Role of Melatonin in Plant Tolerance to Soil Stressors: Salinity, pH and Heavy Metals
Abstract
1. Introduction
2. Salinity Stress Impacts and Tolerance in Plants
2.1. Plant Responses and Tolerance to Salinity Stress
2.2. Melatonin and Salinity Stress
3. pH Stress
3.1. Impact of pH Stress and Tolerance in Plants
3.2. Melatonin and pH Stress
4. Heavy Metal Stress
4.1. Heavy Metal Stress: Impact and Tolerance in Plant
4.2. Melatonin and Heavy Metal Stress
5. Summary and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dubbels, R.; Reiter, R.J.; Klenke, E.; Goebel, A.; Schnakenberg, E.; Ehlers, C.; Schiwara, H.W.; Schloot, W. Melatonin in edible plants identified by radioimmunoassay and by high performance liquid chromatography-mass spectrometry. J. Pineal Res. 1995, 18, 28–31. [Google Scholar] [CrossRef] [PubMed]
- Hattori, A.; Migitaka, H.; Iigo, M.; Itoh, M.; Yamamoto, K.; Ohtani-Kaneko, R.; Hara, M.; Suzuki, T.; Reiter, R.J. Identification of melatonin in plants and its effects on plasma melatonin levels and binding to melatonin receptors in vertebrates. Biochem. Mol. Biol. Int. 1995, 35, 627–634. [Google Scholar]
- Arnao, M.B.; Hernández-Ruiz, J. Melatonin: A new plant hormone and/or a plant master regulator? Trends Plant Sci. 2019, 24, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Galano, A. Melatonin: Exceeding expectations. Physiology 2014, 29, 325–333. [Google Scholar] [CrossRef]
- Malpaux, B.; Migaud, M.; Tricoire, H.; Chemineau, P. Biology of mammalian photoperiodism and the critical role of the pineal gland and melatonin. J. Biol. Rhythms 2001, 16, 336–347. [Google Scholar] [CrossRef]
- Zhao, D.; Yu, Y.; Shen, Y.; Liu, Q.; Zhao, Z.; Sharma, R.; Reiter, R. Melatonin synthesis and function: Evolutionary history in animals and plants. Front. Endocrinol. 2019, 10, 249. [Google Scholar] [CrossRef]
- Weaver, D.R. Melatonin and circadian rhythmicity in vertebrates: Physiological roles and pharmacological effects. In Neurobiology of Sleep and Circadian Rhythms; FW Turek, P.Z., Ed.; Dekker: New York, NY, USA, 1999; pp. 197–262. [Google Scholar]
- Dubocovich, M.L.; Masana, M.I.; Iacob, S.; Sauri, D.M. Melatonin receptor antagonists that differentiate between the human mel1a and mel1b recombinant subtypes are used to assess the pharmacological profile of the rabbit retina Ml1 presynaptic heteroreceptor. Naunyn Schmiedebergs Arch. Pharmacol. 1997, 355, 365–375. [Google Scholar] [CrossRef]
- Ting, K.; Blaylock, N.; Sugden, D.; Delagrange, P.; Scalbert, E.; Wilson, V. Molecular and pharmacological evidence for mt1 melatonin receptor subtype in the tail artery of juvenile wistar rats. Br. J. Pharmacol. 1999, 127, 987–995. [Google Scholar] [CrossRef]
- Dhole, A.M.; Shelat, H.N. Phytomelatonin: A plant hormone for management of stress. J. Anal. Pharm. Res. 2018, 7, 188–190. [Google Scholar] [CrossRef]
- Moustafa-Farag, M.; Almoneafy, A.; Mahmoud, A.; Elkelish, A.; Arnao, M.B.; Li, L.; Ai, S. Melatonin and its protective role against biotic stress impacts on plants. Biomolecules 2019, 10, 54. [Google Scholar] [CrossRef]
- Wang, Y.; Reiter, R.J.; Chan, Z. Phytomelatonin: A universal abiotic stress regulator. J. Exp. Bot. 2018, 69, 963–974. [Google Scholar] [CrossRef] [PubMed]
- Arnao, M.B.; Hernández-Ruiz, J. The multi-regulatory properties of melatonin in plants. In Neurotransmitters in Plants; Ramakrishna, A., Roshchina, V., Eds.; CRC Press: Boca Raton, FL, USA; New York, NY, USA, 2018; pp. 71–101. [Google Scholar]
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef] [PubMed]
- Elkelish, A.A.; Soliman, M.H.; Alhaithloul, H.A.; El-Esawi, M.A. Selenium protects wheat seedlings against salt stress-mediated oxidative damage by up-regulating antioxidants and osmolytes metabolism. Plant Physiol. Biochem. 2019, 137, 144–153. [Google Scholar] [CrossRef]
- Soliman, M.H.; Alayafi, A.A.; El Kelish, A.A.; Abu-Elsaoud, A.M. Acetylsalicylic acid enhance tolerance of phaseolus vulgaris l. to chilling stress, improving photosynthesis, antioxidants and expression of cold stress responsive genes. Bot. Stud. 2018, 59, 6. [Google Scholar] [CrossRef]
- Elkelish, A.A.; Alhaithloul, H.A.S.; Qari, S.H.; Soliman, M.H.; Hasanuzzaman, M. Pretreatment with Trichoderma harzianum alleviates waterlogging-induced growth alterations in tomato seedlings by modulating physiological, biochemical, and molecular mechanisms. J. Environ. Exp. Bot. 2020, 171, 103946. [Google Scholar] [CrossRef]
- Soliman, M.; Alhaithloul, H.A.; Hakeem, K.R.; Alharbi, B.M.; El-Esawi, M.; Elkelish, A. Exogenous nitric oxide mitigates nickel-induced oxidative damage in eggplant by upregulating antioxidants, osmolyte metabolism, and glyoxalase systems. Plants 2019, 8, 562. [Google Scholar] [CrossRef]
- Sreenivasulu, N.; Grimm, B.; Wobus, U.; Weschke, W. Differential response of antioxidant compounds to salinity stress in salt-tolerant and salt-sensitive seedlings of foxtail millet (SetariaItalica). Physiol. Plant. 2000, 109, 435–442. [Google Scholar] [CrossRef]
- El-Esawi, M.A.; Elkelish, A.; Elansary, H.O.; Ali, H.M.; Elshikh, M.; Witczak, J.; Ahmad, M. Genetic transformation and hairy root induction enhance the antioxidant potential of Lactuca serriola L. Oxid. Med. Cell. Longev. 2017, 2017, 5604746. [Google Scholar] [CrossRef]
- Saleh, A.A.; Abdel-Kader, D.Z.; El Elish, A.M. Role of heat shock and salicylic acid in antioxidant homeostasis in mungbean (Vigna radiata L.) plant subjected to heat stress. Am. J. Plant Physiol. 2007, 2, 344–355. [Google Scholar] [CrossRef]
- Farag, M.; Najeeb, U.; Yang, J.; Hu, Z.; Fang, Z.M. Nitric oxide protects carbon assimilation process of watermelon from boron-induced oxidative injury. Plant Physiol. Biochem. 2017, 111, 166–173. [Google Scholar] [CrossRef]
- Mosaad, I.S.; Serag, A.H.; Moustafa-Farag, M.; Seadh, A.K. Effect of exogenous proline application on maize yield and the optimum rate of mineral nitrogen under salinity stress. J. Plant Nutr. 2020, 43, 354–370. [Google Scholar] [CrossRef]
- Moustafa-Farag, M.; Bingsheng, F.; Malangisha Guy, K.; Hu, Z.; Yang, J.; Zhang, M. Activated antioxidant enzymes-reduced malondialdehyde concentration, and improved mineral uptake-promoted watermelon seedlings growth under boron deficiency. J. Plant Nutr. 2016, 39, 1989–2001. [Google Scholar] [CrossRef]
- Kaveh, H.; Nemati, H.; Farsi, M.; Jartoodeh, S.V. How salinity affect germination and emergence of tomato lines. J. Biol. Environ. Sci. 2011, 5, 159–163. [Google Scholar]
- Zörb, C.; Geilfus, C.M.; Dietz, K.J. Salinity and crop yield. Plant Biol. 2019, 21, 31–38. [Google Scholar] [CrossRef]
- Grattan, S.; Grieve, C. Salinity–mineral nutrient relations in horticultural crops. Sci. Hortic. 1998, 78, 127–157. [Google Scholar] [CrossRef]
- Dawood, M.G.; El-Awadi, M.E. Alleviation of salinity stress on Vicia faba L. plants via seed priming with melatonin. Acta Biol. Colomb. 2015, 20, 223–235. [Google Scholar] [CrossRef]
- Ahanger, M.A.; Akram, N.A.; Ashraf, M.; Alyemeni, M.N.; Wijaya, L.; Ahmad, P. Plant Responses to Environmental Stresses—from Gene to Biotechnology. AoB Plants 2017, 9, plx025. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Nahar, K.; Alam, M.M.; Bhowmik, P.C.; Hossain, M.A.; Rahman, M.M.; Prasad, M.N.V.; Ozturk, M.; Fujita, M. Potential use of halophytes to remediate saline soils. BioMed Res. Int. 2014, 2014, 589341. [Google Scholar] [CrossRef]
- Machado, R.; Serralheiro, R. Soil salinity: Effect on vegetable crop growth. management practices to prevent and mitigate soil salinization. Horticulturae 2017, 3, 30. [Google Scholar] [CrossRef]
- Ahmed, D.; Fatima, K.; Saeed, R. Analysis of phenolic and flavonoid contents, and the anti-oxidative potential and lipid peroxidation inhibitory activity of methanolic extract of Carissa opaca roots and its fractions in different solvents. Antioxidants 2014, 3, 671–683. [Google Scholar] [CrossRef]
- Li, C.; Wang, P.; Wei, Z.; Liang, D.; Liu, C.; Yin, L.; Jia, D.; Fu, M.; Ma, F. The mitigation effects of exogenous melatonin on salinity-induced stress in Malus hupehensis. J. Pineal Res. 2012, 53, 298–306. [Google Scholar] [CrossRef]
- Martinez, V.; Nieves-Cordones, M.; Lopez-Delacalle, M.; Rodenas, R.; Mestre, T.C.; Garcia-Sanchez, F.; Rubio, F.; Nortes, P.A.; Mittler, R.; Rivero, R.M. Tolerance to stress combination in tomato plants: New insights in the protective role of melatonin. Molecules 2018, 23, 535. [Google Scholar] [CrossRef]
- Zhou, X.; Zhao, H.; Cao, K.; Hu, L.; Du, T.; Baluška, F.; Zou, Z. Beneficial roles of melatonin on redox regulation of photosynthetic electron transport and synthesis of D1 protein in tomato seedlings under salt stress. Front. Plant Sci. 2016, 7, 1823. [Google Scholar] [CrossRef]
- Wang, L.Y.; Liu, J.L.; Wang, W.X.; Sun, Y. Exogenous melatonin improves growth and photosynthetic capacity of cucumber under salinity-induced stress. Photosynthetica 2016, 54, 19–27. [Google Scholar] [CrossRef]
- Li, H.; Chang, J.; Chen, H.; Wang, Z.; Gu, X.; Wei, C.; Zhang, Y.; Ma, J.; Yang, J.; Zhang, X. Exogenous melatonin confers salt stress tolerance to watermelon by improving photosynthesis and redox homeostasis. Front. Plant Sci. 2017, 8, 295. [Google Scholar] [CrossRef]
- Liu, Z.; Cai, J.S.; Li, J.J.; Lu, G.Y.; Li, C.S.; Fu, G.P.; Zhang, X.K.; Liu, Q.Y.; Zou, X.L.; Cheng, Y. Exogenous application of a low concentration of melatonin enhances salt tolerance in rapeseed (Brassica napus L.) Seedlings. J. Integr. Agric. 2018, 17, 328–335. [Google Scholar]
- Zhang, N.; Zhang, H.J.; Sun, Q.-Q.; Cao, Y.Y.; Li, X.; Zhao, B.; Wu, P.; Guo, Y.D. Proteomic analysis reveals a role of melatonin in promoting cucumber seed germination under high salinity by regulating energy production. Sci. Rep. 2017, 7, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.; Zheng, G.; Li, W.; Wang, Y.; Hu, B.; Wang, H.; Wu, H.; Qian, Y.; Zhu, X.G.; Tan, D.X.; et al. Melatonin delays leaf senescence and enhances salt stress tolerance in rice. J. Pineal Res. 2015, 59, 91–101. [Google Scholar] [CrossRef]
- Li, X.; Yu, B.; Cui, Y.; Yin, Y. Melatonin application confers enhanced salt tolerance by regulating Na+ and Cl− accumulation in rice. Plant Growth Regul. 2017, 83, 441–454. [Google Scholar] [CrossRef]
- Jiang, C.; Cui, Q.; Feng, K.; Xu, D.; Li, C.; Zheng, Q. Melatonin improves antioxidant capacity and ion homeostasis and enhances salt tolerance in maize seedlings. Acta Physiol. Plant. 2016, 38, 1–9. [Google Scholar] [CrossRef]
- Chen, Y.E.; Mao, J.J.; Sun, L.Q.; Huang, B.; Ding, C.B.; Gu, Y.; Liao, J.Q.; Hu, C.; Zhang, Z.W.; Yuan, S.; et al. Exogenous melatonin enhances salt stress tolerance in maize seedlings by improving antioxidant and photosynthetic capacity. Physiol. Plant. 2018, 164, 349–363. [Google Scholar] [CrossRef]
- Wei, W.; Li, Q.T.; Chu, Y.N.; Reiter, R.J.; Yu, X.M.; Zhu, D.H.; Zhang, W.K.; Ma, B.; Lin, Q.; Zhang, J.S.; et al. Melatonin enhances plant growth and abiotic stress tolerance in soybean plants. J. Exp. Bot. 2015, 66, 695–707. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.J.; Zhang, N.; Yang, R.C.; Wang, L.; Sun, Q.Q.; Li, D.B.; Cao, Y.Y.; Weeda, S.; Zhao, B.; Ren, S.; et al. Melatonin promotes seed germination under high salinity by regulating antioxidant systems, ABA and GA4 interaction in cucumber (Cucumis sativus L.). J. Pineal Res. 2014, 57, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Szafrańska, K.; Reiter, R.J.; Posmyk, M.M. Melatonin application to Pisum sativum L. seeds positively influences the function of the photosynthetic apparatus in growing seedlings during paraquat-induced oxidative stress. Front. Plant Sci. 2016, 7, 1663. [Google Scholar] [CrossRef]
- Ke, Q.; Ye, J.; Wang, B.; Ren, J.; Yin, L.; Deng, X.; Wang, S. melatonin mitigates salt stress in wheat seedlings by modulating polyamine metabolism. Int. J. Mol. Sci. 2018, 9, 914. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Xie, Y.; Gu, Q.; Zhao, G.; Zhang, Y.; Cui, W.; Xu, S.; Wang, R.; Shen, W. The atrbohf-dependent regulation of ros signaling is required for melatonin-induced salinity tolerance in Arabidopsis. Free Radic. Biol. Med. 2017, 108, 465–477. [Google Scholar] [CrossRef]
- Rangani, J.; Parida, A.K.; Panda, A.; Kumari, A. Coordinated changes in antioxidative enzymes protect the photosynthetic machinery from salinity induced oxidative damage and confer salt tolerance in an extreme halophyte Salvadora persica L. Front. Plant Sci. 2016, 7, 50. [Google Scholar] [CrossRef]
- Choi, G.H.; Back, K.J.B. Suppression of melatonin 2-Hydroxylase increases melatonin production leading to the enhanced abiotic stress tolerance against cadmium, senescence, salt, and tunicamycin in rice plants. Biomolecules 2019, 9, 589. [Google Scholar] [CrossRef]
- Hwang, O.J.; Back, K. Melatonin deficiency confers tolerance to multiple abiotic stresses in rice via decreased brassinosteroid levels. Int. J. Mol. Sci. 2019, 20, 5173. [Google Scholar] [CrossRef]
- Haydari, M.; Maresca, V.; Rigano, D.; Taleei, A.; Shahnejat-Bushehri, A.A.; Hadian, J.; Sorbo, S.; Guida, M.; Manna, C.; Piscopo, M. Salicylic acid and melatonin alleviate the effects of heat stress on essential oil composition and antioxidant enzyme activity in Mentha× piperita and Mentha arvensis L. Antioxidants 2019, 8, 547. [Google Scholar] [CrossRef]
- Yu, Y.; Wang, A.; Li, X.; Kou, M.; Wang, W.; Chen, X.; Xu, T.; Zhu, M.; Ma, D.; Li, Z.; et al. Melatonin-stimulated triacylglycerol breakdown and energy turnover under salinity stress contributes to the maintenance of plasma membrane H+ atpase activity and K+/Na+ homeostasis in sweet potato. Front. Plant Sci. 2018, 9, 256. [Google Scholar] [CrossRef]
- Wang, P.; Yin, L.; Liang, D.; Li, C.; Ma, F.; Yue, Z. Delayed senescence of apple leaves by exogenous melatonin treatment: Toward regulating the ascorbate–glutathione cycle. J. Pineal Res. 2012, 53, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Zhao, Y.; Yu, X.; Kiprotich, F.; Han, H.; Guan, R.; Wang, R.; Shen, W. Nitric oxide is required for melatonin-enhanced tolerance against salinity stress in rapeseed (Brassica Napus L.) Seedlings. Int. J. Mol. Sci. 2018, 19, 1912. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Long, W.; Zeng, L.; Ding, X.; Cheng, Y.; Zhang, X.; Zou, X. Melatonin-induced transcriptome variation of rapeseed seedlings under salt stress. Int. J. Mol. Sci. 2019, 20, 5355. [Google Scholar] [CrossRef]
- Felle, H.H.; Waller, F.; Molitor, A.; Kogel, K.H. The mycorrhiza fungus Piriformospora indica induces fast root-surface ph signaling and primes systemic alkalinization of the leaf apoplast upon powdery mildew infection. Mol. Plant Microb. Interact. 2009, 22, 1179–1185. [Google Scholar] [CrossRef]
- Felle, H.H.; Herrmann, A.; Schäfer, P.; Hückelhoven, R.; Kogel, K.H. Interactive signal transfer between host and pathogen during successful infection of barley leaves by Blumeria graminis and Bipolaris sorokiniana. J. Plant Physiol. 2008, 165, 52–59. [Google Scholar] [CrossRef]
- Lager, I.; Andréasson, O.; Dunbar, T.L.; Andreasson, E.; Escobar, M.A.; Rasmusson, A.G. Changes in external pH rapidly alter plant gene expression and modulate auxin and elicitor responses. Plant. Cell. Environ. 2010, 33, 1513–1528. [Google Scholar] [CrossRef]
- Läuchli, A.; Grattan, S.R. Soil pH extremes. In Plant Stress Physiology; Shabala, S., Ed.; CABI: Wallingford, CT, USA, 2012; pp. 194–209. [Google Scholar]
- Balks, M.R.; Zabowski, D. Soils in extreme environments. In Celebrating Soil: Discovering Soils and Landscapes; Balks, M.R., Zabowski, D., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 119–142. [Google Scholar]
- Gong, B.; Li, Y.; Li, X.; Bloszies, S.; Wen, D.; Sun, S.; Wei, M.; Yang, F.; Shi, Q.; Wang, X. Sodic alkaline stress mitigation by interaction of nitric oxide and polyamines involves antioxidants and physiological strategies in Solanum lycopersicum. Free Radic. Biol. Med. 2014, 71, 36–48. [Google Scholar] [CrossRef]
- Liu, N.; Jin, Z.; Wang, X.; Wang, S.; Gong, B.; Wen, D.; Wei, M.; Shi, Q. Sodic alkaline stress mitigation with exogenous melatonin involves reactive oxygen metabolism and ion homeostasis in tomato. Sci. Hortic. 2015, 181, 18–25. [Google Scholar] [CrossRef]
- Choudhury, F.K.; Rivero, R.M.; Blumwald, E.; Mittler, R. Reactive oxygen species, abiotic stress and stress combination. Plant J. 2017, 90, 856–867. [Google Scholar] [CrossRef]
- Suzuki, N.; Koussevitzky, S.; Mittler, R.O.N.; Miller, G.A.D. Ros and redox signalling in the response of plants to abiotic stress: Ros and redox signalling in plants. Plant Cell Environ. 2012, 35, 259–270. [Google Scholar] [CrossRef]
- Long, A.; Zhang, J.; Yang, L.T.; Ye, X.; Lai, N.W.; Tan, L.L.; Lin, D.; Chen, L.S. Effects of low pH on photosynthesis, related physiological parameters, and nutrient profiles of Citrus. Front. Plant Sci. 2017, 8, 185. [Google Scholar] [CrossRef] [PubMed]
- Goulding, K.W.T.; Varennes, A. Soil acidification and the importance of liming agricultural soils with particular reference to the United Kingdom. Soil Use Manag. 2016, 32, 390–399. [Google Scholar] [CrossRef]
- Yang, M.; Tan, L.; Xu, Y.; Zhao, Y.; Cheng, F.; Ye, S.; Jiang, W. Effect of low pH and aluminum toxicity on the photosynthetic characteristics of different fast-growing eucalyptus vegetatively propagated clones. PLoS ONE 2015, 10, e0130963. [Google Scholar] [CrossRef]
- Pagani, A.; Mallarino, A.P. Soil pH and crop grain yield as affected by the source and rate of lime. Soil Sci. Soc. Am. J. 2012, 76, 1877–1886. [Google Scholar] [CrossRef]
- Fageria, N.K.; Nascente, A.S. Management of soil acidity of south american soils for sustainable crop production. Adv. Agron. 2014, 128, 221–275. [Google Scholar]
- Fageria, N.K.; Castro, E.M.; Baligar, V.C. Response of upland rice genotypes to soil acidity. In The Red Soils of China: Their Nature, Management and Utilization; Wilson, M.J., He, Z., Yang, X., Eds.; Springer: Dordrecht, The Netherlands, 2004; pp. 219–237. [Google Scholar]
- Fageria, N.K.; Wright, R.J.; Baligar, V.C.; De Sousa, C.M.R. Characterization of physical and chemical properties of varzea soils of Goias State of Brazil. Commun. Soil Sci. Plant Anal. 1991, 22, 1631–1646. [Google Scholar] [CrossRef][Green Version]
- Sumner, D.A. Exotic Pests and Public Policy for Biosecurity: An Introduction and Overview. In Exotic Pests and Diseases, Sumner, D.A., Ed.; Iowa State University Press: Iowa, IA, USA, 2003; pp. 1–6. [Google Scholar]
- Fageria, N.; Baligar, V.; Clark, R. Physiology of Crop Production, 1st ed.; CRC Press: New York, NY, USA, 2006; p. 356. [Google Scholar]
- Fageria, N.K.; Baligar, V.C.; Li, Y.C. Differential soil acidity tolerance of tropical legume cover crops. Commun. Soil Sci. Plant Anal. 2009, 40, 1148–1160. [Google Scholar] [CrossRef]
- Foy, C.D. Soil chemical factors limiting plant root growth. In Limitations to Plant Root Growth; Hatfield, J.L., Stewart, B.A., Eds.; Springer: New York, NY, USA, 1992; pp. 97–149. [Google Scholar]
- Sánchez, P.A.; Salinas, J.G.; Salinas, J.G. Low-input technology for managing oxisols and ultisols in tropical America. Adv. Agron. 1981, 34, 279–406. [Google Scholar]
- Barthod, J.; Rumpel, C.; Calabi-Floody, M.; Mora, M.L.; Bolan, N.S.; Dignac, M.F. Adding worms during composting of organic waste with red mud and fly ash reduces CO2 emissions and increases plant available nutrient contents. J. Environ. Manag. 2018, 222, 207–215. [Google Scholar] [CrossRef]
- Kim, K.; Kim, S.H.; Park, S.M.; Kim, J.; Choi, M. Processes controlling the variations of ph, alkalinity, and CO2 partial pressure in the porewater of coal ash disposal site. J. Hazard. Mater. 2010, 181, 74–81. [Google Scholar] [CrossRef]
- Batra, L. Dehydrogenase activity of normal, saline and alkali soils under different agricultural management systems. J. Indian Soc. Soil Sci. 2004, 52, 160–163. [Google Scholar]
- Sharma, B.R.; Minhas, P.S. Strategies for managing saline/alkali waters for sustainable agricultural production in South Asia. Agric. Water Manag. 2005, 78, 136–151. [Google Scholar] [CrossRef]
- Arshad, M.A.; Coen, G.M. Characterization of soil quality: Physical and chemical criteria. Am. J. Alternat. Agric. 1992, 7, 25–31. [Google Scholar] [CrossRef]
- Pathak, H.; Rao, D.L.N. Carbon and nitrogen mineralization from added organic matter in saline and alkali soils. Soil Biol. Biochem. 1998, 30, 695–702. [Google Scholar] [CrossRef]
- Murtaza, G.; Ghafoor, A.; Qadir, M. Irrigation and soil management strategies for using saline-sodic water in a cotton–wheat rotation. Agric. Water Manag. 2006, 81, 98–114. [Google Scholar] [CrossRef]
- Qadir, M. Crop and irrigation management strategies for saline-sodic soils and waters aimed at environmentally sustainable agriculture. Sci. Total Environ. 2004, 1, 1–19. [Google Scholar] [CrossRef]
- Qadir, M.; Oster, J.D.; Schubert, S.; Noble, A.D.; Sahrawat, K.L. Phytoremediation of Sodic and Saline-Sodic Soils. In Advances in Agronomy; Sparks, D.L., Ed.; Elsevier Science & Technology: San Diego, CA, USA, 2007; pp. 197–247. [Google Scholar]
- Wu, Y.; Li, Y.; Zheng, C.; Zhang, Y.; Sun, Z. Organic amendment application influence soil organism abundance in saline alkali soil. Eur. J. Soil Biol. 2013, 54, 32–40. [Google Scholar] [CrossRef]
- Adcock, D.; McNeill, A.M.; McDonald, G.K.; Armstrong, R.D. Subsoil constraints to crop production on neutral and alkaline soils in south-eastern australia: A review of current knowledge and management strategies. Aust. J. Exp. Agric. 2007, 47, 1245–1261. [Google Scholar] [CrossRef]
- Poeggeler, B.; Hardeland, R. Detection and quantification of melatonin in a dinoflagellate, gonyaulaxpolyedra: Solutions to the problem of methoxyindole destruction in non-vertebrate material. J. Pineal Res. 1994, 17, 1–10. [Google Scholar] [CrossRef]
- Arnao, M.B. Phytomelatonin: Discovery, content, and role in plants. Adv. Bot. 2014, 2014, 1–11. [Google Scholar] [CrossRef]
- Arnao, M.B.; Hernández-Ruiz, J. Growth conditions determine different melatonin levels in Lupinus albus L. J. Pineal Res. 2013, 55, 149–155. [Google Scholar] [CrossRef]
- Debnath, B.; Hussain, M.; Irshad, M.; Mitra, S.; Li, M.; Liu, S.; Qiu, D. Exogenous melatonin mitigates acid rain stress to tomato plants through modulation of leaf ultrastructure, photosynthesis and antioxidant potential. Molecules 2018, 23, 388. [Google Scholar] [CrossRef]
- Xu, J.; Zhang, M.; Liu, G.; Yang, X.; Hou, X. Comparative transcriptome profiling of chilling stress responsiveness in grafted watermelon seedlings. Plant Physiol. Biochem. 2016, 109, 561–570. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Gong, B.; Jin, Z.; Wang, X.; Wei, M.; Yang, F.; Li, Y.; Shi, Q. Sodic alkaline stress mitigation by exogenous melatonin in tomato needs nitric oxide as a downstream signal. J. Plant Physiol. 2015, 186–187, 68–77. [Google Scholar] [CrossRef]
- Gong, X.; Shi, S.; Dou, F.; Song, Y.; Ma, F.; Fangfang, D.; Fengwang, M.; Yi, S.; Xiaoqing, G.; Shuting, S. Exogenous melatonin alleviates alkaline stress in Malus hupehensis Rehd. By regulating the biosynthesis of polyamines. Molecules 2017, 22, 1542. [Google Scholar] [CrossRef]
- Arnao, M.B.; Hernández-Ruiz, J. The physiological function of melatonin in plants. J. Plant Signal. Behav. 2006, 1, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R. Melatonin in plants? Diversity of levels and multiplicity of functions. Front. Plant Sci. 2016, 7, 198. [Google Scholar] [CrossRef]
- Nawaz, M.A.; Huang, Y.; Bie, Z.; Ahmed, W.; Reiter, R.J.; Niu, M.; Hameed, S. Corrigendum: Melatonin: Current status and future perspectives in plant science. Front. Plant Sci. 2016, 7, 714. [Google Scholar] [CrossRef]
- Wen, D.; Gong, B.; Sun, S.; Liu, S.; Wang, X.; Wei, M.; Yang, F.; Li, Y.; Shi, Q. Promoting roles of melatonin in adventitious root development of Solanum lycopersicum L. By regulating auxin and nitric oxide signaling. Front. Plant Sci. 2016, 7, 718. [Google Scholar] [CrossRef]
- Kaur, H.; Mukherjee, S.; Baluska, F.; Bhatla, S.C. Regulatory roles of serotonin and melatonin in abiotic stress tolerance in plants. Plant Signal. Behav. 2015, 10, e1049788. [Google Scholar] [CrossRef]
- Yadav, S.; David, A.; Baluška, F.; Bhatla, S.C. Rapid auxin-induced nitric oxide accumulation and subsequent tyrosine nitration of proteins during adventitious root formation in sunflower hypocotyls. Plant Signal. Behav. 2013, 8, e23196. [Google Scholar] [CrossRef][Green Version]
- Corpas, F.J.; Barroso, J.B. Functions of nitric oxide (NO) in roots during development and under adverse stress conditions. Plants 2015, 4, 240–252. [Google Scholar] [CrossRef]
- Zhang, J.; Zeng, B.; Mao, Y.; Kong, X.; Wang, X.; Yang, Y.; Zhang, J.; Xu, J.; Rengel, Z.; Chen, Q. Melatonin alleviates aluminium toxicity through modulating antioxidative enzymes and enhancing organic acid anion exudation in soybean. Funct. Plant Biol. 2017, 44, 961–968. [Google Scholar] [CrossRef]
- Tiwari, G.; Duraivadivel, P.; Sharma, S.; P, H. 1-aminocyclopropane-1-carboxylic acid deaminase producing beneficial rhizobacteria ameliorate the biomass characters of Panicum maximum Jacq. by mitigating drought and salt stress. Sci. Rep. 2018, 8, 17513. [Google Scholar] [CrossRef]
- DalCorso, G. Heavy Metal Toxicity in Plants. In Plants and Heavy Metals; Furini, A., Ed.; Springer: Dordrecht, The Netherlands, 2012; pp. 1–25. [Google Scholar]
- Arnao, M.B.; Hernandez-Ruiz, J. Melatonin: Synthesis from tryptophan and its role in higher plants. In Amino Acids in Higher Plants; Dmello, J.P.F., Ed.; CABI: Oxfordshire, UK, 2015; pp. 390–435. [Google Scholar]
- Arnao, M.B.; Hernández-Ruiz, J. Melatonin: Plant growth regulator and/or biostimulator during stress? Trends Plant Sci. 2014, 19, 789–797. [Google Scholar] [CrossRef]
- Savvides, A.; Ali, S.; Tester, M.; Fotopoulos, V. Chemical priming of plants against multiple abiotic stresses: Mission possible? Trends Plant Sci. 2016, 21, 329–340. [Google Scholar] [CrossRef] [PubMed]
- Misra, S.; Misra, S.; Gedamu, L.; Gedamu, L. Heavy metal tolerant transgenic Brassica napus L. and Nicotiana tabacum L. plants. Theor Appl. Genet. 1989, 78, 161–168. [Google Scholar] [CrossRef]
- Sunkar, R.; Kapoor, A.; Zhu, J.K. Posttranscriptional induction of two Cu/Zn superoxide dismutase genes in arabidopsis is mediated by downregulation of Mir398 and important for oxidative stress tolerance. Plant Cell 2006, 18, 2051–2065. [Google Scholar] [CrossRef]
- Zhu, Y.L.; Elizabeth, A.H.P.S.; Tarun, A.S.; Weber, S.U.; Jouanin, L.; Terry, N. Cadmium tolerance and accumulation in indian mustard is enhanced by overexpressing γ-glutamylcysteinesynthetase. Plant Physiol. 1999, 121, 1169–1177. [Google Scholar] [CrossRef]
- Hasan, M.K.; Ahammed, G.J.; Yin, L.; Shi, K.; Xia, X.; Zhou, Y.; Yu, J.; Zhou, J. Melatonin mitigates cadmium phytotoxicity through modulation of phytochelatins biosynthesis, vacuolar sequestration, and antioxidant potential in Solanum lycopersicum L. Front. Plant Sci. 2015, 6, 601. [Google Scholar] [CrossRef]
- Zuo, Z.; Sun, L.; Wang, T.; Miao, P.; Zhu, X.; Liu, S.; Song, F.; Mao, H.; Li, X. Melatonin improves the photosynthetic carbon assimilation and antioxidant capacity in wheat exposed to nano-zno stress. Molecules 2017, 22, 1727. [Google Scholar] [CrossRef]
- Posmyk, M.M.; Kuran, H.; Marciniak, K.; Janas, K.M. Presowing seed treatment with melatonin protects red cabbage seedlings against toxic copper ion concentrations. J. Pineal Res. 2008, 45, 24–31. [Google Scholar] [CrossRef]
- Kobylińska, A.; Posmyk, M.M. Melatonin restricts Pb-induced pcd by enhancing Bi-1 expression in tobacco suspension cells. BioMetals 2016, 29, 1059–1074. [Google Scholar] [CrossRef]
- Kobylińska, A.; Reiter, R.J.; Posmyk, M.M. Melatonin protects cultured tobacco cells against lead-induced cell death via inhibition of cytochrome c translocation. Front. Plant Sci. 2017, 8, 1560. [Google Scholar] [CrossRef]
- Li, M.Q.; Hasan, M.K.; Li, C.X.; Ahammed, G.J.; Xia, X.J.; Shi, K.; Zhou, Y.H.; Reiter, R.J.; Yu, J.Q.; Xu, M.X.; et al. Melatonin mediates selenium-induced tolerance to cadmium stress in tomato plants. J. Pineal Res. 2016, 61, 291–302. [Google Scholar] [CrossRef]
- Ni, J.; Wang, Q.; Shah, F.A.; Liu, W.; Wang, D.; Huang, S.; Fu, S.; Wu, L. Exogenous melatonin confers cadmium tolerance by counterbalancing the hydrogen peroxide homeostasis in wheat seedlings. Molecules 2018, 23, 799. [Google Scholar] [CrossRef]
- Gu, Q.; Chen, Z.; Yu, X.; Cui, W.; Pan, J.; Zhao, G.; Xu, S.; Wang, R.; Shen, W. Melatonin confers plant tolerance against cadmium stress via the decrease of cadmium accumulation and reestablishment of microrna-mediated redox homeostasis. Plant Sci. 2017, 261, 28–37. [Google Scholar] [CrossRef]
- Lin, L.; Li, J.; Chen, F.; Liao, M.A.; Tang, Y.; Liang, D.; Xia, H.; Lai, Y.; Wang, X.; Chen, C.; et al. Effects of melatonin on the growth and cadmium characteristics of Cyphomandra betacea Seedlings. Environ. Monit. Assess. 2018, 190, 1–8. [Google Scholar] [CrossRef]
- Huang, P.; Huang, S.; QI’AN, Z.H.A.N.G.; Yan, C.; Lu, X. Physiological mechanism of melatonin regulation in soybeans under aluminum stress. Pak. J. Bot. 2018, 50, 1259–1264. [Google Scholar]
- Nawaz, M.A.; Jiao, Y.; Chen, C.; Shireen, F.; Zheng, Z.; Imtiaz, M.; Bie, Z.; Huang, Y. Melatonin pretreatment improves vanadium stress tolerance of watermelon seedlings by reducing vanadium concentration in the leaves and regulating melatonin biosynthesis and antioxidant-related gene expression. J. Plant Physiol. 2018, 220, 115–127. [Google Scholar] [CrossRef]
- Hacışevki, A.; Baba, B. An overview of melatonin as an antioxidant molecule: A biochemical approach. Mol. Biol. Clin. Pharm. Approaches 2018, 59–85. [Google Scholar] [CrossRef]
- Moustafa-Farag, M.; Mahmoud, A.; Arnao, M.B.; Sheteiwy, M.S.; Dafea, M.; Soltan, M.; Elkelish, A.; Hasanuzzaman, M.; Ai, S.J.A. Melatonin-induced water stress tolerance in plants: Recent advances. Antioxidants 2020, 9, 809. [Google Scholar] [CrossRef]
- Xu, T.; Chen, Y.; Kang, H. Melatonin is a potential target for improving post-harvest preservation of fruits and vegetables. Front. Plant Sci. 2019, 10, 1388. [Google Scholar] [CrossRef]
- Lee, M.C.; Park, J.C.; Kim, D.H.; Kang, S.; Shin, K.H.; Park, H.G.; Han, J.; Lee, J.S. Interrelationship of salinity shift with oxidative stress and lipid metabolism in the monogonont rotifer Brachionus koreanus. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2017, 214, 79–84. [Google Scholar] [CrossRef]
- Zhang, Y. Biological role of ascorbate in plants. In Ascorbic Acid in Plants: Biosynthesis, Regulation and Enhancement; Zhang, Y., Ed.; Springer: New York, NY, USA, 2013; pp. 7–33. [Google Scholar]
- Cai, S.Y.; Zhang, Y.; Xu, Y.P.; Qi, Z.Y.; Li, M.Q.; Ahammed, G.J.; Xia, X.J.; Shi, K.; Zhou, Y.H.; Reiter, R.J.; et al. Hsfa1a upregulates melatonin biosynthesis to confer cadmium tolerance in tomato plants. J. Pineal Res. 2017, 62, e12387. [Google Scholar] [CrossRef]
- Luo, H.; He, C.; Han, L. Heterologous expression of zjomt from Zoysia japonica in Escherichia coli confers aluminum resistance through melatonin production. PLoS ONE 2018, 13, e0196952. [Google Scholar] [CrossRef]
- Hwang, O.J.; Back, K. Melatonin is involved in skotomorphogenesis by regulating brassinosteroid biosynthesis in rice plants. J. Pineal Res. 2018, 65, e12495. [Google Scholar] [CrossRef]
- Moussa, H.R.; Algamal, S.M.A. Does exogenous application of melatonin ameliorate boron toxicity in spinach plants? Int. J. Veg. Sci. 2017, 23, 233–245. [Google Scholar] [CrossRef]
- Erland, L.A.E.; Saxena, P.K.; Murch, S.J. Melatonin in plant signalling and behaviour. Funct. Plant Biol. 2018, 45, 58–69. [Google Scholar] [CrossRef]
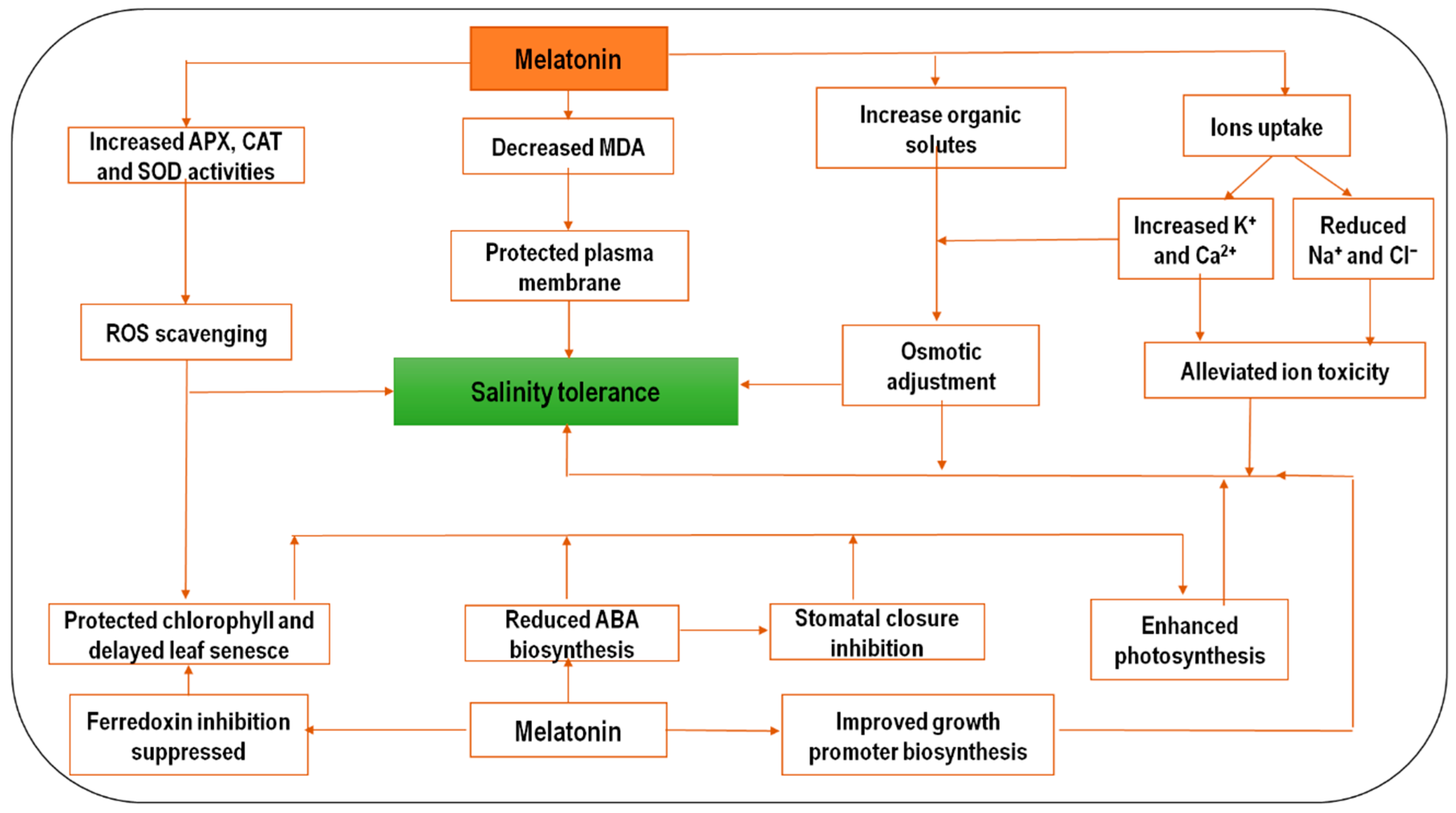
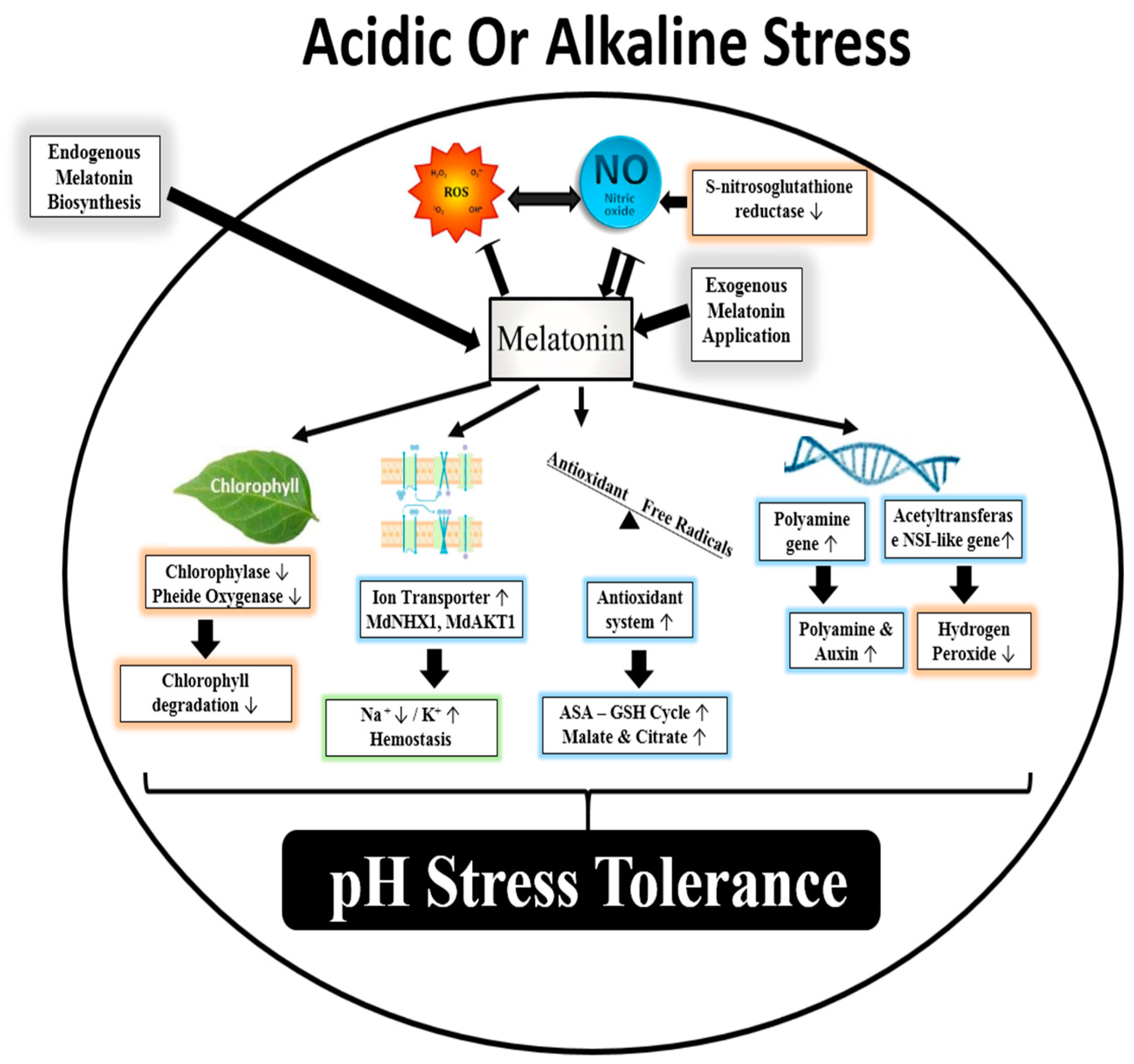
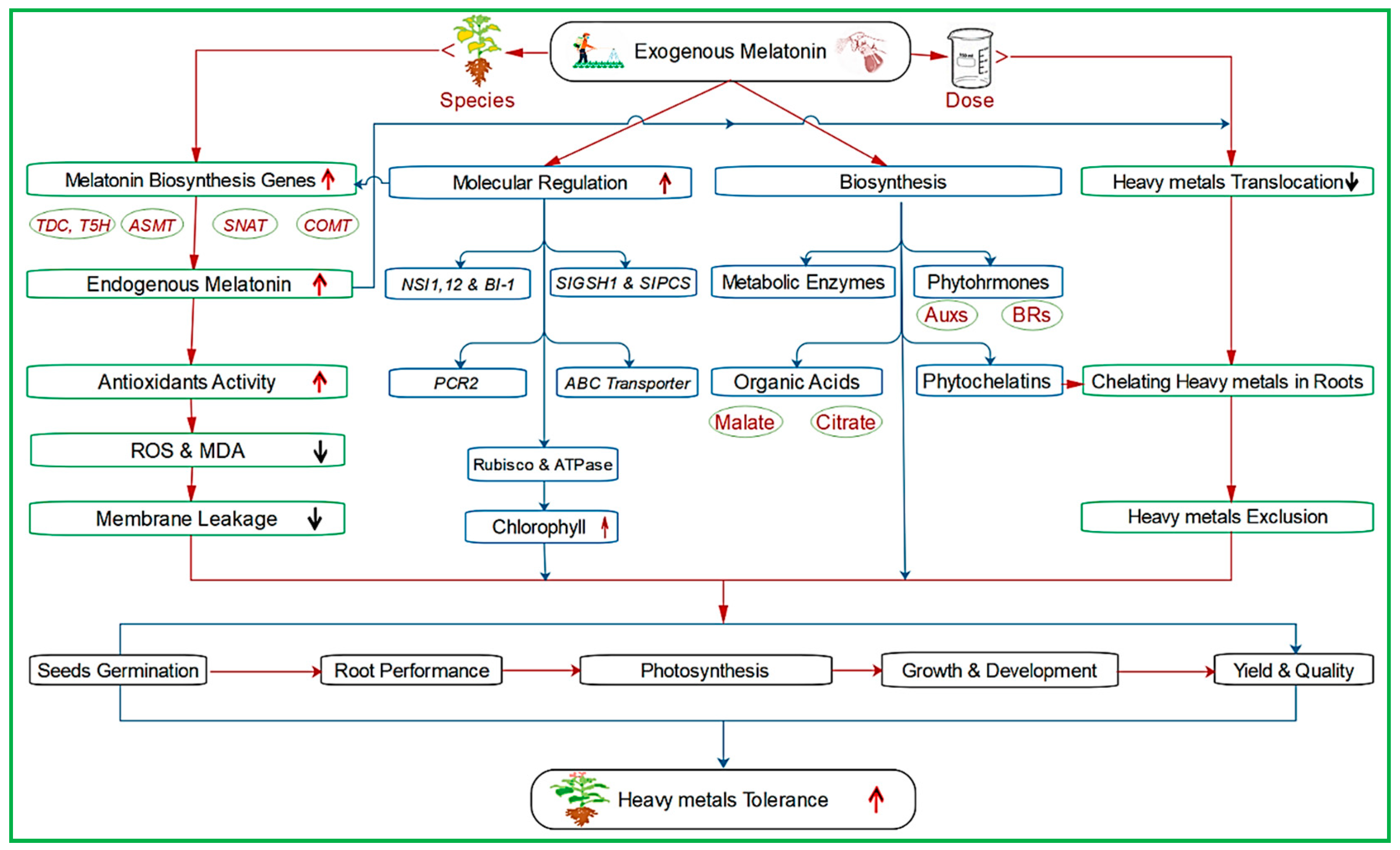
| Common Name | Scientific Name | Stress Treatment | MT Concentration | Findings | References |
|---|---|---|---|---|---|
| Field crops | |||||
| Rice | Oryza sativa | 0.5% NaCl | 0, 10, 20 μM | Antioxidants ↑, leaf senescence and cell death ↓, chlorophyll degradation ↓ | [40] |
| Rice | O. sativa | 150 and 200 mM NaCl | 10–500 μM | Seed germination and root vigor ↑, antioxidant enzymes ↑, Na+ and Cl− contents ↓ | [41] |
| Maize | Zea mays | 100 mM NaCl | 1 μM | Antioxidant enzymes ↑, K+ contents and K+/Na+ ratios ↑, electrolyte leakage ↓, MDA ↓ | [42] |
| Maize | Z. mays | 150 mM NaCl | 0–100 μM | Photosynthesis ↑, antioxidant enzymes ↑, Na+ contents ↓ | [43] |
| Broad bean | Vicia faba | 3.85 and 7.69 dSm−1 diluted seawater | 0100 and 500 μM | Plant growth ↑, RWC ↑, photosynthesis ↑, carbohydrates ↑, phenolic content ↑, IAA ↑, K+,Ca+2, K+/Na+, and Ca+2/Na+ ratios ↑ | [28] |
| Soybean | Glycine max | Soil saturated with 1% (w/v) NaCl | 0–100 μM | Photosynthesis ↑, cell division ↑, carbohydrates ↑, fatty acid ↑, ascorbate ↑, the inhibitory effects on gene expressions ↓ | [44] |
| Rapeseed | Brassica napus | 0.75% NaCl | 0–100 μM 30 μM | Antioxidant enzymes ↑, solute accumulation ↑ | [38] |
| Fruit crops | |||||
| Pingyitiancha | Malus hupehensis | 100 mM NaCl | 0.1 μM | Photosynthesis ↑, ion homeostasis ↑, oxidative damage ↓ | [33] |
| Vegetable crops | |||||
| Tomato | Solanum lycopersicum | 75 mM NaCl | 100 μM | Proteins and membranes protection ↑, antioxidants ↑, photosynthesis ↑ | [34] |
| Tomato | S.lycopersicum | 150 mM NaCl | 0–200 μM | Photosynthesis ↑, ROS ↓ | [35] |
| Cucumber | Cucumis sativus | 150 mM NaCl | 1 μM | Energy production regulation ↑ | [39] |
| Cucumber | C. sativus | 200 mM NaCl | 0–200 μM | Antioxidant enzymes ↑, chlorophyll ↑, photosynthesis ↑ | [36] |
| Cucumber | C. sativus | 150 mM NaCl | (0–500 μM) 1 μM | GA3 biosynthesis ↑, germination rate ↑, ABA ↓, oxidative damage ↓ | [45] |
| Watermelon | Citrullus lanatus | 300 mM NaCl | 50–150 μM | Photosynthesis ↑, antioxidant enzymes ↑, photosystem II efficiency ↑, stomatal closure ↓, oxidative damage ↓ | [37] |
| Common Name | Scientific Name | Stress Type | MT Concentration * | Findings | References |
|---|---|---|---|---|---|
| Lupin | Lupinus albus | pH (3.5 to 8.5) | ? ** | ↑Melatonin | [91] |
| Apple | Malus hupehensis | Alkalinity (pH 8.5 and 8.8) | 5 μM | ↑Polyamines, MDA ↓, ROS ↓, antioxidants ↑, polyamine synthesis genes↑ | [95] |
| Tomato | Solanum lycopersicum | Acidity (pH 2.5) | 100 μM | Photosynthesis ↑, antioxidants↑, ROS ↓ | [92] |
| Common Name | Scientific Name | Stress Concentration | MT Concentration * | Findings | Reference |
|---|---|---|---|---|---|
| Cadmium | |||||
| Wheat | Triticum aestivum | 0.2 mM | 50 μM | Antioxidants enzymes ↑ | [118] |
| Alfalfa | Medicago sativa | 50, 100, and 200 μM | 50 μM | ABC transporter and PCR2 transcripts ↑, Cd accumulation ↓ | [119] |
| Tree tomato | Cyphomandrabetacea | 10 mg·L−1 | 50 μM | Antioxidants ↑, plant biomass ↑ | [120] |
| Tomato | Solanum lycopersicum | 25 and 100 μM | 100 μM | Antioxidants ↑, glutathione and phytochelatins↑ | [112] |
| Tomato | Solanum lycopersicum | 100 μM | 1 μM | Plant growth ↑, electrolyte leakage ↓, photoinhibition ↓ | [117] |
| Lead | |||||
| Tobacco | Nicotiana benthamiana | 15 μM | 200 nM | DNA damage ↓, cell growth, and viability ↑ | [116] |
| Tobacco | Nicotiana benthamiana | 15 μM | 200 nM | Cell proliferation ↑, cell death ↓ | [115] |
| Aluminum | |||||
| Soybean | Glycine max | 300 μM | 100 mM | Antioxidants ↑, photosynthesis ↑, MDA ↓ | [121] |
| Copper | |||||
| Red cabbage | Brassica oleracea var. Capitata f. rubra | 0.5 and 1 mM | 10 μM | Germination and fresh weight ↑, MDA ↓ | [114] |
| Vanadium | |||||
| Watermelon | Citrullus lanatus | 50 mg·L−1 | 0.1 μM | Plant growth ↑, chlorophyll ↑, photosynthesis ↑, antioxidant enzymes ↑, V accumulation ↓, ROS ↓, MDA ↓ | [122] |
| Common Name | Scientific Name | HM Concentration | Melatonin Treatment * | Gene Name | Gene Description | Ref. |
|---|---|---|---|---|---|---|
| Wheat | Triticum aestivum | 0.2 mM Cd | 100 μM | TaASMT1, TaASMT2 and TaTDC | N-acetylserotonin methyltransferase and tryptophan decarboxylase | [118] |
| Watermelon | Citrullus lanatus | 50 mg·L−1 V | 0.1 μM | Cla010664 and Cla004567 | O-methyl transferase and methione S-methyl transferase | [122] |
| Tomato | Solanum lycopersicum | 100 μM Cd | 100 μM | SIGSH1 and SIPCS | Responsible for GSH and PCS in tomato | [112] |
| Alfalafa | Medicago sativa | 100 μM Cd | 50 μM | MsSNAT | M. sativa Serotonin N-acetyltransferase (a melatonin synthetic gene) | [119] |
| Tomato | Solanum lycopersicum | 100 μM Cd | NA | HsfA1a and COMT1 | Heat shock factor A1a and caffeic acid O-methyltransferase 1 | [128] |
| Zoysiagrass | Zoysia japonica | 400 μM Al | NA | ZjOMT | An O-methyltransferase gene cloned from Z. japonica | [129] |
| Soybean | Glycine max | 50 μM Al | 1 μM | NSI1 and NSI2 | Genes encoding acetyltransferase NSI-like (nuclear shuttle protein-interacting) | [103] |
| Tomato | Solanum lycopersicum | 100 μM Cd | 1 μM | TDC, T5H, SNAT, ASMT | Melatonin biosynthetic genes | [117] |
| Tobaco | Nicotiana tabacum | 15 μM Pb | 200 nM | BI-1 | Bax inhibitor-1 protects against apoptosis | [115] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moustafa-Farag, M.; Elkelish, A.; Dafea, M.; Khan, M.; Arnao, M.B.; Abdelhamid, M.T.; El-Ezz, A.A.; Almoneafy, A.; Mahmoud, A.; Awad, M.; et al. Role of Melatonin in Plant Tolerance to Soil Stressors: Salinity, pH and Heavy Metals. Molecules 2020, 25, 5359. https://doi.org/10.3390/molecules25225359
Moustafa-Farag M, Elkelish A, Dafea M, Khan M, Arnao MB, Abdelhamid MT, El-Ezz AA, Almoneafy A, Mahmoud A, Awad M, et al. Role of Melatonin in Plant Tolerance to Soil Stressors: Salinity, pH and Heavy Metals. Molecules. 2020; 25(22):5359. https://doi.org/10.3390/molecules25225359
Chicago/Turabian StyleMoustafa-Farag, Mohamed, Amr Elkelish, Mohamed Dafea, Mumtaz Khan, Marino B. Arnao, Magdi T. Abdelhamid, Aziz Abu El-Ezz, Abdlwareth Almoneafy, Ahmed Mahmoud, Mahrous Awad, and et al. 2020. "Role of Melatonin in Plant Tolerance to Soil Stressors: Salinity, pH and Heavy Metals" Molecules 25, no. 22: 5359. https://doi.org/10.3390/molecules25225359
APA StyleMoustafa-Farag, M., Elkelish, A., Dafea, M., Khan, M., Arnao, M. B., Abdelhamid, M. T., El-Ezz, A. A., Almoneafy, A., Mahmoud, A., Awad, M., Li, L., Wang, Y., Hasanuzzaman, M., & Ai, S. (2020). Role of Melatonin in Plant Tolerance to Soil Stressors: Salinity, pH and Heavy Metals. Molecules, 25(22), 5359. https://doi.org/10.3390/molecules25225359













