Photo-Reduction of CO2 by VIS Light on Polythiophene-ZSM-5 Zeolite Hybrid Photo-Catalyst
Abstract
:1. Introduction
2. Results and Discussion
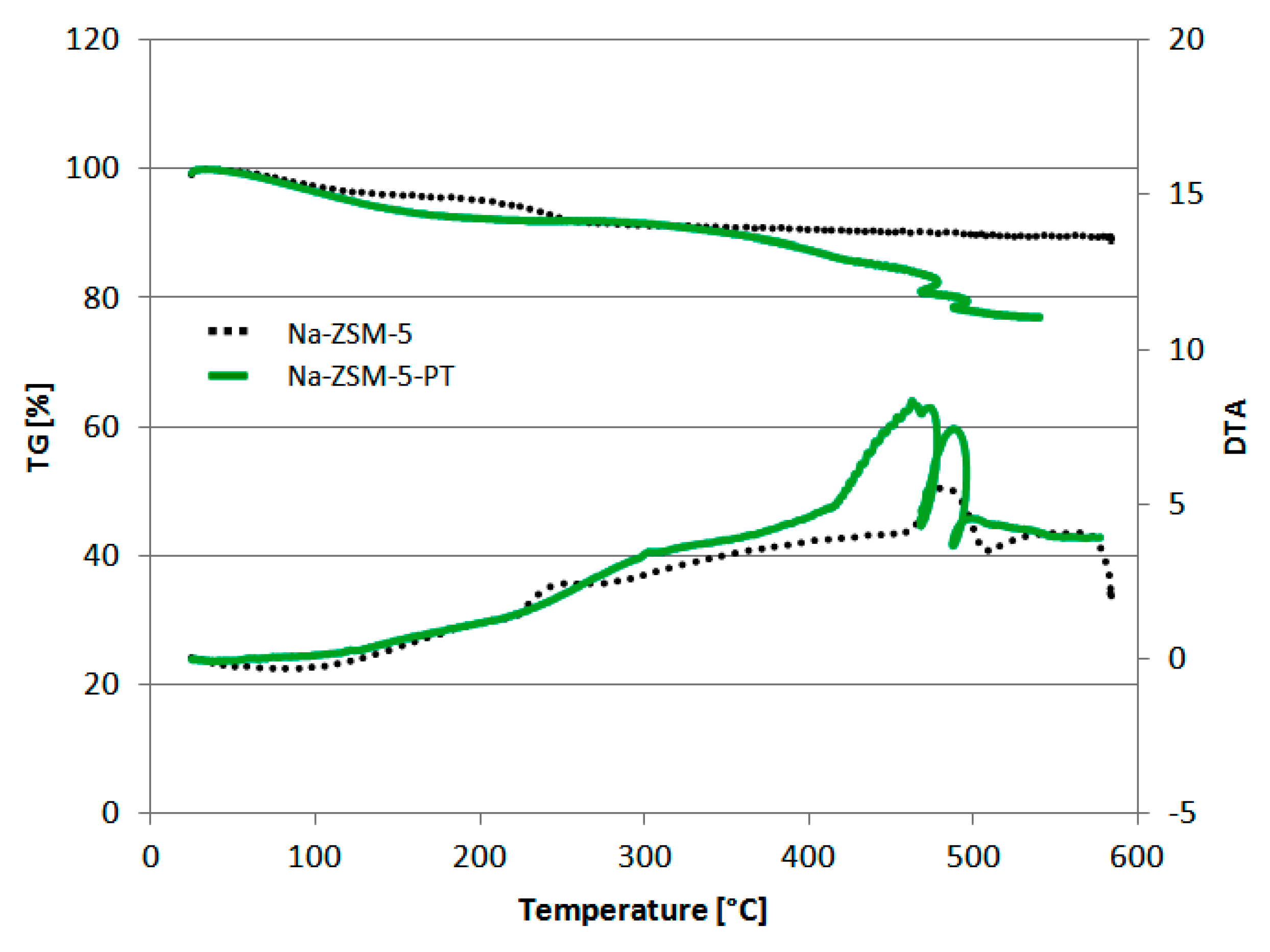
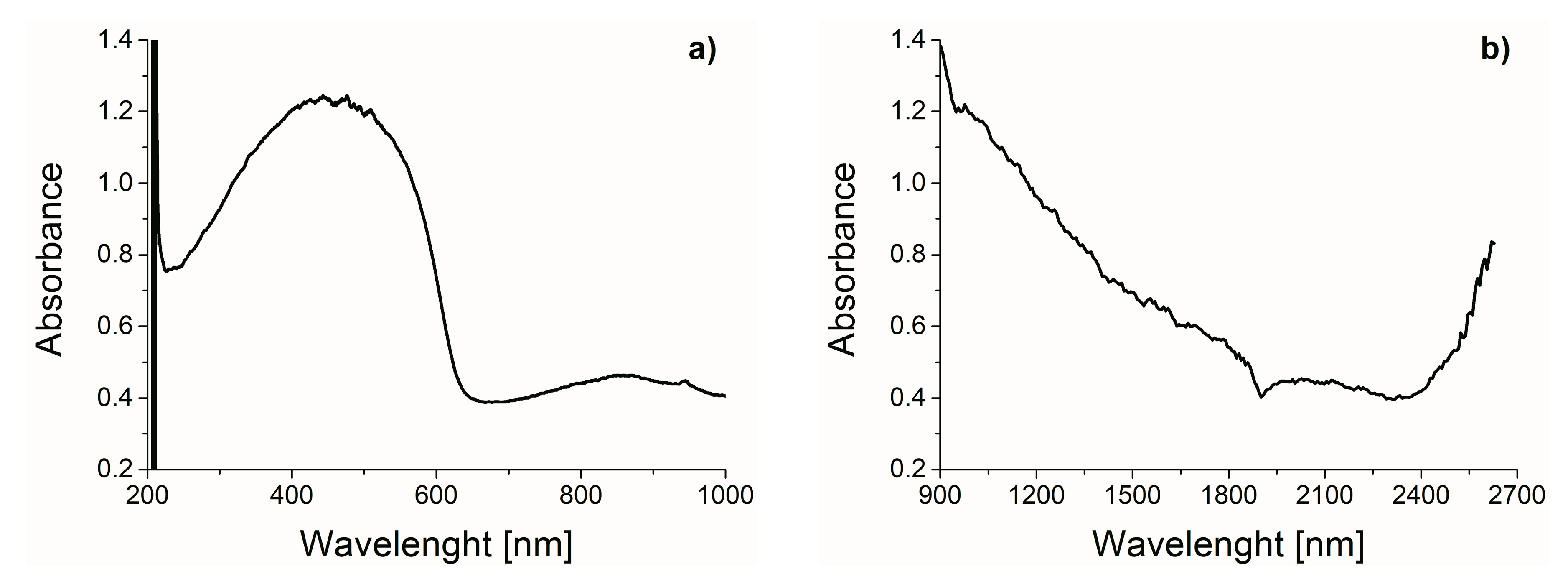
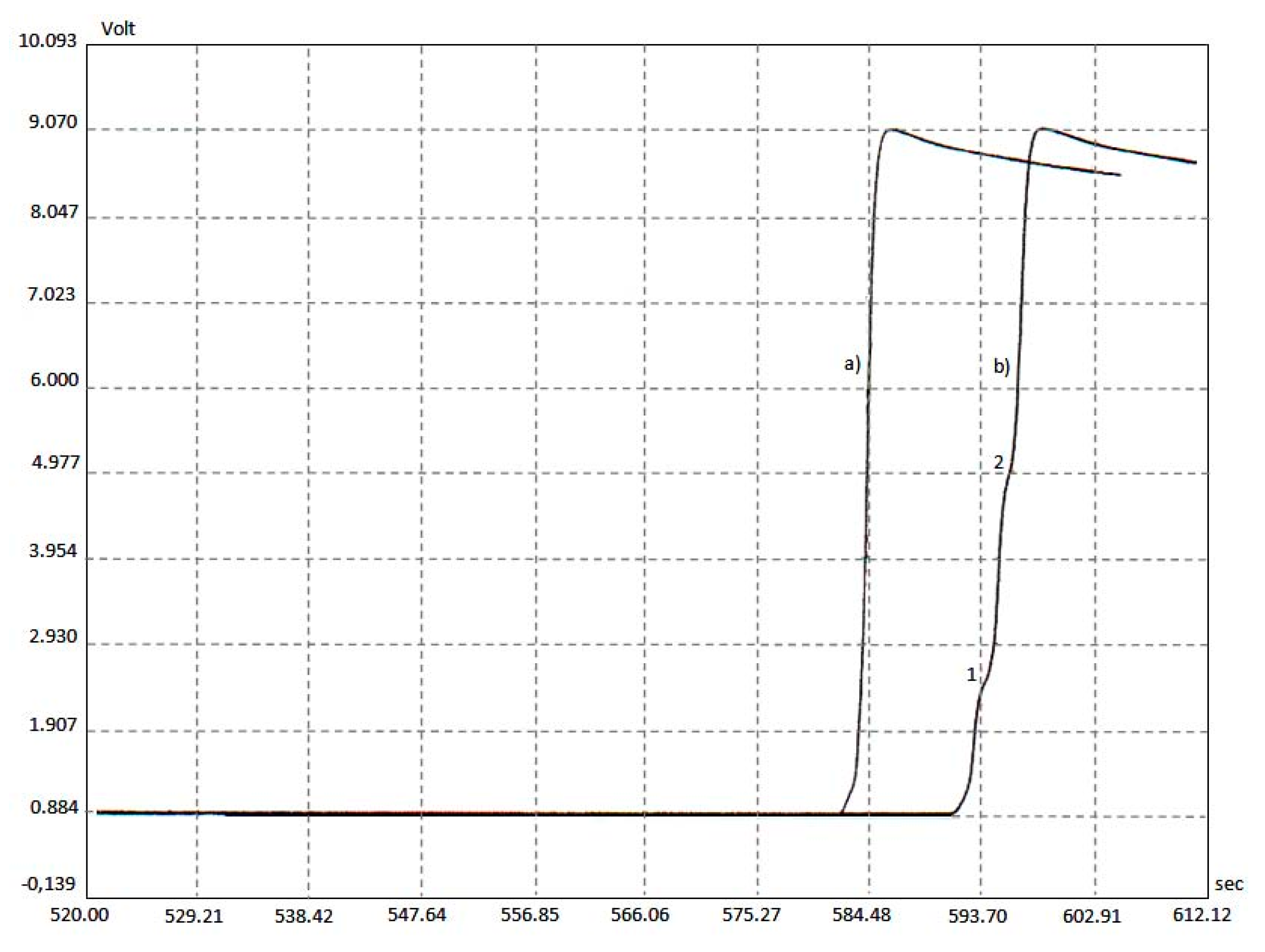
2.1. Characterization of Hybrid Photo-Catalyst Na-ZSM-5-PT
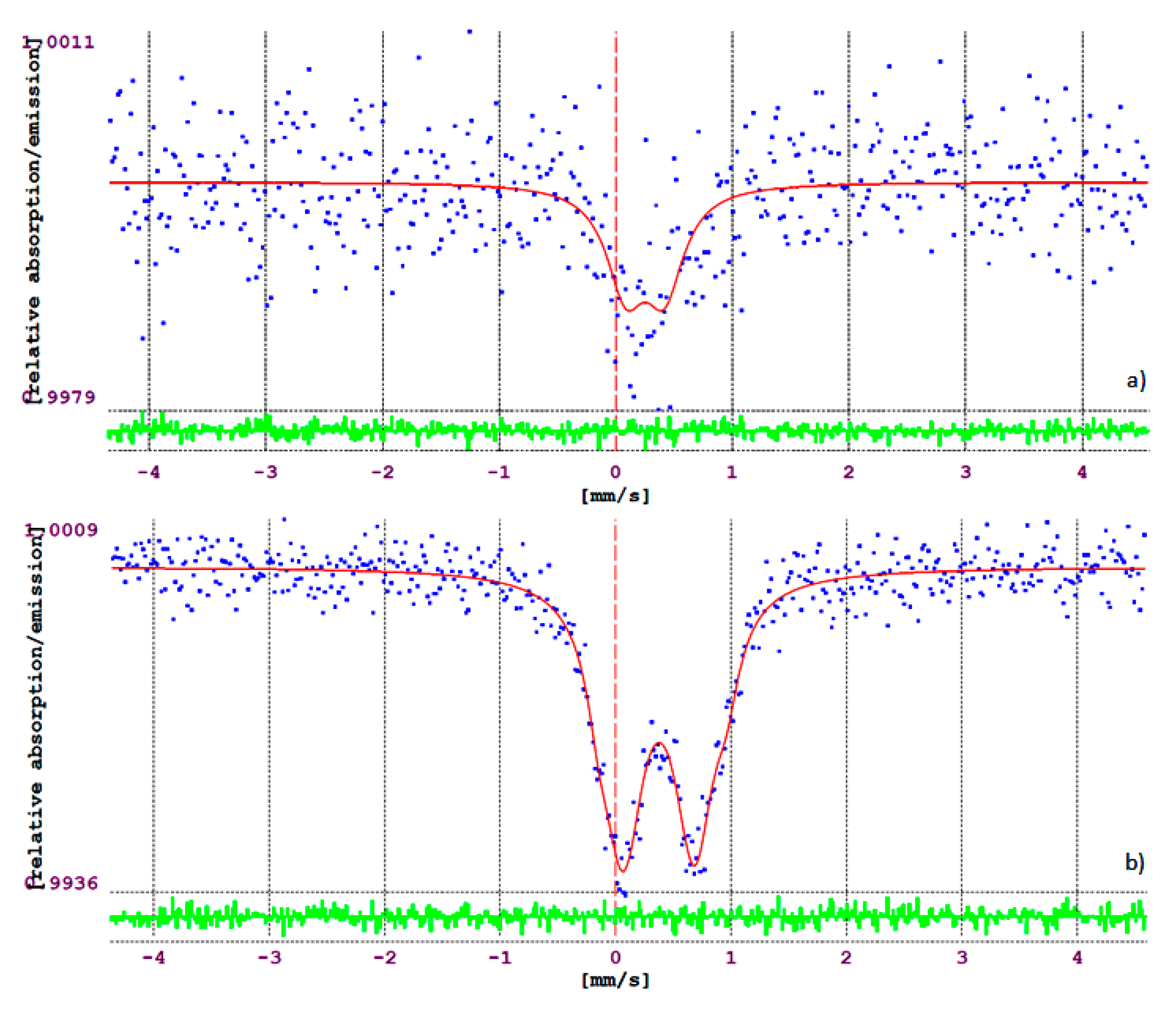
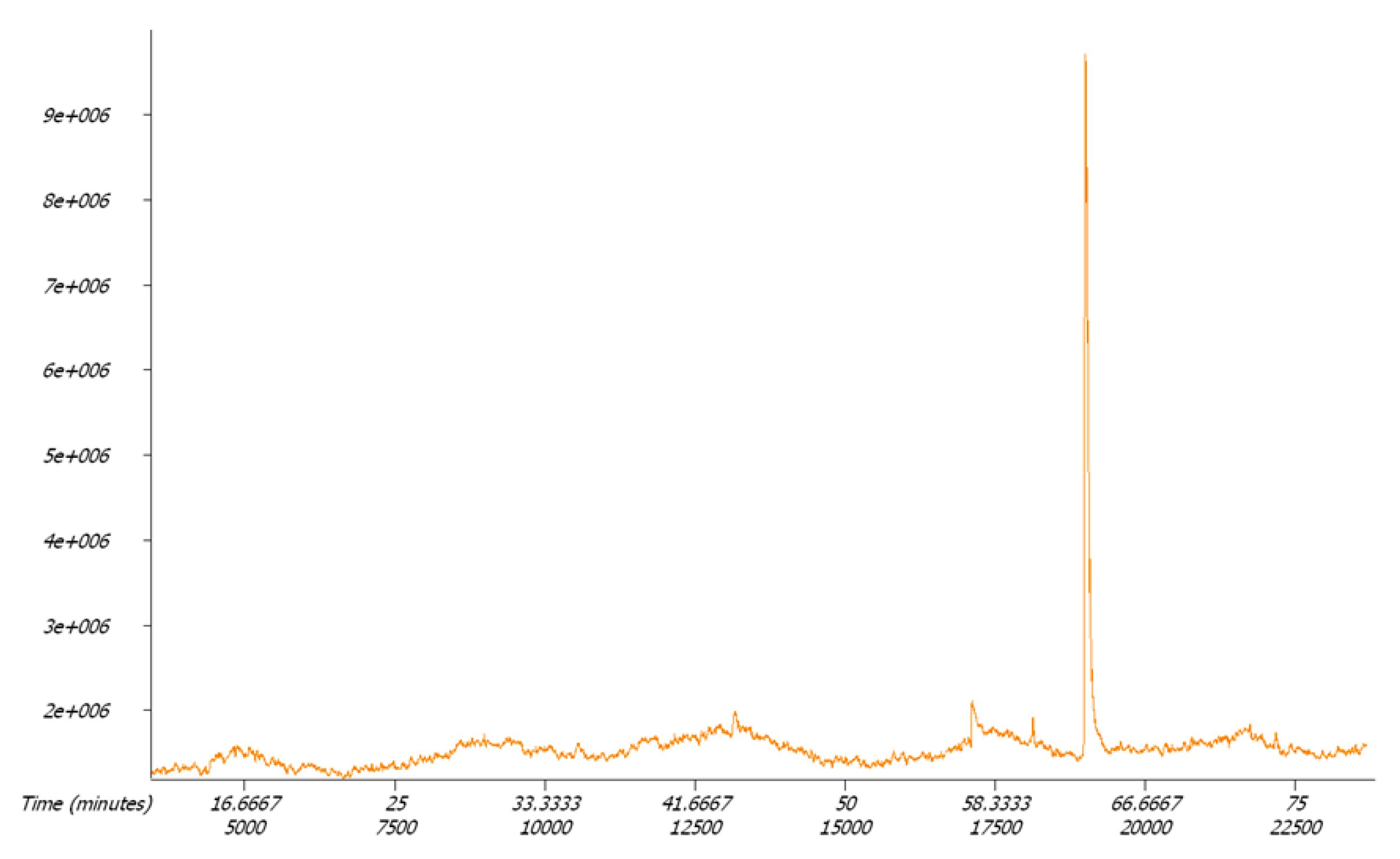
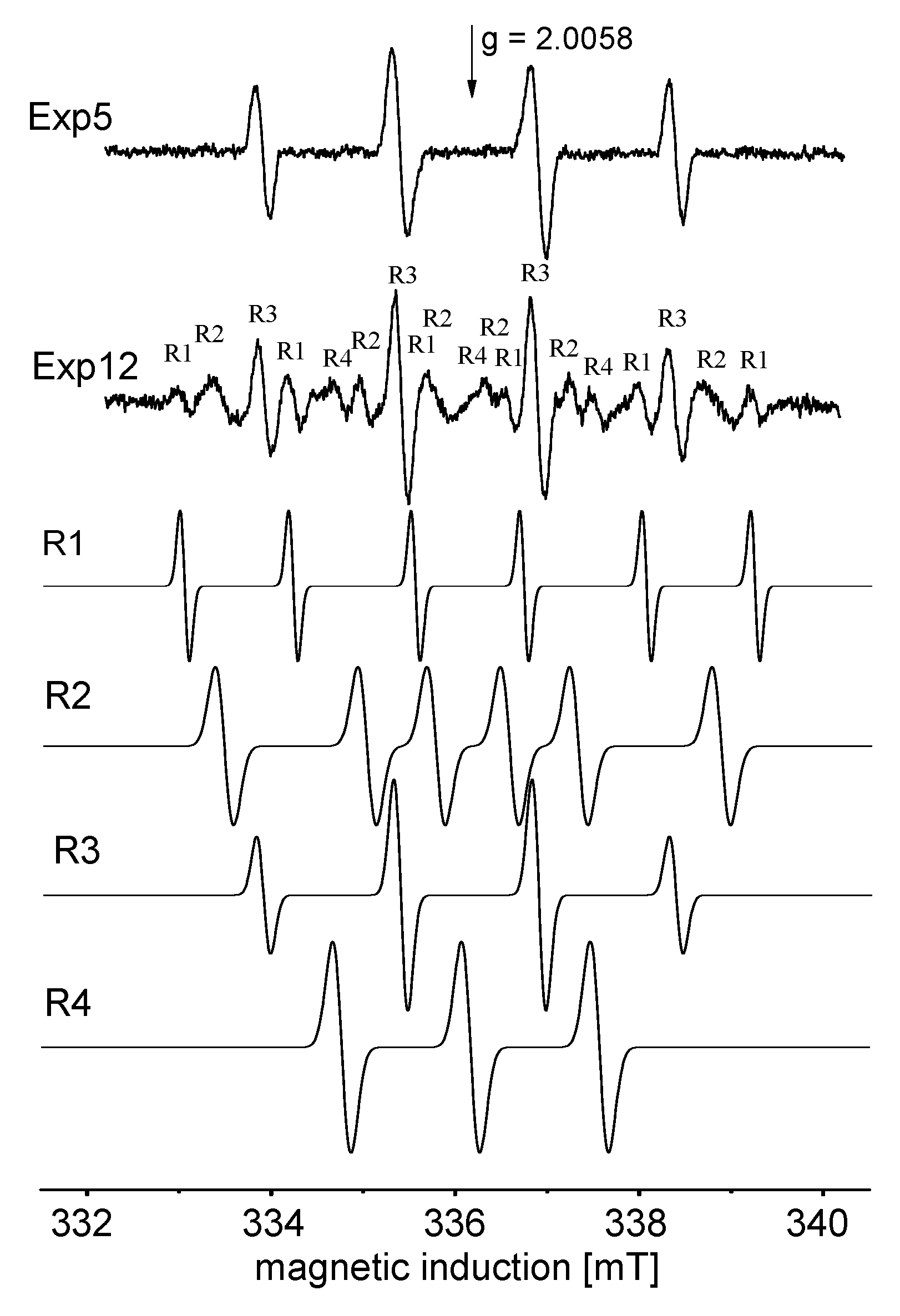
2.2. Detection of Radical Intermediates and Products of CO2 Photo-Reduction in the Aquatic Environment; Photo-Reduction Mechanism
3. Experimental
3.1. Preparation of Photo-Catalyst
3.2. Characterisation of Photo-Catalyst
3.3. Photocatalytical Experiment
3.4. EPR Spin Trapping Experiment
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- National Centers for Environmental Information (NOAA). State of the Climate: Global Analysis for August 2015. Available online: http://www.ncdc.noaa.gov/sotc/global201508 (accessed on 28 January 2016).
- Dlugokencky, E.; Tans, P. NOAA/ESRL. Available online: www.esrl.noaa.gov/gmd/ccgg/trends (accessed on 9 March 2019).
- Tahir, M.; Amin, N.S. Photocatalityc reduction of carbon dioxide with water vapors over montmorillonite modified TiO2 nanocomposites. Appl. Catal. B Environ. 2013, 142–143, 512–522. [Google Scholar] [CrossRef]
- Schulz, M.; Karnahl, M.; Schwalbe, M.; Vos, J.G. The role of the bridging ligand in photocatalytic supramolecular assemblies for the reduction of protons and carbon dioxide. Coord. Chem. Rev. 2012, 256, 1682–1705. [Google Scholar] [CrossRef]
- Lee, D.-S.; Chen, H.-J.; Chen, Y.-W. Photocatalytic reduction of carbon dioxide with water using InNbO4 catalyst with NiO and Co3O4 cocatalysts. J. Phys. Chem. Solids 2012, 73, 661–669. [Google Scholar] [CrossRef]
- Inoue, T.; Akira, F.; Satoshi, K.; Kenichi, H. Photoelectrocatalytic reduction of carbon dioxide in aqueous suspensions of semiconductor powders. Nature 1979, 277, 637–638. [Google Scholar] [CrossRef]
- Jeyalakshmi, V.; Mahalakshmy, R.; Krishnamurthy, K.R.; Viswanathan, B. Photocatalytic reduction of carbon dioxide by water on titania: Role of photophysical and structural properties. Indian J. Chem. 2012, 51A, 411–419. [Google Scholar]
- Anpo, M.; Shima, T.; Kodama, S.; Kubokawa, Y. Photocatalytic hydrogenation of prepyne with water on smal-particle titania: Size quantization effects and reaction intermediates. J. Phys. Chem. 1987, 91, 4305–4310. [Google Scholar] [CrossRef]
- Suib, S.L. New and Future Developments in Catalysis—Solar Photo-Catalysis; Elsevier: Amsterdam, The Netherlands, 2013; pp. 49–62. [Google Scholar]
- Yamashita, H.; Fujii, Y.; Ichihashi, Y.; Zhang, S.G.; Ikeue, K.; Park, D.R.; Koyano, K.; Tatsumi, T.; Anpo, M. Selective formation of CH3OH in photocatalytic reduction of CO2 with H2O on titanium oxides highly dispersed within zeolites and mesoporous molecular sieves. Catal. Today 1998, 45, 221–227. [Google Scholar] [CrossRef]
- Heeger, A.J.; Kivelson, S.; Schrieffer, J.R.; Su, W.P. Solitons in conducting polymers. Rev. Mod. Phys. 1988, 60, 781–850. [Google Scholar] [CrossRef]
- Liu, R.; Liu, Z. Polythiophene: Synthesis in aqueous medium and controllable morphology. Chin. Sci. Bull. 2009, 54, 2028–2032. [Google Scholar] [CrossRef]
- Čík, G.; Hubinová, M.; Šeršeň, F.; Krištín, J.; Antošová, M. Collect: Photocatalytic degradation of 4-chlorophenol by •OH radicals generated by thiophene oligomers incorporated in ZSM-5 zeolite channels. Collect. Czech. Chem. Commun. 2003, 68, 2219–2230. [Google Scholar] [CrossRef]
- Čík, G.; Priesolová, S.; Bujdáková, H.; Šeršeň, F.; Petheöová, T.; Krištín, J. Inactivation of bacteria G+-E. coli by phototic polythiophene incorporated in ZSM-5 zeolite. Chemosphere 2006, 63, 1419–1426. [Google Scholar] [CrossRef]
- Kosová, V. Inluence of Organic and Inorganic Molecules in Zeolite Synthesis. Chem. Listy 2005, 99, 411–420. [Google Scholar]
- Nalwa, N.S. (Ed.) Handbook of Conducting Molecules and Polymers; John Wiley & Sons: Chichester, UK, 1997; Volume 2, p. 331. [Google Scholar]
- Enzel, P.; Bein, T. Polythiophenes and oligothiophenes in zeolite hosts-conjugated nanometer-size filaments. Synth. Met. 1993, 55, 1238–1245. [Google Scholar] [CrossRef]
- Caspar, J.V.; Ramamurthy, V.; Corbin, D.R. Modification of photochemical reactivity by zeolites—Preparation and spectroscopic characterization of polarons and bipolarons of thiopheneoligomers within the channels of pentasil zeolites—The evolution of organic radical ions into conducting polymers. J. Am. Chem. Soc. 1991, 113, 600–610. [Google Scholar] [CrossRef]
- Čík, G.; Šeršeň, F.; Bumbálová, A. Photoassisted production of O2− and H2 in aqueous medium stimulated by polythiophene in ZSM-5 zeolite channels. Collect. Czech. Chem. Commun. 1999, 64, 149–156. [Google Scholar] [CrossRef]
- Kočí, K.; Obalová, L.; Matějová, M.; Plachá, D.; Lacný, Z.; Jirkovský, J.; Šolcová, O. Effect of TiO2 particles size on the photocatalytic reduction of CO2. Appl. Catal. B Environ. 2009, 89, 494–502. [Google Scholar]
- Dvoranová, D.; Barbieriková, Z.; Brezová, V. Radical intermediates in photoinduced reaction on TiO2 (An EPR spin trapping study). Molecules 2014, 19, 17279–17304. [Google Scholar]
- Spin Trap Database. Available online: http://tools.niehs.nih.gov/stdb/ (accessed on 9 March 2019).
- Polovka, M. EPR spectroscopy: A tool to characterize stability and antioxidant properties of food. J. Food Nutr. Res. 2006, 45, 1–11. [Google Scholar]
- Reszka, K.J.; McCormick, M.L.; Buettner, G.R.; Hart, C.M.; Britigan, B.E. Nitric oxide decreases the stability of DMPO spin adducts. Nitric Oxide 2006, 15, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Leaustic, V.; Babonneau, F.; Livage, J. Photoreactivity of tungsten trioxide dispersions: Spin trapping and electron spin resonance detection of radical intermediates. J. Phys. Chem. 1986, 90, 4193–4198. [Google Scholar] [CrossRef]
- Kočí, K.; Obalová, L.; Lacný, Z. Photocatalytic reduction of CO2 over TiO2 based catalysts. Chem. Pap. 2008, 62, 1–9. [Google Scholar] [CrossRef]
- Kaneco, S.; Shimizu, Y.; Ohta, K.; Mizuno, T. Photocatalytic reduction of high pressure carbon dioxide using TiO2 powders with a positive hole scavenger. J. Photochem. Photobiol. A Chem. 1998, 115, 223–226. [Google Scholar] [CrossRef]
- Anpo, M.; Yamashita, H.; Ischihashi, Y.; Ehara, S. Photocatalytic reduction of CO2 with H2O on various titanium-oxide catalysts. J. Electroanal. Chem. 1995, 396, 21–26. [Google Scholar] [CrossRef]
- Sasirekha, N.; Basha, S.J.S.; Shanthi, K. Photocatalytic performance of Ru doped anatase mounted on silica for reduction of carbon dioxide. Appl. Catal. B Environ. 2006, 62, 169–180. [Google Scholar] [CrossRef]
- Guan, T.; Kida, A.; Yoshida, A. Reduction of carbon dioxide with water under concentrated sunlight using photocatalyst combined with Fe-based catalyst. Catal. B Environ. 2003, 41, 387–396. [Google Scholar] [CrossRef]
- Subrahmanyam, M.; Kaneco, S.; Alonso-Vante, N. A screening for photo reduction of carbon dioxide supported on metal oxide catalysts for C1–C3 selectivity. Appl. Catal. B Environ. 1999, 23, 169–174. [Google Scholar] [CrossRef]
- Tseng, I.-H.; Chang, W.-C.; Wu, J.C.S. Photoproduction of CO2 using sol-gel titania-supported copper catalysts. Appl. Catal. B Environ. 2002, 37, 37–48. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compound NaZSM-5-PT is available from the author (G.Č.). |

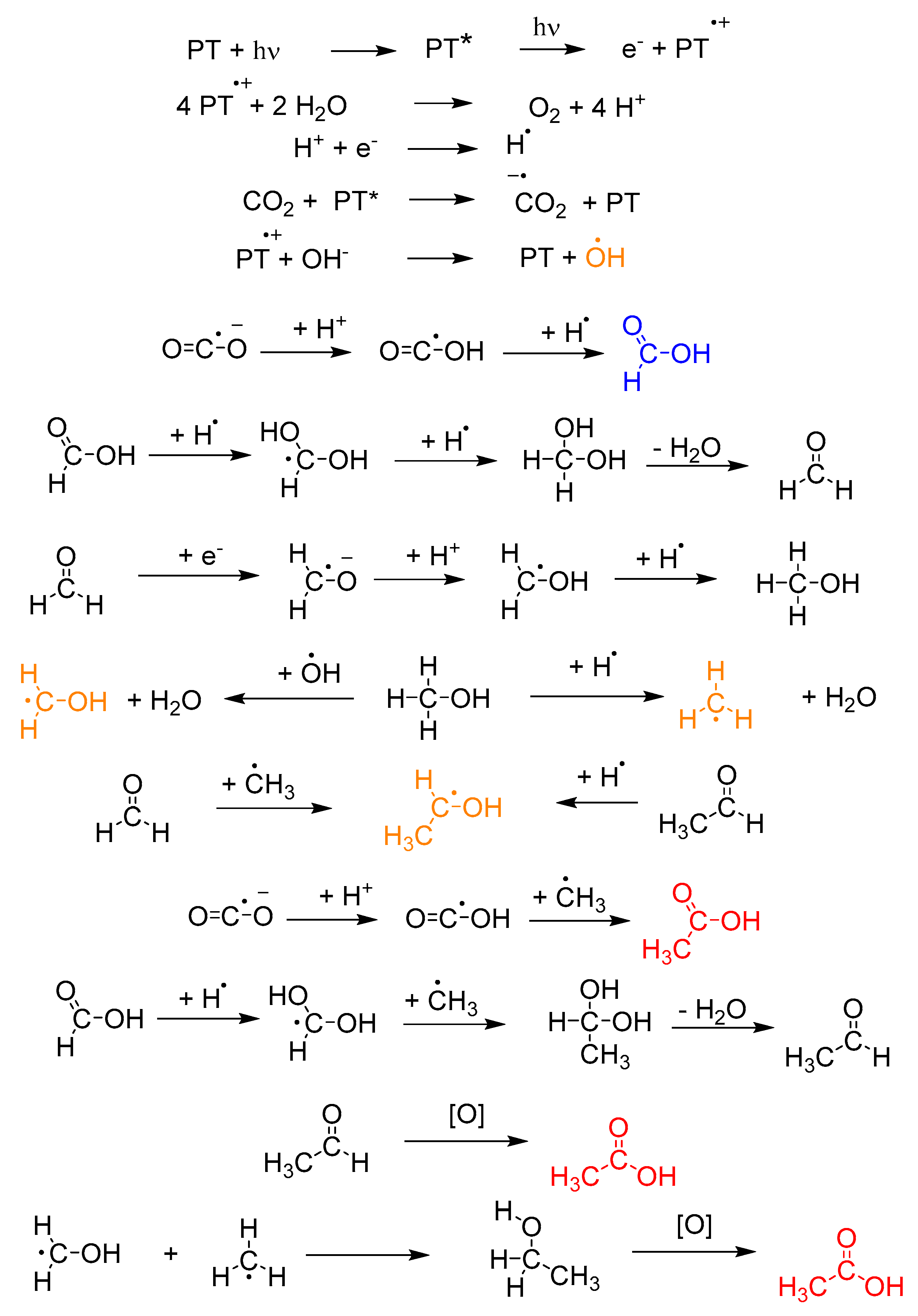
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kianička, J.; Čík, G.; Šeršeň, F.; Špánik, I.; Sokolík, R.; Filo, J. Photo-Reduction of CO2 by VIS Light on Polythiophene-ZSM-5 Zeolite Hybrid Photo-Catalyst. Molecules 2019, 24, 992. https://doi.org/10.3390/molecules24050992
Kianička J, Čík G, Šeršeň F, Špánik I, Sokolík R, Filo J. Photo-Reduction of CO2 by VIS Light on Polythiophene-ZSM-5 Zeolite Hybrid Photo-Catalyst. Molecules. 2019; 24(5):992. https://doi.org/10.3390/molecules24050992
Chicago/Turabian StyleKianička, Jana, Gabriel Čík, František Šeršeň, Ivan Špánik, Robert Sokolík, and Juraj Filo. 2019. "Photo-Reduction of CO2 by VIS Light on Polythiophene-ZSM-5 Zeolite Hybrid Photo-Catalyst" Molecules 24, no. 5: 992. https://doi.org/10.3390/molecules24050992
APA StyleKianička, J., Čík, G., Šeršeň, F., Špánik, I., Sokolík, R., & Filo, J. (2019). Photo-Reduction of CO2 by VIS Light on Polythiophene-ZSM-5 Zeolite Hybrid Photo-Catalyst. Molecules, 24(5), 992. https://doi.org/10.3390/molecules24050992




