Recent Advances in Electrochemiluminescence and Chemiluminescence of Metal Nanoclusters
Abstract
1. Introduction
2. Electrochemiluminescence of Metal NCs
2.1. Metal NCs as ECL Luminophores
2.1.1. Au NCs as ECL Luminophores
2.1.2. Other Monometallic NCs (Ag, Cu, Pt, Ni) as ECL Luminophores
2.2. Metal NCs as ECL Quenchers
2.3. Metal NCs as ECL Catalysts
3. Chemiluminescence of Metal NCs
3.1. Metal NCs as CL Catalysts
3.1.1. The Catalysis of Single Metal NCs
3.1.2. Methods for Improving Catalytic Efficiency of Metal NCs
3.2. Metal NCs as CL Luminophores
3.3. Metal NCs as CL Quenchers
4. Summary and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Halawa, M.I.; Li, B.S.; Xu, G. Novel Synthesis of Thiolated Gold Nanoclusters Induced by Lanthanides for Ultrasensitive and Luminescent Detection of the Potential Anthrax Spores’ Biomarker. ACS Appl. Mater. Interfaces 2020, 12, 32888–32897. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Lin, X.; Shu, T.; Su, L.; Liang, F.; Zhang, X. Self-Assembly of Metal Nanoclusters for Aggregation-Induced Emission. Int. J. Mol. Sci. 2019, 20, 1891. [Google Scholar] [CrossRef]
- Xu, J.; Shang, L. Emerging applications of near-infrared fluorescent metal nanoclusters for biological imaging. Chin. Chem. Lett. 2018, 29, 1436–1444. [Google Scholar] [CrossRef]
- Halawa, M.I.; Wu, F.; Nsabimana, A.; Lou, B.; Xu, G. Inositol directed facile “green” synthesis of fluorescent gold nanoclusters as selective and sensitive detecting probes of ferric ions. Sens. Actuators B Chem. 2018, 257, 980–987. [Google Scholar] [CrossRef]
- Halawa, M.I.; Wu, F.; Fereja, T.H.; Lou, B.; Xu, G. One-pot green synthesis of supramolecular beta-cyclodextrin functionalized gold nanoclusters and their application for highly selective and sensitive fluorescent detection of dopamine. Sens. Actuators B Chem. 2018, 254, 1017–1024. [Google Scholar] [CrossRef]
- Halawa, M.I.; Lai, J.; Xu, G. Gold nanoclusters: Synthetic strategies and recent advances in fluorescent sensing. Mater. Today Nano 2018, 3, 9–27. [Google Scholar] [CrossRef]
- Halawa, M.I.; Gao, W.; Saqib, M.; Kitte, S.A.; Wu, F.; Xu, G. Sensitive detection of alkaline phosphatase by switching on gold nanoclusters fluorescence quenched by pyridoxal phosphate. Biosens. Bioelectron. 2017, 95, 8–14. [Google Scholar] [CrossRef]
- Díez, I.; Pusa, M.; Kulmala, S.; Jiang, H.; Walther, A.; Goldmann, A.S.; Müller, A.H.E.; Ikkala, O.; Ras, R.H.A. Color Tunability and Electrochemiluminescence of Silver Nanoclusters. Angew. Chem. Int. Ed. 2009, 48, 2122–2125. [Google Scholar] [CrossRef]
- Fang, Y.-M.; Song, J.; Li, J.; Wang, M.; Yang, H.; Sun, J.-J.; Chen, G.-N. Electrogenerated chemiluminescence from Au nanoclusters. Chem. Commun. 2011, 47, 2369–2371. [Google Scholar] [CrossRef]
- Chen, Y.; Shen, Y.; Sun, D.; Zhang, H.; Tian, D.; Zhang, J.; Zhu, J.-J. Fabrication of a dispersible graphene/gold nanoclusters hybrid and its potential application in electrogenerated chemiluminescence. Chem. Commun. 2011, 47, 11733–11735. [Google Scholar] [CrossRef]
- Li, L.; Liu, H.; Shen, Y.; Zhang, J.; Zhu, J.J. Electrogenerated Chemiluminescence of Au Nanoclusters for the Detection of Dopamine. Anal. Chem. 2011, 83, 661–665. [Google Scholar] [CrossRef]
- Yuan, D.; Chen, S.; Yuan, R.; Zhang, J.; Zhang, W. An electrogenerated chemiluminescence sensor prepared with a graphene/multiwall carbon nanotube/gold nanocluster hybrid for the determination of phenolic compounds. Analyst 2013, 138, 6001–6006. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.-Y.; Ma, S.; Zhuo, Y.; Chai, Y.; Yuan, R. In Situ Electrochemical Generation of Electrochemiluminescent Silver Naonoclusters on Target-Cycling Synchronized Rolling Circle Amplification Platform for MicroRNA Detection. Anal. Chem. 2016, 88, 3203–3210. [Google Scholar] [CrossRef]
- Zhao, M.; Chen, A.-Y.; Huang, D.; Zhuo, Y.; Chai, Y.-Q.; Yuan, R. Cu Nanoclusters: Novel Electrochemiluminescence Emitters for Bioanalysis. Anal. Chem. 2016, 88, 11527–11532. [Google Scholar] [CrossRef]
- Peng, H.; Jian, M.; Deng, H.; Wang, W.; Huang, Z.; Huang, K.; Liu, A.; Chen, W. Valence States Effect on Electrogenerated Chemiluminescence of Gold Nanocluster. ACS Appl. Mater. Interfaces 2017, 9, 14929–14934. [Google Scholar] [CrossRef]
- Peng, H.; Huang, Z.; Wu, W.; Liu, M.; Huang, K.; Yang, Y.; Deng, H.; Xia, X.-H.; Chen, W. Versatile High-Performance Electrochemiluminescence ELISA Platform Based on a Gold Nanocluster Probe. ACS Appl. Mater. Interfaces 2019, 11, 24812–24819. [Google Scholar] [CrossRef]
- Peng, H.; Huang, Z.; Sheng, Y.; Zhang, X.; Deng, H.; Chen, W.; Liu, J. Pre-oxidation of Gold Nanoclusters Results in a 66% Anodic Electrochemiluminescence Yield and Drives Mechanistic Insights. Angew. Chem. Int. Ed. 2019, 58, 11691–11694. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, B.; Fu, L.; Fu, K.; Zou, G. Efficient and Monochromatic Electrochemiluminescence of Aqueous-Soluble Au Nanoclusters via Host–Guest Recognition. Angew. Chem. Int. Ed. 2019, 58, 6901–6905. [Google Scholar] [CrossRef]
- Peng, H.; Deng, H.; Jian, M.; Liu, A.; Bai, F.; Lin, X.; Chen, W. Electrochemiluminescence sensor based on methionine-modified gold nanoclusters for highly sensitive determination of dopamine released by cells. Microchim. Acta 2016, 184, 735–743. [Google Scholar] [CrossRef]
- Wang, T.; Wang, G.; Padelford, J.W.; Jiang, J.; Wang, G. Near-Infrared Electrogenerated Chemiluminescence from Aqueous Soluble Lipoic Acid Au Nanoclusters. J. Am. Chem. Soc. 2016, 138, 6380–6383. [Google Scholar] [CrossRef]
- Guo, Y.; Pan, X.; Zhang, W.; Hu, Z.; Wong, K.-W.; He, Z.; Li, H.-W. Label-free probes using DNA-templated silver nanoclusters as versatile reporters. Biosens. Bioelectron. 2020, 150, 111926. [Google Scholar] [CrossRef]
- Li, S.; Liu, Y.; Ma, Q. Nanoparticle-based electrochemiluminescence cytosensors for single cell level detection. TrAC Trends Anal. Chem. 2019, 110, 277–292. [Google Scholar] [CrossRef]
- He, S.; Ding, Z. Progress in electrochemistry and electrochemiluminescence of metal clusters. Curr. Opin. Electrochem. 2018, 7, 109–117. [Google Scholar] [CrossRef]
- Rizwan, M.; Mohd-Naim, N.; Ahmed, M.U. Trends and Advances in Electrochemiluminescence Nanobiosensors. Sensors 2018, 18, 166. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Wang, X. Progress of Metal Nanoclusters-based Electrochemiluminescent Analysis. Chin. J. Anal. Chem. 2017, 45, 1776–1785. [Google Scholar] [CrossRef]
- Zhai, Q.; Li, J.; Wang, E. Recent Advances Based on Nanomaterials as Electrochemiluminescence Probes for the Fabrication of Sensors. Chem. Electro.Chem. 2017, 4, 1639–1650. [Google Scholar] [CrossRef]
- Yu, X.; Wang, Q. The determination of copper ions based on sensitized chemiluminescence of silver nanoclusters. Microchim. Acta 2011, 173, 293–298. [Google Scholar] [CrossRef]
- Hu, L.; Yuan, Y.; Zhang, L.; Zhao, J.; Majeed, S.; Xu, G. Copper nanoclusters as peroxidase mimetics and their applications to H2O2 and glucose detection. Anal. Chim. Acta 2013, 762, 83–86. [Google Scholar] [CrossRef]
- Wang, X.-X.; Wu, Q.; Shan, Z.; Huang, Q.-M. BSA-stabilized Au clusters as peroxidase mimetics for use in xanthine detection. Biosens. Bioelectron. 2011, 26, 3614–3619. [Google Scholar] [CrossRef]
- Mokhtarzadeh, E.; Abolhasani, J.; Hassanzadeh, J.; Elham, M.; Jafar, A.; Javad, H. Rhodamine B Chemiluminescence Improved by Mimetic AuCu Alloy Nanoclusters and Ultrasensitive Measurement of H2O2, Glucose and Xanthine. Anal. Sci. 2019, 35, 543–550. [Google Scholar] [CrossRef]
- Yousefzadeh, A.; Abolhasani, J.; Hassanzadeh, J.; Somi, M.H. Ultrasensitive chemiluminescence assay for cimetidine detection based on the synergistic improving effect of Au nanoclusters and graphene quantum dots. Luminescence 2019, 34, 261–271. [Google Scholar] [CrossRef]
- Yousefzadeh, A.; Abolhasani, J.; Hassanzadeh, J.; Somi, M.H. A Highly Efficient Chemiluminescence System Based on an Enhancing Effect of Ag Nanoclusters/Graphene Quantum Dots Mixture for Ultrasensitive Detection of Rabeprazole. Anal. Sci. 2019, 35, 385–391. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, J.; Li, Y.; Han, S. Chemiluminescence of copper nanoclusters and its application for trihexyphenidyl hydrochloride detection. Luminescence 2018, 33, 962–967. [Google Scholar] [CrossRef]
- You, X.; Li, Y. Direct chemiluminescence of fluorescent gold nanoclusters with classic oxidants for hydrogen peroxide sensing. Arab. J. Chem. 2019, 12, 69–74. [Google Scholar] [CrossRef]
- You, X.; Li, Y.; Li, B.; Ma, J. Gold nanoclusters-based chemiluminescence resonance energy transfer method for sensitive and label-free detection of trypsin. Talanta 2016, 147, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Chen, C.; Li, Z.; Yuan, Z.; Lu, C. Hydroxyl radical induced chemiluminescence of hyperbranched polyethyleneimine protected silver nanoclusters and its application in tea polyphenols detection. Anal. Methods 2017, 9, 3114–3120. [Google Scholar] [CrossRef]
- Vahid, B.; Hassanzadeh, J.; Khodakarami, B. CdSe quantum dots-sensitized chemiluminescence system and quenching effect of gold nanoclusters for cyanide detection. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 212, 322–329. [Google Scholar] [CrossRef]
- Zhai, Q.; Xing, H.; Zhang, X.; Li, J.; Wang, E. Enhanced Electrochemiluminescence Behavior of Gold–Silver Bimetallic Nanoclusters and Its Sensing Application for Mercury(II). Anal. Chem. 2017, 89, 7788–7794. [Google Scholar] [CrossRef]
- Yang, F.; Jiang, X.-Y.; Liang, W.-B.; Chai, Y.-Q.; Yuan, R.; Zhuo, Y. 3D Matrix-Arranged AuAg Nanoclusters As Electrochemiluminescence Emitters for Click Chemistry-Driven Signal Switch Bioanalysis. Anal. Chem. 2020, 92, 2566–2572. [Google Scholar] [CrossRef]
- Tang, Y.; Xu, J.; Xiong, C.; Xiao, Y.; Zhang, X.; Wang, S. Enhanced electrochemiluminescence of gold nanoclusters via silver doping and their application for ultrasensitive detection of dopamine. Analyst 2019, 144, 2643–2648. [Google Scholar] [CrossRef]
- Chen, S.; Ma, H.; Padelford, J.W.; Qinchen, W.; Yu, W.; Wang, S.; Kang, X.; Wang, G. Near Infrared Electrochemiluminescence of Rod-Shape 25-Atom AuAg Nanoclusters That Is Hundreds-Fold Stronger Than That of Ru(bpy)3 Standard. J. Am. Chem. Soc. 2019, 141, 9603–9609. [Google Scholar] [CrossRef] [PubMed]
- Goswami, N.; Yao, Q.; Luo, Z.; Li, J.; Chen, T.; Xie, J. Luminescent Metal Nanoclusters with Aggregation-Induced Emission. J. Phys. Chem. Lett. 2016, 7, 962–975. [Google Scholar] [CrossRef]
- Peng, H.; Huang, Z.; Deng, H.; Wu, W.; Huang, K.; Li, Z.; Chen, W.; Liu, J. Dual Enhancement of Gold Nanocluster Electrochemiluminescence: Electrocatalytic Excitation and Aggregation-Induced Emission. Angew. Chem. Int. Ed. 2019, 59, 9982–9985. [Google Scholar] [CrossRef]
- Jiang, H.; Qin, Z.; Zheng, Y.; Liu, L.; Wang, X. Aggregation-Induced Electrochemiluminescence by Metal-Binding Protein Responsive Hydrogel Scaffolds. Small 2019, 15, e1901170. [Google Scholar] [CrossRef]
- Han, S.; Zhang, Z.; Li, S.; Qi, L.; Xu, G. Chemiluminescence and electrochemiluminescence applications of metal nanoclusters. Sci. China Ser. B Chem. 2016, 59, 794–801. [Google Scholar] [CrossRef]
- Zhou, Y.; Chai, Y.-Q.; Yuan, R. Highly Efficient Dual-Polar Electrochemiluminescence from Au25 Nanoclusters: The Next Generation of Multibiomarker Detection in a Single Step. Anal. Chem. 2019, 91, 14618–14623. [Google Scholar] [CrossRef]
- Jiang, W.; Yin, H.; Zhou, Y.; Duan, J.; Li, H.; Wang, M.; Waterhouse, G.I.N.; Ai, S. A novel electrochemiluminescence biosensor for the detection of 5-methylcytosine, TET 1 protein and beta-glucosyltransferase activities based on gold nanoclusters-H2O2 system. Sens. Actuators B Chem. 2018, 274, 144–151. [Google Scholar]
- Li, W.J.; Wang, X.; Jiang, T.; Ma, X.; Tian, H. One-pot synthesis of beta-cyclodextrin modified Au nanoclusters with near-infrared emission. Chem. Commun. 2020, 56, 5580–5583. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Fan, Y.; Zhang, C.; He, Y.; Wei, S. Quenched solid-state electrochemiluminescence of gold nanoclusters and the application in the ultrasensitive detection of concanavalin A. Electrochim. Acta 2017, 228, 195–202. [Google Scholar] [CrossRef]
- Peng, H.-P.; Jian, M.-L.; Huang, Z.-N.; Wang, W.-J.; Deng, H.-H.; Wu, W.-H.; Liu, A.-L.; Xia, X.-H.; Chen, W. Facile electrochemiluminescence sensing platform based on high-quantum-yield gold nanocluster probe for ultrasensitive glutathione detection. Biosens. Bioelectron. 2018, 105, 71–76. [Google Scholar] [CrossRef]
- Kim, J.M.; Jeong, S.; Song, J.K.; Kim, J. Near-infrared electrochemiluminescence from orange fluorescent Au nanoclusters in water. Chem. Commun. 2018, 54, 2838–2841. [Google Scholar] [CrossRef]
- Kang, Y.; Kim, J. Electrochemiluminescence of Glutathione-Stabilized Au Nanoclusters Fractionated by Gel Electrophoresis in Water. ChemElectroChem 2020, 7, 1092–1096. [Google Scholar] [CrossRef]
- Yu, L.; Zhang, Q.; Kang, Q.; Zhang, B.; Shen, D.; Zou, G. Near-Infrared Electrochemiluminescence Immunoassay with Biocompatible Au Nanoclusters as Tags. Anal. Chem. 2020, 92, 7581–7587. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Liu, L.; Wang, X. Red-emitted electrochemiluminescence by yellow fluorescent thioglycol/glutathione dual thiolate co-coated Au nanoclusters. Nanoscale 2017, 9, 9792–9796. [Google Scholar] [CrossRef]
- Wang, T.; Padelford, J.W.; Ma, H.; Gubitosi-Raspino, M.F.; Wang, G. Near-Infrared Electrochemiluminescence from Au Nanoclusters Enhanced by EDTA and Modulated by Ions. ChemElectroChem 2017, 4, 1697–1701. [Google Scholar] [CrossRef][Green Version]
- Wang, T.; Ma, H.; Padelford, J.W.; Lobo, E.; Tran, M.T.; Zhao, F.; Fang, N.; Wang, G. Metal ions-modulated near-infrared electrochemiluminescence from Au nanoclusters enhanced by 4-(2-Hydroxyethyl)-1-piperazineethanesulfonic acid at physiological pH. Electrochim. Acta 2018, 282, 369–376. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, S.; Luo, X.; Chai, Y.; Yuan, R. Ternary Electrochemiluminescence Nanostructure of Au Nanoclusters as a Highly Efficient Signal Label for Ultrasensitive Detection of Cancer Biomarkers. Anal. Chem. 2018, 90, 10024–10030. [Google Scholar] [CrossRef]
- Jia, Y.; Yang, L.; Xue, J.; Ren, X.; Zhang, N.; Fan, D.; Wei, Q.; Ma, H. Highly-branched Cu2O as well-ordered co-reaction accelerator for amplifying electrochemiluminescence response of gold nanoclusters and procalcitonin analysis based on protein bioactivity maintenance. Biosens. Bioelectron. 2019, 144, 111676. [Google Scholar] [CrossRef]
- Jia, Y.; Yang, L.; Xue, J.; Zhang, N.; Fan, D.; Ma, H.; Ren, X.; Hu, L.; Wei, Q. Bioactivity-Protected Electrochemiluminescence Biosensor Using Gold Nanoclusters as the Low-Potential Luminophor and Cu2S Snowflake as Co-reaction Accelerator for Procalcitonin Analysis. ACS Sens. 2019, 4, 1909–1916. [Google Scholar] [CrossRef]
- Zhang, C.; Fan, Y.; Zhang, H.; Chen, S.; Yuan, R. An ultrasensitive signal-on electrochemiluminescence biosensor based on Au nanoclusters for detecting acetylthiocholine. Anal. Bioanal. Chem. 2018, 411, 905–913. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Zhong, X.; Jiang, X.; Zhuo, Y.; Yuan, R.; Wei, S. An ultrasensitive aptasensor based on self-enhanced Au nanoclusters as highly efficient electrochemiluminescence indicator and multi-site landing DNA walker as signal amplification. Biosens. Bioelectron. 2019, 130, 262–268. [Google Scholar] [CrossRef]
- Lv, X.; Ma, H.; Wu, D.; Yan, L.; Ji, L.; Liu, Y.; Pang, X.; Du, B.; Wei, Q. Novel gold nanocluster electrochemiluminescence immunosensors based on nanoporous NiGd–Ni2O3–Gd2O3 alloys. Biosens. Bioelectron. 2016, 75, 142–147. [Google Scholar] [CrossRef]
- Zhang, X.-L.; Li, X.; Li, X.-T.; Gao, Y.; Feng, F.; Yang, G. Electrochemiluminescence sensor for pentoxifylline detection using Au nanoclusters@graphene quantum dots as an amplified electrochemiluminescence luminophore. Sens. Actuators B Chem. 2019, 282, 927–935. [Google Scholar] [CrossRef]
- Zhu, L.; Ye, J.; Yan, M.; Zhu, Q.; Wang, S.; Huang, J.; Sun, J. Electrochemiluminescence Immunosensor Based on Au Nanocluster and Hybridization Chain Reaction Signal Amplification for Ultrasensitive Detection of Cardiac Troponin I. ACS Sens. 2019, 4, 2778–2785. [Google Scholar] [CrossRef]
- Feng, L.; Wu, L.; Xing, F.; Hu, L.; Ren, J.; Qu, X. Novel electrochemiluminescence of silver nanoclusters fabricated on triplex DNA scaffolds for label-free detection of biothiols. Biosens. Bioelectron. 2017, 98, 378–385. [Google Scholar] [CrossRef]
- Jie, G.; Tan, L.; Zhao, Y.; Wang, X. A novel silver nanocluster in situ synthesized as versatile probe for electrochemiluminescence and electrochemical detection of thrombin by multiple signal amplification strategy. Biosens. Bioelectron. 2017, 94, 243–249. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, M.; Zhuo, Y.; Chai, Y.; Xu, W.; Yuan, R. In Situ Electrodeposited Synthesis of Electrochemiluminescent Ag Nanoclusters as Signal Probe for Ultrasensitive Detection of Cyclin-D1 from Cancer Cells. Anal. Chem. 2017, 89, 6787–6793. [Google Scholar] [CrossRef]
- Feng, X.; Han, T.; Xiong, Y.; Wang, S.; Dai, T.; Chen, J.; Zhang, X.; Wang, G. Plasmon-Enhanced Electrochemiluminescence of Silver Nanoclusters for microRNA Detection. ACS Sens. 2019, 4, 1633–1640. [Google Scholar] [CrossRef] [PubMed]
- Song, Q.; Shi, Y.; He, D.; Xu, S.; Ouyang, J. Sequence-Dependent dsDNA-Templated Formation of Fluorescent Copper Nanoparticles. Chem. Eur. J. 2014, 21, 2417–2422. [Google Scholar] [CrossRef]
- Liao, H.; Zhou, Y.; Chai, Y.-Q.; Yuan, R. An ultrasensitive electrochemiluminescence biosensor for detection of MicroRNA by in-situ electrochemically generated copper nanoclusters as luminophore and TiO2 as coreaction accelerator. Biosens. Bioelectron. 2018, 114, 10–14. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, H.; Zhang, H.; Chai, Y.-Q.; Yuan, R. Programmable Modulation of Copper Nanoclusters Electrochemiluminescence via DNA Nanocranes for Ultrasensitive Detection of microRNA. Anal. Chem. 2018, 90, 3543–3549. [Google Scholar] [CrossRef]
- Liu, H.; Gao, X.; Zhuang, X.; Tian, C.; Wang, Z.; Li, Y.; Rogach, A.L. A specific electrochemiluminescence sensor for selective and ultra-sensitive mercury(II) detection based on dithiothreitol functionalized copper nanocluster/carbon nitride nanocomposites. Analyst 2019, 144, 4425–4431. [Google Scholar] [CrossRef]
- Zhuang, X.; Gao, X.; Tian, C.; Cui, D.-L.; Luan, F.; Wang, Z.; Xiong, Y.; Chen, L. Synthesis of europium(iii)-doped copper nanoclusters for electrochemiluminescence bioanalysis. Chem. Commun. 2020, 56, 5755–5758. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wu, W.; Wang, Q.; Xiao, H.; Kuang, Y.; Liu, C. Novel anodic electrochemiluminescence system of Pt nanocluster/graphene hybrids for ultrasensitive detection of Cu2+. J. Electroanal. Chem. 2016, 772, 73–79. [Google Scholar] [CrossRef]
- Babamiri, B.; Salimi, A.; Hallaj, R.; Hasanzadeh, M. Nickel nanoclusters as a novel emitter for molecularly imprinted electrochemiluminescence based sensor toward nanomolar detection of creatinine. Biosens. Bioelectron. 2018, 107, 272–279. [Google Scholar] [CrossRef]
- Ge, J.; Li, C.; Zhao, Y.; Yu, X.; Jie, G. Versatile “on–off” biosensing of thrombin and miRNA based on Ag(i) ion-enhanced or Ag nanocluster-quenched electrochemiluminescence coupled with hybridization chain reaction amplification. Chem. Commun. 2019, 55, 7350–7353. [Google Scholar] [CrossRef]
- Zhu, H.; Ye, J.; Yan, M.; Zhu, Q.; Zhu, H. A wavelength-resolved electrochemiluminescence resonance energy transfer ratiometric immunosensor for detection of cardiac troponin I. Analyst 2019, 144, 6554–6560. [Google Scholar] [CrossRef]
- Luo, Q.-X.; Li, Y.; Liang, R.-P.; Cao, S.-P.; Jin, H.-J.; Qiu, J.-D. Gold nanoclusters enhanced electrochemiluminescence of g-C3N4 for protein kinase activity analysis and inhibition. J. Electroanal. Chem. 2020, 856, 113706. [Google Scholar] [CrossRef]
- Wang, C.; Chen, M.; Han, Q.; Wu, J.; Zhao, X.; Fu, Y. A three-dimensional DNA nanomachine with target recycling amplification technology and multiple electrochemiluminescence resonance energy transfer for sensitive microRNA-141 detection. Biosens. Bioelectron. 2020, 156, 112146. [Google Scholar] [CrossRef]
- Sui, Y.; Deng, M.; Xu, S.; Chen, F. Gold nanocluster-enhanced peroxynitrous acid chemiluminescence for high selectivity sensing of nitrite. RSC Adv. 2015, 5, 13495–13501. [Google Scholar] [CrossRef]
- Yang, D.; He, Y.; Sui, Y.; Chen, F. Determination of catechol in water based on gold nanoclusters-catalyzed chemiluminescence. J. Luminiscence 2017, 187, 186–192. [Google Scholar] [CrossRef]
- Sheng, Y.; Yang, H.; Wang, Y.; Han, L.; Zhao, Y.; Fan, A. Silver nanoclusters-catalyzed luminol chemiluminescence for hydrogen peroxide and uric acid detection. Talanta 2017, 166, 268–274. [Google Scholar] [CrossRef]
- Han, L.; Li, Y.; Fan, A. Improvement of mimetic peroxidase activity of gold nanoclusters on the luminol chemiluminescence reaction by surface modification with ethanediamine. Luminiscence 2018, 33, 751–758. [Google Scholar] [CrossRef]
- Yao, Y.; Wang, X.; Duan, W.; Li, F. A label-free, versatile and low-background chemiluminescence aptasensing strategy based on gold nanocluster catalysis combined with the separation of magnetic beads. Analyst 2018, 143, 709–714. [Google Scholar] [CrossRef]
- Xu, S.; Wang, Y.; Zhou, D.; Kuang, M.; Fang, D.; Yang, W.; Wei, S.; Ma, L. A novel chemiluminescence sensor for sensitive detection of cholesterol based on the peroxidase-like activity of copper nanoclusters. Sci. Rep. 2016, 6, 39157. [Google Scholar] [CrossRef] [PubMed]
- Borghei, Y.-S.; Hosseini, M.; Khoobi, M.; Ganjali, M.R. Copper nanocluster-enhanced luminol chemiluminescence for high-selectivity sensing of tryptophan and phenylalanine. Luminescence 2017, 32, 1045–1050. [Google Scholar] [CrossRef]
- Wu, X.; Hu, X.; Wang, G. Enhanced chemiluminescence of the luminol-hydrogen peroxide system by copper nanoclusters and its analytical application. Chem. Res. Appl. 2016, 28, 1070–1074. [Google Scholar]
- Han, S.; Chen, X. Copper nanoclusters-enhanced chemiluminescence for folic acid and nitrite detection. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 210, 315–320. [Google Scholar] [CrossRef]
- Yousefzadeh, A.; Hassanzadeh, J.; Mousavi, S.M.J.; Yousefzadeh, M. Surface molecular imprinting and powerfully enhanced chemiluminescence emission by Cu nanoclusters/MOF composite for detection of tramadol. Sens. Actuators B Chem. 2019, 286, 154–162. [Google Scholar] [CrossRef]
- Chen, H.; Lin, L.; Li, H.; Li, J.; Lin, J.-M. Aggregation-Induced Structure Transition of Protein-Stabilized Zinc/Copper Nanoclusters for Amplified Chemiluminescence. ACS Nano 2015, 9, 2173–2183. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Peng, W.; You, X. Determination of dopamine by exploiting the catalytic effect of hemoglobin–stabilized gold nanoclusters on the luminol–NaIO4 chemiluminescence system. Microchim. Acta 2017, 184, 3539–3545. [Google Scholar] [CrossRef]
- Yang, N.; He, Y.; Sui, Y.; Chen, F. Gold nanoclusters-catalyzed rhodamine 6G–K3Fe(CN)6 chemiluminescence and its application. Anal. Methods 2016, 8, 7272–7278. [Google Scholar] [CrossRef]
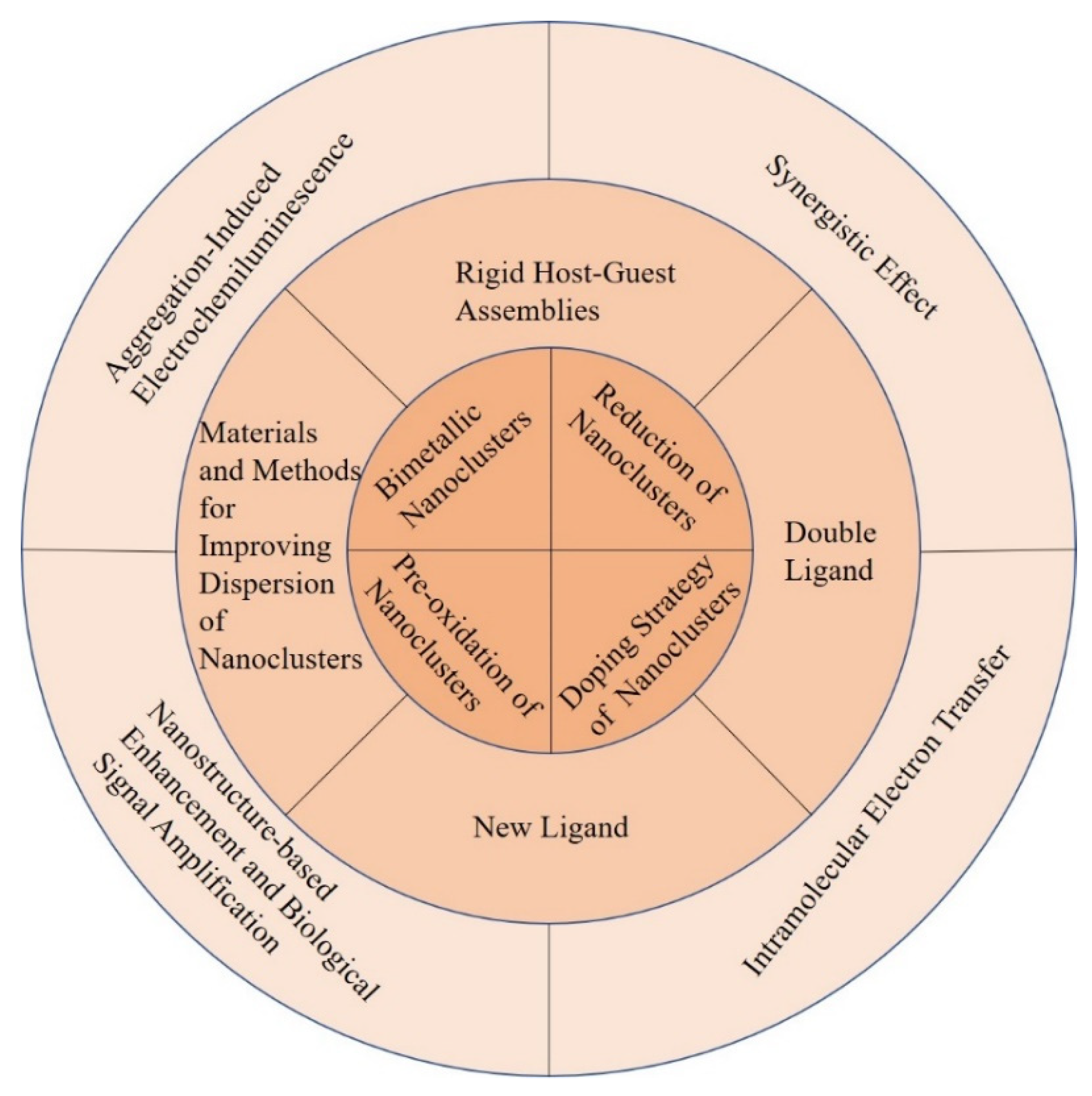
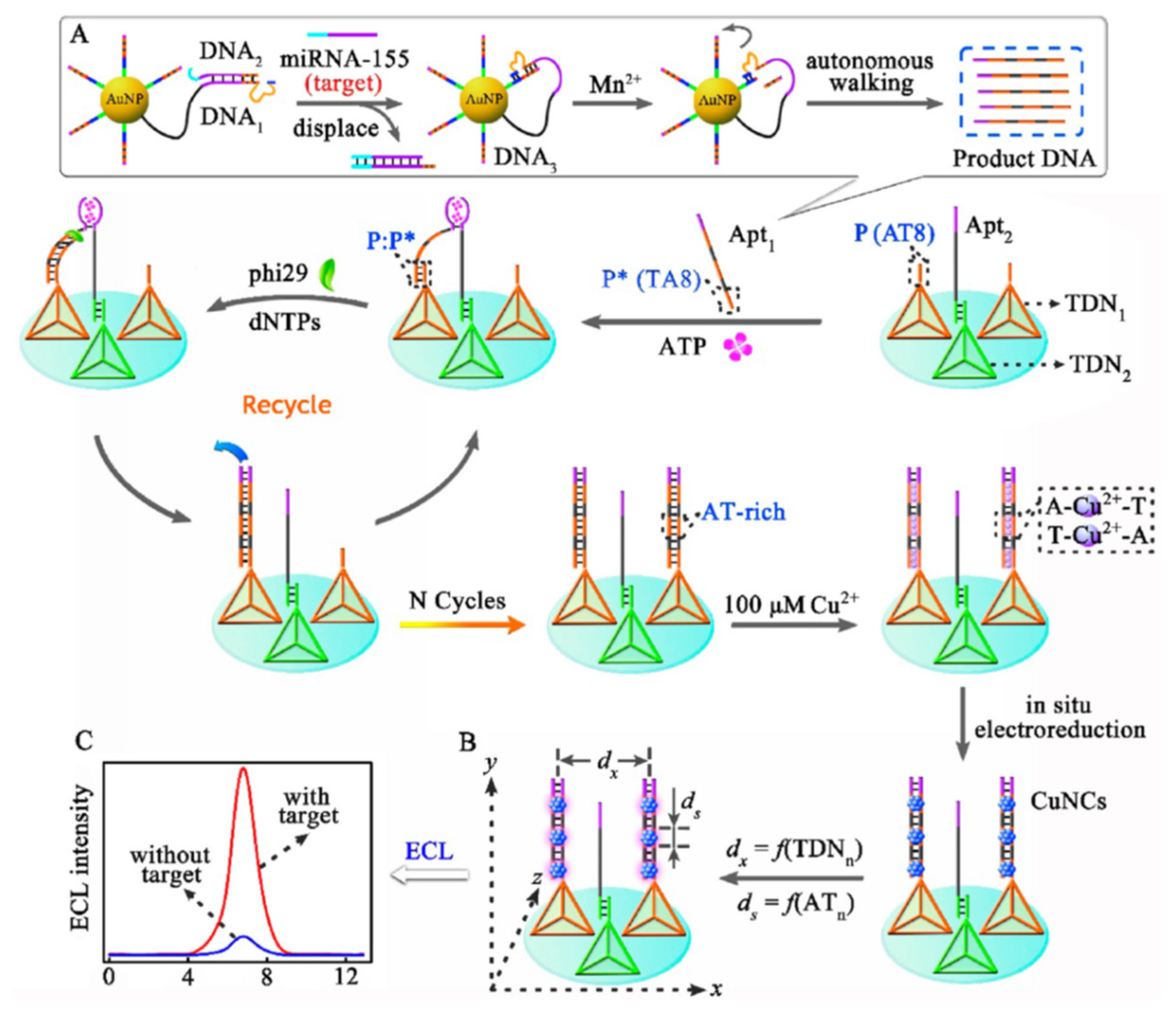
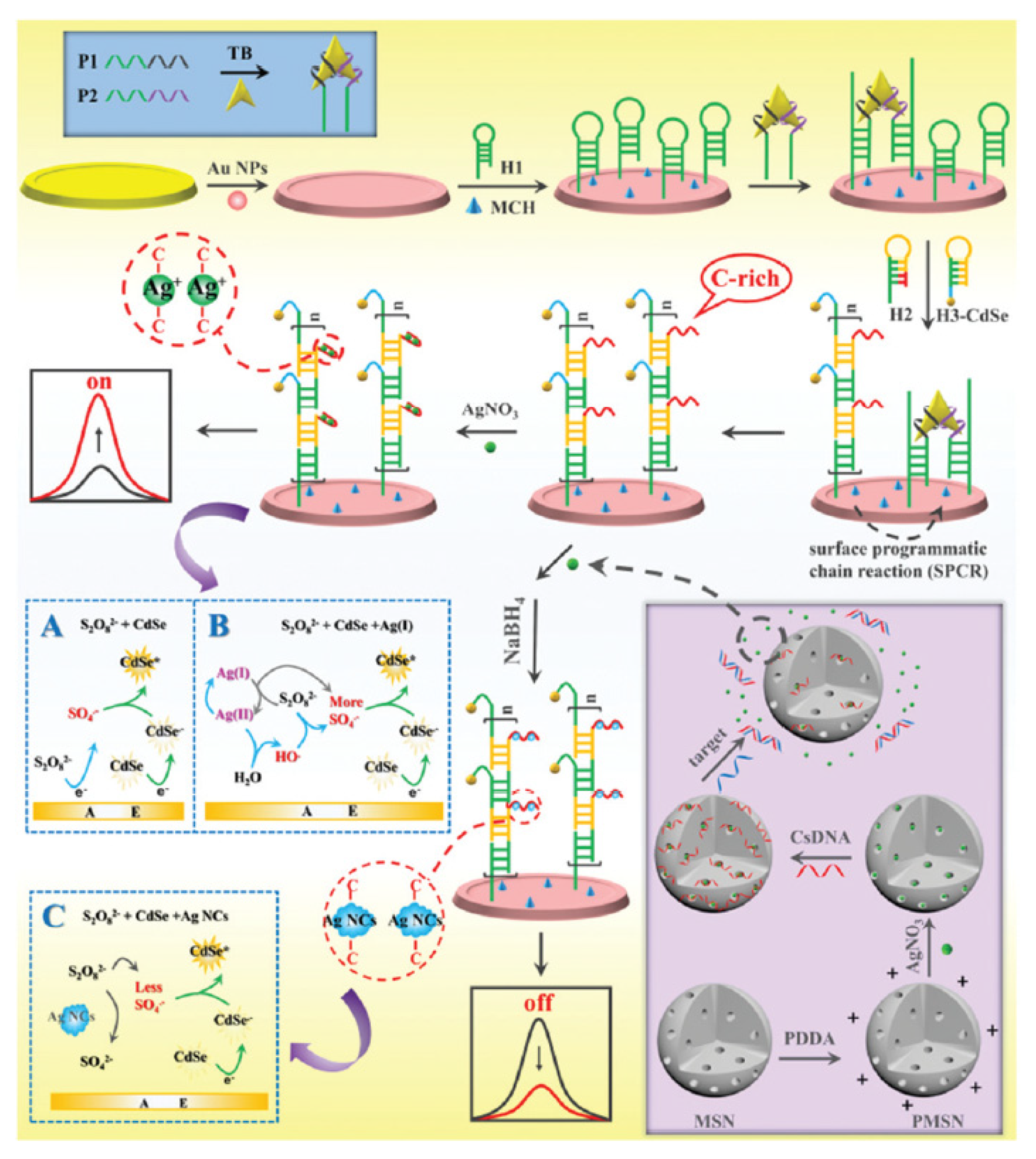
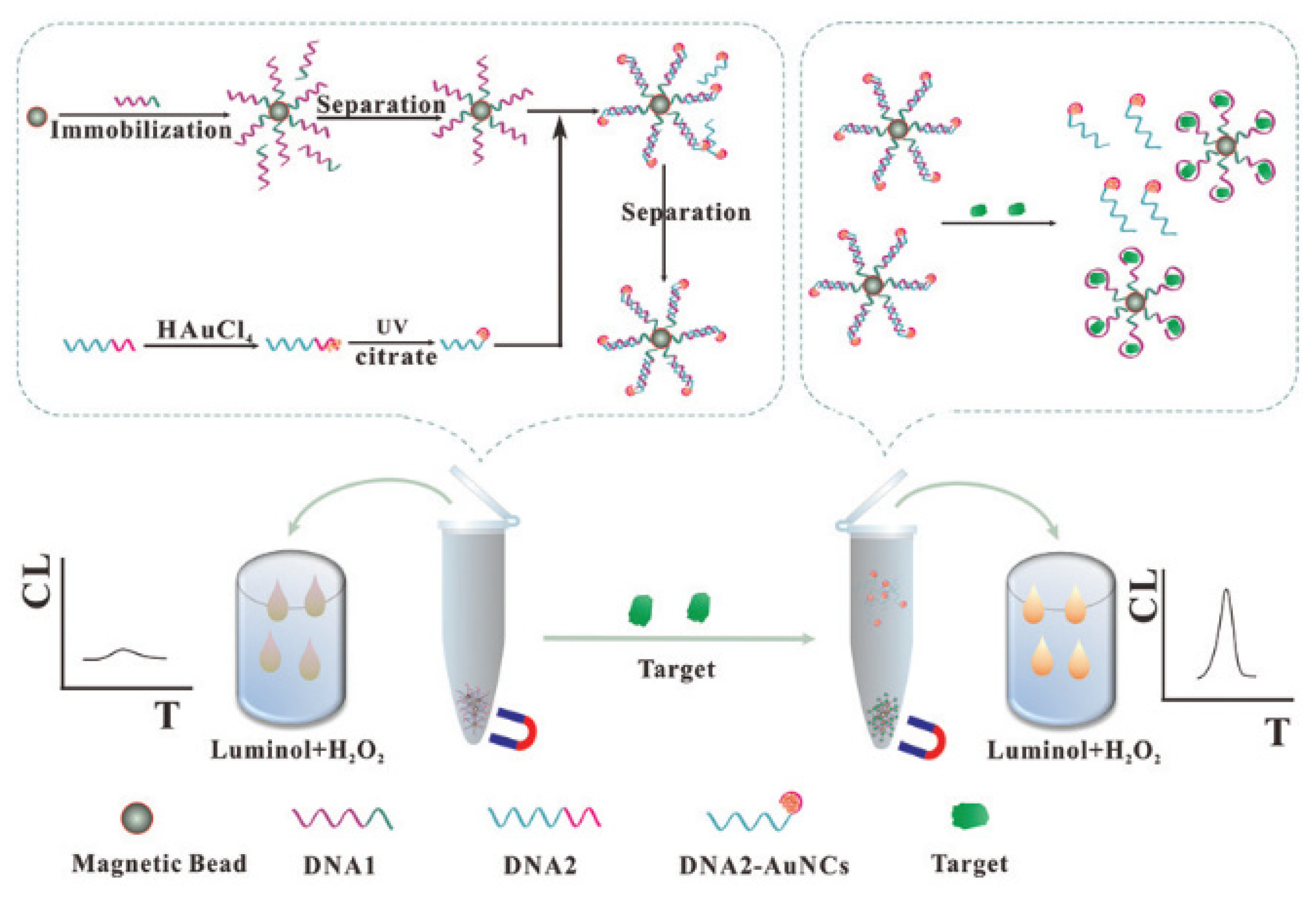
| Position | Methods | ECL System of Au NCs | Ref. |
|---|---|---|---|
| Metal core | Reduction of NCs | N-acetyl-L-cysteine stabilized Au NCs + K2S2O8 | [15] |
| GSH stabilized Au NCs + K2S2O8 | |||
| Pre-oxidation of NCs | L-methionine stabilized Au NCs + TEA | [17] | |
| Dithiothreitol stabilized Au NCs and BSA/Au NCs + TEA | |||
| Doping Strategy of NCs | Doping of Ag into BSA/Au NCs + TEA | [38] | |
| Bimetallic NCs | GSH stabilized AuAg NCs + K2S2O8 | [40] | |
| Au12Ag13 NCs + TPrA | [41] | ||
| Ligand | New Ligand | L-methionine stabilized Au NCs + K2S2O8 | [19] |
| L-methionine stabilized Au NCs + triethanolamine | [53] | ||
| Double Ligand | Thioglycol/glutathione dual ligand-coated Au NCs + TEA | [54] | |
| Rigid Host-Guest Assemblies | Mixed L-arginine (ARG) and 6-aza-2-thiothymine (ATT) protected Au NCs + TPrA | [18] | |
| External environment | Novel coreactants | LA-stabilized Au NCs / EDTA | [55] |
| LA-stabilized Au NCs / HEPES | [56] | ||
| Coreaction accelerator | BSA/AuNCs + K2S2O8, highly-branched Cu2O as the coreaction accelerator | [58] | |
| BSA/AuNCs + TEA, Cu2S snowflake as the coreaction accelerator | [59] | ||
| BSA/AuNCs + K2S2O8, thiocholine produced in situ as the coreaction accelerator | [60] | ||
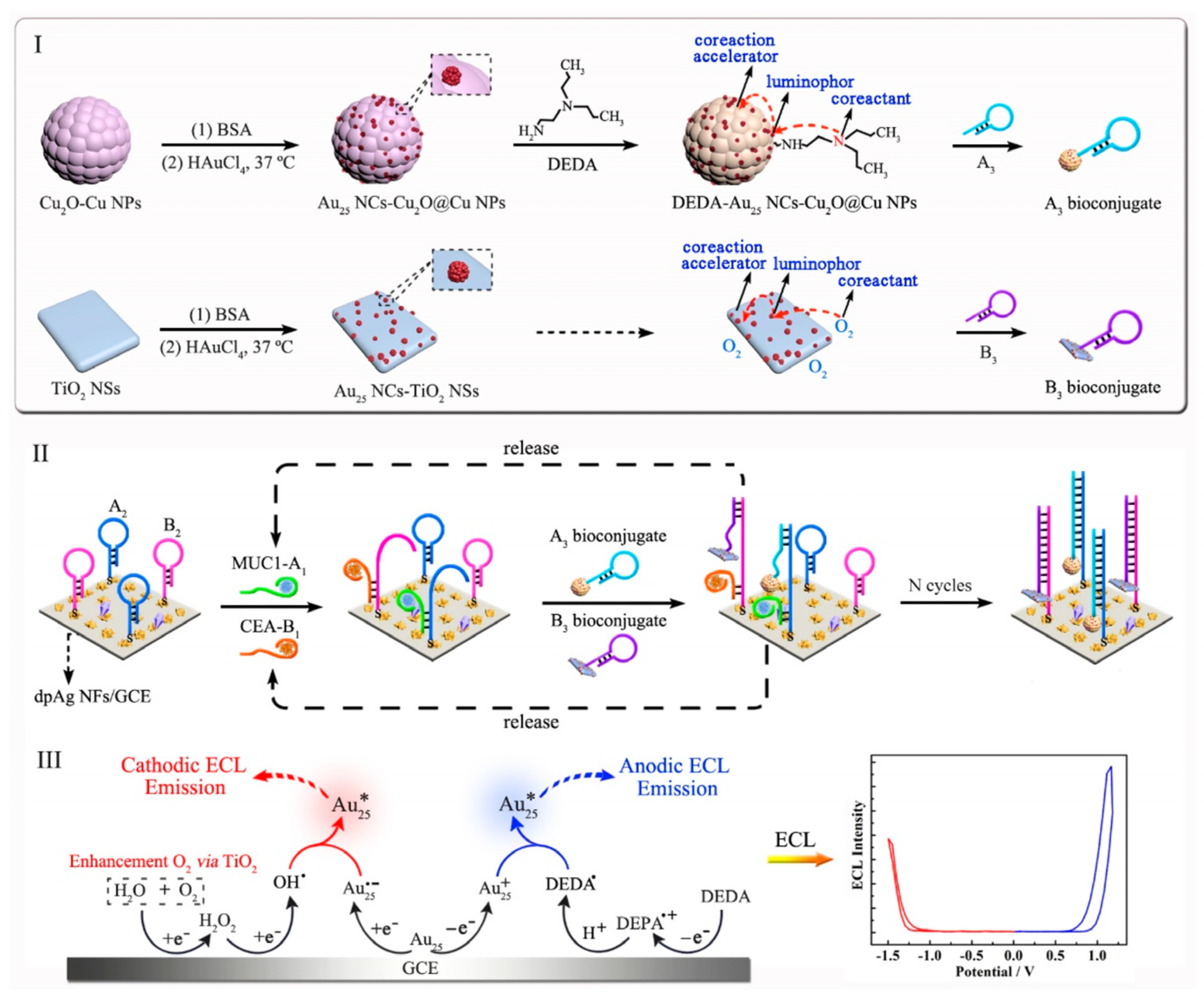
| Intramolecular Electron Transfer: Binary or ternary nanostructure formed by luminophores, coreactants and coreaction accelerators via covalent attachment | Binary nanostructure formed by LA/Au NCs as the luminophore and DEDA as the coreactant | [20] | |
| Binary nanostructure formed by TA/Au NCs as the luminophore and DPEA as the coreactant | [61] | ||
| Ternary nanostructure formed by BSA/Au NCs as the luminophore, TAEA as the coreactant and Pd@CuO nanomaterial as the coreaction accelerator | [57] | ||
| Binary nanostructure formed by BSA/Au NCs as the luminophore, TiO2 nanosheets as the coreaction accelerator with O2 as the coreactant. | [46] | ||
| Ternary nanostructure of BSA/Au NCs- as the luminophore, Cu2O@Cu nanoparticles as the coreaction accelerator, and DEDA as the coreactant. | |||
| Aggregation-Induced ECL | ATT/Au NCs + TEA | [43] | |
| ATP/Au NCs after Ca2+ induced aggregation + HEPES | [44] | ||
| Nanostructure-based Enhancement | BSA/Au NCs + K2S2O8, nanoporous NiGd–Ni2O3–Gd2O3@Au nanoparticles as sensor platform | [62] | |
| Synergistic Effect and ECL resonance energy transfer | Au NCs@ GQDs nanocomposite as the luminophore, GQDs as a donor, Au NCs as an acceptor | [63] | |
| Biological Signal Amplification | Au NCs dual-labeled with hairpin DNA (H1 and H2) + K2S2O8, using HCR signal amplification. | [64] | |
| Position | Methods | ECL system of Ag NCs | Ref. |
| Metal core | Design DNA structure to improve the stability and uniformity of Ag NCs | Triplex DNA templated Ag NCs + Na2S2O8, | [65] |
| External environment | Biological signal amplification | DNA/Ag NCs + K2S2O8, DNAzyme-assisted target recycling and HCR multiple amplification strategy | [66] |
| Coreaction accelerators | Ag NCs + K2S2O8, Fe3O4-CeO2 nanocomposites as coreaction accelerator | [67] | |
| Surface plasmon-enhanced ECL | DNA templated Ag NCs as the luminophore and Au NPs as the localized surface plasmon resonance source | [68] | |
| Metal core | Design DNA structure to improve the stability and less collision of Cu NCs | DNA nanocrane/Cu NCs + K2S2O8 | [71] |
| Recombination | Nanocomposite DTT/Cu NCs / CNNSs + K2S2O8 | [72] | |
| Doping the rich electronic rare earth element into Cu NCs | Eu3+ ion doped GSH stabilized Cu NCs | [73] | |
| External environment | Coreaction accelerators and biological signal amplification | dsDNA/Cu NCs + K2S2O8, TiO2 as coreaction accelerator, exonuclease III-assisted amplification and HCR | [70] |
| Detection Technique | Metal NCs or Probe | Target | Linear Range | LOD | Ref. |
|---|---|---|---|---|---|
| ECL | Graphene/Au NCs + K2S2O8 | H2O2 | 4–24 μM | 2 μM | [10] |
| BSA/Au NCs + K2S2O8 | Dopamine | 2.5–7.5 μM | - | [11] | |
| Graphene/multiwall carbon nanotube/Au NCs + K2S2O8 | Hydroquinone | 1.0–60 μM | 0.3 μM | [12] | |
| Resorcinol | 3.0–70 μM | 1.0 μM | |||
| p-Cresol | 5.0–60 μM | 1.7 μM | |||
| p-Chlorophenol | 4–70 μM | 1.3 μM | |||
| 2-Bromophenol | 5–70 μM | 1.7 μM | |||
| DNA/Ag NCs + K2S2O8 | MicroRNA-21 | 100 aM–100 pM | 22 aM | [13] | |
| BSA/Cu NCs + hydrazine | Dopamine | 1.0 × 10−12–1.0 × 10−8 M | 3.5 × 10−13 M | [14] | |
| Met/Au NCs + K2S2O8 | Dopamine | 0.1–4 μM | 32 nM | [19] | |
| BSA/AuAg NCs + triethylamine | Hg2+ | 10 nM–5 μM | 5 nM | [38] | |
| GSH/AuAg NCs + TEA, Ag NPs as a coreaction accelerator | GSH | 5–200 μM | 0.90 μM | [39] | |
| GSH/AuAg NCs + K2S2O8 | Dopamine | 10 nM–1 mM | 2.3 nM | [40] | |
| Adenosine mono-/di-/triphosphate/Au NCs + TEA | Calmodulin | 0.3–50 μg mL−1 | 0.1 μg mL−1 | [44] | |
| BSA/Au NCs + DEDA | CEA | 1 pg mL−1–1 ng mL−1 | 0.43 pg mL−1 | [46] | |
| MUC1 | 10 fg mL−1–1 ng mL−1 | 5.8 fg mL−1 | |||
| BSA/Au NCs + H2O2 | 5-Methylcytosine-DNA | 0.01–50 nM | 3.46 pM | [47] | |
| Ten-eleven translocation 1 protein | 1–10 μg mL−1 | 0.37 μg mL−1 | |||
| T4 β-glucosyltransferase | 0.5–50 unit mL−1 | 0.176 unit mL−1 | |||
| BSA/Au NCs + TEA | Concanavalin A | 0.004–90 ng mL−1 | 0.001 ng mL−1 | [49] | |
| NAC/Au NCs + K2S2O8 | GSH | 1.0 × 10−9–1.0 × 10−5 M and 1.0 × 10−5–1.0 × 10−1 M | 3.2 × 10−10 M | [50] | |
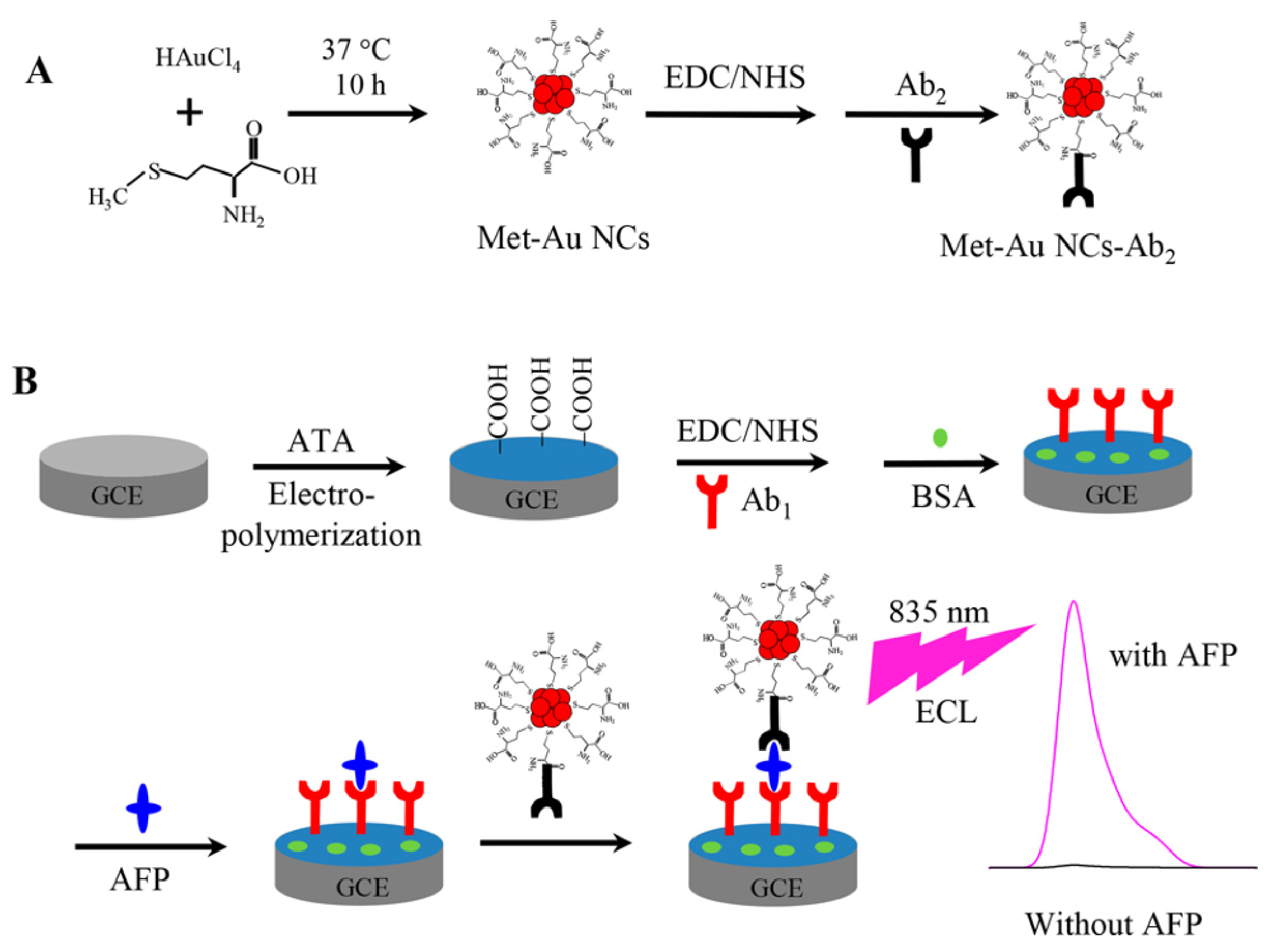
| Met/Au NCs + triethanolamine | α-Fetoprotein | 3 fg mL−1–0.1 ng mL−1 | 1 fg mL−1 | [53] | |
| BSA/Au NCs + TAEA | CEA | 100 fg mL−1–100 ng mL−1 | 16 fg mL−1 | [57] | |
| BSA/Au NCs + K2S2O8 | Procalcitonin | 10 fg mL−1–100 ng mL−1 | 2.90 fg mL−1 | [58] | |
| BSA/Au NCs + TEA | Procalcitonin | 10 fg mL−1–100 ng mL−1 | 2.36 fg mL−1 | [59] | |
| BSA/Au NCs + K2S2O8 | ATCI | 0.50 nM–0.47 mM | 0.17 nM | [60] | |
| TA/Au NCs +DPEA | Mucin 1 | 1 fg mL−1–1 ng mL−1 | 0.54 fg mL−1 | [61] | |
| BSA/Au NCs + K2S2O8 | CEA | 10−4–5 ng mL−1 | 0.03 pg mL−1 | [62] | |
| BSA/Au NCs/GQDs + TEA | Pentoxifylline | 7.0 × 10−7–1.2 × 10−4 M | 9.0 × 10−8 M | [63] | |
| GSH/Au NCs + K2S2O8 | Cardiac troponin I | 5 fg mL−1–50 ng mL−1 | 1.01 fg mL−1 | [64] | |
| Triplex DNA/Ag NCs | Cys | 0.5–50 μM | 0.5 μM | [65] | |
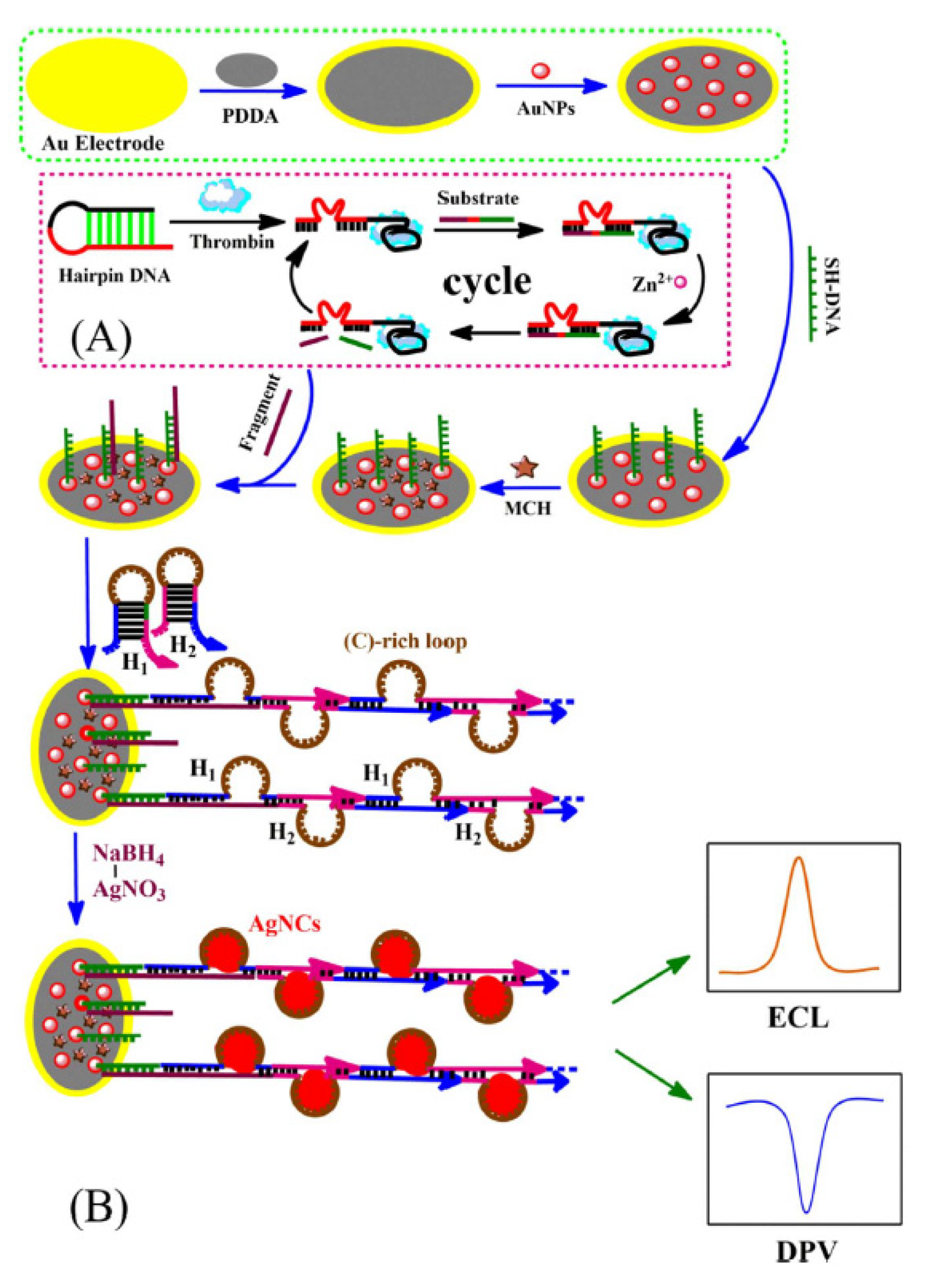
| DNA/Ag NCs + K2S2O8 | Thrombin | 10.0 fM–10.0 nM | 4.5 fM | [66] | |
| Ag NCs + K2S2O8 | CCND1 | 50 fg mL−1–50 ng mL−1 | 28 fg mL−1 | [67] | |
| DNA/AgNCs + K2S2O8 | MicroRNA-21 | 1 aM–104 fM | 0.96 aM | [68] | |
| DNA/Cu NCs + K2S2O8 | MicroRNA-21 | 100 aM–100 pM | 19.05 aM | [70] | |
| DNA/Cu NCs + K2S2O8 | MicroRNA-155 | 100 aM–100 pM | 36 aM | [71] | |
| DTT/Cu NCs + K2S2O8 | Hg2+ | 0.5–10 nM | 0.01 nM | [72] | |
| Eu3+-Cu NCs | Dopamine | 1.0 × 10−11–5.0 × 10−4 M | 1.0 × 10−11 M | [73] | |
| Pt NCs/graphene + TEA | Cu2+ | 1.0 × 10−4–2.0 × 10−1 mg L−1 | 1.0 × 10−4 mg L−1 | [74] | |
| Ni NCs + TPrA | Creatinine | 5 nM–1 mM | 0.5 nM | [75] | |
| DNA/Ag NCs + K2S2O8 | Thrombin | 0.001–1000 pM | 0.165 fM | [76] | |
| MicroRNA-21 | 0–103 pM, 10−5–103 pM | 4.97 aM | |||
| GSH/Au NCs + K2S2O8 | Cardiac troponin I | 50 fg mL−1–50 ng mL− | 9.73 fg mL−1 | [77] | |
| g-C3N4 as a ECL emitter, BSA/Au NCs as catalyst | Protein kinase A | 0.02–20 U mL−1 | 0.005 U mL−1 | [78] | |
| Pt NCs both as the acceptor and donor | MicroRNA-141 | 10 aM–100 nM | 3.3 aM | [79] | |
| CL | Penicillamine/AuCu NCs | H2O2 | 0.2–2000 nM | 0.13 nM | [30] |
| Glucose | 0.1–400 μM | 30 nM | |||
| Xanthine | 0.1–200 μM | 38 nM | |||
| KMnO4 + rhodamine B enhanced by BSA/Au NCs and GQDs | Cimetidine | 0.8–200 ng mL−1 | 0.3 ng mL−1 | [31] | |
| KMnO4 + rhodamine B enhanced by BSA/Ag NCs and GQDs | Rabeprazole | 4–133 ng mL−1 | 1.1 ng mL−1 | [32] | |
| Cysteine/Cu NCs + cerium | Trihexyphenidyl hydrochloride | 0.1–10.0 μM | 49.0 nM | [33] | |
| BSA/Au NCs + KMnO4 | H2O2 | 1.0 ×10−6–1.0 × 10−4M | 5.0×10−7 M | [34] | |
| BSA/AuNCs as an energy acceptor and bis(2,4,6-tri-chlorophenyl)- oxalate–hydrogen peroxide as an energy donor | Trypsin | 0.01–50.0 μg mL−1 | 9 ng mL−1 | [35] | |
| hPEI-AgNCs + H2O2 | Tea polyphenols | 2.52–76.2 μM | 2.52 μM | [36] | |
| H2O2-HCO3− system, CdSe QDs as catalyst, BSA/Au NCs as quencher | Cyanide | 2–225 nM | 0.46 nM | [37] | |
| Au NCs as catalyst | Nitrite | 5 μM–0.1 mM | 4.7 μM | [80] | |
| Au NCs as catalyst | Catechol | 0.1–10 μM | 0.062 μM | [81] | |
| Ag NCs as catalyst | H2O2 | 0.14–100 μM | 0.016 μM | [82] | |
| Uric acid | 2–100 μM | 0.75 μM | |||
| Au NCs as catalyst | Kanamycin | 0.2–4.4 nM | 0.035 nM | [84] | |
| Cu NCs as catalyst | Cholesterol | 0.05–10 mM | 1.5 μM | [85] | |
| Cu NCs as catalyst | Phenylalanine | 1.0 × 10−6–2.7 × 10−5 M | 8.4 × 10−7 M | [86] | |
| Tryptophan | 1.0 × 10−7–3.0 × 10−5 M | 7.5 × 10−8 M | |||
| Cu NCs as catalyst | Tryptophan | 2.0 × 10−7–10−4 M | 6 × 10−8 M | [87] | |
| Cu NCs as catalyst | Nitrite | 1.0–80.0 μM | 0.0954 μM | [88] | |
| Folic acid | 0.1–10.0 μM | 3.0 μM | |||
| Cu NCs@CuMOF as catalyst | Tramadol | 0.0030–2.5 μM | 0.80 nM | [89] | |
| BSA/ZnCu NCs as catalyst | H2O2 | 5.0 × 10−9–1.0 × 10−6 M | 3.0 × 10−10 M | [90] | |
| Hemoglobin/Au NCs as catalyst | Dopamine | 0.3–9.0 nM | 0.1 nM | [91] | |
| Au NCs as catalyst | Bisphenol A | 2.0 × 10−7–1.0 × 10−5 M | 1.0 × 10−6 M | [92] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, S.; Zhao, Y.; Zhang, Z.; Xu, G. Recent Advances in Electrochemiluminescence and Chemiluminescence of Metal Nanoclusters. Molecules 2020, 25, 5208. https://doi.org/10.3390/molecules25215208
Han S, Zhao Y, Zhang Z, Xu G. Recent Advances in Electrochemiluminescence and Chemiluminescence of Metal Nanoclusters. Molecules. 2020; 25(21):5208. https://doi.org/10.3390/molecules25215208
Chicago/Turabian StyleHan, Shuang, Yuhui Zhao, Zhichao Zhang, and Guobao Xu. 2020. "Recent Advances in Electrochemiluminescence and Chemiluminescence of Metal Nanoclusters" Molecules 25, no. 21: 5208. https://doi.org/10.3390/molecules25215208
APA StyleHan, S., Zhao, Y., Zhang, Z., & Xu, G. (2020). Recent Advances in Electrochemiluminescence and Chemiluminescence of Metal Nanoclusters. Molecules, 25(21), 5208. https://doi.org/10.3390/molecules25215208






