A Liposomal Formulation for Improving Solubility and Oral Bioavailability of Nifedipine
Abstract
1. Introduction
2. Results and Discussion
2.1. Preparation of Nifedipine Proliposomes
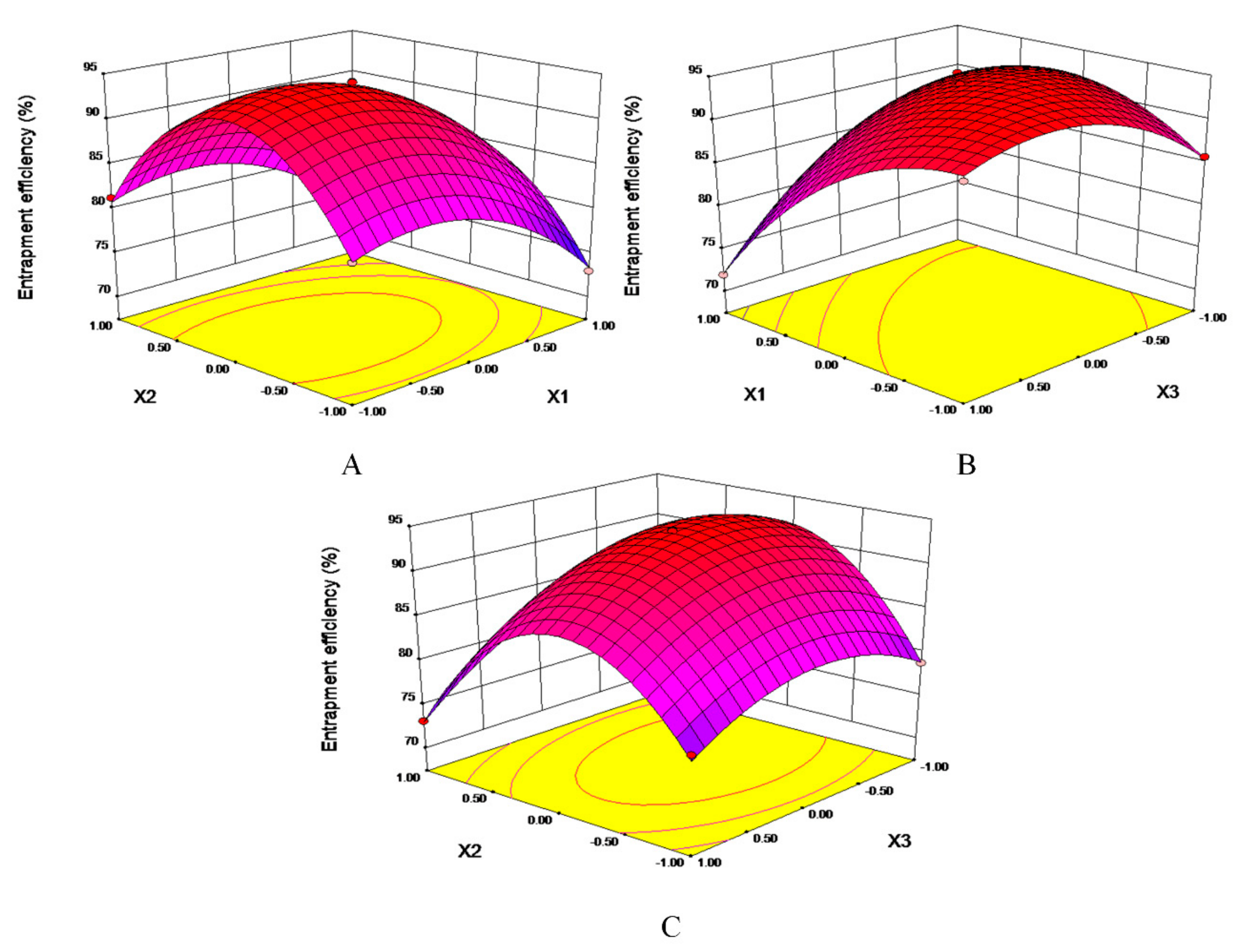
2.2. Optimization of Proliposomes Formulation
1.865 X2X3 − 4.92625 X12 − 11.52125 X22 − 4.47375 X32
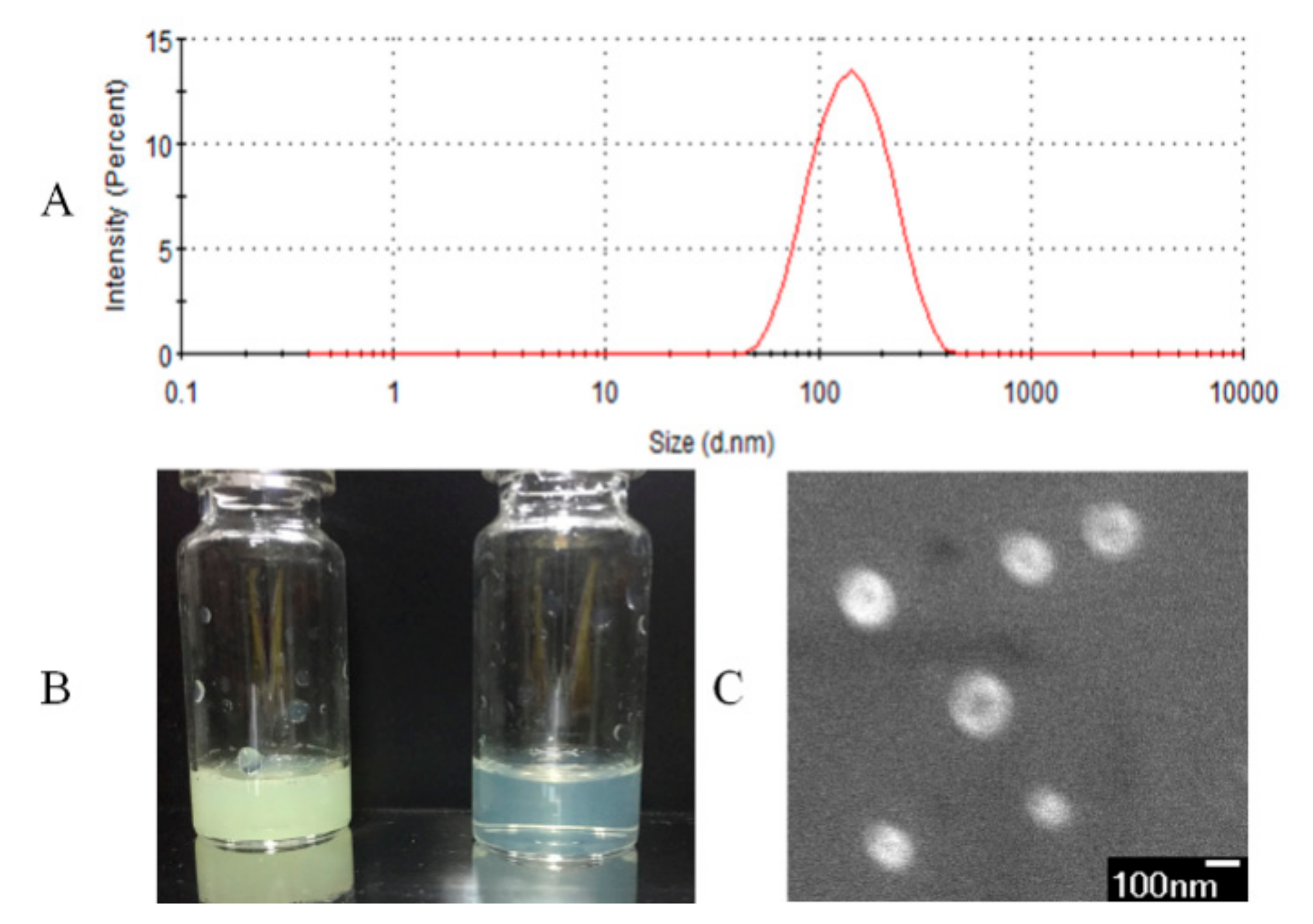
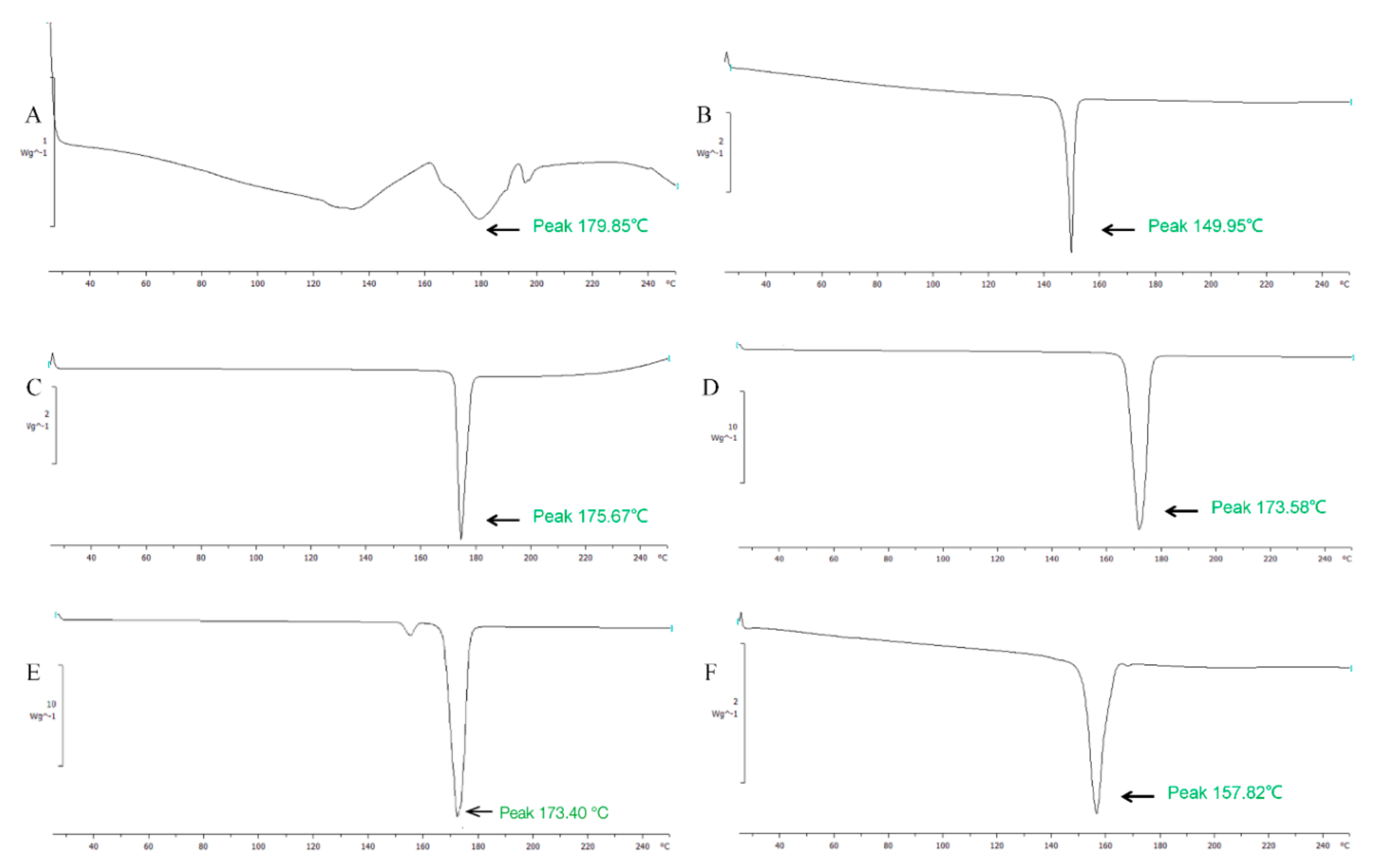
2.3. Characterization of Liposomes
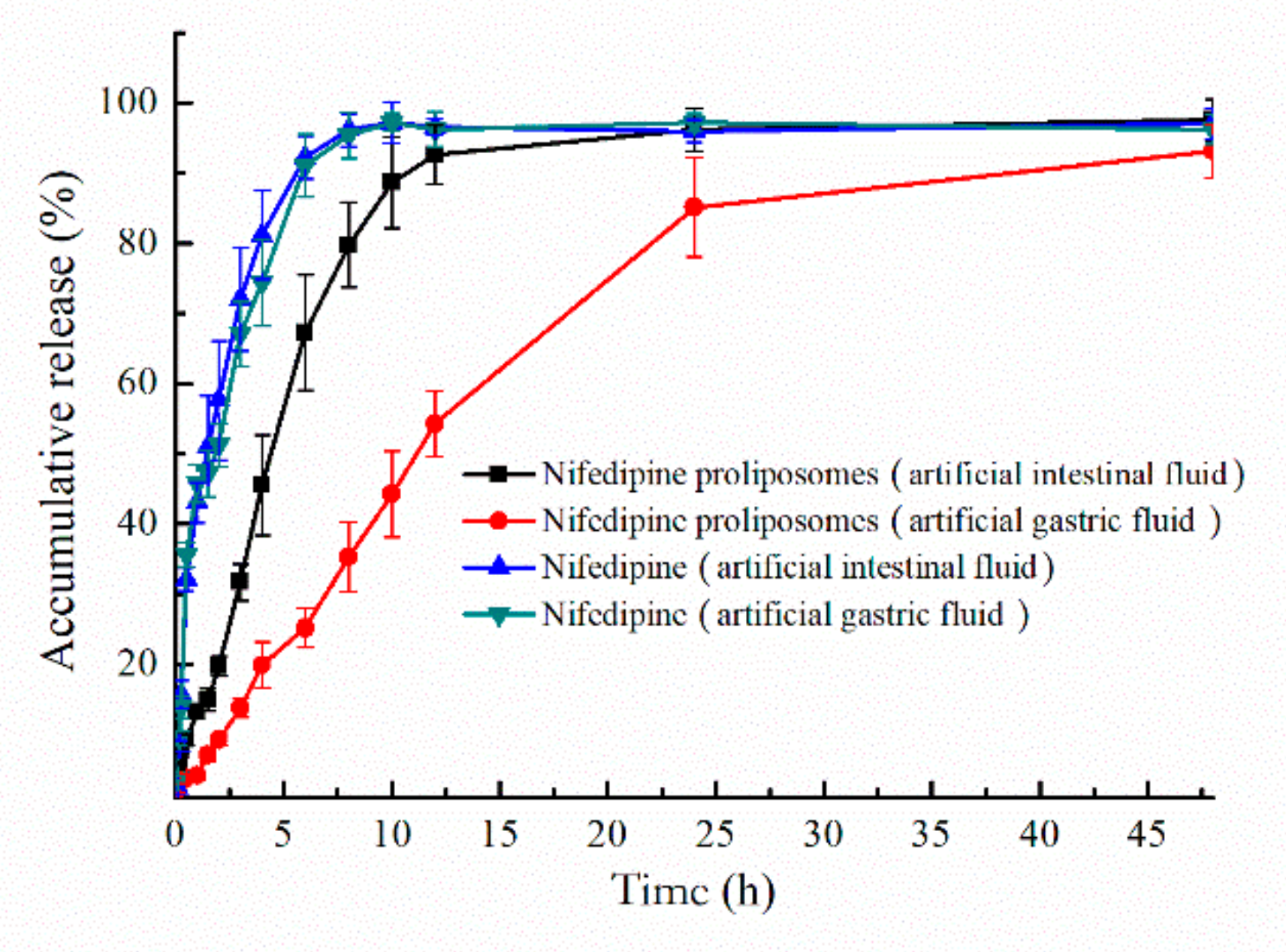
2.4. Drug Release Profiles
2.5. Nifedipine Analysis Method In Vivo
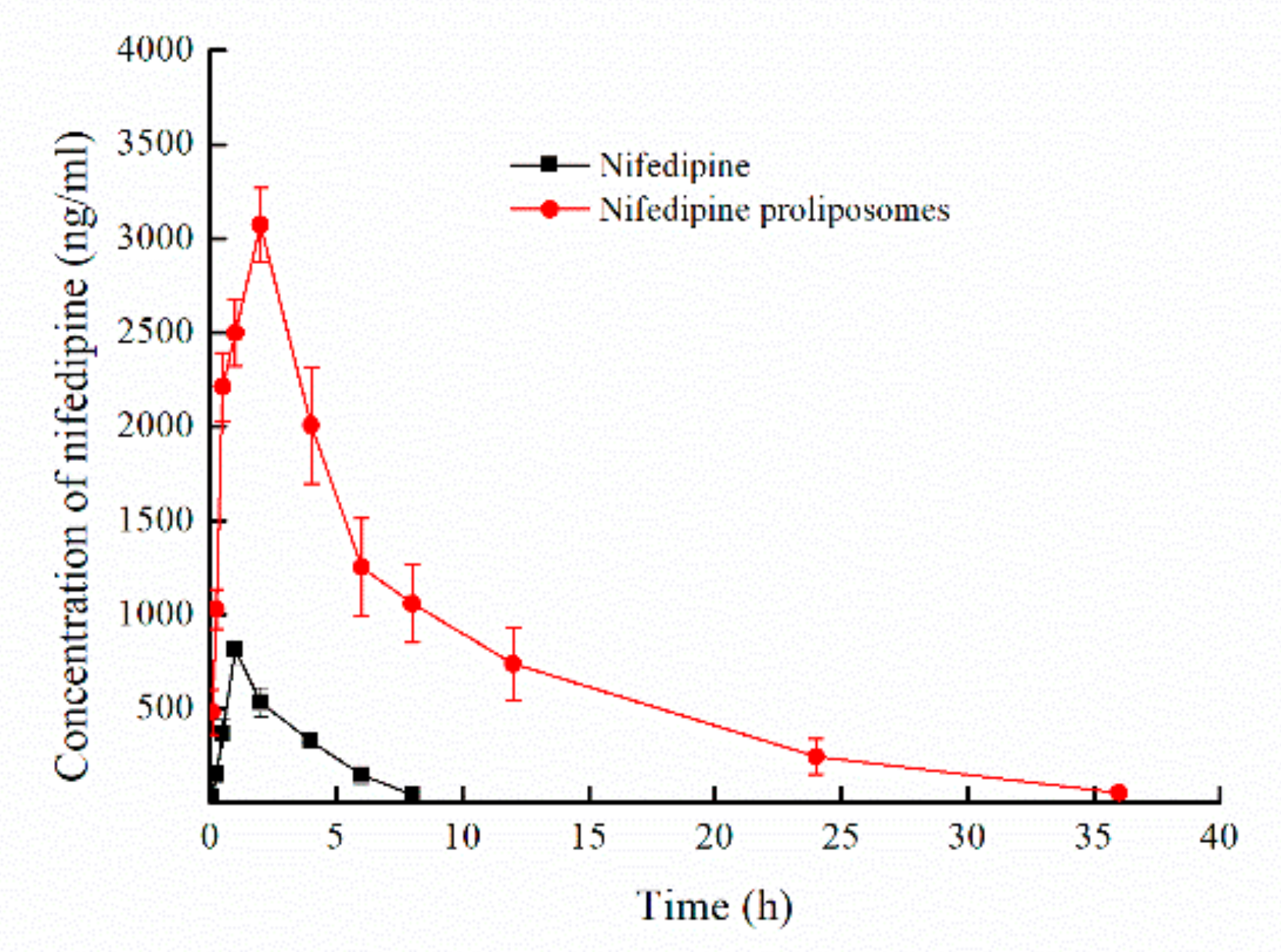
2.6. Pharmacokinetics Study
3. Materials and Methods
3.1. Materials and Animals
3.2. Preparation Proliposomes
3.3. Optimization of Proliposomes Formulation
3.4. Characterization of Proliposomes
3.5. Drug Release Profiles
3.6. Pharmacokinetic Study
3.7. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mantas, A.; Mihranyan, A. Immediate-Release Nifedipine Binary Dry Powder Mixtures with Nanocellulose Featuring Enhanced Solubility and Dissolution Rate. Pharmaceutics 2019, 11, 37. [Google Scholar] [CrossRef]
- Boje, K.M.; Sak, M.; Fung, H.L. Complexation of nifedipine with substituted phenolic ligands. Pharm. Res. 1988, 5, 655–659. [Google Scholar] [CrossRef]
- Weerapol, Y.; Limmatvapirat, S.; Kumpugdee-Vollrath, M.; Sriamornsak, P. Spontaneous emulsification of nifedipine-loaded self-nanoemulsifying drug delivery system. AAPS PharmSciTech 2015, 16, 435–443. [Google Scholar] [CrossRef][Green Version]
- Hecq, J.; Deleers, M.; Fanara, D.; Vranckx, H.; Amighi, K. Preparation and characterization of nanocrystals for solubility and dissolution rate enhancement of nifedipine. Int. J. Pharm. 2005, 299, 167–177. [Google Scholar] [CrossRef]
- Ohshima, H.; Miyagishima, A.; Kurita, T.; Makino, Y.; Iwao, Y.; Sonobe, T.; Itai, S. Freeze-dried nifedipine-lipid nanoparticles with long-term nano-dispersion stability after reconstitution. Int. J. Pharm. 2009, 377, 180–184. [Google Scholar] [CrossRef]
- Ma, J.; Wang, J.; Cheng, Z.; Yin, T.; Teng, H.; Xu, H.; Tang, X.; Cai, C. Sustained-release pellets of nifedipine using microcrystals combined with MCC-based matrix. Drug Dev. Ind. Pharm. 2015, 41, 307–314. [Google Scholar] [CrossRef]
- Baxevanis, F.; Kuiper, J.; Fotaki, N. Impact of presence of excipients in drug analysis in fed-state gastric biorelevant media. Eur. J. Pharm. Biopharm. 2018, 131, 178–188. [Google Scholar] [CrossRef]
- Matsuo, S.; Higashi, K.; Moribe, K. Combination of Roll Grinding and High-Pressure Homogenization Can Prepare Stable Bicelles for Drug Delivery. Nanomaterials 2018, 8, 998. [Google Scholar] [CrossRef]
- Cilurzo, F.; Selmin, F.; Minghetti, P.; Gennari, C.G.; Demartin, F.; Montanari, L. Characterization and physical stability of fast-dissolving microparticles containing nifedipine. Eur. J. Pharm. Biopharm. 2008, 68, 579–588. [Google Scholar] [CrossRef]
- Avila-Salas, F.; Rodriguez Nunez, Y.A.; Marican, A.; Castro, R.I.; Villasenor, J.; Santos, L.S.; Wehinger, S.; Duran-Lara, E.F. Rational Development of a Novel Hydrogel as a pH-Sensitive Controlled Release System for Nifedipine. Polymers 2018, 10, 806. [Google Scholar] [CrossRef]
- Crum, M.; Elkordy, A.A.; Zarara, M.; Elkordy, E.A. In situ lyophilisation of nifedipine directly in hard gelatine capsules. Pharm. Dev. Technol. 2013, 18, 1379–1390. [Google Scholar] [CrossRef]
- Li, D.X.; Kim, J.O.; Oh, D.H.; Lee, W.S.; Hong, M.J.; Kang, J.Y.; Choi, J.S.; Woo, J.S.; Yong, C.S.; Choi, H.G. Development of nifedipine-loaded coated gelatin microcapsule as a long acting oral delivery. Arch. Pharm. Res. 2009, 32, 127–132. [Google Scholar] [CrossRef]
- Secolin, V.A.; Souza, C.R.; Oliveira, W.P. Spray drying of lipid-based systems loaded with Camellia sinensis polyphenols. J. Liposome Res. 2017, 27, 11–20. [Google Scholar] [CrossRef]
- Fong, S.Y.; Brandl, M.; Bauer-Brandl, A. Phospholipid-based solid drug formulations for oral bioavailability enhancement: A meta-analysis. Eur. J. Pharm. Sci. 2015, 80, 89–110. [Google Scholar] [CrossRef]
- Soulairol, I.; Tarlier, N.; Bataille, B.; Cacciaguerra, T.; Sharkawi, T. Spray-dried solid dispersions of nifedipine and vinylcaprolactam/vinylacetate/PEG(6)(0)(0)(0) for compacted oral formulations. Int. J. Pharm. 2015, 481, 140–147. [Google Scholar] [CrossRef]
- Tang, Y.; Zhang, H.; Lu, X.; Jiang, L.; Xi, X.; Liu, J.; Zhu, J. Development and evaluation of a dry powder formulation of liposome-encapsulated oseltamivir phosphate for inhalation. Drug Deliv. 2015, 22, 608–618. [Google Scholar] [CrossRef]
- Wang, Y.; Kho, K.; Cheow, W.S.; Hadinoto, K. A comparison between spray drying and spray freeze drying for dry powder inhaler formulation of drug-loaded lipid-polymer hybrid nanoparticles. Int. J. Pharm. 2012, 424, 98–106. [Google Scholar] [CrossRef]
- Naghibi Beidokhti, H.R.; Ghaffarzadegan, R.; Mirzakhanlouei, S.; Ghazizadeh, L.; Dorkoosh, F.A. Preparation, Characterization, and Optimization of Folic Acid-Chitosan-Methotrexate Core-Shell Nanoparticles by Box-Behnken Design for Tumor-Targeted Drug Delivery. AAPS PharmSciTech 2017, 18, 115–129. [Google Scholar] [CrossRef]
- Funakoshi, Y.; Iwao, Y.; Noguchi, S.; Itai, S. Lipid nanoparticles with no surfactant improve oral absorption rate of poorly water-soluble drug. Int. J. Pharm. 2013, 451, 92–94. [Google Scholar] [CrossRef]
- Elgart, A.; Cherniakov, I.; Aldouby, Y.; Domb, A.J.; Hoffman, A. Lipospheres and pro-nano lipospheres for delivery of poorly water soluble compounds. Chem. Phys. Lipids 2012, 165, 438–453. [Google Scholar] [CrossRef]
- Granada, A.; Tagliari, M.P.; Soldi, V.; Silva, M.A.; Zanetti-Ramos, B.G.; Fernandes, D.; Stulzer, H.K. Nifedipine-loaded polymeric nanocapsules: Validation of a stability-indicating HPLC method to evaluate the drug entrapment efficiency and in vitro release profiles. J. AOAC Int. 2013, 96, 276–281. [Google Scholar] [CrossRef]
- Li, P.; Dai, Y.N.; Zhang, J.P.; Wang, A.Q.; Wei, Q. Chitosan-alginate nanoparticles as a novel drug delivery system for nifedipine. Int. J. Biomed. Sci. IJBS 2008, 4, 221–228. [Google Scholar]
- Hecq, J.; Nollevaux, G.; Deleers, M.; Fanara, D.; Vranckx, H. Nifedipine nanocrystals: Pharmacokinetic evaluation in the rat and permeability studies in Caco-2/HT29-5M21 (co)-cultures. J. Drug Deliv. Sci. Technol. 2006, 16, 437–442. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
| Run | Levels of Independent Factors | Response EE% | ||
|---|---|---|---|---|
| X1 | X2 | X3 | ||
| 1 | −1 | −1 | 0 | 82.06 |
| 2 | 1 | −1 | 0 | 72.95 |
| 3 | −1 | 1 | 0 | 81.37 |
| 4 | 1 | 1 | 0 | 73.27 |
| 5 | −1 | 0 | −1 | 85.79 |
| 6 | 1 | 0 | −1 | 89.12 |
| 7 | −1 | 0 | 1 | 91.02 |
| 8 | 1 | 0 | 1 | 71.93 |
| 9 | 0 | −1 | −1 | 78.85 |
| 10 | 0 | 1 | −1 | 81.36 |
| 11 | 0 | −1 | 1 | 78.15 |
| 12 | 0 | 1 | 1 | 73.15 |
| 13 | 0 | 0 | 0 | 94.12 |
| 14 | 0 | 0 | 0 | 93.41 |
| 15 | 0 | 0 | 0 | 94.05 |
| Source | df | Sum of Squares | Mean Square | F Value | p-Value Prob > F | |
|---|---|---|---|---|---|---|
| Model | 9 | 922.61 | 102.51 | 232.19 | <0.0001 | Significant |
| X1 | 1 | 136.04 | 136.04 | 308.14 | <0.0001 | |
| X2 | 1 | 0.99 | 0.99 | 2.24 | 0.1951 | |
| X3 | 1 | 54.6 | 54.6 | 123.67 | 0.0001 | |
| X1X2 | 1 | 0.26 | 0.26 | 0.58 | 0.4815 | |
| X1X3 | 1 | 125.44 | 125.44 | 284.13 | <0.0001 | |
| X2X3 | 1 | 13.91 | 13.91 | 31.51 | 0.0025 | |
| X12 | 1 | 89.6 | 89.6 | 202.96 | <0.0001 | |
| X22 | 1 | 490.11 | 490.11 | 1110.12 | <0.0001 | |
| X32 | 1 | 73.90 | 73.90 | 167.38 | <0.0001 | |
| Residual | 5 | 2.21 | 0.44 | |||
| Lack of fit | 3 | 1.90 | 0.63 | 4.14 | 0.2007 | No Significant |
| Pure error | 2 | 0.31 | 0.15 | |||
| Coe. total | 14 | 924.81 |
| Time | Medium | Regression Coefficient (R2) | ||||
|---|---|---|---|---|---|---|
| Zero-Order | First-Order | Higuchi | Korsmeyer–Peppas | |||
| Nifedipine | 0–48 h | artificial intestinal fluid | −1.165 | 0.985 | 0.340 | 0.790 |
| 0–48 h | artificial gastric fluid | −1.116 | 0.966 | 0.378 | 0.806 | |
| Proliposomes | 0–48 h | artificial intestinal fluid | 0.149 | 0.980 | 0.799 | 0.827 |
| 0–48 h | artificial gastric fluid | 0.774 | 0.988 | 0.924 | 0.943 | |
| LQC (75 ng/mL) | MQC (500 ng/mL) | HQC (2000 ng/mL) | IS (2000 ng/mL) | |
|---|---|---|---|---|
| Intra-day precision (RSD%) (n = 6) | 8.55 | 5.81 | 10.87 | |
| Inter-day precision (RSD%) (n = 6) | 8.70 | 5.81 | 7.12 | |
| Accuracy (RE%) (n = 6) | 9.26 | 9.44 | 5.41 | |
| Recovery/RSD% (n = 6) | 83.37/2.67 | 83.90/2.86 | 83.11/2.73 | 88.59/4.11 |
| Matrix effect/RSD% (n = 5) | 87.40/4.50 | 87.85/3.99 | 88.76/2.38 | 91.05/2.98 |
| Parameters | Nifedipine | Nifedipine Proliposomes |
|---|---|---|
| Cmax (ng/mL) | 818.20 ± 83.19 | 3074.20 ± 196.77 ** |
| Tmax (h) | 1.00 | 2.00 |
| T1/2β | 1.57 ± 0.18 | 6.61 ± 0.49 ** |
| AUC0–∞ (ng × h/mL) | 2673.97 ± 175.06 | 26970.80 ± 4650.71 ** |
| MRT | 3.0 ± 0.12 | 8.95 ± 0.90 ** |
| V (mL) | 1522.41 ± 223.56 | 646.32 ± 93.63 ** |
| CL (mL/h) | 671.89 ± 38.24 | 68.12 ± 10.96 ** |
| Factors | Range and Level | ||
|---|---|---|---|
| −1 | 0 | 1 | |
| X1 (the mass ratio of drug-to-SPC) | 0.5:1 | 1:1 | 1.5:1 |
| X2 (the mass ratio of SPC-to-Chol) | 2:1 | 4:1 | 6:1 |
| X3 (the concentration of SPC) | 12 mg/mL | 14 mg/mL | 16 mg/mL |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bi, Y.; Lv, B.; Li, L.; Lee, R.J.; Xie, J.; Qiu, Z.; Teng, L. A Liposomal Formulation for Improving Solubility and Oral Bioavailability of Nifedipine. Molecules 2020, 25, 338. https://doi.org/10.3390/molecules25020338
Bi Y, Lv B, Li L, Lee RJ, Xie J, Qiu Z, Teng L. A Liposomal Formulation for Improving Solubility and Oral Bioavailability of Nifedipine. Molecules. 2020; 25(2):338. https://doi.org/10.3390/molecules25020338
Chicago/Turabian StyleBi, Ye, Bingcong Lv, Lianlian Li, Robert J. Lee, Jing Xie, Zhidong Qiu, and Lesheng Teng. 2020. "A Liposomal Formulation for Improving Solubility and Oral Bioavailability of Nifedipine" Molecules 25, no. 2: 338. https://doi.org/10.3390/molecules25020338
APA StyleBi, Y., Lv, B., Li, L., Lee, R. J., Xie, J., Qiu, Z., & Teng, L. (2020). A Liposomal Formulation for Improving Solubility and Oral Bioavailability of Nifedipine. Molecules, 25(2), 338. https://doi.org/10.3390/molecules25020338







