From Waste to Green Applications: The Use of Recovered Gold and Palladium in Catalysis
Abstract
:1. Introduction
2. Gold
2.1. Primary Production of Gold
2.2. Gold from Secondary Sources
2.3. The Role of Gold in Catalysis
2.4. Direct Application of Recovered Gold in Catalysis
3. Palladium
3.1. Primary Production of Palladium
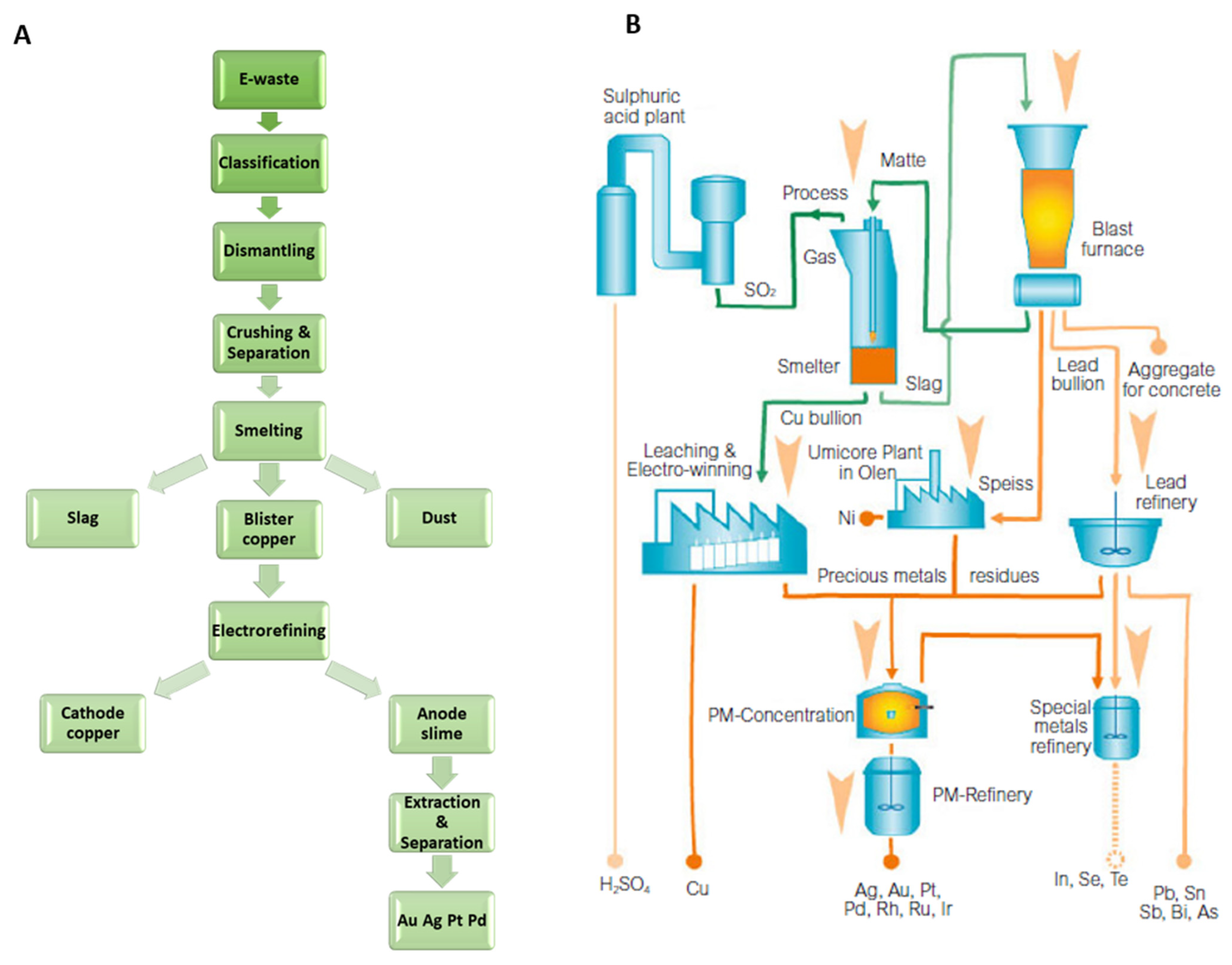
3.2. Palladium from Secondary Sources
3.3. The Role of Palladium in Catalysis
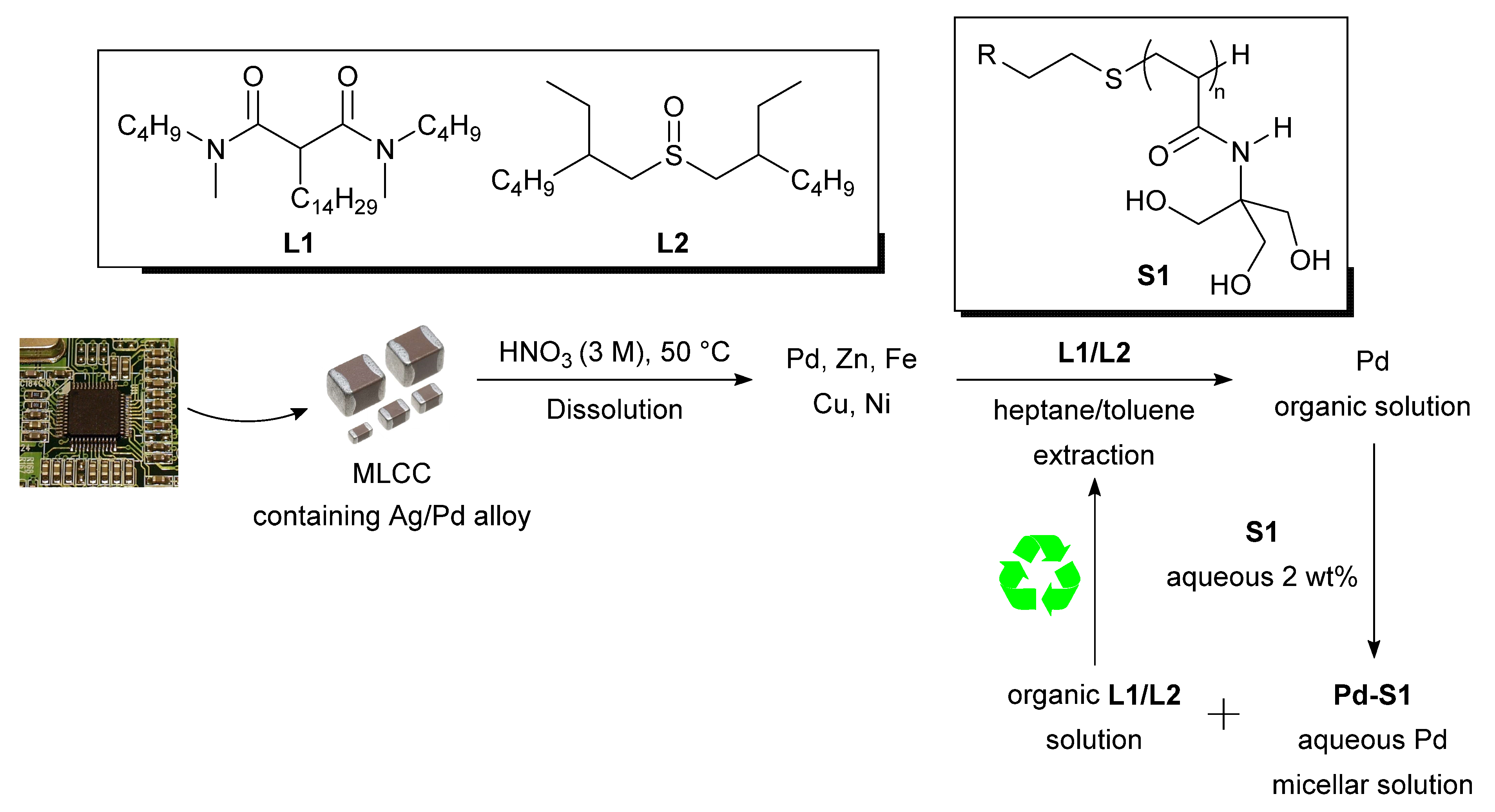
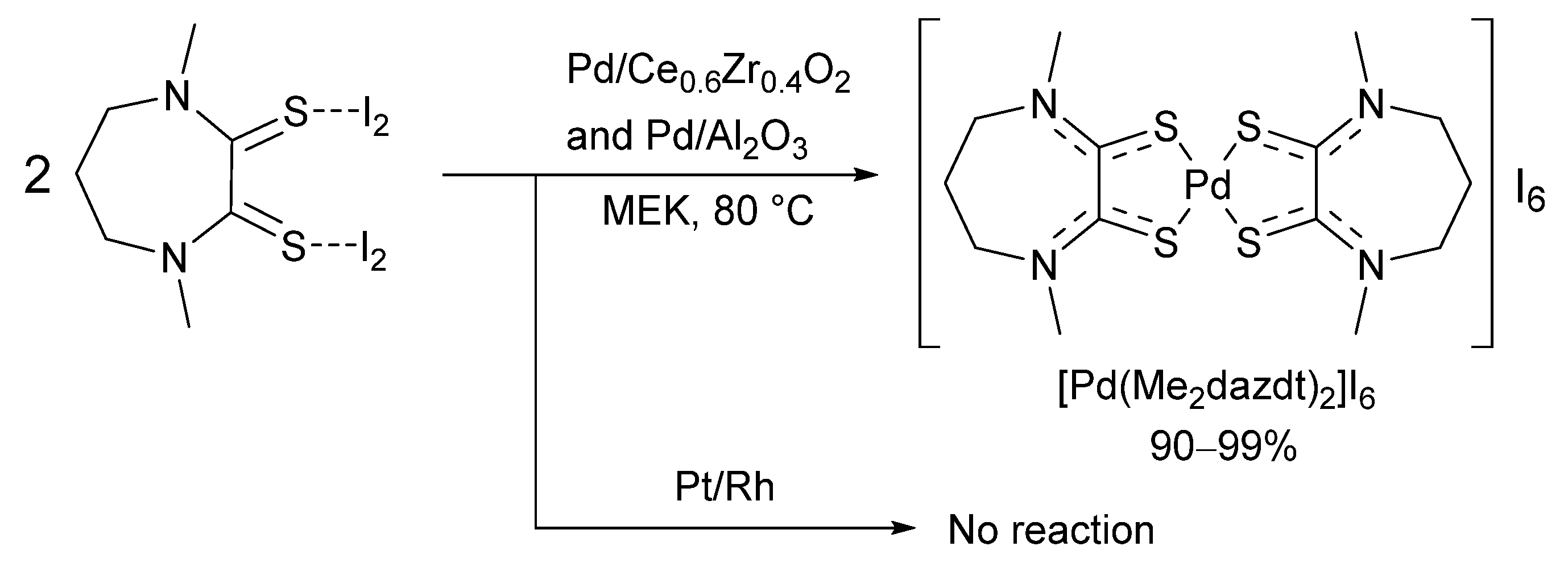
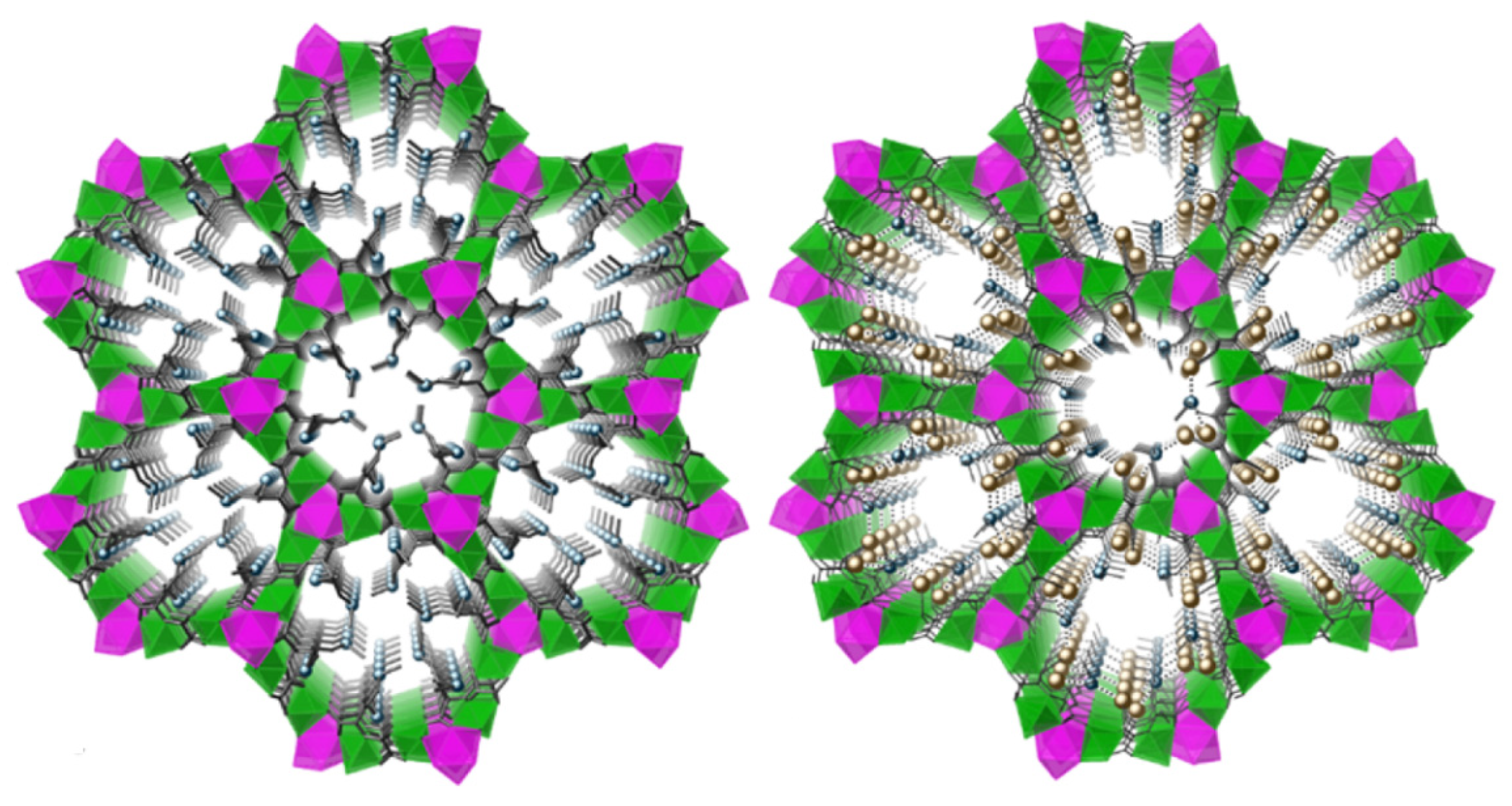
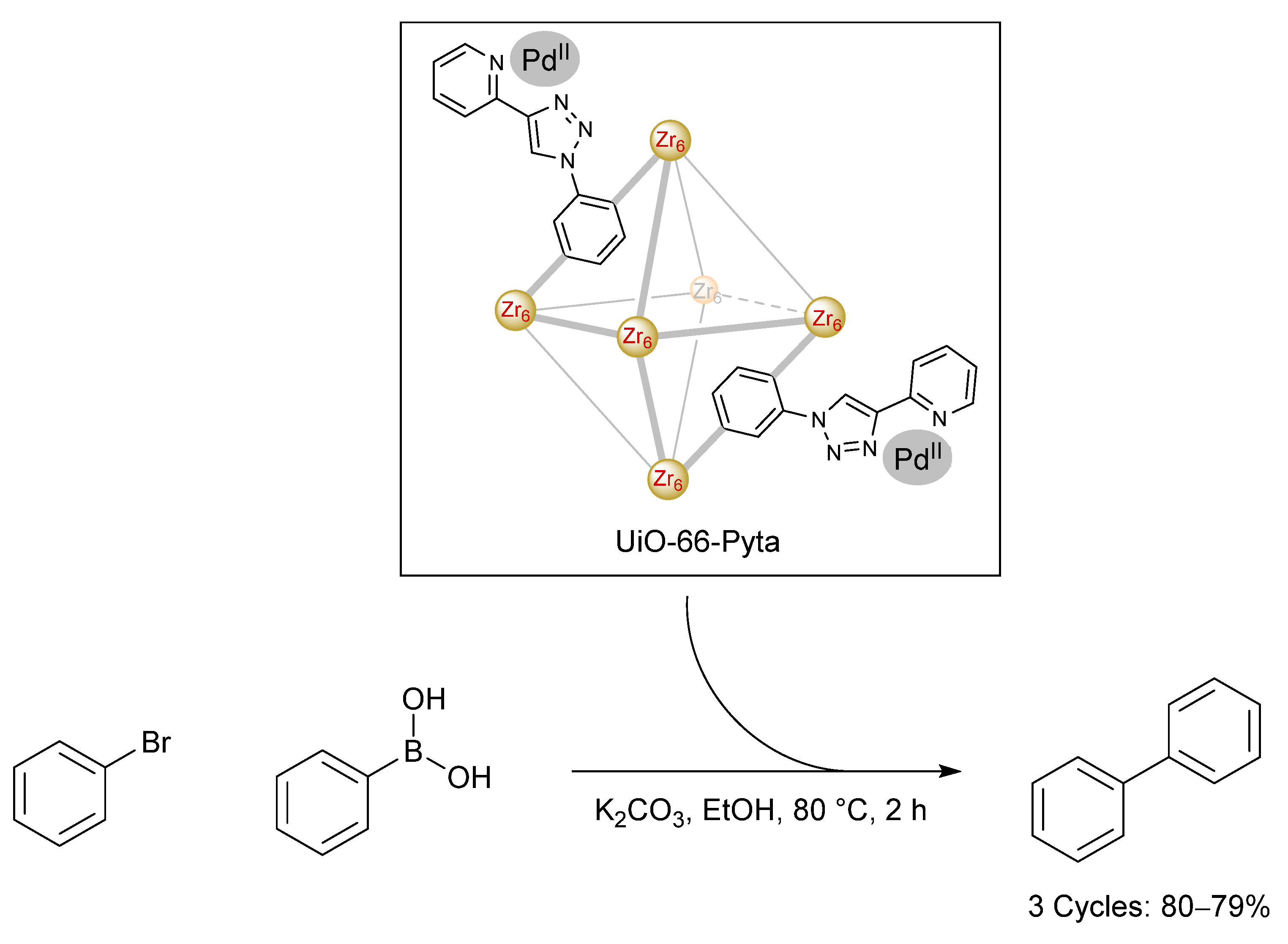
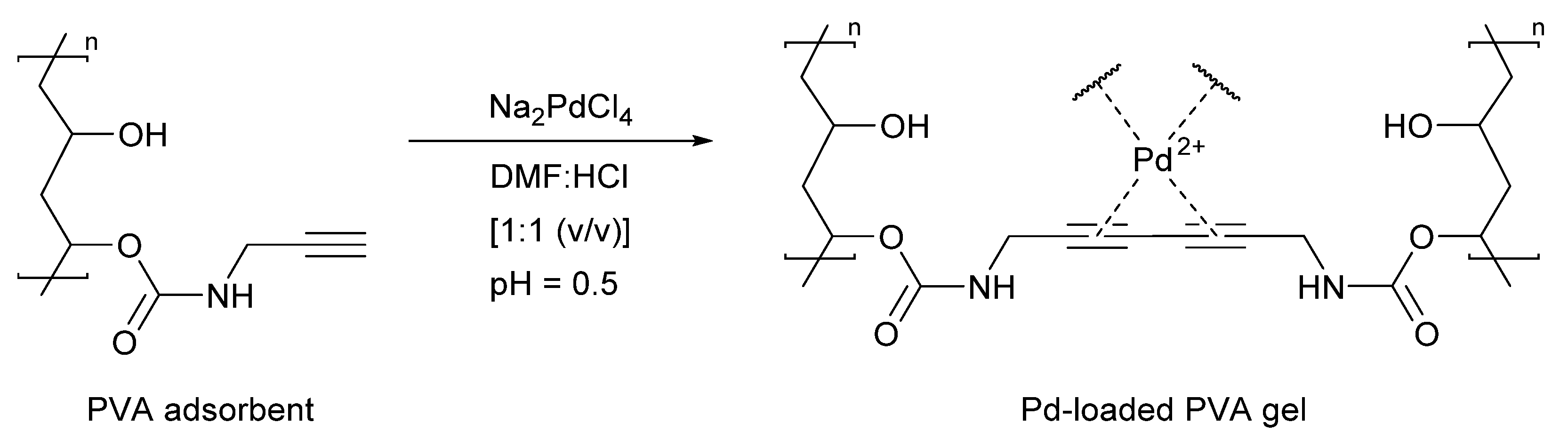
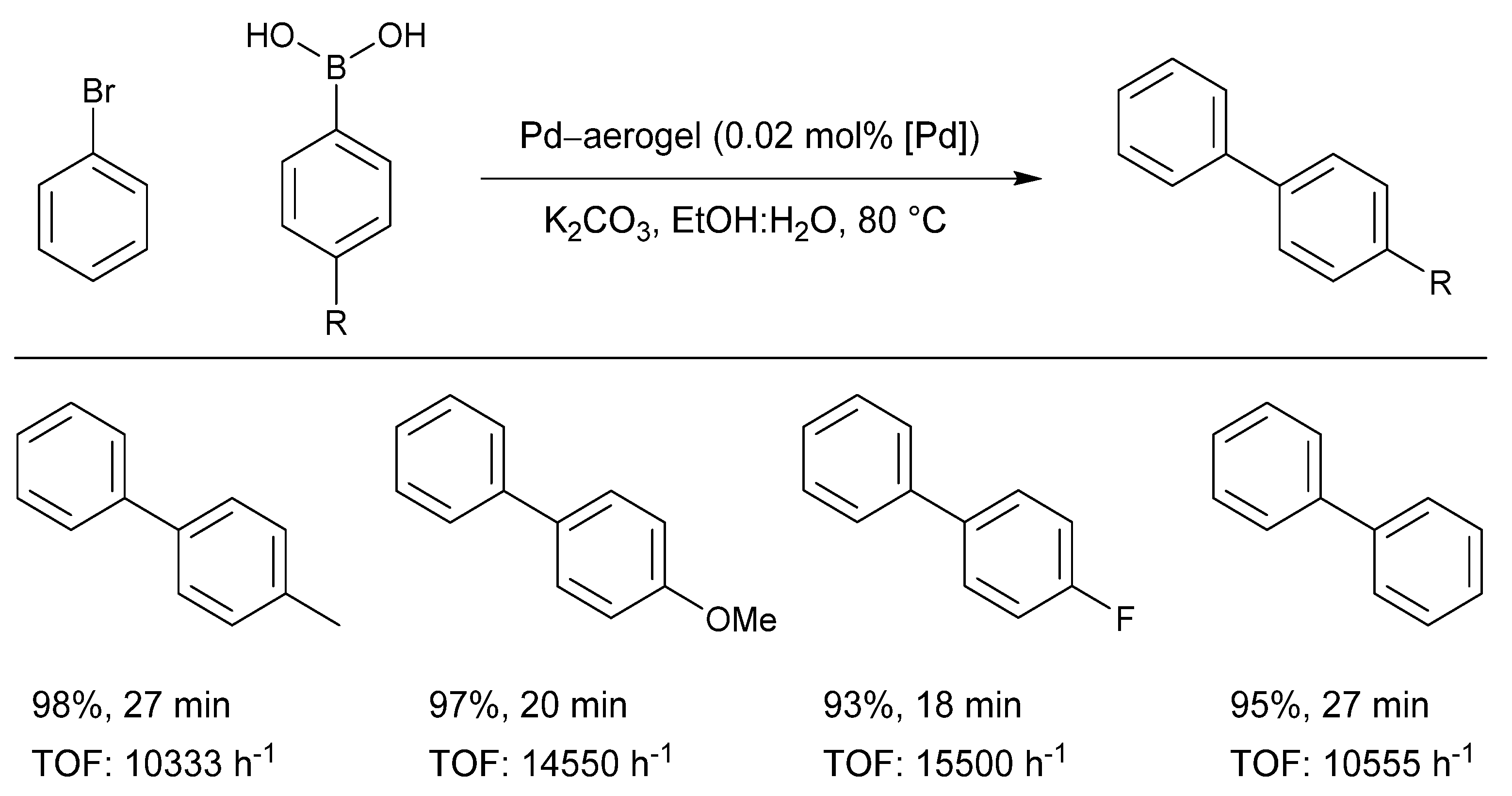
3.4. Direct Application of Recovered Palladium in Catalysis
4. Conclusions and Future Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- European Commission. Report on critical raw materials for the EU, Report of the Ad hoc Working Group on defining critical raw materials. 2015. Available online: https://ec.europa.eu/docsroom/documents/10010/attachments/1/translations/en/renditions/pdf (accessed on 27 August 2021).
- Nuss, P.; Eckelman, M.J. Life cycle assessment of metals: A scientific synthesis. PLoS ONE 2014, 9, e101298. [Google Scholar] [CrossRef]
- Singh, P.K.; Singh, R.S.; Singh, S. Environmental and Social Impacts of Mining and Their Mitigation. In Proceedings of the National Seminar ESIMM-2016, Kolkata, India, 20 September 2016; pp. 17–29. [Google Scholar]
- ACS Green Chemistry Institute: Endangered Elements. 2020. Available online: https://www.acs.org/content/acs/en/greenchemistry/research-innovation/endangered-elements.html (accessed on 9 September 2020).
- Johnson Matthey. PGM Market Report. PGM Mark. Rep. 2019, 1–48. Available online: http://www.platinum.matthey.com/services/market-research/pgm-market-reports (accessed on 9 September 2020).
- Tasker, S.Z.; Standley, E.A.; Jamison, T.F. Recent advances in homogeneous nickel catalysis. Nature 2014, 509, 299–309. [Google Scholar] [CrossRef] [Green Version]
- Mako, T.L.; Byers, J.A. Recent advances in iron-catalysed cross coupling reactions and their mechanistic underpinning. Inorg. Chem. Front. 2016, 3, 766–790. [Google Scholar] [CrossRef]
- Sun, C.L.; Shi, Z.J. Transition-metal-free coupling reactions. Chem. Rev. 2014, 114, 9219–9280. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.J.; Mankad, N.P. C-C and C-X coupling reactions of unactivated alkyl electrophiles using copper catalysis. Chem. Soc. Rev. 2020, 49, 8036–8064. [Google Scholar] [CrossRef]
- Mesganaw, T.; Garg, N.K. Ni- and Fe-catalyzed cross-coupling reactions of phenol derivatives. Org. Process Res. Dev. 2013, 17, 29–39. [Google Scholar] [CrossRef]
- Hazari, N.; Melvin, P.R.; Beromi, M.M. Well-defined nickel and palladium precatalysts for cross-coupling. Nat. Rev. Chem. 2017, 1, 1–17. [Google Scholar] [CrossRef]
- Hewitt, A.; Keel, T.; Tauber, M.; Le-Fiedler, T. The Ups and Downs of Gold Recycling. Available online: https://www.bcg.com/publications/2015/metals-mining-cost-efficiency-ups-and-downs-of-gold-recycling.aspx (accessed on 26 June 2020).
- Geology Page, Mining: What Is Gold Mining? How Is Gold Mined? Available online: http://www.geologypage.com/2019/04/what-is-gold-mining-how-is-gold-mined.html (accessed on 11 June 2020).
- Harris, W. How Gold Works. Available online: https://science.howstuffworks.com/gold3.htm (accessed on 11 June 2020).
- Dozolme, P. Specific and Non-Specific Hazards in Underground Mines. Available online: https://www.thebalancesmb.com/specific-and-non-specific-hazards-in-underground-mines-2367338 (accessed on 11 June 2020).
- Freeport-McMoRan. Indonesia. Available online: https://www.fcx.com/operations/indonesia (accessed on 12 June 2020).
- Extraction and Beneficiation of Ore and Minerals-Gold; U.S. Environmental Protection Agency: Washington, DC, USA, 1994.
- SGS. Cyanide Leaching. Available online: https://www.sgs.sg/en/mining/metallurgy-and-process-design/cyanidation-technologies/cyanide-leaching (accessed on 22 June 2020).
- Brilliant Earth. Gold Mining and the Environment. Available online: https://www.brilliantearth.com/gold-mining-environment/ (accessed on 18 June 2020).
- Freemantle, M. Sodium Cyanide. Available online: https://www.chemistryworld.com/podcasts/sodium-cyanide/4011120.article (accessed on 4 June 2020).
- Esdaile, L.J.; Chalker, J.M. The Mercury Problem in Artisanal and Small-Scale Gold Mining. Chem. A Eur. J. 2018, 24, 6905–6916. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Economic Forum. A New Circular Vision for Electronics Time for a Global Reboot. 2019. Available online: http://www3.weforum.org/docs/WEF_A_New_Circular_Vision_for_Electronics.pdf (accessed on 9 May 2021).
- Wan, R.-Y.; Marc, L.K.; Clayton, R.B. Hydrometallurgical Process for the Recovery of Precious Metal Values from Precious Metal, Ores with Thiosulfate Lixiviant. U.S. Patent 5,354,359, USOO5354359A, 11 October 1994. [Google Scholar]
- Örgül, S.; Atalay, Ü. Gold Extraction from kaymaz gold ore by thiourea leaching. Dev. Miner. Process. 2000, 13, C6–C22. [Google Scholar]
- Norgate, T.; Haque, N. Using life cycle assessment to evaluate some environmental impacts of gold production. J. Clean. Prod. 2012, 29–30, 53–63. [Google Scholar] [CrossRef]
- World Ocean Review. How Much Metal Does the Ore Contain? Available online: https://worldoceanreview.com/en/wor-3/mineral-resources/deposits-and-markets/how-much-metal-does-the-ore-contain/ (accessed on 24 July 2020).
- Angelo, S. Top Primary Silver Miners Average Yield Falls to Record Low. Available online: https://www.moneymetals.com/news/2019/06/14/silver-miners-average-yield-falls-001795 (accessed on 24 July 2020).
- Septoff, A. How the 20 Tons of Mine Waste Per Gold Ring Figure Was Calculated. Available online: https://earthworks.org/publications/how_the_20_tons_of_mine_waste_per_gold_ring_figure_was_calculated/ (accessed on 17 June 2020).
- The World Counts. Environmental Effects of Gold Mining. Available online: https://www.theworldcounts.com/challenges/planet-earth/mining/environmental-effects-of-gold-mining (accessed on 17 June 2020).
- Kramer, S.; Gagosz, F. Gold Catalysis: An Homogeneous Approach; Imperial College Press: London, UK, 2014. [Google Scholar]
- Hagelüken, C.; Corti, C.W. Recycling of gold from electronics: Cost-effective use through “design for recycling”. Gold Bull. 2010, 43, 209–220. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.; Geng, Y.; Hong, J.; Dong, H.; Cui, X.; Sun, M.; Zhang, Q. Life cycle assessment of gold production in China. J. Clean. Prod. 2018, 179, 143–150. [Google Scholar] [CrossRef]
- Tuncuk, A.; Stazi, V.; Akcil, A.; Yazici, E.Y.; Deveci, H. Aqueous metal recovery techniques from e-scrap: Hydrometallurgy in recycling. Miner. Eng. 2012, 25, 28–37. [Google Scholar] [CrossRef]
- European Commission. Waste Electrical & Electronic Equipment (WEEE). Available online: https://ec.europa.eu/environment/waste/weee/index_en.htm, (accessed on 27 May 2020).
- Mudgal, S.; Slater, L. Impacts of gold extraction in the EU. Eur. Com. Eurostat. 2010, 33, 1–44. [Google Scholar]
- Forti, V.; Baldé, C.P.; Kuehr, R.; Bel, G. The Global E-Waste Monitor 2020; United Nations University/United Nations Institute for Training and Research, International Telecommunication Union, and International Solid Waste Association (ISWA). Bonn, Geneva and Rotterdam. 2020. Available online: http://ewastemonitor.info/ (accessed on 30 May 2021).
- Directive 2002/96/EC of the European Parliament and of the Council of 27 January 2003 on waste electrical and electronic equipment (WEEE) - Joint declaration of the European Parliament, the Council and the Commission relating to Article 9. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32002L0096 (accessed on 27 May 2020).
- Directive 2012/19/EU of the European Parliament and of the Council of 4 July 2012 on waste electrical and electronic equipment (WEEE) Text with EEA relevance. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32012L0019 (accessed on 27 May 2020).
- Ding, Y.; Zhang, S.; Liu, B.; Zheng, H.; Chang, C.C. Ekberg, Recovery of precious metals from electronic waste and spent catalysts: A review. Resour. Conserv. Recycl. 2019, 141, 284–298. [Google Scholar] [CrossRef]
- Hagelueken, C. Recycling of Electronic Scrap at Umicore Precious Metals. Acta Metall. Slovaca. 2006, 12, 111–120. [Google Scholar]
- Gold Supply. Processing, Smelting and Refining Gold. Available online: https://www.gold.org/about-gold/gold-supply/gold-refining (accessed on 6 August 2019).
- Rao, M.D.; Singh, K.K.; Morrison, C.A.; Love, J.B. Challenges and opportunities in the recovery of gold from electronic waste. RSC Adv. 2020, 10, 4300–4309. [Google Scholar] [CrossRef] [Green Version]
- Tasker, P.; Plieger, P.; West, L. Metal Complexes for Hydrometallurgy and Extraction. In Comprehensive Coordination Chemistry II; McCleverty, J.A., Meyer, T.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2003; Volume 9, pp. 759–807. [Google Scholar]
- Akcil, A.; Erust, C.; Gahan, C.S.E.; Ozgun, M.; Sahin, M.; Tuncuk, A. Precious metal recovery from waste printed circuit boards using cyanide and non-cyanide lixiviants—A review. Waste Manag. 2015, 45, 258–271. [Google Scholar] [CrossRef]
- Sparrow, G.J.; Woodcock, J.T. Cyanide and Other Lixiviant Leaching Systems for Gold with Some Practical Applications. Min. Proc. Ext. Met. Rev. 1995, 14, 193–247. [Google Scholar] [CrossRef]
- Serpe, A. Green Chemistry for Precious Metals Recovery from WEEE; Elsevier Ltd.: Amsterdam, The Netherlands, 2018; pp. 271–332. [Google Scholar] [CrossRef]
- Hoffmann, J. Advances in the extractive metallurgy of selected rare and precious metals. J. Miner. Met. Mater. Soc. 1991, 43, 18–23. [Google Scholar] [CrossRef]
- Wongsawa, T.; Traiwongsa, N.; Pancharoen, U.; Nootong, K. A review of the recovery of precious metals using ionic liquid extractants in hydrometallurgical processes. Hydrometallurgy 2020, 198, 105488. [Google Scholar] [CrossRef]
- Li, H.; Eksteen, J.; Oraby, E. Hydrometallurgical recovery of metals from waste printed circuit boards (WPCBs): Current status and perspectives – A review. Resour. Conserv. Recycl. 2019, 139, 122–139. [Google Scholar] [CrossRef]
- Trinh, H.B.; Lee, J.C.; Suh, Y.J.; Lee, J. A review on the recycling processes of spent auto-catalysts: Towards the development of sustainable metallurgy. Waste Manag. 2020, 114, 148–165. [Google Scholar] [CrossRef] [PubMed]
- Cyganowski, P. Synthesis of Adsorbents with Anion Exchange and Chelating Properties for Separation and Recovery of Precious Metals—A Review. Solvent Extr. Ion Exch. 2020, 38, 143–165. [Google Scholar] [CrossRef]
- Cui, J.; Zhang, L. Metallurgical recovery of metals from electronic waste: A review. J. Hazard. Mater. 2008, 158, 228–256. [Google Scholar] [CrossRef] [PubMed]
- Norman, R.O.C.; Parr, W.J.E.; Thomas, C.B. The reactions of alkynes, cyclopropanes, and benzene derivatives with gold (III). J. Chem. Soc. Perkin Trans. 1976, 18, 1983–1987. [Google Scholar] [CrossRef]
- Dyker, G. An Eldorado for Homogeneous Catalysis? Org. Synth. Set. 2008, 48, 48–55. [Google Scholar]
- Cambeiro, X.C.; Ahlsten, N.; Larrosa, I. Au-Catalyzed Cross-Coupling of Arenes via Double C-H Activation. J. Am. Chem. Soc. 2015, 137, 15636–15639. [Google Scholar] [CrossRef] [PubMed]
- Seppänen, O.; Aikonen, S.; Muuronen, M.; Alamillo-Ferrer, C.; Burés, J.; Helaja, J. Dual H-bond activation of NHC-Au(i)-Cl complexes with amide functionalized side-arms assisted by H-bond donor substrates or acid additives. Chem. Commun. 2020, 56, 14697–14700. [Google Scholar] [CrossRef]
- Ball, L.T.; Lloyd-Jones, G.C.; Russell, C.A. Gold-catalyzed direct arylation. Science 2012, 337, 1644–1648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ball, L.T.; Lloyd-Jones, G.C.; Russell, C.A. Gold-catalyzed oxidative coupling of arylsilanes and arenes: Origin of selectivity and improved precatalyst. J. Am. Chem. Soc. 2014, 136, 254–264. [Google Scholar] [CrossRef] [PubMed]
- Cai, R.; Lu, M.; Aguilera, E.Y.; Xi, Y.; Akhmedov, N.G.; Petersen, J.L.; Chen, H.; Shi, X. Ligand-Assisted Gold-Catalyzed Cross-Coupling with Aryldiazonium Salts: Redox Gold Catalysis without an External Oxidant. Angew. Chemie Int. Ed. 2015, 54, 8772–8776. [Google Scholar] [CrossRef] [PubMed]
- Miranda-pizarro, J.; Luo, Z.; Moreno, J.J.; Dickie, D.A.; Gunnoe, T.B. Reductive C−C Coupling from Molecular Au(I) Hydrocarbyl Complexes: A Mechanistic Study. J. Am. Chem. Soc. 2020, 143, 2509–2522. [Google Scholar] [CrossRef] [PubMed]
- Hopkinson, M.N.; Tlahuext-Aca, A.; Glorius, F. Merging Visible Light Photoredox and Gold Catalysis. Acc. Chem. Res. 2016, 49, 2261–2272. [Google Scholar] [CrossRef] [PubMed]
- Ciriminna, R.; Falletta, E.; della Pina, C.; Teles, J.H.; Pagliaro, M. Industrial Applications of Gold Catalysis. Angew. Chem. Int. Ed. 2016, 55, 14210–14217. [Google Scholar] [CrossRef] [PubMed]
- Rocchigiani, L.; Bochmann, M. Recent Advances in Gold(III) Chemistry: Structure, Bonding, Reactivity, and Role in Homogeneous Catalysis. Chem. Rev. 2020, 121, 8364–8451. [Google Scholar] [CrossRef]
- Weyrauch, J.P.; Hashmi, A.S.K.; Schuster, A.; Hengst, T.; Schetter, S.; Littmann, A.; Rudolph, M.; Hamzic, M.; Visus, J.; Rominger, F.; et al. Cyclization of propargylic amides: Mild access to oxazole derivatives. Chem. A Eur. J. 2010, 16, 956–963. [Google Scholar] [CrossRef]
- Dyker, G.; Muth, E.; Hashmi, A.S.K.; Ding, L. Gold(III) Chloride-Catalyzed Addition Reactions of Electron-Rich Arenes to Methyl Vinyl Ketone. Adv. Synth. Catal. 2003, 345, 1247–1252. [Google Scholar] [CrossRef]
- Wu, C.Y.; Horibe, T.; Jacobsen, C.B.; Toste, F.D. Stable gold(III) catalysts by oxidative addition of a carbon-carbon bond. Nature 2015, 517, 449–454. [Google Scholar] [CrossRef] [Green Version]
- Harper, M.J.; Emmett, E.J.; Bower, J.F.; Russell, C.A. Oxidative 1,2-Difunctionalization of Ethylene via Gold-Catalyzed Oxyarylation. J. Am. Chem. Soc. 2017, 139, 12386–12389. [Google Scholar] [CrossRef] [Green Version]
- Mon, M.; Ferrando-Soria, J.; Grancha, T.; Fortea-Pérez, F.R.; Gascon, J.; Leyva-Pérez, A.; Armentano, D.; Pardo, E. Selective Gold Recovery and Catalysis in a Highly Flexible Methionine-Decorated Metal-Organic Framework. J. Am. Chem. Soc. 2016, 138, 7864–7867. [Google Scholar] [CrossRef]
- Lahtinen, E.; Kivijärvi, L.; Tatikonda, R.; Väisänen, A.; Rissanen, K.; Haukka, M. Selective Recovery of Gold from Electronic Waste Using 3D-Printed Scavenger. ACS Omega. 2017, 2, 7299–7304. [Google Scholar] [CrossRef]
- Lahtinen, E.; Kukkonen, E.; Kinnunen, V.; Lahtinen, M.; Kinnunen, K.; Suvanto, S.; Vaïsänen, A.; Haukka, M. Gold Nanoparticles on 3D-Printed Filters: From Waste to Catalysts. ACS Omega. 2019, 4, 16891–16898. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Z.; Li, P.; Fang, Y.; Yan, L.; Zhou, W.; Fan, X.; Liu, H. One-step recovery of noble metal ions from oil/water emulsions by chitin nanofibrous membrane for further recycling utilization. Carbohydr. Polym. 2019, 223, 115064. [Google Scholar] [CrossRef]
- Asere, T.G.; Mincke, S.; Folens, K.; Bussche, F.V.; Lapeire, L.; Verbeken, K.; van der Voort, P.; Tessema, D.A.; Laing, G.D.; Stevens, C.V. Dialdehyde carboxymethyl cellulose cross-linked chitosan for the recovery of palladium and platinum from aqueous solution. React. Funct. Polym. 2019, 141, 145–154. [Google Scholar] [CrossRef]
- Cui, Q.; Zhao, H.; Luo, G.; Xu, J. An efficient chitosan/silica composite core-shell microspheres-supported Pd catalyst for aryl iodides sonogashira coupling reactions. Ind. Eng. Chem. Res. 2017, 56, 143–152. [Google Scholar] [CrossRef]
- Mincke, S.; Asere, T.G.; Verheye, I.; Folens, K.; Bussche, F.V.; Lapeire, L.; Verbeken, K.; van der Voort, P.; Tessema, D.A.; Fufa, F.; et al. Functionalized chitosan adsorbents allow recovery of palladium and platinum from acidic aqueous solutions. Green Chem. 2019, 21, 2295–2306. [Google Scholar] [CrossRef]
- Berillo, D.; Cundy, A. 3D-macroporous chitosan-based scaffolds with in situ formed Pd and Pt nanoparticles for nitrophenol reduction. Carbohydr. Polym. 2018, 192, 166–175. [Google Scholar] [CrossRef] [Green Version]
- Glaister, B.J.; Mudd, G.M. The environmental costs of platinum-PGM mining and sustainability: Is the glass half-full or half-empty? Miner. Eng. 2010, 23, 438–450. [Google Scholar] [CrossRef]
- Alison Cowley. Johnson Matthey PGM Market Report. 2020. Available online: http://www.platinum.matthey.com/documents/new-item/pgm-market-reports/pgm-market-report-may-2020.pdf (accessed on 26 March 2021).
- Palladium Price Per Gram. 2020. Available online: https://www.gold.co.uk/palladium-price/palladium-price-per-gram/ (accessed on 9 September 2020).
- Stocks, J.; Blunden, J.R.; Down, C.G. Metal Mining and the Environment; American Geological Institute: Alexandria, VA, USA, 1974. [Google Scholar]
- Johnson Matthey. Johnson Matthey PGM Market Report. PGM Mark. Rep. 2014. Available online: http://www.platinum.matthey.com/documents/new-item/pgmmarketreports/pgmmarketreportmay2014.pdf (accessed on 9 September 2020).
- Hagelüken, C. Recycling the Platinum Group Metals. Platin. Met. Rev. 2012, 56, 29–35. [Google Scholar] [CrossRef]
- Benson, M.; Bennett, C.R.; Patel, M.K.; Harry, J.E.; Cross, M. Collector-metal behaviour in the recovery of platinum-group metals from catalytic converters. Miner. Process. Extr. Metall. 2000, 109, C6–C10. [Google Scholar] [CrossRef]
- Benson, M.; Bennett, C.R.; Harry, J.E.; Patel, M.K.; Cross, M. The recovery mechanism of platinum group metals from catalytic converters in spent automotive exhaust systems. Resour. Conserv. Recycl. 2000, 31, 1–7. [Google Scholar] [CrossRef]
- Amatayakul, W.; Ramnäs, O. Life cycle assessment of a catalytic converter for passenger cars. J. Clean. Prod. 2001, 9, 395–403. [Google Scholar] [CrossRef]
- Sharma, S.; Kumar, A.S.K.; Rajesh, N. A perspective on diverse adsorbent materials to recover precious palladium and the way forward. RSC Adv. 2017, 7, 52133–52142. [Google Scholar] [CrossRef] [Green Version]
- Paiva, A.P. Recycling of palladium from spent catalysts using solvent extraction—Some critical points. Metals 2017, 7, 505. [Google Scholar] [CrossRef] [Green Version]
- Torborg, C.; Beller, M. Recent applications of palladium-catalyzed coupling reactions in the pharmaceutical, agrochemical, and fine chemical industries. Adv. Synth. Catal. 2009, 351, 3027–3043. [Google Scholar] [CrossRef]
- Nicolaou, K.C.; Bulger, P.G.; Sarlah, D. Palladium-catalyzed cross-coupling reactions in total synthesis. Angew. Chemie Int. Ed. 2005, 44, 4442–4489. [Google Scholar] [CrossRef] [PubMed]
- Devendar, P.; Qu, R.Y.; Kang, W.M.; He, B.; Yang, G.F. Palladium-Catalyzed Cross-Coupling Reactions: A Powerful Tool for the Synthesis of Agrochemicals. J. Agric. Food Chem. 2018, 66, 8914–8934. [Google Scholar] [CrossRef] [PubMed]
- Tamao, K.; Miyaura, N.; Hiyama, T.; Shirakawa, E.; Fugami, K.; Kosugi, M.; Muci, A.; Buchwald, S.L.; Miura, M.; Nomura, M. Cross-Coupling Reactions: A Practical Guide; Springer: Berlin, Germany, 2002. [Google Scholar]
- Xu, S.; Kim, E.H.; Wei, A.; Negishi, E.I. Pd- and Ni-catalyzed cross-coupling reactions in the synthesis of organic electronic materials. Sci. Technol. Adv. Mater. 2014, 15, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.G.; Boström, J. Analysis of Past and Present Synthetic Methodologies on Medicinal Chemistry: Where Have All the New Reactions Gone? J. Med. Chem. 2016, 59, 4443–4458. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biffis, A.; Centomo, P.; del Zotto, A.; Zecca, M. Pd Metal Catalysts for Cross-Couplings and Related Reactions in the 21st Century: A Critical Review. Chem. Rev. 2018, 118, 2249–2295. [Google Scholar] [CrossRef] [PubMed]
- Budarin, V.L.; Shuttleworth, P.S.; Clark, J.H.; Luque, R. Industrial Applications of C-C Coupling Reactions. Curr. Org. Synth. 2011, 7, 614–627. [Google Scholar] [CrossRef]
- Roy, D.; Uozumi, Y. Recent Advances in Palladium-Catalyzed Cross-Coupling Reactions at ppm to ppb Molar Catalyst Loadings. Adv. Synth. Catal. 2018, 360, 602–625. [Google Scholar] [CrossRef]
- Molnár, Á. Efficient, selective, and recyclable palladium catalysts in carbon-carbon coupling reactions. Chem. Rev. 2011, 111, 2251–2320. [Google Scholar] [CrossRef]
- Daliran, S.; Ghazagh-Miri, M.; Oveisi, A.R.; Khajeh, M.; Navalón, S.; Âlvaro, M.; Ghaffari-Moghaddam, M.; Delarami, H.S.; García, H. A Pyridyltriazol Functionalized Zirconium Metal-Organic Framework for Selective and Highly Efficient Adsorption of Palladium. ACS Appl. Mater. Interfaces. 2020, 12, 25221–25232. [Google Scholar] [CrossRef]
- Lacanau, V.; Bonneté, F.; Wagner, P.; Schmitt, M.; Meyer, D.; Bihel, F.; Contino-Pépin, C.; Bourgeois, D. From Electronic Waste to Suzuki-Miyaura Cross-Coupling Reaction in Water: Direct Valuation of Recycled Palladium in Catalysis. ChemSusChem 2020, 13, 5224–5230. [Google Scholar] [CrossRef]
- Yang, X.; Chen, J.; Hu, L.; Wei, J.; Shuai, M.; Huang, D.; Yue, G.; Astruc, D.; Zhao, P. Palladium Separation by Pd-Catalyzed Gel Formation via Alkyne Coupling. Chem. Mater. 2019, 31, 7386–7394. [Google Scholar] [CrossRef]
- Serpe, A.; Artizzu, F.; Mercuri, M.L.; Pilia, L.; Deplano, P. Charge transfer complexes of dithioxamides with dihalogens as powerful reagents in the dissolution of noble metals. Coord. Chem. Rev. 2008, 252, 1200–1212. [Google Scholar] [CrossRef]
- Bigoli, F.; Deplano, P.; Mercuri, M.L.; Pellinghelli, M.A.; Pintus, G.; Serpe, A.; Trogu, E.F. A powerful new oxidation agent towards metallic gold powder: N,N′-dimethylperhydrodiàzepine-2,3-dithione (D) bis(diiodine). Synthesis and X-ray structure of [AuDI2]I3. Chem. Commun. 1998, 484, 2351–2352. [Google Scholar] [CrossRef]
- Serpe, A.; Bigoli, F.; Cabras, M.C.; Fornasiero, P.; Graziani, M.; Mercuri, M.L.; Montini, T.; Pilia, L.; Trogu, E.F.; Deplano, P. Pd-dissolution through a mild and effective one-step reaction and its application for Pd-recovery from spent catalytic converters. Chem. Commun. 2005, 8, 1040–1042. [Google Scholar] [CrossRef] [PubMed]
- Deplano, P.; Fornasiero, P.; Graziani, M.; Mercuri, M.L.; Serpe, A.; Trogu, E.F. Method for the Recovery of Palladium. European Patent EP1743044B1, 12 April 2005. [Google Scholar]
- Twigg, M.V. Catalytic control of emissions from cars. Catal. Today 2011, 163, 33–41. [Google Scholar] [CrossRef]
- Gombac, V.; Montini, T.; Falqui, A.; Loche, D.; Prato, M.; Genovese, A.; Mercuri, M.L.; Serpe, A.; Fornasiero, P.; Deplano, P. From: Trash to resource: Recovered-Pd from spent three-way catalysts as a precursor of an effective photo-catalyst for H2 production. Green Chem. 2016, 18, 2745–2752. [Google Scholar] [CrossRef] [Green Version]
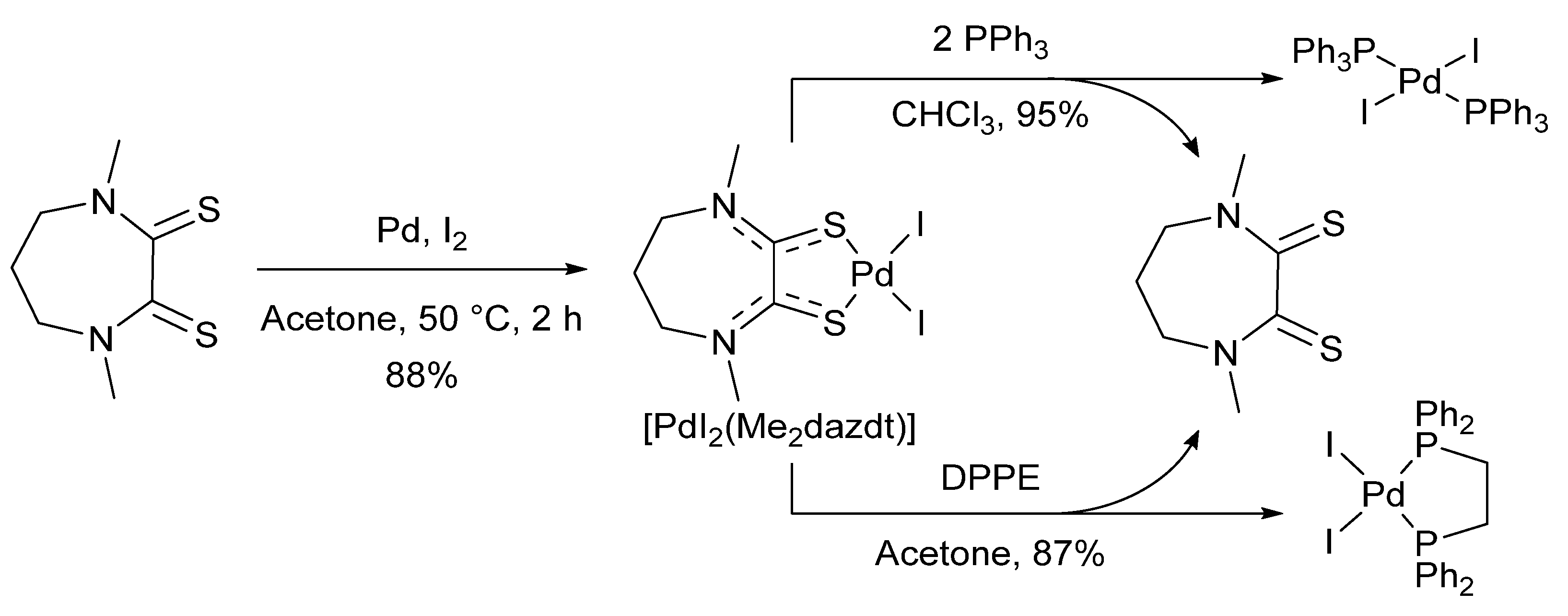
- Jantan, K.A.; Kwok, C.Y.; Chan, K.W.; Marchiò, L.; White, A.J.P.; Deplano, P.; Serpe, A.; Wilton-Ely, J.D.E.T. From recovered metal waste to high-performance palladium catalysts. Green Chem. 2017, 19, 5846–5853. [Google Scholar] [CrossRef] [Green Version]
- Jantan, K.A.; Chan, K.W.; Melis, L.; White, A.J.P.; Marchiò, L.; Deplano, P.; Serpe, A.; Wilton-Ely, J.D.E.T. From Recovered Palladium to Molecular and Nanoscale Catalysts. ACS Sustain. Chem. Eng. 2019, 7, 12389–12398. [Google Scholar] [CrossRef]
- Dick, A.R.; Hull, K.L.; Sanford, M.S. A Highly Selective Catalytic Method for the Oxidative Functionalization of C-H Bonds. J. Am. Chem. Soc. 2004, 126, 2300–2301. [Google Scholar] [CrossRef]
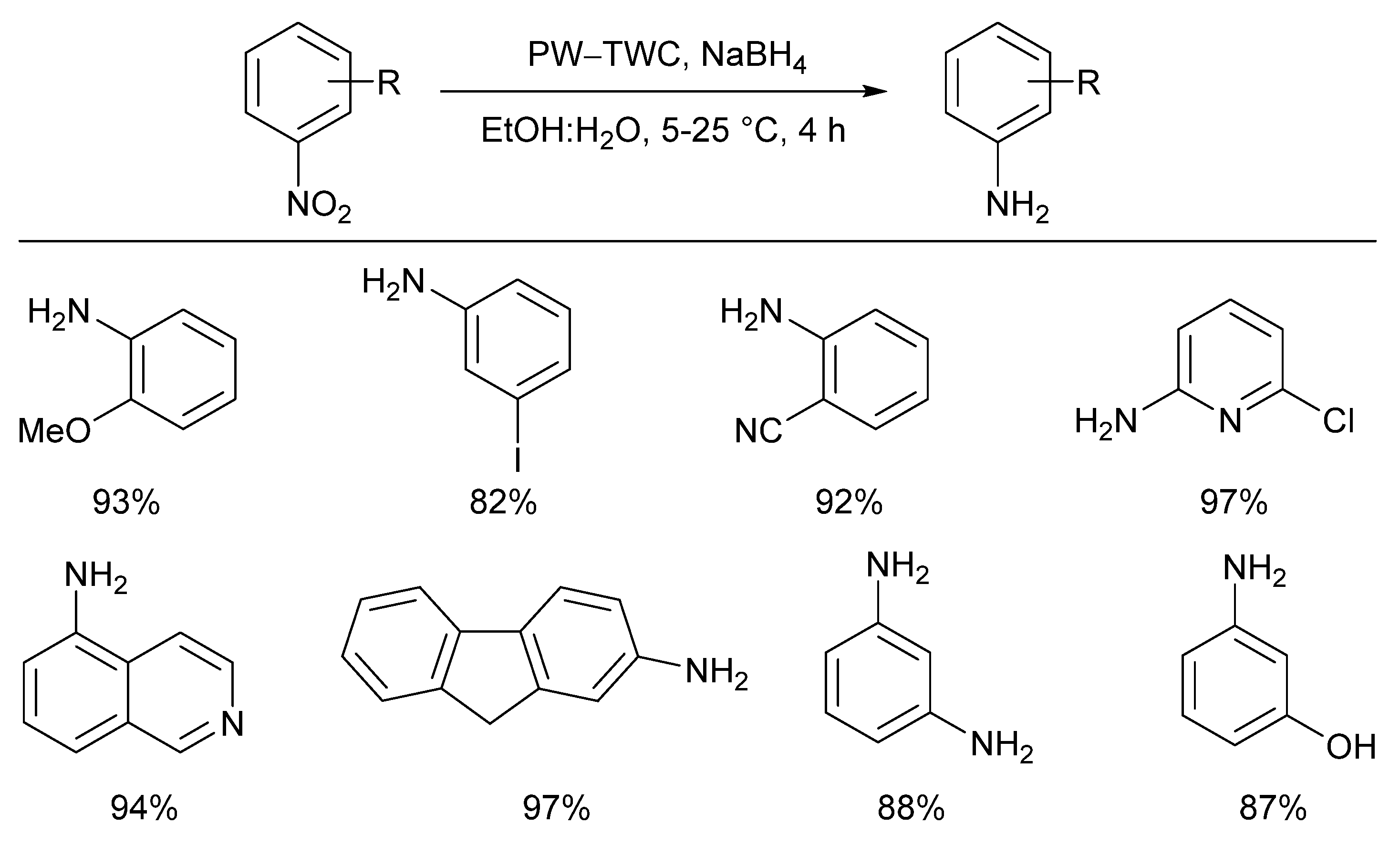
- Genc, H. Efficient reductions of various nitroarenes with scrap automobile catalyst and NaBH4. Catal. Commun. 2015, 67, 64–67. [Google Scholar] [CrossRef]
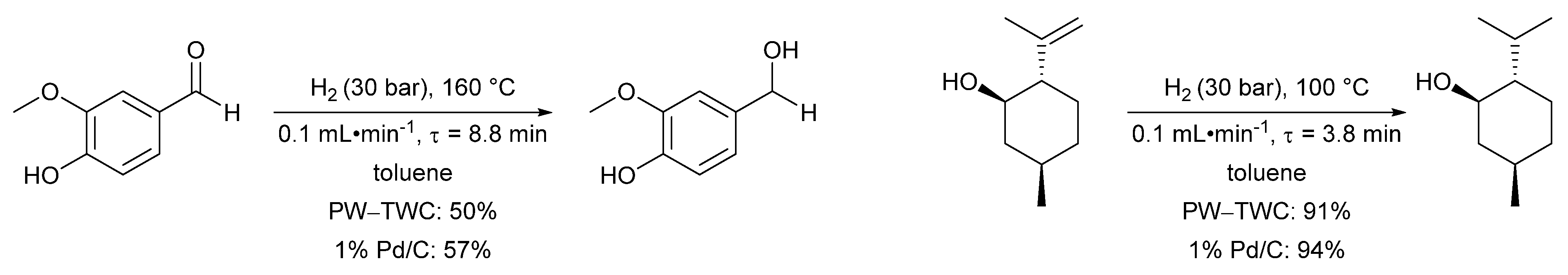
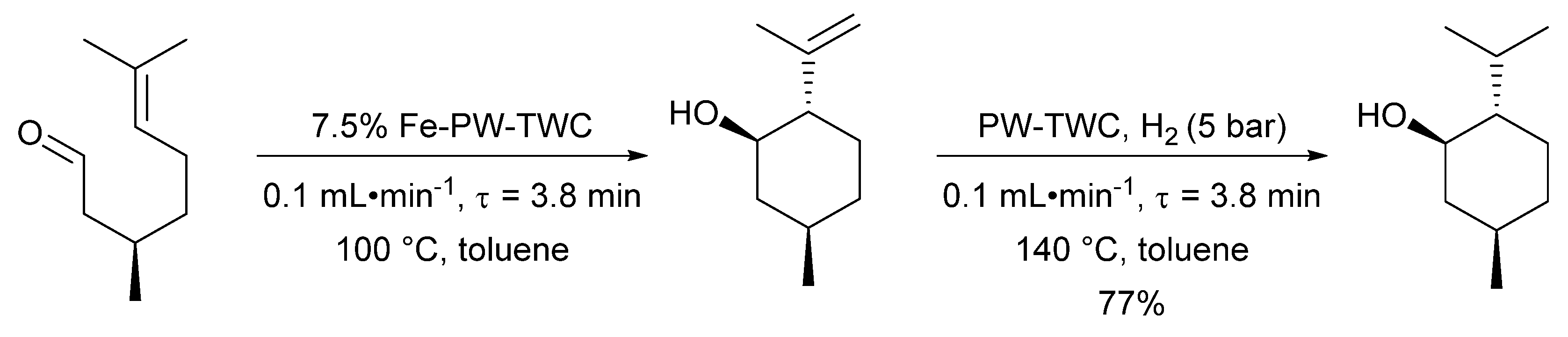
- Cova, C.M.; Zuliani, A.; Manno, R.; Sebastian, V.; Luque, R. Scrap waste automotive converters as efficient catalysts for the continuous-flow hydrogenations of biomass derived chemicals. Green Chem. 2020, 22, 1414–1423. [Google Scholar] [CrossRef]
- Zuliani, A.; Cova, C.M.; Manno, R.; Sebastian, V.; Romero, A.A.; Luque, R. Continuous flow synthesis of menthol: Via tandem cyclisation-hydrogenation of citronellal catalysed by scrap catalytic converters. Green Chem. 2020, 22, 379–387. [Google Scholar] [CrossRef]


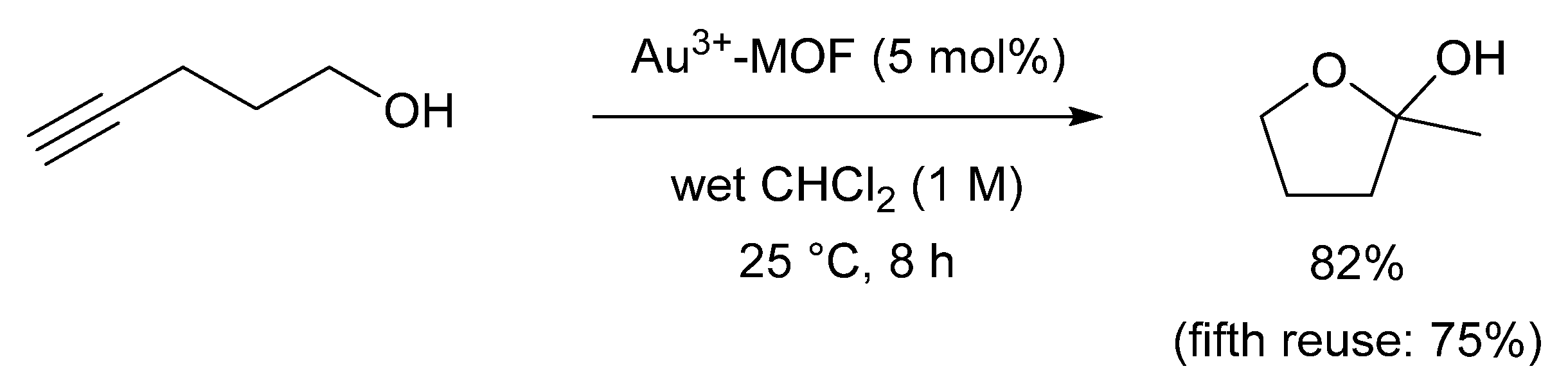


| Lixiviant | Ligand | Oxidant | Gold Complex in Solution | Leaching Conditions |
|---|---|---|---|---|
| Cyanide | CN− | O2 | [Au(CN)2]− | E°: −0.67 V pH > 10, 85 °C |
| Aqua regia | Cl− | HNO3 | [AuCl4]− | E°: 0.96 V pH > 8–11, > 50°C |
| Thiosulfate | S2O32- | O2 or Cu2+ | [Au(S2O3)2]3− | E°: 0.038–0.274 V pH > 8–11, 25 °C |
| Thiourea | S = C(NH2)2 | Fe3+ | [Au{S = C(NH2)2}2]+ | E°: 0.38 V pH 1–2, 25 °C |
| Halide | Cl− | Cl2 | [AuCl4]− | E0: 1.00 V pH < 4, 25 °C a |
| Br− | Br2 | [AuBr4]− | ||
| I− | I2/I3− | [AuI2]− |

| Palladium Source | Yield (%) |
|---|---|
| Pd(OAc)2 | 85 |
| MLCC recovered Pd | 77 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McCarthy, S.; Lee Wei Jie, A.; Braddock, D.C.; Serpe, A.; Wilton-Ely, J.D.E.T. From Waste to Green Applications: The Use of Recovered Gold and Palladium in Catalysis. Molecules 2021, 26, 5217. https://doi.org/10.3390/molecules26175217
McCarthy S, Lee Wei Jie A, Braddock DC, Serpe A, Wilton-Ely JDET. From Waste to Green Applications: The Use of Recovered Gold and Palladium in Catalysis. Molecules. 2021; 26(17):5217. https://doi.org/10.3390/molecules26175217
Chicago/Turabian StyleMcCarthy, Sean, Alvin Lee Wei Jie, D. Christopher Braddock, Angela Serpe, and James D. E. T. Wilton-Ely. 2021. "From Waste to Green Applications: The Use of Recovered Gold and Palladium in Catalysis" Molecules 26, no. 17: 5217. https://doi.org/10.3390/molecules26175217
APA StyleMcCarthy, S., Lee Wei Jie, A., Braddock, D. C., Serpe, A., & Wilton-Ely, J. D. E. T. (2021). From Waste to Green Applications: The Use of Recovered Gold and Palladium in Catalysis. Molecules, 26(17), 5217. https://doi.org/10.3390/molecules26175217