Inhibitory Effect of Fisetin on α-Glucosidase Activity: Kinetic and Molecular Docking Studies
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. α-Glucosidase Inhibition Assay
2.3. α-Glucosidase Kinetic Assay
2.4. Fluorescence Spectrophotometric Measurement
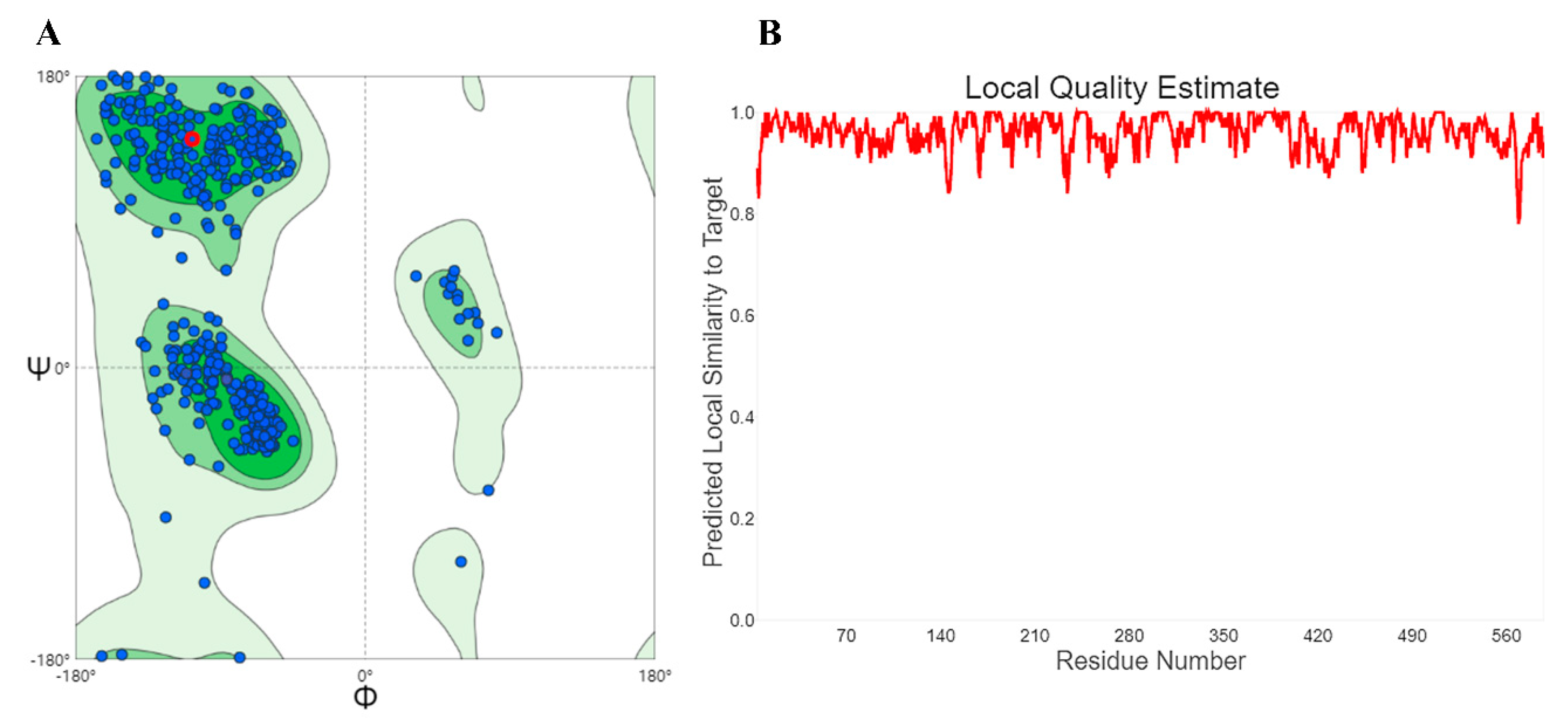
2.5. Homology Modeling and Molecular Docking
3. Results and Discussion
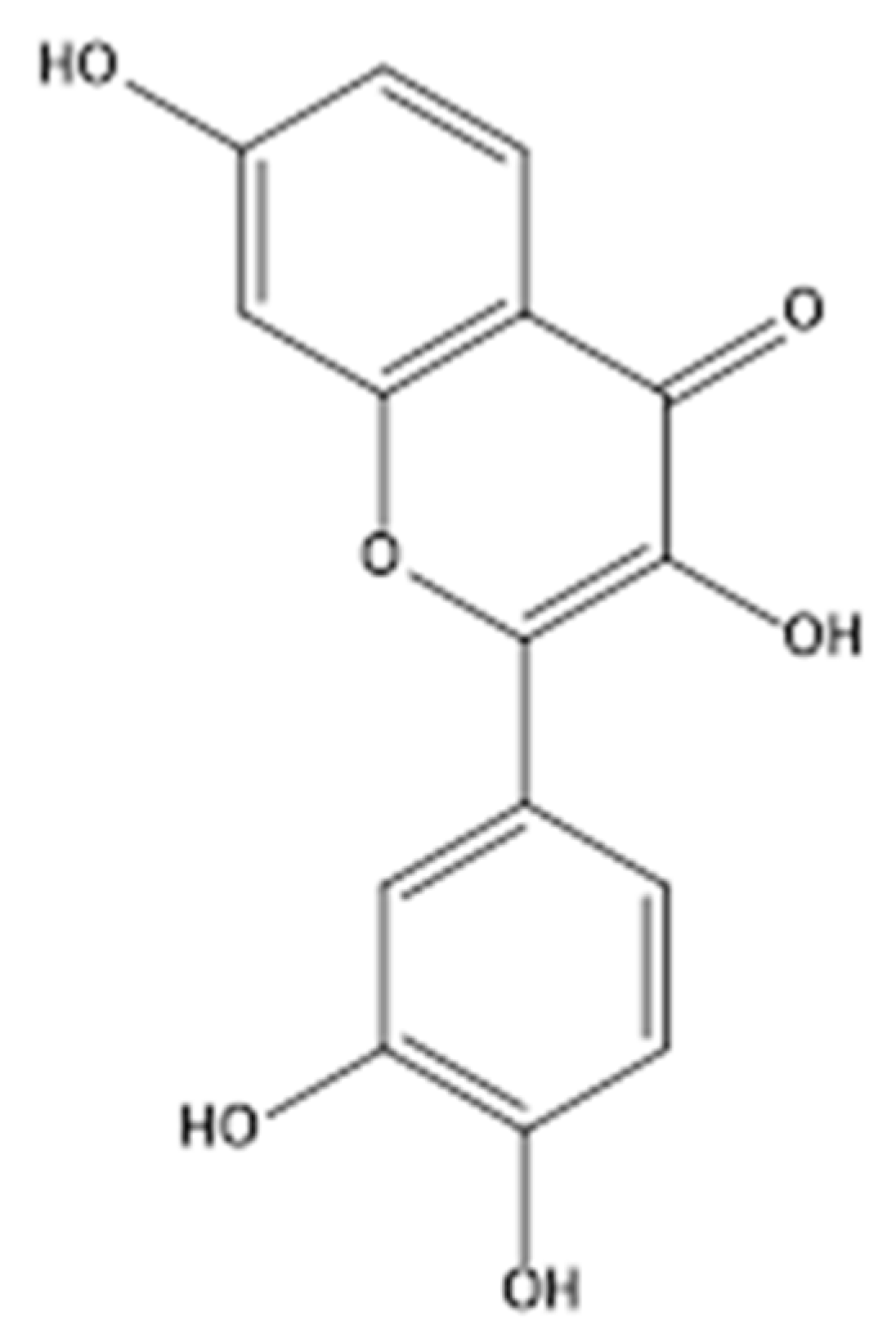
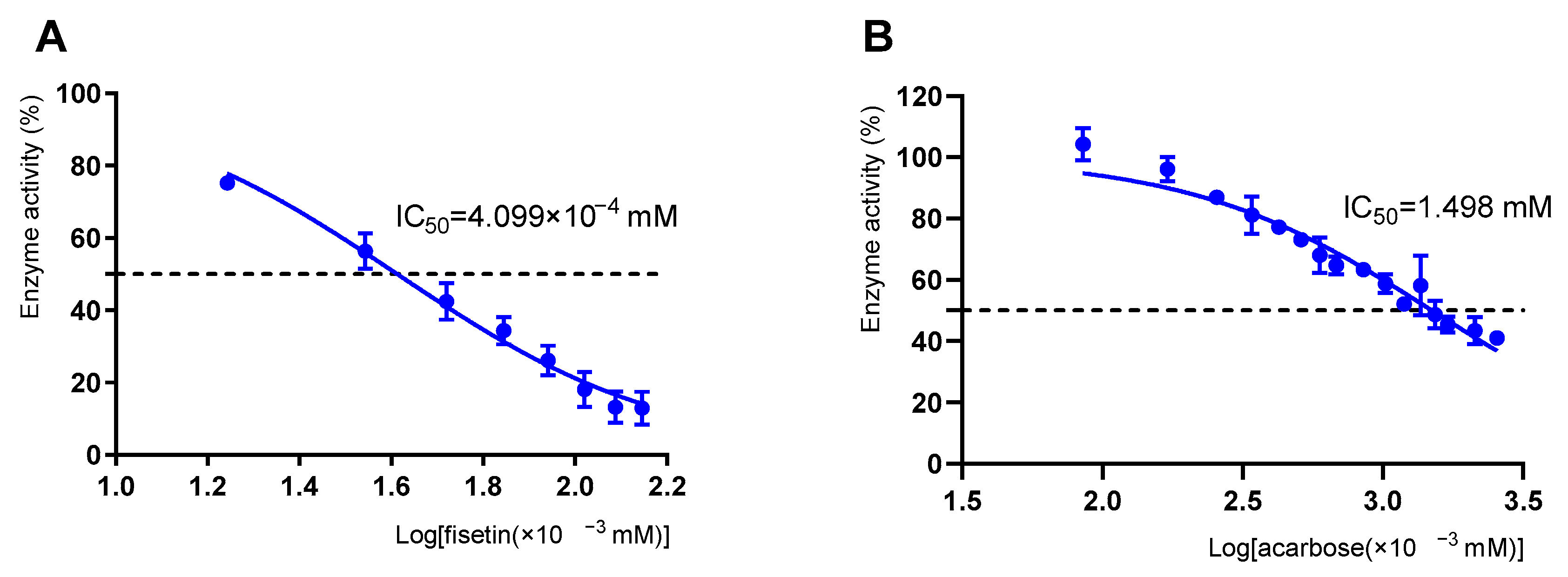
3.1. Inhibitory Effect of Fisetin on α-Glucosidase
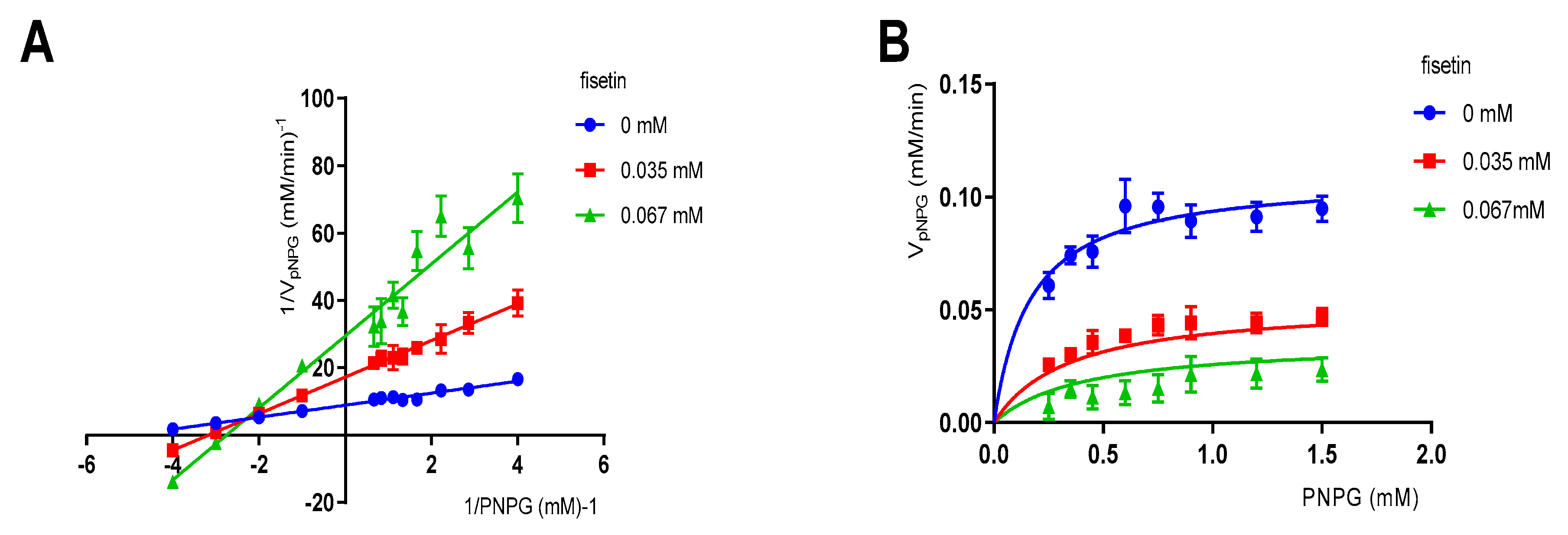
3.2. Enzymatic Kinetic Assay
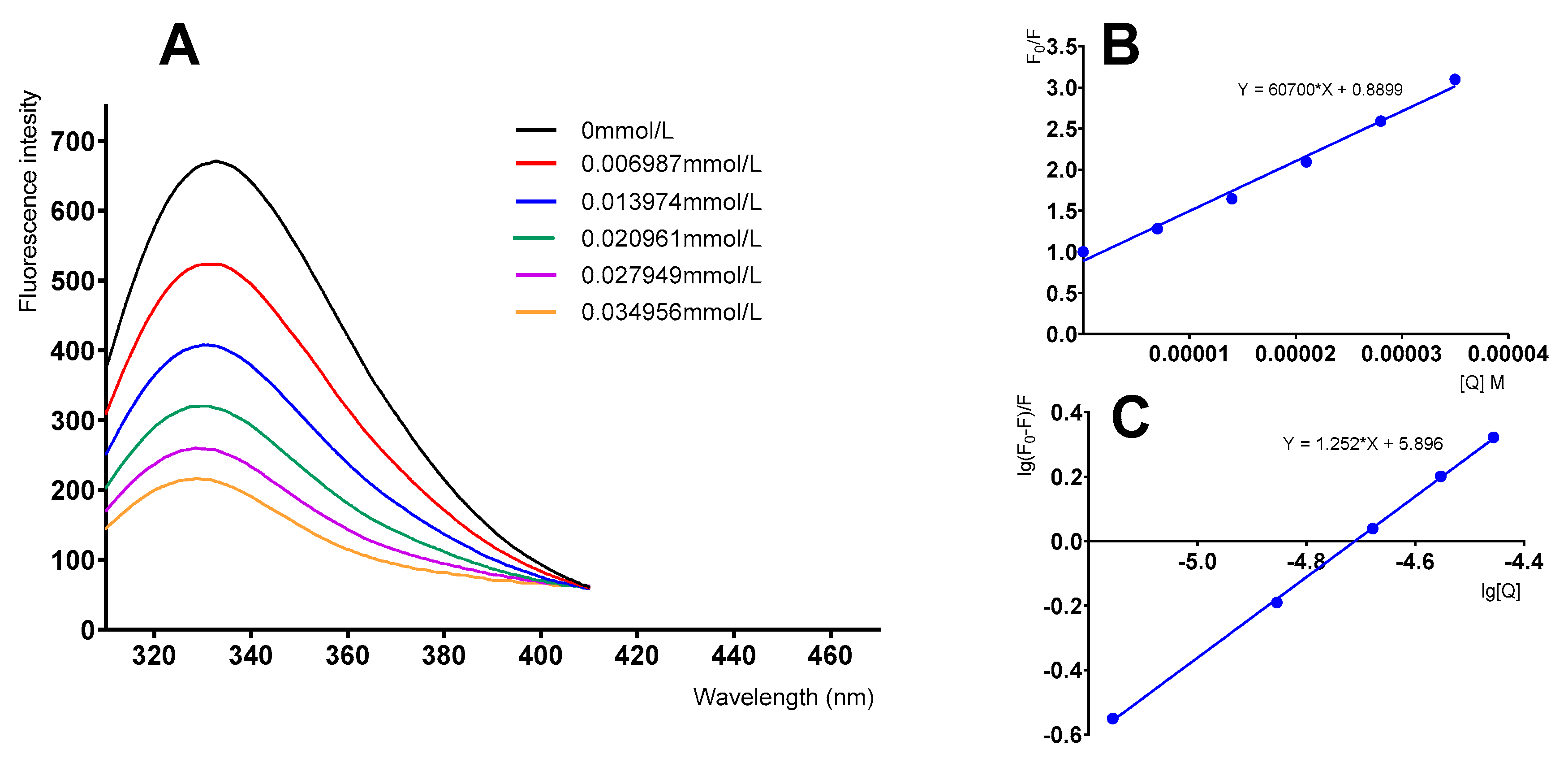
3.3. Fluorescence Quenching Studies of α-Glucosidase by Fisetin
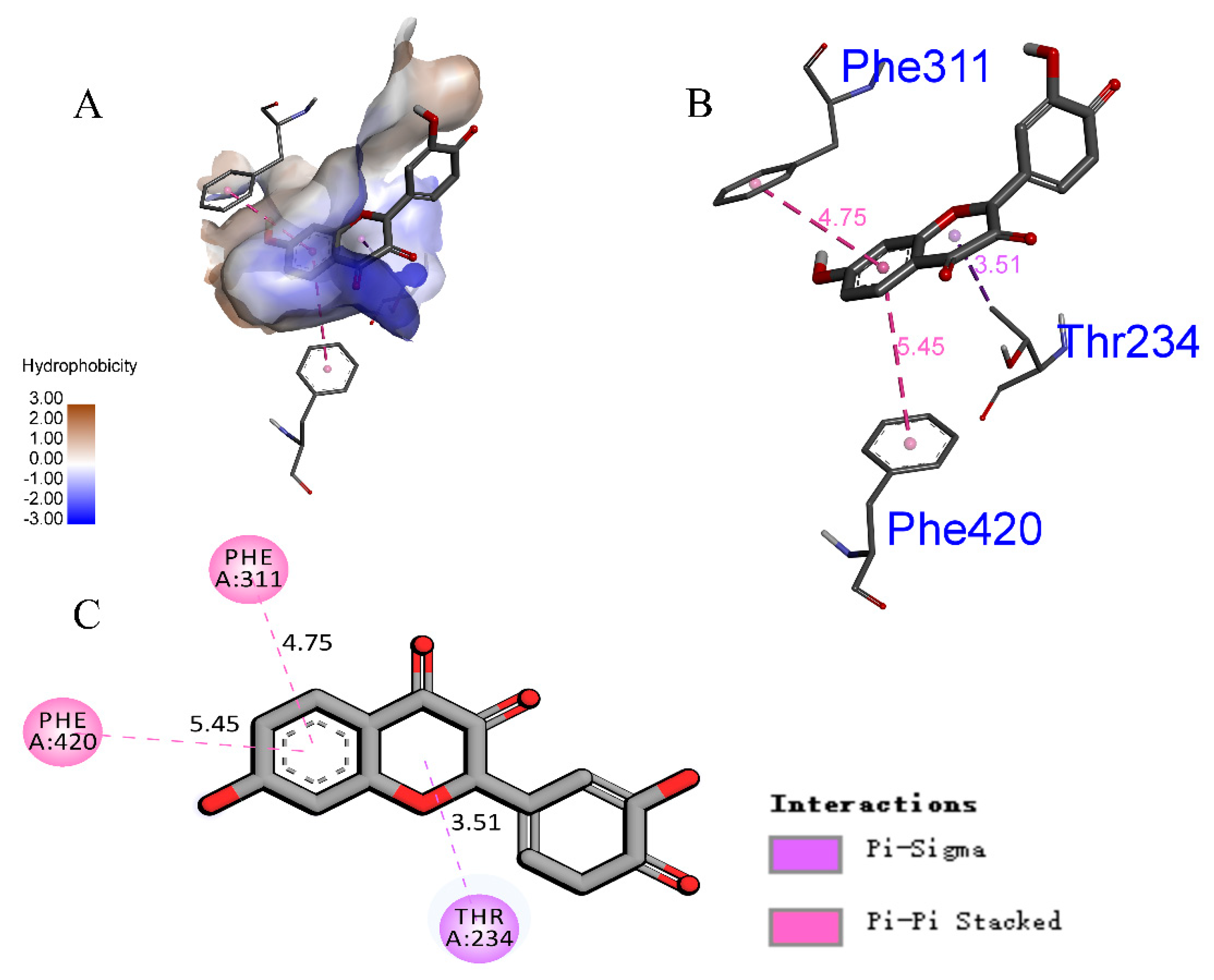
3.4. Homology Modeling and Molecular Docking Analysis of α-Glucosidase
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
Abbreviations
References
- Tian, T.; Chen, G.; Zhang, H.; Yang, F. Personal Glucose Meter for α-Glucosidase Inhibitor Screening Based on the Hydrolysis of Maltose. Molecules 2021, 26, 4638. [Google Scholar] [CrossRef]
- Şöhretoğlu, D.; Sari, S.; Barut, B.; Özel, A. Discovery of potent alpha-glucosidase inhibitor flavonols: Insights into mechanism of action through inhibition kinetics and docking simulations. Bioorg. Chem. 2018, 79, 257–264. [Google Scholar] [CrossRef] [PubMed]
- IDF Diabetes Atlas, 9th ed.; International Diabetes Federation: Brussels, Belgium, 2019; Available online: http://www.diabetesatlas.org (accessed on 13 August 2021).
- Alberti, K.G.M.M.; Zimmet, P.Z. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: Diagnosis and classification of diabetes mellitus. Provisional report of a WHO Consultation. Diabet. Med. 1998, 15, 539–553. [Google Scholar] [CrossRef]
- Asemanipoor, N.; Mohammadi-Khanaposhtani, M.; Moradi, S.; Vahidi, M.; Asadi, M.; Ali Faramarzi, M.; Mahdavi, M.; Biglar, M.; Larijani, B.; Hamedifar, H.; et al. Synthesis and biological evaluation of new benzimidazole-1,2,3-triazole hybrids as potential α-glucosidase inhibitors. Bioorg. Chem. 2019, 95, 103482. [Google Scholar] [CrossRef]
- Chu, Y.; Wu, S.; Hsieh, J. Isolation and characterization of α-glucosidase inhibitory constituents from Rhodiola crenulata. Food Res. Int. 2014, 57, 8–14. [Google Scholar] [CrossRef]
- Ujiroghene, O.J.; Liu, L.; Zhang, S.; Lu, J.; Pang, X.; Lv, J. α-Glucosidase and ACE dual inhibitory protein hydrolysates and peptide fractions of sprouted quinoa yoghurt beverages inoculated with Lactobacillus casei. Food Chem. 2019, 299, 124985. [Google Scholar] [CrossRef]
- Bhandari, M.R.; Jong-Anurakkun, N.; Hong, G.; Kawabata, J. α-Glucosidase and α-amylase inhibitory activities of Nepalese medicinal herb Pakhanbhed (Bergenia ciliata, Haw.). Food Chem. 2008, 106, 247–252. [Google Scholar] [CrossRef]
- Matsui, T.; Ueda, T.; Oki, T.; Sugita, K.; Terahara, N.; Matsumoto, K. α-Glucosidase Inhibitory Action of Natural Acylated Anthocyanins. 1. Survey of Natural Pigments with Potent Inhibitory Activity. J. Agric. Food Chem. 2001, 49, 1948–1951. [Google Scholar] [CrossRef]
- Vitale, M.; Masulli, M.; Rivellese, A.A.; Bonora, E.; Cappellini, F.; Nicolucci, A.; Squatrito, S.; Antenucci, D.; Barrea, A.; Bianchi, C.; et al. Dietary intake and major food sources of polyphenols in people with type 2 diabetes: The TOSCA.IT Study. Eur. J. Nutr. 2018, 57, 679–688. [Google Scholar] [CrossRef]
- Ding, G.; Xu, X.; Li, D.; Chen, Y.; Wang, W.; Ping, D.; Jia, S.; Cao, L. Fisetin inhibits proliferation of pancreatic adenocarcinoma by inducing DNA damage via RFXAP/KDM4A-dependent histone H3K36 demethylation. Cell Death Dis. 2020, 11, 893. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, X.; Deng, J.; Yang, X.; Wang, J.; Wang, Y. Hierarchical porous biochar derived from cotinus coggygria flower by using a novel composite activator for supercapacitors. Chem. Phys. Lett. 2020, 747, 137325. [Google Scholar] [CrossRef]
- Deniz, F.S.S.; Salmas, R.E.; Emerce, E.; Cankaya, I.I.T.; Yusufoglu, H.S.; Orhan, I.E. Evaluation of collagenase, elastase and tyrosinase inhibitory activities of Cotinus coggygria Scop. through in vitro and in silico approaches. S. Afr. J. Bot. 2020, 132, 277–288. [Google Scholar] [CrossRef]
- Higa, S.; Hirano, T.; Kotani, M.; Matsumoto, M.; Fujita, A.; Suemura, M.; Kawase, I.; Tanaka, T. Fisetin, a flavonol, inhibits TH2-type cytokine production by activated human basophils. J. Allergy Clin. Immunol. 2003, 111, 1299–1306. [Google Scholar] [CrossRef] [PubMed]
- Messaadia, L.; Bekkar, Y.; Benamira, M.; Lahmar, H. Predicting the antioxidant activity of some flavonoids of Arbutus plant: A theoretical approach. Chem. Phys. Impact 2020, 1, 100007. [Google Scholar] [CrossRef]
- Myung-Sook, C.; Ji-Young, C.; Eun-Young, K. Fisetin Alleviates Hepatic and Adipocyte Fibrosis and Insulin Resistance in Diet-Induced Obese Mice. J. Med. Food 2020, 23, 1019–1032. [Google Scholar] [CrossRef]
- Kim, A.; Lee, W.; Yun, J. Luteolin and fisetin suppress oxidative stress by modulating sirtuins and forkhead box O3a expression under in vitro diabetic conditions. Nutr. Res. Pract. 2017, 11, 430–434. [Google Scholar] [CrossRef] [Green Version]
- Madeswaran, A.; Umamaheswari, M.; Asokkumar, K.; Sivashanmugam, T.; Subhadradevi, V.; Jagannath, P. Docking studies: In silico lipoxygenase inhibitory activity of some commercially available flavonoids. Bangladesh J. Pharmacol. 2011, 6, 133–138. [Google Scholar] [CrossRef]
- Chen, J.-G.; Wu, S.-W.; Zhang, Q.-F.; Yin, Z.-P.; Zhang, L. α-Glucosidase inhibitory effect of anthocyanins from Cinnamomum camphora fruit: Inhibition kinetics and mechanistic insights through in vitro and in silico studies. Int. J. Biol. Macromol. 2020, 143, 696–703. [Google Scholar] [CrossRef]
- Flores-Bocanegra, L.; Pérez-Vásquez, A.; Torres-Piedra, M.; Bye, R.; Linares, E.; Mata, R. α-Glucosidase Inhibitors from Vauquelinia corymbosa. Molecules 2015, 20, 15330–15342. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.H.; Kim, H.Y.; Yang, S.Y.; Kim, J.B.; Jin, C.H.; Kim, Y.H. Inhibitory activity of (-)-epicatechin-3,5-O-digallate on α-glucosidase and in silico analysis. Int. J. Biol. Macromol. 2017, 107, 1162–1167. [Google Scholar] [CrossRef]
- Wang, G.; He, D.; Li, X.; Li, J.; Peng, Z. Design, synthesis and biological evaluation of novel coumarin thiazole derivatives as α-glucosidase inhibitors. Bioorg. Chem. 2016, 65, 167–174. [Google Scholar] [CrossRef]
- Lin, P.; Zeng, J.; Chen, J.; Nie, X.; Yuan, E.; Wang, X.; Peng, D.; Yin, Z. Synthesis, in vitro inhibitory activity, kinetic study and molecular docking of novel N-alkyl-deoxynojirimycin derivatives as potential α-glucosidase inhibitors. J. Enzym. Inhib. Med. Chem. 2020, 35, 1879–1890. [Google Scholar] [CrossRef]
- Waterhouse, A.; Bertoni, M.; Bienert, S.; Studer, G.; Tauriello, G.; Gumienny, R.; Heer, F.T.; De Beer, T.A.P.; Rempfer, C.; Lorenza, B.; et al. SWISS-MODEL: Homology modelling of protein structures and complexes. Nucleic Acids Res. 2018, 1, W296–W303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, J.; Zhang, G.; Pan, J.; Wang, Y. α-Glucosidase inhibition by luteolin: Kinetics, interaction and molecular docking. Int. J. Biol. Macromol. 2014, 64, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2009, 31, 455–461. [Google Scholar] [CrossRef] [Green Version]
- Wagle, A.; Seong, S.H.; Jung, H.A.; Choi, J.S. Identifying an isoflavone from the root of Pueraria lobata as a potent tyrosinase inhibitor. Food Chem. 2018, 276, 383–389. [Google Scholar] [CrossRef]
- Medina-Pérez, G.; Zaldívar-Ortega, A.K.; Cenobio-Galindo, A.D.J.; Afanador-Barajas, L.N.; Vieyra-Alberto, R.; Estefes-Duarte, J.A.; Campos-Montiel, R.G. Antidiabetic Activity of Cactus Acid Fruit Extracts: Simulated Intestinal Conditions of the Inhibitory Effects on α-amylase and α-glucosidase. Appl. Sci. 2019, 9, 4066. [Google Scholar] [CrossRef] [Green Version]
- Xu, H. Inhibition kinetics of flavonoids on yeast alpha-glucosidase merged with docking simulations. Protein Pept. Lett. 2010, 17, 1270–1279. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Qin, X.; Zhai, Y.; Hao, H.; Lee, J.; Park, Y. Inhibitory effect of hesperetin on α-glucosidase: Molecular dynamics simulation integrating inhibition kinetics. Int. J. Biol. Macromol. 2017, 101, 32–39. [Google Scholar] [CrossRef]
- ALTamimi, J.Z.; BinMowyna, M.N.; AlFaris, N.A.; Alagal, R.I.; El-kott, A.F.; AL-Farga, A.M. Fisetin protects against streptozotocin-induced diabetic cardiomyopathy in rats by suppressing fatty acid oxidation and inhibiting protein kinase R. Saudi Pharm. J. 2021, 29, 27–42. [Google Scholar] [CrossRef]
- Wang, G.; Wang, J.; He, D.; Li, X.; Li, J.; Peng, P. Synthesis, in vitro evaluation and molecular docking studies of novel coumarinisatin derivatives as α-glucosidase inhibitors. Chem. Biol. Drug Des. 2017, 89, 456–463. [Google Scholar] [CrossRef]
- Şöhretoğlu, D.; Sari, S.; Šoral, M.; Barut, B.; Özel, A.; Liptaj, T. Potential of Potentilla inclinata and its polyphenolic compounds in α-glucosidase inhibition: Kinetics and interaction mechanism merged with docking simulations. Int. J. Biol. Macromol. 2018, 79, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Zhang, G.; Zeng, L. Inhibition of α-glucosidase by vitamin D3 and the effect of vitamins B1 and B2. Food Funct. 2015, 7, 982–991. [Google Scholar] [CrossRef]
- Peng, X.; Zhang, G.; Liao, Y.; Gong, D. Inhibitory kinetics and mechanism of kaempferol on α-glucosidase. Food Chem. 2016, 190, 207–215. [Google Scholar] [CrossRef]
- Feroz, S.R.; Mohamad, S.B.; Bakri, Z.S.D.; Malek, S.N.A.; Tayyab, S.; Permyakov, E.A. Probing the interaction of a therapeutic flavonoid, pinostrobin with human serum albumin: Multiple spectroscopic and molecular modeling investigations. PLoS ONE 2013, 8, e76067. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.Q.; Zhou, F.C.; Gao, F.; Bian, J.S.; Shan, F. Comparative Evaluation of Quercetin, Isoquercetin and Rutin as Inhibitors of α-Glucosidase. J. Agric. Food Chem. 2009, 57, 11463–11468. [Google Scholar] [CrossRef] [PubMed]
- Şöhretoğlu, D.; Sari, S.; Özel, A.; Barut, B. α-Glucosidase inhibitory effect of Potentilla astracanica and some isoflavones: Inhibition kinetics and mechanistic insights through in vitro and in silico studies. Int. J. Biol. Macromol. 2017, 105, 1062–1070. [Google Scholar] [CrossRef]
- Park, H.; Hwang, K.Y.; Oh, K.H.; Kim, Y.H.; Lee, J.Y.; Kim, K. Discovery of novel α-glucosidase inhibitors based on the virtual screening with the homology-modeled protein structure. Bioorg. Med. Chem. 2008, 16, 284–292. [Google Scholar] [CrossRef]
- Sichaem, J.; Aree, T.; Lugsanangarm, K.; Tip-Pyang, S. Identification of highly potent alpha-glucosidase inhibitory and antioxidant constituents from Zizyphus rugosa bark: Enzyme kinetic and molecular docking studies with active metabolites. Pharm. Biol. 2017, 55, 1436–1441. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gollapalli, M.; Taha, M.; Ullah, H.; Nawaz, M.; AlMuqarrabun, L.M.R.; Rahim, F.; Qureshi, F.; Mosaddik, A.; Ahmat, N.; Khan, K.M. Synthesis of Bis-indolylmethane sulfonohydrazides derivatives as potent α-Glucosidase inhibitors. Bioorg. Chem. 2018, 80, 112–120. [Google Scholar] [CrossRef]
- Han, L.; Fang, C.; Zhu, R.; Peng, Q.; Li, D.; Wang, M. Inhibitory effect of phloretin on α-glucosidase: Kinetics, interaction mechanism and molecular docking. Int. J. Biol. Macromol. 2016, 95, 520–527. [Google Scholar] [CrossRef] [PubMed]
- Rahim, F.; Ullah, K.; Ullah, H.; Wadood, A.; Taha, M.; Rehman, A.U.; Uddin, I.; Ashraf, M.; Shaukat, A.; Rehman, W.; et al. Triazinoindole analogs as potent inhibitors of α-glucosidase: Synthesis, biological evaluation and molecular docking studies. Bioorg. Chem. 2014, 58, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Vaya, J.; Mahmood, S.; Goldblum, A.; Aviram, M.; Volkova, N.; Shaalan, A.; Musa, R.; Tamir, S. Inhibition of LDL oxidation by flavonoids in relation to their structure and calculated enthalpy. Phytochemistry 2003, 62, 89–99. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shen, B.; Shangguan, X.; Yin, Z.; Wu, S.; Zhang, Q.; Peng, W.; Li, J.; Zhang, L.; Chen, J. Inhibitory Effect of Fisetin on α-Glucosidase Activity: Kinetic and Molecular Docking Studies. Molecules 2021, 26, 5306. https://doi.org/10.3390/molecules26175306
Shen B, Shangguan X, Yin Z, Wu S, Zhang Q, Peng W, Li J, Zhang L, Chen J. Inhibitory Effect of Fisetin on α-Glucosidase Activity: Kinetic and Molecular Docking Studies. Molecules. 2021; 26(17):5306. https://doi.org/10.3390/molecules26175306
Chicago/Turabian StyleShen, Beiyun, Xinchen Shangguan, Zhongping Yin, Shaofu Wu, Qingfeng Zhang, Wenwen Peng, Jingen Li, Lu Zhang, and Jiguang Chen. 2021. "Inhibitory Effect of Fisetin on α-Glucosidase Activity: Kinetic and Molecular Docking Studies" Molecules 26, no. 17: 5306. https://doi.org/10.3390/molecules26175306
APA StyleShen, B., Shangguan, X., Yin, Z., Wu, S., Zhang, Q., Peng, W., Li, J., Zhang, L., & Chen, J. (2021). Inhibitory Effect of Fisetin on α-Glucosidase Activity: Kinetic and Molecular Docking Studies. Molecules, 26(17), 5306. https://doi.org/10.3390/molecules26175306





