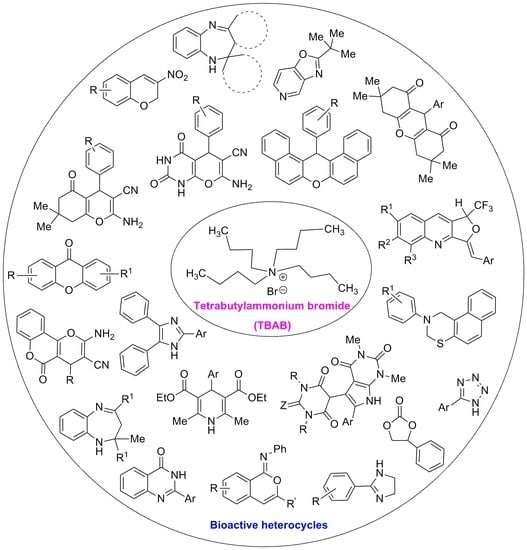
Tetrabutylammonium Bromide (TBAB) Catalyzed Synthesis of Bioactive Heterocycles
Abstract
1. Introduction
2. Applications of TBAB for the Synthesis of Bioactive N-Heterocycles
2.1. Synthesis of 1,4-Dihydropyridines
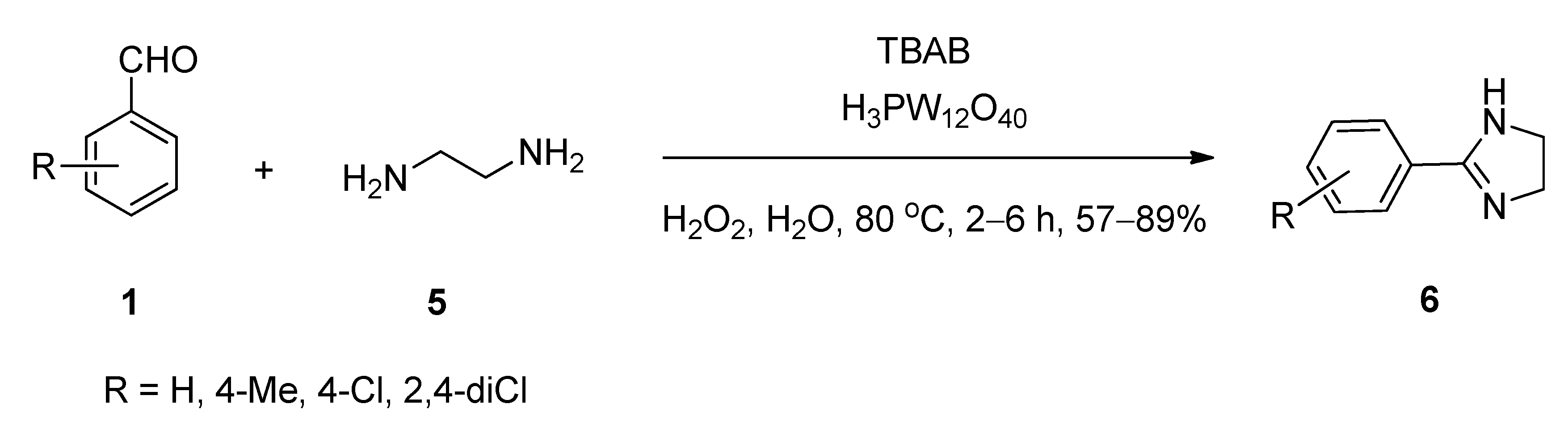
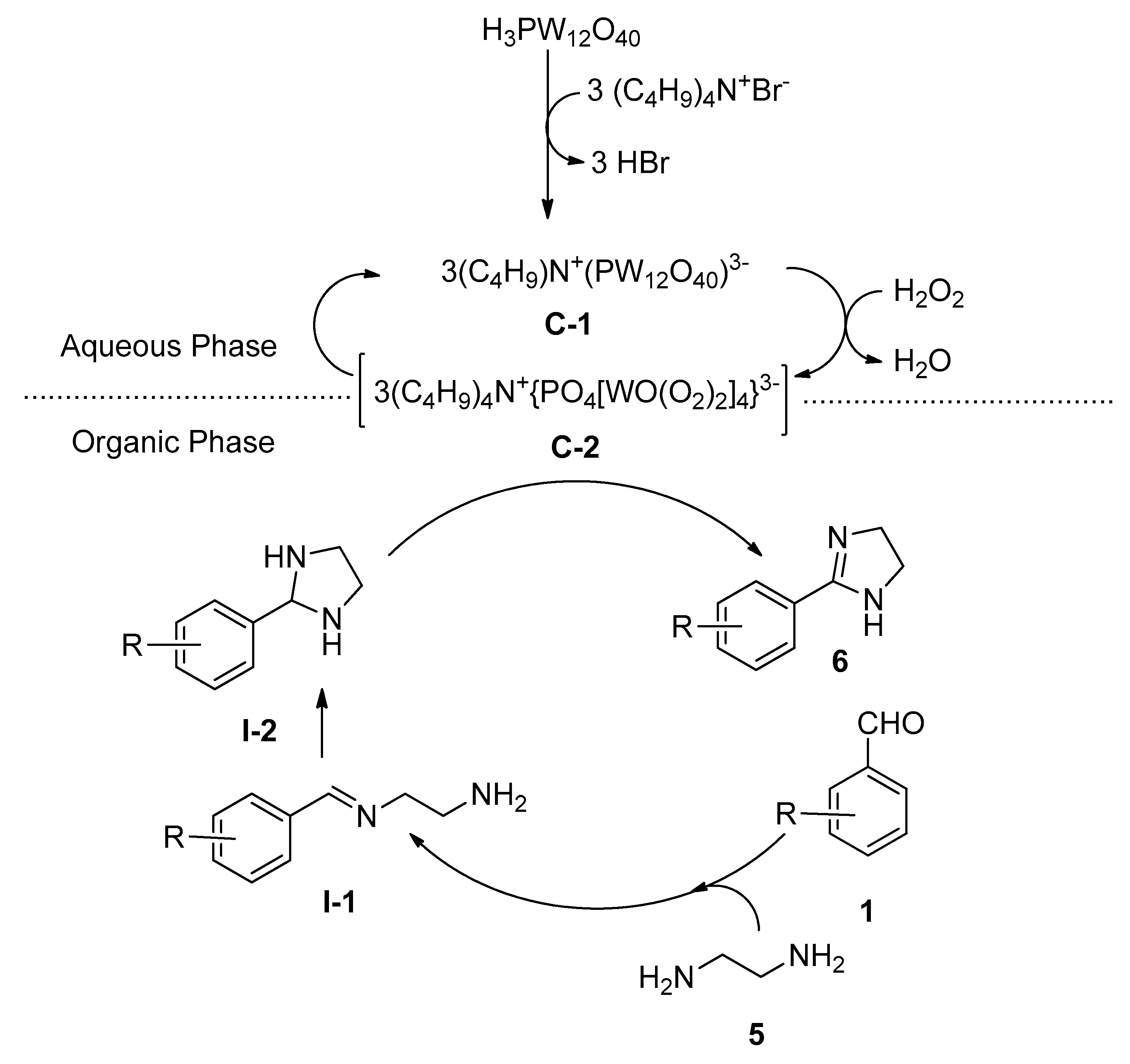
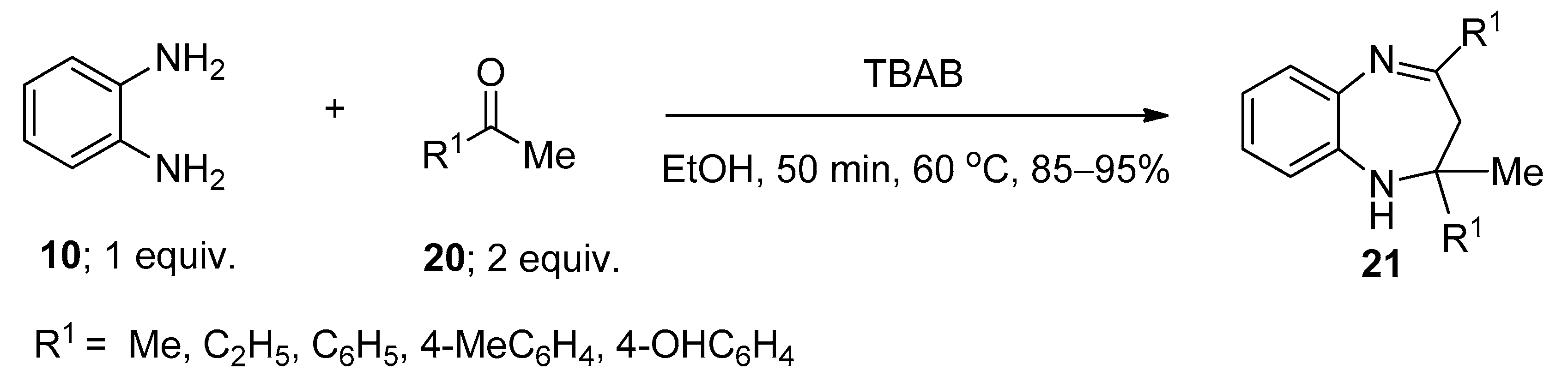
2.2. Synthesis of 2-Substituted Imidazolines
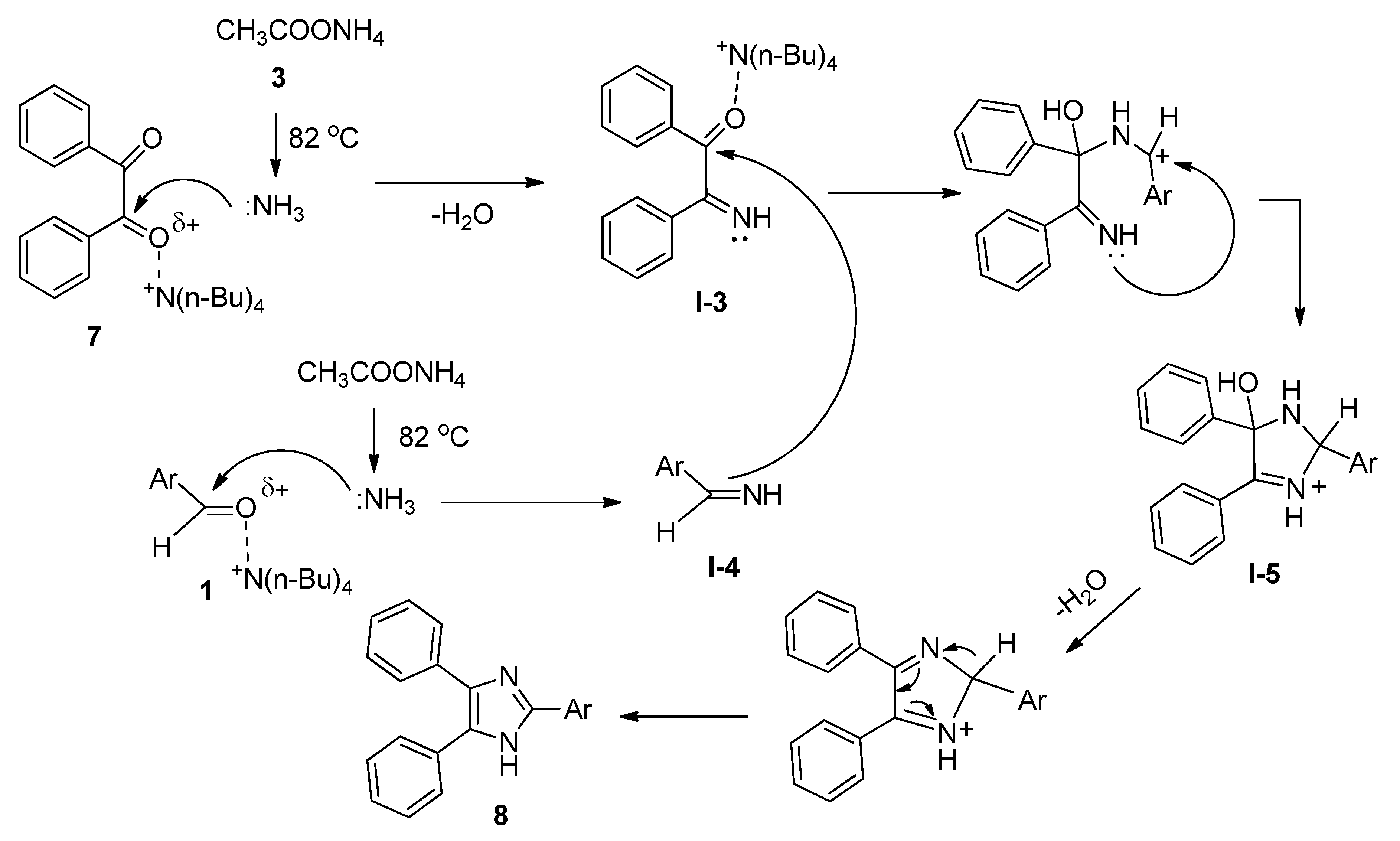
2.3. Synthesis of 2,4,5-Triaryl Imidazoles
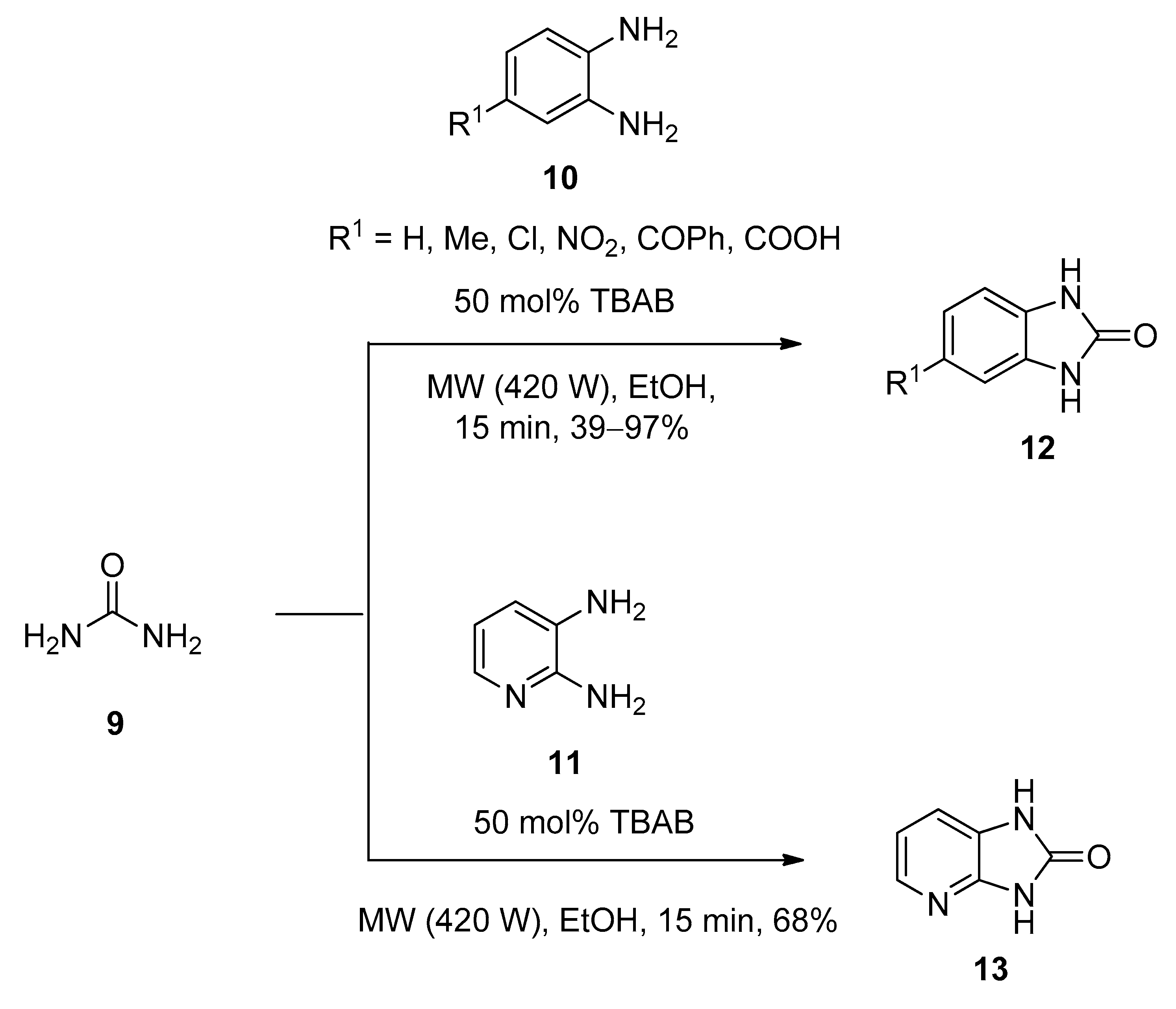
2.4. Synthesis of 1,3-Dihydrobenzimidazol-2-Ones
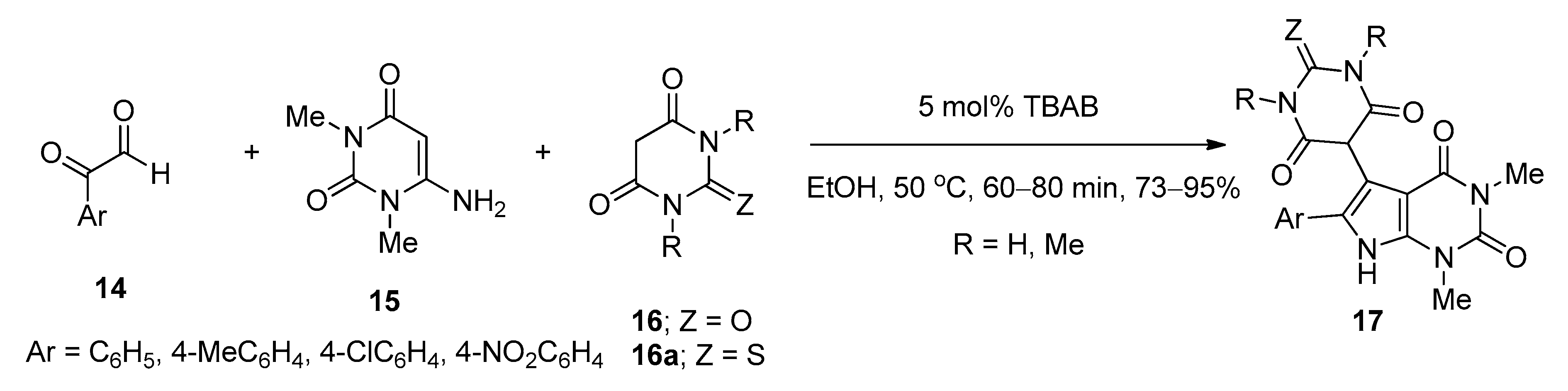
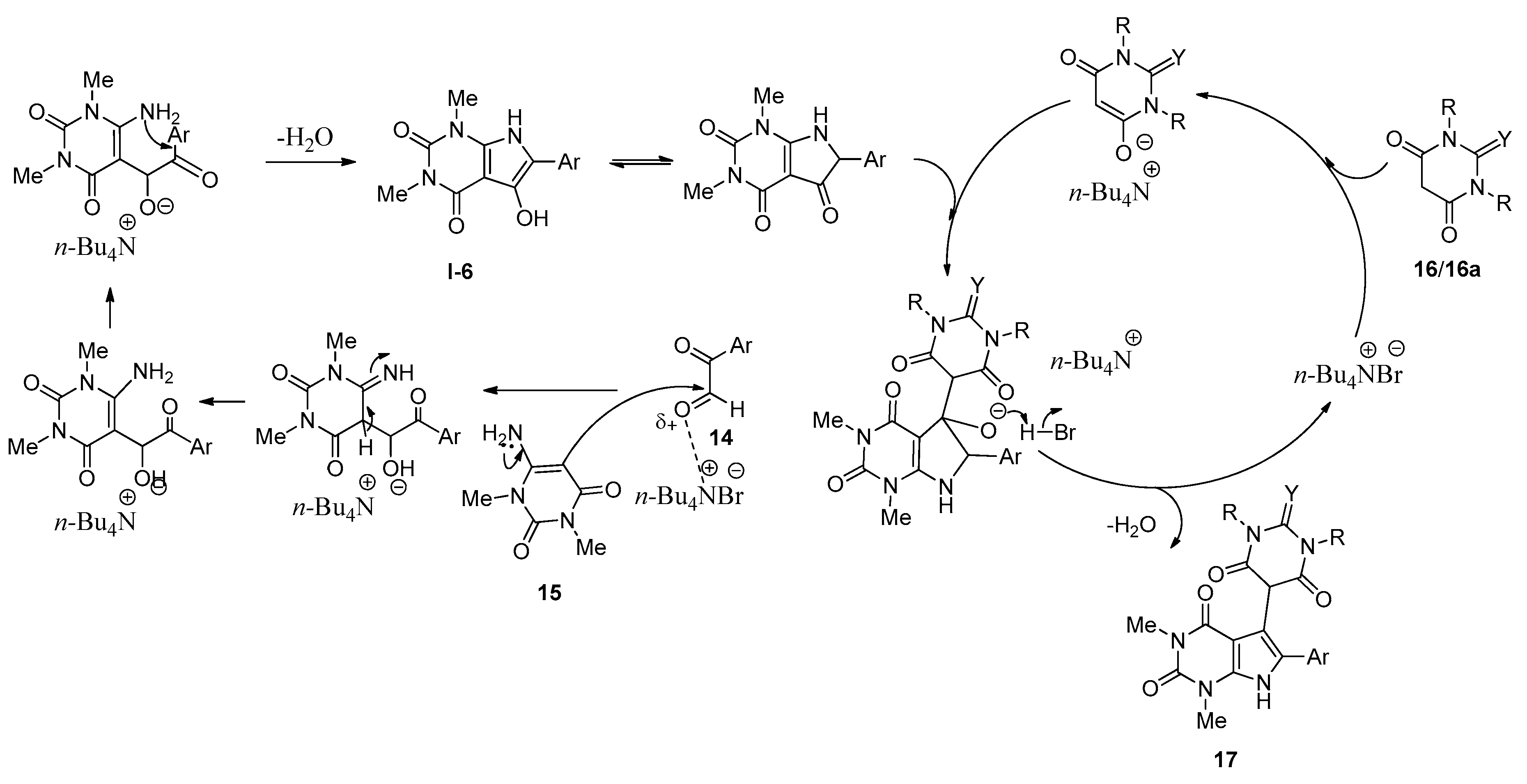
2.5. Synthesis of Pyrrolo[2,3-d]pyrimidine Derivatives
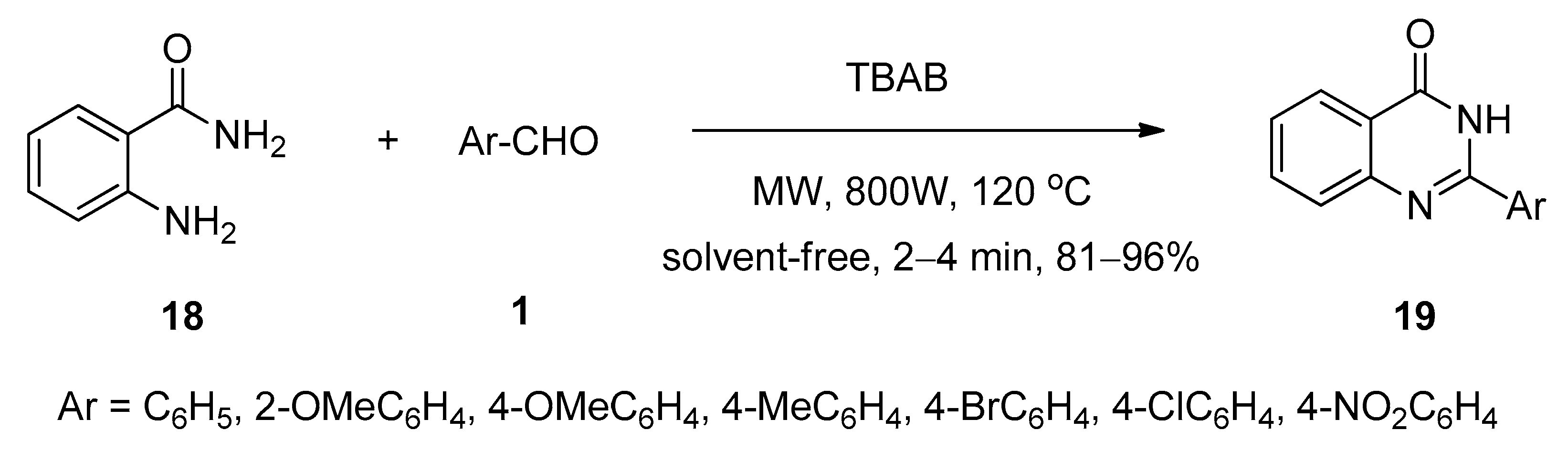
2.6. Synthesis of 2,3-Dihydroquinazolin-4(1H)-Ones
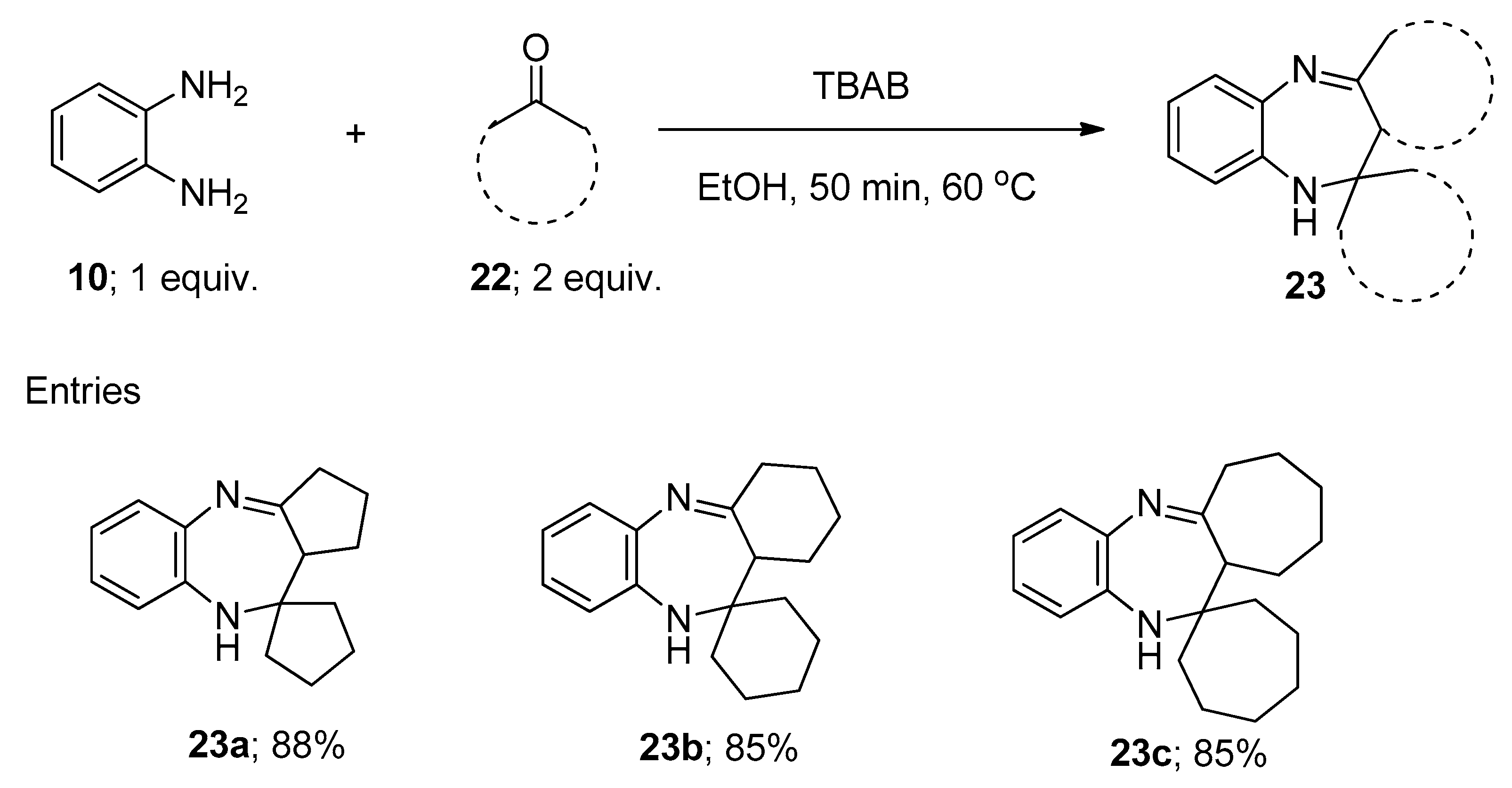
2.7. Synthesis of 1,5-Benzodiazepine Derivatives
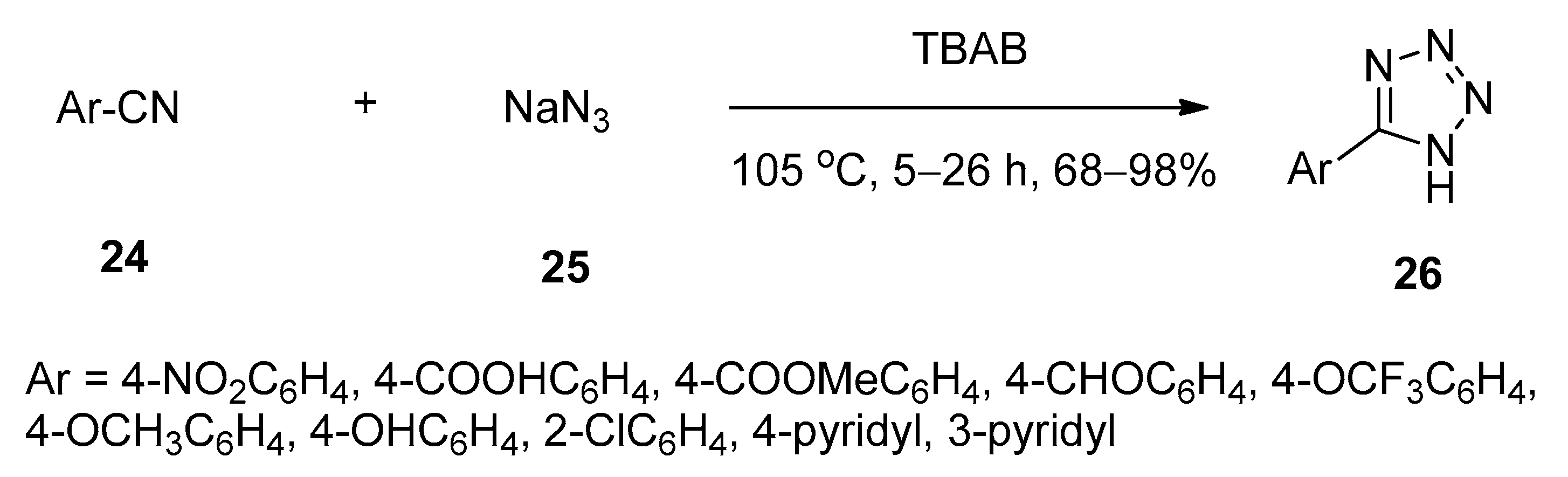
2.8. Synthesis of 5-Substituted 1H-Tetrazoles
3. Applications of TBAB for the Synthesis of Bioactive O-Heterocycles
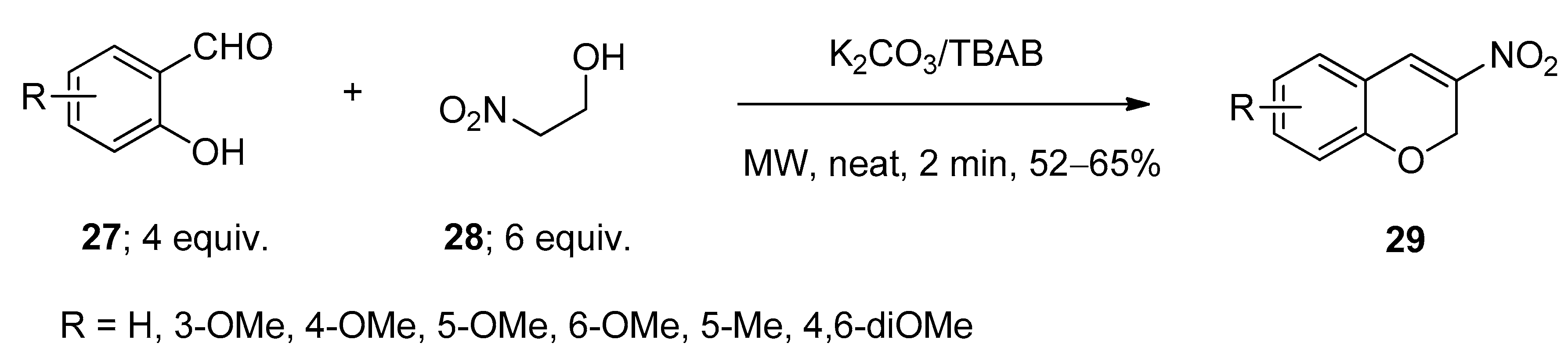
3.1. Synthesis of 3-Nitro-2H-Chromenes
3.2. Synthesis of 3,4-Dihydropyrano[c]chromene
3.3. Synthesis of 4-Phenyl-1,3-Dioxolan-2-One
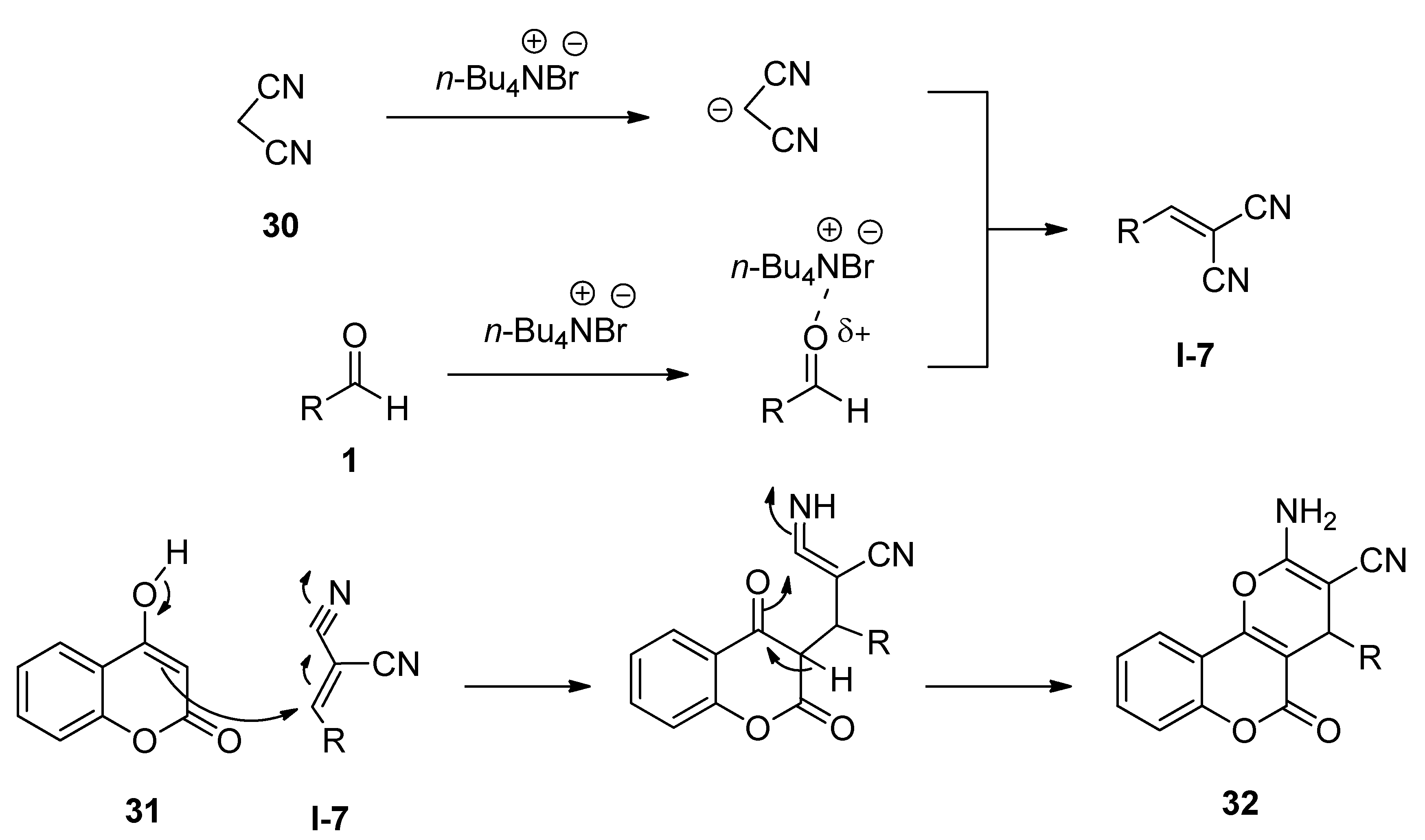
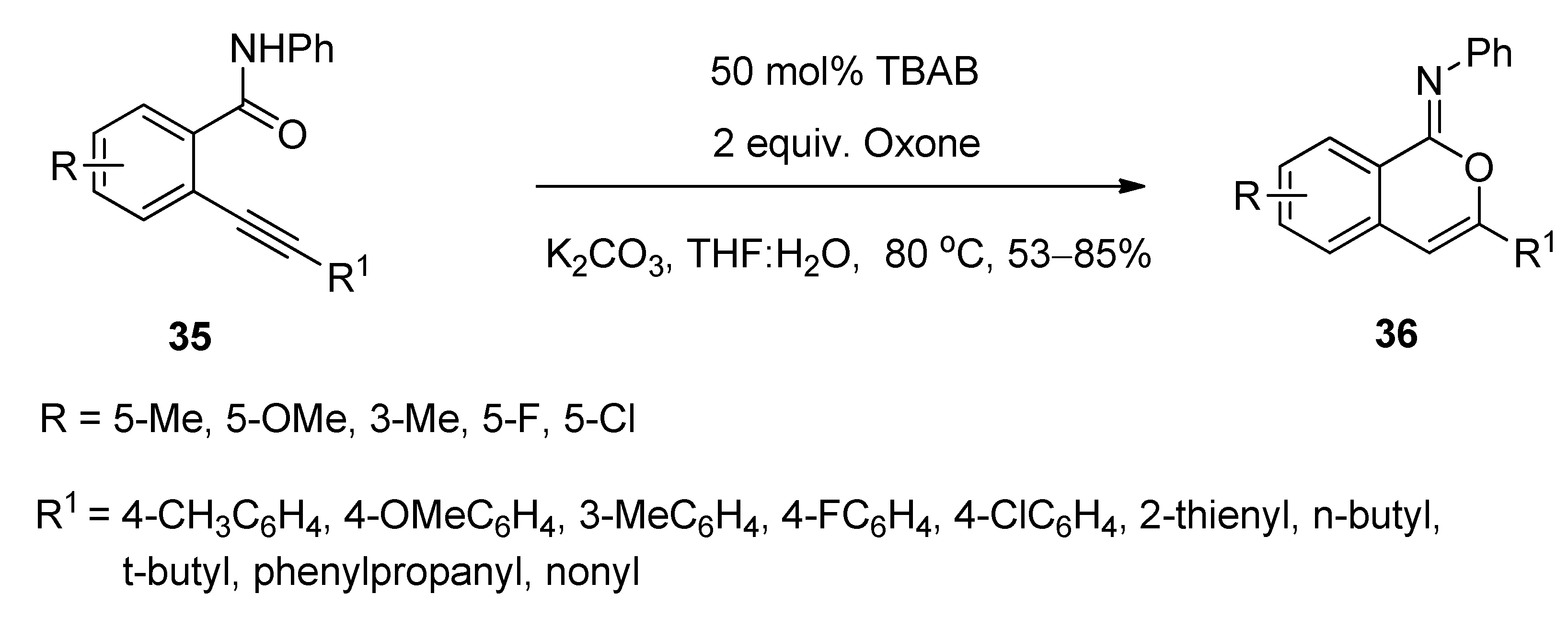
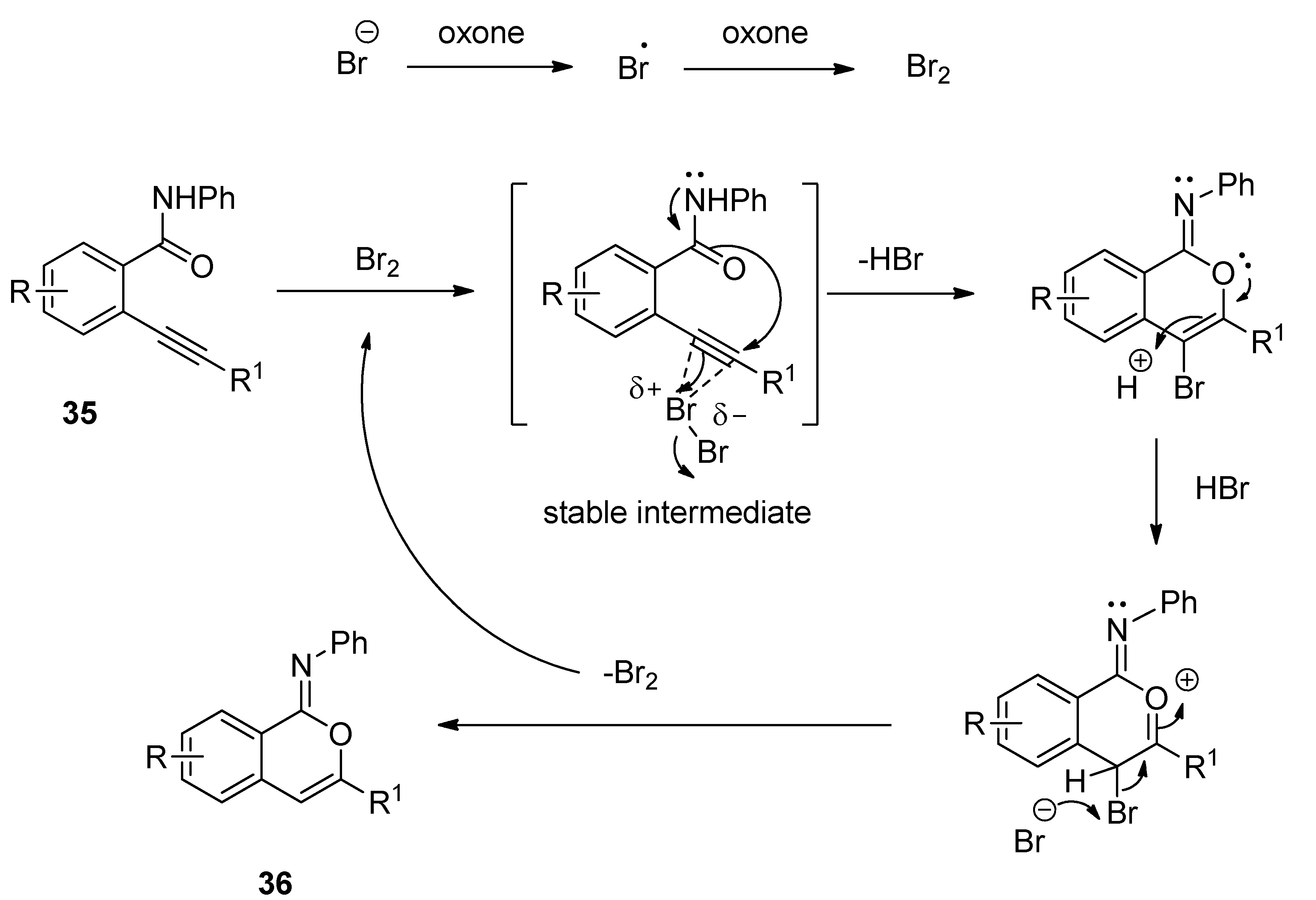
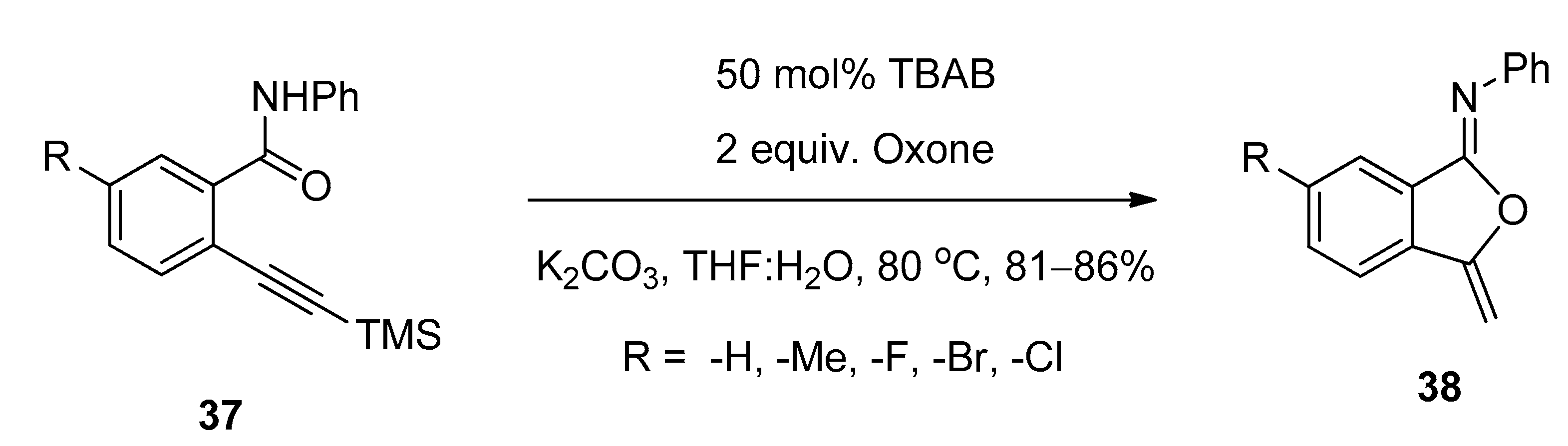
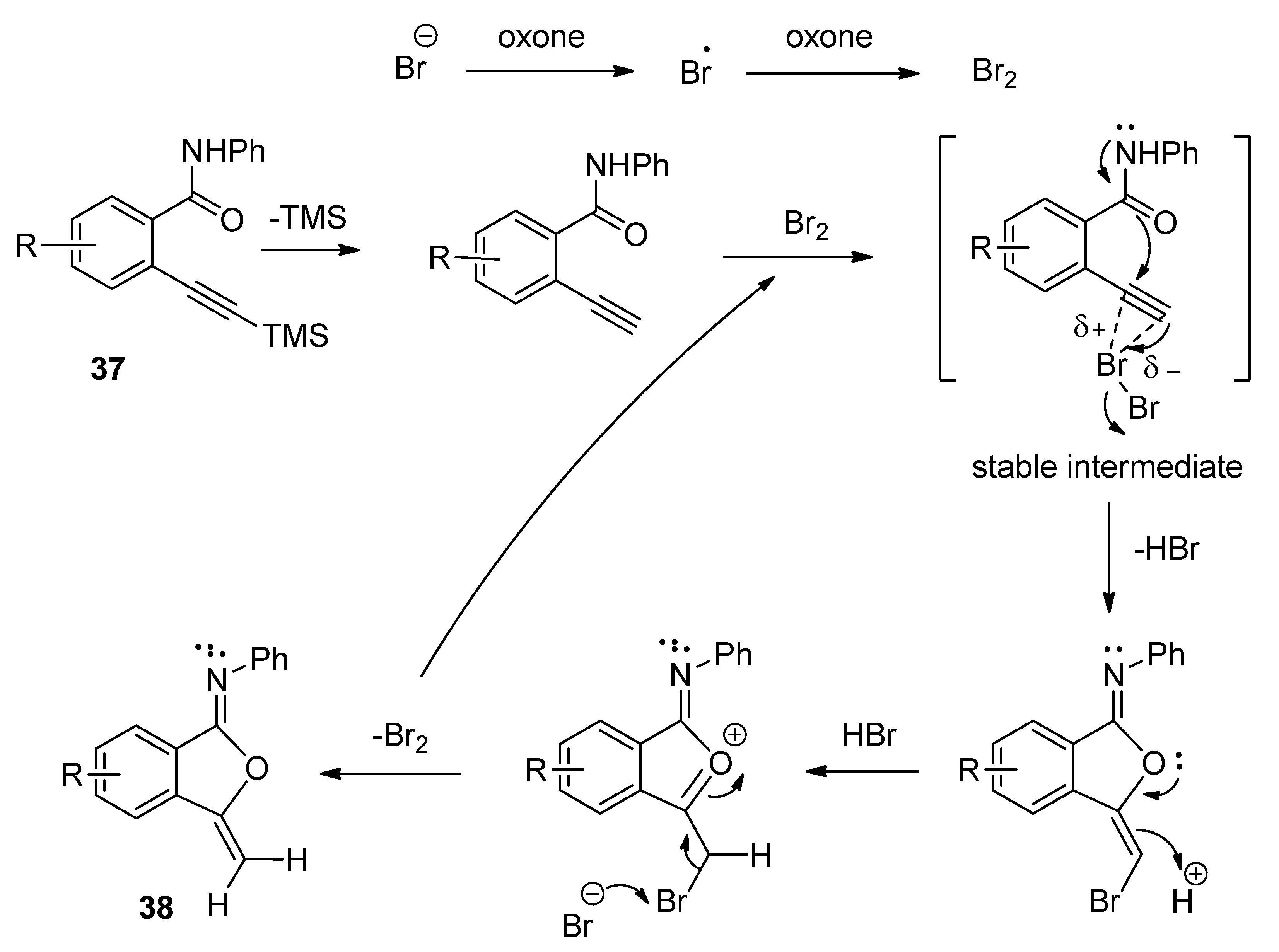
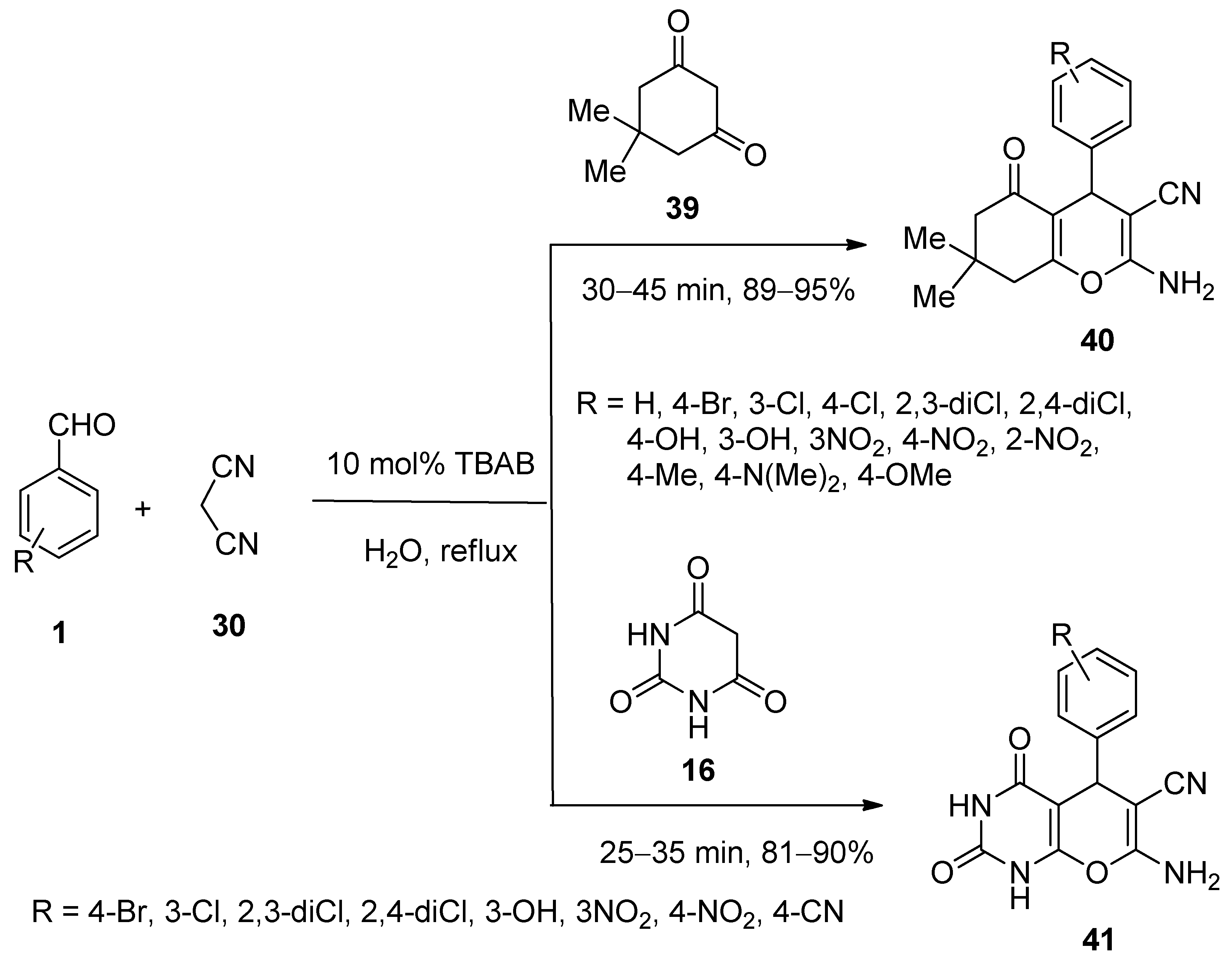
3.4. Synthesis of Isocoumarin-1-Imines and Isobenzofuran-1-Imines
3.5. Synthesis of Tetrahydrobenzo[b]pyran
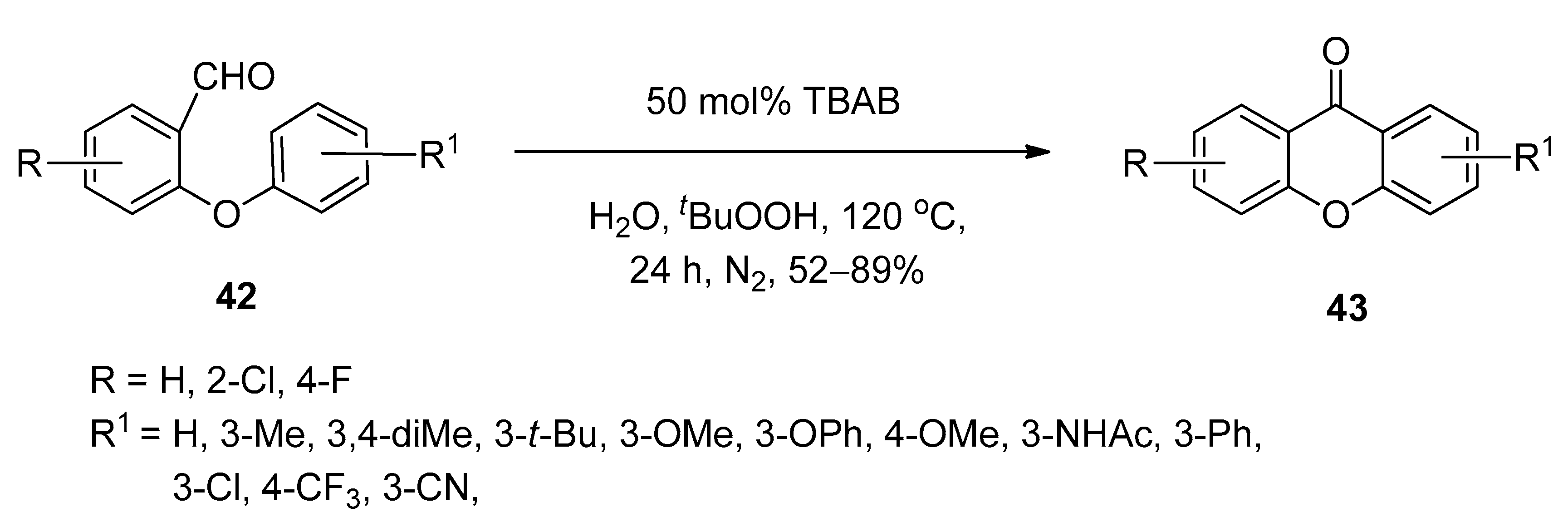
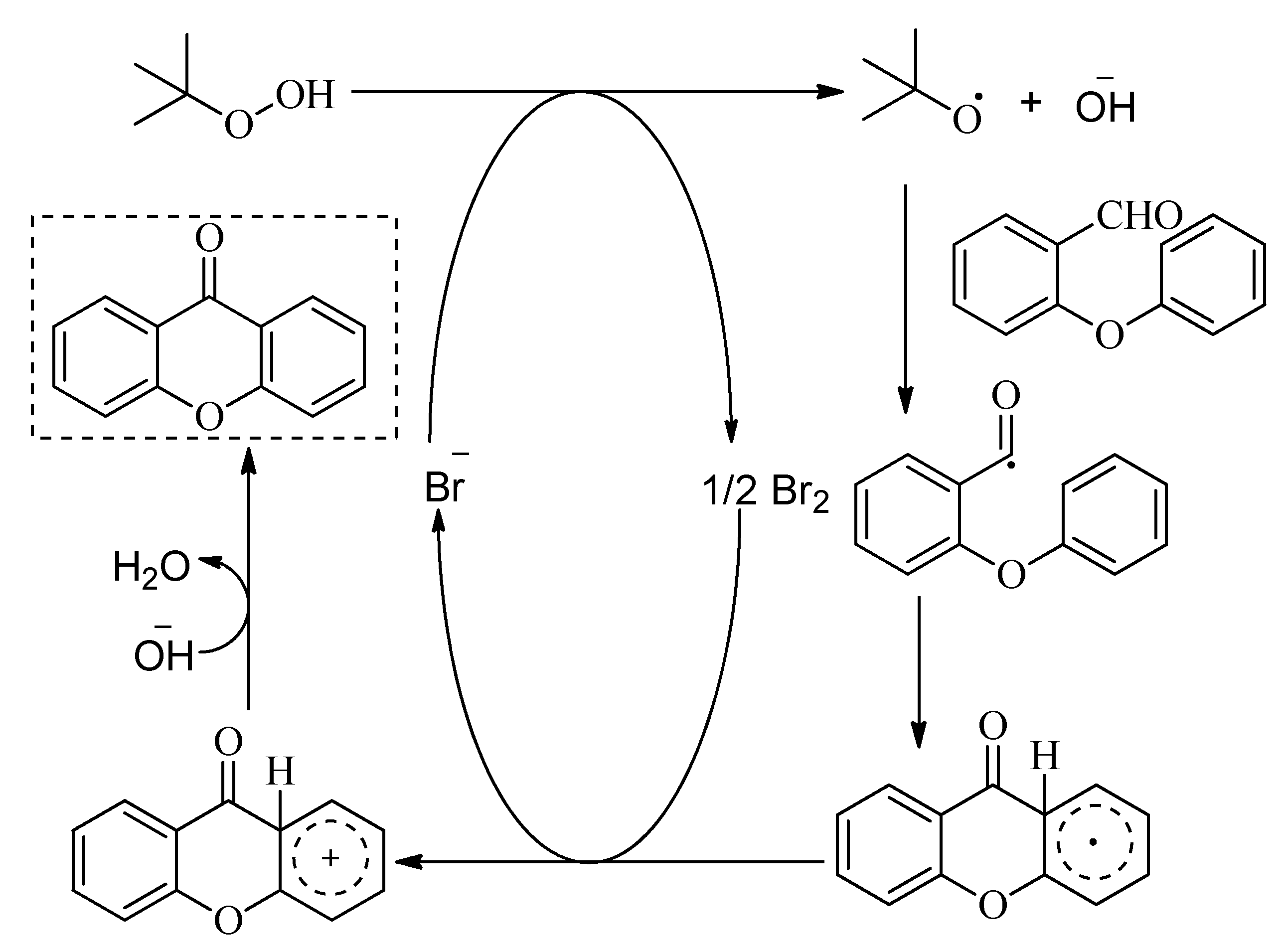
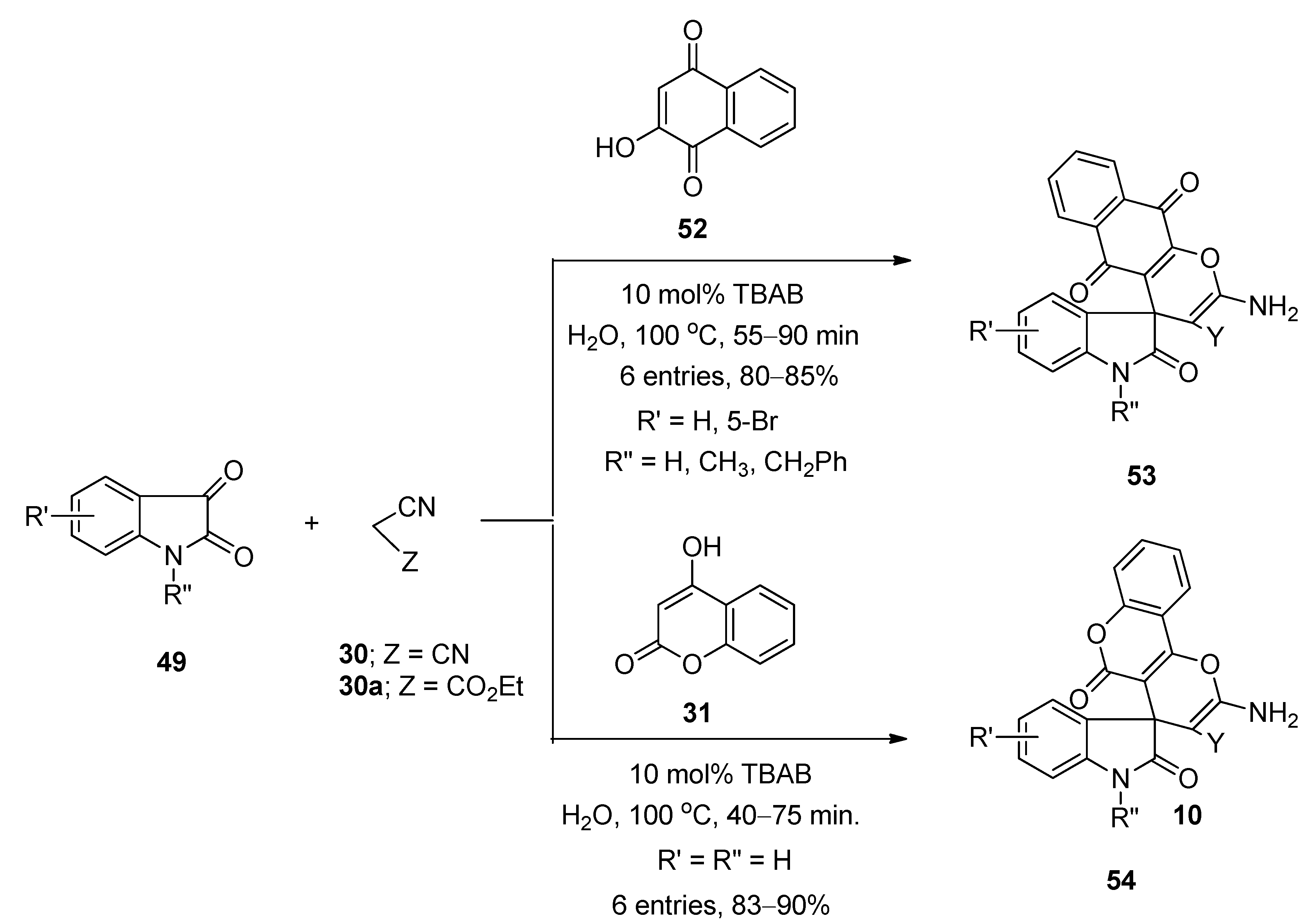
3.6. Synthesis of Xanthones
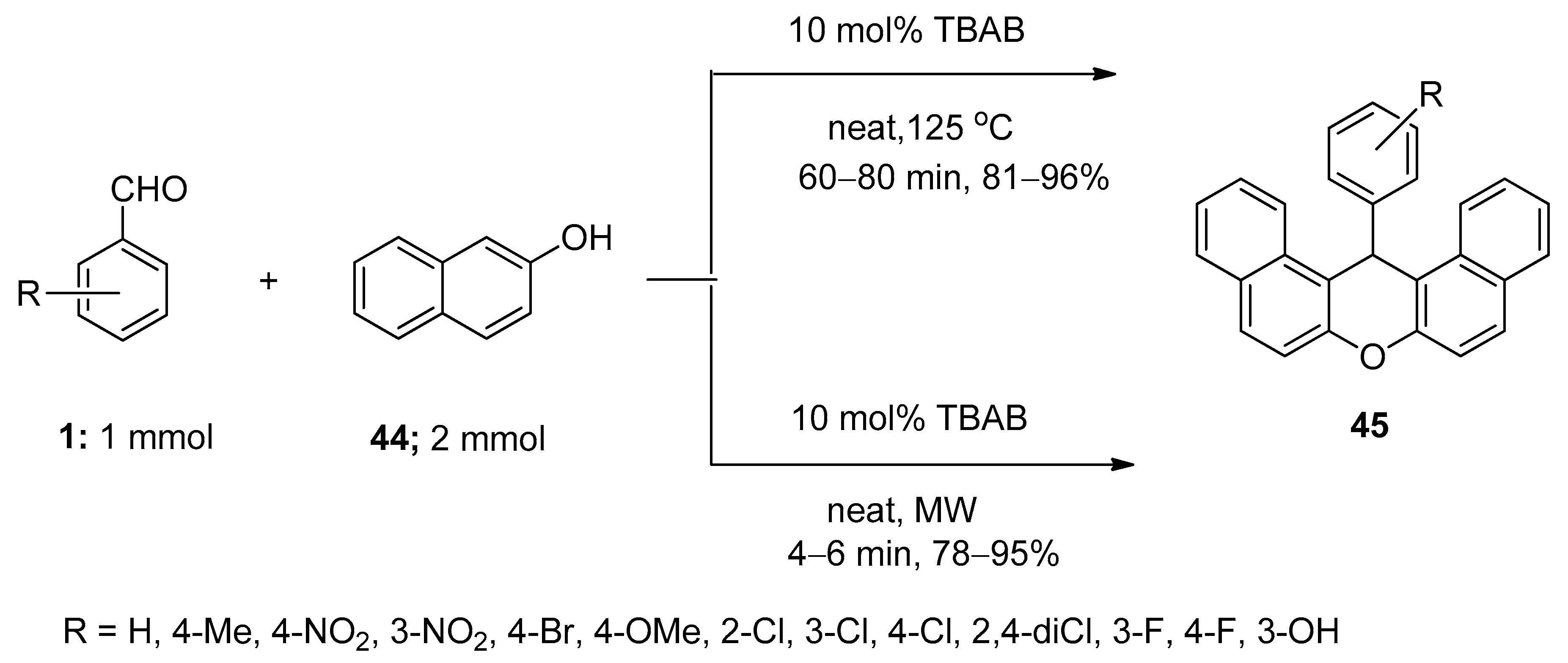
3.7. Synthesis of Aryl-14H-Dibenzo[a.j]xanthenes
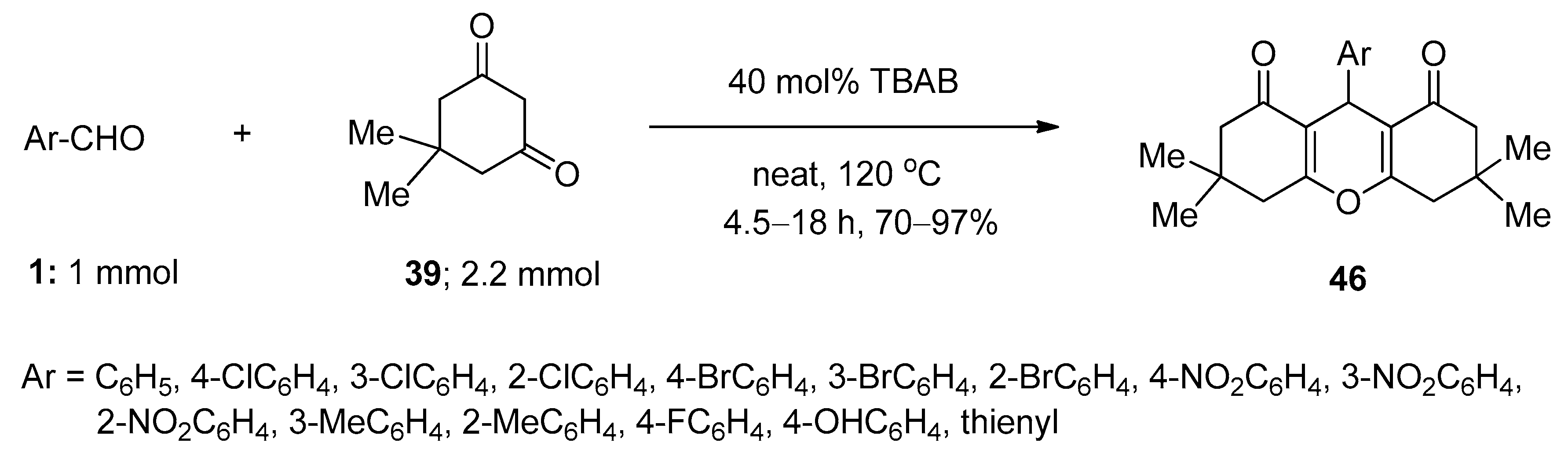
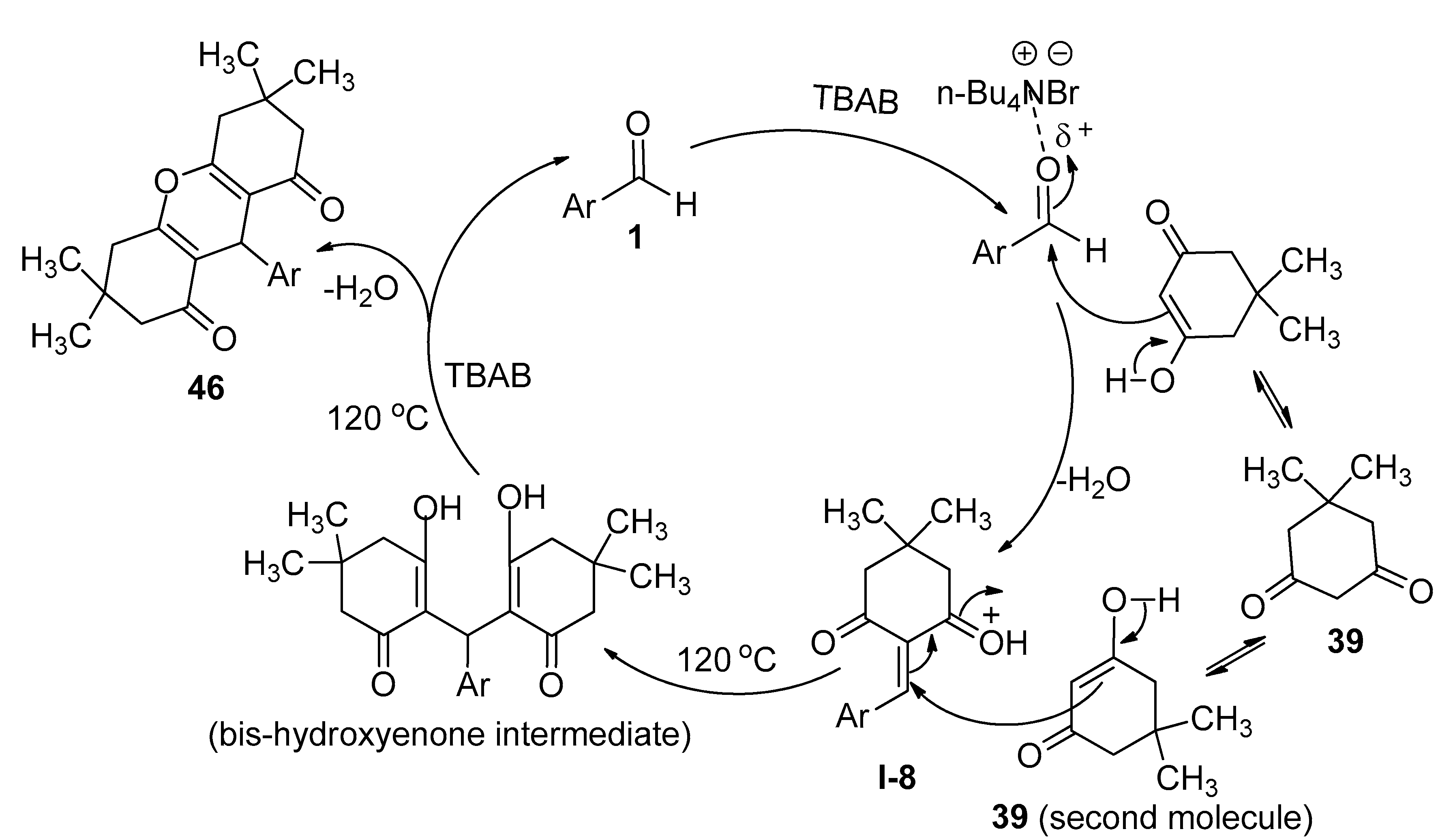
3.8. Synthesis of 1,8-Dioxo-Octahydroxanthenes
4. Applications of TBAB for the Synthesis of Bioactive N- as well as O-Heterocycles
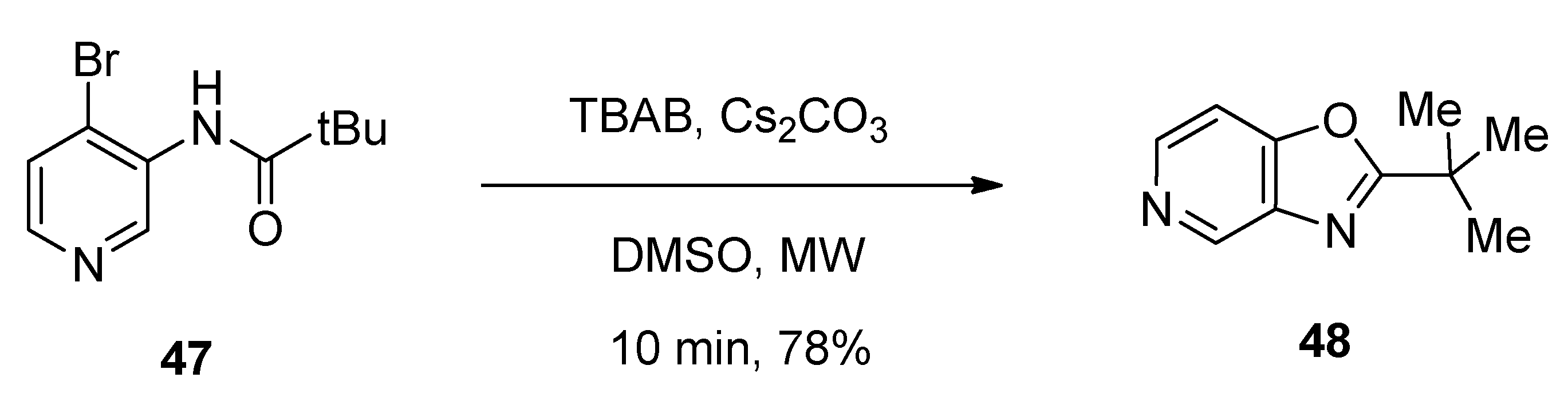
4.1. Synthesis of Oxazolo[4,5-c]pyridine
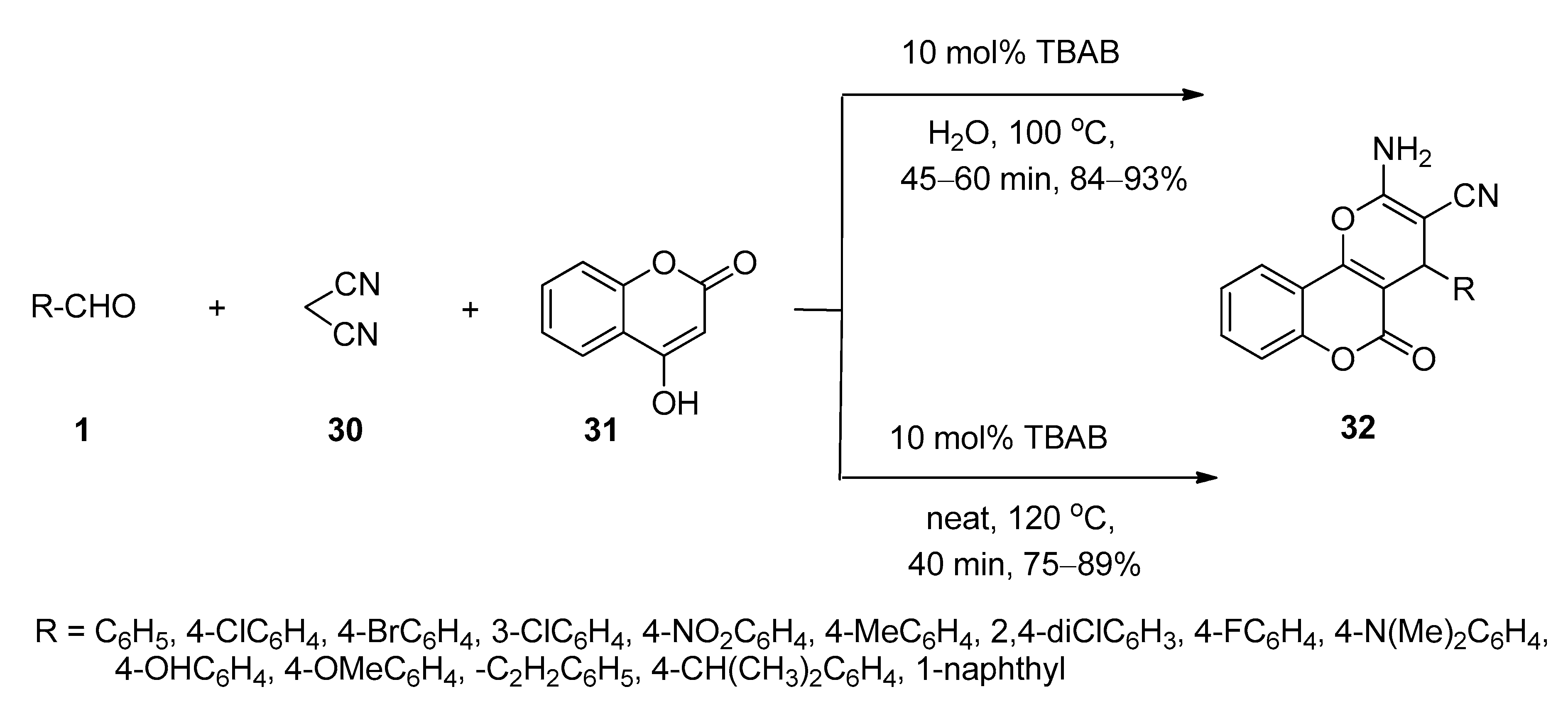
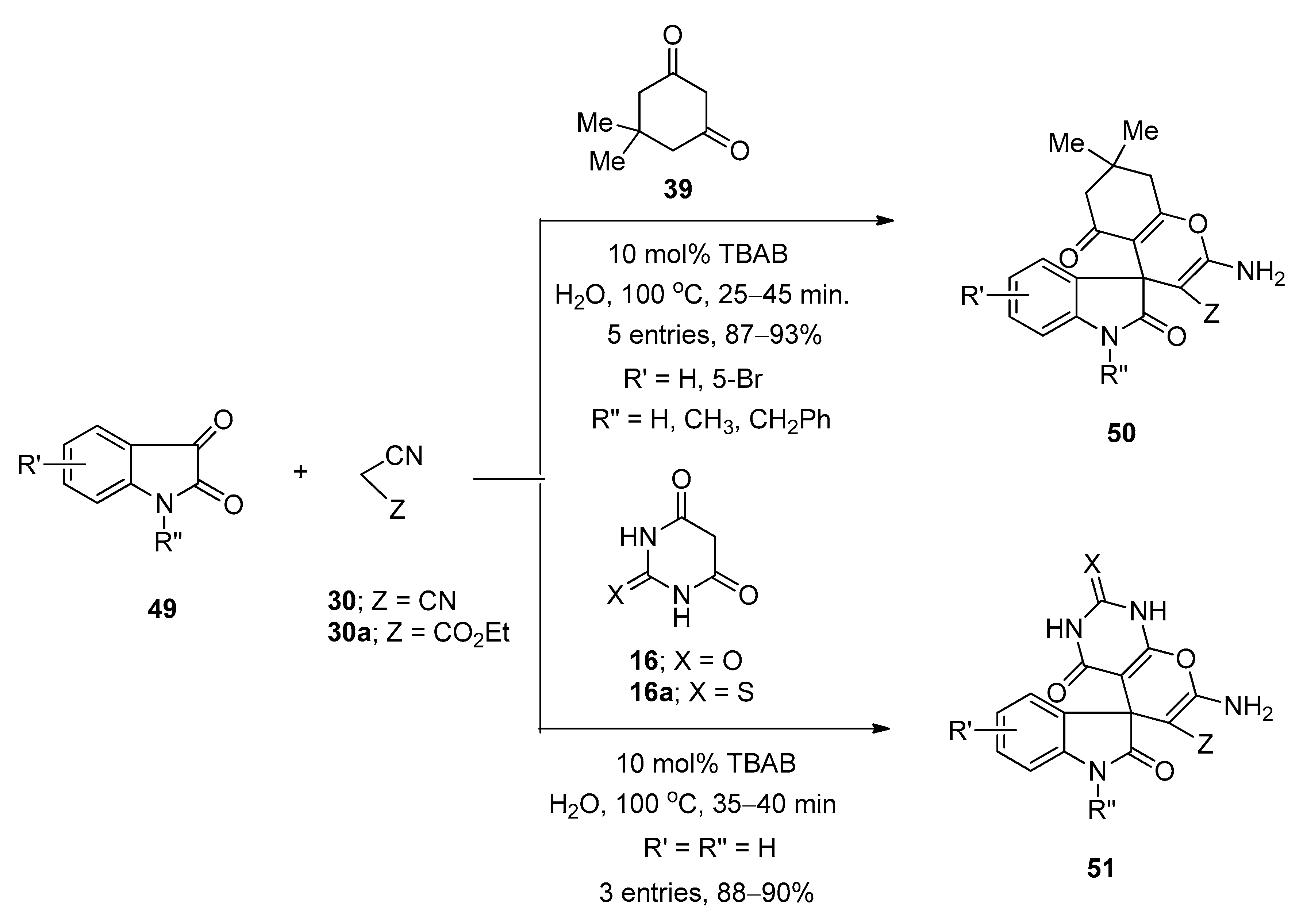
4.2. Synthesis of Pyran-Fused Spirooxindoles
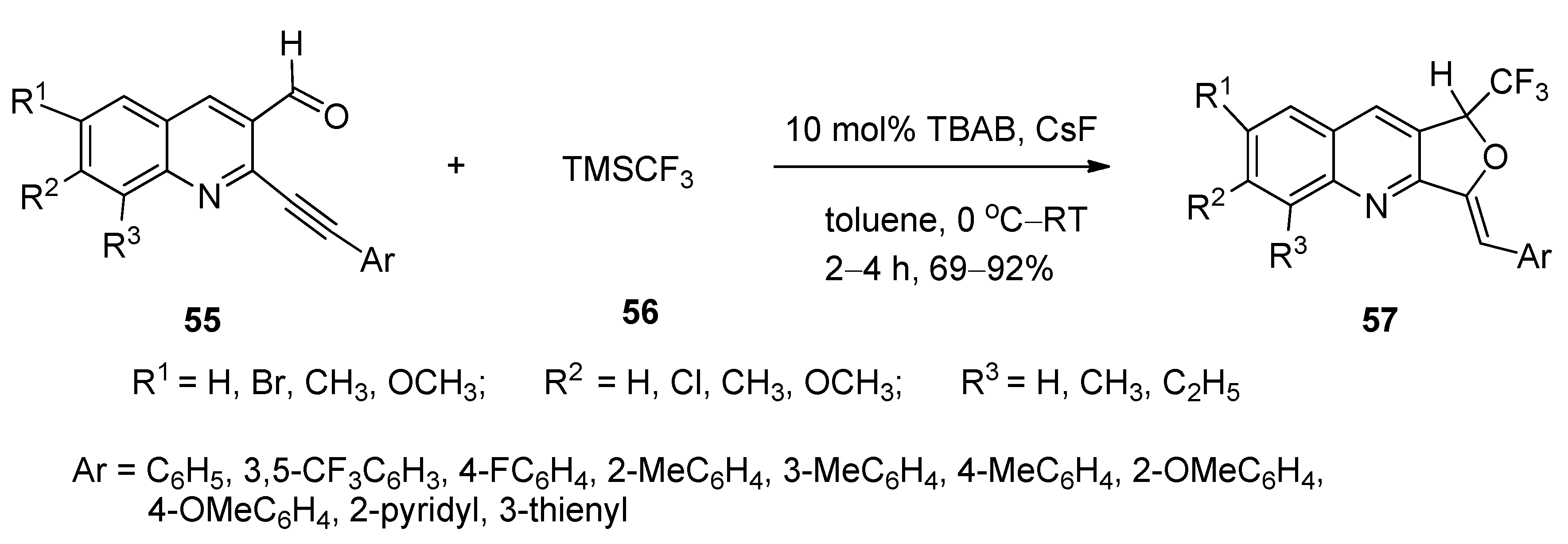
4.3. Synthesis of 1-Trifluoromethyl-3-Alkylidene-1,3-Dihydrofuro[3,4-b]quinolines
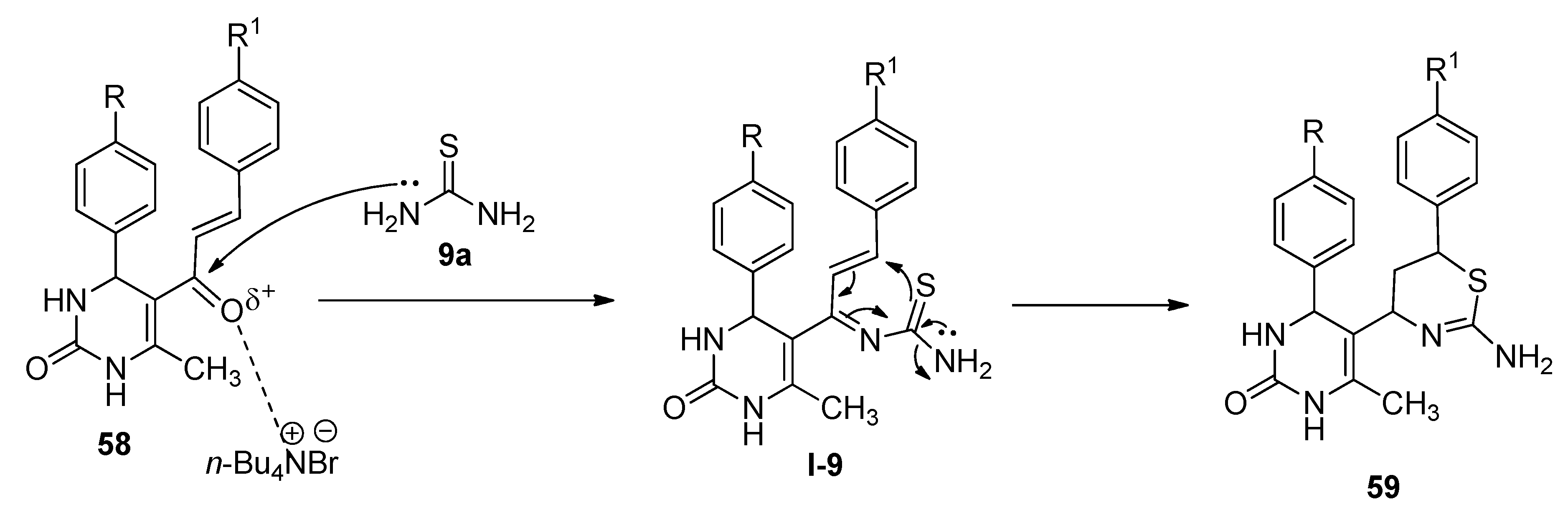
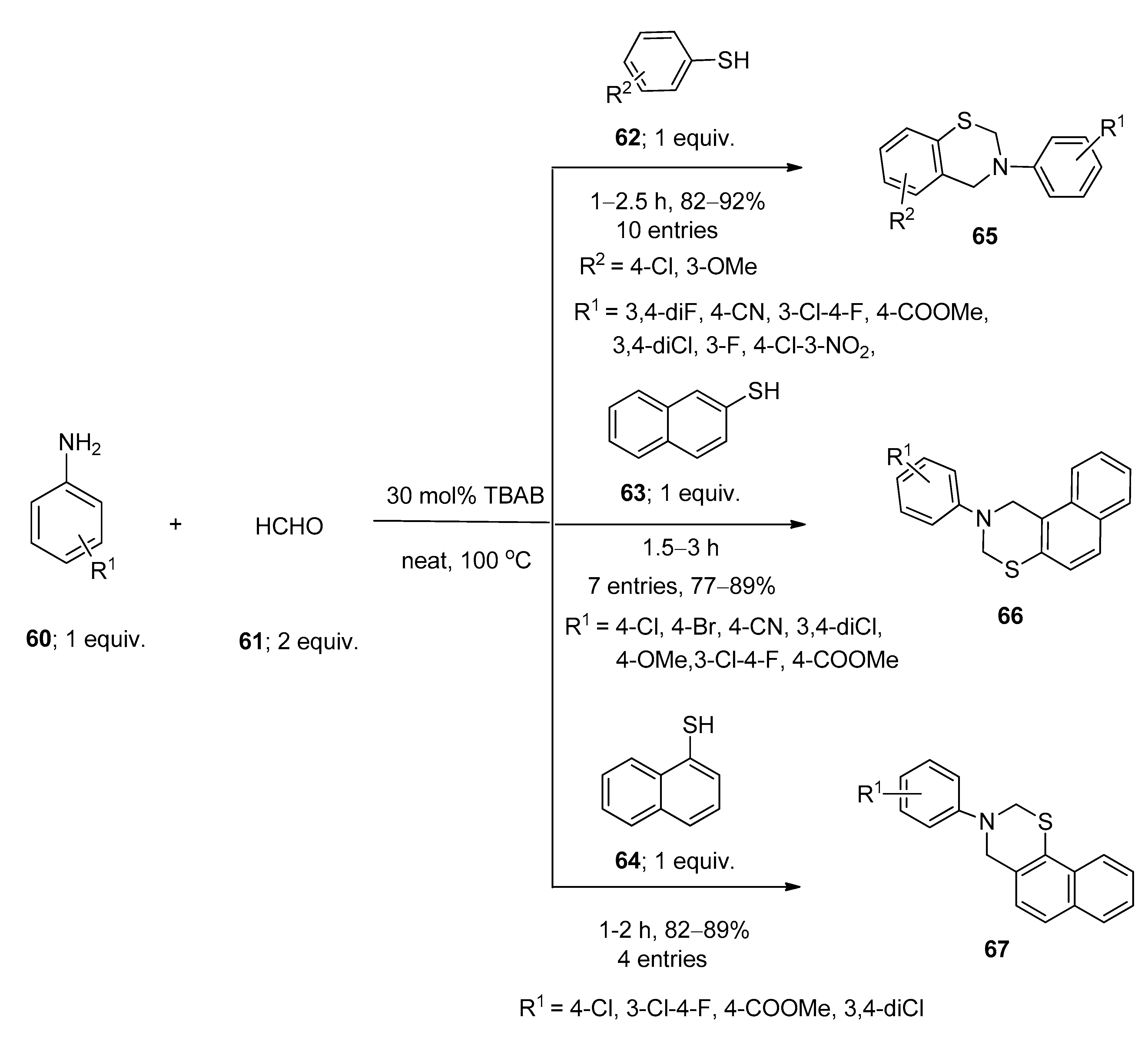
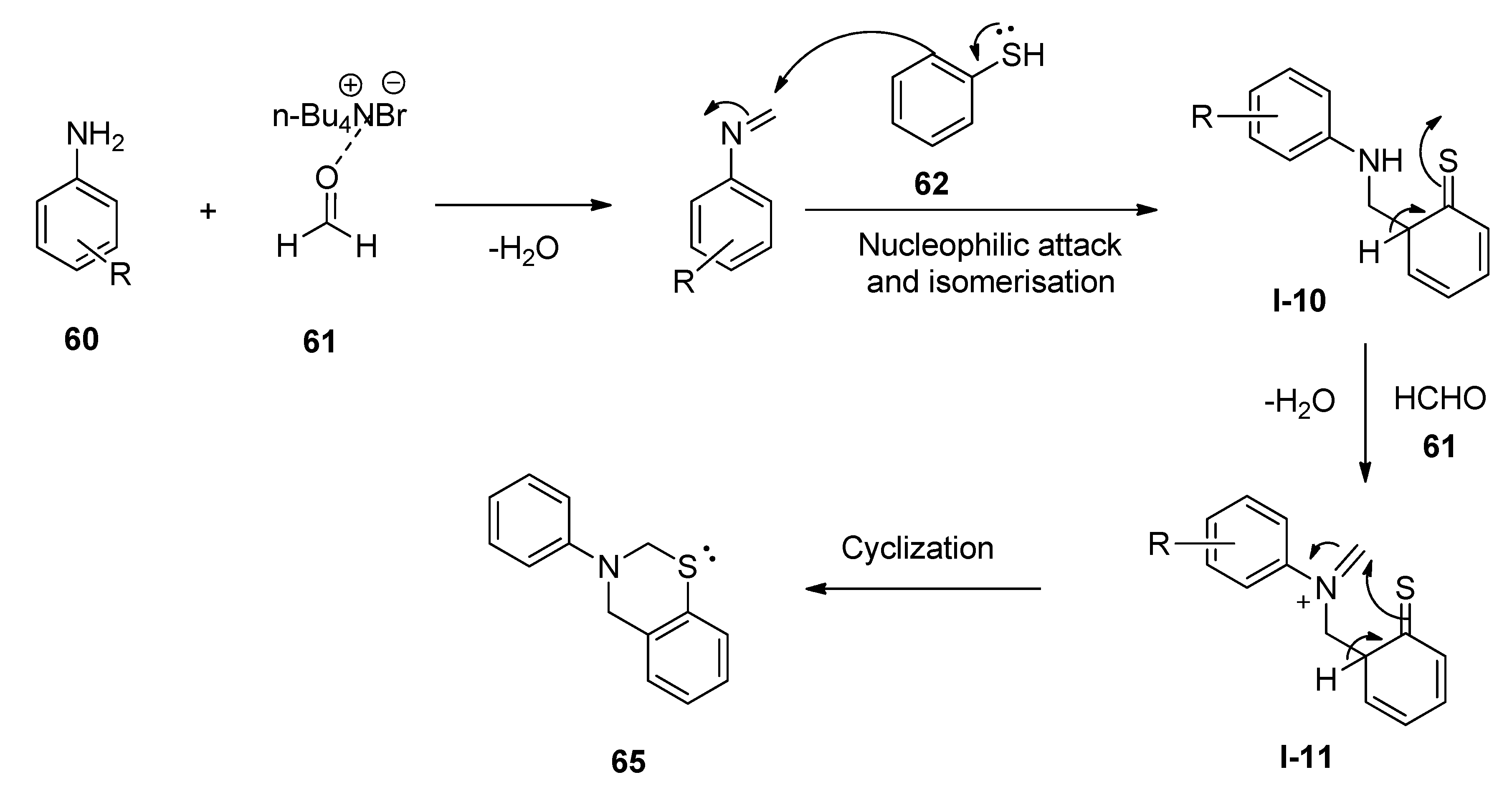
5. Applications of TBAB for the Synthesis of Bioactive N- as well as S-Heterocycles
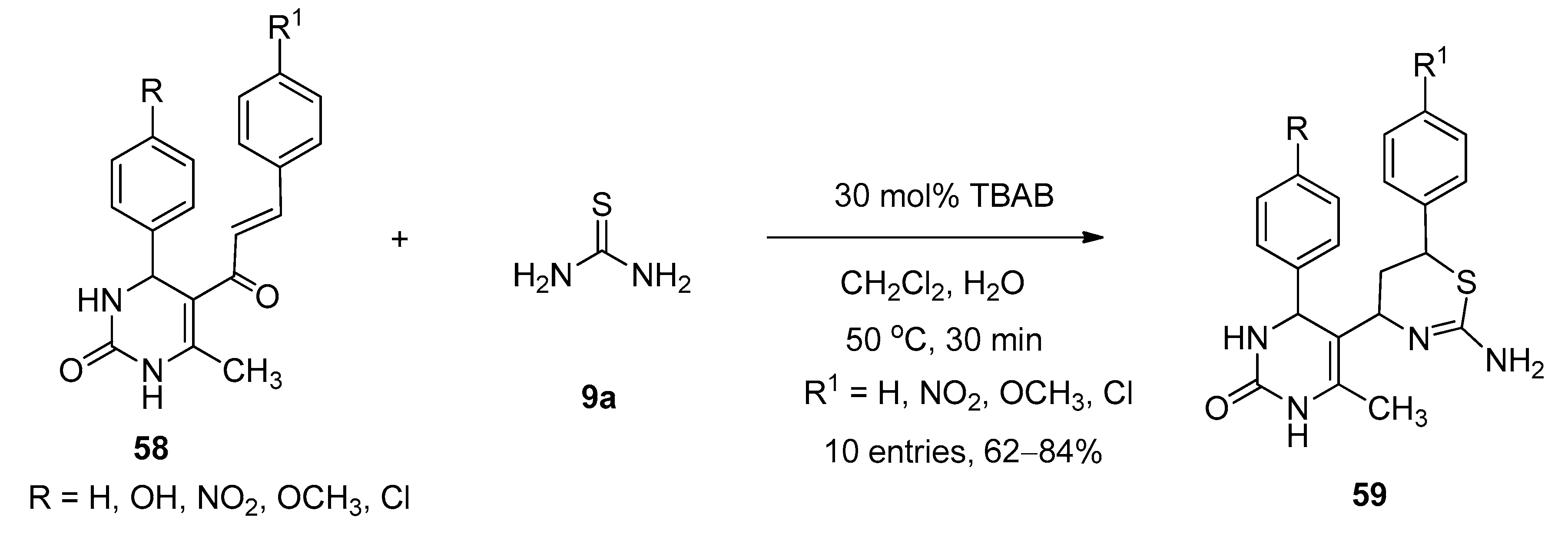
Synthesis of 1,3-Thiazine Derivatives
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Hinkle, J.L.; Cheever, K.H. Brunner & Suddarth’s Textbook of Medical-Surgical Nursing, 13th ed.; Wolters Kluwer Health/Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2014. [Google Scholar]
- Brahmachari, G. Handbook of Pharmaceutical Natural Products, 1st ed.; Wiley-VCH: Weinheim, Germany, 2010. [Google Scholar]
- Abd-Ellah, H.S.; Abdel-Aziz, M.; Shoman, M.E.; Beshr, E.A.M.; Kaoud, T.S.; Ahmed, A.S.F.F. Novel 1,3,4-oxadiazole/oxime hybrids: Synthesis, docking studies and investigation of anti-inflammatory, ulcerogenic liability and analgesic activities. Bioorg. Chem. 2016, 69, 48–63. [Google Scholar] [CrossRef] [PubMed]
- Kitchener, S.; Nasveld, P.; Edstein, M.D. Tafenoquine for the treatment of recurrent Plasmodium vivax malaria. Am. J. Trop. Med. Hyg. 2007, 76, 494–496. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.V.S.; Rajendraprasad, Y.; Mallikarjuna, B.P.; Chandrashekar, S.M.; Kistayya, C. Synthesis of some novel 2-substituted-5-[isopropylthiazole] clubbed 1,2,4-triazole and 1,3,4-oxadiazoles as potential antimicrobial and antitubercular agents. Eur. J. Med. Chem. 2010, 45, 2063–2074. [Google Scholar] [CrossRef] [PubMed]
- Al-Issa, S.A. Synthesis and anticancer activity of some fused pyrimidines and related heterocycles. Saudi Pharm. J. 2013, 21, 305–316. [Google Scholar] [CrossRef] [PubMed]
- Chabukswar, A.R.; Kuchekar, B.S.; Jagdale, S.C.; Lokhande, P.D.; Chabukswar, V.V.; Shisodia, S.U.; Mahabal, R.H.; Londhe, A.M.; Ojha, N.S. Synthesis and evaluation of analgesic, anti-asthmatic activity of (E)-1-(8-hydroxyquinolin-7-yl)-3-phenylprop-2-en-1 ones. Arab. J. Chem. 2016, 9, 704–712. [Google Scholar] [CrossRef]
- Janssens, F.; Torremans, J.; Janssen, M.; Stokbroekx, R.A.; Luyckx, M.; Janssen, P.A. New antihistaminic N-heterocyclic 4-piperidinamines. 1. Synthesis and antihistaminic activity of N-(4-piperidinyl)-1H-benzimidazol-2-amines. J. Med. Chem. 1985, 28, 1925–1933. [Google Scholar] [CrossRef]
- Baldwin, J.J.; Engelhardt, E.L.; Hirschmann, R.; Ponticello, G.S.; Atkinson, J.G.; Wasson, B.K.; Sweet, C.S.; Scriabine, A. Heterocyclic analogues of the antihypertensive beta-adrenergic blocking agent (S)-2-[3-(ter-butylamino)-2-hydroxypropoxy]-3-cyanopyridine. J. Med. Chem. 1980, 23, 65–70. [Google Scholar] [CrossRef]
- Gaikwad, D.D.; Chapolikar, A.D.; Devkate, C.G.; Warad, K.D.; Tayade, A.P.; Pawar, R.P.; Domb, A.J. Synthesis of indazole motifs and their medicinal importance: An overview. Eur. J. Med. Chem. 2015, 90, 707–731. [Google Scholar] [CrossRef]
- Ali, T.E.-S. Synthesis of some novel pyrazolo[3,4-b]pyridine and pyrazolo[3,4-d]pyrimidine derivatives bearing 5,6-diphenyl-1,2,4-triazine moiety as potential antimicrobial agents. Eur. J. Med. Chem. 2009, 44, 4385–4392. [Google Scholar] [CrossRef]
- Matsuoka, H.; Ohi, N.; Mihara, M.; Suzuki, H.; Miyamoto, K.; Maruyama, N.; Tsuji, K.; Kato, N.; Akimoto, T.; Takeda, Y.; et al. Antirheumatic agents: Novel methotrexate derivatives bearing a benzoxazine or benzothiazine moiety. J. Med. Chem. 1997, 40, 105–111. [Google Scholar] [CrossRef]
- Vinodkumar, R.; Vaidya, S.D.; Kumar, B.V.S.; Bhise, U.N.; Bhirud, S.B.; Mashelkar, U.C. Synthesis, anti-bacterial, anti-asthmatic and anti-diabetic activities of novel N-substituted-2-(4-phenylethynyl-phenyl)-1H-benzimidazoles and N-substituted 2[4-(4,4-dimethyl-thiochroman-6-yl-ethynyl)-phenyl)-1H-benzimidazoles. Eur. J. Med. Chem. 2008, 43, 986–995. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, B. Recent developments on ultrasound-assisted one-pot multicomponent synthesis of biologically relevant heterocycles. Ultrason. Sonochem. 2017, 35, 15–35. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wei, P.; Wang, L.; Wang, Q. Design, synthesis, and anti-tobacco mosaic virus (TMV) activity of phenanthroindolizidines and their analogues. J. Agric. Food Chem. 2012, 60, 10212–10219. [Google Scholar] [CrossRef] [PubMed]
- Su, B.; Cai, C.; Deng, M.; Liang, D.; Wang, L.; Wang, Q. Design, synthesis, antiviral activity, and SARs of 13a-substituted phenanthroindolizidine alkaloid derivatives. Bioorg. Med. Chem. Lett. 2014, 24, 2881–2884. [Google Scholar] [CrossRef]
- Banerjee, B. Ultrasound and nano-catalysts: An ideal and sustainable combination to carry out diverse organic transformations. ChemistrySelect 2019, 4, 2484–2500. [Google Scholar] [CrossRef]
- Banik, B.K.; Fernandez, M.; Alvarez, C. Iodine-catalyzed highly efficient Michael reaction of indoles under solvent-free condition. Tetrahedron Lett. 2005, 46, 2479–2482. [Google Scholar] [CrossRef]
- Banerjee, B. Recent developments on organo-bicyclo-bases catalyzed multicomponent synthesis of biologically relevant heterocycles. Curr. Org. Chem. 2018, 22, 208–233. [Google Scholar] [CrossRef]
- Kaur, G.; Bala, K.; Devi, S.; Banerjee, B. Camphorsulfonic acid (CSA): An efficient organocatalyst for the synthesis or derivatization of heterocycles with biologically promising activities. Curr. Green Chem. 2018, 5, 150–167. [Google Scholar] [CrossRef]
- Starks, C.M. Phase-transfer catalysis. I. Heterogeneous reactions involving anion transfer by quaternary ammonium and phosphonium salts. J. Am. Chem. Soc. 1971, 93, 195–199. [Google Scholar] [CrossRef]
- Charette, A.B.; Chinchilla, R.; Nájera, C. Tetrabutylammonium bromide. In Encyclopedia of Reagents for Organic Synthesis; John Wiley & Sons, Ltd.: Chichester, West Sussex, UK, 2007. [Google Scholar]
- Ranu, B.C.; Dey, S.S.; Hajra, A. Catalysis by an ionic liquid: Efficient conjugate addition of thiols to electron deficient alkenes catalyzed by molten tetrabutylammonium bromide under solvent-free conditions. Tetrahedron 2003, 59, 2417–2421. [Google Scholar] [CrossRef]
- Calo, V.; Nacci, A.; Monopoli, A.; Fornaro, A.; Sabbatini, L.; Cioffi, N.; Ditaranto, N. Heck reaction catalyzed by nanosized palladium on chitosan in ionic liquids. Organometallics 2004, 23, 5154–5158. [Google Scholar] [CrossRef]
- Calo, V.; Nacci, A.; Lopez, L.; Mannarini, N. Heck reaction in ionic liquids catalyzed by a Pd–benzothiazole carbene complex. Tetrahedron Lett. 2000, 41, 8973–8976. [Google Scholar] [CrossRef]
- Orlińska, B.J.; Zawadiak, J.M. Copper (II) chloride/tetrabutylammonium bromide catalyzed oxidation of 2,6-diisopropylnaphthalene and 4, 4′-diisopropylbiphenyl. Cent. Eur. J. Chem. 2010, 8, 285–290. [Google Scholar] [CrossRef]
- Wang, F.; Li, X.; Li, Z.; Zhou, S.; Zhang, W. Copper Salts/TBAB-Catalyzed Chemo- and Regioselective β-C(sp3)−H Acyloxylation of Aliphatic Amides. ACS Omega 2019, 4, 331–343. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-H.; Li, J.-L.; Xie, Y.-X. TBAB-promoted ligand-free copper-catalyzed cross-coupling reactions of aryl halides with arylboronic acids. Synthesis 2007, 2007, 984–988. [Google Scholar] [CrossRef]
- Dong, F.; Liu, J.-Q.; Wang, X.-S. An efficient synthesis of biaryl diamides via Ullmann coupling reaction catalyzed by CuI in the presence of Cs2CO3 and TBAB. Res. Chem. Intermed. 2018, 44, 5271–5283. [Google Scholar] [CrossRef]
- Fontaine, P.; Chiaroni, A.; Masson, G.; Zhu, J. One-pot three-component synthesis of α-iminonitriles by ibx/tbab-mediated oxidative Strecker reaction. Org. Lett. 2008, 10, 1509–1512. [Google Scholar] [CrossRef]
- Johnson, C.R.; Ansari, M.I.; Coop, A. Tetrabutylammonium Bromide-Promoted metal-free, efficient, rapid, and scalable synthesis of N-aryl amines. ACS Omega 2018, 3, 10886–10890. [Google Scholar] [CrossRef]
- Dubey, P.K.; Reddy, P.P.; Srinivas, K. A Facile Solvent-Free Synthesis of 1-Alkyl/Aralkyl-2-(1-arylsulfonyl alkyl) benzimidazoles using ‘‘TBAB” as surface catalyst. J. Heterocycl. Chem. 2010, 47, 1317–1322. [Google Scholar] [CrossRef]
- Yao, Y.; Wang, Z.; Wang, B. Tetra-n-butylammonium bromide (TBAB)-initiated carbonylation-peroxidation of styrene derivatives with aldehydes and hydroperoxides. Org. Chem. Front. 2018, 5, 2501–2504. [Google Scholar] [CrossRef]
- Weil, T.; Schreiner, P.R. Organocatalytic alkynylation of aldehydes and ketones under phase-transfer catalytic conditions. Eur. J. Org. Chem. 2005, 2005, 2213–2217. [Google Scholar] [CrossRef]
- Majumdar, K.C.; Sarkar, S.; Ghosh, S. Studies in thio-claisen rearrangement: Regioselective synthesis of thiopyrano[2,3-b]pyran-2-ones and thieno[2,3-b]pyran-2-ones. Synth. Commun. 2004, 34, 1265–1275. [Google Scholar] [CrossRef]
- Wang, C.; Hang, T.; Zhang, H. Microwave-promoted N-alkylation of acridones without solvent. Synth. Commun. 2003, 33, 451–456. [Google Scholar] [CrossRef]
- Liu, Z.-Q.; You, P.-S.; Zhang, L.-D.; Liu, D.-Q.; Liu, S.-S.; Guan, X.-Y. TBAB-catalyzed 1, 6-conjugate sulfonylation of para-quinone methides: A highly efficient approach to unsymmetrical gem-diarylmethyl sulfones in water. Molecules 2020, 25, 539. [Google Scholar] [CrossRef]
- Li, B.; Zhang, Z. POPd/TBAB co-catalyzed Suzuki cross-coupling reaction of heteroaryl chlorides/bromides with 4-fluorophenylboronic acid in water. J. Iran. Chem. Soc. 2016, 13, 637–644. [Google Scholar] [CrossRef]
- Zhao, H.; Cai, M.-Z.; Peng, C.-Y. Stereoselective synthesis of (e)-cinnamonitriles via Heck arylation of acrylonitrile and aryl iodides in water. Synth. Commun. 2002, 32, 3419–3423. [Google Scholar] [CrossRef]
- Youa, E.; Lia, P.; Wang, L. Nickel(0)/tetra-n-butylammonium bromide (TBAB) catalyzed Suzuki-Miyaura reaction. Synthesis 2006, 2006, 1465–1469. [Google Scholar] [CrossRef]
- Sirisha, K.; Achaiah, G.; Reddy, V.M. Facile synthesis and antibacterial, antitubercular, and anticancer activities of novel 1, 4-dihydropyridines. Arch. Pharm. 2010, 343, 342–352. [Google Scholar] [CrossRef]
- Vo, D.; Matome, W.C.; Ramesh, M.; Iqbal, V.; Wolowyk, M.W.; Howlett, S.E.; Knauss, E.E. Syntheses, Calcium channel agoinst-antagonist modulation activities, and voltage-clamp studies of isopropyl 1,4-dihydro-2,6-dimethyl-1-3-nitro-4-pyridinyl-5-carboxylate racemates and enantiomers. J. Med. Chem. 1995, 38, 2851–2859. [Google Scholar] [CrossRef]
- Bazargan, L.; Fouladdel, S.; Shafiee, A.; Amini, M.; Ghaffari, S.M.; Azizi, E. Evaluation of anticancer effects of newly synthesized dihydropyridine derivatives in comparison to verapamil and dozorubicin on T47D parental and resistant cell lines in vitro. Cell. Biol. Toxicol. 2008, 24, 165–174. [Google Scholar] [CrossRef]
- Hilgeroth, A.; Wiese, M.; Billich, A. Synthesis and biological evaluation of the first N-alkyl cage dimeric 4-aryl-1,4-dihydropyridines as novel nonpeptidic HIV-1 protease inhibitors. J. Med. Chem. 1999, 42, 4729–4732. [Google Scholar] [CrossRef] [PubMed]
- Tusell, J.M.; Barron, S.; Serratosa, J. Anticonvulsant activity of calcium channel blockers and calmodulin antagonists in seizures induced by lindane and other convulsant drugs. Brain Res. 1993, 622, 99–104. [Google Scholar] [CrossRef]
- Khoshneviszadeh, M.; Edraki, N.; Javidnia, K.; Alborzi, A.; Pourabbas, B.; Mardanesh, J.; Miri, R. Synthesis and biological evaluation of some new 1,4-dihydropyridines containing different ester substitute and diethyl carbamoyl group as antitubercular agents. Bioorg. Med. Chem. 2009, 17, 1579–1586. [Google Scholar] [CrossRef] [PubMed]
- Vohra, R.K.; Bruneau, C.; Renaud, J.L. Lewis acid-catalysed sequential transformations Straightforward preparation of functional dihydropyridines. Adv. Synth. Catal. 2006, 348, 2571–2574. [Google Scholar] [CrossRef]
- Wang, L.M.; Sheng, J.; Zhang, L.; Han, J.W.; Fan, Z.Y.; Tian, H.; Qian, C.T. Facile Yb(OTf)3-promoted one-pot synthesis of polyhydroquinoline derivatives through Hantzsch reaction. Tetrahedron 2005, 61, 1539–1543. [Google Scholar] [CrossRef]
- Chari, M.A.; Syamasundar, K. Silica gel-NaHSO4-catalysed one-pot synthesis of Hantzsch 1,4-dihydropyridines at ambient temperature. Catal. Commun. 2005, 6, 624–626. [Google Scholar] [CrossRef]
- Sabitha, G.; Arundhathi, K.; Sudhakar, K.; Sastry, B.S.; Yadav, J.S. CeCl3·7H2O- catalyzed one-pot synthesis of Hantzsch 1, 4-dihydropyridines at room temperature. Synth. Commun. 2009, 39, 2843–2851. [Google Scholar] [CrossRef]
- Majid, M.M.; Mina, S.; Narges, K.; Masumeh, Z.; Yahya, S.; Abolghasem, D. Bronsted acid ionic liquid [(CH2)4SO3HMIM] [HSO4] as novel catalyst for one-pot synthesis of Hantzschpolyhydroquinoline derivatives. Synth. Commun. 2010, 40, 523–529. [Google Scholar]
- Heydari, A.; Khaksar, S.; Tajbakhsh, M.; Bijanzadeh, H.R. One-step synthesis of Hantzsch esters and polyhydroquinoline derivatives in fluoro alcohols. J. Flourine Chem. 2009, 130, 609–614. [Google Scholar] [CrossRef]
- Kumar, T.D.A.; Mohan, P.; Subrahmanyam, C.V.S.; Satyanarayana, K. Comparative study of catalytic potential of TBAB, BTEAC, and CTAB in one-pot synthesis of 1,4-dihydropyridines under aqueous medium. Synth. Commun. 2014, 44, 574–582. [Google Scholar] [CrossRef]
- Liu, S.; Li, W.; Pang, Y.; Xiao, H.; Zhou, Y.; Wang, X. Green synthesis of 2-Substituted imidazolines using hydrogen peroxide catalyzed by tungstophosphoric acid and tetrabutylammonium bromide in water. J. Heterocycl. Chem. 2019, 56, 998–1002. [Google Scholar] [CrossRef]
- Dake, S.A.; Kharat, K.R.; Yadav, A.R.; Kendrekar, P.S.; Pawar, R.P. In-vitro antiproliferative activity study of 2, 4, 5-triphenyl-1H-imidazole derivatives. J. Org. Inorg. Chem. 2017, 3, 5. [Google Scholar] [CrossRef]
- Zala, S.P.; Badmanaban, R.; Sen, D.J.; Patel, C.N. Synthesis and biological evaluation of 2, 4, 5-triphenyl-1H-imidazole-1-yl Derivatives. J. Appl. Pharm. Sci. 2012, 2, 202–208. [Google Scholar] [CrossRef]
- Hazelton, J.C.; Iddon, B.; Suschitzky, H.; Woolley, L.H. 2H-benzimidazoles (isobenzimidazoles). Part 10. Synthesis of polysubstitutedo-phenylenediamines and their conversion into heterocycles, particularly 2-substituted benzimidazoles with known or potential anthelminthic activity. Tetrahedron 1995, 51, 10771–10794. [Google Scholar] [CrossRef]
- Wolkenberg, S.E.; Wisnoski, D.D.; Leister, W.H.; Wang, Y.; Zhao, Z.; Lindsley, C.W. Efficient Synthesis of Imidazoles from Aldehydes and 1, 2-Diketones using Microwave Irradiation. Org. Lett. 2004, 6, 1453–1456. [Google Scholar] [CrossRef] [PubMed]
- Roongpisuthipong, A.; Chalermchockcharoenkit, A.; Sirimai, K.; Wanitpongpan, P.; Jaishuen, A.; Foongladda, S.; Kongkergkiat, N.; Prymanee, J. Safety and efficacy of a new imidazole fungicide, Sertaconazole, in the treatment of fungal vulvovaginitis: A comparative study using Fluconazole and Clotrimazole. Asian Biomed. 2010, 4, 443–448. [Google Scholar] [CrossRef][Green Version]
- Radziszewski, B. Ueber die Constitution des Lophins und verwandterVerbindungen. Ber. Der deutsch. Chem. Ges. 1882, 15, 1493–1496. [Google Scholar] [CrossRef]
- Wang, L.M.; Wang, Y.H.; Tian, H.; Yao, Y.F.; Shao, J.H.; Liu, B. Ytterbium triflate as an efficient catalyst for one-pot synthesis of substituted imidazoles through three-component condensation of benzil, aldehydes and ammonium acetate. J. Fluor. Chem. 2006, 127, 1570–1573. [Google Scholar] [CrossRef]
- Siddiqui, S.A.; Narkhede, U.C.; Palimkar, S.S.; Daniel, T.; Loholi, R.J.; Srinivasan, K.V. Room temperature ionic liquid promoted improved and rapid synthesis of 2, 4, 5-triaryl imidazoles from aryl aldehydes and 1, 2-diketones or α-hydroxyketone. Tetrahedron 2005, 61, 3539–3546. [Google Scholar] [CrossRef]
- Frantz, D.E.; Morency, L.; Soheilli, A.; Murry, J.A.; Grabowski, E.J.J.; Tillyer, R.D. Synthesis of Substituted Imidazoles via Organocatalysis. Org. Lett. 2004, 6, 843–846. [Google Scholar] [CrossRef]
- Balalaie, S.; Hashemi, M.M.; Akhbari, M. A novel one-pot synthesis of tetrasubstituted imidazoles under solvent-free conditions and microwave irradiation. Tetrahedron Lett. 2003, 44, 1709–1711. [Google Scholar] [CrossRef]
- Balalaie, S.; Arabanian, A. One-pot synthesis of tetrasubstituted imidazoles catalyzed by zeolite HY and silica gel under microwave irradiation. Green Chem. 2000, 2, 274–276. [Google Scholar] [CrossRef]
- Usyatinsky, A.Y.; Khmelnitsky, Y.L. Microwave-assisted synthesis of substituted imidazoles on a solid support under solvent-free conditions. Tetrahedron Lett. 2000, 41, 5031–5034. [Google Scholar] [CrossRef]
- Heravi, M.M.; Bakhtiari, K.; Oskooie, H.A.; Taheri, S. Synthesis of 2, 4, 5-triaryl-imidazoles catalyzed by NiCl2·6H2O under heterogeneous system. J. Mol. Catal. A Chem. 2007, 263, 279–281. [Google Scholar] [CrossRef]
- Chary, M.V.; Keerthysri, N.C.; Vupallapati, S.V.N.; Lingaiah, N.; Kantevari, S. Tetrabutylammonium bromide (TBAB) in isopropanol: An efficient, novel, neutral and recyclable catalytic system for the synthesis of 2, 4, 5-trisubstituted imidazoles. Catal. Commun. 2008, 9, 2013–2017. [Google Scholar] [CrossRef]
- Aghapoor, K.; Mohsenzadeh, F.; Sayahi, H.; Rastgar, S.; Darabi, H.R. Green synthesis of 1,3-dihydrobenzimidazol-2-ones fromaromatic diamines bymicrowave inatetrabutylammonium bromide-ethanol molten salt paste. Environ. Chem. Lett. 2018, 16, 1109–1116. [Google Scholar] [CrossRef]
- Khalafy, J.; Javahershenas, R. One-pot, three-component synthesis of pyrrolo[2,3-d]pyrimidine derivatives. J. Mex. Chem. Soc. 2018, 62, 62. [Google Scholar] [CrossRef]
- Mannschreck, A.; Koller, H.; Stuhler, G.; Davies, M.A.; Traber, J. The enantiomers of methaqualone and their unequal anticonvulsive activity. Eur. J. Med. Chem. 1984, 19, 381–383. [Google Scholar]
- Xia, Y.; Yang, Z.Y.; Hour, M.J.; Kuo, S.C.; Xia, P.; Bastow, K.F.; Nakanishi, Y.; Nampoothiri, P.; Hackl, T.; Harnel, E.; et al. Antitumor Agents. Part 204:1 Synthesis and Biological Evaluation of Substituted 2-Aryl Quinazolinones. Bioorg. Med. Chem. Lett. 2001, 11, 1193–1196. [Google Scholar] [CrossRef]
- Malamas, M.S.; Miller, J. Quinazoline acetic acids and related analogs as aldose reductase inhibitors. J. Med. Chem. 1991, 34, 1492–1503. [Google Scholar] [CrossRef]
- Hour, M.J.; Huang, L.J.; Kuo, S.C.; Xia, Y.; Bastow, K.; Nakanishi, Y.; Hamel, E.; Lee, K.H. 6-Alkylamino- and 2,3-Dihydro-3′-methoxy-2-phenyl-4-quinazolinones and related compounds: their synthesis, cytotoxicity, and inhibition of tubulin polymerization. J. Med. Chem. 2000, 43, 4479–4487. [Google Scholar] [CrossRef] [PubMed]
- Davoodnia, A.; Allameh, S.; Fakhari, A.R.; Tavakoli-Hoseini, N. Highly efficient solvent-free synthesis of quinazolin-4(3H)-ones and 2, 3-dihydroquinazolin-4(1H)-ones using tetrabutylammonium bromide as novel ionic liquid catalyst. Chin. Chem. Lett. 2010, 21, 550–553. [Google Scholar] [CrossRef]
- Schutz, H. Benzodiazepines; Springer: Heidelberg, Germany, 1982. [Google Scholar]
- Herbert, J.A.; Suschitzky, H. Syntheses of heterocyclic compounds. Part XXIX. Substituted 2, 3-dihydro-1H-1, 5-benzodiazepines. J. Chem. Soc. Perkin Trans. 1974, 1, 2657–2661. [Google Scholar] [CrossRef]
- Yadav, J.S.; Reddy, B.V.S.; Praveenkumar, S.; Nagaiah, K.; Lingaiah, N.; Saiprasad, P.S. Ag3PW12O40: A novel and recyclable heteropoly acid for the synthesis of 1, 5-benzodiazepines under solvent-free conditions. Synthesis 2004, 6, 901–904. [Google Scholar] [CrossRef]
- Jung, D.I.; Choi, T.W.; Kim, Y.Y.; Kim, I.S.; Park, Y.M.; Lee, Y.G.; Jung, D.H. Synthesis of 1, 5-benzodiazepine derivatives. Synth. Commun. 1999, 29, 1941–1951. [Google Scholar]
- Varala, R.; Ramu, E.; Adapa, S.R. Zinc montmorillonite as a reusable heterogeneous catalyst for the synthesis of 2,3-dihydro-1H-1,5-benzodiazepine derivatives. Arkivoc 2006, 13, 171–177. [Google Scholar]
- Curini, M.; Epifano, F.; Marcotullio, M.C.; Rosati, O. Ytterbium triflate promoted synthesis of 1,5-benzodiazepine derivatives. Tetrahedron Lett. 2001, 42, 3193–3195. [Google Scholar] [CrossRef]
- Balakrishna, M.S.; Kaboudin, B. A simple and new method for the synthesis of 1,5-benzodiazepine derivatives on a solid surface. Tetrahedron Lett. 2001, 42, 1127–1129. [Google Scholar] [CrossRef]
- Yadav, J.S.; Reddy, B.V.S.; Eshwaraian, B.; Anuradha, K. Amberlyst-15®: A novel and recyclable reagent for the synthesis of 1, 5-benzodiazepines in ionic liquids. Green Chem. 2002, 4, 592–594. [Google Scholar] [CrossRef]
- Reddy, B.M.; Sreekanth, P.M. An efficient synthesis of 1,5-benzodiazepine derivatives catalyzed by a solid superacid sulfated zirconia. Tetrahedron Lett. 2003, 44, 4447–4449. [Google Scholar] [CrossRef]
- Baseer, M.A.; Khan, A.J. An Efficient one-pot synthesis of 1,5-Benzodiazepine derivatives catalyzed by TBAB under Mild Conditions. J. Chem. 2012, 9, 407–414. [Google Scholar]
- Xie, A.; Cao, M.; Feng, L.; Dong, W. The synthesis of 5-substituted 1H-tetrazoles in molten tetrabutylammonium bromide. J. Chem. Res. 2013, 37, 665–667. [Google Scholar] [CrossRef]
- Koussini, R.; Al-Shihri, A.S. Microwave-assisted synthesis of 3-nitro-2H-chromenes under solvent-less phase-transfer catalytic conditions. Jord. J. Chem. 2008, 3, 103–107. [Google Scholar]
- Brahmachari, G.; Banerjee, B. Facile and one-pot access to diverse and densely functionalized 2-amino-3-cyano-4H-pyrans and pyran-annulated heterocyclic scaffolds via an eco-friendly multicomponent reaction at room temperature using urea as a novel organo-catalyst. ACS Sustain. Chem. Eng. 2014, 2, 411–422. [Google Scholar] [CrossRef]
- Brahmachari, G.; Banerjee, B. Facile and chemically sustainable one-pot synthesis of a wide array of fused O- and N-heterocycles catalyzed by trisodium citrate dihydrate under ambient conditions. Asian J. Org. Chem. 2016, 5, 271–286. [Google Scholar] [CrossRef]
- Brahmachari, G.; Laskar, S.; Banerjee, B. Eco-friendly, one-pot multicomponent synthesis of pyran annulated heterocyclic scaffolds at room temperature using ammonium or sodium formate as non-toxic catalyst. J. Heterocycl. Chem. 2014, 51, E303–E308. [Google Scholar] [CrossRef]
- Khurana, J.M.; Kumar, S. Tetrabutylammonium bromide (TBAB): A neutral and efficient catalyst for the synthesis of biscoumarin and 3, 4-dihydropyrano[c]chromene derivatives in water and solvent-free conditions. Tetrahedron Lett. 2009, 50, 4125–4127. [Google Scholar] [CrossRef]
- Biswas, T.; Mahalingam, V. g-C3N4 and Tetrabutylammonium bromide catalyzed efficient conversion of epoxide to cyclic carbonate under ambient condition. New J. Chem. 2017, 41, 14839–14842. [Google Scholar] [CrossRef]
- Wang, Y.-H.; Ouyang, B.; Qiu, G.; Zhou, H.; Liu, J.-B. Oxidative oxy-cyclization of 2-alkynylbenzamide enabled by TBAB/Oxone: Switchable synthesis of isocoumarin-1-imines and isobenzofuran-1-imine. Org. Biomol. Chem. 2019, 17, 4335–4341. [Google Scholar] [CrossRef]
- Andreani, L.L.; Lapi, E. Aspects and orientations of modern pharmacognosy. Boll. Chim. Farm. 1960, 99, 583–586. [Google Scholar]
- Singh, K.; Singh, J.; Singh, H. A synthetic entry into fused pyran derivatives through carbon transfer reactions of 1, 3-oxazinanes and oxazolidines with carbon nucleophiles. Tetrahedron 1996, 52, 14273–15280. [Google Scholar] [CrossRef]
- Bonsignore, L.; Loy, G.; Secci, D.; Calignano, A. Synthesis and pharmacological activity of 2-oxo-(2H) 1-benzopyran-3-carboxamide derivatives. Eur. J. Med. Chem. 1983, 28, 517–520. [Google Scholar] [CrossRef]
- Mobinikhaledi, A.; Fard, M.A.B. Tetrabutylammonium bromide in water as a green media for the synthesis of pyrano[2, 3-d]pyrimidinone and tetrahydrobenzo[b]pyran derivatives. Acta Chim. Slov. 2010, 57, 931–935. [Google Scholar] [PubMed]
- Rao, H.; Ma, X.; Liu, Q.; Li, Z.; Cao, S.; Li, C.-J. Metal-free oxidative coupling: Xanthone formation via direct annulation of 2-aryloxybenzaldehyde using tetrabutylammonium bromide as a psromoter in aqueous medium. Adv. Synth. Catal. 2013, 355, 2191–2196. [Google Scholar] [CrossRef]
- Khosropour, A.R.; Khodaei, M.M.; Moghannian, H. A facile, simple and convenient method for the synthesis of 14-Alkyl or aryl-14H-dibenzo[a,j]xanthenes catalyzed by p-TSA in solution and solvent-free conditions. Synlett 2005, 2005, 955–958. [Google Scholar] [CrossRef]
- Ko, S.; Yao, C.-F. Heterogeneous catalyst: Amberlyst-15 catalyzes the synthesis of 14-substituted-14H-dibenzo[a,j]xanthenes under solvent-free conditions. Tetrahedron Lett. 2006, 47, 8827–8829. [Google Scholar] [CrossRef]
- Pasha, M.A.; Jayashankara, V.P. Molecular iodine catalyzed synthesis of aryl-14H-dibenzo[a,j]xanthenes under solvent-free condition. Bioorg. Med. Chem. Lett. 2007, 17, 621. [Google Scholar] [CrossRef]
- Das, B.; Ravikanth, B.; Ramu, R.; Laminarayana, K.; Rao, K.B.V. Iodine catalyzed simple and efficient synthesis of 14-aryl or alkyl-14-H-dibenzo[a,j]xanthenes. J. Mol. Catal. A Chem. 2006, 255, 74–77. [Google Scholar] [CrossRef]
- Kantevari, S.; Chary, M.V.; Rudra Das, A.P.; Vuppalapati, S.V.N.; Lingaiah, N. Catalysis by an ionic liquid: Highly efficient solvent-free synthesis of aryl-14H-dibenzo[a.j]xanthenes by molten tetrabutylammonium bromide under conventional and microwave heating. Catal. Commun. 2008, 9, 1575–1578. [Google Scholar] [CrossRef]
- Banerjee, B.; Brahmachari, G. Ammonium chloride catalysed one-pot multicomponent synthesis of 1, 8-dioxo-octahydroxanthenes and N-aryl-1, 8-dioxodecahydroacridines under solvent free conditions. J. Chem. Res. 2014, 38, 745–750. [Google Scholar] [CrossRef]
- Ezabadi, A.; Nazarian, R.; Gholami, M. Tetrabutylammonium bromide: An efficient catalyst for the synthesis of xanthenediones under solvent-free conditions. Orient. J. Chem. 2015, 31, 1559–1563. [Google Scholar] [CrossRef]
- Tjosaas, F.; Kjerstad, I.B.; Fiksdahl, A. Syntheses of oxazolo[4,5-c]pyridine and 6-azaindole. J. Heterocycl. Chem. 2008, 45, 559–562. [Google Scholar] [CrossRef]
- Mobinikhaledi, A.; Foroughifar, N.; Fard, M.A.B. Simple and efficient method for three-component synthesis of spirooxindoles in aqueous and solvent-free media. Synth. Commun. 2011, 41, 441–450. [Google Scholar] [CrossRef]
- Mishra, K.; Singh, J.B.; Gupta, T.; Singh, R.M. TBAB-catalyzed cascade reactions: Facile synthesis of 1-trifluoromethyl-3-alkylidene1, 3-dihydrofuro[3,4-b]quinolines via 5-exo-dig cyclization of o-arylalkynylquinoline aldehydes. Org. Chem. Front. 2017, 4, 1926–1930. [Google Scholar] [CrossRef]
- Bankar, V.V.; Dhankar, R.P. A practical green synthesis of thiazine derivatives using phase transfer catalyst. Rasayan J. Chem. 2018, 11, 1294–1299. [Google Scholar] [CrossRef]
- Saroha, M.; Khanna, G.; Khurana, J.M. Green synthesis of novel naphtho[1,2-e]/benzo[e][1,3] thiazine derivatives via one-pot three-component reaction using tetra n-butyl ammonium bromide. ChemistrySelect 2018, 3, 12560–12562. [Google Scholar] [CrossRef]
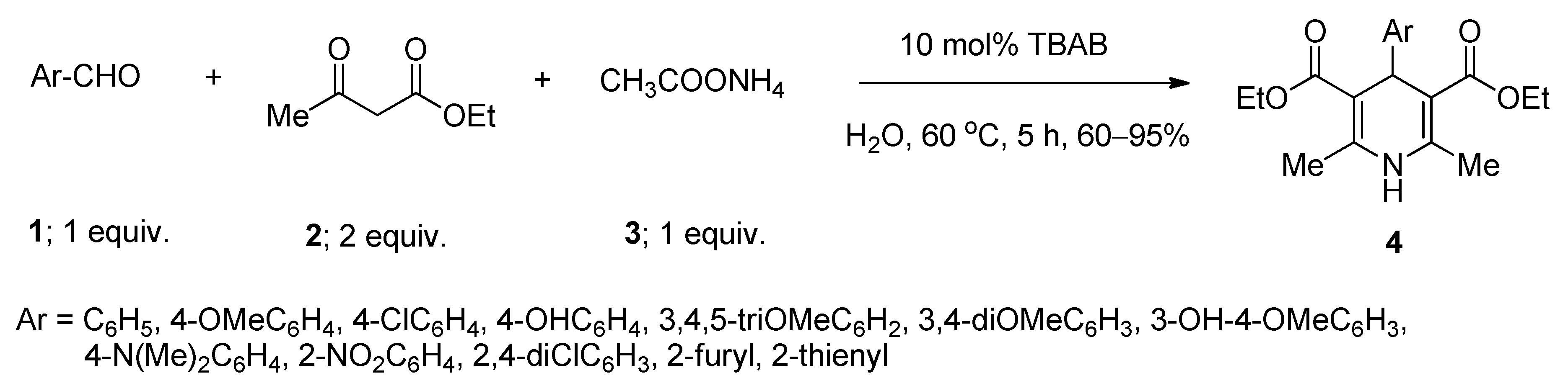
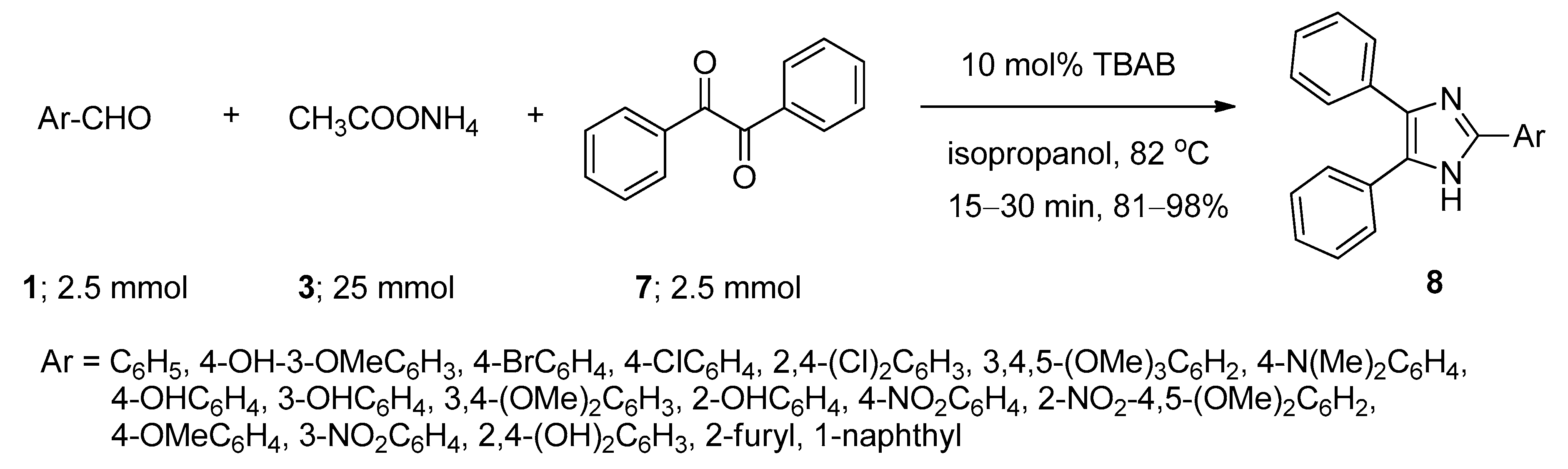

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Banik, B.K.; Banerjee, B.; Kaur, G.; Saroch, S.; Kumar, R. Tetrabutylammonium Bromide (TBAB) Catalyzed Synthesis of Bioactive Heterocycles. Molecules 2020, 25, 5918. https://doi.org/10.3390/molecules25245918
Banik BK, Banerjee B, Kaur G, Saroch S, Kumar R. Tetrabutylammonium Bromide (TBAB) Catalyzed Synthesis of Bioactive Heterocycles. Molecules. 2020; 25(24):5918. https://doi.org/10.3390/molecules25245918
Chicago/Turabian StyleBanik, Bimal Krishna, Bubun Banerjee, Gurpreet Kaur, Shivam Saroch, and Rajat Kumar. 2020. "Tetrabutylammonium Bromide (TBAB) Catalyzed Synthesis of Bioactive Heterocycles" Molecules 25, no. 24: 5918. https://doi.org/10.3390/molecules25245918
APA StyleBanik, B. K., Banerjee, B., Kaur, G., Saroch, S., & Kumar, R. (2020). Tetrabutylammonium Bromide (TBAB) Catalyzed Synthesis of Bioactive Heterocycles. Molecules, 25(24), 5918. https://doi.org/10.3390/molecules25245918