Carbohydrates—Key Players in Tobacco Aroma Formation and Quality Determination
Abstract
1. Introduction
2. Tobacco Carbohydrates
2.1. Tobacco Sugars
2.2. Tobacco Oligosaccharides
2.3. Tobacco Polysaccharides
2.3.1. Starch
2.3.2. Pectins
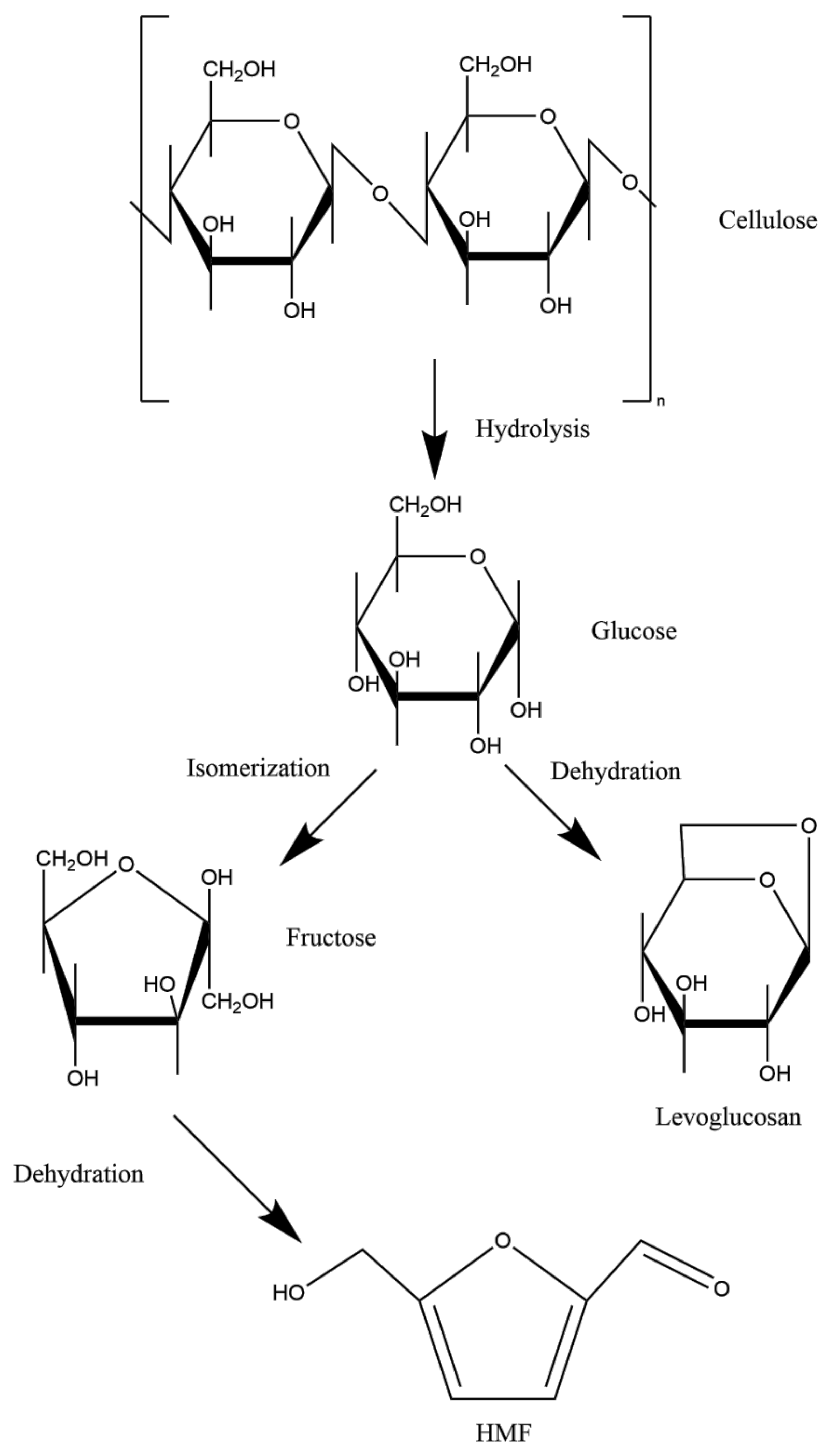
2.3.3. Cellulose
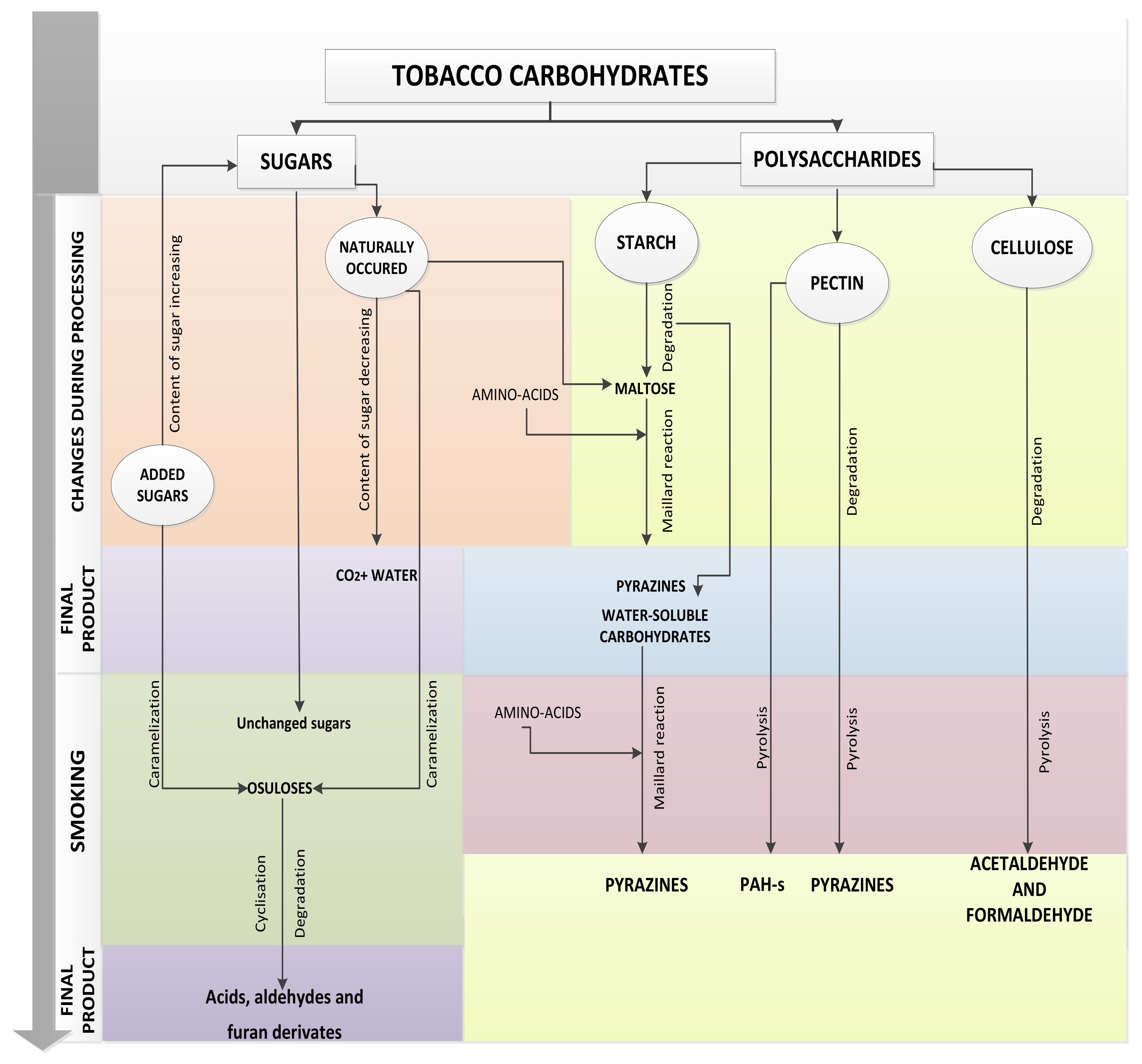
3. Changes during Processing of Tobacco Leaves
3.1. Changes in Content of Reducing Sugars
3.2. Degradation of Starch
3.3. Maillard Reactions
3.4. Caramelization
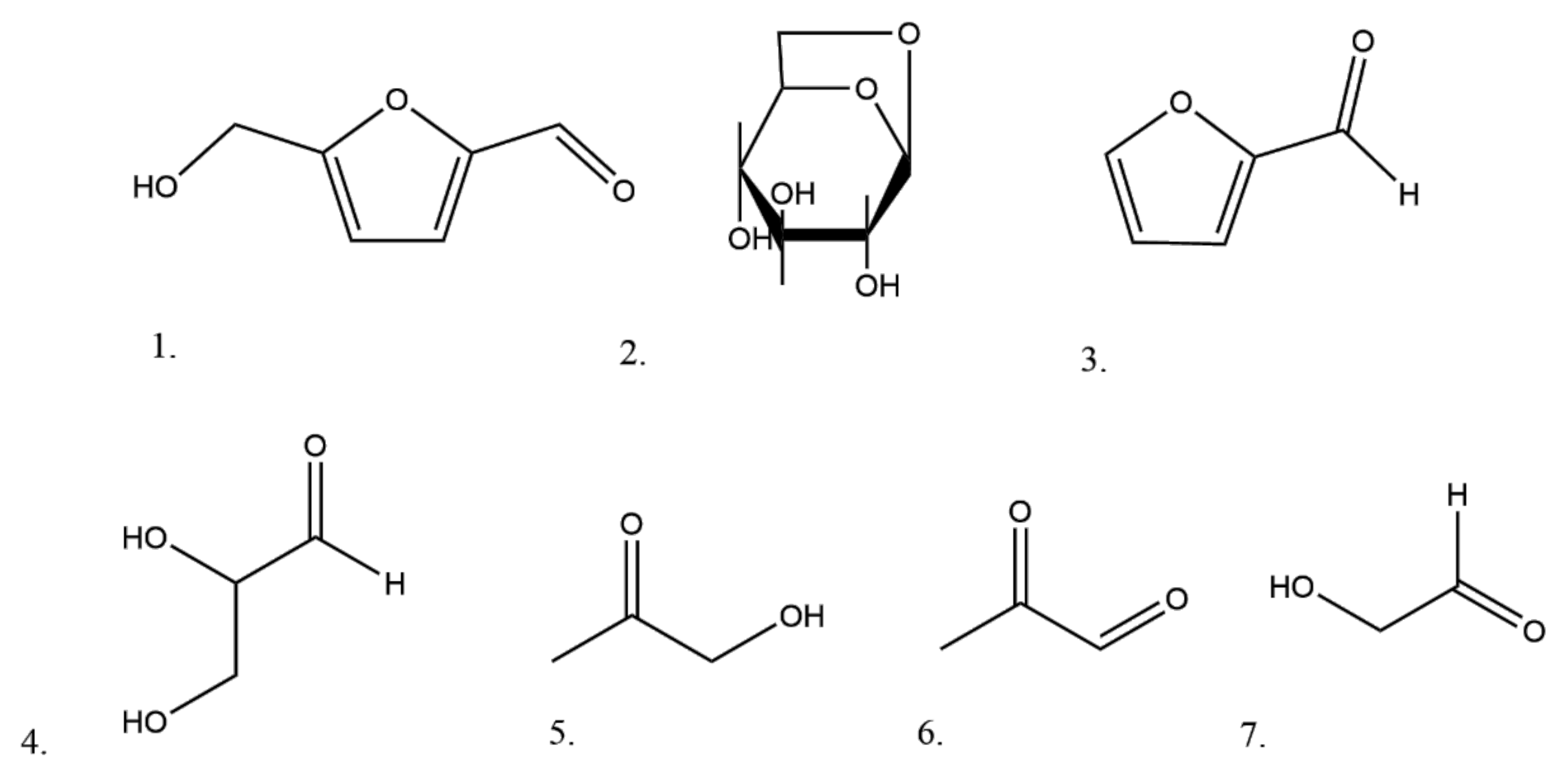
4. Changes during Smoking
Carbohydrates as Precursors of Harmful Compounds
5. Tobacco Carbohydrates as Bioactive Compounds
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tayoub, G.; Sulaiman, H.; Alorfi, M. Determination of nicotine levels in the leaves of some Nicotiana tabacum varieties cultivated in Syria. Herba Pol. 2015, 61, 23–30. [Google Scholar] [CrossRef]
- Banožić, M.; Banjari, I.; Jakovljević, M.; Šubarić, D.; Tomas, S.; Babić, J.; Jokić, S. Optimization of Ultrasound-Assisted Extraction of Some Bioactive Compounds from Tobacco Waste. Molecules 2019, 24, 1611. [Google Scholar] [CrossRef] [PubMed]
- Goubet, I.; Le Quere, J.-L.; Voilley, A.J. Retention of Aroma Compounds by Carbohydrates: Influence of Their Physicochemical Characteristics and of Their Physical State: A Review. J. Agric. Food Chem. 1998, 46, 1981–1990. [Google Scholar] [CrossRef]
- Hall, M.B.M.; Merten, S.D.R. 100-Year Review: Carbohydrates—Characterization, digestion, and utilization. J. Dairy Sci. 2017, 100, 10078–10093. [Google Scholar] [CrossRef]
- Feng, J.-W.; Zheng, S.; Maciel, G.E. EPR Investigations of Charring and Char/Air Interaction of Cellulose, Pectin, and Tobacco. Energ. Fuel. 2004, 18, 560–568. [Google Scholar] [CrossRef]
- Roemer, E.; Schorp, M.K.; Piadé, J.-J.; Seeman, J.I.; Leyden, D.E.; Haussmann, H.J. Scientific assessment of the use of sugars as cigarette tobacco ingredients: A review of published and other publicly available studies. Crit. Rev. Toxicol. 2012, 42, 244–278. [Google Scholar] [CrossRef]
- Xi, Y.X.; Song, J.Z.; Yang, J.; Li, F.; Cai, X.J.; Wang, X.M.; Wei, C.Y. Analysis of flavor precursors and degradation products content in flue-cured tobacco of different color and maturity. Acta Tab. Sin. 2011, 17, 23–30. [Google Scholar]
- Zhu, W.K.; Wang, Y.; Chen, L.Y.; Wang, Z.G.; Li, B.; Wang, B. Effect of two-stage dehydration on retention of characteristic flavor components of flue-cured tobacco in rotary dryer. Dry. Technol. 2016, 34, 1621–1629. [Google Scholar] [CrossRef]
- Mitsui, K.; David, F.; Dumont, E.; Ochiai, N.; Tamura, H.; Sandra, P. LC fractionation followed by pyrolysis GC–MS for the in-depth study of aroma compounds formed during tobacco combustion. J. Anal. Appl. Pyrolysis 2015, 116, 68–74. [Google Scholar] [CrossRef]
- Baker, R.R. The generation of formaldehyde in cigarettes—Overview and recent experiments. Food Chem. Toxicol. 2006, 44, 1799–1822. [Google Scholar] [CrossRef]
- Weeks, W.W. Chemistry of tobacco constituents influences flavor and aroma. Rec. Adv. Tob. Sci. 1985, 11, 175–200. [Google Scholar]
- Schaller, J.P.; Pijnenburg, J.P.M.; Ajithkumar, A.; Tricker, A.R. Evaluation of the Tobacco Heating System 2.2. Part 3: Influence of the tobacco blend on the formation of harmful and potentially harmful constituents of the Tobacco Heating System 2.2 aerosol. Regul. Toxicol. Pharmacol. 2016, 81, S48–S58. [Google Scholar] [CrossRef] [PubMed]
- Ali, H.; Pätzold, R.; Brückner, H. Determination of L- and D-amino acids in smokeless tobacco products and tobacco. Food Chem. 2006, 99, 803–812. [Google Scholar] [CrossRef]
- Leffingwell, J.C. Nitrogen components of leaf and their relationship to smoking quality and aroma. Rec. Adv. Tob. Sci. 1976, 2, 1–31. [Google Scholar]
- Baker, R.R.; Coburn, S.; Liu, C.; Tetteh, J. Pyrolysis of saccharide tobacco ingredients: A TGA-FTIR investigation. J. Anal. Appl. Pyrolysis 2005, 74, 171–180. [Google Scholar] [CrossRef]
- Clarke, M.B.; Bezabeh, D.Z.; Howard, C.T. Determination of Carbohydrates in Tobacco Products by Liquid Chromatography-Mass Spectrometry/Mass Spectrometry: A Comparison with Ion Chromatography and Application to Product Discrimination. J. Agric. Food Chem. 2006, 54, 1975–1981. [Google Scholar] [CrossRef]
- Geiss, O.; Kotzias, D. Tobacco, Cigarettes and Cigarette Smoke an Overview. In Institute for Health and Consumer Protection; Joint Research Center/Institute for Health and Consumer Protection (IHCP): Isra, Italy, 2007; pp. 1–72. [Google Scholar]
- Adam, T.; Ferge, T.; Mitschke, S.; Streibel, T.; Baker, R.R.; Zimmermann, R. Discrimination of three tobacco types (Burley, Virginia and Oriental) by pyrolysis single-photon ionisation? time-of-flight mass spectrometry and advanced statistical methods. Anal. Bioanal. Chem. 2004, 381, 487–499. [Google Scholar] [CrossRef]
- Leffingwell, J.C. Leaf Chemistry BA Basic Chemical Constituents of Tobacco Leaf and Differences among Tobacco Types Reprinted from Tobacco: Production, Chemistry, And Technology; Davis, D.L., Mark, T.N., Eds.; Blackwell Science (Pub.): Oxford, UK, 1999. [Google Scholar]
- Talhout, R.; Opperhuizen, A.; van Amsterdam, J.G.C. Sugars as tobacco ingredient: Effects on mainstream smoke composition. Food Chem. Toxicol. 2006, 44, 1789–1798. [Google Scholar] [CrossRef]
- Wu, M.; Wu, X.L. A study of burley material. Tob. Sci. Technol. 1996, 2, 9–11. [Google Scholar]
- Li, P.; Wu, M.; Xie, J. Changes in Levels of Amino Acids and Basic Components in Burley Tobacco Produced by Roasting. Contrib. Tob. Res. 2003, 20, 459–466. [Google Scholar] [CrossRef]
- Cai, K.; Hu, D.; Lei, B.; Zhao, H.; Pan, W.; Song, B. Determination of carbohydrates in tobacco by pressurized liquid extraction combined with a novel ultrasound-assisted dispersive liquid–liquid microextraction method. Anal. Chim. Acta 2015, 882, 90–100. [Google Scholar] [CrossRef]
- Nagai, A.; Yamamoto, T.; Wariishi, H. Identification of Fructo- and Malto-oligosaccharides in Cured Tobacco Leaves (Nicotiana tabacum). J. Agric. Food Chem. 2012, 60, 6606–6612. [Google Scholar] [CrossRef] [PubMed]
- Zook, C.M.; Patel, P.M.; LaCourse, W.R.; Ralapati, S. Characterization of Tobacco Products by High-Performance Anion Exchange Chromatography−Pulsed Amperometric Detection. J. Agric. Food Chem. 1996, 44, 1773–1779. [Google Scholar] [CrossRef]
- K Department of Health. Permitted Additives to Tobacco Products in the United Kingdom; Department of Health: London, UK, 2003. [Google Scholar]
- EU Tobacco Product Directive TPD. Tobacco Product Directive 2014/40/EU of the European Parliament and of the Council. European Union: Brussels, Belgium, 2014. [Google Scholar]
- Friedmann, M. Food browning and its prevention: An overview. J. Agric. Food Chem. 1996, 44, 631–653. [Google Scholar] [CrossRef]
- Pilon-Smits, E.A.H.; Ebskamp, M.J.M.; Paul, M.J.; Jeuken, M.J.W.; Weisbeek, P.J.; Smeekens, S.C.M. Improved performance of transgenic fructan-accumulating tobacco under drought stress. Plant Physiol. 1995, 107, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Li, T.; Gong, C. A review on starch changes in tobacco leaves during flue-curing. Front. Agric. China 2009, 3, 435–439. [Google Scholar] [CrossRef]
- Ridley, B.L.; O’Neill, M.A.; Mohnen, D. Pectins: Structure, biosynthesis, and oligogalacturonide-related signaling. Phytochemistry 2001, 57, 929–967. [Google Scholar] [CrossRef]
- Lara-Espinoza, C.; Carvajal-Millán, E.; Balandrán-Quintana, R.; López-Franco, Y.; Rascón-Chu, A. Pectin and Pectin-Based Composite Materials: Beyond Food Texture. Molecules 2018, 23, 942. [Google Scholar] [CrossRef]
- Zhang, M.; Zeng, G.; Pan, Y.; Qi, N. Difference research of pectins extracted from tobacco waste by heat reflux extraction and microwave-assisted extraction. Biocatal. Agric. Biotechnol. 2018, 15, 359–363. [Google Scholar] [CrossRef]
- Zhu, X.; Liu, B.; Zheng, S.; Gao, Y. Quantitative and structure analysis of pectin in tobacco by 13C CP/MAS NMR spectroscopy. Anal. Methods 2014, 6, 6407–6413. [Google Scholar] [CrossRef]
- Baliga, V.; Sharma, R.; Miser, D.; McGrath, T.; Hajaligol, M. Physical characterization of pyrolyzed tobacco and tobacco components. J. Anal. Appl. Pyrolysis 2003, 66, 191–215. [Google Scholar] [CrossRef]
- Liu, B.; Li, Y.-M.; Wu, S.-B.; Li, Y.-H.; Deng, S.-S.; Xia, Z.-L. Pyrolysis Characteristic of Tobacco Stem Studied by Py-GC/MS, TG-FTIR, and TG-MS. BioResources 2012, 8, 220–230. [Google Scholar] [CrossRef][Green Version]
- Tso, T.C. Alkaloids in Production, Physiology and Biochemistry of Tobacco Plant. In Tobacco-Production, Chemistry and Technology; Ideals Inc., Beltsville, M.D., Davis, D.L., Nielsen, M.T., Eds.; Blackwell Science: New Jersey, NJ, USA, 1990. [Google Scholar]
- Tuzzin, G.; Godinho, M.; Dettmer, A.; Zattera, A.J. Nanofibrillated cellulose from tobacco industry wastes. Carbohydr. Polym. 2016, 148, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Yang, X.; Dong, C.; Zhang, Z.; Zhang, X.; Zhu, X. Influence of pyrolysis temperature and time on the cellulose fast pyrolysis products: Analytical Py-GC/MS study. J. Anal. Appl. Pyrolysis 2011, 92, 430–438. [Google Scholar] [CrossRef]
- Torikaiu, K.; Uwano, Y.; Nakamori, T.; Tarora, W.; Takahashi, H. Study on tobacco components involved in the pyrolytic generation of selected smoke constituents. Food Chem. Toxicol. 2005, 43, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Sanders, E.B.; Goldsmith, A.I.; Seeman, J.I. A model that distinguishes the pyrolysis of D-glucose, D-fructose, and sucrose from that of cellulose. Application to the understanding of cigarette smoke formation. J. Anal. Appl. Pyrolysis 2002, 66, 29–50. [Google Scholar] [CrossRef]
- Yamaguchi, N.; Suzuki, S.; Makino, A. Starch degradation by alpha-amylase in tobacco leaves during the curing process. Soil Sci. Plant Nutr. 2013, 59, 904–911. [Google Scholar] [CrossRef]
- Abubakar, Y.; Young, J.H.; Johnson, W.H.; Weeks, W.W. Changes in moisture and chemical composition of flue-cured tobacco during curing. Tob. Sci. 2000, 17, 51–58. [Google Scholar] [CrossRef]
- Zilkey, B.F.; Court, W.A.; Binns, M.R.; Basrur, P.K. Chemical Studies on Canadian Tobacco and Tobacco Smok. 1. Tobacco, Tobacco Sheet, and Cigarette Smoke Chemical Analysis on Various Treatments of Bright and Burley Tobacco. Tob. Int. 1982, 184, 83–89. [Google Scholar]
- Pang, T.; Yuan, Z.; Dai, Y.; Wang, C.; Yang, J.; Peng, L.; Xu, G. Identification and determination of glycosides in tobacco leaves by liquid chromatography with atmospheric pressure chemical ionization tandem mass spectrometry. J. Sep. Sci. 2007, 30, 289–296. [Google Scholar] [CrossRef]
- Cerny, C. The Aroma Side of the Maillard Reaction. Ann. N. Y. Acad. Sci. 2008, 1126, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, T.; Schieberle, P. Identification of key aroma compounds generated from cysteine and carbohydrates under roasting conditions. Zeitschrift For Lebensmitteluntersuchung und -Forschung A 1998, 207, 229–236. [Google Scholar] [CrossRef]
- Liu, L.; Wang, X.; Wang, S.; Liu, S.; Jia, Y.; Qin, Y.; Liu, H. Simultaneous quantification of ten Amadori compounds in tobacco using liquid chromatography with tandem mass spectrometry. J. Sep. Sci. 2017, 40, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Jia, C.; Xiu, L.; Mou, D. Simultaneous determination of six kinds of Amadori 305 compounds in tobaccos by LC-MS/MS. J. Chin. Mass. Spectr. Soc. 2015, 36, 45–51. [Google Scholar]
- Zhou, W.; Wang, J.; Wu, D. Determination of important flavour precursor compounds 303 (Amadori compounds) in cigarettes by LC-MS/MS. J. Anal. Chem. 2014, 69, 691–695. [Google Scholar] [CrossRef]
- García-Moreno, M.I.; Benito, J.M.; Mellet, C.O.; Fernández, J.M.G. Chemical and Enzymatic Approaches to Carbohydrate-Derived Spiroketals: Di-D-Fructose Dianhydrides (DFAs). Molecules 2008, 13, 1640–1670. [Google Scholar] [CrossRef] [PubMed]
- Tomasik, P. The thermal decomposition of carbohydrates. Part I. The decomposition of mono-, di-, and oligo-saccharides. Adv. Carbohydr. Chem. Biochem. 1989, 47, 203–278. [Google Scholar]
- Shen, D.K.; Gu, S. The mechanism for thermal decomposition of cellulose and its main products. Bioresourc. Technol. 2009, 100, 6496–6504. [Google Scholar] [CrossRef]
- Husain, Z.; Ansari, K.B.; Chatake, V.S.; Urunkar, Y.; Pandit, A.B.; Joshi, J.B. Valorisation of biomass pellets to renewable fuel and chemicals using pyrolysis: Characterisation of pyrolysis products and its application. Ind. Chem. Eng. 2019, 1–14. [Google Scholar] [CrossRef]
- Van Nierop, L.E.; Talhout, R. Sugar as Tobacco Additive Tastes “Bitter”. J. Addict. Res. Ther. 2016, 7, 10293. [Google Scholar] [CrossRef]
- Jokić, S.; Gagić, T.; Knez, Ž.; Banožić, M.; Škerget, M. Separation of active compounds from tobacco waste using subcritical water extraction. J. Supercrit. Fluids 2019, 153, 104593. [Google Scholar] [CrossRef]
- Martin, C.; Fernandez, T.; Garcia, R.; Carrillo, E.; Marcet, M.; Galbe, M.; Jönsson, L.J. Preparation of hydrolysates from tobacco stalks and ethanolic fermentation by Saccharomyces cerevisiae. World J. Microbiol. Biotechnol. 2002, 18, 857–862. [Google Scholar] [CrossRef][Green Version]
- Azmir, J.; Zaidula, S.M.; Rahmana, M.M.; Sharif, K.M.; Mohamed, A.; Sahena, F.; Jahurul, M.H.A.; Ghafoor, K.; Norulaini, N.A.N.; Omar, A.K.M. Techniques for extraction of bioactive compounds from plant materials: A review. J. Food Eng. 2013, 117, 426–436. [Google Scholar] [CrossRef]
- Cvjetko Bubalo, M.; Vidović, S.; Radojčić Redovniković, I.; Jokić, S. Green solvents for green technologies. J. Chem. Technol. Biotechnol. 2015, 90, 1631–1639. [Google Scholar] [CrossRef]
- Xu, C.P.; Xiao, Y.; Mao, D.B. Antioxidant activities of polysaccharide fractions isolated from burley tobacco flowers. Croat. J. Food Sci. Technol. 2013, 5, 46–52. [Google Scholar]
- Liu, S.; He, P.; Tian, Z.; Li, X.; Xu, C. Ultrasound-Assisted Extraction and Characterization of Polysaccharide From Maryland Tobacco Leaves. J. Chil. Chem. Soc. 2015, 60, 2761–2768. [Google Scholar] [CrossRef][Green Version]
- Popova, V.; Ivanova, T.; Stoyanova, A.; Nikolova, V.; Hristeva, T.; Gochev, V.; Yonchev, Y.; Nikolov, N.; Zheljazkov, V.D. Terpenoids in the Essential Oil and Concentrated Aromatic Products Obtained from Nicotiana glutinosa L. Leaves. Molecules 2020, 25, 30. [Google Scholar] [CrossRef]
- Kroumova, A.B.M.; Zaitlin, D.; Wagner, G.J. Natural variability in acyl moieties of sugar esters produced by certain tobacco and other Solanaceae species. Phytochemistry 2016, 130, 218–227. [Google Scholar] [CrossRef]
- Huang, Z.; Bi, Y.J.; Sha, Y.-F.; Xie, W.Y.; Wu, D.; Liu, B.-Z. Separation and Analysis of Sucrose Esters in Tobacco by Online Liquid Chromatography–Gas Chromatography/Mass Spectrometry. Anal. Sci. 2018, 34, 887–891. [Google Scholar] [CrossRef]
- Puterka, G.J.; Farone, W.; Palmer, T.; Barrington, A. Structure-Function Relationships Affecting the Insecticidal and Miticidal Activity of Sugar Esters. J. Econ. Entomol. 2003, 96, 636–644. [Google Scholar] [CrossRef]
- Cipolla, L.; Peri, F. Carbohydrate-based bioactive compounds for medicinal chemistry applications. Mini Rev. Med. Chem. 2011, 11, 39–54. [Google Scholar] [CrossRef] [PubMed]
| (%) | TOBACCO VARIETY | ||
|---|---|---|---|
| Burley | Virginia | Oriental | |
| Nicotine | 2.07 | 2.26 | 1.18 |
| Sugars | 1.2 | 11.1 | 10.7 |
| Proteins | 10.1 | 7.8 | 7.8 |
| Chlorine | 0.51 | 0.53 | 0.84 |
| Crude Ashes | 18.4 | 12.8 | 16.4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Banožić, M.; Jokić, S.; Ačkar, Đ.; Blažić, M.; Šubarić, D. Carbohydrates—Key Players in Tobacco Aroma Formation and Quality Determination. Molecules 2020, 25, 1734. https://doi.org/10.3390/molecules25071734
Banožić M, Jokić S, Ačkar Đ, Blažić M, Šubarić D. Carbohydrates—Key Players in Tobacco Aroma Formation and Quality Determination. Molecules. 2020; 25(7):1734. https://doi.org/10.3390/molecules25071734
Chicago/Turabian StyleBanožić, Marija, Stela Jokić, Đurđica Ačkar, Marijana Blažić, and Drago Šubarić. 2020. "Carbohydrates—Key Players in Tobacco Aroma Formation and Quality Determination" Molecules 25, no. 7: 1734. https://doi.org/10.3390/molecules25071734
APA StyleBanožić, M., Jokić, S., Ačkar, Đ., Blažić, M., & Šubarić, D. (2020). Carbohydrates—Key Players in Tobacco Aroma Formation and Quality Determination. Molecules, 25(7), 1734. https://doi.org/10.3390/molecules25071734








