Thermodynamics Analysis and Removal of P in a P-(M)-H2O System
Abstract
:1. Introduction
2. Results
2.1. Thermodynamic Analysis of the P-H2O System
2.2. Thermodynamic Analysis of M-P-H2O System
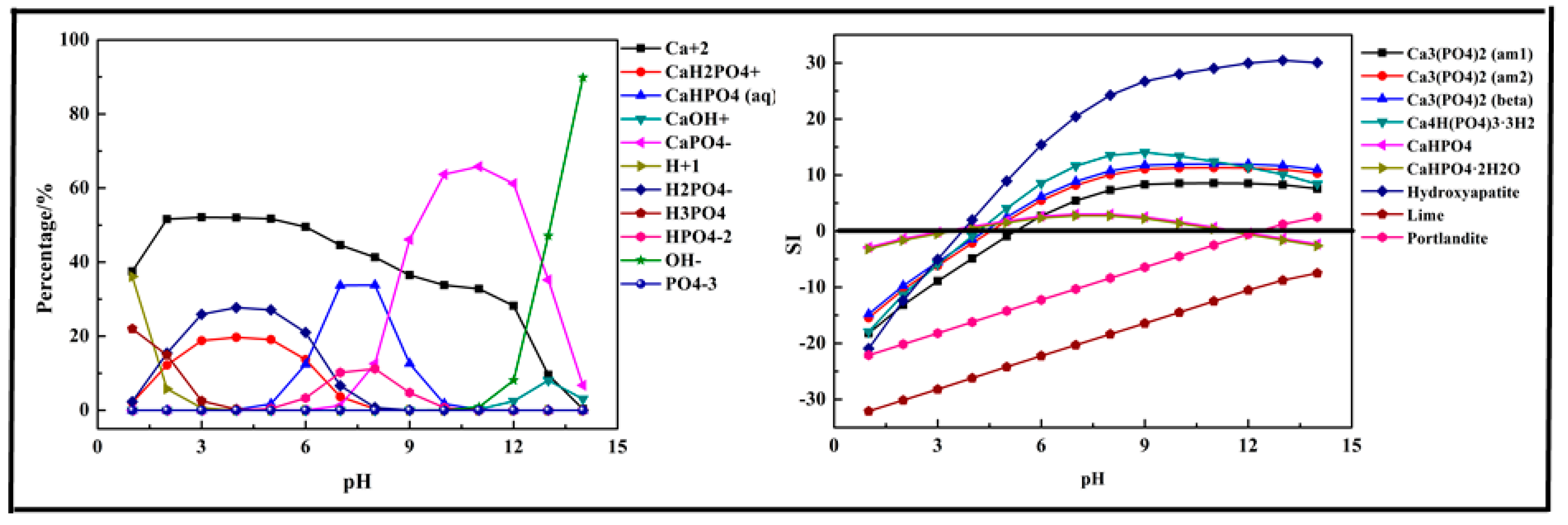
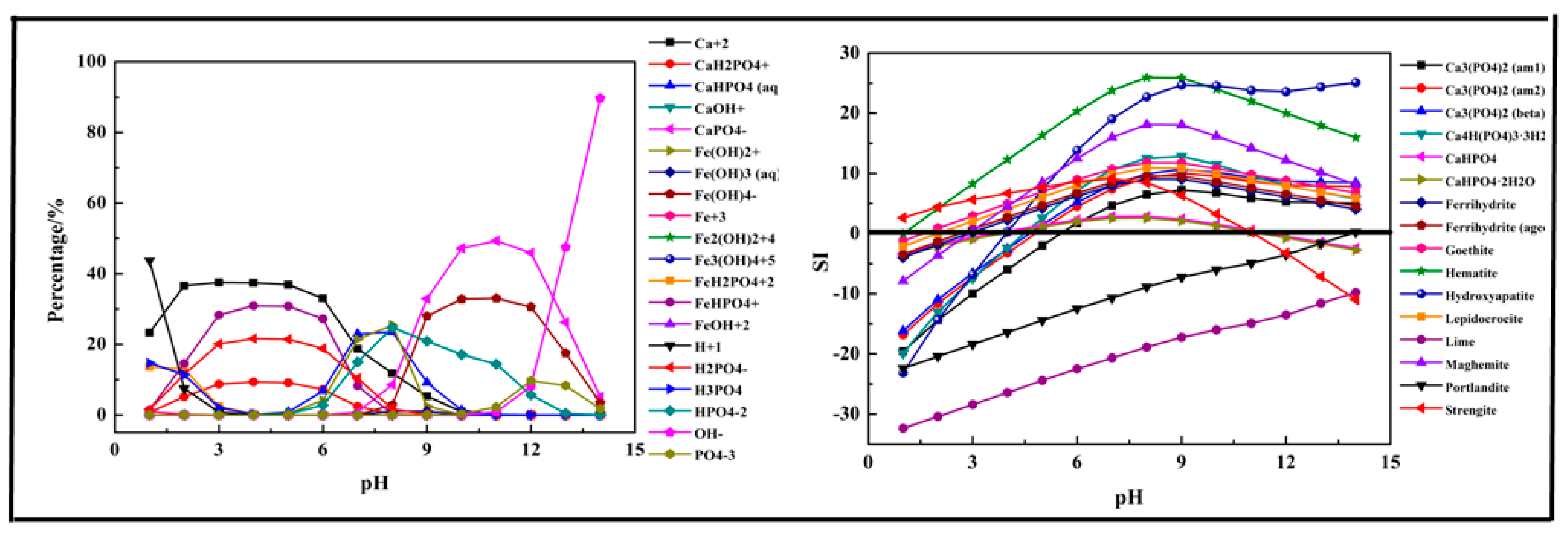
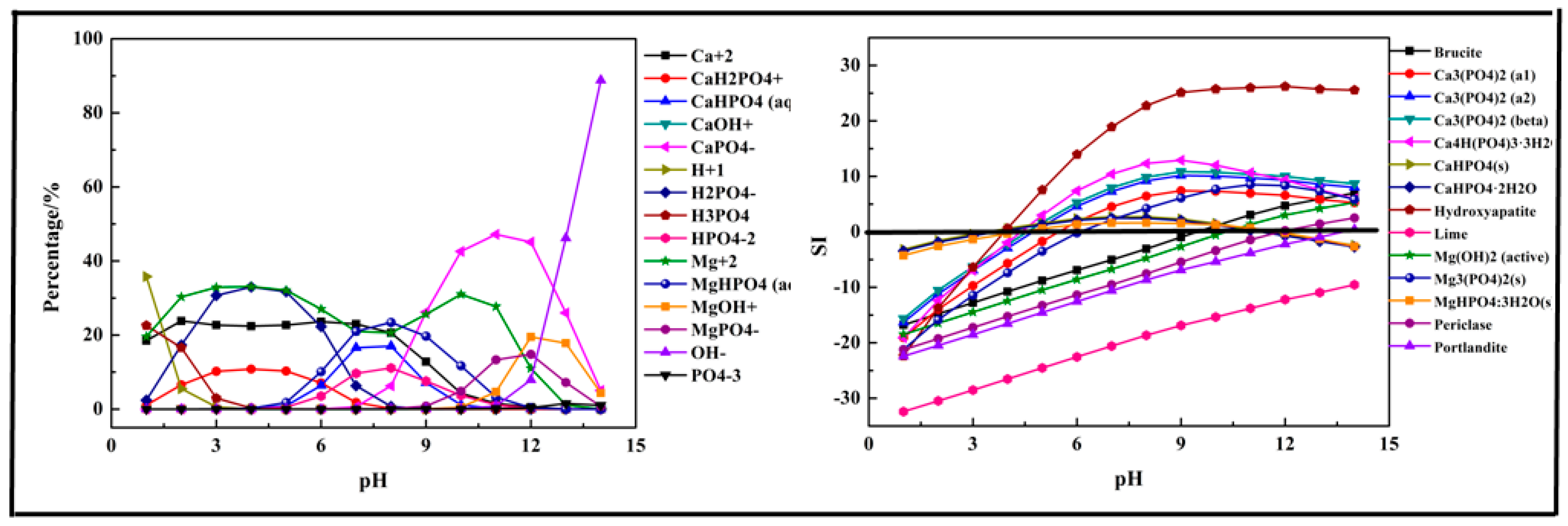
2.2.1. Fe (Ca, Mg)-P-H2O System
2.2.2. Two Salts System
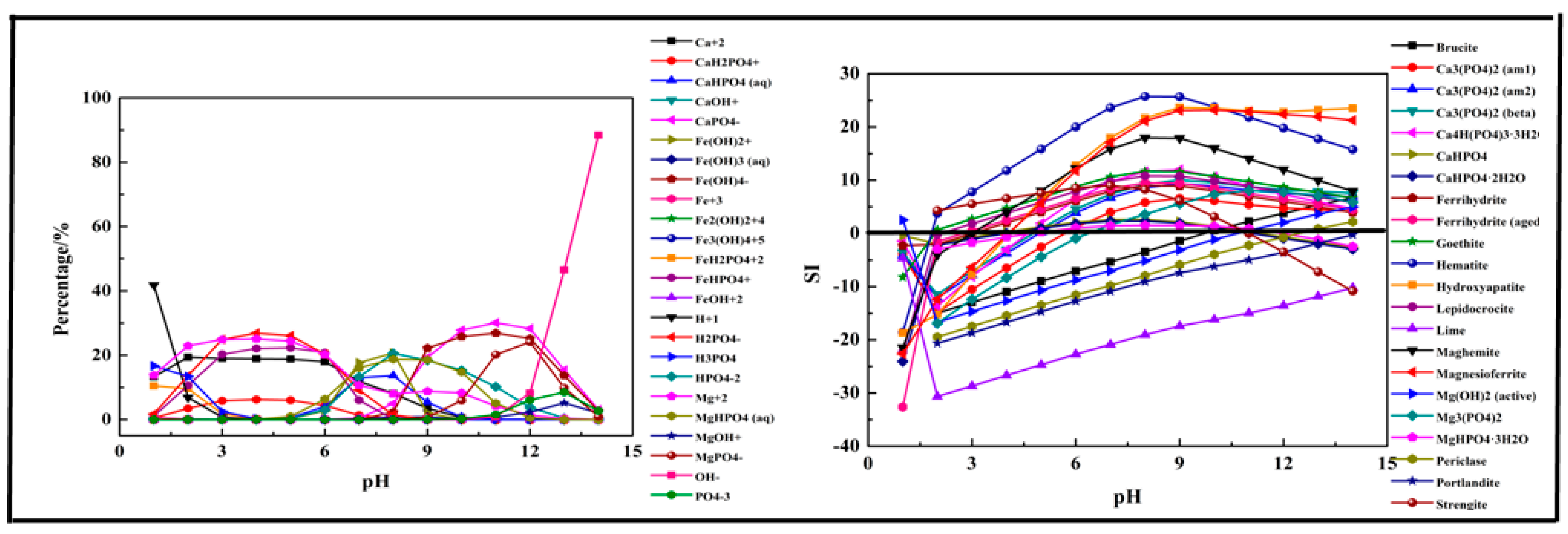
2.2.3. Fe-Ca-Mg-P-H2O System
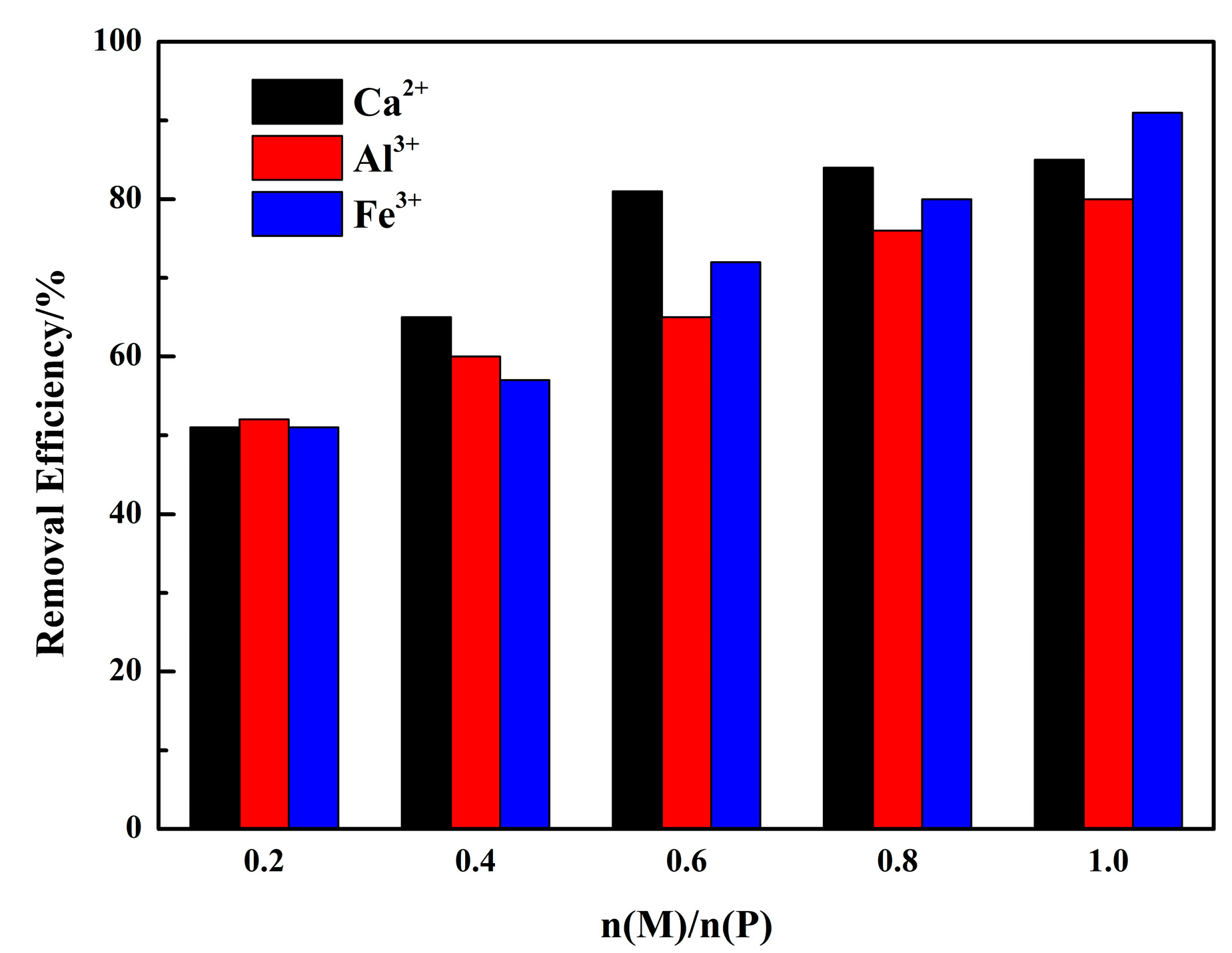
2.3. Phosphorous Removal Experiments
3. Materials and Methods
4. Conclusions
- (1)
- The phosphorus ions existed in the form of H3PO4, H2PO4−, HPO42−, and PO43−. Among them, H2PO4− and HPO42− were the main species in the acidic medium (99% at pH = 5) and alkaline medium (97.9% at pH = 10). In the P-Fe-H2O System ((P) = 0.01 mol/L, (Fe3+) = 0.01 mol/L), H2PO4− was transformed to FeHPO4+ at pH = 0–7 due to the existence of Fe3+ and then transformed into HPO42− at pH > 6 as the Fe3+ was mostly precipitated. In the P-Ca-H2O System ((P) = 0.01 mol/L, (Ca2+) = 0.015 mol/L), the main species in the acidic medium were CaH2PO4+ and HPO42−, and then transformed into CaPO4− at pH > 7. In the P-Mg-H2O System ((P) = 0.01 mol/L, (Mg3+) = 0.015 mol/L), the main species in the acidic medium was H2PO4− and then transformed into MgHPO4 at pH = 5–10, and finally transformed into MgPO4− as pH increased.
- (2)
- The phosphorus was more easily precipitated in the P-Fe-H2O system than the P-Ca-H2O system and P-Mg-H2O system. The suitable pH of the solution for phosphorus precipitation was about 5–10 in all precipitation systems.
- (3)
- The verification experiments (precipitation experiments) with single metal ions confirm that the theoretical analysis can be used to guide the actual experiments.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Okano, K.; Yamamoto, Y.; Takano, H.; Aketo, T.; Honda, K.; Ohtake, H. A simple technology for phosphorus recovery using acid-treated concrete sludge. Sep. Purif. Technol. 2016, 165, 173–178. [Google Scholar] [CrossRef]
- Tran, N.; Drogui, P.; Blais, J.-F.; Mercier, G. Phosphorus removal from spiked municipal wastewater using either electrochemical coagulation or chemical coagulation as tertiary treatment. Sep. Purif. Technol. 2012, 95, 16–25. [Google Scholar] [CrossRef]
- Okano, K.; Uemoto, M.; Kagami, J.; Miura, K.; Aketo, T.; Toda, M.; Honda, K.; Ohtake, H. Novel technique for phosphorus recovery from aqueous solutions using amorphous calcium silicate hydrates (A-CSHs). Water Res. 2013, 47, 2251–2259. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Weidler, P.G.; Berg, U.; Nüesch, R.; Donnert, D. Calcite-seeded crystallization of calcium phosphate for phosphorus recovery. Chemosphere 2006, 63, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Hosni, K.; Ben Moussa, S.; Ben Amor, M. Conditions influencing the removal of phosphate from synthetic wastewater: Influence of the ionic composition. Desalination 2007, 206, 279–285. [Google Scholar] [CrossRef]
- Nguyen, D.D.; Ngo, H.H.; Guo, W.; Nguyen, T.T.; Chang, S.W.; Jang, A.; Yoon, Y.S. Can electrocoagulation process be an appropriate technology for phosphorus removal from municipal wastewater? Sci. Total Environ. 2016, 563–564, 549–556. [Google Scholar] [CrossRef]
- Asselin, M.; Drogui, P.; Benmoussa, H.; Blais, J.-F. Effectiveness of electrocoagulation process in removing organic compounds from slaughterhouse wastewater using monopolar and bipolar electrolytic cells. Chemosphere 2008, 72, 1727–1733. [Google Scholar] [CrossRef]
- Gatsios, E.; Hahladakis, J.N.; Gidarakos, E. Optimization of electrocoagulation (EC) process for the purification of a real industrial wastewater from toxic metals. J. Environ. Manag. 2015, 154, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Wahab, M.A.; Hassine, R.B.; Jellali, S. Removal of phosphorus from aqueous solution by Posidonia oceanica fibers using continuous stirring tank reactor. J. Hazard. Mater. 2011, 189, 577–585. [Google Scholar] [CrossRef]
- Nguyen, D.D.; Ngo, H.H.; Yoon, Y.S. A new hybrid treatment system of bioreactors and electrocoagulation for superior removal of organic and nutrient pollutants from municipal wastewater. Bioresour. Technol. 2014, 153, 116–125. [Google Scholar] [CrossRef]
- Mandel, A.; Zekker, I.; Jaagura, M.; Tenno, T. Enhancement of anoxic phosphorus uptake of denitrifying phosphorus removal process by biomass adaption. Int. J. Environ. Sci. Technol. 2019, 16, 5965–5978. [Google Scholar] [CrossRef]
- Torresi, E.; Tang, K.; Deng, J.; Sund, C.; Smets, B.F.; Christensson, M.; Andersen, H.R. Removal of micropollutants during biological phosphorus removal: Impact of redox conditions in MBBR. Sci. Total Environ. 2019, 663, 496–506. [Google Scholar] [CrossRef]
- Lin, Z.; Wang, Y.; Huang, W.; Wang, J.; Chen, L.; Zhou, J.; He, Q. Single-stage denitrifying phosphorus removal biofilter utilizing intracellular carbon source for advanced nutrient removal and phosphorus recovery. Bioresour. Technol. 2019, 277, 27–36. [Google Scholar] [CrossRef]
- Ge, Y.; Lan, J.; Zhan, C.; Zhou, Y.; Ma, C.; Zhao, L. Biological removal of phosphorus and diversity analysis of microbial community in the enhanced biological phosphorus removal (EBPR) system. Water Environ. J. 2019, 34, 563–574. [Google Scholar] [CrossRef]
- Zou, H.; Wang, Y. Phosphorus removal and recovery from domestic wastewater in a novel process of enhanced biological phosphorus removal coupled with crystallization. Bioresour. Technol. 2016, 211, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Qin, Z.; Shober, A.L.; Scheckel, K.G.; Penn, C.J.; Turner, K.C. Mechanisms of Phosphorus Removal by Phosphorus Sorbing Materials. J. Environ. Qual. 2018, 47, 1232–1241. [Google Scholar] [CrossRef]
- Shi, J.; Yin, D.; Xu, Z.; Song, D.; Cao, F. Fosfomycin removal and phosphorus recovery in a schorl/H2O2 system. RSC Adv. 2016, 6, 68185–68192. [Google Scholar] [CrossRef]
- Zhang, M.; Qiao, S.; Shao, D.; Jin, R.; Zhou, J. Simultaneous nitrogen and phosphorus removal by combined anammox and denitrifying phosphorus removal process. J. Chem. Technol. Biotechnol. 2018, 93, 94–104. [Google Scholar] [CrossRef]
- Shimpo, T.; Yoshikawa, T.; Morita, K. Thermodynamic study of the effect of calcium on removal of phosphorus from silicon by acid leaching treatment. Metall. Mater. Trans. B 2004, 35, 277–284. [Google Scholar] [CrossRef]
- Zhu, M.; Azarov, A.; Monakhov, E.; Tang, K.; Safarian, J. Phosphorus separation from metallurgical-grade silicon by magnesium alloying and acid leaching. Sep. Purif. Technol. 2020, 240, 116614. [Google Scholar] [CrossRef]
- Shu, J.; Wu, H.; Chen, M.; Peng, H.; Li, B.; Liu, R.; Liu, Z.; Wang, B.; Huang, T.; Hu, Z. Fractional removal of manganese and ammonia nitrogen from electrolytic metal manganese residue leachate using carbonate and struvite precipitation. Water Res. 2019, 153, 229–238. [Google Scholar] [CrossRef]
- Shu, J.; Liu, R.; Liu, Z.; Chen, H.; Tao, C. Simultaneous removal of ammonia and manganese from electrolytic metal manganese residue leachate using phosphate salt. J. Clean. Prod. 2016, 135, 468–475. [Google Scholar] [CrossRef]
- Wang, R.; Shu, J.; Chen, M.; Wang, R.; He, D.; Wang, J.; Tang, C.; Han, Y.; Luo, Z. An innovative method for fractionally removing high concentrations of Ni2+, PO43−, TP, COD, and NH4+-N from printed-circuit-board nickel plating wastewater. Sep. Purif. Technol. 2021, 260, 118241. [Google Scholar] [CrossRef]
- Shu, J.; Chen, M.; Wu, H.; Li, B.; Wang, B.; Li, B.; Liu, R.; Liu, Z. An innovative method for synergistic stabilization/solidification of Mn2+, NH4+-N, PO43− and F− in electrolytic manganese residue and phosphogypsum. J. Hazard. Mater. 2019, 376, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Hu, Y.; Huang, D.; Xiong, T.; Li, M.; Balogun, M.S.; Tong, Y. Efficient hydrogen and oxygen evolution electrocatalysis by cobalt and phosphorus dual-doped vanadium nitride nanowires. Mater. Today Chem. 2019, 11, 1–7. [Google Scholar] [CrossRef]
- Xiong, P.; Zhang, Y.; Bao, S.; Huang, J. Precipitation of vanadium using ammonium salt in alkaline and acidic media and the effect of sodium and phosphorus. Hydrometallurgy 2018, 180, 113–120. [Google Scholar] [CrossRef]
- Yang, L.; Liang, X.; Han, Y.; Cai, Y.; Zhao, H.; Sheng, M.; Cao, G. The coupling use of advanced oxidation processes and sequencing batch reactor to reduce nitrification inhibition of industry wastewater: Characterization and optimization. Chem. Eng. J. 2019, 360, 1577–1586. [Google Scholar] [CrossRef]
- Liu, B.; Liu, Z.; Yu, P.; Pan, S.; Xu, Y.; Sun, Y.; Pan, S.-Y.; Yu, Y.; Zheng, H. Enhanced removal of tris(2-chloroethyl) phosphate using a resin-based nanocomposite hydrated iron oxide through a Fenton-like process: Capacity evaluation and pathways. Water Res. 2020, 175, 115655. [Google Scholar] [CrossRef] [PubMed]




| System | Concentration (mol/L) | |||
|---|---|---|---|---|
| P | Fe | Ca | Mg | |
| P-H2O | 0.01–0.09 | |||
| Fe-P-H2O | 0.01 | 0.01 | ||
| Ca-P-H2O | 0.01 | 0.015 | ||
| Mg-P-H2O | 0.01 | 0.015 | ||
| Fe-Mg-P-H2O | 0.01 | 0.005 | 0.0075 | |
| Ca-Mg-P-H2O | 0.01 | 0.0075 | 0.0075 | |
| Fe-Ca-P-H2O | 0.01 | 0.005 | 0.0075 | |
| Fe-Ca-Mg-P-H2O | 0.01 | 0.004 | 0.0045 | 0.0045 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, H.; Guo, J.; Qiu, H.; Wang, C.; Zhang, C.; Hao, Z.; Rao, Y.; Gong, Y. Thermodynamics Analysis and Removal of P in a P-(M)-H2O System. Molecules 2021, 26, 3342. https://doi.org/10.3390/molecules26113342
Peng H, Guo J, Qiu H, Wang C, Zhang C, Hao Z, Rao Y, Gong Y. Thermodynamics Analysis and Removal of P in a P-(M)-H2O System. Molecules. 2021; 26(11):3342. https://doi.org/10.3390/molecules26113342
Chicago/Turabian StylePeng, Hao, Jing Guo, Hongzhi Qiu, Caiqiong Wang, Chenyu Zhang, Zhihui Hao, Yating Rao, and Yanhong Gong. 2021. "Thermodynamics Analysis and Removal of P in a P-(M)-H2O System" Molecules 26, no. 11: 3342. https://doi.org/10.3390/molecules26113342
APA StylePeng, H., Guo, J., Qiu, H., Wang, C., Zhang, C., Hao, Z., Rao, Y., & Gong, Y. (2021). Thermodynamics Analysis and Removal of P in a P-(M)-H2O System. Molecules, 26(11), 3342. https://doi.org/10.3390/molecules26113342







