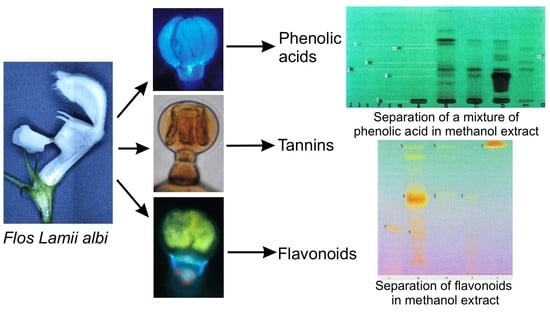
Phenolic Constituents of Lamium album L. subsp. album Flowers: Anatomical, Histochemical, and Phytochemical Study
Abstract
:1. Introduction
2. Results
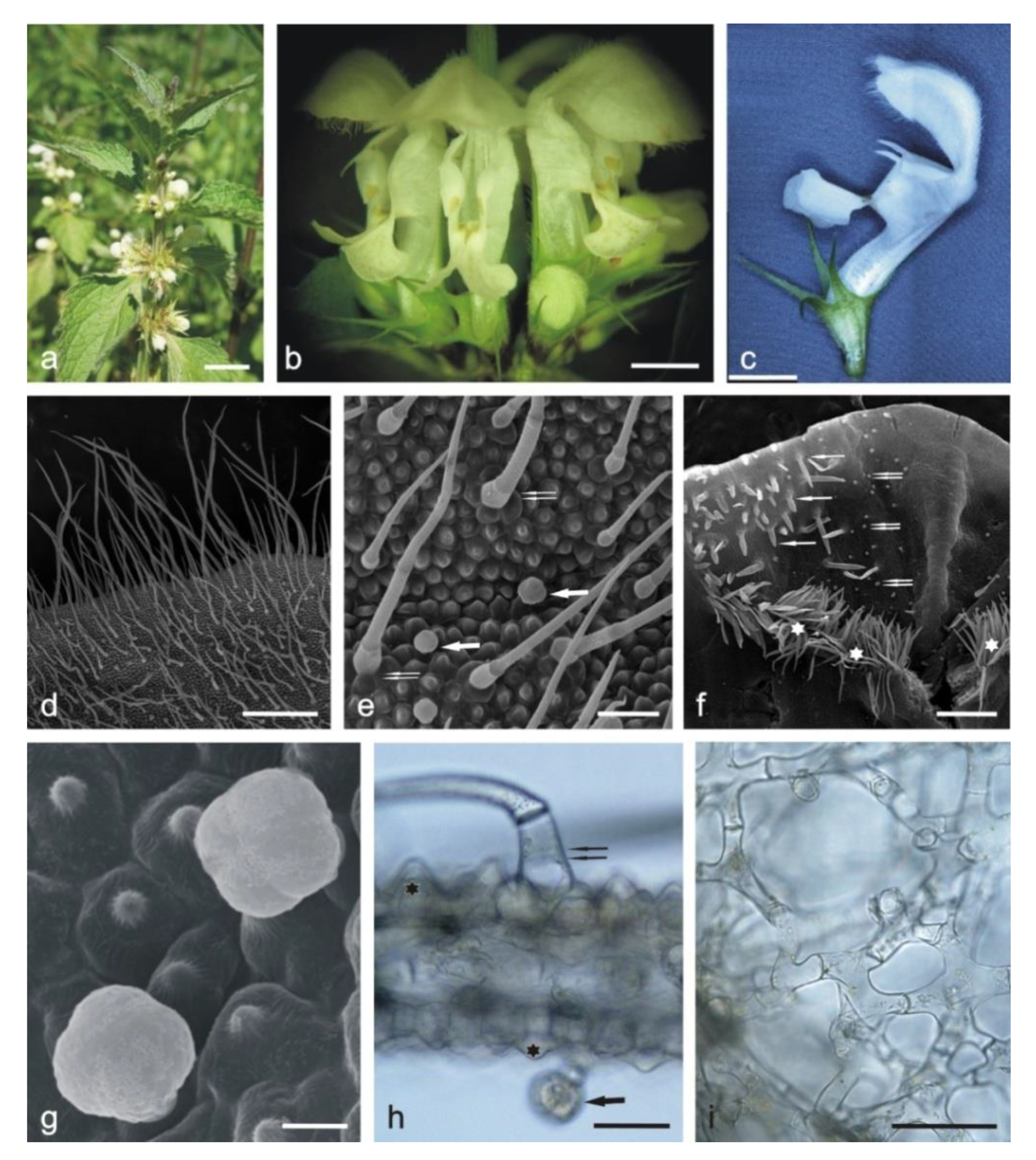
2.1. Anatomical Analyses
2.1.1. Corolla
2.1.2. Calyx
2.1.3. Stamens
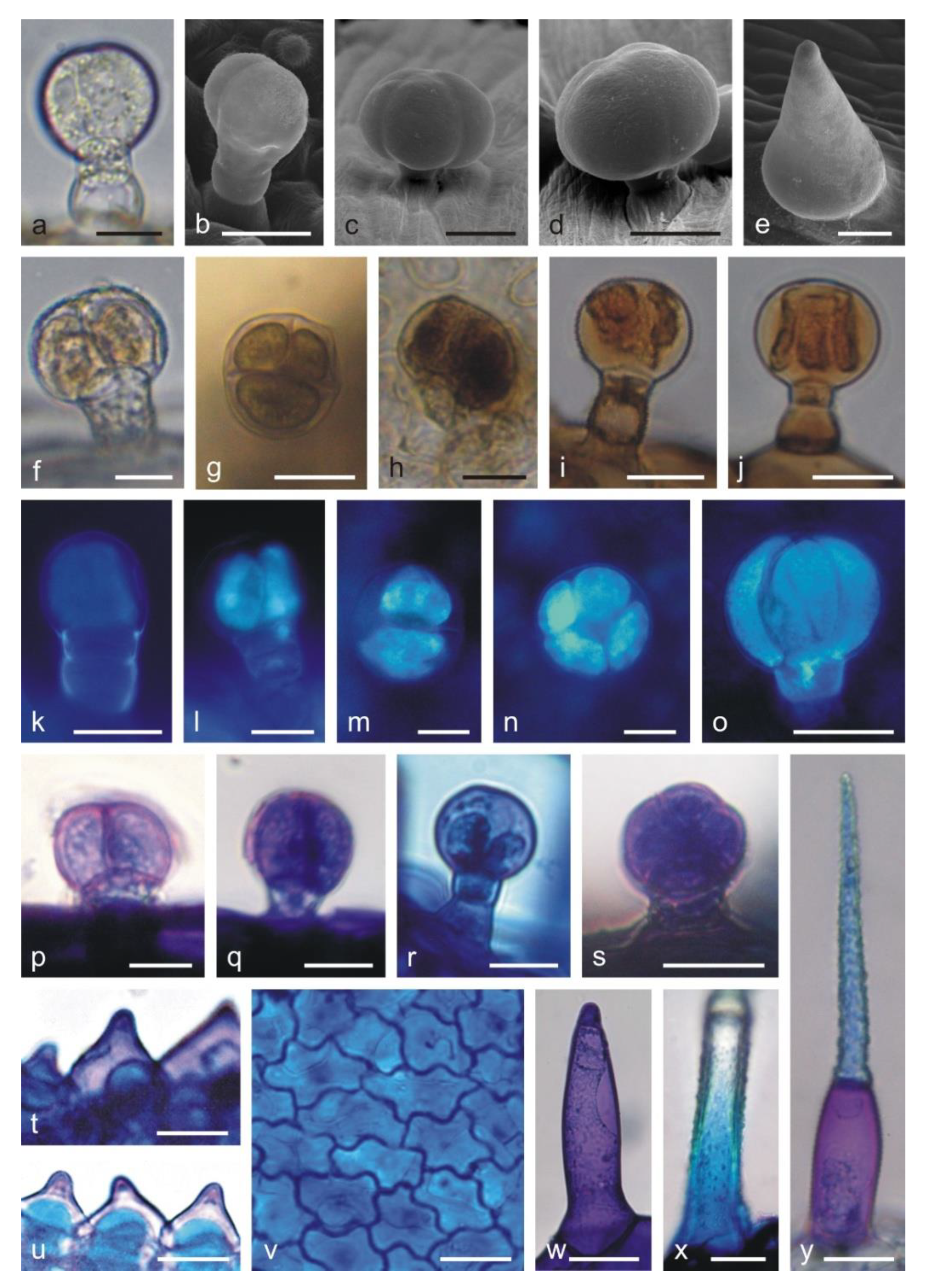
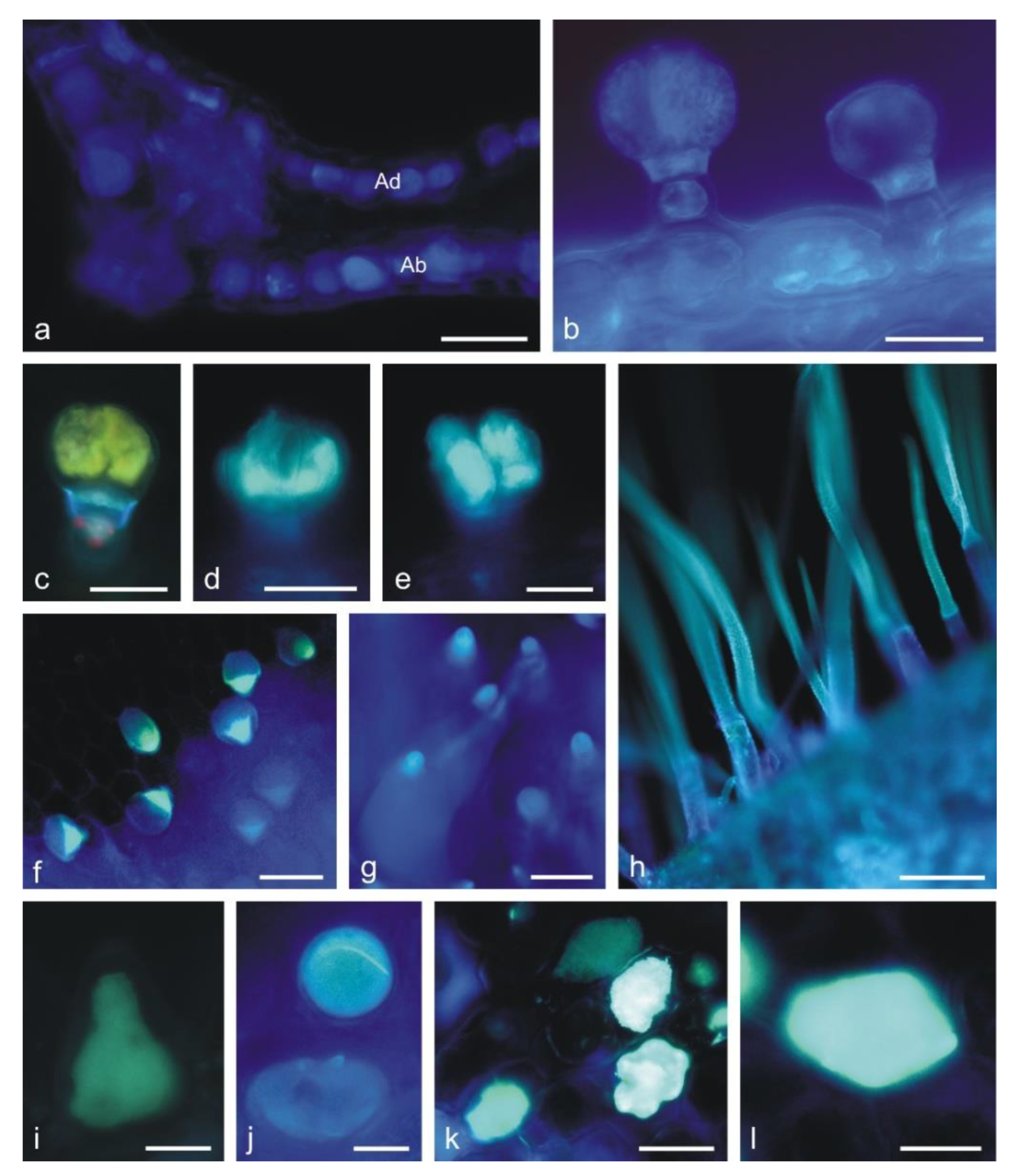
2.2. Histochemical Analyses
2.3. Phytochemical Analyses
3. Discussion
3.1. Anatomy and Histochemistry
3.2. Phytochemistry
4. Materials and Methods
4.1. Histochemical Tests and FM
4.2. SEM
4.3. Phytochemical Analyses
4.3.1. Extract Preparation and Purification
4.3.2. Planar Chromatography Experiments
4.3.3. Analysis of Flavonoids
4.3.4. Visualization of Flavonoids
4.3.5. Analysis of Phenolic Acids
4.3.6. Chromatographic HPLC Experiments
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Mabberley, D.J. The Plant Book, 2nd ed.; Cambridge University Press: Cambridge, UK, 1997. [Google Scholar]
- Yordanova, Z.P.; Zhiponova, M.K.; Iakimova, E.T.; Dimitrova, M.A.; Kapchina-Toteva, V.M. Revealing the reviving secret of the white dead nettle (Lamium album L.). Phytochem. Rev. 2014, 13, 375–389. [Google Scholar] [CrossRef]
- Dengler, J.; Berg, C.; Eisenberg, M.; Isermann, M.; Jansen, F.; Koska, I.; Löbel, S.; Manthey, M.; Pazolt, J.; Spangenberg, A.; et al. New descriptions and typifications of syntaxa within the project ‘Plant communities of Mecklenburg-Vorpommern and their vulnerability’—Part I. Feddes Repertorium Zeitschrift für Botanische Taxonomie und Geobotanik 2003, 114, 587–631. [Google Scholar] [CrossRef]
- Metcalfe, C.R.; Chalk, L. Anatomy of the Dicotyledons; Oxford Press: London, UK, 1972; Volume 2. [Google Scholar]
- Sulborska, A.; Dmitruk, M.; Konarska, A.; Weryszko-Chmielewska, E. Adaptations of Lamium album L. flowers to pollination by Apoidea. Acta Sci. Pol. Hortorum Cultus 2014, 13, 31–43. [Google Scholar]
- Celep, F.; Kahraman, A.; Atalay, Z.; Doğan, M. Morphology, anatomy and trichome properties of Lamium truncatum Boiss. (Lamiaceae) and their systematic implications. Aust. J. Crop Sci. 2011, 5, 147–153. [Google Scholar]
- Özdemir, C.; Baran, P. Morphological, anatomical and cytological investigation on alpine Lamium cymbalariifolium endemic to Turkey. Aust. J. Crop Sci. 2012, 6, 532–540. [Google Scholar]
- Atalay, Z.; Celep, F.; Bara, F.; Doğan, M. Systematic significance of anatomy and trichome morphology in Lamium (Lamioideae; Lamiaceae). Flora 2016, 225, 60–75. [Google Scholar] [CrossRef]
- Baran, P.; Özdemir, C. The morphological and anatomical properties of Lamium lyceum (Lamiaceae), endemic to Turkey. Nord. J. Bot. 2009, 27, 388–396. [Google Scholar] [CrossRef]
- Baran, P.; Özdemir, C. Morphological, anatomical and cytological studies on endemic Lamium pisidicum. Pak. J. Bot. 2013, 45, 73–85. [Google Scholar]
- Turner, N.J.; Łukasz, Ł.J.; Migliorini, P.; Pieroni, A.; Dreon, A.L.; Sacchetti, L.E.; Paoletti, M.G. Edible and Tended Wild Plants, Traditional Ecological Knowledge and Agroecology. Crit. Rev. Plant Sci. 2011, 30, 198–225. [Google Scholar] [CrossRef]
- Pereira, O.R.; Domingues, M.D.R.; Silva, A.M.; Cardoso, S.M. Phenolic constituents of Lamium album: Focus on isoscutellarein derivatives. Food Res. Int. 2012, 48, 330–335. [Google Scholar] [CrossRef]
- Xu, F. Chinese Medicine e.g. for Treating Arthropathy, Comprises Broad Cocklebur, Vervain, Condyle Grass, Motherwort, Saxifrage, Cactus, Mullberry Branch, White Dead Nettle, Boston Ivy, Folium Photiniae, Water Pepper and Chinese Fever Vine; XUFF-Individual; 2008; p. 10. [Google Scholar]
- Paduch, R.; Wójciak-Kosior, M.; Matysik, G. Investigation of biological activity of Lamii albi flos extracts. J. Ethnopharmacol. 2007, 110, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Kelayeh, T.P.S.; Abedinzade, M.; Ghorbani, A. A review on biological effects of Lamium album (white dead nettle) and its components. J. Herbmed Pharm. 2019, 8, 185–193. [Google Scholar] [CrossRef]
- Zhang, H.-J.; Rothwangl, K.; Mesecar, A.D.; Sabahi, A.; Rong, L.; Fong, H.H.S. Lamiridosins, Hepatitis C Virus Entry Inhibitors from Lamium album. J. Nat. Prod. 2009, 72, 2158–2162. [Google Scholar] [CrossRef] [PubMed]
- Valyova, M.S.; Dimitrova, M.A.; Ganeva, Y.A.; Kapchina-Toteva, V.M.; Yordanova, Z.P. Evaluation of antioxidant and free radical scavenging potential of Lamium album L. growing in Bulgaria. J. Pharm. Res. 2011, 4, 945–947. [Google Scholar]
- Pereira, O.R.; Macias, R.I.R.; Perez, M.J.; Marin, J.J.G.; Cardoso, S.M. Protective effects of phenolic constituents from Cytisus mulitflorus, Lamium album L. and Thymus citriodorus on liver cells. J. Funct. Foods 2013, 5, 1170–1179. [Google Scholar] [CrossRef]
- Kokoska, L.; Polesny, Z.; Rada, V.; Nepovim, A.; Vanek, T. Screening of some Siberian medicinal plants for antimicrobial activity. J. Ethnopharmacol. 2002, 82, 51–53. [Google Scholar] [CrossRef]
- Topouzova-Hristova, T.; Moskova-Doumanova, V.; Keremidarska, M.; Doumanov, J.; Miteva, G.; Petkova, B.; Kapchina-Toteva, V. Anticancer effect of plant extracts from Lamium album L. by induction od cell death in vitro. Medicine 2012, 2, 55–59. [Google Scholar]
- Gao, J.; Morgan, G.; Tieu, D.; A Schwalb, T.; Luo, J.Y.; A Wheeler, L.; E Stern, M. ICAM-1 expression predisposes ocular tissues to immune-based inflammation in dry eye patients and Sjögrens syndrome-like MRL/lpr mice. Exp. Eye Res. 2004, 78, 823–835. [Google Scholar] [CrossRef]
- Czerwińska, M.E.; Świerczewska, A.; Woźniak, M.; Kiss, A.K. Bioassay-Guided Isolation of Iridoids and Phenylpropanoids from Aerial Parts of Lamium album and Their Anti-inflammatory Activity in Human Neutrophils. Planta Med. 2017, 83, 1011–1019. [Google Scholar] [CrossRef] [Green Version]
- Croteau, R.; Kutchan, T.M.; Lewis, N.G. Natural products (secondary metabolites). In Biochemistry & Molecular Biology of Plants; Buchanan, B., Gruissem, W., Jones, R., Dekker, M., Eds.; American Society of Plant Physiologists: New York, NY, USA, 2000; pp. 1250–1318. [Google Scholar]
- Lotina-Hennsen, B.; King-Díaz, B.; Aguilar, M.; Terrones, M.G.H. Plant secondary metabolites. Targets and mechanisms of allelopathy. In Allelopathy: A Physiological Proccess with Ecological Implications; Reigosa, M.J., Pedrol, N., Gonzáles, L., Eds.; Springer: Dordrecht, The Netherlands, 2006; pp. 229–265. [Google Scholar]
- Tošić, S.; Stojičić, D.; Slavkovska, V.; Mihailov-Krstev, T.; Zlatković, B.; Budimir, S.; Uzelac, B. Phytochemical composition and biological activities of native and in vitro-propagated Micromeria croatica (Pers.) Schott (Lamiaceae). Planta 2019, 249, 1365–1377. [Google Scholar] [CrossRef]
- Frezza, C.; Venditti, A.; Serafini, M.; Bianco, A. Phytochemistry, chemotaxonomy, ethnopharmacology, and nutraceutics of Lamiaceae. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2019; Volume 62, pp. 125–178. [Google Scholar]
- Salehi, B.; Armstrong, L.; Rescigno, A.; Yeskaliyeva, B.; Seitimova, G.; Beyatli, A.; Jugreet, S.; Mahomoodally, M.F.; Sharopov, F.; Durazzo, A.; et al. Lamium Plants—A Comprehensive Review on Health Benefits and Biological Activities. Molecules 2019, 24, 1913. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caissard, J.-C.; Joly, C.; Bergougnox, V.; Hugueney, P.; Mauriat, M.; Baudino, S. Secretion mechanisms of volatile organic compounds in specialized cells of aromatic plants. Rec. Res. Develop. Cell Biol. 2004, 2, 1–15. [Google Scholar]
- Dmitruk, M.; Sulborska, A.; Żuraw, B.; Stawiarz, E.; Weryszko-Chmielewska, E. Sites of secretion of bioactive compounds in leaves of Dracocephalum moldavica L.: Anatomical, histochemical, and essential oil study. Braz. J. Bot. 2019, 42, 701–715. [Google Scholar] [CrossRef] [Green Version]
- Evert, R.F. Esau’s Plant Anatomy, 3rd ed.; Wiley-Interscience: Hoboken, NJ, USA, 2006. [Google Scholar]
- Bosabalidis, A.M.; Sawidis, T. Glandular and non-glandular hairs in the seasonally dimorphic Origanum dictamnus L. (Lamiaceae) as a means of adaptation to cold stress. Acta Agrobot. 2014, 67, 15–20. [Google Scholar] [CrossRef]
- Werker, E.; Putievsky, E.; Ravid, U.; Dudai, N.; Katzir, I. Glandular Hairs and Essential Oil in Developing Leaves of Ocimum basilicum L. (Lamiaceae). Ann. Bot. 1993, 71, 43–50. [Google Scholar] [CrossRef]
- Naidoo, Y.; Kasim, N.; Heneidak, S.; Nicholas, A.; Naidoo, G. Foliar secretory trichomes of Ocimum obovatum (Lamiaceae): Micromorphological structure and histochemistry. Plant Syst. Evol. 2013, 299, 873–885. [Google Scholar] [CrossRef]
- Liu, M.-Q. Structure and histochemistry of the glandular trichomes on the leaves of Isodon rubescens (Lamiaceae). Afr. J. Biotechnol. 2012, 11, 4069–4078. [Google Scholar] [CrossRef]
- Giuliani, C.; Bini, L.M. Insight into the structure and chemistry of glandular trichomes of Labiatae, with emphasis on subfamily Lamioideae. Plant Syst. Evol. 2008, 276, 199–208. [Google Scholar] [CrossRef]
- Venditti, A.; Bianco, A.; Quassinti, L.; Bramucci, M.; Lupidi, G.; Damiano, S.; Papa, F.; Vittori, S.; Bini, L.M.; Giuliani, C.; et al. Phytochemical Analysis, Biological Activity, and Secretory Structures of Stachys annua (L.) L. subsp. annua (Lamiaceae) from Central Italy. Chem. Biodivers. 2015, 12, 1172–1183. [Google Scholar] [CrossRef]
- Marin, M.; Ascensão, L.; Lakušić, B. Trichomes of Satureja horvatii Šilić (Lamiaceae): Micromorphology and histochemistry. Arch. Biol. Sci. 2012, 64, 995–1000. [Google Scholar] [CrossRef]
- Chwil, M.; Nurzyńska-Wierdak, R.; Chwil, S.; Matraszek, R.; Neugebauerová, J. Histochemistry and micromorphological diversity of glandular trichomes in Melissa officinalis L. leaf epidermis. Acta Sci. Pol. Hortorum Cultus 2016, 15, 153–172. [Google Scholar]
- Haratym, W.; Weryszko-Chmielewska, E. Ultrastructural and histochemical analysis of glandular trichomes of Marrubium vulgare L. (Lamiaceae). Flora Morphol. Distrib. Funct. Ecol. Plants 2017, 231, 11–20. [Google Scholar] [CrossRef]
- Venditti, A.; Bianco, A.; Frezza, C.; Conti, F.; Bini, L.M.; Giuliani, C.; Bramucci, M.; Quassinti, L.; Damiano, S.; Lupidi, G.; et al. Essential oil composition, polar compounds, glandular trichomes and biological activity of Hyssopus officinalis subsp. aristatus (Godr.) Nyman from central Italy. Ind. Crop. Prod. 2015, 77, 253–363. [Google Scholar] [CrossRef]
- Ascensão, L.; Mota, L.; Castro, M.D.M. Glandular Trichomes on the Leaves and Flowers of Plectranthus ornatus: Morphology, Distribution and Histochemistry. Ann. Bot. 1999, 84, 437–447. [Google Scholar] [CrossRef]
- Janošević, D.; Budimar, S.; Alimpić, A.; Marin, P.; Al Sheef, N.; Giweli, A.; Dulitić-Laušević, S. Micromorphology and histochemistry of leaf trichomes of Salvia aegyptica (Lamiaceae). Arch. Biol. Sci. 2016, 68, 291–301. [Google Scholar] [CrossRef]
- Corsi, G. Glandular Hairs of Salvia officinalis: New Data on Morphology, Localization and Histochemistry in Relation to Function. Ann. Bot. 1999, 84, 657–664. [Google Scholar] [CrossRef] [Green Version]
- Jia, P.; Gao, T.; Xin, H. Changes in Structure and Histochemistry of Glandular Trichomes of Thymus quinquecostatus Celak. Sci. World J. 2012, 2012, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Richardson, P.M. The chemistry of the Labiatea: An introduction and overview. In Advances in Labiate Science; Harley, R.M., Reynolds, T., Eds.; Kew, Royal Botanical Gardens: Richmond, UK, 1992. [Google Scholar]
- Usenik, V.; Stampar, F.; Kastelec, D. Phytochemicals in fruits of two Prunus domestica L. plum cultivars during ripening. J. Sci. Food Agric. 2013, 93, 681–692. [Google Scholar] [CrossRef]
- Konarska, A. Morphological, anatomical, and ultrastructural changes in Vaccinium corymbosum fruits during ontogeny. Botany 2015, 93, 589–602. [Google Scholar] [CrossRef] [Green Version]
- Jakobek, L. Interactions of polyphenols with carbohydrates, lipids and proteins. Food Chem. 2015, 175, 556–567. [Google Scholar] [CrossRef]
- Karabourniotis, G.; Liakopoulos, G.; Nikolopoulos, D.; Bresta, P.; Stavroulaki, V.; Sumbele, S. “Carbon gain vs. water saving, growth vs. defence”: Two dilemmas with soluble phenolics as a joker. Plant Sci. 2014, 227, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Padayachee, A.; Netzel, G.; Netzel, M.; Day, L.; Zabaras, D.; Mikkelsen, D.; Gidley, M.J. Binding of polyphenols to plant cell wall analogues—Part 2: Phenolic acids. Food Chem. 2012, 135, 2287–2292. [Google Scholar] [CrossRef] [PubMed]
- Bazzo, Y.M.; Catini, C.D.; Vicentini, F.T.; Cardoso, J.C.; Junior, R.L.C.D.A.; Fonseca, M.J.V. Efficacy of Marigold Extract-Loaded Formulations Against UV-induced Oxidative Stress. J. Pharm. Sci. 2011, 100, 2182–2193. [Google Scholar] [CrossRef]
- Alipieva, K.; Kokubun, T.; Taskova, R.; Evstatieva, L.; Handjieva, N. LC–ESI-MS analysis of iridoid glucosides in Lamium species. Biochem. Syst. Ecol. 2007, 35, 17–22. [Google Scholar] [CrossRef]
- Karabourniotis, G.; Liakopoulos, G. Phenolic compounds in plant cuticles: Physiological and ecophysiological aspects. In Advances in Plant Physiology; Hemantaranjan, A., Ed.; Scientific Publishers: Jodhpur, India, 2005; Volume 8, pp. 33–47. [Google Scholar]
- O’Prey, J.; Brown, J.; Fleming, J.; Harrison, P.R. Effects of dietary flavonoids on major signal transduction pathways in human epithelial cells. Biochem. Pharm. 2003, 66, 2075–2088. [Google Scholar] [CrossRef]
- Spencer, J.P.E.; El Mohsen, M.M.A.; Rice-Evans, C. Cellular uptake and metabolism of flavonoids and their metabolites: Implications for their bioactivity. Arch. Biochem. Biophys. 2004, 423, 148–161. [Google Scholar] [CrossRef]
- Botirov, E.K. Flavonoids and Phenolcarboxylic Acids from Lamium album. Chem. Nat. Compd. 2019, 55, 1159–1160. [Google Scholar] [CrossRef]
- Monteiro, Á.B.; Rodrigues, C.K.D.S.; Nascimento, E.P.D.; Sales, V.D.S.; Delmondes, G.D.A.; Da Costa, M.H.N.; De Oliveira, V.A.P.; Pereira-De-Morais, L.; Boligon, A.A.; Barbosa, R.; et al. Anxiolytic and antidepressant-like effects of Annona coriacea (Mart.) and caffeic acid in mice. Food Chem. Toxicol. 2020, 136, 111049. [Google Scholar] [CrossRef]
- Karatas, O.; Yuce, H.B.; Taskan, M.M.; Gevrek, F.; Yarkac, F.U.; Keskin, A.; Karatas, S.F.O.; Toker, H. The effect of vanillic acid on ligature-induced periodontal disease in Wistar rats. Arch. Oral Biol. 2019, 103, 1–7. [Google Scholar] [CrossRef]
- Tupec, M.; Hýsková, V.; Bělonožníková, K.; Hraníček, J.; Červený, V.; Ryšlavá, H. Characterization of some potential medicinal plants from Central Europe by their antioxidant capacity and the presence of metal elements. Food Biosci. 2017, 20, 43–50. [Google Scholar] [CrossRef]
- Danila, D.; Adriana, T.; Camelia, S.; Valentin, G.; Anca, M. Antioxidant activity of methanolic extracts of Lamium album and Lamium maculatum species from wild populations in the Romanian Eastern Carpathians. Planta Med. 2015, 81. [Google Scholar] [CrossRef]
- Hegi, G. Illustrierte Flora von Mittel-Europa; Carl Hanser Verlag: München, Germany, 1965; Volume 4. [Google Scholar]
- Mennema, J. A Taxonomic Revision of Lamium (Lamiaceae); Leiden Botanical Series; E.J. Brill: Leiden, The Netherlands, 1989; Volume 11. [Google Scholar]
- Kohlmünzer, S. Farmakognozja: Podręcznik dla Studentów Farmacji; PZWL Wydawnictwo Lekarskie: Warszawa, Poland, 2017. [Google Scholar]
- Gahan, P.B. Plant Histochemistry and Cytochemistry; Academic Press: London, UK, 1984. [Google Scholar]
- Gabe, M. Techniques Histologiques; Masson: Paris, France, 1968. [Google Scholar]
- Baker, J.R. Cytological Technique, 5th ed.; Methven: London, UK, 1966. [Google Scholar]
- Gutmann, M. Improved staining procedures for photographic documentation of phenolic deposits in semithin sections of plant tissue. J. Microsc. 1995, 179, 277–281. [Google Scholar] [CrossRef]
- O’Brien, T.P.; McCully, M.E. The Study of Plant Structure: Principles and Selected Methods; Termarcarphi Pty Ltd.: Melbourne, Australia, 1981. [Google Scholar]
- Ribeiro, V.C.; Leitão, C.A.E. Utilisation of Toluidine blue O pH 4.0 and histochemical inferences in plant sections obtained by free-hand. Protoplasma 2019, 257, 993–1008. [Google Scholar] [CrossRef] [PubMed]
- Soukup, A. Selected Simple Methods of Plant Cell Wall Histochemistry and Staining for Light Microscopy. In Plant Cell Morphogenesis; Humana Press: Totowa, NJ, USA, 2014; pp. 25–40. [Google Scholar]
- Buschmann, C.; Langsdorf, G.; Lichtenthaler, H. Imaging of the Blue, Green, and Red Fluorescence Emission of Plants: An Overview. Photosynthetica 2000, 38, 483–491. [Google Scholar] [CrossRef]
- Chaerle, L.; Lenk, S.; Hagenbeek, D.; Buschmann, C.; Van Der Straeten, D. Multicolor fluorescence imaging for early detection of the hypersensitive reaction to tobacco mosaic virus. J. Plant Physiol. 2007, 164, 253–262. [Google Scholar] [CrossRef]
- Huang, S.S.; Kirchoff, B.K.; Liao, J.P. The capitate and peltate glandular trichomes in Lavandula pinnata L. (Lamiaceae): Histochemistry, ultrastructure and secretion. J. Torrey Bot. Soc. 2008, 135, 155–167. [Google Scholar] [CrossRef]
- Charrière-Ladreix, Y. Répartition intracellulaire du secrétat flavonique de Populus nigra L. Planta 1976, 129, 167–174. [Google Scholar] [CrossRef]
- Badria, F.A.; Aboelmaaty, W.S. Plant Histochemistry: A Versatile and Indispensible Tool in Localization of Gene Expression, Enzymes, Cytokines, Secondary Metabolites and Detection of Plants Infection and Pollution. Acta Sci. Pharm. Sci. 2019, 3, 88–100. [Google Scholar] [CrossRef]
- Wójciak-Kosior, M.; Matysik, G.; Skalska, A. Densitometric determination of kinetics of hydrolysis of flavonoid glycosides. J. Planar Chromatogr. Mod. TLC 2004, 17, 286–289. [Google Scholar] [CrossRef]
- Chernetskyy, M.; Woźniak, A.; Skalska-Kamińska, A.; Żuraw, B.; Blicharska, E.; Rejdak, R.; Donica, H.; Weryszko-Chmielewska, E. Structure of Leaves and Phenolic Acids in Kalanchoë daigremontiana Raym.-Hamet & H. Perrier. Acta Sci. Pol. Hortorum Cultus 2018, 17, 137–155. [Google Scholar] [CrossRef]
- Tyszczuk-Rotko, K.; Skalska-Kamińska, A.; Woźniak, A. Voltammetric method using a lead film electrode for the determination of caffeic acid in a plant material. Food Chem. 2011, 125, 1498–1503. [Google Scholar] [CrossRef]
- Matysik, G. Modified programmed multiple gradient development (MGD) in the analysis of complex plant extracts. Chromatographia 1996, 43, 39–43. [Google Scholar] [CrossRef]







| Target Compounds | Stain Reagent | Reaction Color | Trichomes | Papillae | Epidermis Cells | ||
|---|---|---|---|---|---|---|---|
| Peltate | Capitate | Non-Glandular | |||||
| General phenolic compounds | FeCl3 | black, brown | + | + | nd | + | + |
| Toluidine blue O | green or blue | + | + | + | + | + | |
| Tannins | Potassium dichromate | brown | + | + | nd | + | - |
| Toluidine blue O | deep blue | + | + | + | + | + | |
| Phenolic acids | Autofluorescence UV | light blue | + | + | - | + | + |
| Flavonoids | AlCl3 under UV | yellow | + | + | + | + | + |
| Pectins | Toluidine blue O | purple or pink | + | + | + | + | - |
| Elution Step | Development Distance [cm] | Mobile Phase Composition |
|---|---|---|
| 1 | 3 | 6 mL solution A (toluene hexane: formic acid, 7:3: 0.1), 3 mL ethyl acetate, 1 mL methanol |
| 2 | 9 | 5 mL solution A (toluene hexane: formic acid, 7:3: 0.1), 2.5 mL ethyl acetate, 2.5 mL methanol |
| Development Step | Mobile Phase Composition |
|---|---|
| 1, 2 | 3 mL solution A (heptane: dichlorometane, 7:3), 2 mL diisopropyl ether, 0.1 mL 85% formic acid, 1 mL distilled water |
| 3–7 | 4 mL solution A (heptane: dichlorometane, 7:3), 1 mL diisopropyl ether, 0.1 mL 85% formic acid |
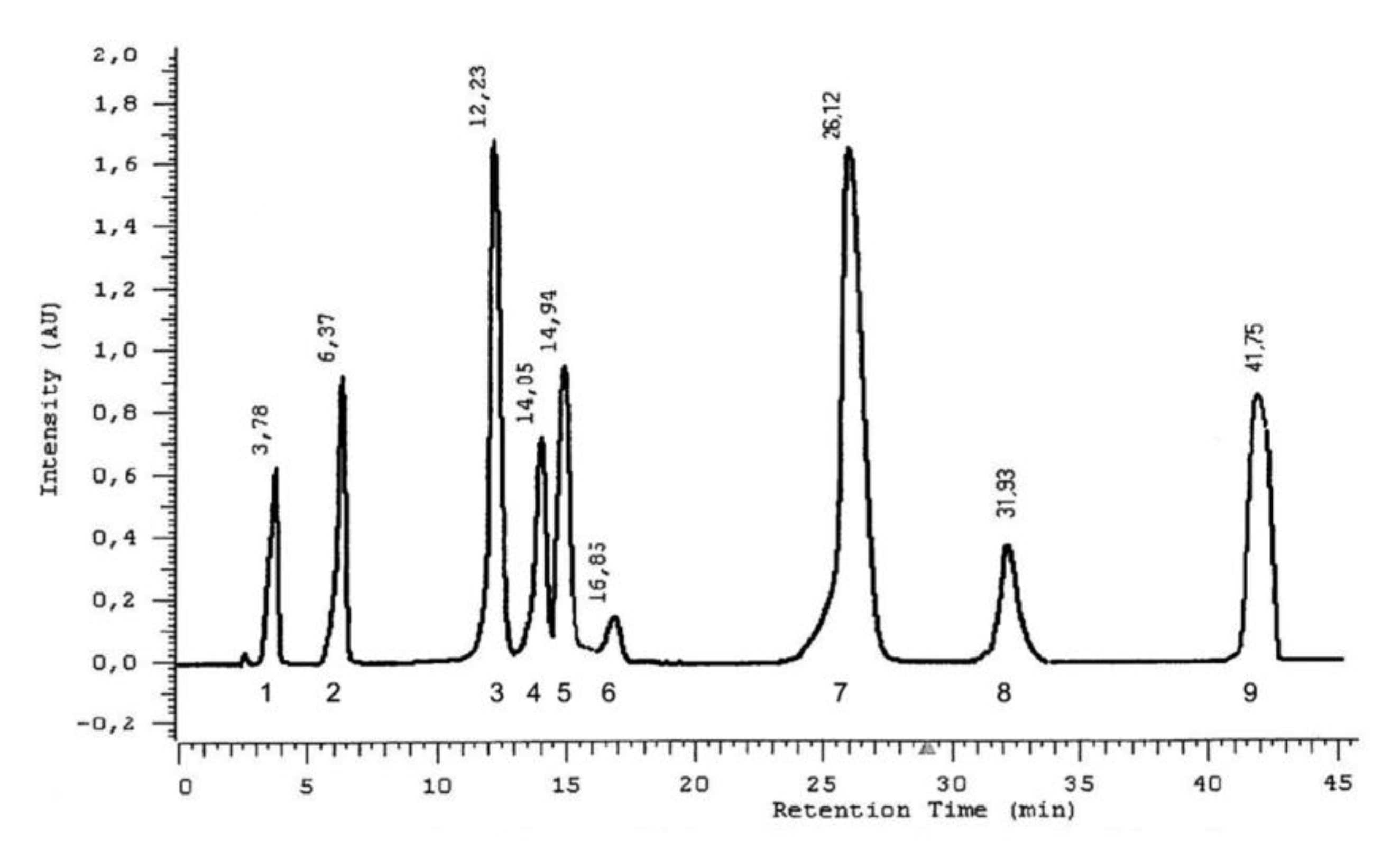
| Phenolic Acid Standard | Retention Time [min] | λmax [nm] |
|---|---|---|
| gallic acid | 3.78 | 227.5; 271.0 |
| protocatechuic acid | 6.37 | 227.5; 258.6; 294.0 |
| gentisic acid | 12.23 | 234.9; 326.9 |
| vanillic acid | 14.05 | 226.4; 260.5; 291.0 |
| caffeic acid | 14.95 | 239.0; 322.6 |
| syringic acid | 16.85 | 224.7; 273.3 |
| p-coumaric acid | 26.12 | 228.7; 308.9 |
| ferulic acid | 31.93 | 234.9; 322.6 |
| chlorogenic acid | 41.75 | 234.9; 300.3 |
Sample Availability: Samples of the compounds are not available from the authors. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sulborska, A.; Konarska, A.; Matysik-Woźniak, A.; Dmitruk, M.; Weryszko-Chmielewska, E.; Skalska-Kamińska, A.; Rejdak, R. Phenolic Constituents of Lamium album L. subsp. album Flowers: Anatomical, Histochemical, and Phytochemical Study. Molecules 2020, 25, 6025. https://doi.org/10.3390/molecules25246025
Sulborska A, Konarska A, Matysik-Woźniak A, Dmitruk M, Weryszko-Chmielewska E, Skalska-Kamińska A, Rejdak R. Phenolic Constituents of Lamium album L. subsp. album Flowers: Anatomical, Histochemical, and Phytochemical Study. Molecules. 2020; 25(24):6025. https://doi.org/10.3390/molecules25246025
Chicago/Turabian StyleSulborska, Aneta, Agata Konarska, Anna Matysik-Woźniak, Marta Dmitruk, Elżbieta Weryszko-Chmielewska, Agnieszka Skalska-Kamińska, and Robert Rejdak. 2020. "Phenolic Constituents of Lamium album L. subsp. album Flowers: Anatomical, Histochemical, and Phytochemical Study" Molecules 25, no. 24: 6025. https://doi.org/10.3390/molecules25246025