An MRM-Based Multiplexed Quantification Assay for Human Adipokines and Apolipoproteins
Abstract
1. Introduction
2. Results
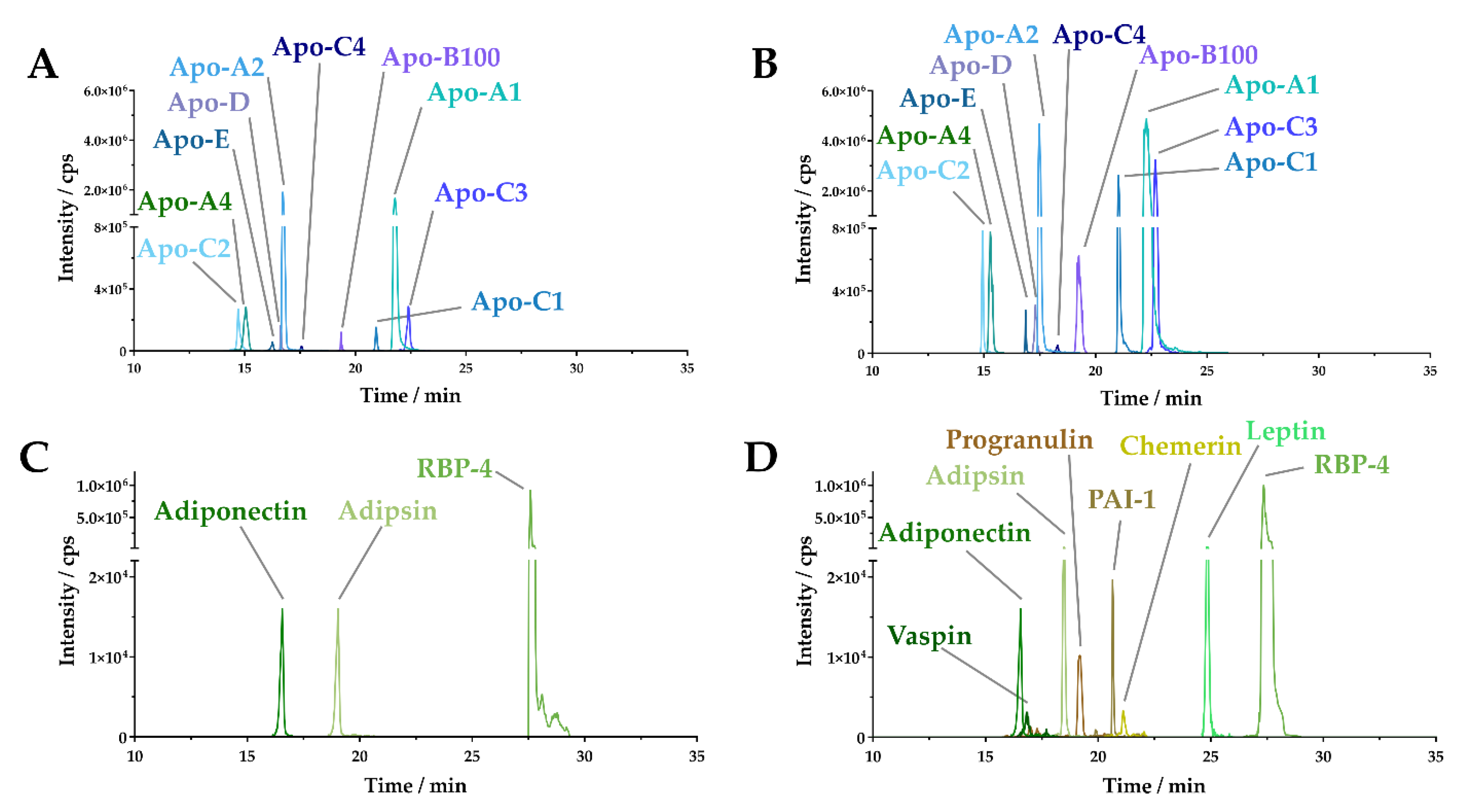
2.1. Sample Preparation and Peptide Selection
2.2. Assay Development
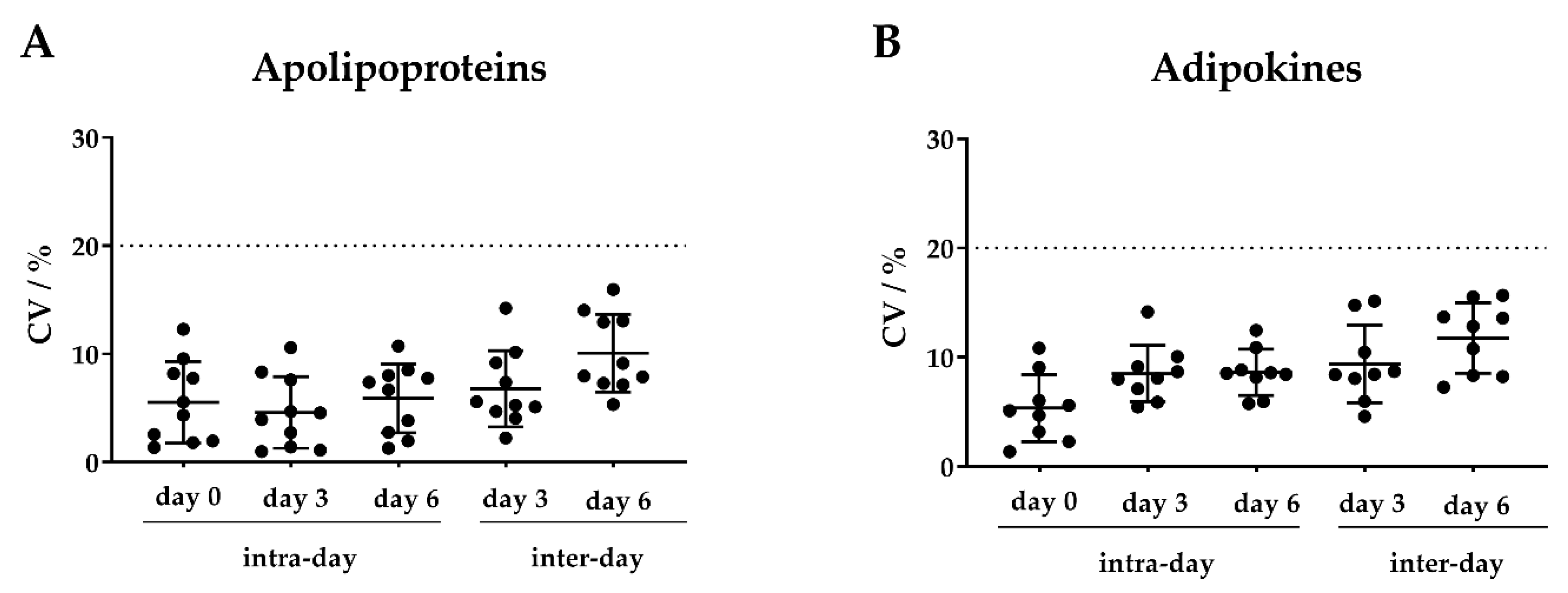
2.2.1. Quantification Characteristics and Reproducibility of the MRM Method
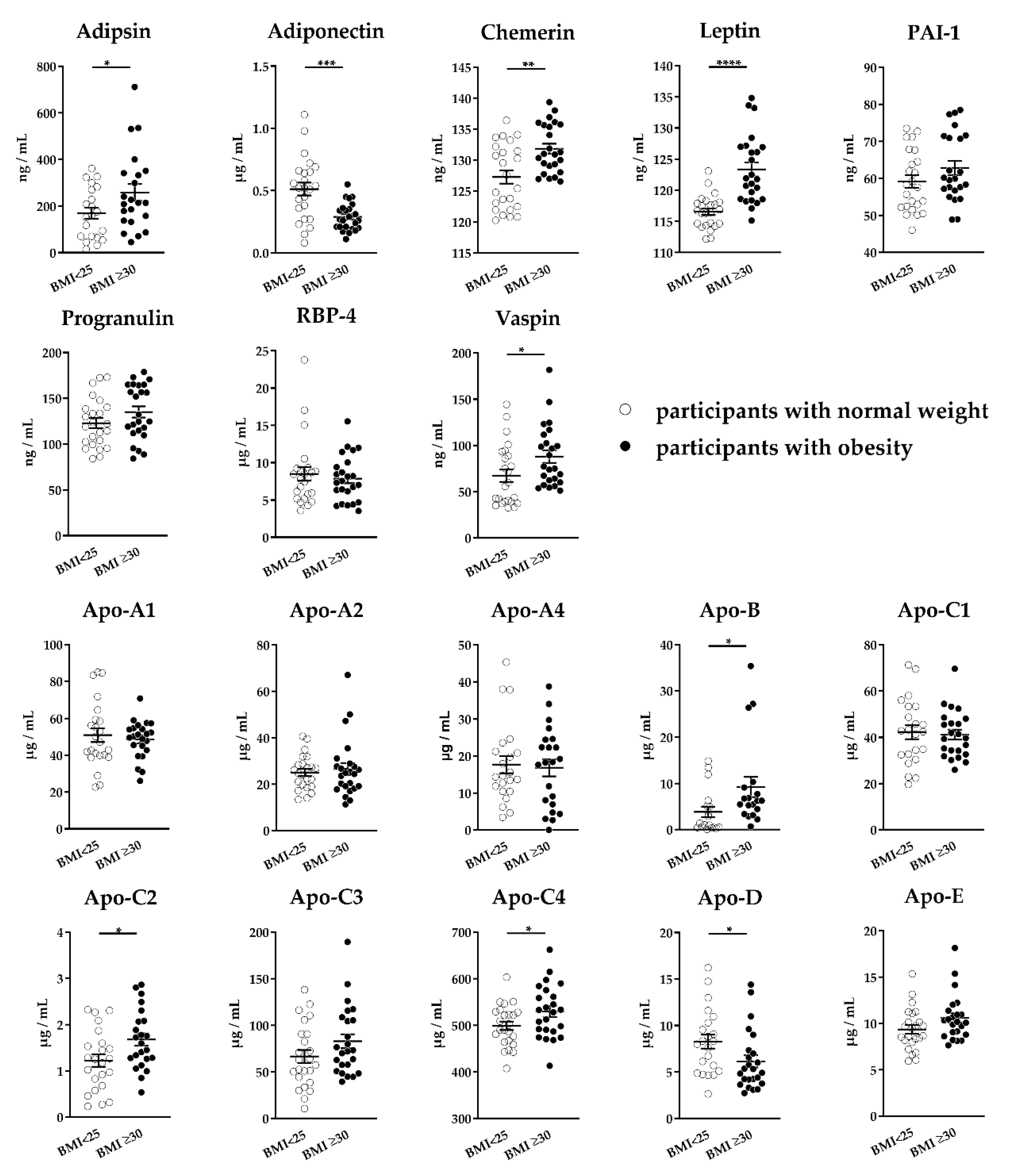
2.2.2. Application of the MRM Assay: Analysis of Human Serum from Adults
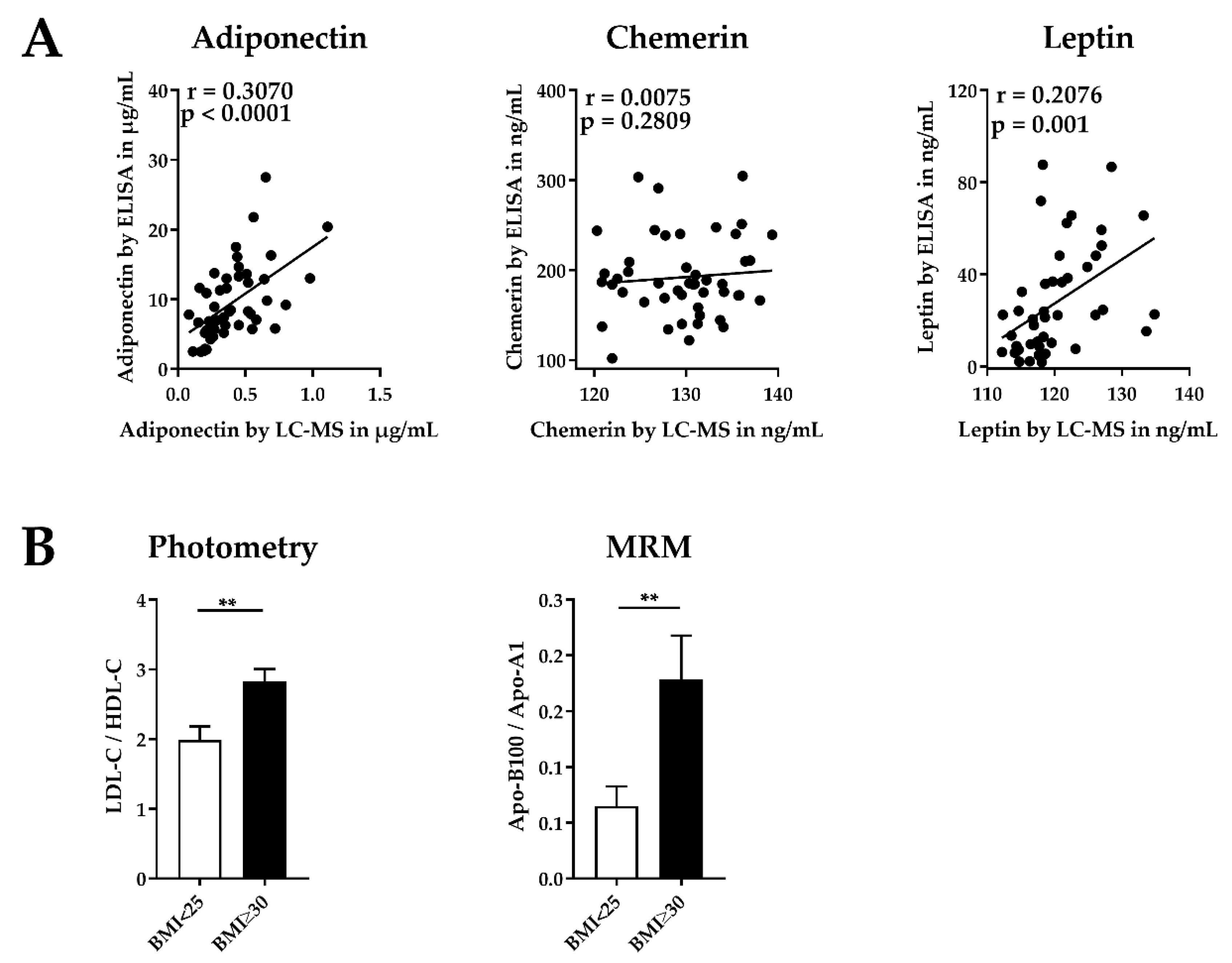
2.2.3. Validation of the MRM Method
2.3. Further Application of the MRM Method
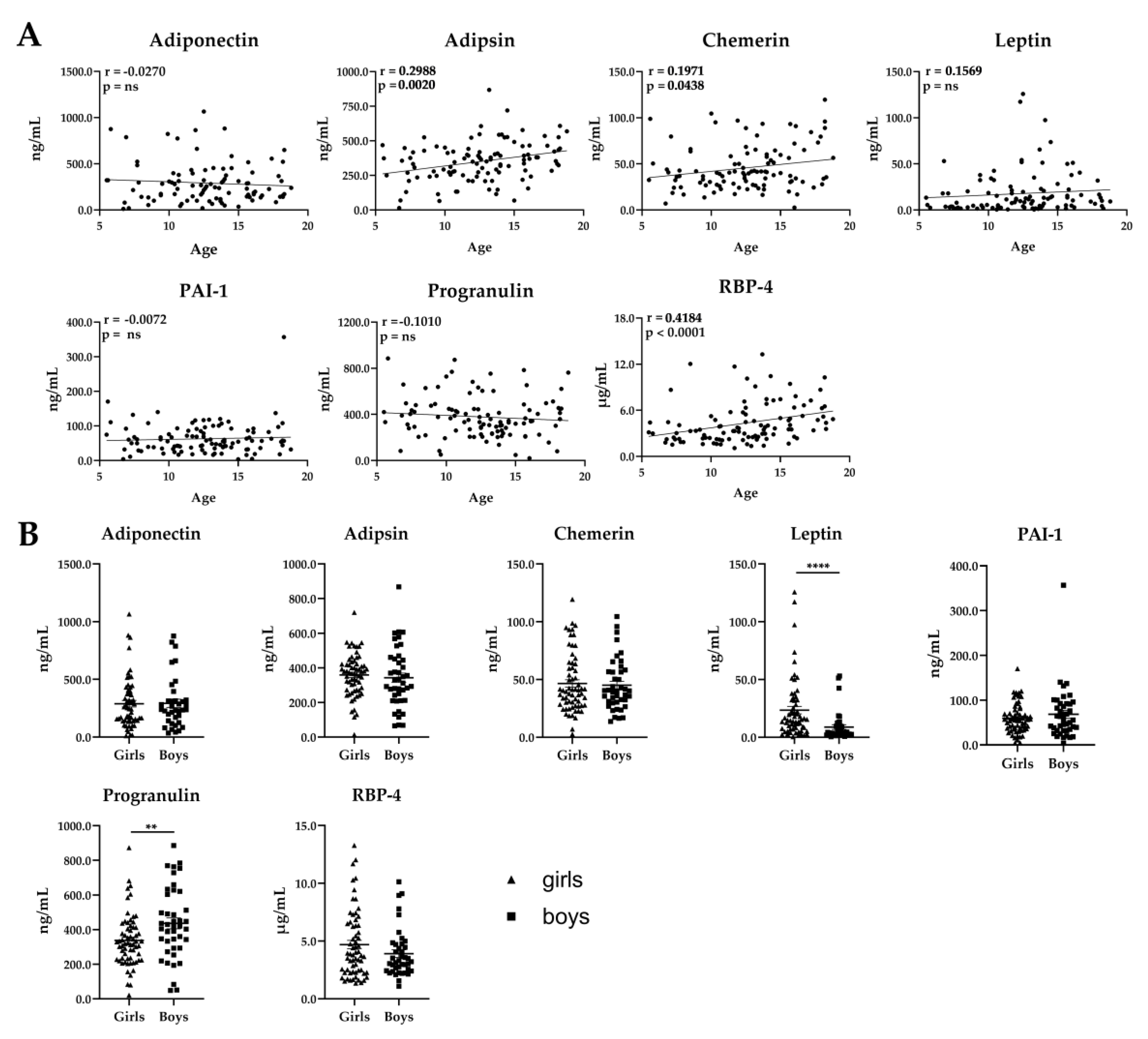
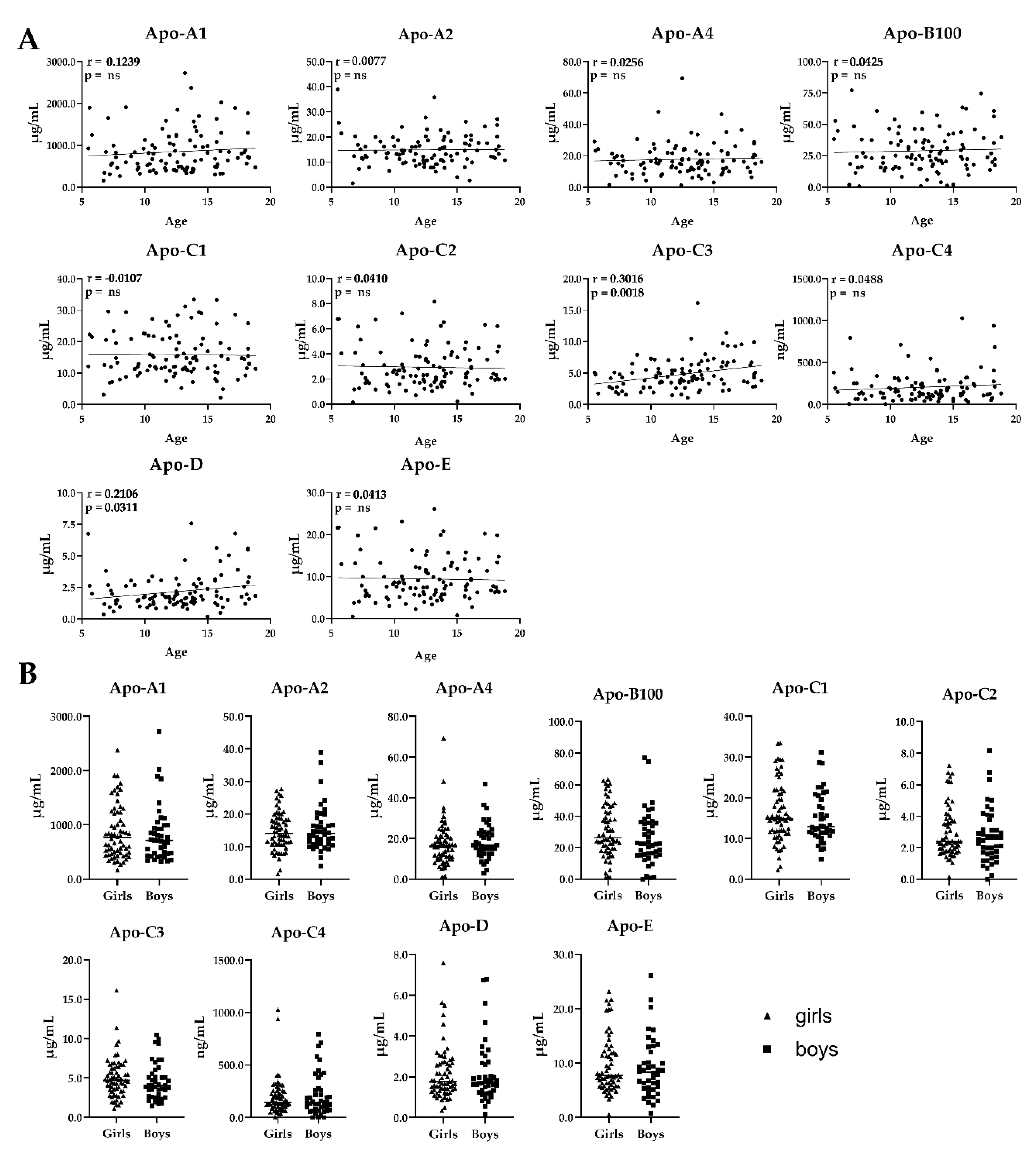
2.3.1. Analysis of Human Serum Samples from Children
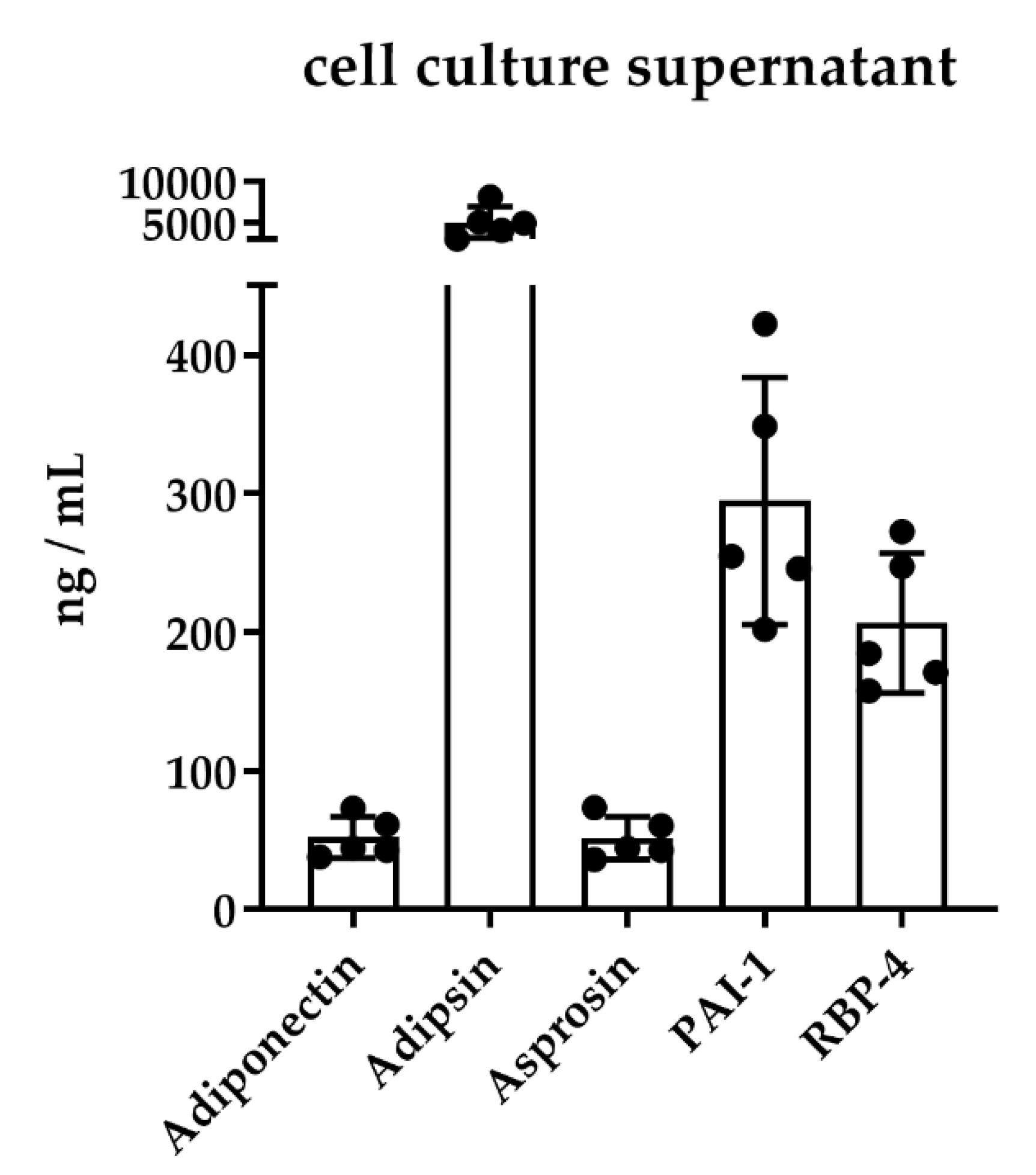
2.3.2. Analysis of Cell Culture Supernatants from Mature Human Adipocytes
3. Discussion
4. Material and Methods
4.1. Ethic Statement
4.2. Reagents Chemicals and Serum Samples
4.3. Human Serum from Adults and Children
4.3.1. Protein Depletion
4.3.2. Proteolytic Cleavage of Human Serum
4.4. Human Preadipocytes (SGBS Cells)
4.4.1. Differentiation of Human Preadipocytes
4.4.2. Proteolytic Cleavage of Cell Culture Supernatant
4.5. NanoRP-UPLC-MS/MS
4.6. Selection of Potential Candidate Peptides
4.7. Data Analysis
4.8. Validation of MRM-Based Method Using ELISA
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cao, H. Adipocytokines in obesity and metabolic disease. J. Endocrinol. 2014, 220, T47–T59. [Google Scholar] [CrossRef] [PubMed]
- Finucane, M.M.; Stevens, G.A.; Cowan, M.J.; Danaei, G.; Lin, J.K.; Paciorek, C.J.; Singh, G.M.; Gutierrez, H.R.; Lu, Y.; Bahalim, A.N.; et al. National, regional, and global trends in body-mass index since 1980: Systematic analysis of health examination surveys and epidemiological studies with 960 country-years and 9·1 million participants. Lancet 2011, 377, 557–567. [Google Scholar] [CrossRef]
- James, P.T.; Leach, R.; Kalamara, E.; Shayeghi, M. The worldwide obesity epidemic. Obesity 2001, 9. [Google Scholar] [CrossRef]
- PH Wilding, J. Causes of obesity. Pract. Diabetes 2001, 18, 288–292. [Google Scholar] [CrossRef]
- Ahima, R.S. Adipose tissue as an endocrine organ. Obesity 2006, 14. [Google Scholar] [CrossRef] [PubMed]
- Fasshauer, M.; Bluher, M. Adipokines in health and disease. Trends Pharm. Sci. 2015, 36, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Fasshauer, M.; Paschke, R. Regulation of adipocytokines and insulin resistance. Diabetologia 2003, 46, 1594–1603. [Google Scholar] [CrossRef]
- Kajimura, S. Adipose tissue in 2016: Advances in the understanding of adipose tissue biology. Nat. Rev. Endocrinol. 2017, 13, 69–70. [Google Scholar] [CrossRef]
- Chacon, M.R.; Miranda, M.; Jensen, C.H.; Fernandez-Real, J.M.; Vilarrasa, N.; Gutierrez, C.; Naf, S.; Gomez, J.M.; Vendrell, J. Human serum levels of fetal antigen 1 (FA1/Dlk1) increase with obesity, are negatively associated with insulin sensitivity and modulate inflammation in vitro. Int. J. Obes. 2008, 32, 1122–1129. [Google Scholar] [CrossRef]
- Madsbad, S. The role of glucagon-like peptide-1 impairment in obesity and potential therapeutic implications. Diabetesobes Metab 2014, 16, 9–21. [Google Scholar] [CrossRef]
- Stulnig, T. Adipositas und die Entzündung des Fettgewebes. J. Klein Endokrin Stoffwechs. 2009, 2, 17–21. [Google Scholar]
- Sommer, G.; Garten, A.; Petzold, S.; Beck-Sickinger, A.G.; Bluher, M.; Stumvoll, M.; Fasshauer, M. Visfatin/PBEF/Nampt: Structure, regulation and potential function of a novel adipokine. Clin. Sci. (Lond) 2008, 115, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Alessi, M.-C.; Poggi, M.; Juhan-Vague, I. Plasminogen activator inhibitor-1, adipose tissue and insulin resistance. Curr Opin. Lipidol. 2007, 18, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Juhan-Vague, I.; Alessi, M.C.; Mavri, A.; Morange, P. Plasminogen activator inhibitor-1, inflammation, obesity, insulin resistance and vascular risk. J. Throm. Haemost. 2003, 1, 1575–1579. [Google Scholar] [CrossRef]
- Landin, K.; Stigendal, L.; Eriksson, E.; Krotkiewski, M.; Risberg, B.; Tengborn, L.; Smith, U. Abdominal obesity is associated with an impaired fibrinolytic activity and elevated plasminogen activator inhibitor-1. Metabolism 1990, 39, 1044–1048. [Google Scholar] [CrossRef]
- Vucenik, I.; Stains, J.P. Obesity and cancer risk: Evidence, mechanisms, and recommendations. Ann. N. Y. Acad. Sci. 2012, 1271, 37–43. [Google Scholar] [CrossRef]
- Wyatt, S.B.; Winters, K.P.; Dubbert, P.M. Overweight and obesity: Prevalence, consequences, and causes of a growing public health problem. Am. J. Med. Sci. 2006, 331, 166–174. [Google Scholar] [CrossRef]
- Donma, M.M.; Donma, O. Apolipoproteins: Biochemistry, methods and clinical significance. Biochem. Educ. 1989, 17, 63–68. [Google Scholar] [CrossRef]
- Dominiczak, M.H.; Caslake, M.J. Apolipoproteins: Metabolic role and clinical biochemistry applications. Ann. Clin. Biochem. 2011, 48, 498–515. [Google Scholar] [CrossRef]
- Agger, S.A.; Marney, L.C.; Hoofnagle, A.N. Simultaneous quantification of apolipoprotein A-I and apolipoprotein B by liquid-chromatography-multiple- reaction-monitoring mass spectrometry. Clin. Chem. 2010, 56, 1804–1813. [Google Scholar] [CrossRef]
- Dittrich, J.; Adam, M.; Maas, H.; Hecht, M.; Reinicke, M.; Ruhaak, L.R.; Cobbaert, C.; Engel, C.; Wirkner, K.; Loffler, M.; et al. Targeted On-line SPE-LC-MS/MS Assay for the Quantitation of 12 Apolipoproteins from Human Blood. Proteomics 2018, 18. [Google Scholar] [CrossRef]
- Oberbach, A.; Schlichting, N.; Neuhaus, J.; Kullnick, Y.; Lehmann, S.; Heinrich, M.; Dietrich, A.; Mohr, F.W.; von Bergen, M.; Baumann, S. Establishing a reliable multiple reaction monitoring-based method for the quantification of obesity-associated comorbidities in serum and adipose tissue requires intensive clinical validation. J. Proteome Res. 2014, 13, 5784–5800. [Google Scholar] [CrossRef] [PubMed]
- Schipper, H.S.; de Jager, W.; van Dijk, M.E.; Meerding, J.; Zelissen, P.M.; Adan, R.A.; Prakken, B.J.; Kalkhoven, E. A multiplex immunoassay for human adipokine profiling. Clin. Chem. 2010, 56, 1320–1328. [Google Scholar] [CrossRef] [PubMed]
- Ali, T.M.; Al Hadidi, K. Chemerin is associated with markers of inflammation and predictors of atherosclerosis in Saudi subjects with metabolic syndrome and type 2 diabetes mellitus. J. Basic Appl. Sci. 2013, 2, 86–95. [Google Scholar] [CrossRef]
- Ziegelmeier, M.; Bachmann, A.; Seeger, J.; Lossner, U.; Kratzsch, J.; Blüher, M.; Stumvoll, M.; Fasshauer, M. Serum levels of adipokine retinol-binding protein-4 in relation to renal function. Diabetes Care 2007, 30, 2588–2592. [Google Scholar] [CrossRef] [PubMed]
- Keshishian, H.; Burgess, M.W.; Gillette, M.A.; Mertins, P.; Clauser, K.R.; Mani, D.R.; Kuhn, E.W.; Farrell, L.A.; Gerszten, R.E.; Carr, S.A. Multiplexed, Quantitative Workflow for Sensitive Biomarker Discovery in Plasma Yields Novel Candidates for Early Myocardial Injury. Mol. Cell Proteom. 2015, 14, 2375–2393. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, M.; Maruoka, S.; Katayose, S. Inverse relationship between plasma adiponectin and leptin concentrations in normal-weight and obese women. Eur. J. Endocrinol. 2002, 147, 173–180. [Google Scholar] [CrossRef]
- Sook Lee, E.; Park, S.S.; Kim, E.; Sook Yoon, Y.; Ahn, H.Y.; Park, C.Y.; Ho Yun, Y.; Woo Oh, S. Association between adiponectin levels and coronary heart disease and mortality: A systematic review and meta-analysis. Int. J. Epidemiol 2013, 42, 1029–1039. [Google Scholar] [CrossRef]
- Sweep, C.; Geurts-Moespot, J.; Grebenschikov, N.; De Witte, J.; Heuvel, J.; Schmitt, M.; Duffy, M.; Jänicke, F.; Kramer, M.; Foekens, J. External quality assessment of trans-European multicentre antigen determinations (enzyme-linked immunosorbent assay) of urokinase-type plasminogen activator (uPA) and its type 1 inhibitor (PAI-1) in human breast cancer tissue extracts. Br. J. Cancer 1998, 78, 1434. [Google Scholar] [CrossRef][Green Version]
- Fain, J.N.; Tagele, B.M.; Cheema, P.; Madan, A.K.; Tichansky, D.S. Release of 12 adipokines by adipose tissue, nonfat cells, and fat cells from obese women. Obesity (Silver Spring) 2010, 18, 890–896. [Google Scholar] [CrossRef]
- Anderson, L.; Hunter, C.L. Quantitative mass spectrometric multiple reaction monitoring assays for major plasma proteins. Mol. Cell Proteom. 2006, 5, 573–588. [Google Scholar] [CrossRef]
- Kuzyk, M.A.; Smith, D.; Yang, J.; Cross, T.J.; Jackson, A.M.; Hardie, D.B.; Anderson, N.L.; Borchers, C.H. Multiple reaction monitoring-based, multiplexed, absolute quantitation of 45 proteins in human plasma. Mol. Cell Proteom. 2009, 8, 1860–1877. [Google Scholar] [CrossRef] [PubMed]
- Yocum, A.K.; Chinnaiyan, A.M. Current affairs in quantitative targeted proteomics: Multiple reaction monitoring-mass spectrometry. Brief. Funct Genom. Proteom. 2009, 8, 145–157. [Google Scholar] [CrossRef] [PubMed]
- Ahn, Y.H.; Shin, P.M.; Oh, N.R.; Park, G.W.; Kim, H.; Yoo, J.S. A lectin-coupled, targeted proteomic mass spectrometry (MRM MS) platform for identification of multiple liver cancer biomarkers in human plasma. J. Proteom. 2012, 75, 5507–5515. [Google Scholar] [CrossRef] [PubMed]
- Anwar, M.A.; Dai, D.L.; Wilson-McManus, J.; Smith, D.; Francis, G.A.; Borchers, C.H.; McManus, B.M.; Hill, J.S.; Cohen Freue, G.V. Multiplexed LC-ESI-MRM-MS-based Assay for Identification of Coronary Artery Disease Biomarkers in Human Plasma. Proteom. Clin. Appl. 2019. [Google Scholar] [CrossRef]
- Arora, A.; Patil, V.; Kundu, P.; Kondaiah, P.; Hegde, A.S.; Arivazhagan, A.; Santosh, V.; Pal, D.; Somasundaram, K. Serum biomarkers identification by iTRAQ and verification by MRM: S100A8/S100A9 levels predict tumor-stroma involvement and prognosis in Glioblastoma. Sci. Rep. 2019, 9, 2749. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.T.; Huang, H.C.; Hsieh, Y.J.; Fu, S.H.; Li, L.; Chen, C.L.; Chu, L.J.; Yu, J.S. Targeting amine- and phenol-containing metabolites in urine by dansylation isotope labeling and liquid chromatography mass spectrometry for evaluation of bladder cancer biomarkers. J. Food Drug Anal. 2019, 27, 460–474. [Google Scholar] [CrossRef]
- Li, X.J.; Hayward, C.; Fong, P.Y.; Dominguez, M.; Hunsucker, S.W.; Lee, L.W.; McLean, M.; Law, S.; Butler, H.; Schirm, M.; et al. A blood-based proteomic classifier for the molecular characterization of pulmonary nodules. Sci. Transl. Med. 2013, 5, 207ra142. [Google Scholar] [CrossRef]
- Liao, H.; Wu, J.; Kuhn, E.; Chin, W.; Chang, B.; Jones, M.D.; O’Neil, S.; Clauser, K.R.; Karl, J.; Hasler, F.; et al. Use of mass spectrometry to identify protein biomarkers of disease severity in the synovial fluid and serum of patients with rheumatoid arthritis. Arthritis Rheum 2004, 50, 3792–3803. [Google Scholar] [CrossRef]
- Mc Ardle, A.; Flatley, B.; Pennington, S.R.; FitzGerald, O. Early biomarkers of joint damage in rheumatoid and psoriatic arthritis. Arthritis Res. 2015, 17, 141. [Google Scholar] [CrossRef]
- Wölter, M.; Okai, C.A.; Smith, D.S.; Ruß, M.; Rath, W.; Pecks, U.; Borchers, C.H.; Glocker, M.O. Maternal Apolipoprotein B100 Serum Levels are Diminished in Pregnancies with Intrauterine Growth Restriction and Differentiate from Controls. Proteom. Clin. Appl 2018, 12, 1800017. [Google Scholar]
- Yang, M.; Yu, Z.; Deng, S.; Chen, X.; Chen, L.; Guo, Z.; Zheng, H.; Chen, L.; Cai, D.; Wen, B.; et al. A Targeted Metabolomics MRM-MS Study on Identifying Potential Hypertension Biomarkers in Human Plasma and Evaluating Acupuncture Effects. Sci. Rep. 2016, 6, 25871. [Google Scholar] [CrossRef] [PubMed]
- Yannell, K.E.; Ferreira, C.R.; Tichy, S.E.; Cooks, R.G. Multiple reaction monitoring (MRM)-profiling with biomarker identification by LC-QTOF to characterize coronary artery disease. Analyst 2018, 143, 5014–5022. [Google Scholar] [CrossRef] [PubMed]
- Wabitsch, M.; Brenner, R.; Melzner, I.; Braun, M.; Möller, P.; Heinze, E.; Debatin, K.-M.; Hauner, H. Characterization of a human preadipocyte cell strain with high capacity for adipose differentiation. Int. J. Obes. 2001, 25, 8. [Google Scholar] [CrossRef] [PubMed]
- Derdemezis, C.S.; Kiortsis, D.N.; Tsimihodimos, V.; Petraki, M.P.; Vezyraki, P.; Elisaf, M.S.; Tselepis, A.D. Effect of plant polyphenols on adipokine secretion from human SGBS adipocytes. Biochem. Res. Int. 2011, 2011, 285618. [Google Scholar] [CrossRef] [PubMed]
- Korner, A.; Wabitsch, M.; Seidel, B.; Fischer-Posovszky, P.; Berthold, A.; Stumvoll, M.; Bluher, M.; Kratzsch, J.; Kiess, W. Adiponectin expression in humans is dependent on differentiation of adipocytes and down-regulated by humoral serum components of high molecular weight. Biochem. Biophys. Res. Commun. 2005, 337, 540–550. [Google Scholar] [CrossRef]
- Wang, B.; Jenkins, J.R.; Trayhurn, P. Expression and secretion of inflammation-related adipokines by human adipocytes differentiated in culture: Integrated response to TNF-α. Am. J. Physiol. 2005. [Google Scholar] [CrossRef]
- Kiyonami, R.; Schoen, A.; Prakash, A.; Peterman, S.; Zabrouskov, V.; Picotti, P.; Aebersold, R.; Huhmer, A.; Domon, B. Increased selectivity, analytical precision, and throughput in targeted proteomics. Mol. Cell Proteom. 2011, 10. [Google Scholar] [CrossRef]
- Urisman, A.; Levin, R.S.; Gordan, J.D.; Webber, J.T.; Hernandez, H.; Ishihama, Y.; Shokat, K.M.; Burlingame, A.L. An Optimized Chromatographic Strategy for Multiplexing In Parallel Reaction Monitoring Mass Spectrometry: Insights from Quantitation of Activated Kinases. Mol. Cell Proteom. 2017, 16, 265–277. [Google Scholar] [CrossRef]
- Burgess, M.W.; Keshishian, H.; Mani, D.R.; Gillette, M.A.; Carr, S.A. Simplified and efficient quantification of low-abundance proteins at very high multiplex via targeted mass spectrometry. Mol. Cell Proteom. 2014, 13, 1137–1149. [Google Scholar] [CrossRef]
- Elshal, M.F.; McCoy, J.P. Multiplex bead array assays: Performance evaluation and comparison of sensitivity to ELISA. Methods 2006, 38, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Mattern, A.; Zellmann, T.; Beck-Sickinger, A.G. Processing, signaling, and physiological function of chemerin. Iubmb Life 2014, 66, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Tilg, H.; Moschen, A.R. Adipocytokines: Mediators linking adipose tissue, inflammation and immunity. Nat. Rev. Immunol. 2006, 6, 772. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.M.; Youn, B.-S.; Lee, H.; Lee, N.; Min, S.-S.; Kwak, S.H.; Lee, H.K.; Park, K.S. Plasma Retinol-Binding Protein-4 Concentrations Are Elevated in Human Subjects With Impaired Glucose Tolerance and Type 2 Diabetes. Diabetes Care 2006, 29, 2457–2461. [Google Scholar] [CrossRef] [PubMed]
- Abola, M.V.; Thompson, C.L.; Chen, Z.; Chak, A.; Berger, N.A.; Kirwan, J.P.; Li, L. Serum levels of retinol-binding protein 4 and risk of colon adenoma. Endocr-Rel Cancer 2015, 22, L1–L4. [Google Scholar] [CrossRef] [PubMed]
- Ülgen, F.; Herder, C.; Kühn, M.C.; Willenberg, H.S.; Schott, M.; Scherbaum, W.A.; Schinner, S. Association of serum levels of retinol-binding protein 4 with male sex but not with insulin resistance in obese patients. Arch. Physiol. Biochem. 2010, 116, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Poveda, N.E.; Garces, M.F.; Ruiz-Linares, C.E.; Varon, D.; Valderrama, S.; Sanchez, E.; Castiblanco-Cortes, A.; Agudelo-Zapata, Y.; Sandoval-Alzate, H.F.; Leal, L.G.; et al. Serum Adipsin Levels throughout Normal Pregnancy and Preeclampsia. Sci. Rep. 2016, 6, 20073. [Google Scholar] [CrossRef]
- Schmid, A.; Hochberg, A.; Berghoff, M.; Schlegel, J.; Karrasch, T.; Kaps, M.; Schaffler, A. Quantification and regulation of adipsin in human cerebrospinal fluid (CSF). Clin. Endocrinol. (Oxf) 2015. [Google Scholar] [CrossRef]
- Meeter, L.H.H.; Patzke, H.; Loewen, G.; Dopper, E.G.P.; Pijnenburg, Y.A.L.; van Minkelen, R.; van Swieten, J.C. Progranulin Levels in Plasma and Cerebrospinal Fluid in Granulin Mutation Carriers. Dement. Geriatr. Cogn. Disord. 2016, 6, 330–340. [Google Scholar] [CrossRef]
- Gibbons, L.; Rollinson, S.; Thompson, J.C.; Robinson, A.; Davidson, Y.S.; Richardson, A.; Neary, D.; Pickering-Brown, S.M.; Snowden, J.S.; Mann, D.M. Plasma levels of progranulin and interleukin-6 in frontotemporal lobar degeneration. Neurobiol Aging 2015, 36, 1603.e1–1603.e4. [Google Scholar] [CrossRef]
- Kadoglou, N.P.; Gkontopoulos, A.; Kapelouzou, A.; Fotiadis, G.; Theofilogiannakos, E.K.; Kottas, G.; Lampropoulos, S. Serum levels of vaspin and visfatin in patients with coronary artery disease—Kozani study. Clin. Chim. Acta 2011, 412, 48–52. [Google Scholar] [CrossRef] [PubMed]
- Youn, B.-S.; Klöting, N.; Kratzsch, J.; Lee, N.; Park, J.W.; Song, E.-S.; Ruschke, K.; Oberbach, A.; Fasshauer, M.; Stumvoll, M.; et al. Serum Vaspin Concentrations in Human Obesity and Type 2 Diabetes. Diabetes 2008, 57, 372–377. [Google Scholar] [CrossRef] [PubMed]
- Aust, G.; Richter, O.; Rohm, S.; Kerner, C.; Hauss, J.; Klöting, N.; Ruschke, K.; Kovacs, P.; Youn, B.-S.; Blüher, M. Vaspin serum concentrations in patients with carotid stenosis. Atherosclerosis 2009, 204, 262–266. [Google Scholar] [CrossRef] [PubMed]
- Auguet, T.; Quintero, Y.; Riesco, D.; Morancho, B.; Terra, X.; Crescenti, A.; Broch, M.; Aguilar, C.; Olona, M.; Porras, J.A.; et al. New adipokines vaspin and omentin. Circulating levels and gene expression in adipose tissue from morbidly obese women. BMC Med. Genet. 2011, 12, 60. [Google Scholar] [CrossRef] [PubMed]
- Hassanin, A.A.M.; Elhusien, A.K.A.; Osman, A.M. Does obesity affect the plasma level of Plasminogen Activator Inhibitor-1? And does CO2 pneumoperitoneum affect it? Egypt J. Anaesth 2013, 29, 203–206. [Google Scholar] [CrossRef][Green Version]
- Lopez-Legarrea, P.; Mansego, M.; Zulet, M.; Alfredo, M. SERPINE1, PAI-1 protein coding gene, methylation levels and epigenetic relationships with adiposity changes in obese subjects with metabolic syndrome features under dietary restriction. J. Clin. Biochem. Nutr. 2013, 53, 139–144. [Google Scholar] [CrossRef]
- Somodi, S.; Seres, I.; Lörincz, H.; Harangi, M.; Fülöp, P.; Paragh, G. Plasminogen Activator Inhibitor-1 Level Correlates with Lipoprotein Subfractions in Obese Nondiabetic Subjects. Int. J. Endocrin 2018, 2018, 9. [Google Scholar] [CrossRef]
- Booth, N.; Simpson, A.; Croll, A.; Bennett, B.; MacGregor, I. Plasminogen activator inhibitor (PAI-1) in plasma and platelets. Br. J. Haematol. 1988, 70, 327–333. [Google Scholar] [CrossRef]
- Wang, C.; Wu, W.K.; Liu, X.; To, K.F.; Chen, G.G.; Yu, J.; Ng, E.K. Increased serum chemerin level promotes cellular invasiveness in gastric cancer: A clinical and experimental study. Peptides 2014, 51, 131–138. [Google Scholar] [CrossRef]
- Haider, D.G.; Schaller, G.; Wolzt, M.; Schindler, K.; Ludvik, B.; Prager, G. Increased Plasma Visfatin Concentrations in Morbidly Obese Subjects Are Reduced after Gastric Banding. J. Clin. Endocrinol. Metab. 2006, 91, 1578–1581. [Google Scholar] [CrossRef]
- Jürimäe, J.; Jürimäe, T. Plasma adiponectin concentration in healthy pre- and postmenopausal women: Relationship with body composition, bone mineral, and metabolic variables. Am. J. Physiol. 2007, 293, E42–E47. [Google Scholar] [CrossRef] [PubMed]
- Minocci, A.; Savia, G.; Lucantoni, R.; Berselli, M.E.; Tagliaferri, M.; Calò, G.; Petroni, M.L.; de Medici, C.; Viberti, G.C.; Liuzzi, A. Leptin plasma concentrations are dependent on body fat distribution in obese patients. Int. J. Obes. 2000, 24, 1139. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.W.; Gingerich, R.; Ostlund, R.E., Jr.; Klein, S. Relation between plasma leptin concentration and body fat, gender, diet, age, and metabolic covariates. J. Clin. Endocrinol. Metab. 1996, 81, 3909–3913. [Google Scholar]
- Dickson, D.W.; Bisceglio, G.; Crook, J.; Swanson, K.; Baker, M.; Finch, N.; Surtees, R.; Crook, R.; Rademakers, R.; Younkin, S.G.; et al. Plasma progranulin levels predict progranulin mutation status in frontotemporal dementia patients and asymptomatic family members. Brain 2009, 132, 583–591. [Google Scholar]
- Bachmann, A.; Seeger, J.; Ziegelmeier, M.; Lössner, U.; Blüher, M.; Stumvoll, M.; Fasshauer, M.; Kratzsch, J.r. Serum Levels of the Adipokine Vaspin in Relation to Metabolic and Renal Parameters. J. Clin. Endocrinol. Metab. 2008, 93, 247–251. [Google Scholar]
- Li, K.; Li, L.; Yang, M.; Liu, H.; Liu, D.; Yang, H.; Boden, G.; Yang, G. Short-term continuous subcutaneous insulin infusion decreases the plasma vaspin levels in patients with type 2 diabetes mellitus concomitant with improvement in insulin sensitivity. Eur. J. Endocrinol. 2011, 164, 905. [Google Scholar] [CrossRef]
- Ouchi, N.; Kihara, S.; Funahashi, T.; Matsuzawa, Y.; Walsh, K. Obesity, adiponectin and vascular inflammatory disease. Curr. Opin. Lipidol. 2003, 14, 561–566. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Du, X.Y.; Zhao, L.; Morser, J.; Leung, L.L. Proteolytic cleavage of chemerin protein is necessary for activation to the active form, Chem157S, which functions as a signaling molecule in glioblastoma. J. Biol. Chem. 2011, 286, 39510–39519. [Google Scholar] [CrossRef]
- Dandona, P.; Aljada, A.; Bandyopadhyay, A. Inflammation: The link between insulin resistance, obesity and diabetes. Trends Immunol. 2004, 25, 4–7. [Google Scholar] [CrossRef]
- Horakova, D.; Azeem, K.; Benesova, R.; Pastucha, D.; Horak, V.; Dumbrovska, L.; Martinek, A.; Novotny, D.; Svagera, Z.; Hobzova, M.; et al. Total and High Molecular Weight Adiponectin Levels and Prediction of Cardiovascular Risk in Diabetic Patients. Int. J. Endocrinol. 2015, 2015, 545068. [Google Scholar] [CrossRef]
- Weigert, J.; Neumeier, M.; Wanninger, J.; Filarsky, M.; Bauer, S.; Wiest, R.; Farkas, S.; Scherer, M.N.; Schäffler, A.; Aslanidis, C.; et al. Systemic chemerin is related to inflammation rather than obesity in type 2 diabetes. Clin. Endocrinol. 2010, 72, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Klöting, N.; Fasshauer, M.; Dietrich, A.; Kovacs, P.; Schön, M.R.; Kern, M.; Stumvoll, M.; Blüher, M. Insulin-sensitive obesity. Am. J. Physiol. 2010, 299, E506–E515. [Google Scholar] [CrossRef] [PubMed]
- Youn, B.-S.; Bang, S.-I.; Klöting, N.; Park, J.W.; Lee, N.; Oh, J.-E.; Pi, K.-B.; Lee, T.H.; Ruschke, K.; Fasshauer, M.; et al. Serum Progranulin Concentrations May Be Associated With Macrophage Infiltration Into Omental Adipose Tissue. Diabetes 2009, 58, 627–636. [Google Scholar] [CrossRef]
- Haider, D.G.; Bohdjalian, A.; Prager, G.; Luger, A.; Schindler, K.; Wolzt, M.; Ludvik, B. Serum Retinol-Binding Protein 4 Is Reduced after Weight Loss in Morbidly Obese Subjects. J. Clin. Endocrinol. Metab. 2007, 92, 1168–1171. [Google Scholar] [CrossRef] [PubMed]
- Volberg, V.; Heggeseth, B.; Harley, K.; Huen, K.; Yousefi, P.; Davé, V.; Tyler, K.; Vedar, M.; Eskenazi, B.; Holland, N. Adiponectin and Leptin Trajectories in Mexican-American Children from Birth to 9 Years of Age. PLoS ONE 2013, 8, e77964. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Liu, X.; Brickman, W.J.; Christoffel, K.K.; Zimmerman, D.; Tsai, H.-J.; Wang, G.; Wang, B.; Li, Z.; Tang, G.; et al. Association of Plasma Leptin Concentrations with Adiposity Measurements in Rural Chinese Adolescents. J. Clin. Endocrinol. Metab. 2009, 94, 3497–3504. [Google Scholar] [CrossRef]
- Schultz, S.; Saalbach, A.; Heiker, J.T.; Meier, R.; Zellmann, T.; Simon, J.C.; Beck-Sickinger, A.G. Proteolytic activation of prochemerin by kallikrein 7 breaks an ionic linkage and results in C-terminal rearrangement. Biochem. J. 2013, 452, 271–280. [Google Scholar] [CrossRef]
- Jouy, F.; Müller, S.A.; Wagner, J.; Otto, W.; von Bergen, M.; Tomm, J.M. Integration of conventional quantitative and phospho-proteomics reveals new elements in activated Jurkat T-cell receptor pathway maintenance. Proteomics 2015, 15, 25–33. [Google Scholar] [CrossRef]
Sample Availability: The mass spectrometry data have been deposited to the PRIDE Archive via PRIDE partner repository (PXD013829). |
| Protein | External Calibration Curve | LOD | LLOQ | ||||
|---|---|---|---|---|---|---|---|
| Slope | Intercept | R2 | Peptide (ng/mL) | Protein (ng/mL) | Peptide (ng/mL) | Protein (ng/mL) | |
| Adipsin | 3.11104 | 1.41106 | 0.97 | 0.63 | 21.63 | 1.88 | 64.88 |
| Adiponectin | 3.22104 | −7.72104 | 0.99 | 0.71 | 13.21 | 2.13 | 39.62 |
| Asprosin | 2.11104 | −4.50106 | 0.96 | 6.02 | 85.00 | 18.05 | 255.00 |
| Chemerin | 9.71104 | −5.97105 | 0.99 | 1.91 | 27.93 | 5.74 | 83.78 |
| Leptin | 9.71104 | −5.97105 | 0.99 | 2.30 | 37.28 | 6.89 | 111.85 |
| PAI-1 | 3.78105 | −1.29106 | 0.99 | 0.34 | 22.53 | 1.03 | 67.59 |
| Progranulin | 7.53104 | 2.08105 | 0.99 | 0.50 | 31.77 | 1.51 | 95.32 |
| RBP-4 | 1.27105 | −2.54106 | 0.99 | 30.09 | 575.25 | 90.27 | 1725.75 |
| Vaspin | 1.20105 | −9.41104 | 0.99 | 0.44 | 23.59 | 1.31 | 70.76 |
| Apo-A1 | 7.32103 | −6.99105 | 0.98 | 46.86 | 1025.93 | 140.57 | 3077.80 |
| Apo-A2 | 5.08104 | 3.12105 | 0.99 | 1.58 | 18.63 | 4.74 | 55.88 |
| Apo-A4 | 1.97104 | 3.55104 | 0.99 | 1.65 | 75.67 | 4.94 | 227.00 |
| Apo-B100 | 1.62104 | 8.44103 | 0.99 | 1.91 | 859.34 | 5.72 | 2578.03 |
| Apo-C1 | 2.69103 | −4.43104 | 0.99 | 4.04 | 31.11 | 12.11 | 93.32 |
| Apo-C2 | 6.02104 | 5.98105 | 0.99 | 1.73 | 18.81 | 5.20 | 56.42 |
| Apo-C3 | 5.00103 | −1.19105 | 0.99 | 20.03 | 180.87 | 60.08 | 542.60 |
| Apo-C4 | 3.65104 | −7.26104 | 0.99 | 1.80 | 24.26 | 5.39 | 72.77 |
| Apo-D | 4.03104 | 2.40105 | 0.99 | 2.06 | 35.46 | 6.18 | 106.38 |
| Apo-E | 1.30104 | −1.47105 | 0.99 | 5.01 | 120.51 | 15.03 | 361.54 |
| Protein | MRM | Literature (ELISA) |
|---|---|---|
| Adipsin | 15.2–711.4 ng/mL | 467–5148 ng/mL [57,58] |
| Adiponectin | 0.1–1.1 µg/mL | 0.02–10.3 µg/mL [70,71,80] |
| Chemerin | 120.3–139.4 ng/mL | 0.6–204.8 ng/mL [24,69,81] |
| Leptin | 112.2–134.84 ng/mL | 1.2–101.3 ng/mL [71,72,81,82] |
| PAI-1 | 46.01–78.5 ng/mL | 1.8–270 ng/mL [65,66,67,68] |
| Progranulin | 84.1–178.9 ng/mL | 5.2–386.0 ng/mL [59,74,83] |
| RBP-4 | 3.5–23.8 µg/mL | 1.5–163 µg/mL [22,55,56,84] |
| Vaspin | 32.6–181.7 ng/mL | 0.3–17.9 ng/mL [61,62,63,64,82] |
| Normal Weight (n = 25) | Obese (n = 25) | |
|---|---|---|
| Sex (w/m) | 21/4 | 16/9 |
| Age (years) | 51.2 ± 18.0 | 54.1 ± 11.2 |
| Body weight (kg) | 61.3 ± 10.2 | 129.0 ± 21.7 |
| BMI (kg/m2) | 21.7 ± 2.5 | 46.6 ± 6.4 |
| HDL-cholesterol (mmol/L) | 1.7 ± 0.4 | 1.3 ± 0.3 |
| LDL-cholesterol (mmol/L) | 3.2 ± 0.8 | 3.6 ± 0.8 |
| Type 2 Diabetes (yes/no) | 6/19 | 19/6 |
| Girls (n = 64) | Boys (n = 44) | |
|---|---|---|
| Age (years) | 12.8 ± 3.6 | 12.2 ± 3.2 |
| BMI (SDS) | −0.3 ± 0.8 | −0.2 ± 0.7 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krieg, L.; Schaffert, A.; Kern, M.; Landgraf, K.; Wabitsch, M.; Beck-Sickinger, A.G.; Körner, A.; Blüher, M.; von Bergen, M.; Schubert, K. An MRM-Based Multiplexed Quantification Assay for Human Adipokines and Apolipoproteins. Molecules 2020, 25, 775. https://doi.org/10.3390/molecules25040775
Krieg L, Schaffert A, Kern M, Landgraf K, Wabitsch M, Beck-Sickinger AG, Körner A, Blüher M, von Bergen M, Schubert K. An MRM-Based Multiplexed Quantification Assay for Human Adipokines and Apolipoproteins. Molecules. 2020; 25(4):775. https://doi.org/10.3390/molecules25040775
Chicago/Turabian StyleKrieg, Laura, Alexandra Schaffert, Matthias Kern, Kathrin Landgraf, Martin Wabitsch, Annette G. Beck-Sickinger, Antje Körner, Matthias Blüher, Martin von Bergen, and Kristin Schubert. 2020. "An MRM-Based Multiplexed Quantification Assay for Human Adipokines and Apolipoproteins" Molecules 25, no. 4: 775. https://doi.org/10.3390/molecules25040775
APA StyleKrieg, L., Schaffert, A., Kern, M., Landgraf, K., Wabitsch, M., Beck-Sickinger, A. G., Körner, A., Blüher, M., von Bergen, M., & Schubert, K. (2020). An MRM-Based Multiplexed Quantification Assay for Human Adipokines and Apolipoproteins. Molecules, 25(4), 775. https://doi.org/10.3390/molecules25040775





