Properties of Polyhexamethylene Guanidine (PHMG) Associated with Fatal Lung Injury in Korea
Abstract
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Collection of Humidifier Disinfectant (HD) Samples
4.2. Quantification of PHMG
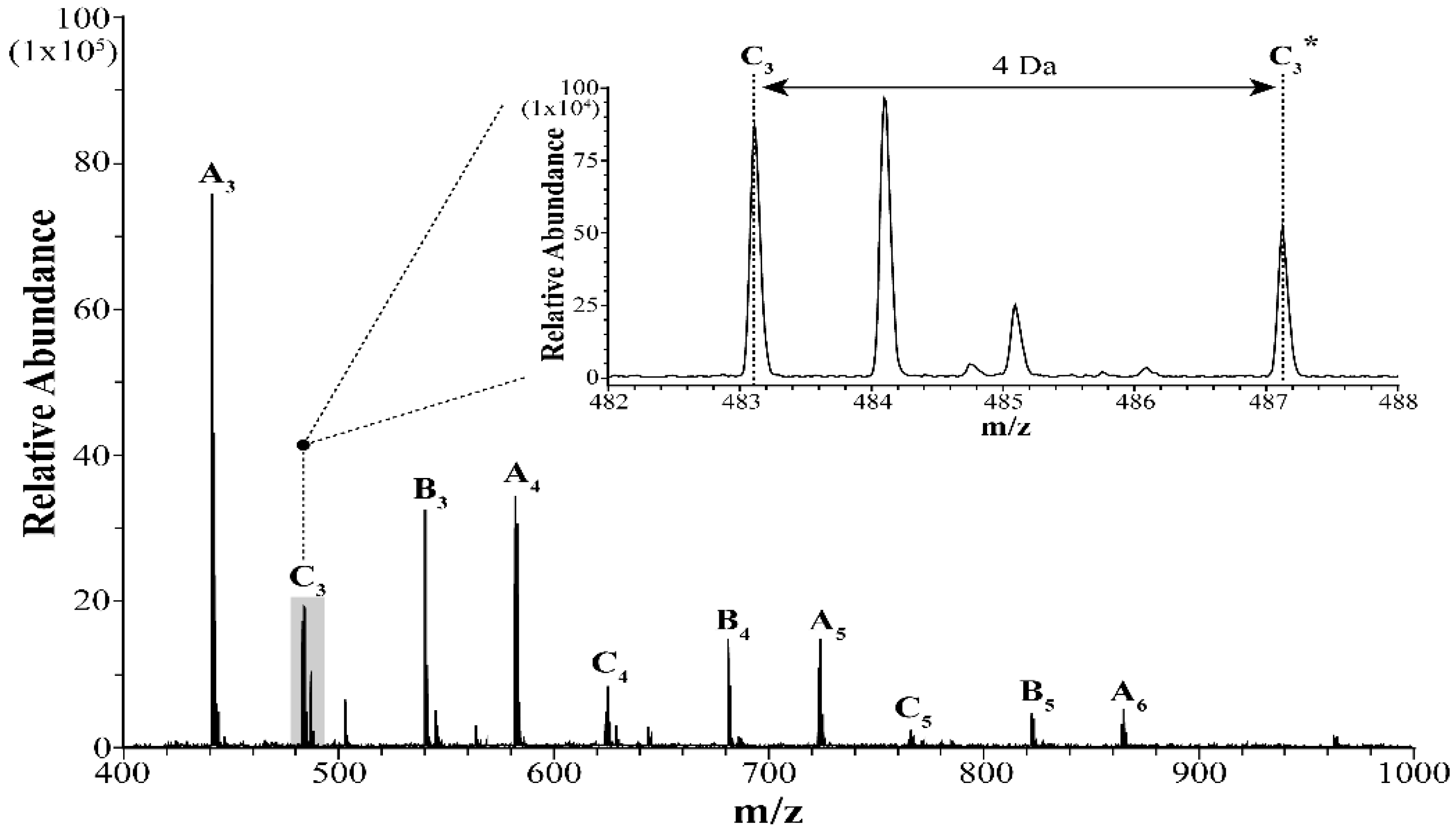
4.3. Detection of the Guanidine Oligomers
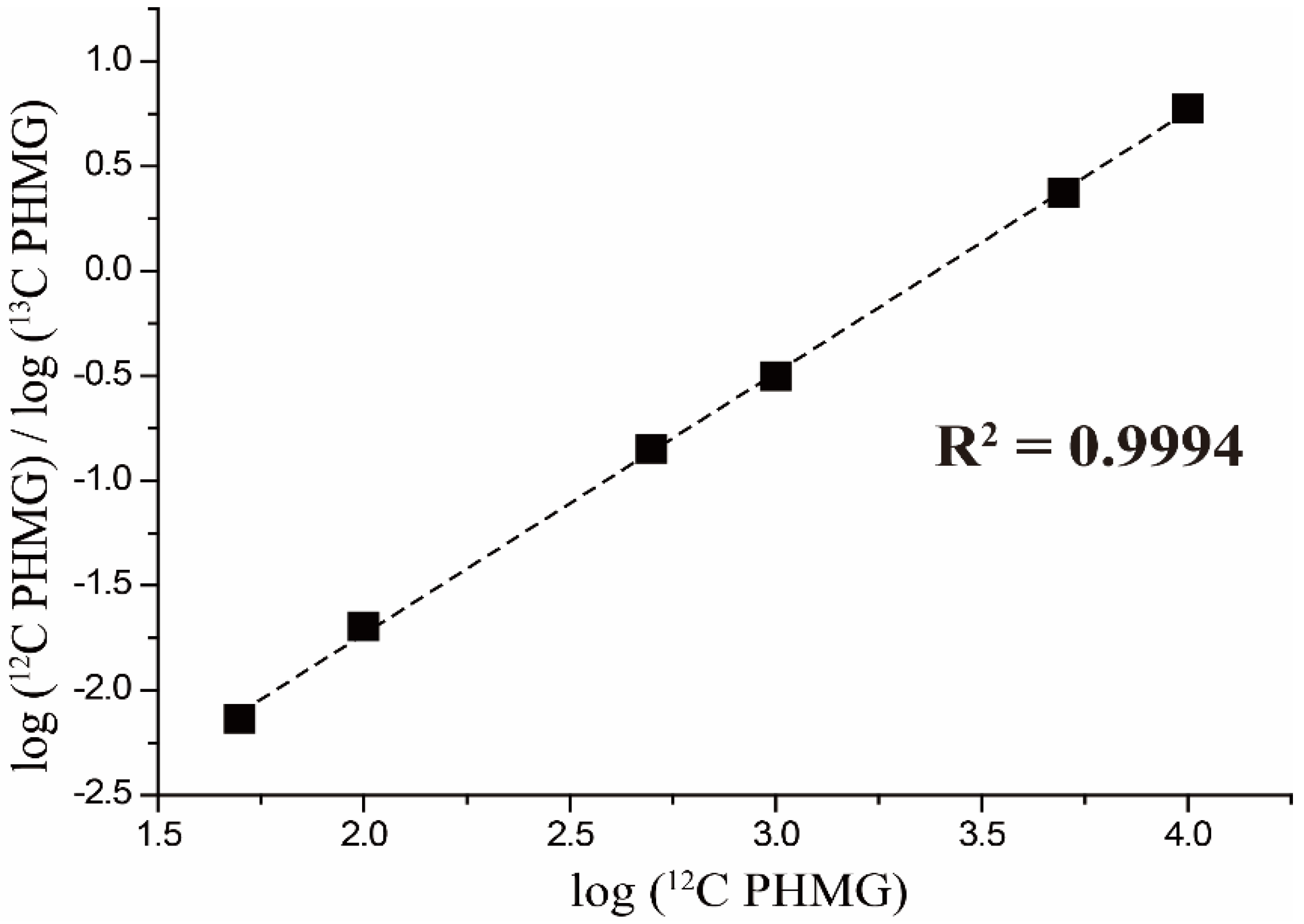
4.4. Calibration Curve
4.5. Average Molecular Mass of PHMG
4.6. Degree of Polymerization of PHMG
4.7. Data Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yang, H.J.; Kim, H.J.; Yu, J.; Lee, E.; Jung, Y.H.; Kim, H.Y.; Seo, J.H.; Kwon, G.Y.; Park, J.H.; Gwack, J. Inhalation toxicity of humidifier disinfectants as a risk factor of children’s interstitial lung disease in Korea: A case-control study. PLoS ONE 2013, 8, e64430. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.W.; Ahn, K.; Yang, H.J.; Lee, S.; Park, J.D.; Kim, W.K.; Kim, J.T.; Kim, H.H.; Rha, Y.H.; Park, Y.M. Humidifier disinfectant–associated children’s interstitial lung disease. Am. J. Resp. Crit. Care Med. 2014, 189, 48–56. [Google Scholar] [CrossRef]
- Kim, H.J.; Lee, M.S.; Hong, S.B.; Huh, J.W.; Do, K.H.; Jang, S.J.; Lim, C.M.; Chae, E.J.; Lee, H.; Jung, M. A cluster of lung injury cases associated with home humidifier use: An epidemiological investigation. Thorax 2014, 69, 703–708. [Google Scholar] [CrossRef] [PubMed]
- Park, D.U.; Ryu, S.H.; Lim, H.K.; Kim, S.K.; Choi, Y.Y.; Ahn, J.J.; Lee, E.; Hong, S.B.; Do, K.H.; Cho, J.l. Types of household humidifier disinfectant and associated risk of lung injury (HDLI) in South Korea. Sci. Total Environ. 2017, 596, 53–60. [Google Scholar] [CrossRef]
- Worley, S.; Sun, G. Biocidal polymers. Trends Polym. Sci. 1996, 11, 364–370. [Google Scholar]
- Tashiro, T. Antibacterial and bacterium adsorbing macromolecules II Macromol. Mater. Eng. 2001, 286, 63–87. [Google Scholar]
- Ascenzi, J.M. Handbook of Disinfectants and Antiseptics; CRC Press: Boca Raton, FL, USA, 1995. [Google Scholar]
- Ohta, S.; Misawa, Y.; Miyamoto, H.; Makino, M.; Nagai, K.; Shiraishi, T.; Nakagawa, Y.; Yamato, S.; Tachikawa, E.; Zenda, H. A comparative study of characteristics of current-type and conventional-type cationic bactericides. Biol. Pharm. Bull. 2001, 24, 1093–1096. [Google Scholar] [CrossRef]
- Messick, C.R.; Pendland, S.L.; Moshirfar, M.; Fiscella, R.G.; Losnedahl, K.J.; Schriever, C.A.; Schreckenberger, P.C. In-vitro activity of polyhexamethylene biguanide (PHMB) against fungal isolates associated with infective keratitis. J. Antimicrob. Chemother. 1999, 44, 297–298. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Anderson, J.; Mueller, S.; Gaines, W.; Kendall, A. Literature review—efficacy of various disinfectants against Legionella in water systems. Water Res. 2002, 36, 4433–4444. [Google Scholar] [CrossRef]
- Wei, D.; Ma, Q.; Guan, Y.; Hu, F.; Zheng, A.; Zhang, X.; Teng, Z.; Jiang, H. Structural characterization and antibacterial activity of oligoguanidine (polyhexamethylene guanidine hydrochloride). Mater. Sci. Eng. C 2009, 29, 1776–1780. [Google Scholar] [CrossRef]
- Hwang, H.J.; Nam, J.; Yang, S.I.; Kwon, J.H.; Oh, H.B. MALDI-TOF analysis of polyhexamethylene guanidine (PHMG) oligomers used as a commercial antibacterial humidifier disinfectant. Bull. Korean Chem. Soc. 2013, 34, 1708–1714. [Google Scholar] [CrossRef]
- Park, D.U. Analysis of Components of Disinfectant Contained in Humidifier Disinfectant Products; Special Commission on Social Disaster Investigation: Seoul, Korean, 2020. (In Korean) [Google Scholar]
- Korean Centers for Disease Control & Prevention (KCDC). Aerosolization of Humidifier Disinfectant; Public Health Weekly Report No. 45; Department of Epidemiology Research: Seoul, Korean, 2020; pp. 831–832. (In Korean) [Google Scholar]
- Park, D.U.; Ryu, S.H.; Roh, H.S.; Lee, E.; Cho, H.J.; Yoon, J.; Lee, S.Y.; Cho, Y.A.; Do, K.H.; Hong, S.J. Association of high-level humidifier disinfectant exposure with lung injury in preschool children. Sci. Total Environ. 2018, 616, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Jiang, J.; Chen, Y. Synthesis and antimicrobial activity of polymeric guanidine and biguanidine salts. Polymer 1999, 40, 6189–6198. [Google Scholar] [CrossRef]
- Feiertag, P.; Albert, M.; Ecker-Eckhofen, E.M.; Hayn, G.; Hönig, H.; Oberwalder, H.W.; Saf, R.; Schmidt, A.; Schmidt, O.; Topchiev, D. Structural characterization of biocidal oligoguanidines. Macromol. Rapid Commun. 2003, 24, 567–570. [Google Scholar] [CrossRef]
- Buxbaum, A.; Kratzer, C.; Graninger, W.; Georgopoulos, A. Antimicrobial and toxicological profile of the new biocide Akacid plus®. J. Antimicrob. Chemother. 2006, 58, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Müller, G.; Kramer, A. Effect of selected wound antiseptics on adult articular cartilage (bovine sesamoid bone) in the presence of Escherichia coli and Staphylococcus aureus. J. Orthop. Res. 2005, 23, 127–133. [Google Scholar] [CrossRef]
- Choi, H.; Kim, K.J.; Lee, D.G. Antifungal activity of the cationic antimicrobial polymer-polyhexamethylene guanidine hydrochloride and its mode of action. Fungal Biol. 2017, 121, 53–60. [Google Scholar] [CrossRef]
- Zhou, Z.; Wei, D.; Guan, Y.; Zheng, A.; Zhong, J. Damage of escherichia coli membrane by bactericidal agent polyhexamethylene guanidine hydrochloride: Micrographic evidences. J. Appl. Microbiol. 2010, 108, 898–907. [Google Scholar] [CrossRef]
- Albert, M.; Feiertag, P.; Hayn, G.; Saf, R.; Hönig, H. Structure−activity relationships of oligoguanidines influence of counterion, diamine, and average molecular weight on biocidal activities. Biomacromolecules 2003, 4, 1811–1817. [Google Scholar] [CrossRef]
- European Chemicals Agency (ECHA). Committee for risk assessment RAC: Annex 1: Background document to the opinion proposing harmonised classification and labelling at community level of ammoniumpentadecafluorooctanoate (APFO). Available online: https://echa.europa.eu/documents/10162/d4685869-d34d-1078-5e5a-e6e92a2736f6 (accessed on 1 June 2020).
- ChemSafetyPro. Comparison of Global Polymer Registration Requirements. Available online: https://www.chemsafetypro.com/Topics/Review/polymer_registration_in_EU_USA_China_Japan_Korea_Taiwan_Philippines.html (accessed on 13 June 2020).
- European Centre for Ecotoxicology and Toxicology of Chemicals (ECETOC). The ECETOC conceptual framework for polymer risk assessment (CF4 Polymers). Available online: http://www.ecetoc.org/wp-content/uploads/2019/06/ECETOC-TR133-1CF4Polymers.pdf (accessed on 1 June 2020).
- Lee, J.H.; Kim, Y.H.; Kwon, J.H. Fatal misuse of humidifier disinfectants in Korea: Importance of screening risk assessment and implications for management of chemicals in consumer products. Environ. Sci. Technol. 2012, 46, 2498–2500. [Google Scholar] [CrossRef]
- National Industrial Chemicals Notification and Assessment Scheme (NICAS). Polyhexamethyleneguanidine Phosphate. Available online: http://www.sudmed.ru/index.php?act=Attach&type=post&id=2882 (accessed on 1 June 2020).
- East, G.; McIntyre, J.; Shao, J. Polybiguanides: Synthesis and characterization of polybiguanides containing hexamethylene groups. Polymer 1997, 38, 3973–3984. [Google Scholar] [CrossRef]
- Ha, Y.; Kwon, J.H. Effects of lipid membrane composition on the distribution of biocidal guanidine oligomer with solid supported lipid membranes. RSC Adv. 2020, 10, 22343–22351. [Google Scholar] [CrossRef]
- Park, D.U.; Friesen, M.C.; Roh, H.S.; Choi, Y.Y.; Ahn, J.J.; Lim, H.K.; Kim, S.K.; Koh, D.H.; Jung, H.J.; Lee, J.H. Estimating retrospective exposure of household humidifier disinfectants. Indoor Air 2015, 25, 631–640. [Google Scholar] [CrossRef]
- Kennedy, G.L., Jr. Toxicity of Hexamethylenediamine (HMDA). Drug Chem. Toxicol. 2005, 28, 15–33. [Google Scholar] [CrossRef] [PubMed]
- Myers, J.L.; Grant, R.L. Development of a chronic inhalation reference value for hexamethylenediamine using an exposure model based on the dihydrochloride salt. Inhal. Toxicol. 2015, 27, 440–449. [Google Scholar] [CrossRef] [PubMed]
- Park, D.U.; Choi, Y.Y.; Ahn, J.J.; Lim, H.K.; Kim, S.K.; Roh, H.S.; Cheong, H.K.; Leem, J.H.; Koh, D.H.; Jung, H.J. Relationship between exposure to household humidifier disinfectants and risk of lung injury: A family-based study. PLoS ONE 2015, 10, e0124610. [Google Scholar] [CrossRef] [PubMed]
- Yoon, D.; Lee, D.; Lee, J.H.; Cha, S.; Oh, H.B. Quantitative analysis of polyhexamethylene guanidine (PHMG) oligomers via matrix-assisted laser desorption/ionization time-of-flight mass spectrometry with an ionic-liquid matrix. Rapid Commun. Mass Sp. 2015, 29, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.; Park, M.; Lee, J.; Song, I.; Ju, Y.; Lee, C.S.; Kwon, J.H.; Moon, B.; Oh, H.B. Quantitative MALDI-TOF mass spectrometric analysis of biocidal polyhexamethylene guanidine (PHMG) oligomers in consumer products. Int. J. Mass Spectrom. 2019, 435, 298–304. [Google Scholar] [CrossRef]
- Nielen, M.W. MALDI time-of-flight mass spectrometry of synthetic polymers. Mass Spectrom. Rev. 1999, 18, 309–344. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
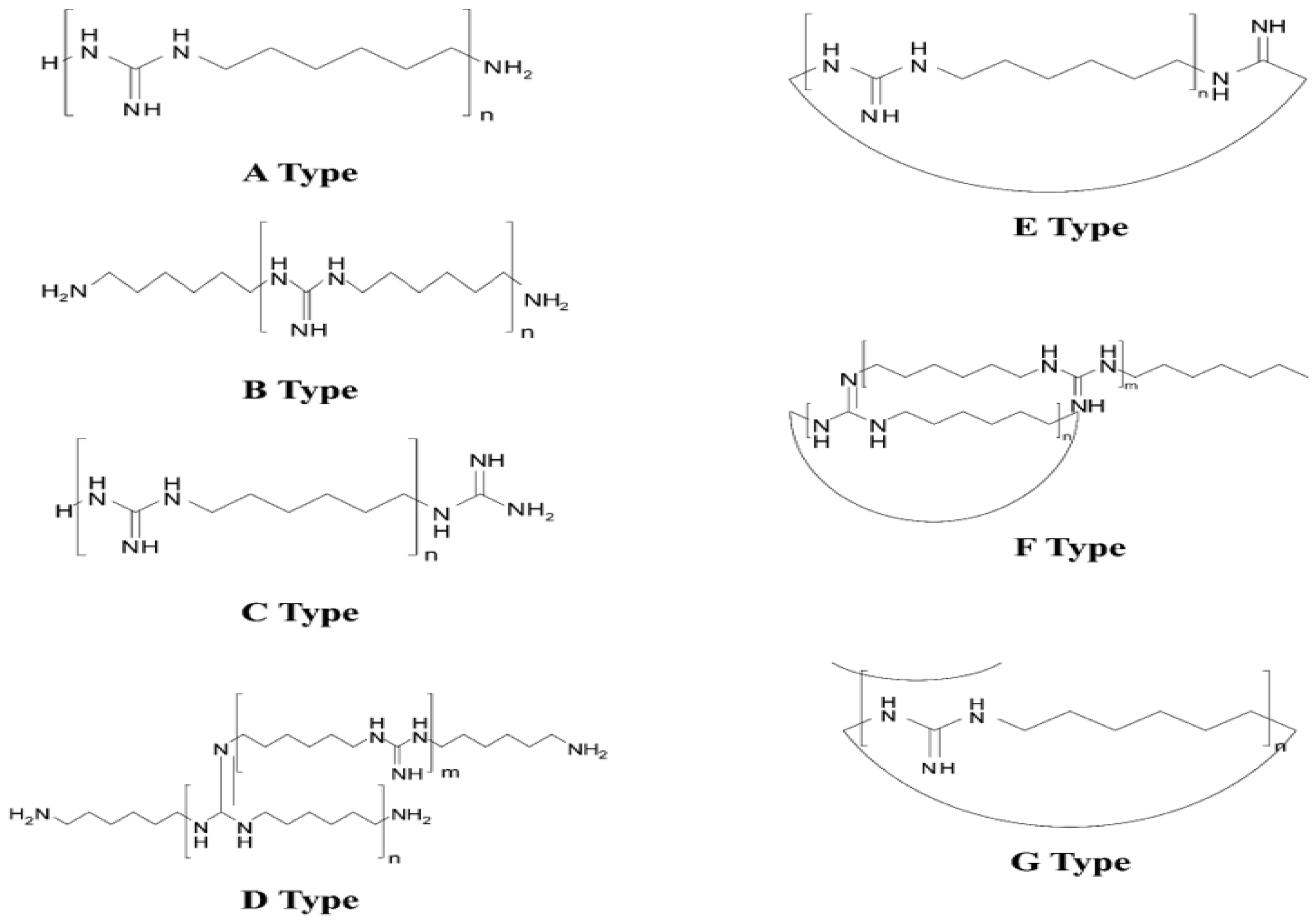
| Brand Name | Sales Period | Total Sales Volume (mL/each) [13] | Cases of HDLI 1 |
|---|---|---|---|
| Oxy Saksak (Old and New versions) | 2000–2011 | 9,561,151 of 500 mL | Yes |
| Lotte Wiselect | 2006–2011 | 110,283 of 1000 mL | Yes |
| Homeplus | 2004–2011 | 296,950 of 550 mL | Yes |
| Atorganic | 2009–2011 | No information | Yes |
| Cefu | 2009–2011 | No information | Yes |
| Vegetable Home Clean Up | 2009–2011 | 106,943 of 1000 mL | No |
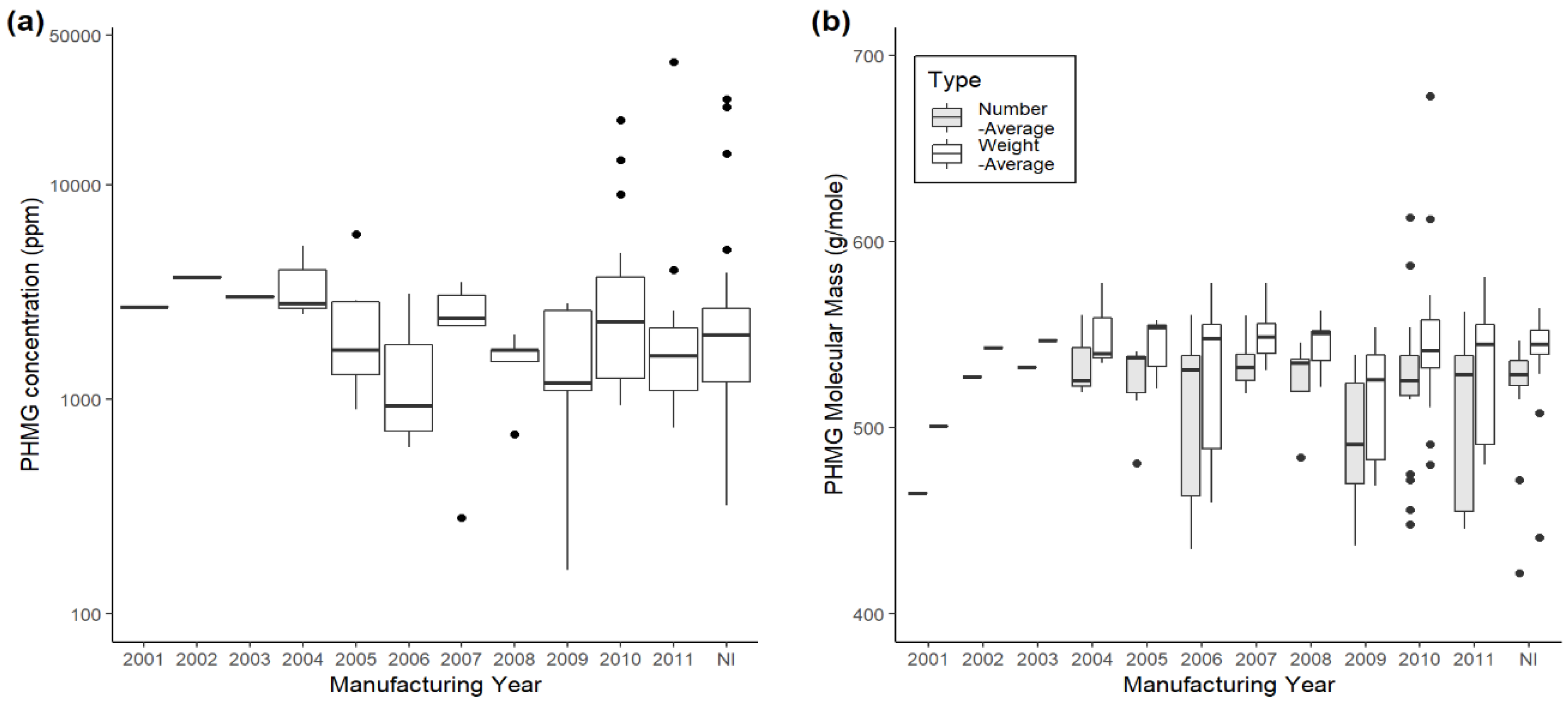
| Brand Name | Year | Number of Sample(s) | Concentration (ppm) | ||
|---|---|---|---|---|---|
| Mean | SD | Range | |||
| Cleanland | NI | 1 | 1500.0 | N/A | N/A |
| Oxy Saksak New | 2004 | 3 | 3500.0 | 1479.9 | 2500–5200 |
| 2005 | 7 | 2400.0 | 1724.3 | 900–5900 | |
| 2006 | 8 | 1431.3 | 820.1 | 670–3100 | |
| 2007 | 7 | 2382.9 | 1051.4 | 280–3500 | |
| 2008 | 4 | 1725.0 | 206.2 | 1500–2000 | |
| 2009 | 7 | 1914.3 | 803.0 | 1100–2800 | |
| 2010 | 14 | 2707.1 | 2025.0 | 1100–9000 | |
| 2011 | 14 | 1595.7 | 592.2 | 740–2600 | |
| NI | 15 | 1737.3 | 964.2 | 320–3900 | |
| Subtotal | 79 | 2051.0 | 1293.0 | 280–9000 | |
| Oxy | 2001 | 1 | 2700.0 | N/A | N/A |
| 2002 | 1 | 3700.0 | N/A | N/A | |
| 2003 | 1 | 3000.0 | N/A | N/A | |
| NI | 1 | 5000.0 | N/A | N/A | |
| Subtotal | 4 | 3600.0 | 1023.1 | 2700–5000 | |
| Vegetable Home Clean Up | 2009 | 1 | 160.0 | N/A | N/A |
| 2010 | 3 | 12,600.0 | 7607.9 | 4800–20,000 | |
| NI | 3 | 20,666.7 | 5859.5 | 14,000–25,000 | |
| Subtotal | 7 | 14,280.0 | 9261.4 | 160–25,000 | |
| Cefu | 2011 | 2 | 19,700.0 | 24,748.7 | 2200–37,200 |
| NI | 1 | 1600.0 | - | 1600 | |
| Subtotal | 3 | 13,666.7 | 20,382.7 | 1600–37,200 | |
| Atorganic | NI | 1 | 560.0 | N/A | N/A |
| Lotte Wiselect | 2006 | 2 | 660.0 | 84.9 | 600–7200 |
| 2008 | 1 | 690.0 | N/A | N/A | |
| 2010 | 3 | 1146.7 | 185.8 | 940–1300 | |
| 2011 | 1 | 1500.0 | N/A | N/A | |
| Subtotal | 7 | 992.9 | 345.9 | 600–1500 | |
| Homeplus | 2009 | 1 | 1200.0 | N/A | N/A |
| 2011 | 6 | 1800.0 | 1143.7 | 1000–4000 | |
| NI | 1 | 2700.0 | N/A | N/A | |
| Subtotal | 8 | 1837.5 | 1048.7 | 1000–4000 | |
| Total | 110 | 3100.9 | 5102.8 | 160–37,200 | |
| Brand Name | Year | Number of Sample (s) | Average Molecular Weight (g/mol) | |||||
|---|---|---|---|---|---|---|---|---|
| Number (Mn) | Weight (Mw) | |||||||
| Mean | SD | Range | Mean | SD | Range | |||
| Cleanland | NI | 1 | 422.0 | - | 422.0 | 441.0 | - | 441.0 |
| Oxy Saksak New | 2004 | 3 | 535.0 | 22.4 | 519.0–560.6 | 551.0 | 23.5 | 535–578 |
| 2005 | 7 | 524.8 | 21.7 | 481.0–541.1 | 544.1 | 14.9 | 521–558 | |
| 2006 | 8 | 503.2 | 53.4 | 435.0–560.8 | 523.4 | 48.7 | 460–578 | |
| 2007 | 7 | 534.4 | 14.3 | 518.5–560.3 | 550.0 | 15.9 | 531–578 | |
| 2008 | 4 | 534.2 | 10.9 | 519.5–545.7 | 550.5 | 11.1 | 536–563 | |
| 2009 | 7 | 501.9 | 37.4 | 437.0–539.0 | 525.0 | 29.6 | 469–554 | |
| 2010 | 14 | 521.3 | 41.7 | 448.0–613.0 | 545.1 | 45.9 | 480–678 | |
| 2011 | 14 | 510.3 | 46.1 | 448.0–562.2 | 532.4 | 39.2 | 480–581 | |
| NI | 15 | 533.1 | 7.3 | 520.3–545.9 | 549.5 | 7.8 | 537–564 | |
| Subtotal | 79 | 520.7 | 35.1 | 435.0–613.0 | 540.6 | 32.5 | 460–678 | |
| Oxy | 2001 | 1 | 465.0 | N/A | N/A | 501.0 | N/A | N/A |
| 2002 | 1 | 527.4 | N/A | N/A | 543.0 | N/A | N/A | |
| 2003 | 1 | 532.7 | N/A | N/A | 547.0 | N/A | N/A | |
| NI | 1 | 531.7 | N/A | N/A | 546.0 | N/A | N/A | |
| Subtotal | 4 | 514.2 | 32.9 | 465.0–532.7 | 534.3 | 22.2 | 501–547 | |
| Vegetable Home Clean Up | 2009 | 1 | 483.0 | N/A | N/A | 483.0 | N/A | N/A |
| 2010 | 3 | 542.4 | 38.9 | 515.2–587.0 | 562.7 | 43.7 | 529–612 | |
| NI | 3 | 516.7 | 1.4 | 515.4–518.3 | 530.0 | 1.0 | 529–531 | |
| Subtotal | 7 | 522.9 | 31.3 | 483.0–587.0 | 537.3 | 38.4 | 483–612 | |
| Cefu | 2011 | 2 | 516.0 | 2.5 | 514.2–517.7 | 528.5 | 0.7 | 528–529 |
| NI | 1 | 472.0 | N/A | N/A | 508.0 | N/A | N/A | |
| Subtotal | 3 | 501.3 | 25.4 | 472.0–517.7 | 521.7 | 11.8 | 508–529 | |
| Atorganic | NI | 1 | 546.9 | N/A | N/A | 562.0 | N/A | N/A |
| Lotte Wiselect | 2006 | 2 | 531.4 | 7.1 | 526.4–536.5 | 548.0 | 8.5 | 542–554 |
| 2008 | 1 | 484.0 | N/A | N/A | 522.0 | N/A | N/A | |
| 2010 | 3 | 517.5 | 39.1 | 475.0–551.9 | 540.3 | 29.0 | 511–569 | |
| 2011 | 1 | 533.0 | N/A | N/A | 549.0 | N/A | N/A | |
| Subtotal | 7 | 518.9 | 28.4 | 475.0–551.9 | 541.1 | 19.5 | 511–569 | |
| Homeplus | 2009 | 1 | 444.0 | N/A | N/A | 475.0 | N/A | N/A |
| 2011 | 6 | 493.9 | 41.1 | 446.0–532.7 | 522.0 | 28.4 | 484–549 | |
| NI | 1 | 527.0 | N/A | N/A | 542.0 | N/A | N/A | |
| Subtotal | 8 | 491.8 | 41.4 | 444.0–532.7 | 518.6 | 30.6 | 475–549 | |
| Total | 110 | 517.2 | 35.9 | 422.0–613.0 | 537.3 | 32.7 | 441–678 | |
| Brand Name | Year | Number of Sample(s) | Mean | SD | Range |
|---|---|---|---|---|---|
| Cleanland | NI | 1 | 2.05 | - | 2.05 |
| Oxy Saksak New | 2004 | 3 | 3.79 | 0.16 | 3.68–3.98 |
| 2005 | 7 | 3.57 | 0.55 | 2.33–3.84 | |
| 2006 | 8 | 3.20 | 0.88 | 2.11–3.98 | |
| 2007 | 7 | 3.79 | 0.10 | 3.68–3.97 | |
| 2008 | 4 | 3.79 | 0.08 | 3.68–3.87 | |
| 2009 | 7 | 3.11 | 0.80 | 2.12–3.82 | |
| 2010 | 14 | 3.38 | 0.66 | 2.17–3.93 | |
| 2011 | 14 | 3.26 | 0.83 | 2.16–3.99 | |
| NI | 15 | 3.78 | 0.05 | 3.69–3.87 | |
| Subtotal | 79 | 3.48 | 0.63 | 2.11–3.99 | |
| Oxy | 2001 | 1 | 2.26 | N/A | N/A |
| 2002 | 1 | 3.74 | N/A | N/A | |
| 2003 | 1 | 3.78 | N/A | N/A | |
| NI | 1 | 3.77 | N/A | N/A | |
| Subtotal | 4 | 3.39 | 0.75 | 2.26–3.78 | |
| Vegetable Home Clean up | 2009 | 1 | 3.43 | N/A | N/A |
| 2010 | 3 | 3.02 | 0.57 | 2.55–3.65 | |
| NI | 3 | 3.66 | 0.01 | 3.66–3.68 | |
| Subtotal | 7 | 3.35 | 0.46 | 2.55–3.68 | |
| Cefu | 2011 | 2 | 3.66 | 0.02 | 3.65–3.67 |
| NI | 1 | 2.29 | N/A | N/A | |
| Subtotal | 3 | 3.20 | 0.79 | 2.29–3.67 | |
| Atorganic | NI | 1 | 3.88 | N/A | N/A |
| Lotte Wiselect | 2006 | 2 | 3.77 | 0.05 | 3.73–3.80 |
| 2008 | 1 | 2.35 | NA | NA | |
| 2010 | 3 | 3.31 | 0.88 | 2.30–3.91 | |
| 2011 | 1 | 3.78 | NA | NA | |
| Subtotal | 7 | 3.37 | 0.72 | 2.30–3.91 | |
| Homeplus | 2009 | 1 | 2.15 | NA | NA |
| 2011 | 6 | 2.99 | 0.85 | 2.16–3.78 | |
| NI | 1 | 3.74 | NA | NA | |
| Subtotal | 8 | 2.98 | 0.83 | 2.15–3.78 | |
| Total | 110 | 3.41 | 0.66 | 2.05–3.99 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, D.-U.; Park, J.; Yang, K.W.; Park, J.-H.; Kwon, J.-H.; Oh, H.B. Properties of Polyhexamethylene Guanidine (PHMG) Associated with Fatal Lung Injury in Korea. Molecules 2020, 25, 3301. https://doi.org/10.3390/molecules25143301
Park D-U, Park J, Yang KW, Park J-H, Kwon J-H, Oh HB. Properties of Polyhexamethylene Guanidine (PHMG) Associated with Fatal Lung Injury in Korea. Molecules. 2020; 25(14):3301. https://doi.org/10.3390/molecules25143301
Chicago/Turabian StylePark, Dong-Uk, Jihoon Park, Kee Won Yang, Ju-Hyun Park, Jung-Hwan Kwon, and Han Bin Oh. 2020. "Properties of Polyhexamethylene Guanidine (PHMG) Associated with Fatal Lung Injury in Korea" Molecules 25, no. 14: 3301. https://doi.org/10.3390/molecules25143301
APA StylePark, D.-U., Park, J., Yang, K. W., Park, J.-H., Kwon, J.-H., & Oh, H. B. (2020). Properties of Polyhexamethylene Guanidine (PHMG) Associated with Fatal Lung Injury in Korea. Molecules, 25(14), 3301. https://doi.org/10.3390/molecules25143301






