Antiangiogenic Activity of Flavonoids: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Results
2.1. Section 1: Systematic Overview
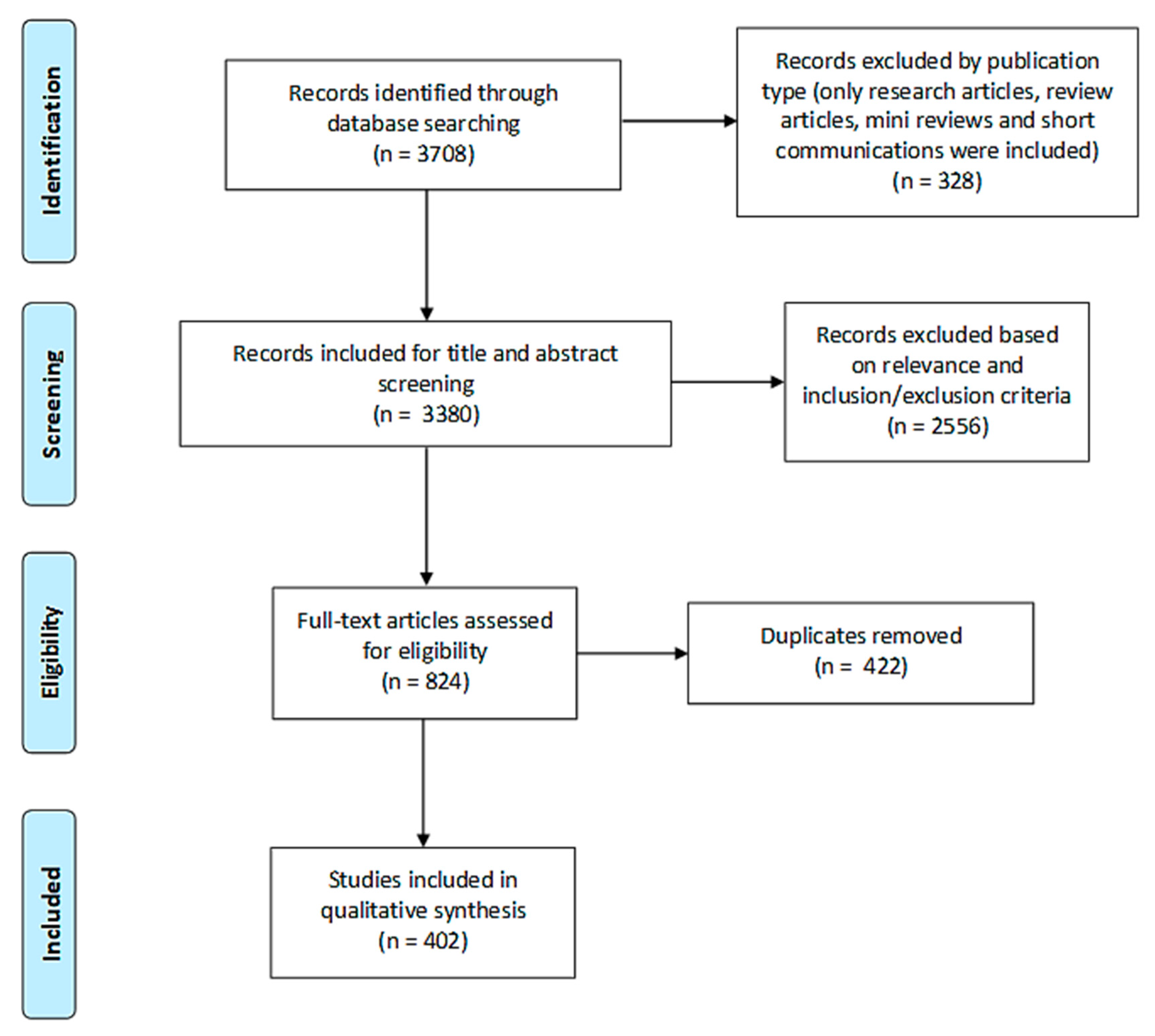
2.1.1. Search Results
2.1.2. Study Characteristics
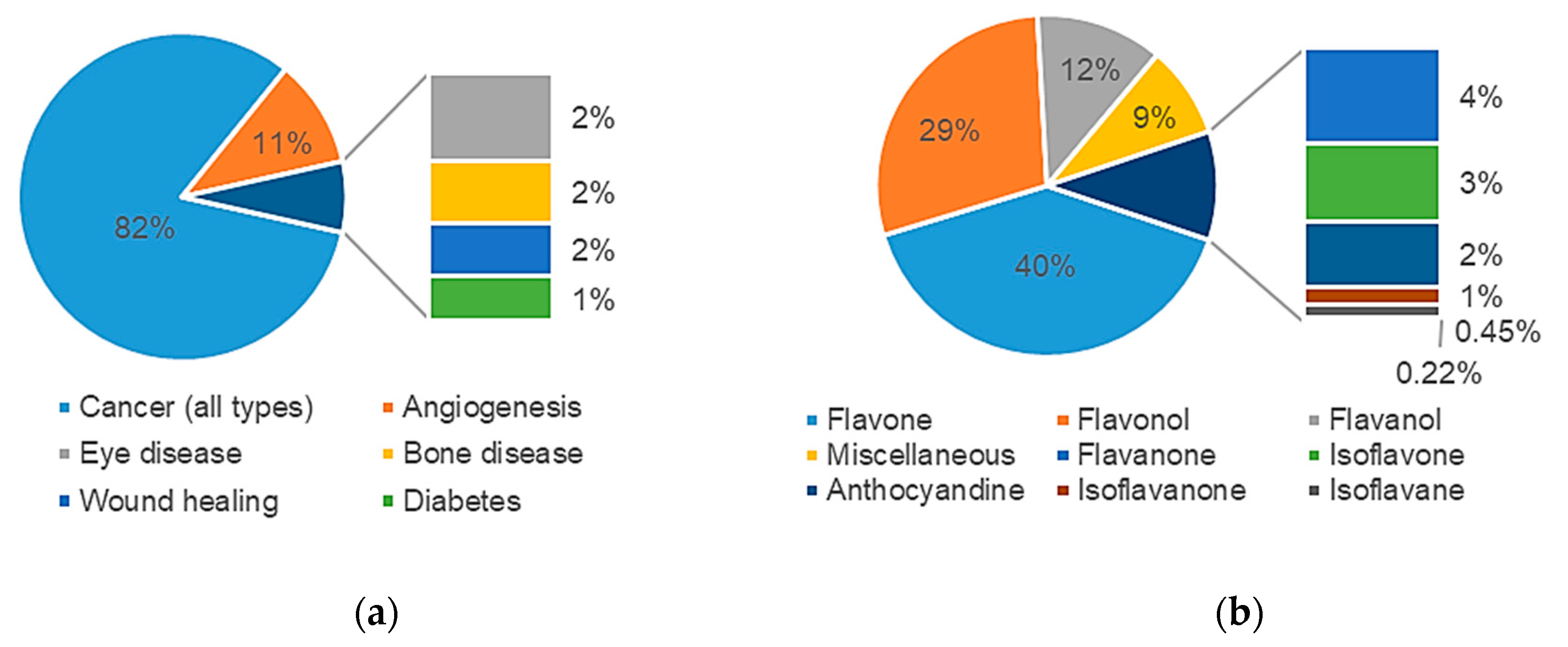
2.1.3. Data Analysis
2.2. Section 2: Meta-Analysis
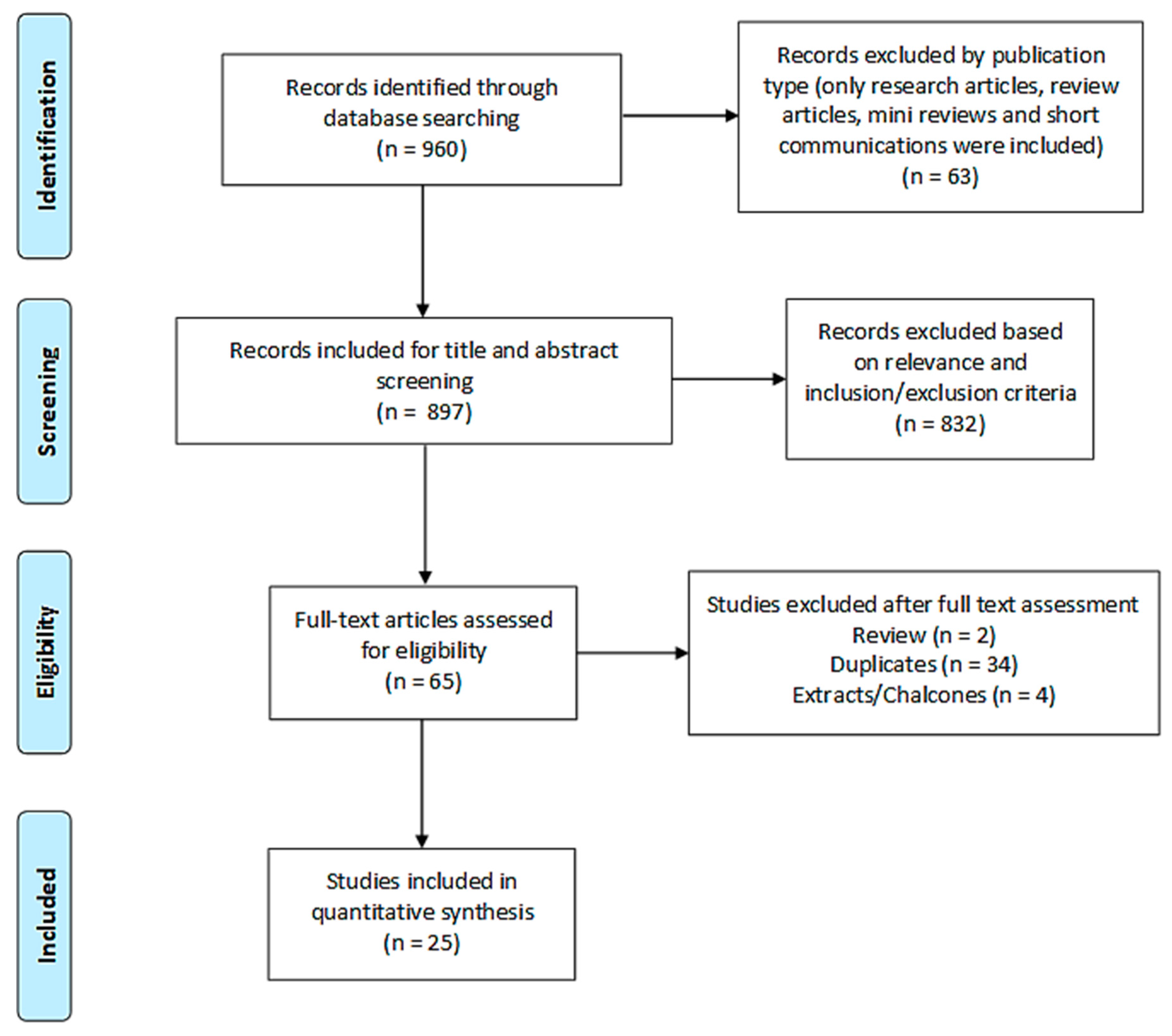
2.2.1. Search Results
2.2.2. Study Characteristics
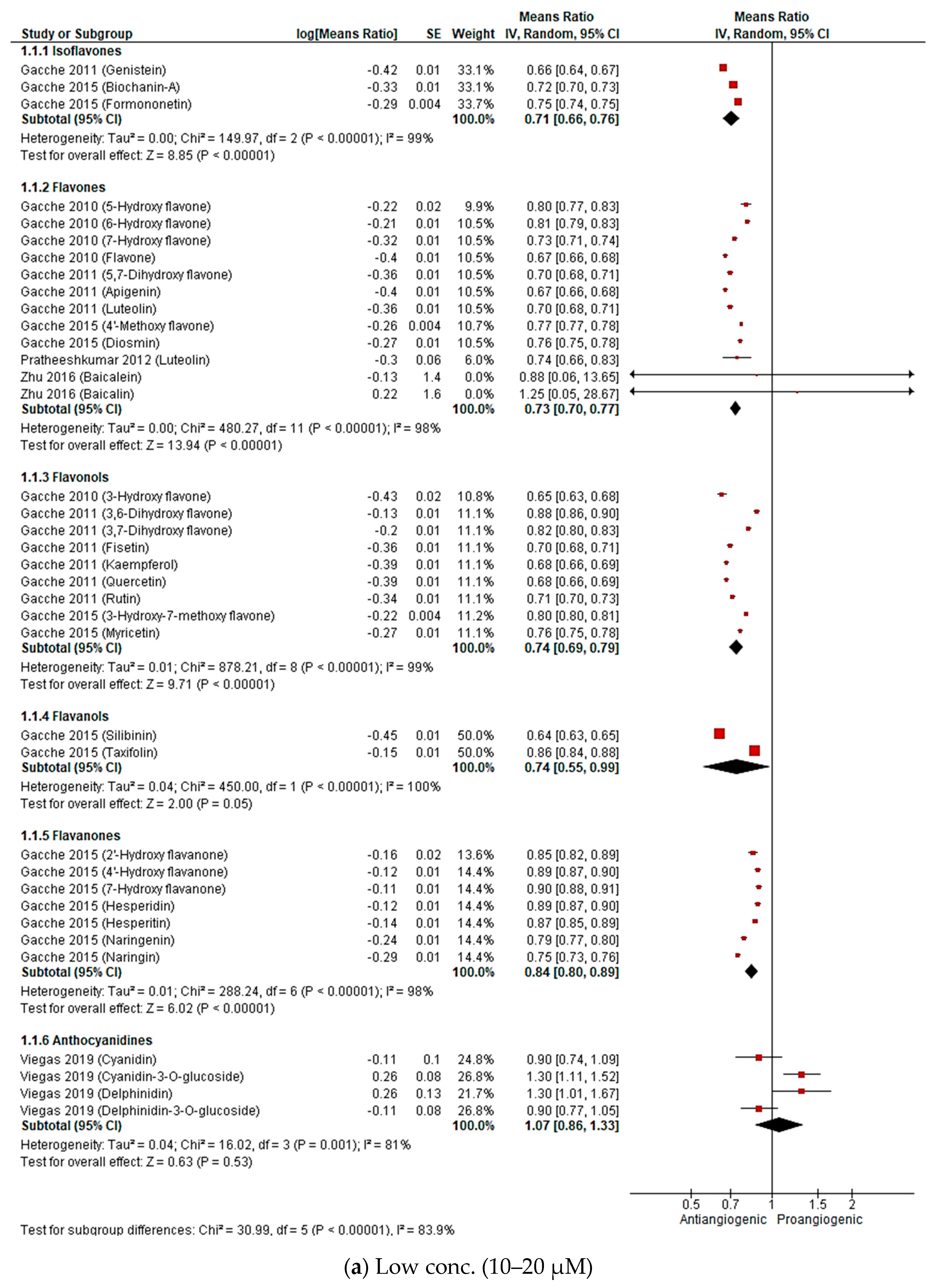
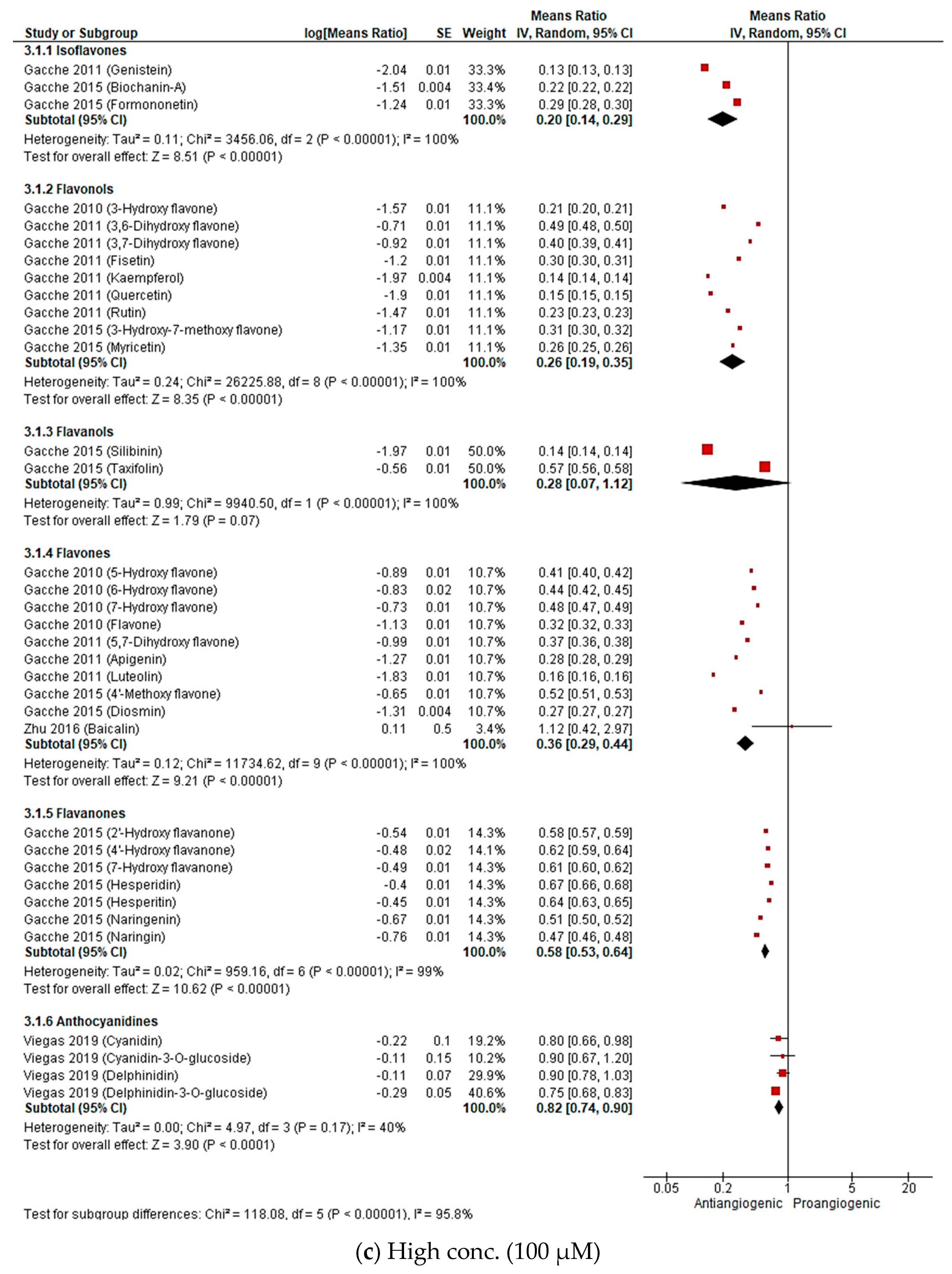
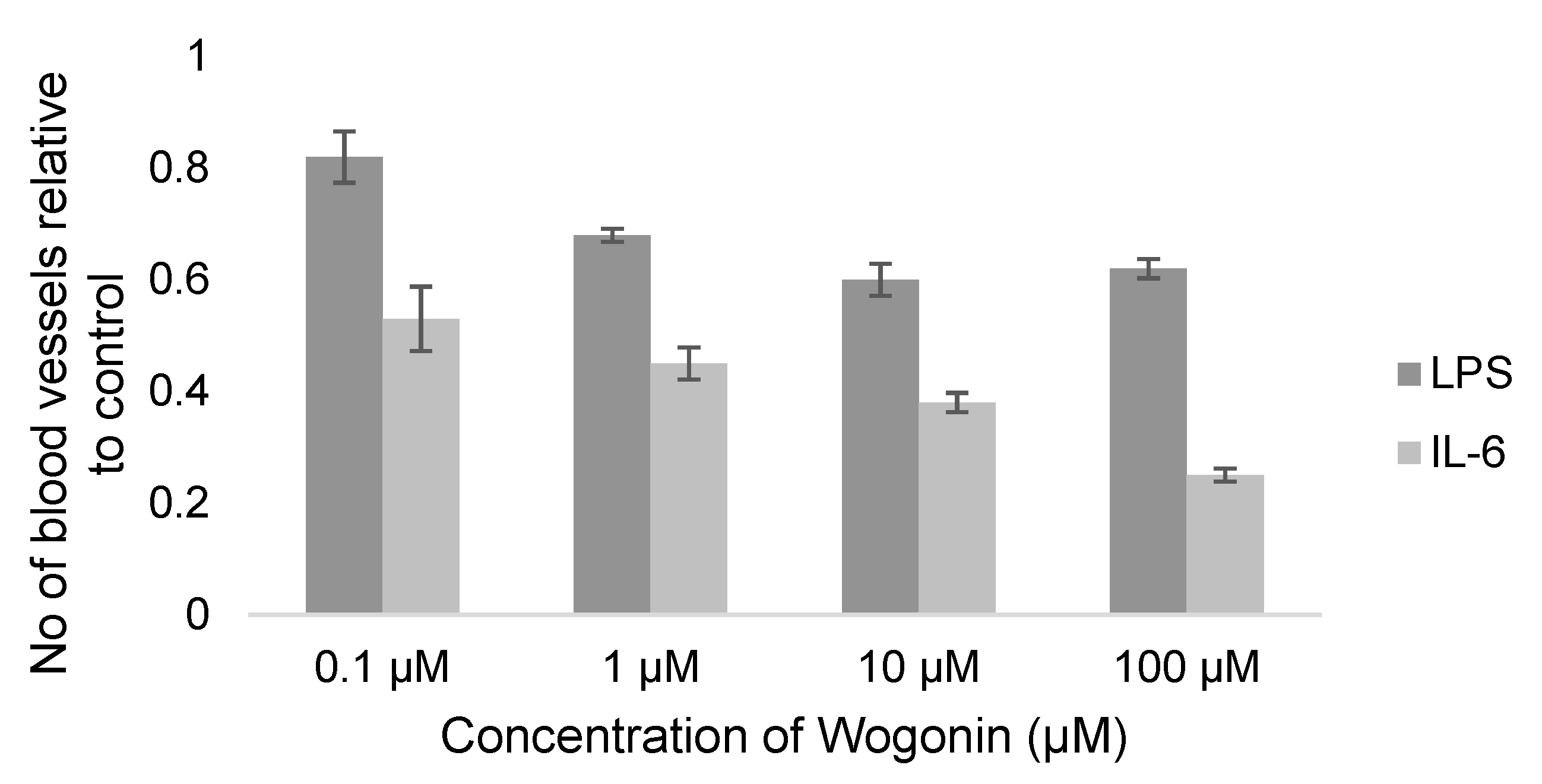
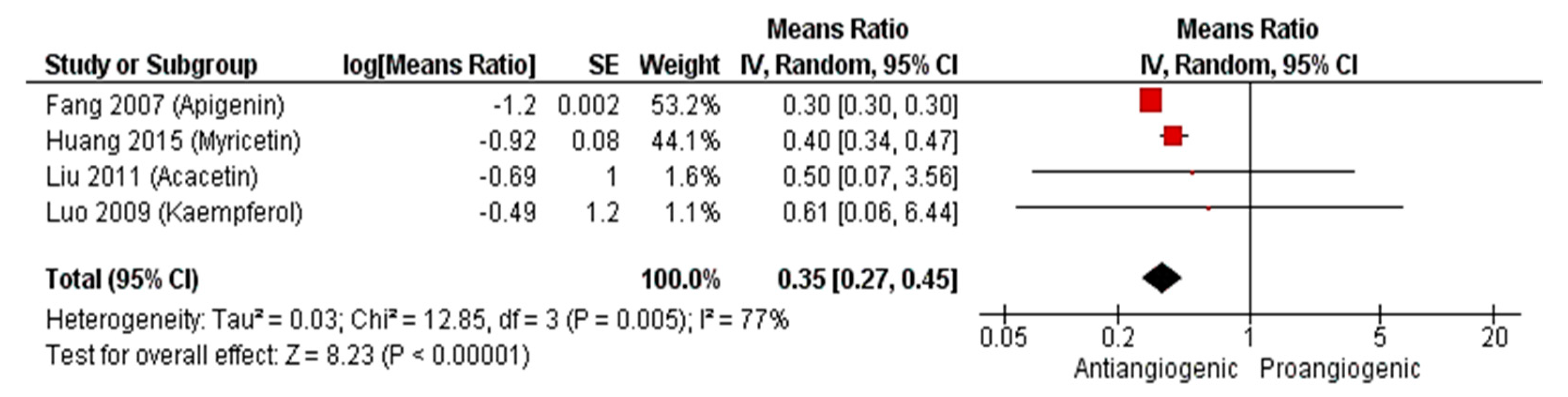
2.2.3. Meta-Analysis (Antiangiogenic Effect of Flavonoids on CAMs)
2.2.4. Sensitivity Analysis
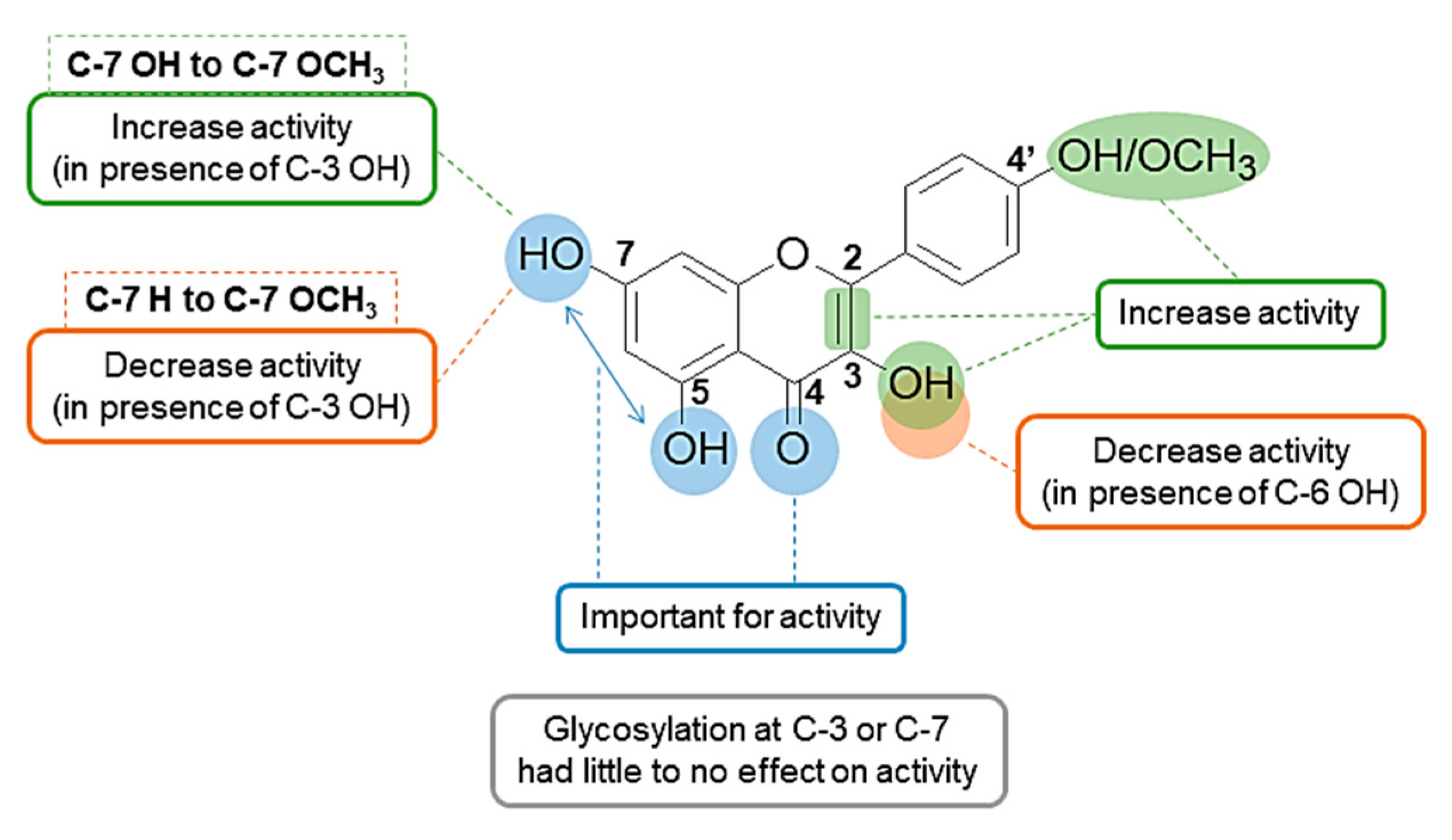
3. Discussion
4. Materials and Methods
4.1. Search Strategy
4.2. Inclusion and Exclusion Criteria
4.3. Data Extraction
4.4. Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carmeliet, P.; Jain, R.K. Molecular Mechanisms and Clinical Applications of Angiogenesis. Nature 2011, 473, 289–307. [Google Scholar] [CrossRef]
- Diniz, C.; Suliburska, J.; Ferreira, I.M. New Insights into the Antiangiogenic and Proangiogenic Properties of Dietary Polyphenols. Mol. Nutr. Food Res. 2017, 61, 1600912. [Google Scholar] [CrossRef]
- Yancopoulos, G.D.; Davis, S.; Gale, N.W.; Rudge, J.S.; Wiegand, S.J.; Holash, J. Vascular-Specific Growth Factors and Blood Vessel Formation. Nature 2000, 407, 242–248. [Google Scholar] [CrossRef]
- Shibuya, M. VEGF-VEGFR System as a Target for Suppressing Inflammation and Other Diseases. Endocr. Metab. Immune Disord. Targets 2015, 15, 135–144. [Google Scholar] [CrossRef]
- Gacche, R.N.; Meshram, R.J. Angiogenic Factors as Potential Drug Target: Efficacy and Limitations of Anti-Angiogenic Therapy. Biochim. Biophys. Acta Rev. Cancer 2014, 1846, 161–179. [Google Scholar] [CrossRef]
- Majewska, I.; Gendaszewska-Darmach, E. Proangiogenic Activity of Plant Extracts in Accelerating Wound Healing a New Face of Old Phytomedicines. Acta Biochim. Polonica 2011, 58, 449–460. [Google Scholar] [CrossRef]
- Mirossay, L.; Varinská, L.; Mojžiš, J. Antiangiogenic Effect of Flavonoids and Chalcones: An Update. Int. J. Mol. Sci. 2018, 19, 27. [Google Scholar] [CrossRef]
- Sulaiman, R.S.; Basavarajappa, H.D.; Corson, T.W. Natural Product Inhibitors of Ocular Angiogenesis. Exp. Eye Res. 2014, 129, 161–171. [Google Scholar] [CrossRef]
- Raffa, D.; Maggio, B.; Raimondi, M.V.; Plescia, F.; Daidone, G. Recent Discoveries of Anticancer Flavonoids. Eur. J. Med. Chem. 2017, 142, 213–228. [Google Scholar] [CrossRef]
- Ravishankar, D.; Rajora, A.K.; Greco, F.; Osborn, H.M.I. Flavonoids as Prospective Compounds for Anti-Cancer Therapy. Int. J. Biochem. Cell Biol. 2013, 45, 2821–2831. [Google Scholar] [CrossRef]
- Abotaleb, M.; Samuel, S.; Varghese, E.; Varghese, S.; Kubatka, P.; Liskova, A.; Büsselberg, D. Flavonoids in Cancer and Apoptosis. Cancers 2018, 11, 28. [Google Scholar] [CrossRef]
- Peluso, I.; Raguzzini, A.; Serafini, M. Effect of Flavonoids on Circulating Levels of TNF-α and IL-6 in Humans: A Systematic Review and Meta-Analysis. Mol. Nutr. Food Res. 2013, 57, 784–801. [Google Scholar] [CrossRef]
- Wang, X.; Ouyang, Y.Y.; Liu, J.; Zhao, G. Flavonoid Intake and Risk of CVD: A Systematic Review and Meta-Analysis of Prospective Cohort Studies. Br. J. Nutr. 2014, 111, 1–11. [Google Scholar] [CrossRef]
- Beking, K.; Vieira, A. Flavonoid Intake and Disability-Adjusted Life Years Due to Alzheimers and Related Dementias: A Population-Based Study Involving Twenty-Three Developed Countries. Public Health Nutr. 2010, 13, 1403–1409. [Google Scholar] [CrossRef]
- Mojzis, J.; Varinska, L.; Mojzisova, G.; Kostova, I.; Mirossay, L. Antiangiogenic Effects of Flavonoids and Chalcones. Pharmacol. Res. 2008, 57, 259–265. [Google Scholar] [CrossRef]
- Jain, R.K.; Schlenger, K.; Höckel, M.; Yuan, F. Quantitative Angiogenesis Assays: Progress and Problems. Nat. Med. 1997, 3, 1203–1208. [Google Scholar] [CrossRef]
- Norrby, K. In Vivo Models of Angiogenesis. J. Cell. Mol. Med. 2006, 10, 588–612. [Google Scholar] [CrossRef]
- Hattori, H.; Okuda, K.; Murase, T.; Shigetsura, Y.; Narise, K.; Semenza, G.L.; Nagasawa, H. Isolation, Identification, and Biological Evaluation of HIF-1-Modulating Compounds from Brazilian Green Propolis. Bioorg. Med. Chem. 2011, 19, 5392–5401. [Google Scholar] [CrossRef]
- Ramchandani, S.; Naz, I.; Lee, J.H.; Khan, M.R.; Ahn, K.S. An Overview of the Potential Antineoplastic Effects of Casticin. Molecules 2020, 25, 1287. [Google Scholar] [CrossRef]
- Yang, D.S.; Li, Z.L.; Peng, W.B.; Yang, Y.P.; Wang, X.; Liu, K.C.; Li, X.L.; Xiao, W.L. Three New Prenylated Flavonoids from Macaranga Denticulata and Their Anticancer Effects. Fitoterapia 2015, 103, 165–170. [Google Scholar] [CrossRef]
- Kashyap, D.; Sharma, A.; Sak, K.; Tuli, H.S.; Buttar, H.S.; Bishayee, A. Fisetin: A Bioactive Phytochemical with Potential for Cancer Prevention and Pharmacotherapy. Life Sci. 2018, 194, 75–87. [Google Scholar] [CrossRef]
- Rengarajan, T.; Yaacob, N.S. The Flavonoid Fisetin as an Anticancer Agent Targeting the Growth Signaling Pathways. Eur. J. Pharmacol. 2016, 789, 8–16. [Google Scholar] [CrossRef]
- Syed, D.N.; Adhami, V.M.; Khan, N.; Khan, M.I.; Mukhtar, H. Exploring the Molecular Targets of Dietary Flavonoid Fisetin in Cancer. Semin. Cancer Biol. 2016, 40, 130–140. [Google Scholar] [CrossRef]
- Lall, R.K.; Adhami, V.M.; Mukhtar, H. Dietary Flavonoid Fisetin for Cancer Prevention and Treatment. Mol. Nutr. Food Res. 2016, 60, 1396–1405. [Google Scholar] [CrossRef]
- Tsai, C.F.; Chen, J.H.; Chang, C.N.; Lu, D.Y.; Chang, P.C.; Wang, S.L.; Yeh, W.L. Fisetin Inhibits Cell Migration via Inducing HO-1 and Reducing MMPs Expression in Breast Cancer Cell Lines. Food Chem. Toxicol. 2018, 120, 528–535. [Google Scholar] [CrossRef]
- Sun, X.; Ma, X.; Li, Q.; Yang, Y.; Xu, X.; Sun, J.; Yu, M.; Cao, K.; Yang, L.; Yang, G.; et al. Anti-cancer Effects of Fisetin on Mammary Carcinoma Cells via Regulation of the PI3K/Akt/MTOR Pathway: In Vitro and in Vivo Studies. Int. J. Mol. Med. 2018, 42, 811–820. [Google Scholar] [CrossRef]
- Li, J.; Gong, X.; Jiang, R.; Lin, D.; Zhou, T.; Zhang, A.; Li, H.; Zhang, X.; Wan, J.; Kuang, G.; et al. Fisetin Inhibited Growth and Metastasis of Triple-Negative Breast Cancer by Reversing Epithelial-to-Mesenchymal Transition via PTEN/Akt/GSK3β Signal Pathway. Front. Pharmacol. 2018, 9, 772. [Google Scholar] [CrossRef]
- Chou, R.H.; Hsieh, S.C.; Yu, Y.L.; Huang, M.H.; Huang, Y.C.; Hsieh, Y.H. Fisetin Inhibits Migration and Invasion of Human Cervical Cancer Cells by Down-Regulating Urokinase Plasminogen Activator Expression through Suppressing the P38 MAPK-Dependent NF-ΚB Signaling Pathway. PLoS ONE 2013, 8, e71983. [Google Scholar] [CrossRef]
- Chen, C.M.; Hsieh, Y.H.; Hwang, J.M.; Jan, H.J.; Hsieh, S.C.; Lin, S.H.; Lai, C.Y. Fisetin Suppresses ADAM9 Expression and Inhibits Invasion of Glioma Cancer Cells through Increased Phosphorylation of ERK1/2. Tumor Biol. 2015, 36, 3407–3415. [Google Scholar] [CrossRef]
- Liu, X.F.; Long, H.J.; Miao, X.Y.; Liu, G.L.; Yao, H.L. Fisetin Inhibits Liver Cancer Growth in a Mouse Model: Relation to Dopamine Receptor. Oncol. Rep. 2017, 38, 53–62. [Google Scholar] [CrossRef]
- Klimaszewska-Wiśniewska, A.; Grzanka, D.; Czajkowska, P.; Hałas-Wiśniewska, M.; Durślewicz, J.; Antosik, P.; Grzanka, A.; Gagat, M. Cellular and Molecular Alterations Induced by Low-dose Fisetin in Human Chronic Myeloid Leukemia Cells. Int. J. Oncol. 2019, 55, 1261–1274. [Google Scholar] [CrossRef]
- Tabasum, S.; Singh, R.P. Fisetin Suppresses Migration, Invasion and Stem-Cell-like Phenotype of Human Non-Small Cell Lung Carcinoma Cells via Attenuation of Epithelial to Mesenchymal Transition. Chem. Biol. Interact. 2019, 303, 14–21. [Google Scholar] [CrossRef]
- Wang, J.; Huang, S. Fisetin Inhibits the Growth and Migration in the A549 Human Lung Cancer Cell Line via the ERK1/2 Pathway. Exp. Ther. Med. 2018, 15, 2667–2673. [Google Scholar] [CrossRef]
- Chien, C.S.; Shen, K.H.; Huang, J.S.; Ko, S.C.; Shih, Y.W. Antimetastatic Potential of Fisetin Involves Inactivation of the PI3K/Akt and JNK Signaling Pathways with Downregulation of MMP-2/9 Expressions in Prostate Cancer PC-3 Cells. Mol. Cell. Biochem. 2010, 333, 169–180. [Google Scholar] [CrossRef]
- Hsieh, M.-H.; Tsai, J.-P.; Yang, S.-F.; Chiou, H.-L.; Lin, C.-L.; Hsieh, Y.-H.; Chang, H.-R. Fisetin Suppresses the Proliferation and Metastasis of Renal Cell Carcinoma through Upregulation of MEK/ERK-Targeting CTSS and ADAM9. Cells 2019, 8, 948. [Google Scholar] [CrossRef]
- Joussen, A.M. Treatment of Corneal Neovascularization with Dietary Isoflavonoids and Flavonoids. Exp. Eye Res. 2000, 71, 483–487. [Google Scholar] [CrossRef]
- Fang, D.; Xiong, Z.; Xu, J.; Yin, J.; Luo, R. Chemopreventive Mechanisms of Galangin against Hepatocellular Carcinoma: A Review. Biomed. Pharmacother. 2019, 109, 2054–2061. [Google Scholar] [CrossRef]
- Kim, J.D.; Liu, L.; Guo, W.; Meydani, M. Chemical Structure of Flavonols in Relation to Modulation of Angiogenesis and Immune-Endothelial Cell Adhesion. J. Nutr. Biochem. 2006, 17, 165–176. [Google Scholar] [CrossRef]
- Chen, D.; Li, D.; Xu, X.; Qiu, S.; Luo, S.; Qiu, E.; Rong, Z.; Zhang, J.; Zheng, D. Galangin Inhibits Epithelial-Mesenchymal Transition and Angiogenesis by Downregulating CD44 in Glioma. J. Cancer 2019, 10, 4499–4508. [Google Scholar] [CrossRef]
- Lei, D.; Zhang, F.; Yao, D.; Xiong, N.; Jiang, X.; Zhao, H. Galangin Increases ERK1/2 Phosphorylation to Decrease ADAM9 Expression and Prevents Invasion in A172 Glioma Cells. Mol. Med. Rep. 2018, 17, 667–673. [Google Scholar] [CrossRef]
- Chien, S.-T.; Shi, M.-D.; Lee, Y.-C.; Te, C.-C.; Shih, Y.-W. Galangin, a Novel Dietary Flavonoid, Attenuates Metastatic Feature via PKC/ERK Signaling Pathway in TPA-Treated Liver Cancer HepG2 Cells. Cancer Cell Int. 2015, 15, 15. [Google Scholar] [CrossRef]
- Huang, H.; Chen, A.Y.; Rojanasakul, Y.; Ye, X.; Rankin, G.O.; Chen, Y.C. Dietary Compounds Galangin and Myricetin Suppress Ovarian Cancer Cell Angiogenesis. J. Funct. Foods 2015, 15, 464–475. [Google Scholar] [CrossRef]
- Cao, J.; Wang, H.; Chen, F.; Fang, J.; Xu, A.; Xi, W.; Zhang, S.; Wu, G.; Wang, Z. Galangin Inhibits Cell Invasion by Suppressing the Epithelial-Mesenchymal Transition and Inducing Apoptosis in Renal Cell Carcinoma. Mol. Med. Rep. 2016, 13, 4238–4244. [Google Scholar] [CrossRef]
- Zhu, Y.; Rao, Q.; Zhang, X.; Zhou, X. Galangin Induced Antitumor Effects in Human Kidney Tumor Cells Mediated via Mitochondrial Mediated Apoptosis, Inhibition of Cell Migration and Invasion and Targeting PI3K/AKT/MTOR Signalling Pathway. J. BUON 2018, 23, 795–799. [Google Scholar]
- Wang, L.; Wang, X.; Chen, H.; Zu, X.; Ma, F.; Liu, K.; Bode, A.M.; Dong, Z.; Kim, D.J. Gossypin Inhibits Gastric Cancer Growth by Direct Targeting AURKA and RSK2. Phyther. Res. 2019, 33, 640–650. [Google Scholar] [CrossRef]
- Li, L.; Fan, P.; Chou, H.; Li, J.; Wang, K.; Li, H. Herbacetin Suppressed MMP9 Mediated Angiogenesis of Malignant Melanoma through Blocking EGFR-ERK/AKT Signaling Pathway. Biochimie 2019, 162, 198–207. [Google Scholar] [CrossRef]
- Jin, X.N.; Yan, E.Z.; Wang, H.M.; Sui, H.J.; Liu, Z.; Gao, W.; Jin, Y. Hyperoside Exerts Anti-Inflammatory and Anti-Arthritic Effects in LPS-Stimulated Human Fibroblast-like Synoviocytes in Vitro and in Mice with Collagen-Induced Arthritis. Acta Pharmacol. Sin. 2016, 37, 674–686. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, T.; Huang, Y.; Wismeijer, D.; Liu, Y. Icariin: Does It Have an Osteoinductive Potential for Bone Tissue Engineering? Phyther. Res. 2014, 28, 498–509. [Google Scholar] [CrossRef]
- Chen, M.; Wu, J.; Luo, Q.; Mo, S.; Lyu, Y.; Wei, Y.; Dong, J. The Anticancer Properties of Herba Epimedii and Its Main Bioactive Componentsicariin and Icariside II. Nutrients 2016, 8, 563. [Google Scholar] [CrossRef]
- Chung, B.H.; Kim, J.D.; Kim, C.K.; Kim, J.W.; Won, M.H.; Lee, H.S.; Dong, M.S.; Ha, K.S.; Kwon, Y.G.; Kim, Y.M. Icariin Stimulates Angiogenesis by Activating the MEK/ERK- and PI3K/Akt/ENOS-Dependent Signal Pathways in Human Endothelial Cells. Biochem. Biophys. Res. Commun. 2008, 376, 404–408. [Google Scholar] [CrossRef]
- Gu, Z.F.; Zhang, Z.T.; Wang, J.Y.; Xu, B.B. Icariin Exerts Inhibitory Effects on the Growth and Metastasis of KYSE70 Human Esophageal Carcinoma Cells via PI3K/AKT and STAT3 Pathways. Environ. Toxicol. Pharmacol. 2017, 54, 7–13. [Google Scholar] [CrossRef]
- Wang, P.; Zhang, J.; Xiong, X.; Yuan, W.; Qin, S.; Cao, W.; Dai, L.; Xie, F.; Li, A.; Liu, Z. Icariin Suppresses Cell Cycle Transition and Cell Migration in Ovarian Cancer Cells. Oncol. Rep. 2019, 41, 2321–2328. [Google Scholar] [CrossRef]
- Singh, W.R.; Devi, H.S.; Kumawat, S.; Sadam, A.; Appukuttan, A.V.; Patel, M.R.; Lingaraju, M.C.; Singh, T.U.; Kumar, D. Angiogenic and MMPs Modulatory Effects of Icariin Improved Cutaneous Wound Healing in Rats. Eur. J. Pharmacol. 2019, 858, 172466. [Google Scholar] [CrossRef]
- Quan, K.; Zhang, X.; Fan, K.; Liu, P.; Yue, Q.; Li, B.; Wu, J.; Liu, B.; Xu, Y.; Hua, W.; et al. Icariside II Induces Cell Cycle Arrest and Apoptosis in Human Glioblastoma Cells through Suppressing Akt Activation and Potentiating FOXO3A Activity. Am. J. Transl. Res. 2017, 9, 2508–2519. [Google Scholar]
- Xing, S.; Yu, W.; Zhang, X.; Luo, Y.; Lei, Z.; Huang, D.; Lin, J.; Huang, Y.; Huang, S.; Nong, F.; et al. Isoviolanthin Extracted from Dendrobium Officinale Reverses TGF-Β1-Mediated Epithelial–Mesenchymal Transition in Hepatocellular Carcinoma Cells via Deactivating the TGF-β/Smad and PI3K/Akt/MTOR Signaling Pathways. Int. J. Mol. Sci. 2018, 19, 1556. [Google Scholar] [CrossRef]
- Chen, A.Y.; Chen, Y.C. A Review of the Dietary Flavonoid, Kaempferol on Human Health and Cancer Chemoprevention. Food Chem. 2013, 138, 2099–2107. [Google Scholar] [CrossRef]
- Kashyap, D.; Sharma, A.; Tuli, H.S.; Sak, K.; Punia, S.; Mukherjee, T.K. Kaempferol—A Dietary Anticancer Molecule with Multiple Mechanisms of Action: Recent Trends and Advancements. J. Funct. Foods 2017, 30, 203–219. [Google Scholar] [CrossRef]
- Chin, H.K.; Horng, C.T.; Liu, Y.S.; Lu, C.C.; Su, C.Y.; Chen, P.S.; Chiu, H.Y.; Tsai, F.J.; Shieh, P.C.; Yang, J.S. Kaempferol Inhibits Angiogenic Ability by Targeting VEGF Receptor-2 and Downregulating the PI3K/AKT, MEK and ERK Pathways in VEGF-Stimulated Human Umbilical Vein Endothelial Cells. Oncol. Rep. 2018, 39, 2351–2357. [Google Scholar] [CrossRef]
- Liang, F.; Han, Y.; Gao, H.; Xin, S.; Chen, S.; Wang, N.; Qin, W.; Zhong, H.; Lin, S.; Yao, X.; et al. Kaempferol Identified by Zebrafish Assay and Fine Fractionations Strategy from Dysosma versipellis Inhibits Angiogenesis through VEGF and FGF Pathways. Sci. Rep. 2015, 5, 14468. [Google Scholar] [CrossRef]
- Kumazawa, S.; Kubota, S.; Yamamoto, H.; Okamura, N.; Sugiyama, Y.; Kobayashi, H.; Nakanishi, M.; Ohta, T. Antiangiogenic Activity of Flavonoids from Melia Azedarach. Nat. Prod. Commun. 2013, 8, 1719–1720. [Google Scholar] [CrossRef]
- Özay, Y.; Güzel, S.; Yumrutaş, Ö.; Pehlivanoğlu, B.; Erdoğdu, İ.H.; Yildirim, Z.; Türk, B.A.; Darcan, S. Wound Healing Effect of Kaempferol in Diabetic and Nondiabetic Rats. J. Surg. Res. 2019, 233, 284–296. [Google Scholar] [CrossRef]
- Sharma, V.; Joseph, C.; Ghosh, S.; Agarwal, A.; Mishra, M.K.; Sen, E. Kaempferol Induces Apoptosis in Glioblastoma Cells through Oxidative Stress. Mol. Cancer Ther. 2007, 6, 2544–2553. [Google Scholar] [CrossRef]
- Shen, S.C.; Lin, C.W.; Lee, H.M.; Chien, L.L.; Chen, Y.C. Lipopolysaccharide plus 12-o-Tetradecanoylphorbol 13-Acetate Induction of Migration and Invasion of Glioma Cells in Vitro and in Vivo: Differential Inhibitory Effects of Flavonoids. Neuroscience 2006, 140, 477–489. [Google Scholar] [CrossRef]
- Qin, Y.; Cui, W.; Yang, X.; Tong, B. Kaempferol Inhibits the Growth and Metastasis of Cholangiocarcinoma in Vitro and in Vivo. Acta Biochim. Biophys. Sin. (Shanghai). 2015, 48, 238–245. [Google Scholar] [CrossRef]
- Zhu, G.; Liu, X.; Li, H.; Yan, Y.; Hong, X.; Lin, Z. Kaempferol Inhibits Proliferation, Migration, and Invasion of Liver Cancer HepG2 Cells by down-Regulation of MicroRNA-21. Int. J. Immunopathol. Pharmacol. 2018, 32, 2058738418814341. [Google Scholar] [CrossRef]
- Jo, E.; Park, S.J.; Choi, Y.S.; Jeon, W.K.; Kim, B.C. Kaempferol Suppresses Transforming Growth Factor-Β1-Induced Epithelial-to-Mesenchymal Transition and Migration of A549 Lung Cancer Cells by Inhibiting Akt1-Mediated Phosphorylation of Smad3 at Threonine-179. Neoplasia 2015, 17, 525–537. [Google Scholar] [CrossRef]
- Labbé, D.; Provençal, M.; Lamy, S.; Boivin, D.; Gingras, D.; Béliveau, R. The Flavonols Quercetin, Kaempferol, and Myricetin Inhibit Hepatocyte Growth. J. Nutr. Biochem. Mol. Genet. Mech. 2009, 139, 646–652. [Google Scholar] [CrossRef]
- Lin, C.W.; Chen, P.N.; Chen, M.K.; Yang, W.E.; Tang, C.H.; Yang, S.F.; Hsieh, Y.S. Kaempferol Reduces Matrix Metalloproteinase-2 Expression by down-Regulating ERK1/2 and the Activator Protein-1 Signaling Pathways in Oral Cancer Cells. PLoS ONE 2013, 8, e80883. [Google Scholar] [CrossRef]
- Chen, H.J.; Lin, C.M.; Lee, C.Y.; Shih, N.C.; Peng, S.F.; Tsuzuki, M.; Amagaya, S.; Huang, W.W.; Yang, J.S. Kaempferol Suppresses Cell Metastasis via Inhibition of the ERK-P38-JNK and AP-1 Signaling Pathways in U-2 OS Human Osteosarcoma Cells. Oncol. Rep. 2013, 30, 925–932. [Google Scholar] [CrossRef]
- Luo, H.; Rankin, G.O.; Liu, L.; Daddysman, M.K.; Jiang, B.H.; Chen, Y.C. Kaempferol Inhibits Angiogenesis and VEGF Expression through Both HIF Dependent and Independent Pathways in Human Ovarian Cancer Cells. Nutr. Cancer 2009, 61, 554–563. [Google Scholar] [CrossRef]
- Lee, J.; Kim, J.H. Kaempferol Inhibits Pancreatic Cancer Cell Growth and Migration through the Blockade of EGFR-Related Pathway in Vitro. PLoS ONE 2016, 11, e0155264. [Google Scholar] [CrossRef]
- Hung, T.W.; Chen, P.N.; Wu, H.C.; Wu, S.W.; Tsai, P.Y.; Hsieh, Y.S.; Chang, H.R. Kaempferol Inhibits the Invasion and Migration of Renal Cancer Cells through the Downregulation of AKT and FAK Pathways. Int. J. Med. Sci. 2017, 14, 984–993. [Google Scholar] [CrossRef]
- Chien, H.W.; Wang, K.; Chang, Y.Y.; Hsieh, Y.H.; Yu, N.Y.; Yang, S.F.; Lin, H.W. Kaempferol Suppresses Cell Migration through the Activation of the ERK Signaling Pathways in ARPE-19 Cells. Environ. Toxicol. 2019, 34, 312–318. [Google Scholar] [CrossRef]
- Clericuzio, M.; Tinello, S.; Burlando, B.; Ranzato, E.; Martinotti, S.; Cornara, L.; La Rocca, A. Flavonoid Oligoglycosides from Ophioglossum vulgatum L. Having Wound Healing Properties. Planta Med. 2012, 78, 1639–1644. [Google Scholar] [CrossRef]
- Zeng, N.; Tong, B.; Zhang, X.; Dou, Y.; Wu, X.; Xia, Y.; Dai, Y.; Wei, Z. Antiarthritis Effect of Morin Is Associated with Inhibition of Synovial Angiogensis. Drug Dev. Res. 2015, 76, 463–473. [Google Scholar] [CrossRef]
- Yue, M.; Zeng, N.; Xia, Y.; Wei, Z.; Dai, Y. Morin Exerts Anti-Arthritic Effects by Attenuating Synovial Angiogenesis via Activation of Peroxisome Proliferator Activated Receptor-γ. Mol. Nutr. Food Res. 2018, 62, 1800202. [Google Scholar] [CrossRef]
- Capitani, N.; Lori, G.; Paoli, P.; Patrussi, L.; Troilo, A.; Baldari, C.T.; Raugei, G.; D’Elios, M.M. LMW-PTP Targeting Potentiates the Effects of Drugs Used in Chronic Lymphocytic Leukemia Therapy. Cancer Cell Int. 2019, 19, 67. [Google Scholar] [CrossRef]
- Li, H.W.; Zou, T.-B.; Jia, Q.; Xia, E.Q.; Cao, W.J.; Liu, W.; He, T.P.; Wang, Q. Anticancer Effects of Morin-7-Sulphate Sodium, a Flavonoid Derivative, in Mouse Melanoma Cells. Biomed. Pharmacother. 2016, 84, 909–916. [Google Scholar] [CrossRef]
- Kang, N.J.; Jung, S.K.; Lee, K.W.; Lee, H.J. Myricetin Is a Potent Chemopreventive Phytochemical in Skin Carcinogenesis. Ann. N. Y. Acad. Sci. 2011, 1229, 124–132. [Google Scholar] [CrossRef]
- Zhou, Z.; Mao, W.; Li, Y.; Qi, C.; He, Y. Myricetin Inhibits Breast Tumor Growth and Angiogenesis by Regulating VEGF/VEGFR2 and P38MAPK Signaling Pathways. Anat. Rec. 2019, 302, 2186–2192. [Google Scholar] [CrossRef]
- Ci, Y.; Zhang, Y.; Liu, Y.; Lu, S.; Cao, J.; Li, H.; Zhang, J.; Huang, Z.; Zhu, X.; Gao, J.; et al. Myricetin Suppresses Breast Cancer Metastasis through Down-Regulating the Activity of Matrix Metalloproteinase (MMP)-2/9. Phyther. Res. 2018, 32, 1373–1381. [Google Scholar] [CrossRef]
- Chiu, W.T.; Shen, S.C.; Chow, J.M.; Lin, C.W.; Shia, L.T.; Chen, Y.C. Contribution of Reactive Oxygen Species to Migration/Invasion of Human Glioblastoma Cells U87 via ERK-Dependent COX-2/PGE2 Activation. Neurobiol. Dis. 2010, 37, 118–129. [Google Scholar] [CrossRef]
- Yamada, N.; Matsushima-Nishiwaki, R.; Kozawa, O. Quercetin Suppresses the Migration of Hepatocellular Carcinoma Cells Stimulated by Hepatocyte Growth Factor or Transforming Growth Factor-α: Attenuation of AKT Signaling Pathway. Arch. Biochem. Biophys. 2020, 682, 108296. [Google Scholar] [CrossRef]
- Ma, H.; Zhu, L.; Ren, J.; Rao, B.; Sha, M.; Kuang, Y.; Shen, W.; Xu, Z. Myricetin Inhibits Migration and Invasion of Hepatocellular Carcinoma MHCC97H Cell Line by Inhibiting the EMT Process. Oncol. Lett. 2019, 18, 6614–6620. [Google Scholar] [CrossRef]
- Shih, Y.W.; Wu, P.F.; Lee, Y.C.; Shi, M.-D.; Chiang, T.A. Myricetin Suppresses Invasion and Migration of Human Lung Adenocarcinoma A549 Cells: Possible Mediation by Blocking the Erk Signaling Pathway. J. Agric. Food Chem. 2009, 57, 3490–3499. [Google Scholar] [CrossRef]
- Ezzati, M.; Yousefi, B.; Velaei, K.; Safa, A. A Review on Anti-Cancer Properties of Quercetin in Breast Cancer. Life Sci. 2020, 248, 117463. [Google Scholar] [CrossRef]
- Tang, S.; Deng, X.; Zhou, J.; Li, Q.; Ge, X.; Miao, L. Pharmacological Basis and New Insights of Quercetin Action in Respect to Its Anti-Cancer Effects. Biomed. Pharmacother. 2020, 121, 109604. [Google Scholar] [CrossRef]
- Kashyap, D.; Mittal, S.; Sak, K.; Singhal, P.; Tuli, H.S. Molecular Mechanisms of Action of Quercetin in Cancer: Recent Advances. Tumor Biol. 2016, 37, 12927–12939. [Google Scholar] [CrossRef]
- Darband, S.G.; Kaviani, M.; Yousefi, B.; Sadighparvar, S.; Pakdel, F.G.; Attari, J.A.; Mohebbi, I.; Naderi, S.; Majidinia, M. Quercetin: A Functional Dietary Flavonoid with Potential Chemo-Preventive Properties in Colorectal Cancer. J. Cell. Physiol. 2018, 233, 6544–6560. [Google Scholar] [CrossRef]
- Song, N.R.; Chung, M.Y.; Kang, N.J.; Seo, S.G.; Jang, T.S.; Lee, H.J.; Lee, K.W. Quercetin Suppresses Invasion and Migration of H-Ras-Transformed MCF10A Human Epithelial Cells by Inhibiting Phosphatidylinositol 3-Kinase. Food Chem. 2014, 142, 66–71. [Google Scholar] [CrossRef]
- Lin, C.; Wu, M.; Dong, J. Quercetin-4′-o-β-d-Glucopyranoside (QODG) Inhibits Angiogenesis by Suppressing VEGFR2-Mediated Signaling in Zebrafish and Endothelial Cells. PLoS ONE 2012, 7, e31708. [Google Scholar] [CrossRef]
- Lee, Y.H.; Tuyet, P.T. Synthesis and Biological Evaluation of Quercetin–Zinc (II) Complex for Anti-Cancer and Anti-Metastasis of Human Bladder Cancer Cells. Vitr. Cell. Dev. Biol. Anim. 2019, 55, 395–404. [Google Scholar] [CrossRef]
- Ravishankar, D.; Watson, K.A.; Boateng, S.Y.; Green, R.J.; Greco, F.; Osborn, H.M.I. Exploring Quercetin and Luteolin Derivatives as Antiangiogenic Agents. Eur. J. Med. Chem. 2015, 97, 259–274. [Google Scholar] [CrossRef]
- Zhao, X.; Wang, Q.; Yang, S.; Chen, C.; Li, X.; Liu, J.; Zou, Z.; Cai, D. Quercetin Inhibits Angiogenesis by Targeting Calcineurin in the Xenograft Model of Human Breast Cancer. Eur. J. Pharmacol. 2016, 781, 60–68. [Google Scholar] [CrossRef]
- Oh, S.J.; Kim, O.; Lee, J.S.; Kim, J.A.; Kim, M.R.; Choi, H.S.; Shim, J.H.; Kang, K.W.; Kim, Y.C. Inhibition of Angiogenesis by Quercetin in Tamoxifen-Resistant Breast Cancer Cells. Food Chem. Toxicol. 2010, 48, 3227–3234. [Google Scholar] [CrossRef]
- Srinivasan, A.; Thangavel, C.; Liu, Y.; Shoyele, S.; Den, R.B.; Selvakumar, P.; Lakshmikuttyamma, A. Quercetin Regulates β-Catenin Signaling and Reduces the Migration of Triple Negative Breast Cancer. Mol. Carcinog. 2016, 55, 743–756. [Google Scholar] [CrossRef]
- Lin, C.W.; Hou, W.C.; Shen, S.C.; Juan, S.H.; Ko, C.H.; Wang, L.M.; Chen, Y.C. Quercetin Inhibition of Tumor Invasion via Suppressing PKCδ/ERK/AP-1-Dependent Matrix Metalloproteinase-9 Activation in Breast Carcinoma Cells. Carcinogenesis 2008, 29, 1807–1815. [Google Scholar] [CrossRef]
- Rivera Rivera, A.; Castillo-Pichardo, L.; Gerena, Y.; Dharmawardhane, S. Anti-Breast Cancer Potential of Quercetin via the Akt/AMPK/Mammalian Target of Rapamycin (MTOR) Signaling Cascade. PLoS ONE 2016, 11, e0157251. [Google Scholar] [CrossRef]
- Zhao, D.; Qin, C.; Fan, X.; Li, Y.; Gu, B. Inhibitory Effects of Quercetin on Angiogenesis in Larval Zebra Fi Sh and Human Umbilical Vein Endothelial Cells. Eur. J. Pharmacol. 2014, 723, 360–367. [Google Scholar] [CrossRef]
- Tan, W.F.; Lin, L.P.; Li, M.H.; Zhang, Y.X.; Tong, Y.G.; Xiao, D.; Ding, J. Quercetin, a Dietary-Derived Flavonoid, Possesses Antiangiogenic Potential. Eur. J. Pharmacol. 2003, 459, 255–262. [Google Scholar] [CrossRef]
- Zhao, X.; Shu, G.; Chen, L.; Mi, X.; Mei, Z.; Deng, X. A Flavonoid Component from Docynia delavayi (Franch.) Schneid Represses Transplanted H22 Hepatoma Growth and Exhibits Low Toxic Effect on Tumor-Bearing Mice. Food Chem. Toxicol. 2012, 50, 3166–3168. [Google Scholar] [CrossRef]
- Lee, L.T.; Huang, Y.T.; Hwang, J.J.; Lee, A.Y.L.; Ke, F.C.; Huang, C.J.; Kandaswami, C.; Lee, P.P.H.; Lee, M.T. Transinactivation of the Epidermal Growth Factor Receptor Tyrosine Kinase and Focal Adhesion Kinase Phosphorylation by Dietary Flavonoids: Effect on Invasive Potential of Human Carcinoma Cells. Biochem. Pharmacol. 2004, 67, 2103–2114. [Google Scholar] [CrossRef]
- Lee, D.E.; Chung, M.Y.; Lim, T.G.; Huh, W.B.; Lee, H.J.; Lee, K.W. Quercetin Suppresses Intracellular Ros Formation, MMP Activation, and Cell Motility in Human Fibrosarcoma Cells. J. Food Sci. 2013, 78. [Google Scholar] [CrossRef]
- Kee, J.Y.; Han, Y.H.; Kim, D.S.; Mun, J.G.; Park, J.; Jeong, M.Y.; Um, J.Y.; Hong, S.H. Inhibitory Effect of Quercetin on Colorectal Lung Metastasis through Inducing Apoptosis, and Suppression of Metastatic Ability. Phytomedicine 2016, 23, 1680–1690. [Google Scholar] [CrossRef]
- Pan, H.C.; Jiang, Q.; Yu, Y.; Mei, J.P.; Cui, Y.K.; Zhao, W.J. Quercetin Promotes Cell Apoptosis and Inhibits the Expression of MMP-9 and Fibronectin via the AKT and ERK Signalling Pathways in Human Glioma Cells. Neurochem. Int. 2015, 80, 60–71. [Google Scholar] [CrossRef]
- Liu, Y.; Tang, Z.G.; Lin, Y.; Qu, X.G.; Lv, W.; Wang, G.-B.; Li, C.L. Effects of Quercetin on Proliferation and Migration of Human Glioblastoma U251 Cells. Biomed. Pharmacother. 2017, 92, 33–38. [Google Scholar] [CrossRef]
- Michaud-Levesque, J.; Bousquet-Gagnon, N.; Béliveau, R. Quercetin Abrogates IL-6/STAT3 Signaling and Inhibits Glioblastoma Cell Line Growth and Migration. Exp. Cell Res. 2012, 318, 925–935. [Google Scholar] [CrossRef]
- da Silva, A.B.; Cerqueira Coelho, P.L.; das Neves Oliveira, M.; Oliveira, J.L.; Oliveira Amparo, J.A.; da Silva, K.C.; Soares, J.R.P.; Pitanga, B.P.S.; dos Santos Souza, C.; de Faria Lopes, G.P.; et al. The Flavonoid Rutin and Its Aglycone Quercetin Modulate the Microglia Inflammatory Profile Improving Antiglioma Activity. Brain. Behav. Immun. 2020, 85, 170–185. [Google Scholar] [CrossRef]
- Liu, Y.; Tang, Z.G.; Yang, J.Q.; Zhou, Y.; Meng, L.H.; Wang, H.; Li, C.L. Low Concentration of Quercetin Antagonizes the Invasion and Angiogenesis of Human Glioblastoma U251 Cells. OncoTargets. Ther. 2017, 10, 4023–4028. [Google Scholar] [CrossRef]
- Lu, J.; Wang, Z.; Li, S.; Xin, Q.; Yuan, M.; Li, H.; Song, X.; Gao, H.; Pervaiz, N.; Sun, X.; et al. Quercetin Inhibits the Migration and Invasion of HCCLM3 Cells by Suppressing the Expression of P-Akt1, Matrix Metalloproteinase (MMP) MMP-2, and MMP-9. Med. Sci. Monit. 2018, 24, 2583–2589. [Google Scholar] [CrossRef]
- Klimaszewska-Wiśniewska, A.; Hałas-Wiśniewska, M.; Izdebska, M.; Gagat, M.; Grzanka, A.; Grzanka, D. Antiproliferative and Antimetastatic Action of Quercetin on A549 Non-Small Cell Lung Cancer Cells through Its Effect on the Cytoskeleton. Acta Histochem. 2017, 119, 99–112. [Google Scholar] [CrossRef]
- Wu, T.C.; Chan, S.T.; Chang, C.N.; Yu, P.S.; Chuang, C.H.; Yeh, S.L. Quercetin and Chrysin Inhibit Nickel-Induced Invasion and Migration by Downregulation of TLR4/NF-ΚB Signaling in A549 cells. Chem. Biol. Interact. 2018, 292, 101–109. [Google Scholar] [CrossRef]
- Lin, Y.C.; Tsai, P.H.; Lin, C.Y.; Cheng, C.H.; Lin, T.H.; Lee, K.P.H.; Huang, K.Y.; Chen, S.H.; Hwang, J.J.; Kandaswami, C.C.; et al. Impact of Flavonoids on Matrix Metalloproteinase Secretion and Invadopodia Formation in Highly Invasive A431-III Cancer Cells. PLoS ONE 2013, 8, e71903. [Google Scholar] [CrossRef]
- Hwang, M.K.; Song, N.R.; Kang, N.J.; Lee, K.W.; Lee, H.J. Activation of Phosphatidylinositol 3-Kinase Is Required for Tumor Necrosis Factor-α-Induced Upregulation of Matrix Metalloproteinase-9: Its Direct Inhibition by Quercetin. Int. J. Biochem. Cell Biol. 2009, 41, 1592–1600. [Google Scholar] [CrossRef]
- Cao, H.H.; Cheng, C.Y.; Su, T.; Fu, X.Q.; Guo, H.; Li, T.; Tse, A.K.W.; Kwan, H.Y.; Yu, H.; Yu, Z.L. Quercetin Inhibits HGF/c-Met Signaling and HGFstimulated Melanoma Cell Migration and Invasion. Mol. Cancer 2015, 14, 103. [Google Scholar] [CrossRef]
- Cao, H.H.; Tse, A.K.W.; Kwan, H.Y.; Yu, H.; Cheng, C.Y.; Su, T.; Fong, W.F.; Yu, Z.L. Quercetin Exerts Anti-Melanoma Activities and Inhibits STAT3 Signaling. Biochem. Pharmacol. 2014, 87, 424–434. [Google Scholar] [CrossRef]
- Zhao, J.; Fang, Z.; Zha, Z.; Sun, Q.; Wang, H.; Sun, M.; Qiao, B. Quercetin Inhibits Cell Viability, Migration and Invasion by Regulating MiR-16/HOXA10 Axis in Oral Cancer. Eur. J. Pharmacol. 2019, 847, 11–18. [Google Scholar] [CrossRef]
- Nam, T.W.; Il Yoo, C.; Kim, H.T.; Kwon, C.H.; Park, J.Y.; Kim, Y.K. The Flavonoid Quercetin Induces Apoptosis and Inhibits Migration through a MAPK-Dependent Mechanism in Osteoblasts. J. Bone Miner. Metab. 2008, 26, 551–560. [Google Scholar] [CrossRef]
- Li, S.; Pei, Y.; Wang, W.; Liu, F.; Zheng, K.; Zhang, X. Quercetin Suppresses the Proliferation and Metastasis of Metastatic Osteosarcoma Cells by Inhibiting Parathyroid Hormone Receptor 1. Biomed. Pharmacother. 2019, 114, 108839. [Google Scholar] [CrossRef]
- Lan, H.; Hong, W.; Fan, P.; Qian, D.; Zhu, J.; Bai, B. Quercetin Inhibits Cell Migration and Invasion in Human Osteosarcoma Cells. Cell. Physiol. Biochem. 2017, 43, 553–567. [Google Scholar] [CrossRef]
- Berndt, K.; Campanile, C.; Muff, R.; Strehler, E.; Born, W.; Fuchs, B. Evaluation of Quercetin as a Potential Drug in Osteosarcoma Treatment. Anticancer Res. 2013, 33, 1297–1306. [Google Scholar]
- Huang, Y.T.; Lee, L.T.; Lee, P.P.H.; Lin, Y.S.; Lee, M.T. Targeting of Focal Adhesion Kinase by Flavonoids and Small-Interfering RNAs Reduces Tumor Cell Migration Ability. Anticancer Res. 2005, 25, 2017–2025. [Google Scholar]
- Yu, D.; Ye, T.; Xiang, Y.; Shi, Z.; Zhang, J.; Lou, B.; Zhang, F.; Chen, B.; Zhou, M. Quercetin Inhibits Epithelial–Mesenchymal Transition, Decreases Invasiveness and Metastasis, and Reverses IL-6 Induced Epithelial–Mesenchymal Transition, Expression of MMP by Inhibiting STAT3 Signaling in Pancreatic Cancer Cells. OncoTargets. Ther. 2017, 10, 4719–4729. [Google Scholar] [CrossRef]
- Bhat, F.A.; Sharmila, G.; Balakrishnan, S.; Arunkumar, R.; Elumalai, P.; Suganya, S.; Raja Singh, P.; Srinivasan, N.; Arunakaran, J. Quercetin Reverses EGF-Induced Epithelial to Mesenchymal Transition and Invasiveness in Prostate Cancer (PC-3) Cell Line via EGFR/PI3K/Akt Pathway. J. Nutr. Biochem. 2014, 25, 1132–1139. [Google Scholar] [CrossRef]
- Yang, F.; Jiang, X.; Song, L.; Wang, H.; Mei, Z.; Xu, Z.; Xing, N. Quercetin Inhibits Angiogenesis through Thrombospondin-1 Upregulation to Antagonize Human Prostate Cancer PC-3 Cell Growth in Vitro and in Vivo. Oncol. Rep. 2016, 35, 1602–1610. [Google Scholar] [CrossRef]
- Song, W.; Zhao, X.; Xu, J.; Zhang, H. Quercetin Inhibits Angiogenesis—Mediated Human Retinoblastoma Growth by Targeting Vascular Endothelial Growth Factor Receptor. Oncol. Lett. 2017, 14, 3343–3348. [Google Scholar] [CrossRef]
- Camero, C.M.; Germanò, M.P.; Rapisarda, A.; D’Angelo, V.; Amira, S.; Benchikh, F.; Braca, A.; De Leo, M. Anti-Angiogenic Activity of Iridoids from Galium Tunetanum. Braz. J. Pharmacogn. 2018, 28, 374–377. [Google Scholar] [CrossRef]
- Ben Sghaier, M.; Pagano, A.; Mousslim, M.; Ammari, Y.; Kovacic, H.; Luis, J. Rutin Inhibits Proliferation, Attenuates Superoxide Production and Decreases Adhesion and Migration of Human Cancerous Cells. Biomed. Pharmacother. 2016, 84, 1972–1978. [Google Scholar] [CrossRef]
- Chen, H.; Miao, Q.; Geng, M.; Liu, J.; Hu, Y.; Tian, L.; Pan, J.; Yang, Y. Anti-Tumor Effect of Rutin on Human Neuroblastoma Cell Lines through Inducing G2/M Cell Cycle Arrest and Promoting Apoptosis. Sci. World J. 2013, 2013. [Google Scholar] [CrossRef]
- Lin, C.M.; Chang, H.; Chen, Y.H.; Li, S.Y.; Wu, I.H.; Chiu, J.H. Protective Role of Wogonin against Lipopolysaccharide-Induced Angiogenesis via VEGFR-2, Not VEGFR-1. Int. Immunopharmacol. 2006, 6, 1690–1698. [Google Scholar] [CrossRef]
- Liu, L.Z.; Jing, Y.; Jiang, L.L.; Jiang, X.E.; Jiang, Y.; Rojanasakul, Y.; Jiang, B.H. Acacetin Inhibits VEGF Expression, Tumor Angiogenesis and Growth through AKT/HIF-1α Pathway. Biochem. Biophys. Res. Commun. 2011, 413, 299–305. [Google Scholar] [CrossRef]
- Zhao, K.; Yuan, Y.; Lin, B.; Miao, Z.; Li, Z.; Guo, Q.; Lu, N. LW-215, a Newly Synthesized Flavonoid, Exhibits Potent Anti-Angiogenic Activity in Vitro and in Vivo. Gene 2018, 642, 533–541. [Google Scholar] [CrossRef]
- Viegas, O.; Faria, M.A.; Sousa, J.B.; Vojtek, M.; Gonçalves-Monteiro, S.; Suliburska, J.; Diniz, C.; Ferreira, I.M.P.L.V.O. Delphinidin-3-O-Glucoside Inhibits Angiogenesis via VEGFR2 Downregulation and Migration through Actin Disruption. J. Funct. Foods 2019, 54, 393–402. [Google Scholar] [CrossRef]
- Rajesh, G.; Harshala, S.; Dhananjay, G.; Jadhav, A.; Vikram, G. Effect of Hydroxyl Substitution of Flavone on Angiogenesis and Free Radical Scavenging Activities: A Structure-Activity Relationship Studies Using Computational Tools. Eur. J. Pharm. Sci. 2010, 39, 37–44. [Google Scholar] [CrossRef]
- Gacche, R.N.; Shegokar, H.D.; Gond, D.S.; Yang, Z.; Jadhav, A.D. Evaluation of Selected Flavonoids as Antiangiogenic, Anticancer, and Radical Scavenging Agents: An Experimental and in Silico Analysis. Cell Biochem. Biophys. 2011, 61, 651–663. [Google Scholar] [CrossRef]
- Gacche, R.N.; Meshram, R.J.; Shegokar, H.D.; Gond, D.S.; Kamble, S.S.; Dhabadge, V.N.; Utage, B.G.; Patil, K.K.; More, R.A. Flavonoids as a Scaffold for Development of Novel Anti-Angiogenic Agents: An Experimental and Computational Enquiry. Arch. Biochem. Biophys. 2015, 577–578, 35–48. [Google Scholar] [CrossRef]
- Chen, Y.; Lu, N.; Ling, Y.; Wang, L.; You, Q.; Li, Z.; Guo, Q. LYG-202, a Newly Synthesized Flavonoid, Exhibits Potent Anti-Angiogenic Activity in Vitro and in Vivo. J. Pharmacol. Sci. 2010, 112, 37–45. [Google Scholar] [CrossRef]
- Huang, Y.; Fang, J.; Lu, W.; Wang, Z.; Wang, Q.; Hou, Y.; Jiang, X.; Reizes, O.; Lathia, J.; Nussinov, R.; et al. A Systems Pharmacology Approach Uncovers Wogonoside as an Angiogenesis Inhibitor of Triple-Negative Breast Cancer by Targeting Hedgehog Signaling. Cell Chem. Biol. 2019, 26, 1143–1158.e6. [Google Scholar] [CrossRef]
- Chen, Y.; Lu, N.; Ling, Y.; Gao, Y.; Wang, L.; Sun, Y.; Qi, Q.; Feng, F.; Liu, W.; Liu, W.; et al. Wogonoside Inhibits Lipopolysaccharide-Induced Angiogenesis in Vitro and in Vivo via Toll-like Receptor 4 Signal Transduction. Toxicology 2009, 259, 10–17. [Google Scholar] [CrossRef]
- Li, X.; Chen, M.; Lei, X.; Huang, M.; Ye, W.; Zhang, R.; Zhang, D. Luteolin Inhibits Angiogenesis by Blocking Gas6/Axl Signaling Pathway. Int. J. Oncol. 2017, 51, 677–685. [Google Scholar] [CrossRef]
- Panda, S.P.; Panigrahy, U.P.; Prasanth, D.; Gorla, U.S.; Guntupalli, C.; Panda, D.P.; Jena, B.R. A Trimethoxy Flavonoid Isolated from Stem Extract of Tabebuia chrysantha Suppresses Angiogenesis in Angiosarcoma. J. Pharm. Pharmacol. 2020, jphp.13272. [Google Scholar] [CrossRef]
- Huang, Y.; Zhao, K.; Hu, Y.; Zhou, Y.; Luo, X.; Li, X.; Wei, L.; Li, Z.; You, Q.; Guo, Q.; et al. Wogonoside Inhibits Angiogenesis in Breast Cancer via Suppressing Wnt/β-Catenin Pathway. Mol. Carcinog. 2016, 55, 1598–1612. [Google Scholar] [CrossRef]
- Bhat, T.A.; Nambiar, D.; Tailor, D.; Pal, A.; Agarwal, R.; Singh, R.P. Acacetin Inhibits in Vitro and in Vivo Angiogenesis and Downregulates Stat Signaling and VEGF Expression. Cancer Prev. Res. 2013, 6, 1128–1139. [Google Scholar] [CrossRef]
- Fang, J.; Zhou, Q.; Liu, L.Z.; Xia, C.; Hu, X.; Shi, X.; Jiang, B.H. Apigenin Inhibits Tumor Angiogenesis through Decreasing HIF-1α and VEGF Expression. Carcinogenesis 2007, 28, 858–864. [Google Scholar] [CrossRef]
- Chen, J.; Chen, A.Y.; Huang, H.; Ye, X.; Rollyson, W.D.; Perry, H.E.; Brown, K.C.; Rojanasakul, Y.; Rankin, G.O.; Dasgupta, P.; et al. The Flavonoid Nobiletin Inhibits Tumor Growth and Angiogenesis of Ovarian Cancers via the Akt Pathway. Int. J. Oncol. 2015, 46, 2629–2638. [Google Scholar] [CrossRef]
- Pratheeshkumar, P.; Son, Y.O.; Budhraja, A.; Wang, X.; Ding, S.; Wang, L.; Hitron, A.; Lee, J.C.; Kim, D.; Divya, S.P.; et al. Luteolin Inhibits Human Prostate Tumor Growth by Suppressing Vascular Endothelial Growth Factor Receptor 2-Mediated Angiogenesis. PLoS ONE 2012, 7, e52279. [Google Scholar] [CrossRef]
- Lin, C.M.; Chang, H.; Chen, Y.H.; Wu, I.H.; Chiu, J.H. Wogonin Inhibits IL-6-Induced Angiogenesis via down-Regulation of VEGF and VEGFR-1, Not VEGFR-2. Planta Med. 2006, 72, 1305–1310. [Google Scholar] [CrossRef]
- Zhu, D.; Wang, S.; Lawless, J.; He, J.; Zheng, Z. Dose Dependent Dual Effect of Baicalin and Herb Huang Qin Extract on Angiogenesis. PLoS ONE 2016, 11, e0167125. [Google Scholar] [CrossRef]
- Favot, L.; Martin, S.; Keravis, T.; Andriantsitohaina, R.; Lugnier, C. Involvement of Cyclin-Dependent Pathway in the Inhibitory Effect of Delphinidin on Angiogenesis. Cardiovasc. Res. 2003, 59, 479–487. [Google Scholar] [CrossRef]
- Hui, M.; Cazet, A.; Nair, R.; Watkins, D.N.; O’Toole, S.A.; Swarbrick, A. The Hedgehog Signalling Pathway in Breast Development, Carcinogenesis and Cancer Therapy. Breast Cancer Res. 2013, 15, 1–14. [Google Scholar] [CrossRef]
- Cao, X.; Geradts, J.; Dewhirst, M.W.; Lo, H.W. Upregulation of VEGF-A and CD24 Gene Expression by the TGLI1 Transcription Factor Contributes to the Aggressive Behavior of Breast Cancer Cells. Oncogene 2012, 31, 104–115. [Google Scholar] [CrossRef]
- Zhang, K.; Lu, J.; Mori, T.; Smith-Powell, L.; Synold, T.W.; Chen, S.; Wen, W. Baicalin Increases VEGF Expression and Angiogenesis by Activating the ERR/PGC-1 Pathway. Cardiovasc. Res. 2011, 89, 426–435. [Google Scholar] [CrossRef]
- Liu, J.-J.; Huang, T.-S.; Cheng, W.-F.; Lu, F.-J. Baicalein and Baicalin Are Potent Inhibitors of Angiogenesis: Inhibition of Endothelial Cell Proliferation, Migration and Differentiation. Int. J. Cancer 2003, 106, 559–565. [Google Scholar] [CrossRef]
- Morbidelli, L. Polyphenol-Based Nutraceuticals for the Control of Angiogenesis: Analysis of the Critical Issues for Human Use. Pharmacol. Res. 2016, 111, 384–393. [Google Scholar] [CrossRef]
- Cockerill, G.W.; Gamble, J.R.; Vadas, M.A. Angiogenesis: Models and Modulators. Int. Rev. Cytol. 1995, 159, 113–160. [Google Scholar] [CrossRef]
- Ribatti, D.; Vacca, A. Models for Studying Angiogenesis in Vivo. Int. J. Biol. Markers 1999, 14, 207–213. [Google Scholar] [CrossRef]
- Staton, C.A.; Stribbling, S.M.; Tazzyman, S.; Hughes, R.; Brown, N.J.; Lewis, C.E. Current Methods for Assaying Angiogenesis in Vitro and in Vivo. Int. J. Experimantal Biol. 2004, 85, 233–248. [Google Scholar] [CrossRef]
- Nguyen, M.; Shing, Y.; Folkman, J. Quantitation of Angiogenesis and Antiangiogenesis in the Chick Embryo Chorioallantoic Membrane. Microvasc. Res. 1994, 47, 31–40. [Google Scholar] [CrossRef]
- Ribatti, D.; Vacca, A.; Roncali, L.; Dammacco, F. The Chick Embryo Chorioallantoic Membrane as a Model for in Vivo Research on Angiogenesis. Int. J. Dev. Biol. 1996, 40, 1189–1197. [Google Scholar] [CrossRef]
- Wang, T.; Li, Q.; Bi, K. Bioactive Flavonoids in Medicinal Plants: Structure, Activity and Biological Fate. Asian J. Pharm. Sci. 2018, 13, 12–23. [Google Scholar] [CrossRef]
- Nguyen, T.T.H.; Moon, Y.H.; Ryu, Y.B.; Kim, Y.M.; Nam, S.H.; Kim, M.S.; Kimura, A.; Kim, D. The Influence of Flavonoid Compounds on the in Vitro Inhibition Study of a Human Fibroblast Collagenase Catalytic Domain Expressed in E. coli. Enzyme Microb. Technol. 2013, 52, 26–31. [Google Scholar] [CrossRef]
- Krych, J.; Gebicka, L. Catalase Is Inhibited by Flavonoids. Int. J. Biol. Macromol. 2013, 58, 148–153. [Google Scholar] [CrossRef]
- Huang, Z.; Fang, F.; Wang, J.; Wong, C.W. Structural Activity Relationship of Flavonoids with Estrogen-Related Receptor Gamma. FEBS Lett. 2010, 584, 22–26. [Google Scholar] [CrossRef]
- Atrahimovich, D.; Vaya, J.; Khatib, S. The Effects and Mechanism of Flavonoid-RePON1 Interactions. Structure-Activity Relationship Study. Bioorganic Med. Chem. 2013, 21, 3348–3355. [Google Scholar] [CrossRef]
- Çelik, H.; Koşar, M. Inhibitory Effects of Dietary Flavonoids on Purified Hepatic NADH-Cytochrome B5 Reductase: Structure-Activity Relationships. Chem. Biol. Interact. 2012, 197, 103–109. [Google Scholar] [CrossRef]
- Sithisarn, P.; Michaelis, M.; Schubert-Zsilavecz, M.; Cinatl, J. Differential Antiviral and Anti-Inflammatory Mechanisms of the Flavonoids Biochanin A and Baicalein in H5N1 Influenza A Virus-Infected Cells. Antiviral Res. 2013, 97, 41–48. [Google Scholar] [CrossRef]
- Kothandan, G.; Gadhe, C.G.; Madhavan, T.; Choi, C.H.; Cho, S.J. Docking and 3D-QSAR (Quantitative Structure Activity Relationship) Studies of Flavones, the Potent Inhibitors of p-Glycoprotein Targeting the Nucleotide Binding Domain. Eur. J. Med. Chem. 2011, 46, 4078–4088. [Google Scholar] [CrossRef]
- Singh, M.; Kaur, M.; Silakari, O. Flavones: An Important Scaffold for Medicinal Chemistry. Eur. J. Med. Chem. 2014, 84, 206–239. [Google Scholar] [CrossRef]
- Haupt, V.J.; Daminelli, S.; Schroeder, M. Drug Promiscuity in PDB: Protein Binding Site Similarity Is Key. PLoS ONE 2013, 8, e65894. [Google Scholar] [CrossRef]
- Lam, I.K.; Alex, D.; Wang, Y.H.; Liu, P.; Liu, A.L.; Du, G.H.; Yuen Lee, S.M. In Vitro and in Vivo Structure and Activity Relationship Analysis of Polymethoxylated Flavonoids: Identifying Sinensetin as a Novel Antiangiogenesis Agent. Mol. Nutr. Food Res. 2012, 56, 945–956. [Google Scholar] [CrossRef]
- Lingen, M.W. Historical Perspective Role of Leukocytes and Endothelial Cells in the Development of Angiogenesis in Inflammation and Wound Healing. Arch. Pathol. Lab. Med. 2001, 125, 67–71. [Google Scholar] [CrossRef]
- Naldini, A.; Carraro, F. Role of Inflammatory Mediators in Angiogenesis. Curr. Drug Targets Inflamm. Allergy 2005, 4, 3–8. [Google Scholar] [CrossRef]
- Kofler, S.; Nickel, T.; Weis, M. Role of Cytokines in Cardiovascular Diseases: A Focus on Endothelial Responses to Inflammation. Clin. Sci. 2005, 108, 205–213. [Google Scholar] [CrossRef]
- Gong, G.; Wang, H.; Kong, X.; Duan, R.; Dong, T.T.X.; Tsim, K.W.K. Flavonoids Are Identified from the Extract of Scutellariae Radix to Suppress Inflammatory-Induced Angiogenic Responses in Cultured RAW 264.7 Macrophages. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef]
- Wu, W.-B.; Hung, D.K.; Chang, F.W.; Ong, E.T.; Chen, B.H. Anti-Inflammatory and Anti-Angiogenic Effects of Flavonoids Isolated from Lycium Barbarum Linnaeus on Human Umbilical Vein Endothelial Cells. Food Funct. 2012, 3, 1068–1081. [Google Scholar] [CrossRef]
- Zhang, F.X.; Kirschning, C.J.; Mancinelli, R.; Xu, X.P.; Jin, Y.; Faure, E.; Mantovani, A.; Rothe, M.; Muzio, M.; Arditi, M. Bacterial Lipopolysaccharide Activates Nuclear Factor-ΚB through Interleukin-1 Signaling Mediators in Cultured Human Dermal Endothelial Cells and Mononuclear Phagocytes. J. Biol. Chem. 1999, 274, 7611–7614. [Google Scholar] [CrossRef]
- Wu, S.H.; Liao, P.Y.; Dong, L.; Chen, Z.Q. Signal Pathway Involved in Inhibition by Lipoxin A4 of Production of Interleukins Induced in Endothelial Cells by Lipopolysaccharide. Inflamm. Res. 2008, 57, 430–437. [Google Scholar] [CrossRef]
- Bachelot, T.; Ray-Coquard, I.; Menetrier-Caux, C.; Rastkha, M.; Duc, A.; Blay, J.Y. Prognostic Value of Serum Levels of Interleukin 6 and of Serum and Plasma Levels of Vascular Endothelial Growth Factor in Hormone-Refractory Metastatic Breast Cancer Patients. Br. J. Cancer 2003, 88, 1721–1726. [Google Scholar] [CrossRef]
- Molostvov, G.; Morris, A.; Rose, P.; Basu, S.; Muller, G. The Effects of Selective Cytokine Inhibitory Drugs (CC-10004 and CC-1088) on VEGF and IL-6 Expression and Apoptosis in Myeloma and Endothelial Cell Co-Cultures. Br. J. Haematol. 2004, 124, 366–375. [Google Scholar] [CrossRef]
- Fang, J.; Xia, C.; Cao, Z.; Zheng, J.Z.; Reed, E.; Jiang, B.-H. Apigenin Inhibits VEGF and HIF-1 Expression via PI3K/AKT/P70S6K1 and HDM2/P53 Pathways. FASEB J. 2005, 19, 342–353. [Google Scholar] [CrossRef]
- Carmeliet, P.; Jain, R.K. Angiogenesis in Cancer and Other Diseases. Nature 2000, 407, 249–257. [Google Scholar] [CrossRef]
- Zimna, A.; Kurpisz, M. Hypoxia-Inducible Factor-1 in Physiological and Pathophysiological Angiogenesis: Applications and Therapies. Biomed. Res. Int. 2015, 2015. [Google Scholar] [CrossRef]
- Lu, K.H.; Chen, P.N.; Hsieh, Y.H.; Lin, C.Y.; Cheng, F.Y.; Chiu, P.C.; Chu, S.C.; Hsieh, Y.S. 3-Hydroxyflavone Inhibits Human Osteosarcoma U2OS and 143B Cells Metastasis by Affecting EMT and Repressing u-PA/MMP-2 via FAK-Src to MEK/ERK and RhoA/MLC2 Pathways and Reduces 143B Tumor Growth in Vivo. Food Chem. Toxicol. 2016, 97, 177–186. [Google Scholar] [CrossRef]
- Liu, L.Z.; Fang, J.; Zhou, Q.; Hu, X.; Shi, X.; Jiang, B.H. Apigenin Inhibits Expression of Vascular Endothelial Growth Factor and Angiogenesis in Human Lung Cancer Cells: Implication of Chemoprevention of Lung Cancer. Mol. Pharmacol. 2005, 68, 635–643. [Google Scholar] [CrossRef]
- Byun, E.B.; Kim, H.M.; Song, H.Y.; Kim, W.S. Hesperidin Structurally Modified by Gamma Irradiation Induces Apoptosis in Murine Melanoma B16BL6 Cells and Inhibits Both Subcutaneous Tumor Growth and Metastasis in C57BL/6 Mice. Food Chem. Toxicol. 2019, 127, 19–30. [Google Scholar] [CrossRef]
- Deep, G.; Kumar, R.; Jain, A.K.; Agarwal, C.; Agarwal, R. Silibinin Inhibits Fibronectin Induced Motility, Invasiveness and Survival in Human Prostate Carcinoma PC3 Cells via Targeting Integrin Signaling. Mutat. Res. Fundam. Mol. Mech. Mutagen. 2014, 768, 35–46. [Google Scholar] [CrossRef]
- Zhao, J.; Xu, J.; Lv, J. Identification of Profilin 1 as the Primary Target for the Anti-Cancer Activities of Furowanin A in Colorectal Cancer. Pharmacol. Rep. 2019, 71, 940–949. [Google Scholar] [CrossRef]
- Deeks, J.J.; Higgins, J.P.; Altman, D.G. Analysing Data and Undertaking Meta-analyses. In Cochrane Handbook for Systematic Reviews of Interventions; John and Wiley and Sons: Hoboken, NJ, USA, 2019; pp. 241–284. [Google Scholar] [CrossRef]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; Estarli, M.; Barrera, E.S.A.; et al. Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) 2015 Statement. Rev. Esp. Nutr. Hum. Diet. 2016, 20, 148–160. [Google Scholar] [CrossRef]
- Huedo-Medina, T.B.; Sánchez-Meca, J.; Marín-Martínez, F.; Botella, J. Assessing Heterogeneity in Meta-Analysis: Q Statistic or I 2 Index? Psychol. Methods 2006, 11, 193–206. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available. |



| Flavonol | Disease | In Vitro Tests | In/Ex Vivo Tests | Author, Year |
|---|---|---|---|---|
| Beturetol | Angiogenesis | CAM | Hisanori Hattori, 2011 [18] | |
| Casticin | Cancer * | Shanaya Ramchandani, 2020 [19] | ||
| Denticulatain | Lung Cancer | ZFM | Da Song Yang, 2015 [20] | |
| Dihydrokaempferide | Angiogenesis | CAM | Hisanori Hattori, 2011 [18] | |
| Fisetin | Cancer * | Dharambir Kashyap, 2018 [21] | ||
| Cancer * | Thamaraiselvan Rengarajan, 2016 [22] | |||
| Cancer * | Deeba N.Syed, 2016 [23] | |||
| Cancer * | Lall K. Rahul, 2016 [24] | |||
| Breast Cancer | In | Cheng Fang Tsai, 2018 [25] | ||
| Breast Cancer | WH, In | Xu Sun, 2018 [26] | ||
| Breast Cancer | WH, In | Mets in mice | Jie Li, 2018 [27] | |
| Cervical Cancer | In | Ruey Hwang Chou, 2013 [28] | ||
| Glioma | In | Chien Min Chen, 2015 [29] | ||
| Hepatic Cancer | In | Xiang Feng Liu, 2017 [30] | ||
| Leukemia | In | Anna Klimaszewska-Wiśniewska, 2019 [31] | ||
| Lung Cancer | WH, In | Saba Tabasum, 2019 [32] | ||
| Lung Cancer | WH, In, Ad | Junjian Wang, 2018 [33] | ||
| Prostate Cancer | WH, In, Ad | Chi Sheng Chien, 2010 [34] | ||
| Renal Cancer | In | Yih Shou Hsieh, 2019 [35] | ||
| Retinopathy | RbCN | A M Joussen, 2000 [36] | ||
| Galangin | Hepatic Cancer * | Dengyang Fang, 2019 [37] | ||
| Angiogenesis | TF, Ad | Jong Deog Kim, 2006 [38] | ||
| Glioma | TF, In | CAM, MD in mice | Daliang Chen, 2019 [39] | |
| Glioma | In | Deqiang Lei, 2018 [40] | ||
| Hepatic Cancer | WH, In, Ad | Shang Tao Chien, 2015 [41] | ||
| Ovarian Cancer | TF | CAM | Haizhi Huang, 2015 [42] | |
| Renal Cancer | WH, In | Jingyi Cao, 2016 [43] | ||
| Renal Cancer | In | Yun Zhu, 2018 [44] | ||
| Gossypin | Gastric Cancer | In | Wang Li, 2019 [45] | |
| Herbacetin | Melanoma | In | Lei Li, 2019 [46] | |
| Hyperoside | Arthritis | WH, In | CIAM in mice | Xiang Nan Jin, 2016 [47] |
| Icariin | Bone disease * | Xin Zhang, 2014 [48] | ||
| Cancer * | Meixia Chen, 2016 [49] | |||
| Angiogenesis | TF, In | RAR | Byung Hee Chung, 2008 [50] | |
| Esophageal Cancer | In | Zhen Fang Gu, 2017 [51] | ||
| Ovarian Cancer | WH | Pengzhen Wang, 2019 [52] | ||
| Wound healing | EWM in rats | Wangkheirakpam Ramdas Singh, 2019 [53] | ||
| Icariside | Cancer * | Meixia Chen, 2016 [49] | ||
| Glioma | WH, In | Kai Quan, 2017 [54] | ||
| Isoviolanthin | Hepatic Cancer | WH, In | Shangping Xing, 2018 [55] | |
| Isosakuranetin | Angiogenesis | CAM | Hisanori Hattori, 2011 [18] | |
| Kaempferol | Cancer * | Allen Y. Chen, 2013 [56] | ||
| Cancer * | Dharambir Kashyap, 2017 [57] | |||
| Angiogenesis | WH, TB, In | Hsien Kuo Chin, 2018 [58] | ||
| Angiogenesis | WH, TB | ZFM | Fang Liang, 2015 [59] | |
| Angiogenesis | CAM | Shigenori Kumazawa, 2013 [60] | ||
| Angiogenesis | TF, Ad | Jong Deog Kim, 2006 [38] | ||
| Diabetes | EWM in rats | Yusuf Özay, 2019 [61] | ||
| Glioma | WH | Vivek Sharma, 2007 [62] | ||
| Glioma | In | Mets in mice | S.C. Shen, 2006 [63] | |
| Hepatic Cancer | WH, In | Mets in mice | Youyou Qin, 2015 [64] | |
| Hepatic Cancer | In | Genglong Zhu, 2018 [65] | ||
| Lung Cancer | WH, In | Eunji Jo, 2015 [66] | ||
| Medulloblastoma | Ad | David Labbé, 2009 [67] | ||
| Oral Cancer | In | Chiao Wen Lin, 2013 [68] | ||
| Osteosarcoma | WH, In, Ad | Hui Jye Chen, 2013 [69] | ||
| Ovarian Cancer | CAM | Haitao Luo, 2009 [70] | ||
| Pancreatic Cancer | In | Jungwhoi Lee, 2016 [71] | ||
| Renal Cancer | WH, In | Mets in mice | Tung Wei Hung, 2017 [72] | |
| Retinal Vascularization | WH, In | Hsiang Wen Chien, 2019 [73] | ||
| Kaempferol-3-O-[(6-caffeoyl)-β- glucopyranosyl (1→3) α-rhamnopyranoside]-7-O-α-rhamnopyranoside | Angiogenesis | WH | Marco Clericuzio, 2012 [74] | |
| Kaempferide | Angiogenesis | CAM | Hisanori Hattori, 2011 [18] | |
| Morin | Arthritis | WH, TB | CIAM in rats | Ni Zeng, 2015 [75] |
| Arthritis | WH, TB | CIAM in rats | Mengfan Yue, 2018 [76] | |
| Leukemia | Ad | Nagaja Capitani, 2019 [77] | ||
| Melanoma | WH | Hua Wen Li, 2016 [78] | ||
| Myricetin | Melanoma * | Nam Joo Kang, 2011 [79] | ||
| Angiogenesis | TF, Ad | Jong Deog Kim, 2006 [38] | ||
| Breast Cancer | In | CAM, MD in mice, RAR | Zhiqing Zhou, 2019 [80] | |
| Breast Cancer | WH, In, Ad | Mets in mice | Yingqian Ci, 2018 [81] | |
| Glioma | WH, In | Wen Ta Chiu, 2010 [82] | ||
| Hepatic Cancer | In | Noriko Yamada, 2020 [83] | ||
| Hepatic Cancer | WH, In | Hongxin Ma, 2019 [84] | ||
| Lung Cancer | WH, In, Ad | Yuan Wei Shih, 2009 [85] | ||
| Medullobalstoma | In, Ad | David Labbé, 2009 [67] | ||
| Ovarian Cancer | TF | CAM | Haizhi Huang, 2015 [42] | |
| Quercetin | Breast Cancer * | Maryam Ezzati, 2020 [86] | ||
| Cancer * | Si-min Tang, 2020 [87] | |||
| Cancer * | Dharambir Kashyap, 2016 [88] | |||
| Colorectal Cancer * | Saber G. Darband, 2018 [89] | |||
| Angiogenesis | WH, In | Nu Ry Song, 2014 [90] | ||
| Angiogenesis | WH, TB | ZFM | Chen Lin, 2012 [91] | |
| Angiogenesis | TF, Ad | Jong Deog Kim, 2006 [38] | ||
| Bladder Cancer | WH, In | Yu Hsiang Lee, 2019 [92] | ||
| Breast Cancer | WH | Divyashree Ravishankar, 2015 [93] | ||
| Breast Cancer | MD in mice | Xin Zhao, 2016 [94] | ||
| Breast Cancer | CAM | Soo Jin Oh, 2010 [95] | ||
| Breast Cancer | WH, In | Asha Srinivasan, 2016 [96] | ||
| Breast Cancer | WH, In | Cheng Wei Lin, 2008 [97] | ||
| Breast Cancer | In | Amilcar Rivera Rivera, 2016 [98] | ||
| Cancer | TF | ZFM | Daxian Zhao, 2014 [99] | |
| Cancer | TF, In | CAM | Wen Fu Tan, 2003 [100] | |
| Cancer | MD in mice | Xiangpei Zhao, 2012 [101] | ||
| Cancer | WH, In | Lung Ta Lee, 2004 [102] | ||
| Cancer | WH | Dong Eun Lee, 2013 [103] | ||
| Colorectal Cancer | WH, In | Mets in mice | Ji Ye Kee, 2016 [104] | |
| Glioma | WH | Hong Chao Pan, 2015 [105] | ||
| Glioma | WH, In | Wen Ta Chiu, 2010 [82] | ||
| Glioma | WH, In | Yue Liu, 2017 [106] | ||
| Glioma | In | Jonathan Michaud-Levesque, 2012 [107] | ||
| Glioma | WH | Alessandra Bispo da Silva, 2020 [108] | ||
| Glioma | WH, TB, In | Yue Liu, 2017 [109] | ||
| Hepatic Cancer | In | Noriko Yamada, 2020 [83] | ||
| Hepatic Cancer | WH, In | Jun Lu, 2018 [110] | ||
| Lung Cancer | WH | Anna Klimaszewska-Wiśniewska, 2017 [111] | ||
| Lung Cancer | In | Tzu Chin Wu, 2018 [112] | ||
| Lung Cancer | In | Yo Chuen Lin, 2013 [113] | ||
| Medulloblastoma | In, Ad | David Labbé, 2009 [67] | ||
| Melanoma | In | Mun Kyung Hwang, 2009 [114] | ||
| Melanoma | In | Hui Hui Cao, 2015 [115] | ||
| Melanoma | WH, In | Mets in mice | Hui Hui Cao, 2014 [116] | |
| Oral Cancer | In | Junfang Zhao, 2019 [117] | ||
| Osteoblasts | In | Tae Wook Nam, 2008 [118] | ||
| Osteosarcoma | WH, In, Ad | Shenglong Li, 2019 [119] | ||
| Osteosarcoma | WH, In | Mets in mice | Haifeng Lan, 2017 [120] | |
| Osteosarcoma | WH, Ad | Kersten Berndt, 2013 [121] | ||
| Pancreatic Cancer | WH, In | Ying Tang Huang, 2005 [122] | ||
| Pancreatic Cancer | WH, In | Yu Dinglai 2017 [123] | ||
| Prostate Cancer | WH, In | Firdous Ahmad Bhat, 2014 [124] | ||
| Prostate Cancer | TF, In | MD in mice | Feiya Yang, 2016 [125] | |
| Retinoblastoma | In | Wei Song, 2017 [126] | ||
| Quercetin-3-O-[(6-caffeoyl)-β-glucopyranosyl(1→3) α-rhamnopyranoside]-7-O-α-rhamnopyranoside | Angiogenesis | WH | Marco Clericuzio, 2012 [74] | |
| Rutin | Angiogenesis | CAM | César Muñoz Camero, 2018 [127] | |
| Angiogenesis | CAM | Shigenori Kumazawa, 2013 [60] | ||
| Cancer | WH, In, Ad | Mohamed ben Sghaier, 2016 [128] | ||
| Glioma | WH | Alessandra Bispo da Silva, 2020 [108] | ||
| Neuroblastoma | WH, In | Hongyan Chen, 2013 [129] | ||
| Author, Year | Flavonoid | Angiogenesis Promoter | Cell Line | Concentration | Time, Duration of Treatment | Results Representation | n |
|---|---|---|---|---|---|---|---|
| Soo Jin Oh, 2010 [95] | Quercetin | NA | TAMR-MCF-7 | 3, 10, 30 µM | NA | Number of branches | 5 to 7 |
| Chiu-Mei Lin, 2006 [130] | Wogonin | LPS (1µg/mL) | NA | 10−5, 10−6, 10−7, 10−8 M | 10th day, 48 h | Percentage of vascular counts (%) | 3 |
| Ling-Zhi Liu, 2011 [131] | Acacetin | NA | OVCAR-3 | 10 µM | 9th day, 4 days | Relative angiogenesis | 5 |
| Kai Zhao, 2018 [132] | Wogonin, LW-215 | NA | NA | Wogonin: 80 ng/CAM, LW-215: 2, 4, 8 ng/CAM | 10th day, 48 h | The number of new vessels (% of control) | 3 |
| Haizhi Huang, 2015 [42] | Galangin, myricetin | NA | OVCAR-3 | G: 40 µM, M: 20 µM | 9th day, 5 days | Blood vessels (%) | 6 |
| Olga Viegas, 2019 [133] | Cyanidin, C-3-O-glucoside, delphinidin, D-3-O-glucoside | NA | NA | 20, 40, 80, 100, 200 µM | 11th day, 48 h | % of control | 5 |
| Wen-fu Tan, 2003 [100] | Quercetin | NA | NA | 25, 50, 100 nmol/10 µL/CAM | 10th day, 48 h | Microscopic pictures | 10 |
| Rajesh Gacche, 2010 [134] | Flavone, 3/5/6/7/-Hydroxy flavone | NA | NA | 10, 50, 100 µM | 10th day, 48 h | Antiangiogenic activity (%) of selected flavonoids | 8 |
| R.N. Gacche, 2011 [135] | 3, 6-DHF, 3, 7-DHF, 5, 7-DHF, apigenin, genistein, kaempferol, luteolin, fisetin, rutin, quercetin | NA | NA | 10, 50, 100 µM in 0.05% DMSO/20 µL/CAM | 10th day, 48 h | Antiangiogenic activity (%) of selected flavonoids | 8 |
| R.N. Gacche, 2015 [136] | 4′-Methoxy flavone, 3-Hydroxy-7-methoxy flavone, Formononetin, Biochanin-A, Diosmin, Hesperitin, Hesperidin, 2′-Hydroxy flavanone, 4′-Hydroxy flavanone, 7-Hydroxy flavanone, Myricetin, Taxifolin, Silibinin, Silymarin, Naringenin, Naringin, Catechin | NA | NA | 10, 50, 100 µM in 0.05% DMSO/20 µL/CAM | 10th day, 48 h | Antiangiogenic activity (%) of selected flavonoids | 8 |
| Yan Chen, 2010 [137] | LYG-202 | NA | NA | 2.4, 12, 60 ng/CAM | 10th day, 48 h | Percentage of vascular counts (% of control) | 10 |
| Hisanori Hattori, 2011 [18] | Beturetol, isosakuranetin | NA | NA | 300 ng/CAM | 5th day, 7 days | Inhibition % of angiogenesis at 300 ng/CAM. | 10 |
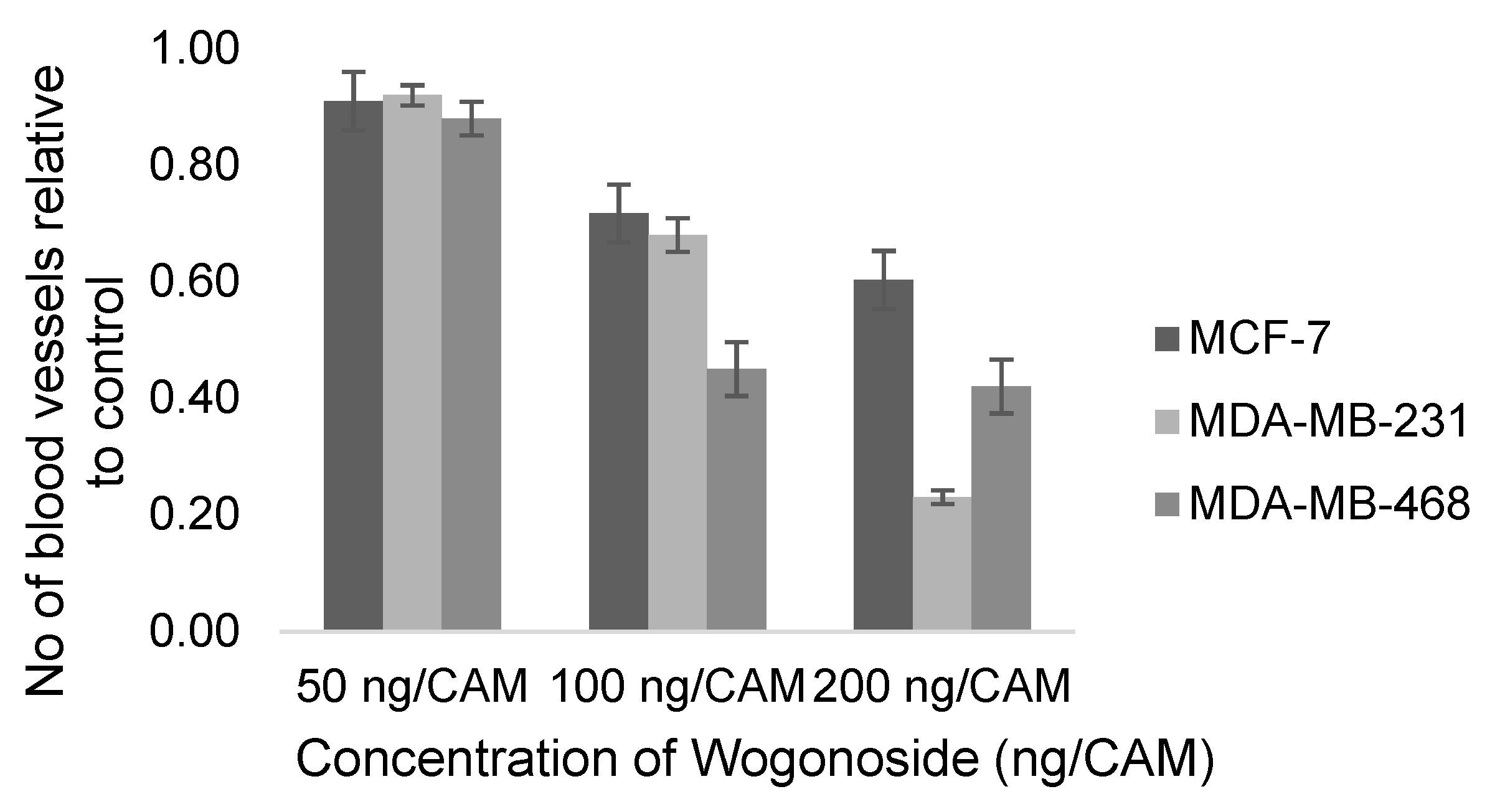
| Yujie Huang, 2019 [138] | Wogonoside | NA | MDA-MB-231, MDA-MB-468 | 50, 100, 200 ng/CAM | 10th day, 48 h | Number of new vessels (% cells) | 3 |
| Yan Chen, 2009 [139] | Wogonoside | LPS (1µg/mL) | NA | 1.5, 15, 150 ng/CAM | 10th day, 48 h | Number of vessels (% of LPS) | 10 |
| Xiaobo Li, 2017 [140] | Luteolin | Gas6 (300 ng/mL) | NA | 10, 20 µM | 6th day, 48 h | Relative vascular density (% of control) | 3 |
| Siva Prasad Panda, 2019 [141] | TMF | NA | EAT | 10, 17, 25 µg/mL | 5th day, 11 days | Microscopic pictures | 5 |
| Yujie Huang, 2016 [142] | Wogonoside | NA | MCF-7 | 50, 100, 200 ng/CAM | 10th day, 48 h | Number of new vessels (% MCF-7) | 3 |
| Tariq A. Bhat, 2013 [143] | Acacetin | NA | NA | 50 µM | 6th day, (every 48 h for 8 days) | % capillary formation | 5 independent areas on CAMs for each treatment |
| Jing Fang, 2007 [144] | Apigenin | NA | OVCAR-3, PC-3 | OVCAR-3: 7.5, 15 µM, PC-3: 10, 20 µM | 9th day, 4 days | Quantification of blood vessels on the CAM | 8 |
| Jianchu Chen, 2015 [145] | Nobiletin | NA | A2780 | 20 µM | 9th day, 5 days | Blood vessel count | 10 |
| Poyil Pratheeshkumar, 2012 [146] | Luteolin | NA | NA | 20, 40 µM | 8th day, 48 h | Relative vascular density | 3 |
| Chiu-Mei Lin, 2006 [147] | Wogonin | IL-6 (10 ng/mL) | NA | 10−5, 10−6, 10−7, 10−8 M | 10th day, 48 h | Percentage of vascular count (%) | 3 |
| Dongqing Zhu, 2016 [148] | Baicalin, baicalein | NA | NA | 0.5, 2, 10, 50 µg/mL and 0.2, 1, 5 mg/mL | 7.5th day, 48 h | Number of new blood vessels | 30 |
| Haitao Luo, 2009 [70] | Kaempferol | NA | OVCAR-3 | 20 µM | 9th day, 5 days | Blood vessel count | 5 |
| Laure Favot, 2003 [149] | Delphinidin | NA | NA | 2, 10, 25, 50 µg | 8th day, 48 h | Microscopic pictures | 5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khater, M.; Greco, F.; Osborn, H.M.I. Antiangiogenic Activity of Flavonoids: A Systematic Review and Meta-Analysis. Molecules 2020, 25, 4712. https://doi.org/10.3390/molecules25204712
Khater M, Greco F, Osborn HMI. Antiangiogenic Activity of Flavonoids: A Systematic Review and Meta-Analysis. Molecules. 2020; 25(20):4712. https://doi.org/10.3390/molecules25204712
Chicago/Turabian StyleKhater, Mai, Francesca Greco, and Helen M. I. Osborn. 2020. "Antiangiogenic Activity of Flavonoids: A Systematic Review and Meta-Analysis" Molecules 25, no. 20: 4712. https://doi.org/10.3390/molecules25204712
APA StyleKhater, M., Greco, F., & Osborn, H. M. I. (2020). Antiangiogenic Activity of Flavonoids: A Systematic Review and Meta-Analysis. Molecules, 25(20), 4712. https://doi.org/10.3390/molecules25204712








