3.1. Synthetic Chemistry
3.1.1. General Considerations
All commercially available starting materials were purchased and used as provided unless otherwise specified. When anhydrous conditions are indicated, anhydrous solvents were used from commercial suppliers. Automated flash column chromatography was performed using a Teledyne ISCO (Lincoln, NE, USA) CombiFlash NextGen system with silica gel-packed columns (RediSep® Rf). Analytical thin-layer chromatography (TLC, commercially available from VWR (Radnor, PA, USA)) was carried out on Merck aluminum-supported silica gel plates (thickness: 200 mm) with fluorescent indicator (F-254). Visualization of compounds on TLC plates was accomplished with UV light (254 nm) and/or with phosphomolybdic acid, ninhydrin, ceric ammonium molybdate, or potassium permanganate staining (Sigma-Aldrich, St. Louis, MO, USA). NMR spectra (1H, 13C, 19F and 31P) were obtained in the Emory University NMR Research Center, directed by Dr. Shaoxiong Wu and Dr. Bing Wang, using either a Bruker INFINITY II 600 MHz spectrometer with cryogenic probe (funded by National Science Foundation grant CHE-1531620), a Varian INOVA 500 MHz spectrometer, a Varian INOVA 400 MHz spectrometer, or a Varian VNMR 400 MHz spectrometer. NMR samples were prepared and processed in deuterated chloroform (CDCl3), deuterated MeOH (CD3OD), deuterated dimethyl sulfoxide (d6-DMSO), or deuterated acetone (d6-acetone) using the residual solvent peak (CDCl3: 1H = 7.26 ppm, 13C = 77.16 ppm; CD3OD: 1H = 3.31 ppm, 13C = 49.00 ppm; d6-DMSO: 1H = 2.50 ppm, 13C = 39.52 ppm; d6-acetone: 1H = 2.05 ppm, 13C = 29.84 ppm) as an internal reference. NMR data are reported to include chemical shifts (δ) reported in ppm, multiplicities indicated as s (singlet), d (doublet), dd (doubled doublet), t (triplet), td (tripled doublet), q (quartet), ddd (doubled doubled doublet), m (multiplet), or br s (broad singlet), coupling constants (J) reported in Hz, and integration normalized to 1 atom. High-resolution mass spectrometry (HRMS) was performed by the Emory University Mass Spectrometry Center, directed by Dr. Fred Strobel. Liquid chromatography–mass spectrometry (LC–MS) was performed on an Agilent 1200 HPLC equipped with a 6120 Quadrupole mass spectrometer (ESI–API) eluting at a rate of 1.00 mL/min with mixtures of HPLC grade MeOH and water or acetonitrile and water (all spiked with 0.1% formic acid) through an analytical, reverse-phase Agilent C18 XDB eclipse column (50 mm × 4.6 mm, 3.5 μM). LC–MS samples were prepared in a solution of 75:25 MeOH/water or 50:50 acetonitrile/water (spiked with 0.1% formic acid), and ultraviolet activity was monitored at 254 nm. Final compound purity was assessed to be ≥95% pure using 1H NMR and LC–MS.
3.1.2. Synthesis of the Per-Acetylated Thiosugar Core
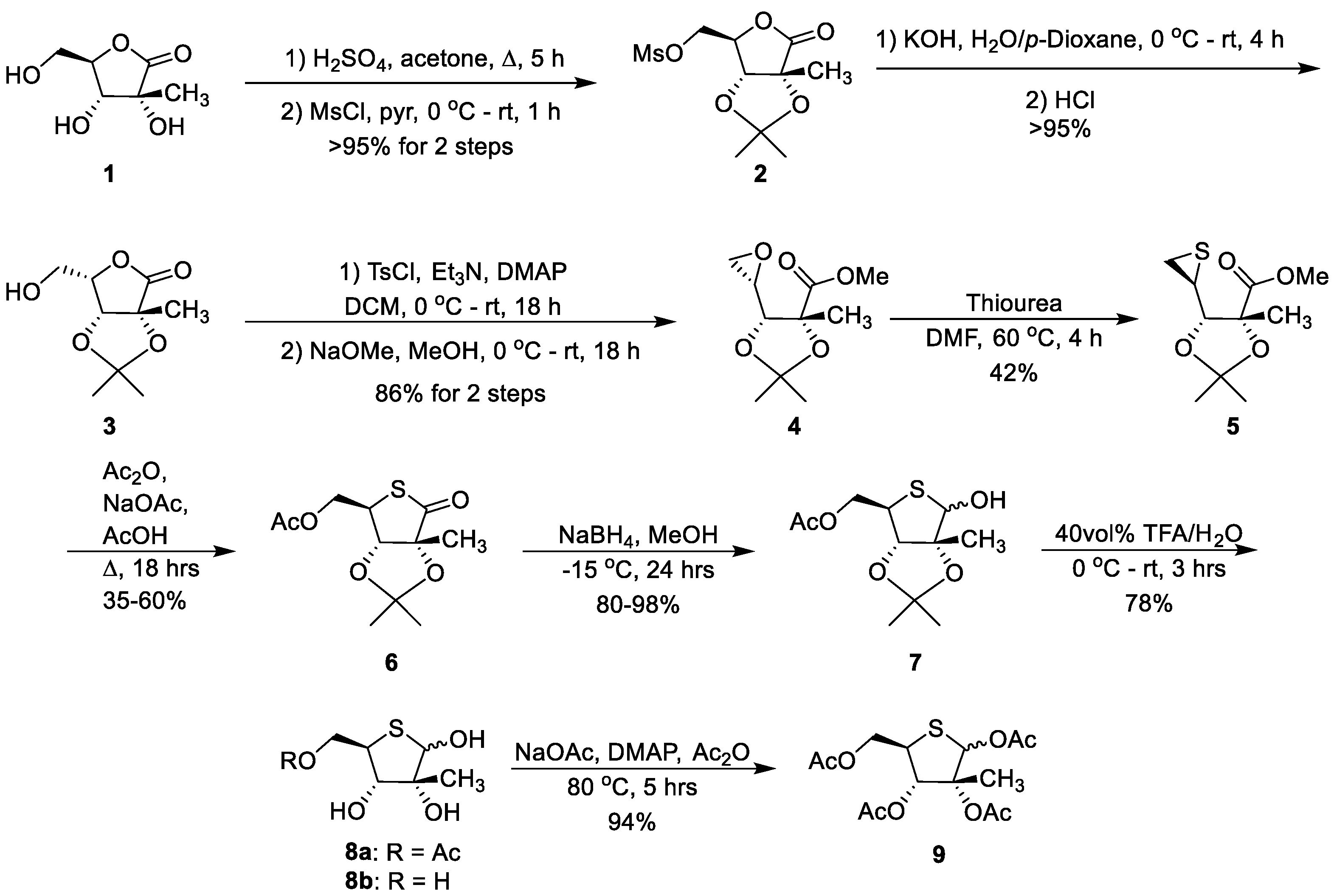
(3a
R,6
R,6a
R)-6-(hydroxymethyl)-2,2,3a-trimethyldihydrofuro [3,4-d][
1,
3]dioxol-4(3a
H)-one (
1.1):
A 500-mL round-bottomed flask equipped with reflux condenser and magnetic stir bar was charged with (3
R,4
R,5
R)-3,4-dihydroxy-5-(hydroxymethyl)-3-methyldihydrofuran-2(3
H)-one (5 g, 30.8 mmol) and acetone (100 mL, 1363 mmol) to give a tan solution. Sulfuric acid (0.25 mL, 4.69 mmol) was added dropwise, and the solution was heated to reflux with stirring. After 4 h, the reaction was cooled with an ice bath and carefully neutralized with solid NaHCO
3 before the reaction mixture was filtered through Celite
®. The crude filtrate was concentrated to afford an off-white solid (5.94 g, 95% yield); NMR spectra were consistent with that previously reported [
56], see
Supplementary Materials.
((3ar,4
r,6ar)-2,2,6a-trimethyl-6-oxotetrahydrofuro[3,4-d][
1,
3]dioxol-4-yl)methyl methanesulfonate (
2):
A 50-mL round-bottomed flask with magnetic stir bar was charged with acetonide
1.1 (5.74 g, 28.4 mmol) and anhydrous pyridine (4.7 mL) under argon to give a colorless solution. The solution was cooled to 0 °C with an ice bath before methanesulfonyl chloride (3.3 mL, 42.6 mmol) was slowly added dropwise with stirring. The reaction was left to stir at 0 °C for approximately 30 min before the ice bath was removed, and the reaction warmed to ambient temperature for another 30 min. Approximately 15 mL of deionized H
2O was added, and the product was extracted with dichloromethane (DCM). The organic layer was washed with 1M HCl followed by saturated NaHCO
3 solution. The organic phase was dried over Na
2SO
4, filtered and concentrated to afford a white solid (7.88 g, 99% yield); NMR spectra were consistent with that previously reported [
56].
(3a
R,6
S,6a
R)-6-(hydroxymethyl)-2,2,3a-trimethyldihydrofuro[3,4-d][
1,
3]dioxol-4(3aH)-one (
3):
A 500-mL round-bottomed flask with magnetic stir bar was charged with mesylate
2 (3.2 g, 11.42 mmol) and 1,4-dioxane (70.5 mL) to give a tan solution. In a separate beaker, solid KOH (1.92 g, 34.2 mmol) was dissolved in deionized water (55.0 mL), and after the heat of dissolution had dissipated the resulting solution was added to the reaction flask with stirring, noting an exotherm. The reaction was allowed to stir at ambient temperature for approximately 4 h. The reaction was acidified with 3 N HCl to pH 1 with stirring, and the crude product was concentrated to dryness. The remaining white solid was triturated with ethyl acetate, and the insoluble material was filtered and further washed with ethyl acetate. The filtrate was concentrated to afford a tan oil (2.29 g, 99% yield); NMR spectra were consistent with those previously reported [
56].
((3a
R,4
S,6a
R)-2,2,6a-trimethyl-6-oxotetrahydrofuro[3,4-d][
1,
3]dioxol-4-yl)methyl 4-methylbenzenesulfonate (
3.1):
A flame-dried 250-mL round-bottom flask with stir bar was charged with argon, lactone
3 (2 g, 9.89 mmol) and DCM (30 mL) to give a colorless solution. The solution was chilled to 0 °C before
N,N-dimethylpyridin-4-amine (0.06 g, 0.495 mmol) and
N-ethyl-
N-isopropylpropan-2-amine (3.5 mL, 19.78 mmol) were added with stirring to give a slightly yellow solution. Lastly, 4-methylbenzene-1-sulfonyl chloride (2.07 g, 10.88 mmol) was slowly added with stirring. The reaction was stirred for 30 min before the ice bath was removed and was then left to stir overnight while warming to ambient temperature. The next morning, TLC (3:2 hexanes:ethyl acetate) indicated the conversion of the starting material to a new UV-active spot, so the reaction was quenched by the addition of cold 1 N HCl. The product was twice extracted with DCM, and the organic extracts were combined and washed with saturated NaHCO
3 solution before being dried over sodium sulfate, filtered and concentrated to afford a brown oil (3.4 g, 93% yield); NMR spectra were consistent with that previously reported [
57].
(4R,5R)-methyl 2,2,4-trimethyl-5-((S)-oxiran-2-yl)-1,3-dioxolane-4-carboxylate (4):
An oven-dried 50-mL round-bottom flask with magnetic stir bar was charged with argon and tosylate 3.1 (3.04 g, 8.53 mmol). The apparatus was chilled with an ice bath before NaOMe (18.8 mL, 9.38 mmol) was added as a solution in methanol dropwise with stirring to give a colorless solution. The reaction was stirred at 0 °C for 1 h before the ice bath was removed, allowing the reaction to warm to ambient temperature while stirring overnight. The reaction was quenched with 10 mL of saturated NH4Cl solution and diluted with 50 mL of deionized water before the product was extracted with ethyl acetate. The organic layers were combined, dried over Na2SO4, filtered and concentrated to afford the crude product, a pale yellow semisolid (1.70 g, 92% yield); 1H NMR (400 MHz, CDCl3) δ 3.76 (s, 3H), 3.58 (d, J = 6.0 Hz, 1H), 3.00 (ddd, J = 6.0, 4.2, 2.7 Hz, 1H), 2.85 (dd, J = 4.8, 4.2 Hz, 1H), 2.71 (dd, J = 4.9, 2.7 Hz, 1H), 1.58 (s, 3H), 1.55 (s, 3H), 1.40 (s, 3H) ppm; 13C NMR (101 MHz, CDCl3) δ 172.7, 111.1, 85.5, 82.3, 52.5, 49.7, 43.5, 26.6, 23.4 ppm.
(4R,5S)-methyl 2,2,4-Trimethyl-5-((S)-thiiran-2-yl)-1,3-dioxolane-4-carboxylate (5):
A 25-mL round-bottom flask with magnetic stir bar was charged with epoxide 4 (0.75 g, 3.47 mmol) and anhydrous DMF (1.88 mL), and the colorless solution was heated to 60 °C. Freshly recrystallized thiourea (0.46 g, 6.00 mmol) was added, and the reaction stirred for 2 h before another half equivalent of thiourea (0.132 g, 1.734 mmol) was added. Monitoring by TLC indicated complete conversion of starting material in 4.5 h. The reaction was quenched by the addition of water and diluted with 25 mL of brine solution before the product was extracted with ethyl acetate. The organic extracts were combined, dried over Na2SO4, filtered and concentrated to afford 1.06 g of crude product. The product mixture was brought up in DCM and filtered over a plug of silica gel, eluting with a 25% ethyl acetate solution in hexanes. The filtrate fractions of interest were concentrated to afford the desired product as a white powder (0.41 g, 51% yield); 1H NMR (400 MHz, CDCl3) δ 3.78 (s, 3H), 3.35 (d, J = 8.4 Hz, 1H), 2.77 (ddd, J = 8.2, 5.9, 4.9 Hz, 1H), 2.53 (dd, J = 6.0, 1.5 Hz, 1H), 2.26 (dd, J = 5.0, 1.4 Hz, 1H), 1.61 (s, 3H), 1.57 (s, 4H), 1.39 (s, 3H) ppm; 13C NMR (101 MHz, CDCl3) δ 172.8, 110.7, 88.9, 84.2, 52.5, 30.2, 26.9, 26.8, 23.8, 22.9 ppm; HRMS (NSI) m/z: [M + H]+ Calcd for C10H17O4S 233.0842; Found 233.0844.
((3a
S,4
R,6a
R)-2,2,6a-trimethyl-6-oxotetrahydrothieno[3,4-d][
1,
3]dioxol-4-yl)methyl acetate (
6):
An oven-dried 250-mL round-bottom flask equipped with stir bar and reflux condenser was charged with thiirane 5 (0.32 g, 1.378 mmol) and sodium acetate (11.75 g, 143 mmol) under argon. Acetic acid (23 mL) was added followed by acetic anhydride (1.3 mL, 13.78 mmol) to give a moist powder. The reaction was heated to reflux, dissolving the mixture, and allowed to stir for 21 h with monitoring by TLC. When the reaction was pulled from heat, the solution solidified. The solid was dissolved by the addition of DCM and careful addition of saturated NaHCO3 solution. The product was extracted with DCM, and the organic extracts were combined, dried over Na2SO4, filtered and concentrated to afford 0.3 g of a brown oil, which was purified via silica gel flash column chromatography (eluted with a 0–15% gradient of ethyl acetate in hexanes). The fractions of interest were combined to afford the desired five-membered product (0.23 g, 65% yield); 1H NMR (400 MHz, CDCl3) δ 4.33 (d, J = 0.7 Hz, 1H), 4.31 (d, J = 6.3 Hz, 1H), 4.31 (d, J = 7.0 Hz, 1H), 4.01 (ddd, J = 7.0, 6.3, 0.7 Hz, 1H), 2.12 (s, 3H), 1.54 (s, 3H), 1.44 (d, J = 0.8 Hz, 3H), 1.40 (d, J = 0.8 Hz, 3H) ppm; 13C NMR (101 MHz, CDCl3) δ 206.1, 170.4, 111.9, 89.9, 82.9, 64.7, 46.2, 27.5, 26.7, 20.9, 20.3 ppm; HRMS (NSI) m/z: [M + Na]+ Calcd for C11H16O5SNa 283.0611; Found 283.0615.
((3a
S,4
R,6a
R)-6-hydroxy-2,2,6a-trimethyltetrahydrothieno[3,4-d][
1,
3]dioxol-4-yl)methyl acetate (
7):
A 250-mL round-bottom flask with magnetic stir bar was charged with thiolactone 6 (0.606 g, 2.328 mmol) and methanol (36 mL) under argon to give a colorless solution. The solution was chilled to −15 °C before NaBH4 (0.176 g, 4.66 mmol) was added under argon with stirring. After one hour, another portion of NaBH4 (0.176 g, 4.66 mmol) was added; two additional portions of the reductant (0.176 g, 4.66 mmol each) were added on the hour for a total of 8 equivs, and the reaction was allowed to stir at −15 °C with monitoring by LC–MS. After the starting material was consumed, the reaction was quenched at −15 °C by the addition of 5 wt% aqueous citric acid solution. The product mixture was then extracted with DCM, and the organic extracts were combined, dried over Na2SO4, filtered and concentrated to afford a lightly colored oil as the crude product, a 7:3 mixture of anomers (0.56 g, 96% yield); 1H NMR (400 MHz, CDCl3) δ 5.11 (d, J = 4.1 Hz, 0.7H, major), 5.10 (d, J = 9.3 Hz, 0.3H, minor), 4.53 (s, 0.7H, major), 4.40 (d, J = 2.0 Hz, 0.3H, minor), 4.35 (d, J = 5.7 Hz, 0.3H, minor), 4.33 (d, J = 5.7 Hz, 0.3H, minor), 4.20–4.13 (m, 1.4H), 3.55–3.51 (m, 0.3H, minor), 3.50–3.46 (m, 0.7H, major), 3.25 (d, J = 9.3 Hz, 0.3H, minor), 2.88 (d, J = 4.3 Hz, 0.7H, major), 2.09 (s, 2.1H, major), 2.08 (s, 0.9H, minor), 1.565 (s, 0.9H, minor), 1.560 (s, 2.1H, major), 1.54 (s, 0.9H, minor), 1.49 (s, 2.1H, major), 1.44 (s, 0.9H, minor), 1.40 (s, 2.1H, major) ppm; 13C NMR (101 MHz, CDCl3) δ 170.9 (major), 170.6 (minor), 113.1 (minor), 111.7 (major), 96.7 (major), 91.9 (major), 91.3 (minor), 90.3 (minor), 88.8 (major), 86.1 (minor), 66.5 (major), 65.7 (minor), 53.9 (major), 48.8 (minor), 28.1 (2C, major), 27.5 (minor), 27.1 (minor), 24.4 (minor), 22.4 (major), 21.1 (major), 21.0 (minor) ppm; HRMS (NSI) m/z: [M + Na]+ Calcd for C11H18O5SNa 285.0767; Found 285.0770.
((2R,3S,4R)-3,4,5-trihydroxy-4-methyltetrahydrothiophen-2-yl)methyl acetate (8a):
A 250-mL round-bottom flask with magnetic stir bar was charged with thiolactol 7 (0.68 g, 2.59 mmol) and 20 mL of an ice-cold 40 vol% aqueous solution of trifluoroacetic acid. The reaction was stirred at ambient temperature for 3 h when TLC (1:4 Hex:EA) showed conversion of starting material to two more polar products. The reaction mixture was concentrated via rotary evaporation followed by azeotropic removal of residual acid via co-evaporation with toluene followed by methanol then ethanol. The resulting oil was purified via silica gel flash column chromatography (eluted with a gradient of 0–15% methanol in ethyl acetate). The fractions of interest were combined for the desired 1,2,3-glycol ([M + Na]+ = 245.0) as a 3:2 mixture of anomers (later fractions were also collected for the 5-deacetylated product (8b, [M + Na]+ = 203.0)) (0.52 g, 90% yield); 1H NMR (399 MHz, CD3OD) δ 5.20 (s, 0.4H, minor), 4.82 (s, 0.6H, major), 4.61–4.47 (m, 1H, both), 4.12 (dd, J = 11.0, 8.2 Hz, 0.6H, major), 4.02 (dd, J = 11.2, 7.9 Hz, 0.4H, minor), 3.80–3.70 (m, 1H, both), 3.53–3.43 (m, 1H, both), 2.05 (s, 1.8H, major), 2.04 (s, 1.2H, minor), 1.33 (s, 1.8H, major), 1.28 (s, 1.2H, minor) ppm; 13C NMR (100 MHz, CD3OD) δ 172.7 (major), 172.6 (minor), 85.6 (major), 85.3 (minor), 82.8 (major), 82.7 (minor), 79.5 (minor), 79.3 (major), 68.3 (major), 67.2 (minor), 49.8 (minor), 48.7 (major), 22.3 (both), 20.9 (major), 20.7 (minor) ppm; HRMS (NSI) m/z: [M + Na]+ Calcd for C8H14O5SNa 245.0454; Found 245.0455.
(3R,4S,5R)-5-(acetoxymethyl)-3-methyltetrahydrothiophene-2,3,4-triyl triacetate (9):
A 50-mL round-bottom flask with magnetic stir bar was charged with a mixture of thioribofuranose derivatives 8a and 8b (0.874 g, 3.93 mmol) which was dried in vacuo overnight. Solid anhydrous NaOAc (0.419 g, 5.11 mmol) was added under argon followed by acetic anhydride (3.7 mL, 39.3 mmol) to give a colorless slurry; finally N,N-dimethylpyridin-4-amine (48 mg, 0.393 mmol) was added. The reaction was heated to 80 °C and allowed to stir approximately 4 h with monitoring by TLC for consumption of starting material. The reaction was quenched by careful addition of saturated aqueous sodium bicarbonate solution. The product was extracted with DCM; the organic layers were combined, dried over Na2SO4, filtered and concentrated to a thin yellow-brown oil. The oil was brought up in DCM and purified via silica gel flash column chromatography (eluted with a 0–30% gradient of ethyl acetate in hexanes). The fractions of interest were combined and concentrated to afford the desired product as a 2:1 mixture of anomers (1.29 g, 94% yield); 1H NMR (400 MHz, CDCl3) δ 6.44 (s, 0.7H, major), 6.28 (s, 0.3H, minor), 5.35 (d, J = 3.2 Hz, 0.3H, minor), 5.34 (d, J = 9.3 Hz, 0.7H, major), 4.36–4.38 (m, 1.4H), 4.17–4.09 (m, 1.4H), 3.80–3.72 (m, 1.4H), 2.18 (s, 2H, major), 2.11 (s, 2H, major), 2.10 (s, 3H, both), 2.083 (s, 1H, minor), 2.078 (s, 1H, minor), 2.04 (s, 2H, major), 2.03 (s, 1H, minor), 1.74 (s, 1H, minor), 1.58 (s, 2H, major) ppm; 13C NMR (151 MHz, CDCl3) δ 170.5, 170.2, 169.8, 169.3, 88.0, 78.8, 78.0, 65.5, 44.8, 22.1, 21.3, 21.0, 20.8, 17.0 ppm; HRMS (ESI) m/z: [M + Na]+ Calcd for C14H20O8SNa 371.0771; Found 371.0768.
3.1.3. Synthesis of the 4′-Thionucleosides
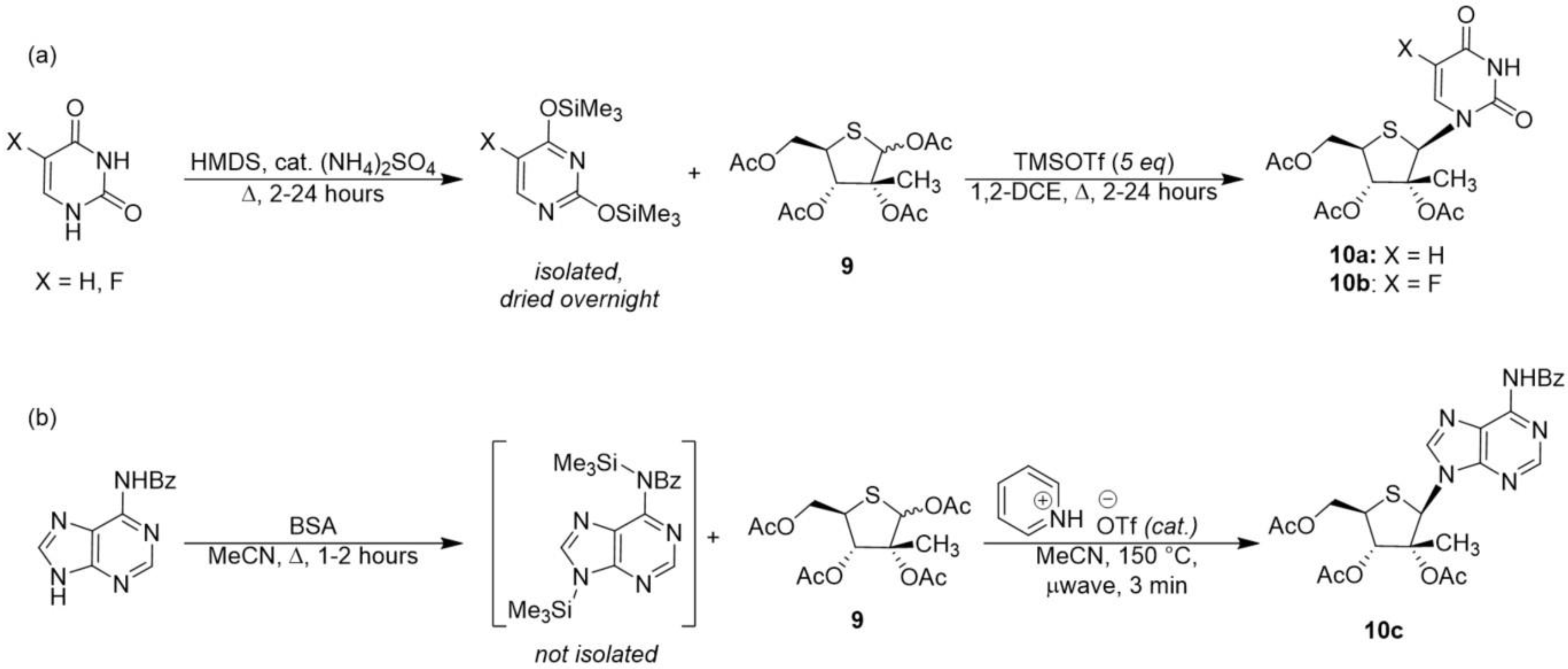
General Procedure for Nucleoside Synthesis Using HMDS and TMSOTf
The nucleobase of interest (5–6 eq) was suspended in anhydrous hexamethyldisilazane (HMDS, 0.5 M in nucleobase) with catalytic ammonium sulfate (7.5 mol% relative to nucleobase) and heated to reflux under argon until the reaction clarified (1–2 h). The reaction mixture was then carefully concentrated to dryness and placed under high vacuum overnight, after which time the silylated intermediate was brought up in 1,2-DCE (1 M in nucleobase) under argon and treated with (3R,4S,5R)-5-(acetoxymethyl)-3-methyltetrahydrothiophene-2,3,4-triyl triacetate 9 (1 eq) and TMSOTf, (5.5 eq). The reaction was heated to 60 °C and left to stir until starting material was consumed by TLC. The reaction was allowed to cool to ambient temperature before being quenched with a saturated aqueous solution of NaHCO3, and the product was extracted with DCM. The organic layer was dried over Na2SO4, filtered, concentrated and purified via silica gel flash column chromatography (eluted with 0–10% methanol gradient in DCM) to afford the glycosylated product, which was subsequently deprotected by stirring with a 7 N solution of ammonia in methanol overnight in a sealed vessel. The next day, the volatiles were removed, and the residue was purified via silica gel flash column chromatography (eluted with 0–15% methanol gradient in DCM) to afford the desired 4′-thionucleoside analog.
1-((2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)-3-methyltetrahydrothiophen-2-yl)pyrimidine-2,4(1H,3H)-dione (11a):
Following the general procedure, uracil (0.78 g, 6.95 mmol) was glycosylated with 9 (484 mg, 1.39 mmol) and subsequently deprotected to afford the desired product (245 mg, 64% yield over two steps); 1H NMR (399 MHz, CD3OD) δ 8.36 (d, J = 8.2 Hz, 1H), 5.95 (s, 1H), 5.75 (d, J = 8.1 Hz, 1H), 3.99–3.90 (m, 2H), 3.73 (d, J = 9.5 Hz, 1H), 3.48 (ddd, J = 9.4, 4.7, 2.9 Hz, 1H), 1.17 (s, 3H) ppm; 13C NMR (100 MHz, CD3OD) δ 165.4, 152.8, 143.8, 102.6, 83.2, 77.3, 68.0, 61.6, 53.6, 21.0 ppm; HRMS (NSI) m/z: [M + Na]+ Calcd for C10H14O5N2SNa 297.0516; Found 297.0508.
1-((2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)-3-methyltetrahydrothiophen-2-yl)-5-fluoropyrimidine-2,4(1H,3H)-dione (11b):
Following the general procedure, 5-fluorouracil (195 mg, 1.50 mmol) was glycosylated with 9 (80 mg, 0.23 mmol) and subsequently deprotected to afford the desired product (50 mg, 75% yield over two steps); 1H NMR (399 MHz, CD3OD) δ 8.72 (d, 3JH–F = 7.4 Hz, 1H), 5.90 (d, 5JH–F = 1.5 Hz, 1H), 3.98 (dd, J = 11.9, 3.6 Hz, 1H), 3.90 (dd, J = 11.8, 2.5 Hz, 1H), 3.79 (d, J = 9.4 Hz, 1H), 3.44 (ddd, J = 9.4, 3.5, 2.5 Hz, 1H), 1.20 (s, 3H) ppm; 13C NMR (100 MHz, CD3OD) δ 159.3 (d, 2JC–F = 26.1 Hz), 151.7, 141.2 (d, 1JC–F = 233.6 Hz), 127.8 (d, 2JC–F = 35.8 Hz), 83.4, 76.7, 68.9, 60.6, 53.4, 20.7 ppm; 19F NMR (376 MHz, CD3OD) δ-167.3 (d, 3JF–H = 6.9 Hz) ppm; HRMS (NSI) m/z: [M – H]- Calcd for C10H12O5N2FS 291.0456; Found 291.0458.
4-amino-1-((2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)-3-methyltetrahydrothiophen-2-yl)-5-fluoropyrimidin-2(1H)-one (11e):
Following the general procedure, N4-benzoyl-5-fluorocytosine (380 mg, 1.63 mmol) was glycosylated with 9 (97 mg, 0.28 mmol) and subsequently deprotected to afford the desired product (67 mg, 83% yield over two steps); 1H NMR (399 MHz, CD3OD) δ 8.65 (d, 3JH–F = 7.3 Hz, 1H), 6.02 (d, 5JH–F = 1.9 Hz, 1H), 3.98 (dd, J = 11.9, 3.6 Hz, 1H), 3.91 (dd, J = 11.9, 2.5 Hz, 1H), 3.78 (d, J = 9.4 Hz, 1H), 3.45 (ddd, J = 9.4, 3.6, 2.6 Hz, 1H), 1.15 (s, 3H) ppm; 13C NMR (100 MHz, CD3OD) δ 159.2 (d, 2JC–F = 14.0 Hz), 157.4, 137.9 (d, 1JC–F = 243.7 Hz), 128.4 (d, 2JC–F = 33.1 Hz), 83.4, 76.7, 69.3, 60.8, 53.3, 20.7 ppm; 19F NMR (376 MHz, CD3OD) δ-166.8 (d, 3JF–H = 7.2 Hz) ppm; HRMS (NSI) m/z: [M – H]− Calcd for C10H13O4N3FS 290.0616; Found 290.0619.
4-amino-1-((2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)-3-methyltetrahydrothiophen-2-yl)pyrimidin-2(1H)-one (11f):
Following the general procedure, N4-acetylcytosine (0.5 g, 3.27 mmol) was glycosylated with 9 (256 mg, 0.74 mmol) and subsequently deprotected to afford the desired product (104 mg, 52% yield over two steps); 1H NMR (400 MHz, CD3OD) δ 8.36 (d, J = 7.6 Hz, 1H), 6.11 (s, 1H), 5.93 (d, J = 7.5 Hz, 1H), 3.97–3.92 (m, 2H), 3.72 (d, J = 9.5 Hz, 1H), 3.47 (dt, J = 9.5, 3.6 Hz, 2H), 1.12 (s, 3H) ppm; 13C NMR (101 MHz, CD3OD) δ 167.1, 159.0, 144.4, 96.2, 83.4, 77.2, 68.6, 61.5, 53.3, 20.7 ppm; HRMS (NSI) m/z: [M + H]+ Calcd for C10H16O4N3S 274.0856; Found 274.0855.
General procedure for Nucleoside Synthesis via the Microwave-Assisted Glycosylation of Nucleobases with BSA and Pyridinium Triflate.
The nucleobase of interest (1.1 eq) was suspended in anhydrous acetonitrile (0.2 M in nucleobase) under argon and treated with N,O-bis(trimethylsilyl)acetamide (2.2 eq relative to nucleobase), and the reaction was heated to 75 °C for 1–2 h. Meanwhile, an oven-dried microwave vial was charged with argon, catalytic pyridinium triflate (5 mol% relative to glycosyl donor), and (3R,4S,5R)-5-(acetoxymethyl)-3-methyltetrahydrothiophene-2,3,4-triyl triacetate 9 (1 eq) as a 0.4 M solution in anhydrous acetonitrile. After cooling to ambient temperature, the crude silylated intermediate was delivered to the microwave vial under argon. The suspension stirred briefly before being irradiated to 150 °C until starting material was consumed by TLC. The reaction mixture was concentrated to dryness and purified via silica gel flash column chromatography (eluted with 0–10% methanol gradient in DCM) to afford the glycosylated product, which was subsequently deprotected by stirring with a 7 N solution of ammonia in methanol overnight in a sealed vessel. The next day, the volatiles were removed, and the residue was purified via silica gel flash column chromatography (eluted with 0–15% methanol in DCM) to afford the desired 4′-thionucleoside analog.
(2R,3R,4S,5R)-2-(6-amino-9H-purin-9-yl)-5-(hydroxymethyl)-3-methyltetrahydrothiophene-3,4-diol (11c):
Following the microwave-assisted general procedure, N6-benzoyladenine (52 mg, 0.22 mmol) was glycosylated with 9 (70 mg, 0.2 mmol) and subsequently deprotected to afford the desired product (42 mg, 71% yield over two steps); 1H NMR (400 MHz, CD3OD) δ 8.68 (s, 1H), 8.22 (s, 1H), 5.83 (s, 1H), 4.10 (d, J = 9.4 Hz, 1H), 4.05–3.97 (m, 2H), 3.61 (ddd, J = 9.4, 4.2, 3.2 Hz, 1H), 0.92 (s, 3H) ppm; 13C NMR (101 MHz, CD3OD) δ 157.4, 153.9, 151.1, 142.2, 83.4, 77.1, 66.4, 61.8, 53.5, 21.0 ppm; HRMS (NSI) m/z: [M + H]+ Calcd for C11H16O3N5S 298.0968; Found 298.0969.
2-((2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)-3-methyltetrahydrothiophen-2-yl)-1,2,4-triazine-3,5(2H,4H)-dione (11d):
Following the microwave-assisted general procedure, 6-azauracil (18 mg, 0.158 mmol) was glycosylated with 9 (42 mg, 0.12 mmol) and subsequently deprotected to afford the desired product (20 mg, 65% yield over two steps); 1H NMR (400 MHz, CD3OD) δ 7.52 (s, 1H), 6.01 (s, 1H), 4.07 (dd, J = 10.9, 3.7 Hz, 1H), 3.98 (d, J = 8.9 Hz, 1H), 3.74 (dd, J = 10.9, 8.6 Hz, 1H), 3.51 (td, J = 8.7, 3.7 Hz, 1H), 1.19 (s, 3H) ppm; 13C NMR (126 MHz, CD3OD) δ 158.3, 150.3, 137.3, 83.4, 79.4, 69.8, 65.8, 53.9, 20.7 ppm; HRMS (NSI) m/z: [M – H]– Calcd for C9H12O5N3S 274.0492; Found 274.0505.
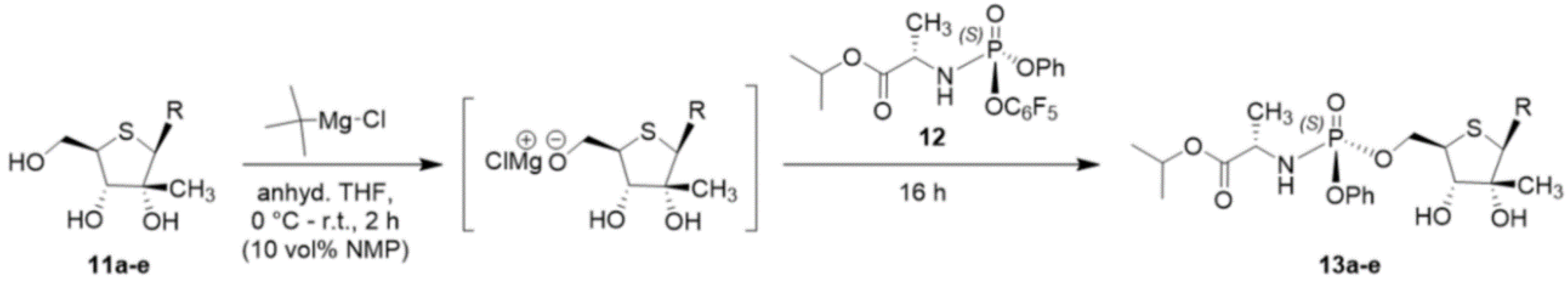
3.1.4. Synthesis of the Phosphoramidate Monophosphate Prodrugs
General Procedure for the Phosphoramidate Coupling Reaction
The 4′-thionucleoside of interest (1 eq) was dried in vacuo overnight and then dissolved in anhydrous THF (0.2 M) with or without 10 vol%
N-methylpyrrolidone co-solvent, depending on solubility. The solution was cooled to 0 °C before being dropwise treated with tert-butylmagnesium chloride (1.2–2 eq) as a 1 M solution in THF under argon, resulting in a thick white slurry. The ice bath was removed, and the deprotonation proceeded while slowly warming to ambient temperature over 2–3 h. The intermediate magnesium alkoxide was then dropwise treated with (
S)-isopropyl 2-(((
S)-(perfluorophenoxy)(phenoxy)phosphoryl)amino)propanoate
12 [
39] (1–2 eq) as a 0.4 M solution in anhydrous THF, and the resulting slurry stirred at ambient temperature overnight. The next day, the reaction was quenched by the addition of saturated aqueous NH
4Cl solution, and the product was extracted with DCM. The organic layers were combined, dried over Na
2SO
4, filtered, concentrated and purified via silica gel flash column chromatography (eluting with 0–15% methanol in DCM) to afford the desired nucleoside phosphoramidate.
(2S)-isopropyl 2-(((((2R,3S,4R,5R)-5-(2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-3,4-dihydroxy-4-methyltetrahydrothiophen-2-yl)methoxy)(phenoxy)phosphoryl)amino)propanoate (13a):
Following the general phosphoramidation procedure, thionucleoside 11a (51 mg, 0.19 mmol) was treated with 12 (84 mg, 0.19 mmol) to afford the desired product (45 mg, 45% yield); 1H NMR (600 MHz, CD3OD) δ 8.08 (d, J = 8.2 Hz, 1H), 7.37 (t, J = 7.9 Hz, 2H), 7.29–7.26 (m, 2H), 7.23–7.17 (m, 1H), 5.99 (s, 1H), 5.68 (d, J = 8.1 Hz, 1H), 4.97 (app hept, J = 6.3 Hz, 1H), 4.56–4.53 (m, 1H), 4.49–4.46 (m, 1H), 3.93 (dq, J = 9.7, 7.1 Hz, 1H), 3.70 (d, J = 9.6 Hz, 1H), 3.68–3.65 (m, 1H), 1.36 (d, J = 7.1 Hz, 3H), 1.22 (d, J = 6.3 Hz, 6H), 1.17 (s, 3H) ppm; 13C NMR (151 MHz, CD3OD) δ 174.4 (d, 3JC–P = 5.4 Hz), 165.7, 152.9, 152.1 (d, 2JC–P = 6.7 Hz), 143.6, 130.9, 126.2, 121.3 (d, 3JC–P = 4.9 Hz), 102.9, 82.8, 77.6, 70.2, 68.2, 67.6 (d, 2JC–P = 5.1 Hz), 51.7, 51.1 (d, 2JC–P = 8.4 Hz), 22.0, 21.9, 20.7, 20.6 ppm; 31P NMR (121 MHz, CD3OD) δ 3.6 ppm; HRMS (ESI) m/z: [M + H]+ Calcd for C22H31N3O9PS 544.1513; Found 544.1511.
(S)-isopropyl 2-(((S)-(((2R,3S,4R,5R)-5-(5-fluoro-2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-3,4-dihydroxy-4-methyltetrahydrothiophen-2-yl)methoxy)(phenoxy)phosphoryl)amino)propanoate (13b):
Following the general phosphoramidation procedure, thionucleoside 11b (38 mg, 0.13 mmol) was treated with 12 (59 mg, 0.13 mmol) to afford the desired product (16 mg, 22% yield); 1H NMR (500 MHz, CD3OD) δ 8.21 (d, 3JH–F = 6.8 Hz, 1H), 7.37–7.324(m, 2H), 7.27–7.25 (m, 2H), 7.20–7.17 (m, 1H), 5.96 (d, 5JH–F = 1.7 Hz, 1H), 4.97 (app hept, J = 6.2 Hz, 1H), 4.55–4.52 (m, 1H), 4.50–4.46 (m, 1H), 3.92 (dq, J = 10.0, 7.1 Hz, 1H), 3.71 (d, J = 9.6 Hz, 1H), 3.67–3.63 (m, 1H), 1.35 (d, J = 7.1 Hz, 3H), 1.22 (d, J = 6.3 Hz, 6H), 1.20 (s, 3H) ppm; 13C NMR (151 MHz, MeOD) δ 174.3 (d, 3JC–P = 5.4 Hz), 159.0 (d, 2JC–F = 26.4 Hz), 152.0 (d, 2JC–P = 6.5 Hz), 151.5, 141.2 (d, 1JC–F = 235.4 Hz), 130.7, 127.1 (d, 2JC–F = 35.0 Hz), 126.1, 121.3 (d, 3JC–P = 4.8 Hz), 82.7, 77.4, 70.1, 68.6, 67.3 (d, 2JC–P = 4.8 Hz), 51.6, 51.0 (d, 2JC–P = 8.6 Hz), 22.0, 21.9, 20.62, 20.59 (d, 3JC–P = 2.1 Hz) ppm; 19F NMR (282 MHz, CD3OD) δ-165.6 (d, 3JF–H = 6.3 Hz); 31P NMR (121 MHz, CD3OD) δ 3.7 ppm; HRMS (NSI) m/z: [M + H]+ Calcd for C22H30N3O9FPS 562.1419; Found 562.1411.
(S)-isopropyl 2-(((S)-(((2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-dihydroxy-4-methyltetrahydrothiophen-2-yl)methoxy)(phenoxy)phosphoryl)amino)propanoate (13c):
Following the general phosphoramidation procedure, thionucleoside 11c (86 mg, 0.29 mmol) was treated with 12 (131 mg, 0.29 mmol) to afford the desired product after two rounds of purification (40 mg, 25% yield); 1H NMR (300 MHz, CD3OD) δ 8.44 (s, 1H), 8.22 (s, 1H), 7.43–7.31 (m, 2H), 7.35–7.24 (m, 2H), 7.25–7.13 (m, 1H), 5.87 (s, 1H), 5.05–4.90 (m, 1H), 4.66–4.52 (m, 2H), 4.11 (d, J = 9.5 Hz, 1H), 3.94 (dq, J = 9.9, 7.1 Hz, 1H), 3.86–3.73 (m, 1H), 1.37 (dd, J = 7.1, 0.8 Hz, 3H), 1.20 (d, J = 6.2 Hz, 3H), 1.15 (d, J = 6.3 Hz, 3H), 0.93 (s, 3H) ppm; 13C NMR (151 MHz, CD3OD) δ 174.4 (d, 3JC–P = 5.5 Hz), 157.5, 154.0, 152.2 (d, 2JC–P = 6.7 Hz), 151.2, 141.9, 130.8, 126.2, 121.4 (d, 3JC–P = 4.8 Hz), 120.2, 83.0, 77.7, 70.1, 68.4 (d, 2JC–P = 5.0 Hz), 66.4, 51.7, 51.2 (d, 2JC–P = 8.5 Hz), 22.0, 21.9, 20.9, 20.7 (d, 3JC–P = 6.3 Hz) ppm; 31P NMR (121 MHz, CD3OD) δ 3.7 ppm; HRMS (ESI) m/z: [M + H]+ Calcd for C23H32N6O7PS 567.1785; Found 567.1778.
(2S)-isopropyl 2-(((((2R,3S,4R,5R)-5-(3,5-dioxo-4,5-dihydro-1,2,4-triazin-2(3H)-yl)-3,4-dihydroxy-4-methyltetrahydrothiophen-2-yl)methoxy)(phenoxy)phosphoryl)amino)propanoate (13d):
Following the general phosphoramidation procedure, thionucleoside 11d (60 mg, 0.22 mmol) was treated with 12 (100 mg, 0.22 mmol) to afford the desired product (27 mg, 23% yield); 1H NMR (600 MHz, CD3OD) δ 7.49 (s, 1H), 7.35 (t, J = 7.9 Hz, 2H), 7.27–7.21 (m, 2H), 7.21–7.15 (m, 1H), 6.03 (s, 1H), 4.95 (dq, J = 12.5, 6.3 Hz, 1H), 4.59 (ddd, J = 10.0, 6.0, 3.9 Hz, 1H), 4.28–4.21 (m, 1H), 4.04 (d, J = 8.7 Hz, 1H), 3.90 (dq, J = 9.7, 7.1 Hz, 1H), 3.64 (td, J = 8.9, 3.9 Hz, 1H), 1.34 (d, J = 7.1 Hz, 3H), 1.22 (d, J = 6.3 Hz, 6H), 1.20 (s, 3H) ppm; 13C NMR (151 MHz, CD3OD) δ 174.4 (d, 3JC–P = 5.9 Hz), 158.3, 152.2 (d, 2JC–P = 6.9 Hz), 150.2, 137.5, 130.7, 126.1, 121.4 (d, 3JC–P = 4.8 Hz), 83.3, 79.3, 70.9 (d, 2JC–P = 5.3 Hz), 70.1, 51.6, 51.5 (d, 2JC–P = 8.0 Hz), 22.0, 21.9, 20.7, 20.6 (d, 3JC–P = 6.1 Hz) ppm; 31P NMR (121 MHz, CD3OD) δ 3.3 ppm; HRMS (ESI-) m/z: [M + Cl]− Calcd for C21H29N4O9ClPS 579.1087; Found 579.1090.
Isopropyl (2S)-2-[[[(2R,3S,4R,5R)-5-(4-amino-5-fluoro-2-oxo-pyrimidin-1-yl)-3,4-dihydroxy-4-methyl-tetrahydrothiophen-2-yl]methoxy-phenoxy-phosphoryl]amino]propanoate (13e):
Following the general phosphoramidation procedure, thionucleoside 11e (72 mg, 0.25 mmol) was treated with 12 (224 mg, 0.5 mmol) to afford the desired product after three rounds of purification (12 mg, 8% yield); 1H NMR (600 MHz, MeOD) δ 8.19 (d, 3JH–F = 6.8 Hz, 1H), 7.39–7.33 (m, 2H), 7.29–7.26 (m, 2H), 7.20–7.18 (m, 1H), 6.09 (d, 5JH–F = 1.8 Hz, 1H), 4.97 (app hept, J = 6.3 Hz, 1H), 4.57–4.52 (m, 1H), 4.50–4.46 (m, 1H), 3.93 (dq, J = 10.1, 7.1 Hz, 1H), 3.69 (d, J = 9.7 Hz, 1H), 3.67–3.63 (m, 1H), 1.35 (d, J = 7.1 Hz, 3H), 1.22 (d, J = 6.2 Hz, 3H), 1.21 (d, J = 6.3 Hz, 3H), 1.15 (s, 3H) ppm; 13C NMR (151 MHz, CD3OD) δ 174.4 (d, 3JC–P = 5.4 Hz), 159.3 (d, 2JC–F = 14.0 Hz), 157.4, 152.2 (d, 2JC–P = 6.6 Hz), 138.0 (d, 1JC–F = 244.9 Hz), 130.8, 127.9 (d, 2JC–F = 32.8 Hz), 126.1, 121.4 (d, 3JC–P = 4.9 Hz), 82.9, 77.4, 70.2, 69.3, 67.6 (d, 2JC–P = 4.8 Hz), 51.7, 51.0 (d, 2JC–P = 8.6 Hz), 22.0, 21.9, 20.63, 20.59 ppm; 19F NMR (282 MHz, CD3OD) δ-165.7 (d, 3JF–H = 5.6 Hz) ppm; 31P NMR (121 MHz, CD3OD) δ 3.7 ppm; HRMS (NSI) m/z: [M + H]+ Calcd for C22H31N4O8FPS 561.1579; Found 561.1578.
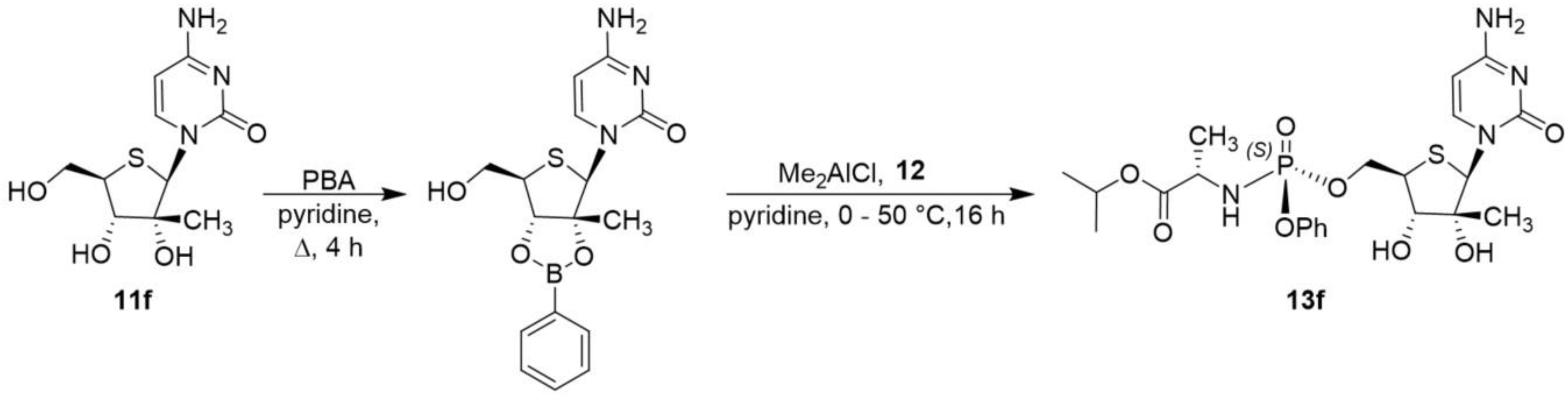
Isopropyl (2S)-2-[[[(2R,3S,4R,5R)-5-(4-amino-2-oxo-pyrimidin-1-yl)-3,4-dihydroxy-4-methyl-tetrahydrothiophen-2-yl]methoxy-phenoxy-phosphoryl]amino]propanoate (13f):
An oven-dried 50-mL round-bottom flask with stir bar and reflux condenser was charged with thionucleoside 11f (80.5 mg, 0.29 mmol), and the apparatus and solid were dried overnight in vacuo. The flask was then charged with argon, phenylboronic acid (38 mg, 0.31 mmol), and anhydrous pyridine (0.75 mL) to give a yellow solution. The reaction was heated to 100 °C for approximately 4 h before the volatiles were removed. The crude yellow semi-solid was dried briefly in vacuo before the flask was again charged with argon followed by 12 (160 mg, 0.35 mmol) and anhydrous pyridine (1 mL) to give a yellow solution. The solution was chilled to 0 °C before Me2AlCl (0.15 mL, 0.15 mmol) was dropwise added as a 1 M solution in hexanes, and the reaction was warmed to 50 °C with stirring overnight. The next morning, the reaction was cooled to ambient temperature before being quenched with 0.4 mL of 30 wt% aqueous L-tartaric acid solution. The reaction mixture was then diluted with ethyl acetate and aqueous brine solution before being extracted with ethyl acetate. The organic extracts were combined, dried over Na2SO4, filtered, concentrated and purified via two rounds of silica gel flash column chromatography (eluted with a gradient of 0–15% methanol in DCM) to afford the desired product (59 mg, 37% yield); 1H NMR (600 MHz, CD3OD) δ 8.09 (d, J = 7.5 Hz, 1H), 7.41–7.35 (m, 2H), 7.31–7.26 (m, 2H), 7.23–7.17 (m, 1H), 6.16 (s, 1H), 5.90 (d, J = 7.4 Hz, 1H), 4.97 (app hept, J = 6.3 Hz, 1H), 4.57–4.51 (m, 1H), 4.50–4.43 (m, 1H), 3.93 (dq, J = 9.9, 7.1 Hz, 1H), 3.67 (br, 1H), 1.36 (d, J = 7.1 Hz, 3H), 1.22 (d, J = 6.2 Hz, 3H), 1.21 (d, J = 6.3 Hz, 3H), 1.12 (s, 3H) ppm; 13C NMR (151 MHz, CD3OD) δ 174.4 (d, 3JC–P = 5.4 Hz), 167.1, 159.0, 152.2 (d, 2JC–P = 6.7 Hz), 144.1, 130.9, 126.2, 121.3 (d, 3JC–P = 4.8 Hz), 96.5, 82.9, 77.5, 70.2, 68.7, 67.7 (d, 2JC–P = 5.0 Hz), 51.7, 50.9 (d, 2JC–P = 8.6 Hz), 22.0, 21.9, 20.7, 20.6 ppm; 31P NMR (121 MHz, CD3OD) δ 3.5 ppm; HRMS (NSI) m/z: [M + H]+ Calcd for C22H32N4O8PS 543.1673; Found 543.1676.