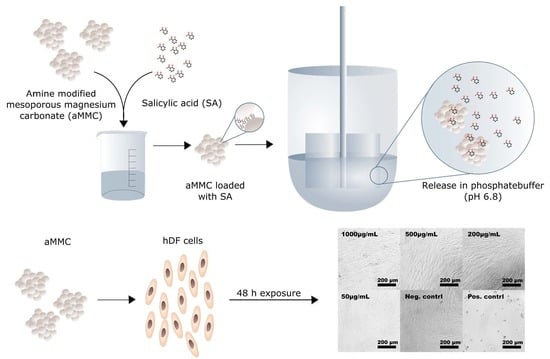
Exploring the Use of Amine Modified Mesoporous Magnesium Carbonate for the Delivery of Salicylic Acid in Topical Formulations: In Vitro Cytotoxicity and Drug Release Studies
Abstract
1. Introduction
2. Results and Discussion
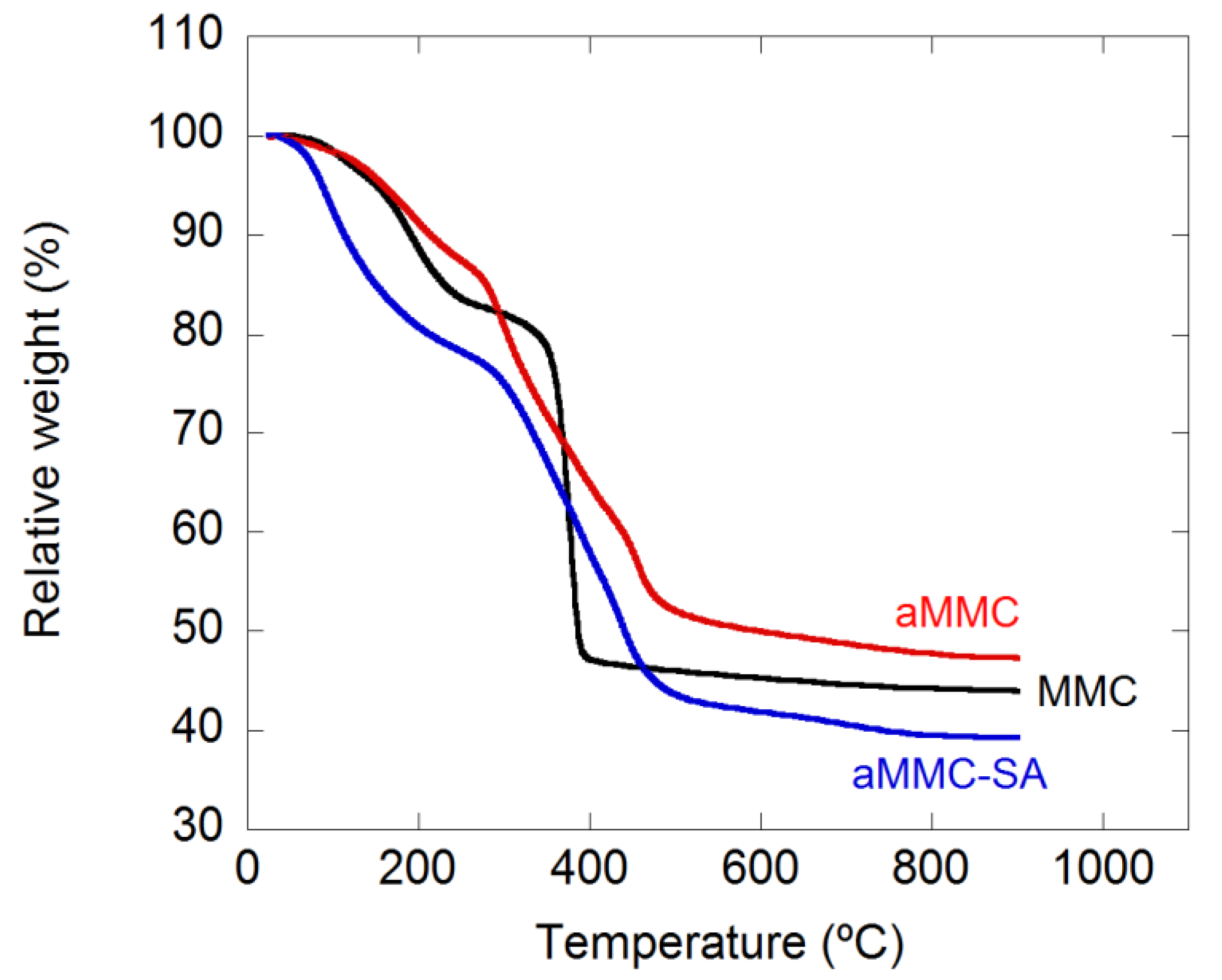
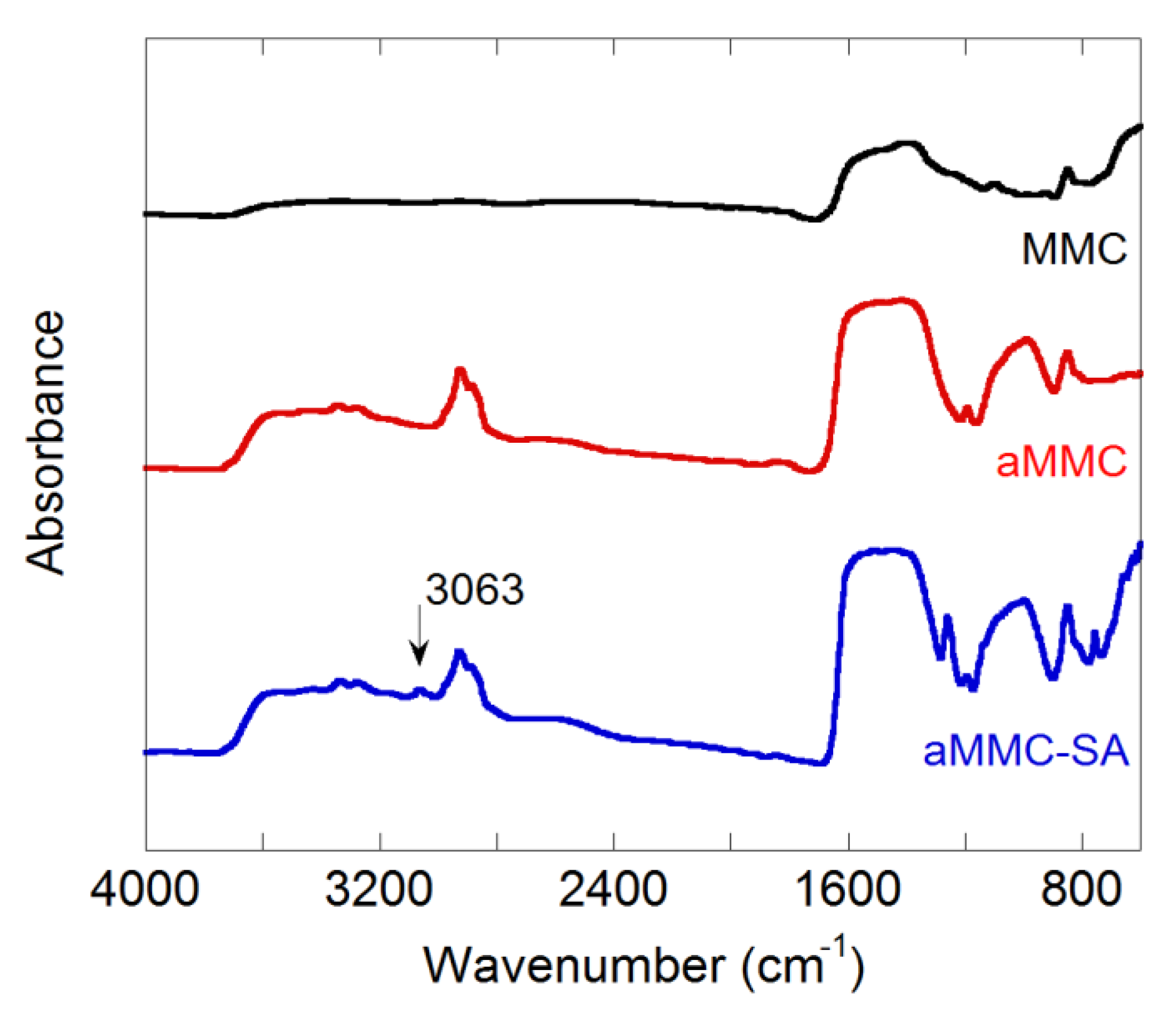
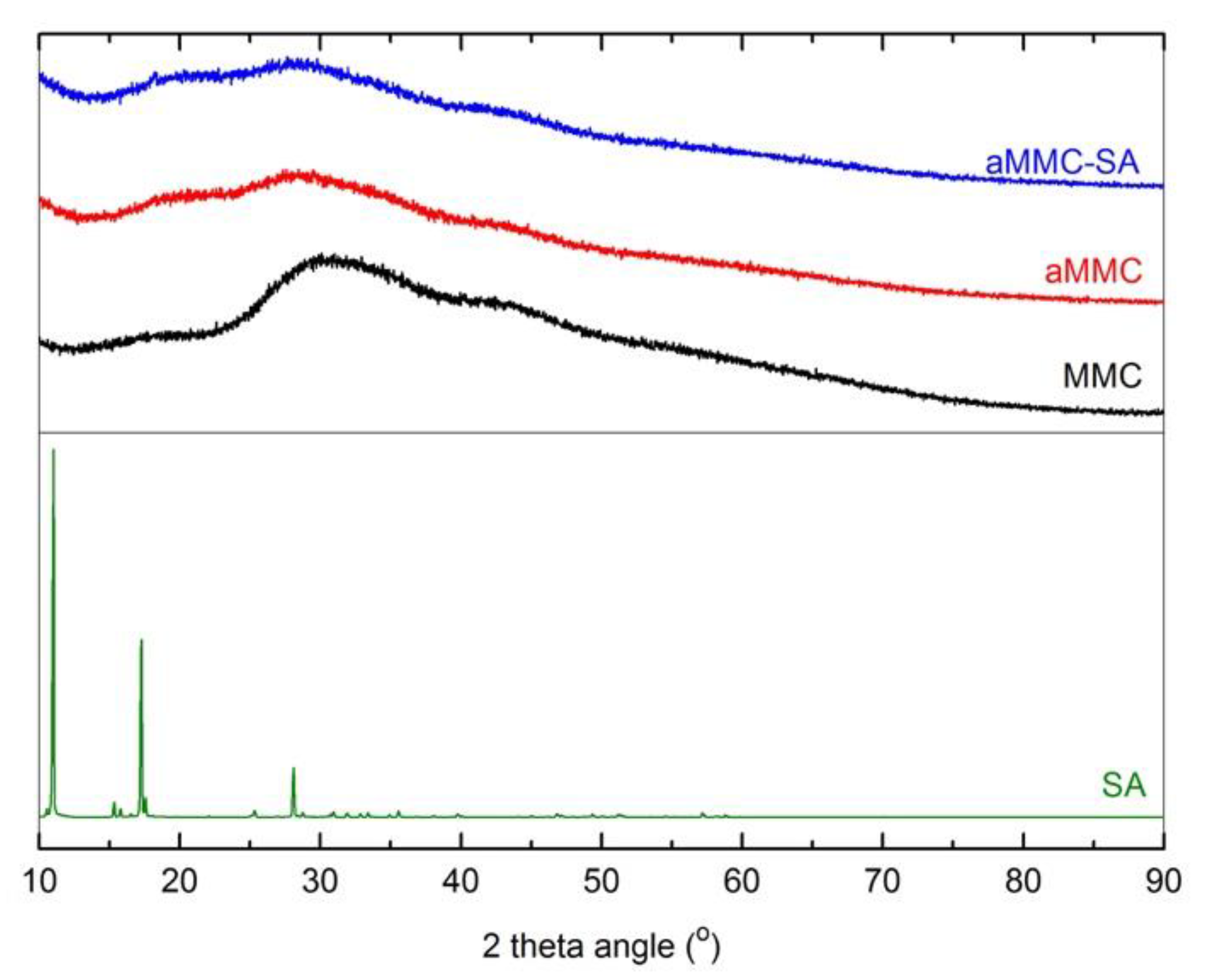
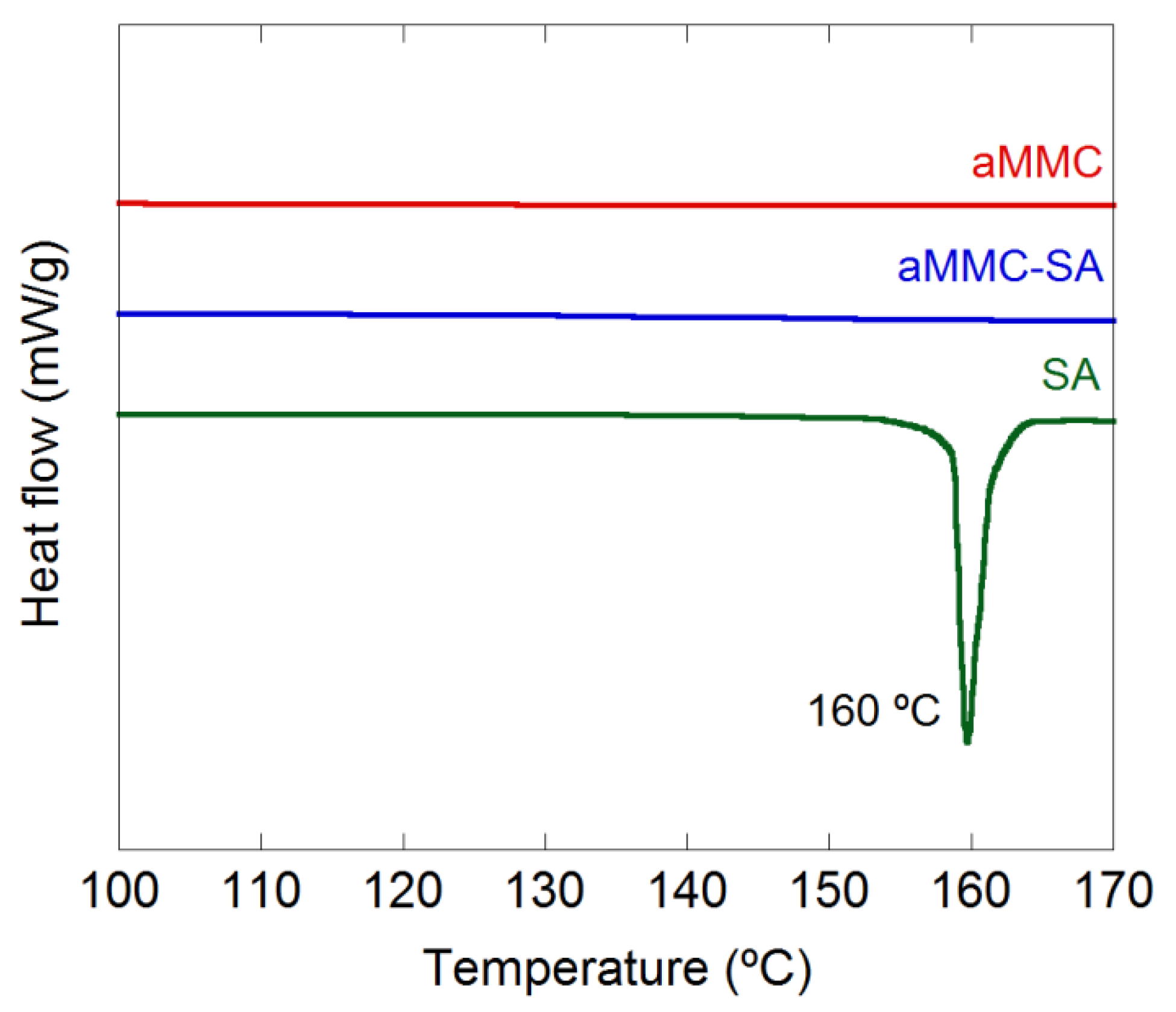
2.1. Material Characterization
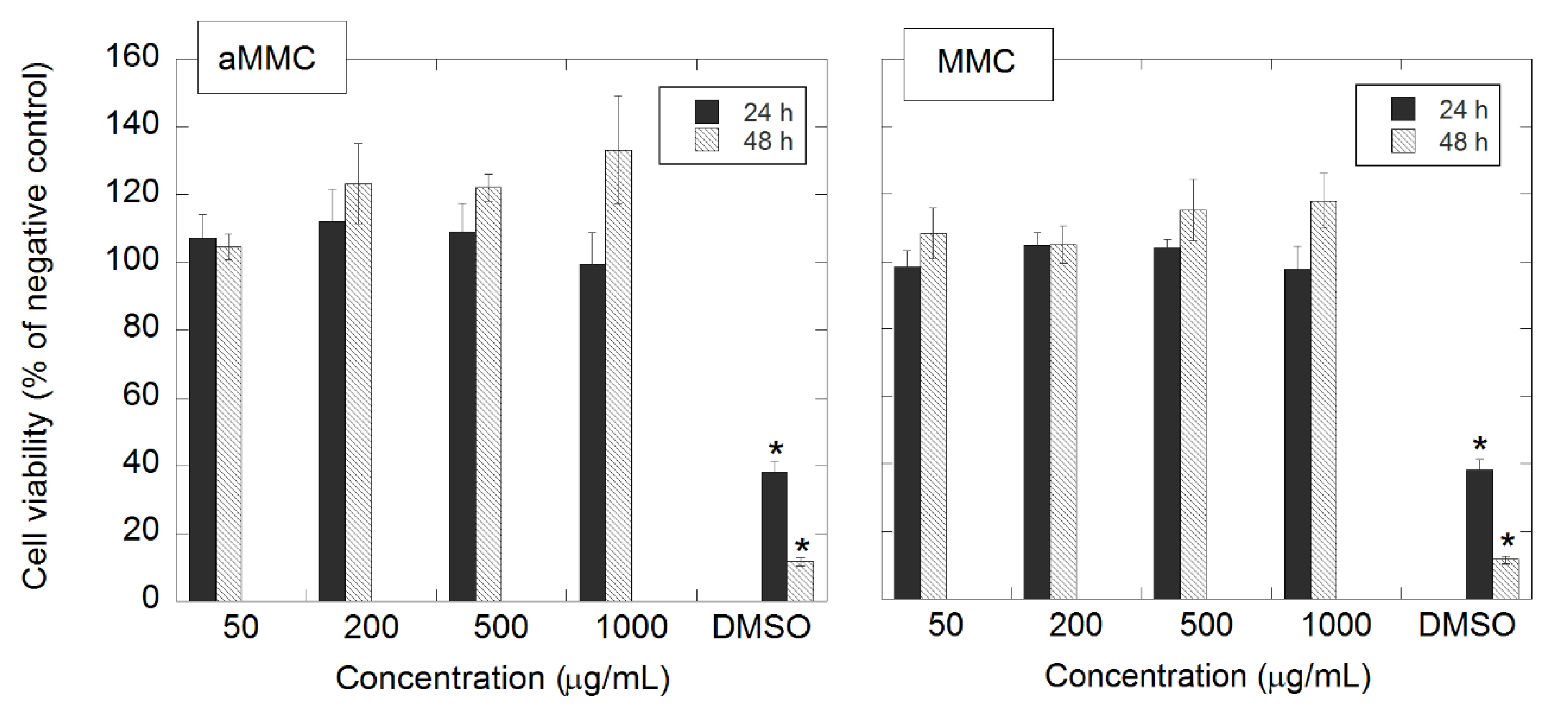
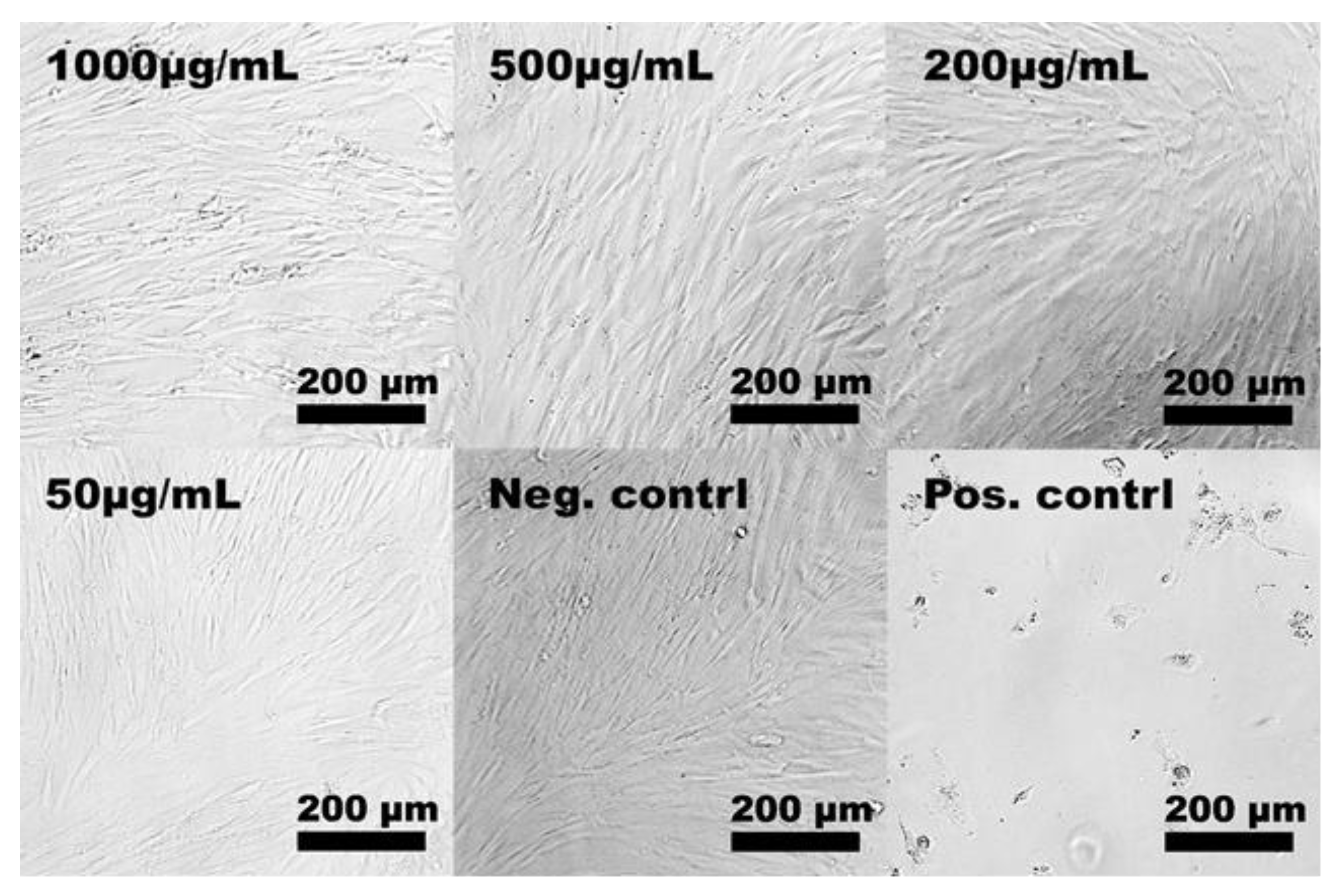
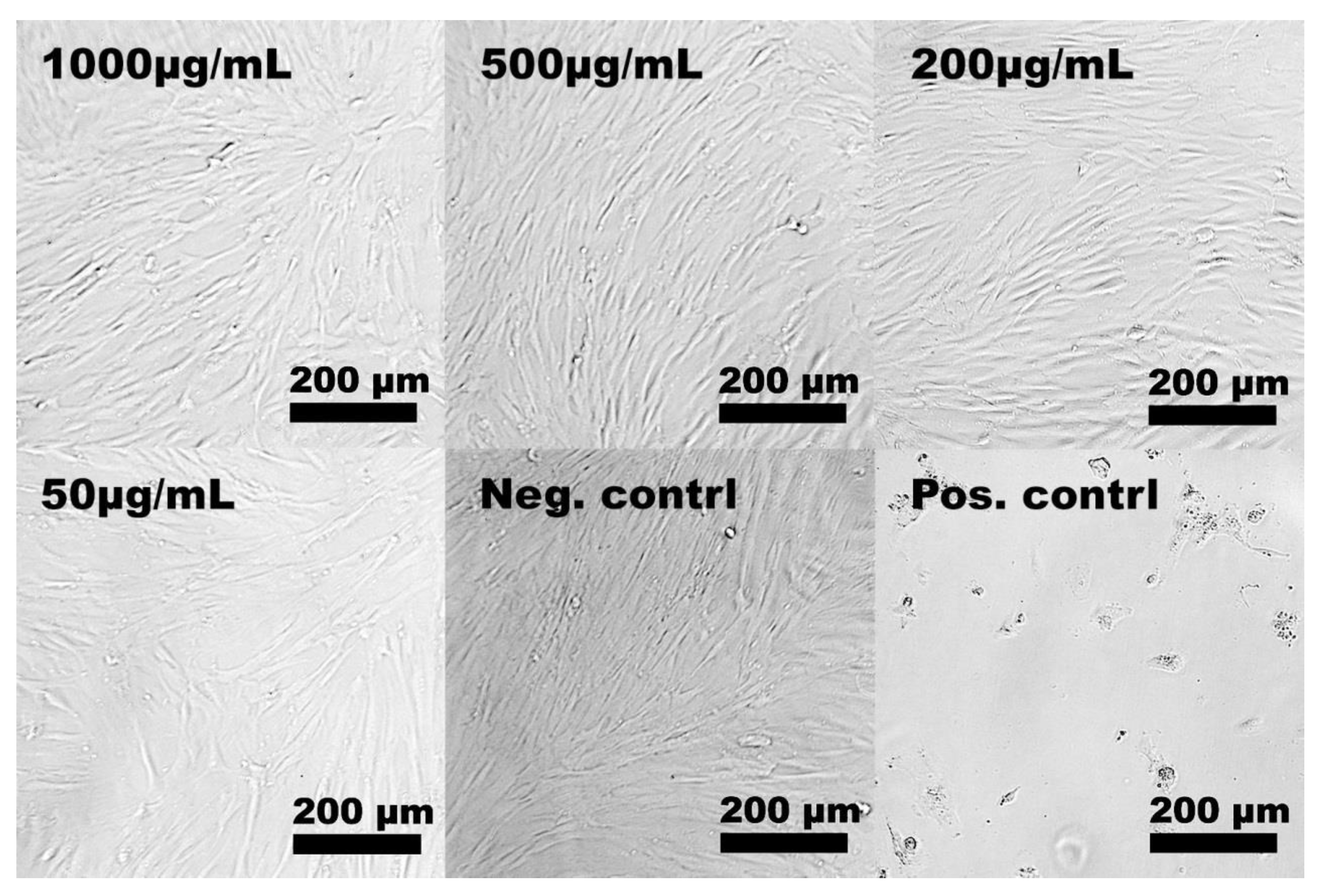
2.2. In Vitro Cytotoxicity
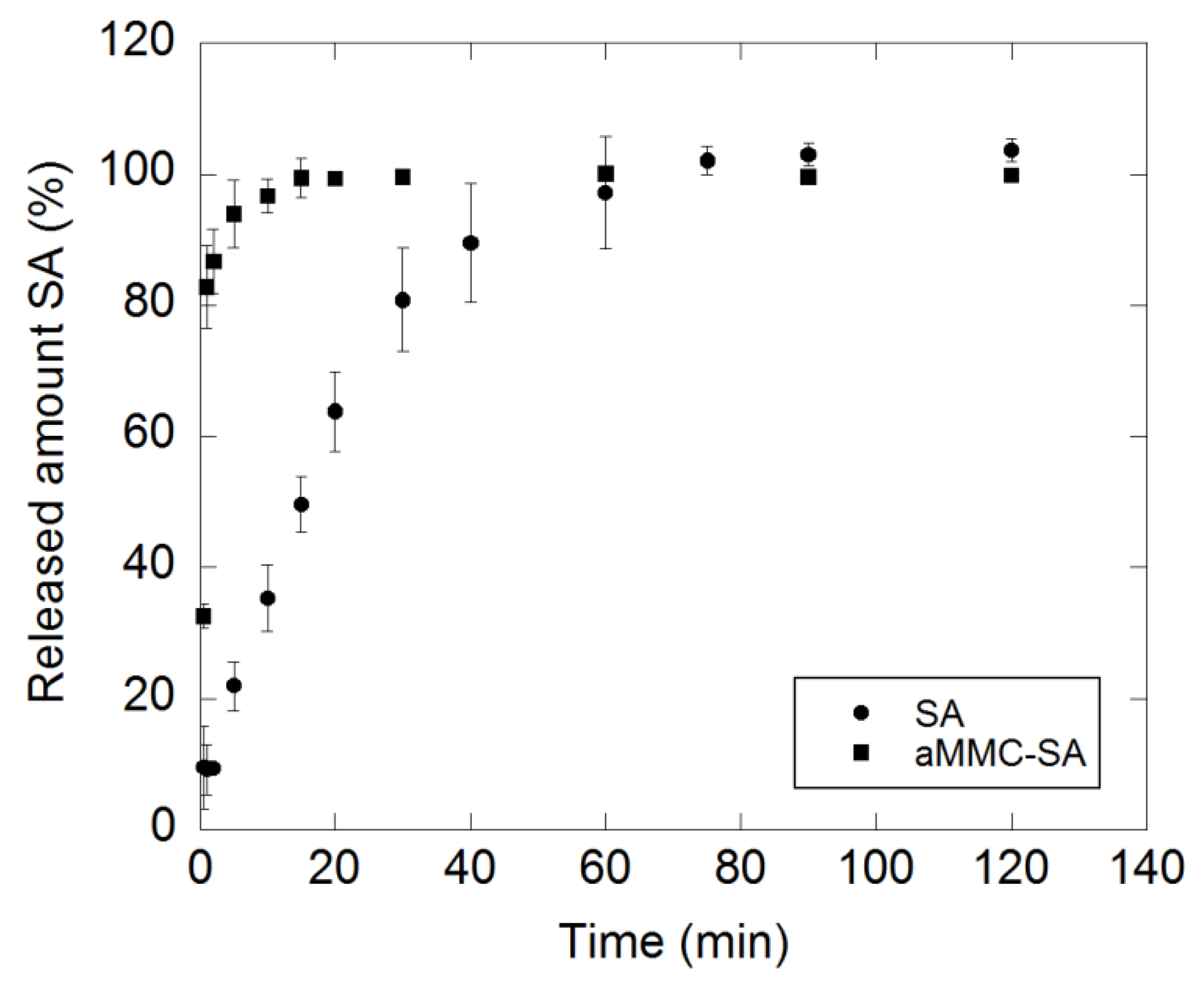
2.3. Drug-Release Test
3. Materials and Methods
3.1. Synthesis of MMC
3.2. Amine Modification of MMC
3.3. Material Characterization
3.3.1. N2 Sorption Analysis
3.3.2. X-ray Powder Diffraction (XRD)
3.3.3. Differential Scanning Calorimetry (DSC)
3.3.4. Thermal Gravimetric Analysis (TGA)
3.3.5. Fourier Transform Infrared Spectroscopy (FTIR)
3.3.6. Scanning Electron Microscopy (SEM)
3.4. In Vitro Cytotoxicity
3.4.1. Alamar Blue Assay
3.4.2. Light Microscopy
3.4.3. Statistical Analysis
3.5. Drug Loading Procedure and Release Test
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lin, A.N.; Nakatsui, T. Salicylic acid revisited. Int. J. Dermatol. 1998, 37, 335–342. [Google Scholar]
- Tosti, A.; Grimes, P.E.; Pia de Padova, M. Color Atlas of Chemical Peels; Philipp, M., Blasig, E., Eds.; Springer: Heidelberg, Germany, 2006. [Google Scholar]
- Arif, T. Salicylic acid as a peeling agent: A comprehensive review. Clin. Cosmet. Investig. Dermatol. 2015, 8, 455–461. [Google Scholar] [CrossRef]
- Flower, R.; Gryglewski, R.; Herbaczynska-Cedro, K.; Vane, J.R. Effects of Anti-inflammatory Drugs on Prostaglandin Biosynthesis. Nat. New Biol. 1972, 238, 104–106. [Google Scholar] [CrossRef]
- Madan, R.K.; Levitt, J.M. A review of toxicity from topical salicylic acid preparations. J. Am. Acad. Dermatol. 2014, 70, 788–792. [Google Scholar] [CrossRef]
- Kraft, J.; Freiman, A. Management of acne. Can. Med. Assoc. 2011, 183, E430–E435. [Google Scholar] [CrossRef]
- Marczyk, B.; Mucha, P.; Budzisz, E.; Rotsztejn, H. Comparative study of the effect of 50% pyruvic and 30% salicylic peels on the skin lipid film in patients with acne vulgaris. J. Cosmet. Dermatol. 2014, 13, 15–21. [Google Scholar] [CrossRef]
- Lebwohl, M. The role of salicylic acid in the treatment of psoriasis. Int. J. Dermatol. 1999, 38, 16–24. [Google Scholar] [CrossRef]
- Elewski, B.E. Clinical Diagnosis of Common Scalp Disorders Mycotic Conditions. J. Investig. Dermatol. Symp. Proc. 2005, 10, 190–193. [Google Scholar] [CrossRef]
- Dasgupta, Q.; Chatterjee, K.; Madras, G. Controlled Release of Salicylic Acid from Biodegradable Cross-Linked Polyesters. Mol. Pharm. 2015, 12, 3479–3489. [Google Scholar] [CrossRef]
- Vilaça, N.; Morais-Santos, F.; Machado, A.F.; Sirkecioğlu, A.; Pereira, M.F.R.; Sardo, M.; Rocha, J.; Parpot, P.; Fonseca, A.M.; Baltazar, F.; et al. Micro- and mesoporous structures as drug delivery carriers for salicylic acid. J. Phys. Chem. C 2015, 119, 3589–3595. [Google Scholar] [CrossRef]
- Kurczewska, J.; Pecyna, P.; Ratajczak, M.; Gajęcka, M.; Schroeder, G. Halloysite nanotubes as carriers of vancomycin in alginate-based wound dressing. Saudi Pharm. J. 2017, 25, 911–920. [Google Scholar] [CrossRef]
- Forsgren, J.; Frykstrand, S.; Grandfield, K.; Mihranyan, A.; Strømme, M. A Template-Free, Ultra-Adsorbing, High Surface Area Carbonate Nanostructure. PLoS ONE 2013, 8, e68486. [Google Scholar] [CrossRef]
- Frykstrand, S.; Forsgren, J.; Mihranyan, A.; Strømme, M. On the pore forming mechanism of Upsalite, a micro- and mesoporous magnesium carbonate. Micropor. Mesopor. Mat. 2014, 190, 99–104. [Google Scholar] [CrossRef]
- Cheung, O.; Zhang, P.; Frykstrand, S.; Zheng, H.; Yang, T.; Sommariva, M.; Zou, X.; Strømme, M. Nanostructure and pore size control of template-free synthesised mesoporous magnesium carbonate. RSC Adv. 2016, 6, 74241–74249. [Google Scholar] [CrossRef]
- US Food and Drug Administration. U.S. Department of Helth & Human Services; US Food and Drug Administration: Silver Spring, MD, USA, 2018.
- Wade, A.; Weller, P.J. Handbook Pharmaceutical Excipients, 6th ed.; Rowe, R.C., Sheskey, P.J., Quinn, M.E., Eds.; Pharmaceutical Press: London, UK, 2009; ISBN 978-0-12-088479-7. [Google Scholar]
- Frykstrand, S.; Forsgren, J.; Zhang, P.; Strømme, M.; Ferraz, N. Cytotoxicity, in Vivo Skin Irritation and Acute Systemic Toxicity of the Mesoporous Magnesium Carbonate Upsalite®. J. Biomater. Nanobiotechnol. 2015, 6, 257–266. [Google Scholar] [CrossRef]
- Welch, K.; Latifzada, M.A.; Frykstrand, S.; Strømme, M. Investigation of the Antibacterial Effect of Mesoporous Magnesium Carbonate. ACS Omega 2016, 1, 907–914. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Forsgren, J.; Strømme, M. Stabilisation of amorphous ibuprofen in Upsalite, a mesoporous magnesium carbonate, as an approach to increasing the aqueous solubility of poorly soluble drugs. Int. J. Pharm. 2014, 472, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Zardán Gómez de la Torre, T.; Welch, K.; Bergström, C.; Strømme, M. Supersaturation of poorly soluble drugs induced by mesoporous magnesium carbonate. Eur. J. Pharm. Sci. 2016, 93, 468–474. [Google Scholar] [CrossRef]
- Yang, J.; Alvebratt, C.; Zhang, P.; Zardán Gómez de la Torre, T.; Strømme, M.; Bergström, C.; Welch, K. Enhanced release of poorly water-soluble drugs from synergy between mesoporous magnesium carbonate and polymers. Int. J. Pharm. 2017, 525, 183–190. [Google Scholar] [CrossRef]
- Bacsik, Z.; Atluri, R.; Garcia-Bennett, A.E.; Hedin, N. Temperature-induced uptake of CO2 and formation of carbamates in mesocaged silica modified with n-propylamines. Langmuir 2010, 26, 10013–10024. [Google Scholar] [CrossRef]
- Bacsik, Z.; Ahlsten, N.; Ziadi, A.; Zhao, G.; Garcia-Bennett, A.E.; Martin-Matute, B.; Hedin, N. Mechanisms and Kinetics for Sorption of CO2 on Bicontinuous Mesoporous Silica Modified with n-Propylamine. Langmuir 2011, 27, 11118–11128. [Google Scholar] [CrossRef]
- Braz, W.R.; Rocha, N.L.; de Faria, E.H.; e Silva, M.L.A.; Ciuffi, K.J.; Tavares, D.C.; Furtado, R.A.; Rocha, L.A.; Nassar, E.J. Incorporation of anti-in fl ammatory agent into mesoporous silica. Nanotechnology 2016, 27, 385103. [Google Scholar] [CrossRef]
- Jambhrunkar, S.; Qu, Z.; Popat, A.; Yang, J.; Noonan, O.; Acauan, L.; Ahmad Nor, Y.; Yu, C.; Karmakar, S. Effect of surface functionality of silica nanoparticles on cellular uptake and cytotoxicity. Mol. Pharm. 2014, 11, 3642–3655. [Google Scholar] [CrossRef]
- Lee, D.H.; Choi, M.; Yu, B.W.; Ryoo, R. Organic functionalization of mesopore walls in hierarchically porous zeolites. Chem. Commun. 2009, 1, 74–76. [Google Scholar] [CrossRef]
- Datt, A.; El-Maazawi, I.; Larsen, S.C. Aspirin loading and release from MCM-41 functionalized with aminopropyl groups via Co-condensation or postsynthesis modification methods. J. Phys. Chem. C 2012, 116, 18358–18366. [Google Scholar] [CrossRef]
- Vall, M.; Zhang, P.; Gao, A.; Frykstrand, S.; Cheung, O.; Strømme, M. Effects of amine modification of mesoporous magnesium carbonate on controlled drug release. Int. J. Pharm. 2017, 524, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Pochard, I.; Vall, M.; Erikssson, J.; Farineau, C.; Cheung, O.; Frykstrand, S.; Welch, K.; Strømme, M. Amine-functionalised mesoporous magnesium carbonate: Dielectric spectroscopy studies of interactions with water and stabilitye. Mater. Chem. Phys. 2018, 216, 332–338. [Google Scholar] [CrossRef]
- Vall, M.; Strømme, M.; Cheung, O. Amine-Modified Mesoporous Magnesium Carbonate as an Effective Adsorbent for Azo Dyes. ACS Omega 2019, 4, 2973–2979. [Google Scholar] [CrossRef]
- Gatoo, M.A.; Naseem, S.; Arfat, M.Y.; Mahmood, D.A.; Qasim, K.; Zubair, S. Physicochemical Properties of Nanomaterials: Implication in Associated Toxic Manifestations. Biomed Res. Int. 2014, 2014, 1–8. [Google Scholar] [CrossRef]
- Zhang, P.; Zardán Gómez De La Torre, T.; Forsgren, J.; Bergström, C.A.S.; Strømme, M. Diffusion-Controlled Drug Release from the Mesoporous Magnesium Carbonate Upsalite®. J. Pharm. Sci. 2016, 105, 657–663. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of gases in multimolecular layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |

| SBET (m2/g) | Pore Diameter (nm) | Pore Volume (cm3/g) | Particle Size (μm) a | |
|---|---|---|---|---|
| MMC | 519 | 6.7 | 1.05 | 270 |
| aMMC | 509 | 5.5 | 0.70 | 19 |
| aMMC-SA | 441 | 5.5 | 0.57 | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vall, M.; Ferraz, N.; Cheung, O.; Strømme, M.; Zardán Gómez de la Torre, T. Exploring the Use of Amine Modified Mesoporous Magnesium Carbonate for the Delivery of Salicylic Acid in Topical Formulations: In Vitro Cytotoxicity and Drug Release Studies. Molecules 2019, 24, 1820. https://doi.org/10.3390/molecules24091820
Vall M, Ferraz N, Cheung O, Strømme M, Zardán Gómez de la Torre T. Exploring the Use of Amine Modified Mesoporous Magnesium Carbonate for the Delivery of Salicylic Acid in Topical Formulations: In Vitro Cytotoxicity and Drug Release Studies. Molecules. 2019; 24(9):1820. https://doi.org/10.3390/molecules24091820
Chicago/Turabian StyleVall, Maria, Natalia Ferraz, Ocean Cheung, Maria Strømme, and Teresa Zardán Gómez de la Torre. 2019. "Exploring the Use of Amine Modified Mesoporous Magnesium Carbonate for the Delivery of Salicylic Acid in Topical Formulations: In Vitro Cytotoxicity and Drug Release Studies" Molecules 24, no. 9: 1820. https://doi.org/10.3390/molecules24091820
APA StyleVall, M., Ferraz, N., Cheung, O., Strømme, M., & Zardán Gómez de la Torre, T. (2019). Exploring the Use of Amine Modified Mesoporous Magnesium Carbonate for the Delivery of Salicylic Acid in Topical Formulations: In Vitro Cytotoxicity and Drug Release Studies. Molecules, 24(9), 1820. https://doi.org/10.3390/molecules24091820