Comparison of Two Process Schemes Combining Hydrothermal Treatment and Acidogenic Fermentation of Source-Separated Organics
Abstract
1. Introduction
2. Results and Discussion
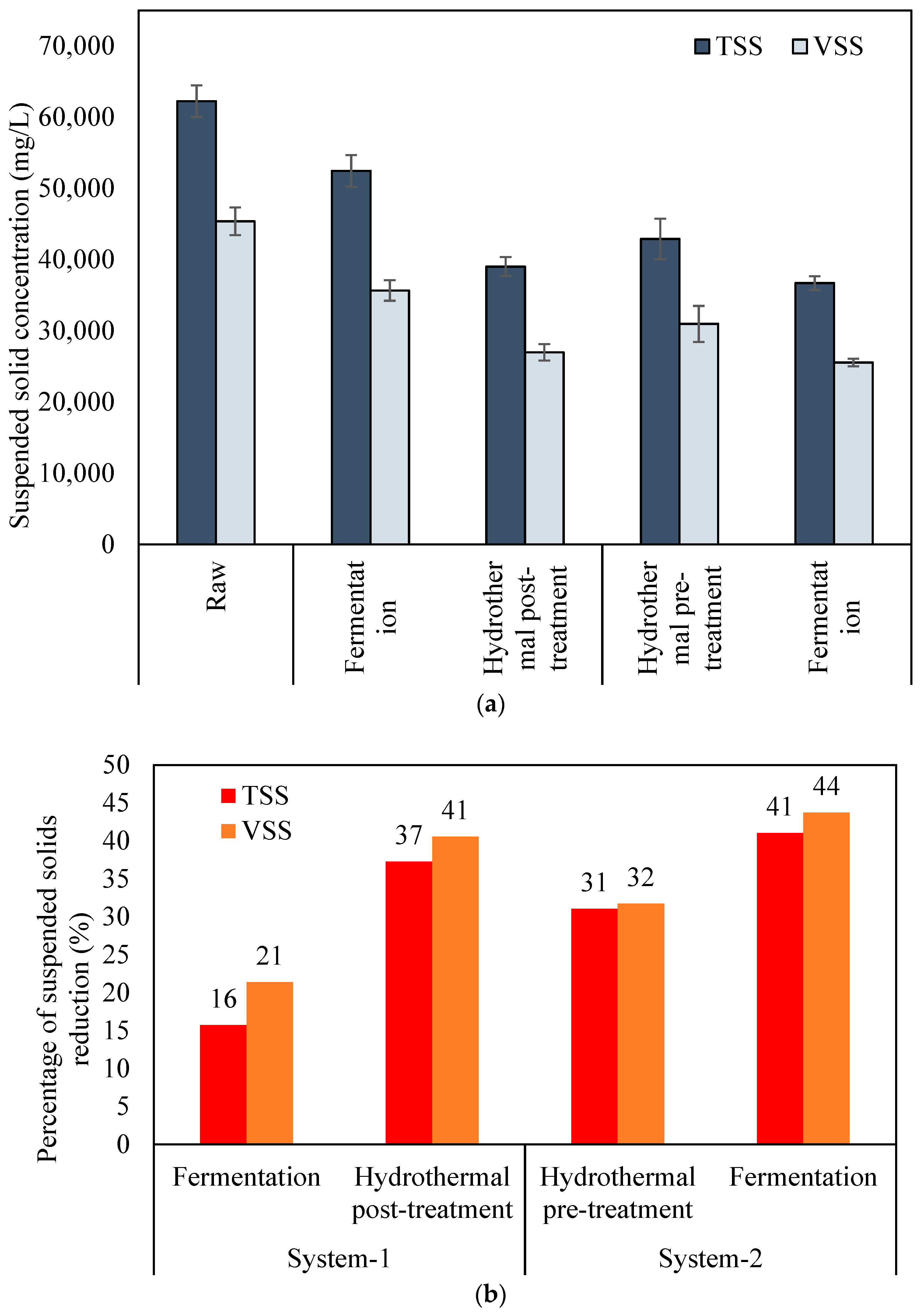
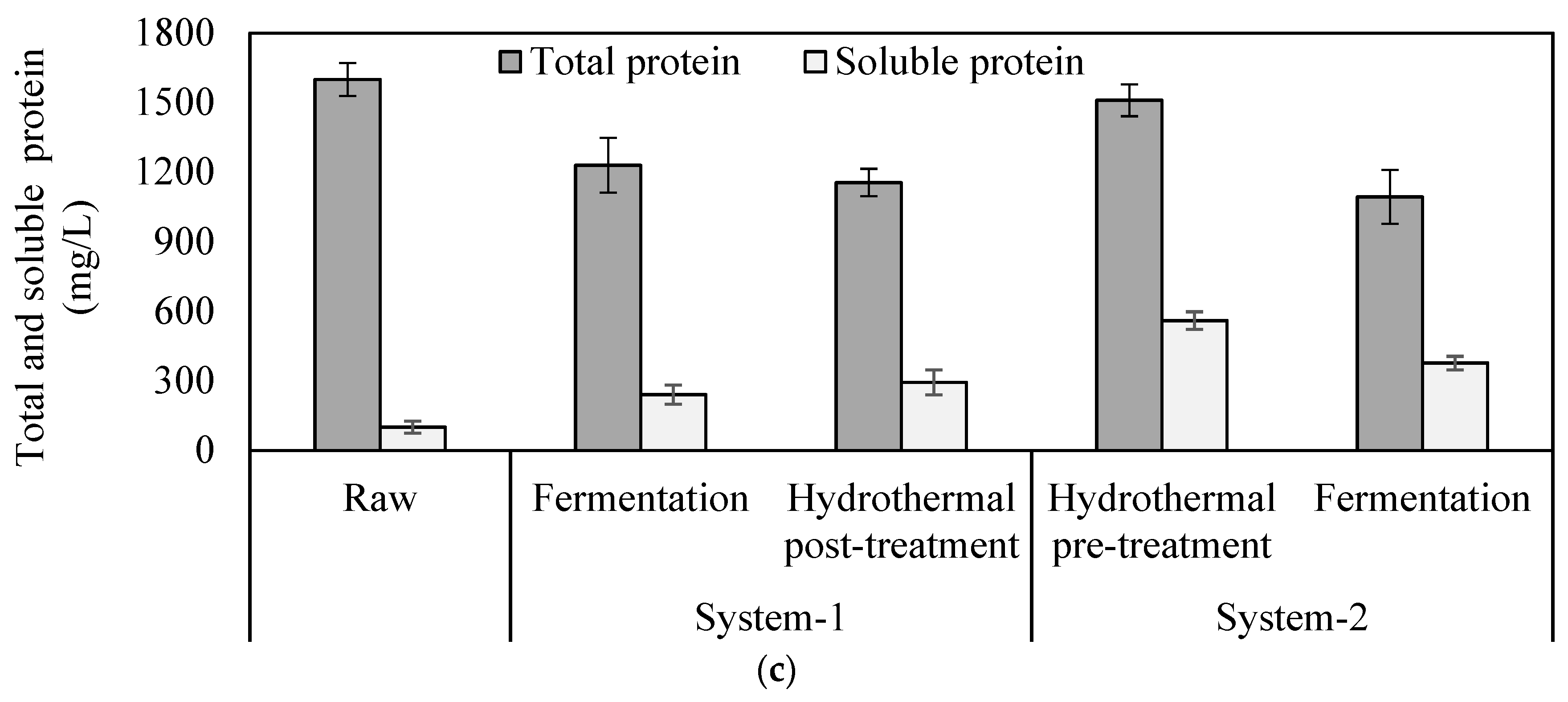
2.1. Solubilization of Particulate Matters
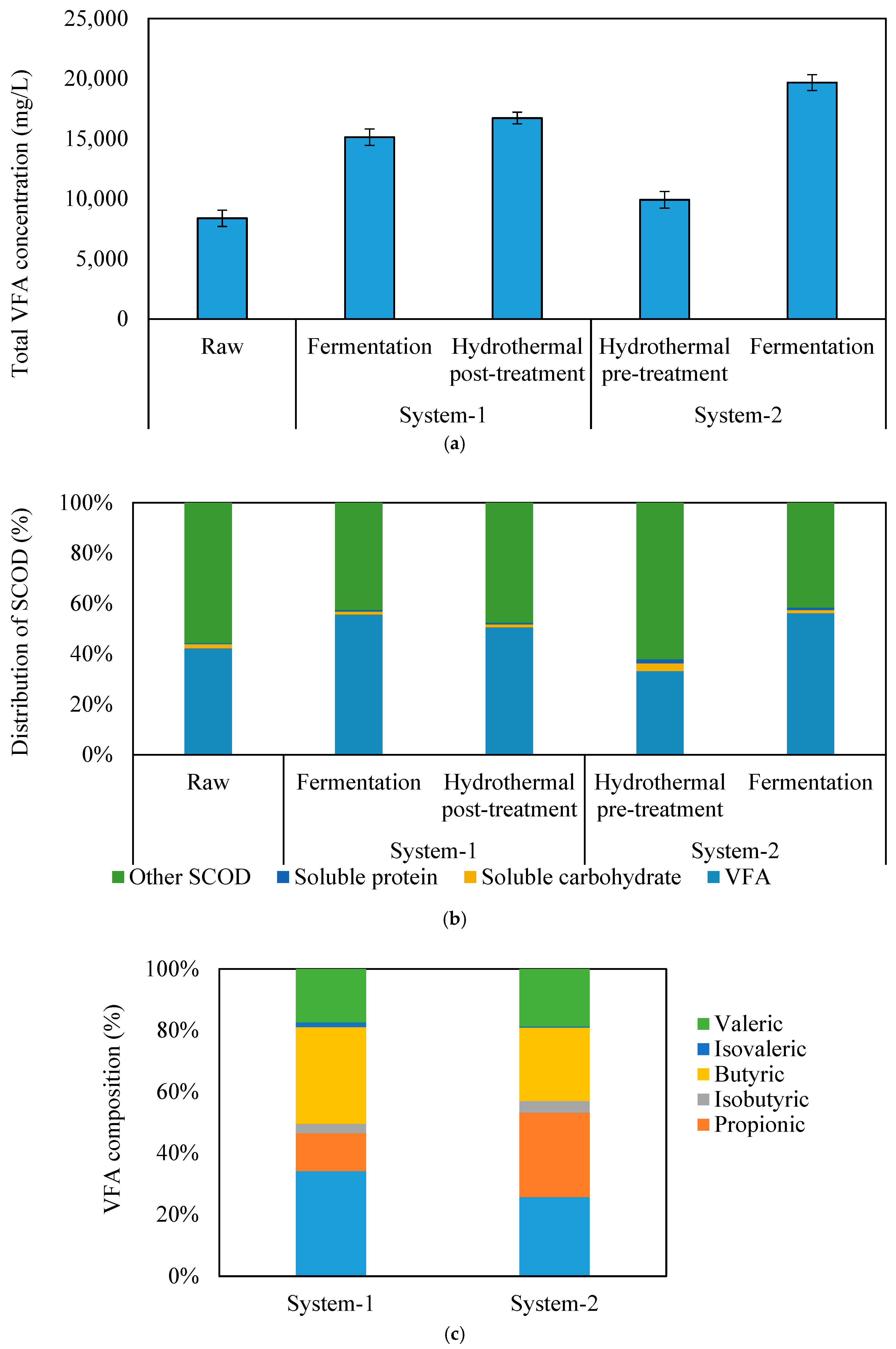
2.2. Yields and Distribution of VFAs
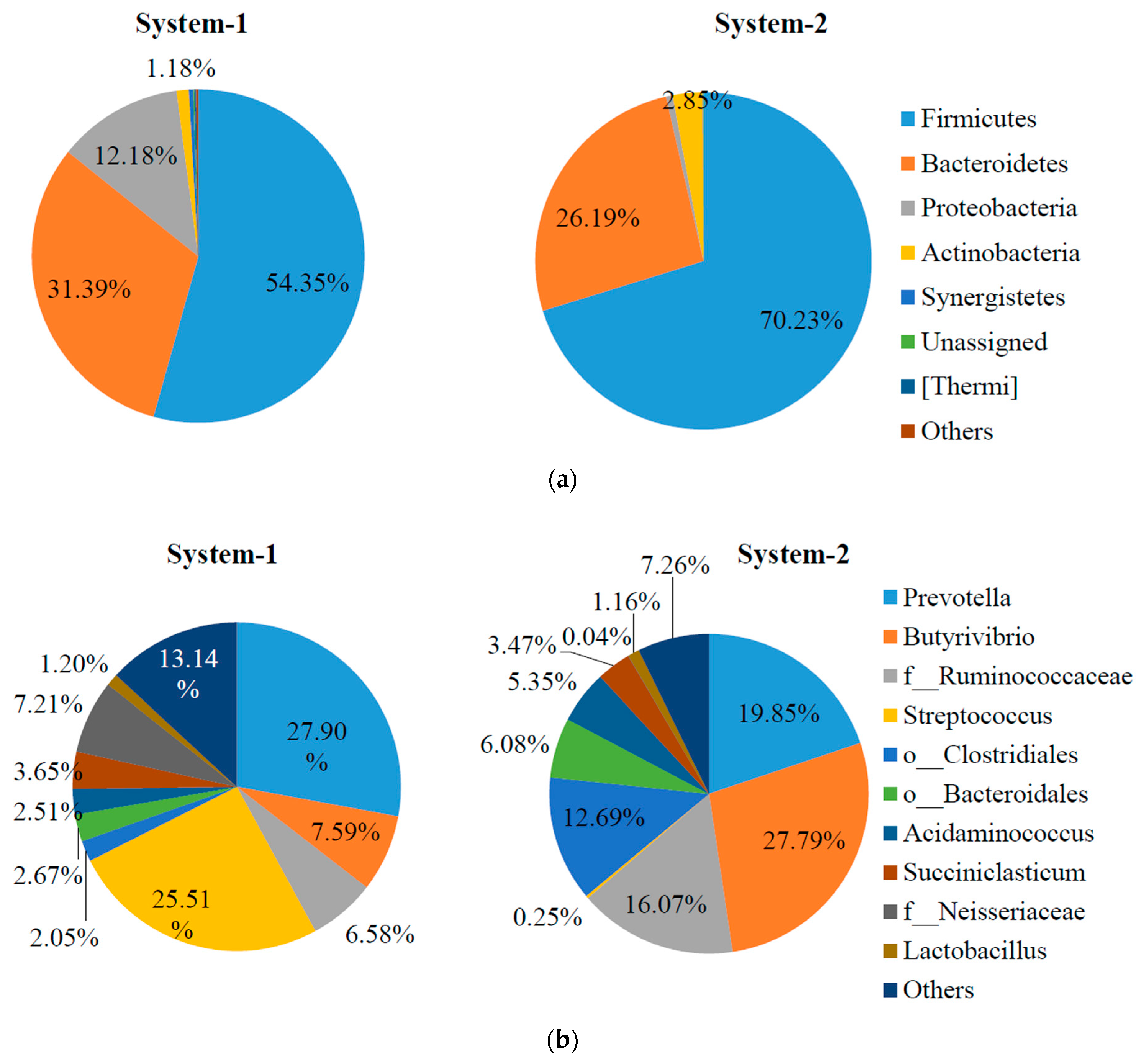
2.3. Microbial Community
3. Materials and Methods
3.1. SSO and Inoculum
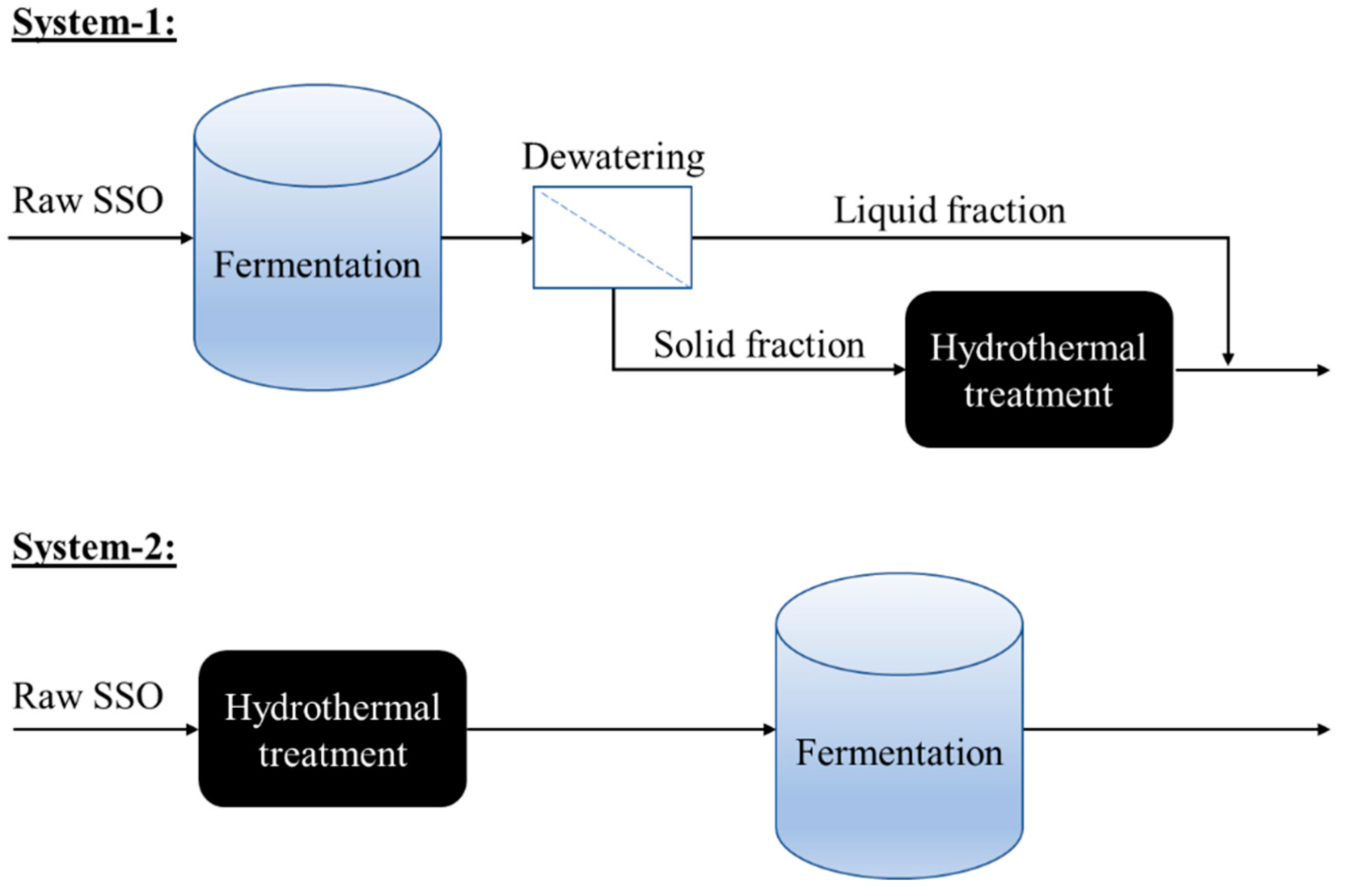
3.2. Experimental Design and Set-Up
3.3. Analytical Methods
3.4. Microbial Community Analysis
3.5. Calculations
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Xu, F.; Li, Y.; Ge, X.; Yang, L.; Li, Y. Anaerobic digestion of food waste–challenges and opportunities. Bioresour. Technol. 2018, 247, 1047–1058. [Google Scholar] [CrossRef] [PubMed]
- Paritosh, K.; Kushwaha, S.K.; Yadav, M.; Pareek, N.; Chawade, A.; Vivekanand, V. Food waste to energy: An overview of sustainable approaches for food waste management and nutrient recycling. Biomed. Res. Int. 2017, 2017, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Luk, G.K.; Bekmuradov, V. Energy products from source-separated organic waste. WIT Trans. Ecol. Environ. 2014, 180, 469–478. [Google Scholar]
- Hosseini Koupaie, E.; Azizi, A.; Bazyar Lakeh, A.A.; Hafez, H.; Elbeshbishy, E. Comparison of liquid and dewatered digestate as inoculum for anaerobic digestion of organic solid wastes. Waste Manag. 2019, 87, 228–236. [Google Scholar]
- Lee, W.S.; Chua, A.S.M.; Yeoh, H.K.; Ngoh, G.C. A review of the production and applications of waste-derived volatile fatty acids. Chem. Eng. J. 2014, 235, 83–99. [Google Scholar] [CrossRef]
- Cesaro, A.; Belgiorno, V. Pretreatment methods to improve anaerobic biodegradability of organic municipal solid waste fractions. Chem. Eng. J. 2014, 240, 24–37. [Google Scholar] [CrossRef]
- Zheng, Y.; Zhao, J.; Xu, F.; Li, Y. Pretreatment of lignocellulosic biomass for enhanced biogas production. Prog. Energy Combust. Sci. 2014, 42, 35–53. [Google Scholar] [CrossRef]
- Dhar, B.R.; Nakhla, G.; Ray, M.B. Techno-economic evaluation of ultrasound and thermal pretreatments for enhanced anaerobic digestion of municipal waste activated sludge. Waste Manag. 2012, 32, 542–549. [Google Scholar] [CrossRef] [PubMed]
- Ariunbaatar, J.; Panico, A.; Esposito, G.; Pirozzi, F.; Lens, P.N.L. Pretreatment methods to enhance anaerobic digestion of organic solid waste. Appl. Energy 2014, 123, 143–156. [Google Scholar] [CrossRef]
- Yin, J.; Wang, K.; Yang, Y.; Shen, D.; Wang, M.; Mo, H. Improving production of volatile fatty acids from food waste fermentation by hydrothermal pretreatment. Bioresour. Technol. 2014, 171, 323–329. [Google Scholar] [CrossRef]
- Yu, X.; Yin, J.; Wang, K.; Shen, D.; Long, Y.; Chen, T. Enhancing Food Waste Hydrolysis and the Production Rate of Volatile Fatty Acids by Prefermentation and Hydrothermal Pretreatments. Energy Fuels 2016, 30, 4002–4008. [Google Scholar]
- Ding, L.; Cheng, J.; Qiao, D.; Yue, L.; Li, Y.Y.; Zhou, J.; Cen, K. Investigating hydrothermal pretreatment of food waste for two-stage fermentative hydrogen and methane co-production. Bioresour. Technol. 2017, 241, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Razavi, A.S.; Hosseini Koupaie, E.; Azizi, A.; Hafez, H.; Elbeshbishy, E. Hydrothermal pretreatment of source separated organics for enhanced solubilization and biomethane recovery. Bioresour. Technol. 2018, 274, 502–511. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Jin, Y. Effects of thermal pretreatment on acidification phase during two-phase batch anaerobic digestion of kitchen waste. Renew. Energy 2015, 77, 550–557. [Google Scholar] [CrossRef]
- Dhar, B.R.; Elbeshbishy, E.; Hafez, H.; Lee, H.S. Hydrogen production from sugar beet juice using an integrated biohydrogen process of dark fermentation and microbial electrolysis cell. Bioresour. Technol. 2015, 198, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Xia, A.; Cheng, J.; Murphy, J.D. Innovation in biological production and upgrading of methane and hydrogen for use as gaseous transport biofuel. Biotechnol. Adv. 2016, 34, 451–472. [Google Scholar] [CrossRef] [PubMed]
- Elbeshbishy, E.; Hafez, H.; Dhar, B.R.; Nakhla, G. Single and combined effect of various pretreatment methods for biohydrogen production from food waste. Int. J. Hydrogen Energy 2011, 36, 11379–11387. [Google Scholar] [CrossRef]
- Cheng, J.; Ding, L.; Xia, A.; Lin, R.; Li, Y.; Zhou, J.; Cen, K. Hydrogen production using amino acids obtained by protein degradation in waste biomass by combined dark- and photo-fermentation. Bioresour. Technol. 2015, 179, 13–19. [Google Scholar] [CrossRef]
- Gonzalez-garcia, R.A.; Mccubbin, T.; Navone, L.; Stowers, C.; Nielsen, L.K.; Marcellin, E. Microbial Propionic Acid Production. Fermentation 2017, 3, 21. [Google Scholar] [CrossRef]
- Sundberg, C.; Franke-Whittle, I.H.; Kauppi, S.; Yu, D.; Romantschuk, M.; Insam, H.; Jönsson, H. Characterisation of source-separated household waste intended for composting. Bioresour. Technol. 2011, 102, 2859–2867. [Google Scholar] [CrossRef]
- Jin, Y.; Li, Y.; Li, J. Influence of thermal pretreatment on physical and chemical properties of kitchen waste and the efficiency of anaerobic digestion. J. Environ. Manag. 2016, 180, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Krajmalinik-Brown, R.; Zhang, H.; Rittmann, B.E. An electron-flow model can predict complex redox reactions in mixed-culture fermentative BioH2: Microbial ecology evidence. Biotechnol. Bioeng. 2009, 104, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Emerson, E.L.; Weimer, P.J. Fermentation of model hemicelluloses by Prevotella strains and Butyrivibrio fibrisolvens in pure culture and in ruminal enrichment cultures. Appl. Microbiol. Biotechnol. 2017, 101, 4269–4278. [Google Scholar] [CrossRef] [PubMed]
- Rogosa, M. Acidaminococcus gen. n., Acidaminococcus fermentans sp. n., anaerobic gram-negative diplococci using amino acids as the sole energy source for growth. J. Bacteriol. 1969, 98, 756–766. [Google Scholar]
- Cabrol, L.; Marone, A.; Tapia-Venegas, E.; Steyer, J.; Ruiz-Filippi, G.; Trably, E. Microbial ecology of fermentative hydrogen producing bioprocesses: useful insights for driving the ecosystem function. FEMS Microbiol. Rev. 2017, 41, 158–181. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- DuBois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric Method for Determination of Sugars and Related Substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Frolund, B.; Griebe, T.; Nielsen, P.H. Enzymatic activity in the activated-sludge floc matrix. Appl. Microbiol. Biotechnol. 1995, 43, 755–761. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: high-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef]
- Rideout, J.R.; He, Y.; Navas-Molina, J.A.; Walters, W.A.; Ursell, L.K.; Gibbons, S.M.; Chase, J.; McDonald, D.; Gonzalez, A.; Robbins-Pianka, A.; et al. Subsampled open-reference clustering creates consistent, comprehensive OTU definitions and scales to billions of sequences. PeerJ. 2014, 2. [Google Scholar] [CrossRef]
- Angelidaki, I.; Sanders, W. Assessment of the anaerobic biodegradability of macropollutants. Rev. Environ. Sci. Bio/Technology 2004, 3, 117–129. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |

| Substrate | Hydrothermal Process Condition | Hydrothermal Process Severity Index | Fermentation Condition | VFA Yield | Increase in VFA | Reference |
|---|---|---|---|---|---|---|
| Food waste from canteen | 160 °C, 30 min | 3.2 | Batch, 30 °C, 15 days | 910 mg/g VSremoved | 55% | [10] |
| Restaurant, canteen, dining hall food waste | 160 °C, 20 min | 3.1 | Batch, Fermentation: 35 °C, 48 h | 450 mg/g VS | 240% | [12] |
| Food waste from canteen | 160 °C, 30 min | 3.2 | Batch, 30 °C, 21 days | 740 mg/g VSSremoved | 130% | [11] |
| Kitchen waste from canteen | 120 °C, 50 min | 2.3 | Batch, 35 °C, 21 days | 32 mg/g VS | No increase | [14] |
| Kitchen waste from canteen | 160 °C, 120 min | 3.8 | - | 280 mmol/L | 120% | [21] |
| 1.6 mmol/gVSS | ||||||
| SSO | 170 °C, 30 min | 3.5 | Semi-continuous, fed once a day, 38 °C, HRT = 3 days | 433 mg/gVSS | 135% | This study |
| 570 mg/gVSSremoved |
| Parameters | SSO | Inoculum |
|---|---|---|
| pH | 5.55 ± 0.1 | 7.9 ± 0.1 |
| TS mg/L | 65,300 ± 1900 | 15,700 ± 100 |
| VS mg/L | 47,900 ± 1304 | 9250 ± 150 |
| VS/TS (%) | 72 ± 0.3 | 59 ± 1 |
| TSS mg/L | 62,000 ± 3500 | 15,400 ± 300 |
| VSS mg/L | 45,400 ± 2000 | 10,000 ± 200 |
| VSS/TSS (%) | 72.9 ± 1.9 | 65.2 ± 0.3 |
| TCOD mg/L | 99,200 ± 3200 | 18,000 ± 200 |
| SCOD mg/L | 32,500 ± 900 | 500 ± 70 |
| Total carbohydrates (mg/L glucose) | 12,500 ± 450 | NA |
| Soluble carbohydrates (mg/L glucose) | 450 ± 30 | NA |
| Total protein mg/L | 1600 ± 150 | NA |
| Soluble protein mg/L | 100 ± 15 | NA |
| Total nitrogen (mg/L N) | 3100 ± 607 | 1300 ± 40 |
| Total phosphate (mg/L PO4−) | 1250 ± 354 | 1350 ± 30 |
| Alkalinity (mg/L CaCO3) | 5600 ± 300 | 4700 ± 15 |
| Ammonia (NH3-N) | 900 ± 300 | 700 ± 50 |
| VFA (mg/L HoAc) | 8400 ± 680 | 680 ± 20 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, L.; Hosseini Koupaie, E.; Azizi, A.; Bazyar Lakeh, A.A.; Dhar, B.R.; Hafez, H.; Elbeshbishy, E. Comparison of Two Process Schemes Combining Hydrothermal Treatment and Acidogenic Fermentation of Source-Separated Organics. Molecules 2019, 24, 1466. https://doi.org/10.3390/molecules24081466
Lin L, Hosseini Koupaie E, Azizi A, Bazyar Lakeh AA, Dhar BR, Hafez H, Elbeshbishy E. Comparison of Two Process Schemes Combining Hydrothermal Treatment and Acidogenic Fermentation of Source-Separated Organics. Molecules. 2019; 24(8):1466. https://doi.org/10.3390/molecules24081466
Chicago/Turabian StyleLin, Long, Ehssan Hosseini Koupaie, Armineh Azizi, Amir Abbas Bazyar Lakeh, Bipro R. Dhar, Hisham Hafez, and Elsayed Elbeshbishy. 2019. "Comparison of Two Process Schemes Combining Hydrothermal Treatment and Acidogenic Fermentation of Source-Separated Organics" Molecules 24, no. 8: 1466. https://doi.org/10.3390/molecules24081466
APA StyleLin, L., Hosseini Koupaie, E., Azizi, A., Bazyar Lakeh, A. A., Dhar, B. R., Hafez, H., & Elbeshbishy, E. (2019). Comparison of Two Process Schemes Combining Hydrothermal Treatment and Acidogenic Fermentation of Source-Separated Organics. Molecules, 24(8), 1466. https://doi.org/10.3390/molecules24081466







