Effect of Cation Structure in Quinolinium-Based Ionic Liquids on the Solubility in Aromatic Sulfur Compounds or Heptane: Thermodynamic Study on Phase Diagrams
Abstract
:1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Materials
3.2. Karl–Fischer Analysis
3.3. Phase Equilibria Apparatus and Measurements
3.4. Correlation with NRTL Equation
4. Conclusions and Future Perspectives
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Carnahan, N.F.; Salager, J.L.; Antón, R.; Dávila, A. Properties of resins extracted from Boscan crude oil and their effect on the stability of asphaltenes in Boscan and Hamaca crude oils. Energy Fuels 1999, 13, 309–314. [Google Scholar] [CrossRef]
- Kulkarni, P.S.; Afonso, C.A.M. Deep desulfurization of diesel fuel using ionic liquids: Current status and future challenges. Green Chem. 2010, 12, 1139–1149. [Google Scholar] [CrossRef]
- Abro, R.; Abdeltawab, A.A.; Al-Deyab, S.S.; Yu, G.; Qazi, A.B.; Gao, S.; Chen, X. A review of extractive desulfurization of fuel oils using ionic liquids. RSC Adv. 2014, 4, 35302–35317. [Google Scholar] [CrossRef]
- Kurnia, K.A.; Pinho, S.P.; Coutinho, J.A.P. Designing ionic liquids for absorptive cooling. Green Chem. 2014, 16, 3741–3745. [Google Scholar] [CrossRef]
- Larriba, M.; Navarro, P.; Gonzalez-Miquel, M.; Omar, S.; Palomar, J.; Garcia, J.; Rodríguez, F. Dicyanamide-based ionic liquids in the liquid–liquid extraction of aromatics from alkanes: Experimental evaluation and computational predictions. Chem. Eng. Res. Des. 2016, 109, 561–572. [Google Scholar] [CrossRef]
- Karadas, F.; Atilhan, M.; Aparicio, S. Review on the use of Ionic Liquids (ILs) as alternative fluids for CO2 capture and natural gas sweetening. Energy Fuels 2010, 24, 5817–5828. [Google Scholar] [CrossRef]
- Paduszyński, K.; Królikowski, M.; Zawadzki, M.; Orzeł, P. Computer-aided molecular design of new task-specific ionic liquids for extractive desulfurization of gasoline. ACS Sustain. Chem. Eng. 2017, 5, 9032–9042. [Google Scholar] [CrossRef]
- Królikowski, M.; Królikowska, M.; Więckowski, M.; Piłowski, A. The influence of the ionic liquids functionalization on interaction in binary systems with organic solutes and water-thermodynamic data of activity coefficients at infinite dilution. J. Chem. Thermodyn. 2020, 147, 106117. [Google Scholar] [CrossRef]
- Sobota, M.; Dohnal, V.; Vrbka, P. Activity Coefficients at Infinite Dilution of Organic Solutes in the Ionic Liquid 1-Ethyl-3-methyl-imidazolium Nitrate. J. Phys. Chem. B 2009, 113, 4323–4332. [Google Scholar] [CrossRef]
- Martins, M.A.; Coutinho, J.A.P.; Pinho, S.P.; Domańska, U. Measurements of Activity Coefficients at Infinite Dilutionof Organic Solutes and Water on Polar Imidazolium-Based Ionic Liquids. J. Chem. Thermodyn. 2015, 91, 194–203. [Google Scholar] [CrossRef]
- Paduszyński, K. In Silico Calculation of Infinite Dilution Activity Coefficients of Molecular Solutes in Ionic Liquids: Critical Review of Current Methods and New Models Based on Three Machine Learning Algorithms. J. Chem. Inf. Model. 2016, 56, 1420–1437. [Google Scholar] [CrossRef]
- Królikowski, M.; Więckowski, M.; Zawadzki, M. Separation of organosulfur compounds from heptane by liquid–liquid extraction with tricyanomethanide based ionic liquids. Experimental data and NRTL correlation. J. Chem. Thermodyn. 2020, 149, 106149. [Google Scholar] [CrossRef]
- Paduszyński, K.; Królikowska, M. Extensive Evaluation of Performance of the COSMO-RS Approach in Capturing Liquid-Liquid Equilibria of Binary Mixtures of Ionic Liquids with Molecular Compounds. Ind. Eng. Chem. Res. 2020, 59, 11851–11863. [Google Scholar] [CrossRef]
- Domańska, U.; Roguszewska, M.; Królikowski, M.; Ramjugernath, D.; Naidoo, P. Phase equilibria study of binary systems comprising an (ionic liquid + hydrocarbon). J. Chem. Thermodyn. 2015, 83, 90–96. [Google Scholar] [CrossRef]
- Marciniak, A.; Wlazło, M. Ternary (liquid + liquid) equilibria of {trifluorotris (perfluo-roethyl) phosphate based ionic liquids + thiophene + heptane}: Part 2. J. Chem. Thermodyn. 2015, 86, 196–201. [Google Scholar] [CrossRef]
- Rodríguez, H.; Francisco, M.; Soto, A.; Arce, A. Liquid–liquid equilibrium and interfacial tension of the ternary system heptane + thiophene + 1-ethyl-3-methylimidazolium bis (trifluoro-methanesulfonyl) imide. Fluid Phase Equilib. 2010, 298, 240–245. [Google Scholar] [CrossRef]
- Marciniak, A.; Królikowski, M. Ternary liquid-liquid equilibria of bis(trifluoromethyl-sulfonyl)-amide based ionic liquids + thiophene + n-heptane. The influence of cation structure. Fluid Phase Equilib. 2012, 321, 59–63. [Google Scholar] [CrossRef]
- Domańska, U.; Zawadzki, M.; Tshibangu, M.; Ramjugernath, D.; Letcher, T.M. Phase equilibria study of {N-butylquinolinium bis{(trifluoromethyl)sulfonyl}imide + aromatic hydrocarbons, or an alcohol} binary systems. J. Chem. Thermodyn. 2010, 42, 1180–1186. [Google Scholar] [CrossRef]
- Domańska, U.; Zawadzki, M. Thermodynamic properties of the N-butylisoquinolinium bis(trifluoromethylsulfonyl)imide. J. Chem. Thermodyn. 2011, 43, 989–995. [Google Scholar] [CrossRef]
- Domańska, U.; Zawadzki, M.; Zwolińska, M. Thermophysical properties and phase equilibria study of the binary systems {N-hexylquinolinium bis(trifluoromethylsulfonyl)imide + aromatic hydrocarbons, or an alcohol}. J. Chem. Thermodyn. 2011, 43, 775–781. [Google Scholar] [CrossRef]
- Domańska, U.; Zawadzki, M.; Tshibangu, M.; Ramjugernath, D.; Letcher, T.M. Phase equilibria study of {N-hexylisoquinolinium bis{(trifluoromethyl)sulfonyl}imide + aromatic hydrocarbons or an alcohol} binary systems. J. Phys. Chem. B 2011, 115, 4003–4010. [Google Scholar] [CrossRef]
- Zawadzki, M.; Domańska, U. Thermodynamic properties of the N-octylquinolinium bis{(trifluoromethyl)sulfonyl}imide. J. Chem. Thermodyn. 2012, 48, 276–283. [Google Scholar] [CrossRef]
- Domańska, U.; Zawadzki, M.; Królikowski, M.; Lewandrowska, A. Phase equilibria study of binary and ternary mixtures of {N-octylisoquinolinium bis{(trifluoromethyl)sulfonyl}-imide + hydrocarbon, or an alcohol, or water}. Chem. Eng. J. 2012, 181–182, 63–71. [Google Scholar]
- Holbrey, J.D.; López-Martin, I.; Rothenberg, G.; Seddon, K.S.; Silvero, G.; Zheng, X. Desulfurisation of oils using ionic liquids: Selection of cationic and anionic components to enhance extraction efficiency. Green Chem. 2008, 10, 87–92. [Google Scholar] [CrossRef]
- Lei, Z.; Arlt, W.; Wasserscheid, P. Selection of entrainers in the 1-hexene/n-hexane system with a limited solubility. Fluid Phase Equilib. 2007, 260, 29–35. [Google Scholar] [CrossRef]
- Prausnitz, J.M.; Lichtenthaler, R.M.; Gomez de Azevedo, E. Molecular Thermodynamics of Fluid Phase Equilibria, 3rd ed.; Prentice Hall: Upper Saddle River, NJ, USA, 1999. [Google Scholar]
Sample Availability: Samples of the compounds [BQuin][NTf2], [HQuin][NTf2] and [OQuin][NTf2]) are available from the authors. |
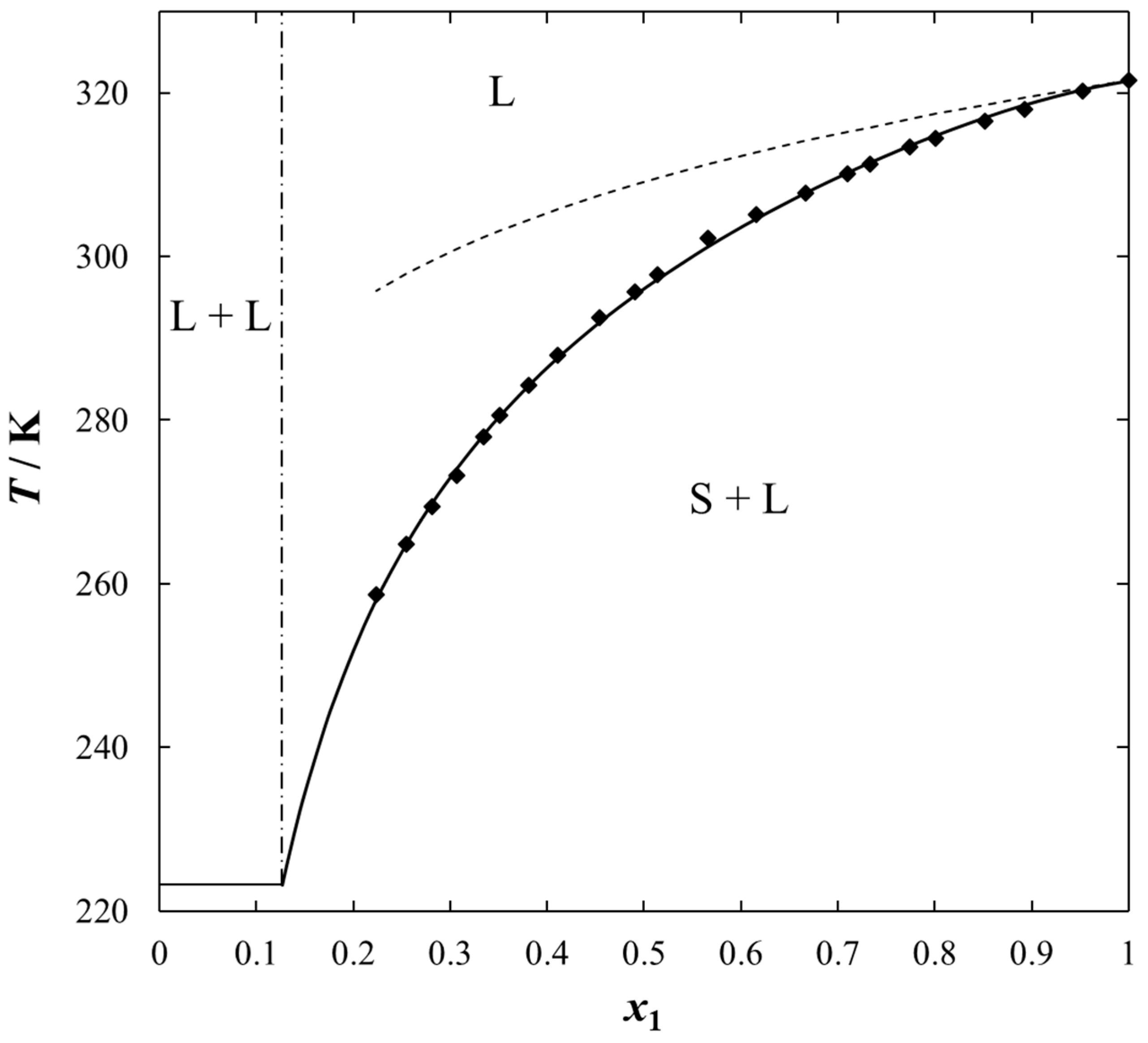

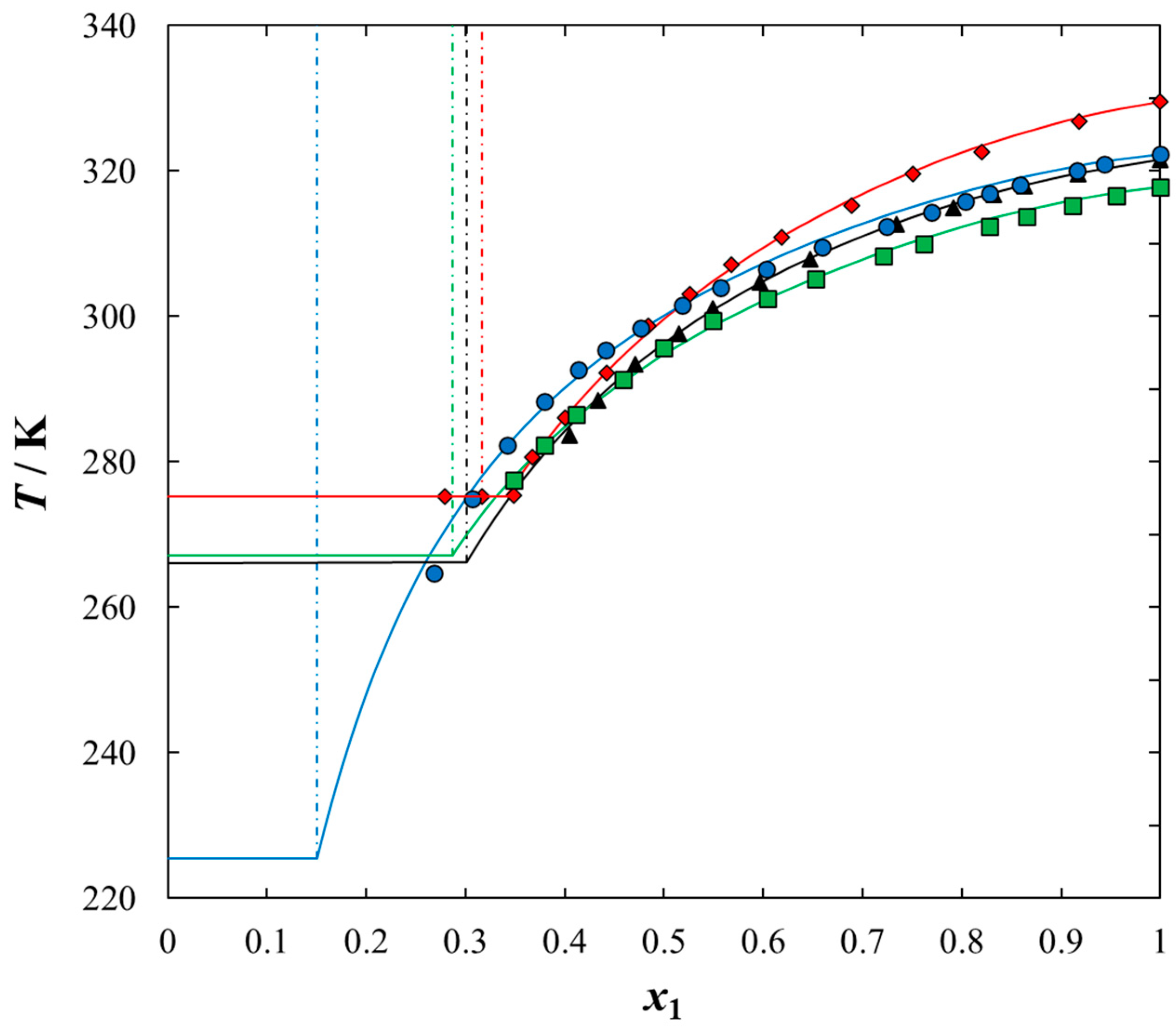
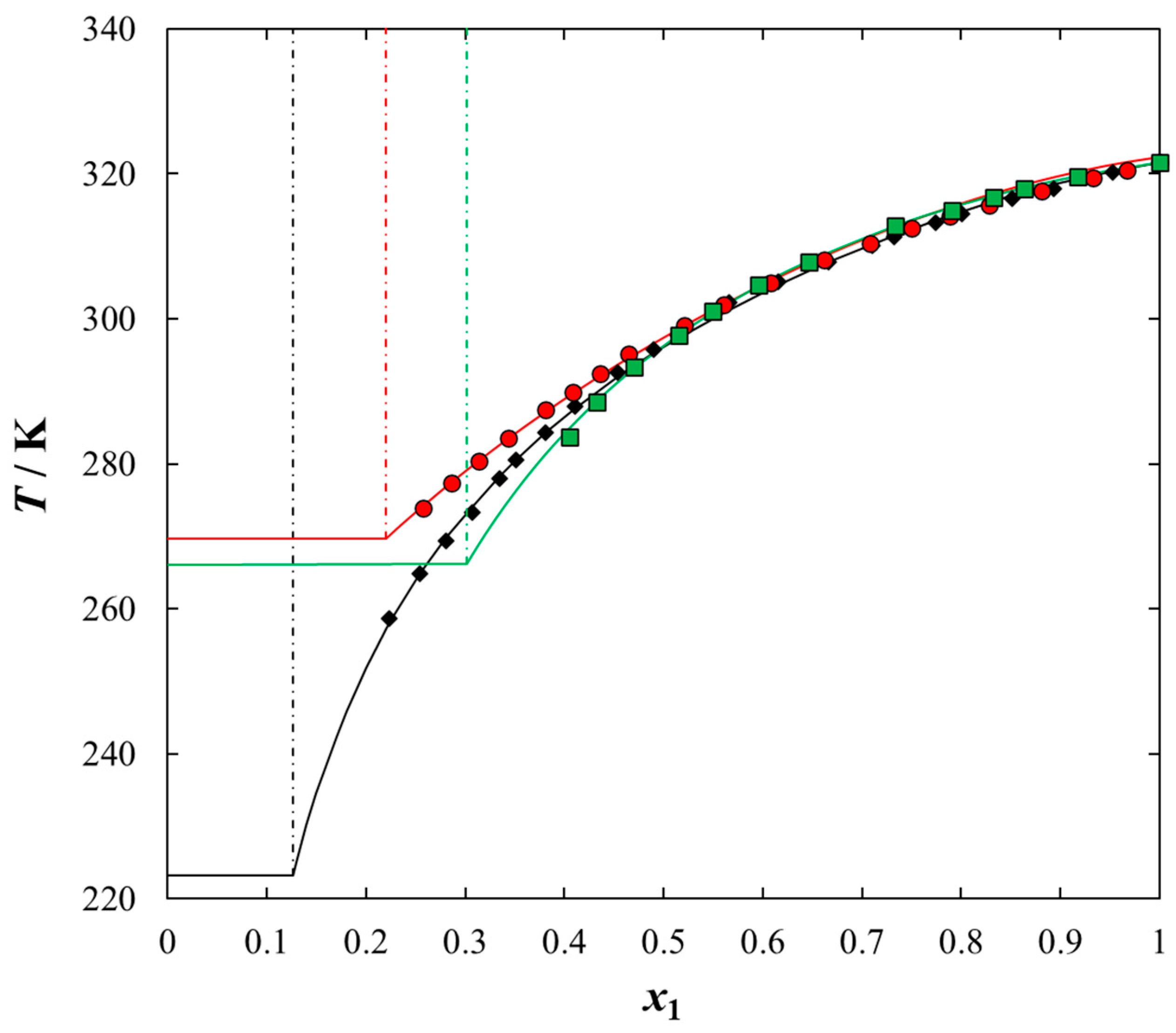

| x1 | TSLE/K | γ1 | x1 | TSLE/K | γ1 |
|---|---|---|---|---|---|
| [BiQuin][NTf2] (1) + thiophene (2) | |||||
| 1.0000 | 321.5 | 1.00 | 0.5139 | 297.8 | 0.48 |
| 0.9527 | 320.2 | 0.99 | 0.4904 | 295.7 | 0.44 |
| 0.8930 | 318.0 | 0.95 | 0.4541 | 292.5 | 0.38 |
| 0.8515 | 316.5 | 0.92 | 0.4106 | 287.9 | 0.32 |
| 0.8008 | 314.5 | 0.87 | 0.3808 | 284.2 | 0.27 |
| 0.7746 | 313.3 | 0.83 | 0.3514 | 280.5 | 0.23 |
| 0.7328 | 311.3 | 0.78 | 0.3347 | 277.9 | 0.20 |
| 0.7105 | 310.1 | 0.75 | 0.3066 | 273.3 | 0.16 |
| 0.6664 | 307.8 | 0.69 | 0.2808 | 269.4 | 0.13 |
| 0.6153 | 305.1 | 0.62 | 0.2543 | 264.8 | 0.10 |
| 0.5665 | 302.2 | 0.55 | 0.2231 | 258.7 | 0.06 |
| [BQuin][NTf2] (1) + thiophene (2) | |||||
| 1.0000 | 329.6 | 1.00 | 0.6235 | 311.4 | 0.62 |
| 0.9807 | 329.2 | 1.00 | 0.5497 | 305.8 | 0.51 |
| 0.9664 | 328.8 | 0.99 | 0.4948 | 300.6 | 0.42 |
| 0.9476 | 328.3 | 0.99 | 0.4499 | 296.3 | 0.35 |
| 0.9106 | 327.0 | 0.97 | 0.4094 | 291.0 | 0.29 |
| 0.8736 | 325.6 | 0.94 | 0.3621 | 284.5 | 0.22 |
| 0.8415 | 324.2 | 0.91 | 0.3279 | 278.8 | 0.16 |
| 0.8216 | 323.1 | 0.89 | 0.3080 | 275.2 | 0.14 |
| 0.7919 | 321.5 | 0.85 | 0.2542 | 264.5 | 0.07 |
| 0.7309 | 317.9 | 0.78 | 0.2226 | 255.2 | 0.04 |
| 0.6972 | 316.0 | 0.73 | |||
| [HQuin][NTf2] (1) + thiophene (2) | |||||
| 1.0000 | 317.9 | 1.00 | 0.5226 | 299.0 | 0.42 |
| 0.9431 | 316.8 | 0.98 | 0.4878 | 296.5 | 0.36 |
| 0.8968 | 315.5 | 0.95 | 0.4625 | 294.8 | 0.32 |
| 0.8472 | 313.7 | 0.90 | 0.4144 | 290.7 | 0.25 |
| 0.8091 | 312.4 | 0.85 | 0.3633 | 285.6 | 0.18 |
| 0.7695 | 311.1 | 0.80 | 0.3367 | 282.2 | 0.15 |
| 0.7284 | 309.4 | 0.74 | 0.2917 | 276.1 | 0.09 |
| 0.6662 | 306.8 | 0.64 | 0.2638 | 271.5 | 0.07 |
| 0.6292 | 305.2 | 0.58 | 0.2398 | 267.0 | 0.05 |
| 0.5830 | 302.7 | 0.51 | 0.2146 | 262.5 | 0.03 |
| 0.5523 | 300.8 | 0.46 | 0.1860 | 256.3 | 0.02 |
| [OQuin][NTf2] (1) + thiophene (2) | |||||
| 1.0000 | 322.3 | 1.00 | 0.4361 | 297.1 | 0.29 |
| 0.9436 | 321.4 | 0.98 | 0.4093 | 294.6 | 0.25 |
| 0.8762 | 319.3 | 0.93 | 0.3762 | 291.0 | 0.20 |
| 0.7590 | 314.7 | 0.79 | 0.3319 | 285.0 | 0.14 |
| 0.6824 | 311.3 | 0.68 | 0.3028 | 280.7 | 0.10 |
| 0.6244 | 308.4 | 0.59 | 0.2694 | 275.0 | 0.07 |
| 0.5934 | 306.8 | 0.54 | 0.2345 | 268.5 | 0.04 |
| 0.5319 | 303.5 | 0.44 | 0.2093 | 263.0 | 0.02 |
| 0.5061 | 301.9 | 0.40 | 0.1861 | 256.3 | 0.01 |
| 0.4734 | 300.0 | 0.35 | |||
| x1 | TSLE/K | γ1 | x1 | TSLE/K | γ1 |
|---|---|---|---|---|---|
| [BiQuin][NTf2] (1) + benzothiophene (2) | |||||
| 1.0000 | 321.5 | 1.00 | 0.5960 | 304.7 | 0.64 |
| 0.9175 | 319.6 | 0.98 | 0.5495 | 301.1 | 0.56 |
| 0.8633 | 317.9 | 0.96 | 0.5153 | 297.7 | 0.49 |
| 0.8325 | 316.8 | 0.94 | 0.4704 | 293.4 | 0.40 |
| 0.7910 | 315.0 | 0.90 | 0.4332 | 288.6 | 0.33 |
| 0.7337 | 312.8 | 0.84 | 0.4052 | 283.6 | 0.27 |
| 0.6469 | 307.9 | 0.72 | |||
| [BQuin][NTf2] (1) + benzothiophene (2) | |||||
| 1.0000 | 329.6 | 1.00 | 0.4839 | 298.7 | 0.37 |
| 0.9185 | 326.8 | 0.98 | 0.4419 | 292.3 | 0.29 |
| 0.8200 | 322.7 | 0.90 | 0.4002 | 286.0 | 0.22 |
| 0.7509 | 319.6 | 0.82 | 0.3675 | 280.7 | 0.16 |
| 0.6885 | 315.3 | 0.73 | 0.3481 | 275.4 | 0.13 |
| 0.6181 | 310.9 | 0.62 | 0.3166 | 275.2 | |
| 0.5674 | 307.1 | 0.53 | 0.2786 | 275.2 | |
| 0.5261 | 303.0 | 0.45 | |||
| [HQuin][NTf2] (1) + benzothiophene (2) | |||||
| 1.0000 | 317.9 | 1.00 | 0.6037 | 302.4 | 0.48 |
| 0.9559 | 316.7 | 0.99 | 0.5493 | 299.5 | 0.39 |
| 0.9112 | 315.3 | 0.96 | 0.4990 | 295.8 | 0.30 |
| 0.8650 | 313.7 | 0.91 | 0.4586 | 291.4 | 0.24 |
| 0.8277 | 312.4 | 0.86 | 0.4112 | 286.5 | 0.17 |
| 0.7619 | 310.0 | 0.76 | 0.3797 | 282.4 | 0.13 |
| 0.7205 | 308.3 | 0.69 | 0.3481 | 277.5 | 0.09 |
| 0.6521 | 305.1 | 0.57 | |||
| [OQuin][NTf2] (1) + benzothiophene (2) | |||||
| 1.0000 | 322.3 | 1.00 | 0.5573 | 304.0 | 0.45 |
| 0.9439 | 320.9 | 0.99 | 0.5180 | 301.6 | 0.38 |
| 0.9160 | 320.0 | 0.97 | 0.4763 | 298.4 | 0.31 |
| 0.8591 | 318.1 | 0.92 | 0.4415 | 295.4 | 0.25 |
| 0.8276 | 316.9 | 0.88 | 0.4137 | 292.7 | 0.21 |
| 0.8039 | 315.8 | 0.85 | 0.3790 | 288.3 | 0.15 |
| 0.7693 | 314.3 | 0.80 | 0.3418 | 282.3 | 0.10 |
| 0.7239 | 312.4 | 0.74 | 0.3069 | 275.0 | 0.07 |
| 0.6591 | 309.5 | 0.63 | 0.2683 | 264.8 | 0.03 |
| 0.6028 | 306.5 | 0.53 | |||
| x1 | TSLE/K | γ1 | x1 | TSLE/K | γ1 |
|---|---|---|---|---|---|
| [BiQuin][NTf2] (1) + 2-methylthiophene (2) | |||||
| 1.0000 | 321.5 | 1.00 | 0.5601 | 302.0 | 0.56 |
| 0.9665 | 320.5 | 1.00 | 0.5204 | 299.1 | 0.50 |
| 0.9334 | 319.4 | 0.99 | 0.4646 | 295.1 | 0.43 |
| 0.8815 | 317.7 | 0.96 | 0.4359 | 292.5 | 0.39 |
| 0.8280 | 315.6 | 0.91 | 0.4083 | 290.0 | 0.35 |
| 0.7882 | 314.1 | 0.87 | 0.3813 | 287.4 | 0.32 |
| 0.7505 | 312.5 | 0.82 | 0.3434 | 283.6 | 0.28 |
| 0.7082 | 310.5 | 0.77 | 0.3135 | 280.4 | 0.24 |
| 0.6620 | 308.2 | 0.71 | 0.2865 | 277.4 | 0.22 |
| 0.6081 | 305.0 | 0.63 | 0.2576 | 274.0 | 0.19 |
| x1 | TSLE/K | x1 | TSLE/K | TLLE/K |
|---|---|---|---|---|
| [BiQuin][NTf2] (1) + heptane (2) | ||||
| 1.0000 | 321.51 | 0.9684 | 321.49 | 324.54 |
| 0.9833 | 321.49 | 0.9640 | 321.49 | 337.15 |
| 0.9490 | 321.49 | 361.29 | ||
| [BQuin][NTf2] (1) + heptane (2) | ||||
| 1.000 | 329.6 | 0.967 | 328.4 | 333.3 |
| 0.988 | 329.0 | 0.956 | 328.4 | 355.6 |
| 0.975 | 328.4 | 0.967 | 328.4 | 333.3 |
| [HQuin][NTf2] (1) + heptane (2) | ||||
| 1.000 | 317.9 | 0.958 | 314.0 | 321.3 |
| 0.985 | 316.8 | 0.946 | 314.0 | 348.0 |
| 0.971 | 315.3 | |||
| [OQuin][NTf2] (1) + heptane (2) | ||||
| 1.000 | 322.3 | 0.942 | 318.0 | 326.0 |
| 0.982 | 321.4 | 0.923 | 318.0 | 360.1 |
| 0.961 | 320.5 | |||
| System | NRTL Parameters | RMSD | ||
|---|---|---|---|---|
| [BiQuin][NTf2] (1) + thiophene (2) | −4591.78 | −2579.41 | 0.78 | 0.64 |
| [BQuin][NTf2] (1) + thiophene (2) | −5915.62 | −2776.95 | 0.71 | 0.45 |
| [HQuin][NTf2] (1) + thiophene (2) | −5797.59 | −3159.81 | 0.62 | 0.56 |
| [OQuin][NTf2] (1) + thiophene (2) | −6318.13 | −3125.06 | 0.62 | 0.69 |
| [BiQuin][NTf2] (1) + benzothiophene (2) | −8888.07 | −2032.34 | 0.46 | 0.55 |
| [BQuin][NTf2] (1) + thiophene (2) | −11,775.80 | −2937.43 | 0.45 | 0.78 |
| [HQuin][NTf2] (1) + thiophene (2) | −11,068.53 | −3916.45 | 0.45 | 0.80 |
| [OQuin][NTf2] (1) + thiophene (2) | −12,273.10 | −3527.76 | 0.45 | 1.05 |
| [BiQuin][NTf2] (1) + 2-methylthiophene (2) | −2000.44 | −4000.62 | 0.20 | 0.85 |
| Structure | Name, Abbreviation, CAS No. |
|---|---|
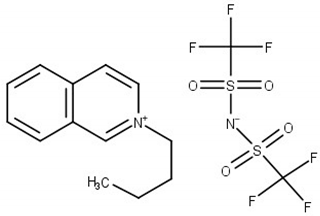 | N-butylisoquinolinium bis(trifluoromethylsulfonyl)imide, [BiQuin][NTf2] M = 466.42 g·mol−1 CAS No. 957763-47-2 |
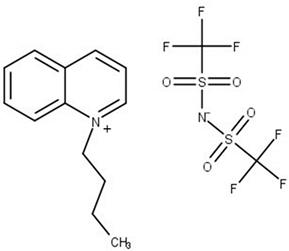 | N-butylquinolinium bis(trifluoromethylsulfonyl)imide, [BQuin][NTf2] M = 466.42 g·mol−1 CAS No. 1289382-58-6 |
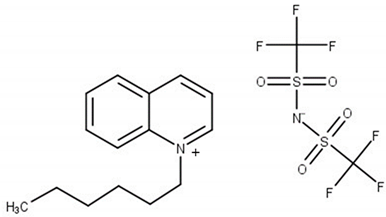 | N-hexylquinolinium bis(trifluoromethylsulfonyl)imide, [HQuin][NTf2] M = 494.47 g·mol−1 CAS No. 1263302-30-2 |
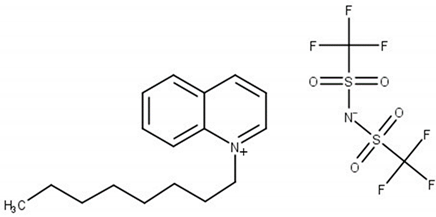 | N-octylquinolinium bis(trifluoromethylsulfonyl)imide, [OQuin][NTf2] M = 522.52 g·mol−1 CAS No. 868671-34-5 |
| Sample | Source | Initial Mole Fraction Purity (wt%) | Purification Method | Final Mole Fraction Purity (wt%) | Analysis Method | Water Content/ppm |
|---|---|---|---|---|---|---|
| [BiQuin][NTf2] | own synthesis | - | crystallization, vacuum heating | 97.0 | Karl–Fischer, 1H-NMR | 290 |
| [BQuin][NTf2] | own synthesis | - | crystallization, vacuum heating | 97.0 | Karl–Fischer, 1H-NMR | 280 |
| [HQuin][NTf2] | own synthesis | - | crystallization, vacuum heating | 97.0 | Karl–Fischer, 1H-NMR | 280 |
| [OQuin][NTf2] | own synthesis | - | crystallization, vacuum heating | 97.0 | Karl–Fischer, 1H-NMR | 250 |
| heptane | Sigma-Aldrich b | 99.0 | - | 99.0 | - | 50 |
| thiophene | Sigma-Aldrich b | 99.0 | - | 99.0 | - | 70 |
| 2-methylthiophene | Sigma-Aldrich b | 98.0 | - | 98.0 | - | 110 |
| benzothiophene | Sigma-Aldrich b | 98.0 | - | 98.0 | - | 150 |
| Ionic Liquid | Tg,1/K | ΔCp(g),1/J mol−1 K−1 | Tfus/K | ΔfusH/kJ mol−1 |
|---|---|---|---|---|
| [BiQuin][NTf2] [19] | 217.9 | 332.4 | 321.0 | 46.13 |
| [BQuin][NTf2] [18] | 199.2 | 105.9 | 329.1 | 44.14 |
| [HQuin][NTf2] [20] | 219.3 | 317.7 | 317.2 | 63.54 |
| [OQuin][NTf2] [22] | 219.4 | 423 | 321.3 | 62.91 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Królikowska, M.; Królikowski, M.; Domańska, U. Effect of Cation Structure in Quinolinium-Based Ionic Liquids on the Solubility in Aromatic Sulfur Compounds or Heptane: Thermodynamic Study on Phase Diagrams. Molecules 2020, 25, 5687. https://doi.org/10.3390/molecules25235687
Królikowska M, Królikowski M, Domańska U. Effect of Cation Structure in Quinolinium-Based Ionic Liquids on the Solubility in Aromatic Sulfur Compounds or Heptane: Thermodynamic Study on Phase Diagrams. Molecules. 2020; 25(23):5687. https://doi.org/10.3390/molecules25235687
Chicago/Turabian StyleKrólikowska, Marta, Marek Królikowski, and Urszula Domańska. 2020. "Effect of Cation Structure in Quinolinium-Based Ionic Liquids on the Solubility in Aromatic Sulfur Compounds or Heptane: Thermodynamic Study on Phase Diagrams" Molecules 25, no. 23: 5687. https://doi.org/10.3390/molecules25235687
APA StyleKrólikowska, M., Królikowski, M., & Domańska, U. (2020). Effect of Cation Structure in Quinolinium-Based Ionic Liquids on the Solubility in Aromatic Sulfur Compounds or Heptane: Thermodynamic Study on Phase Diagrams. Molecules, 25(23), 5687. https://doi.org/10.3390/molecules25235687





