Trichothecin Inhibits Cancer-Related Features in Colorectal Cancer Development by Targeting STAT3
Abstract
:1. Introduction
2. Results
2.1. TCN Induces Cell Apoptosis of Human Colorectal Cancer HCT 116 Cells
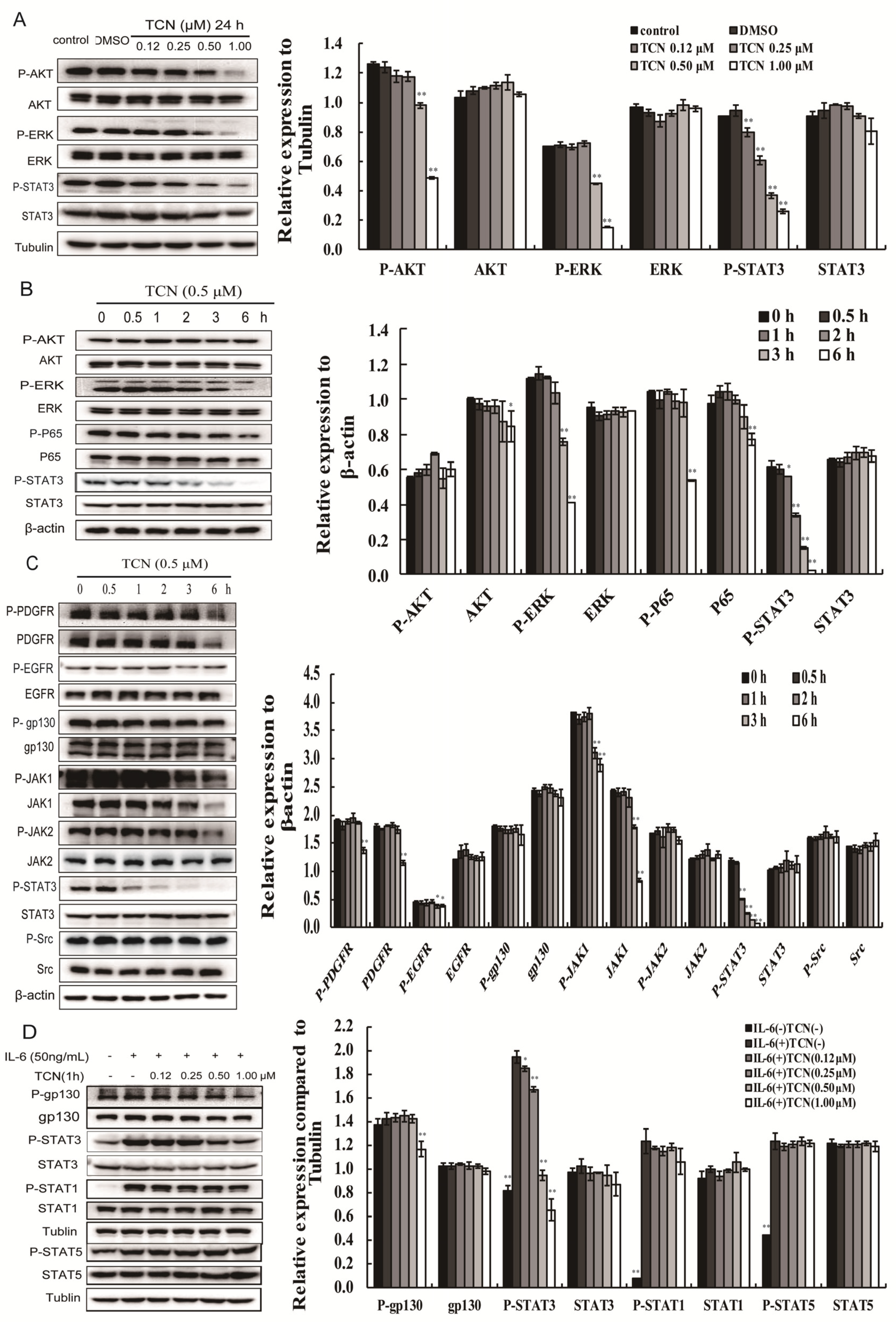
2.2. TCN Preferentially Inhibits Activation of STAT3
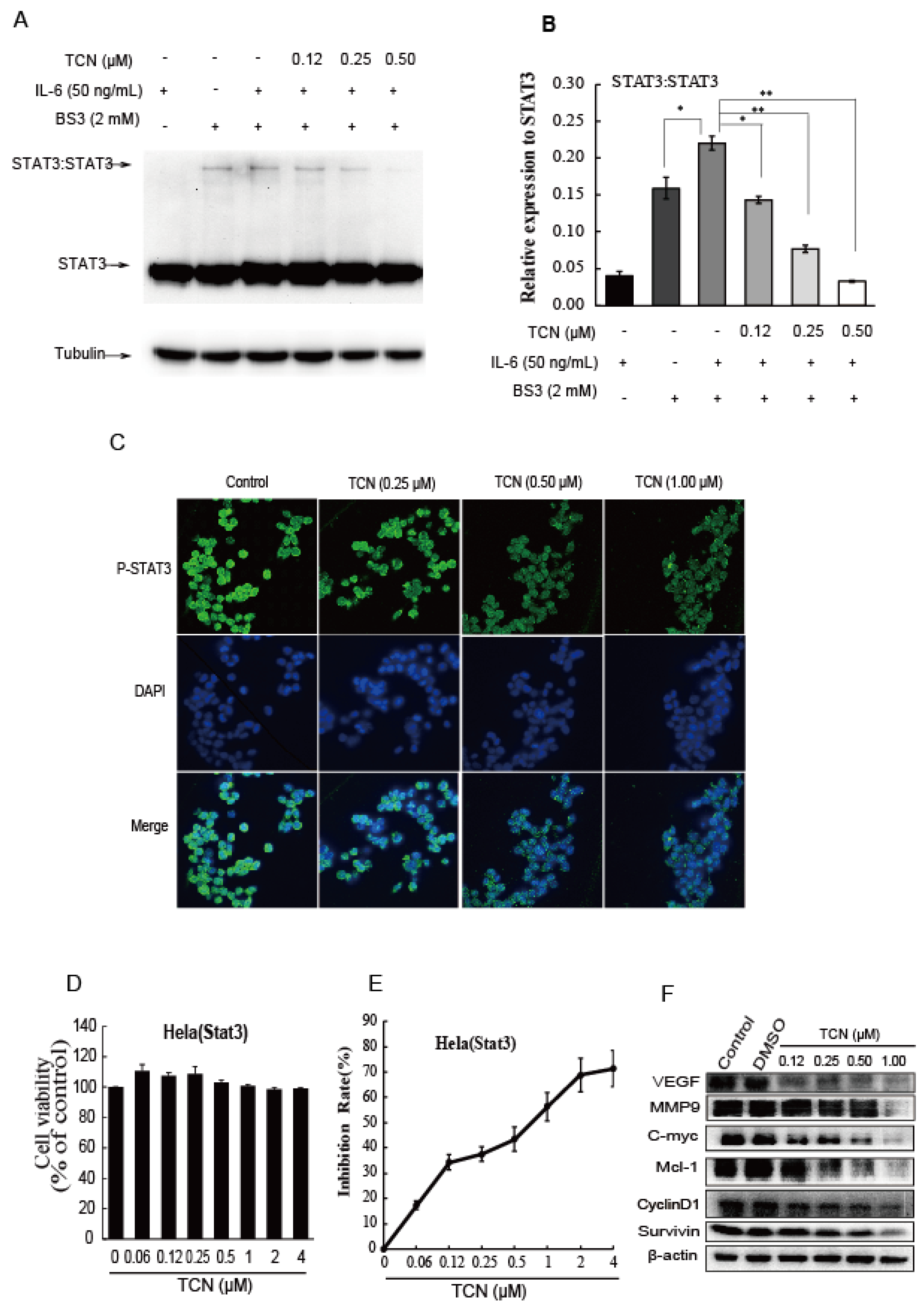
2.3. TCN Inhibits the Dimerization of STAT3 and Blocks STAT3 Nuclear Translocation
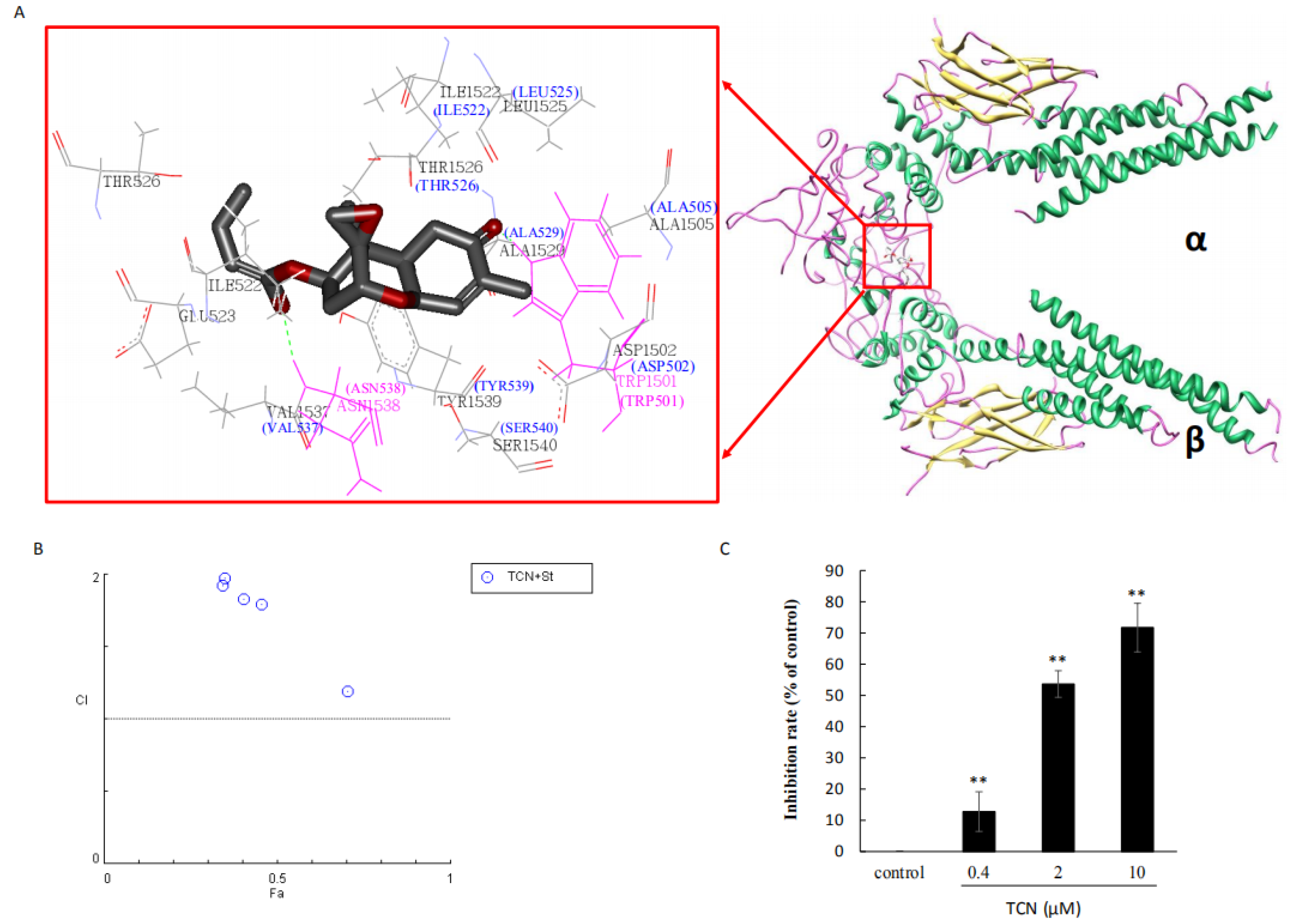
2.4. TCN Binds to the SH2 Domain of STAT3
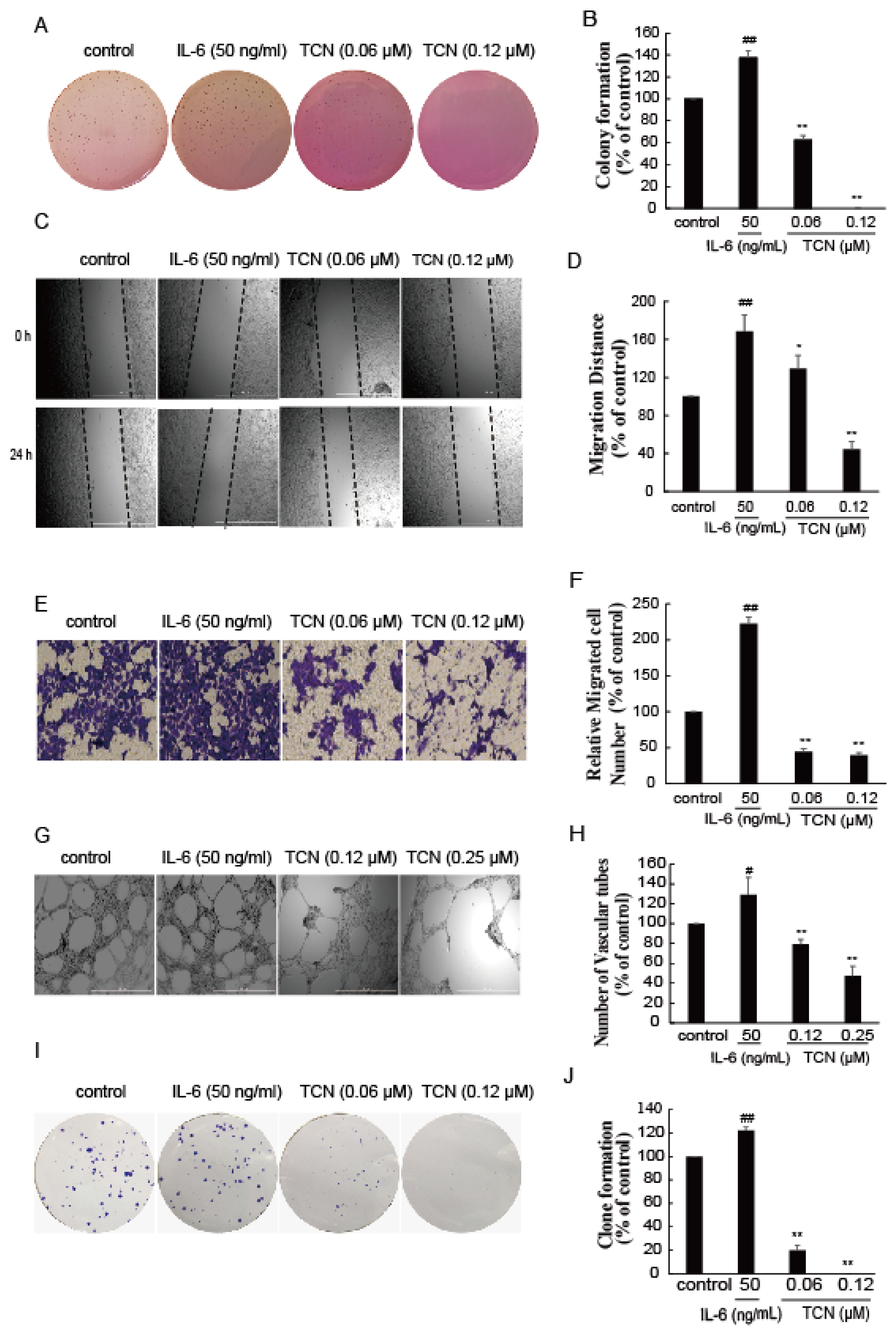
2.5. TCN Inhibits Cell Metastasis and Angiogenesis In Vitro
3. Discussion
4. Materials and Methods
4.1. Compound
4.2. Cell and Cell Culture
4.3. Cell Viability Assay
4.4. Clonogenic Assay
4.5. Cell Cycle Analysis
4.6. Annexin V-FITC/PI Double-Staining Assay
4.7. Soft Agar Colony Formation Assay
4.8. Wound Healing Assay
4.9. Transwell Invasion Assay
4.10. HUVEC Tube Formation Assay
4.11. Molecular Modeling
4.12. Luciferase Report Gene Assay
4.13. Immunofluorescence Assay
4.14. Fluorescence Polarization Assays
4.15. Tyrosine Kinase Activity Assay
4.16. Western Blotting Analysis
4.17. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Aggarwal, B.B.; Sethi, G.; Ahn, K.S.; Sandur, S.K.; Pandey, M.K.; Kunnumakkara, A.B.; Sung, B.; Ichikawa, H. Targeting signal-transducer-and-activator-of-transcription-3 for prevention and therapy of cancer: modern target but ancient solution. Ann. N. Y. Acad. Sci. 2006, 1091, 151–169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Becker, S.; Groner, B.; Muller, C.W. Three-dimensional structure of the Stat3beta homodimer bound to DNA. Nature 1998, 394, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Kortylewski, M.; Yu, H. Stat3 as a Potential Target for Cancer Immunotherapy. J. Immunother. 2007, 30, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Niu, G.; Wright, K.L.; Huang, M.; Song, L.; Haura, E.; Turkson, J.; Zhang, S.; Wang, T.; Sinibaldi, D.; Coppola, D.; et al. Constitutive Stat3 activity up-regulates VEGF expression and tumor angiogenesis. Oncogene 2002, 21, 2000–2008. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bromberg, J. Stat proteins and oncogenesis. J. Clin. Invest. 2002, 109, 1139–1142. [Google Scholar] [CrossRef] [PubMed]
- Buettner, R.; Mora, L.B.; Jove, R. Activated STAT signaling in human tumors provides novel molecular targets for therapeutic intervention. Clin. Cancer Res. 2002, 8, 945–954. [Google Scholar]
- Darnell, J.E. Transcription factors as targets for cancer therapy. Nat. Rev. Cancer 2002, 2, 740–749. [Google Scholar] [CrossRef]
- Yue, P.; Turkson, J. Targeting STAT3 in cancer: how successful are we? Expert Opin. Investig. Drugs 2009, 18, 45–56. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Crowe, P.; Goldstein, D.; Yang, J.-L. STAT3 inhibition, a novel approach to enhancing targeted therapy in human cancers. Int. J. Oncol. 2012, 41, 1181–1191. [Google Scholar] [CrossRef] [Green Version]
- Alonzi, T. Induced somatic inactivation of STAT3 in mice triggers the development of a fulminant form of enterocolitis. Cytokine 2004, 26, 45–56. [Google Scholar] [CrossRef]
- Mantovani, A.; Allavena, P.; Sica, A.; Balkwill, F.R. Cancer-related inflammation. Nature 2008, 454, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Zhuang, G.; Cao, Y.; Du, P.; Kim, H.J.; Settleman, J. Drug resistance via feedback activation of Stat3 in oncogene-addicted cancer cells. Cancer Cell 2014, 26, 207–221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, C.; Li, H.; Lin, H.-J.; Yang, S.; Lin, J.; Liang, G. Feedback Activation of STAT3 as a Cancer Drug-Resistance Mechanism. Trends Pharmacol. Sci. 2016, 37, 47–61. [Google Scholar] [CrossRef] [PubMed]
- Bromberg, J.F.; Wrzeszczynska, M.H.; Devgan, G.; Zhao, Y.; Pestell, R.G.; Albanese, C.; E Darnell, J. Stat3 as an Oncogene. Cell 1999, 98, 295–303. [Google Scholar] [CrossRef] [Green Version]
- Calò, V.; Migliavacca, M.; Bazan, V.; Macaluso, M.; Buscemi, M.; Gebbia, N.; Russo, A. STAT proteins: From normal control of cellular events to tumorigenesis. J. Cell. Physiol. 2003, 197, 157–168. [Google Scholar] [CrossRef]
- Schlessinger, K.; Levy, D.E. Malignant transformation but not normal cell growth depends on signal transducer and activator of transcription 3. Cancer Res. 2005, 65, 5828–5834. [Google Scholar] [CrossRef] [Green Version]
- Guanizo, A.C.; Fernando, C.D.; Garama, D.J.; Gough, D.J. STAT3: a multifaceted oncoprotein. Growth Factors 2018, 36, 1–14. [Google Scholar] [CrossRef]
- Yu, H.; Jove, R. The STATs of cancer — new molecular targets come of age. Nat. Rev. Cancer 2004, 4, 97–105. [Google Scholar] [CrossRef]
- Inghirami, G.; Chiarle, R.; Simmons, W.; Piva, R.; Schlessinger, K.; Levy, D.E. New and Old Functions of STAT3: A Pivitol Target for Individualized Treatment of Cancer. Cell Cycle 2005, 4, 1131–1133. [Google Scholar] [CrossRef]
- Alvarez, J.V.; Greulich, H.; Sellers, W.R.; Meyerson, M.; Frank, D.A. Signal transducer and activator of transcription 3 is required for the oncogenic effects of non-small-cell lung cancer-associated mutations of the epidermal growth factor receptor. Cancer Res. 2006. [Google Scholar] [CrossRef] [Green Version]
- Becker, T.M.; Boyd, S.C.; Mijatov, B.; Gowrishankar, K.; Snoyman, S.; Pupo, G.M.; Scolyer, R.A.; Mann, G.J.; Kefford, R.; Zhang, X.D.; et al. Mutant B-RAF-Mcl-1 survival signaling depends on the STAT3 transcription factor. Oncogene 2013, 33, 1158–1166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, A.; Hirpara, J.L.; Pervaiz, S.; Eu, J.-Q.; Sethi, G.; Goh, B.C. Do STAT3 inhibitors have potential in the future for cancer therapy? Expert Opin. Investig. Drugs 2017, 26, 1–5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niu, G.; Heller, R.; Catlett-Falcone, R.; Coppola, D.; Jaroszeski, M.; Dalton, W.; Jove, R.; Yu, H. Gene therapy with dominant-negative Stat3 suppresses growth of the murine melanoma B16 tumor in vivo. Cancer Res. 1999, 59, 5059–5063. [Google Scholar] [PubMed]
- Xu, G.; Zhang, C.; Zhang, J. Dominant negative STAT3 suppresses the growth and invasion capability of human lung cancer cells. Mol. Med. Rep. 2009, 2, 819–824. [Google Scholar] [CrossRef] [Green Version]
- Klosek, S.K.; Nakashiro, K.-I.; Hara, S.; Goda, H.; Hamakawa, H. Stat3 as a molecular target in RNA interference-based treatment of oral squamous cell carcinoma. Oncol. Rep. 2008, 20, 873–878. [Google Scholar] [PubMed]
- Qian, W.; Guan, W.; Gao, Y.; Tan, J.; Qiao, Z.; Huang, H.; Xia, C. Inhibition of STAT3 by RNA interference suppresses angiogenesis in colorectal carcinoma. Braz. J. Med Boil. Res. 2011, 44, 1222–1230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Du, J.; Liu, Q.; Zhang, Y. Down-regulation of STAT3 expression using vector-based RNA interference promotes apoptosis in Hepatocarcinoma cells. Artif. Cells. Nanomed. Biotechnol. 2015, 44, 1–5. [Google Scholar] [CrossRef]
- Hong, D.; Kurzrock, R.; Kim, Y.; Woessner, R.; Younes, A.; Nemunaitis, J.; Fowler, N.; Zhou, T.; Schmidt, J.; Jo, M.; et al. AZD9150, a next-generation antisense oligonucleotide inhibitor of STAT3 with early evidence of clinical activity in lymphoma and lung cancer. Sci. Transl. Med. 2015. [Google Scholar] [CrossRef] [Green Version]
- Reilley, M.J.; McCoon, P.; Cook, C.; Lyne, P.D.; Kurzrock, R.; Kim, Y.; Woessner, R.; Younes, A.; Nemunaitis, J.; Fowler, N.H.; et al. STAT3 antisense oligonucleotide AZD9150 in a subset of patients with heavily pretreated lymphoma: results of a phase 1b trial. J. Immunother. Cancer 2018. [Google Scholar] [CrossRef] [Green Version]
- Turkson, J.; Ryan, D.; Kim, J.S.; Zhang, Y.; Chen, Z.; Haura, E.; Laudano, A.; Sebti, S.; Hamilton, A.D.; Jove, R. Phosphotyrosyl Peptides Block Stat3-mediated DNA Binding Activity, Gene Regulation, and Cell Transformation. J. Boil. Chem. 2001, 276, 45443–45455. [Google Scholar] [CrossRef] [Green Version]
- Turkson, J.; Kim, J.S.; Zhang, S.; Yuan, J.; Huang, M.; Glenn, M.; Haura, E.; Sebti, S.; Hamilton, A.D.; Jove, R. Novel peptidomimetic inhibitors of signal transducer and activator of transcription 3 dimerization and biological activity. Mol. Cancer Ther. 2004, 3, 261–269. [Google Scholar] [PubMed]
- Mandal, P.K.; Gao, F.; Lu, Z.; Ren, Z.; Ramesh, R.; Birtwistle, J.S.; Kaluarachchi, K.K.; Chen, X.; Bast, R.C.; Liao, W.S.; et al. Potent and Selective Phosphopeptide Mimetic Prodrugs Targeted to the Src Homology 2 (SH2) Domain of Signal Transducer and Activator of Transcription 3. J. Med. Chem. 2011, 54, 3549–3563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, H.; Wang, R.; Wang, S.; Lin, J. A low-molecular-weight compound discovered through virtual database screening inhibits Stat3 function in breast cancer cells. Proc. Natl. Acad. Sci. USA 2005, 102, 4700–4705. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siddiquee, K.; Zhang, S.; Guida, W.C.; Blaskovich, M.A.; Greedy, B.; Lawrence, H.R.; Yip, M.L.R.; Jove, R.; McLaughlin, M.M.; Lawrence, N.J.; et al. Selective chemical probe inhibitor of Stat3, identified through structure-based virtual screening, induces antitumor activity. Proc. Natl. Acad. Sci. USA 2007, 104, 7391–7396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akiyama, Y.; Ashizawa, T.; Miyata, H.; Ishii, H.; Oshita, C.; Matsuno, K.; Masuda, Y.; Furuya, T.; Okawara, T.; Otsuka, M.; et al. Antitumor activity of a novel small molecule STAT3 inhibitor against a human lymphoma cell line with high STAT3 activation. Int. J. Oncol. 2011, 38, 1245–1252. [Google Scholar] [CrossRef] [Green Version]
- Couto, J.I.; Bear, M.D.; Lin, J.; Pennell, M.L.; Kulp, S.K.; Kisseberth, W.C.; London, C. Biologic activity of the novel small molecule STAT3 inhibitor LLL12 against canine osteosarcoma cell lines. BMC Veter.-Res. 2012. [Google Scholar] [CrossRef] [Green Version]
- Bharti, A.C.; Donato, N.; Aggarwal, B.B. Curcumin (diferuloylmethane) inhibits constitutive and IL-6-inducible STAT3 phosphorylation in human multiple myeloma cells. J. Immunol. 2003, 171, 3863–3871. [Google Scholar] [CrossRef]
- Ball, D.P.; Lewis, A.M.; Williams, D.; Resetca, D.; Wilson, D.J.; Gunning, P.T. Signal transducer and activator of transcription 3 (STAT3) inhibitor, S3I-201, acts as a potent and non-selective alkylating agent. Oncotarget 2016, 7, 20669–20679. [Google Scholar] [CrossRef]
- Li, Z.; Zhu, T.; Xu, Y.; Wu, C.; Chen, J.; Ren, Y.; Kong, L.; Sun, S.; Guo, W.; Wang, Y.; et al. A novel STAT3 inhibitor, HJC0152, exerts potent antitumor activity in glioblastoma. Am. J. Cancer Res. 2019, 9, 699–713. [Google Scholar]
- Okusaka, T.; Ueno, H.; Ikeda, M.; Mitsunaga, S.; Ozaka, M.; Ishii, H.; Yokosuka, O.; Ooka, Y.; Yoshimoto, R.; Yanagihara, Y.; et al. Phase 1 and pharmacological trial of OPB-31121, a signal transducer and activator of transcription-3 inhibitor, in patients with advanced hepatocellular carcinoma. Hepatol. Res. 2015, 45, 1283–1291. [Google Scholar] [CrossRef]
- Ogura, M.; Uchida, T.; Terui, Y.; Hayakawa, F.; Kobayashi, Y.; Taniwaki, M.; Takamatsu, Y.; Naoe, T.; Tobinai, K.; Munakata, W.; et al. Phase I study of OPB-51602, an oral inhibitor of signal transducer and activator of transcription 3, in patients with relapsed/refractory hematological malignancies. Cancer Sci. 2015, 106, 896–901. [Google Scholar] [CrossRef] [PubMed]
- Orlova, A.; Wagner, C.; De Araujo, E.; Bajusz, D.; Neubauer, H.A.; Herling, M.; Gunning, P.T.; Keserű, G.M.; Moriggl, R.H. Direct Targeting Options for STAT3 and STAT5 in Cancer. Cancers 2019, 1930. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bai, L.; Zhou, H.; Xu, R.; Zhao, Y.; Chinnaswamy, K.; McEachern, D.; Chen, J.; Yang, C.-Y.; Liu, Z.; Wang, M.; et al. A Potent and Selective Small-Molecule Degrader of STAT3 Achieves Complete Tumor Regression In Vivo. Cancer Cell 2019, 36, 498–511.e17. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, G.; Ma, J.; Zeng, Y.; Ma, W.; Zhao, P.-J. Endophytic fungus Trichothecium roseum LZ93 antagonizing pathogenic fungi in vitro and its secondary metabolites. J. Microbiol. 2010, 48, 784–790. [Google Scholar] [CrossRef]
- Wang, W.; Liu, H.; Wang, S.; Hao, X.; Li, L. A diterpenoid derivative 15-oxospiramilactone inhibits Wnt/beta-catenin signaling and colon cancer cell tumorigenesis. Cell Res. 2011, 21, 730–740. [Google Scholar] [CrossRef]
- Du, R.H.; Cui, J.T.; Wang, T.; Zhang, A.H.; Tan, R.X. Trichothecin induces apoptosis of HepG2 cells via caspase-9 mediated activation of the mitochondrial death pathway. Toxicon 2012, 59, 143–150. [Google Scholar] [CrossRef]
- Elmore, S.A. Apoptosis: A Review of Programmed Cell Death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef]
- Inoue, S.; Browne, G.; Melino, G.; Cohen, G.M. Ordering of caspases in cells undergoing apoptosis by the intrinsic pathway. Cell Death Differ. 2009, 16, 1053–1061. [Google Scholar] [CrossRef] [Green Version]
- Diamantopoulos, P.T.; Sofotasiou, M.; Papadopoulou, V.; Polonyfi, K.; Iliakis, T.; Viniou, N.-A. PARP1-Driven Apoptosis in Chronic Lymphocytic Leukemia. BioMed Res. Int. 2014, 2014, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Shabbir, A.; Cox, A.; Rodriguez-Menocal, L.; Salgado, M.; Van Badiavas, E. Mesenchymal Stem Cell Exosomes Induce Proliferation and Migration of Normal and Chronic Wound Fibroblasts, and Enhance Angiogenesis In Vitro. Stem Cells Dev. 2015, 24, 1635–1647. [Google Scholar] [CrossRef]
- Geiger, J.L.; Grandis, J.R.; Bauman, J.E. The STAT3 pathway as a therapeutic target in head and neck cancer: Barriers and innovations. Oral Oncol. 2015, 56, 84–92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McDermott, M.W. Effect of p53 Status and STAT1 on Chemotherapy-Induced, Fas-Mediated Apoptosis in Colorectal Cancer. Cancer Res. 2005, 65, 8951–8960. [Google Scholar] [CrossRef] [Green Version]
- Xiong, A.; Yang, Z.; Shen, Y.; Zhou, J.; Shen, Q. Transcription Factor STAT3 as a Novel Molecular Target for Cancer Prevention. Cancers 2014, 6, 926–957. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verhoeven, Y.; Tilborghs, S.; Jacobs, J.; De Waele, J.; Quatannens, D.; Deben, C.; Prenen, H.; Pauwels, P.; Trinh, X.B.; Wouters, A.; et al. The potential and controversy of targeting STAT family members in cancer. Semin. Cancer Boil. 2020, 60, 41–56. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.-G.; Chen, J.R.; Hernandez, L.; Alvord, W.G.; Stewart, C.L. Dual control of LIF expression and LIF receptor function regulate Stat3 activation at the onset of uterine receptivity and embryo implantation. Proc. Natl. Acad. Sci. USA 2001, 98, 8680–8685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Li, P.-K.; Li, C.; Lin, J. Inhibition of STAT3 Signaling Blocks the Anti-apoptotic Activity of IL-6 in Human Liver Cancer Cells. J. Boil. Chem. 2010, 285, 27429–27439. [Google Scholar] [CrossRef] [Green Version]
- Kesanakurti, D.; Chetty, C.; Dinh, D.H.; Gujrati, M.; Rao, J.S. Role of MMP-2 in the regulation of IL-6/Stat3 survival signaling via interaction with alpha5beta1 integrin in glioma. Oncogene 2013, 32, 327–340. [Google Scholar] [CrossRef] [Green Version]
- Darnell, J.; Kerr, I.; Stark, G. Jak-STAT pathways and transcriptional activation in response to IFNs and other extracellular signaling proteins. Science 1994, 264, 1415–1421. [Google Scholar] [CrossRef] [Green Version]
- Zhong, Z.; Wen, Z.; Darnell, J. Stat3: a STAT family member activated by tyrosine phosphorylation in response to epidermal growth factor and interleukin-6. Science 1994, 264, 95–98. [Google Scholar] [CrossRef]
- Schust, J.; Sperl, B.; Hollis, A.; Mayer, T.U.; Berg, T. Stattic: A Small-Molecule Inhibitor of STAT3 Activation and Dimerization. Chem. Boil. 2006, 13, 1235–1242. [Google Scholar] [CrossRef] [Green Version]
- Osugi, T.; Oshima, Y.; Fujio, Y.; Funamoto, M.; Yamashita, A.; Negoro, S.; Kunisada, K.; Izumi, M.; Nakaoka, Y.; Hirota, H.; et al. Cardiac-specific Activation of Signal Transducer and Activator of Transcription 3 Promotes Vascular Formation in the Heart. J. Boil. Chem. 2001, 277, 6676–6681. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turkson, J. STAT proteins as novel targets for cancer drug discovery. Expert Opin. Ther. Targets 2004, 8, 409–422. [Google Scholar] [CrossRef] [PubMed]
- Miyoshi, K.; Takaishi, M.; Nakajima, K.; Ikeda, M.; Kanda, T.; Tarutani, M.; Iiyama, T.; Asao, N.; DiGiovanni, J.; Sano, S. Stat3 as a Therapeutic Target for the Treatment of Psoriasis: A Clinical Feasibility Study with STA-21, a Stat3 Inhibitor. J. Investig. Dermatol. 2011, 131, 108–117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koskela, H.L.; Eldfors, S.; Ellonen, P.; Van Adrichem, A.J.; Kuusanmäki, H.I.; Andersson, E.; Lagström, S.; Clemente, M.J.; Olson, T.; Jalkanen, S.E.; et al. Somatic STAT3 mutations in large granular lymphocytic leukemia. New Engl. J. Med. 2012, 366, 1905–1913. [Google Scholar] [CrossRef] [Green Version]
- Takatsuki, A.; Tamura, G.; Arima, K. Mode of action of trichothecin on multiplication of newcastle disease virus in cultured cells. J. Antibiot. 1969, 22, 241–247. [Google Scholar] [CrossRef]
- Barbacid, M.; Fresno, M.; Vázquez, D. Inhibitors of polypeptide elongation on yeast polysomes. J. Antibiot. 1975, 28, 453–462. [Google Scholar] [CrossRef] [Green Version]
- Fresno, M.; Carrasco, L.; Vazquez, D.; Escudero, M.F. Initiation of the Polypeptide Chain by Reticulocyte Cell-Free Systems. Survey of Different Inhibitors of Translation. JBIC J. Boil. Inorg. Chem. 1976, 68, 355–364. [Google Scholar] [CrossRef]
- McLaughlin, J.; Bin-Umer, M.A.; Tortora, A.; Mendez, N.; McCormick, S.P.; Tumer, N.E. A genome-wide screen in Saccharomyces cerevisiae reveals a critical role for the mitochondria in the toxicity of a trichothecene mycotoxin. Proc. Natl. Acad. Sci. USA 2009, 106, 21883–21888. [Google Scholar] [CrossRef] [Green Version]
- Su, J.; Zhao, P.; Kong, L.; Li, X.; Yan, J.; Zeng, Y.; Li, Y. Trichothecin induces cell death in NF-kappaB constitutively activated human cancer cells via inhibition of IKKbeta phosphorylation. PLoS ONE 2013, 8, e71333. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.; Liao, X.; Agarwal, M.K.; Barnes, L.; Auron, P.E.; Stark, G.R. Unphosphorylated STAT3 accumulates in response to IL-6 and activates transcription by binding to NFkappaB. Genes. Dev. 2007, 21, 1396–1408. [Google Scholar] [CrossRef] [Green Version]
- Wang, T.; Niu, G.; Kortylewski, M.; Burdelya, L.; Shain, K.; Zhang, S.; Bhattacharya, R.; Gabrilovich, D.; Heller, R.; Coppola, M.; et al. Regulation of the innate and adaptive immune responses by Stat-3 signaling in tumor cells. Nat. Med. 2003, 10, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Haura, E.B.; Turkson, J.; Jove, R. Mechanisms of Disease: insights into the emerging role of signal transducers and activators of transcription in cancer. Nat. Clin. Pr. Oncol. 2005, 2, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Yuan, F.; Qiao, L.; Chen, Y.; Qi, X.; Liu, Y.; Li, D.; Gu, Q.; Li, J.; Liu, M. AS1041, a Novel Synthesized Derivative of Marine Natural Compound Aspergiolide A, Arrests Cell Cycle, Induces Apoptosis, and Inhibits ERK Activation in K562 Cells. Mar. Drugs 2017, 346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.; Yue, P.; Fletcher, S.; Zhao, W.; Gunning, P.T.; Turkson, J. A novel small-molecule disrupts Stat3 SH2 domain–phosphotyrosine interactions and Stat3-dependent tumor processes. Biochem. Pharmacol. 2010, 79, 1398–1409. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, Z.; Mao, X.; Mertens, C.; Krishnaraj, R.; Qin, J.; Mandal, P.; Romanowski, M.J.; McMurray, J.S.; Chen, X. Crystal structure of unphosphorylated STAT3 core fragment. Biochem. Biophys. Res. Commun. 2008, 374, 1–5. [Google Scholar] [CrossRef]
- Spitzer, R.; Jain, A.N. Surflex-Dock: Docking benchmarks and real-world application. J. Comput. Mol. Des. 2012, 26, 687–699. [Google Scholar] [CrossRef]
- Lis, C.; Rubner, S.; Roatsch, M.; Berg, A.; Gilcrest, T.; Fu, D.; Nguyen, E.; Schmidt, A.-M.; Krautscheid, H.; Meiler, J.; et al. Development of Erasin: a chromone-based STAT3 inhibitor which induces apoptosis in Erlotinib-resistant lung cancer cells. Sci. Rep. 2017. [Google Scholar] [CrossRef] [Green Version]
- He, C.-X.; Ai, J.; Xing, W.-Q.; Chen, Y.; Zhang, H.-T.; Huang, M.; Hu, Y.-H.; Ding, J.; Geng, M.-Y. Yhhu3813 is a novel selective inhibitor of c-Met Kinase that inhibits c-Met-dependent neoplastic phenotypes of human cancer cells. Acta Pharmacol. Sin. 2013, 35, 89–97. [Google Scholar] [CrossRef] [Green Version]
Sample Availability: Samples of the compounds are not available from authors. |


| Compounds | Concentration (μM) | Inhibition Rate(%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Flt-1 | KDR | PDGFR-α | PDGFR-β | EGFR | ErbB4 | c-Src | ABL | EPH-A2 | IGF1R | ||
| TCN | 20.0 | 0 | 16.4 | 23.1 | 1.0 | 11.6 | 21.4 | 0.0 | 0.0 | 0.0 | 0.0 |
| Su11248 | 1.0 | 66.6 | 81.8 | 68.4 | 80.2 | ||||||
| BIBW2992 | 1.0 | 84.7 | 84.2 | ||||||||
| Dasatinib | 1.0 | 63.2 | 56.7 | 76.2 | |||||||
| AEW541 | 1.0 | 56.6 | |||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qi, X.; Li, M.; Zhang, X.-m.; Dai, X.-f.; Cui, J.; Li, D.-h.; Gu, Q.-q.; Lv, Z.-h.; Li, J. Trichothecin Inhibits Cancer-Related Features in Colorectal Cancer Development by Targeting STAT3. Molecules 2020, 25, 2306. https://doi.org/10.3390/molecules25102306
Qi X, Li M, Zhang X-m, Dai X-f, Cui J, Li D-h, Gu Q-q, Lv Z-h, Li J. Trichothecin Inhibits Cancer-Related Features in Colorectal Cancer Development by Targeting STAT3. Molecules. 2020; 25(10):2306. https://doi.org/10.3390/molecules25102306
Chicago/Turabian StyleQi, Xin, Meng Li, Xiao-min Zhang, Xiu-fen Dai, Jian Cui, De-hai Li, Qian-qun Gu, Zhi-hua Lv, and Jing Li. 2020. "Trichothecin Inhibits Cancer-Related Features in Colorectal Cancer Development by Targeting STAT3" Molecules 25, no. 10: 2306. https://doi.org/10.3390/molecules25102306
APA StyleQi, X., Li, M., Zhang, X.-m., Dai, X.-f., Cui, J., Li, D.-h., Gu, Q.-q., Lv, Z.-h., & Li, J. (2020). Trichothecin Inhibits Cancer-Related Features in Colorectal Cancer Development by Targeting STAT3. Molecules, 25(10), 2306. https://doi.org/10.3390/molecules25102306






