Synthesis and Characterization of Functionalized Nanosilica for Zinc Ion Mitigation; Experimental and Computational Investigations
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemical and Methods
2.2. Characterization Techniques
2.3. Procedure of Zn(II) Adsorption
2.4. Synthesis of Grafted Silica
2.5. DFT Simulations
3. Results and Discussion
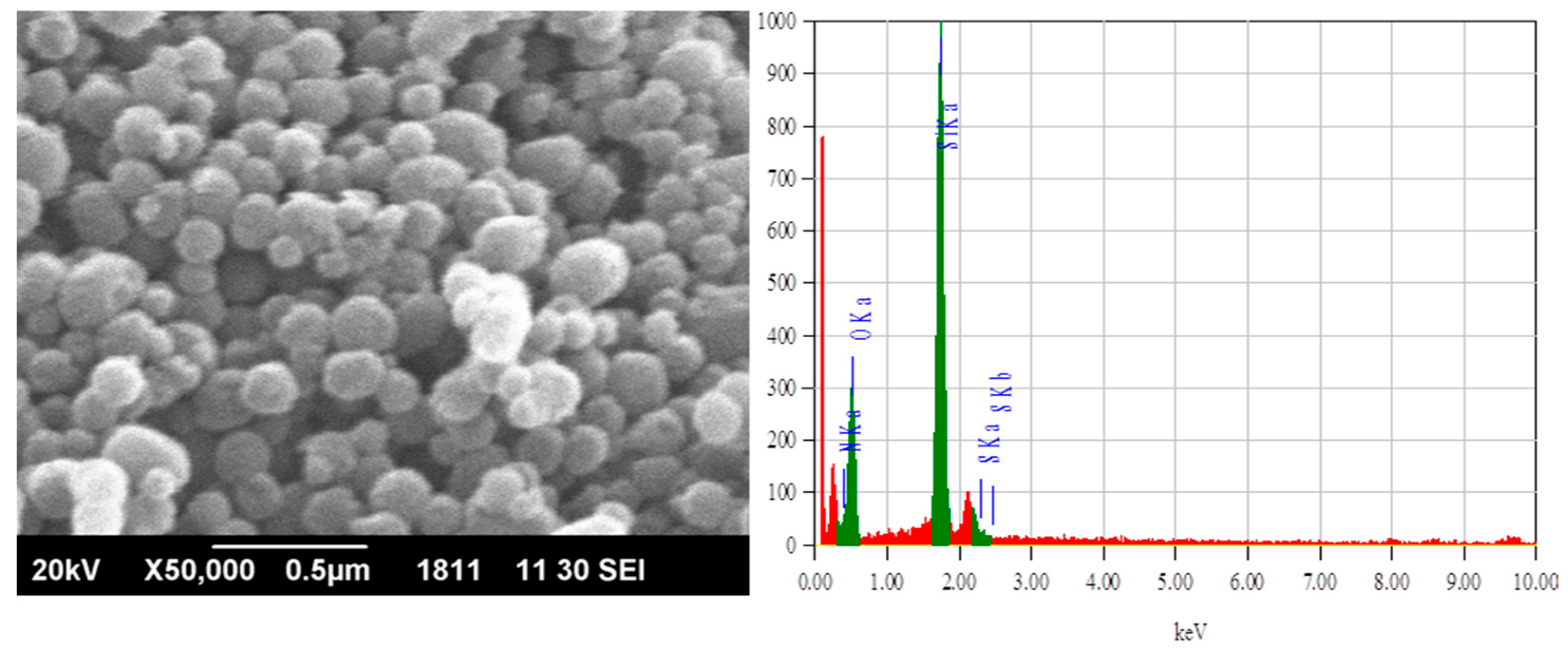
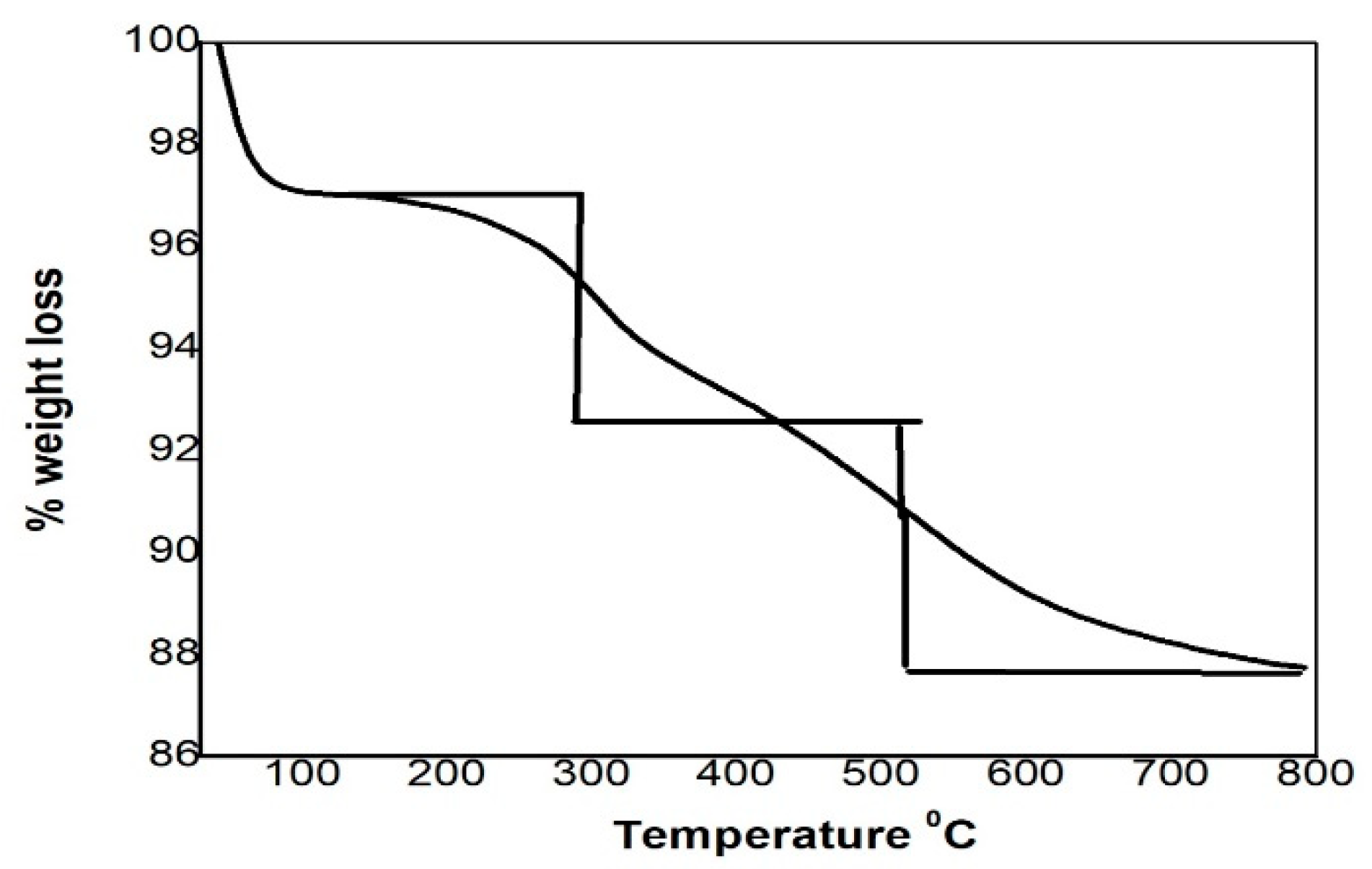
3.1. Adsorbent Characterization
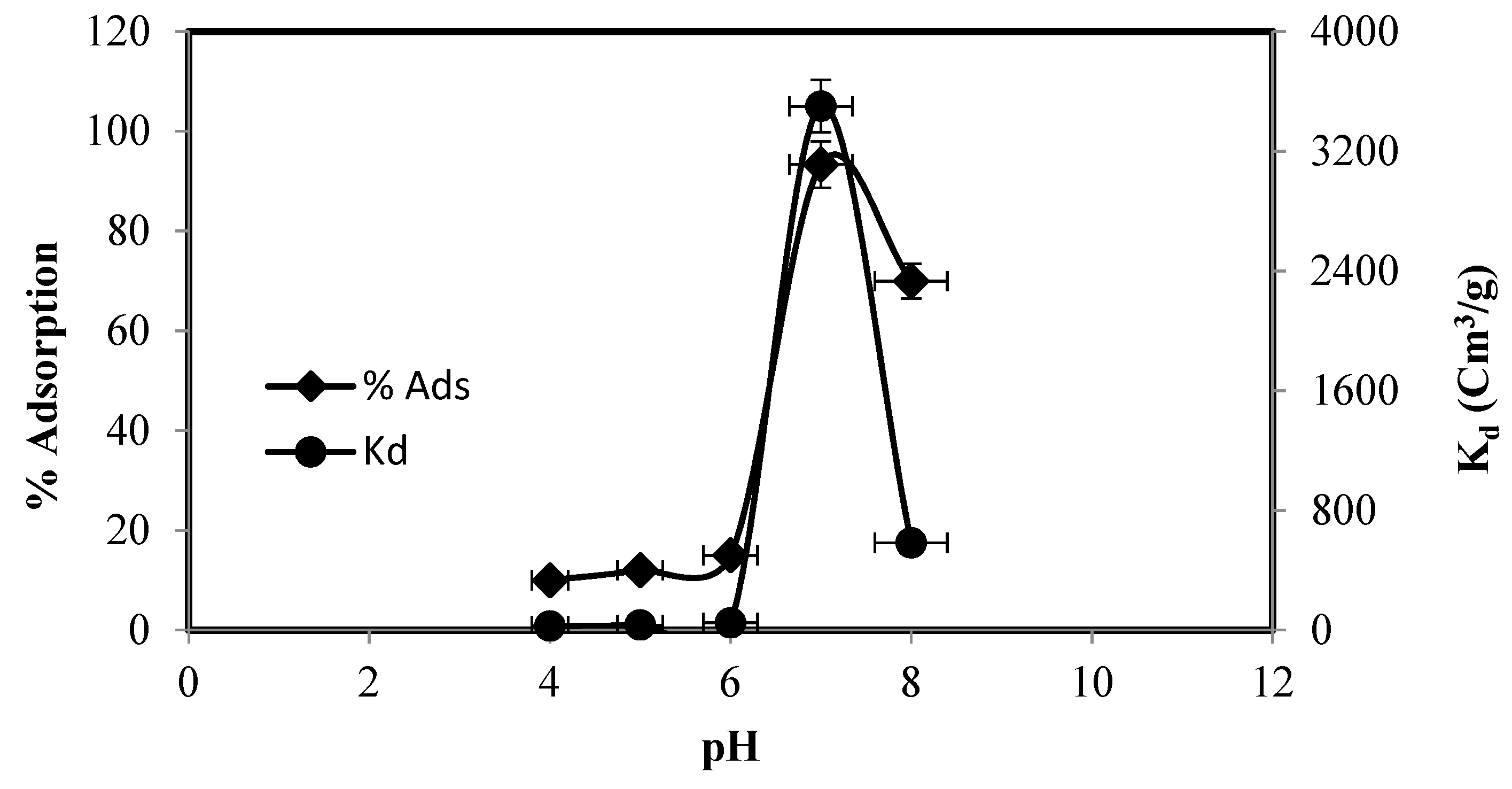
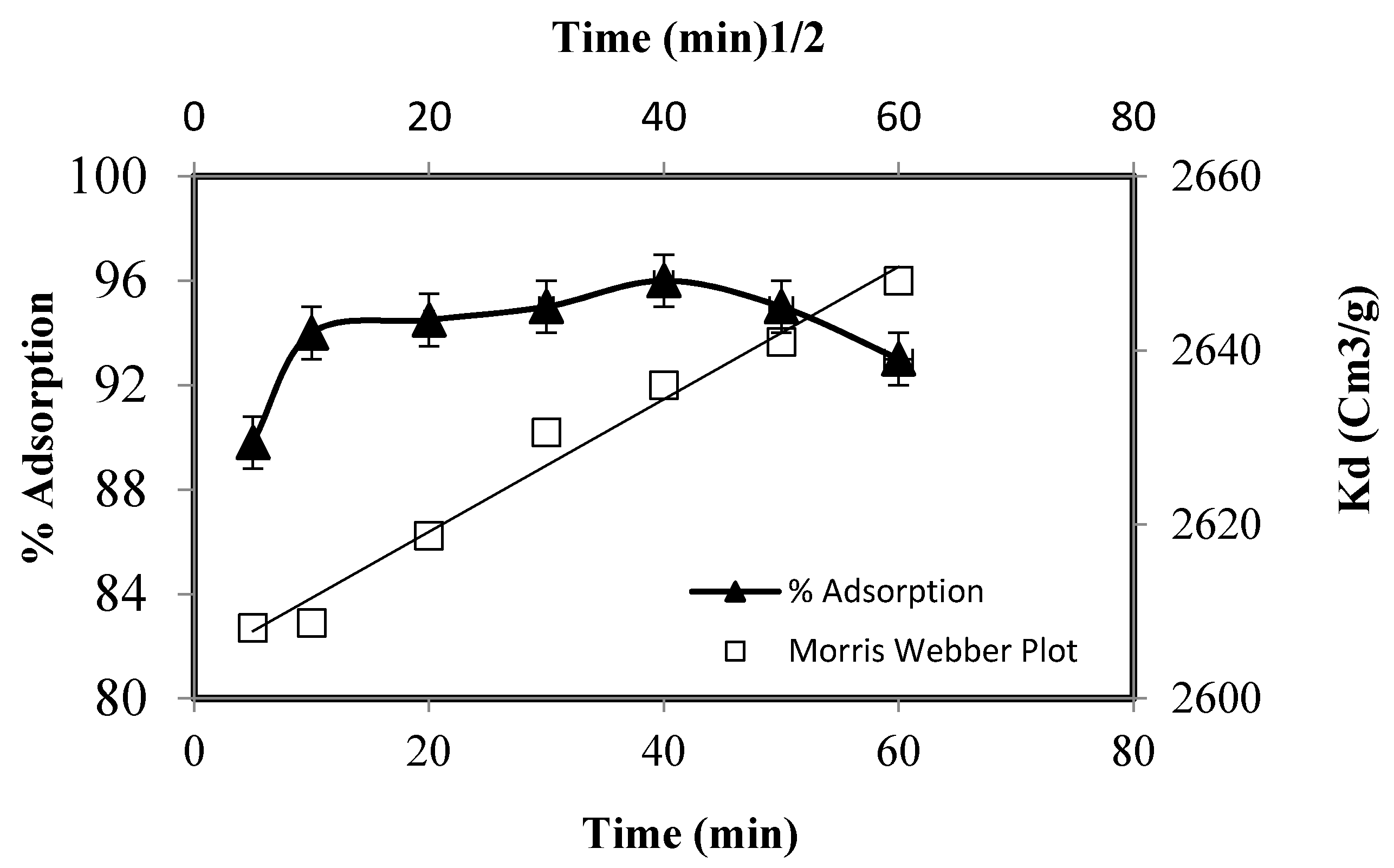
3.2. Zn(II) Adsorption on Functionalized Silica
3.3. Zn(II) Adsorption in the Presence of Other Ions and Sorbent Regeneration
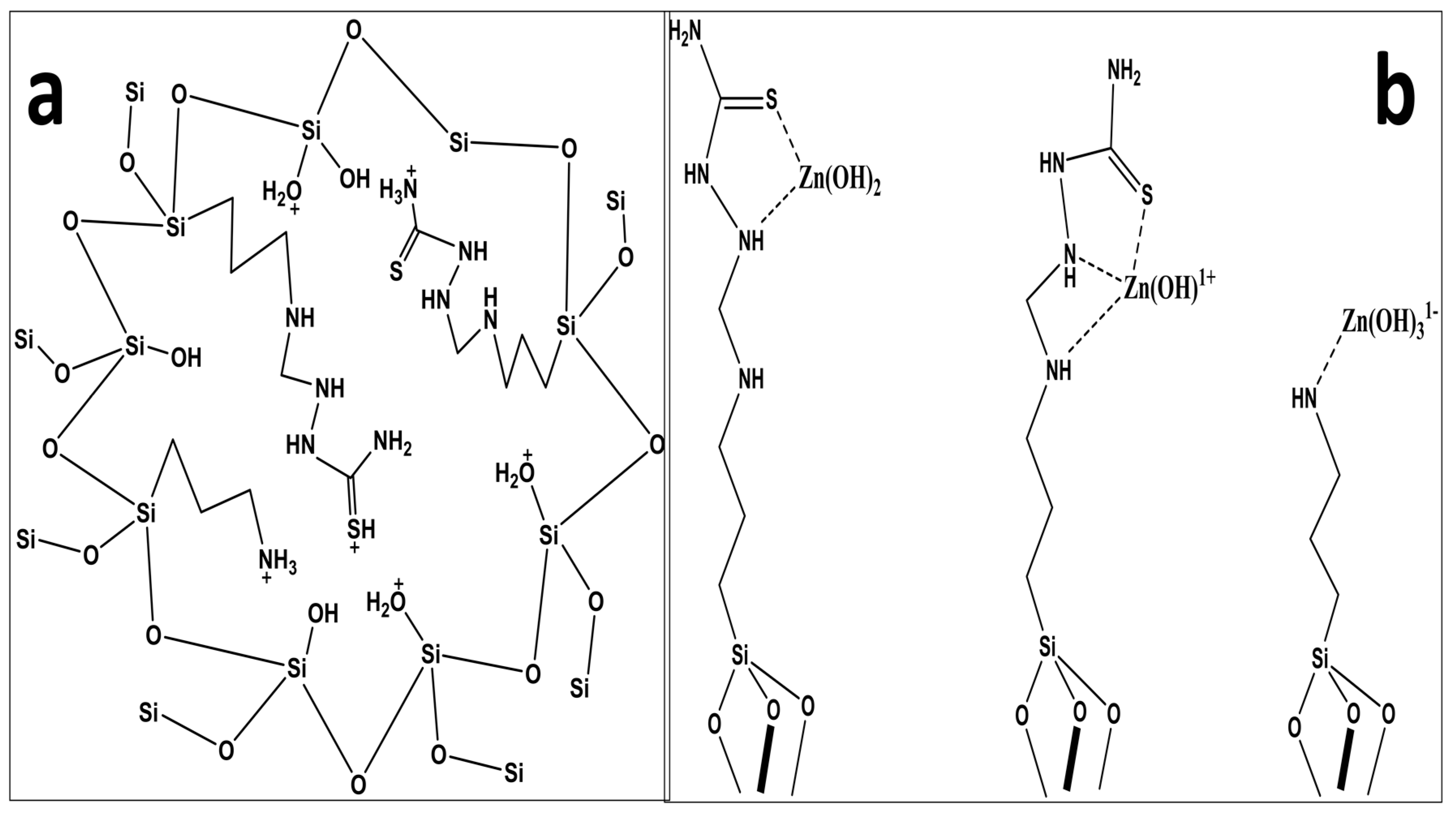
3.4. DFT-Based Thermodynamic Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- He, Y.; Liu, Q.; Hu, J.; Zhao, C.; Peng, C.; Yang, Q.; Wang, H.; Liu, H. Efficient removal of Pb (II) by amine functionalized porous organic polymer through post-synthetic modification. Sep. Purif. Technol. 2017, 180, 142–148. [Google Scholar] [CrossRef]
- Mahmud, H.N.M.E.; Huq, A.O.; Binti Yahya, R. The removal of heavy metal ions from wastewater/aqueous solution using polypyrrole-based adsorbents: A review. RSC Adv. 2016, 6, 14778–14791. [Google Scholar] [CrossRef]
- Hao, S.; Verlotta, A.; Aprea, P.; Pepe, F.; Caputo, D.; Zhu, W. Optimal synthesis of Amino functionalized mesoporous silicas for the adsorption of heavy metal ions. Micropor. Mesopor. Mater. 2016, 236, 250–259. [Google Scholar] [CrossRef]
- Sather, A.C.; Berryman, O.B.; Rebek, J., Jr. Selective recognition and extraction of the uranyl ion. J. Am. Chem. Soc. 2010, 132, 13572–13574. [Google Scholar] [CrossRef] [PubMed]
- Hou, H.; Yu, D.; Hu, G. Preparation and properties of ion-imprinted hollow particles for the selective adsorption of silver ions. Langmuir 2015, 31, 1376–1384. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Vilar, V.J.; Botelho, C.M.R.; Boaventura, A. A review of the use of red mud as adsorbent forthe removal of toxic pollutants from water and wastewater. Environ. Technol. 2011, 32, 231–249. [Google Scholar] [CrossRef]
- Aljerf, L. efficiency extraction of bromocresol purple dye and heavy metals as chromium from industrial effluent by adsorption onto a modified surface of zeolite: Kinetics and equilibrium study. J. Environ. Manag. 2018, 225, 120–132. [Google Scholar] [CrossRef]
- Tao, H.C.; Zhang, H.R.; Li, J.B.; Ding, W.Y. Biomass based activated carbon obtained from sludge and sugarcane bagasse for removing lead ion from wastewater. Bioresour. Technol. 2015, 192, 611–617. [Google Scholar] [CrossRef]
- Nayak, A.; Bhushan, B.; Gupta, V.; Sharma, P. Chemically activated carbon from lignocellulosic wastes for heavy metal wastewater remediation: Effect of activation conditions. J. Colloid Interface Sci. 2017, 493, 228–240. [Google Scholar] [CrossRef]
- Tavakoli, O.; Goodarzi, V.; Saeb, M.R.; Mahmoodi, N.M.; Borja, R. Competitive removal of heavy metal ions from squid oil under isothermal condition by CR11 chelate ion exchanger. J. Hazard. Mater. 2017, 334, 256–266. [Google Scholar] [CrossRef]
- Samiey, B.; Cheng, C.H.; Wu, J. Organic-inorganic hybrid polymers as adsorbents for removal of heavy metal ions from solutions: A review. Materials 2014, 7, 673–726. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, M.E.; Hafez, O.F.; Alrefaay, A.; Osman, M.M. Performance evaluation of hybrid inorganic/organic adsorbents in removal and preconcentration of heavy metals from drinking and industrial waste water. Desalination 2010, 253, 9–15. [Google Scholar] [CrossRef]
- Soylak, M.; Unsal, Y.E. Chromium and iron determinations in food and herbal plant samples by atomic absorption spectrometry after solid phase extraction on single-walled carbon nanotubes (SWCNTs) disk. Food Chem. Toxicol. 2010, 48, 1511–1515. [Google Scholar] [CrossRef] [PubMed]
- Sierra, I.; Pérez-Quintanilla, D. Heavy metal complexation on hybrid mesoporous silica: An approach to analytical applications. Chem. Soc. Rev. 2013, 42, 3792–3807. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for Drinking-Water Quality, 4th ed.; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Shafiabadi, M.; Dashti, A.; Tayebi, H.A. Removal of Hg (II) from aqueous solution using polypyrrole/SBA-15 nanocomposite: Experimental and modeling. Synth. Metals 2016, 212, 154–160. [Google Scholar] [CrossRef]
- Ahmad, R.; Ahmed, M. Removal of Zn(II) ions from aqueous solutions using BPHA-impregnated polyurethane foam. J. Chin. Chem. Soc. 2008, 55, 147–154. [Google Scholar] [CrossRef]
- Yang, H.; Xu, R.; Xue, X.; Li, F.; Li, G. Hybrid surfactant-templated mesoporous silica formed in ethanol and its applications for heavy metal removal. J. Hazard. Mater. 2008, 152, 690–698. [Google Scholar] [CrossRef]
- Hossain, K.Z.; Mercier, L. Intra frame work metal ion adsorption in ligand functionalized mesoporous silica. Adv. Mater. 2002, 14, 1053–1056. [Google Scholar] [CrossRef]
- Pérez-Quintanilla, D.; Sánchez, A.; del Hierro, I.; Fajardo, M.; Sierra, I. Preparation, characterization, and Zn2+ adsorption behavior of chemically modified MCM-41 with 5-mercapto-1-methyltetrazole. J. Colloid Interface Sci. 2007, 313, 551–562. [Google Scholar] [CrossRef]
- Frisch, M.; Trucks, G.; Schlegel, H.; Scuseria, G.; Robb, M.; Cheeseman, J.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.; et al. Gaussian 09, Revision A. 02; Gaussian Inc.: Wallingford, CT, USA, 2009; pp. 1372–1377. [Google Scholar]
- Heidari, A.; Younesi, H.; Mehraban, Z. Removal of Ni (II), Cd (II), and Pb (II) from a ternary aqueous solution by amino functionalized mesoporous and nano mesoporous silica. Chem. Eng. J. 2009, 153, 70–79. [Google Scholar] [CrossRef]
- Marenich, A.V.; Cramer, C.J.; Truhlar, D.G. Universal Solvation Model Based on Solute Electron Density and on a Continuum Model of the Solvent Defined by the Bulk Dielectric Constant and Atomic Surface Tensions. J. Phys. Chem. B 2009, 113, 6378–6396. [Google Scholar] [CrossRef] [PubMed]
- Arakaki, L.; Filha, V.A.; Germano, A.; Santos, S.; Fonseca, M.; Sousa, K.; Espínola, J.; Arakaki, T. Silica gel modified with ethylenediamine and succinic acid–adsorption and calorimetry of cations in aqueous solution. Thermochim. Acta. 2013, 556, 34–40. [Google Scholar] [CrossRef]
- Arce, V.B.; Scotto, J.; Allegretti, P.E.; Melo, M.A., Jr.; Airoldi, C.; Salum, M.L.; Erra-Balsells, R.; Pis Diez, R.; Mártire, D.O. Combined experimental and computational investigation of the fluorescence quenching of riboflavin by cinnamic alcohol chemisorbed on silica nanoparticles. J. Phys. Chem. C 2014, 118, 15348–15355. [Google Scholar] [CrossRef]
- Sakkayawong, N.; Thiravetyan, P.; Nakbanpote, W. Adsorption mechanism of synthetic reactive dye wastewater by chitosan. J. Colloid Interface Sci. 2005, 286, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Akhavan, B.; Jarvis, K.; Majewski, P. Plasma Polymer Functionalized Silica Particles for Heavy Metals Removal. ACS App. mater. Interfaces 2015, 7, 4265–4274. [Google Scholar]
- De Zoubov, N.; Pourbaix, M. Atlas of Electrochemical Equilibria in Aqueous Solutions; Pourbaix, M., Ed.; National Association of Corrosion Engineers; Ceberlcor: Houston, TX, USA, 1966; pp. 406–413. [Google Scholar]
- Kaya, A.; Ören, A.H. Adsorption of Zinc from Aqueous Solutions to Bentonite. J. Hazard. Mater. 2005, 125, 183–189. [Google Scholar] [CrossRef]
- Algarra, M.; Jiménez, M.V.; Rodríguez-Castellón, E.; Jiménez-López, A.; Jiménez-Jiménez, J. Heavy metals removal from electroplating wastewater by aminopropyl-Si MCM-41. Chemosphere 2005, 59, 779–786. [Google Scholar] [CrossRef]
- Xie, X.; Tian, Y.; Qin, Z.; Yu, Q.; Wei, H.; Wang, D.; Li, X.; Wang, X. Complexation of Manganese with Glutarimidedioxime: Implication for Extraction Uranium from Seawater. Sci. Rep. 2017, 7, 43503. [Google Scholar] [CrossRef]
- Niu, Y.; Yang, J.; Qu, R.; Gao, Y.; Du, N.; Chen, H.; Sun, C.; Wang, W. Synthesis of Silica-Gel-Supported Sulfur-Capped PAMAM Dendrimers for Efficient Hg(II) Adsorption: Experimental and DFT Study. Ind. Eng. Chem. Res. 2016, 55, 3679–3688. [Google Scholar] [CrossRef]
- Remko, M.; Broer, R.; Remková, A.; Duijnen, P.T.V. How strong are Ca2+–heparin and Zn2+–heparin interactions? Chem. Phys. Lett. 2015, 621, 12–17. [Google Scholar] [CrossRef]
- Carreño, A.; Schott, E.; Zarate, X.; Manriquez, J.M.; Vega, J.C.; Mardones, M.; Cowley, A.H.; Chavez, I.; Hinestroza, J.P.; Arratia-Perez, R. DFT studies on coordination models for adsorption essays of Cu(II) and Ni(II) solutions in modified silica gel with iminodiacetic groups. Chem. Pap. 2017, 71, 1019–1030. [Google Scholar] [CrossRef]
- Hartmann, M.; Clark, T.; Eldik, R. Hydration and Water Exchange of Zinc(II) Ions. Application of Density theory. J. Am. Chem. Soc. 1997, 119, 7843–7850. [Google Scholar] [CrossRef]
- Melo, D.Q.; Neto, V.O.; Oliveira, J.T.; Barros, A.L.; Gomes, E.C.; Raulino, G.S.; Longuinotti, E.; Nascimento, R.F. Adsorption equilibria of Cu2+, Zn2+, and Cd2+ on EDTA-functionalized silica spheres. J. Chem. Eng. Data 2013, 58, 798–806. [Google Scholar] [CrossRef]
- Dąbrowski, A. Adsorption—From theory to practice. Adv. Colloid Interface Sci. 2001, 93, 135–224. [Google Scholar] [CrossRef]
- Mousavi, S.J.; Parvini, M.; Ghorbani, M. Experimental design data for the zinc ions adsorption based on Mesoporous modified chitosan using Central Composite Design method. Carbohydr. Polym. 2018, 188, 197–212. [Google Scholar] [CrossRef]
- Singh, K.; Mohan, S. Kinetic studies of the sucrose adsorption onto an alumina interface. App. Surf. Sci. 2004, 221, 308–318. [Google Scholar] [CrossRef]
- Hanif, M.A.; Nadeem, R.; Bhatti, H.N.; Ahmad, N.R.; Ansari, T.M. Ni(II) bio sorption by Cassia fistula (Golden Shower) biomass. J. Hazard Matter. 2007, 139, 345–355. [Google Scholar] [CrossRef]
- Quintelas, C.; Rocha, Z.; Silva, B.; Fonseca, B.; Figueiredo, H.; Tavares, T. Removal of Cd (II), Cr (VI), Fe (III) and Ni (II) from aqueous solutions by an E. coli biofilm supported on kaolin. Chem. Eng. J. 2009, 149, 319–324. [Google Scholar] [CrossRef]
- Tran, L.; Wu, P.; Zhu, Y.; Liu, S.; Zhu, N. Comparative study of Hg(II) adsorption by thiol- and hydroxyl-containing bifunctional montmorillonite and vermiculite. App. Surf. Sci. 2015, 356, 91–101. [Google Scholar] [CrossRef]
- Liu, Y.; Gao, M.; Gu, Z.; Luo, Z.; Ye, Y.; Lu, L. Comparison between the removal of phenol and catechol by modified montmorillonite with two novel hydroxyl-containing Gemini surfactants. J. Hazard. Mater. 2014, 267, 71–80. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |

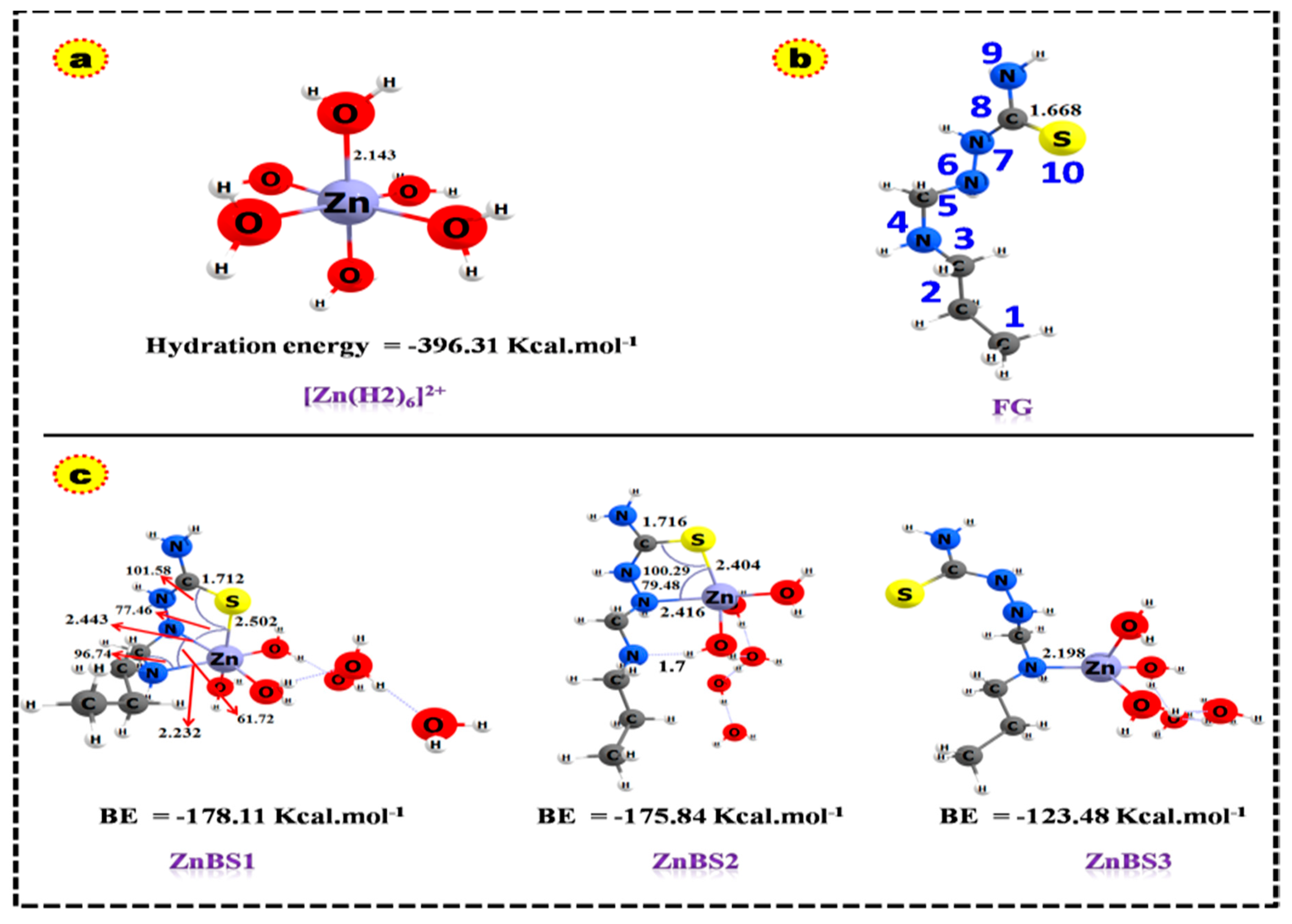
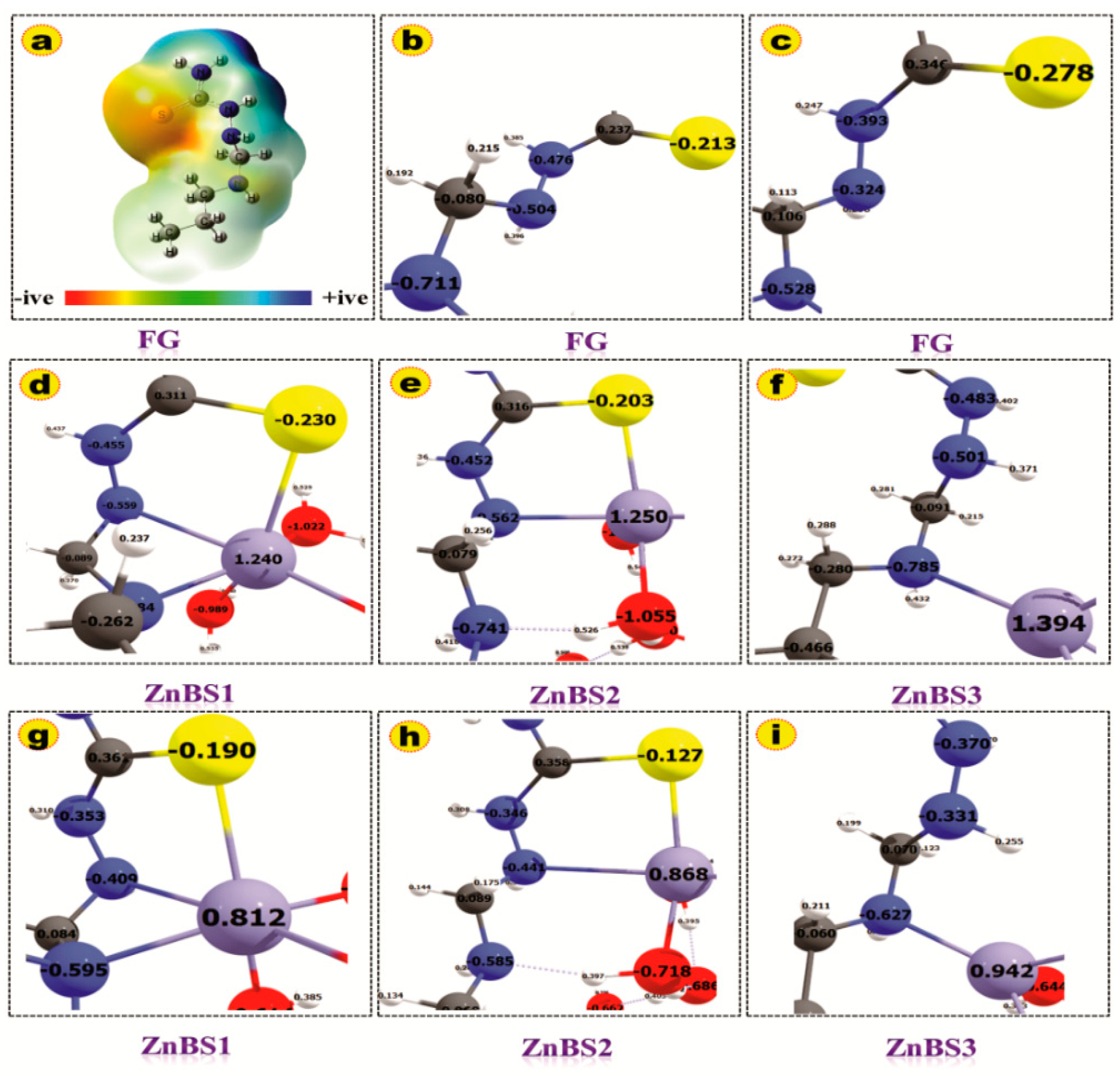
| Specie | Bond Lengths (Å) | |||||
| C=S | Zn—O | 4N—Zn | 6N—Zn | 10S—Zn | 4N…HO | |
| FG | 1.67 | |||||
| FG-Zn-1 | 1.71 | 2.23 | 2.44 | 2.50 | ||
| FG-Zn-2 | 1.72 | 2.42 | 2.40 | 1.7 | ||
| FG-Zn-3 | 2.20 | |||||
| [Zn(H2O)6]2+ | 2.14 | |||||
| Specie | Bond Angles (°) | |||||
| 5C—4N—Zn | 4N—Zn—6N | 6N—Zn—10S | 8C—10S—Zn | 5C—4N—Zn | ||
| FG-Zn-1 | 96.74 | 61.72 | 77.46 | 101.58 | ||
| FG-Zn-2 | 79.48 | 100.29 | ||||
| FG-Zn-3 | 110.7 | |||||
| Complex | ||||||||
|---|---|---|---|---|---|---|---|---|
| Gas Phase | Solvent Phase | Gas Phase | Solvent Phase | Gas Phase | Solvent Phase | Gas Phase | Solvent Phase | |
| Zn(H2O)6 | −396.3 | −80.91 | −394.37 | −78.91 | −362.61 | −69.43 | −218.23 | −65.31 |
| ZnBS1 | −178.11 | −59.89 | −169.82 | −57.45 | −140.76 | −49.81 | −97.46 | −51.31 |
| ZnBS2 | −175.84 | −58.90 | −168.35 | −54.71 | −140.84 | −45.72 | −92.25 | −49.10 |
| ZnBS3 | −123.48 | −41.37 | −117.77 | −40.3 | −86.66 | −37.6 | −104.34 | −54.4 |
| Isotherm Model | Constants | Values |
|---|---|---|
| Langmuir isotherm | Q (µmol g−1) | 20,000 |
| b × 103 (L mol−1) | 3.8 | |
| R2 | 0.99 | |
| Freundlich isotherm | 1/n | 0.1627 |
| Cm (mmol g−1) | 1.18 | |
| R2 | 0.64 | |
| D-R isotherm | β (KJ2 mol−2) | −0.0015 |
| Xm (mmol g−1) | 27.20 | |
| Es (KJ mol−1) | 18.25 | |
| R2 | 0.97 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ali, Z.; Ahmad, R.; Farooq, W.A.; Khan, A.; Khan, A.A.; Bibi, S.; Adalat, B.; Almutairi, M.A.; Yaqub, N.; Atif, M. Synthesis and Characterization of Functionalized Nanosilica for Zinc Ion Mitigation; Experimental and Computational Investigations. Molecules 2020, 25, 5534. https://doi.org/10.3390/molecules25235534
Ali Z, Ahmad R, Farooq WA, Khan A, Khan AA, Bibi S, Adalat B, Almutairi MA, Yaqub N, Atif M. Synthesis and Characterization of Functionalized Nanosilica for Zinc Ion Mitigation; Experimental and Computational Investigations. Molecules. 2020; 25(23):5534. https://doi.org/10.3390/molecules25235534
Chicago/Turabian StyleAli, Zarshad, Rashid Ahmad, W. Aslam Farooq, Aslam Khan, Adnan Ali Khan, Saira Bibi, Bushra Adalat, Mona A. Almutairi, Nafeesah Yaqub, and Muhammad Atif. 2020. "Synthesis and Characterization of Functionalized Nanosilica for Zinc Ion Mitigation; Experimental and Computational Investigations" Molecules 25, no. 23: 5534. https://doi.org/10.3390/molecules25235534
APA StyleAli, Z., Ahmad, R., Farooq, W. A., Khan, A., Khan, A. A., Bibi, S., Adalat, B., Almutairi, M. A., Yaqub, N., & Atif, M. (2020). Synthesis and Characterization of Functionalized Nanosilica for Zinc Ion Mitigation; Experimental and Computational Investigations. Molecules, 25(23), 5534. https://doi.org/10.3390/molecules25235534






