Medicinal Activities and Nanomedicine Delivery Strategies for Brucea javanica Oil and Its Molecular Components
Abstract
1. Introduction
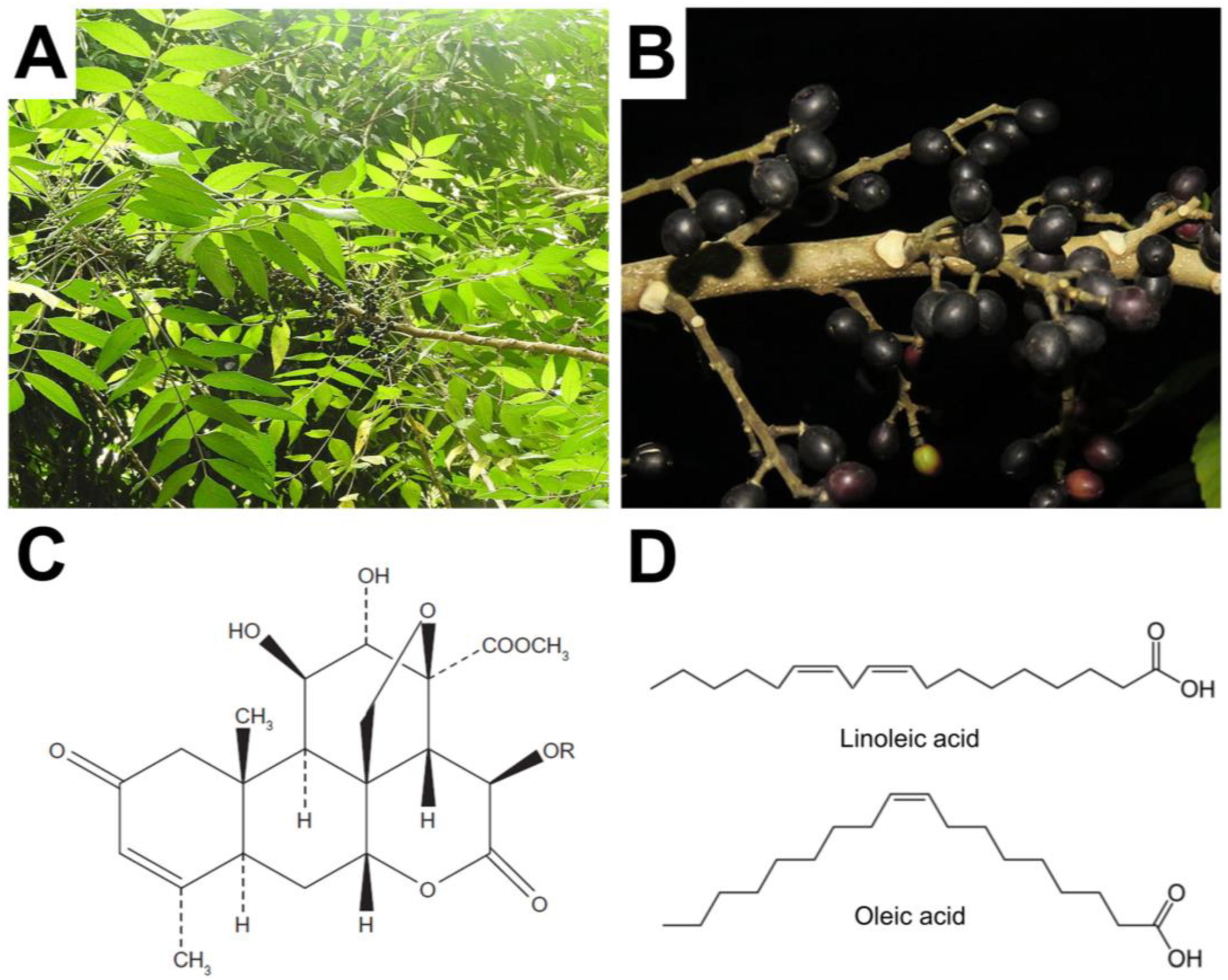
2. Overview of Brucea javanica Oil and Molecular Components
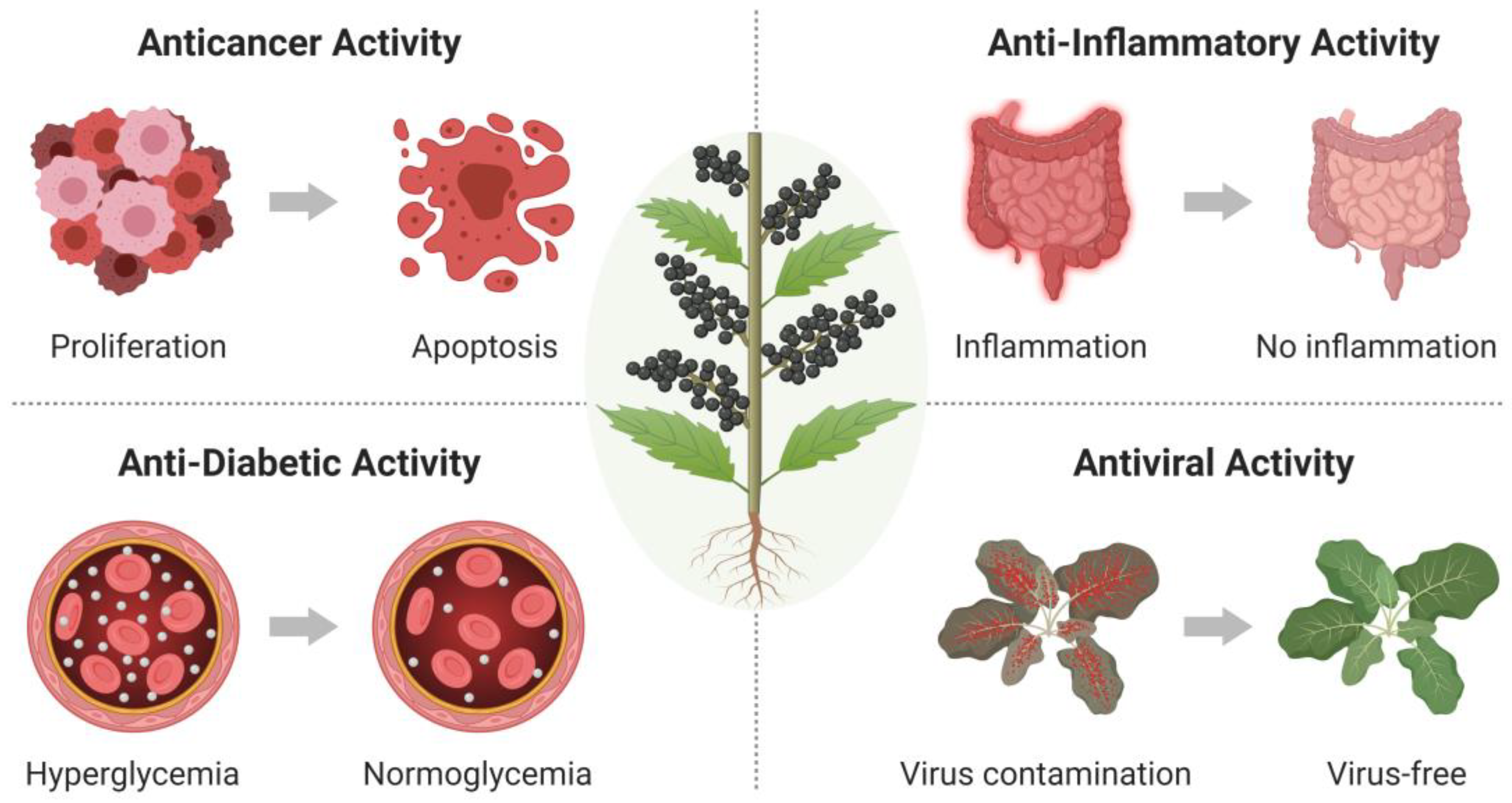
2.1. Anticancer Activity
2.2. Anti-Inflammatory Activity
2.3. Anti-Diabetic Activity
2.4. Antiviral Activity
3. Nanomedicine Delivery Strategies
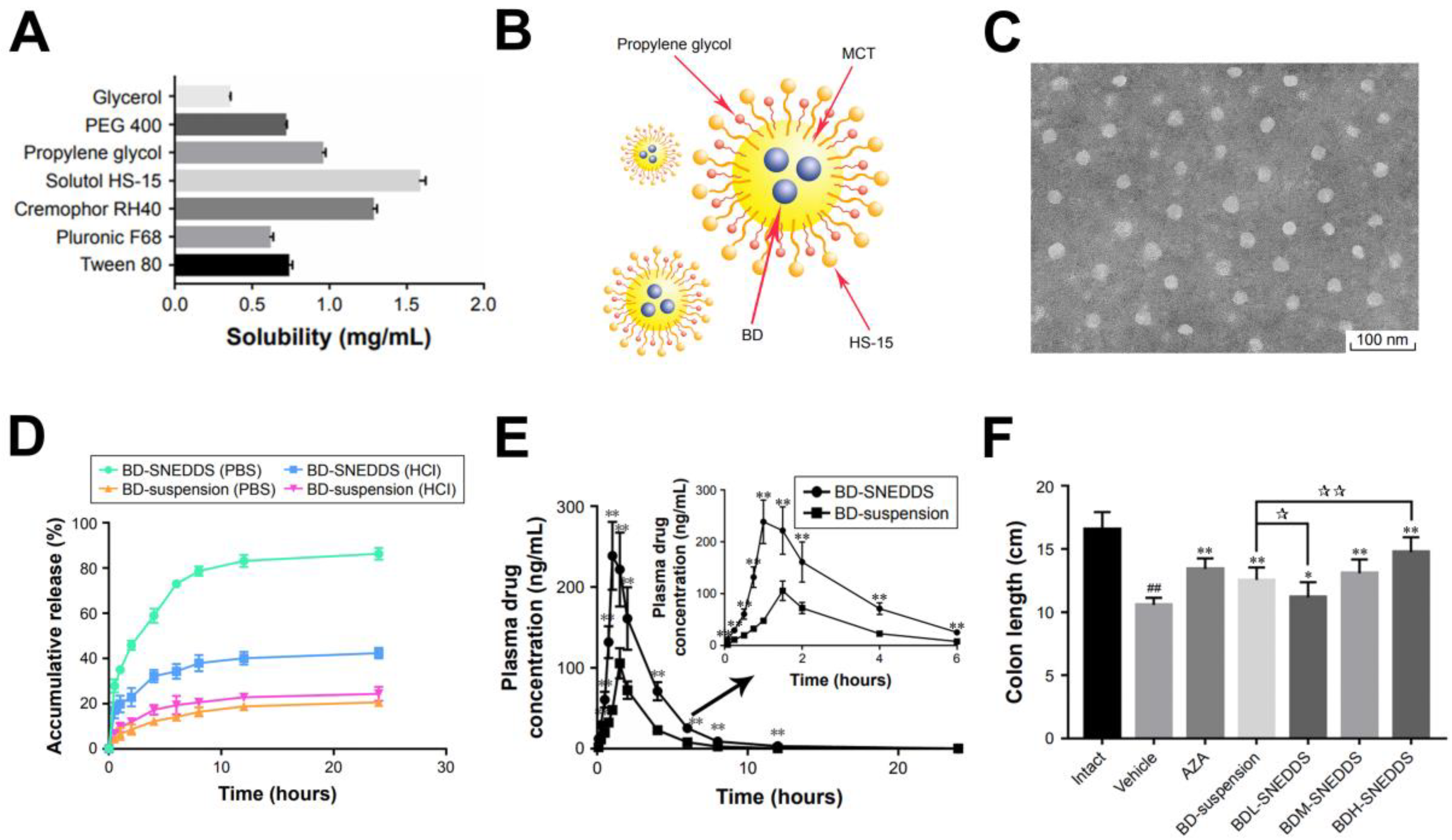
3.1. Nanoemulsions
3.1.1. BJO Encapsulation
3.1.2. Quassinoid Encapsulation
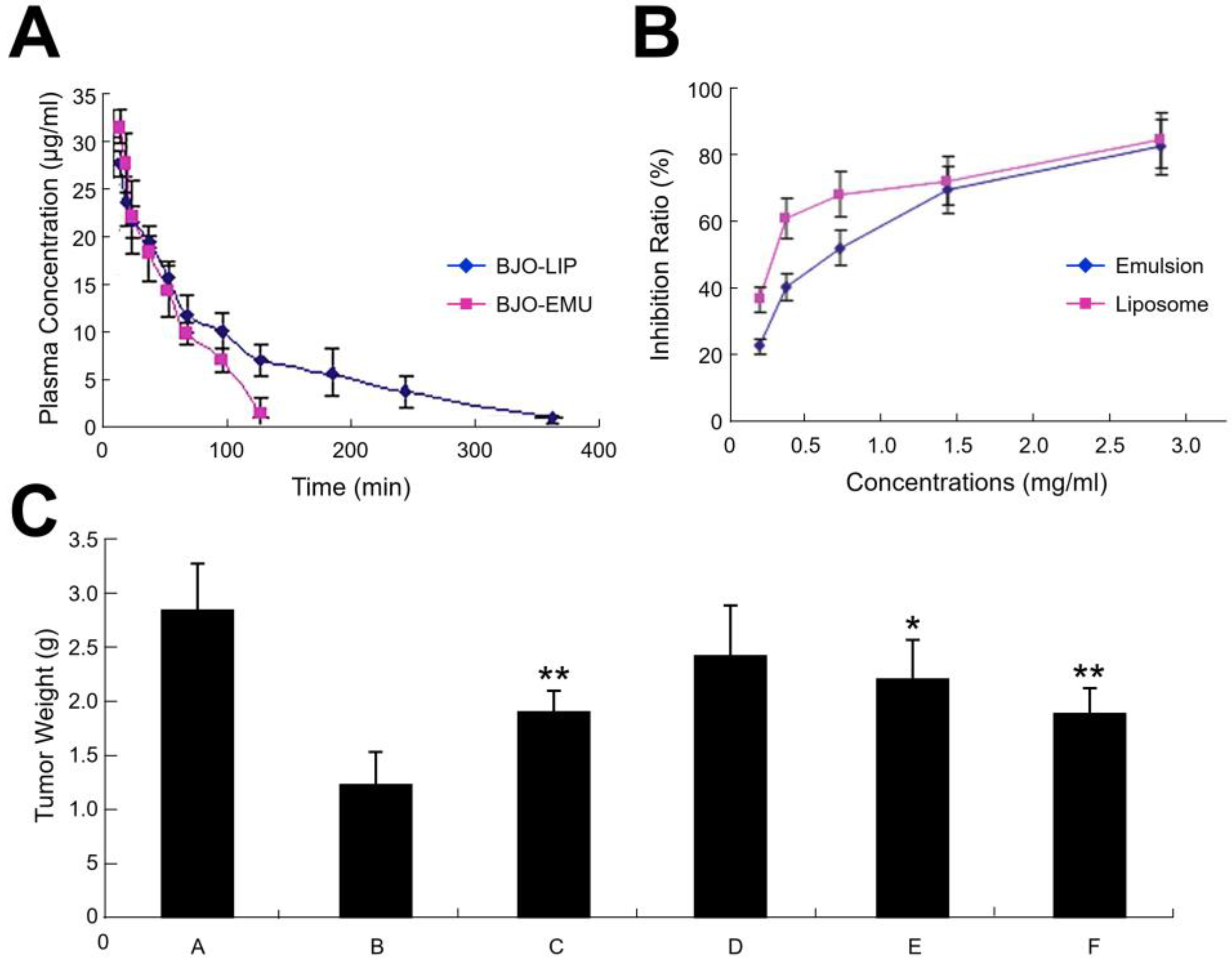
3.2. Liposomes
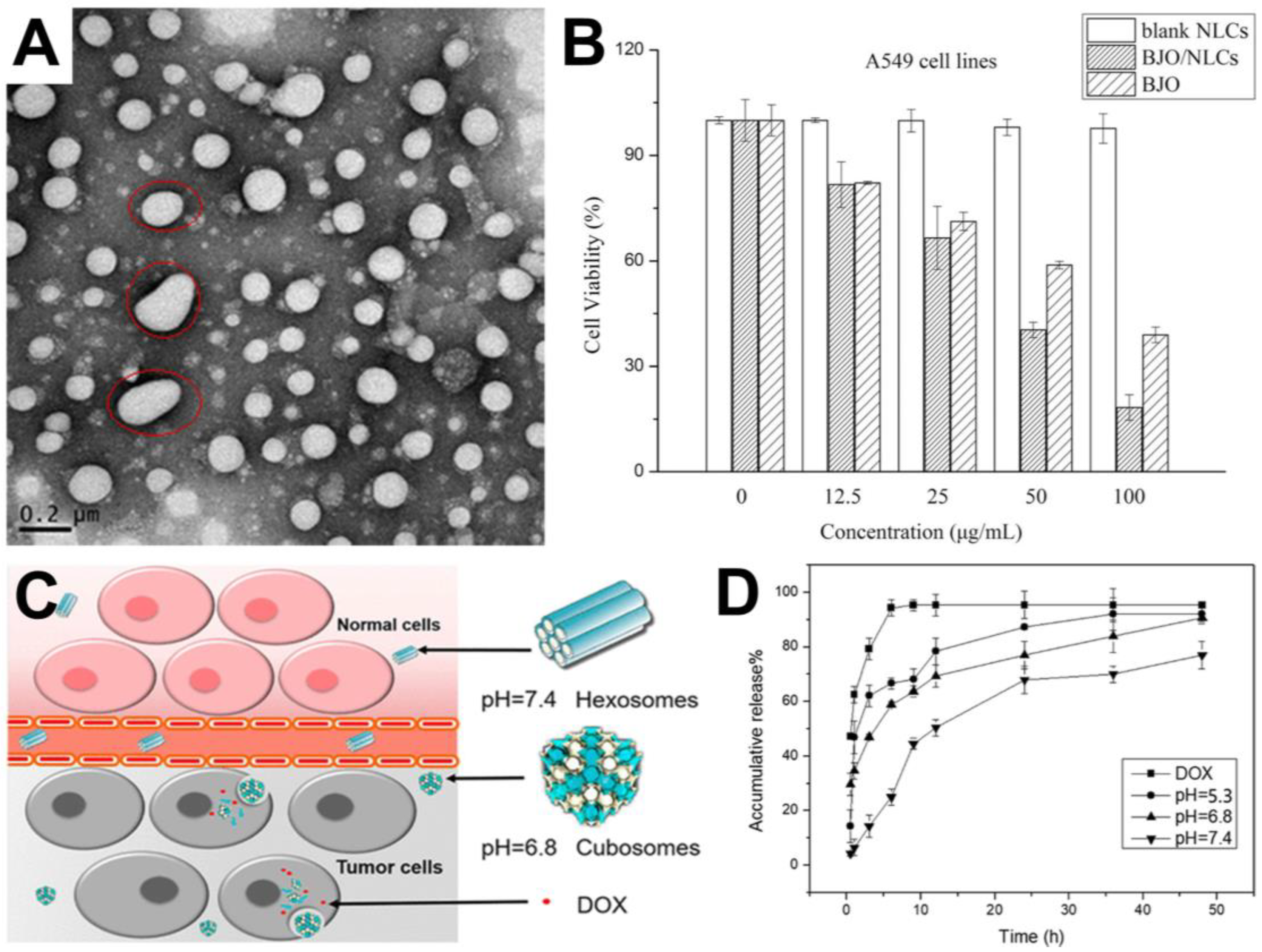
3.3. Nanostructured Lipid Carriers
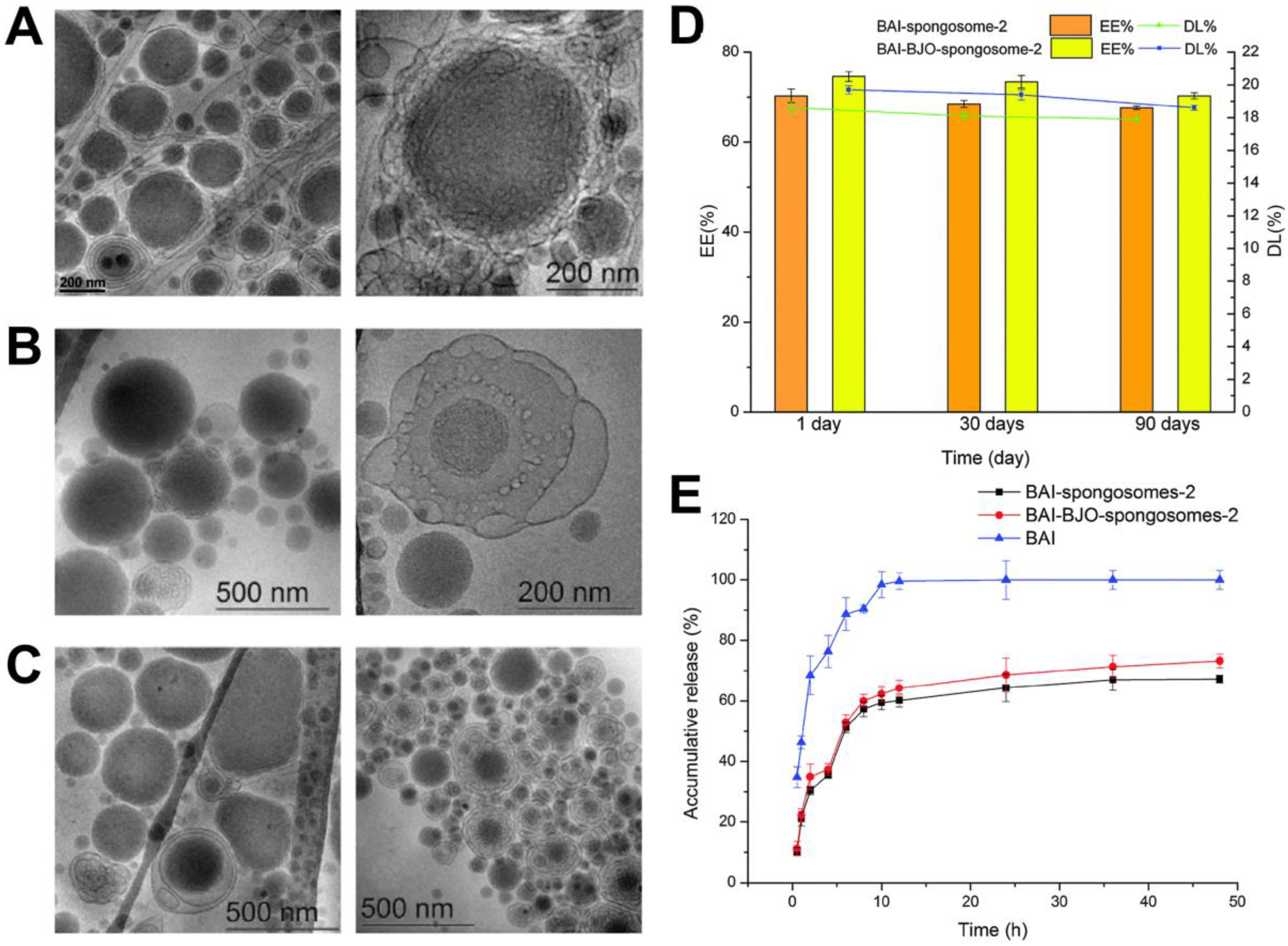
3.4. Spongosomes
4. Conclusions and Outlook
Author Contributions
Funding
Conflicts of Interest
References
- Man, S.; Gao, W.; Wei, C.; Liu, C.-X. Anticancer drugs from traditional toxic Chinese medicines. Phytother. Res. 2012, 26, 1449–1465. [Google Scholar] [CrossRef] [PubMed]
- Ablat, A.; Mohamad, J.; Awang, K.; Shilpi, J.A.; Arya, A. Evaluation of antidiabetic and antioxidant properties of Brucea javanica seed. Sci. World J. 2014, 2014, 86130. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Li, C.; Zhang, Y.; Wen, Q.; Ren, D. Phytochemical and biological activities of an anticancer plant medicine: Brucea javanica. Anti-Cancer Agents Med. Chem. 2014, 14, 440–458. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Chen, R.; Wang, S.; Tan, W.; Hu, Y.; Peng, X.; Wang, Y. Chemical components, pharmacological properties, and nanoparticulate delivery systems of Brucea javanica. Int. J. Nanomed. 2013, 8, 85–92. [Google Scholar] [CrossRef]
- Yan, Z.; Guo, G.-F.; Zhang, B. Research of Brucea javanica against cancer. Chin. J. Integr. Med. 2017, 23, 153–160. [Google Scholar] [CrossRef]
- Tang, W.; Eisenbrand, G. Brucea javanica (L.) Merr. In Chinese Drugs of Plant Origin: Chemistry, Pharmacology, and Use in Traditional and Modern Medicine; Tang, W., Eisenbrand, G., Eds.; Springer: Berlin, Germany, 1992; pp. 207–222. ISBN 978-3-642-73739-8. [Google Scholar]
- Samy, J.; Sugumaran, M.; Lee, K.L.W. Herbs of Malaysia: An Introduction to the Medicinal, Culinary, Aromatic and Cosmetic Use of Herbs; Wong, K.M., Ed.; Times Editions: Shah Alam, Malaysia, 2005; ISBN 9833001793. [Google Scholar]
- Yan, Z.; Zhang, B.; Huang, Y.; Qiu, H.; Chen, P.; Guo, G.F. Involvement of autophagy inhibition in Brucea javanica oil emulsion-induced colon cancer cell death. Oncol. Lett. 2015, 9, 1425–1431. [Google Scholar] [CrossRef]
- Vieira, I.J.C.; Braz-Filho, R. Quassinoids: Structural diversity, biological activity and synthetic studies. In Studies in Natural Products Chemistry; Atta-ur-Rahman, Ed.; Elsevier: Amsterdam, The Netherlands, 2006; Volume 33, pp. 433–492. ISBN 978-0-444-52717-2. [Google Scholar]
- Alves, I.A.; Miranda, H.M.; Soares, L.A.; Randau, K.P. Simaroubaceae family: Botany, chemical composition and biological activities. Rev. Bras. Farmacogn. 2014, 24, 481–501. [Google Scholar] [CrossRef]
- Yu, Y.-L.; Lu, Y.; Tang, X.; Cui, F.-D. Formulation, preparation and evaluation of an intravenous emulsion containing Brucea javanica oil and Coix seed oil for anti-tumor application. Biol. Pharm. Bull. 2008, 31, 673–680. [Google Scholar] [CrossRef]
- Fuhong, D.; Xiang, G.; Haiying, L.; Jiangye, W.; Xueming, G.; Wenxiao, C. Evaluation of efficacy and safety for Brucea javanica oil emulsion in the control of the malignant pleural effusions via thoracic perfusion. BMC Cancer 2018, 18, 411. [Google Scholar] [CrossRef]
- Xunyi, N.; Dalin, H.; Jiangong, D. Treatment of prostatic carcinoma (stage C to D) with fructus bruceae emulsion. J. Clin. Urol. 1998, 12, R737.25. [Google Scholar]
- Shan, G.-Y.; Zhang, S.; Li, G.-W.; Chen, Y.-S.; Liu, X.-A.; Wang, J.-K. Clinical evaluation of oral Fructus bruceae oil combined with radiotherapy for the treatment of esophageal cancer. Chin. J. Integr. Med. 2011, 17, 933–936. [Google Scholar] [CrossRef] [PubMed]
- Ji, Z.-Q.; Huang, X.-E.; Wu, X.-Y.; Liu, J.; Wang, L.; Tang, J.-H. Safety of Brucea javanica and Cantharidin combined with chemotherapy for treatment of NSCLC patients. Asian Pac. J. Cancer Prev. 2014, 15, 8603–8605. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.-R.; Liu, S.-Y.; Zhu, J.-L.; Zhang, D.; Wang, K.-H. Efficacy of Brucea javanica oil emulsion injection combined with the chemotherapy for treating gastric cancer: A systematic review and meta-analysis. Evid. Based Complement. Alternat. Med. 2018, 2018, 6350782. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Wu, Z.; Liu, X.; Ni, R.; Zhu, X.; Ma, L.; Liu, J. Preparation, safety, pharmacokinetics, and pharmacodynamics of liposomes containing Brucea javanica oil. AAPS PharmSciTech 2010, 11, 878–884. [Google Scholar] [CrossRef][Green Version]
- Xiang, Q.; Zhou, L.; Zhang, H.; Zhang, H.; Yao, C.; Hang, Y. The pharmacokinetic studies of Brucea javanica oil microemulsions in rats. Zhongyaocai 2007, 30, 1113–1115. [Google Scholar]
- Zhang, Y.; Zhang, L.; Zhang, Q.; Zhang, X.; Zhang, T.; Wang, B. Enhanced gastric therapeutic effects of Brucea javanica oil and its gastroretentive drug delivery system compared to commercial products in pharmacokinetics study. Drug Des. Devel. Ther. 2018, 12, 535–544. [Google Scholar] [CrossRef]
- Tan, T.; Lai, C.-J.-S.; Zeng, S.-L.; Li, P.; Liu, E.-H. Comprehensive profiling and characterization of quassinoids from the seeds of Brucea javanica via segment and exposure strategy coupled with modified mass defect filter. Anal. Bioanal. Chem. 2016, 408, 527–533. [Google Scholar] [CrossRef]
- Ye, Q.-M.; Bai, L.-L.; Hu, S.-Z.; Tian, H.-Y.; Ruan, L.-J.; Tan, Y.-F.; Hu, L.-P.; Ye, W.-C.; Zhang, D.-M.; Jiang, R.-W. Isolation, chemotaxonomic significance and cytotoxic effects of quassinoids from Brucea javanica. Fitoterapia 2015, 105, 66–72. [Google Scholar] [CrossRef]
- Zhang, H.; Yang, J.Y.; Zhou, F.; Wang, L.H.; Zhang, W.; Sha, S.; Wu, C.F. Seed oil of Brucea javanica induces apoptotic death of acute myeloid leukemia cells via both the death receptors and the mitochondrial-related pathways. Evid. Based Complement. Alternat. Med. 2011, 2011, 965016. [Google Scholar] [CrossRef]
- Chen, J.-H.; Kim, S.-H.; Fan, P.-W.; Liu, C.-Y.; Hsieh, C.-H.; Fang, K. The aqueous extract of Chinese medicinal herb Brucea javanica suppresses the growth of human liver cancer and the derived stem-like cells by apoptosis. Drug Des. Devel. Ther. 2016, 10, 2003–2013. [Google Scholar]
- Chen, C.; Wang, B. Brucea javanica oil emulsion alleviates cachexia induced by Lewis lung cancer cells in mice. J. Drug Target. 2018, 26, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Cao, Y.; Liu, H.-Y.; Fu, Z.-D.; Han, R. Experimental studies on the apoptosis of HL-60 cells induced by Brucea javanica oil emulsion. Zhongguo Zhongyao Zazhi 2003, 28, 759–762. [Google Scholar] [PubMed]
- Tang, T.; Meng, L.; Chen, L.; Ding, J. Reversal of multidrug resistance and inhibition of DNA topoisomerase II by emulsion of seed oil of Brucea Javanica. Chin. Pharmacol. Bull. 2001, 17, 534–539. [Google Scholar]
- Lou, G.-G.; Yao, H.-P.; Xie, L.-P. Brucea javanica oil induces apoptosis in T24 bladder cancer cells via upregulation of caspase-3, caspase-9, and inhibition of NF-κB and COX-2 expressions. Am. J. Chin. Med. 2010, 38, 613–624. [Google Scholar] [CrossRef]
- Pazouki, S.; Baty, J.; Wallace, H.; Coleman, C. Utilization of extracellular lipids by HT29/219 cancer cells in culture. Lipids 1992, 27, 827–834. [Google Scholar] [CrossRef]
- Ringbom, T.; Huss, U.; Stenholm, Å.; Flock, S.; Skattebøl, L.; Perera, P.; Bohlin, L. COX-2 inhibitory effects of naturally occurring and modified fatty acids. J. Nat. Prod. 2001, 64, 745–749. [Google Scholar] [CrossRef]
- Jiang, L.; Wang, W.; He, Q.; Wu, Y.; Lu, Z.; Sun, J.; Liu, Z.; Shao, Y.; Wang, A. Oleic acid induces apoptosis and autophagy in the treatment of tongue squamous cell carcinomas. Sci. Rep. 2017, 7, 11277. [Google Scholar] [CrossRef]
- Wang, S.; Hu, H.; Zhong, B.; Shi, D.; Qing, X.; Cheng, C.; Deng, X.; Zhang, Z.; Shao, Z. Bruceine D inhibits tumor growth and stem cell-like traits of osteosarcoma through inhibition of STAT3 signaling pathway. Cancer Med. 2019, 8, 7345–7358. [Google Scholar] [CrossRef]
- Luo, C.; Wang, Y.; Wei, C.; Chen, Y.; Ji, Z. The anti-migration and anti-invasion effects of bruceine D in human triple-negative breast cancer MDA-MB-231 cells. Exp. Ther. Med. 2020, 19, 273–279. [Google Scholar] [CrossRef]
- Lau, S.T.; Lin, Z.-X.; Liao, Y.; Zhao, M.; Cheng, C.H.; Leung, P.S. Brucein D induces apoptosis in pancreatic adenocarcinoma cell line PANC-1 through the activation of p38-mitogen activated protein kinase. Cancer Lett. 2009, 281, 42–52. [Google Scholar] [CrossRef]
- Xie, J.H.; Lai, Z.Q.; Zheng, X.H.; Xian, Y.F.; Li, Q.; Ip, S.P.; Xie, Y.L.; Chen, J.N.; Su, Z.R.; Lin, Z.X. Apoptosis induced by bruceine D in human non-small-cell lung cancer cells involves mitochondrial ROS-mediated death signaling. Int. J. Mol. Med. 2019, 44, 2015–2026. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Yuan, X.; Qu, Y.; Li, X.; Wu, G.; Li, C.; Zu, X.; Yang, N.; Ke, X.; Zhou, J. Bruceine D inhibits hepatocellular carcinoma growth by targeting β-catenin/jagged1 pathways. Cancer Lett. 2017, 403, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Groner, B.; von Manstein, V. Jak Stat signaling and cancer: Opportunities, benefits and side effects of targeted inhibition. Mol. Cell. Endocrinol. 2017, 451, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Moon, S.J.; Jeong, B.C.; Kim, H.J.; Lim, J.E.; Kim, H.-J.; Kwon, G.Y.; Jackman, J.A.; Kim, J.H. Bruceantin targets HSP90 to overcome resistance to hormone therapy in castration-resistant prostate cancer. Theranostics 2021, 11, 958–973. [Google Scholar] [CrossRef]
- Fiaschetti, G.; Grotzer, M.; Shalaby, T.; Castelletti, D.; Arcaro, A. Quassinoids: From traditional drugs to new cancer therapeutics. Curr. Med. Chem. 2011, 18, 316–328. [Google Scholar] [CrossRef]
- Yu, X.-Q.; Shang, X.-Y.; Huang, X.-X.; Yao, G.-D.; Song, S.-J. Brusatol: A potential anti-tumor quassinoid from Brucea javanica. Chin. Herb. Med. 2020. [Google Scholar] [CrossRef]
- Baumgart, D.C.; Sandborn, W.J. Inflammatory bowel disease: Clinical aspects and established and evolving therapies. Lancet 2007, 369, 1641–1657. [Google Scholar] [CrossRef]
- Huang, Y.-F.; Li, Q.-P.; Dou, Y.-X.; Wang, T.-T.; Qu, C.; Liang, J.-L.; Lin, Z.-X.; Huang, X.-Q.; Su, Z.-R.; Chen, J.-N. Therapeutic effect of Brucea javanica oil emulsion on experimental Crohn’s disease in rats: Involvement of TLR4/NF-κB signaling pathway. Biomed. Pharmacother. 2019, 114, 108766. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.-C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef]
- Hoesel, B.; Schmid, J.A. The complexity of NF-κB signaling in inflammation and cancer. Mol. Cancer 2013, 12, 86. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Gaynor, R.B. Therapeutic potential of inhibition of the NF-κB pathway in the treatment of inflammation and cancer. J. Clin. Investig. 2001, 107, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-F.; Zhou, J.-T.; Qu, C.; Dou, Y.-X.; Huang, Q.-H.; Lin, Z.-X.; Xian, Y.-F.; Xie, J.-H.; Xie, Y.-L.; Lai, X.-P. Anti-inflammatory effects of Brucea javanica oil emulsion by suppressing NF-κB activation on dextran sulfate sodium-induced ulcerative colitis in mice. J. Ethnopharmacol. 2017, 198, 389–398. [Google Scholar] [CrossRef]
- Li, Q.; Yang, L.; Fan, L.; Liang, C.; Wang, Q.; Wen, H.; Dai, J.; Li, X.; Zhang, Y. Activity of Brucea javanica oil emulsion against gastric ulcers in rodents. Asian J. Pharm. Sci. 2018, 13, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wang, T.; Dou, Y.; Huang, Y.; Qu, C.; Gao, J.; Huang, Z.; Xie, Y.; Huang, P.; Lin, Z. Brusatol ameliorates 2, 4, 6-trinitrobenzenesulfonic acid-induced experimental colitis in rats: Involvement of NF-κB pathway and NLRP3 inflammasome. Int. Immunopharmacol. 2018, 64, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Hall, I.H.; Lee, K.H.; Imakura, Y.; Okano, M.; Johnson, A. Anti-inflammatory agents III: Structure–activity relationships of brusatol and related quassinoids. J. Pharm. Sci. 1983, 72, 1282–1284. [Google Scholar] [CrossRef]
- Magdalon, J.; Vinolo, M.A.; Rodrigues, H.G.; Paschoal, V.A.; Torres, R.P.; Mancini-Filho, J.; Calder, P.C.; Hatanaka, E.; Curi, R. Oral administration of oleic or linoleic acids modulates the production of inflammatory mediators by rat macrophages. Lipids 2012, 47, 803–812. [Google Scholar] [CrossRef]
- Shamsul, K.; Tajuddin, A.; Mazina, M. Tumbuhan Ubatan Tradisional Malaysia; Institut Biosains Universiti Pertanian Malaysia: Serdang, Malaysia, 2003. [Google Scholar]
- Ablat, A.; Halabi, M.F.; Mohamad, J.; Hasnan, M.H.H.; Hazni, H.; Teh, S.-H.; Shilpi, J.A.; Mohamed, Z.; Awang, K. Antidiabetic effects of Brucea javanica seeds in type 2 diabetic rats. BMC Complement. Altern. Med. 2017, 17, 94. [Google Scholar] [CrossRef]
- Auberval, N.; Dal, S.; Bietiger, W.; Seyfritz, E.; Peluso, J.; Muller, C.; Zhao, M.; Marchioni, E.; Pinget, M.; Jeandidier, N. Oxidative stress type influences the properties of antioxidants containing polyphenols in RINm5F beta cells. Evid. Based Complement. Alternat. Med. 2015, 2015, 859048. [Google Scholar] [CrossRef]
- NoorShahida, A.; Wong, T.W.; Choo, C.Y. Hypoglycemic effect of quassinoids from Brucea javanica (L.) Merr (Simaroubaceae) seeds. J. Ethnopharmacol. 2009, 124, 586–591. [Google Scholar] [CrossRef]
- Pallas, V.; García, J.A. How do plant viruses induce disease? Interactions and interference with host components. J. Gen. Virol. 2011, 92, 2691–2705. [Google Scholar] [CrossRef]
- Shen, J.-G.; Zhang, Z.; Wu, Z.; Xie, L.; Lin, Q. Antiviral effect of Ailanthus altissima and Brucea javanica on tobacco mosaic virus. Zhongguo Zhongyao Zazhi 2007, 32, 27–29. [Google Scholar]
- Shen, J.G.; Zhang, Z.K.; Wu, Z.J.; Ouyang, M.A.; Xie, L.H.; Lin, Q.Y. Antiphytoviral activity of bruceine-D from Brucea javanica seeds. Pest Manag. Sci. 2008, 64, 191–196. [Google Scholar] [CrossRef]
- Yan, X.-H.; Chen, J.; Di, Y.-T.; Fang, X.; Dong, J.-H.; Sang, P.; Wang, Y.-H.; He, H.-P.; Zhang, Z.-K.; Hao, X.-J. Anti-tobacco mosaic virus (TMV) quassinoids from Brucea javanica (L.) Merr. J. Agric. Food Chem. 2010, 58, 1572–1577. [Google Scholar] [CrossRef] [PubMed]
- Ryu, S.M.; Kwon, J.; Seo, Y.H.; Song, E.G.; Hong, S.S.; Kim, B.S.; Hong, J.S.; Ryu, K.H.; Lee, D. Quassinoids isolated from Brucea javanica inhibit pepper mottle virus in pepper. Virus Res. 2017, 227, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Okano, M.; Fukamiya, N.; Tagahara, K.; Cosentino, M.; Lee, T.T.-Y.; Morris-Natschke, S.; Lee, K.-H. Anti-HIV activity of quassinoids. Bioorg. Med. Chem. Lett. 1996, 6, 701–706. [Google Scholar] [CrossRef]
- Apers, S.; Cimanga, K.; Berghe, D.V.; Van Meenen, E.; Longanga, A.O.; Foriers, A.; Vlietinck, A.; Pieters, L. Antiviral activity of simalikalactone D, a quassinoid from Quassia africana. Planta Med. 2002, 68, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Kreilgaard, M. Influence of microemulsions on cutaneous drug delivery. Adv. Drug Deliv. Rev. 2002, 54, S77–S98. [Google Scholar] [CrossRef]
- Lawrence, M.J.; Rees, G.D. Microemulsion-based media as novel drug delivery systems. Adv. Drug Deliv. Rev. 2000, 45, 89–121. [Google Scholar] [CrossRef]
- Yang, F.; Yu, X.H.; Qiao, F.; Cheng, L.H.; Chen, G.; Long, X.; Wang, X.R.; Li, X.L.; Liang, R.C.; Chen, Y.Z. Formulation and characterization of Brucea javanica oil microemulsion for improving safety. Drug Dev. Ind. Pharm. 2014, 40, 266–277. [Google Scholar] [CrossRef]
- Wang, Y.; Li, Y.; Wu, J.; Shen, Q. Characterisation and evaluation of self-microemulsifying drug delivery system of Brucea javanica oil. Micro Nano Lett. 2012, 7, 256–261. [Google Scholar] [CrossRef]
- Shao, A.; Chen, G.; Jiang, N.; Li, Y.; Zhang, X.; Wen, L.; Yang, F.; Wei, S. Development and evaluation of self-microemulsifying liquid and granule formulations of Brucea javanica oil. Arch. Pharmacal Res. 2013, 36, 993–1003. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Su, H.; Wen, L.; Shao, A.; Yang, F.; Chen, G. Enhanced anticancer effect of Brucea javanica oil by solidified self-microemulsifying drug delivery system. J. Drug Deliv. Sci. Technol. 2018, 48, 266–273. [Google Scholar] [CrossRef]
- Liu, T.-T.; Mu, L.-Q.; Dai, W.; Wang, C.-B.; Liu, X.-Y.; Xiang, D.-X. Preparation, characterization, and evaluation of antitumor effect of Brucea javanica oil cationic nanoemulsions. Int. J. Nanomed. 2016, 11, 2515–2529. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Tan, L.; Xie, J.; Lai, Z.; Huang, Y.; Qu, C.; Luo, D.; Lin, Z.; Huang, P.; Su, Z. Characterization of brusatol self-microemulsifying drug delivery system and its therapeutic effect against dextran sodium sulfate-induced ulcerative colitis in mice. Drug Deliv. 2017, 24, 1667–1679. [Google Scholar] [CrossRef] [PubMed]
- Dou, Y.-X.; Zhou, J.-T.; Wang, T.-T.; Huang, Y.-F.; Chen, V.P.; Xie, Y.-L.; Lin, Z.-X.; Gao, J.-S.; Su, Z.-R.; Zeng, H.-F. Self-nanoemulsifying drug delivery system of bruceine D: A new approach for anti-ulcerative colitis. Int. J. Nanomed. 2018, 13, 5887–5907. [Google Scholar] [CrossRef] [PubMed]
- Allen, T.M.; Hansen, C.B.; de Menezes, D.E.L. Pharmacokinetics of long-circulating liposomes. Adv. Drug Deliv. Rev. 1995, 16, 267–284. [Google Scholar] [CrossRef]
- Gao, W.; Hu, C.-M.J.; Fang, R.H.; Zhang, L. Liposome-like nanostructures for drug delivery. J. Mater. Chem. B 2013, 1, 6569–6585. [Google Scholar] [CrossRef]
- Ye, H.; Liu, X.; Sun, J.; Zhu, S.; Zhu, Y.; Chang, S. Enhanced therapeutic efficacy of LHRHa-targeted Brucea javanica oil liposomes for ovarian cancer. BMC Cancer 2016, 16, 831. [Google Scholar] [CrossRef]
- Yue, Y.; Yang, Y.; Shi, L.; Wang, Z. Suppression of human hepatocellular cancer cell proliferation by Brucea javanica oil-loaded liposomes via induction of apoptosis. Arch. Med. Sci. 2015, 11, 856–862. [Google Scholar] [CrossRef]
- Huang, Z.-R.; Hu, S.-C.; Yang, Y.-L.; Fang, J.-Y. Development and evaluation of lipid nanoparticles for camptothecin delivery: A comparison of solid lipid nanoparticles, nanostructured lipid carriers, and lipid emulsion. Acta Pharmacol. Sin. 2008, 29, 1094–1102. [Google Scholar] [CrossRef]
- Lv, W.; Zhao, S.; Yu, H.; Li, N.; Garamus, V.M.; Chen, Y.; Yin, P.; Zhang, R.; Gong, Y.; Zou, A. Brucea javanica oil-loaded nanostructure lipid carriers (BJO NLCs): Preparation, characterization and in vitro evaluation. Colloids Surf. Physicochem. Eng. Aspects 2016, 504, 312–319. [Google Scholar] [CrossRef]
- Müller, R.; Radtke, M.; Wissing, S. Nanostructured lipid matrices for improved microencapsulation of drugs. Int. J. Pharm. 2002, 242, 121–128. [Google Scholar] [CrossRef]
- Jia, L.-J.; Zhang, D.-R.; Li, Z.-Y.; Feng, F.-F.; Wang, Y.-C.; Dai, W.-T.; Duan, C.-X.; Zhang, Q. Preparation and characterization of silybin-loaded nanostructured lipid carriers. Drug Deliv. 2010, 17, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Bondì, M.L.; Craparo, E.F.; Giammona, G.; Cervello, M.; Azzolina, A.; Diana, P.; Martorana, A.; Cirrincione, G. Nanostructured lipid carriers-containing anticancer compounds: Preparation, characterization, and cytotoxicity studies. Drug Deliv. 2007, 14, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Angelova, A.; Hu, F.; Garamus, V.M.; Peng, C.; Li, N.; Liu, J.; Liu, D.; Zou, A. pH responsiveness of hexosomes and cubosomes for combined delivery of Brucea javanica Oil and Doxorubicin. Langmuir 2019, 35, 14532–14542. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Angelova, A.; Angelov, B.; Drechsler, M.; Garamus, V.M.; Willumeit-Roemer, R.; Zou, A. Sterically stabilized spongosomes for multidrug delivery of anticancer nanomedicines. J. Mater. Chem. B 2015, 3, 7734–7744. [Google Scholar] [CrossRef] [PubMed]
- Zou, A.; Li, Y.; Chen, Y.; Angelova, A.; Garamus, V.M.; Li, N.; Drechsler, M.; Angelov, B.; Gong, Y. Self-assembled stable sponge-type nanocarries for Brucea javanica oil delivery. Colloids Surf. B Biointerfaces 2017, 153, 310–319. [Google Scholar] [CrossRef] [PubMed]
- Baek, J.; Jeong, H.; Ham, Y.; Jo, Y.H.; Choi, M.; Kang, M.; Son, B.; Choi, S.; Ryu, H.W.; Kim, J. Improvement of spinal muscular atrophy via correction of the SMN2 splicing defect by Brucea javanica (L.) Merr. extract and bruceine D. Phytomedicine 2019, 65, 153089. [Google Scholar] [CrossRef] [PubMed]
- Lahrita, L.; Moriai, K.; Iwata, R.; Itoh, K.; Kato, E. Quassinoids in Brucea javanica are potent stimulators of lipolysis in adipocytes. Fitoterapia 2019, 137, 104250. [Google Scholar] [CrossRef]
- Mao, G.; Tian, Y.; Sun, Z.; Ou, J.; Xu, H. Bruceine D isolated from Brucea javanica (L.) Merr. as a systemic feeding deterrent for three major lepidopteran pests. J. Agric. Food Chem. 2019, 67, 4232–4239. [Google Scholar] [CrossRef]
- Nakao, R.; Mizukami, C.; Kawamura, Y.; Bawm, S.; Yamasaki, M.; Maede, Y.; Matsuura, H.; Nabeta, K.; Nonaka, N.; Oku, Y. Evaluation of efficacy of bruceine A, a natural quassinoid compound extracted from a medicinal plant, Brucea javanica, for canine babesiosis. J. Vet. Med. Sci. 2009, 71, 33–41. [Google Scholar] [CrossRef] [PubMed]
| Nanomedicine Class | Development Status | Main Effects |
|---|---|---|
| Nanoemulsions | In vivo treatment efficacy in mouse and rat models |
|
| Liposomes | In vivo treatment efficacy in mouse model |
|
| Nanostructured Lipid Carriers | In vitro drug release and cancer cell inhibition |
|
| Spongosomes | In vitro drug release and cancer cell inhibition |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoon, B.K.; Lim, Z.Y.; Jeon, W.-Y.; Cho, N.-J.; Kim, J.H.; Jackman, J.A. Medicinal Activities and Nanomedicine Delivery Strategies for Brucea javanica Oil and Its Molecular Components. Molecules 2020, 25, 5414. https://doi.org/10.3390/molecules25225414
Yoon BK, Lim ZY, Jeon W-Y, Cho N-J, Kim JH, Jackman JA. Medicinal Activities and Nanomedicine Delivery Strategies for Brucea javanica Oil and Its Molecular Components. Molecules. 2020; 25(22):5414. https://doi.org/10.3390/molecules25225414
Chicago/Turabian StyleYoon, Bo Kyeong, Zheng Yi Lim, Won-Yong Jeon, Nam-Joon Cho, Jeong Hoon Kim, and Joshua A. Jackman. 2020. "Medicinal Activities and Nanomedicine Delivery Strategies for Brucea javanica Oil and Its Molecular Components" Molecules 25, no. 22: 5414. https://doi.org/10.3390/molecules25225414
APA StyleYoon, B. K., Lim, Z. Y., Jeon, W.-Y., Cho, N.-J., Kim, J. H., & Jackman, J. A. (2020). Medicinal Activities and Nanomedicine Delivery Strategies for Brucea javanica Oil and Its Molecular Components. Molecules, 25(22), 5414. https://doi.org/10.3390/molecules25225414







