Recent Advances on Metal-Free, Visible-Light- Induced Catalysis for Assembling Nitrogen- and Oxygen-Based Heterocyclic Scaffolds
Abstract
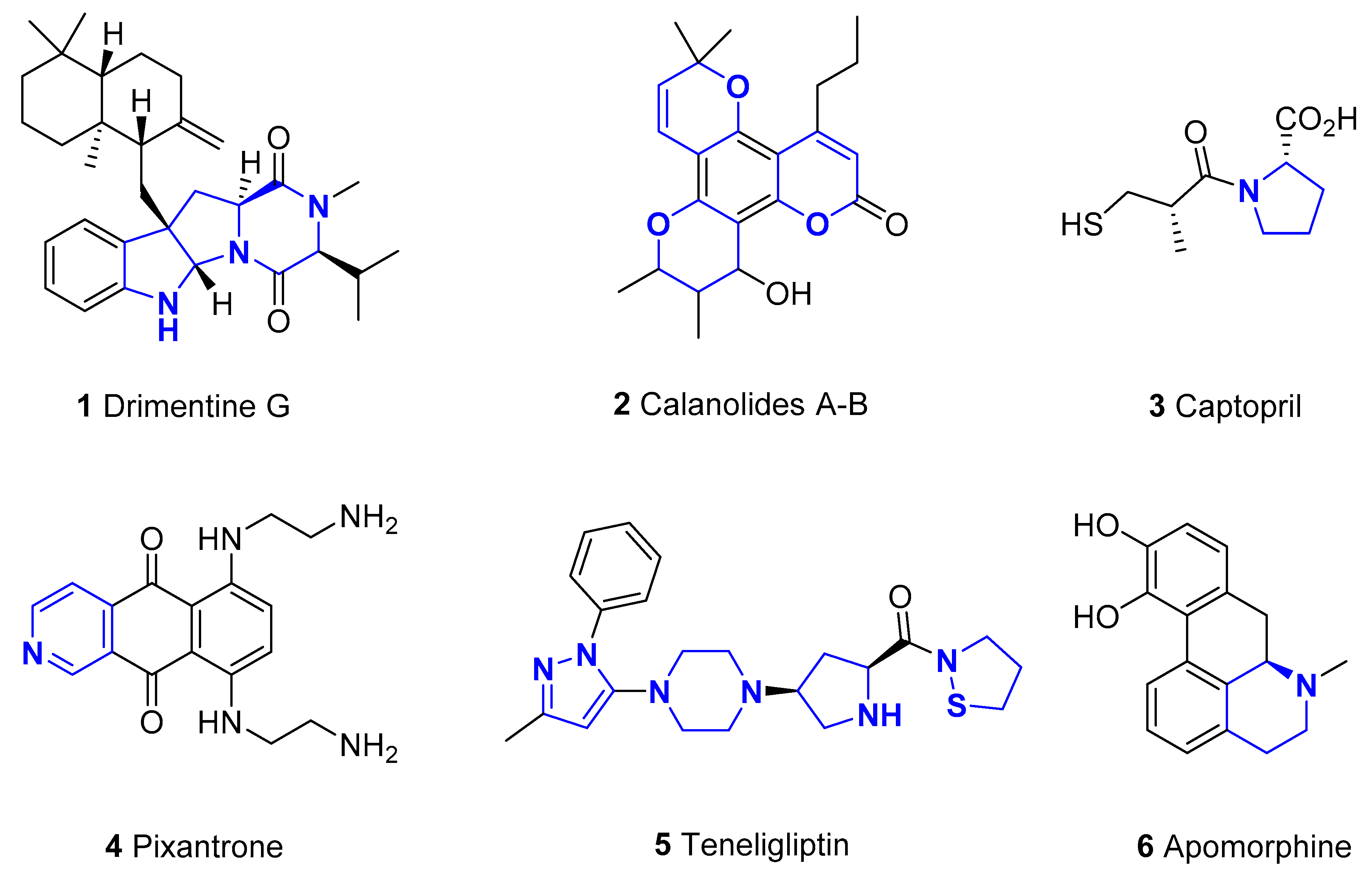
1. Introduction
2. Application of Visible-Light-Mediated Catalysis in Synthesis of Heterocyclic Compounds
2.1. N-containing Heterocycles
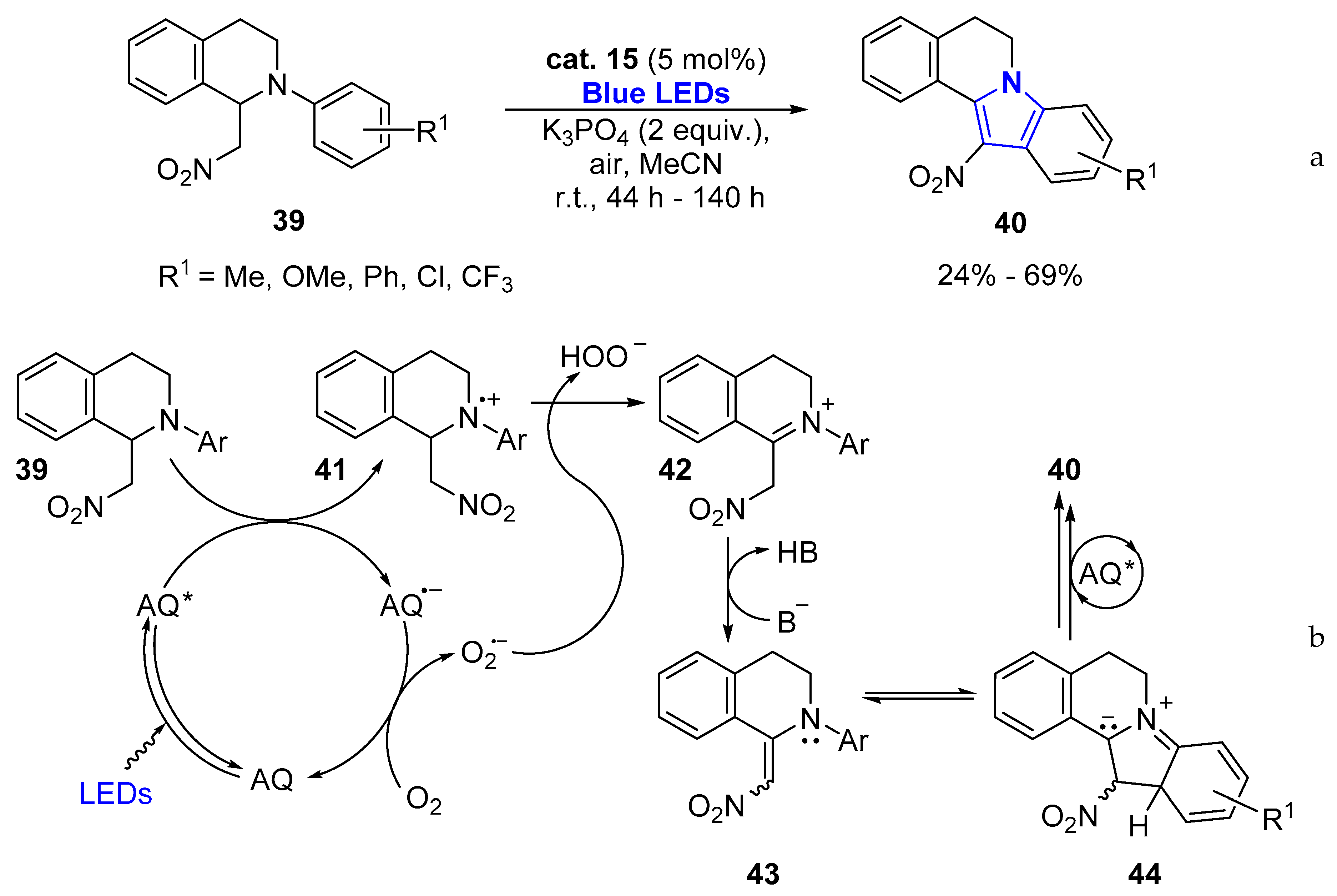
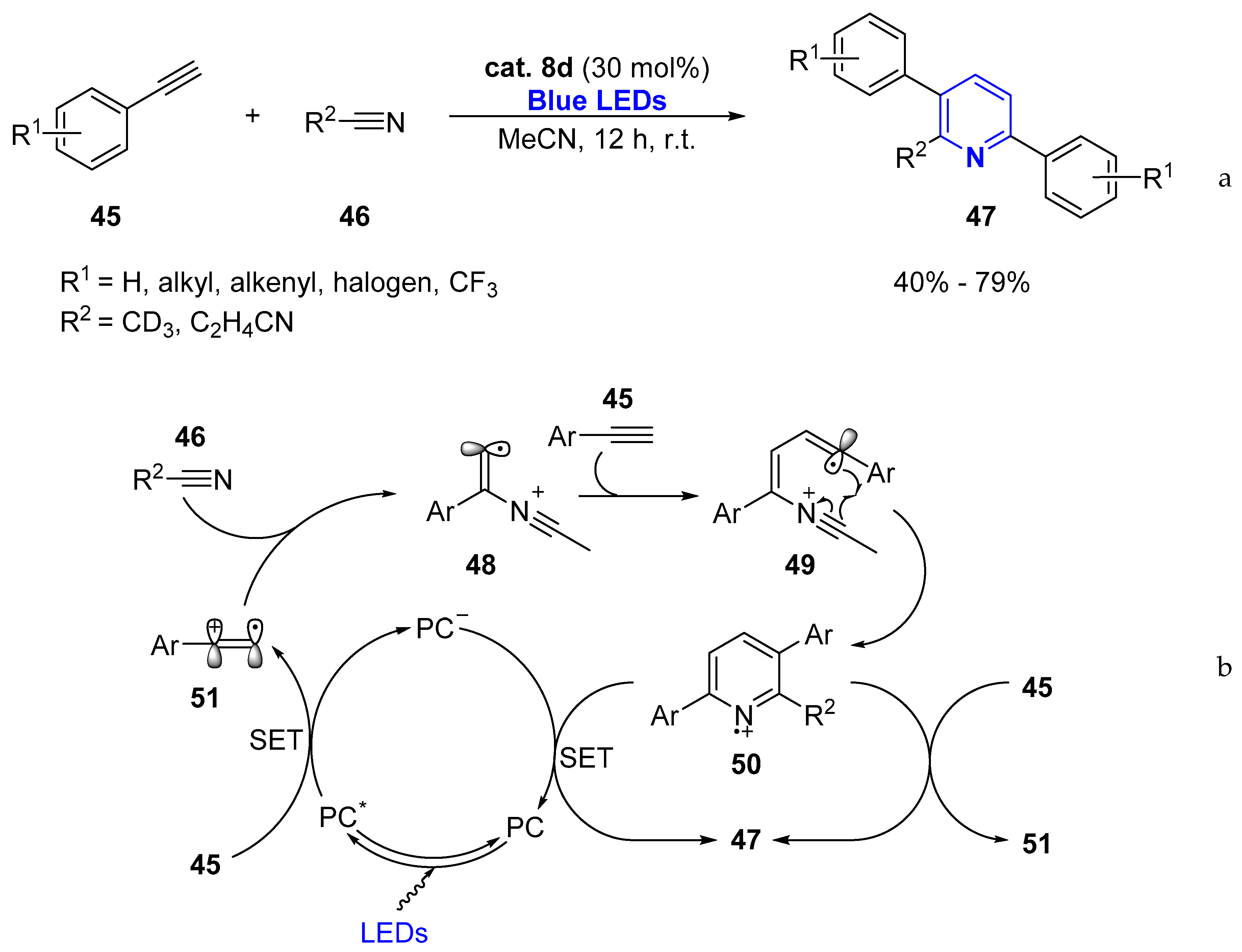
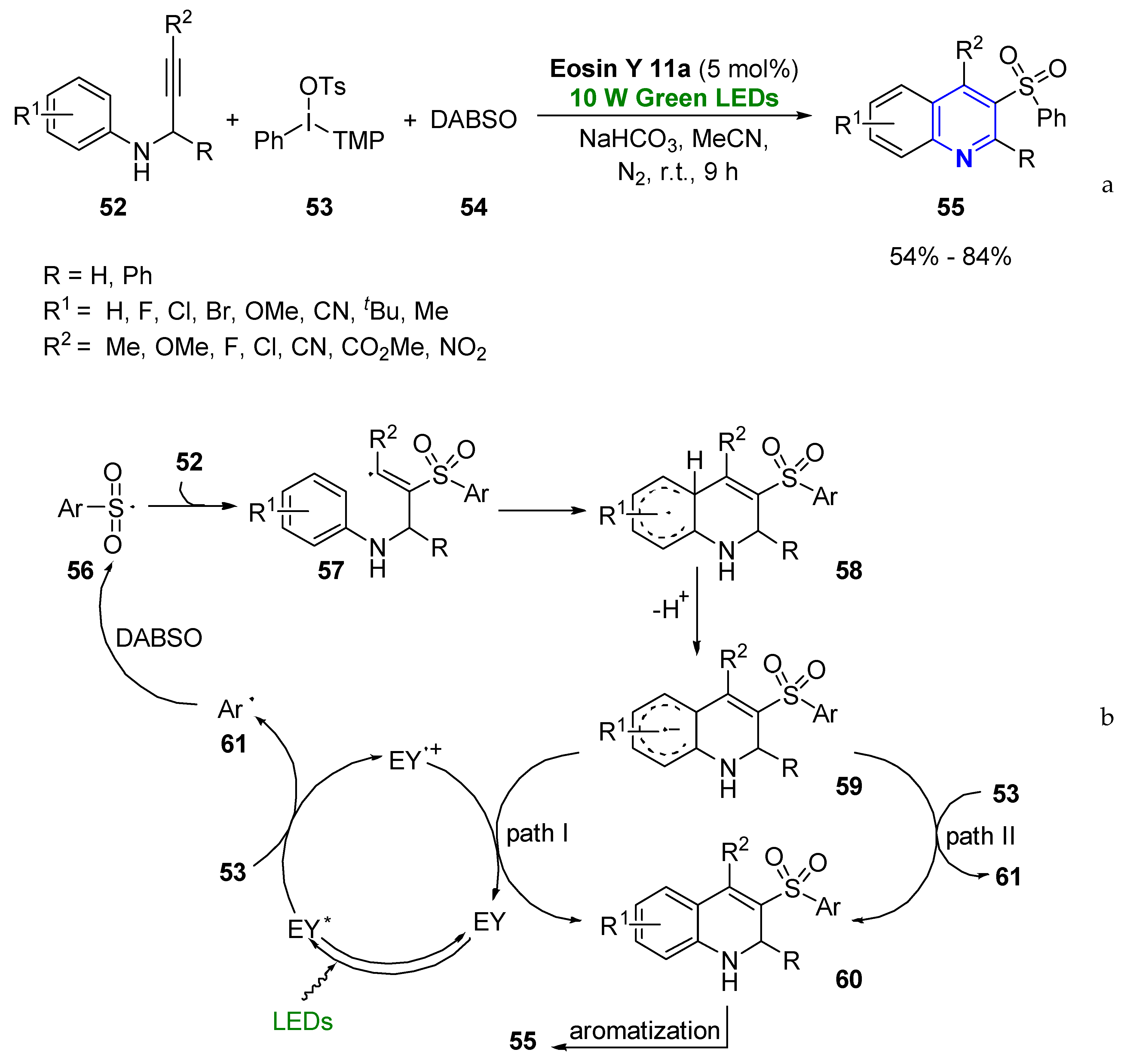
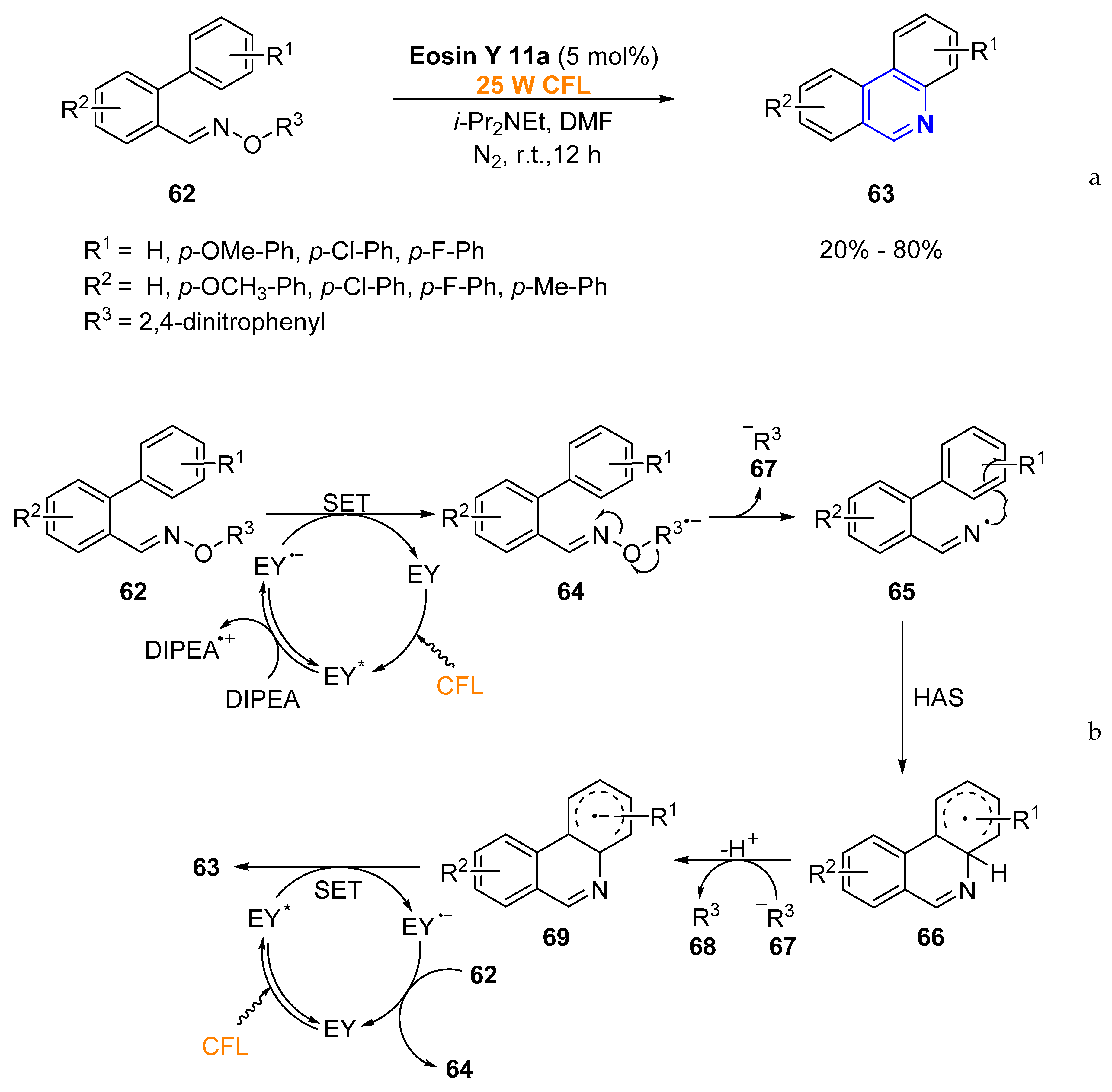
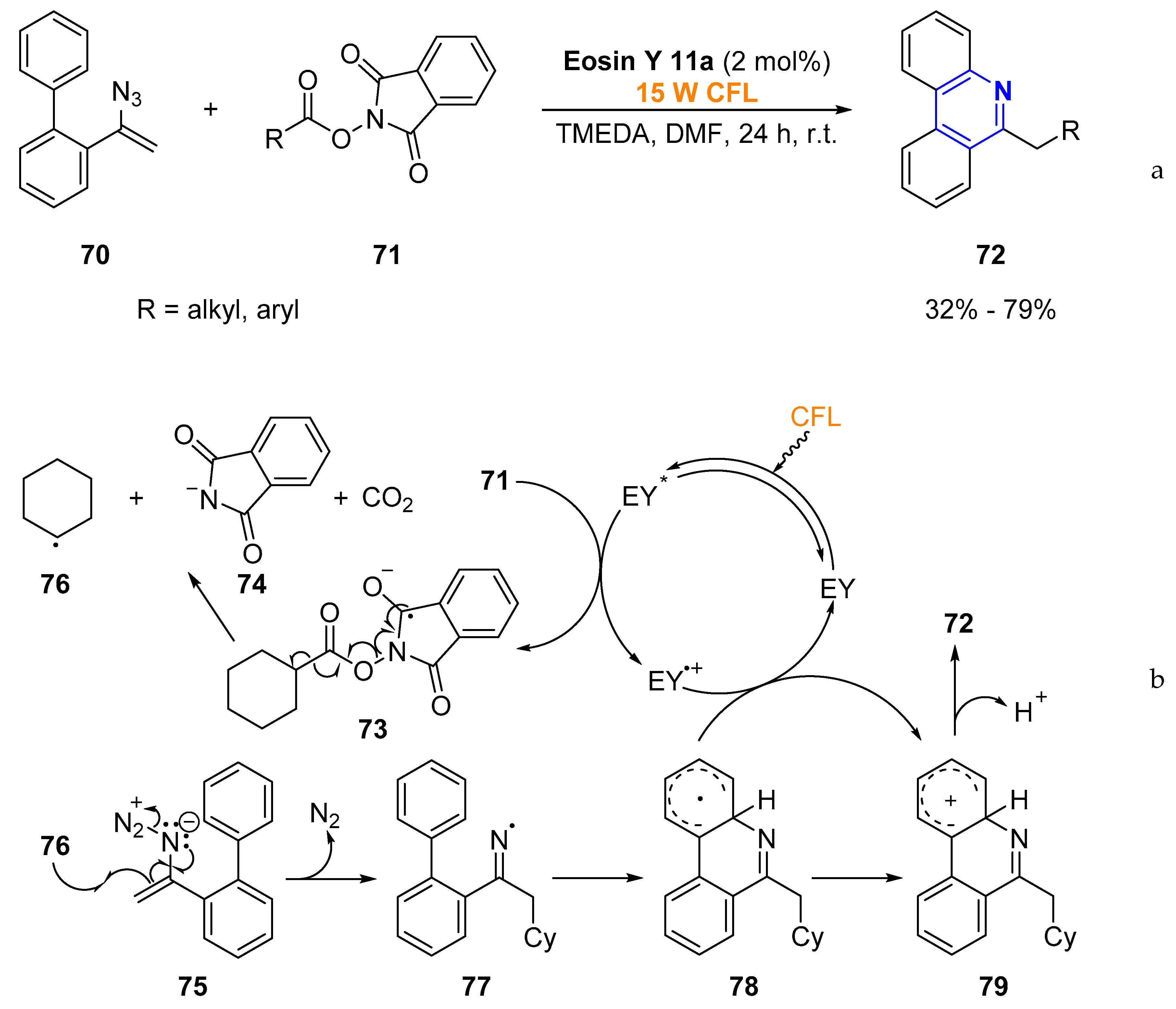
2.1.1. Aromatic Heterocycles
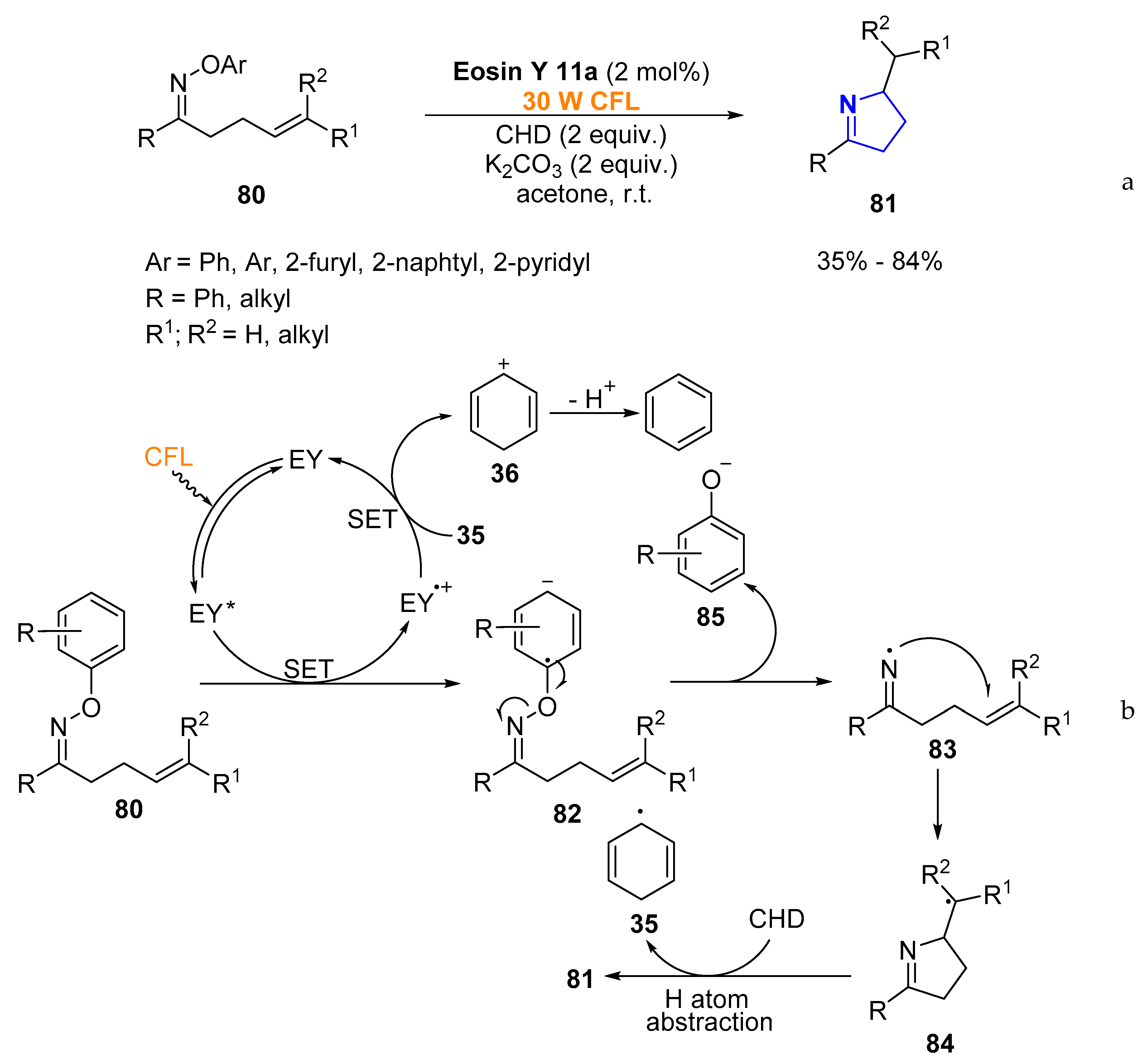
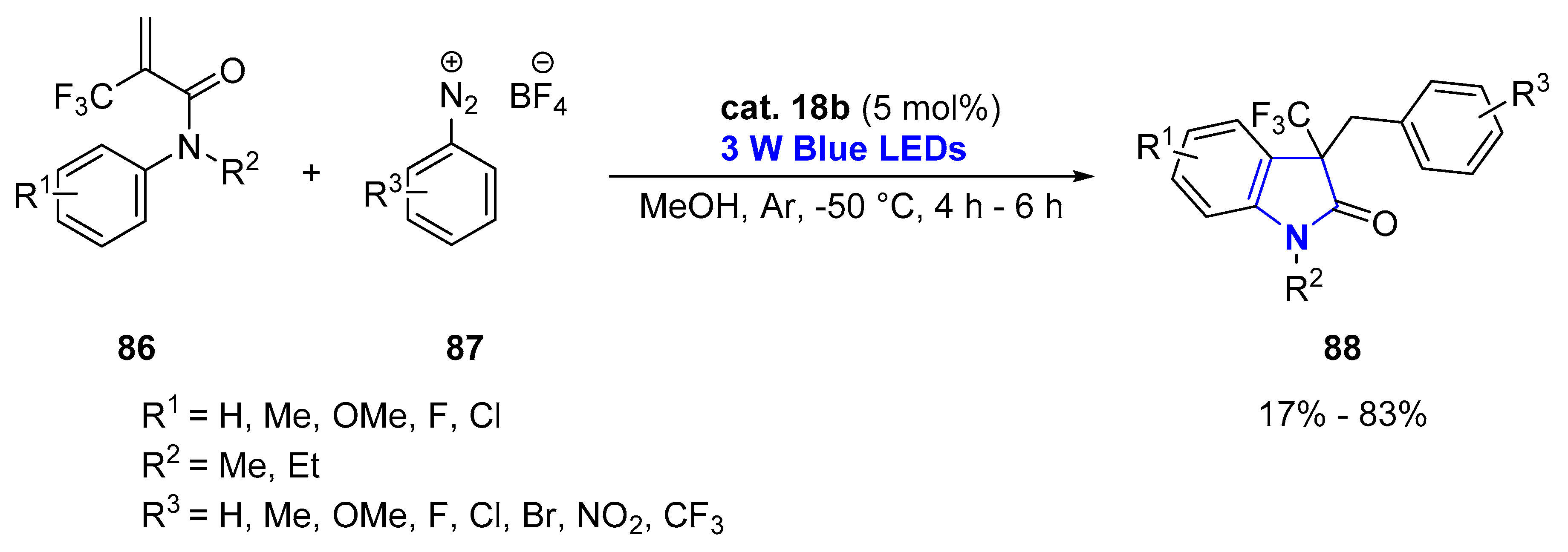
2.1.2. Non-Aromatic Heterocycles
2.2. O-Containing Heterocycles
2.2.1. Aromatic Heterocycles
2.2.2. Non-Aromatic Heterocycles
2.3. Heterocycles Bearing More Than One Heteroatom
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Joule, J.A.; Mills, K. Heterocyclic Chemistry, 5th ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2010. [Google Scholar]
- Pozharskii, A.F.; Soldatenkov, A.T.; Katritzky, A.R. Heterocycles in Life and Society: An Introduction to Heterocyclic Chemistry, Biochemistry and Applications, 2nd ed.; Wiley: Hoboken, NJ, USA, 2011. [Google Scholar]
- Eicher, T.; Hauptmann, S.; Speicher, A. The Chemistry of Heterocycles: Structures, Reactions, Synthesis, and Applications; 3rd Completely Revised and Enlarged ed.; Wiley-VCH: Weinheim, Germany, 2013. [Google Scholar]
- Creagh, T.; Ruckle, J.L.; Tolbert, D.T.; Giltner, J.; Eiznhamer, D.A.; Dutta, B.; Flavin, M.T.; Xu, Z.Q. Safety and pharmacokinetics of single doses of (+)-Calanolide a, a novel, naturally occurring nonnucleoside reverse transcriptase inhibitor, in healthy, human immunodeficiency virus-negative human subjects. Antimicrob. Agents Chemother. 2001, 45, 1379–1386. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef]
- Sun, Y.; Li, R.; Zhang, W.; Li, A. Total synthesis of Indotertine A and Drimentines A, F, and G. Angew. Chem. Int. Ed. 2013, 52, 9201–9204. [Google Scholar] [CrossRef]
- Yajima, T.; Yajima, K.; Hayashi, M.; Takahashi, H.; Yasuda, K. Efficacy and safety of teneligliptin in addition to insulin therapy in type 2 diabetes mellitus patients on hemodialysis evaluated by continuous glucose monitoring. Diabetes Res. Clin. Pract. 2016, 122, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S. Visible-Light-Active Photocatalysis: Nanostructured Catalyst Design, Mechanisms, and Applications; Wiley-VCH: Weinheim, Germany, 2018. [Google Scholar]
- Stephenson, C.R.J.; Yoon, T.P.; MacMillan, D.W.C. Visible Light Photocatalysis in Organic Chemistry; Wiley-VCH: Weinheim, Germany, 2018. [Google Scholar]
- Parsons, A.F. An Introduction to Free Radical Chemistry; Wiley-Blackwell: Hoboken, NJ, USA, 2000. [Google Scholar]
- Togo, H. Advanced Free Radical Reactions for Organic Synthesis; Elsevier Science: Amsterdam, The Netherlands, 2004. [Google Scholar]
- Romero, N.A.; Nicewicz, D.A. Organic Photoredox Catalysis. Chem. Rev. 2016, 116, 10075–10166. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Studer, A. Recent advances in the synthesis of nitrogen heterocycles via radical cascade reactions using isonitriles as radical acceptors. Chem. Soc. Rev. 2015, 44, 3505–3521. [Google Scholar] [CrossRef]
- Chen, J.R.; Hu, X.Q.; Lu, L.Q.; Xiao, W.J. Exploration of Visible-Light Photocatalysis in Heterocycle Synthesis and Functionalization: Reaction Design and Beyond. Acc. Chem. Res. 2016, 49, 1911–1923. [Google Scholar] [CrossRef]
- Tanoury, G. Photochemical Synthesis of Azaheterocycles. Synthesis 2016, 48, 2009–2025. [Google Scholar] [CrossRef]
- Zhou, L.; Lokman Hossain, M.; Xiao, T. Synthesis of N-Containing Heterocyclic Compounds Using Visible-light Photoredox Catalysis. Chem. Rec. 2016, 16, 319–334. [Google Scholar] [CrossRef] [PubMed]
- Budén, M.E.; Bardagi, J.I.; Rossi, R.A. Constructing Heterocycles by Visible Light Photocatalysis. Curr. Org. Synth. 2017, 14, 398–429. [Google Scholar]
- Xuan, J.; Studer, A. Radical cascade cyclization of 1,n-enynes and diynes for the synthesis of carbocycles and heterocycles. Chem. Soc. Rev. 2017, 46, 4329–4346. [Google Scholar] [CrossRef] [PubMed]
- Bogdos, M.K.; Pinard, E.; Murphy, J.A. Applications of organocatalysed visible-light photoredox reactions for medicinal chemistry. Beilstein J. Org. Chem. 2018, 14, 2035–2064. [Google Scholar] [CrossRef] [PubMed]
- Xuan, J.; Lu, L.-Q.; Chen, J.-R.; Xiao, W.-J. Visible-Light-Driven Photoredox Catalysis in the Construction of Carbocyclic and Heterocyclic Ring Systems. Eur. J. Org. Chem. 2013, 2013, 6755–6770. [Google Scholar] [CrossRef]
- Zhang, W.; Cue, B.W. Green Techniques for Organic Synthesis and Medicinal Chemistry; Wiley: Hoboken, NJ, USA, 2012. [Google Scholar]
- Richter, G.W.; Solez, K. Transition Metal Toxicity; Academic Press: Cambridge, MA, USA, 1990; Volume 31. [Google Scholar]
- Wang, S.; Shi, X. Molecular Mechanisms of Metal Toxicity and Carcinogenesis. Mol. Cell. Biochem. 2001, 222, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Egorova, K.S.; Ananikov, V.P. Toxicity of Metal Compounds: Knowledge and Myths. Organometallics 2017, 36, 4071–4090. [Google Scholar] [CrossRef]
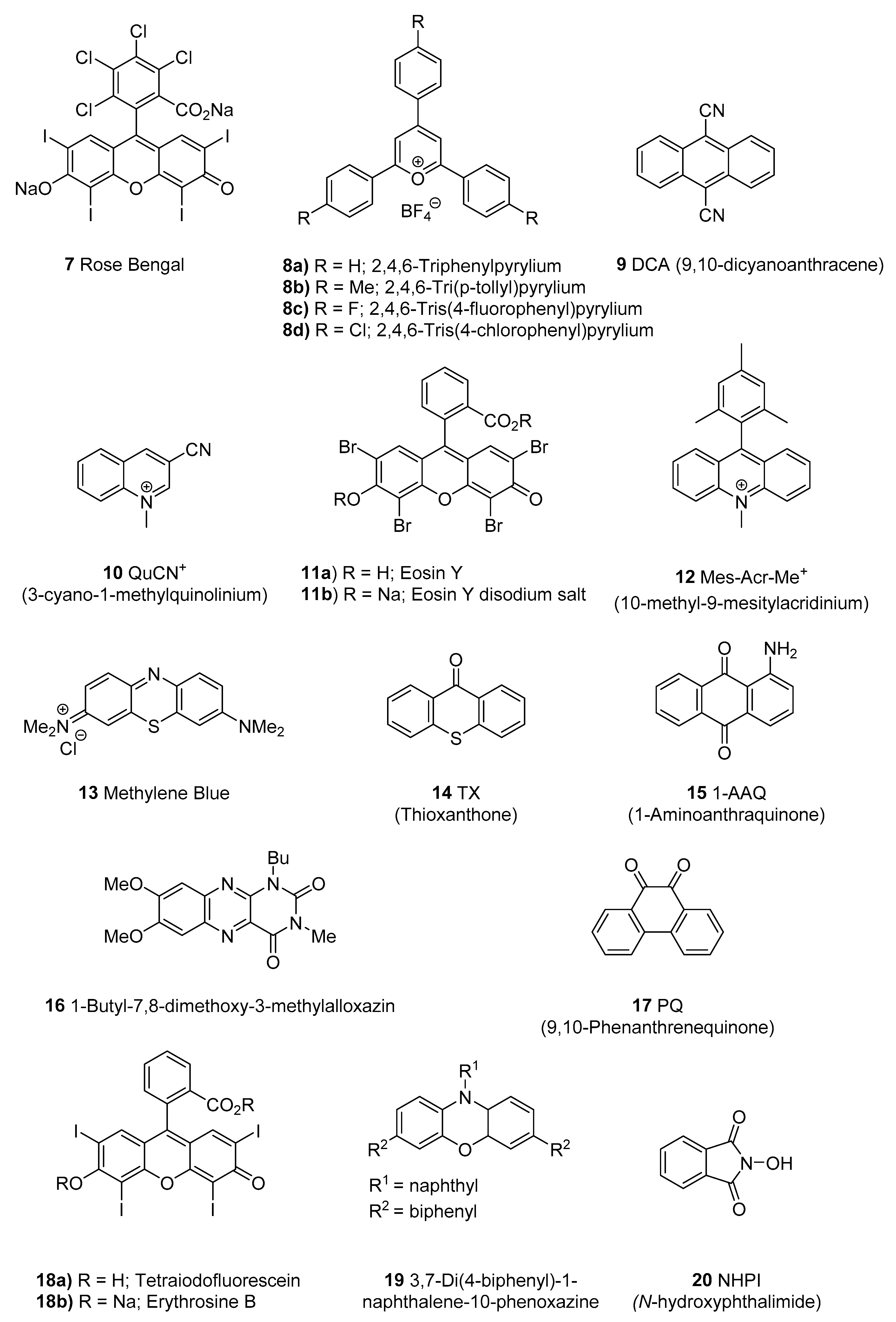
- Hari, D.P.; Konig, B. Synthetic applications of eosin Y in photoredox catalysis. Chem. Commun. 2014, 50, 6688–6699. [Google Scholar] [CrossRef]
- Srivastava, V.; Singh, P.P. Eosin Y catalysed photoredox synthesis: A review. RSC Adv. 2017, 7, 31377–31392. [Google Scholar] [CrossRef]
- Ray, S.; Samanta, P.K.; Biswas, P. Visible-Light-Active Photocatalysis: Nanostrucured Catalyst Design, Mechanisms, and Applicatios; Wilech-VCH: Weinheim, Germany, 2018; pp. 393–415. [Google Scholar]
- Taylor, A.P.; Robinson, R.P.; Fobian, Y.M.; Blakemore, D.C.; Jones, L.H.; Fadeyi, O. Modern advances in heterocyclic chemistry in drug discovery. Org. Biomol. Chem. 2016, 14, 6611–6637. [Google Scholar] [CrossRef]
- Baumann, M.; Baxendale, I.R. An overview of the synthetic routes to the best selling drugs containing 6-membered heterocycles. Beilstein J. Org. Chem. 2013, 9, 2265–2319. [Google Scholar] [CrossRef]
- Minozzi, M.; Nanni, D.; Spagnolo, P. From azides to nitrogen-centered radicals: Applications of azide radical chemistry to organic synthesis. Chem. Eur. J. 2009, 15, 7830–7840. [Google Scholar] [CrossRef]
- Funk, C.D. Leukotriene modifiers as potential therapeutics for cardiovascular disease. Nat. Rev. Drug Discov. 2005, 4, 664–672. [Google Scholar] [CrossRef] [PubMed]
- Vitaku, E.; Smith, D.T.; Njardarson, J.T. Analysis of the structural diversity, substitution patterns, and frequency of nitrogen heterocycles among U.S. FDA approved pharmaceuticals. J. Med. Chem. 2014, 57, 10257–10274. [Google Scholar] [CrossRef] [PubMed]
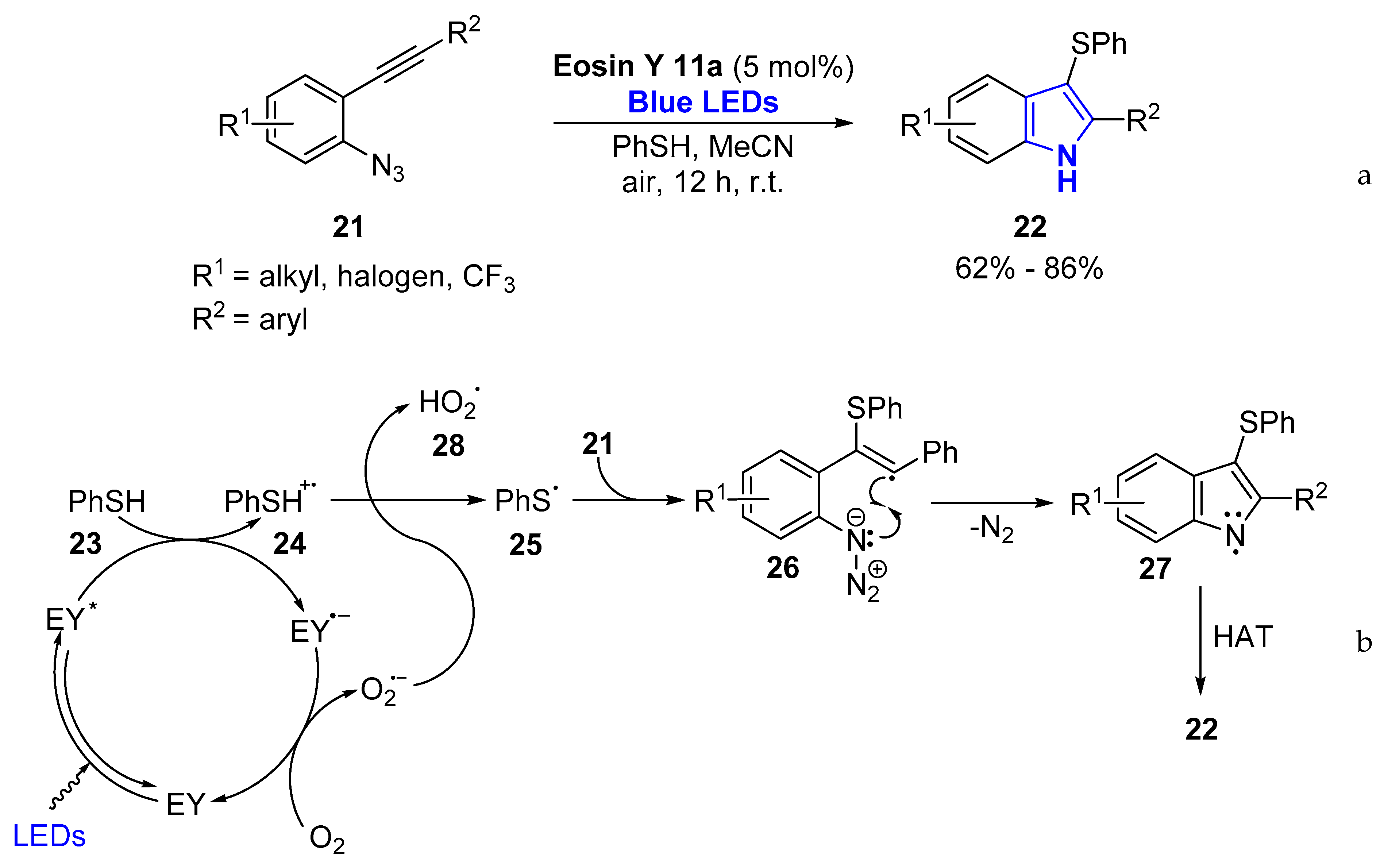
- Tambe, S.D.; Rohokale, R.S.; Kshirsagar, U.A. Visible-Light-Mediated Eosin Y Photoredox-Catalyzed Vicinal Thioamination of Alkynes: Radical Cascade Annulation Strategy for 2-Substituted-3-sulfenylindoles. Eur. J. Org. Chem. 2018, 2117–2121. [Google Scholar] [CrossRef]
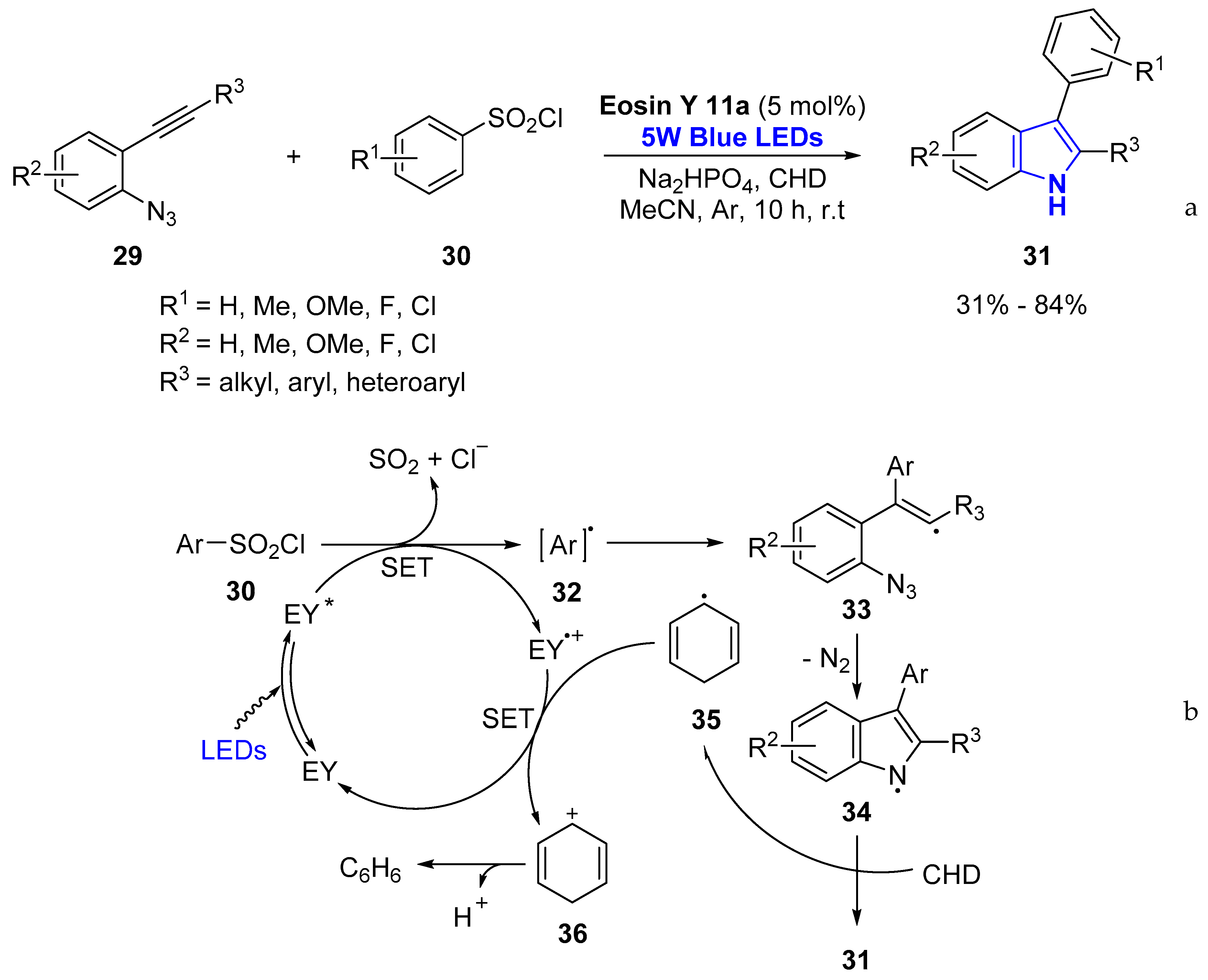
- Gu, L.; Jin, C.; Wang, W.; He, Y.; Yang, G.; Li, G. Transition-metal-free, visible-light induced cyclization of arylsulfonyl chlorides with o-azidoarylalkynes: A regiospecific route to unsymmetrical 2,3-disubstituted indoles. Chem. Commun. 2017, 53, 4203–4206. [Google Scholar] [CrossRef]
- Ojima, I. Fluorine in Medicinal Chemistry and Chemical Biology; Wiley-Blackwell: Chichester, UK, 2009. [Google Scholar]
- Jana, S.; Verma, A.; Kadu, R.; Kumar, S. Visible-light-induced oxidant and metal-free dehydrogenative cascade trifluoromethylation and oxidation of 1,6-enynes with water. Chem. Sci. 2017, 8, 6633–6644. [Google Scholar] [CrossRef]
- Nicolaou, K.C.; Chen, J.S. The art of total synthesis through cascade reactions. Chem. Soc. Rev. 2009, 38, 2993–3009. [Google Scholar] [CrossRef] [PubMed]
- Rusch, F.; Unkel, L.N.; Alpers, D.; Hoffmann, F.; Brasholz, M. A visible light photocatalytic cross-dehydrogenative coupling/dehydrogenation/6pi-cyclization/oxidation cascade: Synthesis of 12-nitroindoloisoquinolines from 2-aryltetrahydroisoquinolines. Chem. Eur. J. 2015, 21, 8336–8340. [Google Scholar] [CrossRef]
- Wang, K.; Meng, L.G.; Wang, L. Visible-Light-Promoted [2+2+2] Cyclization of Alkynes with Nitriles to Pyridines Using Pyrylium Salts as Photoredox Catalysts. Org. Lett. 2017, 19, 1958–1961. [Google Scholar] [CrossRef]
- Sun, D.; Yin, K.; Zhang, R. Visible-light-induced multicomponent cascade cycloaddition involving N-propargyl aromatic amines, diaryliodonium salts and sulfur dioxide: Rapid access to 3-arylsulfonylquinolines. Chem. Commun. 2018, 54, 1335–1338. [Google Scholar] [CrossRef]
- Pan, C.; Han, J.; Zhang, H.; Zhu, C. Radical arylalkoxycarbonylation of 2-isocyanobiphenyl with carbazates: Dual C-C bond formation toward phenanthridine-6-carboxylates. J. Org. Chem. 2014, 79, 5374–5378. [Google Scholar] [CrossRef]
- Liu, X.; Qing, Z.; Cheng, P.; Zheng, X.; Zeng, J.; Xie, H. Metal-Free Photoredox Catalyzed Cyclization of O-(2,4-Dinitrophenyl)oximes to Phenanthridines. Molecules 2016, 21, 1690. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.C.; Zhang, J.Y.; Zhang, J.J.; Duan, X.H.; Guo, L.N. Metal-Free, Visible-Light-Promoted Decarboxylative Radical Cyclization of Vinyl Azides with N-Acyloxyphthalimides. J. Org. Chem. 2018, 83, 1598–1605. [Google Scholar] [CrossRef]
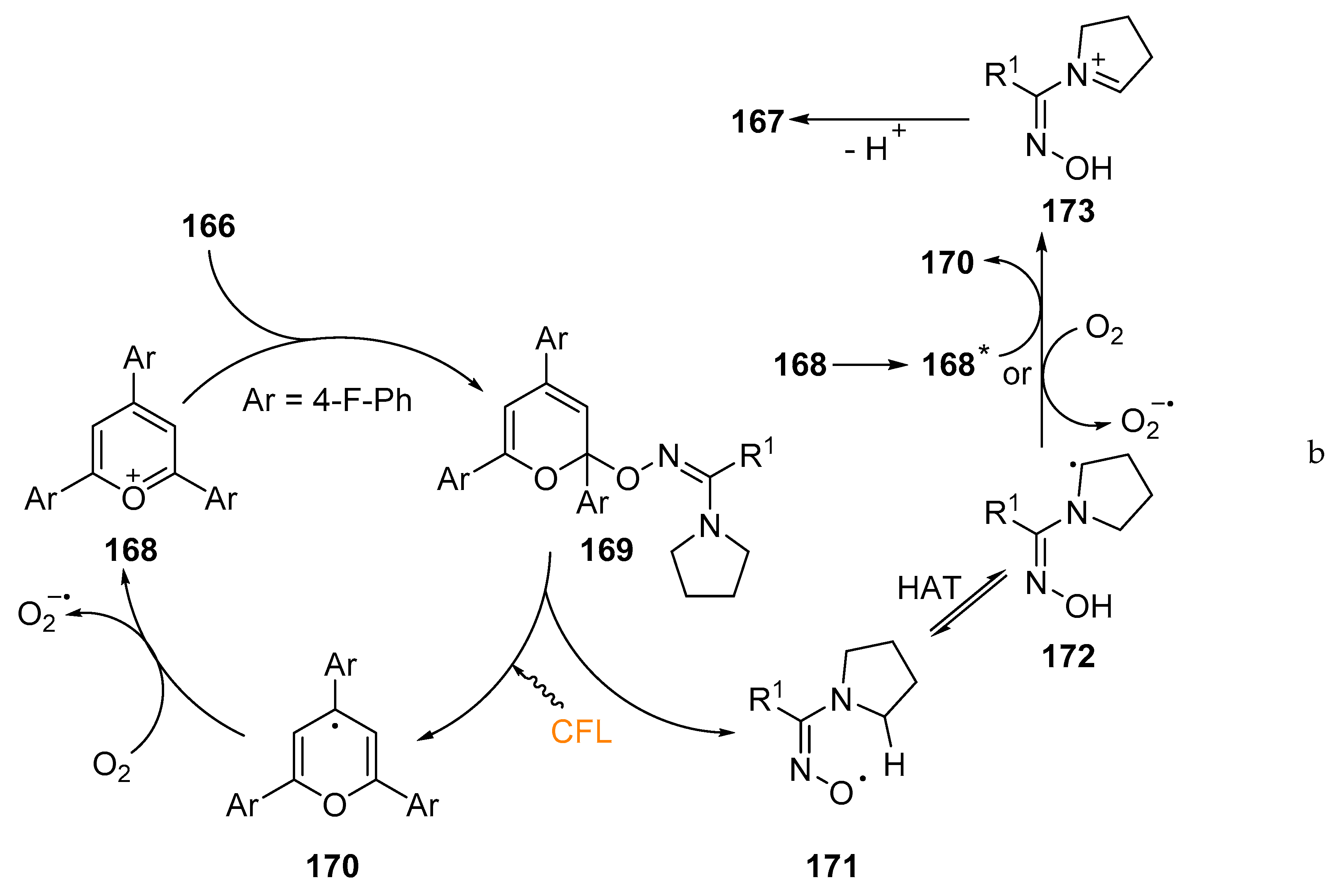
- Davies, J.; Booth, S.G.; Essafi, S.; Dryfe, R.A.; Leonori, D. Visible-Light-Mediated Generation of Nitrogen-Centered Radicals: Metal-Free Hydroimination and Iminohydroxylation Cyclization Reactions. Angew. Chem. Int. Ed. 2015, 54, 14017–14021. [Google Scholar] [CrossRef] [PubMed]
- Gonda, Z.; Béke, F.; Tischler, O.; Petró, M.; Novák, Z.; Tóth, B.L. Erythrosine B Catalyzed Visible-Light Photoredox Arylation-Cyclization of N-Alkyl-N-aryl-2-(trifluoromethyl)acrylamides to 3-(Trifluoromethyl)indolin-2-one Derivatives. Eur. J. Org. Chem. 2017, 2017, 2112–2117. [Google Scholar] [CrossRef]
- Zou, S.; Geng, S.; Chen, L.; Wang, H.; Huang, F. Visible light driven metal-free intramolecular cyclization: A facile synthesis of 3-position substituted 3,4-dihydroisoquinolin-1(2H)-one. Org. Biomol. Chem. 2019, 17, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Carreira, E.M.; Kvaerno, L. Classics in Stereoselective Synthesis; Wiley-VCH: Weinheim, Germany, 2009. [Google Scholar]
- Clay, A.; Vallavoju, N.; Krishnan, R.; Ugrinov, A.; Sivaguru, J. Metal-Free Visible Light-Mediated Photocatalysis: Controlling Intramolecular [2+2] Photocycloaddition of Enones through Axial Chirality. J. Org. Chem. 2016, 81, 7191–7200. [Google Scholar] [CrossRef] [PubMed]
- Xin, J.-R.; Guo, J.-T.; Vigliaturo, D.; He, Y.-H.; Guan, Z. Metal-free visible light driven synthesis of tetrahydroquinoline derivatives utilizing Rose Bengal. Tetrahedron 2017, 73, 4627–4633. [Google Scholar] [CrossRef]
- Zhu, S.; Das, A.; Bui, L.; Zhou, H.; Curran, D.P.; Rueping, M. Oxygen Switch in Visible-Light Photoredox Catalysis: Radical Additions and Cyclizations and Unexpected C–C-Bond Cleavage Reactions. J. Am. Chem. Soc. 2013, 135, 1823–1829. [Google Scholar] [CrossRef]
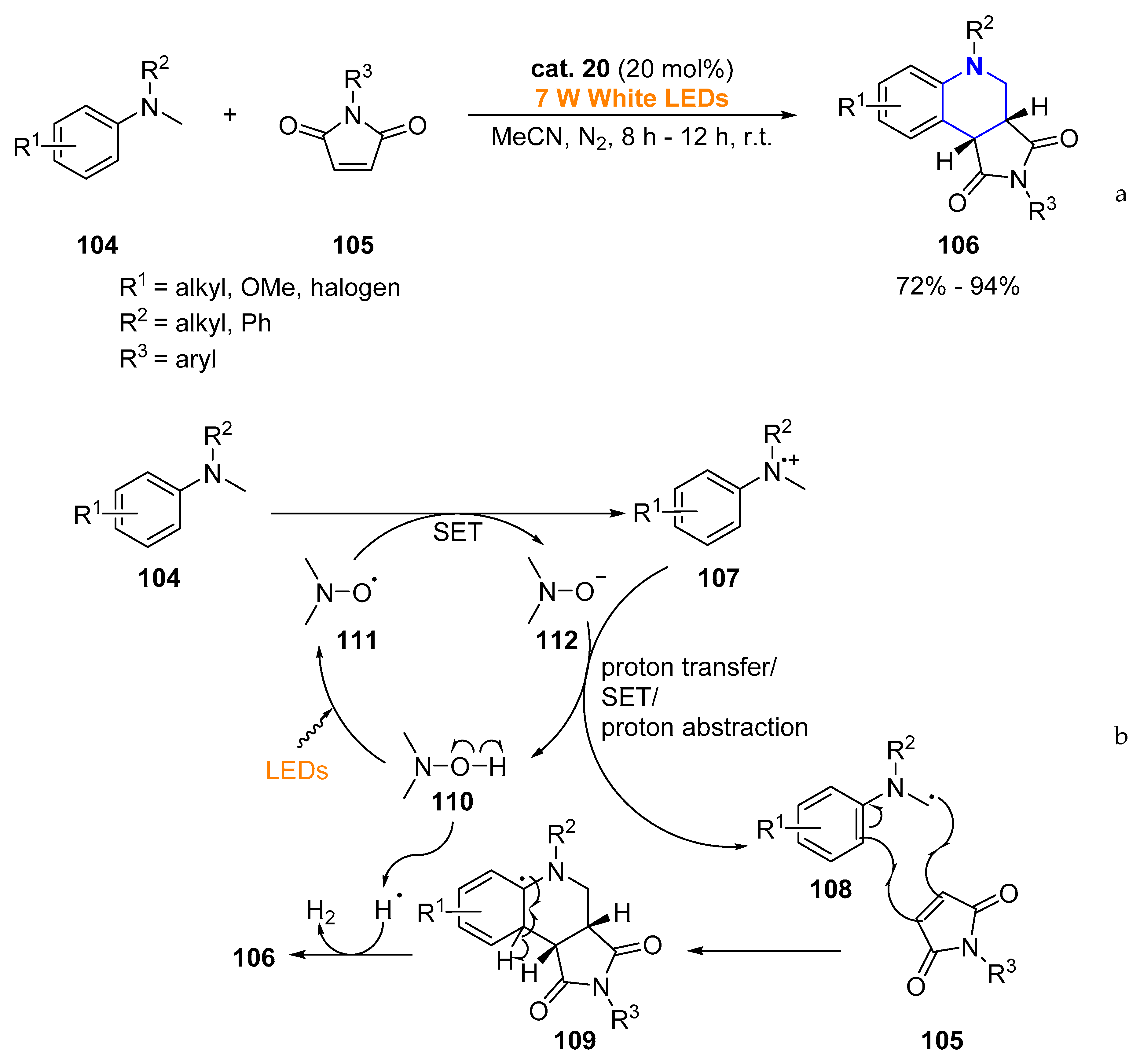
- Liang, Z.; Xu, S.; Tian, W.; Zhang, R. Eosin Y-catalyzed visible-light-mediated aerobic oxidative cyclization of N,N-dimethylanilines with maleimides. Beilstein J. Org. Chem. 2015, 11, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Yadav, A.K.; Yadav, L.D.S. Visible light photoredox catalysis with N-hydroxyphthalimide for [4+2] cyclization between N-methylanilines and maleimides. Tetrahedron Lett. 2017, 58, 552–555. [Google Scholar] [CrossRef]
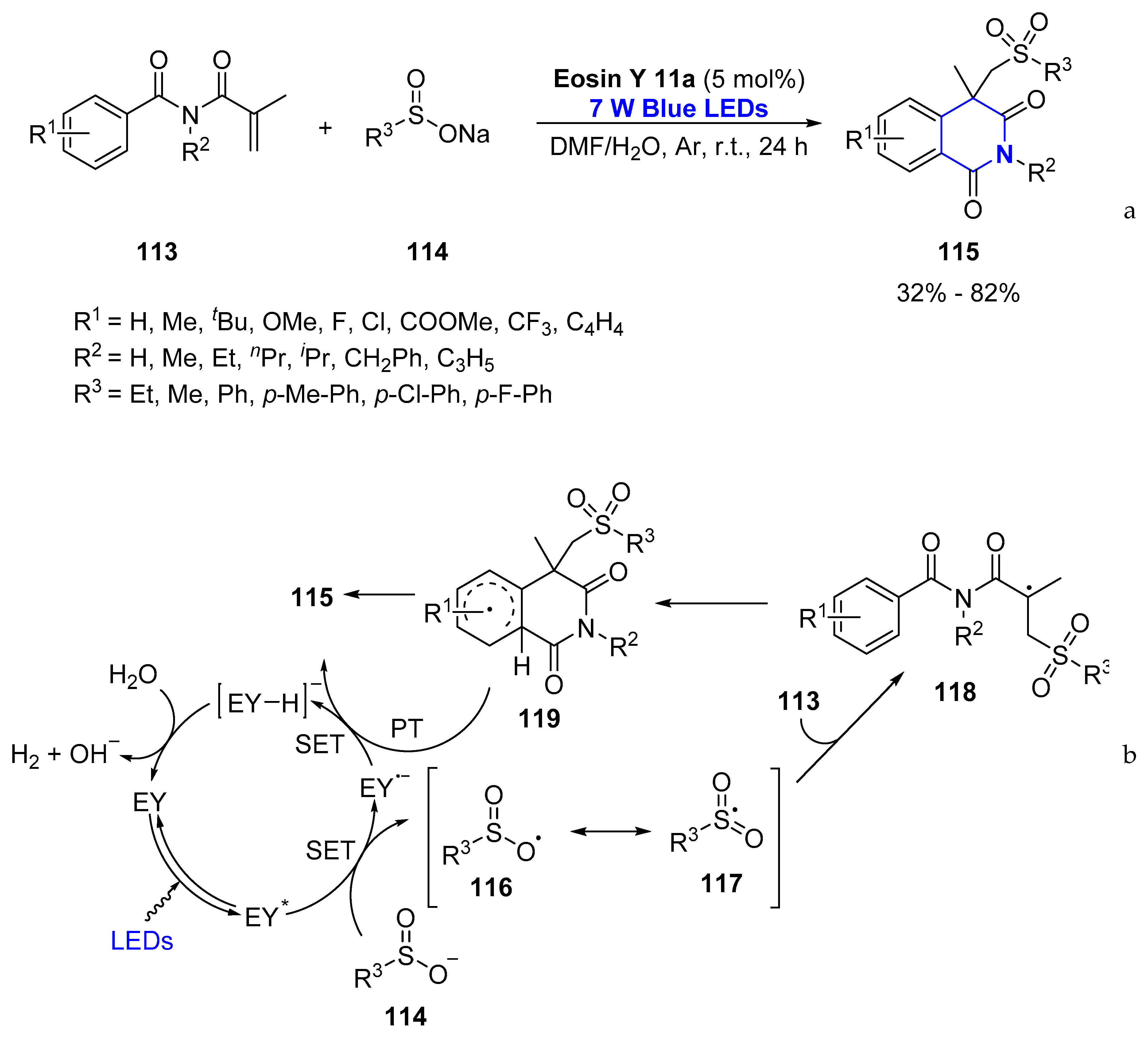
- Zuo, K.-L.; He, Y.-H.; Guan, Z. Metal-Free Visible-Light Photocatalytic Tandem Radical Addition-Cyclization Strategy for the Synthesis of Sulfonyl-Containing Isoquinolinediones. Eur. J. Org. Chem. 2019, 2019, 939–948. [Google Scholar] [CrossRef]
- Martins, P.; Jesus, J.; Santos, S.; Raposo, L.R.; Roma-Rodrigues, C.; Baptista, P.V.; Fernandes, A.R. Heterocyclic Anticancer Compounds: Recent Advances and the Paradigm Shift towards the Use of Nanomedicine’s Tool Box. Molecules 2015, 20, 16852–16891. [Google Scholar] [CrossRef] [PubMed]
- Cossy, J.; Guérinot, A. Advances in Heterocyclic Chemistry. Natural Products Containing Oxygen Heterocycles—Synthetic Advances Between 1990 and 2015. Adv. Heterocycl. Chem. 2016, 119, 107–142. [Google Scholar]
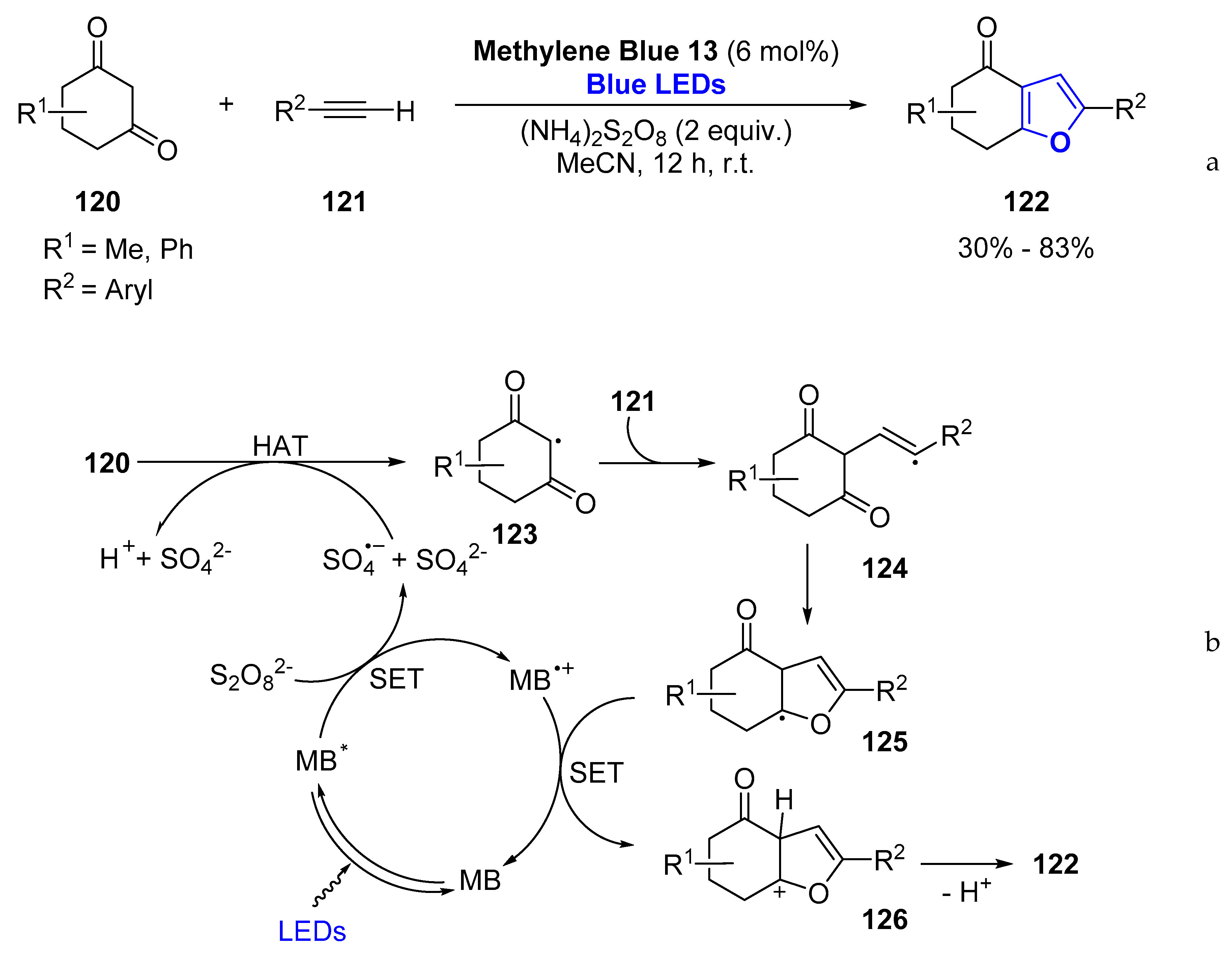
- Shao, A.; Luo, X.; Chiang, C.W.; Gao, M.; Lei, A. Furans Accessed through Visible-Light-Mediated Oxidative [3+2] Cycloaddition of Enols and Alkynes. Chemistry 2017, 23, 17874–17878. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Wang, K.L.; Liu, M.; She, Z.G.; Wang, C.Y. Bioactive steroid derivatives and butyrolactone derivatives from a gorgonian-derived Aspergillus sp. fungus. Chem. Biodivers. 2015, 12, 1398–1406. [Google Scholar] [CrossRef] [PubMed]
- Niu, X.; Dahse, H.M.; Menzel, K.D.; Lozach, O.; Walther, G.; Meijer, L.; Grabley, S.; Sattler, I. Butyrolactone I derivatives from Aspergillus terreus carrying an unusual sulfate moiety. J. Nat. Prod. 2008, 71, 689–692. [Google Scholar] [CrossRef]
- Cavanaugh, C.L.; Nicewicz, D.A. Synthesis of alpha-Benzyloxyamino-gamma-butyrolactones via a Polar Radical Crossover Cycloaddition Reaction. Org. Lett. 2015, 17, 6082–6085. [Google Scholar] [CrossRef]
- Romero, N.A.; Nicewicz, D.A. Mechanistic insight into the photoredox catalysis of anti-markovnikov alkene hydrofunctionalization reactions. J. Am. Chem. Soc. 2014, 136, 17024–17035. [Google Scholar] [CrossRef]
- Zhang, Q.B.; Ban, Y.L.; Zhou, D.G.; Zhou, P.P.; Wu, L.Z.; Liu, Q. Preparation of alpha-Acyloxy Ketones via Visible-Light-Driven Aerobic Oxo-Acyloxylation of Olefins with Carboxylic Acids. Org. Lett. 2016, 18, 5256–5259. [Google Scholar] [CrossRef]
- Kouser, F.; Sharma, V.K.; Rizvi, M.; Sultan, S.; Chalotra, N.; Gupta, V.K.; Nandi, U.; Shah, B.A. Stereoselective synthesis of 3,4-di-substituted mercaptolactones via photoredox-catalyzed radical addition of thiophenols. Tetrahedron Lett. 2018, 59, 2161–2166. [Google Scholar] [CrossRef]
- Gesmundo, N.J.; Grandjean, J.M.; Nicewicz, D.A. Amide and amine nucleophiles in polar radical crossover cycloadditions: Synthesis of gamma-lactams and pyrrolidines. Org. Lett. 2015, 17, 1316–1319. [Google Scholar] [CrossRef] [PubMed]
- Morse, P.D.; Nguyen, T.M.; Cruz, C.L.; Nicewicz, D.A. Enantioselective counter-anions in photoredox catalysis: The asymmetric cation radical Diels-Alder reaction. Tetrahedron 2018, 74, 3266–3272. [Google Scholar] [CrossRef] [PubMed]
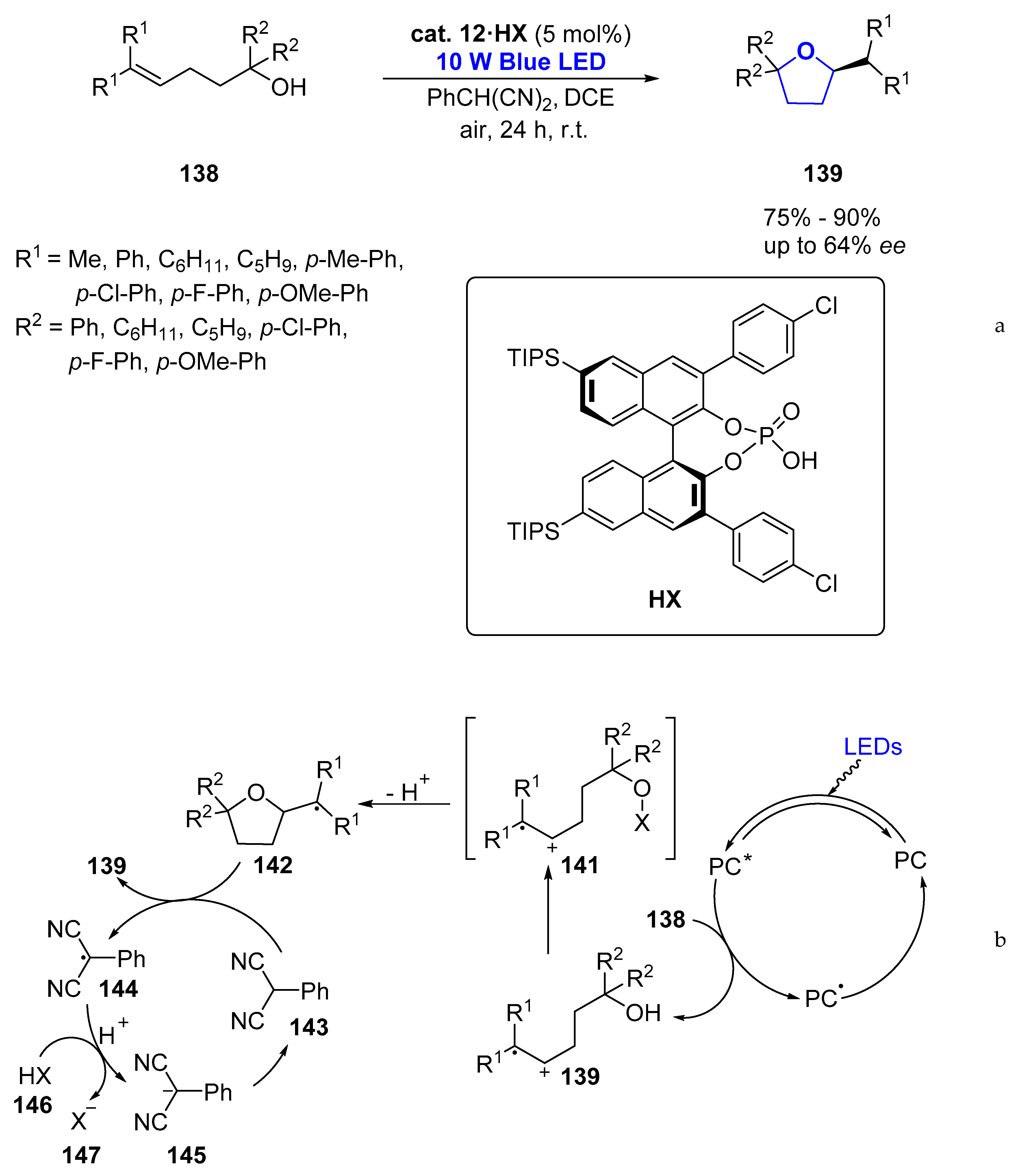
- Yang, Z.; Li, H.; Li, S.; Zhang, M.-T.; Luo, S. A chiral ion-pair photoredox organocatalyst: Enantioselective anti-Markovnikov hydroetherification of alkenols. Organ. Chem. Front. 2017, 4, 1037–1041. [Google Scholar] [CrossRef]
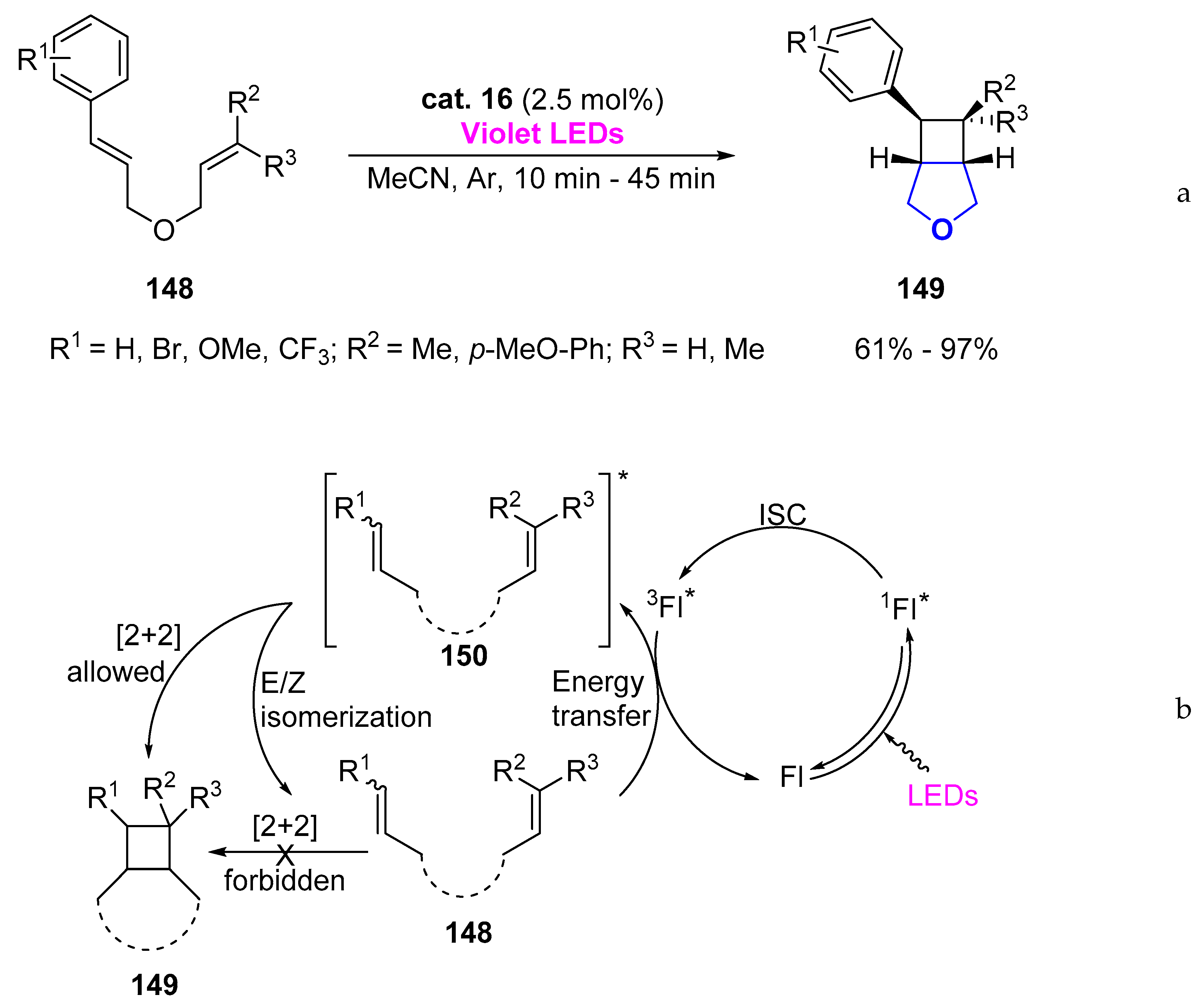
- Jirásek, M.; Straková, K.; Neveselý, T.; Svobodová, E.; Rottnerová, Z.; Cibulka, R. Flavin-Mediated Visible-Light [2+2] Photocycloaddition of Nitrogen- and Sulfur-Containing Dienes. Eur. J. Org. Chem. 2017, 2017, 2139–2146. [Google Scholar] [CrossRef]
- Mojr, V.; Pitrová, G.; Straková, K.; Prukała, D.; Brazevic, S.; Svobodová, E.; Hoskovcová, I.; Burdziński, G.; Slanina, T.; Sikorski, M.; et al. Flavin Photocatalysts for Visible-Light [2+2] Cycloadditions: Structure, Reactivity and Reaction Mechanism. ChemCatChem 2018, 10, 849–858. [Google Scholar] [CrossRef]
- Mojr, V.; Svobodova, E.; Strakova, K.; Nevesely, T.; Chudoba, J.; Dvorakova, H.; Cibulka, R. Tailoring flavins for visible light photocatalysis: Organocatalytic [2+2] cycloadditions mediated by a flavin derivative and visible light. Chem. Commun. 2015, 51, 12036–12039. [Google Scholar] [CrossRef] [PubMed]
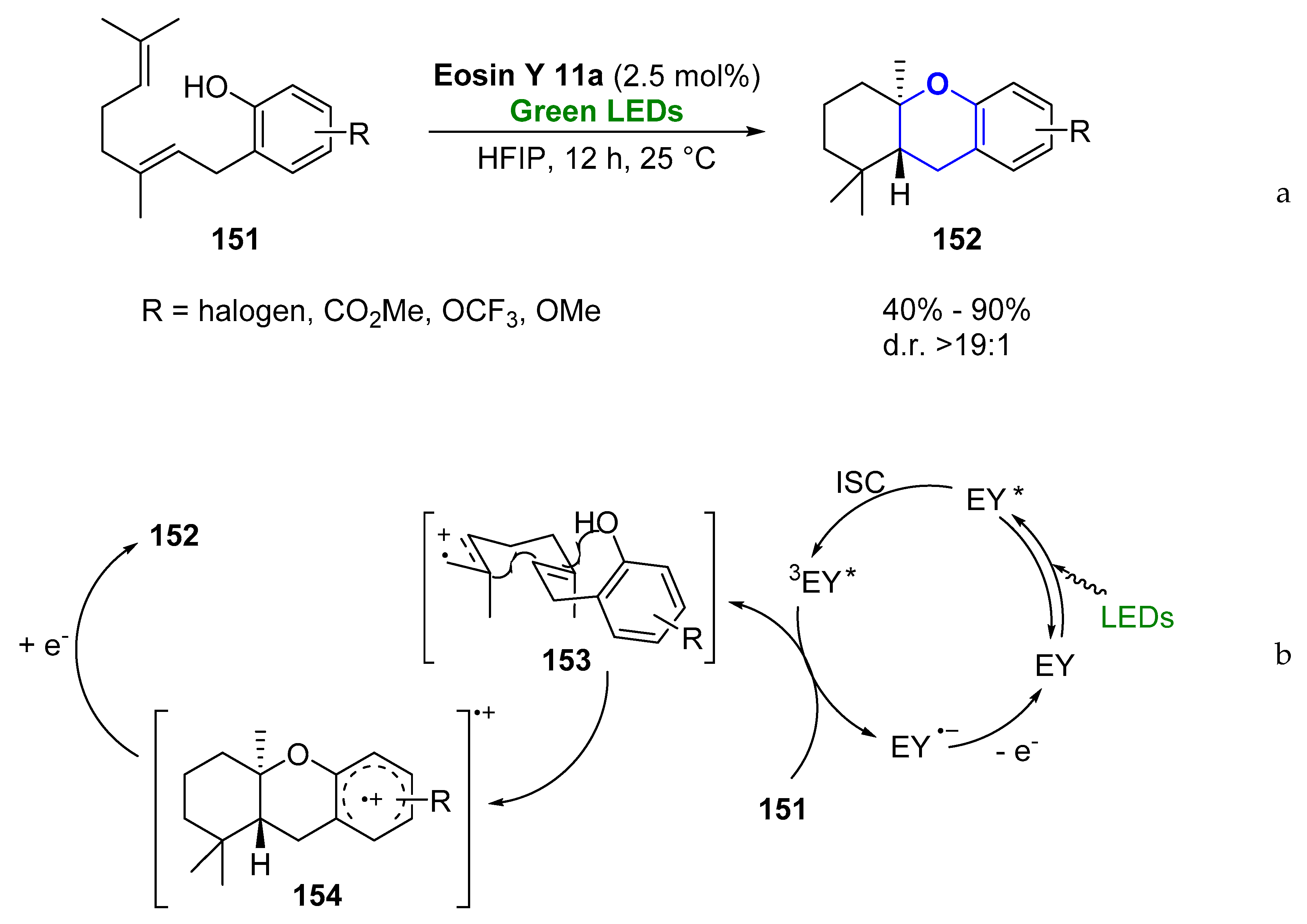
- Yoder, R.A.; Johnston, J.N. A Case Study in Biomimetic Total Synthesis: Polyolefin Carbocyclizations to Terpenes and Steroids. Chem. Rev. 2005, 105, 4730–4756. [Google Scholar] [CrossRef]
- Justicia, J.; Alvarez de Cienfuegos, L.; Campana, A.G.; Miguel, D.; Jakoby, V.; Gansauer, A.; Cuerva, J.M. Bioinspired terpene synthesis: A radical approach. Chem. Soc. Rev. 2011, 40, 3525–3537. [Google Scholar] [CrossRef]
- Yang, Z.; Li, H.; Zhang, L.; Zhang, M.T.; Cheng, J.P.; Luo, S. Organic Photocatalytic Cyclization of Polyenes: A Visible-Light-Mediated Radical Cascade Approach. Chem. Eur. J. 2015, 21, 14723–14727. [Google Scholar] [CrossRef]
- Yang, W.; Yang, S.; Li, P.; Wang, L. Visible-light initiated oxidative cyclization of phenyl propiolates with sulfinic acids to coumarin derivatives under metal-free conditions. Chem. Commun. 2015, 51, 7520–7523. [Google Scholar] [CrossRef]
- Hashimoto, T.; Maruoka, K. Recent advances of catalytic asymmetric 1,3-dipolar cycloadditions. Chem. Rev. 2015, 115, 5366–5412. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Gao, F.; Yang, C.; Gao, G.L.; Zhao, Y.; Gao, Y.; Xia, W. Visible-Light-Mediated Anti-Regioselective Nitrone 1,3-Dipolar Cycloaddition Reaction and Synthesis of Bisindolylmethanes. Org. Lett. 2017, 19, 5086–5089. [Google Scholar] [CrossRef]
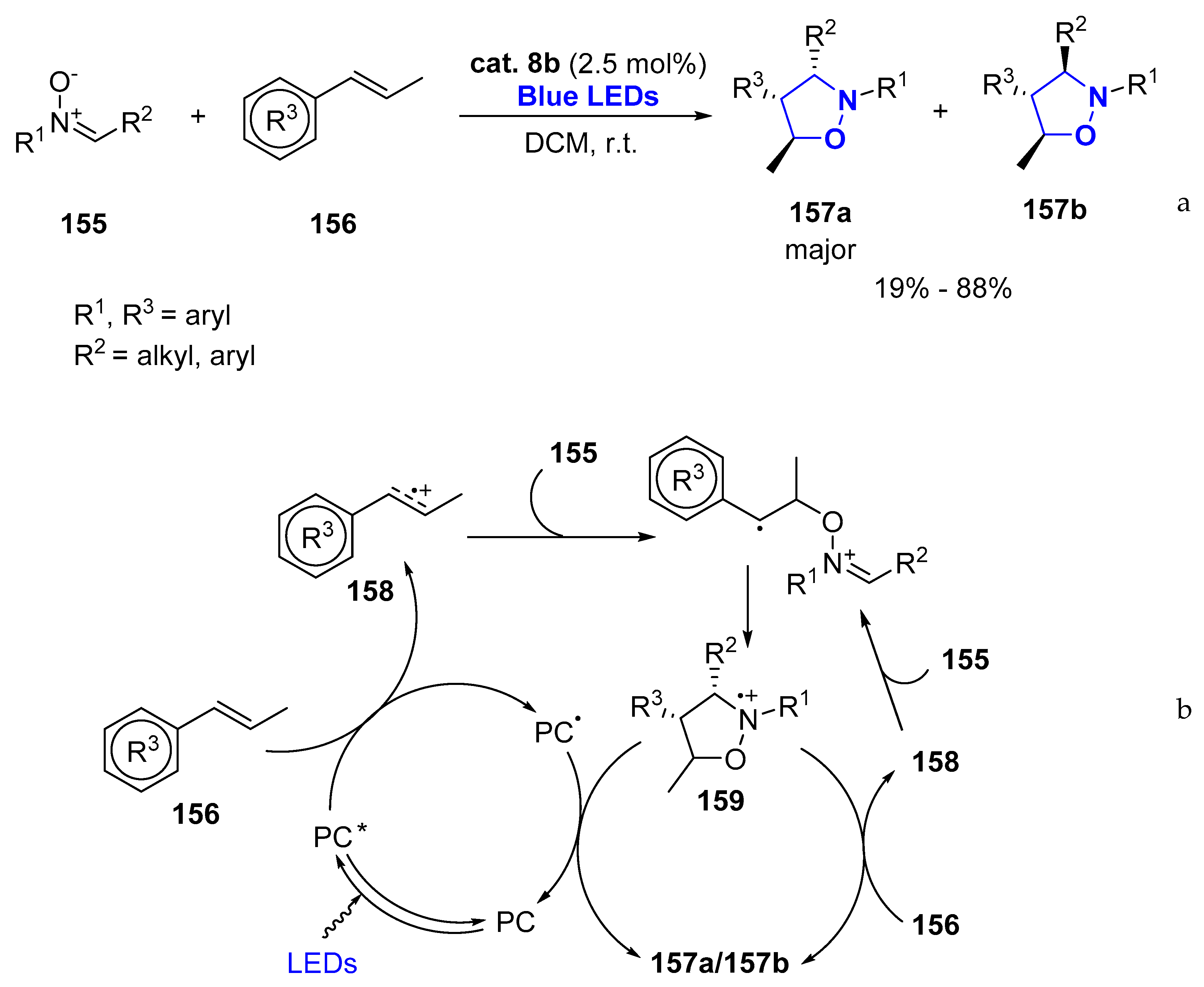
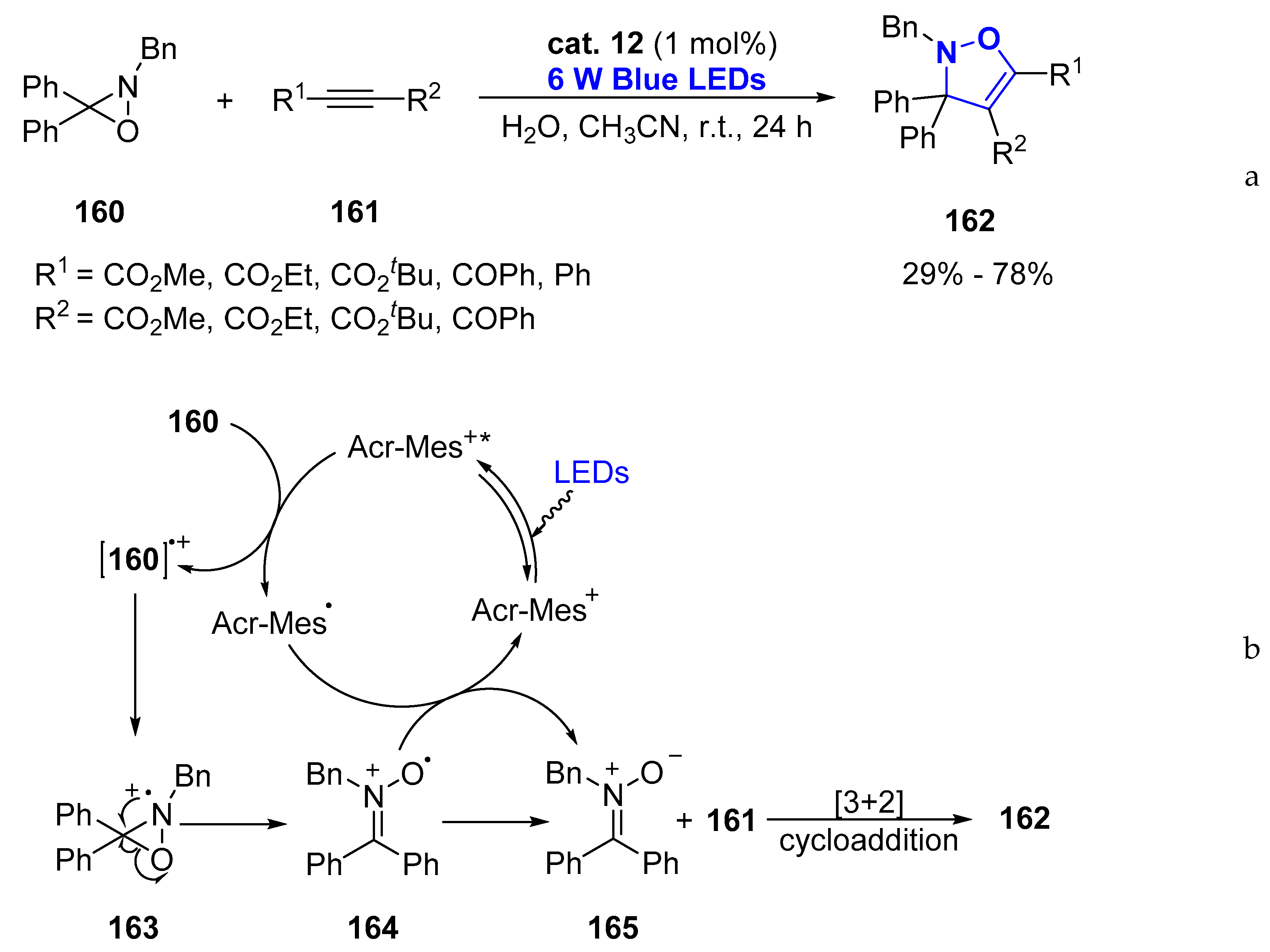
- Jang, G.S.; Lee, J.; Seo, J.; Woo, S.K. Synthesis of 4-Isoxazolines via Visible-Light Photoredox-Catalyzed [3+2] Cycloaddition of Oxaziridines with Alkynes. Org. Lett. 2017, 19, 6448–6451. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Cui, L.S.; Zhong, C.; Jiang, Z.Q.; Liao, L.S. Asymmetric design of bipolar host materials with novel 1,2,4-oxadiazole unit in blue phosphorescent device. Org. Lett. 2014, 16, 1622–1625. [Google Scholar] [CrossRef]
- Soni, V.K.; Kim, J.; Cho, E.J. Organocatalytic Oxidative Cyclization of Amidoximes for the Synthesis of 1,2,4-Oxadiazolines. Adv. Synth. Catal. 2018, 360, 2626–2631. [Google Scholar] [CrossRef]
- Tilvi, S.; Singh, K.S. Synthesis of Oxazole, Oxazoline and Isoxazoline Derived Marine Natural Products: A Review. Curr. Organ. Chem. 2016, 20, 898–929. [Google Scholar] [CrossRef]
- Hargaden, G.C.; Guiry, P.J. Recent applications of oxazoline-containing ligands in asymmetric catalysis. Chem. Rev. 2009, 109, 2505–2550. [Google Scholar] [CrossRef] [PubMed]
- Morse, P.D.; Nicewicz, D.A. Divergent Regioselectivity in Photoredox-Catalyzed Hydrofunctionalization Reactions of Unsaturated Amides and Thioamides. Chem. Sci. 2015, 6, 270–274. [Google Scholar] [CrossRef]
- Petzold, D.; Nitschke, P.; Brandl, F.; Scheidler, V.; Dick, B.; Gschwind, R.M.; Konig, B. Visible-Light-Mediated Liberation and In Situ Conversion of Fluorophosgene. Chem. Eur. J. 2019, 25, 361–366. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Sugiura, Y.; Yamaguchi, E.; Tada, N.; Itoh, A. Synthetic Method for the Preparation of Quinazolines by the Oxidation of Amines Using Singlet Oxygen. Asian J. Org. Chem. 2017, 6, 432–435. [Google Scholar] [CrossRef]
- Riemer, D.; Schilling, W.; Goetz, A.; Zhang, Y.; Gehrke, S.; Tkach, I.; Hollóczki, O.; Das, S. CO2-Catalyzed Efficient Dehydrogenation of Amines with Detailed Mechanistic and Kinetic Studies. ACS Catal. 2018, 8, 11679–11687. [Google Scholar] [CrossRef]
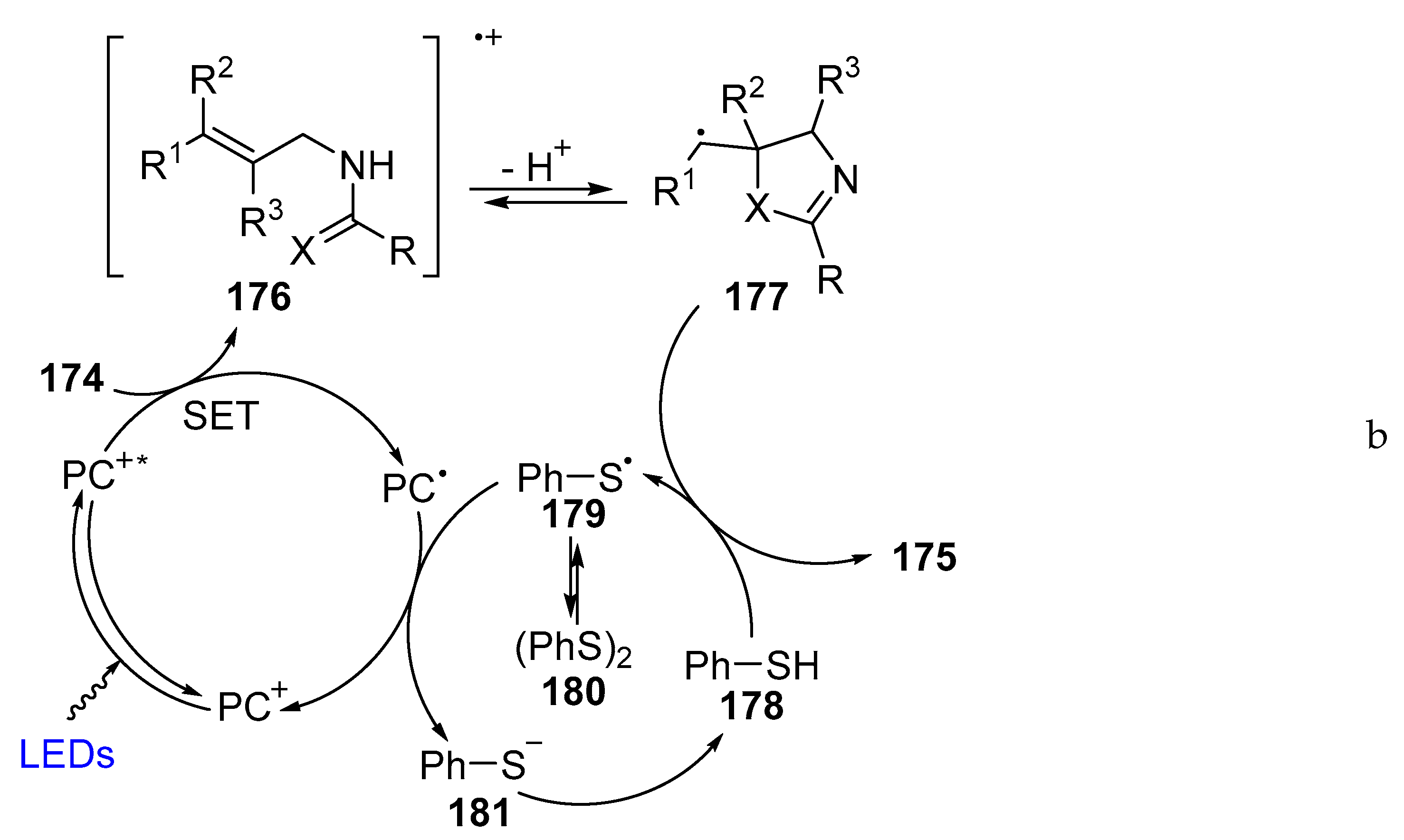
- Shen, Z.C.; Yang, P.; Tang, Y. Transition Metal-Free Visible Light-Driven Photoredox Oxidative Annulation of Arylamidines. J. Org. Chem. 2016, 81, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.X. Enantioselective biotransformations of nitriles in organic synthesis. Acc. Chem. Res. 2015, 48, 602–611. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Fang, X.; Zhuang, S.; Liu, P.; Sun, P. Photoredox Catalysis: Construction of Polyheterocycles via Alkoxycarbonylation/Addition/Cyclization Sequence. Org. Lett. 2017, 19, 3580–3583. [Google Scholar] [CrossRef] [PubMed]
- Borpatra, P.J.; Deb, M.L.; Baruah, P.K. Visible light-promoted metal-free intramolecular cross dehydrogenative coupling approach to 1,3-oxazines. Tetrahedron Lett. 2017, 58, 4006–4010. [Google Scholar] [CrossRef]
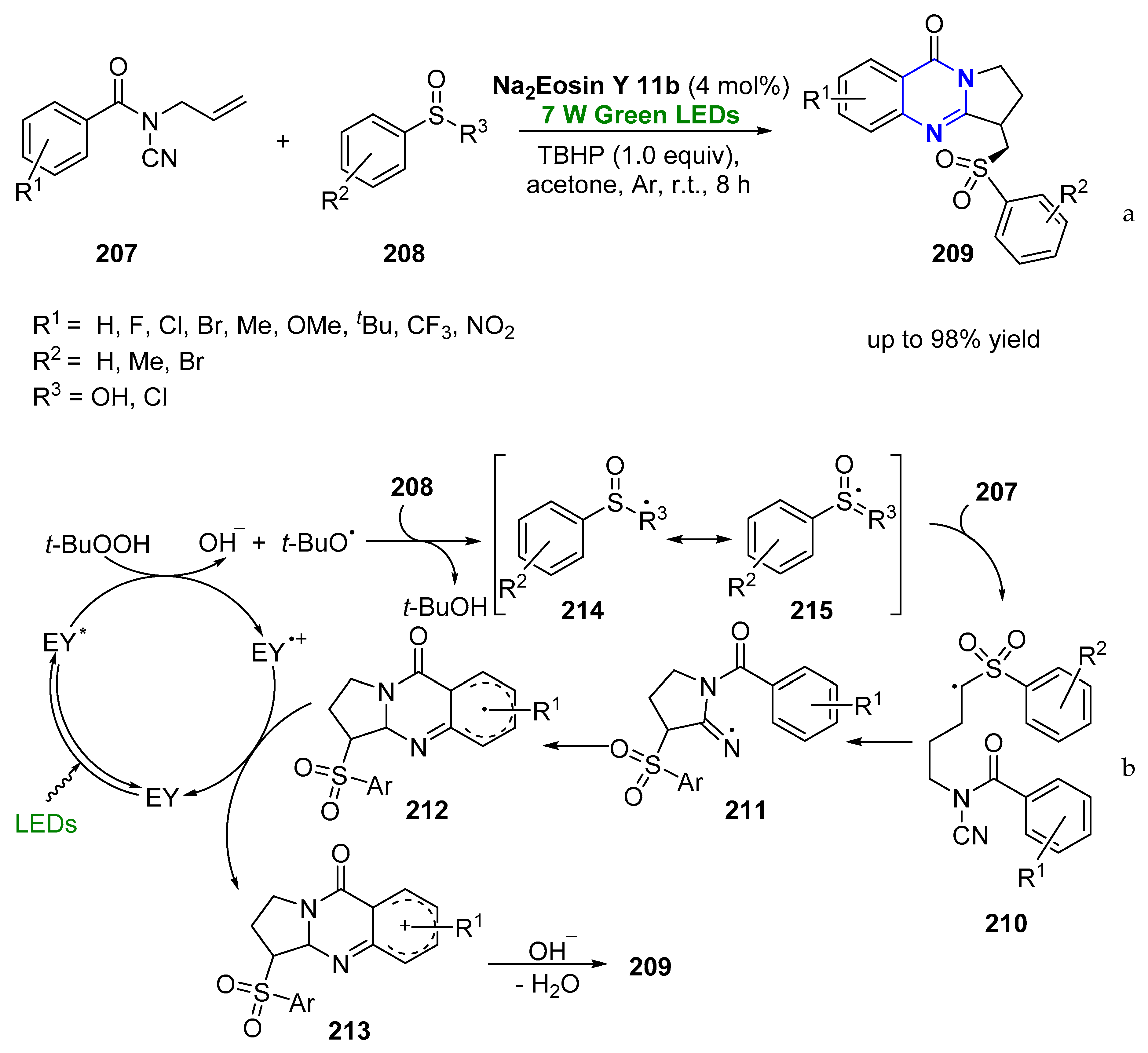
- Qian, P.; Deng, Y.; Mei, H.; Han, J.; Zhou, J.; Pan, Y. Visible-Light Photoredox Catalyzed Oxidative/Reductive Cyclization Reaction of N-Cyanamide Alkenes for the Synthesis of Sulfonated Quinazolinones. Org. Lett. 2017, 19, 4798–4801. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.; Srivastava, M.; Rai, P.; Tripathi, B.P.; Mishra, A.; Singh, J.; Singh, J. Oxidative organophotoredox catalysis: A regioselective synthesis of 2-nitro substituted imidazopyridines and 3-substituted indoles, initiated by visible light. New J. Chem. 2016, 40, 9694–9701. [Google Scholar] [CrossRef]
- Yadav, L.; Kapoorr, R.; Singh, S.; Tripathi, S. Photocatalytic Oxidative Heterocyclization of Semicarbazones: An Efficient Approach for the Synthesis of 1,3,4-Oxadiazoles. Synlett 2015, 26, 1201–1206. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
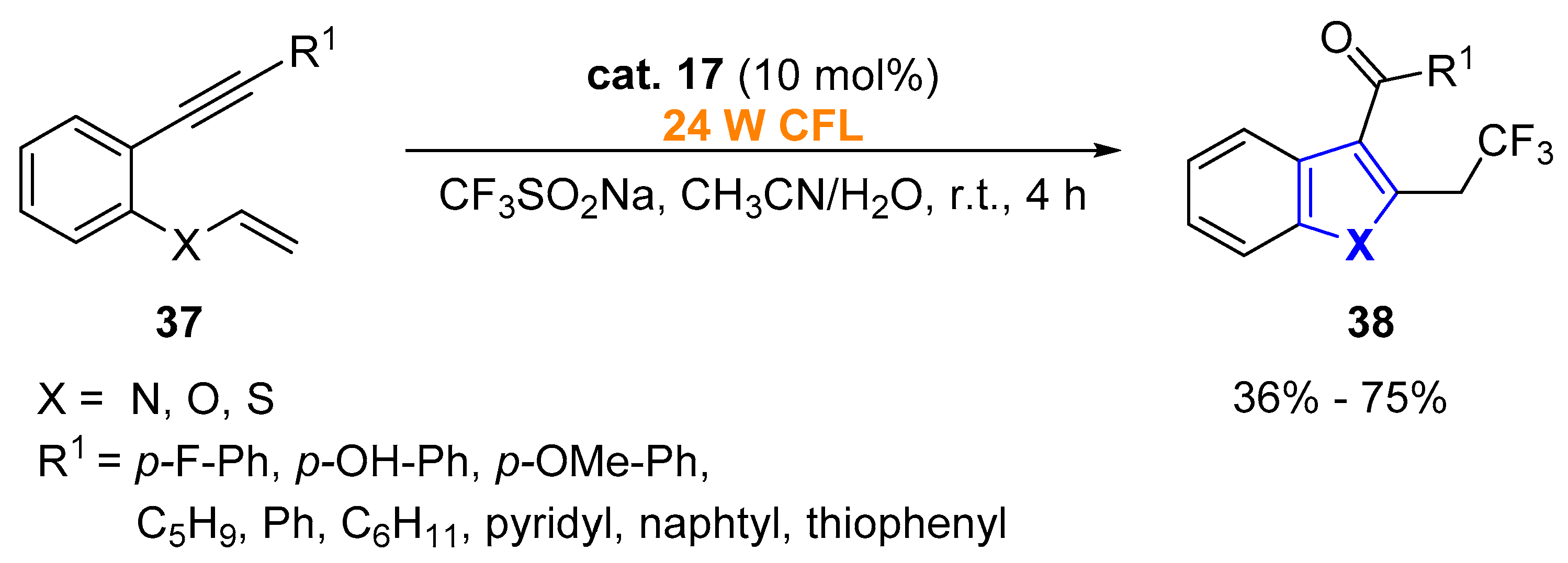




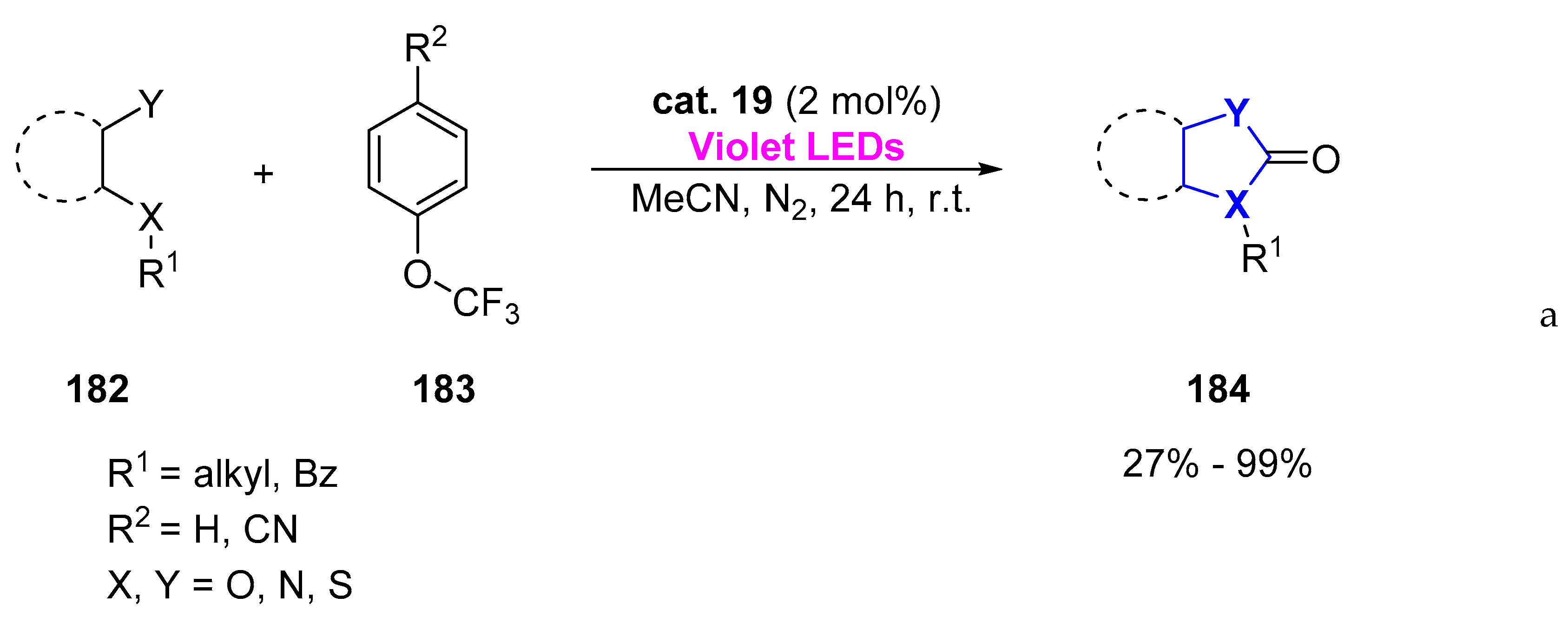


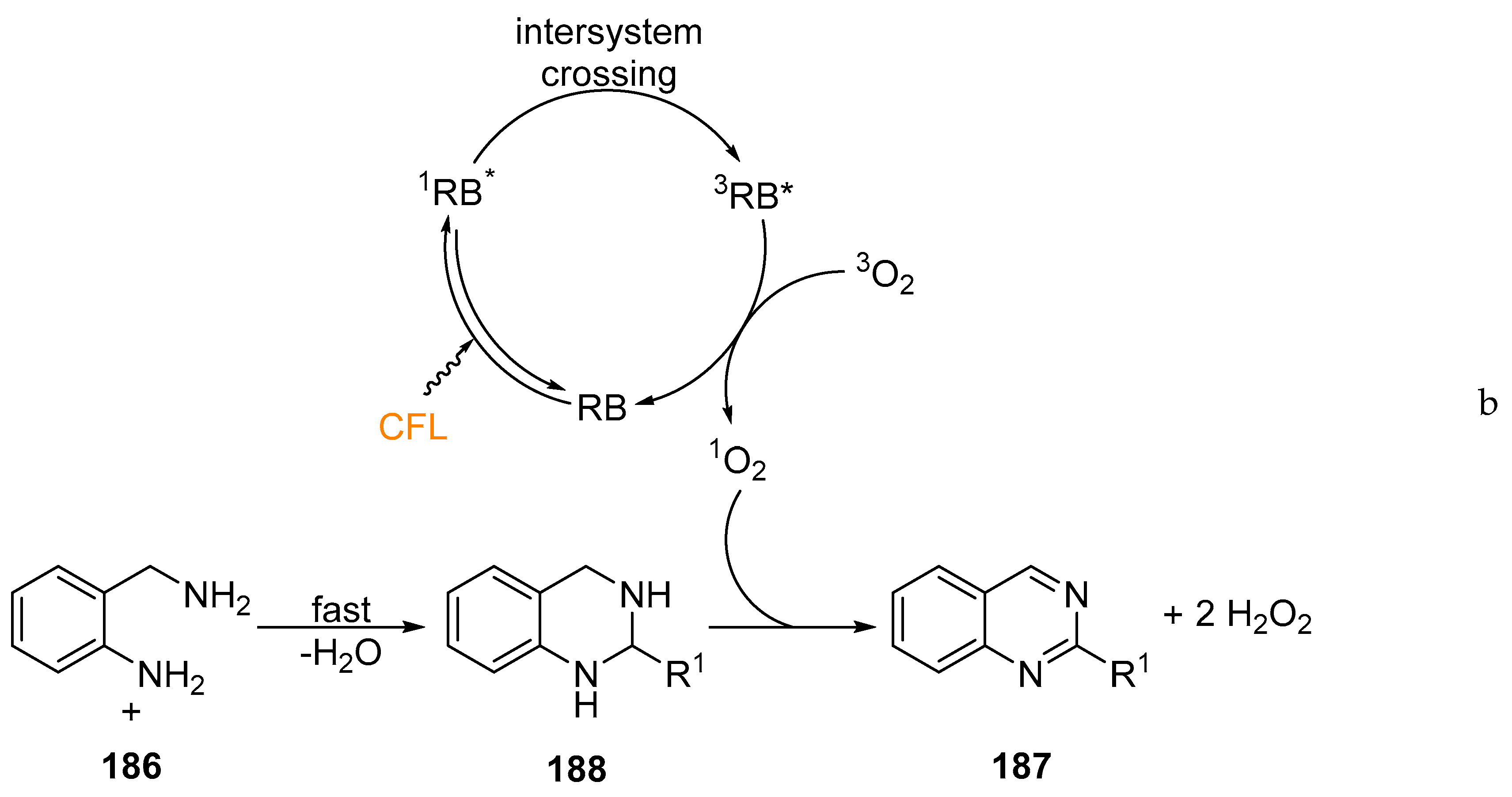

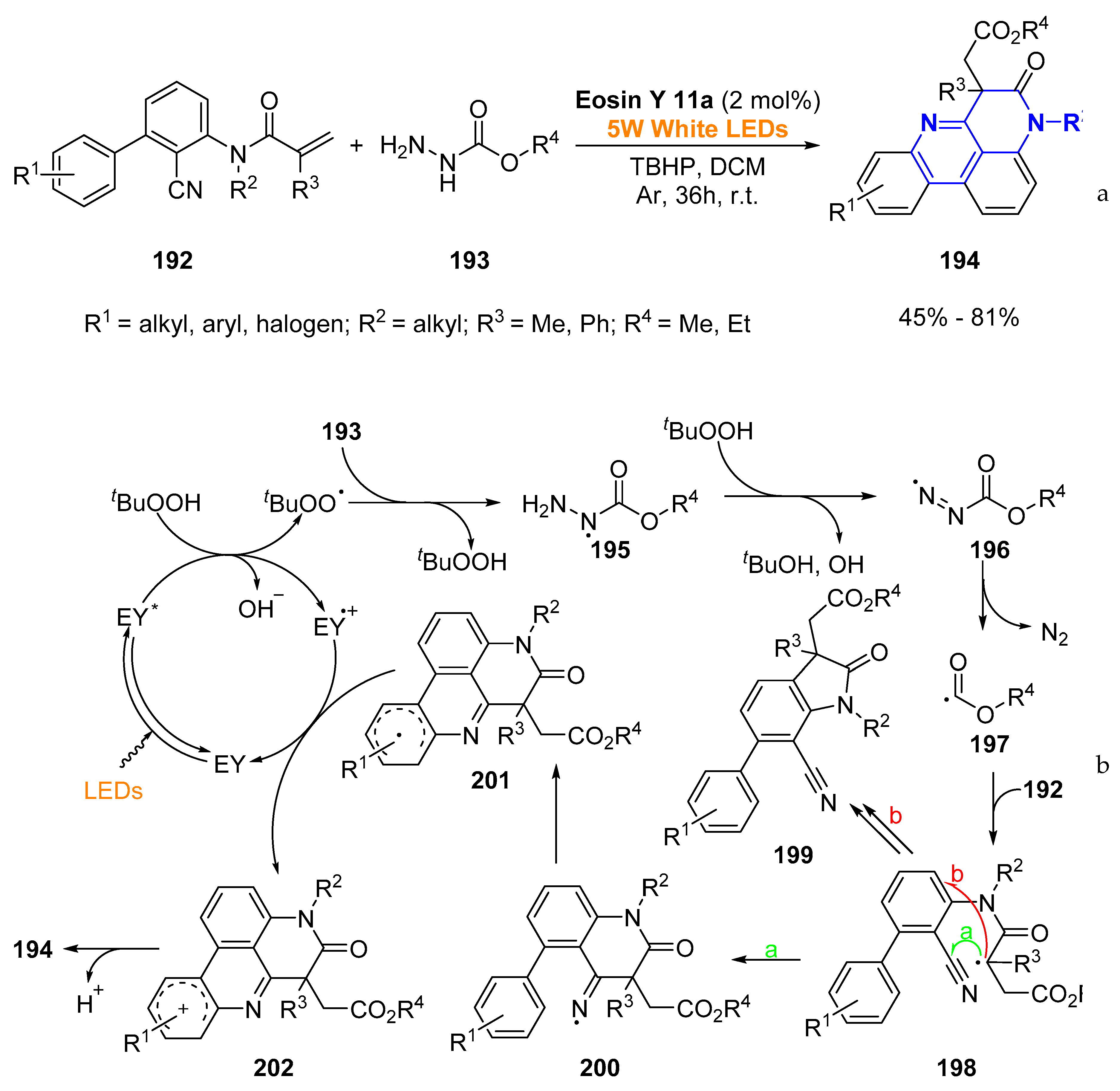
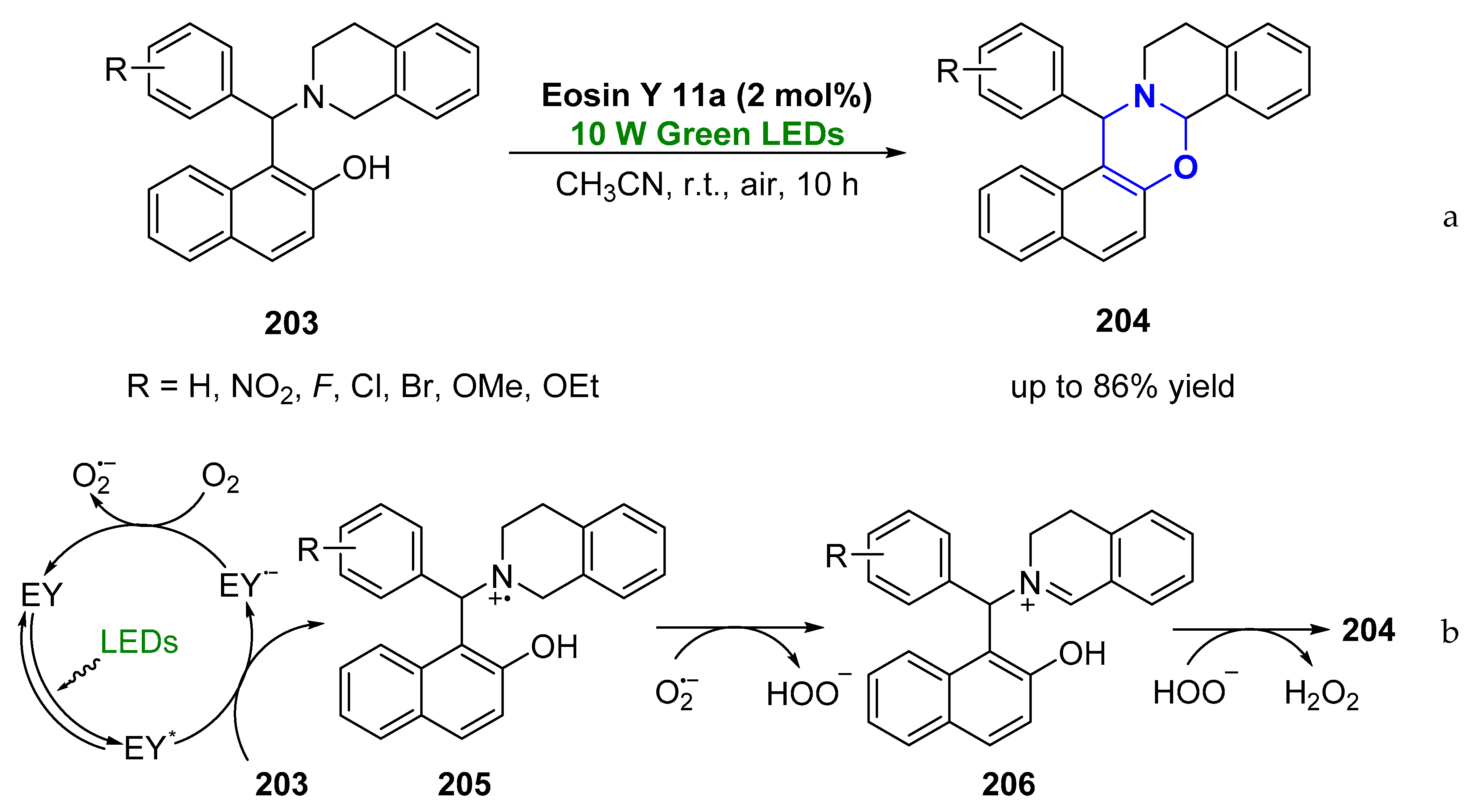
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pawlowski, R.; Stanek, F.; Stodulski, M. Recent Advances on Metal-Free, Visible-Light- Induced Catalysis for Assembling Nitrogen- and Oxygen-Based Heterocyclic Scaffolds. Molecules 2019, 24, 1533. https://doi.org/10.3390/molecules24081533
Pawlowski R, Stanek F, Stodulski M. Recent Advances on Metal-Free, Visible-Light- Induced Catalysis for Assembling Nitrogen- and Oxygen-Based Heterocyclic Scaffolds. Molecules. 2019; 24(8):1533. https://doi.org/10.3390/molecules24081533
Chicago/Turabian StylePawlowski, Robert, Filip Stanek, and Maciej Stodulski. 2019. "Recent Advances on Metal-Free, Visible-Light- Induced Catalysis for Assembling Nitrogen- and Oxygen-Based Heterocyclic Scaffolds" Molecules 24, no. 8: 1533. https://doi.org/10.3390/molecules24081533
APA StylePawlowski, R., Stanek, F., & Stodulski, M. (2019). Recent Advances on Metal-Free, Visible-Light- Induced Catalysis for Assembling Nitrogen- and Oxygen-Based Heterocyclic Scaffolds. Molecules, 24(8), 1533. https://doi.org/10.3390/molecules24081533





