Photoinduced DNA Cleavage and Photocytotoxic of Phenanthroline-Based Ligand Ruthenium Compounds
Abstract
1. Introduction
2. Results and Discussion
2.1. Synthesis and Characterization
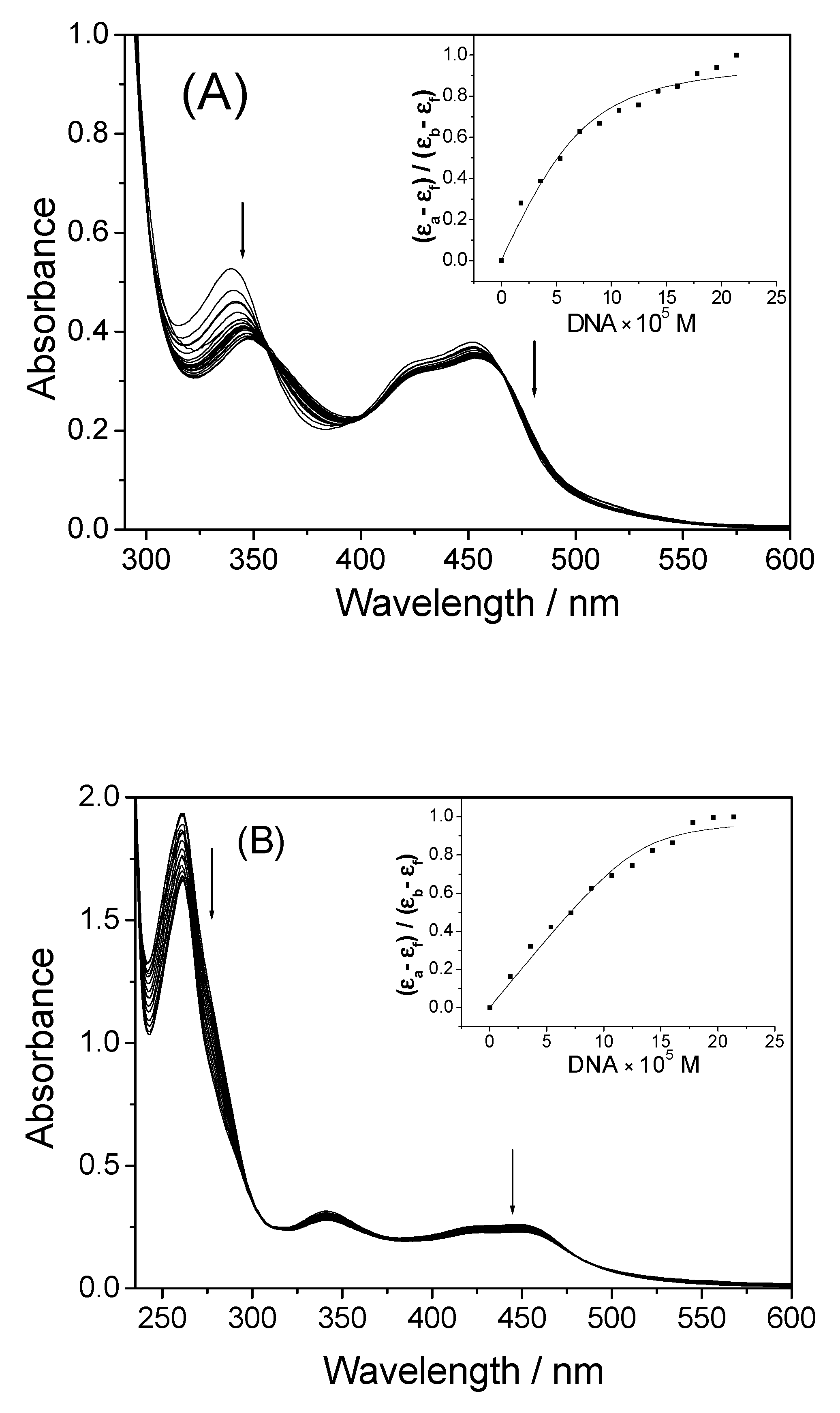
2.2. UV-Vis Spectra
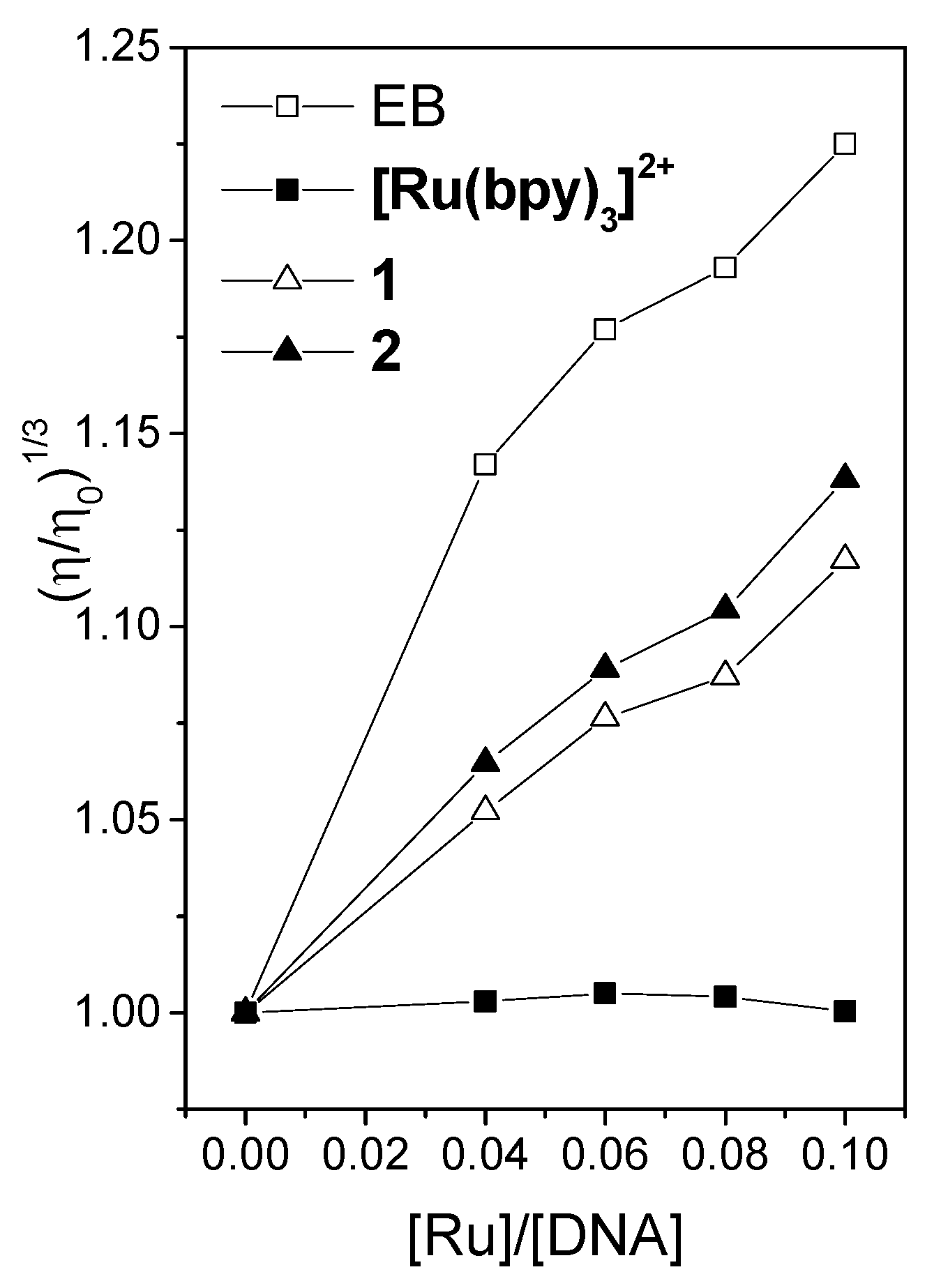
2.3. Viscosity Measurements
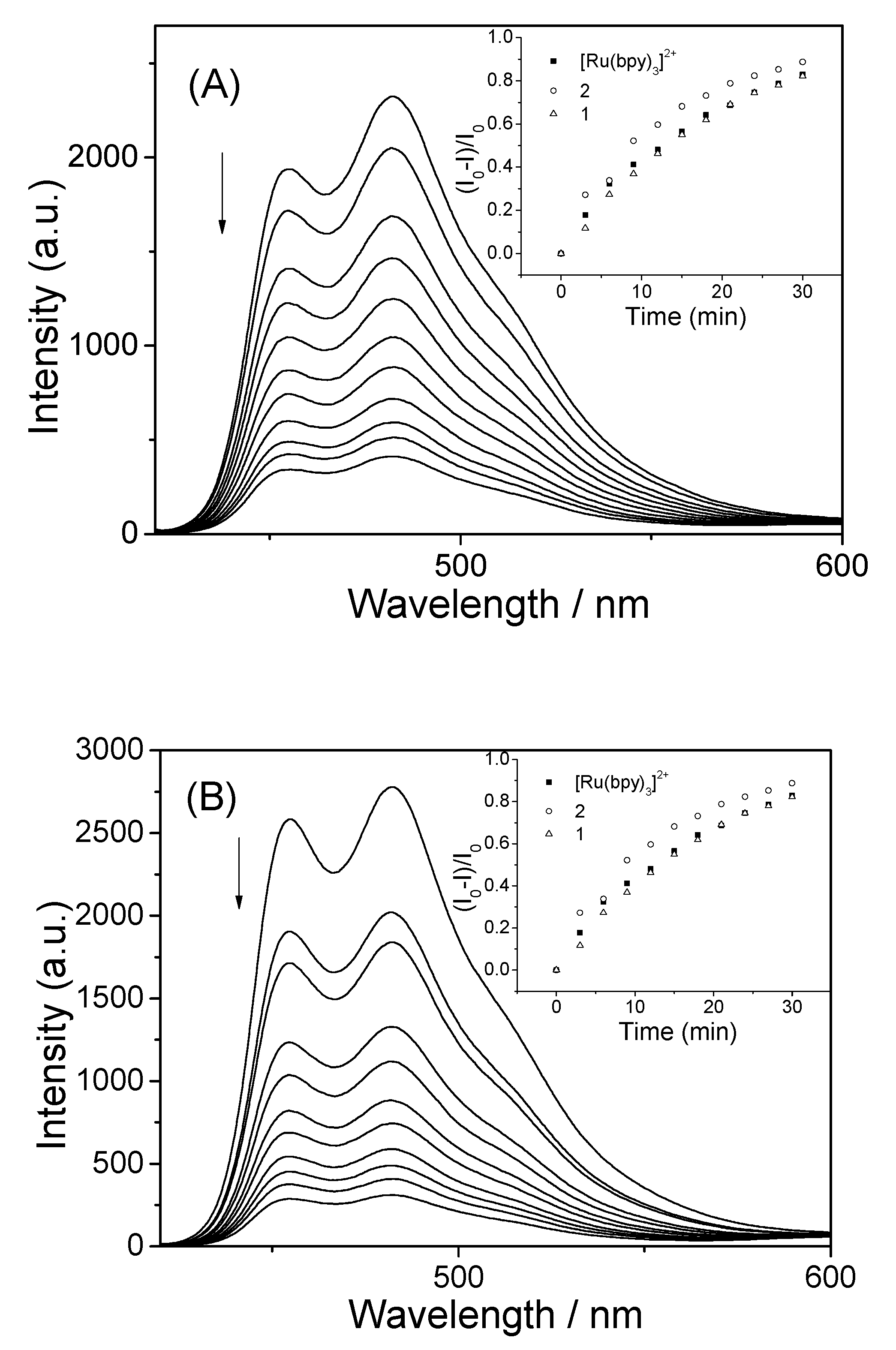
2.4. Emission Titration Experiments
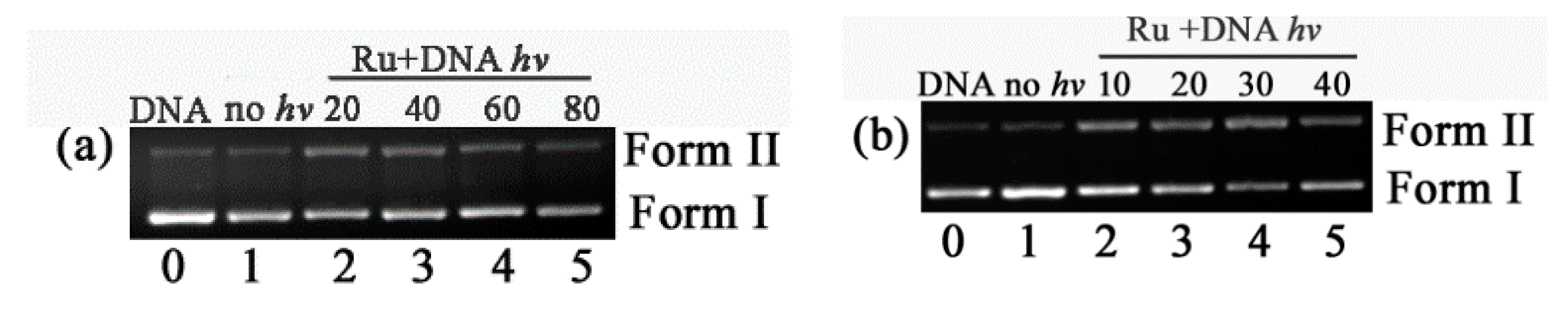
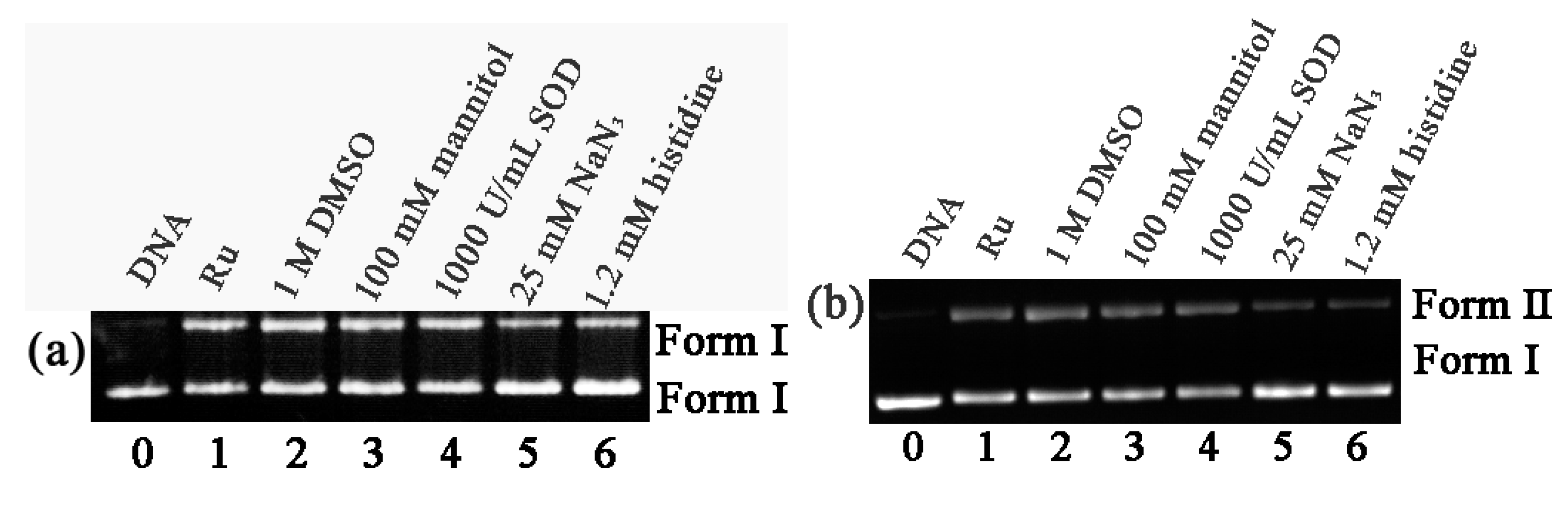
2.5. DNA Photocleavage Studies
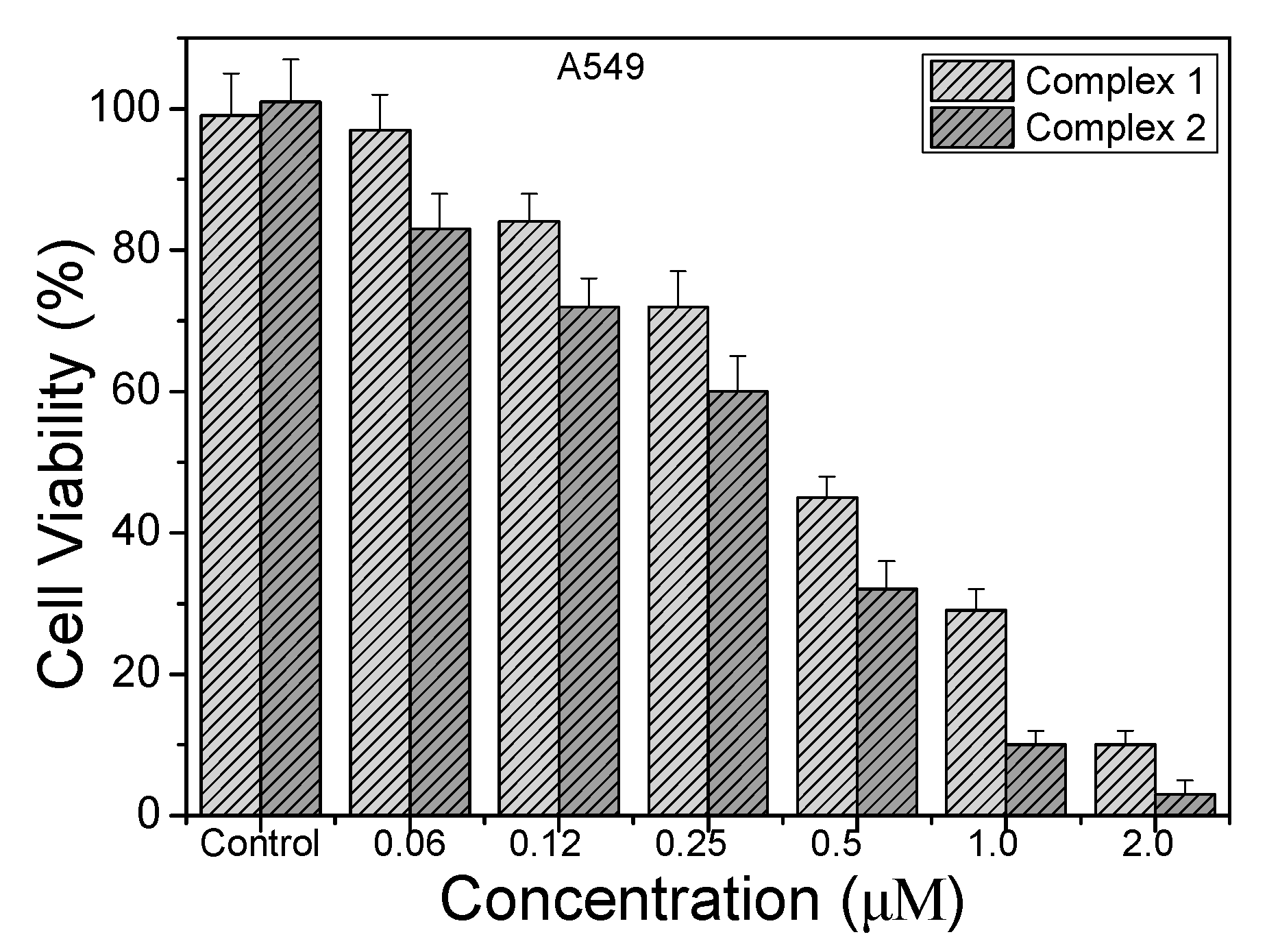
2.6. Photocytotoxicity
3. Materials and Methods
3.1. Instrumentation
3.2. DNA Interactions
3.3. Photoinduced DNA Cleavage
3.4. Singlet Oxygen Quantum Yield Measurement
3.5. Photocytotoxicity
3.6. Synthesis
3.6.1. 2-(5-(1,10-Phenanthroline))-1H-naphtha[2,3]imidazole (pni)
3.6.2. Synthesis of Complexes 1 and 2
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Zeng, L.L.; Gupta, P.; Chen, Y.; Wang, E.; Ji, L.N.; Chao, H.; Chen, Z.S. The development of anticancer ruthenium (II) complexes: From single molecule compounds to nanomaterials. Chem. Soc. Rev. 2017, 46, 5771–5804. [Google Scholar] [CrossRef]
- Furrer, J.; Süss-Fink, G. Thiolato-bridged dinuclear arene ruthenium complexes and their potential as anticancer drugs. Coord. Chem. Rev. 2016, 309, 36–50. [Google Scholar] [CrossRef]
- Li, F.; Collins, J.G.; Keene, F.R. Ruthenium complexes as antimicrobial agents. Chem. Soc. Rev. 2015, 44, 2529–2542. [Google Scholar] [CrossRef]
- Shi, G.; Monro, S.; Hennigar, R.; Colpitts, J.; Fong, J.; Kasimova, K.; Yin, H.; DeCoste, R.; Spencer, C.; Chamberlain, L. Ru(II) dyads derived from α-oligothiophenes: A new class of potent and versatile photosensitizers for PDT. Coord. Chem. Rev. 2015, 282, 127–138. [Google Scholar] [CrossRef]
- Liu, H.K.; Sadler, P.J. Metal complexes as DNA intercalators. Acc. Chem. Res. 2011, 44, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Genereux, J.C.; Barton, J.K. Mechanisms for DNA charge transport. Chem. Rev. 2010, 110, 1642–1662. [Google Scholar] [CrossRef] [PubMed]
- Keene, F.R.; Smith, J.A.; Collins, J.G. Metal complexes as structure-selective binding agents for nucleic acids. Coord. Chem. Rev. 2009, 253, 2021–2035. [Google Scholar] [CrossRef]
- Dervan, P.B. Molecular recognition of DNA by small molecules. Bioorg. Med. Chem. 2001, 9, 2215–2235. [Google Scholar] [CrossRef]
- Lippert, B. Cisplatin: Chemistry and Biochemistry of a Leading Anti-Cancer Drug; Wiley-VCH: Weinheim, Germany, 1999. [Google Scholar]
- Reedijk, J. Improved understanding in platinium antitumour chemistry. Chem. Commun. 1996, 1996, 801–806. [Google Scholar] [CrossRef]
- Guo, Z.J.; Sadler, P.J. Medicinal inorganic chemistry. Adv. Inorg. Chem. 2000, 49, 183–306. [Google Scholar]
- Roberts, J.D.; Peroutka, J.; Farrell, N. Cellular pharmacology of polynuclear platinum anti-cancer agents. J. Inorg. Biochem. 1999, 77, 51–57. [Google Scholar] [CrossRef]
- Martnez, R.; Chacn-Garca, L. The search of DNA-intercalators as. antitumoral drugs: What it worked and what did not work. Curr. Med. Chem. 2005, 12, 127–151. [Google Scholar] [CrossRef]
- Wan, D.; Tang, B.; Wang, Y.J.; Guo, B.H.; Yin, H.; Yi, Q.Y.; Liu, Y.J. Synthesis and anticancer properties of ruthenium (II) complexes as potent apoptosis inducers through mitochondrial disruption. Eur. J. Med. Chem. 2017, 139, 180–190. [Google Scholar] [CrossRef] [PubMed]
- Tang, B.; Shen, F.; Wan, D.; Guo, B.H.; Wang, Y.J.; Yi, Q.Y.; Liu, Y.J. DNA-binding; molecular docking studies and biological activity studies of ruthenium (II) polypyridyl complexes. RSC Adv. 2017, 405, 228–234. [Google Scholar] [CrossRef]
- Wan, D.; Lai, S.H.; Zeng, C.C.; Zhang, C.; Tang, B.; Liu, Y.J. Ruthenium (II) polypyridyl complexes: Synthesis, characterization and anticancer activity studies on BEL-7402 cells. J. Inorg. Biochem. 2017, 173, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Clark, M.J.E. Ruthenium and Other Metal Complexes in Cancer Chemotherapy; Springer: Berlin/Heidelberg, Germany, 1989. [Google Scholar]
- Fan, C.D.; Wu, Q.; Chen, T.F.; Zhang, Y.B.; Zheng, W.J.; Wang, Q.; Mei, W.J. Arene ruthenium (II) complexes induce S-phase arrest in MG-63 cells through stabilization of c-Myc G-quadruplex DNA. Med. Chem. Commun. 2014, 5, 597–602. [Google Scholar] [CrossRef]
- Fernández, R.; Melchart, M.; Habtemariam, A.; Parsons, S.; Sadler, P.J. Use of Chelating Ligands to Tune the Reactive Site of Half-Sandwich Ruthenium (II)–Arene Anticancer Complexes. Chem. Eur. J. 2004, 10, 5173–5179. [Google Scholar] [CrossRef] [PubMed]
- Levina, A.; Mitra, A.; Lay, P.A. Recent developments in ruthenium anticancer drugs. Metallomics 2009, 1, 458–470. [Google Scholar] [CrossRef]
- Gill, M.R.; Thomas, J.A. Ruthenium (II) polypyridyl complexes and DNA—From structural probes to cellular imaging and therapeutics. Chem. Soc. Rev. 2012, 41, 3179–3192. [Google Scholar] [CrossRef]
- Yao, J.L.; Gao, X.; Sun, W.L.; Fan, X.Z.; Shi, S.; Yao, T.M. A naked-eye on–off–on molecular “light switch” based on a reversible “conformational switch” of G-quadruplex DNA. Inorg. Chem. 2012, 51, 12591–12593. [Google Scholar] [CrossRef]
- Baggaley, E.; Gill, M.R.; Green, N.H.; Turton, D.; Sazanovich, I.V.; Botchway, S.W.; Smythe, C.; Haycock, J.W.; Weinstein, J.A.; Thomas, J.A. Dinuclear Ruthenium(II) Complexes as Two-Photon, Time-Resolved Emission Microscopy Probes for Cellular DNA. Angew. Chem. Int. Ed. Engl. 2014, 53, 3367–3371. [Google Scholar] [CrossRef]
- Gill, M.R.; Gracia-Lara, J.; Foster, S.J.; Smythe, C.; Battaglia, G.; Thomas, J.A. A ruthenium (II) polypyridyl complex for direct imaging of DNA structure in living cells. Nat. Chem. 2009, 1, 662–667. [Google Scholar] [CrossRef] [PubMed]
- Hartshorn, R.M.; Barton, J.K. Novel dipyridophenazine complexes of ruthenium (II): Exploring luminescent reporters of DNA. J. Am. Chem. Soc. 1992, 114, 5919–5925. [Google Scholar] [CrossRef]
- Ghosh, G.; Colon, K.L.; Fuller, A.; Sainuddin, T.; Bradner, E.; McCain, J.; Monro, S.M.A.; Yin, H.M.; Hetu, M.W.; Cameron, C.G.; et al. Cyclometalated Ruthenium(II) Complexes Derived from α-Oligothiophenes as Highly Selective Cytotoxic or Photocytotoxic Agents. Inorg. Chem. 2018, 57, 7694–7712. [Google Scholar]
- Silva-Caldeira, P.P.; Oliveira Junior, A.C.A.; Pereira-Maia, E.C. Photocytotoxic Activity of Ruthenium(II) Complexes with Phenanthroline-Hydrazone Ligands. Molecules 2021, 26, 2084–2095. [Google Scholar] [CrossRef]
- Rizvi, I.; Anbil, S.; Alagic, N.; Celli, J.P.; Zheng, L.Z.; Palanisami, A.; Glidden, M.D.; Pogue, B.W.; Hasan, T. PDT Dose Parameters Impact Tumoricidal Durability and Cell Death Pathways in a 3D Ovarian Cancer Model. Photochem. Photobiol. 2013, 89, 942–952. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Guan, R.; Zhang, C.; Huang, J.; Ji, L.N.; Chao, H. Two-photon luminescent metal complexes for bioimaging and cancer phototherapy. Coord. Chem. Rev. 2016, 310, 16–40. [Google Scholar] [CrossRef]
- Yu, H.J.; Liu, J.P.; Hao, Z.F.; He, J.; Sun, M.; Hu, S.; Yu, L.; Chao, H. Synthesis, characterization and biological evaluation of ruthenium(II) complexes [Ru(dtzp)(dppz)Cl]+ and [Ru(dtzp)(dppz)CH3CN]2+ for photodynamic therapy. Dyes Pigment. 2017, 136, 416–426. [Google Scholar] [CrossRef]
- Zhang, J.X.; Zhou, J.W.; Chan, C.F.; Lau, T.C.K.; Kwong, D.W.J.; Tam, H.L.; Mak, N.K.; Wong, K.L.; Wong, W.K. Comparative Studies of the Cellular Uptake, Subcellular Localization, and Cytotoxic and Phototoxic Antitumor Properties of Ruthenium(II)−Porphyrin Conjugates with Different Linkers. Bioconjug Chem. 2012, 23, 1623–1638. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.L.; Kuang, S.; Li, G.Y.; Jin, C.Z.; Ji, L.N.; Ji, C.H. A GSH-activatable ruthenium(II)-azo photosensitizer for twophoton photodynamic therapy. Chem. Commun. 2017, 53, 1977–1980. [Google Scholar] [CrossRef]
- Yip, A.M.H.; Lo, K.K.W. Luminescent rhenium(I), ruthenium(II), and iridium(III) polypyridine complexes containing a poly(ethylene glycol) pendant or bioorthogonal reaction group as biological probes and photocytotoxic agents. Coord. Chem. Rev. 2018, 361, 138–163. [Google Scholar] [CrossRef]
- Huang, H.Y.; Zhang, P.Y.; Yu, B.L.; Jin, C.Z.; Ji, L.N.; Chao, H. Synthesis, characterization and biological evaluation of mixed-ligand ruthenium(ii) complexes for photodynamic therapy. Dalton Trans. 2015, 44, 17335–17345. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.Y.; Yu, B.L.; Zhang, P.Y.; Huang, J.J.; Chen, Y.; Ji, L.N.; Chao, H. Chiral Highly Charged Ruthenium(II) Polypyridyl Complexes as Lysosome-Localized Photosensitizers for Two-Photon Photodynamic Therapy. Angew. Chem. Int. Ed. 2015, 54, 14049–14052. [Google Scholar] [CrossRef] [PubMed]
- Karges, J.; Kuang, S.; Maschietto, F.; Blacque, O.; Ciofini, I.; Chao, H.; Gasser, G. Rationally designed ruthenium complexes for 1-and 2-photon photodynamic therapy. Nat. Comm. 2020, 11, 3262. [Google Scholar] [CrossRef] [PubMed]
- Ellahioui, Y.; Patra, M.; Mari, C.; Kaabi, R.; Karges, J.; Gasser, G.; Gómez-Ruiz, S. Mesoporous silica nanoparticles functionalised with a photoactive ruthenium(ii) complex: Exploring the formulation of a metal-based photodynamic therapy photosensitiser. Dalton Trans. 2019, 48, 5940–5951. [Google Scholar] [CrossRef] [PubMed]
- Conti, L.; Bencini, A.; Ferrante, C.; Gellini, C.; Paoli, P. Highly charged ruthenium (II) polypyridyl complexes as effective photosensitizer in photodynamic therapy. Chem. Eur. J. 2019, 25, 10606–10615. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liao, X.; Xiong, K.; Kuang, S.; Jin, C.; Ji, L.N.; Chao, H. Boosting two-photon photodynamic therapy with mitochondria-targeting ruthenium–glucose conjugates. Chem. Commun. 2020, 56, 5839–5842. [Google Scholar] [CrossRef]
- McGhee, J.D.; von Hippel, P.H. Theoretical aspects of DNA-protein interactions: Co-operative and non-cooperative binding of large ligands to a one-dimensional homogeneous lattice. J. Mol. Biol. 1974, 86, 469–489. [Google Scholar] [CrossRef]
- Ambroise, A.; Maiya, B.G. Ruthenium (II) complexes of 6; 7-dicyanodipyridoquinoxaline: Synthesis; luminescence studies and DNA interaction. Inorg. Chem. 2000, 39, 4256–4263. [Google Scholar] [CrossRef]
- Liu, J.G.; Ye, B.H.; Li, H.; Ji, L.N.; Li, R.H.; Zhou, J.Y. Synthesis; characterization and DNA-binding properties of novel dipyridophenazine complex of ruthenium (II):[Ru(IP)2(DPPZ)]2+. J. Inorg. Biochem. 1999, 73, 117–122. [Google Scholar] [CrossRef]
- Ji, L.N.; Zou, X.H.; Liu, J.G. Shape-and enantioselective interaction of Ru (II)/Co (III) polypyridyl complexes with DNA. Coord. Chem. Rev. 2001, 216–217, 513–536. [Google Scholar] [CrossRef]
- Cohen, G.; Eisenberg, H. Viscosity and sedimentation study of sonicated DNA–proflavine complexes. Biopolymers 1969, 8, 45–55. [Google Scholar] [CrossRef]
- Satyanarayana, S.; Dabroniak, J.C.; Chaires, J.B. Neither. DELTA.-nor. LAMBDA.-tris (phenanthroline) ruthenium (II) binds to DNA by classical intercalation. Biochemistry 1992, 31, 9319–9324. [Google Scholar] [CrossRef] [PubMed]
- Satyanarayana, S.; Dabroniak, J.C.; Chaires, J.B. Tris(phenanthroline) ruthenium (II) enantiomer interactions with DNA: Mode and specificity of binding. Biochemistry 1993, 32, 2573–2584. [Google Scholar] [CrossRef] [PubMed]
- Burya, S.J.; Lutterman, D.A.; Turro, C. Absence of quenching by [Fe(CN)6]4− is not proof of DNA intercalation. Chem. Comm. 2011, 47, 1848–1850. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, F.R.; Merkel, P.B.; Kearns, D.R. Unambiguous evidence for the participation of singlet oxygen in photodynamic oxidation of amino acids. Photochem. Photobiol. 1972, 16, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Gao, F.; Yang, W.Y.; Zhou, Z.X.; Lin, J.Q.; Ji, L.N. StructureActivity Relationship of Polypyridyl Ruthenium(II) Complexes as DNA Intercalators, DNA Photocleavage Reagents, and DNA Topoisomerase and RNA Polymerase Inhibitors. Chem. Biodivers. 2013, 10, 367–384. [Google Scholar] [CrossRef]
- Tan, L.F.; Chao, H. DNA-binding and photocleavage studies of mixed polypyridyl ruthenium(II) complexes with calf thymus DNA. Inorg. Chim. Acta 2007, 360, 2016–2022. [Google Scholar] [CrossRef]
- Abdel-Shafi, A.A.; Beer, P.D.; Mortimer, R.J.; Wilkinson, F. Photosensitized Generation of Singlet Oxygen from Vinyl Linked Benzo-Crown-Ether−Bipyridyl Ruthenium(II) Complexes. J. Phys. Chem. A 2000, 104, 192–202. [Google Scholar] [CrossRef]
- Albani, B.A.; Peña, B.; Leed, N.A.; de Paula, N.A.B.G.; Pavani, C.; Baptista, M.S.; Dunbar, K.R.; Turro, C. Marked Improvement in Photoinduced Cell Death bya New Tris-Heteroleptic Complex with Dual Action: Singlet Oxygen Sensitization and Ligand Dissociation. J. Am. Chem. Soc. 2014, 136, 17095–17101. [Google Scholar] [CrossRef]
- Yu, H.J.; Huang, S.M.; Li, L.Y.; Jia, H.N.; Chao, H.; Mao, Z.W.; Liu, J.Z.; Ji, L.N. Synthesis, DNA-binding and photocleavage studies of ruthenium complexes [Ru(bpy)2(mitatp)]2+ and [Ru(bpy)2(nitatp)]2+. J. Inorg. Biochem. 2009, 103, 881–890. [Google Scholar] [CrossRef] [PubMed]
- Bolger, J.; Gourdon, A.; Ishow, E.; Launay, J.P. Mononuclear and Binuclear Tetrapyrido [3;2-a:2′;3′-c:3″; 2″-h:2‴; 3‴-j] phenazine (tpphz) Ruthenium and Osmium Complexes. Inorg. Chem. 1996, 35, 2937–2944. [Google Scholar] [CrossRef]
- Ishow, E.; Gourdon, A.; Launay, J.P. Observation of supramolecular π–π dimerization of a dinuclear ruthenium complex by 1H NMR and ESMS. Chem. Commun. 1998, 17, 1909–1910. [Google Scholar] [CrossRef]
- Zaid, A.; Sun, J.S.; Nguyen, C.H.; Bisagni, E.; Garestier, T.; Grierson, D.S.; Zain, R. Triple-Helix Directed Cleavage of Double-Stranded DNA by Benzoquinoquinoxaline-1; 10-phenanthroline Conjugates. ChemBioChem 2004, 5, 1550–1557. [Google Scholar] [CrossRef]
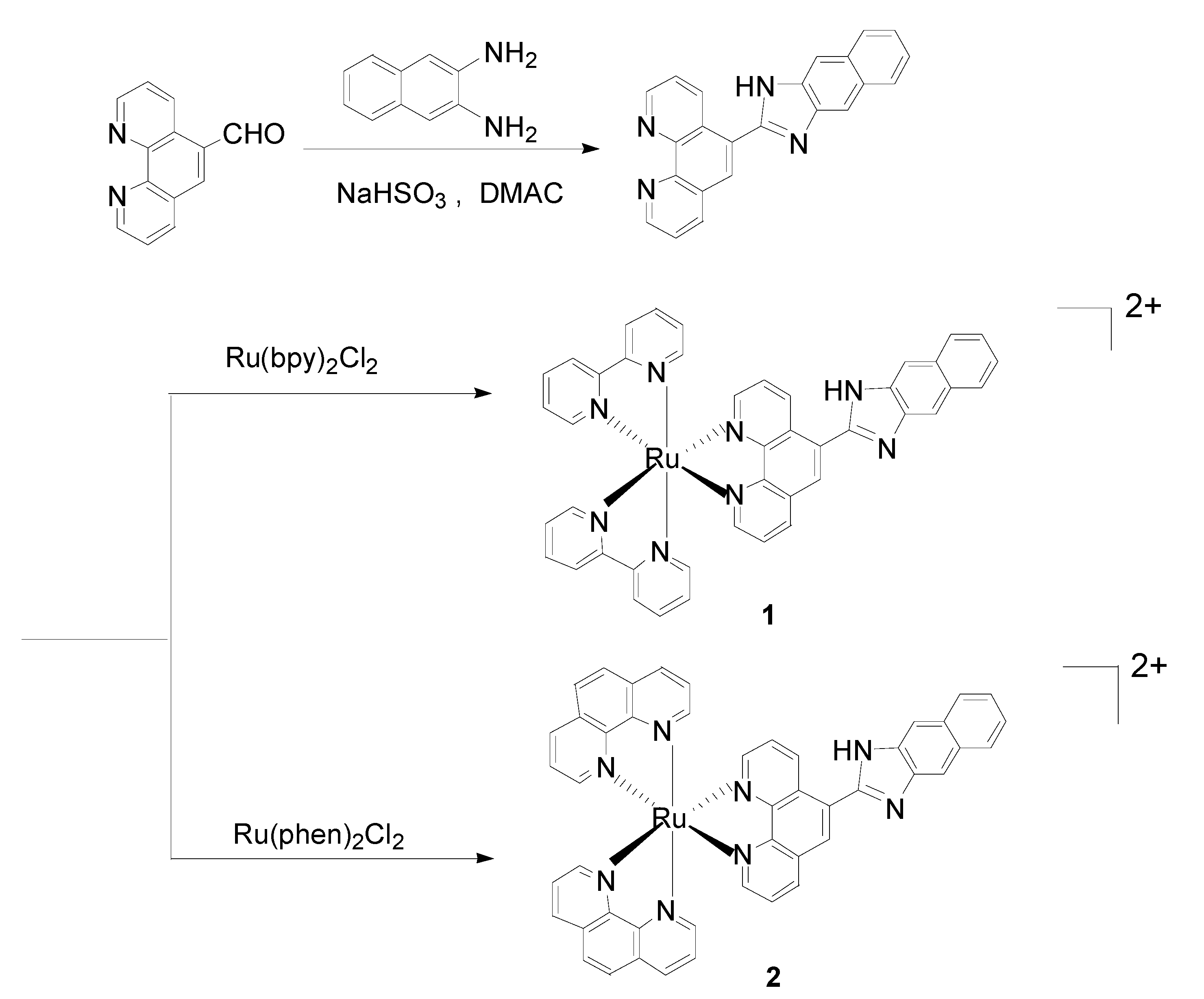


| Complex | IC50 (μM) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| HeLa | A549 | A375 | |||||||
| Dark | Light | PI | Dark | Light | PI | Dark | Light | PI | |
| 1 | >100 | 0.48 ± 0.13 | 208 | >100 | 0.44 ± 0.17 | 227 | >100 | 0.41 ± 0.13 | 244 |
| 2 | >100 | 0.25 ± 0.02 | 400 | >100 | 0.28 ± 0.05 | 357 | >100 | 0.26 ± 0.03 | 385 |
| [Ru(bpy)3]2+ [32] | >300 | 161 ± 5.62 | 1.86 | >300 | 152 ± 4.34 | 1.97 | |||
| [Ru(bpy)2dppn]2+ [52] | 110 ± 28 | 0.39 ± 0.06 | 282 | ||||||
| Cisplatin | 45.75 ± 3.31 | 43.61 ± 5.52 | 1.05 | 38.27 ± 2.46 | 33.81 ± 5.17 | 1.13 | 33.86 ± 3.82 | 30.63 ± 5.47 | 1.11 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, X.; Liu, N.-Y.; Deng, Y.-Q.; Wang, S.; Liu, T.; Liu, X.-W. Photoinduced DNA Cleavage and Photocytotoxic of Phenanthroline-Based Ligand Ruthenium Compounds. Molecules 2021, 26, 3471. https://doi.org/10.3390/molecules26113471
Hu X, Liu N-Y, Deng Y-Q, Wang S, Liu T, Liu X-W. Photoinduced DNA Cleavage and Photocytotoxic of Phenanthroline-Based Ligand Ruthenium Compounds. Molecules. 2021; 26(11):3471. https://doi.org/10.3390/molecules26113471
Chicago/Turabian StyleHu, Xia, Ning-Yi Liu, Yuan-Qing Deng, Shan Wang, Ting Liu, and Xue-Wen Liu. 2021. "Photoinduced DNA Cleavage and Photocytotoxic of Phenanthroline-Based Ligand Ruthenium Compounds" Molecules 26, no. 11: 3471. https://doi.org/10.3390/molecules26113471
APA StyleHu, X., Liu, N.-Y., Deng, Y.-Q., Wang, S., Liu, T., & Liu, X.-W. (2021). Photoinduced DNA Cleavage and Photocytotoxic of Phenanthroline-Based Ligand Ruthenium Compounds. Molecules, 26(11), 3471. https://doi.org/10.3390/molecules26113471






