Simultaneous Natural Deep Eutectic Solvent-Based Ultrasonic-Assisted Extraction of Bioactive Compounds of Cinnamon Bark and Sappan Wood as a Dipeptidyl Peptidase IV Inhibitor
Abstract
1. Introduction
2. Results and Discussions
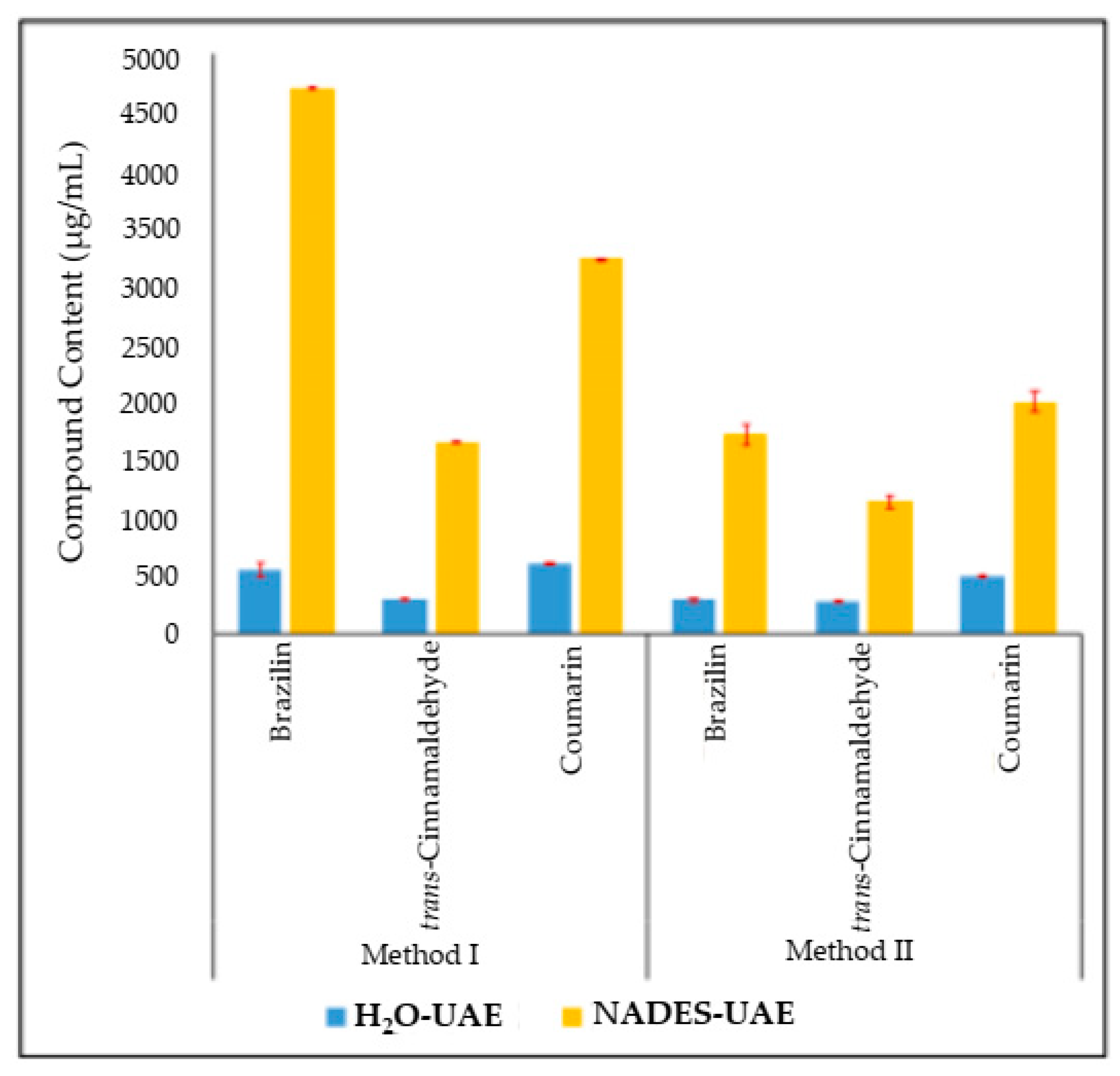
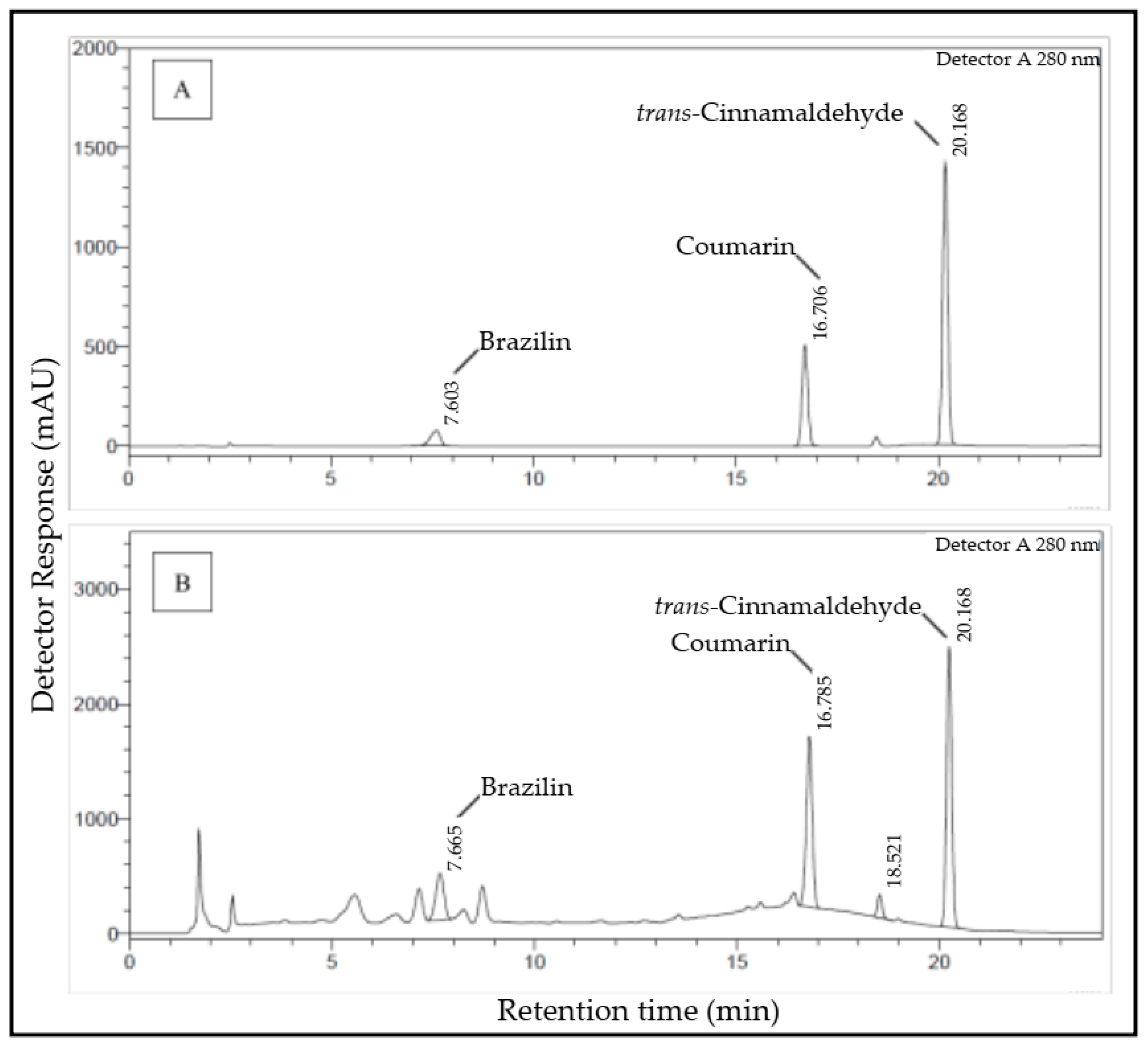
2.1. Simultaneous NADES-UAE Process and Marker Compound Determination
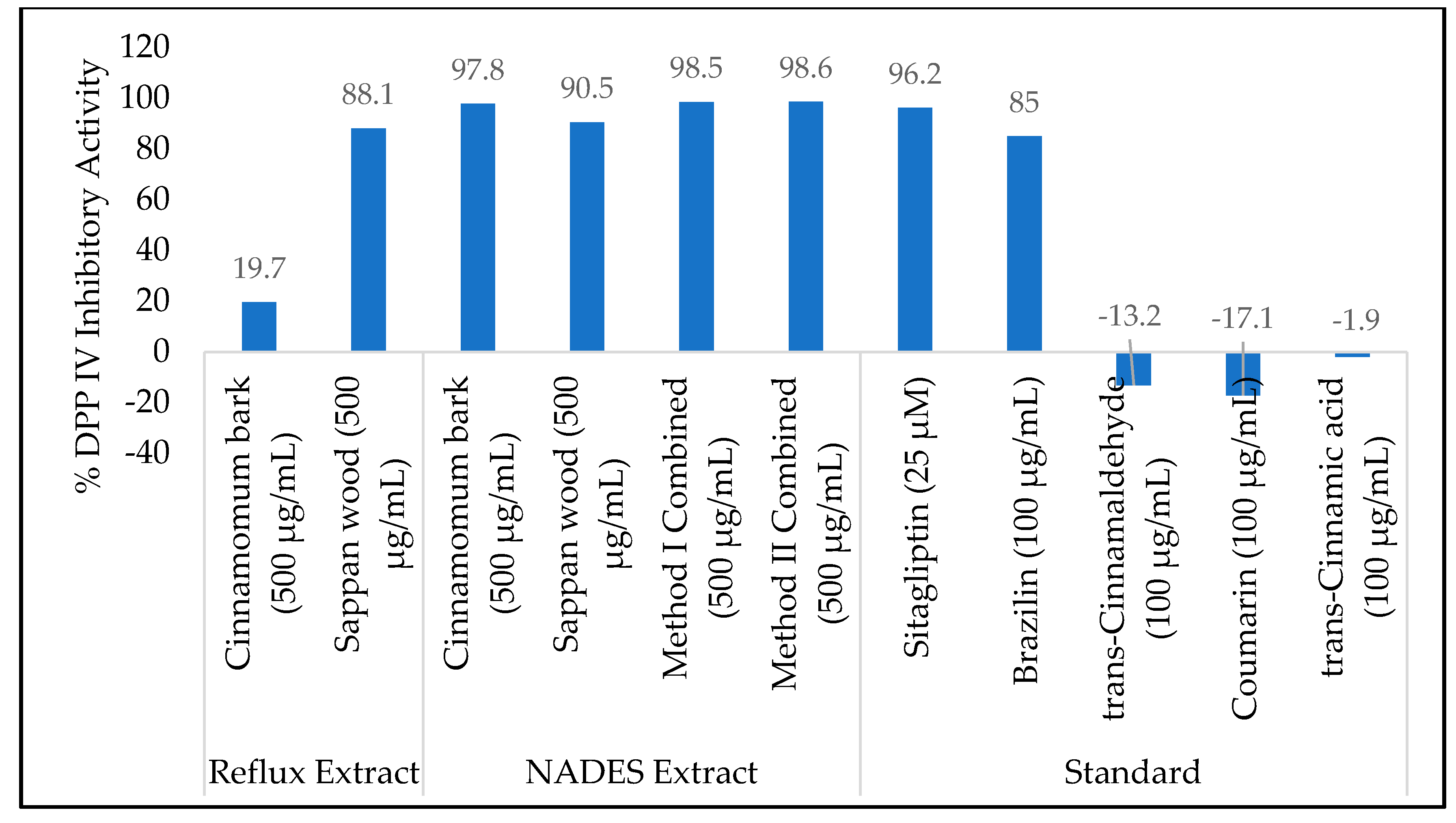
2.2. In Vitro DPP IV Inhibitory Activity Assay
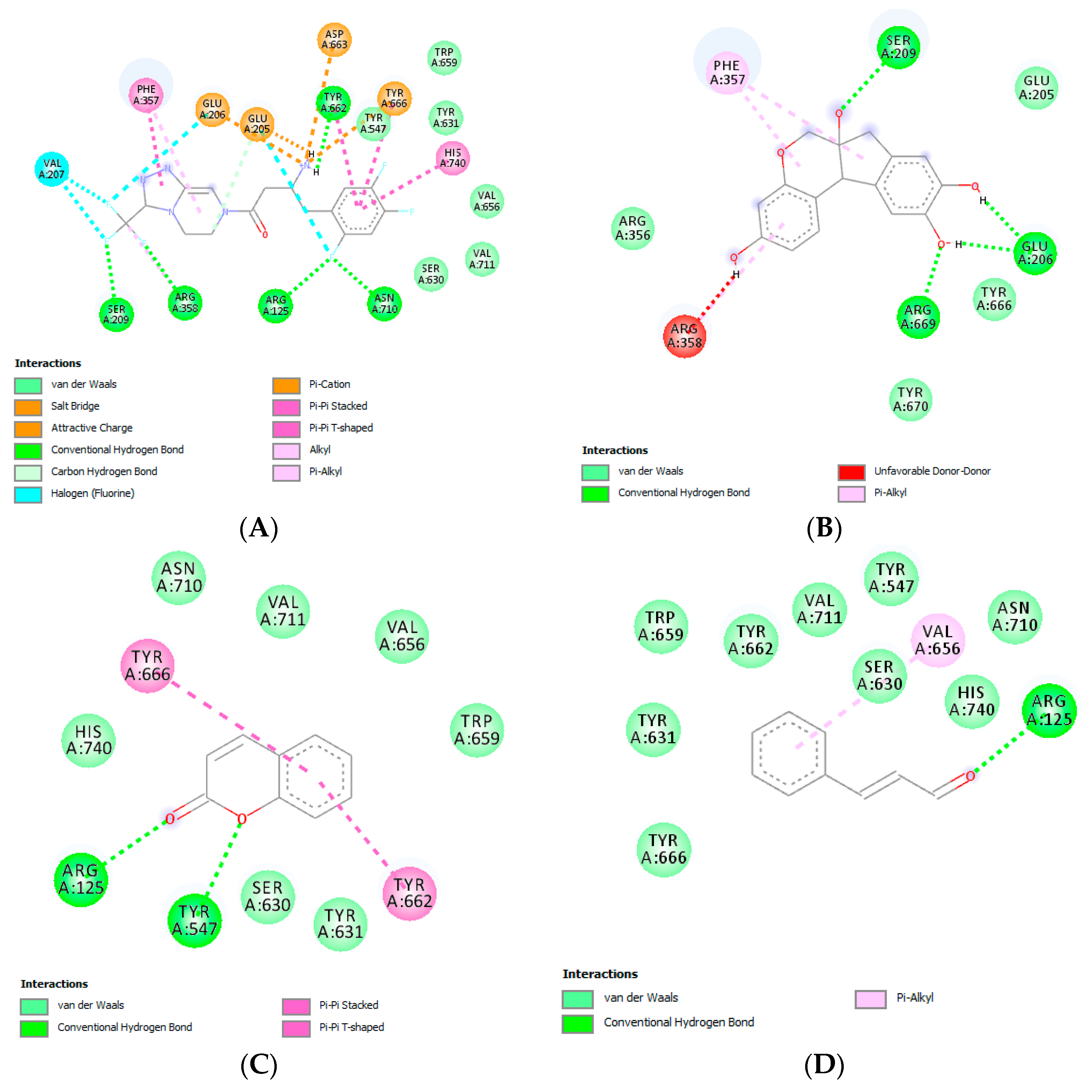
2.3. In Silico Molecular Docking Study
3. Materials and Methods
3.1. Materials
3.2. Equipment
3.3. Conventional and Non-Conventional Extraction Process
3.4. Simultaneous NADES-UAE Process for Extraction of Marker Compounds from a Combination of Samples
3.5. Analysis of Marker Compounds
3.6. In Vitro Dipeptidyl Peptidase IV Activity Assay
3.7. In Silico Molecular Docking Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- American Diabetes Association. Classification and diagnosis of diabetes. Diabetes Care 2017, 40, S11–S24. [Google Scholar] [CrossRef]
- Chaudhury, A.; Duvoor, C.; Dendi, V.S.R.; Kraleti, S.; Chada, A.; Ravilla, R.; Marco, A.; Shekhawat, N.S.; Montales, M.T.; Kuriakose, K.; et al. Clinical Review of Antidiabetic Drugs: Implications for Type 2 Diabetes Mellitus Management. Front. Endocrinol. 2017, 8, 1–12. [Google Scholar] [CrossRef]
- Tella, S.H.; Rendell, M.S. DPP-4 inhibitors: Focus on safety. Expert Opin. Drug Saf. 2014, 14, 127–140. [Google Scholar] [CrossRef]
- Lahrita, L.; Kato, E.; Kawabata, J. Uncovering potential of Indonesian medicinal plants on glucose uptake enhancement and lipid suppression in 3T3-L1 adipocytes. J. Ethnopharmacol. 2015, 168, 229–236. [Google Scholar] [CrossRef]
- You, E.-J.; Khil, L.-Y.; Kwak, W.-J.; Won, H.-S.; Chae, S.-H.; Lee, B.-H.; Moon, C.-K. Effects of brazilin on the production of fructose-2,6-bisphosphate in rat hepatocytes. J. Ethnopharmacol. 2005, 102, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Ranasinghe, P.; Galappatthy, P.; Constantine, G.R.; Jayawardena, R.; Weeratunga, H.D.; Premakumara, G.; Katulanda, P. Cinnamomum zeylanicum (Ceylon cinnamon) as a potential pharmaceutical agent for type-2 diabetes mellitus: Study protocol for a randomized controlled trial. Trials 2017, 18, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Tulin, E.K.C.B.; Loreto, M.T.P. Alpha-Glucosidase Inhibitory Activity and Fractionation of Bioactive Compounds from bark Extracts of Sibucao (Caesalpinia sappan L.) In the Philippines. Pharmacogn. J. 2017, 9, 356–360. [Google Scholar] [CrossRef]
- Setyaningsih, E.P.; Saputri, F.C.; Mun’Im, A. The Antidiabetic Effectivity of Indonesian Plants Extracts via DPP-IV Inhibitory Mechanism. J. Young- Pharm. 2019, 11, 161–164. [Google Scholar] [CrossRef]
- Kim, S.H.; Hyun, S.H.; Choung, S.Y. Anti-diabetic effect of cinnamon extract on blood glucose in db/db mice. J. Ethnopharmacol. 2006, 104, 119–123. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Polansky, M.M.; Anderson, R.A. Cinnamon extract and polyphenols affect the expression of tristetraprolin, insulin receptor, and glucose transporter 4 in mouse 3T3-L1 adipocytes. Arch. Biochem. Biophys. 2007, 459, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Lin, L.; Ye, W. Techniques for extraction and isolation of natural products: A comprehensive review. Chin. Med. 2018, 13, 20. [Google Scholar] [CrossRef] [PubMed]
- Jeliński, T.; Przybyłek, M.; Cysewski, P. Natural Deep Eutectic Solvents as Agents for Improving Solubility, Stability and Delivery of Curcumin. Pharm. Res. 2019, 36, 116. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Friesen, J.B.; McAlpine, J.B.; Lankin, D.C.; Chen, S.-N.; Pauli, G.F. Natural Deep Eutectic Solvents: Properties, Applications, and Perspectives. J. Nat. Prod. 2018, 81, 679–690. [Google Scholar] [CrossRef] [PubMed]
- Chemat, F.; Rombaut, N.; Sicaire, A.-G.; Meullemiestre, A.; Fabiano-Tixier, A.-S.; Abert-Vian, M. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason. Sonochemistry 2017, 34, 540–560. [Google Scholar] [CrossRef] [PubMed]
- Zardo, I.; Sobczyk, A.D.E.; Marczak, L.D.F.; Sarkis, J.R. Optimization of Ultrasound Assisted Extraction of Phenolic Compounds from Sunflower Seed Cake Using Response Surface Methodology. Waste Biomass Valorization 2017, 10, 33–44. [Google Scholar] [CrossRef]
- Savic-Gajic, I.M.; Savic, I.M.; Boskov, I.; Žerajić, S.; Markovic, I.; Gajic, D. Optimization of Ultrasound-Assisted Extraction of Phenolic Compounds from Black Locust (Robiniae pseudoacaciae) Flowers and Comparison with Conventional Methods. Antioxidants 2019, 8, 248. [Google Scholar] [CrossRef] [PubMed]
- Savic, I.M.; Gajic, I.M.S. Optimization of ultrasound-assisted extraction of polyphenols from wheatgrass (Triticum aestivum L.). J. Food Sci. Technol. 2020, 57, 2809–2818. [Google Scholar] [CrossRef]
- Wang, L.; Bai, M.; Qin, Y.; Liu, B.; Wang, Y.; Zhou, Y. Application of Ionic Liquid-Based Ultrasonic-Assisted Extraction of Flavonoids from Bamboo Leaves. Molecules 2018, 23, 2309. [Google Scholar] [CrossRef]
- Ahmad, I.; Pertiwi, A.S.; Kembaren, Y.H.; Rahman, A.; Mun’im, A. Application of natural deep eutectic solvent-based ultrasonic assisted extraction of total polyphenolic and caffeine content from coffe beans (Coffea Beans L.) for instant food products. J. Appl. Pharm. Sci. 2018, 81, 38–43. [Google Scholar]
- Sakti, A.S.; Saputri, F.C.; Mun’Im, A. Optimization of choline chloride-glycerol based natural deep eutectic solvent for extraction bioactive substances from Cinnamomum burmannii barks and Caesalpinia sappan heartwoods. Heliyon 2019, 5, e02915. [Google Scholar] [CrossRef]
- Cayman Chemical Company. DPP (IV) Inhibitor Screening Assay Kit; Cayman Chemical Company: Ann Arbor, MI, USA, 2017. [Google Scholar]
- Arulmozhiraja, S.; Matsuo, N.; Ishitsubo, E.; Okazaki, S.; Shimano, H.; Tokiwa, H. Comparative Binding Analysis of Dipeptidyl Peptidase IV (DPP-4) with Antidiabetic Drugs – An Ab Initio Fragment Molecular Orbital Study. PLoS ONE 2016, 11, e0166275. [Google Scholar] [CrossRef] [PubMed]
- Aryati, W.D.; Nadhira, A.; Febianli, D.; Fransisca, F.; Mun’Im, A. Natural deep eutectic solvents ultrasound-assisted extraction (NADES-UAE) of trans-cinnamaldehyde and coumarin from cinnamon bark [Cinnamomum burmannii (Nees T. Nees) Blume]. J. Res. Pharm. 2020, 24, 389–398. [Google Scholar] [CrossRef]
- Setiawan, H.; Angela, I.L.; Wijaya, O.; Mun’Im, A. Application of Natural Deep Eutectic Solvents (NADES) for Sappan Wood (Caesalpinia sappan L.) Extraction to Test for Inhibition of DPP IV Activity. J. Res. Pharm. 2020, 24, 380–388. [Google Scholar] [CrossRef]
- Matheeussen, V.; Jungraithmayr, W.; De Meester, I. Dipeptidyl peptidase 4 as a therapeutic target in ischemia/reperfusion injury. Pharmacol. Ther. 2012, 136, 267–282. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wu, Y.; Deng, Y.; Kim, B.; Pierce, L.; Krilov, G.; Lupyan, D.; Robinson, S.; Dahlgren, M.K.; Greenwood, J.; et al. Accurate and Reliable Prediction of Relative Ligand Binding Potency in Prospective Drug Discovery by Way of a Modern Free-Energy Calculation Protocol and Force Field. J. Am. Chem. Soc. 2015, 137, 2695–2703. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef] [PubMed]
- BIOVA DS. Discovery Studio Visualizer; BIOVA: San Diego, CA, USA, 2020. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |

| Extract or Standards | IC50 (µg/mL) |
|---|---|
| Cinnamon NADES | 205.0 |
| Sappan NADES | 1254.0 |
| Combined NADES Method-1 | 37.5 |
| Combined NADES Method-2 | 353.6 |
| Sappan Reflux | 82.0 |
| Brazilin | 9.9 |
| Sitagliptin | 5 × 10−3 |
| Ligand Compound | ΔG (kcal/mol) | Inhibition Constant | Interaction |
|---|---|---|---|
| Sitagliptin | −9.60 | 92.10 nM | Arg125, Glu205, Glu206, Val207, Ser209, Phe357, Arg358, Tyr547, Ser630, Tyr631, Val656, Trp659, Tyr662, Asp663, Tyr666, Asn710, Val711, His740 |
| Brazilin | −6.35 | 22.06 µM | Glu205, Glu206, Ser209, Arg356, Phe357, Arg358, Tyr666, Arg669, Tyr670 |
| Coumarin | −5.46 | 99.97 µM | Arg125, Tyr547, Ser630, Tyr631, Tyr662, Val656, Trp659, Tyr666, Asn710, Val711, His740 |
| trans-Cinnamaldehyde | −4.95 | 237.03 µM | Arg125, Tyr547, Ser630, Tyr631, Val656, Trp659, Tyr662, Tyr666, Asn710, Val711, His740 |
| trans-Cinnamic acid | −4.16 | 898.84 µM | Arg125, Glu205, Tyr547, Ser630, Tyr631, Val656, Trp659, Tyr662, Tyr666, Val711, His740 |
| Optimum Extraction Conditions | Method and Extraction Time | Choline Chloride–Glycerol Ratio | Sample–NADES Ratio | Water Added |
|---|---|---|---|---|
| Method I (optimum conditions for brazilin) | UAE, for 50 min | 2:1 w/w | 1:2 w/w | 47.57% |
| Method II (optimum conditions for trans-cinnamaldehyde and coumarin) | UAE, for 30 min | 2:1 w/w | 1:8 w/w | 20.00% |
| No | Time (min) | ddH2O (Acetic Acid) | Acetonitrile |
|---|---|---|---|
| 1 | 0 | 84.5% (0.3%) | 14.5% |
| 2 | 8 | 74.5% (0.3%) | 24.5% |
| 3 | 16 | 50.0% (0.04%) | 50.0% |
| 4 | 24 | 40.0% (0.04%) | 60.0% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad, I.; Arifianti, A.E.; Sakti, A.S.; Saputri, F.C.; Mun’im, A. Simultaneous Natural Deep Eutectic Solvent-Based Ultrasonic-Assisted Extraction of Bioactive Compounds of Cinnamon Bark and Sappan Wood as a Dipeptidyl Peptidase IV Inhibitor. Molecules 2020, 25, 3832. https://doi.org/10.3390/molecules25173832
Ahmad I, Arifianti AE, Sakti AS, Saputri FC, Mun’im A. Simultaneous Natural Deep Eutectic Solvent-Based Ultrasonic-Assisted Extraction of Bioactive Compounds of Cinnamon Bark and Sappan Wood as a Dipeptidyl Peptidase IV Inhibitor. Molecules. 2020; 25(17):3832. https://doi.org/10.3390/molecules25173832
Chicago/Turabian StyleAhmad, Islamudin, Ayun Erwina Arifianti, Aditya Sindu Sakti, Fadlina Chany Saputri, and Abdul Mun’im. 2020. "Simultaneous Natural Deep Eutectic Solvent-Based Ultrasonic-Assisted Extraction of Bioactive Compounds of Cinnamon Bark and Sappan Wood as a Dipeptidyl Peptidase IV Inhibitor" Molecules 25, no. 17: 3832. https://doi.org/10.3390/molecules25173832
APA StyleAhmad, I., Arifianti, A. E., Sakti, A. S., Saputri, F. C., & Mun’im, A. (2020). Simultaneous Natural Deep Eutectic Solvent-Based Ultrasonic-Assisted Extraction of Bioactive Compounds of Cinnamon Bark and Sappan Wood as a Dipeptidyl Peptidase IV Inhibitor. Molecules, 25(17), 3832. https://doi.org/10.3390/molecules25173832







