Kaempferol Facilitated Extinction Learning in Contextual Fear Conditioned Rats via Inhibition of Fatty-Acid Amide Hydrolase
Abstract
1. Introduction
2. Results
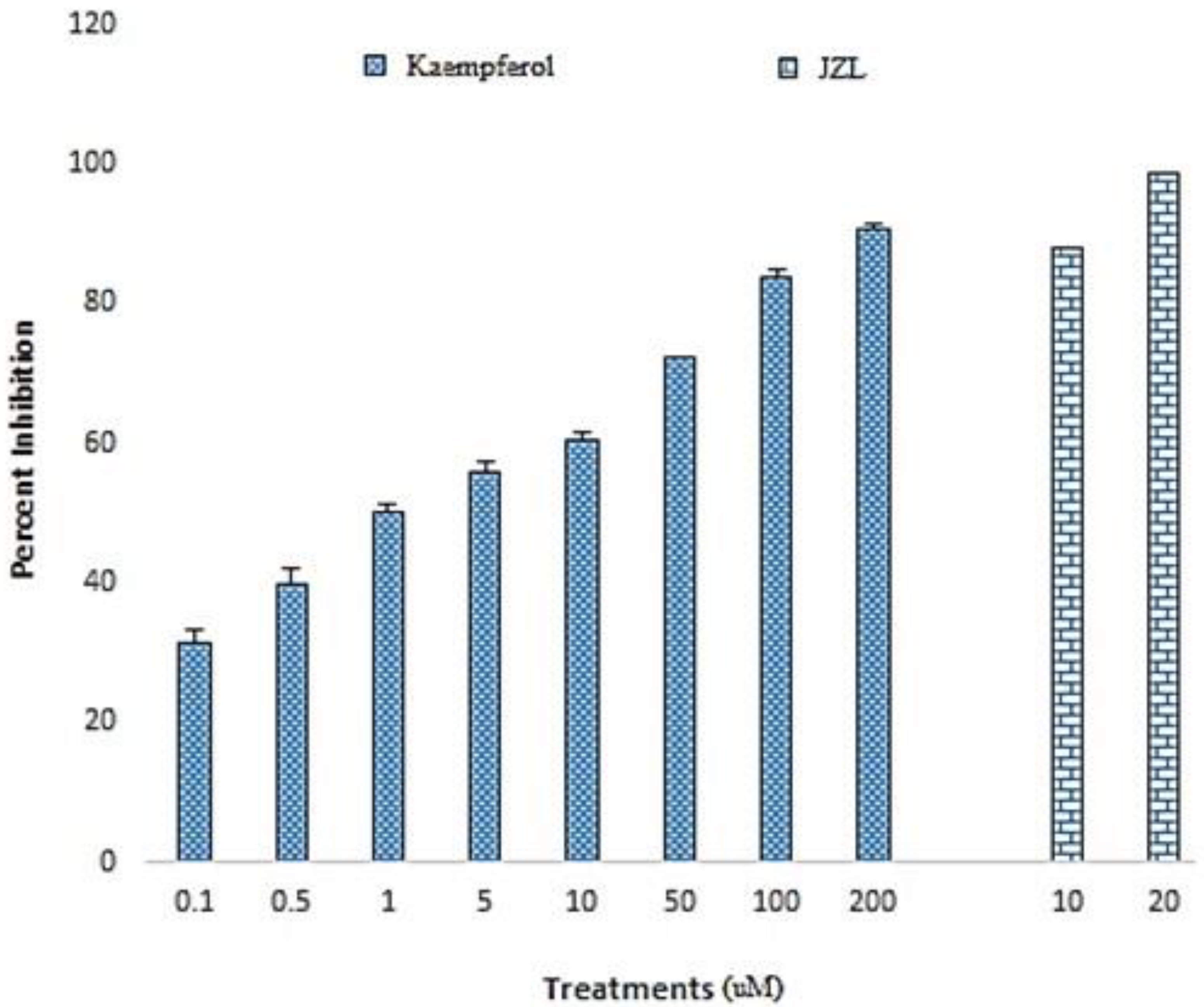
2.1. FAAH Inhibitory Activity
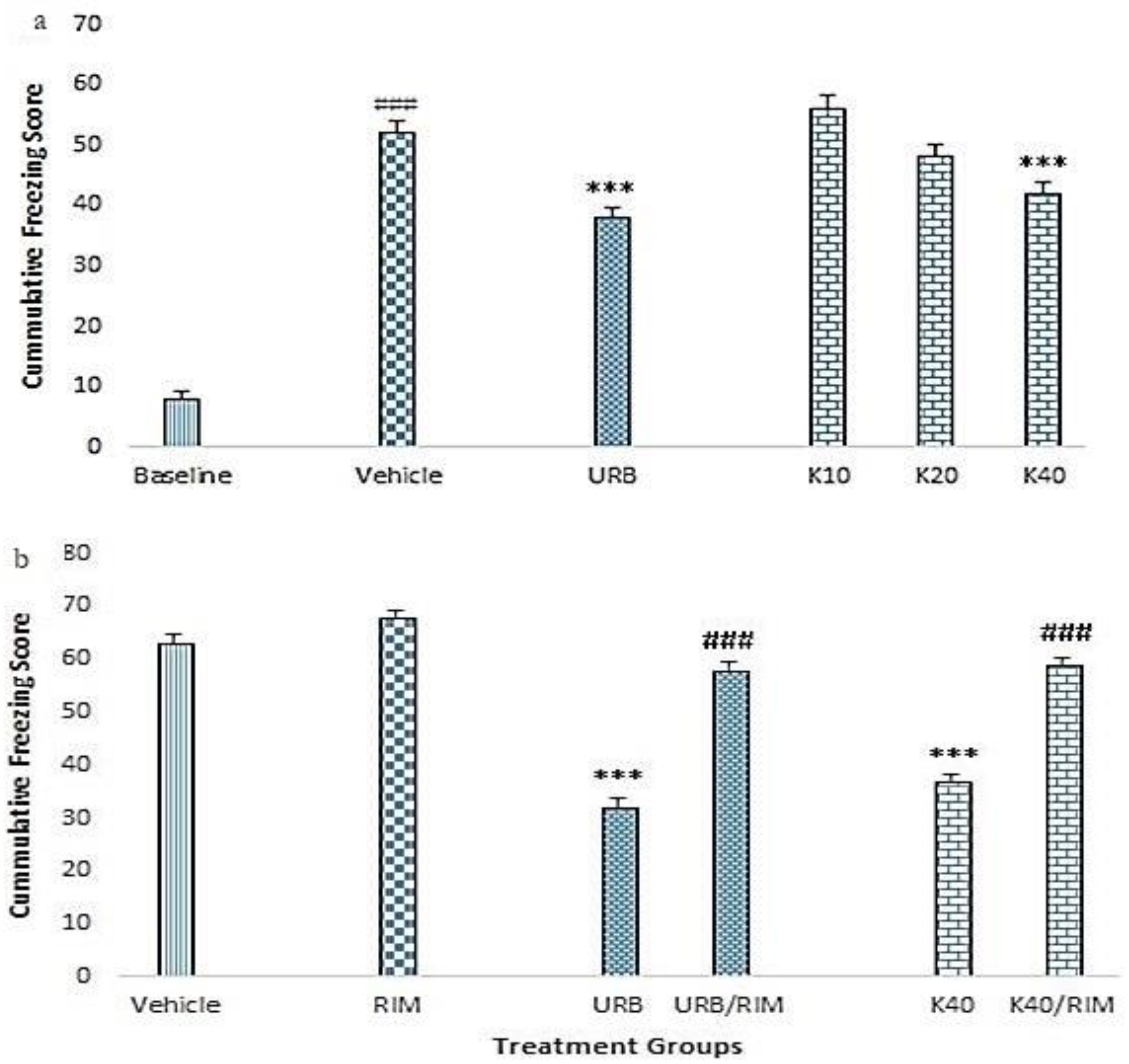
2.2. Contextual Fear Conditioning (CFC)
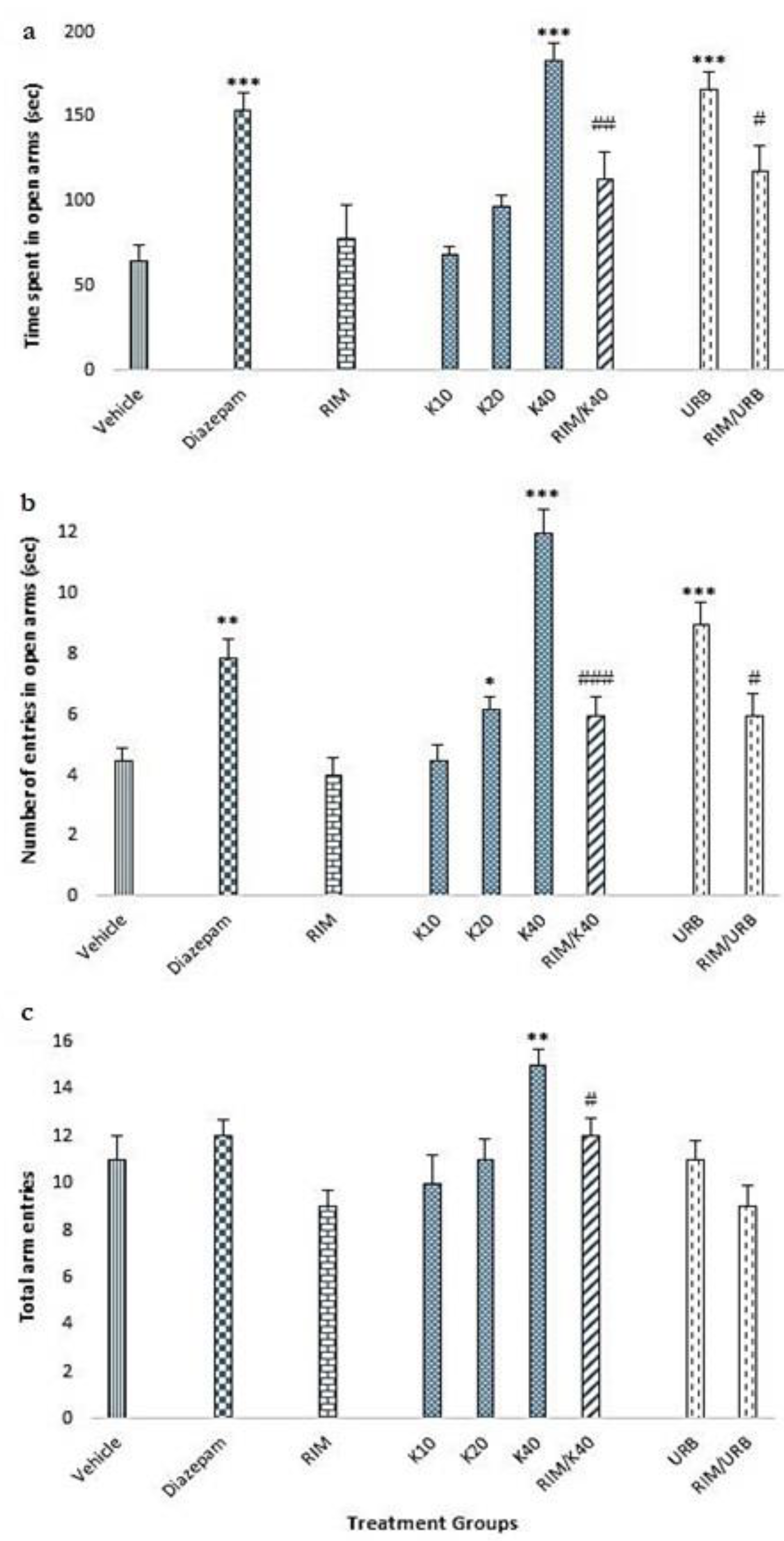
2.3. Elevated Plus Maze Test
2.4. Corticosterone Levels
2.5. In Silico Study
3. Discussion
4. Methodology
4.1. Drugs and Chemicals
4.2. Animals
4.3. FAAH Enzyme Inhibition Assay
4.4. Behavioral Studies
4.4.1. Contextual Fear Conditioning
Habituation
Acquisition
Extinction
4.4.2. Elevated Plus-Maze (EPM)
4.5. Serum Corticosterone Levels
4.6. In Silico Analysis
4.6.1. Protein Preparation
4.6.2. Ligand Preparation
4.6.3. Molecular Docking
4.6.4. Prime/MM-GBSA Simulation
4.7. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Joels, M.; Baram, T.Z. The neuro-symphony of stress. Nat. Rev. Neurosci. 2009, 10, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Taber, K.H.; Hurley, R.A. Endocannabinoids: Stress, anxiety, and fear. J. Neuropsychiatry Clin. Neurosci. 2009, 21. [Google Scholar] [CrossRef] [PubMed]
- Shin, L.M.; Liberzon, I. The neurocircuitry of fear, stress, and anxiety disorders. Neuropsychopharmacology 2010, 35, 169. [Google Scholar] [CrossRef] [PubMed]
- Bisson, J.I.; Cosgrove, S.; Lewis, C.; Roberts, N.P. Post-traumatic stress disorder. BMJ 2015, 351. [Google Scholar] [CrossRef]
- Singewald, N.; Schmuckermair, C.; Whittle, N.; Holmes, A.; Ressler, K. Pharmacology of cognitive enhancers for exposure-based therapy of fear, anxiety and trauma-related disorders. Pharmacol. Ther. 2015, 149, 150–190. [Google Scholar] [CrossRef]
- Davidson, R.J. Anxiety and affective style: Role of prefrontal cortex and amygdala. Biol. Psychiatry 2002, 51, 68–80. [Google Scholar] [CrossRef]
- Phelps, E.A.; O’Connor, K.J.; Gatenby, J.C.; Gore, J.C.; Grillon, C.; Davis, M. Activation of the left amygdala to a cognitive representation of fear. Nat. Neurosci. 2001, 4, 437–441. [Google Scholar] [CrossRef]
- Lutz, B. Endocannabinoid signals in the control of emotion. Curr. Opin. Pharmacol. 2009, 9, 46–52. [Google Scholar] [CrossRef]
- Segev, A.; Korem, N.; Zer-Aviv, T.M.; Abush, H.; Lange, R.; Sauber, G.; Hillard, C.J.; Akirav, I. Role of endocannabinoids in the hippocampus and amygdala in emotional memory and plasticity. Neuropsychopharmacology 2018, 43, 2017–2027. [Google Scholar] [CrossRef]
- Howlett, A.C.; Abood, M.E. CB1 and CB2 receptor pharmacology. In Advances in Pharmacology; Elsevier: Amsterdam, The Netherlands, 2017; Volume 80, pp. 169–206. [Google Scholar]
- Hourani, W.; Alexander, S.P.H. Cannabinoid ligands, receptors and enzymes: Pharmacological tools and therapeutic potential. Brain Neurosci. Adv. 2018, 2, 2398212818783908. [Google Scholar] [CrossRef]
- Zanettini, C.; Panlilio, L.V.; Aliczki, M.; Goldberg, S.R.; Haller, J.; Yasar, S. Effects of endocannabinoid system modulation on cognitive and emotional behavior. Front. Behav. Neurosci. 2011, 5, 57. [Google Scholar] [CrossRef]
- Hill, M.N.; Gorzalka, B.B. Pharmacological enhancement of cannabinoid CB1 receptor activity elicits an antidepressant-like response in the rat forced swim test. Eur. Neuropsychopharmacol. 2005, 15, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Piomelli, D.; Duranti, A.; Tontini, A.; Mor, M.; Tarzia, G. Modulation of Anxiety through Blockade of Anandamide Hydrolysis. U.S Patent US7,176,201B2, 13 February 2007. [Google Scholar]
- Rubino, T.; Realini, N.; Castiglioni, C.; Guidali, C.; Vigano, D.; Marras, E.; Petrosino, S.; Perletti, G.; Maccarrone, M.; Di Marzo, V.; et al. Role in anxiety behavior of the endocannabinoid system in the prefrontal cortex. Cereb. Cortex 2007, 18, 1292–1301. [Google Scholar] [CrossRef]
- Rubino, T.; Guidali, C.; Viganò, D.; Realini, N.; Valenti, M.; Massi, P.; Parolaro, D. CB1 receptor stimulation in specific brain areas differently modulate anxiety-related behaviour. Neuropharmacology 2008, 54, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Moreira, F.A.; Kaiser, N.; Monory, K.; Lutz, B. Reduced anxiety-like behaviour induced by genetic and pharmacological inhibition of the endocannabinoid-degrading enzyme fatty acid amide hydrolase (FAAH) is mediated by CB1 receptors. Neuropharmacology 2008, 54, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Kinsey, S.G.; O’Neal, S.T.; Long, J.Z.; Cravatt, B.F.; Lichtman, A.H. Inhibition of endocannabinoid catabolic enzymes elicits anxiolytic-like effects in the marble burying assay. Pharmacol. Biochem. Behav. 2011, 98, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Roelke, C.T.; Rademacher, D.J.; Hillard, C.J. Inhibition of restraint stress-induced neural and behavioural activation by endogenous cannabinoid signalling. Eur. J. Neurosci. 2005, 21, 1057–1069. [Google Scholar] [CrossRef]
- Hill, M.N.; Gorzalka, B.B. The endocannabinoid system and the treatment of mood and anxiety disorders. CNS Neurol. Disord. Drug Targets 2009, 8, 451–458. [Google Scholar] [CrossRef]
- Cota, D. The role of the endocannabinoid system in the regulation of hypothalamic-pituitary-adrenal axis activity. J. Neuroendocr. 2008, 20, 35–38. [Google Scholar] [CrossRef]
- Steiner, M.A.; Wotjak, C.T. Role of the endocannabinoid system in regulation of the hypothalamic-pituitary-adrenocortical axis. Prog. Brain Res. 2008, 170, 397–432. [Google Scholar]
- Jucá, M.M.; Filho, F.M.S.C.; De Almeida, J.C.; Mesquita, D.D.S.; Barriga, J.R.D.M.; Dias, K.C.F.; Barbosa, T.M.; Vasconcelos, L.C.; Leal, L.K.A.M.; Ribeiro, J.E.; et al. Flavonoids: Biological activities and therapeutic potential. Nat. Prod. Res. 2018, 34, 692–705. [Google Scholar] [CrossRef]
- Lenman, A.; Fowler, C.J. Interaction of ligands for the peroxisome proliferator-activated receptor γ with the endocannabinoid system. Br. J. Pharmacol. 2007, 151, 1343–1351. [Google Scholar] [CrossRef] [PubMed]
- Thors, L.; Eriksson, J.; Fowler, C.J. Inhibition of the cellular uptake of anandamide by genistein and its analogue daidzein in cells with different levels of fatty acid amide hydrolase-driven uptake. Br. J. Pharmacol. 2007, 152, 744–750. [Google Scholar] [CrossRef] [PubMed]
- Thors, L.; Burston, J.; Alter, B.J.; McKinney, M.; Cravatt, B.; Ross, R.; Pertwee, R.G.; Gereau, R.; Wiley, J.L.; Fowler, C. Biochanin A, a naturally occurring inhibitor of fatty acid amide hydrolase. Br. J. Pharmacol. 2010, 160, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Alam, W.; Khan, H.; Shah, M.A.; Cauli, O.; Saso, L. Kaempferol as a dietary anti-inflammatory agent: Current therapeutic standing. Molecules 2020, 25, 4073. [Google Scholar] [CrossRef] [PubMed]
- Imran, M.; Salehi, B.; Sharifi-Rad, J.; Gondal, T.A.; Saeed, F.; Imran, A.; Shahbaz, M.; Fokou, P.V.T.; Arshad, M.U.; Khan, H.; et al. Kaempferol: A key emphasis to its anticancer potential. Molecules 2019, 24, 2277. [Google Scholar] [CrossRef]
- Kim, H.-Y.; Kim, O.-H.; Sung, M.-K. Effects of phenol-depleted and phenol-rich diets on blood markers of oxidative stress, and urinary excretion of quercetin and kaempferol in healthy volunteers. J. Am. Coll. Nutr. 2003, 22, 217–223. [Google Scholar] [CrossRef]
- Ren, J.; Lu, Y.; Qian, Y.; Chen, B.; Wu, T.; Ji, G. Recent progress regarding kaempferol for the treatment of various diseases. Exp. Ther. Med. 2019, 18, 2759–2776. [Google Scholar] [CrossRef]
- Park, S.-H.; Sim, Y.-B.; Han, P.-L.; Lee, J.-K.; Suh, H.-W. Antidepressant-like effect of kaempferol and quercitirin, isolated from opuntia ficus-indica var. saboten. Exp. Neurobiol. 2010, 19, 30–38. [Google Scholar] [CrossRef]
- Grundmann, O.; Nakajima, J.-I.; Kamata, K.; Seo, S.; Butterweck, V. Kaempferol from the leaves of Apocynum venetum possesses anxiolytic activities in the elevated plus maze test in mice. Phytomedicine 2009, 16, 295–302. [Google Scholar] [CrossRef]
- Babaei, P.; Kouhestani, S.; Jafari, A. Kaempferol attenuates cognitive deficit via regulating oxidative stress and neuroinflammation in an ovariectomized rat model of sporadic dementia. Neural Regen. Res. 2018, 13, 1827–1832. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.S.; Stiff, C.; Lazerwith, S.E.; Kesten, S.R.; Fay, L.K.; Morris, M.; Beidler, D.; Liimatta, M.B.; Smith, S.E.; Dudley, D.T.; et al. Discovery of PF-04457845: A highly potent, orally bioavailable, and selective urea FAAH inhibitor. ACS Med. Chem. Lett. 2011, 2, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, S.; Tripathi, R.K.P.; Ayyannan, S.R. Scaffold hopping-guided design of some isatin based rigid analogs as fatty acid amide hydrolase inhibitors: Synthesis and evaluation. Biomed. Pharmacother. 2018, 107, 1611–1623. [Google Scholar] [CrossRef] [PubMed]
- Doran, C.M.; Kinchin, I. A review of the economic impact of mental illness. Aust. Heal. Rev. 2019, 43, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Pitman, R.K.; Rasmusson, A.M.; Koenen, K.C.; Shin, L.M.; Orr, S.P.; Gilbertson, M.W.; Milad, M.R.; Liberzon, I. Biological studies of post-traumatic stress disorder. Nat. Rev. Neurosci. 2012, 13, 769–787. [Google Scholar] [CrossRef]
- Riebe, C.J.; Wotjak, C.T. Endocannabinoids and stress. Stress 2011, 14, 384–397. [Google Scholar] [CrossRef]
- Patel, S.; Hill, M.N.; Hillard, C.J. Effects of phytocannabinoids on anxiety, mood, and the endocrine system. In Handbook of Cannabis; Oxford University Press: Oxford, UK, 2014; pp. 189–207. [Google Scholar]
- Thors, L.; Belghiti, M.; Fowler, C.J. Inhibition of fatty acid amide hydrolase by kaempferol and related naturally occurring flavonoids. Br. J. Pharmacol. 2008, 155, 244–252. [Google Scholar] [CrossRef]
- McDannald, M.A.; Galarce, E.M. Measuring Pavlovian fear with conditioned freezing and conditioned suppression reveals different roles for the basolateral amygdala. Brain Res. 2011, 1374, 82–89. [Google Scholar] [CrossRef]
- Milad, M.R.; Rauch, S.L.; Pitman, R.K.; Quirk, G.J. Fear extinction in rats: Implications for human brain imaging and anxiety disorders. Biol. Psychol. 2006, 73, 61–71. [Google Scholar] [CrossRef]
- Careaga, M.B.L.; Girardi, C.E.N.; Suchecki, D. Understanding posttraumatic stress disorder through fear conditioning, extinction and reconsolidation. Neurosci. Biobehav. Rev. 2016, 71, 48–57. [Google Scholar] [CrossRef]
- Quirk, G.J.; Mueller, D. Neural mechanisms of extinction learning and retrieval. Neuropsychopharmacology 2007, 33, 56–72. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-H.; Knapska, E.; Orsini, C.A.; Rabinak, C.A.; Zimmerman, J.M.; Maren, S. Fear extinction in rodents. Curr. Protoc. Neurosci. 2009, 47, 8.23.1–8.23.17. [Google Scholar] [CrossRef] [PubMed]
- Singewald, N.; Holmes, A. Rodent models of impaired fear extinction. Psychopharmacology 2019, 236, 21–32. [Google Scholar] [CrossRef]
- Fidelman, S.; Zer-Aviv, T.M.; Lange, R.; Hillard, C.J.; Akirav, I. Chronic treatment with URB597 ameliorates post-stress symptoms in a rat model of PTSD. Eur. Neuropsychopharmacol. 2018, 28, 630–642. [Google Scholar] [CrossRef] [PubMed]
- Chhatwal, J.P.; Ressler, K.J. Modulation of fear and anxiety by the endogenous cannabinoid system. CNS Spectrums 2007, 12, 211–220. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhang, Y.; Ouyang, K.; Lipina, T.V.; Wang, H.; Zhou, Q. Conditioned stimulus presentations alter anxiety level in fear-conditioned mice. Mol. Brain 2019, 12, 28. [Google Scholar] [CrossRef]
- Ponder, C.A.; Kliethermes, C.L.; Drew, M.R.; Müller, J.; Das, K.; Risbrough, V.B.; Crabbe, J.C.; Gilliam, T.C.; Palmer, A.A. Selection for contextual fear conditioning affects anxiety-like behaviors and gene expression. Genes Brain Behav. 2007, 6, 736–749. [Google Scholar] [CrossRef]
- Burman, M.A.; Szolusha, K.; Bind, R.; Kerney, K.; Boger, D.L.; Bilsky, E.J. FAAH inhibitor OL-135 disrupts contextual, but not auditory, fear conditioning in rats. Behav. Brain Res. 2016, 308, 1–5. [Google Scholar] [CrossRef]
- Haller, J.; Goldberg, S.R.; Pelczer, K.G.; Aliczki, M.; Panlilio, L.V. The effects of anandamide signaling enhanced by the FAAH inhibitor URB597 on coping styles in rats. Psychopharmacology 2013, 230, 353–362. [Google Scholar] [CrossRef]
- Bitencourt, R.M.; Pamplona, F.A.; Takahashi, R.N. Facilitation of contextual fear memory extinction and anti-anxiogenic effects of AM404 and cannabidiol in conditioned rats. Eur. Neuropsychopharmacol. 2008, 18, 849–859. [Google Scholar] [CrossRef]
- Gunduz-Cinar, O.; MacPherson, K.P.; Cinar, R.; Gamble-George, J.; Sugden, K.; Williams, B.; Godlewski, G.; Ramikie, T.S.; Gorka, A.X.; Alapafuja, S.O.; et al. Convergent translational evidence of a role for anandamide in amygdala-mediated fear extinction, threat processing and stress-reactivity. Mol. Psychiatry 2012, 18, 813–823. [Google Scholar] [CrossRef] [PubMed]
- Haller, J.; Barna, I.; Barsvari, B.; Pelczer, K.G.; Yasar, S.; Panlilio, L.; Goldberg, S. Interactions between environmental aversiveness and the anxiolytic effects of enhanced cannabinoid signaling by FAAH inhibition in rats. Psychopharmacology 2009, 204, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Pamplona, F.A.; Takahashi, R.N. WIN 55212-2 impairs contextual fear conditioning through the activation of CB1 cannabinoid receptors. Neurosci. Lett. 2006, 397, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Vissiennon, C.; Nieber, K.; Kelber, O.; Butterweck, V. Route of administration determines the anxiolytic activity of the flavonols kaempferol, quercetin and myricetin—Are they prodrugs? J. Nutr. Biochem. 2012, 23, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Scherma, M.; Medalie, J.; Fratta, W.; Vadivel, S.K.; Makriyannis, A.; Piomelli, D.; Mikics, E.; Haller, J.; Yasar, S.; Tanda, G.; et al. The endogenous cannabinoid anandamide has effects on motivation and anxiety that are revealed by fatty acid amide hydrolase (FAAH) inhibition. Neuropharmacology 2008, 54, 129–140. [Google Scholar] [CrossRef]
- Bluett, R.J.; Gamble-George, J.; Hermanson, D.J.; Hartley, N.D.; Marnett, L.J.; Patel, S. Central anandamide deficiency predicts stress-induced anxiety: Behavioral reversal through endocannabinoid augmentation. Transl. Psychiatry 2014, 4, e408. [Google Scholar] [CrossRef]
- Bedse, G.; Colangeli, R.; Lavecchia, A.M.; Romano, A.; Altieri, F.; Cifani, C.; Cassano, T.; Egaetani, S. Role of the basolateral amygdala in mediating the effects of the fatty acid amide hydrolase inhibitor URB597 on HPA axis response to stress. Eur. Neuropsychopharmacol. 2014, 24, 1511–1523. [Google Scholar] [CrossRef]
- Zaman, N.; Ahmad, H.; Abid, M.A.; Rahman, N.-U.; Roome, T.; Abbas, G. Acetaminophen (paracetamol) facilitated extinction lear ning in contextual fear conditioned rats. Lett. Drug Des. Discov. 2017, 14, 898–903. [Google Scholar] [CrossRef]
- Mayo, L.M.; Asratian, A.; Lindé, J.; Morena, M.; Haataja, R.; Hammar, V.; Augier, G.; Hill, M.N.; Heilig, M. Elevated anandamide, enhanced recall of fear extinction, and attenuated stress responses following inhibition of fatty acid amide hydrolase: A randomized, controlled experimental medicine trial. Biol. Psychiatry 2019, 87, 538–547. [Google Scholar] [CrossRef]
- Min, X.; Thibault, S.T.; Porter, A.C.; Gustin, D.J.; Carlson, T.J.; Xu, H.; Lindstrom, M.; Xu, G.; Uyeda, C.; Ma, Z.; et al. Discovery and molecular basis of potent noncovalent inhibitors of fatty acid amide hydrolase (FAAH). Proc. Natl. Acad. Sci. USA 2011, 108, 7379–7384. [Google Scholar] [CrossRef]
- Seierstad, M.; Breitenbucher, J.G. Discovery and development of fatty acid amide hydrolase (FAAH) Inhibitors. J. Med. Chem. 2008, 51, 7327–7343. [Google Scholar] [CrossRef] [PubMed]
- Boger, D.L.; Miyauchi, H.; Du, W.; Hardouin, C.; Fecik, R.A.; Cheng, H.; Hwang, I.; Hedrick, M.P.; Leung, D.; Acevedo, O.; et al. Discovery of a potent, selective, and efficacious class of reversible α-ketoheterocycle inhibitors of fatty acid amide hydrolase effective as analgesics. J. Med. Chem. 2005, 48, 1849–1856. [Google Scholar] [CrossRef] [PubMed]
- Kage, K.L.; Richardson, P.L.; Traphagen, L.; Severin, J.; Pereda-Lopez, A.; Lübben, T.; Davis-Taber, R.; Vos, M.H.; Bartley, D.; Walter, K.; et al. A high throughput fluorescent assay for measuring the activity of fatty acid amide hydrolase. J. Neurosci. Methods 2007, 161, 47–54. [Google Scholar] [CrossRef]
- Berardi, A.; Trezza, V.; Palmery, M.; Trabace, L.; Cuomo, V.; Campolongo, P. An updated animal model capturing both the cognitive and emotional features of post-traumatic stress disorder (PTSD). Front. Behav. Neurosci. 2014, 8, 142. [Google Scholar] [CrossRef] [PubMed]
- Pellow, S.; Chopin, P.; File, S.E.; Briley, M. Validation of open: Closed arm entries in an elevated plus-maze as a measure of anxiety in the rat. J. Neurosci. Methods 1985, 14, 149–167. [Google Scholar] [CrossRef]
- Walf, A.A.; Frye, C.A. The use of the elevated plus maze as an assay of anxiety-related behavior in rodents. Nat. Protoc. 2007, 2, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Schrödinger. 3: Maestro; LLC: New York, NY, USA, 2019. [Google Scholar]
- Zhang, J.; Hao, Q.; Liu, X.; Jing, Z.; Jia, W.-Q.; Wang, S.-Q.; Xu, W.-R.; Cheng, X.-C.; Wang, R. Molecular docking, 3D-QSAR and structural optimization on imidazo-pyridine derivatives dually targeting AT1 and PPARγ. Oncotarget 2017, 8, 25612–25627. [Google Scholar] [CrossRef][Green Version]
- Borkotoky, S.; Meena, C.K.; Murali, A. Interaction analysis of T7 RNA polymerase with heparin and its low molecular weight derivatives—An in silico approach. Bioinform. Biol. Insights 2016, 10, 155–166. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |

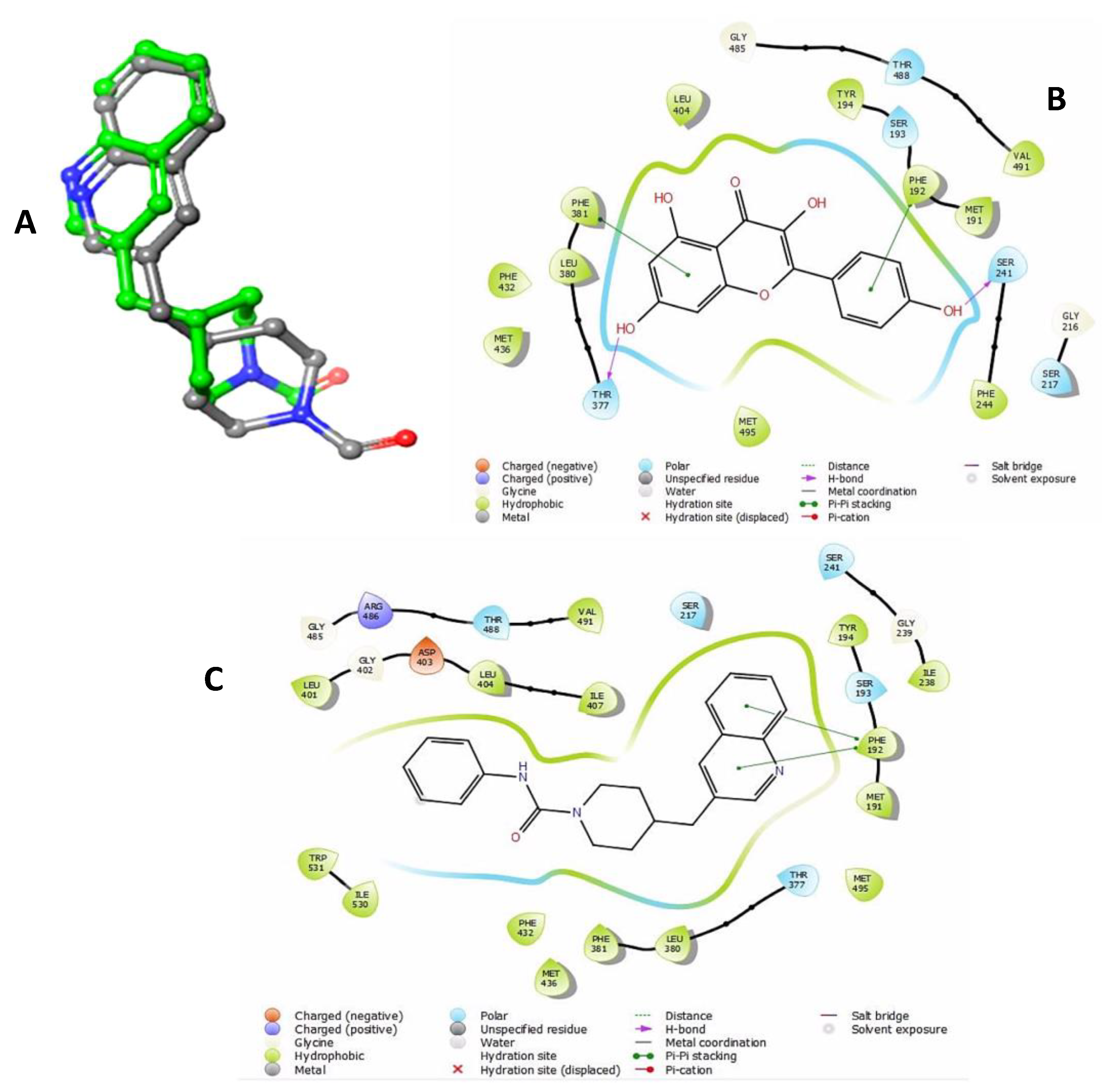

| Target | Compound | Glide Score (Kcal/mol) | Prime MM/GBSA ΔGbind (Kcal/mol) | Interacting Residues | Interaction Types |
|---|---|---|---|---|---|
| FAAH | PF-750 | −8.575 | −54.46 | PHE192 | Pi–Pi stacking |
| Kaempferol | −9.199 | −63.92 | SER241, PHE192, PHE381, THR377 | H-bonding, Pi–Pi stacking |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad, H.; Rauf, K.; Zada, W.; McCarthy, M.; Abbas, G.; Anwar, F.; Shah, A.J. Kaempferol Facilitated Extinction Learning in Contextual Fear Conditioned Rats via Inhibition of Fatty-Acid Amide Hydrolase. Molecules 2020, 25, 4683. https://doi.org/10.3390/molecules25204683
Ahmad H, Rauf K, Zada W, McCarthy M, Abbas G, Anwar F, Shah AJ. Kaempferol Facilitated Extinction Learning in Contextual Fear Conditioned Rats via Inhibition of Fatty-Acid Amide Hydrolase. Molecules. 2020; 25(20):4683. https://doi.org/10.3390/molecules25204683
Chicago/Turabian StyleAhmad, Hammad, Khalid Rauf, Wahid Zada, Margaret McCarthy, Ghulam Abbas, Fareeha Anwar, and Abdul Jabbar Shah. 2020. "Kaempferol Facilitated Extinction Learning in Contextual Fear Conditioned Rats via Inhibition of Fatty-Acid Amide Hydrolase" Molecules 25, no. 20: 4683. https://doi.org/10.3390/molecules25204683
APA StyleAhmad, H., Rauf, K., Zada, W., McCarthy, M., Abbas, G., Anwar, F., & Shah, A. J. (2020). Kaempferol Facilitated Extinction Learning in Contextual Fear Conditioned Rats via Inhibition of Fatty-Acid Amide Hydrolase. Molecules, 25(20), 4683. https://doi.org/10.3390/molecules25204683






