Physiological and Oxidative Stress Responses of Lettuce to Cleomside A: A Thiohydroximate, as a New Allelochemical from Cleome arabica L.
Abstract
1. Introduction
2. Results and Discussion
2.1. Structural Elucidation of Secondary Metabolites
2.2. Phytotoxicity Assays
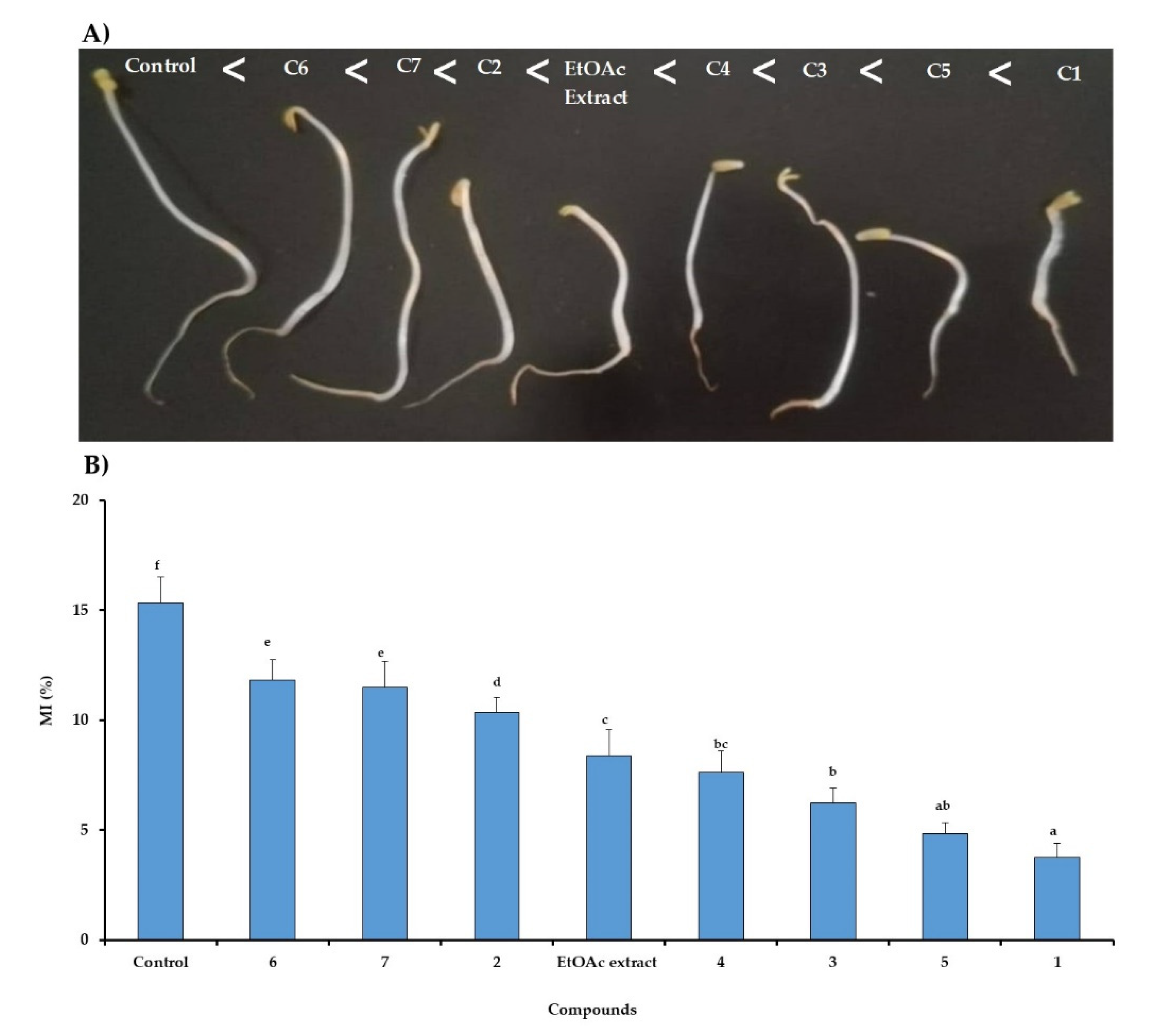
2.3. Mitotic Index in Lettuce Roots
2.4. Oxidative Stress Markers
2.4.1. Membrane Integrity
2.4.2. Hydrogen Peroxide (H2O2) Content
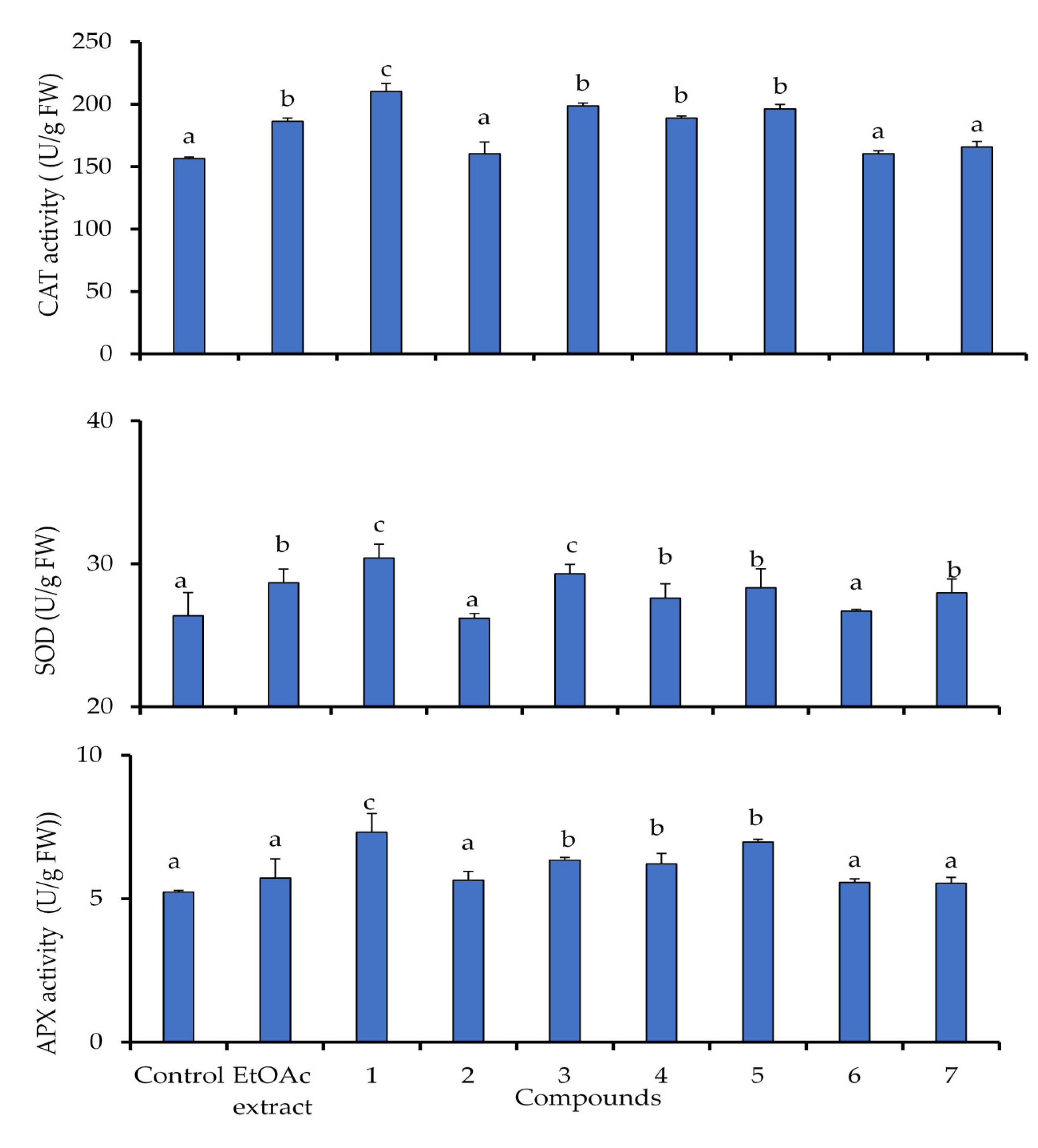
2.5. Activation of Antioxidant Defensive Enzymes in Lettuce
2.6. Correlation between Phytotoxic Potential and Antioxidant Parameters
2.7. Cluster and Principal Compound Analysis
3. Materials and Methods
3.1. Plant Material
3.2. Isolation and Identification of Bioactive Compound
3.2.1. General Experimental Procedures
3.2.2. Extraction and Isolation
3.3. Phytotoxic Assays
3.4. Cytogenotoxicity Test
3.5. Oxidative Stress Response of Lettuce to C. arabica Extract and Its Identified Active Compounds
3.5.1. Determination of Electrolyte Leakage (EL)
3.5.2. Lipid Peroxidation
3.5.3. Determination of Hydrogen Peroxide (H2O2) Content
3.6. Antioxidant Enzyme Activities
3.7. Statistical Analyses
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Van Evert, F.K.; Fountas, S.; Jakovetic, D.; Crnojevic, V.; Travlos, I.; Kempenaar, C. Big data for weed control and crop protection. Weed Res. 2017, 57, 218–233. [Google Scholar] [CrossRef]
- Dayan, F.E.; Duke, S.O. Discovery for new herbicide sites of action by quantification of plant primary metabolite and enzyme pools. Engineering 2020, 6, 509–514. [Google Scholar] [CrossRef]
- Ladhari, A.; Omezzine, F.; DellaGreca, M.; Zarrelli, A.; Zuppolini, S.; Haouala, R. Phytotoxic activity of Cleome arabica L. and its principal discovered active compounds. S. Afr. J. Bot. 2013, 88, 341–351. [Google Scholar] [CrossRef]
- Macías, F.A.; Oliveros-Bastidas, A.; Marín, D.; Chinchilla, N.; Castellano, D.; Molinillo, J.M.G. Evidence for an allelopathic interaction between rye and wild oats. J. Agric. Food Chem. 2014, 62, 9450–9457. [Google Scholar] [CrossRef] [PubMed]
- Takhi, D.; Ouinten, M.; Yousfi, M. Study of antimicrobial activity of secondary metabolites extracted from spontaneous plants from the area of Laghouat, Algeria. Adv. Environ. Biol. 2011, 5, 469–476. [Google Scholar]
- Khalafallah, A.K.; Mohamed, A.H.; Yousof, A.H.; Hussien, T.A.; Mohamed-Elamir, F.H.; Shinji, O. Damarane triterpene from Cleome arabica. Pharmacogn. Res. 2009, 1, 162–165. [Google Scholar]
- Ismail, S.I.; Ito, H.; Selloum, L.; Bouriche, H.; Yoshida, T. Constituents. of Cleome arabica leaves and twigs. Nat. Med. 2005, 59, 53. [Google Scholar]
- Djeridane, A.; Yousfi, M.; Brunel, J.M.; Stocker, P. Isolation and characterization of a new steroid derivative as a powerful antioxidant from Cleome arabica in screening the in vitro antioxidant capacity of 18 Algerian medicinal plants. Food Chem. Toxicol. 2010, 48, 2599–2606. [Google Scholar] [CrossRef]
- Bouriche, H.; Arnhold, J. Effect of Cleome arabica leaf extract treated by naringinase on human neutrophil chemotaxis. Nat. Prod. Commun. 2010, 5, 415–418. [Google Scholar] [CrossRef]
- McDanell, R.; McLean, A.E.M.; Hanley, A.B.; Heaney, R.K.; Fenwick, G.R. Chemical and biological properties of indole glucosinolates (glucobrassicins): A review. Food Chem. Toxicol. 1988, 26, 59–70. [Google Scholar] [CrossRef]
- Agerbirk, N.; Petersen, B.L.; Olsen, C.E.; Halkier, B.A.; Nielsen, J.N.J. 1,4-Dimethoxyglucobrassicin in Barbarea and 4-hydroxyglucobrassicin in Arabidopsis and Brassica. Agric. Food Chem. 2001, 49, 1502–1507. [Google Scholar] [CrossRef]
- Das, S.; Tyagi, A.K.; Singhal, K.K. Effect of Glucosinolates of Taramira (Eruca Sativa) oil cake on nutrient utilization and growth of crossbred calves. Asian-Aust. J. Anim. Sci. 2003, 16, 813–817. [Google Scholar] [CrossRef]
- Fahey, J.W.; Zalcmann, A.T.; Talalay, P. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 2001, 56, 5–51. [Google Scholar] [CrossRef]
- Ladhari, A.; Omezzine, F.; Haouala, R. The impact of Tunisian Capparidaceae species on cytological, physiological and biochemical mechanisms in lettuce. S. Afr. J. Bot. 2014, 93, 222–230. [Google Scholar] [CrossRef]
- Romero-Romero, T.; Sanchez-Nieto, S.; Sanjuan-Badillo, A.; Amaua, A.L.; Cruz-Ortega, R. Comparative effects of allelochemical and water stress in roots of Lycopersicon esculentum Mill. (Solanaceae). Plant Sci. 2005, 168, 1059–1066. [Google Scholar] [CrossRef]
- DellaGreca, M.; Mancino, A.; Previtera, L.; Zarrelli, A.; Zuppolini, S. Lignans from Phillyrea angustifolia L. Phytochem. Lett. 2011, 4, 118–121. [Google Scholar] [CrossRef]
- Ribeiro, A.; Pilo-Veloso, D. Trypanocidal flavanoids from Trixis vauthieri. J. Nat. Prod. 1997, 60, 836–838. [Google Scholar] [CrossRef]
- Sharaf, M.; El-Ansari, M.A.; Saleh, N.A.M. Flavonoids of four cleome and three capparis species. Biochem. Syst. Ecol. 1997, 25, 161–166. [Google Scholar] [CrossRef]
- Sekine, K.; Hase, T.; Sato, N. Reversible DNA compaction by sulfite reductase regulates transcriptional activity of chloroplast nucleoids. J. Biol. Chem. 2002, 277, 24399–24404. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E. Plant Physiology, 5th ed.; Sinauer Associates: Sunderland, UK, 2010; p. 782. [Google Scholar]
- Grubb, C.D.; Abel, S. Glucosinolate metabolism and its control. Trends Plant Sci. 2006, 11, 89–100. [Google Scholar] [CrossRef]
- Moreland, D.E.; Novitski, W.P. Interference by flavone and flavonols with chloroplast-mediated electron transport and phosphorylation. Phytochemistry 1988, 27, 3359–3365. [Google Scholar] [CrossRef]
- Stenlid, G. Flavonoids as inhibitors of the formation of adenosine triphosphate in plant mitochondria. Phytochemistry 1970, 9, 2251–2256. [Google Scholar] [CrossRef]
- Blum, U. Plant–Plant Allelopathic Interactions: Partitioning and Seedling Effects of Phenolic Acids as Related to Their Physicochemical and Conditional Properties, 1st ed.; Springer: London, UK, 2014. [Google Scholar] [CrossRef]
- Rojas, E.; Herrera, L.A.; Sordo, M.; Gonsebatt, M.E.; Montero, R.; Rodriguez, R.; Ostrosky-Wegman, P. Mitotic index and cell proliferation kinetics for identification of antineoplastic activity. Anti-Cancer Drugs 1993, 4, 637–640. [Google Scholar] [CrossRef] [PubMed]
- Hallak, A.M.G.; Davide, L.C.; Souza, I.F. Effects of sorghum (Sorghum bicolor L.) root exudates on the cell cycle of the bean plant (Phaseolus vulgaris L.) root. Genet. Mol. Biol. 1999, 22, 95–99. [Google Scholar] [CrossRef]
- Sánchez-Moreiras, A.M.; De La Pena, T.C.; Reigosa, M.J. The natural compound benzoxazolin-2(3H)-one selectively retards cell cycle in lettuce root meristems. Phytochemistry 2008, 69, 2172–2179. [Google Scholar] [CrossRef]
- Nishida, N.; Tamotsu, S.; Nagata, N.; Saito, C.; Sakai, A. Allelopathic effects of volatile monoterpenoids produced by Salvia leucophylla: Inhibition of cell proliferation and DNA synthesis in the root apical meristem of Brassica campestris seedlings. J. Chem. Ecol. 2005, 31, 1187–1203. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, R.C.; Feitoza, R.B.B.; Lima, H.R.P.; De Carvalho, G.M. Phytotoxic effects of phenolic compounds on Calopogonium mucunoides (Fabaceae) roots. Aust. J. Bot. 2015, 63, 679–686. [Google Scholar] [CrossRef]
- Maness, P.C.; Smolinski, S.; Blake, D.M.; Huang, Z.; Wolfrum, E.J.; Jacoby, W.A. Bactericidal activity of photocatalytic TiO2 reaction: Toward an understanding of its killing mechanism. Appl. Environ. Microbiol. 1999, 65, 4094–4098. [Google Scholar] [CrossRef] [PubMed]
- Weir, T.L.; Park, S.W.; Vivanco, M. Biochemical and physiological mechanisms mediated by allelochemicals. Curr. Opin. Plant Biol. 2004, 7, 472–479. [Google Scholar] [CrossRef]
- Harminder, P.; Batish, D.R.; Kaur, S.; Arora, K.; Kohli, R.K. α-Pinene inhibits growth and induces oxidative stress in roots. Ann. Bot. 2006, 98, 1261–1269. [Google Scholar] [CrossRef]
- Yu, J.Q.; Ye, S.F.; Zhang, M.F.; Hu, W.H. Effects of root exudates, aqueous root extracts of cucumber (Cucumis sativus L.) and allelochemicals on photosynthesis and antioxidant enzymes in cucumber. Biochem. Syst. Ecol. 2003, 31, 129–139. [Google Scholar] [CrossRef]
- Ye, S.F.; Zhou, Y.H.; Sun, Y.; Zou, L.Y.; Yu, J.Q. Cinnamic acid causes oxidative stress in cucumber roots, and promotes incidence of Fusarium wilt. Environ. Exp. Bot. 2006, 56, 255–262. [Google Scholar] [CrossRef]
- Inderjit; Duke, S.O. Ecophysiological aspects of allelopathy. Planta 2003, 217, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Tigre, R.C.; Pereira, E.C.; da Silva, N.H.; Vicente, C.; Legaz, M.E. Potential phenolic bioherbicides from Cladonia verticillaris produce ultrastructural changes in Lactuca sativa seedlings. S. Afr. J. Bot. 2015, 98, 16–25. [Google Scholar] [CrossRef]
- Liu, M.; Lu, S. Plastoquinone and ubiquinone in plants: Biosynthesis, physiological function and metabolic engineering. Front. Plant Sci. 2016, 7, 1–18. [Google Scholar] [CrossRef]
- Macias, F.A.; Varela, R.M.; Torres, A.; Galindo, J.L.G.; Molinillo, J.M.G. Allelochemicals from sunflowers: Chemistry, bioactivity and applications. In Chemical Ecology of Plants: Allelopathy in Aquatic and Terrestrial Ecosystems, 1st ed.; Inderjit, Mallik, A.U., Eds.; Springer: Basel, Switzerland, 2002. [Google Scholar] [CrossRef]
- Oracz, K.; Bailly, C.; Gniazdowska, A.; Come, D.; Corbineau, F.; Bogatek, R. Induction of oxidative stress by sunflower phytotoxins in germinating mustard seeds. J. Chem. Ecol. 2007, 33, 251–264. [Google Scholar] [CrossRef]
- Farhoudi, R.; Lee, D.J. Allelopathic effects of barley (Hordeum vulgare) on sucrose synthase activity, lipid peroxidation and antioxidant enzymatic activities of Hordeum spontoneum and Avena ludoviciana. Proc. Natl. Acad. Sci. India B. 2013, 83, 447–452. [Google Scholar] [CrossRef]
- Talukdar, D. Allelopathic effects of Lantana camara L. on Lathyrus sativus L.: Oxidative imbalance and cytogenetic consequences. Allelopath. J. 2013, 31, 71–90. [Google Scholar]
- Devi, S.R.; Prasad, M.N.V. Ferulic acid mediated changes in oxidative enzymes of maize seedlings: Implications in growth. Biol. Plant. 1996, 38, 387–395. [Google Scholar] [CrossRef]
- Maffei, M.; Bertea, C.M.; Garneri, F.; Scannerini, S. Effect of benzoic acid hydroxy and methoxy ring substituents during cucumber (Cucumis sativus L.) germination. I. Isocitrate lyase and catalase activity. Plant Sci. 1999, 141, 139–147. [Google Scholar] [CrossRef]
- Zeng, R.S.; Luo, S.M.; Shi, Y.H.; Shi, M.B.; Tu, C.Y. Physiological and biochemical mechanism of allelopathy of secalonic acid F on higher plants. Agron. J. 2001, 93, 72–79. [Google Scholar] [CrossRef]
- Sánchez-Moreiras, A.M.; Reigosa, M.J. Whole plant response of lettuce after root exposure to BOA (2(3H)benzoxazolinone). J. Chem. Ecol. 2005, 31, 2689–2703. [Google Scholar] [CrossRef] [PubMed]
- Vainola, A.; Repo, T. Impedance spectroscopy in frost hardiness evaluation of Rhododendron leaves. Ann. Bot. 2000, 86, 799–805. [Google Scholar] [CrossRef]
- Pottier-Alapetite, G. Angiospermes-Dicotyledones, Apétales-Dialypétales; Flore de la Tunisie: Tunis, Tunisie, 1979. [Google Scholar]
- Chiapuso, G.; Sanchez, A.M.; Reigosa, M.J.; Gonzaiez, L.; Pellissier, F. Do germination indices adequately reflect allelochemical effects on the germination process? J. Chem. Ecol. 1997, 23, 2445–2453. [Google Scholar] [CrossRef]
- Chung, I.M.; Ahn, J.K.; Yun, S.J. Assessment of allelopathic potential of barnyardgrass (Echinochloa crus-galli) on rice (Oryza sativa L.) cultivars. Crop Prot. 2001, 20, 921–928. [Google Scholar] [CrossRef]
- Koodkaew, I.; Sunohara, Y.; Matsuyama, S.; Matsumoto, H. Phytotoxic action mechanism of hapalocyclamide in lettuce seedlings. Plant Physiol. Biochem. 2012, 58, 23–28. [Google Scholar] [CrossRef]
- Shalata, A.; Neumann, P.M. Exogenous ascorbic acid (vitamin C) increases resistance to salt stress and reduces lipid peroxidation. J. Exp. Bot. 2001, 52, 2207–2211. [Google Scholar] [CrossRef]
- Alia, K.V.; Prasad, S.K.; Pardha, S.P. Effect of zinc on free radicals and proline in Brassica and Cajanus. Phytochemistry 1995, 42, 45–47. [Google Scholar] [CrossRef]
- Velikova, V.; Yordanov, I.; Edreva, A. Oxidative stress and some antioxidant systems in acid rain-treated bean plants: Protective role of exogenous polyamines. Plant Sci. 2000, 151, 59–66. [Google Scholar] [CrossRef]
- Giannopolitis, C.N.; Ries, S.K. Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol. 1977, 59, 309–314. [Google Scholar] [CrossRef]
- Cakmak, I.; Marschner, H. Magnesium deficiency and high light intensity enhance activities of superoxide dismutase, ascorbate peroxidase and glutathione reductase in bean leaves. Plant Physiol. 1992, 98, 1222–1227. [Google Scholar] [CrossRef] [PubMed]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 1981, 22, 867–888. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
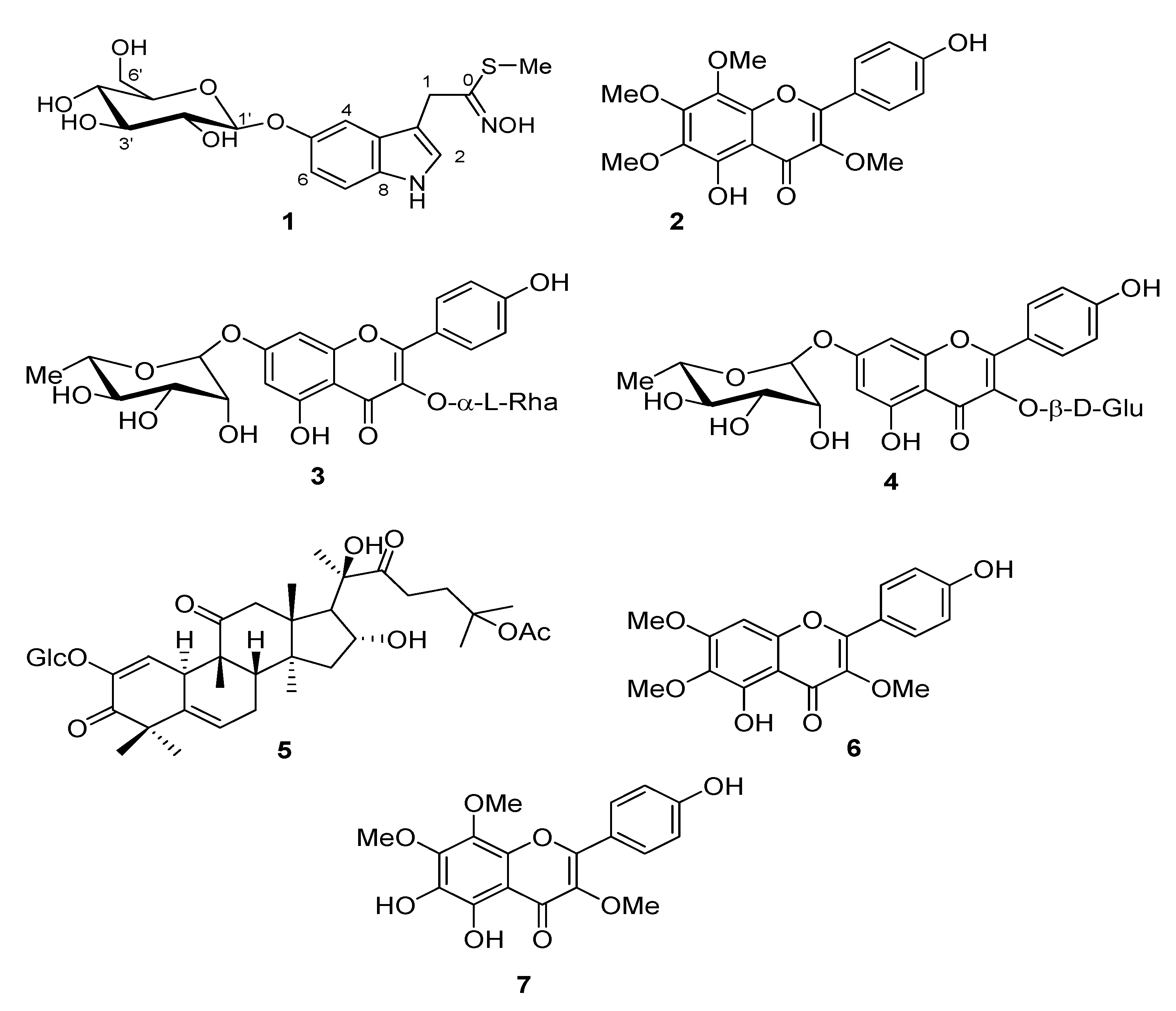




| No. | 1 | ||
|---|---|---|---|
| δC (mult.) | δH a | HMBC Correlations | |
| SMe | 12.6 (q) | 2.24 s | 0 |
| 0 | 171.0 (s) | ||
| 1 | 39.0 (t) | 4.78, d (14.4) 4.50, d (14.4) | 0, 2, 3, 9 |
| 2 | 124.7 (d) | 7.10, s | 1, 3, 8, 9 |
| 3 | 113.5 (s) | ||
| 4 | 107.8 (d) | 7.02 (overlapped) | 5, 8, 9 |
| 5 | 153.5 (s) | ||
| 6 | 104.9 (d) | 6.80, br d (7.1) | 4, 7, 8 |
| 7 | 123.9 (d) | 7.02 (overlapped) | 5, 8 |
| 8 | 140.5 (s) | ||
| 9 | 119.0 (s) | ||
| 1′ | 102.5 (d) | 5.10, d (7.1) | 5, 3′ |
| 2′ | 75.8 (d) | 3.53, m | 4′ |
| 3′ | 78.8 (d) | 3.44, m | 5′ |
| 4′ | 72.0 (d) | 3.42, m | 6′ |
| 5′ | 78.8 (d) | 3.61, m | 1′ |
| 6′ | 63.1 (t) | 3.92, dd (13.5, 10.2) 3.75, dd (12.0, 5.4) | 5′ |
| Concentrations (µg mL−1) | |||||||
|---|---|---|---|---|---|---|---|
| Treatments | 50 | 100 | 200 | 400 | 800 | IC50 | |
| EtOAc Extract | 89.3 ± 3.4 c | 77.6 ± 1.5 c | 70.7 ± 4.9 c | 69.6 ± 1.3 c | 71.9 ± 3.6 b | >800 | |
| Compounds | 1 | 26.3 ± 2.1 a | 25.1 ± 1.7 a | 15.3 ± 0.9 a | 00.0 ± 0.0 a | 00.0 ± 0.0 a | <50 |
| 2 | 93.2 ± 1.4 c | 90.7 ± 5.6 d | 89.6 ± 1.2 d | 91.3 ± 2.6 d | 70.4 ± 5.1 b | >800 | |
| 3 | 86.3 ± 4.3 c | 80.4 ± 4.7 c | 78.7 ± 2.7 c | 66.3 ± 3.6 c | 60.4 ± 3.9 b | >800 | |
| 4 | 93.6 ± 2.9 c | 86.7 ± 4.3 c | 82.9 ± 3.9 d | 80.4 ± 3.4 d | 77.3 ± 5.3 b | >800 | |
| 5 | 65.3 ± 3.4 b | 51.1 ± 3.6 b | 30.4 ± 1.6 b | 25.0 ± 2.3 b | 00.0 ± 0.0 a | 91.4 | |
| 6 | 96.3 ± 4.9 c | 94.2 ± 5.4 d | 94.6 ± 4.3 d | 88.2 ± 3.2 d | 80.4 ± 4.1 b | >800 | |
| 7 | 93.4 ± 3.6 c | 93.7 ± 4.6 d | 91.9 ± 6.4 d | 90.7 ± 5.4 d | 90.7 ± 2.6 c | >800 | |
| EL (%) | MDA (nmol g−1 FW) | H2O2 (nmol g−1 FW) | ||
|---|---|---|---|---|
| Control | 150.6 ± 2.6 b | 50.3 ± 2.3 a | 4.6 ± 1.5 a | |
| EtOAc | 177.3 ± 5.1 b c | 64.2 ± 1.9 b | 12.4 ± 2.1 b | |
| Compounds | 1 | 210.6 ± 3.9 c | 70.2 ± 4.5 c | 15.3 ± 2.6 c |
| 2 | 170.3 ± 1.6 b c | 62.3 ± 2.4 b | 10.2 ± 2.4 b | |
| 3 | 201.6 ± 2.5 c | 71.6 ± 2.1 c | 14.9 ± 3.4 c | |
| 4 | 183.7 ± 2.6 bc | 64.7 ± 3.7 b | 12.3 ± 1.9 b | |
| 5 | 200.9 ± 3.1 c | 72.6 ± 5.9 c | 15.1 ± 0.6 c | |
| 6 | 124.3 ± 0.9 a | 43.1 ± 3.1 a | 5.2 ± 0.5 a | |
| 7 | 154.9 ± 1.3 b | 54.3 ± 7.3 a | 6.1 ± 0.5 a |
| GI | RL | SL | MI | EL | MDA | H2O2 | CAT | SOD | APX | |
|---|---|---|---|---|---|---|---|---|---|---|
| IG | ||||||||||
| RL | 0.920 ** | |||||||||
| SL | 0.929 ** | 0.947 ** | ||||||||
| MI | 0.699 * | 0.924 ** | 0.843 ** | |||||||
| EL | 0.433 | 0.631 | 0.494 | −0.880 ** | ||||||
| MDA | 0.322 | 0.528 | 0.377 | −0.874 ** | 0.978 ** | |||||
| H2O2 | 0.282 | 0.471 | 0.304 | −0.951 ** | 0.947 ** | 0.962 ** | ||||
| CAT | 0.349 | 0.555 | 0.429 | −0.946 ** | 0.900 ** | 0.854 ** | 0.922 ** | |||
| SOD | 0.267 | 0.464 | 0.374 | −0.813 ** | 0.765 * | 0.690 * | 0.747 * | 0.907 ** | ||
| APX | 0.514 | 0.570 | 0.518 | −0.934 ** | 0.842 ** | 0.781 * | 0.847 ** | 0.905 ** | 0.755 * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ladhari, A.; Andolfi, A.; DellaGreca, M. Physiological and Oxidative Stress Responses of Lettuce to Cleomside A: A Thiohydroximate, as a New Allelochemical from Cleome arabica L. Molecules 2020, 25, 4461. https://doi.org/10.3390/molecules25194461
Ladhari A, Andolfi A, DellaGreca M. Physiological and Oxidative Stress Responses of Lettuce to Cleomside A: A Thiohydroximate, as a New Allelochemical from Cleome arabica L. Molecules. 2020; 25(19):4461. https://doi.org/10.3390/molecules25194461
Chicago/Turabian StyleLadhari, Afef, Anna Andolfi, and Marina DellaGreca. 2020. "Physiological and Oxidative Stress Responses of Lettuce to Cleomside A: A Thiohydroximate, as a New Allelochemical from Cleome arabica L." Molecules 25, no. 19: 4461. https://doi.org/10.3390/molecules25194461
APA StyleLadhari, A., Andolfi, A., & DellaGreca, M. (2020). Physiological and Oxidative Stress Responses of Lettuce to Cleomside A: A Thiohydroximate, as a New Allelochemical from Cleome arabica L. Molecules, 25(19), 4461. https://doi.org/10.3390/molecules25194461







