Perfume and Flavor Engineering: A Chemical Engineering Perspective
Abstract
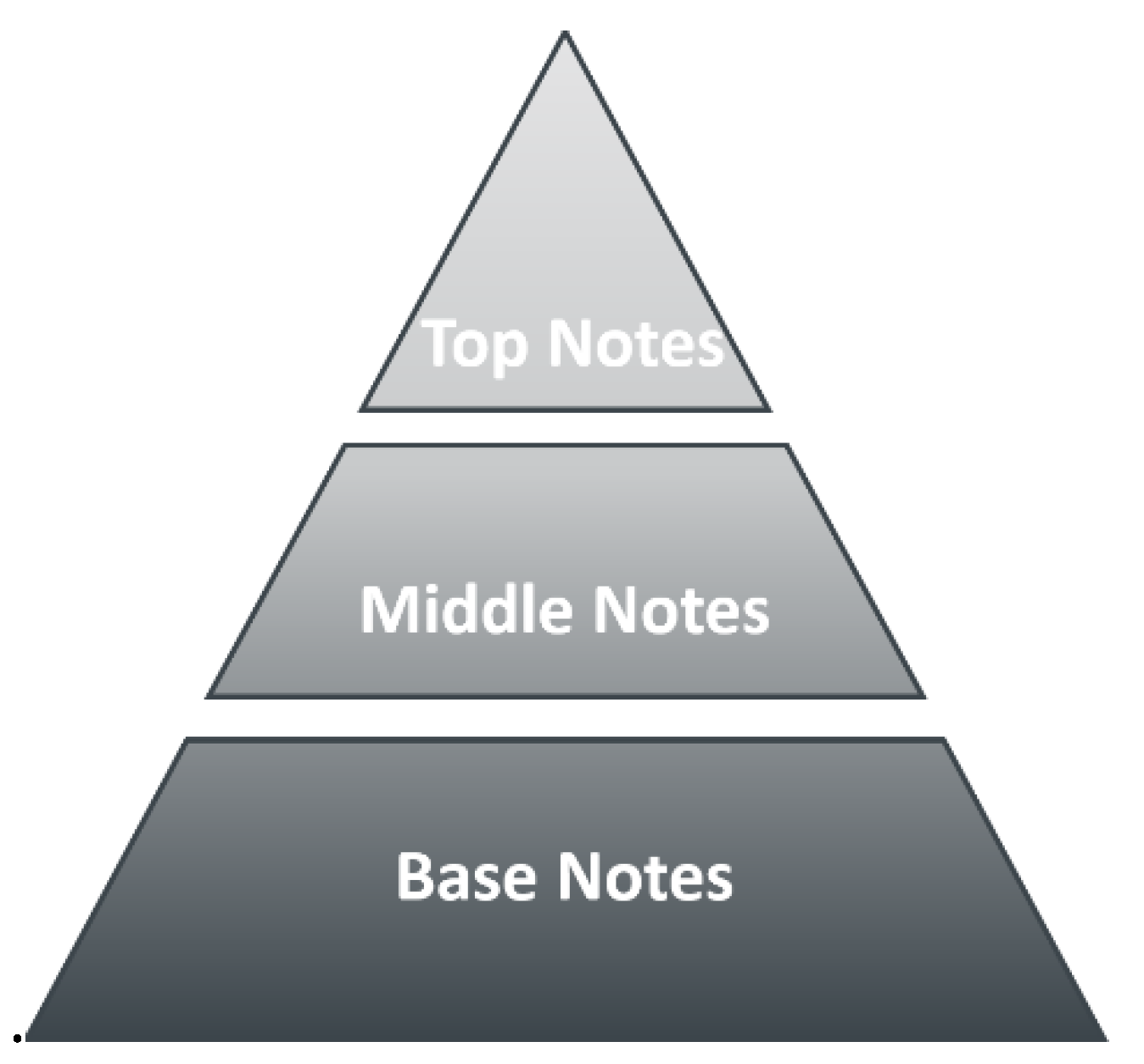
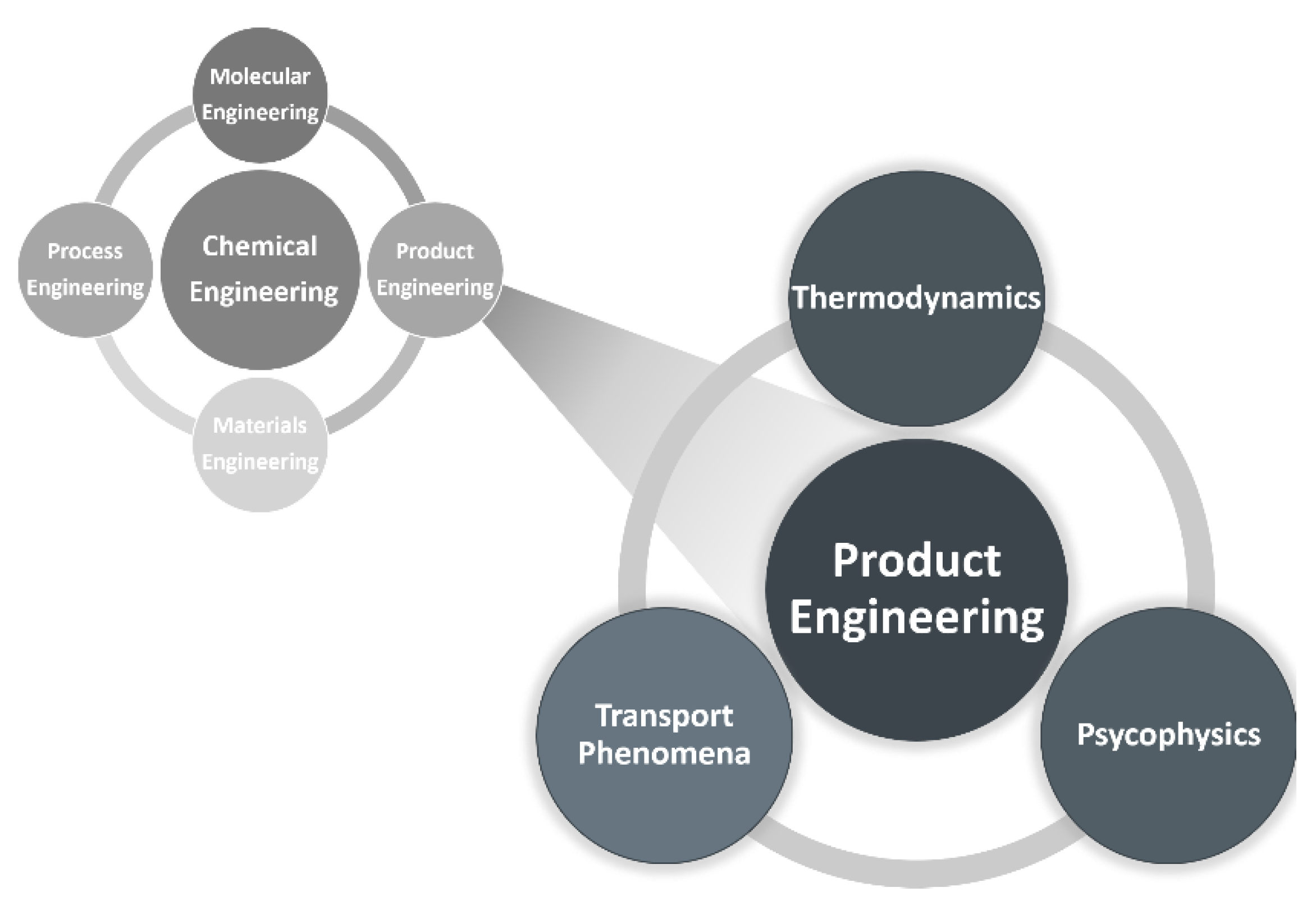
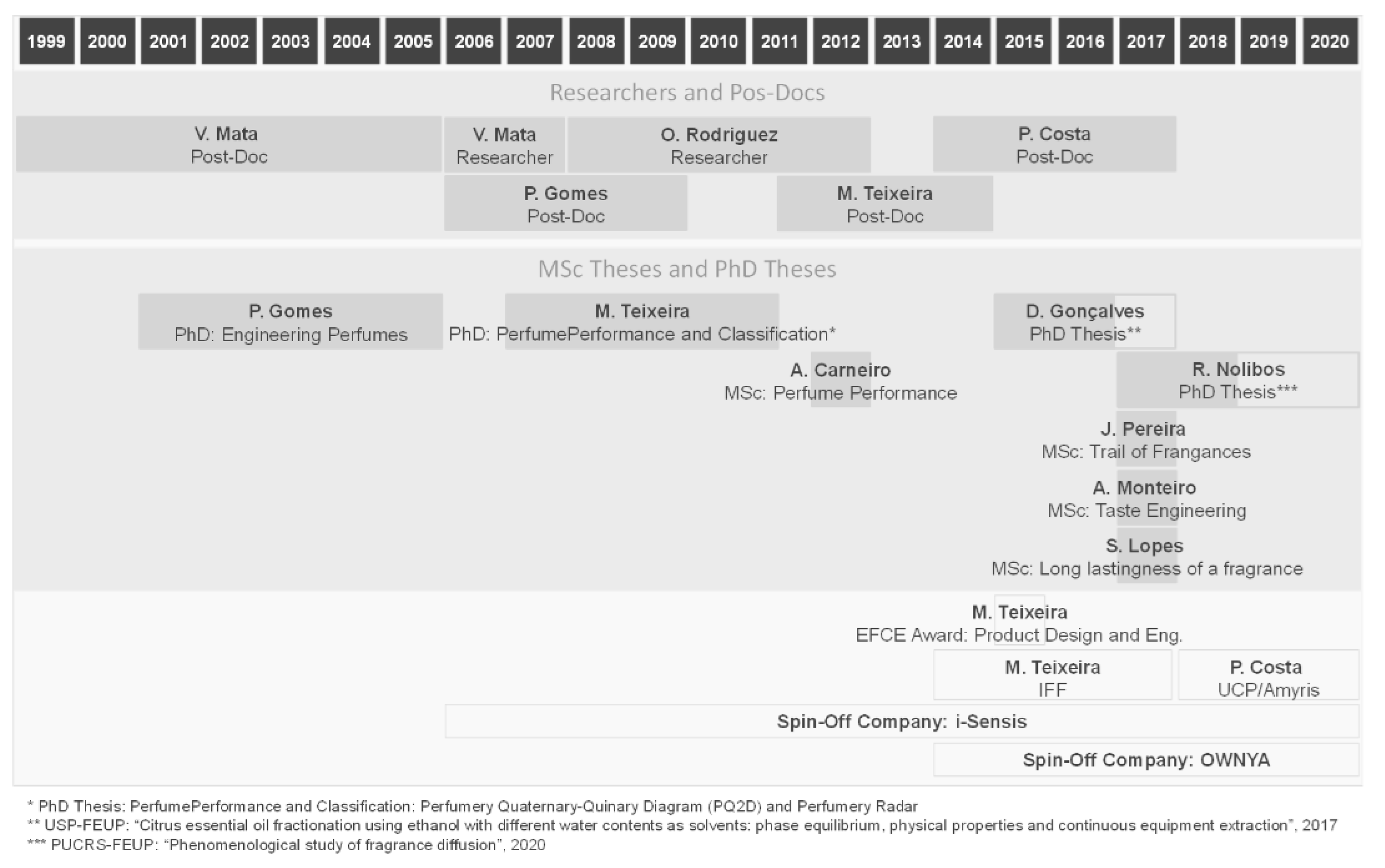
1. Looking Back: The Beginning of Perfume Engineering at LSRE. What Do We Smell?
2. The Perception of Odors
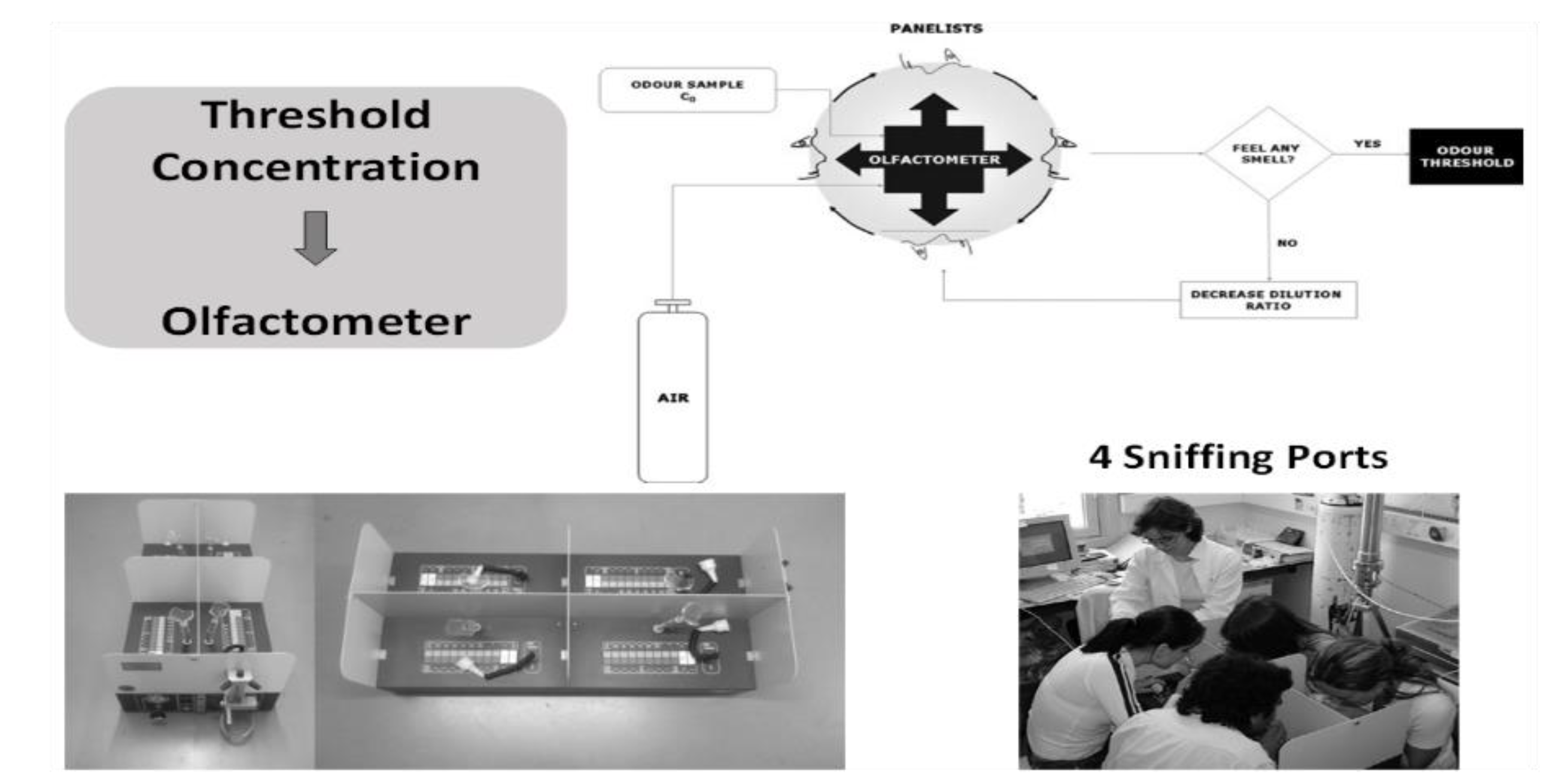
2.1. Odor Thresholds
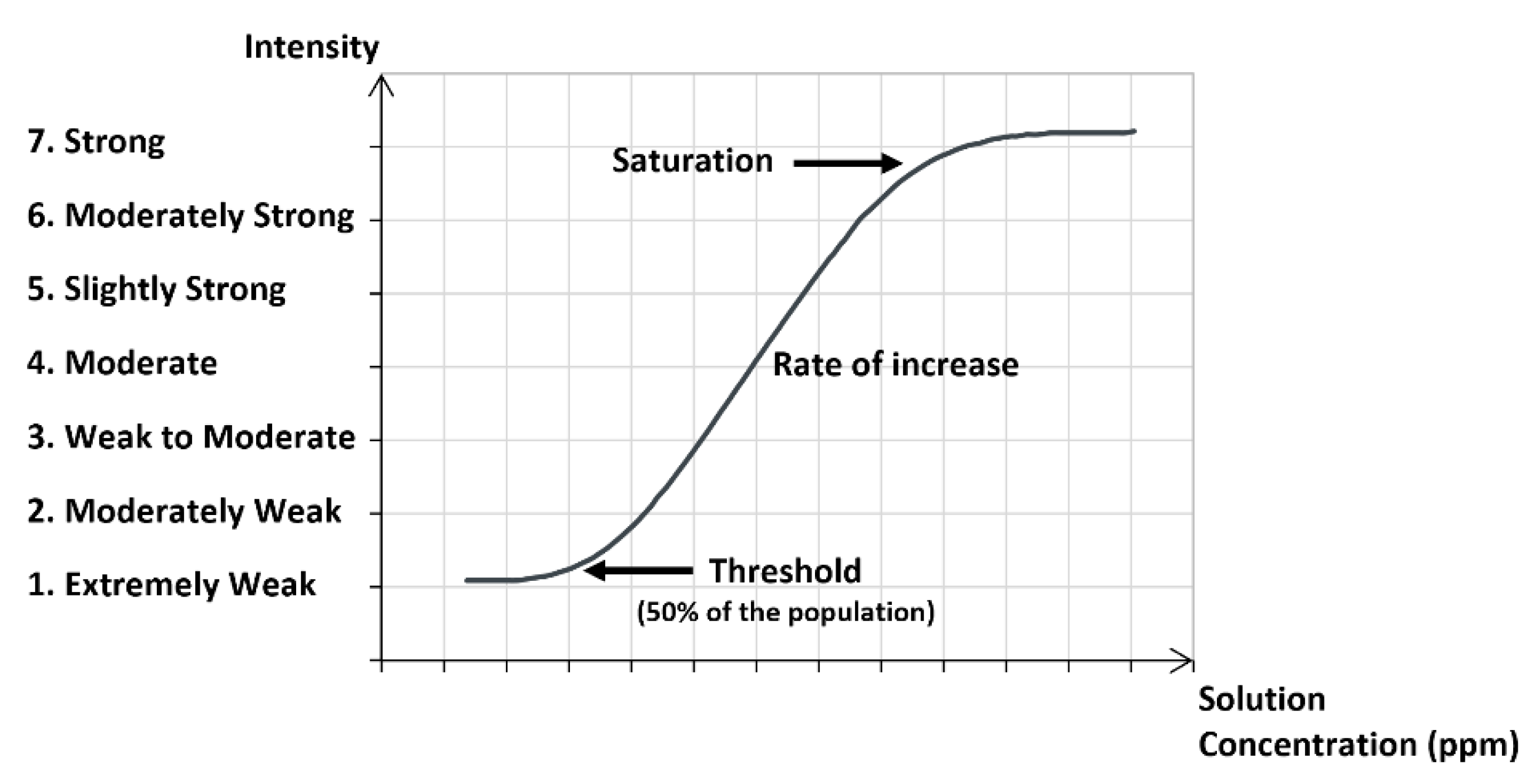
2.2. Odor Intensity Models
2.3. Odor Character Model for a Mixture—The Strongest Component Model
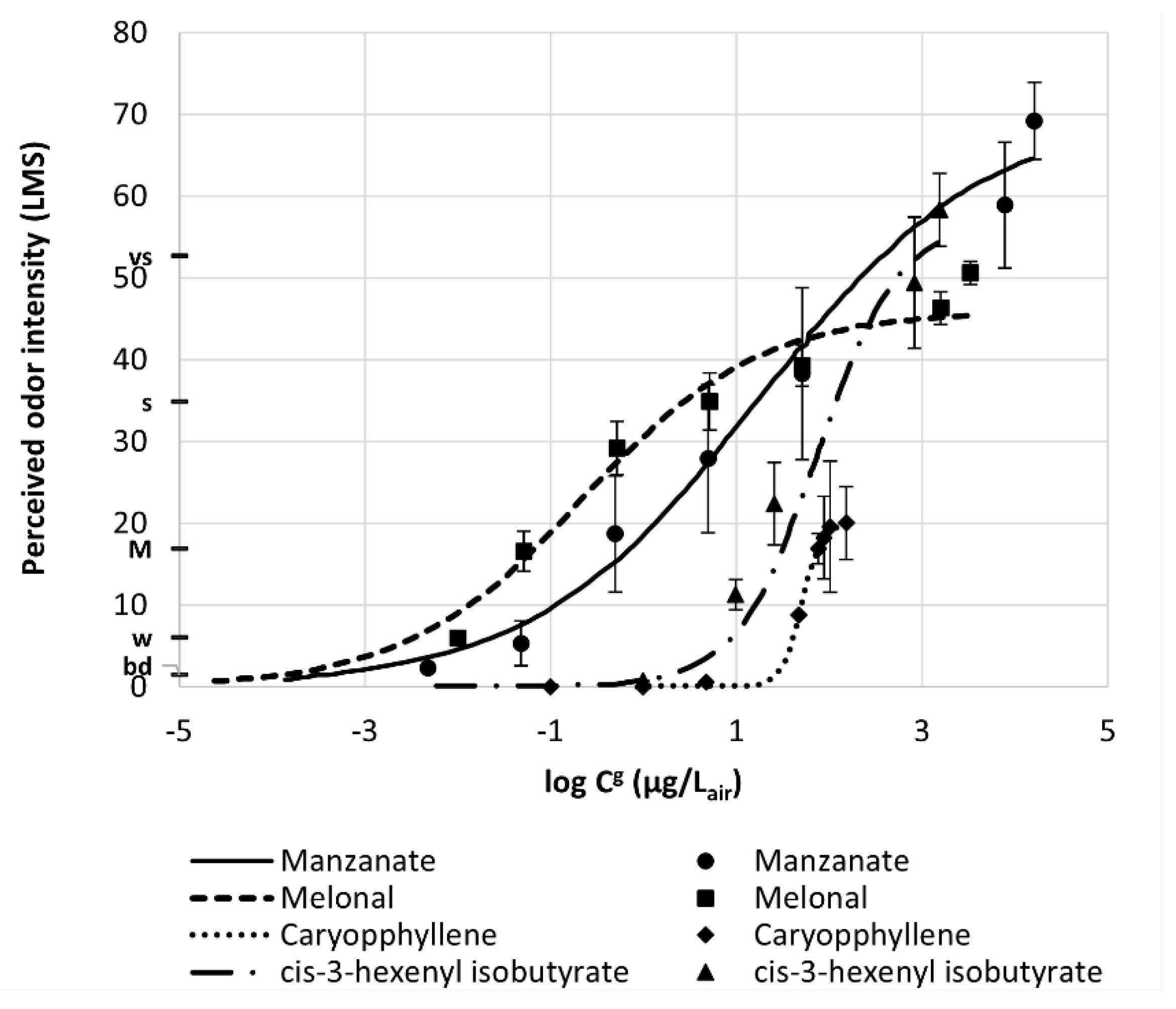
2.4. Sensory Dose/Response Curve
2.5. Evaporation of Perfumes: Modeling Vapor-Liquid Equilibrium (VLE)
2.6. Prediction of Odor Detection Threshold (ODT)
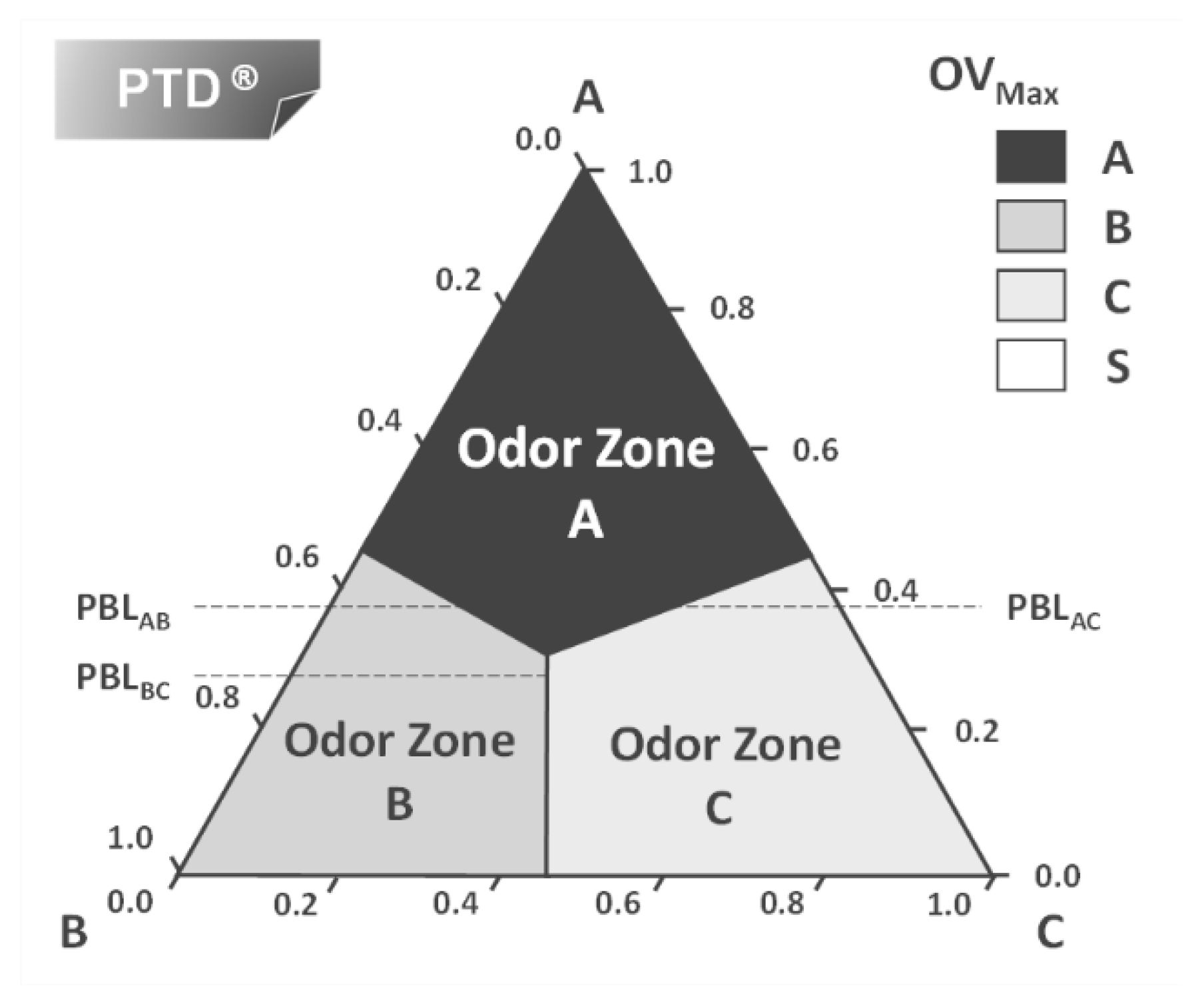
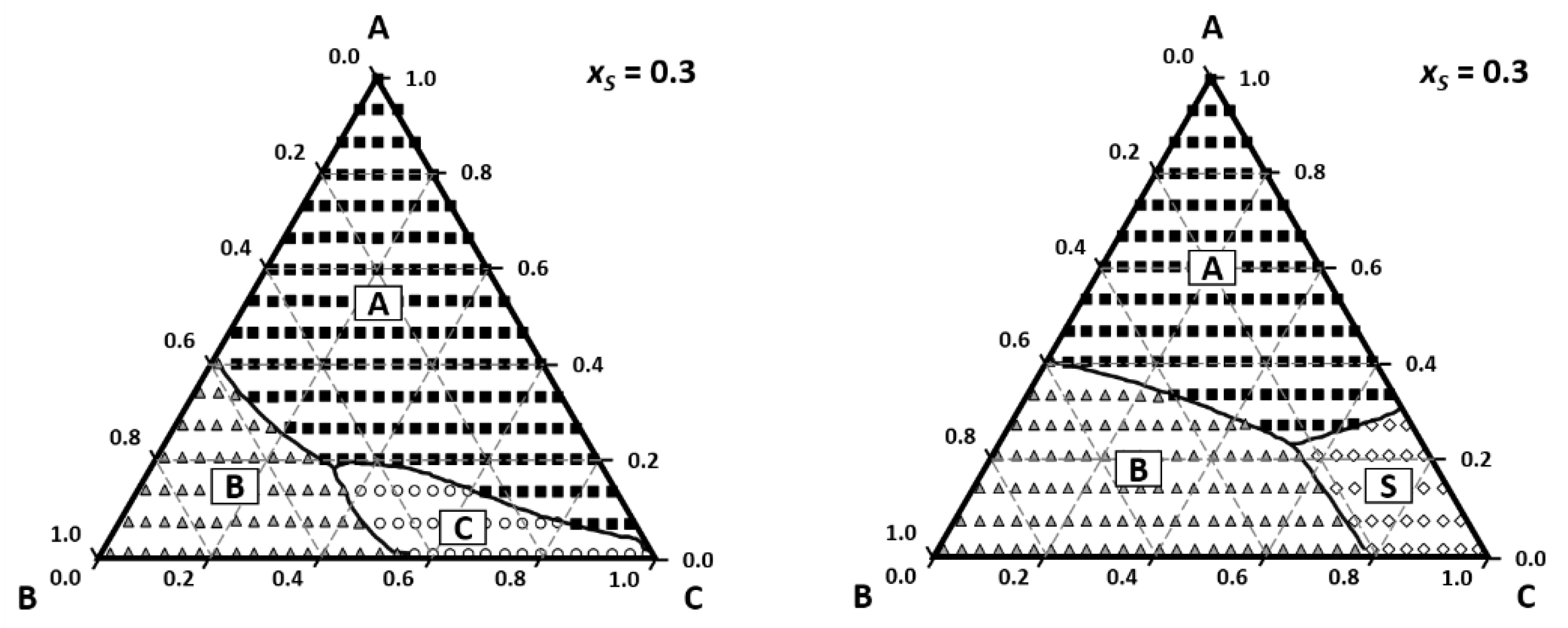
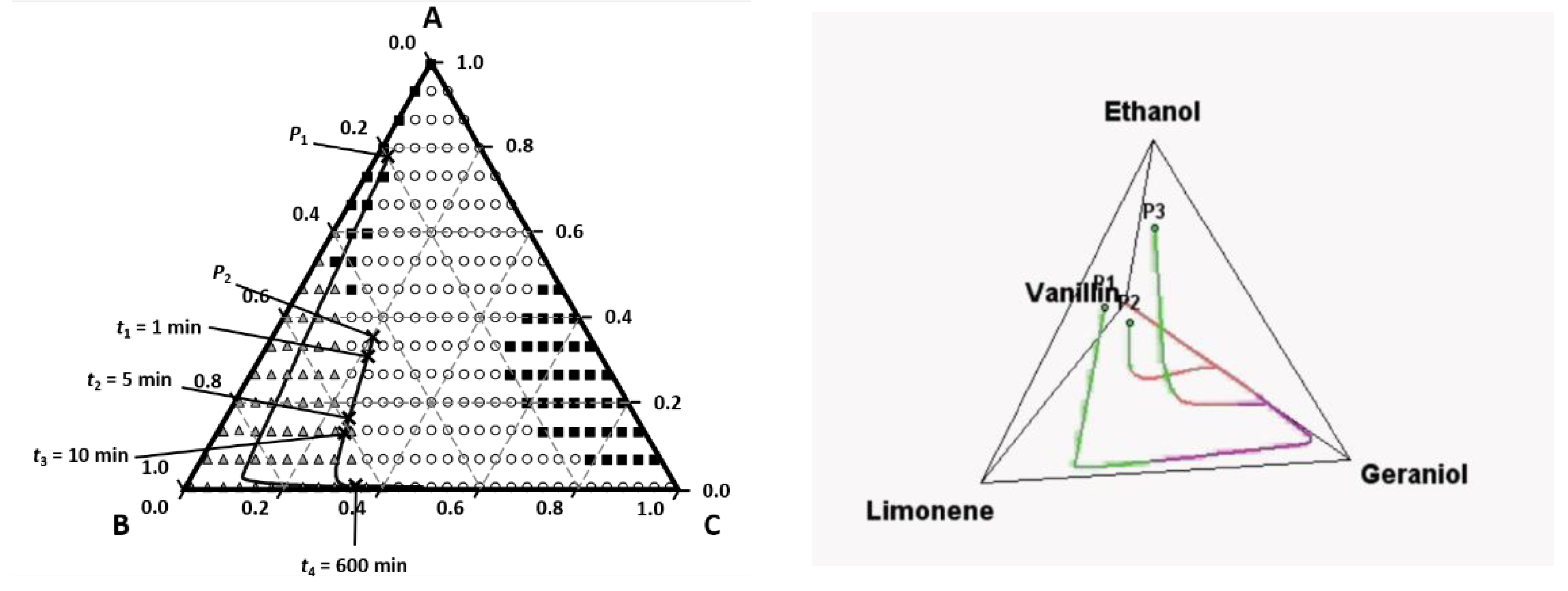
3. Perfumery Ternary Diagram (PTD)
- (i)
- a database of vapor pressure for perfumery raw materials (PRM);
- (ii)
- a database of ODT for PRM and
- (iii)
- a tool to calculate activity coefficients for PRM.
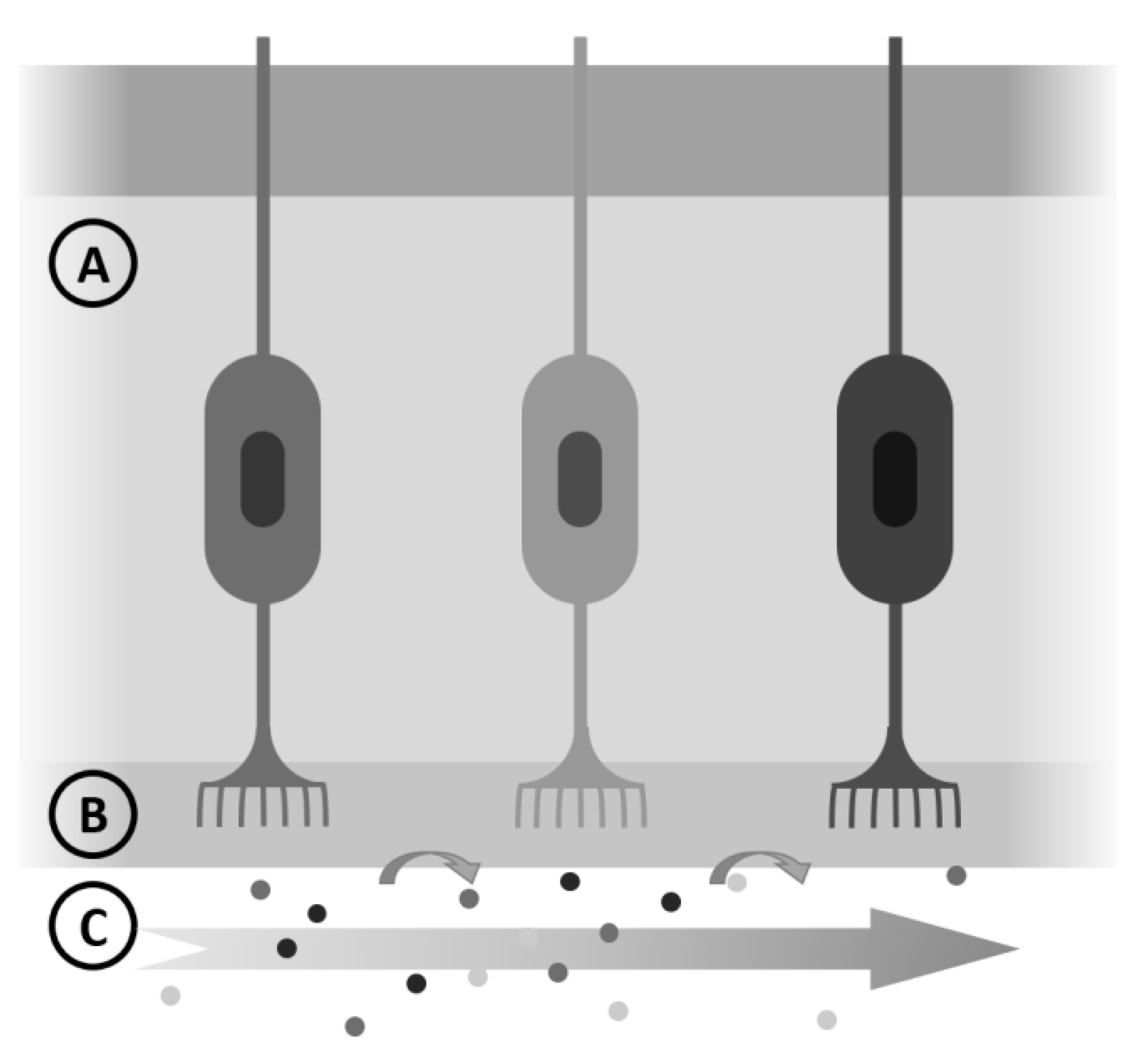
4. Diffusion and Performance of Perfumes
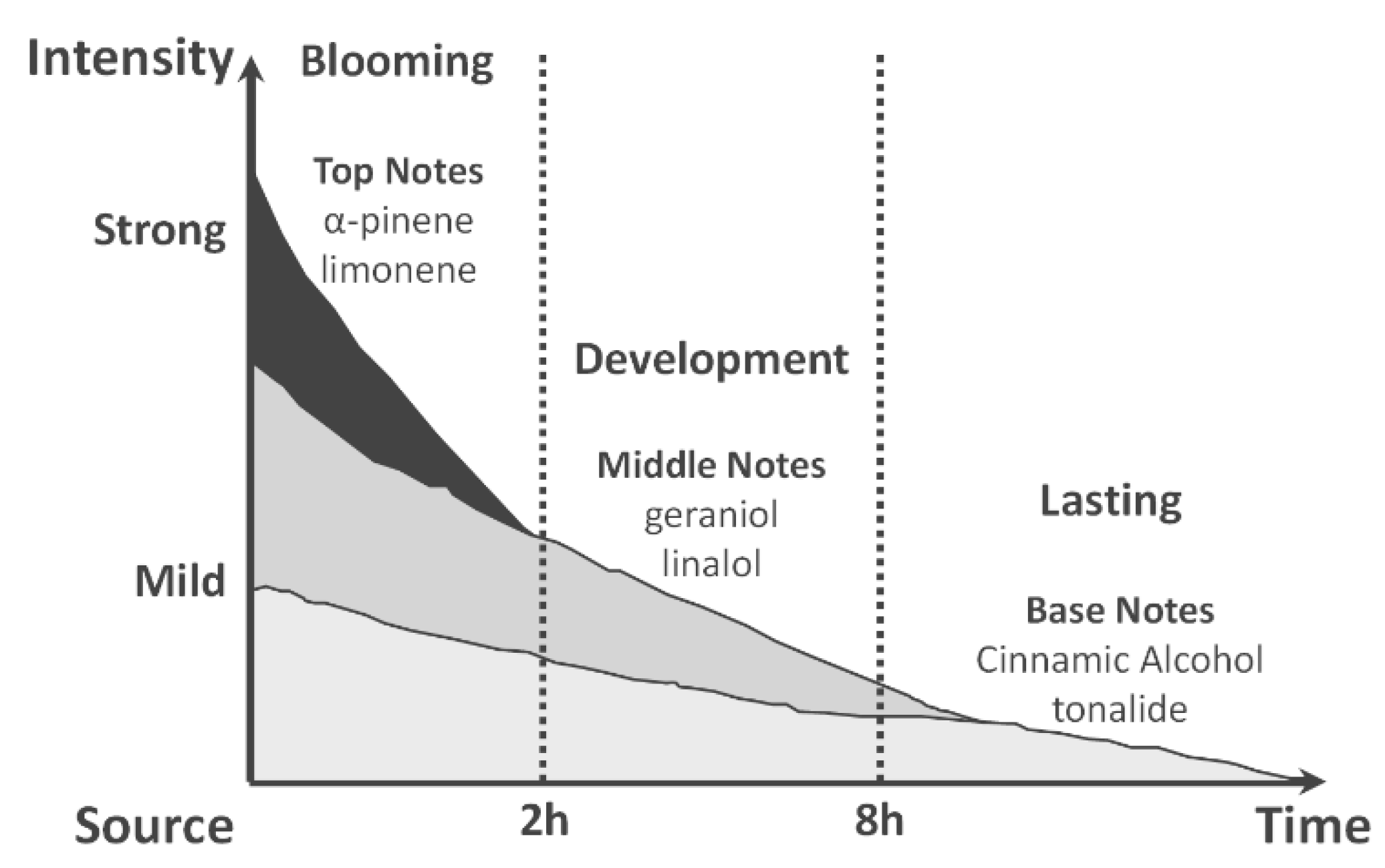
4.1. Perfume Performance
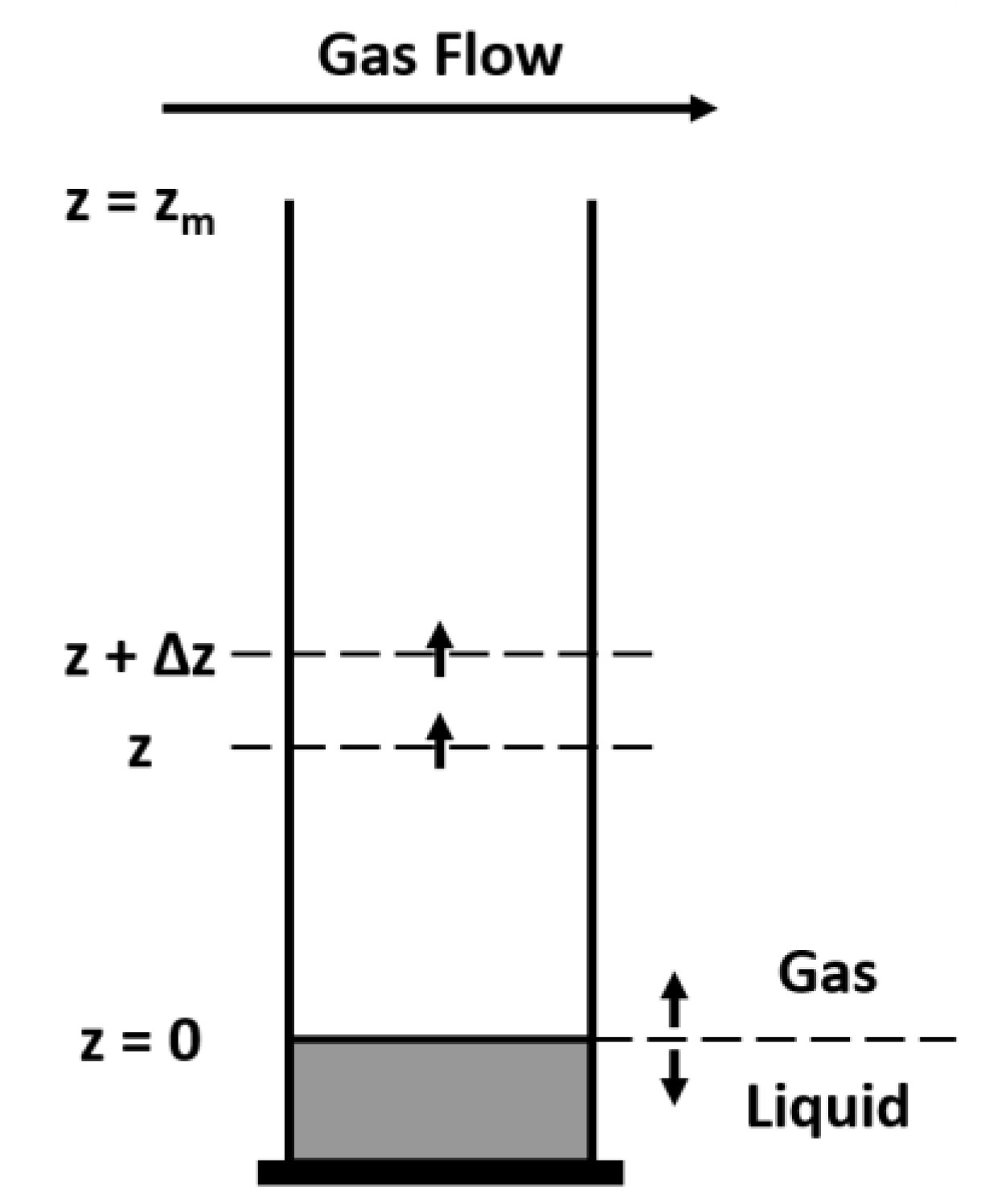
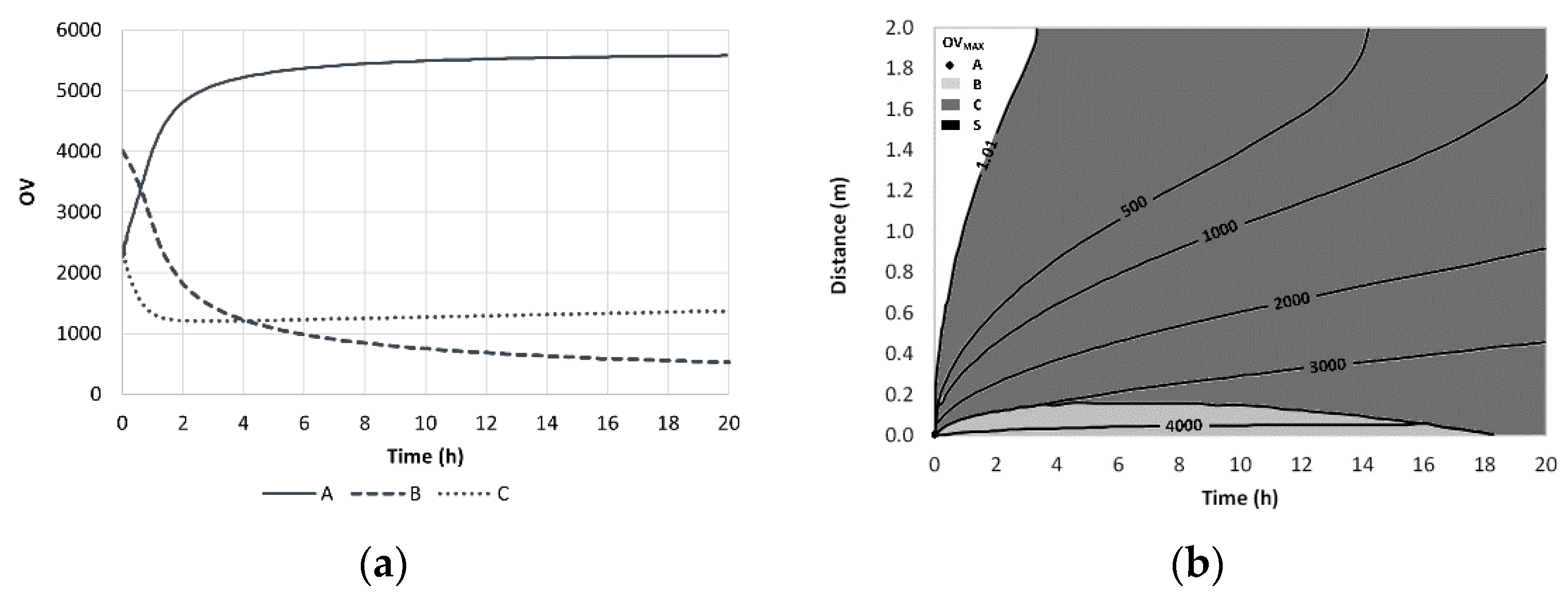
4.2. A Simple 1D Diffusion Model
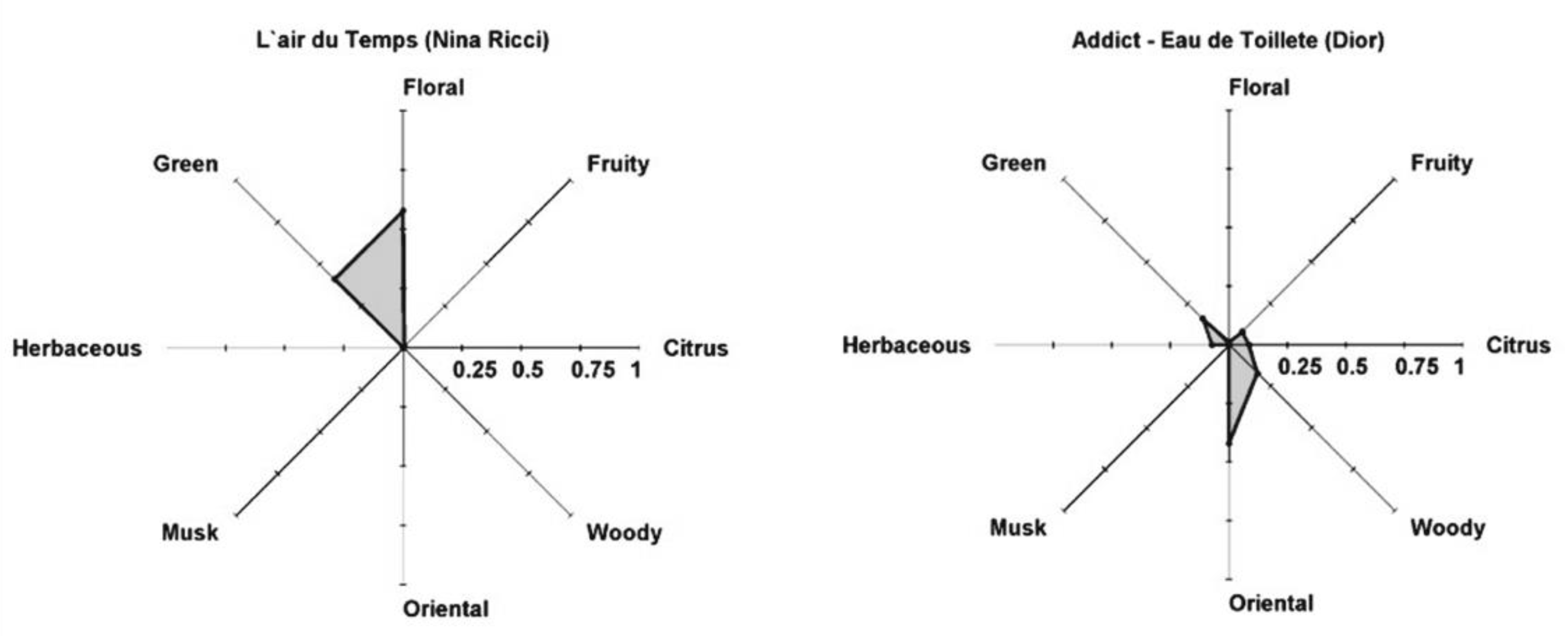
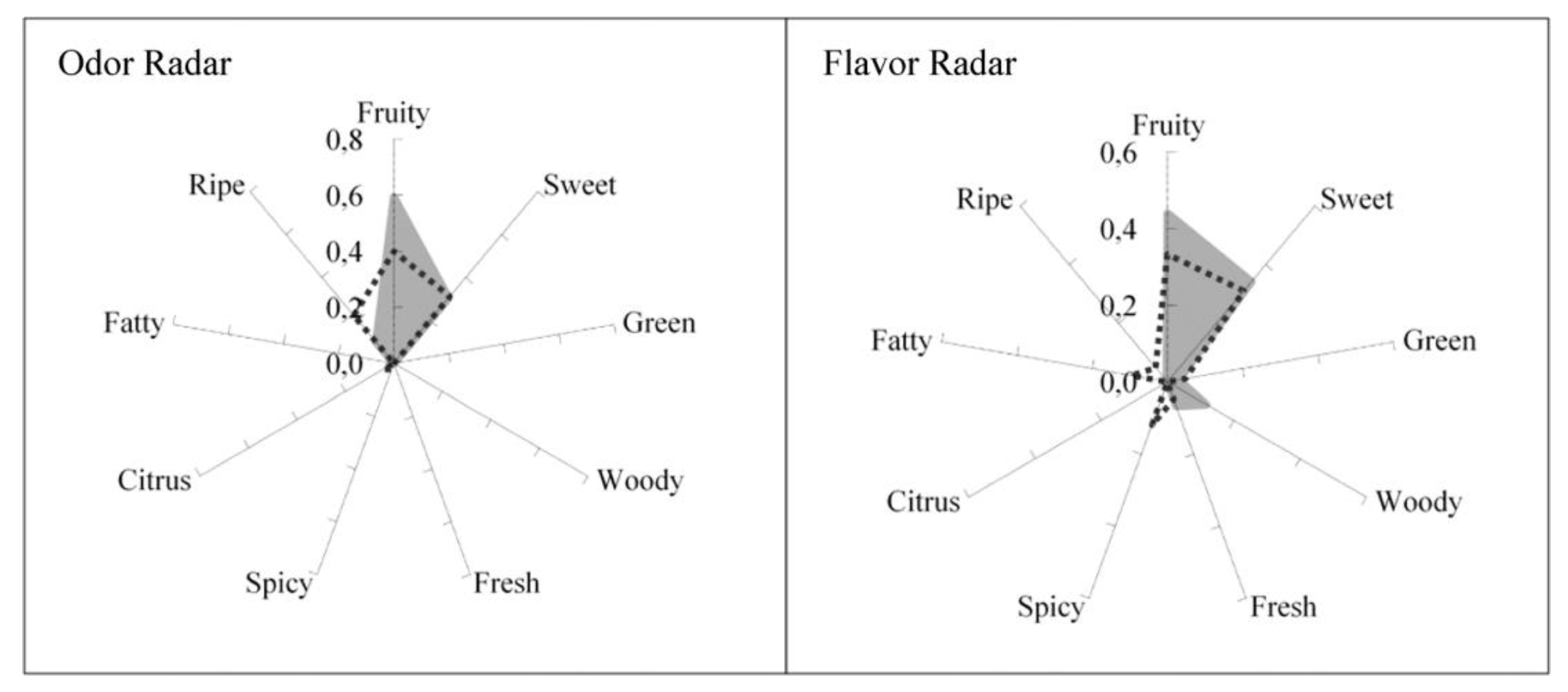
5. Perfume Classification and Perfumery Radar
- (i)
- Classification of pure fragrances in j = 8 olfactory families: citrus, fruity, floral, green, herbaceous, musk, oriental, woody;
- (ii)
- prediction of the odor intensity for each fragrance i, OVi;
- (iii)
- calculation of the odor value for each family , and normalization ;
- (iv)
- plotting the perfumery radar.
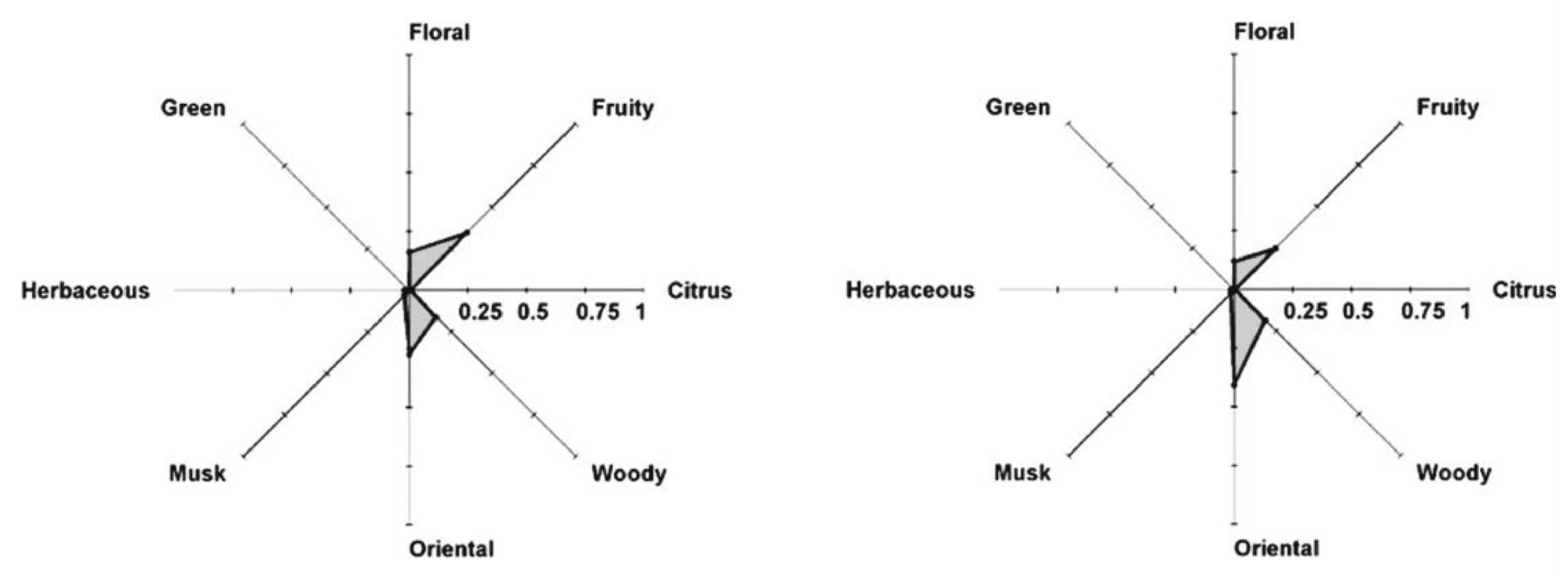
6. The Effect of Matrix and Skin
6.1. Effect of Matrix (Glycerine, Dipropylene Glycol, Skin Lotion)
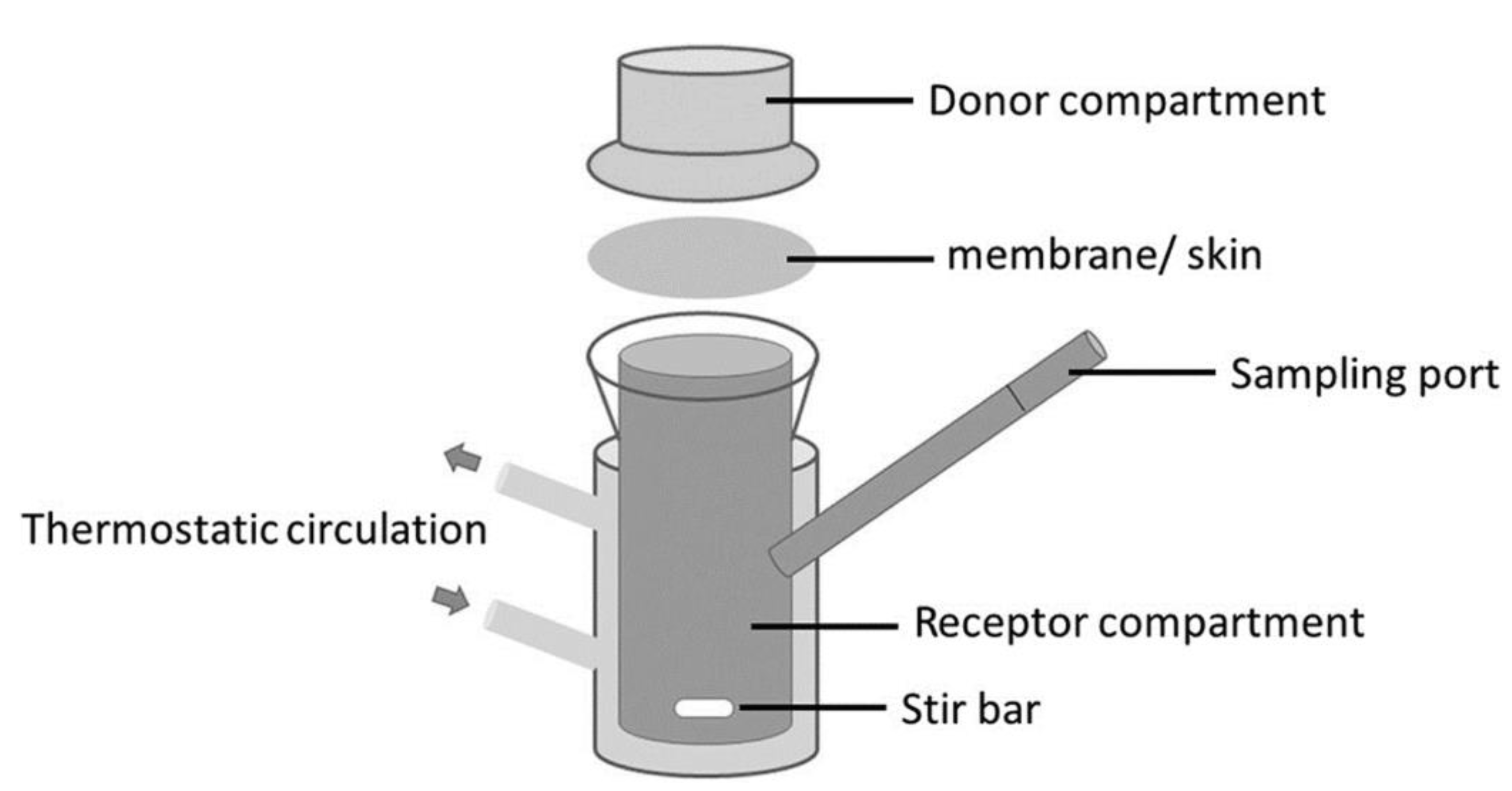
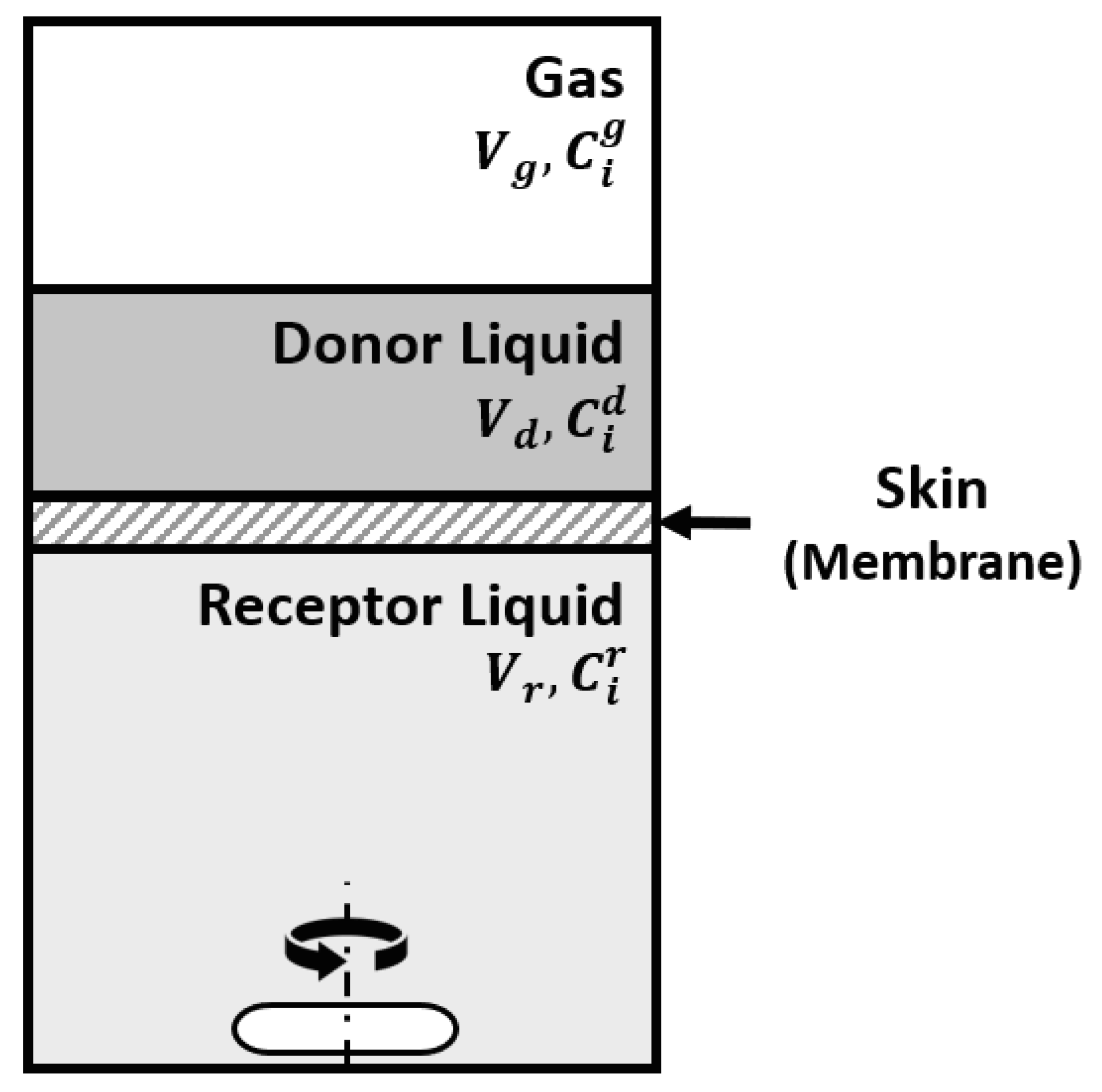
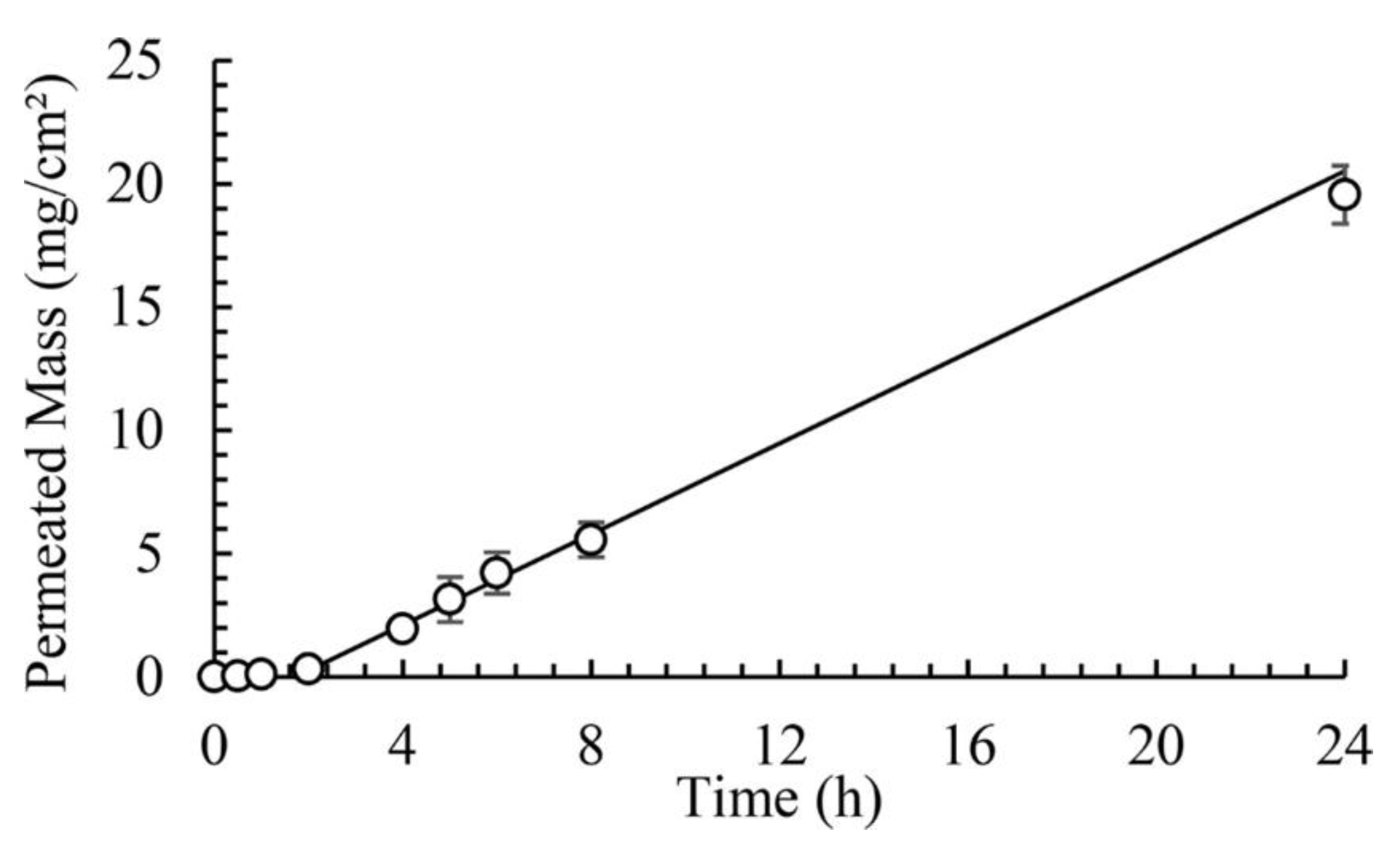
6.2. The Effect of Skin
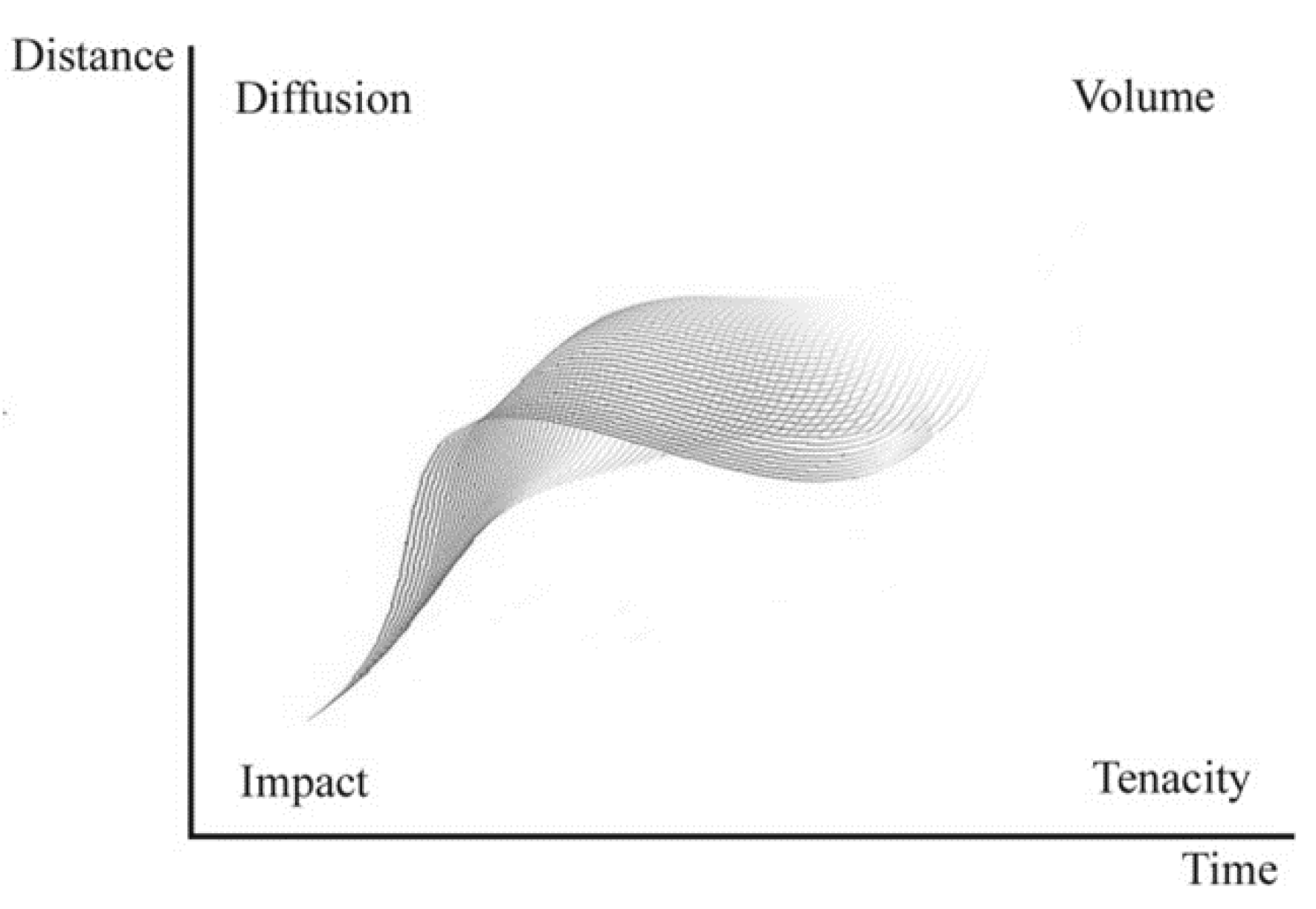
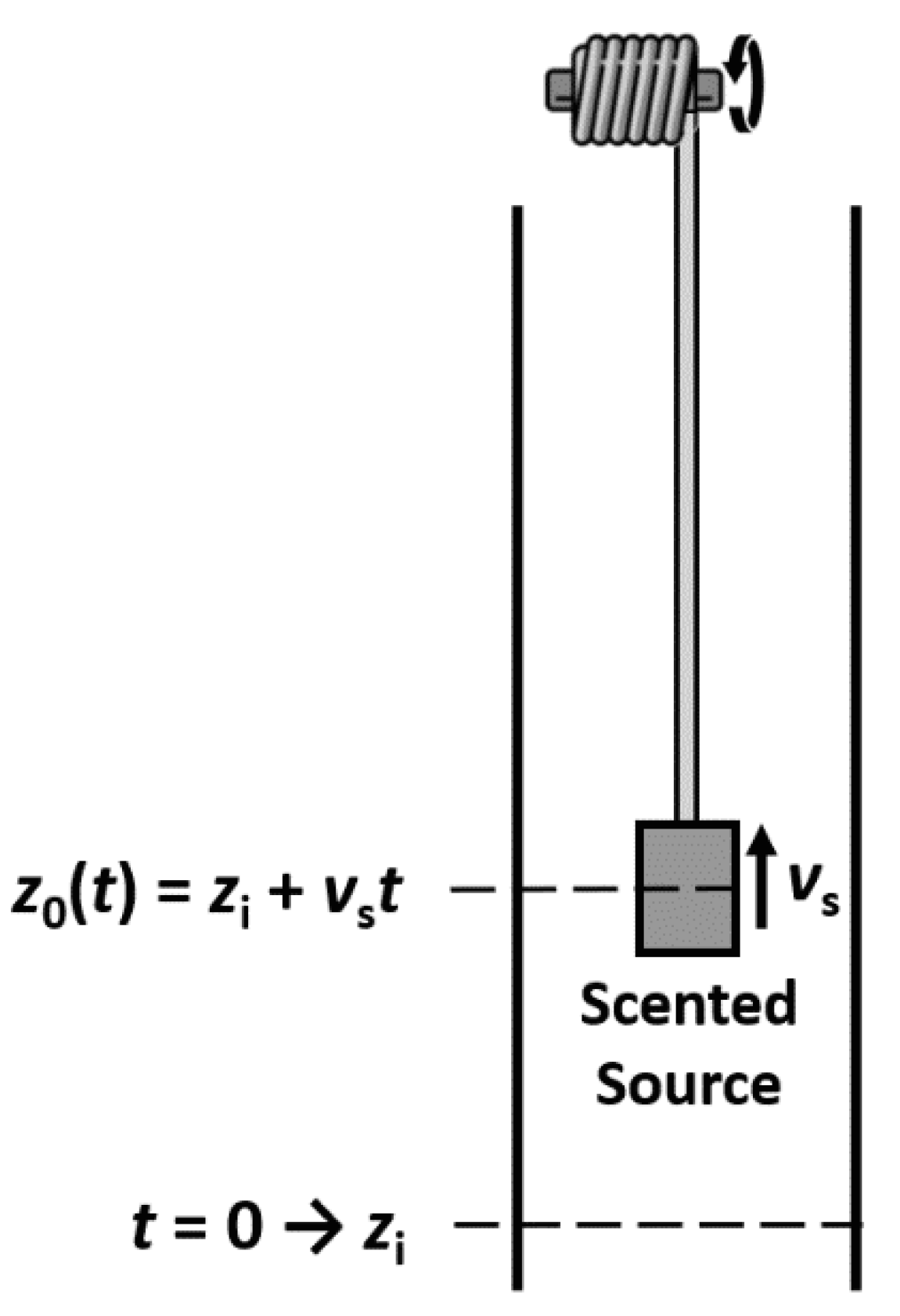
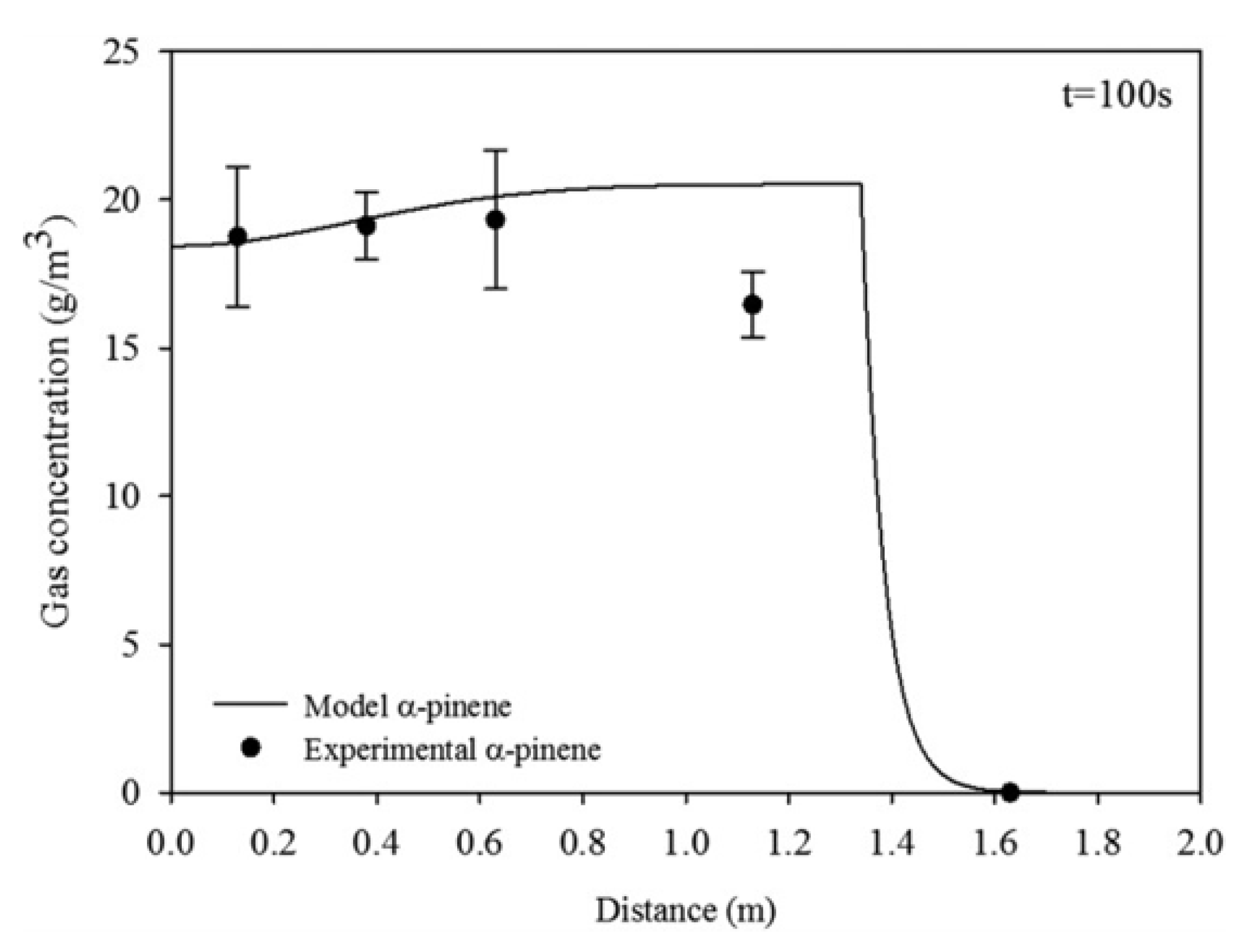
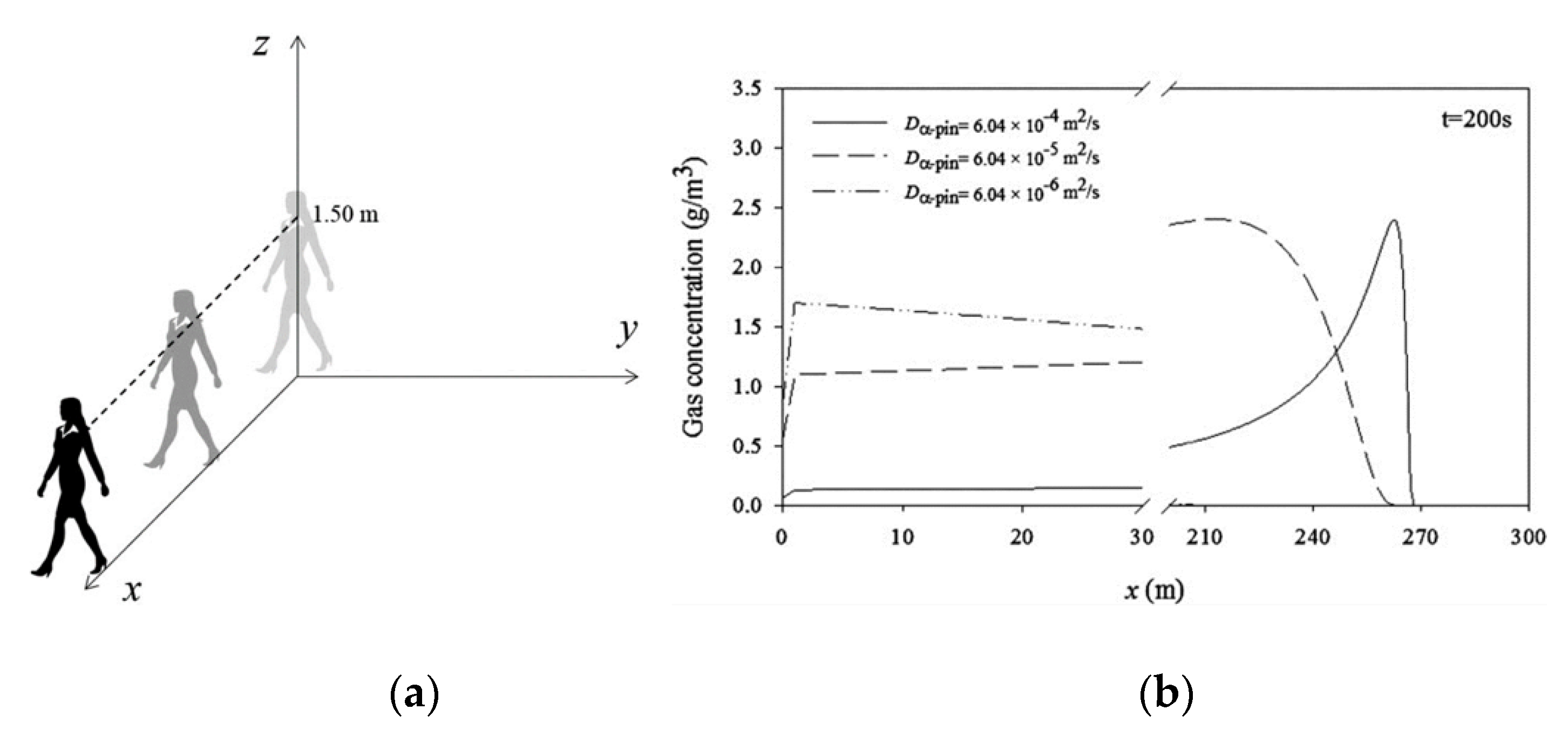
7. The Trail of Perfumes
8. Flavor Engineering
9. Looking Ahead
- (i)
- The importance of diffusivity on the trail of perfumes or sillage
- (ii)
- Formulation of perfumes and fragranced products
- (iii)
- Artificial intelligence (AI) and perfume design
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Carles, J. A method of creation in perfumery. Soap Perfum. Cosmet. 1962, 35, 328–335. [Google Scholar]
- Leffingwell & Associates. Available online: http://www.leffingwell.com/top_10.htm (accessed on 6 April 2021).
- Cussler, E.; Moggridge, G. Chemical Product Design, 2nd ed.; Cambridge University Press: Cambridge, UK, 2011. [Google Scholar]
- Costa, R.; Moggridge, G.D.; Saraiva, P.M. Chemical product engineering: An emerging paradigm within chemical engineering. AIChE J. 2006, 52, 1976–1986. [Google Scholar] [CrossRef]
- Rodrigues, A.E.; Da Silva, F.A.; Granato, M.; Lamia, N.; Grande, C.; Gomes, P.S. Development of propane/propene separations: From molecular simulation to process development. In Proceedings of the XXXIII Reunion Iberica de Adsorcion, Madrid, Spain, 22–24 September 2008. [Google Scholar]
- Teixeira, M.A.; Rodríguez, O.; Gomes, P.; Mata, V.; Rodrigues, A.E. Perfume Engineering, Butterworth-Heineman; Elsevier: Oxford, UK, 2013. [Google Scholar]
- Axel, R. Scents and Sensibility: A molecular logic of olfactory perception (Nobel lecture). Angew. Chem. Int. Ed. 2005, 44, 6110–6127. [Google Scholar] [CrossRef]
- Buck, L.B. Unraveling the sense of smell (Nobel lecture). Angew. Chem. Int. Ed. 2005, 44, 6128–6140. [Google Scholar] [CrossRef] [PubMed]
- Devos, M.; Rouault, J.; Laffort, P. Standardized Olfactory Power Law Exponents; Editions Universitaires Sciences: Dijon, France, 2002. [Google Scholar]
- Devos, M.; Patte, F.; Roualt, J.; Laffort, P.; van Gemert, L.J. Standardized Human Olfactory Thresholds; Oxford University Press, Inc.: New York, NY, USA, 1990; pp. 1–165. [Google Scholar]
- Van Gemert, L.J. Odour Thresholds. In Complications of Odour Threshold Values in Air, Water and Other Media (Edition 2003); Oliemans Punter & Partners BV: Utrecht, The Netherlands, 2003. [Google Scholar]
- Calkin, R.; Jellinek, S. Perfumery: Practice and Principles; John Wiley & Sons: New York, NY, USA, 1994. [Google Scholar]
- Stevens, S.S. On the physiological law. Psychol. Rev. 1957, 64, 153–181. [Google Scholar] [CrossRef]
- Wakayama, H.; Sakasai, M.; Yoshikawa, K.; Inoue, M. Method for Predicting Odor Intensity of Perfumery Raw Materials Using Dose−Response Curve Database. Ind. Eng. Chem. Res. 2019, 58, 15036–15044. [Google Scholar] [CrossRef]
- Green, B.G.; Shaffer, G.S.; Gilmore, M.M. Derivation and Evaluation of a Semantic Scale of Oral Sensation Magnitude with Apparent Ratio Properties. Chem. Senses 1993, 18, 683–702. [Google Scholar] [CrossRef]
- Green, B.G.; Dalton, P.; Cowart, B.; Shaffer, G.; Rankin, K.; Higgins, J. Evaluating the ‘Labeled Magnitude Scale’ for Measuring Sensations of Taste and Smell. Chem. Senses 1996, 21, 323–334. [Google Scholar] [CrossRef]
- Teixeira, M.A.; Rodriguez, O.; Mota, F.L.; Macedo, E.A.; Rodrigues, A.E. Evaluation of group-contribution methods to predict VLE and odor intensity of fragrances. Ind. Eng. Chem. Res. 2011, 50, 9390–9402. [Google Scholar] [CrossRef]
- Xavier, V.; Staudt, P.; Soares, R.D.P. Predicting VLE and odor intensity of matrices containing fragrances with COSMO-SAC. Ind. Eng. Chem. Res. 2020, 59, 2145–2154. [Google Scholar] [CrossRef]
- Teixeira, M.A.; Rodriguez, O.; Rodrigues, A.E.; Selway, R.; Riveroll, M.; Chieffi, A. Prediction and experimental measurement of the odor intensity of fragrance mixtures. Ind. Eng. Chem. Res. 2013, 52, 963–971. [Google Scholar] [CrossRef]
- Rodriguez, O.; Teixeira MARodrigues, A.E. Prediction of odor detection thresholds using partition coefficients. Flavor Fragr. J. 2011, 26, 421–428. [Google Scholar] [CrossRef]
- Mata, V.G.; Gomes, P.B.; Rodrigues, A.E. Science behind perfume design. In Proceedings of the Second European Symposium on Product Technology (Product Design and Technology), Groningen, The Netherlands, 15 September 2004. [Google Scholar]
- Mata, V.G.; Gomes, P.B.; Rodrigues, A.E. Engineering perfumes. AIChE J. 2005, 51, 2834–2852. [Google Scholar] [CrossRef]
- Mata, V.G.; Gomes, P.B.; Rodrigues, A.E. Perfumery ternary diagram (PTD): A new concept applied to the optimization of perfume compositions. Flavour Fragr. J. 2005, 20, 465–471. [Google Scholar] [CrossRef]
- Mata, V.G.; Gomes, P.B.; Rodrigues, A.E. A new methodology for the definition of odor zones in perfumery ternary diagrams. AIChE J. 2006, 52, 2938–2948. [Google Scholar] [CrossRef]
- Teixeira, M.; Rodriguez, O.; Mata, V.; Rodrigues, A.E. Perfumery Quaternary Diagrams for Engineering Perfumes. AIChE J. 2009, 55, 2171–2185. [Google Scholar] [CrossRef]
- Teixeira, M.A. Perfume Performance and Classification: Perfumery Quaternary-Quinary Diagram (PQ2D) and Perfumery Radar. Ph.D. Thesis, University of Porto, Porto, Portugal, 2011. [Google Scholar]
- Teixeira, M.A.; Rodriguez, O.; Mata, V.G.; Rodrigues, A.E. The diffusion of perfume mixtures and odor performance. Chem. Eng. Sci. 2009, 64, 2570–2589. [Google Scholar] [CrossRef]
- Pereira, J.; Costa, P.; Loureiro, J.M.; Rodrigues, A.E. Modelling Diffusion of Fragrances: A Radial Perspective. Can. J. Chem. Eng. 2019, 97, 351–360. [Google Scholar] [CrossRef]
- Almeida, R.N.; Rodrigues, A.E.; Vargas, R.M.; Cassel, D. Radial diffusion model for fragrance materials: Prediction and validation. AIChE J. 2021. under review. [Google Scholar]
- Edwards, M. The Fragrance Wheel. Available online: http://www.fragrancedirectory.info/usadirectory/ (accessed on 31 October 2012).
- DROM. Drom Fragrance Circle. Available online: http://www.drom.com/ (accessed on 31 December 2011).
- Brechbill, G.O. A Reference Book on Fragrance Ingredients; Creative Endeavor Books: Hackensack, NJ, USA, 2006. [Google Scholar]
- Surburg, H.; Panten, J. Common Fragrance and Flavors Materials-Preparation, Properties and Uses; Wiley-VCH: Weinheim, Germany, 2006. [Google Scholar]
- The Good Scents Company. Available online: http://www.thegoodscentscompany.com/ (accessed on 31 May 2010).
- AIHA. Odor Thresholds for Chemicals with Established Health Standards; American Institute Hygiene Association: Fairfax, VA, USA, 2013. [Google Scholar]
- Teixeira, M.; Rodriguez, O.; Rodrigues, A.E. Perfumery Radar: A predictive tool for Perfume Family classification. Ind. Eng. Chem. Res. 2010, 49, 11764–11777. [Google Scholar] [CrossRef]
- Costa, P.; Velasco, C.; Loureiro, J.; Rodrigues, A.E. Effect of cosmetic matrices on the release and odor profiles of the supercritical CO2 extract of Origanum majorana L. Int. J. Cosmet. Sci. 2016, 38, 364–374. [Google Scholar] [CrossRef]
- Costa, P.; Teixeira, M.; Lièvre, Y.; Loureiro, J.; Rodrigues, A.E. Modelling fragrance components release from a simplified matrix used in toiletries and household products. Ind. Eng. Chem. Res. 2015, 54, 11720–11731. [Google Scholar] [CrossRef]
- Costa, P.; Teixeira, M.; Mestre, G.; Carneiro, L.; Loureiro, J.; Rodrigues, A.E. Predicting Vapor-Phase Concentrations for the Assessment of the Odor Perception of Fragrance Chemicals Diluted in Mineral Oil. Ind. Eng. Chem. Res. 2017, 56, 8767–8777. [Google Scholar] [CrossRef]
- Almeida, R.; Costa, P.; Pereira, J.; Cassel, E.; Rodrigues, A.E. Evaporation and permeation of fragrances applied to skin. Ind. Eng. Chem. Res. 2019, 58, 9644–9650. [Google Scholar] [CrossRef]
- Wicke, E.; Kallenbach, R. Die Oberflachendiffusion von Kohlendioxyd in aktiven Kohlen. Kolloid Z. 1941, 97, 135–151. [Google Scholar] [CrossRef]
- Almeida, R.N.; Hartz, J.G.; Costa, P.; Rodrigues, A.E.; Vargas, R.M.; Cassel, E. Permeability coefficients and vapor pressure determination for fragrance materials. Int. J. Cosmet. Sci. 2021, 43, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Behan, J.M.; MacMaster, A.; Perring, K.; Tuck, K.M. Insight into how skin changes perfume. Int. J. Cosmet. Sci. 1996, 18, 237–246. [Google Scholar] [CrossRef]
- Vuilleumier, J.; Flament, I.; Sauvegrain, P. Headspace analysis study of evaporation rate of perfume ingredients applied onto skin. Int. J. Cosmet. Sci. 1995, 17, 61–76. [Google Scholar] [CrossRef]
- Schwarzenbach, R.; Bertschi, L. Models to assess perfume diffusion from skin. Int. J. Cosmet. Sci. 2001, 23, 85–98. [Google Scholar] [CrossRef]
- Tibaldi, R.; ten Berge, W.; Drolet, D. Dermal absorption of chemicals: Estimation by IH SkinPerm. J. Occup. Environ. Hyg. 2014, 11, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Benaim, C.; Brahms, J. Sillage in perfumery. In Proceedings of the World Perfumery Congress, Nice, France, 5–7 June 2018. [Google Scholar]
- Pereira, J.; Costa, P.; Coimbra, M.C.; Rodrigues, A.E. The trail of perfumes. AIChE J. 2018, 64, 2890–2897. [Google Scholar] [CrossRef]
- Luetkenhaus, M.; Saint-Paul, A.; Singer, M.; Bensamou, S.; Carrasco, C. Device for Assessing the Sillage of Fragrance Materials. U.S. Patent 10,794,798, 6 October 2020. [Google Scholar]
- Monteiro, A.; Costa, P.; Loureiro, J.; Rodrigues, A.E. Flavour engineering—A methodology to predict sensory qualities of flavored products. Ind. Eng. Chem. Res. 2018, 57, 8115–8123. [Google Scholar] [CrossRef]
- Paulino, B.N.; Sales, A.; de Oliveira Felipe, L.; Pastore, G.M.; Molina, G.; Bicas, J.L. Biotechnological production of non-volatile flavor compounds. Curr. Opin. Food Sci. 2021, 41, 26–35. [Google Scholar] [CrossRef]
- Menis-Henrique, M.E.C. Methodologies to advance the understanding of flavor chemistry. Curr. Opin. Food Sci. 2020, 33, 131–135. [Google Scholar] [CrossRef]
- Panda, S.; Chen, J.; Benjamin, O. Development of model mouth for food oral processing studies: Present challenges and scopes. Innov. Food Sci. Emerg. Technol. 2020, 66, 102524. [Google Scholar] [CrossRef]
- Gupta, S.; Khan, S.; Muzafar, M.; Kushwaha, M.; Yadav, A.; Gupta, A.P. Encapsulation: Entrapping essential oil/flavors/aromas in food, Chapter 6. In Encapsulations; Elsevier: Amsterdam, The Netherlands, 2016; pp. 229–268. [Google Scholar]
- Tovar, T.M.; Mahlez, J.J.; Knox, C.K.; LeVan, D.M. 110th Anniversary: Molecular Structure Effects on Mass Transfer of C10 Hydrocarbons in BPL Activated Carbon. Ind. Eng. Chem. Res. 2019, 58, 15271–15279. [Google Scholar] [CrossRef]
- Jeylisse Castañer, J.; Ramirez, C.A. Binary gas diffusivity estimates from transient, one-dimensional sublimation–diffusion experiments in a spherical enclosure. Chem. Eng. Commun. 2018, 205, 1167–1192. [Google Scholar] [CrossRef]
- Jaime, M.D.S.; Maisonet, S.; Ramírez, C. Effect of Liquid Phase Composition on the Experimental Determination of Binary Gas Diffusivities in an Isothermal Stefan Diffusion Column. Chem. Eng. Commun. 2020, 12, 1658–1684. [Google Scholar] [CrossRef]
- Mookerjee, B.D.; Trenkle, R.W.; Wilson, R.A. System for Perfume Creation Using Aroma Emission Analysis from a Living Fruit and Flower in Close Proximity. U.S. Patent 5,367,899, 29 November 1994. [Google Scholar]
- Mookerjee, B.D.; Patel, S.M.; Trenkle, R.W.; Wilson, R.A. Aura of aroma: A novel technology to study the emission of fragrance from the skin. In Flavors and Fragrances; Swift, K.A.D., Ed.; Woohead Publishing Limited: Cambridge, UK, 2005. [Google Scholar]
- Quellet, C.; Alchenberger, A. Method and Device for the Characterization of Perfume Composition. European Patent Application EP 3521823 A1, 2019. [Google Scholar]
- Zhau, W.; Doro, F.; Teixeira, M. A rapid approach to optimize the design of fragrances for fabric care products. Flavor Fragr. J. 2020, 35, 167–173. [Google Scholar]
- Teixeira, M.; Unno, M.; Helweg, H.; Kay, L.M.; Shin, J.C. High Performing, High Impact Bloom Accord. International Patent Application PCT WO 2018/071897 A1, 19 April 2018. [Google Scholar]
- Rodrigues, S.; Martins, I.; Fernandes, I.; Gomes, P.; Mata, M.; Barreiro, F.; Rodrigues, A.E. Scentfashion: Microencapsulated perfumes for textile application. Chem. Eng. J. 2009, 149, 463–477. [Google Scholar] [CrossRef]
- Sharkawy, A.; Fernandes, I.; Barreiro, M.; Rodrigues, A.E.; Shoeib, T. Aroma-loaded microcapsules with antibacterial activity for eco-friendly textile application: Synthesis, characterization, release and green grafting. Ind. Eng. Chem. Res. 2017, 56, 5516–5526. [Google Scholar] [CrossRef]
- Lopes, S.; Pinheiro, C.; Costa, P.; Fernandes, I.; Barreiro, F.; Rodrigues, A.E. Chitosan-cellulose particles as deliver vehicles for limonene fragrance. Ind. Crop. Prod. 2019, 139, 111407. [Google Scholar] [CrossRef]
- Pauling, L.; Delbruck, M. The nature of the intermolecular forces operative in biological processes. Science 1940, 92, 77–79. [Google Scholar] [CrossRef]
- Wright, R.H. Stereochemical and vibrational theory of odour. Nature 1972, 239, 226. [Google Scholar] [CrossRef] [PubMed]
- Wright, R.H. Odor and molecular vibration: Neural coding of olfactory information. J. Theor. Biol. 1977, 64, 473–474. [Google Scholar] [CrossRef]
- Dyson, G.M. The scientific basis of odor. Chem. Ind. 1938, 57, 647–651. [Google Scholar] [CrossRef]
- Turin, L. A Method for the Calculation of Odor Character from Molecular Structure. J. Theor. Biol. 2002, 216, 367–385. [Google Scholar] [CrossRef]
- Meierhenrich, U.; Golebiowski, J.; Fernandez, X.; Cabrol-Bass, D. The molecular basis of olfactory chemoreception. Angew. Chem. Int. Ed. 2004, 43, 6410–6412. [Google Scholar] [CrossRef]
- Brookes, J.; Horsfield, A.; Stoneham, A. Odour character differences for enantiomers correlate with molecular flexibility. J. R. Soc. Interface 2009, 6, 75–86. [Google Scholar] [CrossRef]
- Laing, D.; Legha, P.; Jinks, A.; Hutchinson, I. Relationship between Molecular Structure, Concentration and Odor Qualities of Oxygenated Aliphatic Molecules. Chem. Senses 2003, 28, 57–69. [Google Scholar] [CrossRef]
- Clarke, R. Why the world wants controls over Artificial Intelligence. Comput. Law Secur. Rev. 2019, 35, 423–433. [Google Scholar] [CrossRef]
- Chastrette, M.; Thomas-Danguin, T.; Rallet, E. Modelling the Human Olfactory Stimulus-Response Function. Chem. Senses 1998, 23, 181–196. [Google Scholar] [CrossRef] [PubMed]
- Behrens, M.; Briand, L.; de March, C.A.; Matsunami, H.; Yamashita, A.; Meyerhof, W.; Weyand, S. Structure–Function Relationships of Olfactory and Taste Receptors. Chem. Senses 2018, 43, 81–87. [Google Scholar] [CrossRef]
- Zhang, L.; Mao, H.; Liu, L.; Du, J.; Gani, R. A machine learning based computer-aided molecular design/screening methodology for fragrance molecules. Comput. Chem. Eng. 2018, 115, 295–308. [Google Scholar] [CrossRef]
- Yu, P.; Low, M.Y.; Zhou, W. Design of experiments and regression modelling in food flavour and sensory analysis: A review. Trends Food Sci. Technol. 2018, 71, 202–215. [Google Scholar] [CrossRef]
- Sanchez-Lengeling, B.; Wei, J.N.; Lee, B.K.; Gerkin, R.C.; Aspuru-Guzik, A.; Wiltschko, A.B. Machine Learning for Scent: Learning Generalizable Perceptual Representations of Small Molecules. arXiv 2019, arXiv:1910.10685. [Google Scholar]
- Dário Gutiérrez, E.; Dhurandhar, A.; Keller, A.; Meyer, P.; Cecchi, G.A. Predicting natural language descriptions of mono-molecular odorants. Nat. Commun. 2018, 9, 4979. [Google Scholar] [CrossRef]
- Keller, A.; Gerkin, R.C.; Guan, Y.; Dhurandhar, A.; Turu, G.; Szalai, B.; Mainland, J.D.; Ihara, Y.; Yu, C.W.; Wolfinger, R.; et al. Predicting human olfactory perception from chemical features of odor molecules. Science 2017, 355, 820–826. [Google Scholar] [CrossRef]
- Zhang, X.; Zhou, T.; Ng, K.M. Optimization-based Cosmetic Formulation: Integration of Mechanistic Model, Surrogate Model, and Heuristics. AIChE J. 2021, 67, e17064. [Google Scholar] [CrossRef]
- Santana, V.; Márcio, M.; Loureiro, J.M.; Ribeiro, A.M.; Rodrigues, A.E.; Nogueira, I. Optimal fragrances formulation using a deep learning neural network architecture: A novel systematic approach. Comput. Chem. Eng. 2021, 150. [Google Scholar] [CrossRef]
| Species | Oil (wt%) | Aura of Aroma® (wt%) | VLE Gas-Phase Composition (wt%) |
|---|---|---|---|
| Benzyl acetone | 0.02 | 0.03 | 0.17 |
| Benzyl acetate | 0.20 | 5.20 | 5.55 |
| Linalool | 2.20 | 34.10 | 57.05 |
| Raspberry ketone | 11.90 | 1.70 | 4.11 |
| 2-Tridecanone | 0.02 | 5.50 | 0.04 |
| 2-Pentadecanone | 69.00 | 33.50 | 25.62 |
| Ethyl myristate | 14.80 | 8.50 | 4.57 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodrigues, A.E.; Nogueira, I.; Faria, R.P.V. Perfume and Flavor Engineering: A Chemical Engineering Perspective. Molecules 2021, 26, 3095. https://doi.org/10.3390/molecules26113095
Rodrigues AE, Nogueira I, Faria RPV. Perfume and Flavor Engineering: A Chemical Engineering Perspective. Molecules. 2021; 26(11):3095. https://doi.org/10.3390/molecules26113095
Chicago/Turabian StyleRodrigues, Alírio E., Idelfonso Nogueira, and Rui P. V. Faria. 2021. "Perfume and Flavor Engineering: A Chemical Engineering Perspective" Molecules 26, no. 11: 3095. https://doi.org/10.3390/molecules26113095
APA StyleRodrigues, A. E., Nogueira, I., & Faria, R. P. V. (2021). Perfume and Flavor Engineering: A Chemical Engineering Perspective. Molecules, 26(11), 3095. https://doi.org/10.3390/molecules26113095








