Selective Upregulation by Theanine of Slc38a1 Expression in Neural Stem Cell for Brain Wellness
Abstract
1. Introduction
2. Theanine on Neural Stem Cells (NSCs)
2.1. Neural Progenitor Cells (NPCs)
2.2. Modulation by Glu of Neurogenesis
2.3. Activation by Theanine of Embryonic Neurogenesis
2.4. Theanine on Adult Neurogenesis
3. Correlation between Theanine and GLN Transporters
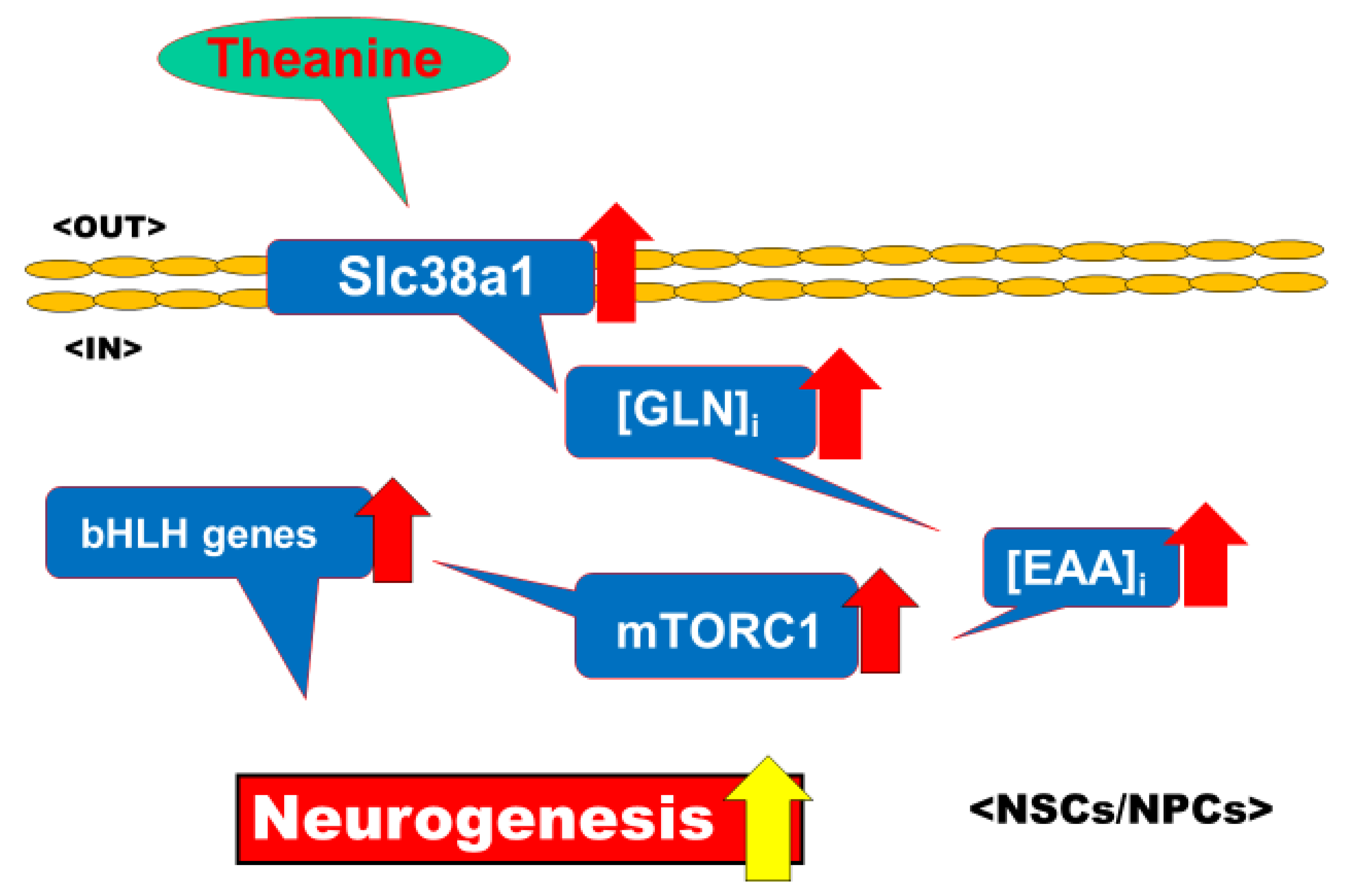
3.1. Selective Upregulation by Theanine of Slc38a1 Expression in NPCs
3.2. Usefulness of Pluripotent P19 Cells as NPC Model
3.3. Stimulation by Theanine of Slc38a1 Promoter in P19 Cells
3.4. Establishment of Stable Transfectants of Slc38a1 in P19 Cells
4. Roles of GLN in the Brain
4.1. Replenishment of Amino Acid Neurotransmitter Pools
4.2. Modulation of Ammonia Homeostasis
4.3. Pharmacological Profiles of [3H]GLN Uptake
4.4. Activation by GLN of mTOR Signaling
4.5. Significance of Selective Upregulation of Slc38a1 Expression
4.6. Mechanisms Underlying Upregulation of Slc38a1 Expression
4.7. Conditional Knockout Mice Devoid of Slc38a1 from Neurons
5. Theanine for Human Brain Wellness
6. Conclusions
Funding
Conflicts of Interest
Abbreviations
References
- Biegon, A.; Liraz-Zaltsman, S.; Shohami, E. Stimulation of N-methyl-d-aspartae receptors by exogenous and endogenous ligands improves outcome of brain injury. Curr. Opin. Neurol. 2018, 31, 687–692. [Google Scholar] [CrossRef]
- Ashihara, H. Occurrence, biosynthesis and metabolism of theanine (γ-glutamyl-l-ethylamide) in plants: A comprehensive review. Nat. Prod. Commun. 2015, 10, 803–810. [Google Scholar] [CrossRef]
- Sakato, Y. Studies on chemical constituents of tea. Part III. On a new amide theanine. Nippon Nogeikagaku Kaishi 1949, 23, 262–267. (In Japanese) [Google Scholar] [CrossRef]
- Ikegaya, K.; Takayanagi, H.; Anan, T. Chemical composition of Matcha. Tea Res. J. 1984, 60, 79–81. [Google Scholar] [CrossRef]
- Yoneda, Y.; Kuramoto, N.; Kawada, K. The role of glutamine in neurogenesis promoted by the green tea amino acid theanine in neural progenitor cells for brain health. Neurochem. Int. 2019, 129, 104505. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhao, B. Natural antioxidants in prevention and management of Alzheimer’s disease. Front. Biosci. Elite Ed. 2012, 4, 794–808. [Google Scholar] [CrossRef]
- Kakuda, T.; Nozawa, A.; Sugimoto, A.; Niino, H. Inhibition by L-theanine of binding of [3H]AMPA, [3H]kainate, and [3H]MDL 105,519 to glutamate receptors. Biosci. Biotechnol. Biochem. 2002, 66, 2683–2686. [Google Scholar] [CrossRef]
- Unno, K.; Fujitani, K.; Takamori, N.; Takabayashi, F.; Maeda, K.; Miyazaki, H.; Tanida, N.; Iguchi, K.; Shimoi, K.; Hoshino, M. Theanine intake improves the shortened lifespan, cognitive dysfunction and behavioural depression that are induced by chronic psychosocial stress in mice. Free Radic. Res. 2011, 45, 966–974. [Google Scholar] [CrossRef]
- Takarada, T.; Nakamichi, N.; Kakuda, T.; Nakazato, R.; Kokubo, H.; Ikeno, S.; Nakamura, S.; Hinoi, E.; Yoneda, Y. Daily oral intake of L-theanine prevents the decline of 5-bromo-2′-deoxyuridine incorporation in hippocampal dentate gyrus with concomitant alleviation of behavioral abnormalities in adult mice with severe traumatic stress. J. Pharmacol. Sci. 2015, 127, 292–297. [Google Scholar] [CrossRef]
- Nguyen, B.T.; Sharma, N.; Shin, E.J.; Jeong, J.H.; Lee, S.H.; Jang, C.G.; Nah, S.Y.; Nabeshima, T.; Yoneda, Y.; Kim, H.C. Theanine attenuates memory impairments induced by klotho gene depletion in mice. Food Funct. 2019, 10, 325–332. [Google Scholar] [CrossRef]
- Yoneda, Y. An L-glutamine transporter isoform for neurogenesis facilitated by L-theanine. Neurochem. Res. 2017, 42, 2686–2697. [Google Scholar] [CrossRef] [PubMed]
- Gage, F.H.; Coates, P.W.; Palmer, T.D.; Kuhn, H.G.; Fisher, L.J.; Suhonen, J.O.; Peterson, D.A.; Suhr, S.T.; Ray, J. Survival and differentiation of adult neuronal progenitor cells transplanted to the adult brain. Proc. Natl. Acad. Sci. USA 1995, 92, 11879–11883. [Google Scholar] [CrossRef] [PubMed]
- Suhonen, J.O.; Peterson, D.A.; Ray, J.; Gage, F.H. Differentiation of adult hippocampus-derived progenitors into olfactory neurons in vivo. Nature 1996, 383, 624–627. [Google Scholar] [CrossRef] [PubMed]
- Doetsch, F.; Caille, I.; Lim, D.A.; Garcia-Verdugo, J.M.; Alvarez-Buylla, A. Subventricular zone astrocytes are neural stem cell in the adult mammalian brain. Cell 1999, 97, 703–716. [Google Scholar] [CrossRef]
- Temple, S.; Alvarez-Buylla, A. Stem cells in the adult mammalian central nervous system. Curr. Opin. Neurobiol. 1999, 9, 135–141. [Google Scholar] [CrossRef]
- Yoneyama, M.; Fukui, M.; Nakamichi, N.; Kitayama, T.; Taniura, H.; Yoneda, Y. Activation of GABAA receptors facilitates astroglial differentiation induced by ciliary neurotrophic factor in neural progenitors isolated from fetal rat brain. J. Neurochem. 2007, 100, 1667–1679. [Google Scholar]
- Fukui, M.; Nakamichi, N.; Yoneyama, M.; Ozawa, S.; Fujimori, S.; Takahata, Y.; Nakamura, N.; Taniura, H.; Yoneda, Y. Modulation of cellular proliferation and differentiation through GABAB receptors expressed by undifferentiated neural progenitor cells isolated from fetal mouse brain. J. Cell. Physiol. 2008, 216, 507–519. [Google Scholar] [CrossRef]
- Kitayama, T.; Yoneyama, M.; Yoneda, Y. Possible regulation by N-methyl-D-aspartate receptors of proliferative progenitor cells expressed in adult mouse hippocampal dentate gyrus. J. Neurochem. 2003, 84, 767–780. [Google Scholar] [CrossRef]
- Kitayama, T.; Yoneyama, M.; Tamaki, K.; Yoneda, Y. Regulation of neuronal differentiation by N-methyl-D-aspartate receptors expressed in neural progenitor cells isolated from adult mouse hippocampus. J. Neurosci. Res. 2004, 76, 599–612. [Google Scholar] [CrossRef]
- Nakamichi, N.; Takarada, T.; Yoneda, Y. Neurogenesis mediated by γ-aminobutyric acid and glutamate signaling. J. Pharmacol. Sci. 2009, 110, 133–149. [Google Scholar] [CrossRef]
- Takarada, T.; Ogura, M.; Nakamichi, N.; Kakuda, T.; Nakazato, R.; Kokubo, H.; Ikeno, S.; Nakamura, S.; Kutsukake, T.; Hinoi, E.; et al. Upregulation of Slc38a1 gene along with promotion of neurosphere growth and subsequent neuronal specification in undifferentiated neural progenitor cells exposed to L-theanine. Neurochem. Res. 2016, 41, 5–15. [Google Scholar] [CrossRef]
- Takarada, T.; Nakamichi, N.; Nakazato, R.; Kakuda, T.; Kokubo, H.; Ikeno, S.; Nakamura, S.; Kuramoto, N.; Hinoi, E.; Yoneda, Y. Possible activation by the green tea amino acid L-theanine of mammalian target of rapamycin signaling in undifferentiated neural progenitor cells in vitro. Biochem. Biophys. Rep. 2016, 5, 89–95. [Google Scholar] [CrossRef]
- Takeda, A.; Sakamoto, K.; Tamano, H.; Fukura, K.; Inui, N.; Suh, S.W.; Won, S.J.; Yokogishi, H. Facilitated neurogenesis in the developing hippocampus after intake of L-theanine, an amino acid in tea leaves, and object recognition memory. Cell. Mol. Neurobiol. 2011, 31, 1079–1088. [Google Scholar] [CrossRef]
- Altman, J.; Das, G.D. Autoradiographic and histological evidence of postnatal hippocampal neurogenesis in rats. J. Comp. Neurol. 1965, 124, 319–336. [Google Scholar] [CrossRef]
- Johansson, C.B.; Momma, S.; Clarke, D.L.; Risling, M.; Lendahl, U.; Frisen, J. Identification of a neural stem cell in the adult mammalian central nervous system. Cell 1999, 96, 25–34. [Google Scholar] [CrossRef]
- Mignone, J.L.; Kukekov, V.; Chiang, A.S.; Steindler, D.; Enikolopov, G. Neural stem and progenitor cells in nestin-GFP transgenic mice. J. Comp. Neurol. 2004, 469, 311–324. [Google Scholar] [CrossRef]
- Dennis, C.V.; Sheahan, P.J.; Graeber, M.B.; Sheedy, D.L.; Kril, J.J.; Sutherland, G.T. Microglial proliferation in the brain of chronic alcoholics with hepatic encephalopathy. Metab. Brain Dis. 2014, 29, 1027–1039. [Google Scholar] [CrossRef]
- Doorn, K.J.; Drukarch, B.; van Dam, A.M.; Lucassen, P.J. Hippocampal proliferation is increased in presymptomatic Parkinson’s disease and due to microglia. Neural Plast. 2014, 2014, 9591254. [Google Scholar] [CrossRef]
- Boldrini, M.; Fulmore, C.A.; Tartt, A.N.; Simeon, L.R.; Pavlova, I.; Poposka, V.; Rosoklija, G.B.; Stankov, A.; Arango, V.; Dwork, A.J.; et al. Human hippocampal neurogenesis persists throughout aging. Cell Stem Cell 2018, 22, 589–599. [Google Scholar] [CrossRef]
- Moreno-Jimenez, E.P.; Flor-Garcia, M.; Terreros-Roncal, J.; Rabano, A.; Cafini, F.; Pallas-Bazarra, N.; Avila, J.; Llorens-Martin, M. Adult hippocampal neurogenesis is abundant in neurologically healthy subjects and drops sharply in patients with Alzheimer’s disease. Nat. Med. 2019, 25, 554–560. [Google Scholar] [CrossRef]
- Tobin, M.K.; Musaraca, K.; Disouky, A.; Shetti, A.; Bheri, A.; Honer, W.G.; Kim, N.; Dawe, R.J.; Bennett, D.A.; Arfanakis, K.; et al. Human hippocampal neurogenesis persists in aged adults and Alzheimer’s disease patients. Cell Stem Cell 2019, 24, P974–P982. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, O.; Liebl, J.; Bernard, S.; Alkass, K.; Yeung, M.S.; Steier, P.; Kutschera, W.; Johnson, L.; Landen, M.; Druid, H.; et al. The age of olfactory bulb neurons in humans. Neuron 2012, 74, 634–639. [Google Scholar] [CrossRef] [PubMed]
- McBurney, M.W.; Rogers, B.J. Isolation of male embryonal carcinoma cells and their chromosome replication patterns. Dev. Biol. 1982, 89, 503–508. [Google Scholar] [CrossRef]
- Jones-Villeneuve, E.M.; McBurney, M.W.; Rogers, K.A.; Kalnins, V.I. Retinoic acid induces embryonal carcinoma cells to differentiate into neurons and glial cells. J. Cell Biol. 1982, 94, 253–262. [Google Scholar] [CrossRef]
- Ogura, M.; Kakuda, T.; Takarada, T.; Nakamichi, N.; Fukumori, R.; Kim, Y.H.; Hinoi, E.; Yoneda, Y. Promotion of both proliferation and differentiation in pluripotent P19 cells with stable overexpression of the GLN transporter Slc38a1. PLoS ONE 2012, 7, e48270. [Google Scholar] [CrossRef]
- Ogura, M.; Nakamichi, N.; Takano, K.; Oikawa, H.; Kambe, Y.; Ohno, Y.; Taniura, H.; Yoneda, Y. Functional expression of a GLN transporter responsive to down-regulation by lipopolysaccharide through reduced promoter activity in cultured rat neocortical astrocytes. J. Neurosci. Res. 2006, 83, 1447–1460. [Google Scholar] [CrossRef]
- Ogura, M.; Taniura, H.; Nakamichi, N.; Yoneda, Y. Upregulation of the GLN transporter through transactivation mediated by cAMP/protein kinase A signals toward exacerbation of vulnerability to oxidative stress in rat neocortical astrocytes. J. Cell. Physiol. 2007, 212, 375–385. [Google Scholar] [CrossRef]
- Butterworth, R.F. Pathophysiology of brain dysfunction in hyperammonemic syndromes: The many faces of glutamine. Mol. Gen. Met. 2014, 113, 113–117. [Google Scholar] [CrossRef]
- Rae, C.; Hare, N.; Bubb, W.A.; McEwan, S.R.; Broer, A.; McQuillan, J.A.; Balcar, V.J.; Conigrave, A.D.; Broer, S. Inhibition of glutamine transporter depletes glutamate and GABA neurotransmitter pools: Further evidence for metabolic compartmentation. J. Neurochem. 2003, 85, 503–514. [Google Scholar] [CrossRef]
- Wang, L.; Maher, T.J.; Wurtman, R.J. Oral L0glutamine increases GABA levels in striatal tissue and extracellular fluid. FASEB J. 2007, 21, 1227–1232. [Google Scholar] [CrossRef]
- Qureshi, T.; Sorensen, C.; Berghuis, P.; Jensen, V.; Dobszay, M.B.; Farkas, T.; Dalen, K.T.; Guo, C.; Hassel, B.; Utheim, T.P.; et al. The glutamine transporter Slc38a1 regulates GABAergic neurotransmission and synaptic plasticity. Cereb. Cortex 2019, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Schousboe, A.; Hertz, L.; Svenneby, G.; Kvamme, E. Phosphate activated glutaminase activity and GLN uptake in primary cultures of astrocytes. J. Neurochem. 1979, 32, 943–950. [Google Scholar] [CrossRef] [PubMed]
- Laake, J.H.; Slyngstad, T.A.; Haug, F.M.; Otterson, O.P. Glutamine from glial cells is essential for the maintenance of the nerve terminal pool of glutamate; immunological evidence from hippocampal slice cultures. J. Neurosci. 1999, 88, 1137–1151. [Google Scholar] [CrossRef]
- Olalla, L.; Gutierrez, A.; Campos, J.A.; Khan, Z.U.; Alonso, F.J.; Segura, J.A.; Marquez, J.; Aledo, J.C. Nuclear localization of L-type glutaminase in mammalian brain. J. Biol. Chem. 2002, 277, 38939–38944. [Google Scholar] [CrossRef]
- Marquez, J.; Tosina, M.; de la Rosa, V.; Segura, J.A.; Alonso, F.J.; Mates, J.M.; Campos-Sandoval, J.A. New insights into brain glutaminases: Beyond their role on glutamatergic transmission. Neurochem. Int. 2009, 55, 64–70. [Google Scholar] [CrossRef]
- Tsuge, H.; Sano, S.; Hayakawa, T.; Kakuda, T.; Unno, T. L-theanine, gamma-glutamylethylamide, is metabolized by renal phosphate-independent glutaminase. Biochim. Biophys. Acta 2003, 1620, 47–53. [Google Scholar] [CrossRef]
- Balcar, V.J.; Johnston, G.A.R. High affinity uptake of L-GLN in rat brain slices. J. Neurochem. 1975, 24, 875–879. [Google Scholar] [CrossRef]
- Nagaraja, T.N.; Brookes, N. GLN transport in mouse cerebral astrocytes. J. Neurochem. 1996, 66, 1665–1674. [Google Scholar] [CrossRef]
- Albers, A.; Broer, A.; Wagner, C.A.; Setiawan, I.; Lang, P.A.; Kranz, E.U.; Lang, F.; Broer, S. Na+ transport by the neural GLN transporter ATA1. Pflugers Arch. 2001, 443, 92–101. [Google Scholar] [CrossRef]
- Chaudhry, F.A.; Schmitz, D.; Reimer, R.J.; Larsson, P.; Gray, A.T.; Nicoll, R.; Kavanaugh, M.; Edwards, R.H. GLN uptake by neurons: Interaction of protons with system A transporters. J. Neurosci. 2002, 22, 62–72. [Google Scholar] [CrossRef]
- Kakuda, T.; Hinoi, E.; Abe, A.; Nozawa, A.; Ogura, M.; Yoneda, Y. L-theanine, an ingredient of green tea, inhibits [3H]GLN transport in neurons and astroglia in rat brain. J. Neurosci. Res. 2008, 86, 1846–1856. [Google Scholar] [CrossRef] [PubMed]
- Jhanwar-Uniyal, M.; Amin, A.G.; Cooper, J.B.; Das, K.; Schmidt, M.H.; Murali, R. Discrete signaling mechanisms of mTORC1 and mTORC2: Connected yet apart in cellular and molecular aspects. Adv. Biol. Reg. 2017, 64, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Fumarola, C.; La Monica, S.; Guidotti, G.G. Amino acid signaling through the mammalian target of rapamycin (mTOR) pathway: Role of GLN and of cell shrinkage. J. Cell. Physiol. 2005, 204, 155–165. [Google Scholar] [CrossRef]
- Nicklin, P.; Bergman, P.; Zhang, B.; Triantafellow, E.; Wang, H.; Nyfeler, B.; Yang, H.; Hild, M.; Kung, C.; Wilson, C.; et al. Bidirectional transport of amino acids regulates mTOR and autophagy. Cell 2009, 136, 521–534. [Google Scholar] [CrossRef]
- Russell, R.C.; Fang, C.; Guan, K.L. An emerging role for TOR signaling in mammalian tissue and stem cell physiology. Dev. Camb. Eng. 2011, 138, 3343–3356. [Google Scholar] [CrossRef]
- Endo, M.; Antonyak, M.A.; Cerione, R.A. Cdc42-mTOR signaling pathway controls Nes5 and Pax6 expression in retinoic acid-dependent neural differentiation. J. Biol. Chem. 2009, 284, 5107–5118. [Google Scholar] [CrossRef]
- Tee, A.R.; Sampson, J.R.; Pal, D.K.; Bateman, J.M. The role of mTOR signaling in neurogenesis, insights from tuberous sclerosis complex. Semin. Cell. Dev. Biol. 2016, 52, 12–20. [Google Scholar] [CrossRef]
- LiCausi, F.; Hartman, N.W. Role of mTOR complexes in neurogenesis. Int. J. Mol. Sci. 2018, 19, 1544. [Google Scholar] [CrossRef]
- Cangiano, A. Denervation supersensitivity as a model for the neural control of muscle. Neuroscience 1985, 14, 963–971. [Google Scholar] [CrossRef]
- Nardone, R.; Holler, Y.; Thomschewski, A.; Holler, P.; Lochner, P.; Golaszewski, S.; Brigo, F.; Trinka, E. Serotonergic transmission after spinal injury. J. Neural Transm. 2015, 122, 279–295. [Google Scholar] [CrossRef]
- Duran, R.V.; Oppliger, W.; Robitaille, A.M.; Heiserich, L.; Skendaj, R.; Gottlieb, E.; Hall, M.N. Glutaminolysis activates Rag-mTORC1 signaling. Mol. Cell 2012, 47, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Yamada, D.; Kawabe, K.; Tosa, I.; Tsukamoto, S.; Nakazato, R.; Kou, M.; Fujikawa, K.; Nakamura, S.; Ono, M.; Oohashi, T.; et al. Inhibition of the glutamine transporter SNAT1 confers neuroprotection in mice by modulating the mTOR-autophagy system. Commun. Biol. 2019, 2, 346. [Google Scholar] [CrossRef] [PubMed]
- Buckley, K.M.; Hess, D.L.; Sazonova, I.Y.; Periyasamy-Thandavan, S.; Barrett, J.R.; Kirks, R.; Grace, H.; Kondrikova, G.; Johnson, M.H.; Hess, D.C.; et al. Rapamycin up-regulation of autophagy reduces infarct size and improves outcomes in both permanent MCAL, and embolic MCAO, murine models of stroke. Exp. Transl. Stroke Med. 2014, 6, 8. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Sun, F.; Wang, J.; Mao, X.; Xie, L.; Yang, S.H.; Su, D.M.; Simpkins, J.M.; Greenberg, D.A.; Jin, K. mTOR signaling inhibition modulates macrophage/microglia-mediated neuroinflammation and secondary injury via regulatory T cells after focal ischemia. J. Immunol. 2014, 192, 6009–6019. [Google Scholar] [CrossRef]
- Wu, M.; Zhang, H.; Kai, J.; Dong, J.; Xu, Z.; Wong, M.; Zeng, L.H. Rapamycin prevents cerebral stroke by modulating apoptosis nd autophagy in penumbra in rats. Ann. Clin. Transl. Neurosci. 2017, 5, 138–146. [Google Scholar] [CrossRef]
- Ogura, M.; Takarada, T.; Nakamichi, N.; Kawagoe, H.; Sako, A.; Nakazato, R.; Yoneda, Y. Exacerbated vulnerability to oxidative stress in astrocytic C6 glioma cells with stable overexpression of the GLN transporter slc38a1. Neurochem. Int. 2011, 58, 504–511. [Google Scholar] [CrossRef]
- Kimura, K.; Ozeki, M.; Juneja, L.R.; Ohira, H. L-theanine reduces psychological and physiological stress response. Biol. Psychol. 2007, 74, 39–45. [Google Scholar] [CrossRef]
- Yoto, A.; Motoki, M.; Murano, S.; Yokogoshi, H. Effects of L-theanine or caffeine intake on changes in blood pressure under physical and psychological stresses. J. Physiol. Anthropol. 2012, 31, 28. [Google Scholar] [CrossRef]
- Hidese, S.; Ota, M.; Wakabayashi, C.; Noda, T.; Ozawa, H.; Okubo, T.; Kunugi, H. Effects of chronic l-theanine administration in patients with major depressive disorder: An open-label study. Acta Neuropsychiatr. 2017, 29, 72–79. [Google Scholar] [CrossRef]
- Lyon, M.R.; Kapoor, M.P.; Juneja, L.R. The effects of L-L-theanine (Sun L-theanine) on objective sleep quality in boys with attention deficit hyperactivity disorder (ADHD): A randomized, double-blind, placebo-controlled clinical trial. Altern. Med. Rev. 2011, 16, 348–354. [Google Scholar]
- Barrett, J.R.; Tracy, D.K.; Giaroli, G. To sleep or not sleep: A systemic review of the literature of pharmacological treatments of insomnia in children and adolescents with attention deficit/hyperactivity disorder. J. Child Adolesc. Psychopharmacol. 2013, 23, 640–647. [Google Scholar] [CrossRef] [PubMed]
- Rao, T.P.; Ozeki, M.; Juneja, L.R. In search of a safe natural sleep aid. J. Am. Coll. Nutr. 2015, 34, 436–447. [Google Scholar] [CrossRef] [PubMed]
- Ritsner, M.S.; Miodownik, C.; Ratner, Y.; Shleifer, T.; Mar, M.; Pintov, L.; Lerner, V. L-theanine relieves positive, activation, and anxiety symptoms in patients with schizophrenia and schizoaffective disorders: An 8-week, randomized. double-blind, placebo-controlled, 2-center study. J. Clin. Psychiatr. 2011, 72, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Miodownik, C.; Maayan, R.; Ratner, Y.; Lerner, V.; Pintov, L.; Mar, M.; Weizman, A.; Ritsner, M.S. Serum levels of brain-derived neurotrophic factor and cortisol to sulfate of dehydroepiandrosterone molar ratio associated with clinical response to L-theanine as augmentation of antipsychotic therapy in schizophrenia and schizoaffective disorder patients. Clin. Neuropharmacol. 2011, 34, 155–160. [Google Scholar] [CrossRef]
- Lardner, A.L. Neurobiological effects of the green tea constituent theanine and its potential role in the treatment of psychiatric and neurodegenerative disorders. Nutr. Neurosci. 2014, 17, 145–155. [Google Scholar] [CrossRef]
- Yokogoshi, H.; Kobayashi, M.; Mochizuki, M.; Terashima, T. Effect of L-theanine, γ-glutamylethylamide, on brain monoamines and striatal dopamine release in conscious rats. Neurochem. Res. 1998, 23, 667–673. [Google Scholar] [CrossRef]
- Kakuda, T. Neuroprotective effects of theanine and its preventive effects on cognitive dysfunction. Pharmacol. Res. 2011, 64, 162–168. [Google Scholar] [CrossRef]
- Olthof, M.R.; Hollman, P.C.H.; Katan, M.B. Chlorogenic acid and caffeic acid are absorbed in humans. J. Nutr. 2001, 131, 66–71. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jimenez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef]
- Kim, Y.; Keogh, J.B.; Clifton, P.M. Polyphenols and glycemic control. Nutrients 2016, 8, 17. [Google Scholar] [CrossRef]
- Squillaro, T.; Schettino, C.; Sampaolo, S.; Galderisi, U.; Di Iorio, G.; Giordano, A.; Melone, M.A.B. Adult-onset brain tumors and neurodegeneration: Are polyphenols protective? J. Cell. Physiol. 2018, 233, 3955–3967. [Google Scholar] [CrossRef] [PubMed]
- Yoneda, Y. An urgent need for validated nutraceutical sciences. J. Pharmacol. Ther. Forecast. 2018, 1, 1001. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoneda, Y.; Kawada, K.; Kuramoto, N. Selective Upregulation by Theanine of Slc38a1 Expression in Neural Stem Cell for Brain Wellness. Molecules 2020, 25, 347. https://doi.org/10.3390/molecules25020347
Yoneda Y, Kawada K, Kuramoto N. Selective Upregulation by Theanine of Slc38a1 Expression in Neural Stem Cell for Brain Wellness. Molecules. 2020; 25(2):347. https://doi.org/10.3390/molecules25020347
Chicago/Turabian StyleYoneda, Yukio, Koichi Kawada, and Nobuyuki Kuramoto. 2020. "Selective Upregulation by Theanine of Slc38a1 Expression in Neural Stem Cell for Brain Wellness" Molecules 25, no. 2: 347. https://doi.org/10.3390/molecules25020347
APA StyleYoneda, Y., Kawada, K., & Kuramoto, N. (2020). Selective Upregulation by Theanine of Slc38a1 Expression in Neural Stem Cell for Brain Wellness. Molecules, 25(2), 347. https://doi.org/10.3390/molecules25020347






