A Golgi Apparatus-Targeting, Naphthalimide-Based Fluorescent Molecular Probe for the Selective Sensing of Formaldehyde
Abstract
:1. Introduction
2. Results and Discussion
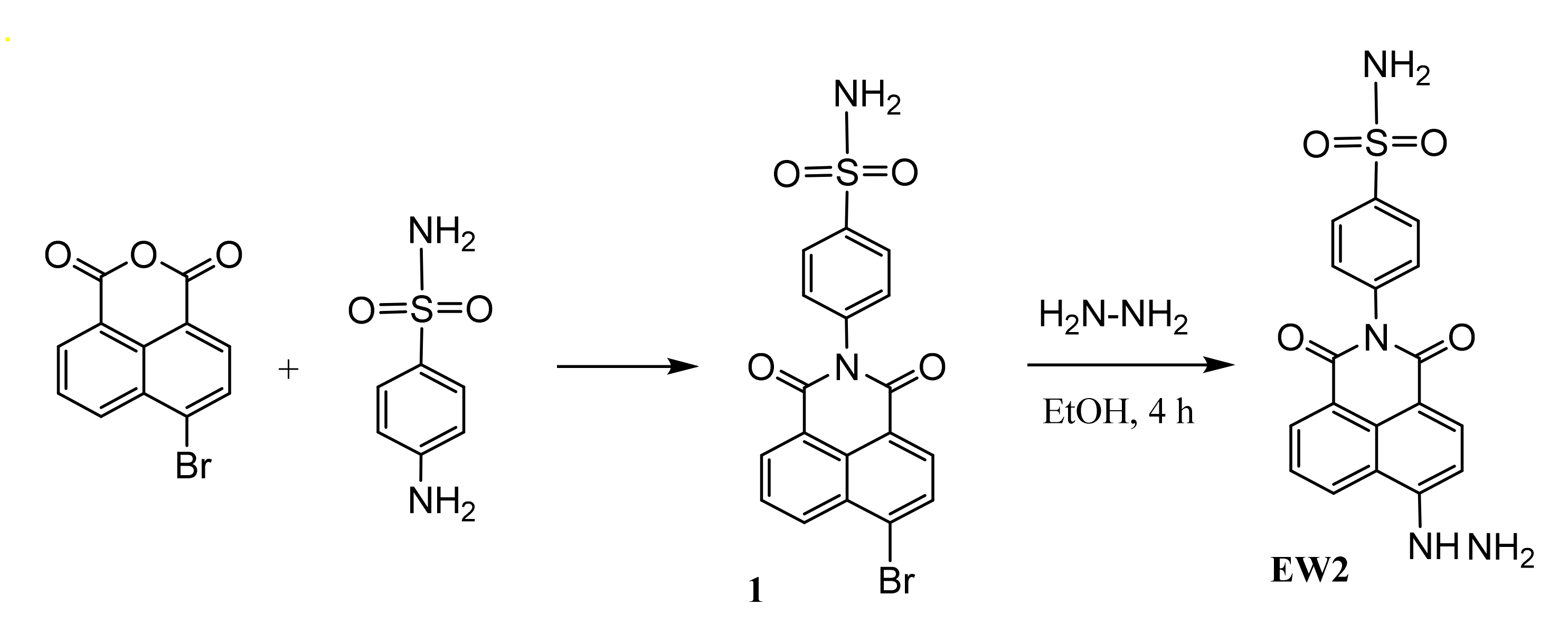
2.1. Design and Synthesis of Probe EW2
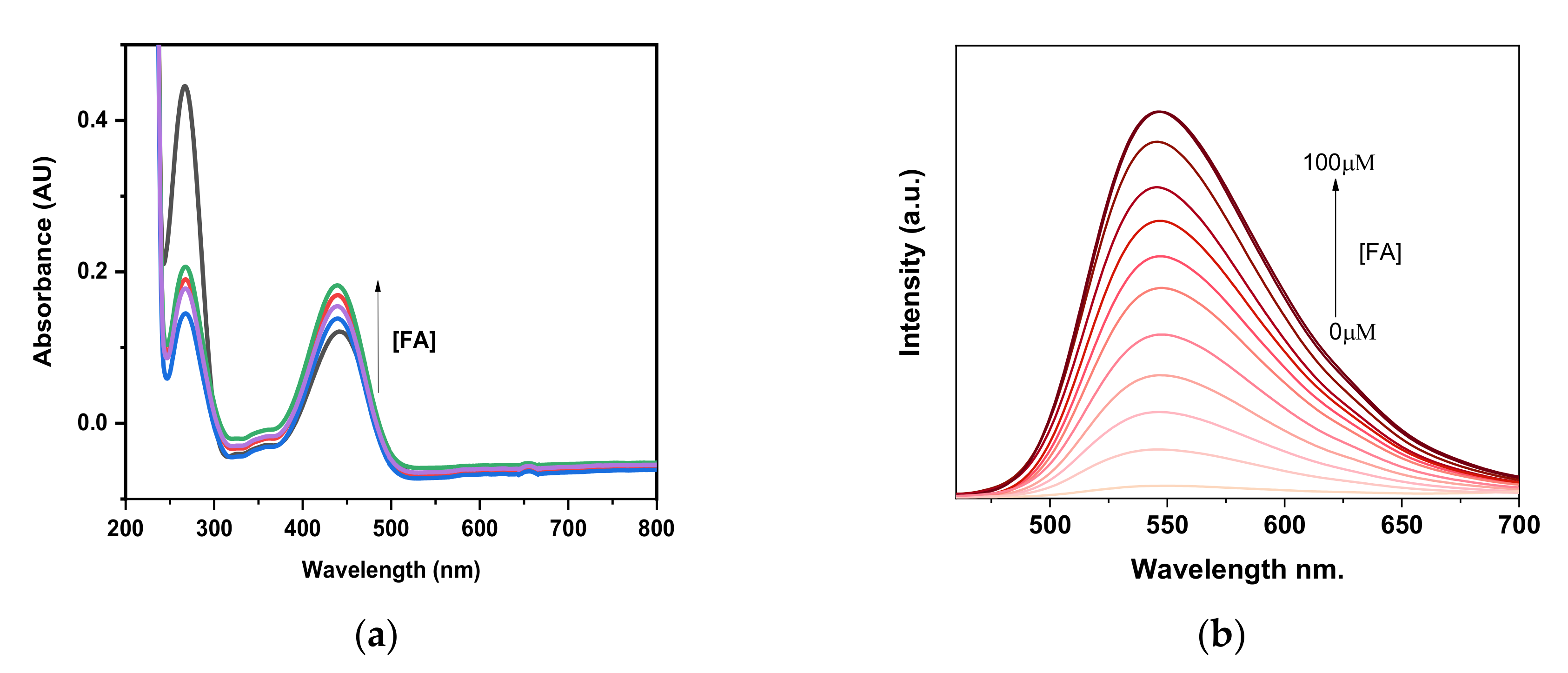
2.2. Optical Properties of Probe EW2
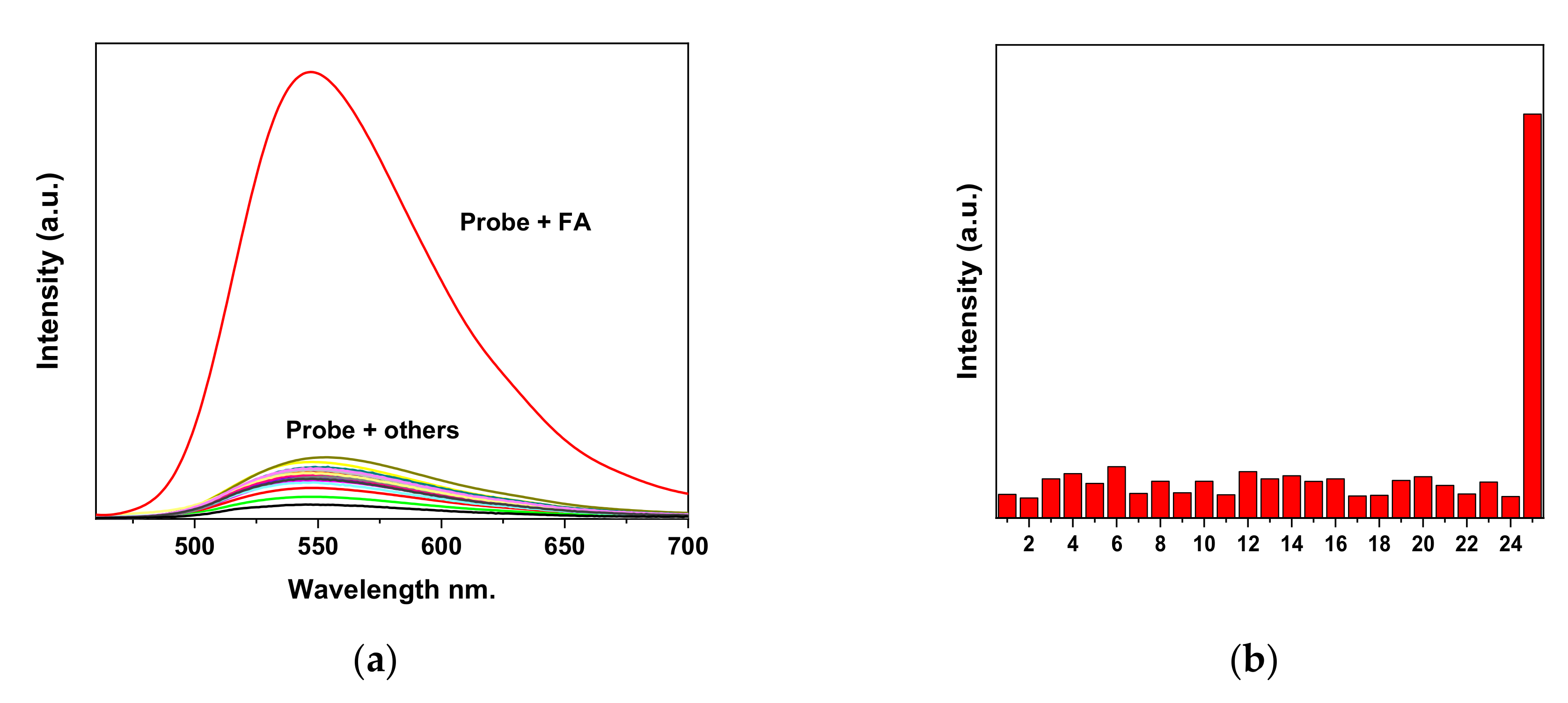
2.3. Selectivity of EW2 towards FA
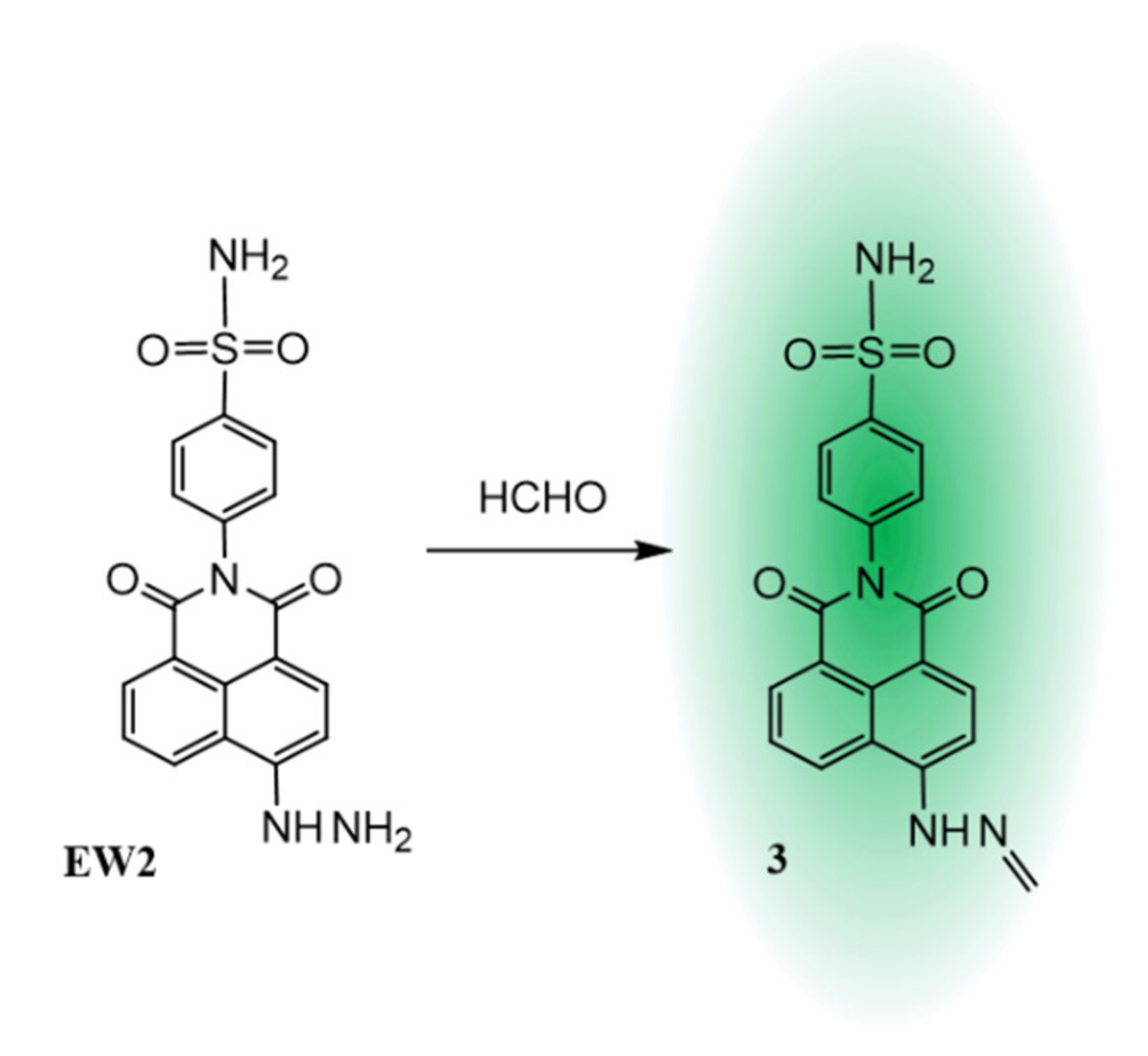
2.4. Proposed Sensing Mechanism
2.5. Detection of Formaldehyde in Food Samples
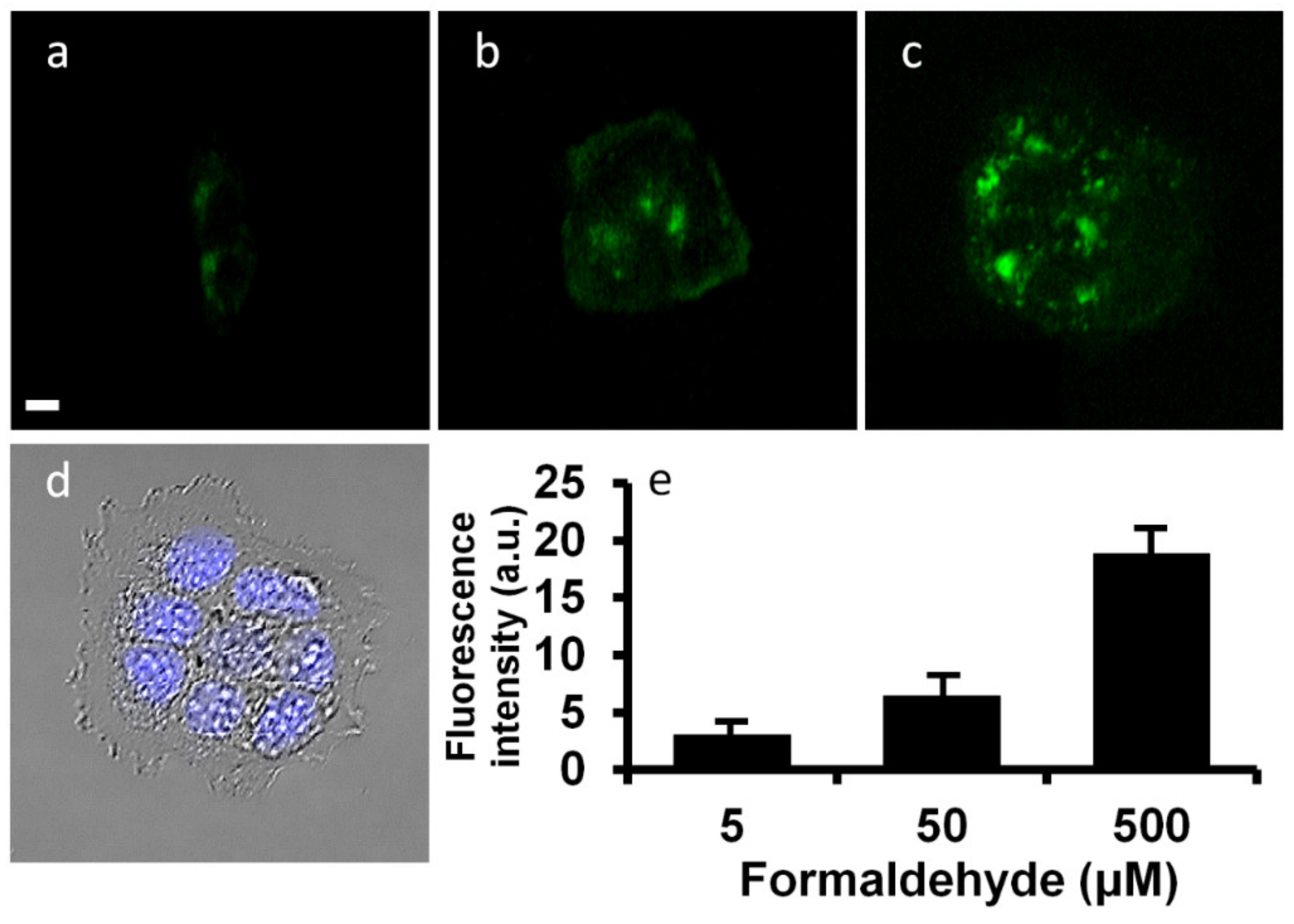
2.6. Cell Imaging of the Probe
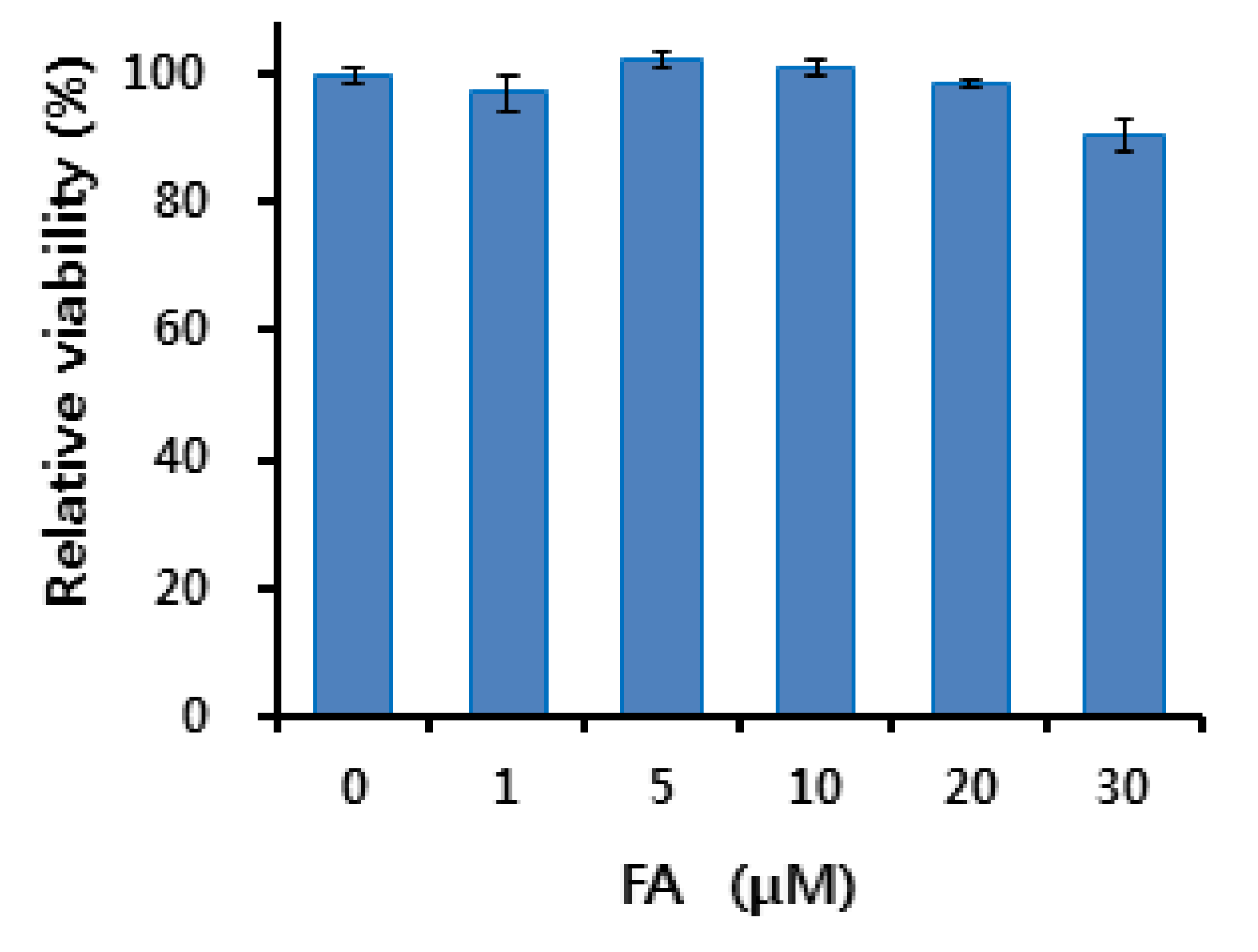
2.7. Cytotoxicity Studies
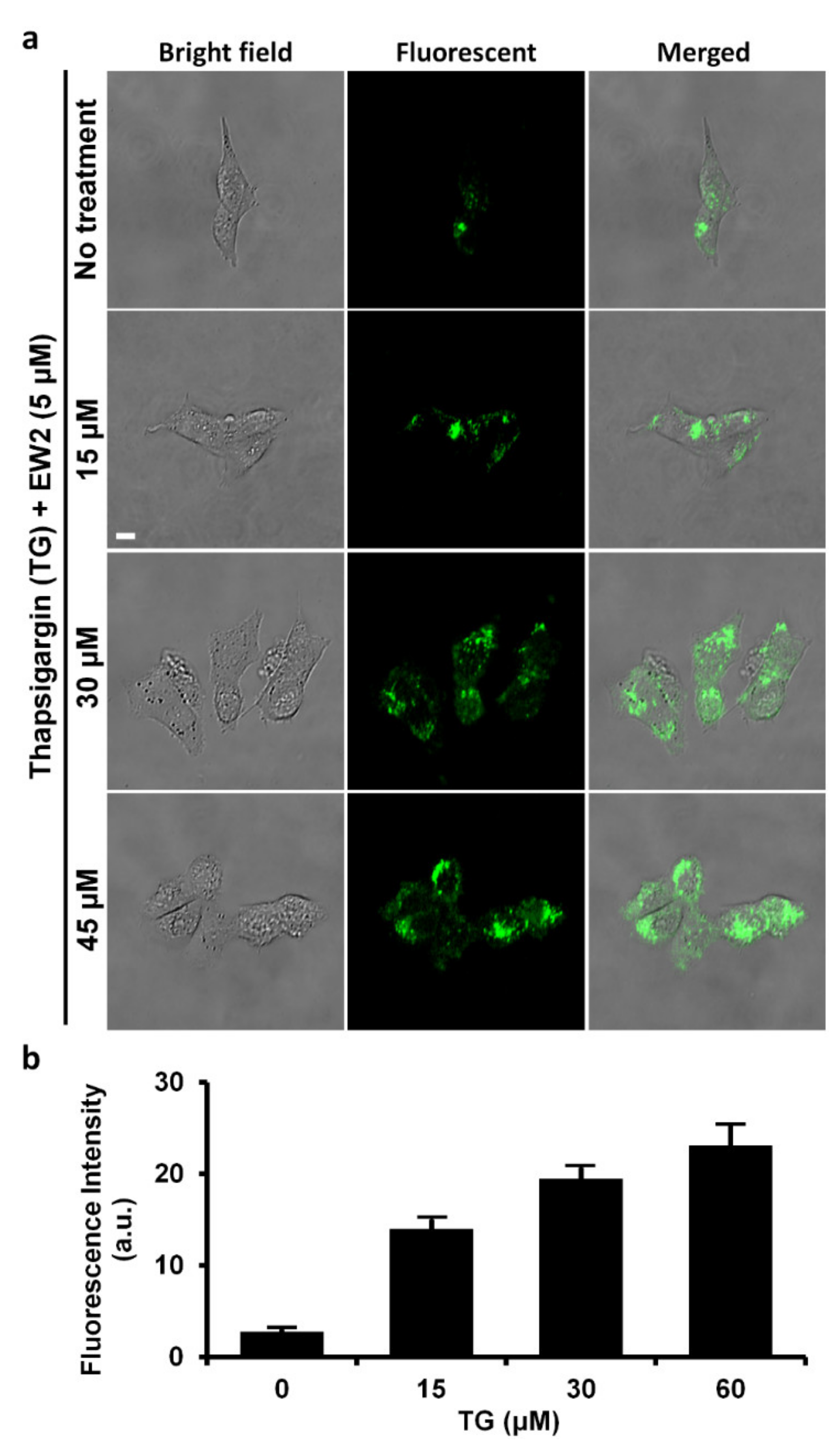
2.8. Inhibition Studies of the Probe
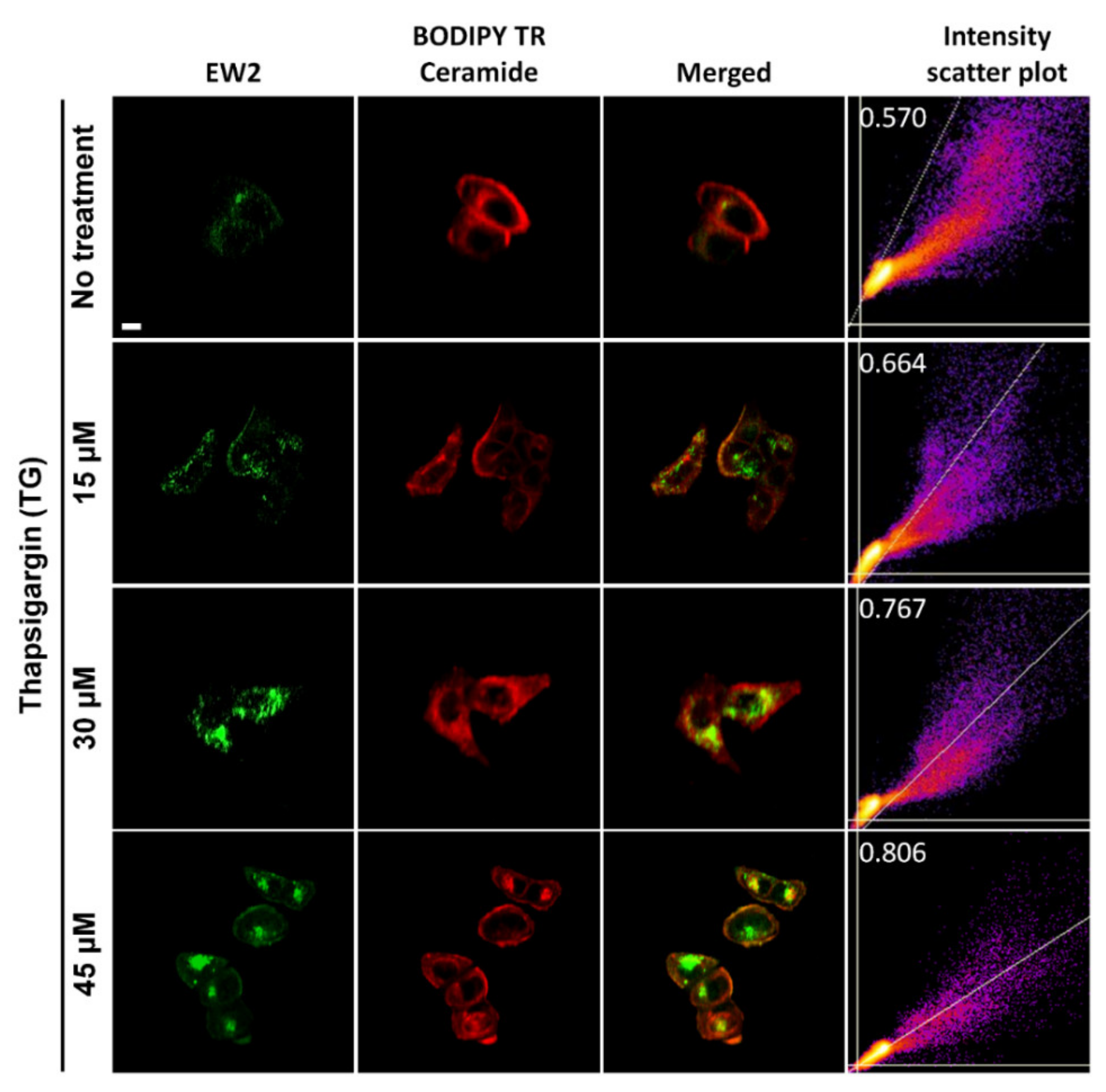
2.9. Colocalization Experiment
2.10. Imaging Endogenous FA in Cells
3. Materials and Methods
3.1. Materials and Instrumentation
3.2. Synthesis of Compound 1
3.3. Synthesis of Probe EW2
3.4. Absorption and Fluorescent Titration Measurements
3.5. Preparation of Formaldehyde Source from Dried Shiitake Mushrooms
3.6. Detection of Formaldehyde Source from Dried Shiitake Mushrooms
3.7. Preparation of Onion Extract and Detection of Formaldehyde
3.8. Cell Imaging
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
Sample Availability
References
- Zhou, Y.; Yan, J.; Zhang, N.; Li, D.; Xiao, S.; Zheng, K. A ratiometric fluorescent probe for formaldehyde in aqueous solution, serum and air using aza-cope reaction. Sens. Actuators B Chem. 2018, 258, 156–162. [Google Scholar] [CrossRef]
- El Sayed, S.; Pascual, L.; Licchelli, M.; Martínez-Máñez, R.; Gil, S.; Costero, A.M.; Sancenón, F. Chromogenic Detection of Aqueous Formaldehyde Using Functionalized Silica Nanoparticles. ACS Appl. Mater. Inter. 2016, 8, 14318–14322. [Google Scholar] [CrossRef]
- Wang, T.; Gao, X.; Tong, J.; Chen, L.C. Determination of formaldehyde in beer based on cloud point extraction using 2,4-dinitrophenylhydrazine as derivative reagent. Food Chem. 2012, 131, 11577–11582. [Google Scholar] [CrossRef]
- Chiou, J.; Leung, A.H.H.; Lee, H.W.; Wong, W.T. Rapid testing methods for food contaminants and toxicants. J. Integr. Agric. 2015, 14, 2243–2264. [Google Scholar] [CrossRef] [Green Version]
- Salthammer, T.; Mentese, S.; Marutzky, R. Formaldehyde in the Indoor Environment. Chem. Rev. 2010, 110, 2536–2572. [Google Scholar] [CrossRef] [PubMed]
- Unzeta, M.; Sol, M.; Boada, M.; Hernandez, M. Semicarbazide-sensitive amine oxidase (SSAO) and its possible contribution to vascular damage in Alzheimer’s disease. J. Neural Transm. 2007, 114, 857–862. [Google Scholar] [CrossRef]
- Hauptmann, M.; Stewart, P.A.; Lubin, J.H.; Beane Freeman, L.E.; Hornung, R.W.; Herrick, R.F.; Hoover, R.N.; Fraumeni, J.F., Jr.; Blair, A.; Hayes, R.B. Mortality From Lymphohematopoietic Malignancies and Brain Cancer Among Embalmers Exposed to Formaldehyde. Natl. Cancer Inst. 2009, 101, 1696–1708. [Google Scholar] [CrossRef]
- Afkhami, A.; Bagheri, H. Preconcentration of trace amounts of formaldehyde from water, biological and food samples using an efficient nanosized solid phase, and its determination by a novel kinetic method. Microchim. Acta. 2012, 176, 217–227. [Google Scholar] [CrossRef]
- Luo, W.; Li, H.; Zhang, Y.; Ang, C.Y.W. Determination of formaldehyde in blood plasma by high-performance liquid chromatography with fluorescence detection. J. Chromatogr. B Biomed. Sci. Appl. 2001, 753, 253–257. [Google Scholar] [CrossRef]
- Yu, P.H.; Cauglin, C.; Wempe, K.L.; Gubisne-Haberle, D. A novel sensitive high-performance liquid chromatography/electrochemical procedure for measuring formaldehyde produced from oxidative deamination of methylamine and in biological samples. Anal. Biochem. 2003, 318, 285–290. [Google Scholar] [CrossRef]
- Kato, S.; Burke, P.J.; Koch, T.H.; Bierbaum, V.M. Formaldehyde in Human Cancer Cells: Detection by Preconcentration-Chemical Ionization Mass Spectrometry. Anal. Chem. 2001, 73, 2992–2997. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.; Dodani, S.C.; Chang, C.J. Reaction-based small-molecule fluorescent probes for chemoselective bioimaging. Nat. Chem. 2012, 4, 973–984. [Google Scholar] [CrossRef]
- Yang, Y.; Zhao, Q.; Feng, W.; Li, F. Luminescent Chemodosimeters for Bioimaging. Chem. Rev. 2013, 113, 192–270. [Google Scholar] [CrossRef]
- Chen, X.; Lee, K.A.; Ha, E.M.; Lee, K.M.; Seo, Y.Y.; Choi, H.K.; Kim, H.N.; Kim, M.J.; Cho, C.S.; Lee, S.Y.; et al. A specific and sensitive method for detection of hypochlorous acid for the imaging of microbe-induced HOCl production. Chem. Commun. 2011, 47, 4373–4375. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Kong, X.; Xu, A.; Dong, B.; Lin, W. Development of a Two-Photon Fluorescent Probe for Imaging of Endogenous Formaldehyde in Living Tissues. Angew. Chem. Int. Ed. 2016, 55, 3356–3359. [Google Scholar] [CrossRef]
- Roth, A.; Li, H.; Anorma, C.; Chan, J. A Reaction-Based Fluorescent Probe for Imaging of Formaldehyde in Living Cells. J. Am. Chem. Soc. 2015, 137, 10890–10893. [Google Scholar] [CrossRef]
- Xu, A.; Tang, Y.; Lin, W. Development of a mitochondrial-targeted two-photon fluorescence turn-on probe for formaldehyde and its bio-imaging applications in living cells and tissues. New J. Chem. 2018, 42, 8325–8329. [Google Scholar] [CrossRef]
- Xin, F.; Tian, Y.; Gao, C.; Guo, B.; Wu, Y.; Zhao, J.; Jing, J.; Zhang, X. A two-photon fluorescent probe for basal formaldehyde imaging in zebrafish and visualization of mitochondrial damage induced by FA stress. Analyst 2019, 144, 2297–2303. [Google Scholar] [CrossRef]
- Kim, H.M.; Cho, B.R. Small-Molecule Two-Photon Probes for Bioimaging Applications. Chem. Rev. 2015, 115, 5014–5055. [Google Scholar] [CrossRef]
- Hui, W.; Zixu, H.; Yuyun, Y.; Jiao, Z.; Wei, Z.; Wen, Z.; Ping, L.; Bo, T. Ratiometric fluorescence imaging of Golgi H2O2 reveals a correlation between Golgi oxidative stress and hypertension. Chem. Sci. 2019, 10, 10876–10880. [Google Scholar]
- Zhu, H.; Liang, C.; Cai, X.; Zhang, H.; Liu, C.; Jia, P.; Li, Z.; Yu, Y.; Zhang, X.; Sheng, W.; et al. Rational design of a targetable fluorescent probe for visualizing H2S production under golgi stress response elicited by monensin. Anal. Chem. 2020, 92, 1883–1889. [Google Scholar] [CrossRef] [PubMed]
- Brewer, T.F.; Chang, C.J. An Aza-Cope Reactivity-Based Fluorescent Probe for Imaging Formaldehyde in Living Cells. J. Am. Chem. Soc. 2015, 137, 10886–10889. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liang, X.-G.; Chen, B.; Shao, L.-X.; Cheng, J.; Huang, M.-Z.; Chen, Y.; Hu, Y.-Z.; Han, Y.-F.; Han, F.; Li, X. A Fluorogenic Probe for Ultrafast and Reversible Detection of Formaldehyde in Neurovascular Tissues. Theranostics 2017, 7, 2305–2313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Sample | Determined FA (μM) | Spiked FA (μM) | Total FA Found (μM) | Recovery (%) |
|---|---|---|---|---|
| Mushrooms | 5.2 | 5 | 8.2 | 81 |
| 10 | 12.1 | 85 | ||
| 15 | 20.5 | 103 | ||
| Onions | 4.8 | 5 | 7.5 | 80 |
| 10 | 11.1 | 83 | ||
| 15 | 18.3 | 99 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fortibui, M.M.; Lim, W.; Lee, S.; Park, S.; Kim, J. A Golgi Apparatus-Targeting, Naphthalimide-Based Fluorescent Molecular Probe for the Selective Sensing of Formaldehyde. Molecules 2021, 26, 4980. https://doi.org/10.3390/molecules26164980
Fortibui MM, Lim W, Lee S, Park S, Kim J. A Golgi Apparatus-Targeting, Naphthalimide-Based Fluorescent Molecular Probe for the Selective Sensing of Formaldehyde. Molecules. 2021; 26(16):4980. https://doi.org/10.3390/molecules26164980
Chicago/Turabian StyleFortibui, Maxine Mambo, Wanyoung Lim, Sohyun Lee, Sungsu Park, and Jinheung Kim. 2021. "A Golgi Apparatus-Targeting, Naphthalimide-Based Fluorescent Molecular Probe for the Selective Sensing of Formaldehyde" Molecules 26, no. 16: 4980. https://doi.org/10.3390/molecules26164980
APA StyleFortibui, M. M., Lim, W., Lee, S., Park, S., & Kim, J. (2021). A Golgi Apparatus-Targeting, Naphthalimide-Based Fluorescent Molecular Probe for the Selective Sensing of Formaldehyde. Molecules, 26(16), 4980. https://doi.org/10.3390/molecules26164980









