Synthesis, Characterization, In Vitro Anticancer Potentiality, and Antimicrobial Activities of Novel Peptide–Glycyrrhetinic-Acid-Based Derivatives
Abstract
:1. Introduction
2. Results and Discussion
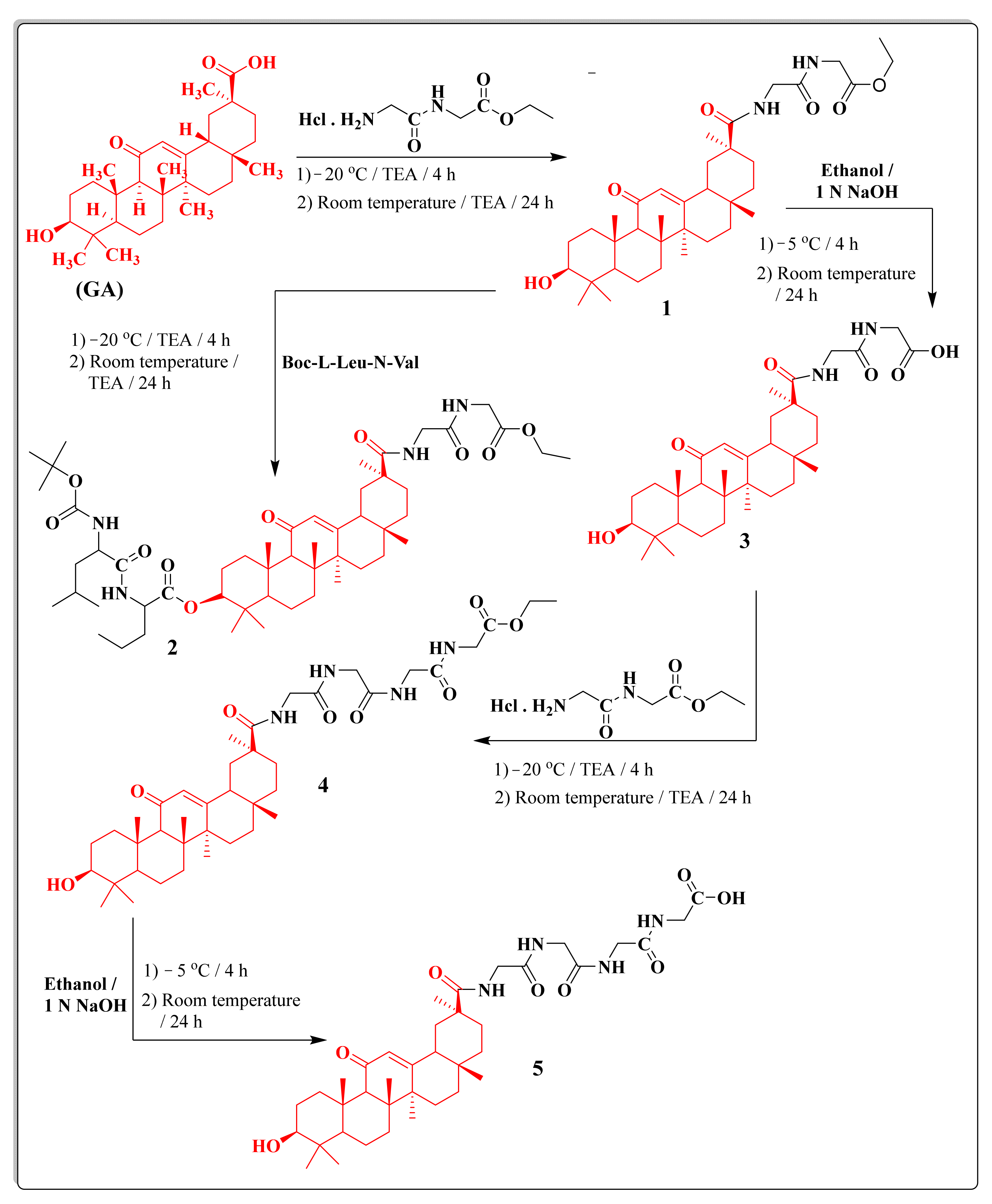
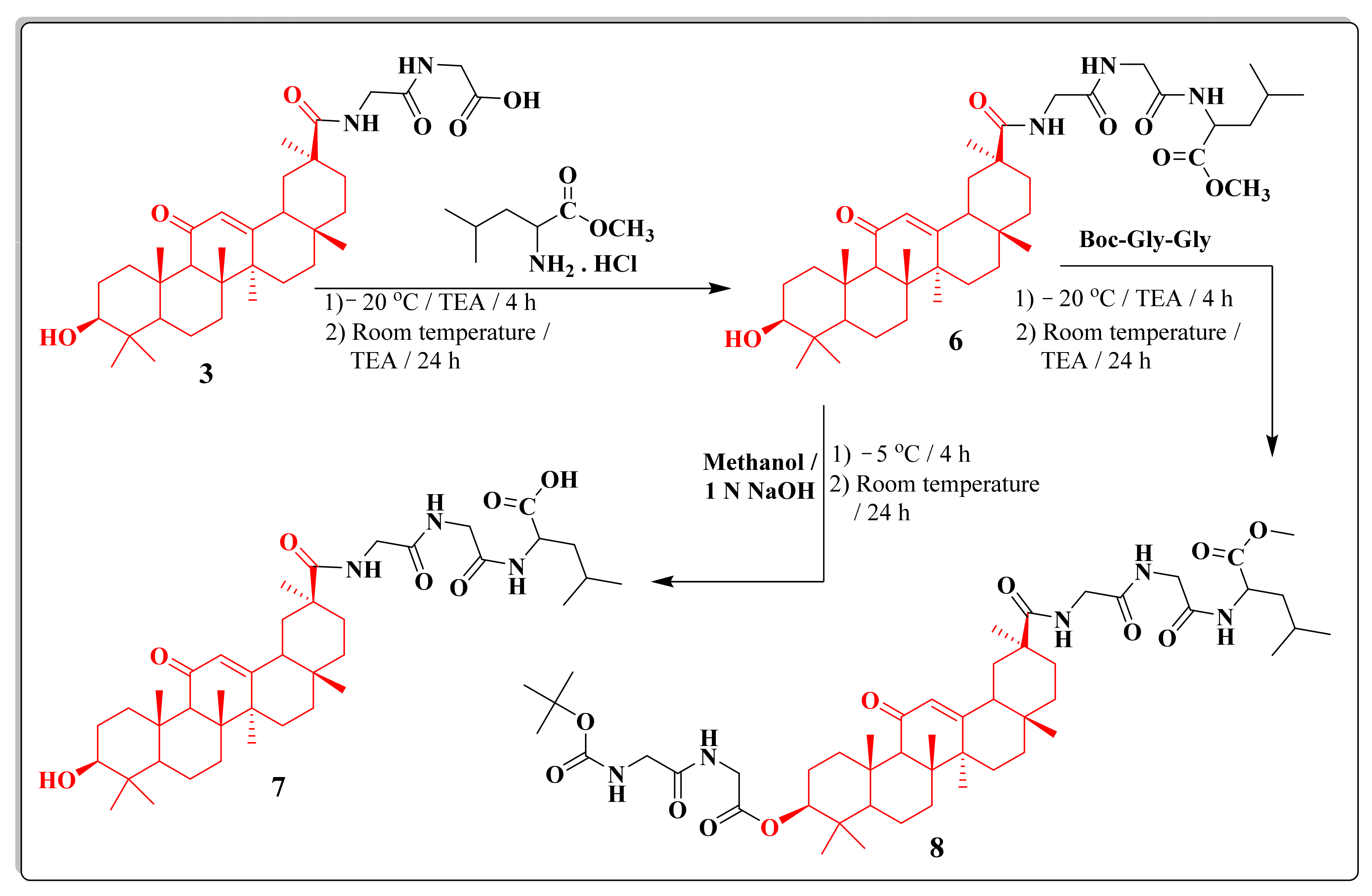
2.1. Chemistry
2.2. Biological Activity
2.2.1. Anticancer Evaluation
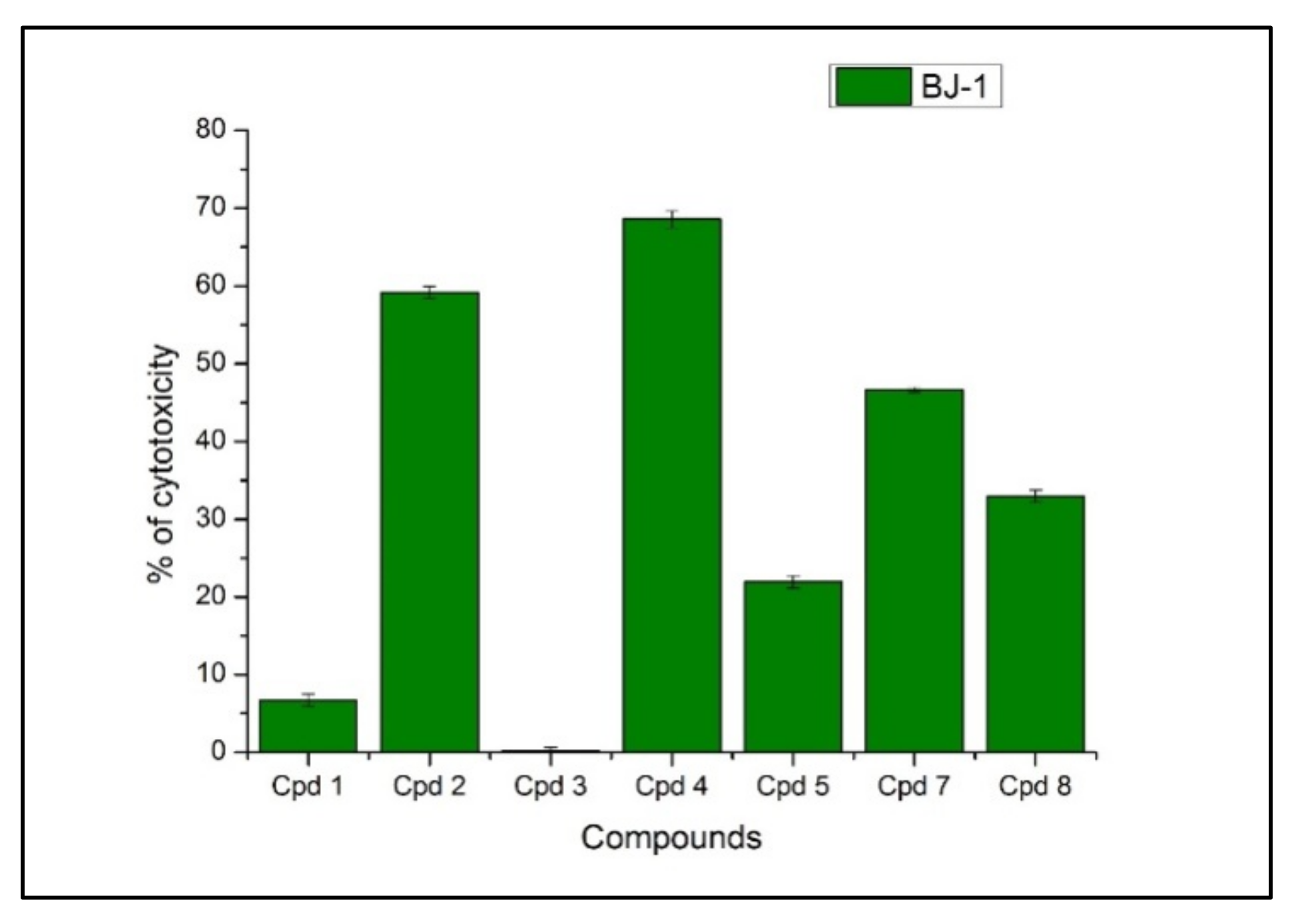
2.2.2. The Effect of the GA Peptides against the Normal Skin Fibroblasts Cell Line BJ-1
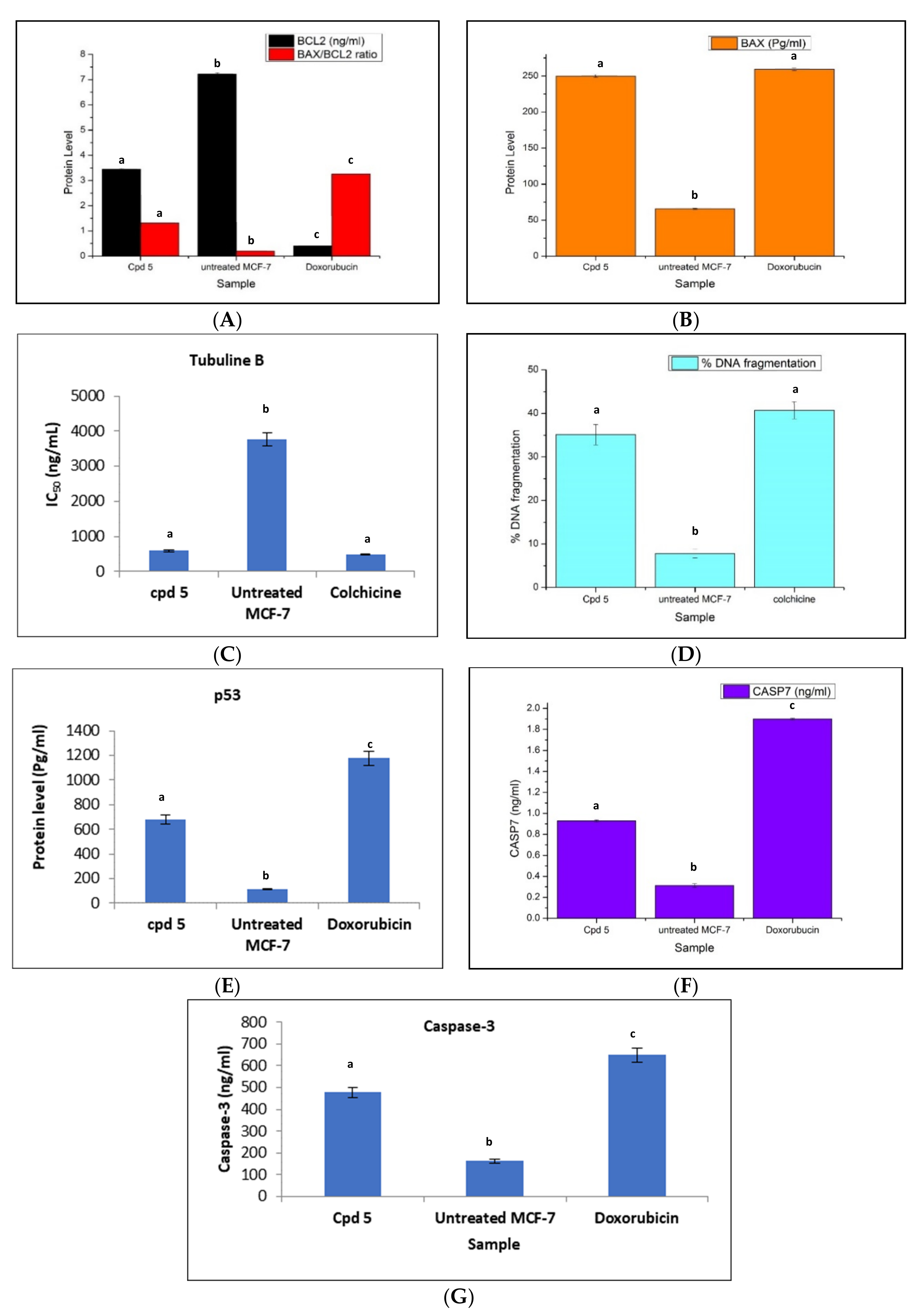
2.2.3. The Effect of the GA Peptide 5 on Bax, Bcl-2, and p53 Levels
2.2.4. Effect of the Peptide 5 on the Level of Caspase-3, Caspase-7, Tubulin Polymerization (TubB), and % of DNA Fragmentation
2.2.5. Antimicrobial Activity
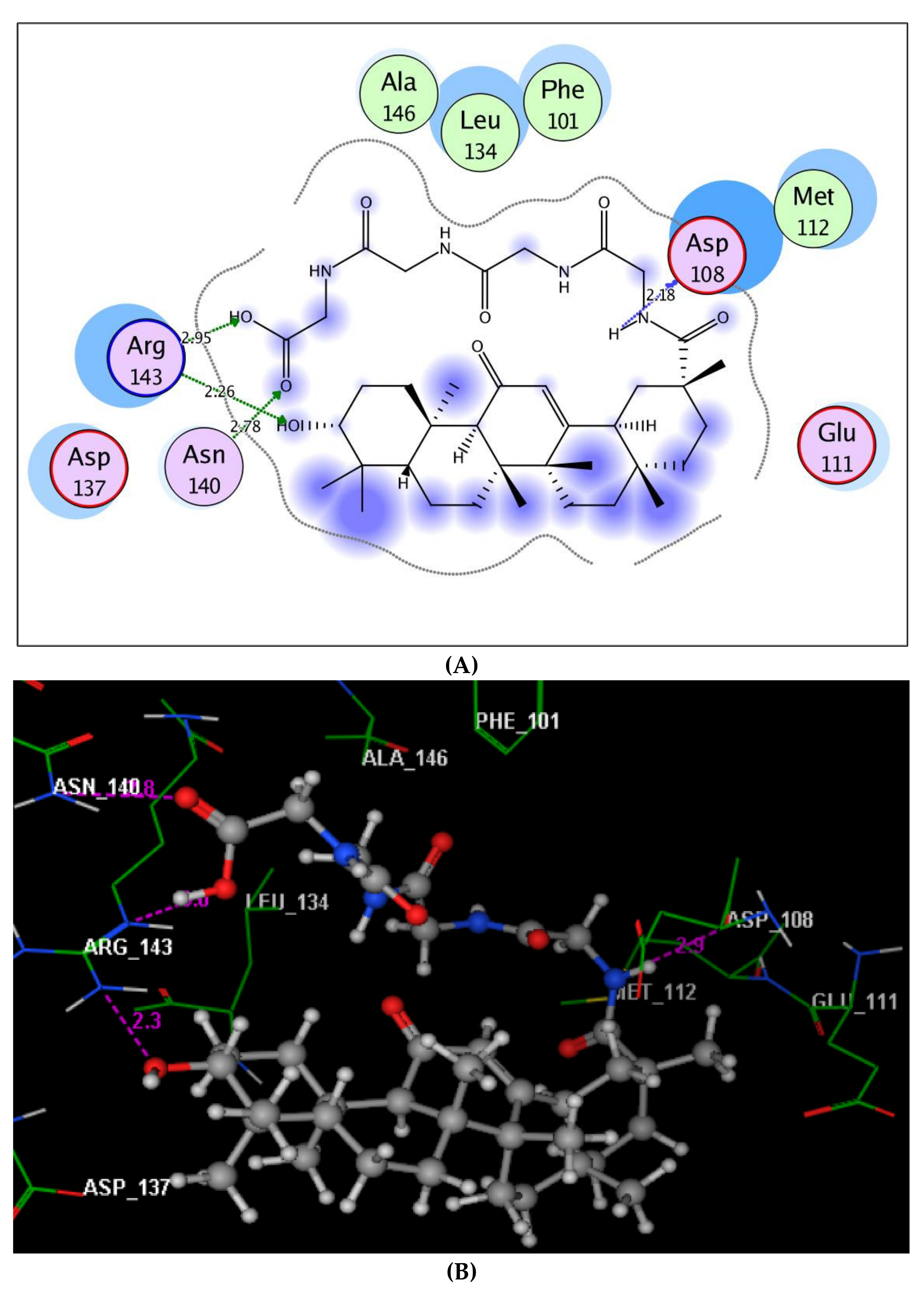
2.3. Molecular Docking Study on Caspase-3 and Bcl-2
3. Materials and Methods
3.1. Chemistry
3.1.1. Synthesis of Amino Acid Ester Hydrochlorides and Boc-Protected Amino Acids
3.1.2. Synthesis of OH-GA-Gly-Gly-OEt; Ethyl 2-(2-(10-Hydroxy-2,4a,6a,6b,9,9,12a-heptamethyl-13-oxo-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicene-2-carboxamido)acetamido)acetate, (1)
3.1.3. Synthesis of Boc-l-Leu-N-Val-GA-GLy-Gly-Oet; 11-(2-(2-Ethoxy-2-oxoethylamino)-2-oxoethylcarbamoyl)-4,4,6a,6b,8a,11,14b-heptamethyl-14-oxo-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,14,14a,14b-icosahydropicen-3-yl 2-(2-(tert-Butoxycarbonylamino)-4-methylpentanamido)pentanoate, (2)
3.1.4. Synthesis of OH-GA-Gly-Gly-COOH; 2-(2-(10-Hydroxy-2,4a,6a,6b,9,9,12a-heptamethyl-13-oxo-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicene-2-carboxamido)acetamido)acetic acid; (3)
3.1.5. Synthesis of OH-GA-Gly-Gly- Gly-Gly-OEt; Ethyl 1-(10-Hydroxy-2,4a,6a,6b,9,9,12a-heptamethyl-13-oxo-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicen-2-yl)-1,4,7,10-tetraoxo-2,5,8,11-tetraazatridecan-13-oate; (4)
3.1.6. Synthesis of OH-GA-Gly-Gly-Gly-Gly–COOH; 1-(10-Hydroxy-2,4a,6a,6b,9,9,12a-heptamethyl-13-oxo-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicen-2-yl)-1,4,7,10-tetraoxo-2,5,8,11-tetraazatridecan-13-oic Acid; (5)
3.1.7. Synthesis of OH-GLA-GLy-Gly-l-Leu-Ome(Sh-12);Methyl 2-(2-(2-(10-Hydroxy-2,4a,6a,6b,9,9,12a-heptamethyl-13-oxo-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicene-2-carboxamido)acetamido)acetamido)-4-methylpentanoate; (6)
3.1.8. Synthesis of OH-GA-Gly-Gly-l-Leu-COOH; 2-(2-(2-(10-Hydroxy-2, 4a, 6a, 6b, 9, 9, 12a-heptamethyl-13-oxo-1, 2, 3, 4, 4a, 5, 6, 6a, 6b, 7, 8, 8a, 9, 10, 11, 12, 12a, 12b, 13, 14b-icosahydropicene-2-carboxamido) acetamido) acetamido)-4-methylpentanoic Acid; (7)
3.1.9. Synthesis of Boc-Gly-Gly-GA-Gly-Gly-l-Leu-Ome; Methyl 2-(2-(2-(10-(2-(2-(Tert-butoxycarbonylamino)acetamido)acetoxy)-2,4a,6a,6b,9,9,12a-heptamethyl-13-oxo-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicene-2-carboxamido)acetamido)acetamido)-4-methylpentanoate, (8)
3.2. Biological Activities
3.2.1. Cytotoxicity
3.2.2. Estimation of Bcl-2 Level
3.2.3. Estimation of Bax Level
3.2.4. Estimation of Human p53 Level
3.2.5. Calculation of IC50Values
3.2.6. Human CASP-3(Caspase-3) Estimation
3.2.7. Human CASP-7 (Caspase-7) Estimation
3.2.8. Enzyme-Linked Immunosorbent Assay Kit for Tubulin Beta (TUBb)
3.2.9. Estimation of DNA Fragmentation through DPA Assay
3.2.10. In Vitro Antimicrobial Activity
3.3. Molecular Docking Study
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Huang, M.; Lu, J.-J.; Ding, J. Natural Products in Cancer Therapy: Past, Present and Future. Nat. Prod. Bioprospecting 2021, 11, 5–13. [Google Scholar] [CrossRef]
- Singh, S.; Sharma, B.; Kanwar, S.S.; Kumar, A. Lead Phytochemicals for Anticancer Drug Development. Front. Plant Sci. 2016, 7, 1667. [Google Scholar] [CrossRef] [Green Version]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef]
- Huang, M.-Y.; Zhang, L.-L.; Ding, J.; Lu, J.-J. Anticancer drug discovery from Chinese medicinal herbs. Chin. Med. 2018, 13, 35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khwaza, V.; Mlala, S.; Oyedeji, O.; Aderibigbe, B. Pentacyclic Triterpenoids with Nitrogen-Containing Heterocyclic Moiety, Privileged Hybrids in Anticancer Drug Discovery. Molecules 2021, 26, 2401. [Google Scholar] [CrossRef] [PubMed]
- Borková, L.; Frydrych, I.; Jakubcová, N.; Adámek, R.; Lišková, B.; Gurská, S.; Medvedíková, M.; Hajdúch, M.; Urban, M. Synthesis and biological evaluation of triterpenoid thiazoles derived from betulonic acid, dihydrobetulonic acid, and ursonic acid.Eur. J. Med. Chem. 2020, 185, 111806. [Google Scholar] [CrossRef] [PubMed]
- Kamble, S.M.; Goyal, S.N.; Patil, C.R. Multifunctional pentacyclic triterpenoids as adjuvants in cancer chemotherapy: A review. RSC Adv. 2014, 4, 33370–33382. [Google Scholar] [CrossRef]
- Herrera-España, A.D.; Us-Martín, J.; Hernández-Ortega, S.; Mirón-López, G.; Quijano, L.; Villanueva-Toledo, J.R.; Mena-Rejón, G.J. Synthesis, structure analysis and activity against breast and cervix cancer cells of a triterpenoid thiazole derived from ochraceolide A. J. Mol. Struct. 2020, 1204, 127555. [Google Scholar] [CrossRef]
- Ikeda, T.; Yokomizo, K.; Okawa, M.; Tsuchihashi, R.; Kinjo, J.; Nohara, T.; Uyeda, M. Anti-herpes Virus Type 1 Activity of Oleanane-Type Triterpenoids. Biol. Pharm. Bull. 2005, 28, 1779–1781. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.; Ji, L.; Yu, C.; Chen, Q.; Ge, Q.; Lu, Y. MiR-423-5p Regulates Cells Apoptosis and Extracellular Matrix Degradation via Nucleotide-Binding, Leucine-Rich Repeat Containing X1 (NLRX1) in Interleukin 1 beta (IL-1β)-Induced Human Nucleus Pulposus Cells. Med. Sci. Monit. 2020, 26, e922497. [Google Scholar] [CrossRef]
- Manu, K.A.; Kuttan, G. Ursolic acid induces apoptosis by activating p53 and caspase-3 gene expressions and suppressing NF-κB mediated activation of bcl-2 in B16F-10 melanoma cells. Int. Immunopharmacol. 2008, 8, 974–981. [Google Scholar] [CrossRef]
- Pengyue, Z.; Tao, G.; Hongyun, H.; Liqiang, Y.; Yihao, D. Breviscapine confers a neuroprotective efficacy against transient focal cerebral ischemia by attenuating neuronal and astrocytic autophagy in the penumbra. Biomed. Pharmacother. 2017, 90, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Byun, H.S.; Zhou, W.; Park, I.; Kang, K.; Lee, S.-R.; Piao, X.; Park, J.B.; Kwon, T.K.; Na, M.; Hur, G.M. C-27-carboxylated oleanane triterpenoids up-regulate TRAIL DISC assembly via p38 MAPK and CHOP-mediated DR5 expression in human glioblastoma cells. Biochem. Pharmacol. 2018, 158, 243–260. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhao, C.; Jou, D.; Lü, J.; Zhang, C.; Lin, L.; Lin, J. Ursolic acid inhibits the growth of colon cancer-initiating cells by targeting STAT3. Anticancer. Res. 2013, 33, 4279–4284. [Google Scholar]
- Gomaa, A.A.; Abdel-Wadood, Y.A. The potential of glycyrrhizin and licorice extract in combating COVID-19 and associated conditions. Phytomedicine Plus 2021, 1, 100043. [Google Scholar] [CrossRef]
- Baltina, L.; Kondratenko, R. Glycyrrhizic Acid Derivatives as New Antiviral and Immune Modulating Agents. Curr. Bioact. Compd. 2021, 17, 41–58. [Google Scholar] [CrossRef]
- Kowalska, A.; Kalinowska-Lis, U. 18 β-Glycyrrhetinic acid: Its core biological properties and dermatological applications. Int. J. Cosmet. Sci. 2019, 41, 325–331. [Google Scholar] [CrossRef] [Green Version]
- Bordbar, N.; Karimi, M.H.; Amirghofran, Z. The effect of glycyrrhizin on maturation and T cell stimulating activity of dendritic cells. Cell. Immunol. 2012, 280, 44–49. [Google Scholar] [CrossRef]
- Shamsa, F.; Ohtsuki, K.; Hasanzadeh, E.; Rezazadeh, S. The anti-inflammatory and anti-viral effects of an ethnic medicine: Glycyrrhizin. J. Med. Plant Res. 2010, 9, 1–28. [Google Scholar]
- Wang, Y.M.; Du, G.Q. Glycyrrhizic acid prevents enteritis through reduction of NF-κB p65 and p38MAPK expression in rat. Mol. Med. Rep. 2016, 13, 3639–3646. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Shen, Y.; Qiu, R.; Chen, Z.; Chen, Z.; Chen, W. 18 β-glycyrrhetinic acid exhibits potent antitumor effects against colo-rectal cancer via inhibition of cell proliferation and migration. Int. J. Oncol. 2017, 51, 615–624. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Noshita, T.; Yu, T.; Kidachi, Y.; Kamiie, K.; Umetsu, H.; Ryoyama, K. Novel effects of glycyrrhetinic acid on the central nervous system tumorigenic progenitor cells: Induction of actin disruption and tumor cell-selective toxicity. Eur. J. Med. Chem. 2010, 45, 2943–2948. [Google Scholar] [CrossRef]
- Wang, X.-F.; Zhou, Q.-M.; Lu, Y.-Y.; Zhang, H.; Huang, S.; Su, S.-B. Glycyrrhetinic acid potently suppresses breast cancer invasion and metastasis by impairing the p38 MAPK-AP1 signaling axis. Expert Opin. Ther. Targets 2015, 19, 577–587. [Google Scholar] [CrossRef]
- Haghshenas, V.; Fakhari, S.; Mirzaie, S.; Rahmani, M.; Farhadifar, F.; Pirzadeh, S.; Jalili, A. Glycyrrhetinic Acid Inhibits Cell Growth and Induces Apoptosis in Ovarian Cancer A2780 Cells. Adv. Pharm. Bull. 2014, 4, 437–441. [Google Scholar] [CrossRef]
- Pirzadeh, S.; Fakhari, S.; Jalili, A.; Mirzai, S.; Ghaderi, B.; Haghshenas, V. Glycyrrhetinic acid induces apoptosis in leukemic HL60 cells through upregulating of CD95/CD178. Int. J. Mol. Cell. Med. 2014, 3, 272–278. [Google Scholar] [PubMed]
- Khalaf, H.S.; Naglah, A.M.; Al-Omar, M.A.; Moustafa, G.O.; Awad, H.M.; Bakheit, A.H. Synthesis, docking, computational studies, and antimicrobial evaluations of new dipeptide derivatives based on nicotinoylglycylglycine hydrazide. Molecules 2020, 25, 3589. [Google Scholar] [CrossRef] [PubMed]
- Moustafa, G.O. Synthesis of Dibenzofurans Possessing Anti-Allergy, Antioxidant, Anti-Inflammatory, Antimalarial and Treatment of Skin Conditions. Egypt. J. Chem. 2021, 64, 9–10. [Google Scholar] [CrossRef]
- Moustafa, G.O. Therapeutic Potentials of Cyclic Peptides as Promising Anticancer Drugs. Egypt. J. Chem. 2021. [Google Scholar] [CrossRef]
- Naglah, A.M.; Moustafa, G.O.; A Elhenawy, A.; Mounier, M.M.; El-Sayed, H.; A Al-Omar, M.; A Almehizia, A.; A Bhat, M. Nα-1, 3-Benzenedicarbonyl-Bis-(Amino Acid) and Dipeptide Candidates: Synthesis, Cytotoxic, Antimicrobial and Molecular Docking Investigation. Drug Des. Dev. Ther. 2021, 15, 1315–1332. [Google Scholar] [CrossRef]
- Hassan, A.S.; Askar, A.A.; Nossier, E.S.; Naglah, A.M.; Moustafa, G.O.; Al-Omar, M.A. Antibacterial Evaluation, In Silico Char-acters and Molecular Docking of Schiff Bases Derived from 5-aminopyrazoles. Molecules 2019, 24, 3130. [Google Scholar] [CrossRef] [Green Version]
- Bhattachary, B.; Mukherjee, S. Cancer Therapy Using Antibiotics. J. Cancer Res. Ther. 2015, 6, 849–858. [Google Scholar] [CrossRef] [Green Version]
- Tacar, O.; Sriamornsak, P.; Dass, C.R. Doxorubicin: An update on anticancer molecular action, toxicity and novel drug delivery systems. J. Pharm. Pharmacol. 2012, 65, 157–170. [Google Scholar] [CrossRef]
- Abd El-Meguid, E.A.; Moustafa, G.O.; Awad, H.M.; Zaki, E.R.; Nossier, E.S. Novel benzothiazole hybrids targeting EGFR: Design, synthesis, biological evaluation and molecular docking studies. J. Mol. Struct. 2021, 1240, 130595. [Google Scholar] [CrossRef]
- Amin, K.M.; Syam, Y.M.; Anwar, M.M.; Ali, H.I.; Abdel-Ghani, T.M.; Serry, A.M. Synthesis and molecular docking study of new benzofuran and furo[3,2-g]chromone-based cytotoxic agents against breast cancer and p38α MAP kinase inhibitors. Bioorg. Chem. 2018, 76, 487–500. [Google Scholar] [CrossRef]
- Mohamed, F.H.; Shalaby, A.M.; Soliman, H.A.; Abdelazem, A.Z.; Mounier, M.M.; Nossier, E.S.; Moustafa, G.O. Design, Synthesis and Molecular Docking Studies of Novel Cyclic Pentapeptides Based on Phthaloyl Chloride with Expected Anticancer Ac-tivity. Egypt. J. Chem. 2020, 63, 1723–1736. [Google Scholar] [CrossRef]
- Abo-Ghalia, M.H.; Moustafa, G.O.; Amr, A.E.; Naglah, A.M.; Elsayed, E.A.; Bakheit, A.H. Anticancer activities and 3D-QSAR studies of some new synthesized macrocyclic heptapeptide derivatives. Molecules 2020, 25, 1253. [Google Scholar] [CrossRef] [Green Version]
- Kalmouch, A.; Radwan, M.A.A.; Omran, M.M.; Sharaky, M.; Moustafa, G.O. Synthesis of novel 2, 3’-bipyrrole derivatives from chalcone and amino acids as antitumor agents. Egypt. J. Chem. 2020, 63, 4409–4421. [Google Scholar]
- Moustafa, G.O.; Al-Wasidi, A.S.; Naglah, A.M.; Refat, M.S. Isolation and Synthesis of Dibenzofuran Derivatives Possessing Anticancer Activities: A Review. Egypt. J. Chem. 2020, 63, 2355–2367. [Google Scholar] [CrossRef]
- A Elhenawy, A.; Al-Harbi, L.; O Moustafa, G.; A El-Gazzar, M.; Abdel-Rahman, R.F.; Salim, A.E. Synthesis, comparative docking, and pharmacological activity of naproxen amino acid derivatives as possible anti-inflammatory and analgesic agents. Drug Des. Dev. Ther. 2019, 13, 1773–1790. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hassan, A.S.; Moustafa, G.O.; Morsy, N.M.; Abdou, A.M.; Hafez, T.S. Design, synthesis and antibacterial activity of N-aryl-3-(arylamino)-5-(((5-substituted furan-2-yl)methylene)amino)-1H-pyrazole-4-carboxamide as Nitrofurantoin® ana-logues. Egypt. J. Chem. 2020, 63, 4469–4481. [Google Scholar] [CrossRef]
- Moustafa, G.; Al-Wasidi, A.S.; Naglah, A.; Kalmouch, A.; Adam, A.M.A.; Refat, M. Preparation of Cr2O3, MnO2, Fe2O3, NiO, CuO, and ZnO oxides using their glycine complexes as precursors for in situ thermal decomposition. Egypt. J. Chem. 2020, 63, 8–9. [Google Scholar] [CrossRef]
- Al-Wasidi, A.S.; Naglah, A.M.; Refat, M.S.; El-Megharbel, S.M.; Kalmouch, A.; Moustafa, G.O. Synthesis, spectroscopic charac-terization and antimicrobial studies of Mn(II), Co(II), Ni(II), Cr(III) and Fe(III) melatonin drug complexes. Egypt. J. Chem. 2020, 63, 1469–1481. [Google Scholar]
- Al-Wasidi, A.S.; Wafeek, M.; Abd El-Ghaffar, H.A.; Naglah, A.M.; Kalmouch, A.; Hamed, M.; Moustafa, G.O. Effect of Density on Growth Hormone and Some Physiological Parameters and its Relation to Growth Performance. Egypt. J. Chem. 2020, 63, 1575–1584. [Google Scholar]
- Wu, P.; Tu, B.; Liang, J.; Guo, S.; Cao, N.; Chen, S.; Luo, Z.; Li, J.; Zheng, W.; Tang, X.; et al. Synthesis and biological evaluation of pentacyclic triterpenoid derivatives as potential novel anti-bacterial agents. Bioorg. Chem. 2021, 109, 104692. [Google Scholar] [CrossRef]
- Long, D.R.; Mead, J.; Hendricks, J.M.; Hardy, M.E.; Voyich, J.M. 18β-Glycyrrhetinic Acid Inhibits Methicillin-Resistant Staphylococcus aureus Survival and Attenuates Virulence Gene Expression. Antimicrob. Agents Chemother. 2012, 57, 241–247. [Google Scholar] [CrossRef] [Green Version]
- Li, H.-E.; Qiu, J.-Z.; Yang, Z.-Q.; Dong, J.; Wang, J.-F.; Luo, M.-J.; Pan, J.; Dai, X.-H.; Zhang, Y.; Song, B.-L.; et al. Glycyrrhetinic acid protects mice from Staphylococcus aureus pneumonia. Fitoter. 2012, 83, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Oyama, K.; Kawada-Matsuo, M.; Oogai, Y.; Hayashi, T.; Nakamura, N.; Komatsuzawa, H. Antibacterial Effects of Glycyrrhetinic Acid and Its Derivatives on Staphylococcus aureus. PLoS ONE 2016, 11, e0165831. [Google Scholar] [CrossRef] [Green Version]
- Kassem, A.F.; Moustafa, G.O.; Nossier, E.; Khalaf, H.S.; Mounier, M.M.; Al-Yousef, S.A.; Mahmoud, S.Y. In vitro anticancer potentiality and molecular modelling study of novel amino acid derivatives based on N1,N3-bis-(1-hydrazinyl-1-oxopropan-2-yl) isophthalamide. J. Enzym. Inhib. Med. Chem. 2019, 34, 1247–1258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pfeffer, C.M.; Singh, A.T.K. Apoptosis: A Target for Anticancer Therapy. Int. J. Mol. Sci. 2018, 19, 448. [Google Scholar] [CrossRef] [Green Version]
- Zaman, S.; Wang, R.; Gandhi, V. Targeting the apoptosis pathway in hematologic malignancies. Leuk. Lymphoma 2014, 55, 1980–1992. [Google Scholar] [CrossRef]
- Lukin, D.J.; Carvajal, L.A.; Liu, W.-J.; Resnick-Silverman, L.; Manfredi, J.J. p53 Promotes Cell Survival due to the Reversibility of Its Cell-Cycle Checkpoints. Mol. Cancer Res. 2014, 13, 16–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feng, B.; Zhao, C.; Li, J.; Yu, J.; Zhang, Y.; Zhang, X.; Tian, T.; Zhao, L. The Novel Synthetic Triterpene Methyl 3β-O-[4-(2-Aminoethylamino)-4-oxo-butyryl]olean-12-ene-28-oate Inhibits Breast Tumor Cell Growth in Vitro and in Vivo. Chem. Pharm. Bull. 2020, 68, 962–970. [Google Scholar] [CrossRef]
- He, Y.; Sun, J.; Nan, M.; Wang, X.; Lin, H.; Lv, G.; Li, Y.; Duan, M.; Ye, H. Succinyl rotundic acid inhibits growth and promotes apoptosis in the HeLa cervical cancer cell line. Mater. Express 2020, 10, 571–577. [Google Scholar] [CrossRef]
- Hassan, A.S.; Moustafa, G.O.; Awad, H.M.; Nossier, E.S.; Mady, M.F. Design, Synthesis, Anticancer Evaluation, Enzymatic As-says, and a Molecular Modeling Study of Novel Pyrazole–Indole Hybrids. ACS Omega 2021, 6, 12361–12374. [Google Scholar] [CrossRef]
- Srour, A.M.; Ahmed, N.S.; El-Karim, S.S.A.; Anwar, M.M.; El-Hallouty, S.M. Design, synthesis, biological evaluation, QSAR analysis and molecular modelling of new thiazol-benzimidazoles as EGFR inhibitors. Bioorganic Med. Chem. 2020, 28, 115657. [Google Scholar] [CrossRef]
- Zhu, G.; Pan, C.; Bei, J.-X.; Li, B.; Liang, C.; Xu, Y.; Fu, X. Mutant p53 in Cancer Progression and Targeted Therapies. Front. Oncol. 2020, 10. [Google Scholar] [CrossRef]
- Ibrahim, H.S.; Eldehna, W.M.; Abdel-Aziz, H.A.; Elaasser, M.; Abdel-Aziz, M. Improvement of antibacterial activity of some sulfa drugs through linkage to certain phthalazin-1(2H)-one scaffolds. Eur. J. Med. Chem. 2014, 85, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Dürst, U.N.; Bruder, E.; Egloff, L.; Wüst, J.; Schneider, J.; O Hirzel, H. Micrococcus luteus: A rare pathogen of valve prosthesis endocarditis. Zeitschrift Kardiologie 1991, 80, 294–298. [Google Scholar]
- Nossier, E.S.; El-Hallouty, S.M.; Zaki, E.R. Synthesis, anticancer evaluation and molecular modeling of some substituted thiazol-idinonyl and thiazolyl pyrazole derivatives. Int. J. Pharm. Sci. 2015, 7, 353–359. [Google Scholar]
- Amr, A.E.-G.E.; Elsayed, E.A.; Al-Omar, M.A.; Eldin, H.B.; Nossier, E.S.; Abdallah, M.M. Design, Synthesis, Anticancer Evaluation and Molecular Modeling of Novel Estrogen Derivatives. Molecules 2019, 24, 416. [Google Scholar] [CrossRef] [Green Version]
- Nossier, E.; El-Karim, S.S.A.; Khalifa, N.M.; El-Sayed, A.S.; Hassan, E.S.I.; El-Hallouty, S.M. Kinase Inhibitory Activities and Molecular Docking of a Novel Series of Anticancer Pyrazole Derivatives. Molecules 2018, 23, 3074. [Google Scholar] [CrossRef] [Green Version]
- Feeney, B.; Pop, C.; Swartz, P.; Mattos, A.C.; Clark, A.C. Role of Loop Bundle Hydrogen Bonds in the Maturation and Activity of (Pro)caspase-3. Biochemistry 2006, 45, 13249–13263. [Google Scholar] [CrossRef] [Green Version]
- Souers, A.J.; Leverson, J.D.; Boghaert, E.R.; Ackler, S.L.; Catron, N.D.; Chen, J.; Dayton, B.D.; Ding, H.; Enschede, S.H.; Fairbrother, W.J.; et al. ABT-199, a potent and selective BCL-2 inhibitor, achieves antitumor activity while sparing platelets. Nat. Med. 2013, 19, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Barbareschi, M.; Caffo, O.; Veronese, S.M.; Leek, R.D.; Fina, P.; Fox, S.; Bonzanini, M.; Girlando, S.; Morelli, L.; Eccher, C.; et al. Bcl-2 and p53 expression in node-negative breast carcinoma: A study with long-term follow-up. Hum. Pathol. 1996, 27, 1149–1155. [Google Scholar] [CrossRef]
- Onur, R.; Semerciöz, A.; Orhan, I.; Yekeler, H. The effects of melatonin and the antioxidant defense system on apoptosis regulator proteins (Bax and Bcl-2) in experimentally induced varicocele. Urol. Res. 2004, 32, 204–208. [Google Scholar] [CrossRef]
- Thomas, M.D.; McIntosh, G.G.; Anderson, J.J.; McKenna, D.M.; Parr, A.H.; Johnstone, R.; Lennard, T.W.; Horne, C.H.; Angus, B. A novel quantitative immunoassay system for p53 using antibodies selected for optimum designation of p53 status. J. Clin. Pathol. 1997, 50, 143–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dawood, D.H.; Nossier, E.S.; Ali, M.M.; Mahmoud, A.E. Synthesis and molecular docking study of new pyrazole derivatives as potent anti-breast cancer agents targeting VEGFR-2 kinase. Bioorganic Chem. 2020, 101, 103916. [Google Scholar] [CrossRef]
- Denault, J.-B.; Salvesen, G.S. Human Caspase-7 Activity and Regulation by Its N-terminal Peptide. J. Biol. Chem. 2003, 278, 34042–34050. [Google Scholar] [CrossRef] [Green Version]
- Liliom, K.; Lehotzky, A.; Molnar, A.; Ovádi, J. Characterization of Tubulin-Alkaloid Interactions by Enzyme-Linked Immunosorbent Assay. Anal. Biochem. 1995, 228, 18–26. [Google Scholar] [CrossRef]
- Chakraborty, S.P.; Karmahapatra, S.; Sahu, S.K.; Pramanik, P.; Roy, S. Amelioratory Effect of Nanoconjugated Vancomycin on Spleen during VRSA-Induced Oxidative Stress. Pathol. Res. Int. 2011, 2011, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Burton, K.A. study of the conditions and mechanism of the diphenylamine reaction for the colorimetric estimation of deoxyri-bonucleic acid. Biochem. J. 1956, 62, 315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gonelimali, F.D.; Lin, J.; Miao, W.; Xuan, J.; Charles, F.; Chen, M.; Hatab, S.R. Antimicrobial Properties and Mechanism of Action of Some Plant Extracts Against Food Pathogens and Spoilage Microorganisms. Front. Microbiol. 2018, 9, 1639. [Google Scholar] [CrossRef] [PubMed]
- Zaidan, M.R.S.; Rain, A.N.; Badrul, A.R.; Adlin, A.; Norazah, A.; Zakiah, I. In vitro screening of five local medicinal plants for antibacterial activity using disc diffusion method. Trop. Biomed. 2005, 22, 165–170. [Google Scholar] [PubMed]


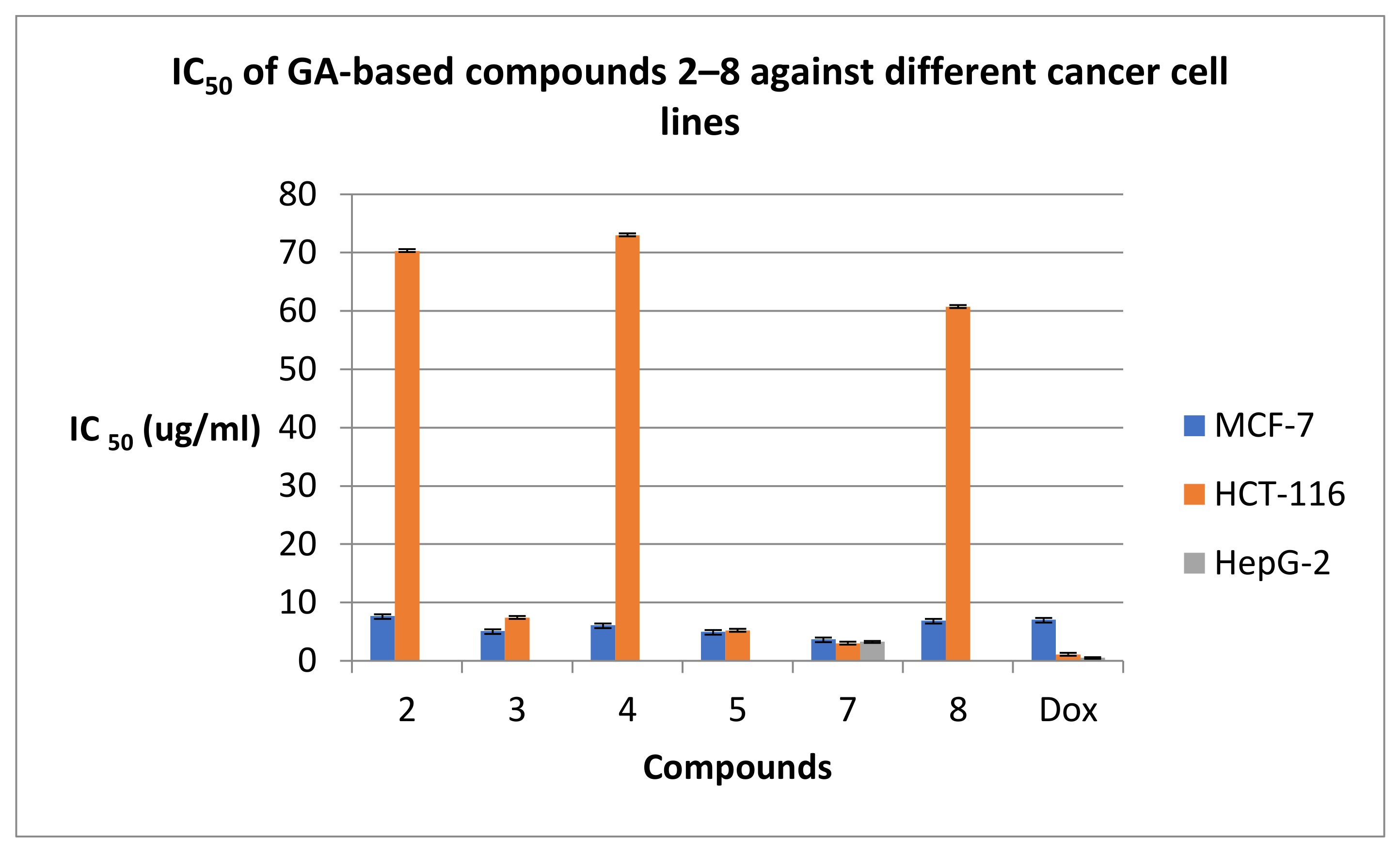

| Compd. No. | IC50 (µg/mL) ± SD | ||
|---|---|---|---|
| MCF-7 | HCT-116 | HepG-2 | |
| 2 | 7.70 ± 1.3 | 70.30 ± 0.9 | --- |
| 3 | 5.1 ± 0.7 | 7.40 ± 0.4 | --- |
| 4 | 6.10 ± 0.4 | 73.0 ± 1.4 | --- |
| 5 | 5.0 ± 0.3 | 5.2 ± 0.8 | --- |
| 7 | 3.70 ± 0.2 | 3.0 ± 1.1 | 3.30 ± 0.1 |
| 8 | 6.90 ± 1.1 | 60.70 ± 0.6 | --- |
| DOX | 7.07 ± 1 | 1.09 ± 3 | 0.54 ± 1 |
| Pathogenic Microbial Strains | Diameter of Inhibition Zone (mm) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 7 | 8 | GM | KC | ||
| Gram-positive bacteria | Streptococcus pneumonia RCMB 010010 | 15 ± 0.16 | 12 ± 0.82 | 17 ± 0.21 | 11 ± 0.24 | 16 ± 0.12 | - | 12 ± 0.22 | 27.2 ± 0.80 | |
| Staphylococcus aureus ATCC25923 | 15 ± 0.16 | 17 ± 0.11 | 17 ± 0.32 | 10 ± 0.13 | 15 ± 0.15 | - | 13 ± 0.24 | 25.4 ± 0.18 | ||
| Micrococcus Luteus | 30 ± 0.21 | 30 ± 0.13 | 30 ± 0.25 | 29 ± 0.13 | 30 ± 0.15 | 30 ± 0.11 | 30 ± 0.17 | 24.4 ± 0.72 | ||
| Gram-negative bacteria | Escherichia coli ATCC25922 | 20 ± 0.25 | 18 ± 0.14 | 16 ± 0.31 | 13 ± 0.15 | 18 ± 0.23 | 15 ± 0.21 | 15 ± 0.16 | 26.3 ± 0.15 | |
| Pseudomonas aeruginosa ATCC7853 | 18 ± 0.15 | 15 ± 0.25 | 15 ± 0.18 | 13 ± 0.16 | 17 ± 0.16 | 13 ± 0.14 | 14 ± 0.16 | 24.41 ± 0.18 | ||
| Proteus vulgaris RCMB 010085 | - | - | 17 ± 0.17 | 12 ± 0.23 | 10 ± 0.17 | 12 ± 0.14 | - | 20.0 ± 0.30 | ||
| Yeast | Candida albicans | - | - | 12 ± 0.23 | 15 ± 0.12 | 13 ± 0.17 | - | - | 22.8 ± 0.10 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moustafa, G.O.; Shalaby, A.; Naglah, A.M.; Mounier, M.M.; El-Sayed, H.; Anwar, M.M.; Nossier, E.S. Synthesis, Characterization, In Vitro Anticancer Potentiality, and Antimicrobial Activities of Novel Peptide–Glycyrrhetinic-Acid-Based Derivatives. Molecules 2021, 26, 4573. https://doi.org/10.3390/molecules26154573
Moustafa GO, Shalaby A, Naglah AM, Mounier MM, El-Sayed H, Anwar MM, Nossier ES. Synthesis, Characterization, In Vitro Anticancer Potentiality, and Antimicrobial Activities of Novel Peptide–Glycyrrhetinic-Acid-Based Derivatives. Molecules. 2021; 26(15):4573. https://doi.org/10.3390/molecules26154573
Chicago/Turabian StyleMoustafa, Gaber O., Ahmed Shalaby, Ahmed M. Naglah, Marwa M. Mounier, Heba El-Sayed, Manal M. Anwar, and Eman S. Nossier. 2021. "Synthesis, Characterization, In Vitro Anticancer Potentiality, and Antimicrobial Activities of Novel Peptide–Glycyrrhetinic-Acid-Based Derivatives" Molecules 26, no. 15: 4573. https://doi.org/10.3390/molecules26154573
APA StyleMoustafa, G. O., Shalaby, A., Naglah, A. M., Mounier, M. M., El-Sayed, H., Anwar, M. M., & Nossier, E. S. (2021). Synthesis, Characterization, In Vitro Anticancer Potentiality, and Antimicrobial Activities of Novel Peptide–Glycyrrhetinic-Acid-Based Derivatives. Molecules, 26(15), 4573. https://doi.org/10.3390/molecules26154573







