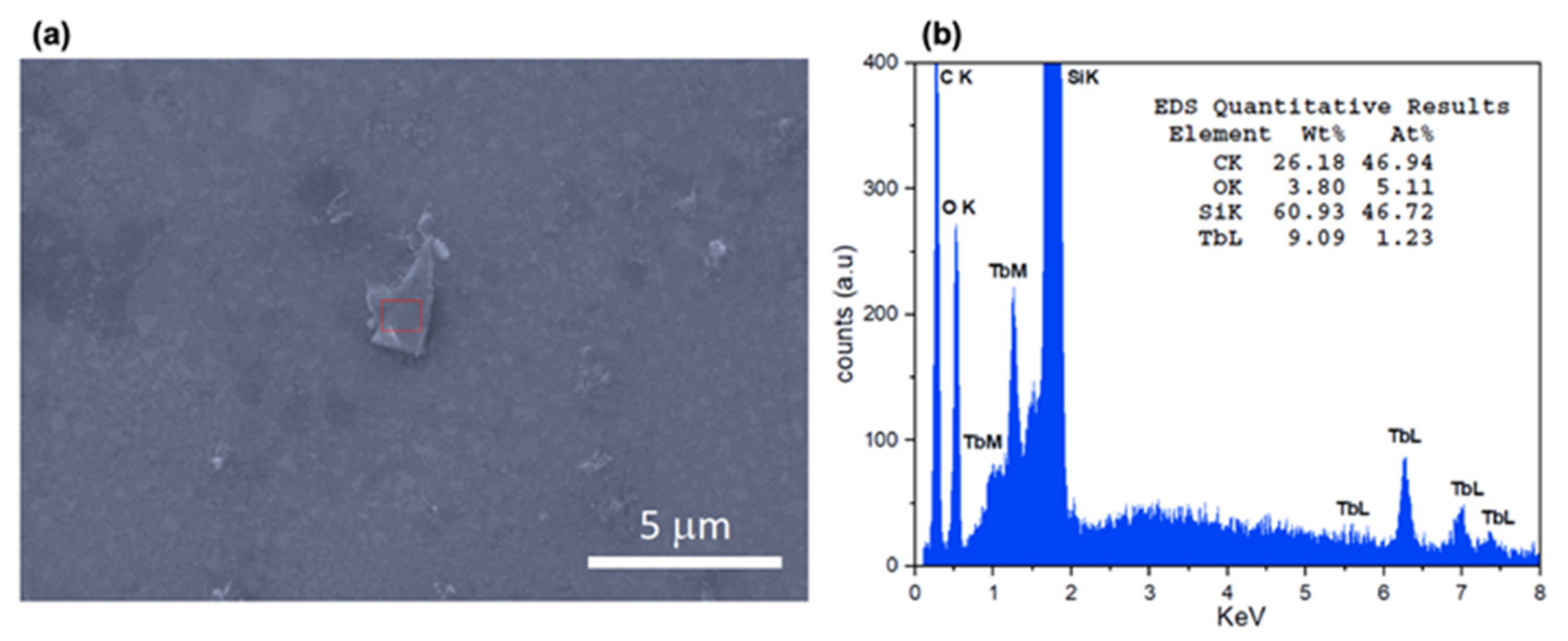
2.2. Exfoliation and Deposition of Tb-2D MOF Flakes onto Silicon
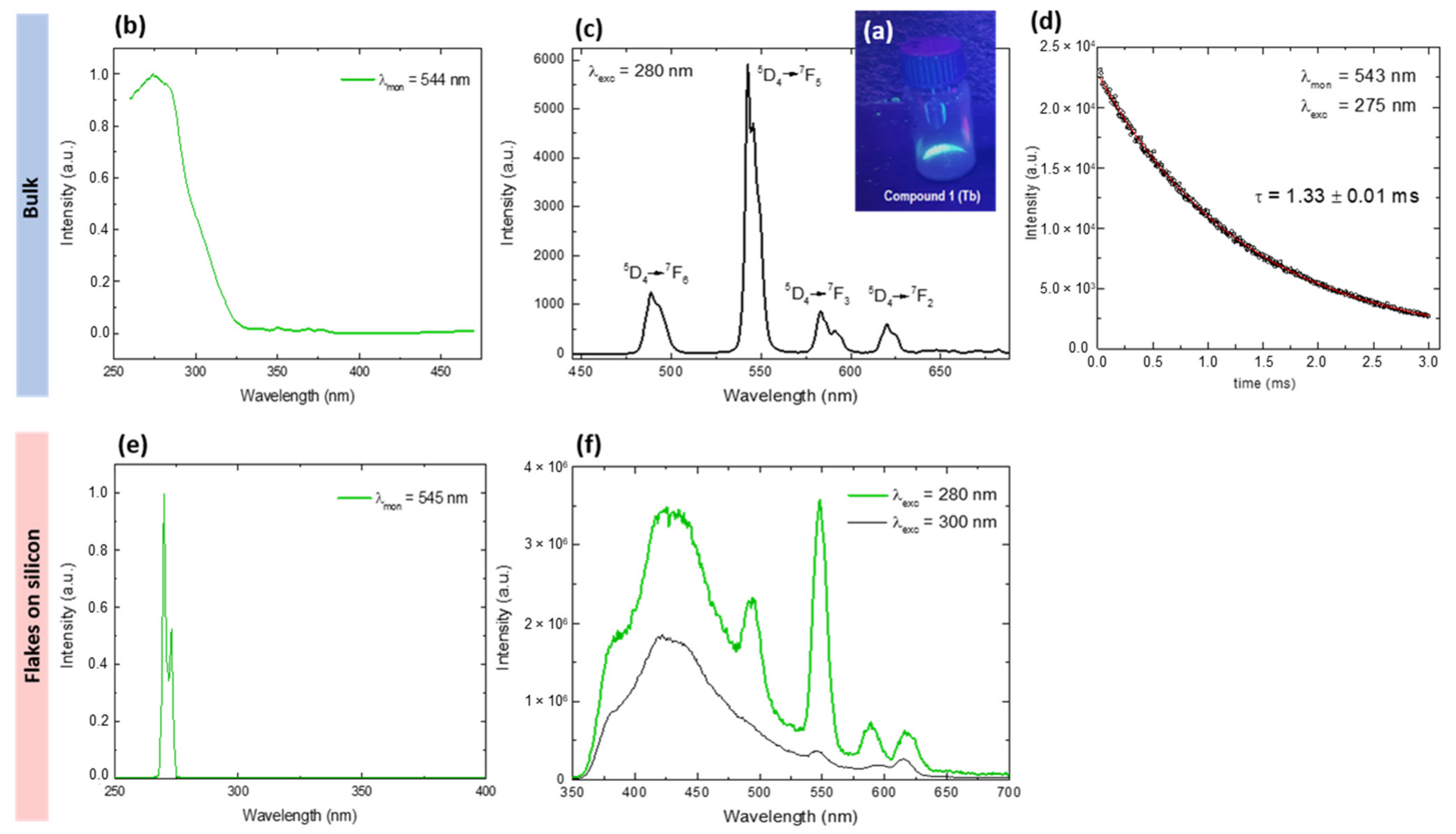
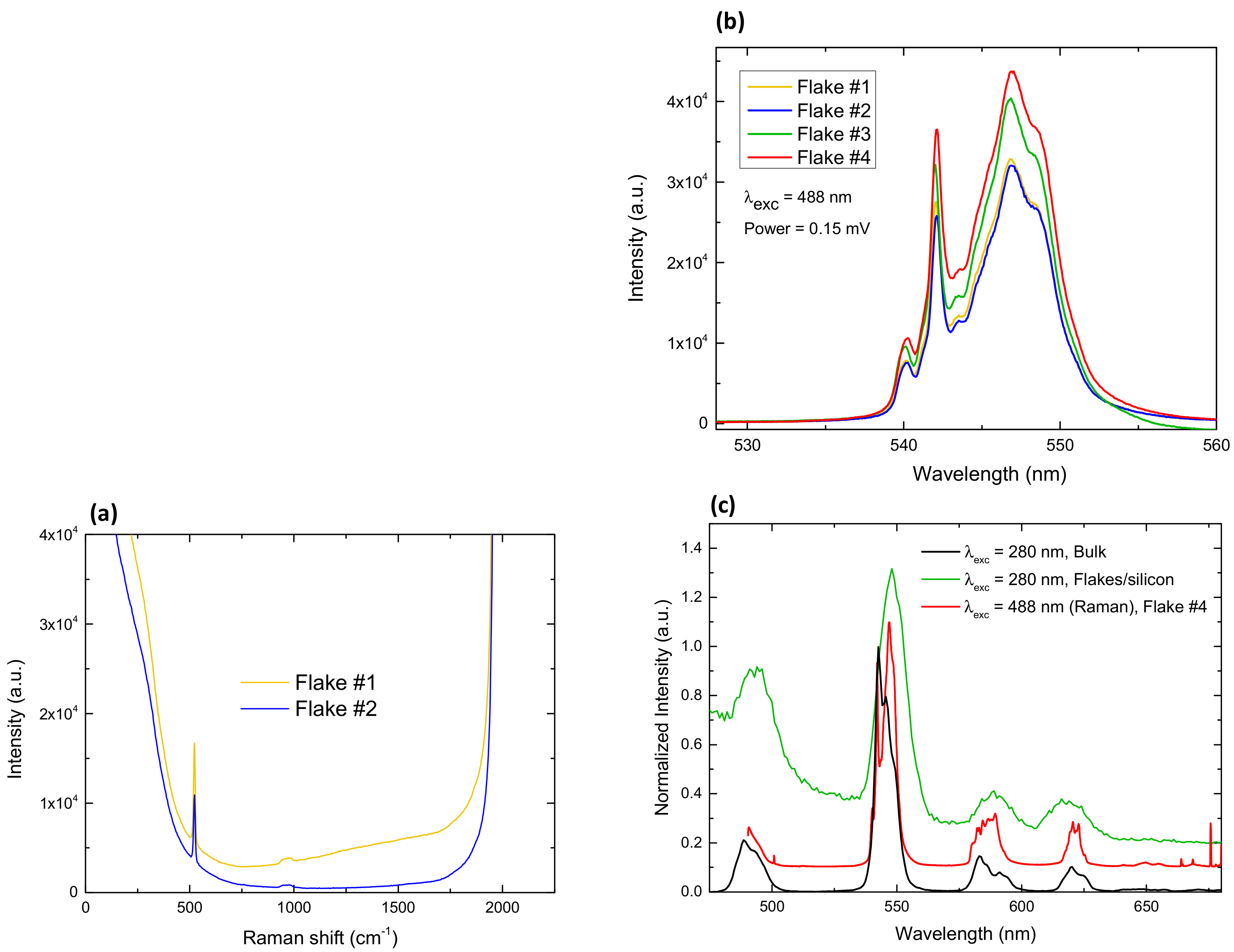
2.3. Luminescence and Raman Spectroscopy
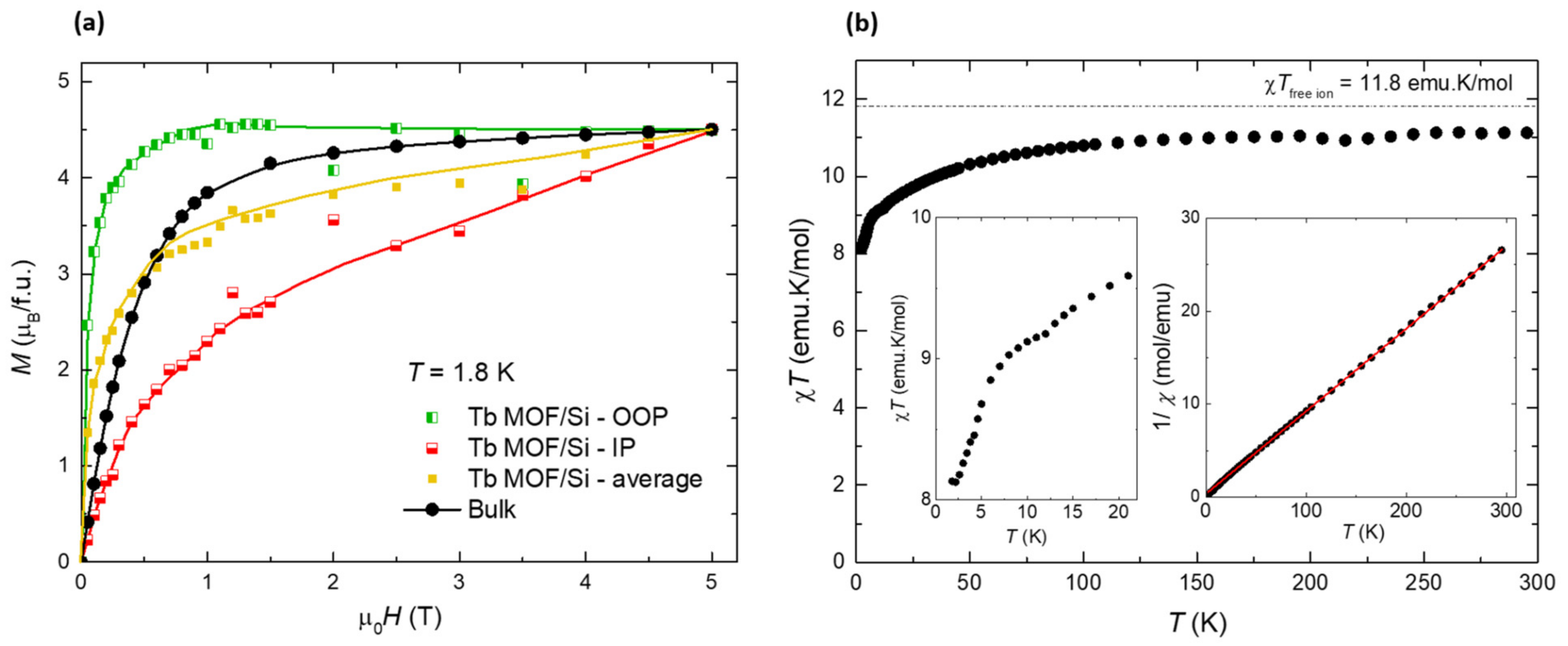
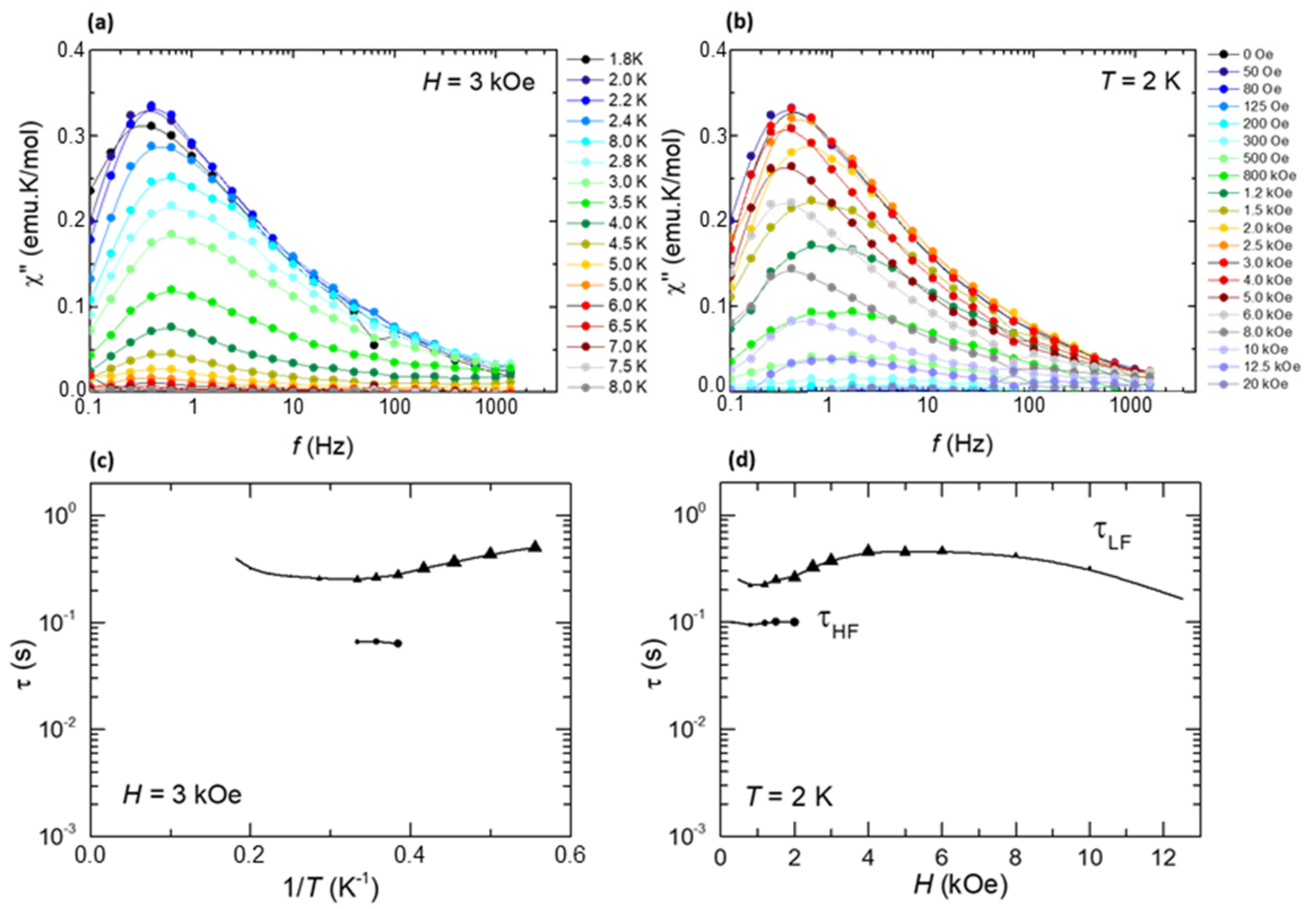
2.4. Magnetic Characterization
3. Experimental Methods
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Vijayakumar, S.; Nagamuthu, S.; Muralidharan, G. Supercapacitor Studies on NiO Nanoflakes Synthesized Through a Microwave Route. ACS Appl. Mater. Interfaces 2013, 5, 2188–2196. [Google Scholar] [CrossRef] [PubMed]
- Mendoza-Sánchez, B.; Gogotsi, Y. Synthesis of Two-Dimensional Materials for Capacitive Energy Storage. Adv. Mater. 2016, 28, 6104–6135. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Guo, Y.; Ma, W.; Zhou, Z. 2 D Materials for Electrochemical Energy Storage: Design, Preparation, and Application. ChemSusChem 2020, 13, 1155–1171. [Google Scholar] [CrossRef]
- Lin, L.; Chen, J.; Liu, D.; Li, X.; Wallace, G.G.; Zhang, S. Engineering 2D Materials: A Viable Pathway for Improved Electrochemical Energy Storage. Adv. Energy Mater. 2020, 10, 2002621. [Google Scholar] [CrossRef]
- Heard, C.J.; Cejka, J.; Opanasenko, M.; Nachtigall, P.; Centi, G.; Perathoner, S. 2D Oxide Nanomaterials to Adress the Energy Transition and Catalysis. Adv. Mater. 2018, 31, 1801712. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Chen, S.; Duo, Y.; Zhu, Y.; Fan, T.; Zou, Q.; Qu, M.; Lin, Z.; Zhao, J.; Li, Y.; et al. Biocompatible Two- Dimensional Titanium Nanosheets for Multimodal Imaging-Guided Cancer Theranostics. ACS Appl. Mater. Interfaces 2019, 11, 22129–22140. [Google Scholar] [CrossRef]
- Navarro-Moratalla, J.O.I.E.; Manãs-Valero, S.; Pinilla-Cienfuegos, E.; Castellanos-Gomez, A.; Quereda, J.; Rubio-Bollinger, G.; Chirolli, L.; Silva-Guillén, J.A.; Agraït, N.; Steele, G.A.; et al. Enhanced superconductivity in atomically thin TaS2. Nat. Commun. 2016, 7, 11043. [Google Scholar] [CrossRef] [Green Version]
- Huang, B.; Clark, G.; Navarro-Moratalla, E.; Klein, D.R.; Cheng, R.; Seyler, K.L.; Zhong, D.; Schmidgall, E.; McGuire, M.; Cobden, D.H.; et al. Layer-dependent ferromagnetism in a van der Waals crystal down to the monolayer limit. Nat. Cell Biol. 2017, 546, 270–273. [Google Scholar] [CrossRef] [Green Version]
- Miró, P.; Audiffred, M.; Heine, T. An atlas of two-dimensional materials. Chem. Soc. Rev. 2014, 43, 6537–6554. [Google Scholar] [CrossRef]
- Coronado, E. Molecular magnetism: From chemical design to spin control in molecules, materials and devices. Nat. Rev. Mater. 2019, 5, 87–104. [Google Scholar] [CrossRef]
- Wang, B.; Jin, J.; Ding, B.; Han, X.; Han, A.; Liu, J. General Approach to Metal-Organic Framework Nanosheets With Controllable Thickness by Using Metal Hydroxides as Precursors. Front. Mater. 2020, 7, 37. [Google Scholar] [CrossRef] [Green Version]
- Ding, G.; Wang, Y.; Zhang, G.; Zhou, K.; Zeng, K.; Li, Z.; Zhou, Y.; Zhang, C.; Chen, X.; Han, S.-T. 2D Metal-Organic Framework Nanosheets with Time-Dependent and Multilevel Memristive Switching. Adv. Funct. Mater. 2019, 29, 1806637. [Google Scholar] [CrossRef]
- Zhao, M.; Huang, Y.; Peng, Y.; Huang, Z.; Ma, Q.; Zhang, H. Two-dimensional metal–organic framework nanosheets: Synthesis and applications. Chem. Soc. Rev. 2018, 47, 6267–6295. [Google Scholar] [CrossRef] [PubMed]
- Yin, D.-D.; Chen, Q.; Meng, Y.-S.; Sun, H.-L.; Zhang, Y.-Q.; Gao, S. Slow magnetic relaxation in a novel carboxylate/oxalate/hydroxyl bridged dysprosium layer. Chem. Sci. 2015, 6, 3095–3101. [Google Scholar] [CrossRef] [Green Version]
- Mínguez Espallargas, G.; Coronado, E. Magnetic functionalities in MOFs: From the framework to the pore. Chem. Soc. Rev. 2018, 47, 533–557. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartual-Murgui, C.; Akou, A.; Thibault, C.; Molnár, G.; Vieu, C.; Salmon, L.; Bousseksou, A. Spin-crossover metal–organic frameworks: Promising materials for designing gas sensors. J. Mater. Chem. C 2015, 3, 1277–1285. [Google Scholar] [CrossRef]
- Guo, F.-S.; Leng, J.-D.; Liu, J.-L.; Meng, Z.-S.; Tong, M.-L. Polynuclear and Polymeric Gadolinium Acetate Derivatives with Large Magnetocaloric Effect. Inorg. Chem. 2012, 51, 405–413. [Google Scholar] [CrossRef]
- Baldovi, J.J.; Coronado, E.; Gaita-Arino, A. A SIM-MOF: Three-Dimensional Organisation of Single-Ion Magnets with Anion-Exchange Capabilities. Chem. Eur. J. 2014, 20, 10695–10702. [Google Scholar] [CrossRef]
- Zhang, X.; Vieru, V.; Feng, X.; Liu, J.-L.; Zhang, Z.; Na, B.; Shi, W.; Wang, B.-W.; Powell, A.K.; Chibotaru, L.F.; et al. Influence of Guest Exchange on the Magnetization Dynamics of Dilanthanide Single-Molecule-Magnet Nodes within a Metal-Organic Framework. Angew. Chem. Int. Ed. 2015, 54, 9861–9865. [Google Scholar] [CrossRef] [Green Version]
- Huang, G.; Fernandez-Garcia, G.; Badiane, I.; Camarra, M.; Freslon, S.; Guillou, O.; Daiguebonne, C.; Totti, F.; Cador, O.; Guizouarn, T.; et al. Magnetic Slow Relaxation in a Metal-Organic Framework Made of Chains of Ferromagnetically Coupled Single-Molecule Magnets. Chem. Eur. J. 2018, 24, 6983–6991. [Google Scholar] [CrossRef] [Green Version]
- Dogaheh, S.G.; Heras Ojea, M.J.; Piquer, L.R.; Artús Suàrez, L.; Khanmohammadi, H.; Aromí, G.; Sañudo, E.C. Co II and Cu II Fluorescent Complexes with Acridine-Based Ligands. Eur. J. Inorg. Chem. 2016, 2016, 3314–3321. [Google Scholar] [CrossRef] [Green Version]
- Gholizadeh Dogaheh, S.; Khanmohammadi, H.; Sañudo, E.C. Double-decker luminescent ytterbium and erbium SMMs with symmetric and asymmetric Schiff base ligands. New J. Chem. 2017, 41, 10101–10111. [Google Scholar] [CrossRef] [Green Version]
- Menelaou, M.; Ouharrou, F.; Rodríguez, L.; Roubeau, O.; Teat, S.J.; Aliaga-Alcalde, N. Dy(III)- and Yb(III)-curcuminoid compounds: Original fluorescent single-ion magnet and magnetic near-IR luminescent species. Chemistry 2012, 18, 11545–11549. [Google Scholar] [CrossRef]
- Soussi, K.; Jung, J.; Pointillart, F.; Le Guennic, B.; Lefeuvre, B.; Golhen, S.; Cador, O.; Guyot, Y.; Maury, O.; Ouahab, L. Magnetic and photo-physical investigations into Dy-III and Yb-III complexes involving tetrathiafulvalene ligand. Inorg. Chem. Front. 2015, 2, 1105–1117. [Google Scholar] [CrossRef]
- Pointillart, F.; Le Guennic, B.; Cauchy, T.; Golhen, S.; Cador, O.; Maury, O.; Ouahab, L. A series of tetrathiafulvalene-based lanthanide complexes displaying either single molecule magnet or luminescence—Direct magnetic and photo-physical correlations in the ytterbium analogue. Inorg. Chem. 2013, 52, 5978–5990. [Google Scholar] [CrossRef]
- Marin, R.; Brunet, G.; Murugesu, M. Shining new light on multifunctional lanthanide single-molecule magnets. Angew. Chemie Int. Ed. 2019. [Google Scholar]
- Jia, J.H.; Li, Q.W.; Chen, Y.C.; Liu, J.L.; Tong, M.L. Luminescent single-molecule magnets based on lanthanides: Design strategies, recent advances and magneto-luminescent studies. Coord. Chem. Rev. 2019, 378, 365–381. [Google Scholar] [CrossRef]
- Benmansour, S.; Hernández-Paredes, A.; Mondal, A.; López Martínez, G.; Canet-Ferrer, J.; Konar, S.; Gómez-García, C.J. Slow relaxation of the magnetization, reversible solvent exchange and luminescence in 2D anilato-based frameworks. Chem. Commun. 2020, 56, 9862–9865. [Google Scholar] [CrossRef] [PubMed]
- Salinas Uber, J.; Estrader, M.; Mathonière, C.; Clérac, R.; Roubeau, O.; Aromí, G. Photochromic Performance of Two Cu(II)-One-Dimensional Solvatomorphs Controlled by Intermolecular Interactions. Cryst. Growth Des. 2016, 16, 4026–4033. [Google Scholar] [CrossRef] [Green Version]
- Estrader, M.; Salinas Uber, J.; Barrios, L.A.; Garcia, J.; Lloyd-Williams, P.; Roubeau, O.; Teat, S.J.; Aromí, G. A Magneto-optical Molecular Device: Interplay of Spin Crossover, Luminescence, Photomagnetism, and Photochromism. Angew. Chem. Int. Ed. 2017, 56, 15622–15627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartolome, E.; Bartolome, J.; Sedona, F.; Lobo-Checa, J.; Forrer, D.; Herrero-Albillos, J.; Piantek, M.; Herrero-Martin, J.; Betto, D.; Velez-Fort, E.; et al. Enhanced Magnetism through Oxygenation of FePc/Ag(110) Monolayer Phases. J. Phys. Chem. C 2020, 124, 13993–14006. [Google Scholar] [CrossRef]
- Li, Q.-P.; Yan, B. Luminescent GaN semiconductor based on surface modification with lanthanide complexes through an ionic liquid bridge. RSC Adv. 2012, 2, 10840–10843. [Google Scholar] [CrossRef]
- Bahuleyan, B.K.; Toussaint, K.; Rinnert, H.; Vallon, R.; Molinari, M.; Chuburu, F.; Cadiou, C. Silicon Wafer Functionalization with a Luminescent Tb(III) Coordination Complex: Synthesis, Characterization, and Application to the Optical Detection of NO in the Gas Phase. Molecules 2019, 24, 1914. [Google Scholar] [CrossRef] [Green Version]
- Heinke, L.; Tu, M.; Wannapaiboon, S.; Fischer, R.; Woll, C. Surface-mounted metal-organic frameworks for applications in sensing and separation. Microporous Mesoporous Mater. 2015, 216, 200–215. [Google Scholar] [CrossRef]
- Doustkhah, E.; Hassandoost, R.; Khataee, A.; Luque, R.; Assadi, M.H.N. Hard-templated metal–organic frameworks for advanced applications. Chem. Soc. Rev. 2021, 50, 2927–2953. [Google Scholar] [CrossRef] [PubMed]
- Ashworth, D.J.; Foster, J.A. Metal–organic framework nanosheets (MONs): A new dimension in materials chemistry. J. Mater. Chem. A 2018, 6, 16292–16307. [Google Scholar] [CrossRef] [Green Version]
- Pepi, B.; Contreras-Pereda, N.; Suárez-García, S.; Hayatia, P.; Benmansourb, S.; Pascal, R.; Morsalid, A.; Ruiz-Molina, D. Solvent-tuned ultrasonic synthesis of 2D coordination polymer nanostructures and flakes. Ultrason. Sonochem. 2021, 72, 105425. [Google Scholar] [CrossRef] [PubMed]
- León-Alcaide, L.; López-Cabrelles, J.; Espallargas, G.M.; Coronado, E. 2D magnetic MOFs with micron-lateral size by liquid exfoliation. Chem. Commun. 2020, 56, 7657–7660. [Google Scholar] [CrossRef]
- González, J.; Sevilla, P.; Gabarró-Riera, G.; Jover, J.; Echeverría, J.; Fuertes, S.; Arauzo, A.; Bartolomé, E.; Sañudo, E.C. A multifunctional Dysprosium-carboxylato 2D metallorganic framework. Angew. Chem. Int. Ed. 2021, 60, 12001–12006. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.-M.; Xiong, M.; Zhang, D.-Q.; Du, M.; Zhu, D.-B. Two- and three-dimensional lanthanide–organic frameworks constructed using 1-hydro-6-oxopyridine-3-carboxylate and oxalate ligands. Dalton Trans. 2009, 5666–5672. [Google Scholar] [CrossRef] [PubMed]
- López-Cabrelles, J.; Mañas-Valero, S.; Vitórica-Yrezábal, I.J.; Bereciartua, P.J.; Waerenborgh, J.A.; Rodríguez-Velamazán, J.C.; Vieira, B.J.C.; Davidovikj, D.; Steeneken, P.G.; Zant, H.S.J.; et al. Isoreticular two-dimensional magnetic coordination polymers prepared through pre-synthetic ligand functionalization. Nat. Chem. 2018, 10, 1001–1007. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Temple, P.A.; Hathaway, C.E. Multiphonon Raman Spectra of Silicon. Phys. Rev. B 1973, 7, 3685. [Google Scholar] [CrossRef]
- González, J.C.; Mestres, N.; Puig, T.; Gázquez, J.; Sandiumenge, F.; Obradors, X.; Usoskin, A.; Jooss, C.; Freyhardt, H.C.; Feenstra, R. Biaxial texture analysis of YBa2Cu3O7-coated conductors by micro-Raman spectroscopy. Phys. Rev. B 2004, 70, 094525. [Google Scholar] [CrossRef]


| Formula | [Tb(MeCOO)(PhCOO)2] (1) |
| CCDC | 2095560 |
| Crystal System | Monoclinic |
| Space Group | P21/c |
| Z | 4 |
| a (Å) | 16.3617(9) |
| b (Å) | 12.6659(7) |
| c (Å) | 7.2496(4) |
| Alfao | 90 |
| Betao | 101.073(2) |
| Gammao | 90 |
| V (Å3) | 1474.41(14) |
| T (K) | 100(2) |
| X-ray | ALS |
| R (wR) | 0.0258 (0.0563) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bartolomé, E.; Arauzo, A.; Herce, S.; Palau, A.; Mestres, N.; Fuertes, S.; Sevilla, P.; Settineri, N.S.; Navarro-Spreafico, L.; González, J.; et al. Luminescent and Magnetic Tb-MOF Flakes Deposited on Silicon. Molecules 2021, 26, 5503. https://doi.org/10.3390/molecules26185503
Bartolomé E, Arauzo A, Herce S, Palau A, Mestres N, Fuertes S, Sevilla P, Settineri NS, Navarro-Spreafico L, González J, et al. Luminescent and Magnetic Tb-MOF Flakes Deposited on Silicon. Molecules. 2021; 26(18):5503. https://doi.org/10.3390/molecules26185503
Chicago/Turabian StyleBartolomé, Elena, Ana Arauzo, Sergio Herce, Anna Palau, Narcis Mestres, Sara Fuertes, Pablo Sevilla, Nicholas S. Settineri, Laura Navarro-Spreafico, Jonay González, and et al. 2021. "Luminescent and Magnetic Tb-MOF Flakes Deposited on Silicon" Molecules 26, no. 18: 5503. https://doi.org/10.3390/molecules26185503
APA StyleBartolomé, E., Arauzo, A., Herce, S., Palau, A., Mestres, N., Fuertes, S., Sevilla, P., Settineri, N. S., Navarro-Spreafico, L., González, J., & Sañudo, E. C. (2021). Luminescent and Magnetic Tb-MOF Flakes Deposited on Silicon. Molecules, 26(18), 5503. https://doi.org/10.3390/molecules26185503











