Genome-Based Insights into the Production of Carotenoids by Antarctic Bacteria, Planococcus sp. ANT_H30 and Rhodococcus sp. ANT_H53B
Abstract
1. Introduction
2. Results and Discussion
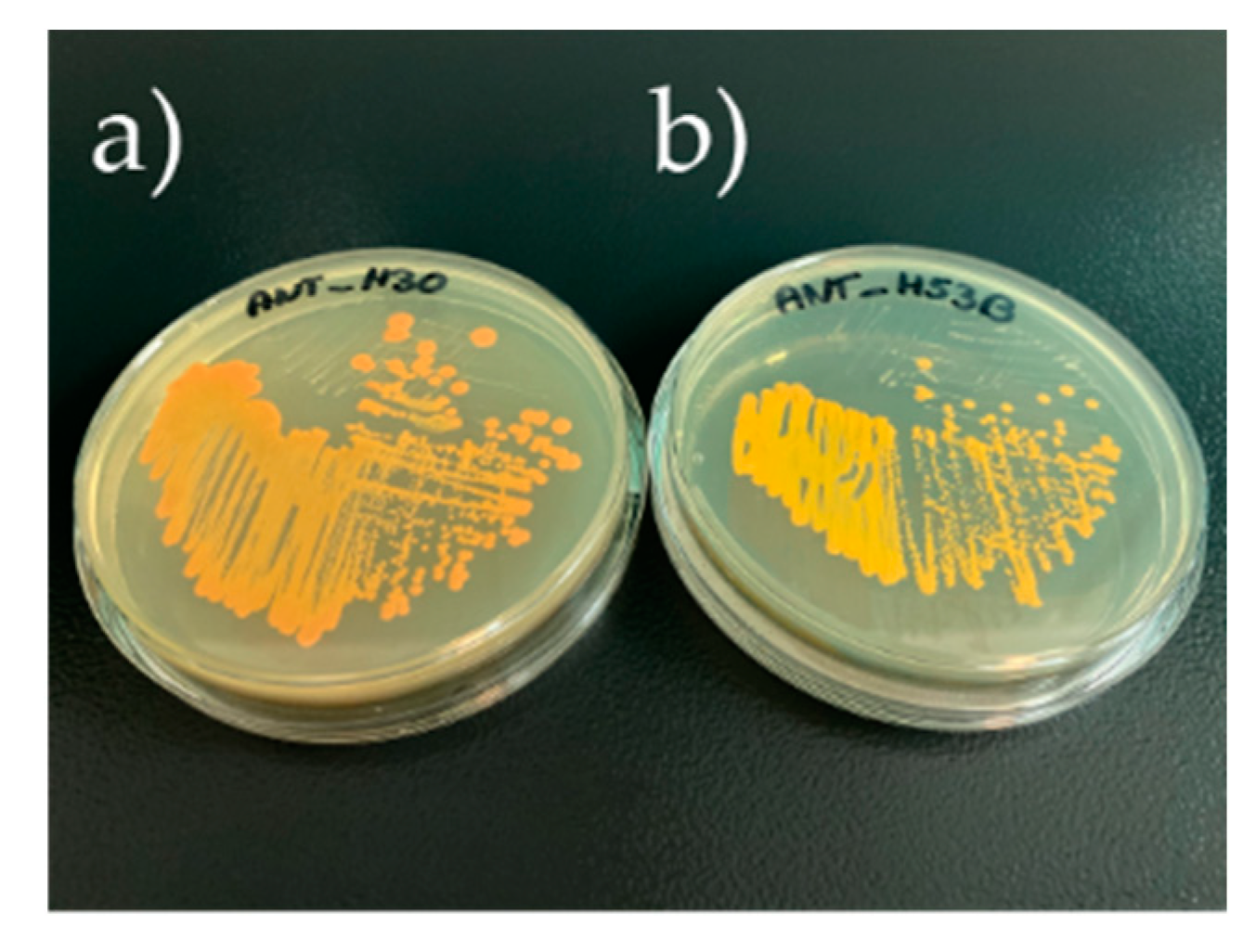
2.1. General Physiological Characterization of Planococcus sp. ANT_H30 and Rhodococcus sp. ANT_H53B
2.2. Genomic Characterization of Bacterial Strains and Identification of Carotenoid Biosynthesis Gene Clusters
2.2.1. Genomes Sequencing and Overall Genomic Characterization
2.2.2. Identification of Carotenoids Biosynthesis Gene Clusters
2.2.3. Genome-Based Insight into the Metabolic Potential
- (i)
- benzoates and ethylbenzene—using e.g., 4-methoxybenzoate monooxygenase (EC: 1.14.99.15) (GenBank accession number: FQ188_15500), P-hydroxybenzoate 3-monooxygenase (EC: 1.14.13.2) (GenBank accession number: FQ188_16125), benzoate 1,2-dioxygenase (EC: 1.14.12.10) (GenBank accession number: FQ188_16155), protocatechuate 3,4-dioxygenase (EC: 1.13.11.3) (GenBank accession number: FQ188_10175), and hydroxyquinol 1,2-dioxygenase (EC: 1.13.11.37) (GenBank accession number: FQ188_18125);
- (ii)
- aminobenzoates—with the use of amidase (EC: 3.5.1.4) (GenBank accession numbers: FQ188_09805, FQ188_11165, and FQ188_16180), and monooxygenase (EC: 1.14.13.-) (GenBank accession numbers: FQ188_11040 and FQ188_17850);
- (iii)
- fluorobenzoates—using carboxymethylenebutenolidase (EC: 3.1.1.45) (GenBank accession numbers: FQ188_13875, FQ188_14120, FQ188_15865, and FQ188_18215);
- (iv)
- toluene and xylene—using benzaldehyde dehydrogenase (EC: 1.2.1.28) (GenBank accession number: FQ188_11010), maleylacetate reductase (EC: 1.3.1.32) (GenBank accession number: FQ188_18130), and catechol 1,2-dioxygenase (EC: 1.13.11.1) (GenBank accession number: FQ188_16170);
- (v)
- nitro compounds, such as nitrotoluene, atrazine, caprolactam—using dihydropteridine reductase (EC: 1.5.1.34) (GenBank accession number: FQ188_00250), and N-ethylmaleimide reductase (GenBank accession number: FQ188_00250);
- (vi)
- halogenated compounds, like dioxins, chloroalkanes, chloroalkenes, chlorocyclohexane, and chlorobenzene—using 2-haloacid dehalogenase (EC: 3.8.1.2) (GenBank accession numbers: FQ188_04145, FQ188_19570, and FQ188_19725), 2,4-dichlorophenol 6-monooxygenase (EC: 1.14.13.20) (GenBank accession numbers: FQ188_15475 and FQ188_18135).
2.2.4. Biosafety Considerations of Planococcus sp. ANT_H30 and Rhodococcus sp. ANT_H53B
2.3. Chemical Identification of Synthesized Carotenoids
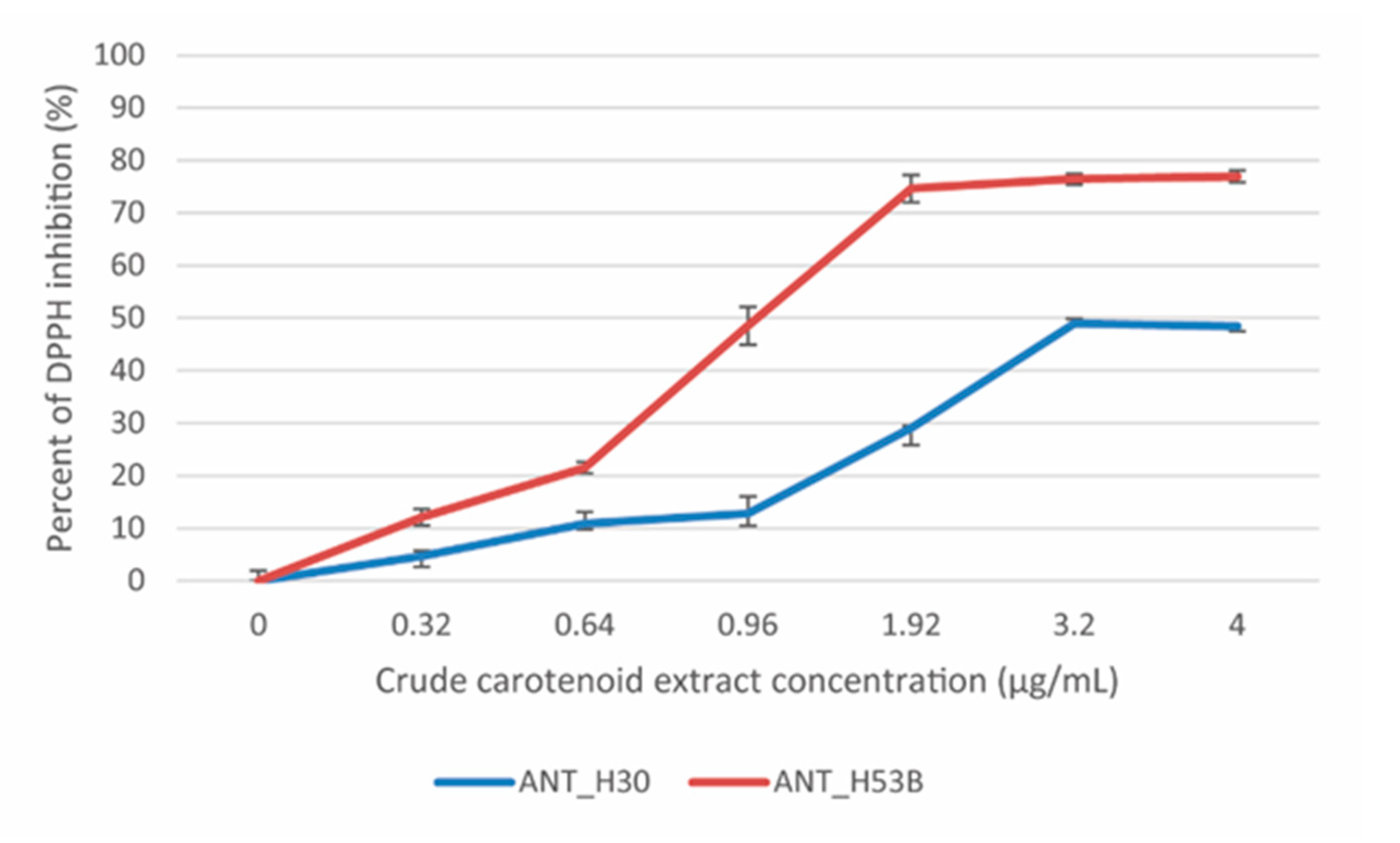
2.4. Free Radical Scavenging Activity
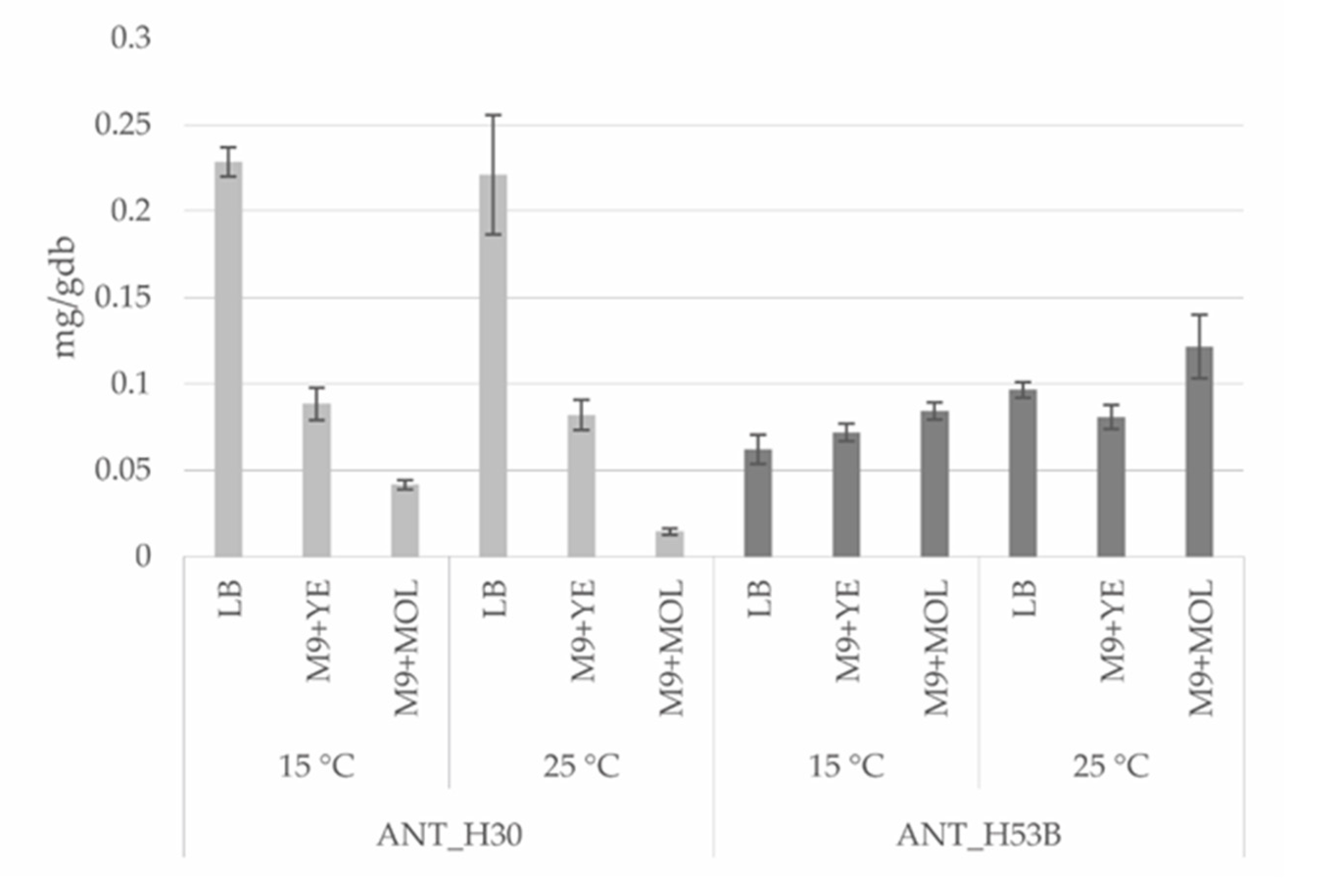
2.5. Optimization of Production of Carotenoids
3. Materials and Methods
3.1. Bacterial Strains and Culture Conditions
3.2. Detection of Siderophores and Surfactants
3.3. Draft Genome Sequencing
3.4. Bioinformatics
3.5. Antibiotic Susceptibility Testing
3.6. Extraction of Carotenoids from Bacterial Culture
3.7. Qualitative Analysis of Carotenoids
3.7.1. Ultraperformance Liquid Chromatography (UPLC)
3.7.2. Gas Chromatography-Mass Spectrometry (GC-MS)
3.8. Quantitative Analysis of Carotenoids
3.9. Free Radical Scavenging Activity
3.10. Nucleotide Sequence Accession Numbers
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ruiz, B.; Chávez, A.; Forero, A.; García-Huante, Y.; Romero, A.; Sánchez, M.; Rocha, D.; Sánchez, B.; Rodríguez-Sanoja, R.; Sánchez, S.; et al. Production of microbial secondary metabolites: Regulation by the carbon source. Crit. Rev. Microbiol. 2010, 36, 146–167. [Google Scholar] [CrossRef]
- Reis-Mansur, M.C.P.P.; Cardoso-Rurr, J.S.; Silva, J.V.M.A.; de Souza, G.R.; Cardoso, V.D.S.; Mansoldo, F.R.P.; Pinheiro, Y.; Schultz, J.; Lopez Balottin, L.B.; da Silva, A.J.R.; et al. Carotenoids from UV-resistant Antarctic Microbacterium sp. LEMMJ01. Sci. Rep. 2019, 9, 1–14. [Google Scholar] [CrossRef]
- Dieser, M.; Greenwood, M.; Foreman, C.M. Carotenoid pigmentation in Antarctic heterotrophic bacteria as a strategy to withstand environmental stresses. Arctic. Antarct. Alp. Res. 2010, 42, 396–405. [Google Scholar] [CrossRef]
- Tuncer, P.B.; Büyükleblebici, S.; Eken, A.; Taşdemir, U.; Durmaz, E.; Büyükleblebici, O.; Çoşkun, E. Comparison of cryoprotective effects of lycopene and cysteamine in different cryoprotectants on bull semen and fertility results. Reprod. Domest. Anim. 2014, 49, 746–752. [Google Scholar] [CrossRef]
- Nagarajan, J.; Ramakrishnan, R.; Raghunandan, M.E.; Galanakis, C.M.; Krishnamurthy, N.P. Chapter 8–Carotenoids. In Nutraceutical and Functional Food Components-Effects of Innovative Processing Techniques; Galanakis, C.M., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 259–296. ISBN 978-0-12-805257-0. [Google Scholar]
- Sánchez, S.; Ruiz, B.; Rodríguez-Sanoja, R.; Flores-Cotera, L.B. Microbial production of carotenoids. In Microbial Production of Food Ingredients, Enzymes and Nutraceuticals; Elsevier: Amsterdam, The Netherlands, 2013; pp. 194–234. ISBN 9780857093431. [Google Scholar] [CrossRef]
- Sandmann, G. Carotenoids of biotechnological importance. In Advances in Biochemical Engineering/Biotechnology; Scheper, T., Ed.; Springer: Cham, Switzerland, 2015; Volume 148, pp. 449–467. ISBN 978-3-319-20107-8. [Google Scholar] [CrossRef]
- Cavicchioli, R.; Charlton, T.; Ertan, H.; Omar, S.M.; Siddiqui, K.S.; Williams, T.J. Biotechnological uses of enzymes from psychrophiles. Microb. Biotechnol. 2011, 4, 449–460. [Google Scholar] [CrossRef]
- Maj, A.; Dziewit, L.; Drewniak, L.; Garstka, M.; Krucon, T.; Piatkowska, K.; Gieczewska, K.; Czarnecki, J.; Furmanczyk, E.; Lasek, R.; et al. In vivo creation of plasmid pCRT01 and its use for the construction of carotenoid-producing Paracoccus spp. strains that grow efficiently on industrial wastes. Microb. Cell Fact. 2020, 19, 1–14. [Google Scholar] [CrossRef]
- Kim, J.W.; Choi, B.H.; Kim, J.H.; Kang, H.J.; Ryu, H.; Lee, P.C. Complete genome sequence of Planococcus faecalis AJ003 T, the type species of the genus Planococcus and a microbial C30 carotenoid producer. J. Biotechnol. 2018, 266, 72–76. [Google Scholar] [CrossRef]
- Umeno, D.; Tobias, A.V.; Arnold, F.H. Evolution of the C30 carotenoid synthase CrtM for function in a C40 pathway. J. Bacteriol. 2002, 184, 6690–6699. [Google Scholar] [CrossRef]
- Atam, S.I.; Singh, L.; Dube, S.; Reddy, G.S.N.; Shivaji, S. Psychrophilic Planococcus maitriensis sp.nov. from Antarctica. Syst. Appl. Microbiol. 2003, 26, 505–510. [Google Scholar] [CrossRef]
- Majumdar, S.; Priyadarshinee, R.; Kumar, A.; Mandal, T.; Dasgupta Mandal, D. Exploring Planococcus sp. TRC1, a bacterial isolate, for carotenoid pigment production and detoxification of paper mill effluent in immobilized fluidized bed reactor. J. Clean. Prod. 2019, 211, 1389–1402. [Google Scholar] [CrossRef]
- Majidzadeh, M.; Fatahi-Bafghi, M. Current taxonomy of Rhodococcus species and their role in infections. Eur. J. Clin. Microbiol. Infect. Dis. 2018, 37, 2045–2062. [Google Scholar] [CrossRef] [PubMed]
- Bej, A.K.; Saul, D.; Aislabie, J. Cold-tolerant alkane-degrading Rhodococcus species from Antarctica. Polar Biol. 2000, 23, 100–105. [Google Scholar] [CrossRef]
- Espuny, M.J.; Egjido, S.; Mercadè, M.E.; Manresa, A. Characterization of trehalose tetraester produced by a waste lube oil degrader Rhodococcus sp. 51T7. Toxicol. Environ. Chem. 1995, 48, 83–88. [Google Scholar] [CrossRef]
- Le, R.K.; Wells, T.; Das, P.; Meng, X.; Stoklosa, R.J.; Bhalla, A.; Hodge, D.B.; Yuan, J.S.; Ragauskas, A.J. Conversion of corn stover alkaline pre-treatment waste streams into biodiesel via Rhodococci. RSC Adv. 2017, 7, 4108–4115. [Google Scholar] [CrossRef]
- Mahan, K.M.; Le, R.K.; Wells, T.; Anderson, S.; Yuan, J.S.; Stoklosa, R.J.; Bhalla, A.; Hodge, D.B.; Ragauskas, A.J. Production of single cell protein from agro-waste using Rhodococcus opacus. J. Ind. Microbiol. Biotechnol. 2018, 45, 795–801. [Google Scholar] [CrossRef] [PubMed]
- Bell, K.S.; Philp, J.C.; Aw, D.W.J.; Christofi, N. The genus Rhodococcus. J. Appl. Microbiol. 1998, 85, 195–210. [Google Scholar] [CrossRef]
- Chen, Y.; Xie, B.; Yang, J.; Chen, J.; Sun, Z. Identification of microbial carotenoids and isoprenoid quinones from Rhodococcus sp. B7740 and its stability in the presence of iron in model gastric conditions. Food Chem. 2018, 240, 201–211. [Google Scholar] [CrossRef]
- Romaniuk, K.; Ciok, A.; Decewicz, P.; Uhrynowski, W.; Budzik, K.; Nieckarz, M.; Pawlowska, J.; Zdanowski, M.K.; Bartosik, D.; Dziewit, L. Insight into heavy metal resistome of soil psychrotolerant bacteria originating from King George Island (Antarctica). Polar Biol. 2018, 41, 1319–1333. [Google Scholar] [CrossRef]
- Ravikumar, S.; Woo, H.M.; Choi, J.-i. Analysis of novel antioxidant sesquarterpenes (C35 terpenes) produced in recombinant Corynebacterium glutamicum. Appl. Biochem. Biotechnol. 2018, 186, 525–534. [Google Scholar] [CrossRef]
- Perez-Fons, L.; Steiger, S.; Khaneja, R.; Bramley, P.M.; Cutting, S.M.; Sandmann, G.; Fraser, P.D. Identification and the developmental formation of carotenoid pigments in the yellow/orange Bacillus spore-formers. Biochim. Biophys. Acta-Mol. Cell Biol. Lipids 2011, 1811, 177–185. [Google Scholar] [CrossRef]
- Hupert-Kocurek, K.; Guzik, U.; Wojcieszyńska, D. Characterization of catechol 2,3-dioxygenase from Planococcus sp. strain S5 induced by high phenol concentration. Acta Biochim. Pol. 2012, 59, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Liu, Y.H.; Luo, N.; Zhang, X.Y.; Luan, T.G.; Hu, J.M.; Wang, Z.Y.; Wu, P.C.; Chen, M.J.; Lu, J.Q. Biodegradation of benzene and its derivatives by a psychrotolerant and moderately haloalkaliphilic Planococcus sp. strain ZD22. Res. Microbiol. 2006, 157, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.H.; Joe, M.H.; Kim, D.H.; Park, H.; Choi, J.I.; Lim, S. Complete genome sequence of Planococcus sp. PAMC21323 isolated from Antarctica and its metabolic potential to detoxify pollutants. Stand. Genomic Sci. 2018, 13, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Palatinszky, M.; Herbold, C.; Jehmlich, N.; Pogoda, M.; Han, P.; Von Bergen, M.; Lagkouvardos, I.; Karst, S.M.; Galushko, A.; Koch, H.; et al. Cyanate as an energy source for nitrifiers. Nature 2015, 524, 105–108. [Google Scholar] [CrossRef]
- Howden, A.J.M.; Preston, G.M. Nitrilase enzymes and their role in plant-microbe interactions. Microb. Biotechnol. 2009, 2, 441–451. [Google Scholar] [CrossRef]
- Blasco, R.; Martínez-Luque, M.; Madrid, M.P.; Castillo, F.; Moreno-Vivián, C. Rhodococcus sp. RB1 grows in the presence of high nitrate and nitrite concentrations and assimilates nitrate in moderately saline environments. Arch. Microbiol. 2001, 175, 435–440. [Google Scholar] [CrossRef]
- Akhtar, N.; Ghauri, M.A.; Akhtar, K.; Parveen, S.; Farooq, M.; Ali, A.; Schierack, P. Comparative analysis of draft genome sequence of Rhodococcus sp. Eu-32 with other Rhodococcus species for its taxonomic status and sulfur metabolism potential. Curr. Microbiol. 2019, 76, 1207–1214. [Google Scholar] [CrossRef]
- Nguyen, Q.T.; Trinco, G.; Binda, C.; Mattevi, A.; Fraaije, M.W. Discovery and characterization of an F420-dependent glucose-6-phosphate dehydrogenase (Rh-FGD1) from Rhodococcus jostii RHA1. Appl. Microbiol. Biotechnol. 2017, 101, 2831–2842. [Google Scholar] [CrossRef]
- Greening, C.; Ahmed, F.H.; Mohamed, A.E.; Lee, B.M.; Pandey, G.; Warden, A.C.; Scott, C.; Oakeshott, J.G.; Taylor, M.C.; Jackson, C.J. Physiology, biochemistry, and applications of F420- and Fo-dependent redox Reactions. Microbiol. Mol. Biol. Rev. 2016, 80, 451–493. [Google Scholar] [CrossRef]
- Zhang, Y.; Qin, F.; Qiao, J.; Li, G.; Shen, C.; Huang, T.; Hu, Z. Draft genome sequence of Rhodococcus sp. strain P14, a biodegrader of high-molecular-weight polycyclic aromatic hydrocarbons. J. Bacteriol. 2012, 194, 3546. [Google Scholar] [CrossRef]
- Chen, B.S.; Otten, L.G.; Resch, V.; Muyzer, G.; Hanefeld, U. Draft genome sequence of Rhodococcus rhodochrous strain ATCC 17895. Stand. Genom. Sci. 2013, 9, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Vikram, S.; Kumar, S.; Subramanian, S.; Raghava, G.P.S. Draft genome sequence of the nitrophenol-degrading actinomycete Rhodococcus imtechensis RKJ300. J. Bacteriol. 2012, 194, 3543. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Franzetti, A.; Gandolfi, I.; Bestetti, G.; Smyth, T.J.P.; Banat, I.M. Production and applications of trehalose lipid biosurfactants. Eur. J. Lipid Sci. Technol. 2010, 112, 617–627. [Google Scholar] [CrossRef]
- Stes, E.; Francis, I.; Pertry, I.; Dolzblasz, A.; Depuydt, S.; Vereecke, D. The leafy gall syndrome induced by Rhodococcus fascians. FEMS Microbiol. Lett. 2013, 342, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Anastasi, E.; MacArthur, I.; Scortti, M.; Alvarez, S.; Giguére, S.; Vázquez-Boland, J.A. Pangenome and phylogenomic analysis of the pathogenic actinobacterium Rhodococcus equi. Genome Biol. Evol. 2016, 8, 3140–3148. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Morichaud, Z.; Chen, S.; Leonetti, J.P.; Brodolin, K. Mycobacterium tuberculosis RbpA protein is a new type of transcriptional activator that stabilizes the σ a-containing RNA polymerase holoenzyme. Nucleic Acids Res. 2012, 40, 6547–6557. [Google Scholar] [CrossRef]
- Michel, A.; Agerer, F.; Hauck, C.R.; Herrmann, M.; Ullrich, J.; Hacker, J.; Ohlsen, K. Global regulatory impact of ClpP protease of Staphylococcus aureus on regulons involved in virulence, oxidative stress response, autolysis, and DNA repair. J. Bacteriol. 2006, 188, 5783–5796. [Google Scholar] [CrossRef]
- Wall, D.M.; Duffy, P.S.; DuPont, C.; Prescott, J.F.; Meijer, W.G. Isocitrate lyase activity is required for virulence of the intracellular pathogen Rhodococcus equi. Infect. Immun. 2005, 73, 6736–6741. [Google Scholar] [CrossRef]
- Popova, A.V.; Andreeva, A.S. Carotenoid-lipid Interactions. In Advances in Planar Lipid Bilayers and Liposomes; Iglič, A., Rappolt, M., Kulkarni, C., Eds.; Academic Press: Cambridge, MA, USA, 2013; pp. 215–236. [Google Scholar] [CrossRef]
- Molyneux, P. The use of the stable free radical diphenylpicryl-hydrazyl (DPPH) for estimating antioxidant activity. Songklanakarin J. Sci. Technol. 2004, 26, 211–219. [Google Scholar] [CrossRef]
- Asker, D.; Beppu, T.; Ueda, K. Unique diversity of carotenoid-producing bacteria isolated from Misasa, a radioactive site in Japan. Appl. Microbiol. Biotechnol. 2007, 77, 383–392. [Google Scholar] [CrossRef]
- Vila, E.; Hornero-Méndez, D.; Azziz, G.; Lareo, C.; Saravia, V. Carotenoids from heterotrophic bacteria isolated from Fildes Peninsula, King George Island, Antarctica. Biotechnol. Rep. 2019, 21, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ram, S.; Mitra, M.; Shah, F.; Tirkey, S.R.; Mishra, S. Bacteria as an alternate biofactory for carotenoid production: A review of its applications, opportunities and challenges. J. Funct. Foods 2020, 67, 103867. [Google Scholar] [CrossRef]
- Asker, D. Isolation and characterization of a novel, highly selective astaxanthin-producing marine bacterium. J. Agric. Food Chem. 2017, 65, 9101–9109. [Google Scholar] [CrossRef] [PubMed]
- Elbing, K.; Brent, R. Media preparation and bacteriological tools. Curr. Protoc. Mol. Biol. 2002, 59, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Bultreys, A.; Gheysen, I. Production and comparison of peptide siderophores from strains of distantly related pathovars of Pseudomonas syringae and Pseudomonas viridiflava LMG 2352. Appl. Environ. Microbiol. 2000, 66, 325–331. [Google Scholar] [CrossRef][Green Version]
- Schwyn, B.; Neilands, J.B. Universal chemical assay for the detection and determination of siderophores. Anal. Biochem. 1987, 160, 47–56. [Google Scholar] [CrossRef]
- Sambrook, J.; Maniatis, T.; Fritsch, E.F.; Russell, D.W. Russell Molecular cloning: A laboratory manual, III; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2001; ISBN 9781936113415. [Google Scholar] [CrossRef][Green Version]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 2011, 17, 10. [Google Scholar] [CrossRef]
- Aziz, R.K.; Bartels, D.; Best, A.A.; DeJongh, M.; Disz, T.; Edwards, R.A.; Formsma, K.; Gerdes, S.; Glass, E.M.; Kubal, M.; et al. The RAST Server: Rapid annotations using subsystems technology. BMC Genom. 2008, 9, 75. [Google Scholar] [CrossRef]
- Wattam, A.R.; Davis, J.J.; Assaf, R.; Boisvert, S.; Brettin, T.; Bun, C.; Conrad, N.; Dietrich, E.M.; Disz, T.; Gabbard, J.L.; et al. Improvements to PATRIC, the all-bacterial bioinformatics database and analysis resource center. Nucleic Acids Res. 2017, 45, D535–D542. [Google Scholar] [CrossRef]
- Altschul, S.F.; Madden, T.L.; Schaffer, A.A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped BLAST and PSI-BLAST: A new generation of protein database search programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef]
- Overbeek, R.; Olson, R.; Pusch, G.D.; Olsen, G.J.; Davis, J.J.; Disz, T.; Edwards, R.A.; Gerdes, S.; Parrello, B.; Shukla, M.; et al. The SEED and the rapid annotation of microbial genomes using subsystems technology (RAST). Nucleic Acids Res. 2014, 42, D206–D214. [Google Scholar] [CrossRef] [PubMed]
- Moriya, Y.; Itoh, M.; Okuda, S.; Yoshizawa, A.C.; Kanehisa, M. KAAS: An automatic genome annotation and pathway reconstruction server. Nucleic Acids Res. 2007, 35, W182–W185. [Google Scholar] [CrossRef] [PubMed]
- Blin, K.; Shaw, S.; Steinke, K.; Villebro, R.; Ziemert, N.; Lee, S.Y.; Medema, M.H.; Weber, T. antiSMASH 5.0: Updates to the secondary metabolite genome mining pipeline. Nucleic Acids Res. 2019, 47, 81–87. [Google Scholar] [CrossRef]
- Kanehisa, M.; Sato, Y.; Morishima, K. BlastKOALA and GhostKOALA: KEGG tools for functional characterization of genome and metagenome sequences. J. Mol. Biol. 2016, 428, 726–731. [Google Scholar] [CrossRef]
- Liu, B.; Zheng, D.; Jin, Q.; Chen, L.; Yang, J. VFDB 2019: A comparative pathogenomic platform with an interactive web interface. Nucleic Acids Res. 2019, 47, 687–692. [Google Scholar] [CrossRef]
- Jia, B.; Raphenya, A.R.; Alcock, B.; Waglechner, N.; Guo, P.; Tsang, K.K.; Lago, B.A.; Dave, B.M.; Pereira, S.; Sharma, A.N.; et al. CARD 2017: Expansion and model-centric curation of the comprehensive antibiotic resistance database. Nucleic Acids Res. 2017, 45, D566–D573. [Google Scholar] [CrossRef]
- EUCAST European Committee on Antimicrobial Susceptibility Testing (EUCAST). Available online: http://www.eucast.org (accessed on 30 January 2020).
- Skupień, J.; Wójtowicz, J.; Kowalewska, Ł.; Mazur, R.; Garstka, M.; Gieczewska, K.; Mostowska, A. Dark-chilling induces substantial structural changes and modifies galactolipid and carotenoid composition during chloroplast biogenesis in cucumber (Cucumis sativus L.) cotyledons. Plant. Physiol. Biochem. 2017, 111, 107–118. [Google Scholar] [CrossRef]
- Szakiel, A.; Voutquenne-Nazabadioko, L.; Henry, M. Isolation and biological activities of lyoniside from rhizomes and stems of Vaccinium myrtillus. Phytochem. Lett. 2011, 4, 138–143. [Google Scholar] [CrossRef]
Sample Availability: Samples of the carotenoids produced by both strains are available from the authors. |

| Feature | Calculation | |
|---|---|---|
| Strain | ANT_H30 | ANT_H53B |
| Number of Contigs | 22 | 37 |
| Estimated Genome Size (bp) | 3,636,638 | 5,176,448 |
| GC Content (%) | 40.8% | 64.87% |
| Number of Genes | 3591 | 4889 |
| Number of Proteins with Functional Assignments | 2562 | 3379 |
| Number of Proteins with Enzyme Commission (EC) Number Assignments | 874 | 1149 |
| Number of Transfer RNA (tRNA) Genes | 59 | 46 |
| Number of Regulatory RNA Genes | 23 | 12 |
| Strain | Gene | GenBank Accession Number | Encoded Protein | Reference Protein | Amino Acids Identity |
|---|---|---|---|---|---|
| ANT_H30 | crtP | FQ085_05070; FQ085_10685 | Diapolycopene oxygenase (EC: 1.14.99.44) | AUO94_02190; AUO94_13335 | 98%; 98% |
| ANT_H30 | crtM | FQ085_05075 | Dehydrosqualene synthase (EC: 2.5.1.96) | AUO94_02185 | 98% |
| ANT_H30 | crtN | FQ085_05080 | Dehydrosqualene desaturase (EC: 1.3.8.2) | AUO94_02180 | 98% |
| ANT_H30 | crtNc | FQ085_05085 | 4,4′-diapolycopene oxidase (EC: 1.14.99.44) | AUO94_02175 | 98% |
| ANT_H53B | crtE | FQ188_09125 | Geranylgeranyl diphosphate synthase (EC: 2.5.1.29) | NY08_684 | 95% |
| ANT_H53B | crtY | FQ188_06685 | Lycopene beta-cyclase (EC: 5.5.1.19) | NY08_1078 | 93% |
| ANT_H53B | crtO | FQ188_09100 | Beta-carotene ketolase (EC: 1.14.99.63) | NY08_689 | 95% |
| ANT_H53B | crtI | FQ188_09130, FQ188_15555 | Phytoene dehydrogenase (EC: 1.14.99.-) | NY08_683; NY08_2230 | 94%; 90% |
| ANT_H53B | crtB | FQ188_09140 | Phytoene synthase (EC: 2.5.1.32) | NY08_680 | 95% |
| ANT_H53B | crtZ | FQ188_15555 | Carotene hydroxylase (EC: 1.14.13.129) | NY08_4121 | 95% |
| ANT_H53B | crtU | FQ188_19840 | Phi-Carotenoid synthase (EC: 1.3.99.39) | NY08_3769 | 96% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Styczynski, M.; Rogowska, A.; Gieczewska, K.; Garstka, M.; Szakiel, A.; Dziewit, L. Genome-Based Insights into the Production of Carotenoids by Antarctic Bacteria, Planococcus sp. ANT_H30 and Rhodococcus sp. ANT_H53B. Molecules 2020, 25, 4357. https://doi.org/10.3390/molecules25194357
Styczynski M, Rogowska A, Gieczewska K, Garstka M, Szakiel A, Dziewit L. Genome-Based Insights into the Production of Carotenoids by Antarctic Bacteria, Planococcus sp. ANT_H30 and Rhodococcus sp. ANT_H53B. Molecules. 2020; 25(19):4357. https://doi.org/10.3390/molecules25194357
Chicago/Turabian StyleStyczynski, Michal, Agata Rogowska, Katarzyna Gieczewska, Maciej Garstka, Anna Szakiel, and Lukasz Dziewit. 2020. "Genome-Based Insights into the Production of Carotenoids by Antarctic Bacteria, Planococcus sp. ANT_H30 and Rhodococcus sp. ANT_H53B" Molecules 25, no. 19: 4357. https://doi.org/10.3390/molecules25194357
APA StyleStyczynski, M., Rogowska, A., Gieczewska, K., Garstka, M., Szakiel, A., & Dziewit, L. (2020). Genome-Based Insights into the Production of Carotenoids by Antarctic Bacteria, Planococcus sp. ANT_H30 and Rhodococcus sp. ANT_H53B. Molecules, 25(19), 4357. https://doi.org/10.3390/molecules25194357








