The Role of Percent Volume Buried in the Characterization of Copper(I) Complexes for Lighting Purposes
Abstract
1. Introduction
2. Results and Discussion
2.1. General Strategies
2.2. Macroscopic Level
2.3. Case Study 1: Importance of Steric Effects
2.4. Case Study 2—Investigating Electronic Effects
2.5. Case Study 3—Differentiating Steric from Electronic Factors
3. Materials and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Strohmeier, W.; Müller, F.-J. Klassifizierung phosphorhaltiger Liganden in Metallcarbonyl-Derivaten nach der π-Acceptorstärke. Chemische Berichte 1967, 100, 2812–2821. [Google Scholar] [CrossRef]
- Tolman, C.A. Steric effects of phosphorus ligands in organometallic chemistry and homogeneous catalysis. Chem. Rev. 1977, 77, 313–348. [Google Scholar] [CrossRef]
- Poater, A.; Ragone, F.; Giudice, S.; Costabile, C.; Dorta, R.; Nolan, S.P.; Cavallo, L. Thermodynamics of N-Heterocyclic Carbene Dimerization: The Balance of Sterics and Electronics. Organometallics 2008, 27, 2679–2681. [Google Scholar] [CrossRef]
- Poater, A.; Ragone, F.; Mariz, R.; Dorta, R.; Cavallo, L. Comparing the Enantioselective Power of Steric and Electrostatic Effects in Transition-Metal-Catalyzed Asymmetric Synthesis. Chem. Eur. J. 2010, 16, 14348–14353. [Google Scholar] [CrossRef] [PubMed]
- Falivene, L.; Cao, Z.; Petta, A.; Serra, L.; Poater, A.; Oliva, R.; Scarano, V.; Cavallo, L. Towards the online computer-aided design of catalytic pockets. Nat. Chem. 2019, 11, 872–879. [Google Scholar] [CrossRef]
- Clavier, H.; Correa, A.; Cavallo, L.; Escudero-Adán, E.C.; Benet-Buchholz, J.; Slawin, A.M.Z.; Nolan, S.P. [Pd(NHC)(allyl)Cl] Complexes: Synthesis and Determination of the NHC Percent Buried Volume (%Vbur) Steric Parameter. Eur. J. Inorg. Chem. 2009, 2009, 1767–1773. [Google Scholar] [CrossRef]
- Clavier, H.; Nolan, S.P. Percent buried volume for phosphine and N-heterocyclic carbene ligands: Steric properties in organometallic chemistry. Chem. Commun. 2010, 46, 841–861. [Google Scholar] [CrossRef]
- Gómez-Suárez, A.; Nelson, D.J.; Nolan, S.P. Quantifying and understanding the steric properties of N-heterocyclic carbenes. Chem. Commun. 2017, 53, 2650–2660. [Google Scholar] [CrossRef]
- Cervantes-Reyes, A.; Rominger, F.; Hashmi, A.S.K. Sterically Demanding AgI and CuI N-Heterocyclic Carbene Complexes: Synthesis, Structures, Steric Parameters, and Catalytic Activity. Chem. Eur. J. 2020, 26, 5530–5540. [Google Scholar] [CrossRef]
- Armaroli, N.; Accorsi, G.; Holler, M.; Moudam, O.; Nierengarten, J.-F.; Zhou, Z.; Wegh, R.T.; Welter, R. Highly Luminescent CuI Complexes for Light-Emitting Electrochemical Cells. Adv. Mater. 2006, 18, 1313–1316. [Google Scholar] [CrossRef]
- Costa, R.D.; Tordera, D.; Ortí, E.; Bolink, H.J.; Schönle, J.; Graber, S.; Housecroft, C.E.; Constable, E.C.; Zampese, J.A. Copper(I) complexes for sustainable light-emitting electrochemical cells. J. Mater. Chem. 2011, 21, 16108–16118. [Google Scholar] [CrossRef]
- Costa, R.D.; Ortí, E.; Bolink, H.J.; Monti, F.; Accorsi, G.; Armaroli, N. Luminescent Ionic Transition-Metal Complexes for Light-Emitting Electrochemical Cells. Angew. Chem. Int. Ed. 2012, 51, 8178–8211. [Google Scholar] [CrossRef] [PubMed]
- Hofbeck, T.; Monkowius, U.; Yersin, H. Highly Efficient Luminescence of Cu(I) Compounds: Thermally Activated Delayed Fluorescence Combined with Short-Lived Phosphorescence. J. Am. Chem. Soc. 2015, 137, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Elie, M.; Sguerra, F.; Di Meo, F.; Weber, M.D.; Marion, R.; Grimault, A.; Lohier, J.-F.; Stallivieri, A.; Brosseau, A.; Pansu, R.B.; et al. Designing NHC–Copper(I) Dipyridylamine Complexes for Blue Light-Emitting Electrochemical Cells. ACS Appl. Mater. Interfaces 2016, 8, 14678–14691. [Google Scholar] [CrossRef] [PubMed]
- Hamze, R.; Shi, S.; Kapper, S.C.; Muthiah Ravinson, D.S.; Estergreen, L.; Jung, M.-C.; Tadle, A.C.; Haiges, R.; Djurovich, P.I.; Peltier, J.L.; et al. “Quick-Silver” from a Systematic Study of Highly Luminescent, Two-Coordinate, d10 Coinage Metal Complexes. J. Am. Chem. Soc. 2019, 141, 8616–8626. [Google Scholar] [CrossRef] [PubMed]
- Hamze, R.; Peltier, J.L.; Sylvinson, D.; Jung, M.; Cardenas, J.; Haiges, R.; Soleilhavoup, M.; Jazzar, R.; Djurovich, P.I.; Bertrand, G.; et al. Eliminating nonradiative decay in Cu(I) emitters:>99% quantum efficiency and microsecond lifetime. Science 2019, 363, 601–606. [Google Scholar] [CrossRef]
- Keller, S.; Constable, E.C.; Housecroft, C.E.; Neuburger, M.; Prescimone, A.; Longo, G.; Pertegás, A.; Sessolo, M.; Bolink, H.J. [Cu(bpy)(P^P)]+ containing light-emitting electrochemical cells: Improving performance through simple substitution. Dalton Trans. 2014, 43, 16593–16596. [Google Scholar] [CrossRef]
- Keller, S.; Pertegás, A.; Longo, G.; Martínez, L.; Cerdá, J.; Junquera-Hernández, J.M.; Prescimone, A.; Constable, E.C.; Housecroft, C.E.; Ortí, E.; et al. Shine bright or live long: Substituent effects in [Cu(N^N)(P^P)]+-based light-emitting electrochemical cells where N^N is a 6-substituted 2,2′-bipyridine. J. Mater. Chem. C 2016, 4, 3857–3871. [Google Scholar] [CrossRef]
- Keller, S.; Alkan-Zambada, M.; Prescimone, A.; Constable, E.C.; Housecroft, C.E. Extended π-Systems in Diimine Ligands in [Cu(P^P)(N^N)][PF6] Complexes: From 2,2′-Bipyridine to 2-(Pyridin-2-yl)Quinoline. Crystals 2020, 10, 255. [Google Scholar] [CrossRef]
- Smith, C.S.; Branham, C.W.; Marquardt, B.J.; Mann, K.R. Oxygen Gas Sensing by Luminescence Quenching in Crystals of Cu(xantphos)(phen)+ Complexes. J. Am. Chem. Soc. 2010, 132, 14079–14085. [Google Scholar] [CrossRef]
- Li, X.-L.; Ai, Y.-B.; Yang, B.; Chen, J.; Tan, M.; Xin, X.-L.; Shi, Y.-H. Syntheses, structures and photophysical properties of a series of luminescent copper(I) mixed-ligand complexes. Polyhedron 2012, 35, 47–54. [Google Scholar] [CrossRef]
- Tsubomura, T.; Kimura, K.; Nishikawa, M.; Tsukuda, T. Structures and photophysical properties of copper(I) complexes bearing diphenylphenanthroline and bis(diphenylphosphino)alkane: The effect of phenyl groups on the phenanthroline ligand. Dalton Trans. 2015, 44, 7554–7562. [Google Scholar] [CrossRef] [PubMed]
- Cabrera, A.R.; Gonzalez, I.A.; Cortés-Arriagada, D.; Natali, M.; Berke, H.; Daniliuc, C.G.; Camarada, M.B.; Toro-Labbé, A.; Rojas, R.S.; Salas, C.O. Synthesis of new phosphorescent imidoyl-indazol and phosphine mixed ligand Cu(I) complexes – structural characterization and photophysical properties. RSC Adv. 2016, 6, 5141–5153. [Google Scholar] [CrossRef]
- Brunner, F.; Martínez-Sarti, L.; Keller, S.; Pertegás, A.; Prescimone, A.; Constable, E.C.; Bolink, H.J.; Housecroft, C.E. Peripheral halo-functionalization in [Cu(N^N)(P^P)]+ emitters: Influence on the performances of light-emitting electrochemical cells. Dalton Trans. 2016, 45, 15180–15192. [Google Scholar] [CrossRef] [PubMed]
- Keller, S.; Brunner, F.; Junquera-Hernández, J.M.; Pertegás, A.; La-Placa, M.-G.; Prescimone, A.; Constable, E.C.; Bolink, H.J.; Ortí, E.; Housecroft, C.E. CF3 Substitution of [Cu(P^P)(bpy)][PF6] Complexes: Effects on Photophysical Properties and Light-Emitting Electrochemical Cell Performance. ChemPlusChem 2018, 83, 217–229. [Google Scholar] [CrossRef] [PubMed]
- Alkan-Zambada, M.; Keller, S.; Martínez-Sarti, L.; Prescimone, A.; Junquera-Hernández, J.M.; Constable, E.C.; Bolink, H.J.; Sessolo, M.; Ortí, E.; Housecroft, C.E. [Cu(P^P)(N^N)][PF6] compounds with bis(phosphane) and 6-alkoxy, 6-alkylthio, 6-phenyloxy and 6-phenylthio-substituted 2,2′-bipyridine ligands for light-emitting electrochemical cells. J. Mater. Chem. C 2018, 6, 8460–8471. [Google Scholar] [CrossRef]
- Fresta, E.; Volpi, G.; Milanesio, M.; Garino, C.; Barolo, C.; Costa, R.D. Novel Ligand and Device Designs for Stable Light-Emitting Electrochemical Cells Based on Heteroleptic Copper(I) Complexes. Inorg. Chem. 2018, 57, 10469–10479. [Google Scholar] [CrossRef]
- Groom, C.R.; Bruno, I.J.; Lightfoot, M.P.; Ward, S.C. The Cambridge Structural Database. Acta Crystallogr. 2016, B72, 171–179. [Google Scholar] [CrossRef]
- Bruno, I.J.; Cole, J.C.; Edgington, P.R.; Kessler, M.; Macrae, C.F.; McCabe, P.; Pearson, J.; Taylor, R. New software for searching the Cambridge Structural Database and visualizing crystal structures. Acta Crystallogr. 2002, B58, 389–397. [Google Scholar] [CrossRef]
- Linfoot, C.L.; Leitl, M.J.; Richardson, P.; Rausch, A.F.; Chepelin, O.; White, F.J.; Yersin, H.; Robertson, N. Thermally Activated Delayed Fluorescence (TADF) and Enhancing Photoluminescence Quantum Yields of [CuI(diimine)(diphosphine)]+ Complexes—Photophysical, Structural, and Computational Studies. Inorg. Chem. 2014, 53, 10854–10861. [Google Scholar] [CrossRef]
- Nishikawa, M.; Wakita, Y.; Nishi, T.; Miura, T.; Tsubomura, T. Long-lived and oxygen-responsive photoluminescence in the solid state of copper(i) complexes bearing fluorinated diphosphine and bipyridine ligands. Dalton Trans. 2015, 44, 9170–9181. [Google Scholar] [CrossRef] [PubMed]
- Keller, S.; Prescimone, A.; Bolink, H.; Sessolo, M.; Longo, G.; Martínez-Sarti, L.; Junquera-Hernández, J.M.; Constable, E.C.; Ortí, E.; Housecroft, C.E. Luminescent copper(i) complexes with bisphosphane and halogen-substituted 2,2′-bipyridine ligands. Dalton Trans. 2018, 47, 14263–14276. [Google Scholar] [CrossRef] [PubMed]
- Brunner, F.; Babaei, A.; Pertegás, A.; Junquera-Hernández, J.M.; Prescimone, A.; Constable, E.C.; Bolink, H.J.; Sessolo, M.; Ortí, E.; Housecroft, C.E. Phosphane tuning in heteroleptic [Cu(N^N)(P^P)]+ complexes for light-emitting electrochemical cells. Dalton Trans. 2019, 48, 446–460. [Google Scholar] [CrossRef] [PubMed]
- Keller, S.; Prescimone, A.; Constable, E.C.; Housecroft, C.E. Copper(i) and silver(i) complexes of 9,9-dimethyl-4,5-bis(di-tert-butylphosphino)xanthene: Photophysical properties and structural rigidity under pressure. Photochem. Photobiol. Sci. 2018, 17, 375–385. [Google Scholar] [CrossRef]
- Brunner, F.; Graber, S.; Baumgartner, Y.; Häussinger, D.; Prescimone, A.; Constable, E.C.; Housecroft, C.E. The effects of introducing sterically demanding aryl substituents in [Cu(N^N)(P^P)]+ complexes. Dalton Trans. 2017, 46, 6379–6391. [Google Scholar] [CrossRef]
- Mazzeo, F.; Brunner, F.; Prescimone, A.; Constable, C.E.; Housecroft, E.C. Intra-Cation versus Inter-Cation π-Contacts in [Cu(P^P)(N^N)][PF6] Complexes. Crystals 2020, 10, 1. [Google Scholar] [CrossRef]
- Nohara, I.; Keller, A.; Tarassenko, N.; Prescimone, A.; Constable, C.E.; Housecroft, E.C. Heteroleptic [Cu(P^P)(N^N)][PF6] Compounds with Isomeric Dibromo-1,10-Phenanthroline Ligands. Inorganics 2020, 8, 4. [Google Scholar] [CrossRef]
- Keller, S.; Bantle, M.; Prescimone, A.; Constable, C.E.; Housecroft, E.C. Hinged and Wide: A New P^P Ligand for Emissive [Cu(P^P)(N^N)][PF6] Complexes. Molecules 2019, 24, 3934. [Google Scholar] [CrossRef]
- Murray, N.S.; Keller, S.; Constable, E.C.; Housecroft, C.E.; Neuburger, M.; Prescimone, A. [Cu(N^N)(P^P)]+ complexes with 2,2′:6′,2′′-terpyridine ligands as the N^N domain. Dalton Trans. 2015, 44, 7626–7633. [Google Scholar] [CrossRef]
- Weber, M.D.; Viciano-Chumillas, M.; Armentano, D.; Cano, J.; Costa, R.D. σ-Hammett parameter: A strategy to enhance both photo- and electro-luminescence features of heteroleptic copper(I) complexes. Dalton Trans. 2017, 46, 6312–6323. [Google Scholar] [CrossRef]
- Nishikawa, M.; Tsubomura, T. Photophysical Properties of a Copper(I) Complex Bearing 4,4′-Dimethoxy-2,2′-bipyridine and 1,3-Bis(diphenylphosphino)propane. Bull. Chem. Soc. Jpn. 2014, 87, 912–914. [Google Scholar] [CrossRef]
- Arnosti, N.; Brunner, F.; Susic, I.; Keller, S.; Junquera-Hernández, J.M.; Prescimone, A.; Bolink, H.J.; Sessolo, M.; Ortí, E.; Housecroft, C.E.; et al. Remote Modification of Bidentate Phosphane Ligands Controlling the Photonic Properties in Their Complexes: Enhanced Performance of [Cu(RN-xantphos)(N^N)][PF6] in Light-Emitting Electrochemical Cells. Adv. Opt. Mater. 2020, 1901689. [Google Scholar] [CrossRef]
- González, I.A.; Henríquez, M.A.; Cortés-Arriagada, D.; Natali, M.; Daniliuc, C.G.; Dreyse, P.; Maze, J.; Rojas, R.S.; Salas, C.O.; Cabrera, A.R. Heteroleptic Cu(i) complexes bearing methoxycarbonyl-imidoylindazole and POP ligands—An experimental and theoretical study of their photophysical properties. New J. Chem. 2018, 42, 12576–12586. [Google Scholar] [CrossRef]
- Yang, M.; Li, X.-N.; Jia, J.-H.; Chen, X.-L.; Lu, C.-Z. A rationally designed vapoluminescent compound with adsorptive channels and responsive luminophores for volatile organic compounds (VOCs). Dalton Trans. 2019, 48, 1179–1183. [Google Scholar] [CrossRef]
- Zhu, N.; Wang, G.; Lin, S.; Li, Z.-F.; Xin, X.-L.; Yang, Y.-P.; Liu, M.; Jin, Q.-H. New discovery in crystallography: Correlation of terahertz time-domain spectra with crystal structures and photoluminescence properties of mononuclear/binuclear diimine–Cu(i)-phosphine complexes. CrystEngComm 2019, 21, 4275–4288. [Google Scholar] [CrossRef]
- Kuang, X.-N.; Lin, S.; Liu, J.-M.; Han, H.-L.; Liu, M.; Xin, X.-L.; Yang, Y.-P.; Li, Z.-F.; Jin, Q.-H.; Li, S.-F.; et al. Heteroleptic Cu(I) complexes with aromatic diimines and phosphines: Synthesis, structure, photophysical properties and THz time domain spectroscopy. Polyhedron 2019, 165, 51–62. [Google Scholar] [CrossRef]
- Liu, R.; Huang, M.-M.; Yao, X.-X.; Li, H.-H.; Yang, F.-L.; Li, X.-L. Synthesis, structures and aggregation-induced emissive properties of copper(I) complexes with 1H-imidazo[4,5-f][1,10]phenanthroline derivative and diphosphine as ligands. Inorg. Chim. Acta 2015, 434, 172–180. [Google Scholar] [CrossRef]
- Meyer, M.; Brunner, F.; Prescimone, A.; Constable, E.C.; Housecroft, C.E. Chimera Diimine Ligands in Emissive [Cu(P^P)(N^N)][PF6] Complexes. Inorganics 2020, 8, 33. [Google Scholar] [CrossRef]
Sample Availability: No new compounds were prepared in this work; no samples are available from the authors. |
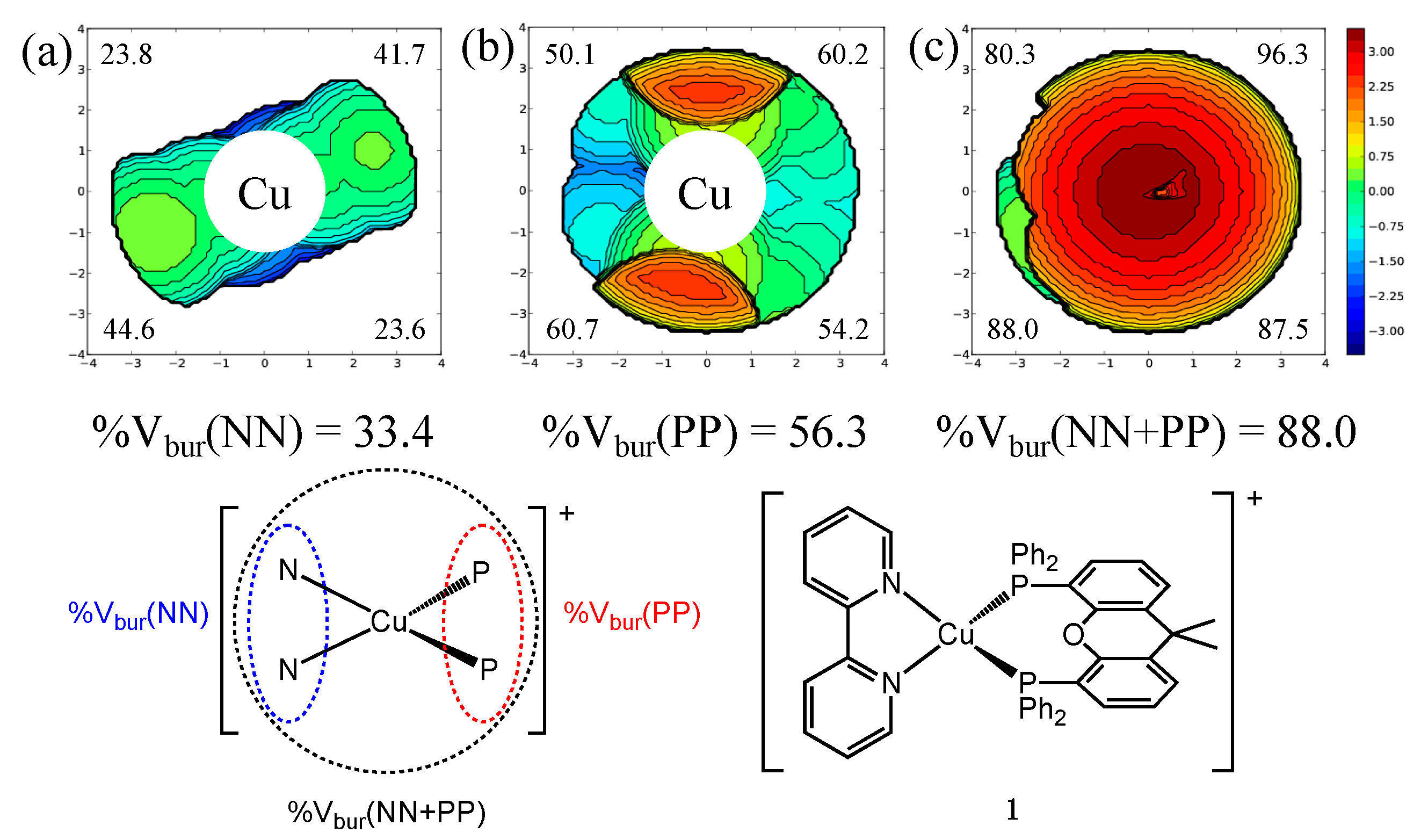
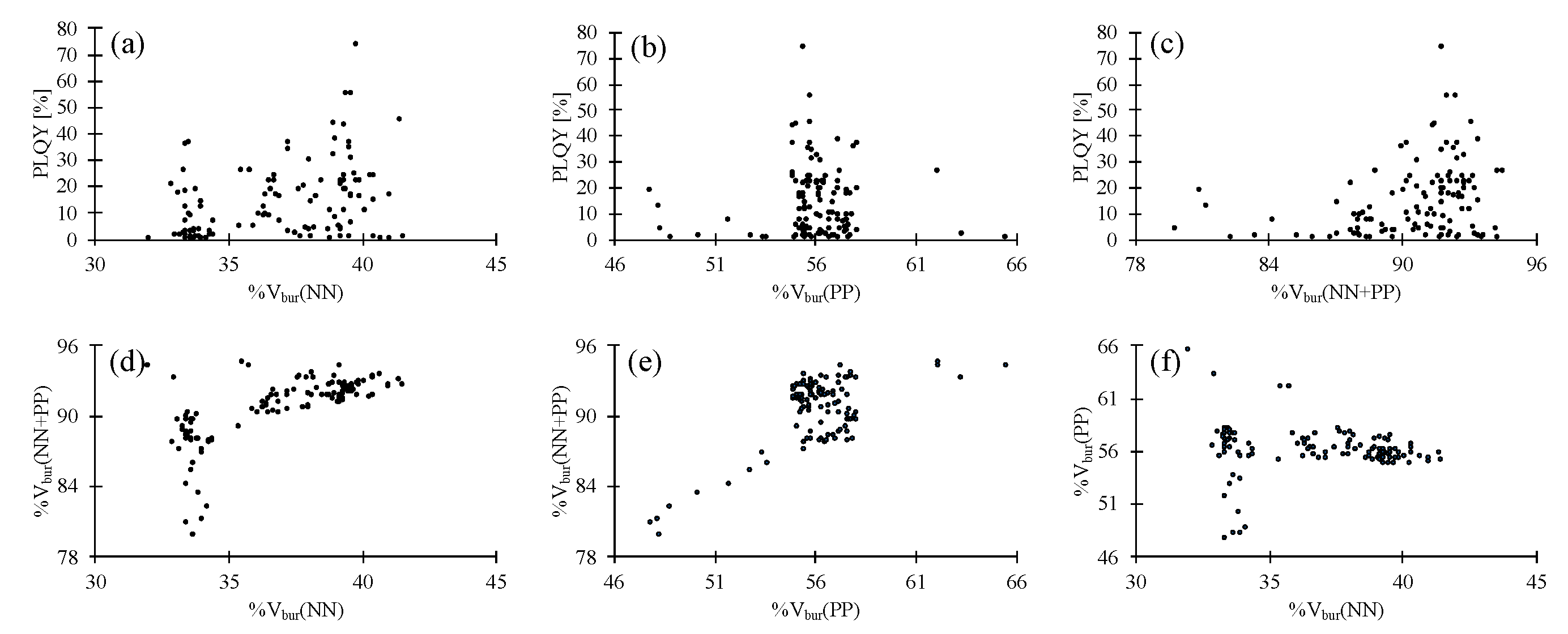
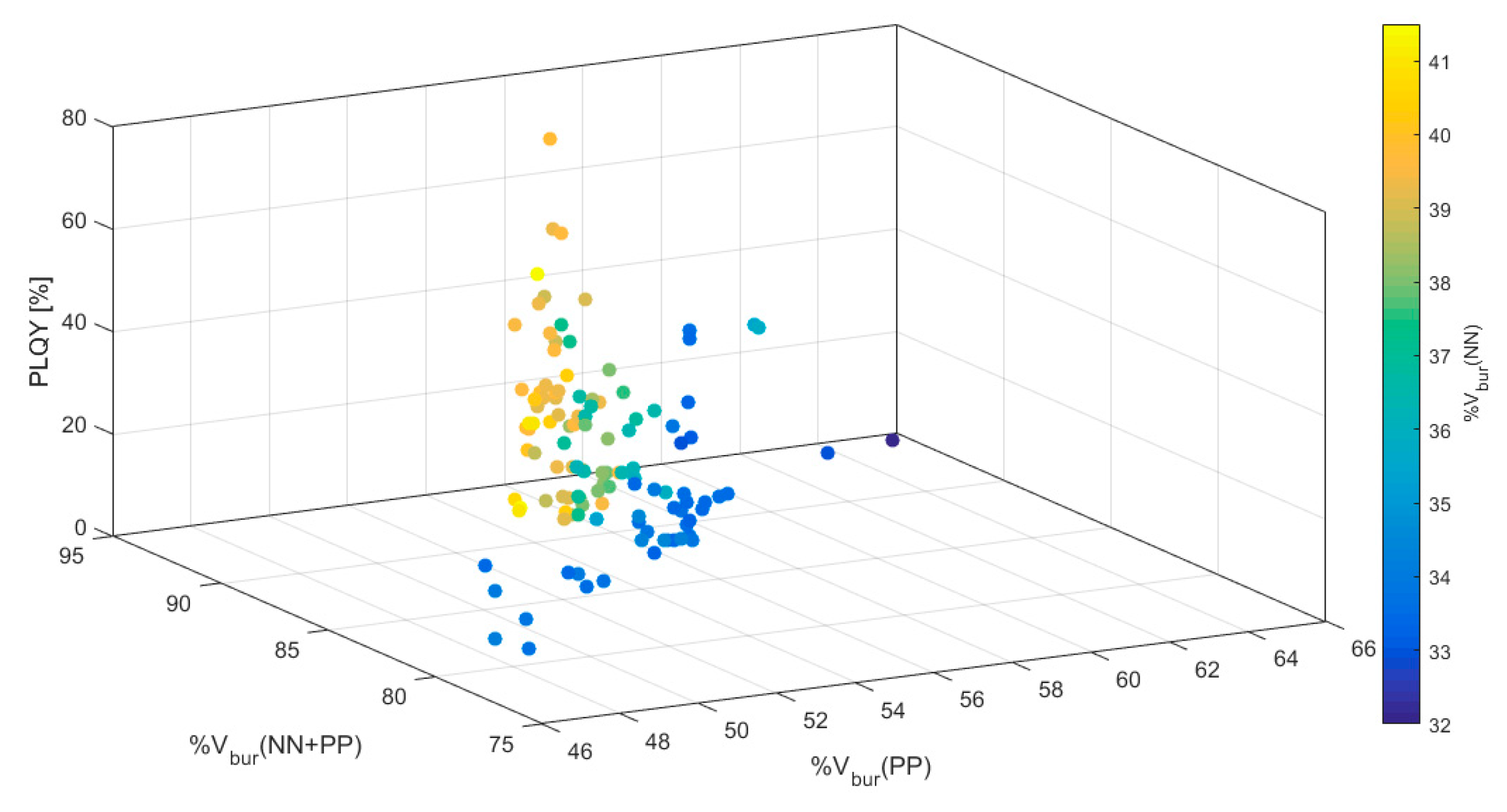
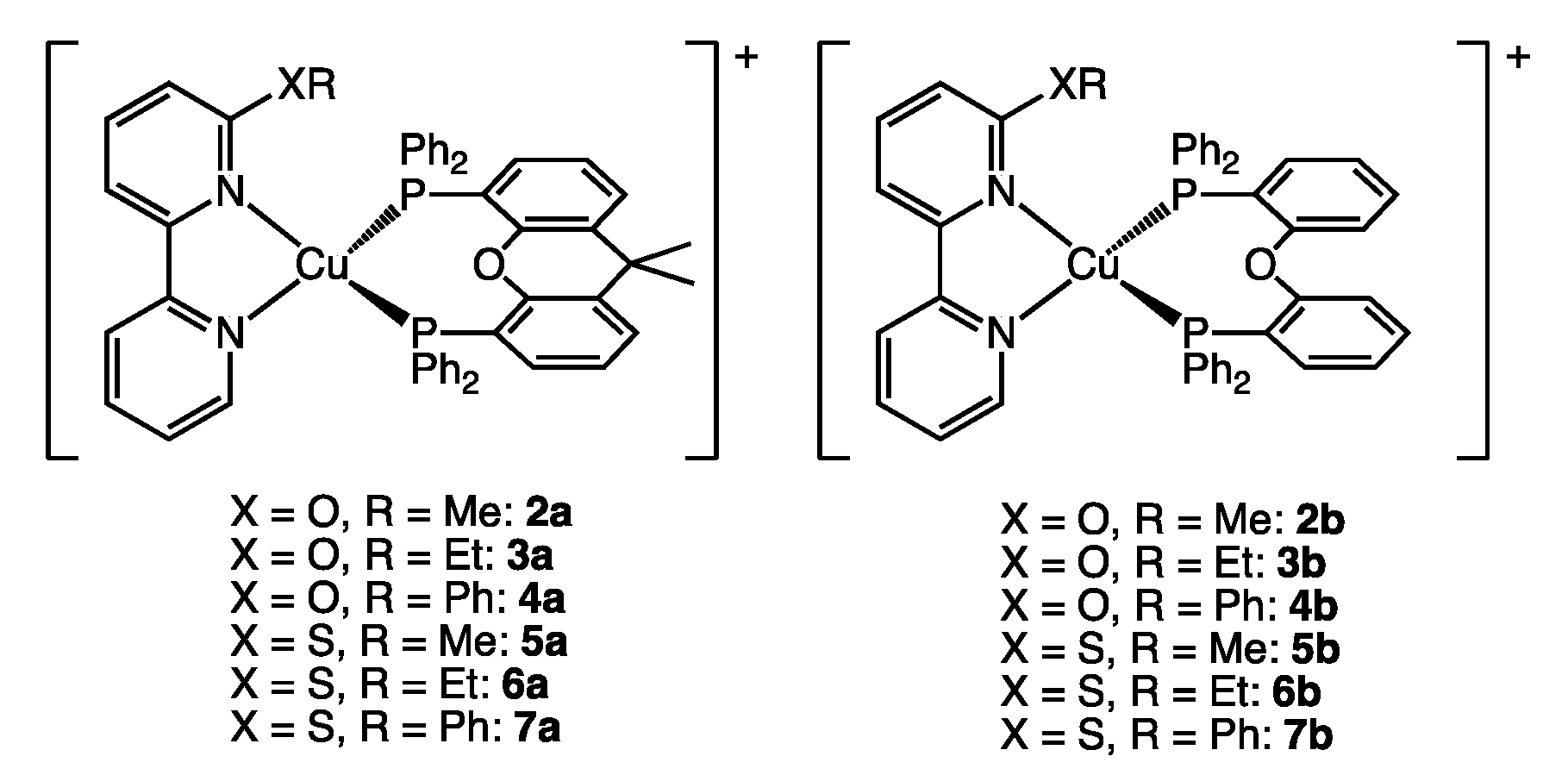
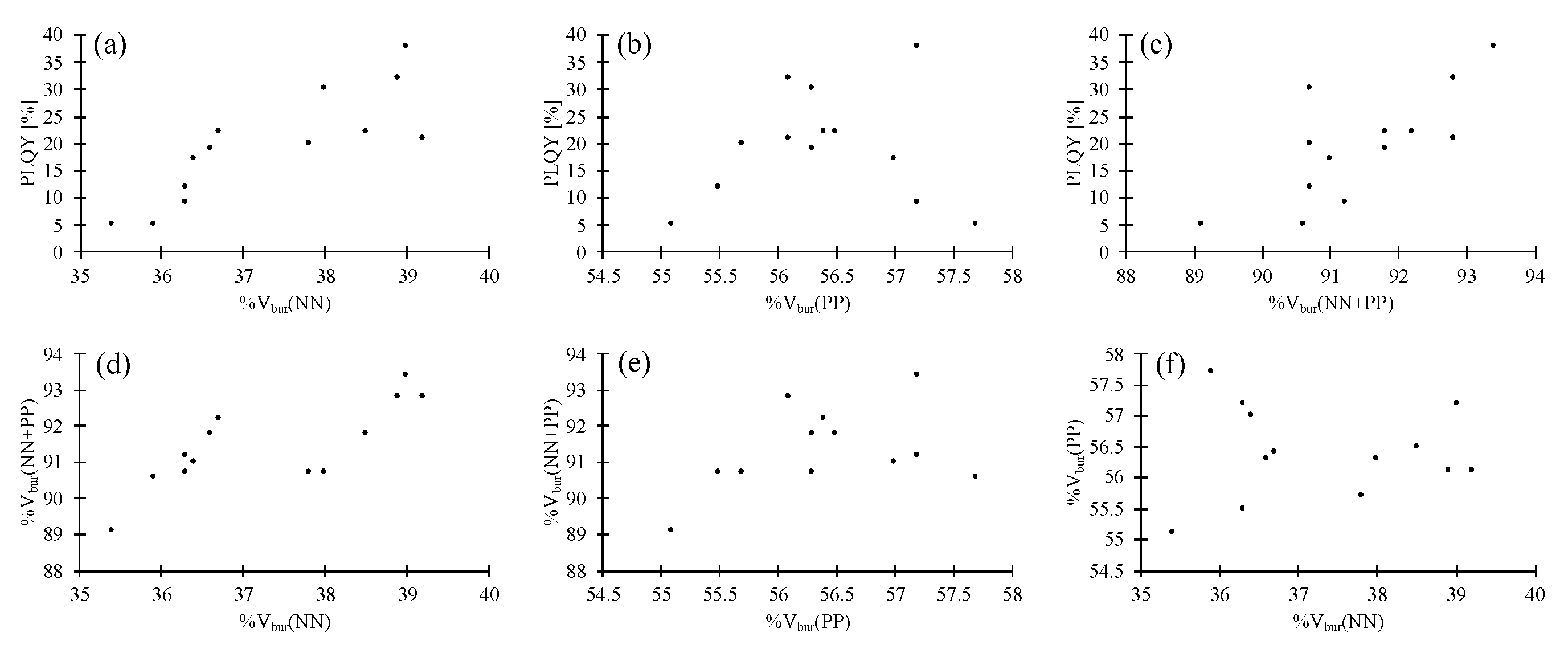
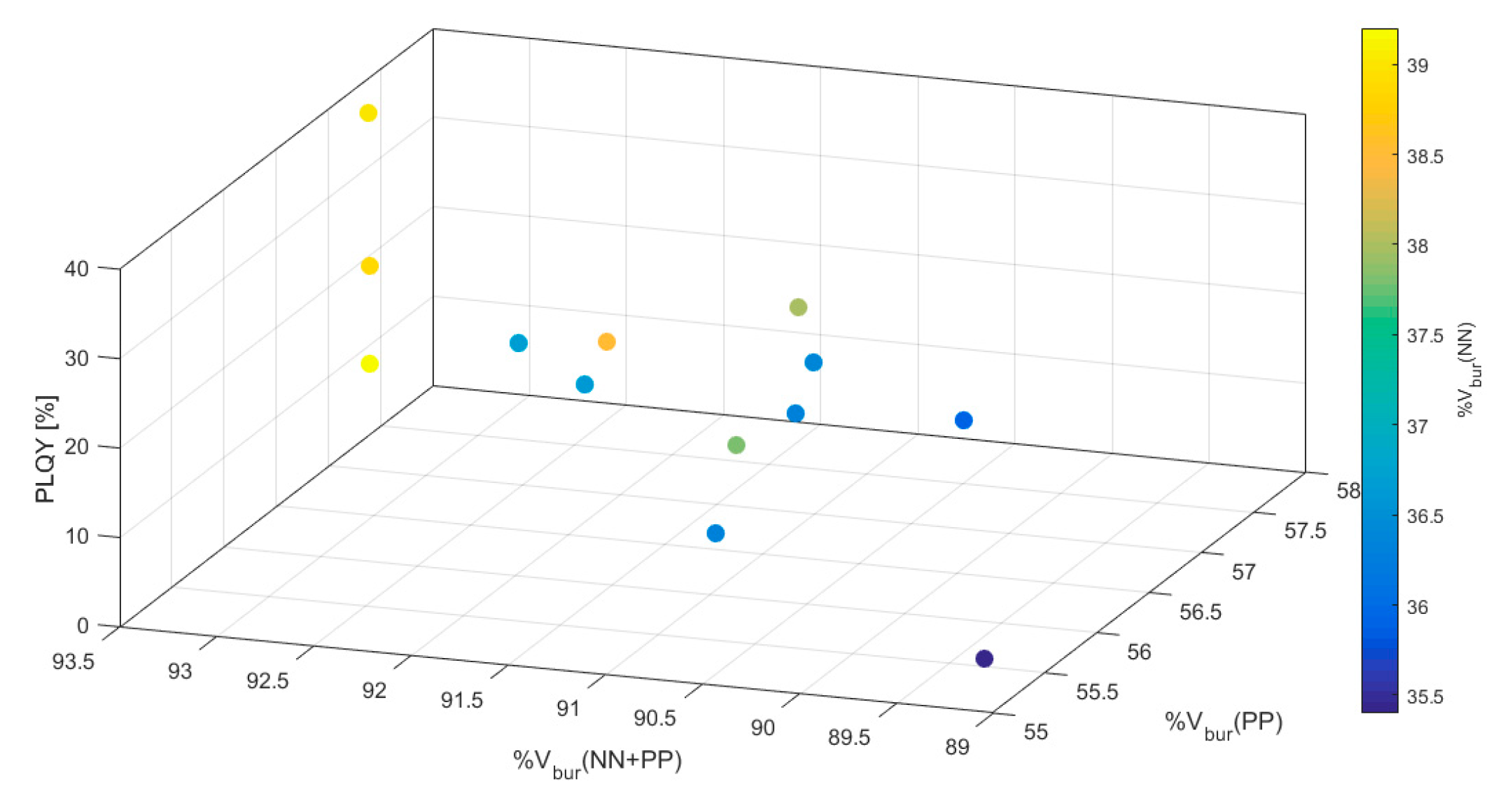
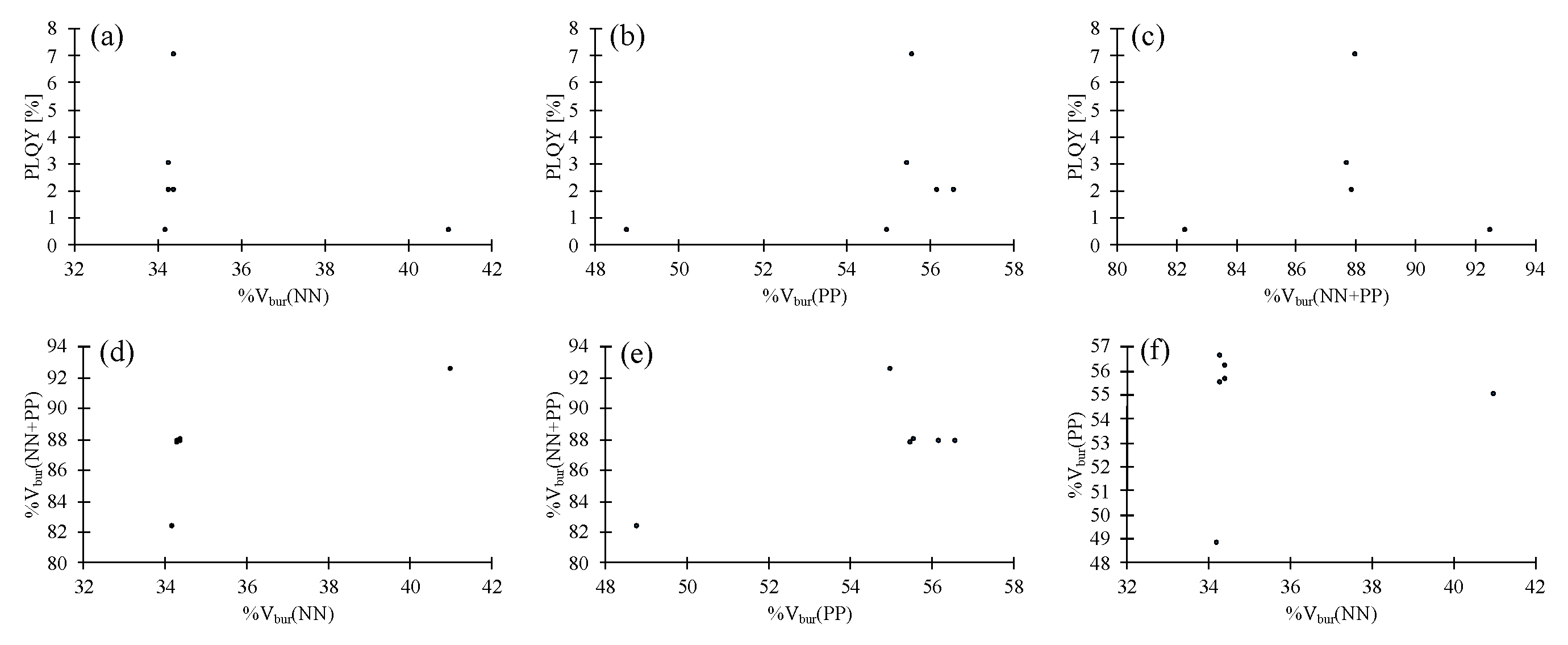
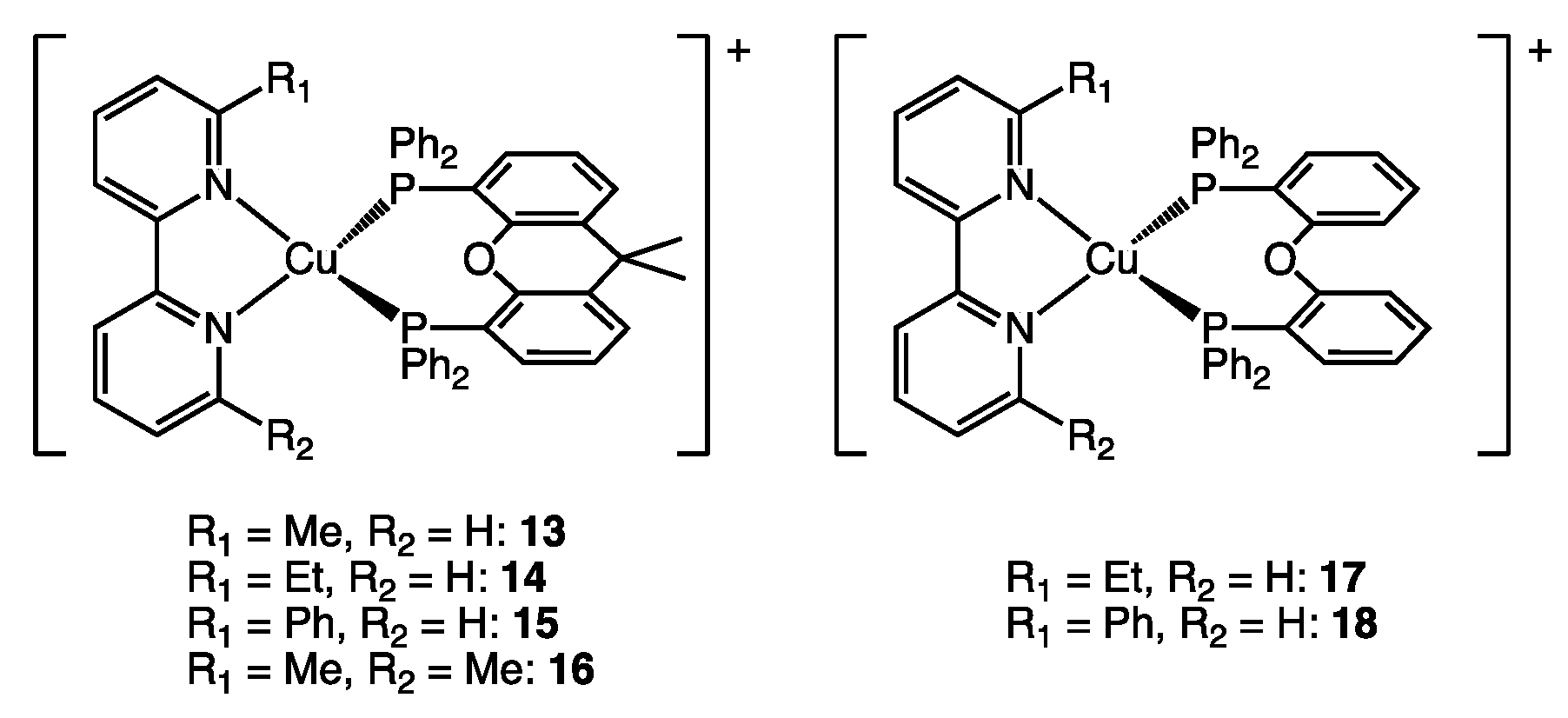

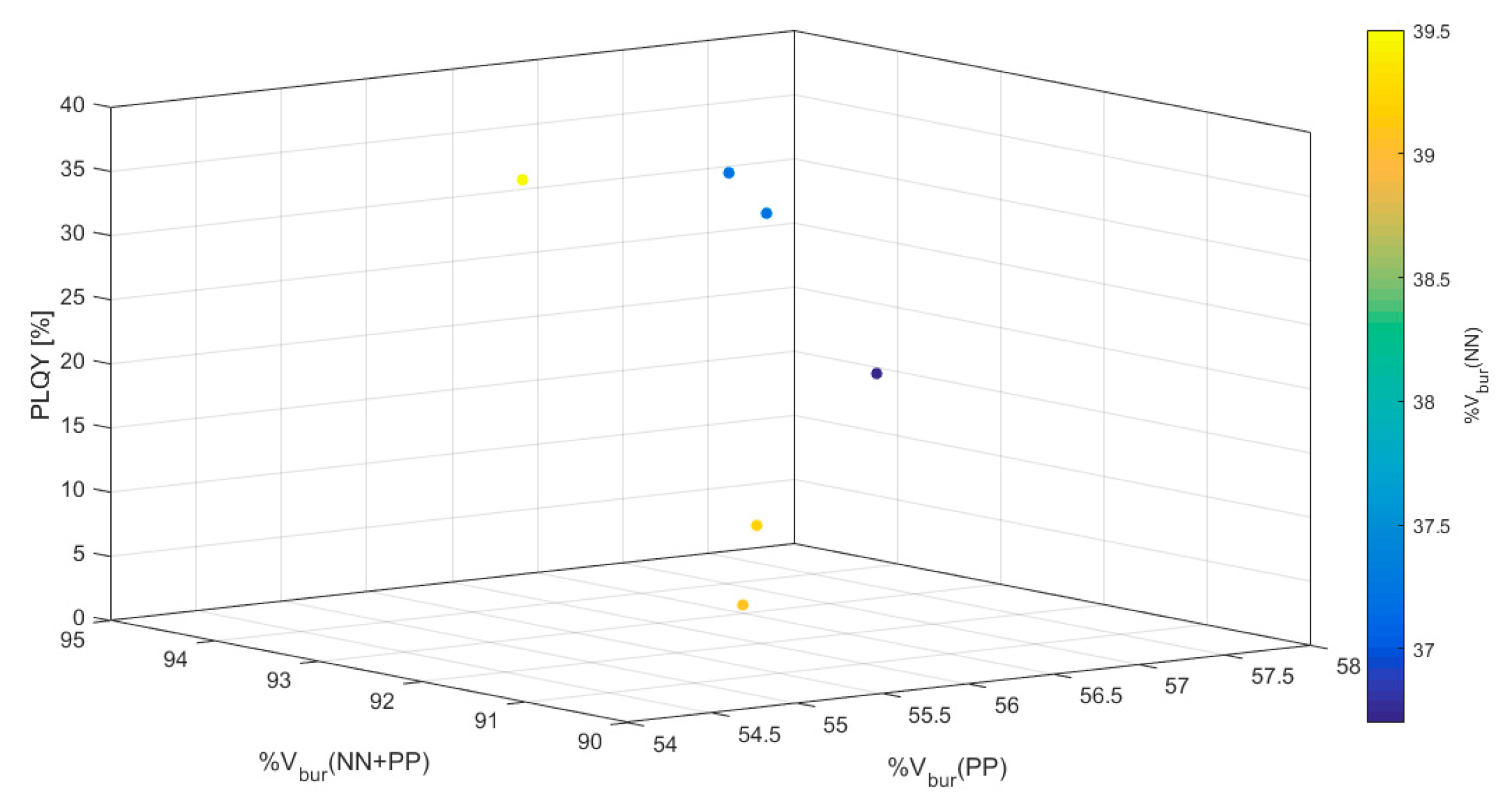
| Complex | PLQY [%] | %Vbur(NN) | %Vbur(PP) | %Vbur(NN + PP) |
|---|---|---|---|---|
| 2a | 19 | 36.6 | 56.3 | 91.8 |
| 2b | 17 | 36.4 | 57.0 | 91.0 |
| 3a | 22 | 36.7 | 56.4 | 92.2 |
| 3b | 9 | 36.3 | 57.2 | 91.2 |
| 4a | 12 | 36.3 | 55.5 | 90.7 |
| 4b a | 5 | 35.4/35.9 | 55.1/57.7 | 89.1/90.6 |
| 5a | 21 | 39.2 | 56.1 | 92.8 |
| 5b | 30 | 38.0 | 56.3 | 90.7 |
| 6a | 32 | 38.9 | 56.1 | 92.8 |
| 6b | 22 | 38.5 | 56.5 | 91.8 |
| 7a | 38 | 39.0 | 57.2 | 93.4 |
| 7b | 20 | 37.8 | 55.7 | 90.7 |
| Complex | PLQY [%] | %Vbur(NN) | %Vbur(PP) | %Vbur(NN + PP) |
|---|---|---|---|---|
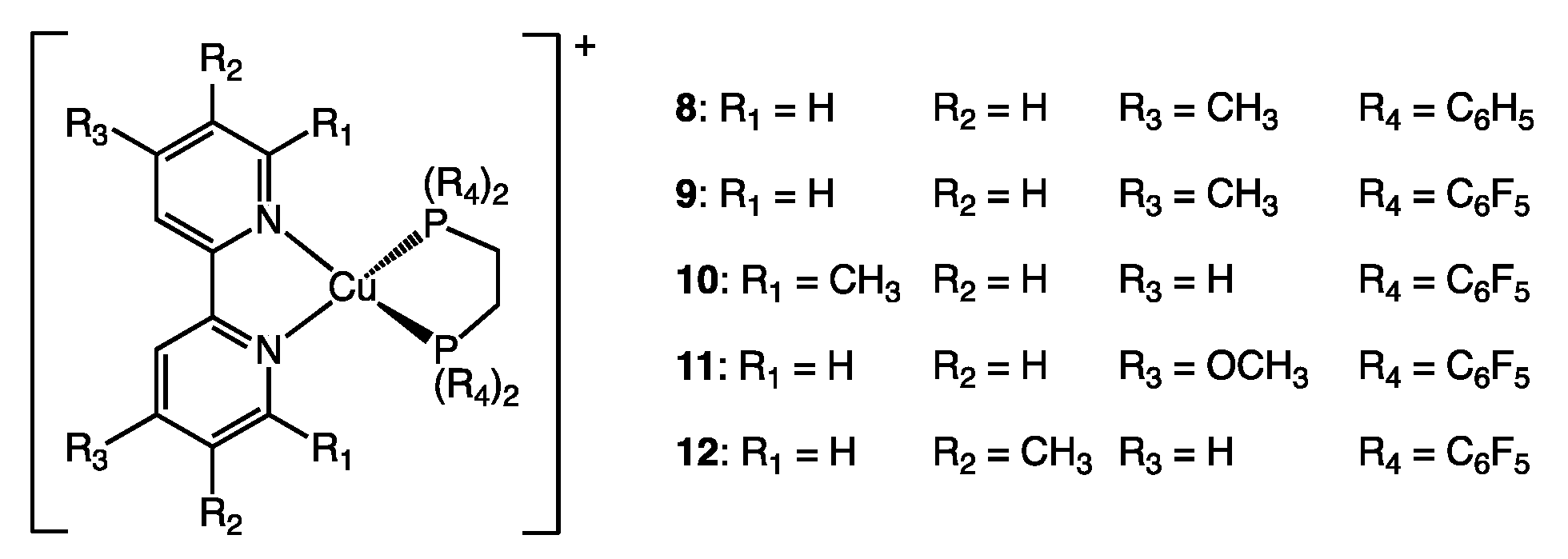
| 8 | 0.5 (0.5) a | 34.2 | 48.8 | 82.3 |
| 9 | 7 (8) a | 34.4 | 55.6 | 88.0 |
| 10 | 0.5 (9) a | 41.0 | 55.0 | 92.5 |
| 11 | 3 (3) a | 34.3 | 55.5 | 87.7 |
| 12 b | 2 (2) a | 34.3/34.4 | 56.6/56.2 | 87.9/87.9 |
| Complex | PLQY [%] | %Vbur(NN) | %Vbur(PP) | %Vbur(NN + PP) |
|---|---|---|---|---|
| 13 | 34 | 37.2 | 55.9 | 91.8 |
| 14 | 37 | 37.2 | 55.8 | 92.0 |
| 15 | 3.7 | 39.2 | 57.3 | 94.2 |
| 16 | 37 | 39.5 | 54.9 | 92.5 |
| 17 | 24 | 36.7 | 55.7 | 90.4 |
| 18 | 5.2 | 39.1 | 55.4 | 91.2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alkan-Zambada, M.; Constable, E.C.; Housecroft, C.E. The Role of Percent Volume Buried in the Characterization of Copper(I) Complexes for Lighting Purposes. Molecules 2020, 25, 2647. https://doi.org/10.3390/molecules25112647
Alkan-Zambada M, Constable EC, Housecroft CE. The Role of Percent Volume Buried in the Characterization of Copper(I) Complexes for Lighting Purposes. Molecules. 2020; 25(11):2647. https://doi.org/10.3390/molecules25112647
Chicago/Turabian StyleAlkan-Zambada, Murat, Edwin C. Constable, and Catherine E. Housecroft. 2020. "The Role of Percent Volume Buried in the Characterization of Copper(I) Complexes for Lighting Purposes" Molecules 25, no. 11: 2647. https://doi.org/10.3390/molecules25112647
APA StyleAlkan-Zambada, M., Constable, E. C., & Housecroft, C. E. (2020). The Role of Percent Volume Buried in the Characterization of Copper(I) Complexes for Lighting Purposes. Molecules, 25(11), 2647. https://doi.org/10.3390/molecules25112647









