Pimpinella anisum Essential Oil Nanoemulsion Toxicity against Tribolium castaneum? Shedding Light on Its Interactions with Aspartate Aminotransferase and Alanine Aminotransferase by Molecular Docking
Abstract
1. Introduction
2. Results
2.1. GC-MS Analysis of P. anisum Essential Oil and Nanoemulsion
2.2. Charachterization of Nanoemulsion
2.3. Toxicity Assays and Sub-Lethal Toxicity Assays
2.4. Biochemical Assays
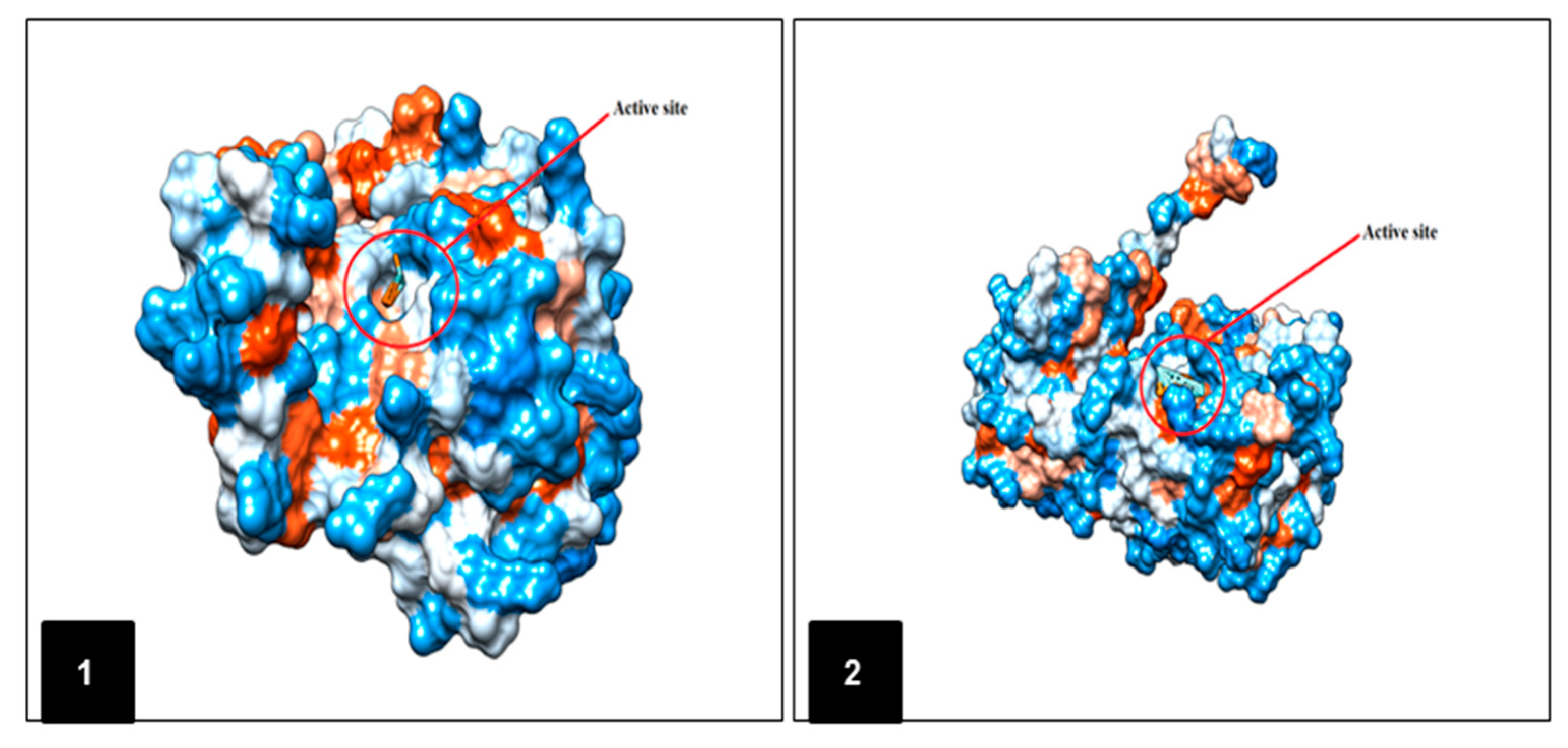
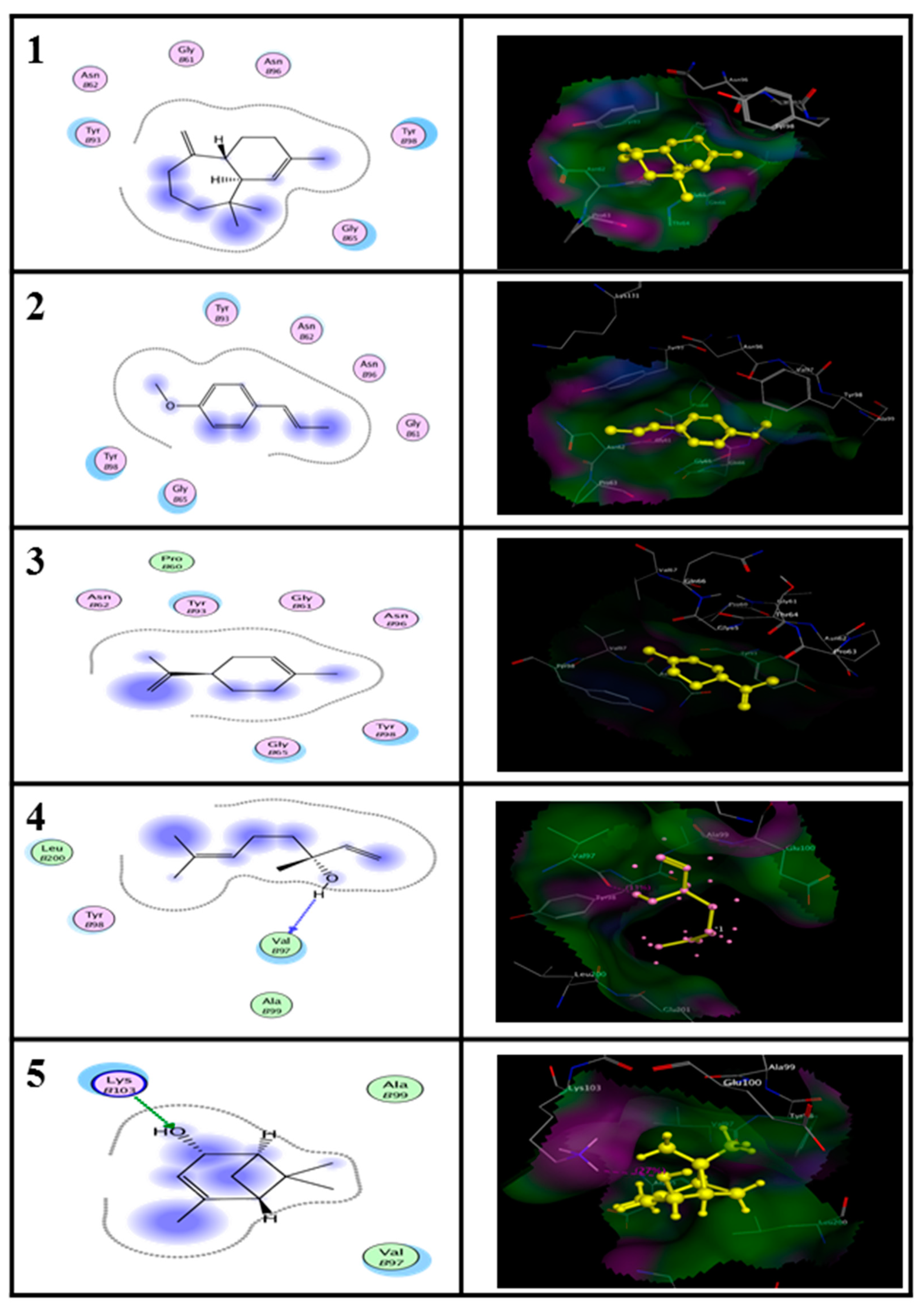
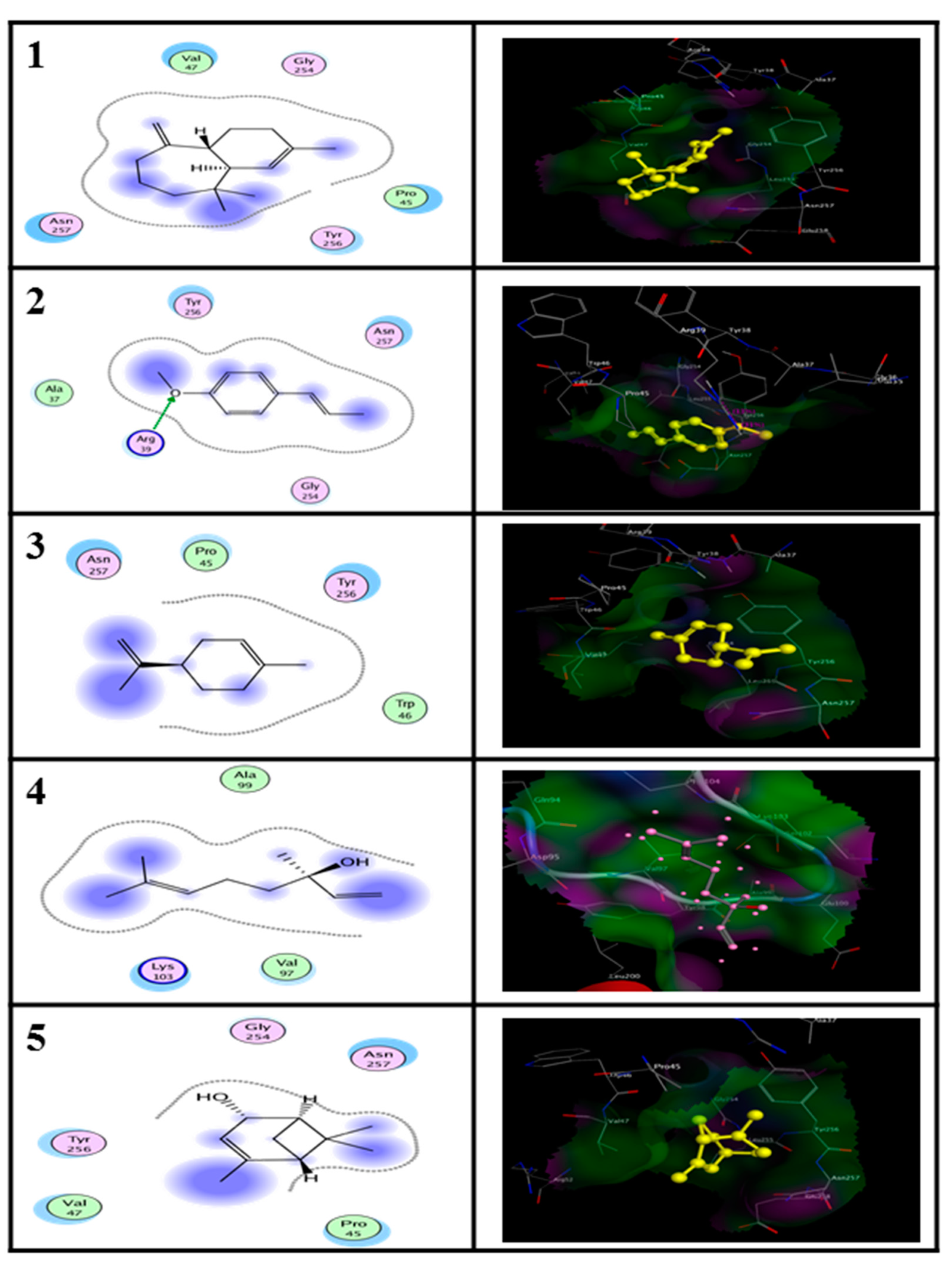
2.5. In Silico Molecular Docking Prediction of the Binding Site of Main Compounds of the EO and NE on Key Enzymes
3. Discussion
4. Materials and Methods
4.1. Insect Rearing
4.2. Essential Oil
4.3. Nanoemulsion Preparation and Characterization
4.4. Gascromatography Coupled with Mass Spectrometry
4.5. Toxicactivity Assay
4.6. Evaluation of the Potential Mode of Action of Aniseed EO and NE
4.7. In Vitro Biochemical Assays
4.8. In Silico Molecular Docking Assay
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Pretty, J.; Bharucha, Z.P. Integrated pest management for sustainable intensification of agriculture in Asia and Africa. Insects 2015, 6, 152–182. [Google Scholar] [CrossRef] [PubMed]
- Ponsankar, A.; Vasantha-Srinivasan, P.; Senthil-Nathan, S.; Thanigaivel, A.; Edwin, E.-S.; Selin-Rani, S.; Kalaivani, K.; Hunter, W.B.; Alessandro, R.T.; Abdel-Megeed, A.; et al. Target and non-target toxicity of botanical insecticide derived from Couroupita guianensis L. flower against generalist herbivore, Spodoptera litura Fab. and an earthworm, Eisenia foetida Savigny. Ecotoxicol. Environ. Saf. 2016, 133, 260–270. [Google Scholar] [CrossRef]
- Brouwer, M.; Kromhout, H.; Vermeulen, R.; Duyzer, J.; Kramer, H.; Hazeu, G.; De Snoo, G.; Huss, A. Assessment of residential environmental exposure to pesticides from agricultural fields in the Netherlands. J. Expo. Sci. Environ. Epidemiol. 2018, 28, 173–181. [Google Scholar] [CrossRef]
- Pavela, R.; Benelli, G. Essential Oils as Ecofriendly Biopesticides? Challenges and Constraints. Trends Plant Sci. 2016, 21, 1000–1007. [Google Scholar] [CrossRef]
- Pavela, R.; Benelli, G. Ethnobotanical knowledge on botanical repellents employed in the African region against mosquito vectors—A review. Exp. Parasitol. 2016, 167, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Pavela, R.; Maggi, F.; Lupidi, G.; Cianfaglione, K.; Dauvergne, X.; Bruno, M.; Benelli, G. Efficacy of sea fennel (Crithmum maritimum L., Apiaceae) essential oils against Culex quinquefasciatus Say and Spodoptera littoralis (Boisd.). Ind. Crop. Prod. 2017, 109, 603–610. [Google Scholar] [CrossRef]
- Rizzo, R.; Lo Verde, G.; Sinacori, M.; Maggi, F.; Cappellacci, L.; Petrelli, R.; Vittori, S.; Morshedloo, M.R.; Fofie, N.G.B.Y.; Benelli, G. Developing green insecticides to manage olive fruit flies? Ingestion toxicity of four essential oils in protein baits on Bactrocera oleae. Ind. Crop. Prod. 2020, 143, 111884. [Google Scholar] [CrossRef]
- Kavallieratos, N.G.; Boukouvala, M.C.; Ntalli, N.; Skourti, A.; Karagianni, E.S.; Nika, E.P.; Kontodimas, D.C.; Cappellacci, L.; Petrelli, R.; Cianfaglione, K.; et al. Effectiveness of eight essential oils against two key stored-product beetles, Prostephanus truncatus (Horn) and Trogoderma granarium Everts. Food Chem. Toxicol. 2020, 139. [Google Scholar] [CrossRef] [PubMed]
- Benelli, G.; Pavela, R.; Giordani, C.; Casettari, L.; Curzi, G.; Cappellacci, L.; Petrelli, R.; Maggi, F. Acute and sub-lethal toxicity of eight essential oils of commercial interest against the filariasis mosquito Culex quinquefasciatus and the housefly Musca domestica. Ind. Crop. Prod. 2018, 112, 668–680. [Google Scholar] [CrossRef]
- Tunç, I.; Berger, B.M.; Erler, F.; Dagli, F. Ovicidal activity of essential oils from five plants against two stored-product insects. J. Stored Prod. Res. 2000, 36, 161–168. [Google Scholar] [CrossRef]
- Evergetis, E.; Michaelakis, A.; Haroutounian, S.A. Essential Oils of Umbelliferae (Apiaceae) Family Taxa as Emerging Potent Agents for Mosquito Control. In Integrated Pest Management and Pest Control; Larramendy, M.L., Soloneski, S., Eds.; IntechOpen: Rijeka, Croatia, 2012. [Google Scholar]
- Kanda, D.; Kaur, S.; Koul, O. A comparative study of monoterpenoids and phenylpropanoids from essential oils against stored grain insects: Acute toxins or feeding deterrents. J. Pest Sci. 2017, 90, 531–545. [Google Scholar] [CrossRef]
- Toloza, A.C.; Zygadlo, J.; Biurrun, F.; Rotman, A.; Picollo, M.I. Bioactivity of Argentinean essential oils against permethrin-resistant head lice, Pediculus humanus capitis. J. Insect Sci. 2010, 10. [Google Scholar] [CrossRef] [PubMed]
- Locci, E.; Lai, S.; Piras, A.; Marongiu, B.; Lai, A. 13C-CPMAS and 1H-NMR study of the inclusion complexes of β-cyclodextrin with carvacrol, thymol, and eugenol prepared in supercritical carbon dioxide. Chem. Biodivers. 2004, 1, 1354–1366. [Google Scholar] [CrossRef] [PubMed]
- Mastelić, J.; Jerković, I.; Blažević, I.; Poljak-Blaži, M.; Borović, S.; Ivančić-Baće, I.; Smrěcki, V.; Žarković, N.; Brčić-Kostic, K.; Vikić-Topić, D.; et al. Comparative study on the antioxidant and biological activities of carvacrol, thymol, and eugenol derivatives. J. Agric. Food Chem. 2008, 56, 3989–3996. [Google Scholar] [CrossRef]
- Parisi, C.; Vigani, M.; Rodríguez-Cerezo, E. Agricultural nanotechnologies: What are the current possibilities? Nano Today 2015, 10, 124–127. [Google Scholar] [CrossRef]
- Cespi, M.; Quassinti, L.; Perinelli, D.R.; Bramucci, M.; Iannarelli, R.; Papa, F.; Ricciutelli, M.; Bonacucina, G.; Palmieri, G.F.; Maggi, F. Microemulsions enhance the shelf-life and processability of Smyrnium olusatrum L. essential oil. Flavour Fragr. J. 2017, 32, 159–164. [Google Scholar] [CrossRef]
- Campolo, O.; Cherif, A.; Ricupero, M.; Siscaro, G.; Grissa-Lebdi, K.; Russo, A.; Cucci, L.M.; Di Pietro, P.; Satriano, C.; Desneux, N.; et al. Citrus peel essential oil nanoformulations to control the tomato borer, Tuta absoluta: Chemical properties and biological activity. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Nenaah, G.E. Chemical composition, toxicity and growth inhibitory activities of essential oils of three Achillea species and their nano-emulsions against Tribolium castaneum (Herbst). Ind. Crop. Prod. 2014, 53, 252–260. [Google Scholar] [CrossRef]
- Pavela, R.; Benelli, G.; Pavoni, L.; Bonacucina, G.; Cespi, M.; Cianfaglione, K.; Bajalan, I.; Morshedloo, M.R.; Lupidi, G.; Romano, D.; et al. Microemulsions for delivery of Apiaceae essential oils—Towards highly effective and eco-friendly mosquito larvicides? Ind. Crop. Prod. 2019, 129, 631–640. [Google Scholar] [CrossRef]
- Al-Bayati, F.A. Synergistic antibacterial activity between Thymus vulgaris and Pimpinella anisum essential oils and methanol extracts. J. Ethnopharmacol. 2008, 116, 403–406. [Google Scholar] [CrossRef]
- Shokri, A.; Hatami, T.; Khamforoush, M. Near critical carbon dioxide extraction of Anise (Pimpinella Anisum L.) seed: Mathematical and artificial neural network modeling. J. Supercrit. Fluids 2011, 58, 49–57. [Google Scholar] [CrossRef]
- Ullah, H.; Honermeier, B. Fruit yield, essential oil concentration and composition of three anise cultivars (Pimpinella anisum L.) in relation to sowing date, sowing rate and locations. Ind. Crop. Prod. 2013, 42, 489–499. [Google Scholar] [CrossRef]
- Iannarelli, R.; Caprioli, G.; Sut, S.; Dall’Acqua, S.; Fiorini, D.; Vittori, S.; Maggi, F. Valorizing overlooked local crops in the era of globalization: The case of aniseed (Pimpinella anisum L.) from Castignano (central Italy). Ind. Crop. Prod. 2017, 104, 99–110. [Google Scholar] [CrossRef]
- Dorman, H.J.D.; Deans, S.G. Antimicrobial agents from plants: Antibacterial activity of plant volatile oils. J. Appl. Microbiol. 2000, 88, 308–316. [Google Scholar] [CrossRef]
- Omidbaigi, R.; Hadjiakhoondi, A.; Saharkhiz, M. Changes in content and chemical composition of Pimpinella anisum oil at various harvest time. J. Essent. Oil-Bear. Plants 2003, 6, 46–50. [Google Scholar] [CrossRef]
- Samojlik, I.; Mijatović, V.; Petković, S.; Škrbić, B.; Božin, B. The influence of essential oil of aniseed (Pimpinella anisum, L.) on drug effects on the central nervous system. Fitoterapia 2012, 83, 1466–1473. [Google Scholar] [CrossRef]
- Pavela, R. Insecticidal properties of Pimpinella anisum essential oils against the Culex quinquefasciatus and the non-target organism Daphnia magna. J. Asia. Pac. Entomol. 2014, 17, 287–293. [Google Scholar] [CrossRef]
- Hashem, A.S.; Awadalla, S.S.; Zayed, G.M.; Maggi, F.; Benelli, G. Pimpinella anisum essential oil nanoemulsions against Tribolium castaneum—Insecticidal activity and mode of action. Environ. Sci. Pollut. Res. 2018, 25, 18802–18812. [Google Scholar] [CrossRef]
- Olle, M.; Bender, I. The content of oils in umbelliferous crops and its formation. Agron. Res. 2010, 8, 687–696. [Google Scholar]
- Saxena, S.N.; Sharma, Y.K.; Rathore, S.S.; Singh, K.K.; Barnwal, P.; Saxena, R.; Upadhyaya, P.; Anwer, M.M. Effect of cryogenic grinding on volatile oil, oleoresin content and anti-oxidant properties of coriander (Coriandrum sativum L.) genotypes. J. Food Sci. Technol. 2015, 52, 568–573. [Google Scholar] [CrossRef]
- Orav, A.; Raal, A.; Arak, E. Essential oil composition of Pimpinella anisum L. fruits from various European countries. Nat. Prod. Res. 2008, 22, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Beyer, E.M.; Azizi, A.; Honermeier, B. Effects of sowing time and sowing density on fruit yield, essential oil concentration and composition of anise (Pimpinella anisum L.) under field conditions in Germany. Z. fur Arznei- und Gewurzpflanzen 2011, 16, 26–33. [Google Scholar]
- Iannarelli, R.; Marinelli, O.; Morelli, M.B.; Santoni, G.; Amantini, C.; Nabissi, M.; Maggi, F. Aniseed (Pimpinella anisum L.) essential oil reduces pro-inflammatory cytokines and stimulates mucus secretion in primary airway bronchial and tracheal epithelial cell lines. Ind. Crop. Prod. 2018, 114, 81–86. [Google Scholar] [CrossRef]
- Benelli, G.; Pavela, R.; Iannarelli, R.; Petrelli, R.; Cappellacci, L.; Cianfaglione, K.; Afshar, F.H.; Nicoletti, M.; Canale, A.; Maggi, F. Synergized mixtures of Apiaceae essential oils and related plant-borne compounds: Larvicidal effectiveness on the filariasis vector Culex quinquefasciatus Say. Ind. Crop. Prod. 2017, 96, 186–195. [Google Scholar] [CrossRef]
- Silver, K.; Jiang, H.; Fu, J.; Phillips, T.W.; Beeman, R.W.; Park, Y. The Tribolium castaneum cell line TcA: A new tool kit for cell biology. Sci. Rep. 2014, 4. [Google Scholar] [CrossRef]
- Rodrigues, V.M.; Rosa, P.T.V.; Marques, M.O.M.; Petenate, A.J.; Meireles, M.A.A. Supercritical Extraction of Essential Oil from Aniseed (Pimpinella anisum L.) Using CO2: Solubility, Kinetics, and Composition Data. J. Agric. Food Chem. 2003, 51, 1518–1523. [Google Scholar] [CrossRef]
- Foroughi, A.; Pournaghi, P.; Najafi, F.; Zangeneh, A.; Zangeneh, M.M.; Moradi, R. Evaluation of antibacterial activity and phytochemical screening of Pimpinella anisem’s essential oil. Int. J. Pharmacogn. Phytochem. Res. 2016, 8, 1886–1890. [Google Scholar]
- Khanjari, A.; Bahonar, A.; Noori, N.; Siahkalmahaleh, M.R.; Rezaeigolestani, M.; Asgarian, Z.; Khanjari, J. In vitro antibacterial activity of Pimpinella anisum essential oil and its influence on microbial, chemical, and sensorial properties of minced beef during refrigerated storage. J. Food Saf. 2019, 39. [Google Scholar] [CrossRef]
- Ksouda, G.; Sellimi, S.; Merlier, F.; Falcimaigne-cordin, A.; Thomasset, B.; Nasri, M.; Hajji, M. Composition, antibacterial and antioxidant activities of Pimpinella saxifraga essential oil and application to cheese preservation as coating additive. Food Chem. 2019, 288, 47–56. [Google Scholar] [CrossRef]
- Romdhane, M.; Tizaoui, C. The kinetic modelling of a steam distillation unit for the extraction of aniseed (Pimpinella anisum) essential oil. J. Chem. Technol. Biotechnol. 2005, 80, 759–766. [Google Scholar] [CrossRef]
- Ozel, A. Anise (Pimpinella anisum): Changes in yields and component composition on harvesting at different stages of plant maturity. Exp. Agric. 2009, 45, 117–126. [Google Scholar] [CrossRef]
- Mahfouz, S.A.; Sharaf-Eldin, M.A. Effect of mineral vs. biofertilizer on growth, yield, and essential oil content of fennel (Foeniculum vulgare Mill.). Int. Agrophysics 2007, 21, 361–366. [Google Scholar]
- Shi, B.; Wang, Z.; Wen, H. Research on the strengths of electrostatic and van der Waals interactions in ionic liquids. J. Mol. Liq. 2017, 241, 486–488. [Google Scholar] [CrossRef]
- Hoeller, S.; Sperger, A.; Valenta, C. Lecithin based nanoemulsions: A comparative study of the influence of non-ionic surfactants and the cationic phytosphingosine on physicochemical behaviour and skin permeation. Int. J. Pharm. 2009, 370, 181–186. [Google Scholar] [CrossRef]
- Arancibia, C.; Navarro-Lisboa, R.; Zúñiga, R.N.; Matiacevich, S. Application of CMC as thickener on nanoemulsions based on olive oil: Physical properties and stability. Int. J. Polym. Sci. 2016, 2016. [Google Scholar] [CrossRef]
- Kotha, P.; Rayalu, J.; Sai Gopal Divi, V.R.; Dakinedi, S.; Dowlathabad, M. Modelling simulation phylogenetics of leukemia FMS tyrosine kinase 3 (FLT3). Online J. Bioinforma. 2015, 16, 8–17. [Google Scholar]
- Wang, S.-Q.; Du, Q.-S.; Huang, R.-B.; Zhang, D.-W.; Chou, K.-C. Insights from investigating the interaction of oseltamivir (Tamiflu) with neuraminidase of the 2009 H1N1 swine flu virus. Biochem. Biophys. Res. Commun. 2009, 386, 432–436. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, S.-Q.; Xu, W.-R.; Wang, R.-L.; Chou, K.-C. Design novel dual agonists for treating type-2 diabetes by targeting peroxisome proliferator-activated receptors with core hopping approach. PLoS ONE 2012, 7. [Google Scholar] [CrossRef]
- Chou, K.-C.; Chen, N.Y. The biological functions of low-frequency phonons. Sci. Sin. 1977, 20, 447–457. [Google Scholar]
- Lovell, S.C.; Davis, I.W.; Arendall, W.B., 3rd; Bakker, P.I.W.; Word, J.; Prisant, M.; Richardson, J.; Richardson, D.C. Structure validation by Calpha geometry: Phi, psi and Cbeta deviation. Proteins Struct. Funct. Genet. 2003, 50, 437–450. [Google Scholar] [CrossRef]
- Lashgari, A.; Mashayekhi, S.; Javadzadeh, M.; Marzban, R. Effect of Mentha piperita and Cuminum cyminum essential oil on Tribolium castaneum and Sitophilus oryzae. Arch. Phytopathol. Plant Prot. 2014, 47, 324–329. [Google Scholar] [CrossRef]
- Pavela, R. History, presence and perspective of using plant extracts as commercial botanical insecticides and farm products for protection against insects—A review. Plant Prot. Sci. 2016, 52, 229–241. [Google Scholar] [CrossRef]
- Sahaf, B.Z.; Moharamipour, S. Comparative study on deterrency of Carum copticum C. B. Clarke and Vitex pseudo-negundo (Hausskn.) Hand.-Mzt essential oils on feeding behavior of Tribolium castaneum (Herbst). Iran. Journla Med. Aromat. Plants 2009, 24, 385–395. [Google Scholar]
- Ebadollah, A.; Mahboubi, M. Insecticidal Activity of the Essential Oil Isolated from Azilia eryngioides (Pau) Hedge et Lamond Against Two Beetle Pests. Chil. J. Agric. Res. 2011, 71, 406–411. [Google Scholar] [CrossRef][Green Version]
- Islam, M.; Hasan, M.M.; Xiong, W.; Zhang, S.; Lei, C. Fumigant and repellent activities of essential oil from Coriandrum sativum (L.) (Apiaceae) against red flour beetle Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). J. Pest Sci. 2009, 82, 171–177. [Google Scholar] [CrossRef]
- Oliveira, A.P.; Santana, A.S.; Santana, E.D.R.; Lima, A.P.S.; Faro, R.R.N.; Nunes, R.S.; Lima, A.D.; Blank, A.F.; Araújo, A.P.A.; Cristaldo, P.F.; et al. Nanoformulation prototype of the essential oil of Lippia sidoides and thymol to population management of Sitophilus zeamais (Coleoptera: Curculionidae). Ind. Crop. Prod. 2017, 107, 198–205. [Google Scholar] [CrossRef]
- Golden, G.; Quinn, E.; Shaaya, E.; Kostyukovsky, M.; Poverenov, E. Coarse and nano emulsions for effective delivery of the natural pest control agent pulegone for stored grain protection. Pest Manag. Sci. 2018, 74, 820–827. [Google Scholar] [CrossRef]
- Pant, M.; Dubey, S.; Patanjali, P.K.; Naik, S.N.; Sharma, S. Insecticidal activity of eucalyptus oil nanoemulsion with karanja and jatropha aqueous filtrates. Int. Biodeterior. Biodegrad. 2014, 91, 119–127. [Google Scholar] [CrossRef]
- Chang, C.L.; Cho, I.L.K.; Li, Q.X. Insecticidal activity of basil oil, trans-anethole, estragole, and linalool to adult fruit flies of Ceratitis capitata, Bactrocera dorsalis, and Bactrocera cucurbitae. J. Econ. Entomol. 2009, 102, 203–209. [Google Scholar] [CrossRef]
- Heshmati Afshar, F.; Maggi, F.; Iannarelli, R.; Cianfaglione, K.; Isman, M.B. Comparative toxicity of Helosciadium nodiflorum essential oils and combinations of their main constituents against the cabbage looper, Trichoplusia ni (Lepidoptera). Ind. Crop. Prod. 2017, 98, 46–52. [Google Scholar] [CrossRef]
- Karr, L.L.; Coats, J.R. Insecticidal Properties of d-Limonene. J. Pestic. Sci. 1988, 13, 287–290. [Google Scholar] [CrossRef]
- Ainane, A.; Khammour, F.; Charaf, S.; Elabboubi, M.; Elkouali, M.; Talbi, M.; Benhima, R.; Cherroud, S.; Ainane, T. Chemical composition and insecticidal activity of five essential oils: Cedrus atlantica, Citrus limonum, Rosmarinus officinalis, Syzygium aromaticum and Eucalyptus globules. Mater. Today Proc. 2019, 13, 474–485. [Google Scholar] [CrossRef]
- Arrese, E.L.; Soulages, J.L. Insect fat body: Energy, metabolism, and regulation. Annu. Rev. Entomol. 2010, 55, 207–225. [Google Scholar] [CrossRef] [PubMed]
- Bajda, M.; Łoś, A.; Merska-Kazanowska, M. Effect of amphotericin B on the biochemical markers in the haemolymph of honey bees. Med. Weter. 2014, 70, 766–769. [Google Scholar]
- Nation, J. Insect Physiology and Biochemistry; CRC Press: London, UK, 2008. [Google Scholar]
- Mardani-Talaee, M.; Rahimi, V.; Zibaee, A. Effects of host plants on digestive enzymatic activities and some components involved in intermediary metabolism of Chrysodeixis chalcites (Lepidoptera: Noctuidae). J. Entomol. Acarol. Res. 2014, 46, 96–101. [Google Scholar] [CrossRef]
- Zhang, Y.; Dong, X.; Liu, J.; Hu, M.; Zhong, G.; Geng, P.; Yi, X. Molecular Cloning, Expression and Molecular Modeling of Chemosensory Protein from Spodoptera litura and Its Binding Properties with Rhodojaponin III. PLoS ONE 2012, 7. [Google Scholar] [CrossRef]
- El Kholy, S.; Makarem, H.; Abdel-Latif, A.; Seif, A. Physiological and biochemical effects of some essential oils on the granary weevil, Sitophilus granarius (L.) (Coleoptera: Curculionidae). Egypt. J. Exp. Biolology (Zool.) 2015, 11, 117. [Google Scholar]
- Surendra Nath, B. Changes in carbohydrate metabolism in hemolymph and fat body of the silkworm, Bombyx mori L., exposed to organophosphorus insecticides. Pestic. Biochem. Physiol. 2000, 68, 127–137. [Google Scholar] [CrossRef]
- Saleem, M.A.; Shakoori, A.R. Permethrin- and malathion-induced macromolecular abnormalities in adult Tribolium castaneum (herbst). Arch. Insect Biochem. Physiol. 1987, 5, 45–55. [Google Scholar] [CrossRef]
- Shakoori, F.R.; Feroz, A.; Riaz, T. Effect of sub-lethal doses of phosphine on macromolecular concentrations and metabolites of adult beetles of stored grain pest, Trogoderma granarium, previously exposed to phosphine. Pak. J. Zool. 2016, 48, 583–588. [Google Scholar]
- Ali, N.S.; Ali, S.S.; Shakoori, A.R. Effects of sublethal doses of talstar on biochemical components of malathion-resistant and -susceptible adults of rhyzopertha dominica. Pak. J. Zool. 2011, 43, 879–887. [Google Scholar]
- Hamouda, T.; Hayes, M.M.; Cao, Z.; Tonda, R.; Johnson, K.; Wright, D.C.; Brisker, J.; Baker, J.R., Jr. A novel surfactant nanoemulsion with broad-spectrum sporicidal activity against Bacillus species. J. Infect. Dis. 1999, 180, 1939–1949. [Google Scholar] [CrossRef]
- Joe, M.M.; Bradeeba, K.; Parthasarathi, R.; Sivakumaar, P.K.; Chauhan, P.S.; Tipayno, S.; Benson, A.; Sa, T. Development of surfactin based nanoemulsion formulation from selected cooking oils: Evaluation for antimicrobial activity against selected food associated microorganisms. J. Taiwan Inst. Chem. Eng. 2012, 43, 172–180. [Google Scholar] [CrossRef]
- Shukla, R.; Singh, P.; Prakash, B.; Kumar, A.; Mishra, P.K.; Dubey, N.K. Efficacy of essential oils of Lippia alba (Mill.) N.E. Brown and Callistemon lanceolatus (Sm.) Sweet and their major constituents on mortality, oviposition and feeding behaviour of pulse beetle, Callosobruchus chinensis L. J. Sci. Food Agric. 2011, 91, 2277–2283. [Google Scholar]
- Farrar, R.R.; Barbour, J.D.; Kennedy, G.G. Quantifying Food Consumption and Growth in Insects. Ann. Entomol. Soc. Am. 1989, 82, 593–598. [Google Scholar] [CrossRef]
- Hafiz, A.; Riaz, T.; Shakoori, F.R. Metabolic Profile of a Stored Grain Pest Trogoderma granarium Exposed to Deltamethrin. Pak. J. Zool. 2016, 49, 183–188. [Google Scholar] [CrossRef]
- Liao, M.; Xiao, J.-J.; Zhou, L.-J.; Yao, X.; Tang, F.; Hua, R.-M.; Wu, X.-W.; Cao, H.-Q. Chemical composition, insecticidal and biochemical effects of Melaleuca alternifolia essential oil on the Helicoverpa armigera. J. Appl. Entomol. 2017, 141, 721–728. [Google Scholar] [CrossRef]
- Keppanan, R.; Sivaperumal, S.; Chadra Kanta, D.; Akutse, K.S.; Wang, L. Molecular docking of protease from Metarhizium anisopliae and their toxic effect against model insect Galleria mellonella. Pestic. Biochem. Physiol. 2017, 138, 8–14. [Google Scholar] [CrossRef]
- Abbott, W.S. A method of computing the effectiveness of an insecticide. 1925. J. Am. Mosq. Control Assoc. 1987, 3, 302–303. [Google Scholar]
- Finney, D.J. Probit Analysis, 3rd ed.; Cambridge University Press: Cambridge, UK, 1971. [Google Scholar]
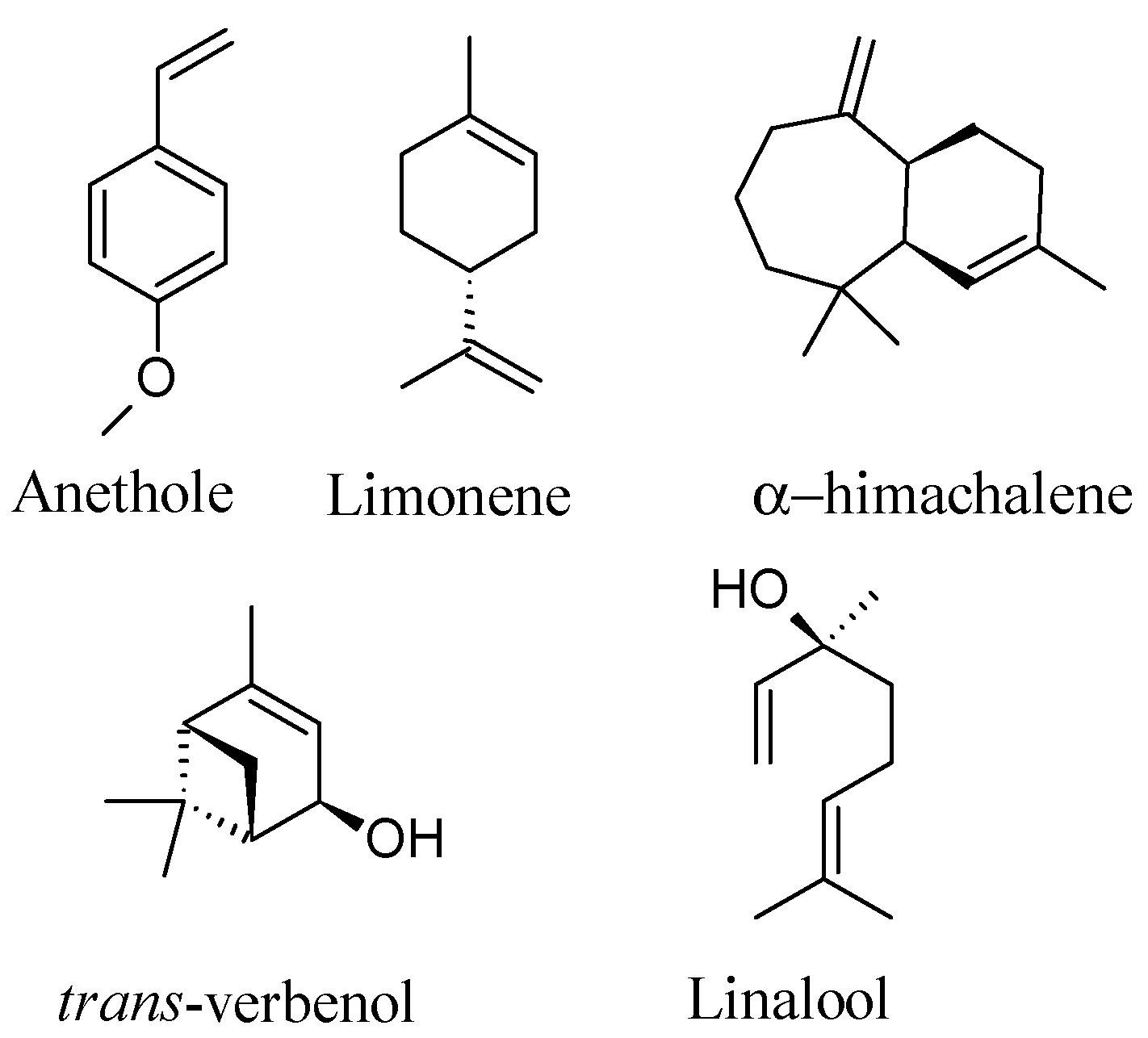
| Retention Time | Compounds | KI * | Compound EO (mg/g) | Compound NE (mg/g) |
|---|---|---|---|---|
| 8.2 | Limonene | 1.202 (1210) | 55.7 ± 0.1 | 03.2 ± 0.1 |
| 8.4 | 1,8-Cineole | 1.207 (1220) | 03.6 ± 0.1 | 0.3 ± 0.1 |
| 20.3 | Linalool | 1.557 (1556) | 16.4 ± 0.2 | 01.9 ± 0.2 |
| 20.5 | Linalyl formate | 1.563 (1579) | 09.2 ± 0.1 | 01.1 ± 0.2 |
| 23.8 | Methyl chavicol | 1.674 (1683) | 08.1 ± 0.2 | 01.1 ± 0.2 |
| 24.1 | trans-Verbenol | 1.685 (1683) | 24.7 ± 0.2 | 01.6 ± 0.1 |
| 24.2 | α-Himachalene | 1.688 (1690) | 25.2 ± 0.2 | 03.5 ± 0.1 |
| 25.5 | Geranial | 1.734 (1740) | 08.7 ± 0.1 | 01.2 ± 0.2 |
| 28.4 | (E)-Anethole | 1.839 (1847) | 801.0 ± 0.2 | 102.1 ± 0.1 |
| 33.1 | p-anisaldehyde | 2.023 (2020) | 09.9 ± 0.1 | 01.3 ± 0.1 |
| 33.5 | (E)-cinnamaldehyde | 2.030 (2030) | 08.6 ± 0.1 | 01.2 ± 0.2 |
| 36.5 | epi-α--Cadinol | 2.168 (2165) | 08.1 ± 0.2 | 01.0 ± 0.2 |
| 36.6 | Eugenol | 2.170 (2186) | 08.9 ± 0.1 | 01.2 ± 0.1 |
| 44.6 | Acetyl-isoeugenol | 2.475 (2400) | 11.3 ± 0.2 | 01.1 ± 0.1 |
| Total | 999.4 | 121.8 |
| Treatment | Concentration (%, v/v) | RGR ± SD (mg/mg/day) | RCR ± SD (mg/mg/day) | ECI ± SD (%) | FDI ± SD (%) |
|---|---|---|---|---|---|
| P. anisum EO | 1 | −0.25 ± 0.06 b | 1.05 ± 0.11b | −17.24 ± 0.84 b | 19.04 ± 0.45 c |
| 2 | −0.37 ± 0.28 b | 0.79 ± 0.03 b | −34.48 ± 0.21 c | 38.09 ± 0.23 b | |
| 3 | −0.42 ± 0.05 b | 0.52 ± 0.54c | −51.72 ± 0.23 d | 57.13 ± 0.04 b | |
| 4 | −0.64 ± 0.21 c | 0.26 ± 0.21c | −68.96 ± 0.56 e | 76.18 ± 0.52 a | |
| Control | 0.13 ± 0.04 a | 3.88 ± 0.06 a | 34.87 ± 0.21 a | 0.00 ± 0.00 d | |
| LC50 (%, v/v) = 2.1 a (1.8 − 2.9) b; slope = 2.37 | |||||
| P. anisum NE | 1 | 0.21 ± 0.18 a | 3.31 ± 0.46 a | −0.30 ± 1.85 b | 0.39 ± 0.21 b |
| 2 | 0.14 ± 0.05 b | 2.48 ± 0.15 a | −0.61 ± 1.01 b | 0.79 ± 0.29 b | |
| 3 | 0.04 ± 0.06 c | 1.65 ± 0.16 b | −0.91 ± 0.32 c | 1.19 ± 0.34 a | |
| 4 | −0.02 ± 0.02 d | 0.83 ± 1.23 c | −1.22 ± 0.34 c | 1.59 ± 0.56 a | |
| Control | 0.13 ± 0.04 b | 3.88 ± 0.06 a | 34.87 ± 0.21 a | 0.00 ± 0.00 c | |
| LC50 (%, v/v) = 9.8 a (8.6 − 12.7) b; slope = 2.43 | |||||
| Parameter | Control | P. anisum Essential Oil | Variation (%) * | P. anisum Nanoemulsion | Variation (%) * |
|---|---|---|---|---|---|
| ALT (U/mL) | 64.8 | 76.38 | −17.87 | 59.42 | +8.31 |
| AST (U/mL) | 92.5 | 64.23 | +30.56 | 37.85 | +59.32 |
| Glucose (mg/dL) | 102 | 19.84 | +80.54 | 14.49 | +85.79 |
| Total protein (mg/dL) | 1.96 | 2.27 | −15.81 | 0.93 | +52.08 |
| Total lipid (mg %) | 562.5 | 645 | −14.75 | 510 | +9.33 |
| Enzyme | Ligand | Binding Energy (kcal/M) | RMSD (A°) |
|---|---|---|---|
| Alanine aminotransferase (ALT) | (E)-anethole | −11.93 | 3.67 |
| Limonene | −11.26 | 1.69 | |
| alpha-himachalene | −12.03 | 2.94 | |
| trans-Verbenol | −10.42 | 2.42 | |
| Aspartate aminotransferase (AST) | (E)-anethole | −11.51 | 2.43 |
| Limonene | −9.34 | 2.12 | |
| alpha-himachalene | −9.38 | 3.01 | |
| trans-Verbenol | −7.95 | 2.74 |
Sample Availability: Samples of the compounds are not available from the authors. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hashem, A.S.; Ramadan, M.M.; Abdel-Hady, A.A.A.; Sut, S.; Maggi, F.; Dall’Acqua, S. Pimpinella anisum Essential Oil Nanoemulsion Toxicity against Tribolium castaneum? Shedding Light on Its Interactions with Aspartate Aminotransferase and Alanine Aminotransferase by Molecular Docking. Molecules 2020, 25, 4841. https://doi.org/10.3390/molecules25204841
Hashem AS, Ramadan MM, Abdel-Hady AAA, Sut S, Maggi F, Dall’Acqua S. Pimpinella anisum Essential Oil Nanoemulsion Toxicity against Tribolium castaneum? Shedding Light on Its Interactions with Aspartate Aminotransferase and Alanine Aminotransferase by Molecular Docking. Molecules. 2020; 25(20):4841. https://doi.org/10.3390/molecules25204841
Chicago/Turabian StyleHashem, Ahmed S., Marwa M. Ramadan, Amira A. A. Abdel-Hady, Stefania Sut, Filippo Maggi, and Stefano Dall’Acqua. 2020. "Pimpinella anisum Essential Oil Nanoemulsion Toxicity against Tribolium castaneum? Shedding Light on Its Interactions with Aspartate Aminotransferase and Alanine Aminotransferase by Molecular Docking" Molecules 25, no. 20: 4841. https://doi.org/10.3390/molecules25204841
APA StyleHashem, A. S., Ramadan, M. M., Abdel-Hady, A. A. A., Sut, S., Maggi, F., & Dall’Acqua, S. (2020). Pimpinella anisum Essential Oil Nanoemulsion Toxicity against Tribolium castaneum? Shedding Light on Its Interactions with Aspartate Aminotransferase and Alanine Aminotransferase by Molecular Docking. Molecules, 25(20), 4841. https://doi.org/10.3390/molecules25204841










