Flame-Made Calcium Phosphate Nanoparticles with High Drug Loading for Delivery of Biologics
Abstract
1. Introduction
2. Results and Discussion
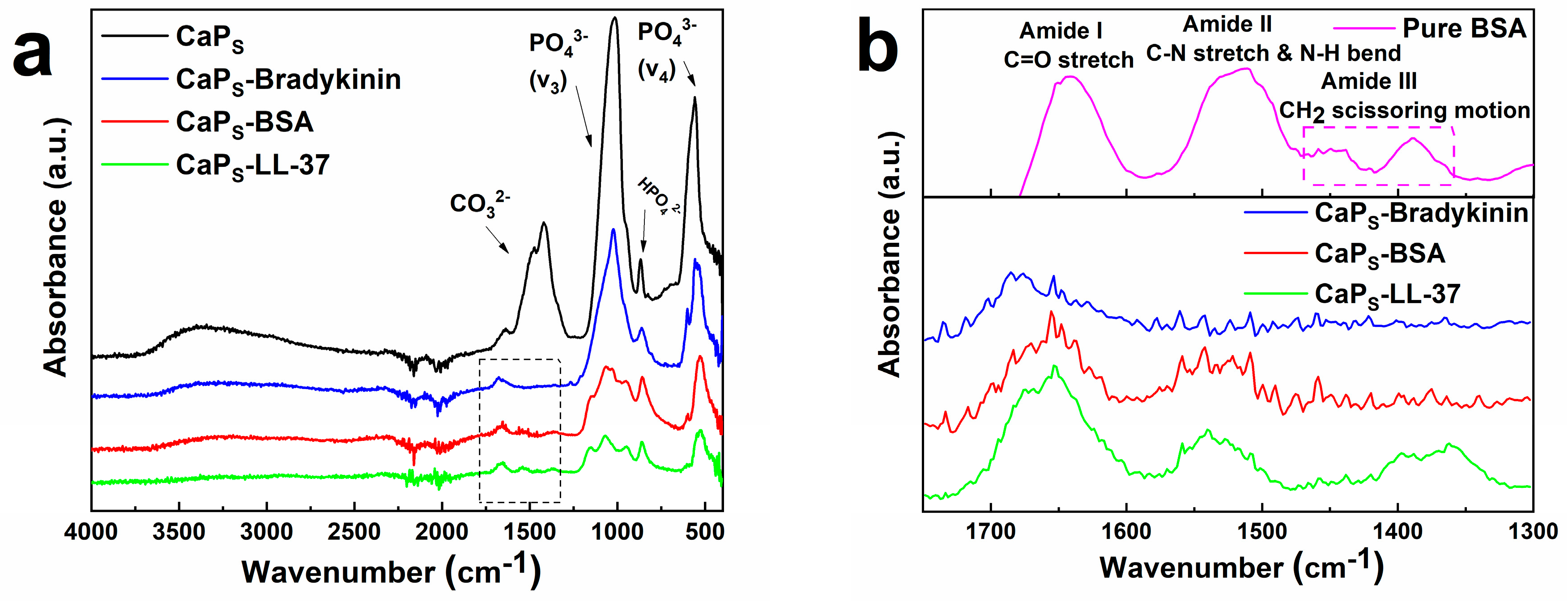
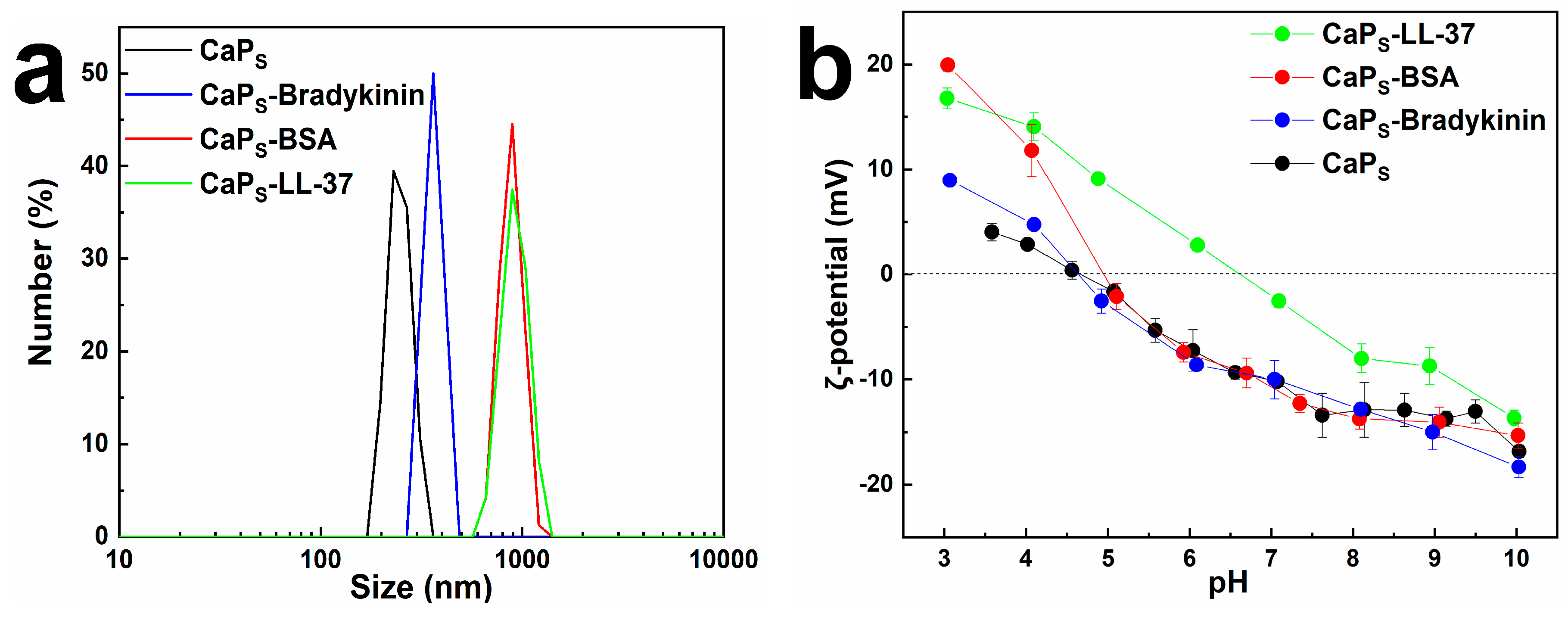
2.1. Particle Morphology
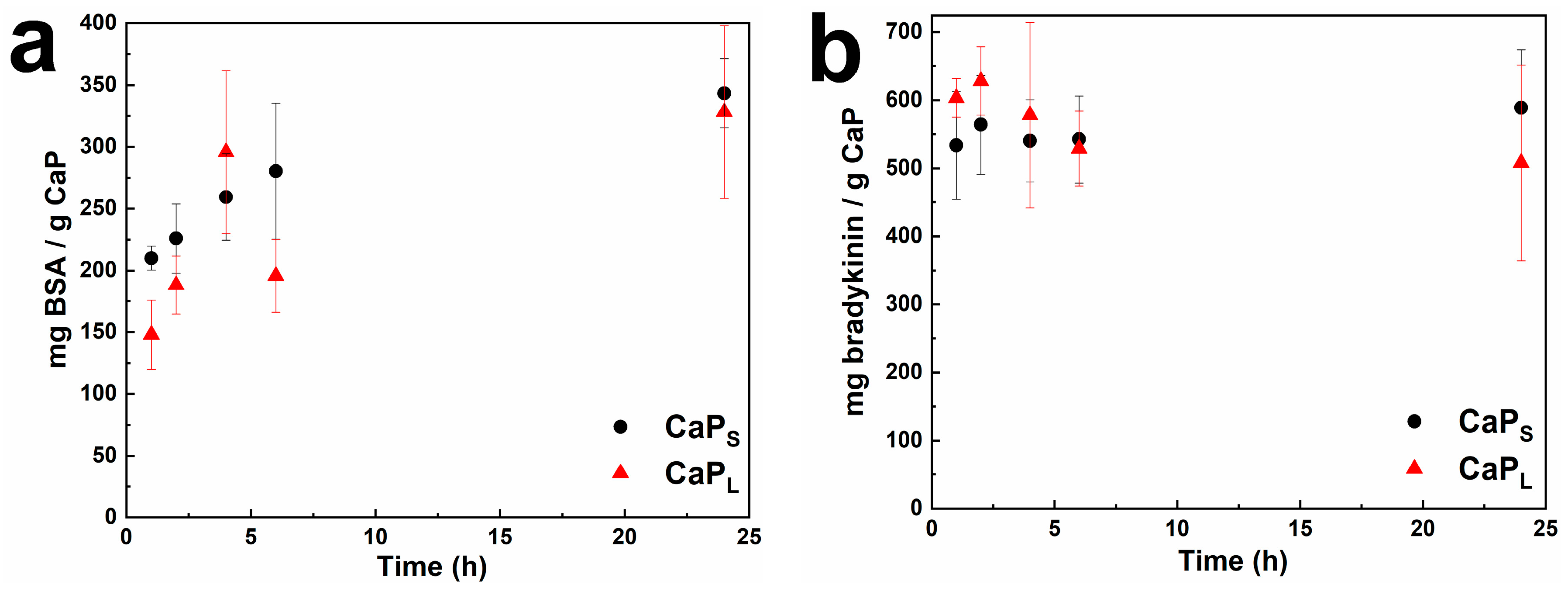
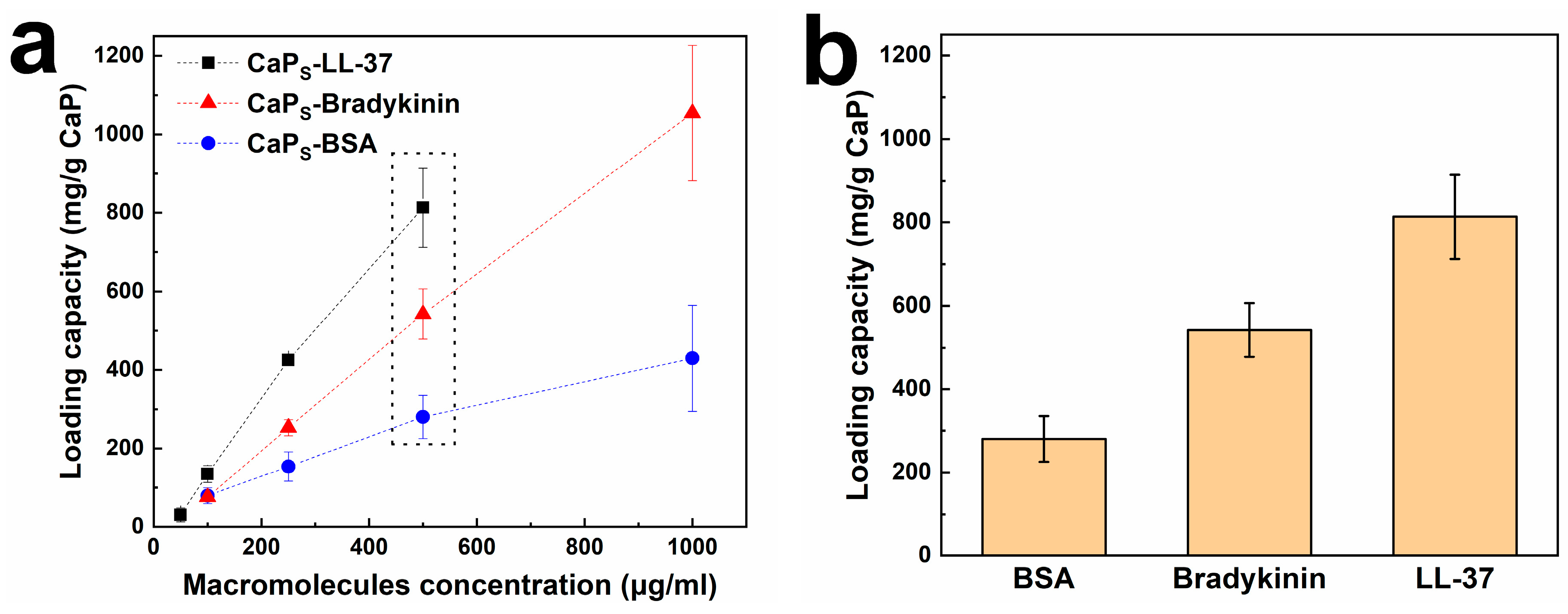
2.2. Loading Macromolecules on CaP Nanoparticles: Effect of Incubation Time and Macromolecule Concentration
2.3. Assessement of LL-37 Stability and Antimicrobial Assays
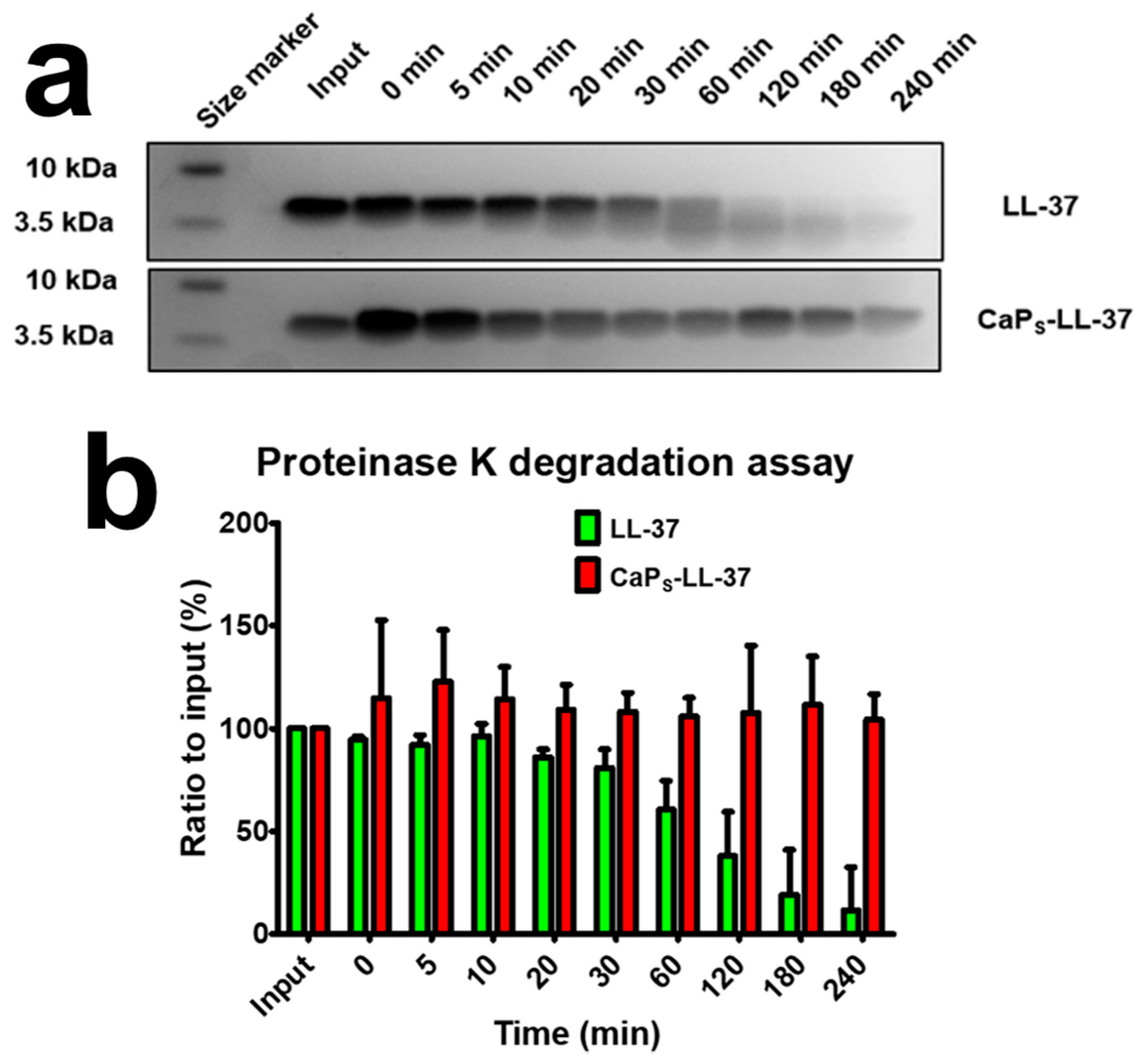
2.3.1. LL-37 Loading on CaP Nanoparticles Keeps LL-37 Intact from Degradation by Proteinase K
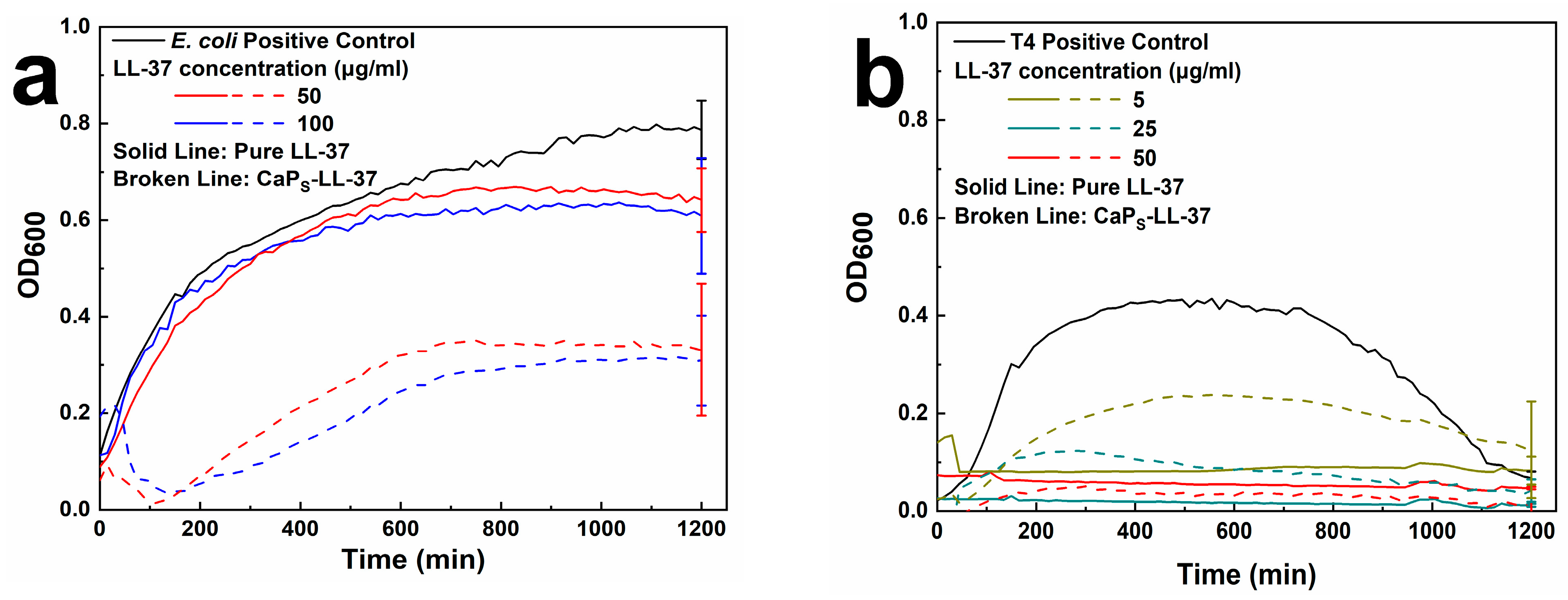
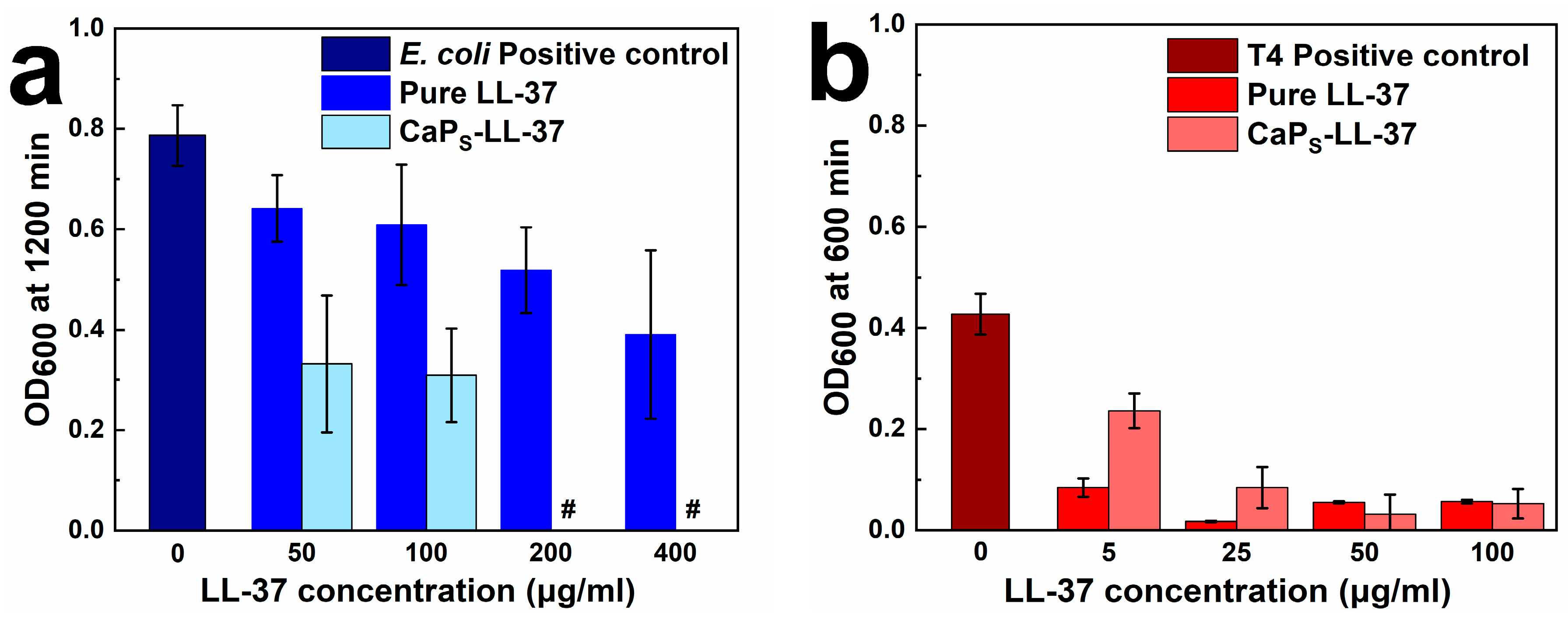
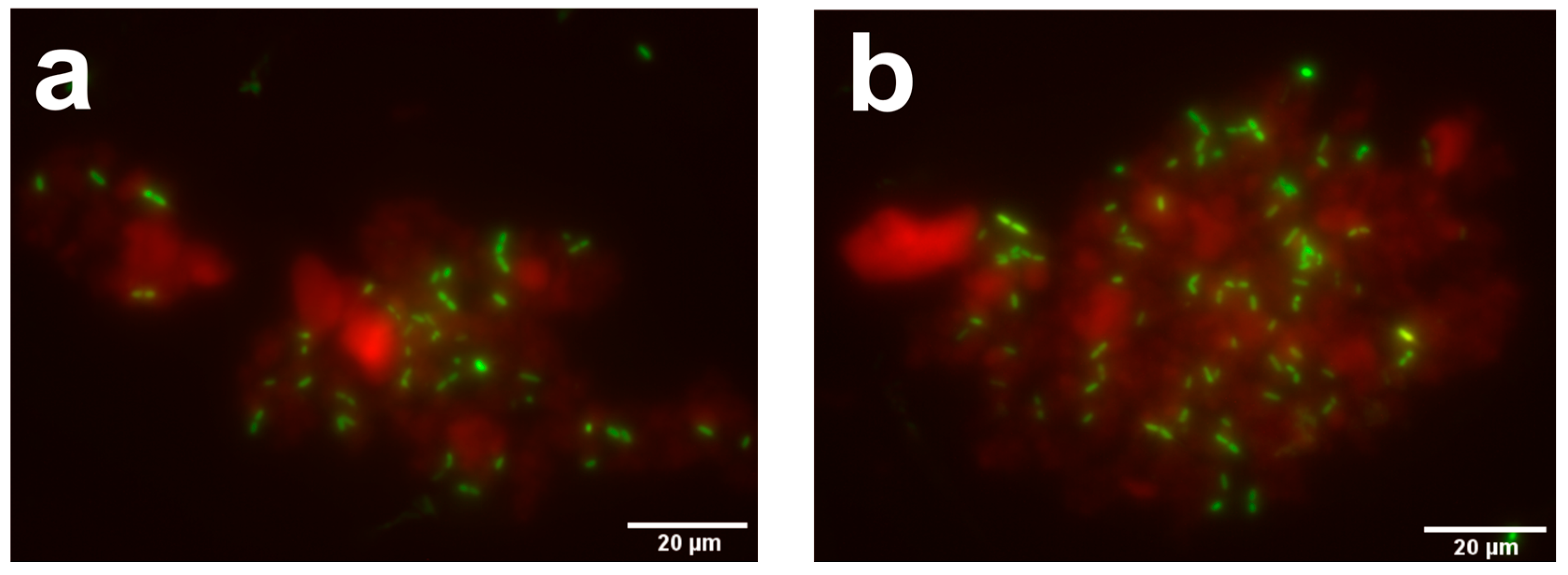
2.3.2. Antimicrobial Activity of LL-37-Loaded CaPS
3. Materials and Methods
3.1. Particle Synthesis and Characterization
3.2. Preparation of Nanoparticles Suspension and Macromolecules Adsorption
3.3. Proteolysis and Growth Curves Analysis
3.3.1. Degradation Assay
3.3.2. SDS-PAGE
3.3.3. Growth Curves
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Biosimilars in the EU: Information Guide for Healthcare Professionals; European Medicines Agency: Amsterdam, The Netherlands, 2019.
- Mullard, A. 2019 FDA drug approvals. Nat. Rev. Drug Discov. 2020, 19, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Anselmo, A.C.; Gokarn, Y.; Mitragotri, S. Non-invasive delivery strategies for biologics. Nat. Rev. Drug Discov. 2019, 18, 19–40. [Google Scholar] [CrossRef]
- Soares, S.; Sousa, J.; Pais, A.; Vitorino, C. Nanomedicine: Principles, Properties, and Regulatory Issues. Front. Chem. 2018, 6, 360. [Google Scholar] [CrossRef] [PubMed]
- Markwalter, C.E.; Pagels, R.F.; Hejazi, A.N.; Gordon, A.G.R.; Thompson, A.L.; Prud’homme, R.K. Polymeric Nanocarrier Formulations of Biologics Using Inverse Flash NanoPrecipitation. AAPS J. 2020, 22, 18. [Google Scholar] [CrossRef] [PubMed]
- Kamaly, N.; Yameen, B.; Wu, J.; Farokhzad, O.C. Degradable Controlled-Release Polymers and Polymeric Nanoparticles: Mechanisms of Controlling Drug Release. Chem. Rev. 2016, 116, 2602–2663. [Google Scholar] [CrossRef]
- Malmsten, M. Inorganic nanomaterials as delivery systems for proteins, peptides, DNA, and siRNA. Curr. Opin. Colloid Interface Sci. 2013, 18, 468–480. [Google Scholar] [CrossRef]
- Liang, R.; Wei, M.; Evans, D.G.; Duan, X. Inorganic nanomaterials for bioimaging, targeted drug delivery and therapeutics. Chem. Commun. 2014, 50, 14071–14081. [Google Scholar] [CrossRef]
- Bisso, S.; Mura, S.; Castagner, B.; Couvreur, P.; Leroux, J.-C. Dual delivery of nucleic acids and PEGylated-bisphosphonates via calcium phosphate nanoparticles. Eur. J. Pharm. Biopharm. 2019, 142, 142–152. [Google Scholar] [CrossRef]
- Bhakta, G.; Mitra, S.; Maitra, A. DNA encapsulated magnesium and manganous phosphate nanoparticles: Potential non-viral vectors for gene delivery. Biomaterials 2005, 26, 2157–2163. [Google Scholar] [CrossRef]
- Yu, J.; Hao, R.; Sheng, F.; Xu, L.; Li, G.; Hou, Y. Hollow manganese phosphate nanoparticles as smart multifunctional probes for cancer cell targeted magnetic resonance imaging and drug delivery. Nano Res. 2012, 5, 679–694. [Google Scholar] [CrossRef]
- Zhuang, X.; Wu, T.; Zhao, Y.; Hu, X.; Bao, Y.; Guo, Y.; Song, Q.; Li, G.; Tan, S.; Zhang, Z. Lipid-enveloped zinc phosphate hybrid nanoparticles for codelivery of H-2Kb and H-2Db-restricted antigenic peptides and monophosphoryl lipid A to induce antitumor immunity against melanoma. J. Control. Release 2016, 228, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Deng, L.; Maeno-Hikichi, Y.; Lai, M.; Chang, S.; Chen, G.; Zhang, J. Formation of an Endophilin-Ca2+ Channel Complex Is Critical for Clathrin-Mediated Synaptic Vesicle Endocytosis. Cell 2003, 115, 37–48. [Google Scholar] [CrossRef]
- Levingstone, T.J.; Herbaj, S.; Dunne, N.J. Calcium Phosphate Nanoparticles for Therapeutic Applications in Bone Regeneration. Nanomaterials 2019, 9, 1570. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Dong, R.; Park, Y.; Bohner, M.; Zhang, X.; Ting, K.; Soo, C.; Wu, B.M. Controlled release of NELL-1 protein from chitosan/hydroxyapatite-modified TCP particles. Int. J. Pharm. 2016, 511, 79–89. [Google Scholar] [CrossRef]
- Miragoli, M.; Ceriotti, P.; Iafisco, M.; Vacchiano, M.; Salvarani, N.; Alogna, A.; Carullo, P.; Ramirez-Rodríguez, G.B.; Patrício, T.; Esposti, L.D.; et al. Inhalation of peptide-loaded nanoparticles improves heart failure. Sci. Transl. Med. 2018, 10, eaan6205. [Google Scholar] [CrossRef]
- Kojima, S.; Nagata, F.; Inagaki, M.; Kugimiya, S.; Kato, K. Avidin-adsorbed peptide–calcium phosphate composites exhibiting high biotin-binding activity. New J. Chem. 2019, 43, 427–435. [Google Scholar] [CrossRef]
- Kojima, S.; Nagata, F.; Kugimiya, S.; Kato, K. Synthesis of peptide-containing calcium phosphate nanoparticles exhibiting highly selective adsorption of various proteins. Appl. Surf. Sci. 2018, 458, 438–445. [Google Scholar] [CrossRef]
- Vanegas Sáenz, J.R.; Tenkumo, T.; Kamano, Y.; Egusa, H.; Sasaki, K. Amiloride-enhanced gene transfection of octa-arginine functionalized calcium phosphate nanoparticles. PLoS ONE 2017, 12, e0188347. [Google Scholar] [CrossRef]
- Huang, X.; Andina, D.; Leroux, J.-C.; Castagner, B. Preparation of PEGylated chelator-stabilized calcium phosphate nanoparticles for liver-targeted delivery of nucleic acid. J. Control. Release 2015, 213, e116. [Google Scholar] [CrossRef]
- Hua, S.; de Matos, M.B.C.; Metselaar, J.M.; Storm, G. Current Trends and Challenges in the Clinical Translation of Nanoparticulate Nanomedicines: Pathways for Translational Development and Commercialization. Front. Pharmacol. 2018, 9, 790. [Google Scholar] [CrossRef]
- Wu, J.; Kamaly, N.; Shi, J.; Zhao, L.; Xiao, Z.; Hollett, G.; John, R.; Ray, S.; Xu, X.; Zhang, X.; et al. Development of Multinuclear Polymeric Nanoparticles as Robust Protein Nanocarriers. Angew. Chem. Int. Ed. 2014, 53, 8975–8979. [Google Scholar] [CrossRef] [PubMed]
- Allen, S.; Osorio, O.; Liu, Y.-G.; Scott, E. Facile assembly and loading of theranostic polymersomes via multi-impingement flash nanoprecipitation. J. Control. Release 2017, 262, 91–103. [Google Scholar] [CrossRef] [PubMed]
- Pratsinis, S.E. Aerosol-based technologies in nanoscale manufacturing: From functional materials to devices through core chemical engineering. AIChE J. 2010, 56, 3028–3035. [Google Scholar] [CrossRef]
- Mohn, D.; Doebelin, N.; Tadier, S.; Bernabei, R.E.; Luechinger, N.A.; Stark, W.J.; Bohner, M. Reactivity of calcium phosphate nanoparticles prepared by flame spray synthesis as precursors for calcium phosphate cements. J. Mater. Chem. 2011, 21, 13963–13972. [Google Scholar] [CrossRef]
- Parent, M.; Baradari, H.; Champion, E.; Damia, C.; Viana-Trecant, M. Design of calcium phosphate ceramics for drug delivery applications in bone diseases: A review of the parameters affecting the loading and release of the therapeutic substance. J. Control. Release 2017, 252, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Tonigold, M.; Simon, J.; Estupiñán, D.; Kokkinopoulou, M.; Reinholz, J.; Kintzel, U.; Kaltbeitzel, A.; Renz, P.; Domogalla, M.P.; Steinbrink, K.; et al. Pre-adsorption of antibodies enables targeting of nanocarriers despite a biomolecular corona. Nat. Nanotechnol. 2018, 13, 862–869. [Google Scholar] [CrossRef]
- Mishra, D.K.; Shandilya, R.; Mishra, P.K. Lipid based nanocarriers: A translational perspective. Nanomedicine 2018, 14, 2023–2050. [Google Scholar] [CrossRef]
- Kuroda, K.; Okumura, K.; Isogai, H.; Isogai, E. The Human Cathelicidin Antimicrobial Peptide LL-37 and Mimics are Potential Anticancer Drugs. Front. Oncol. 2015, 5, 144. [Google Scholar] [CrossRef]
- Chen, C.H.; Lu, T.K. Development and Challenges of Antimicrobial Peptides for Therapeutic Applications. Antibiotics 2020, 9, 24. [Google Scholar] [CrossRef]
- Braun, K.; Pochert, A.; Lindén, M.; Davoudi, M.; Schmidtchen, A.; Nordström, R.; Malmsten, M. Membrane interactions of mesoporous silica nanoparticles as carriers of antimicrobial peptides. J. Colloid Interface Sci. 2016, 475, 161–170. [Google Scholar] [CrossRef]
- Fumakia, M.; Ho, E.A. Nanoparticles Encapsulated with LL37 and Serpin A1 Promotes Wound Healing and Synergistically Enhances Antibacterial Activity. Mol. Pharm. 2016, 13, 2318–2331. [Google Scholar] [CrossRef] [PubMed]
- Comune, M.; Rai, A.; Chereddy, K.K.; Pinto, S.; Aday, S.; Ferreira, A.F.; Zonari, A.; Blersch, J.; Cunha, R.; Rodrigues, R.; et al. Antimicrobial peptide-gold nanoscale therapeutic formulation with high skin regenerative potential. J. Control. Release 2017, 262, 58–71. [Google Scholar] [CrossRef] [PubMed]
- Martin-Serrano, Á.; Gómez, R.; Ortega, P.; de la Mata, F.J. Nanosystems as Vehicles for the Delivery of Antimicrobial Peptides (AMPs). Pharmaceutics 2019, 11, 448. [Google Scholar] [CrossRef] [PubMed]
- Vignoni, M.; de Alwis Weerasekera, H.; Simpson, M.J.; Phopase, J.; Mah, T.-F.; Griffith, M.; Alarcon, E.I.; Scaiano, J.C. LL37 peptide@silver nanoparticles: Combining the best of the two worlds for skin infection control. Nanoscale 2014, 6, 5725–5728. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Orue, I.; Gainza, G.; Girbau, C.; Alonso, R.; Aguirre, J.J.; Pedraz, J.L.; Igartua, M.; Hernandez, R.M. LL37 loaded nanostructured lipid carriers (NLC): A new strategy for the topical treatment of chronic wounds. Eur. J. Pharm. Biopharm. 2016, 108, 310–316. [Google Scholar] [CrossRef]
- Chereddy, K.K.; Her, C.-H.; Comune, M.; Moia, C.; Lopes, A.; Porporato, P.E.; Vanacker, J.; Lam, M.C.; Steinstraesser, L.; Sonveaux, P.; et al. PLGA nanoparticles loaded with host defense peptide LL37 promote wound healing. J. Control. Release 2014, 194, 138–147. [Google Scholar] [CrossRef]
- Hanifi, A.; Fathi, M.H.; Mir Mohammad Sadeghi, H. Effect of strontium ions substitution on gene delivery related properties of calcium phosphate nanoparticles. J. Mater. Sci. Mater. Med. 2010, 21, 2601–2609. [Google Scholar] [CrossRef]
- Mädler, L.; Kammler, H.K.; Mueller, R.; Pratsinis, S.E. Controlled synthesis of nanostructured particles by flame spray pyrolysis. J. Aerosol Sci. 2002, 33, 369–389. [Google Scholar] [CrossRef]
- Ataol, S.; Tezcaner, A.; Duygulu, O.; Keskin, D.; Machin, N.E. Synthesis and characterization of nanosized calcium phosphates by flame spray pyrolysis, and their effect on osteogenic differentiation of stem cells. J. Nanopart. Res. 2015, 17, 95. [Google Scholar] [CrossRef]
- Loher, S.; Stark, W.J.; Maciejewski, M.; Baiker, A.; Pratsinis, S.E.; Reichardt, D.; Maspero, F.; Krumeich, F.; Günther, D. Fluoro-apatite and Calcium Phosphate Nanoparticles by Flame Synthesis. Chem. Mater. 2005, 17, 36–42. [Google Scholar] [CrossRef]
- Swain, S.K.; Sarkar, D. Study of BSA protein adsorption/release on hydroxyapatite nanoparticles. Appl. Surf. Sci. 2013, 286, 99–103. [Google Scholar] [CrossRef]
- Starsich, F.H.L.; Herrmann, I.K.; Pratsinis, S.E. Nanoparticles for Biomedicine: Coagulation during Synthesis and Applications. Annu. Rev. Chem. Biomol. Eng. 2019, 10, 155–174. [Google Scholar] [CrossRef] [PubMed]
- Mavropoulos, E.; Costa, A.M.; Costa, L.T.; Achete, C.A.; Mello, A.; Granjeiro, J.M.; Rossi, A.M. Adsorption and bioactivity studies of albumin onto hydroxyapatite surface. Colloids Surf. B 2011, 83, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Yan, C.; Zhang, X.; Shi, D.; Chi, L.; Luo, G.; Deng, J. Antimicrobial peptide modification enhances the gene delivery and bactericidal efficiency of gold nanoparticles for accelerating diabetic wound healing. Biomater. Sci. 2018, 6, 2757–2772. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zong, G.; He, L.; Zhang, Y.; Liu, C.; Wang, L. Effects of Fumed and Mesoporous Silica Nanoparticles on the Properties of Sylgard 184 Polydimethylsiloxane. Micromachines 2015, 6, 855–864. [Google Scholar] [CrossRef]
- Ozhukil Kollath, V.; Mullens, S.; Luyten, J.; Traina, K.; Cloots, R. Protein–calcium phosphate nanocomposites: Benchmarking protein loading via physical and chemical modifications against co-precipitation. RSC Adv. 2015, 5, 55625–55632. [Google Scholar] [CrossRef]
- Alhazmi, H. FT-IR Spectroscopy for the Identification of Binding Sites and Measurements of the Binding Interactions of Important Metal Ions with Bovine Serum Albumin. Sci. Pharm. 2019, 87, 5. [Google Scholar] [CrossRef]
- Spyrogianni, A.; Herrmann, I.K.; Lucas, M.S.; Leroux, J.-C.; Sotiriou, G.A. Quantitative analysis of the deposited nanoparticle dose on cell cultures by optical absorption spectroscopy. Nanomedicine 2016, 11, 2483–2496. [Google Scholar] [CrossRef]
- Oren, Z.; Lerman, J.C.; Gudmundsson, G.H.; Agerberth, B.; Shai, Y. Structure and organization of the human antimicrobial peptide LL-37 in phospholipid membranes: Relevance to the molecular basis for its non-cell-selective activity. Biochem. J. 1999, 341, 501. [Google Scholar] [CrossRef]
- Stevenson, K.; McVey, A.F.; Clark, I.B.N.; Swain, P.S.; Pilizota, T. General calibration of microbial growth in microplate readers. Sci. Rep. 2016, 6, 38828. [Google Scholar] [CrossRef]
- Mellroth, P.; Daniels, R.; Eberhardt, A.; Rönnlund, D.; Blom, H.; Widengren, J.; Normark, S.; Henriques-Normark, B. LytA, Major Autolysin of Streptococcus pneumoniae, Requires Access to Nascent Peptidoglycan. J. Biol. Chem. 2012, 287, 11018–11029. [Google Scholar] [CrossRef] [PubMed]
- Leszczynska, K.; Namiot, D.; Byfield, F.J.; Cruz, K.; Zendzian-Piotrowska, M.; Fein, D.E.; Savage, P.B.; Diamond, S.; McCulloch, C.A.; Janmey, P.A.; et al. Antibacterial activity of the human host defence peptide LL-37 and selected synthetic cationic lipids against bacteria associated with oral and upper respiratory tract infections. J. Antimicrob. Chemother. 2013, 68, 610–618. [Google Scholar] [CrossRef] [PubMed]
- Wnorowska, U.; Niemirowicz, K.; Myint, M.; Diamond, S.L.; Wróblewska, M.; Savage, P.B.; Janmey, P.A.; Bucki, R. Bactericidal Activities of Cathelicidin LL-37 and Select Cationic Lipids against the Hypervirulent Pseudomonas aeruginosa Strain LESB58. Antimicrob. Agents Chemother. 2015, 59, 3808–3815. [Google Scholar] [CrossRef] [PubMed]
- Song, F.; Li, Y.; Wang, S.; Zhang, L.; Chen, Q. Multifunctional dual-mesoporous silica nanoparticles loaded with a protein and dual antitumor drugs as a targeted delivery system. New J. Chem. 2019, 43, 17284–17297. [Google Scholar] [CrossRef]
- Ching Lau, C.; Reardon, P.J.T.; Campbell Knowles, J.; Tang, J. Phase-Tunable Calcium Phosphate Biomaterials Synthesis and Application in Protein Delivery. ACS Biomater. Sci. Eng. 2015, 1, 947–954. [Google Scholar] [CrossRef]
Sample Availability: Samples of the CaPS and CaPL compounds are available from the authors. |

| Sample | O2 Dispersion Gas Flow Rate (L/min) | Precursor Flow Rate (mL/min) | Ca and P Concentration (M) | Precursor Concentration in Flame (mmolCa+P/LO2disp) | SSA (m2/g) | dBET (nm) |
|---|---|---|---|---|---|---|
| CaPL | 3 | 8 | 0.2 | 0.533 | 73 | 26 |
| CaPS | 8 | 3 | 0.1 | 0.0375 | 246 | 8 |
| Nanocarrier | Hydrodynamic Diameter (nm) | Pathogen | LL-37 Loading (mg/g particle) | LL-37 MIC (μg/mL) | NPs MIC (μg/mL) | Ref |
|---|---|---|---|---|---|---|
| PLGA | 163 | E. coli (ATCC 25922) | 1.02 ± 0.06 | No significant antimicrobial activity | [37] | |
| Nanostructured Lipid Carriers | 220.6 | E. coli (ATCC 25922) | 16.76 ± 0.07 | 20 (72% killing) | 1193 | [36] |
| Solid Lipid nanoparticles | 232.2 (loaded particles) | E. coli (ATCC 25922) S. aureus (ATCC 25923) | 8.48–16.32 | 2–3 (65–72% killing) | 184–236 | [32] |
| 2–3 (42–47% killing) | ||||||
| Au | 10.47 ± 1.89 | Methicillin-resistant S. aureus | 0.56 | 0.9 | 1536 | [45] |
| Mesoporous SiO2 | 307.9 * 294.6 * | E. coli (ATCC 25922) | 24.1 ** 129.7 ** | 2.2 | 93 | [31] |
| 44.9 | 346 | |||||
| Calcium Phosphate | 230 (dBET = 8 nm) | S. pneumoniae (T4, ATCC BAA-34) E. coli (HVM52) | 813.4 ± 101.3 | 50 | 61 | This work |
| 200 | 246 | |||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsikourkitoudi, V.; Karlsson, J.; Merkl, P.; Loh, E.; Henriques-Normark, B.; Sotiriou, G.A. Flame-Made Calcium Phosphate Nanoparticles with High Drug Loading for Delivery of Biologics. Molecules 2020, 25, 1747. https://doi.org/10.3390/molecules25071747
Tsikourkitoudi V, Karlsson J, Merkl P, Loh E, Henriques-Normark B, Sotiriou GA. Flame-Made Calcium Phosphate Nanoparticles with High Drug Loading for Delivery of Biologics. Molecules. 2020; 25(7):1747. https://doi.org/10.3390/molecules25071747
Chicago/Turabian StyleTsikourkitoudi, Vasiliki, Jens Karlsson, Padryk Merkl, Edmund Loh, Birgitta Henriques-Normark, and Georgios A. Sotiriou. 2020. "Flame-Made Calcium Phosphate Nanoparticles with High Drug Loading for Delivery of Biologics" Molecules 25, no. 7: 1747. https://doi.org/10.3390/molecules25071747
APA StyleTsikourkitoudi, V., Karlsson, J., Merkl, P., Loh, E., Henriques-Normark, B., & Sotiriou, G. A. (2020). Flame-Made Calcium Phosphate Nanoparticles with High Drug Loading for Delivery of Biologics. Molecules, 25(7), 1747. https://doi.org/10.3390/molecules25071747






