Recent Advances in Micro/Nanomaterial-Based Aptamer Selection Strategies
Abstract
1. Introduction
2. Aptamer Discovery Strategies for Molecule Detection and Monitoring
Basic Process of Aptamer Discovery
3. Current Aptamer Discovery Strategies to Detect Chemicals and Biomolecules
3.1. Bead-Mediated Aptamer Selection
3.2. Carbon Nanomaterial-Mediated Aptamer Selection
3.3. Other Nanoparticle-Mediated Aptamer Selection
4. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, Z.; Mohamed, M.A.; Vinu Mohan, A.M.; Zhu, Z.; Sharma, V.; Mishra, G.K.; Mishra, R.K. Application of electrochemical aptasensors toward clinical diagnostics, food, and environmental monitoring: Review. Sensors 2019, 19, 5435. [Google Scholar] [CrossRef] [PubMed]
- Stanciu, L.A.; Wei, Q.; Barui, A.K.; Mohammad, N. Recent advances in aptamer-based biosensors for global health applications. Annu. Rev. Biomed. Eng. 2021, 23, 433–459. [Google Scholar] [CrossRef] [PubMed]
- Tuerk, C.; Gold, L. Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science 1990, 249, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Ellington, A.D.; Szostak, J.W. In vitro selection of RNA molecules that bind specific ligands. Nature 1990, 346, 818–822. [Google Scholar] [CrossRef]
- Bayat, P.; Nosrati, R.; Alibolandi, M.; Rafatpanah, H.; Abnous, K.; Khedri, M.; Ramezani, M. SELEX methods on the road to protein targeting with nucleic acid aptamers. Biochimie 2018, 154, 132–155. [Google Scholar] [CrossRef]
- Boussebayle, A.; Groher, F.; Suess, B. RNA-based Capture-SELEX for the selection of small molecule-binding aptamers. Methods 2019, 161, 10–15. [Google Scholar] [CrossRef]
- Sefah, K.; Shangguan, D.; Xiong, X.; O’Donoghue, M.B.; Tan, W. Development of DNA aptamers using Cell-SELEX. Nat. Protoc. 2010, 5, 1169–1185. [Google Scholar] [CrossRef]
- Barman, J. Targeting cancer cells using aptamers: Cell-SELEX approach and recent advancements. RSC Adv. 2015, 5, 11724–11732. [Google Scholar] [CrossRef]
- Li, S.; Xu, H.; Ding, H.; Huang, Y.; Cao, X.; Yang, G.; Li, J.; Xie, Z.; Meng, Y.; Li, X.; et al. Identification of an aptamer targeting hnRNP A1 by tissue slide-based SELEX. J. Pathol. 2009, 218, 327–336. [Google Scholar] [CrossRef]
- Tsai, Y.-C.; Lin, C.-S.; Lin, C.-N.; Hsu, F.-F.; Lee, G.-B. Screening aptamers targeting the cell membranes of clinical cancer tissues on an integrated microfluidic system. Sens. Actuators B Chem. 2021, 330, 129334. [Google Scholar] [CrossRef]
- Lin, C.S.; Tsai, Y.C.; Hsu, K.F.; Lee, G.B. Optimization of aptamer selection on an automated microfluidic system with cancer tissues. Lab Chip 2021, 21, 725–734. [Google Scholar] [CrossRef] [PubMed]
- Shigdar, S.; Lv, L.; Wang, L.; Duan, W. Application of aptamers in histopathology. In Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2016; Volume 1380, pp. 191–196. [Google Scholar]
- Li, F.; Wang, Q.; Zhang, H.; Deng, T.; Feng, P.; Hu, B.; Jiang, Y.; Cao, L. Characterization of a DNA aptamer for ovarian cancer clinical tissue recognition and in vivo imaging. Cell. Physiol. Biochem. 2018, 51, 2564–2574. [Google Scholar] [CrossRef]
- Guan, B.; Zhang, X. Aptamers as versatile ligands for biomedical and pharmaceutical applications. Int. J. Nanomed. 2020, 15, 1059–1071. [Google Scholar] [CrossRef]
- Nguyen, V.T.; Kwon, Y.S.; Kim, J.H.; Gu, M.B. Multiple GO-SELEX for efficient screening of flexible aptamers. Chem Commun. 2014, 50, 10513–10516. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Kou, Q.; Wu, P.; Sun, Q.; Wu, J.; Le, T. Selection and application of DNA aptamers against sulfaquinoxaline assisted by graphene oxide-based SELEX. Food Anal. Methods 2020, 14, 250–259. [Google Scholar] [CrossRef]
- Xing, L.; Zhang, Y.; Yang, J. Graphene oxide-assisted non-immobilized SELEX of chiral drug ephedrine aptamers and the analytical binding mechanism. Biochem. Biophys. Res. Commun. 2019, 514, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Mosing, R.K.; Mendonsa, S.D.; Bowser, M.T. Capillary electrophoresis-SELEX selection of aptamers with affinity for HIV-1 reverse transcriptase. Anal. Chem. 2005, 77, 6107–6112. [Google Scholar] [CrossRef] [PubMed]
- Hamedani, N.S.; Müller, J.C. Capillary electrophoresis for the selection of DNA aptamers recognizing activated protein C. In Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2016; Volume 1380, pp. 61–75. [Google Scholar]
- Zhu, C.; Yang, G.; Ghulam, M.; Li, L.; Qu, F. Evolution of multi-functional capillary electrophoresis for high-efficiency selection of aptamers. Biotechnol. Adv. 2019, 37, 8. [Google Scholar] [CrossRef] [PubMed]
- Guo, K.T.; Ziemer, G.; Paul, A.; Wendel, H.P. Cell-SELEX, novel perspectives of aptamer-based therapeutics. Int. J. Mol. Sci. 2008, 9, 668–678. [Google Scholar] [CrossRef]
- Kaur, H. Recent developments in cell-SELEX technology for aptamer selection. Biochimica Biophysica Acta Gen. Subj. 2018, 1862, 2323–2329. [Google Scholar] [CrossRef]
- Kim, Y.; Liu, C.; Tan, W. Aptamers generated by Cell SELEX for biomarker discovery. Biomark. Med. 2009, 3, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Mayer, G.; Ahmed, M.S.; Dolf, A.; Endl, E.; Knolle, P.A.; Famulok, M. Fluorescence activated cell sorting for aptamer SELEX with cell mixtures. Nat. Protoc. 2010, 5, 1993–2004. [Google Scholar] [CrossRef] [PubMed]
- McDonald, J.; Howells, L.M.; Brown, K.; Thompson, J.P.; Lambert, D.G. Use of preparative fluorescence-activated cell sorting (FACS) to profile opioid receptor and peptide mRNA expression on human granulocytes, lymphocytes, and monocytes. Br. J. Anaesth. 2010, 105, 707–708. [Google Scholar]
- Nishimoto, K.P.; Newkirk, D.; Hou, S.; Fruehauf, J.; Nelson, E.L. Fluorescence activated cell sorting (FAGS) using RNAlater to minimize RNA degradation and perturbation of mRNA expression from cells involved in initial host microbe interactions. J. Microbiol. Methods 2007, 70, 205–208. [Google Scholar] [CrossRef] [PubMed]
- Golden, M.C.; Collins, B.D.; Willis, M.C.; Koch, T.H. Diagnostic potential of PhotoSELEX-evolved ssDNA aptamers. J. Biotechnol. 2000, 81, 167–178. [Google Scholar] [CrossRef]
- Dunn, M.; Jimenez, R.; Chaput, J. Analysis of aptamer discovery and technology. Nat. Rev. Chem. 2017, 1, 0076. [Google Scholar] [CrossRef]
- Cole, K.H.; Lupták, A. High-throughput methods in aptamer discovery and analysis. Methods Enzymol. 2019, 621, 329–346. [Google Scholar]
- Ni, S.; Zhuo, Z.; Pan, Y.; Yu, Y.; Li, F.; Liu, J.; Wang, L.; Wu, X.; Li, D.; Wan, Y.; et al. Recent progress in aptamer discoveries and modifications for therapeutic applications. ACS Appl. Mater. Interfaces 2021, 13, 9500–9519. [Google Scholar] [CrossRef]
- Lyu, C.; Khan, I.M.; Wang, Z. Capture-SELEX for aptamer selection: A short review. Talanta 2021, 229, 122274. [Google Scholar] [CrossRef] [PubMed]
- Morihiro, K.; Kasahara, Y.; Obika, S. Biological applications of xeno nucleic acids. Mol. Biosyst. 2017, 13, 235–245. [Google Scholar] [CrossRef]
- Taylor, A.I.; Holliger, P. Selecting fully-modified XNA aptamers using synthetic genetics. Curr. Protoc. Chem. Biol. 2018, 10, e44. [Google Scholar] [CrossRef]
- Gatto, B.; Palumbo, M.; Sissi, C. Nucleic acid aptamers based on the G-quadruplex structure: Therapeutic and diagnostic potential. Curr. Med. Chem. 2009, 16, 1248–1265. [Google Scholar] [CrossRef]
- Boucard, D.; Toulmé, J.J.; Di Primo, C. Bimodal loop-loop interactions increase the affinity of RNA aptamers for HIV-1 RNA structures. Biochemistry 2006, 45, 1518–1524. [Google Scholar] [CrossRef]
- Chovelon, B.; Durand, G.; Dausse, E.; Toulmé, J.J.; Faure, P.; Peyrin, E.; Ravelet, C. ELAKCA: Enzyme-linked aptamer kissing complex assay as a small molecule sensing platform. Anal. Chem. 2016, 88, 2570–2575. [Google Scholar] [CrossRef]
- Goux, E.; Dausse, E.; Guieu, V.; Azéma, L.; Durand, G.; Henry, M.; Choisnard, L.; Toulmé, J.J.; Ravelet, C.; Peyrin, E. A colorimetric nanosensor based on a selective target-responsive aptamer kissing complex. Nanoscale 2017, 9, 4048–4052. [Google Scholar] [CrossRef] [PubMed]
- Tucker, W.O.; Shum, K.T.; Tanner, J.A. G-quadruplex DNA aptamers and their ligands: Structure, function and application. Curr. Pharm. Des. 2012, 18, 2014–2026. [Google Scholar] [CrossRef]
- Li, L.; Xu, S.; Peng, X.; Ji, Y.; Yan, H.; Cui, C.; Li, X.; Pan, X.; Yang, L.; Qiu, L.; et al. Engineering G-quadruplex aptamer to modulate its binding specificity. Natl. Sci. Rev. 2020, 8, nwaa202. [Google Scholar] [CrossRef] [PubMed]
- Bing, T.; Zheng, W.; Zhang, X.; Shen, L.; Liu, X.; Wang, F.; Cui, J.; Cao, Z.; Shangguan, D. Triplex-Quadruplex structural scaffold: A new binding structure of aptamer. Sci. Rep. 2017, 7, 15467. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.Y.; Kwok, C.K. Specific binding of a d-RNA G-quadruplex structure with an l-RNA aptamer. Angew. Chem. 2020, 59, 5293–5297. [Google Scholar] [CrossRef] [PubMed]
- Roxo, C.; Kotkowiak, W.; Pasternak, A. G-Quadruplex-forming aptamers-characteristics, applications, and perspectives. Molecules 2019, 24, 3781. [Google Scholar] [CrossRef] [PubMed]
- Strehlitz, B.; Stoltenburg, R.; Nikolaus, N.; Strehlitz, B. Capture-SELEX: Selection of DNA aptamers for aminoglycoside antibiotics. J. Anal. Methods Chem. 2012, 2012, 415697. [Google Scholar]
- Li, W.; Luo, Y.; Gao, T.; Yang, L.; Wang, J.; Pei, R. In vitro selection of DNA aptamers for a small-molecule porphyrin by gold nanoparticle-based SELEX. J. Mol. Evol. 2019, 87, 231–239. [Google Scholar] [CrossRef]
- Wu, Y.; Zhan, S.; Wang, L.; Zhou, P. Selection of a DNA aptamer for cadmium detection based on cationic polymer mediated aggregation of gold nanoparticles. Analyst 2014, 139, 1550–1561. [Google Scholar] [CrossRef]
- Zhao, X.; Johnson, J.K. Simulation of adsorption of DNA on carbon nanotubes. J. Am. Chem. Soc. 2007, 129, 10438–10445. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.R.; Johnson, A.T.; Klein, M.L. Probing the structure of DNA-carbon nanotube hybrids with molecular dynamics. Nano Lett. 2008, 8, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Husale, B.S.; Sahoo, S.; Radenovic, A.; Traversi, F.; Annibale, P.; Kis, A. ssDNA binding reveals the atomic structure of graphene. Langmuir 2010, 26, 18078–18082. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Lei, X.; Tu, Y.; Tan, Z.J.; Song, B.; Fang, H. Dynamic cooperation of hydrogen binding and π stacking in ssDNA adsorption on graphene oxide. Chemistry 2017, 23, 13100–13104. [Google Scholar] [CrossRef] [PubMed]
- Park, J.W.; Tatavarty, R.; Kim, D.W.; Jung, H.T.; Gu, M.B. Immobilization-Free screening of aptamers assisted by graphene oxide. Chem. Commun. 2012, 48, 2071–2073. [Google Scholar] [CrossRef]
- Gu, H.; Duan, N.; Xia, Y.; Hun, X.; Wang, H.; Wang, Z. Magnetic separation-based multiple SELEX for effectively selecting aptamers against saxitoxin, domoic acid and tetrodotoxin. J. Agric. Food Chem. 2018, 66, 9801–9809. [Google Scholar] [CrossRef]
- Jo, M.; Ahn, J.Y.; Lee, J.; Lee, S.; Hong, S.W.; Yoo, J.W.; Kang, J.; Dua, P.; Lee, D.K.; Hong, S.; et al. Development of single-stranded DNA aptamers for specific Bisphenol A detection. Oligonucleotides 2011, 21, 85–91. [Google Scholar] [CrossRef]
- Niazi, J.H.; Lee, S.J.; Kim, Y.S.; Gu, M.B. ssDNA aptamers that selectively bind oxytetracycline. Bioorg. Med. Chem. 2008, 16, 1254–1261. [Google Scholar] [CrossRef]
- Kiani, Z.; Shafiei, M.; Rahimi-Moghaddam, P.; Karkhane, A.A.; Ebrahimi, S.A. In vitro selection and characterization of deoxyribonucleic acid aptamers for digoxin. Anal. Chim. Acta 2012, 748, 67–72. [Google Scholar] [CrossRef]
- Liu, Y.-G.; Chen, Y. High-Efficiency thermal asymmetric interlaced PCR for amplification of unknown flanking sequences. Biotechniques 2007, 43, 5. [Google Scholar] [CrossRef]
- Higuchi, R.G.; Ochman, H. Production of single-stranded DNA templates by exonuclease digestion following the polymerase chain reaction. Nucleic Acids Res. 1989, 17, 5865. [Google Scholar] [CrossRef]
- Espelund, M.; Stacy, R.A.; Jakobsen, K.S. A simple method for generating single-stranded DNA probes labeled to high activities. Nucleic Acids Res. 1990, 18, 6157–6158. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yue, H.; Chen, J.; Chem, X.; Wang, X.; Zhang, Y.; Zhou, N. Systematic screening and optimization of single-stranded DNA aptamer specific for N-acetylneuraminic acid: A comparative study. Sens. Actuators B Chem. 2021, 344, 130270. [Google Scholar] [CrossRef]
- Ando, T.; Yamamoto, M.; Yokoyama, T.; Horiuchi, D.; Kawakami, T. In vitro selection generates RNA aptamer that antagonizes PCSK9-LDLR interaction and recovers cellular LDL uptake. J. Biosci. Bioeng. 2021, 131, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.; Zhang, H.; Zhou, J.; Ma, Q.; Yan, W.; Zhao, L.; Wu, S.; Chen, H. Aptamer-Targeting of Aleutian mink disease virus (AMDV) can be an effective strategy to inhibit virus replication. Sci. Rep. 2021, 11, 4649. [Google Scholar] [CrossRef]
- Kohlberger, M.; Wildner, S.; Regl, C.; Huber, C.G.; Gadermaier, G. Rituximab-specific DNA aptamers are able to selectively recognize heat-treated antibodies. PLoS ONE 2020, 15, e0241560. [Google Scholar] [CrossRef]
- Qiao, N.; Li, J.; Wu, X.; Diao, D.; Zhao, J.; Li, J.; Ren, X.; Ding, X.; Shangguan, D.; Lou, X. Speeding up in vitro discovery of structure-switching aptamers via magnetic cross-linking precipitation. Anal. Chem. 2019, 91, 13383–13389. [Google Scholar] [CrossRef]
- Wei, H.; Cai, R.; Yue, H.; Tian, Y.; Zhou, N. Screening and application of a truncated aptamer for high-sensitive fluorescent detection of metronidazole. Anal. Chim. Acta 2020, 1128, 203–210. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Q.; Luo, Y.; Gao, T.; Zhao, Y.; Pei, R. In vitro selection of ssDNA aptamers that can specifically recognize and differentiate riboflavin and its derivative FAD. Talanta 2019, 204, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Yu, H.; Alkhamis, O.; Liu, Y.; Canoura, J.; Fu, F.; Xiao, Y. In vitro isolation of class-specific oligonucleotide-based small-molecule receptors. Nucleic Acids Res. 2019, 47, e71. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, T.; Rathore, A.S.; Jha, S.K. The selection of highly specific and selective aptamers using modified SELEX and their use in process analytical techniques for Lucentis bioproduction. RSC Adv. 2020, 10, 28906. [Google Scholar] [CrossRef]
- Yu, H.; Luo, Y.; Alkhamis, O.; Canoura, J.; Yu, B.; Xiao, Y. Isolation of natural DNA aptamers for challenging small-molecule targets, cannabinoids. Anal. Chem. 2021, 93, 3172–3180. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Yang, W.; Alkhamis, O.; Canoura, J.; Yang, K.A.; Xiao, Y. In vitro isolation of small-molecule-binding aptamers with intrinsic dye-displacement functionality. Nucleic Acids Res. 2018, 46, e43. [Google Scholar] [CrossRef]
- Yang, L.; Ding, P.; Luo, Y.; Wang, J.; Lv, H.; Li, W.; Cao, Y.; Pei, R. Exploration of catalytic nucleic acids on porphyrin metalation and peroxidase activity by in vitro selection of aptamers for N-methyl mesoporphyrin IX. ACS Comb. Sci. 2019, 21, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Gao, T.; Li, W.; Luo, Y.; Ullah, S.; Fang, X.; Cao, Y.; Pei, R. Ni-Nitrilotriacetic acid affinity SELEX method for selection of DNA aptamers specific to the N-cadherin protein. ACS Comb. Sci. 2020, 22, 867–872. [Google Scholar] [CrossRef]
- Song, S.; Wang, X.; Xu, K.; Li, Q.; Ning, L.; Yang, X. Selection of highly specific aptamers to Vibrio parahaemolyticus using cell-SELEX powered by functionalized graphene oxide and rolling circle amplification. Anal. Chim. Acta 2019, 1052, 153–162. [Google Scholar] [CrossRef]
- Gu, H.; Duan, N.; Wu, S.; Hao, L.; Xia, Y.; Ma, X.; Wang, Z. Graphene oxide-assisted non-immobilized SELEX of okdaic acid aptamer and the analytical application of aptasensor. Sci. Rep. 2016, 6, 21665. [Google Scholar] [CrossRef]
- Islam, M.M.; Ghielmetti, V.M.; Allen, P.B. Graphene oxide assisted light-up aptamer selection against Thioflavin T for label-free detection of microRNA. Sci. Rep. 2021, 11, 4291. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.S.; Kim, E.J.; Park, T.K.; Bae, D.W.; Cha, S.S.; Kim, T.W.; Kim, Y.P. Gold nanoparticle-assisted SELEX as a visual monitoring platform for the development of small molecule-binding DNA aptasensors. Biosens. Bioelectron. 2021, 191, 113468. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, B.; Kalyani, N.; Anand, A.; Khan, E.; Das, S.; Bansal, V.; Kumar, A.; Sharma, T.K. GOLD SELEX: A novel SELEX approach for the development of high-affinity aptamers against small molecules without residual activity. Microchim. Acta 2020, 187, 618. [Google Scholar] [CrossRef]
- Ye, H.; Duan, N.; Wu, S.; Tan, G.; Gu, H.; Li, J.; Wang, H.; Wang, Z. Orientation selection of broad-spectrum aptamers against lipopolysaccharides based on capture-SELEX by using magnetic nanoparticles. Microchim. Acta 2017, 184, 4235–4242. [Google Scholar] [CrossRef]
- Ma, Y.; Li, X.; Li, W.; Liu, Z. Glycan-Imprinted magnetic nanoparticle-based SELEX for efficient screening of glycoprotein-binding aptamers. ACS Appl. Mater. Interfaces 2018, 10, 40918–40926. [Google Scholar] [CrossRef]
- Ma, Y.; Li, W.; Xing, R.; Li, P.; Liu, Z. Epitope-imprinted magnetic nanoparticles as a general platform for efficient in vitro evolution of protein-binding aptamers. ACS Sens. 2020, 5, 2537–2544. [Google Scholar] [CrossRef]
- Zhang, H.; Li, X.; Huang, A.; Yan, Z.; Chen, Y.; Bie, Z. PEI-Assisted boronate affinity magnetic nanoparticle-based SELEX for efficient in vitro evolution of saponin-binding aptamers. RSC Adv. 2021, 11, 8775–8781. [Google Scholar] [CrossRef]
- Jia, W.; Li, H.; Wilkop, T.; Liu, X.; Yu, X.; Cheng, Q.; Xu, D.; Chen, H.Y. Silver decahedral nanoparticles empowered SPR imaging-SELEX for high throughput screening of aptamers with real-time assessment. Biosens. Bioelectron. 2018, 109, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.L.; Wan, Y.T.; Tang, M.; Pang, D.W.; Zhang, Z.L. Multifunctional screening platform for the highly efficient discovery of aptamers with high affinity and specificity. Anal. Chem. 2017, 89, 6535–6542. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.L.; Xiang, M.Q.; Tang, M.; Pang, D.W.; Zhang, Z.L. Ebola virus aptamers: From highly efficient selection to application on magnetism-controlled chips. Anal. Chem. 2019, 91, 3367–3373. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Yang, L.; Liang, X.; Dong, T.; Liu, H. Bare magnetic nanoparticles as fluorescence quenchers for detection of thrombin. Analyst 2015, 140, 4114–4120. [Google Scholar] [CrossRef]
- Song, C.; Wang, G.Y.; Kong, D.M. A facile fluorescence method for versatile biomolecular detection based on pristine α-Fe2O3 nanoparticle-induced fluorescence quenching. Biosens. Bioelectron. 2015, 68, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.A.; Chávez, J.L.; Chushak, Y.; Chapleau, R.R.; Hagen, J.; Kelley-Loughnane, N. Tunable stringency aptamer selection and gold nanoparticle assay for detection of cortisol. Anal. Bioanal. Chem. 2014, 406, 4637–4647. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Wang, J.; Zhang, M.; Shi, G. Selection of a structure-switching aptamer for the specific methotrexate detection. ACS Sens. 2021. [Google Scholar] [CrossRef]
- Tan, Y.; Guo, Q.; Xie, Q.; Wang, K.; Yuan, B.; Zhou, Y.; Liu, J.; Huang, J.; He, X.; Yang, X.; et al. Single-Walled carbon nanotubes (SWCNTs)-assisted cell-systematic evolution of ligands by exponential enrichment (cell-SELEX) for improving screening efficiency. Anal. Chem. 2014, 86, 9466–9472. [Google Scholar] [CrossRef]
- Zhu, Y.; Cai, Y.; Xu, L.; Zheng, L.; Wang, L.; Qi, B.; Xu, C. Building an aptamer/graphene oxide FRET biosensor for one-step detection of bisphenol A. ACS Appl. Mater. Interfaces 2015, 7, 7492–7496. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.; Qian, Y.; Wu, P.; Zhang, H.; Cai, C. Fluorescence quenching of graphene oxide combined with the site-specific cleavage of restriction endonuclease for deoxyribonucleic acid demethylase activity assay. Anal. Chim. Acta 2015, 869, 74–80. [Google Scholar] [CrossRef]
- Wei, W.; Gao, C.; Xiong, Y.; Zhang, Y.; Liu, S.; Pu, Y. A fluorescence method for detection of DNA and DNA methylation based on graphene oxide and restriction endonuclease HpaII. Talanta 2015, 131, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Baig, N.; Kammakakam, I.; Falath, W. Nanomaterials, a review of synthesis methods, properties, recent progress, and challenges. Mater. Adv. 2021, 2, 1821–1871. [Google Scholar] [CrossRef]
- Yeh, Y.C.; Creran, B.; Rotello, V.M. Gold nanoparticles, preparation, properties, and applications in bionanotechnology. Nanoscale 2012, 4, 1871–1880. [Google Scholar] [CrossRef]
- Liu, J. Adsorption of DNA onto gold nanoparticles and graphene oxide, surface science and applications. Phys. Chem. Chem. Phys. 2012, 14, 10485–10496. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; He, L.; Wang, J.; Fang, X.; Zhang, L. Developing a combined strategy for monitoring the progress of aptamer selection. Analyst 2017, 142, 3136–3139. [Google Scholar] [CrossRef] [PubMed]
- Kolm, C.; Cervenka, I.; Aschl, U.J.; Baumann, N.; Jakwerth, S.; Krska, R.; Mach, R.L.; Sommer, R.; DeRosa, M.C.; Kirschner, A.; et al. DNA aptamers against bacterial cells can be efficiently selected by a SELEX process using state-of-the art qPCR and ultra-deep sequencing. Sci. Rep. 2020, 10, 20917. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, S.A. Molecular imprinting polymers and their composites, a promising material for diverse applications. Biomater. Sci. 2017, 5, 388. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Chen, Y.; Liu, Z. Boronate affinity materials for separation and molecular recognition, structure, properties and applications. Chem. Soc. Rev. 2015, 44, 8097–8123. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; He, H. Synthesis and applications of boronate affinity materials, from class selectivity to biomimetic specificity. Acc. Chem. Res. 2017, 50, 2185–2193. [Google Scholar] [CrossRef] [PubMed]
- Amendola, V.; Bakr, O.M.; Stellacci, F. A study of the Surface plasmon resonance of silver nanoparticles by the discrete dipole approximation method, effect of shape, size, structure, and assembly. Plasmonics 2010, 5, 85–97. [Google Scholar] [CrossRef]
- Rebe Raz, S.; Leontaridou, M.; Bremer, M.G.; Peters, R.; Weigel, S. Development of surface plasmon resonance-based sensor for detection of silver nanoparticles in food and the environment. Anal. Bioanal. Chem. 2012, 403, 2843–2850. [Google Scholar] [CrossRef]
- Emami, N.; Ferdousi, R. AptaNet as a deep learning approach for aptamer-protein interaction prediction. Sci. Rep. 2021, 11, 6074. [Google Scholar] [CrossRef]
- Li, J.; Ma, X.; Li, X.; Gu, J. PPAI: A web server for predicting protein-aptamer interactions. BMC Bioinform. 2020, 21, 236. [Google Scholar] [CrossRef]
- Torkamanian-Afshar, M.; Lanjanian, H.; Nematzadeh, S.; Tabarzad, M.; Najafi, A.; Kiani, F.; Masoudi-Nejad, A. RPINBASE: An online toolbox to extract features for predicting RNA-protein interactions. Genomics 2020, 112, 2623–2632. [Google Scholar] [CrossRef]
- Biesiada, M.; Pachulska-Wieczorek, K.; Adamiak, R.W.; Purzycka, K.J. RNAComposer and RNA 3D structure prediction for nanotechnology. Methods 2016, 103, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, J.; Huang, Y.; Xiao, Y. 3dRNA v2.0: An Updated web server for RNA 3D structure prediction. Int. J. Mol. Sci. 2019, 20, 4116. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Xu, X.; Chen, S.J. Predicting RNA Structure with Vfold. In Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2017; Volume 1654, pp. 3–15. [Google Scholar]
- Boniecki, M.J.; Lach, G.; Dawson, W.K.; Tomala, K.; Lukasz, P.; Soltysinski, T.; Rother, K.M.; Bujnicki, J.M. SimRNA: A coarse-grained method for RNA folding simulations and 3D structure prediction. Nucleic Acids Res. 2016, 44, e63. [Google Scholar] [CrossRef]
- Yamasaki, S.; Amemiya, T.; Yabuki, Y.; Horimoto, K.; Fukui, K. ToGo-WF: Prediction of RNA tertiary structures and RNA-RNA/protein interactions using the KNIME workflow. J. Comput. Aided Mol. Des. 2019, 33, 497–507. [Google Scholar] [CrossRef]
- Gruber, A.R.; Bernhart, S.H.; Lorenz, R. The ViennaRNA web services. In Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2015; Volume 1269, pp. 307–326. [Google Scholar]
- Zuker, M. Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res. 2003, 31, 3406–3415. [Google Scholar] [CrossRef]
- Andronescu, M.; Aguirre-Hernández, R.; Condon, A.; Hoos, H.H. RNAsoft: A suite of RNA secondary structure prediction and design software tools. Nucleic Acids Res. 2003, 31, 3416–3422. [Google Scholar] [CrossRef]
- Bellaousov, S.; Reuter, J.S.; Seetin, M.G.; Mathews, D.H. RNAstructure: Web servers for RNA secondary structure prediction and analysis. Nucleic Acids Res. 2013, 41, W471–W474. [Google Scholar] [CrossRef] [PubMed]
- Zadeh, J.N.; Steenberg, C.D.; Bois, J.S.; Wolfe, B.R.; Pierce, M.B.; Khan, A.R.; Dirks, R.M.; Pierce, N.A. NUPACK: Analysis and design of nucleic acid systems. J. Comput. Chem. 2011, 32, 170–173. [Google Scholar] [CrossRef] [PubMed]
- Pierce, B.G.; Wiehe, K.; Hwang, H.; Kim, B.H.; Vreven, T.; Weng, Z. ZDOCK server: Interactive docking prediction of protein-protein complexes and symmetric multimers. Bioinformatics 2014, 30, 1771–1773. [Google Scholar] [CrossRef]
- Huang, S.Y.; Zou, X. MDockPP: A hierarchical approach for protein-protein docking and its application to CAPRI rounds 15–19. Proteins 2010, 78, 3096–3103. [Google Scholar] [CrossRef] [PubMed]
- Forli, S.; Huey, R.; Pique, M.E.; Sanner, M.F.; Goodsell, D.S.; Olson, A.J. Computational protein-ligand docking and virtual drug screening with the AutoDock suite. Nat. Protoc. 2016, 11, 905–919. [Google Scholar] [CrossRef] [PubMed]
- Seeliger, D.; de Groot, B.L. Ligand docking and binding site analysis with PyMOL and Autodock/Vina. J. Comput. Aided Mol. Des. 2010, 24, 417–422. [Google Scholar] [CrossRef]
- Hung, L.Y.; Wang, C.H.; Hsu, K.F.; Chou, C.Y.; Lee, G.B. An on-chip Cell-SELEX process for automatic selection of high-affinity aptamers specific to different histologically classified ovarian cancer cells. Lab Chip 2014, 14, 4017–4028. [Google Scholar] [CrossRef] [PubMed]
- Tapia-Alveal, C.; Olsen, T.R.; Worgall, T.S. Personalized immunoglobulin aptamers for detection of multiple myeloma minimal residual disease in serum. Commun. Biol. 2020, 3, 781. [Google Scholar] [CrossRef] [PubMed]
- Gopinathan, P.; Hung, L.Y.; Wang, C.H.; Chiang, N.J.; Wang, Y.C.; Shan, Y.S.; Lee, G.B. Automated selection of aptamers against cholangiocarcinoma cells on an integrated microfluidic platform. Biomicrofluidics 2017, 11, 044101. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.X.; Lai, P.X.; Mao, J.Y.; Chu, H.W.; Unnikrishnan, B.; Anand, A.; Huang, C.C. Supramolecular aptamers on graphene oxide for efficient inhibition of thrombin activity. Front. Chem. 2019, 7, 280. [Google Scholar] [CrossRef]
- Ranganathan, S.V.; Halvorsen, K.; Myers, C.A.; Robertson, N.M.; Yigit, M.V.; Chen, A.A. Complex thermodynamic behavior of single-stranded nucleic acid adsorption to graphene surfaces. Langmuir 2016, 32, 6028–6034. [Google Scholar] [CrossRef]
| Company | Website |
|---|---|
| Optimer™ | https://aptamergroup.com/ (accessed on 24 August 2021) |
| Creative Biolabs | https://www.creative-biolabs.com/ (accessed on 24 August 2021) |
| Oak Biosciences | https://www.oakbiosciences.com/ (accessed on 24 August 2021) |
| TriLink Biotechnologies | https://www.trilinkbiotech.com/ (accessed on 24 August 2021) |
| Novaptech | https://novaptech.com/ (accessed on 24 August 2021) |
| BasePair Biotechnologies | https://www.basepairbio.com/ (accessed on 24 August 2021) |
| Aptagen | https://www.aptagen.com/ (accessed on 24 August 2021) |
| Aptamer sciences | http://aptsci.com/en/about-us/overview/ (accessed on 24 August 2021) |
| Novaptech | https://novaptech.com/ (accessed on 24 August 2021) |
| AptaTargets | http://aptatargets.com/ (accessed on 24 August 2021) |
| RIBOMIC | https://www.ribomic.com/eng/technology.php/ (accessed on 24 August 2021) |
| NeoVentures Biotechnology | https://neoaptamers.com/ (accessed on 24 August 2021) |
| TAGCyx Biotechnologies | https://tagcux/com/en/ (accessed on 24 August 2021) |
| Raptamer | https://raptamer.com/ (accessed on 24 August 2021) |
| Classification | Material | Target Molecule(s) | Kd Value | Kd Value-Measuring Method | Characterization Method | Features | Reference |
|---|---|---|---|---|---|---|---|
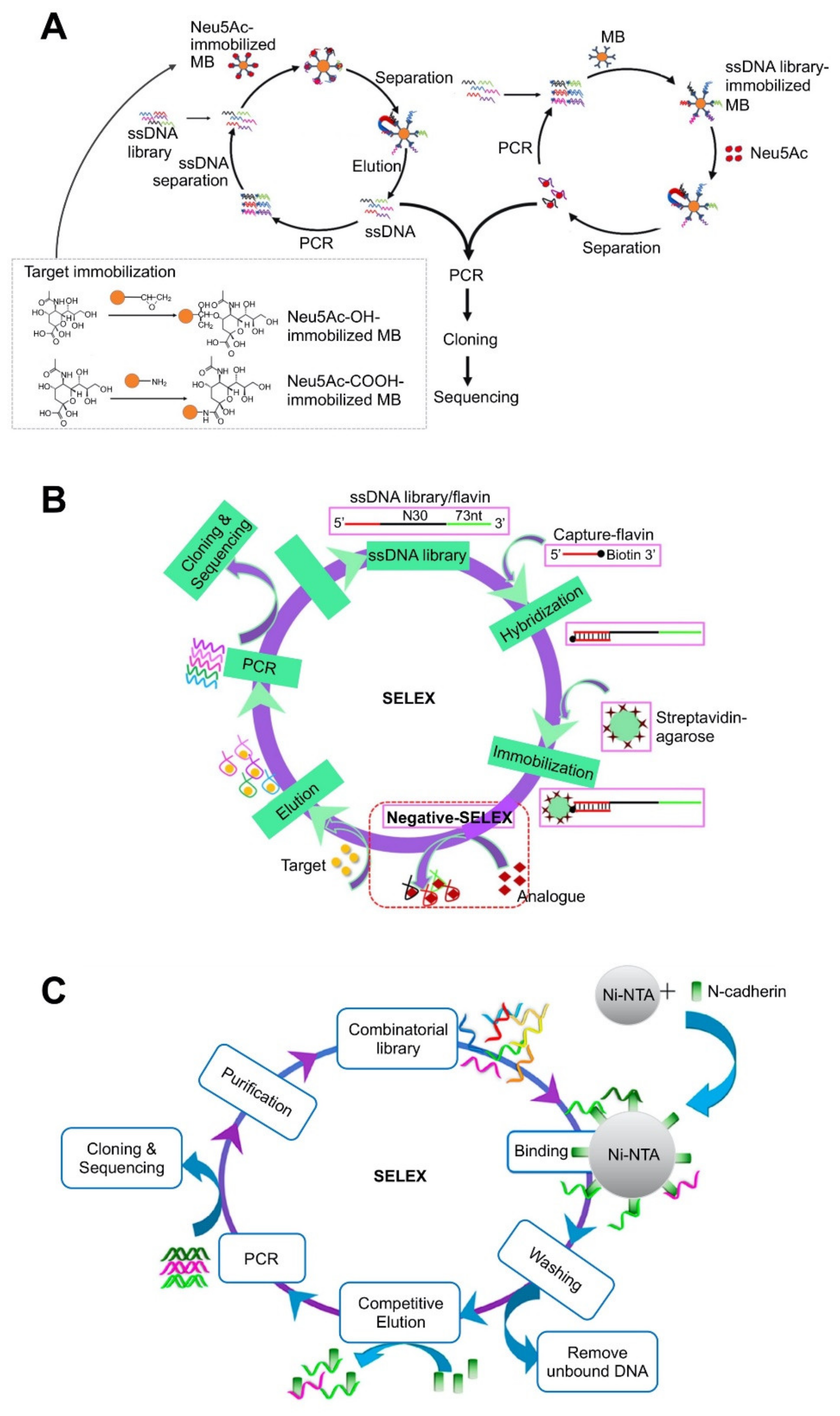
| Bead-based platforms | Magnetic bead (MB) | N-acetylneuraminic acid | 55.71 ± 12.29 nM | Fluorescence assay by labeling aptamer with FAM at the 5′-end | Prediction of two-dimensional (2D) structure using Mfold. Three-dimensional (3D) structure analysis by RNAComposer. | Use of both immobilization strategies using target-immobilized MBs and aptamer-immobilized MBs. | [58]; Figure 1A |
| MB | PCSK9 | 70 ± 10 nM | Biolayer interferometry assay | Target-binding analysis by qPCR and chemiluminescence. | Target-immobilization strategy. RNA aptamer. | [59] | |
| MB | Aleutian mink disease virus (AMDV) | 247 ± 62.5 nM | Enzyme-linked oligonucleotide assay (ELONA) | Prediction of the 3D structures using 3dRNA-v2.0. | Use of recombinant AMDV VP2 protein as a target. Monitoring the SELEX process by quantitative PCR (qPCR). | [60] | |
| MB | Rituximab | 8.8 nM | ELISA using biotinylated aptamer | Prediction of 2D structure using Mfold. Structural analysis by circular dichroism (CD). | Target-immobilization strategy. Immobilization of rituximab on the protein A-coated MB for Fab orientation and easy detachment of target-bound aptamer from MB by heating at every round. | [61] | |
| MB | Human alpha-thrombin (Tb), human serum albumin (hSA) | 1.2–20 nM (for hSA), 33 nM (for Tb) | MB-based qPCR method, ELONA, fluorescence evanescent wave biosensor. | Specificity using Cy3/biotinylated probe and MB-based fluorescence measurements. Prediction of 2D structure using Mfold. | Use of cross-linking reaction by binding between amino groups of target proteins and activated carboxylic acid groups of MBs. Short screening time (four cycles). Non-immobilization strategy. | [62] | |
| MB | Metronidazole | 77.22 ± 11.27 nM | Fluorescence assay by labeling aptamer with FAM at the 5′-end. | Homology analysis by DNAMAN and Snap Gene. Prediction of secondary structures by RNA Structure software and Mfold Web Server. Simulation of tertiary structure by PyMoL 1.7.6 software. | Library-immobilized SELEX. | [63] | |
| Agarose bead | Flavin adenine dinucleotide (FAD) | 0.61 ± 0.04 (for riboflavin), 0.44 ± 0.02 for FAD) | Monitoring the fluorescence of flavin after binding with aptamer. | Prediction of 2D structure using Mfold. | Library-immobilized SELEX. | [64]; Figure 1B | |
| Agarose bead | Butylone, ethylone, alpha-PVP | 6.9 μM (for ethylone), 9.5 μM (butylone), 21 μM (PVP) | Isothermal titration calorimetry (ITC) | Specificity analysis by gel-elution assay. | Selection of multiple molecule–targeting aptamer. | [65] | |
| Agarose bead | Lucentis | 25.72 ± 4.2 nM | Thermofluorimetric and non-faradaic impedance spectroscopy (NFIS) analysis. | Specificity analysis by NFIS analysis. | Use of stringent condition at each cycle for selecting aptamers with high affinity. | [66] | |
| Agarose bead | (−)-trans-Δ9-tetrahydrocannabinol (THC), UR-144 and XLR-11, two widely abused synthetic cannabinoids | 61 ± 25 nM (for THC), 310 ± 70 nM (for XLR-11), 127 ± 32 nM (for UR-144) | ITC | Septicity analysis by strand-displacement fluorescence assay. | Library-immobilized strategy. A binding buffer containing organic solvent (i.e., 2.6% methanol or 5% DMSO) was used to increase their solubility. | [67] | |
| Agarose bead | 3,4- methylenedioxypyrovalerone (MDPV) | 6.1 ± 0.2 μM | ITC | Cy7-displacement assay | Library-immobilized strategy, immobilization of biotinylated cDNA:library duplex on bead. | [68] | |
| Agarose bead | N-Methyl Mesoporphyrin IX | Prediction of 2D structure using Mfold. Structural analysis by CD and PAGE. | Immobilization of biotinylated DNA:ssDNA duplex on streptavidin-coated agarose bead. Selection pressure: decrease of the concentrations of ssDNA and the target. | [69] | |||
| Ni | N-cadherin derived from the human protein (Met1-Ala724) | 93 nM | Fluorescence assay by labeling aptamer with FAM. | Immobilization of His-tagged protein on Ni-NTA column. | [70]; Figure 1C | ||
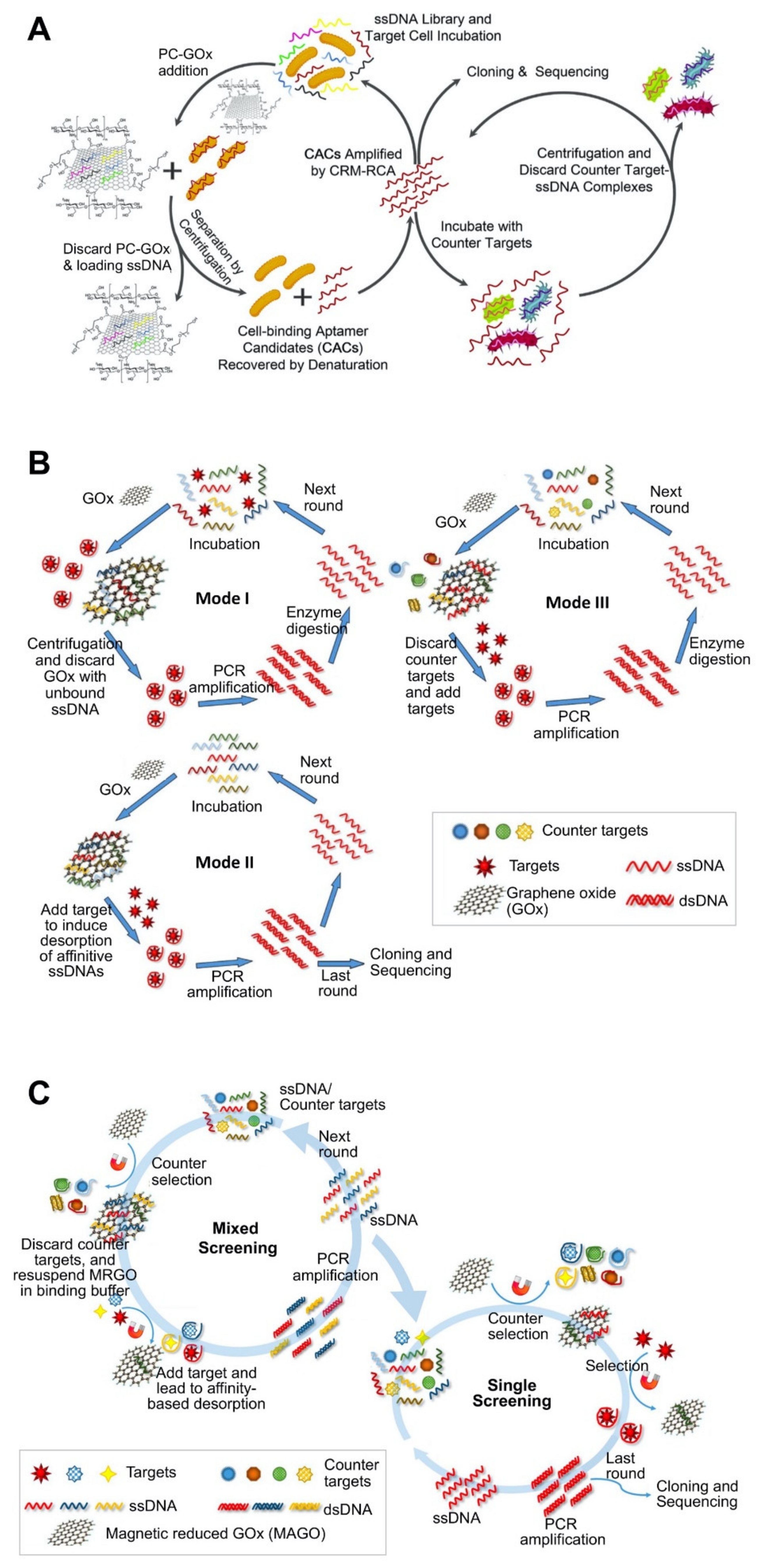
| Carbon nanomaterial–based platforms | GOx | Vibrio parahaemolyticus | 10.3 ± 2.5 nM | Flow cytometry using 5′ FITC-labeled aptamer | Prediction of 2D structure using Mfold. | Non-immobilization strategy. Use of GOx functionalized with poly ethylene glycol and chitosan for enhanced water solubility and biocompatibility. | [71]; Figure 2A |
| GOx | Okadaic acid (OA) | 40 nM | Fluorescence assay by labeling aptamer with FAM at the 5′-end. | 2D and 3D structure using RNA structure. | Screening of aptamer sequentially through three modes of SELEX. Use of oligo-free strategy during the early rounds, oligo-immobilizing strategy during last rounds, and counter-section strategy in the final rounds. Selection pressure: the decreasing amounts and shortening incubation time of ssDNA and OA reduced the probability of binding between ssDNA and OA. | [72]; Figure 2B | |
| GOx | Domoic acid (DA), saxitoxin (STX), and tetrodotoxin (TTX) | 62.07 ± 19.97 nM (for DA), 44.12 ± 15.38 nM (for TTX), 61.44 ± 23.18 nM (for STX) | Use of FAM-labeled aptamer immobilized on GOx. | Prediction of 2D structure using Mfold. | Use of magnetic-reduced GOx (MRGO) platform for simple and specific partitioning. Employing the quenching property of GOx against fluorophore. | [51]; Figure 2C | |
| GOx | Thioflavin T | ND | ND | 2D structure prediction by Nupack software. CD spectroscopy. Isothermal strand displacement amplification (SDA). | Non-immobilization strategy. | [73] | |
| GOx | Sulfaquinoxaline | 82.54 nM | Fluorescence assay by labeling aptamer with TAMRA. | Prediction of 2D structure using Mfold. | Use of the quenching ability of GOx against fluorophore. Use of TMARA-labeled aptamer to monitor the optimization of SELEX. | [16] | |
| Other nanomaterial-based platforms | AuNP | Zinc (II)-Protoporphyrin IX (ZnPPIX) | 9.53 ± 1.86 μM | Fluorescence assay by labeling aptamer with FAM. | Prediction of 2D structure using Mfold. CD spectroscopy. | Optimization of adsorbing ssDNA library on AuNPs (60 mM NaCl and incubation time of 75 min). | [44] |
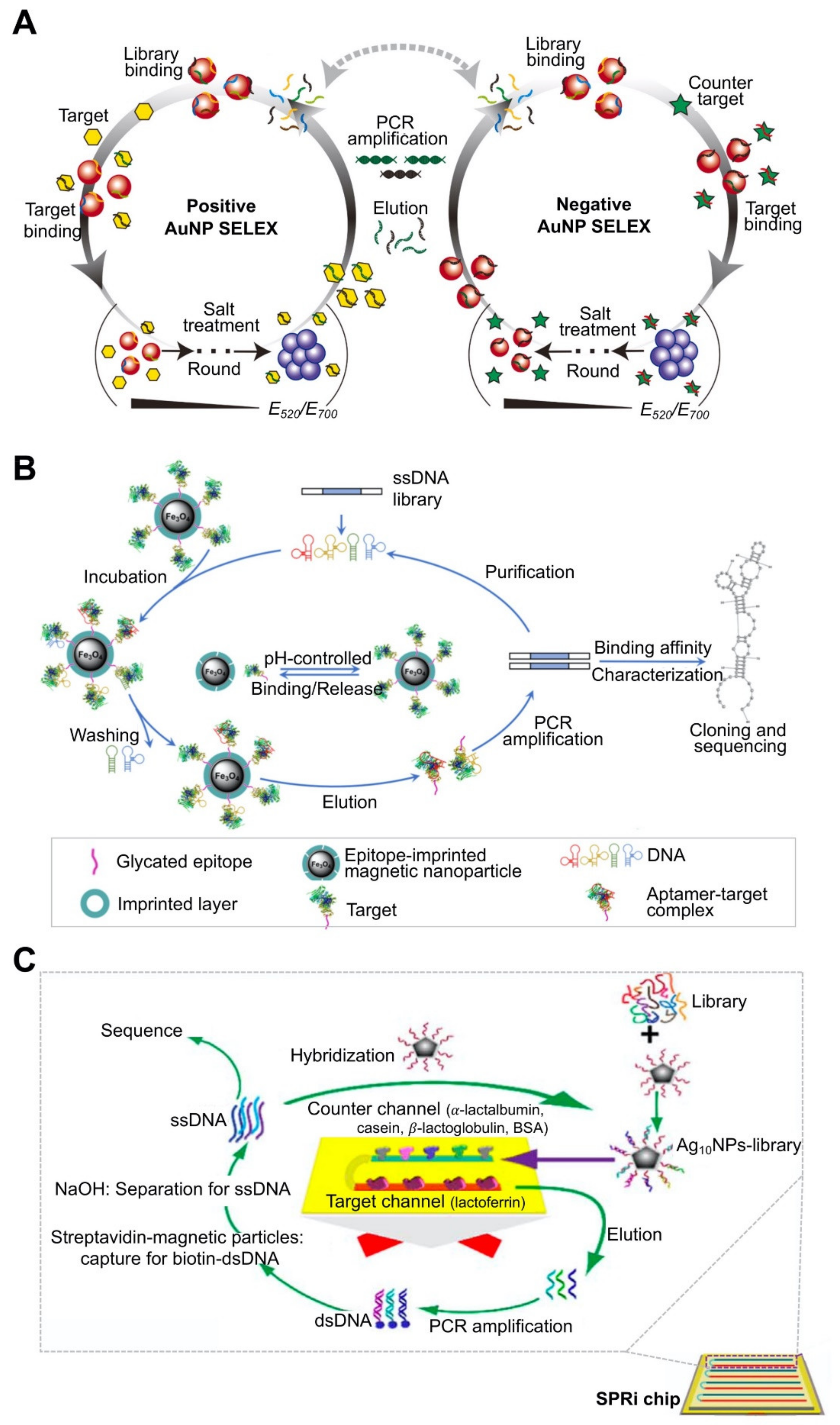
| AuNP | Brassinolide (BL), bisphenol A (BPA) | 17.3 nM (for BL), 37.9 nM (for BPA) | GNP-based colorimetric assays. | ITC. CD spectroscopy. | Monitoring of the progress of SELEX by salt-induced NP aggregation. Improvement of Kd value and specificity of truncated aptamer compared to full-length aptamer. | [74]; Figure 3A | |
| AuNP | Dichlorvos (DV) | 42.3 nM | ITC, NanoZyme (mimic peroxidase activity of GNPs)-based colorimetric assay. | CD spectroscopy. Determination of primary sequence homology among candidate aptamers using CLUSTALW. 2D structure prediction by Nupack software. | Aptamer-NanoZyme (AuNP having peroxidase mimic activity)-based colorimetric assay, colorimetry (AuNP, TMB), nanozyme. | [75] | |
| Magnetic nanoparticle (MNP) | Lipopolysaccharides (LPS) | 102 ± 17 nM | Fluorescence assay by labeling aptamer with FAM. | 2D structure prediction by DNAMAN 8. | Library-immobilization strategy. | [76] | |
| MNP | RNase B, transferrin | 91 ± 30 nM (for RNase B), 88 ± 31 nM (for transferrin) | Capillary electrophoresis (CE) equipped with laser-induced fluorescence (LIF) assay. | Selectivity analysis by CE-LIF assay. | Use of glycan-imprinted MNP. Target-immobilization strategy. | [77] | |
| MNP | Myoglobin (Mb), β2-microglobulin | 46.3 ± 10.4 nM (for Mb), 36.7 ± 11.4 nM (for β2-microglobulin) | Enzyme-linked oligosorbent assay | Enzyme-linked oligosorbent assay. | Use of epitope-imprinted MNP. Target-immobilization strategy. Short screening time (three cycles, takes 1 day). | [78]; Figure 3B | |
| MNP | Saponin (ginsenoside Re and Rb1) | 2.3 ± 0.3 μM (for Re), 4.9 ± 0.3 μM (for Rb1) | Fluorescence assay by labeling aptamer with FAM | 2D structure analysis. | Use of polyethyleneimine-assisted boronate affinity MNPs. | [79] | |
| Silver nanoparticle (AgNP) | Lactoferrin (Lac) | 0.953 ± 0.114 nM | Surface plasmon resonance measurement. | Specificity using surface plasmon resonance imaging, structure prediction by Oligoanalyzer 3.1. | Use of AgNP-conjugated oligos. Target immobilization on microarray. Enabled real-time monitoring of binding properties and specificity between the target and the aptamer. | [80]; Figure 3C |
| Classification | Material | Advantage | Limitation |
|---|---|---|---|
| Bead-based platform | Magnetic bead | Simple partitioning of unbound and bound oligos using magnet | Required a conjugation process for target or oligo-immobilization strategy |
| Agarose bead | Enables a microcolumn bead-based SELEX Reducing non-specific binding | Limitation of high-throughput selection | |
| Nickel bead | Enables a microcolumn bead-based SELEX Reducing non-specific binding enables a relatively free interaction of target and oligos by His-tag for lifting targets on solid substrate | Need for additional His-tagging of target | |
| Carbon-based nanomaterial platform | Graphene oxide (GOx) | Enables a free interaction of target and oligo Simple removal of unbound oligos using selective adsorption of ssDNA by GOx Enables fabrication of GOx with additional functionality (e.g., magnetic reduced GOx) Enables optimization of SELEX conditions using the fluorescent quenching property of GOx | Potential failure of separation by low water solubility |
| Gold nanoparticle | Enables a free interaction of target and oligo Simple removal of unbound oligos using selective adsorption of ssDNA by GOx Enables real-time monitoring SELEX process via color change by salt-induced AuNP aggregation | Required a conjugation process for target | |
| Magnetic nanoparticle | Simple partitioning of unbound and bound oligos using magnet | Need for a conjugation process for target or oligo-immobilization strategy | |
| Silver nanoparticle | Enables a development of SELEX strategy combined with other plasmonic sensing strategy (e.g., SPRi) | Required a conjugation process for target |
| Purpose | Software | Features | Website | Reference |
|---|---|---|---|---|
| Aptamer-protein interaction prediction | AptaNet | Use of balancing technique and a deep neural network | https://github.com/nedaemami/AptaNet (accessed on 24 August 2021) | [101] |
| PPAI | Use of the abstracted sequence features and the machine learning framework | http://39.96.85.9/ PPAI (accessed on 24 August 2021) | [102] | |
| RNA-protein interaction | RPINBASE | Predicting the interactions between RNAs and proteins by applying machine learning approaches | http://rpinbase.com/Explore (accessed on 24 August 2021) | [103] |
| Aptamer 3D structure prediction | RNAComposer | Based on the machine translation principle and operates on the RNA FRABASE database | http://rnacomposer.cs.put.poznan.pl/(accessed on 24 August 2021) | [104] |
| 3dRNA | Automated method of building RNA 3D structures from sequences and 2D structures by using the smallest secondary elements. | http://biophy.hust.edu.cn/3dRNA (accessed on 24 August 2021) | [105] | |
| Vfold3D | Template-based coarse-grained structure prediction model | http://rna.physics.missouri.edu/ vfold3D/ (accessed on 24 August 2021) | [106] | |
| simRNA | Simulations of RNA conformational dynamics (folding, unfolding, multiple chain complex formation, etc.) | https://genesilico.pl/SimRNAweb (accessed on 24 August 2021) | [107] | |
| ToGo-WF | Prediction of RNA 3D structures and RNA-RNA/protein interactions using the KNIME workflow | https://togo.medals.jp/active_local_rna_prediction.eng.html (accessed on 24 August 2021) | [108] | |
| Aptamer 2D structure prediction | ViennaRNA | Calculation of either minimum free energy or partition functions | https://www.tbi.univie.ac.at/RNA/ (accessed on 24 August 2021) | [109] |
| Mfold | Use of free energy minimization method | http://www.unafold.org/ (accessed on 24 August 2021) | [110] | |
| RNAsoft (AveRNA) | Energy-based, pseudoknot-free RNA secondary structure prediction | http://www.rnasoft.ca/ (accessed on 24 August 2021) | [111] | |
| RNAstructure | Use of free energy minimization method | https://rna.urmc.rochester.edu/RNAstructureWeb/ (accessed on 24 August 2021) | [112] | |
| NUPACK | Calculation of the partition function and minimum free energy secondary structure | http://www.nupack.org/ (accessed on 24 August 2021) | [113] | |
| Molecular docking | ZDOCK | Fast Fourier transform (FFT) algorithm to search and obtain all the binding poses | https://zdock.umassmed.edu/ (accessed on 24 August 2021) | [114] |
| MDockPP | FFT algorithm to collect all putative binding poses | https://zougrouptoolkit.missouri.edu/MDockPP/ (accessed on 24 August 2021) | [115] | |
| AutoDock | Calculation of the free energy to score binding poses | http://autodock.scripps.edu/ (accessed on 24 August 2021) | [116] | |
| AutoDock Vina | Use of an empirical scoring function to score the binding poses | http://vina.scripps.edu/ (accessed on 24 August 2021) | [117] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, D.-M.; Go, M.-J.; Lee, J.; Na, D.; Yoo, S.-M. Recent Advances in Micro/Nanomaterial-Based Aptamer Selection Strategies. Molecules 2021, 26, 5187. https://doi.org/10.3390/molecules26175187
Kim D-M, Go M-J, Lee J, Na D, Yoo S-M. Recent Advances in Micro/Nanomaterial-Based Aptamer Selection Strategies. Molecules. 2021; 26(17):5187. https://doi.org/10.3390/molecules26175187
Chicago/Turabian StyleKim, Dong-Min, Myeong-June Go, Jingyu Lee, Dokyun Na, and Seung-Min Yoo. 2021. "Recent Advances in Micro/Nanomaterial-Based Aptamer Selection Strategies" Molecules 26, no. 17: 5187. https://doi.org/10.3390/molecules26175187
APA StyleKim, D.-M., Go, M.-J., Lee, J., Na, D., & Yoo, S.-M. (2021). Recent Advances in Micro/Nanomaterial-Based Aptamer Selection Strategies. Molecules, 26(17), 5187. https://doi.org/10.3390/molecules26175187






