Transcriptomic Analysis of Vibrio parahaemolyticus Reveals Different Virulence Gene Expression in Response to Benzyl Isothiocyanate
Abstract
1. Introduction
2. Results
2.1. Antibacterial Tests
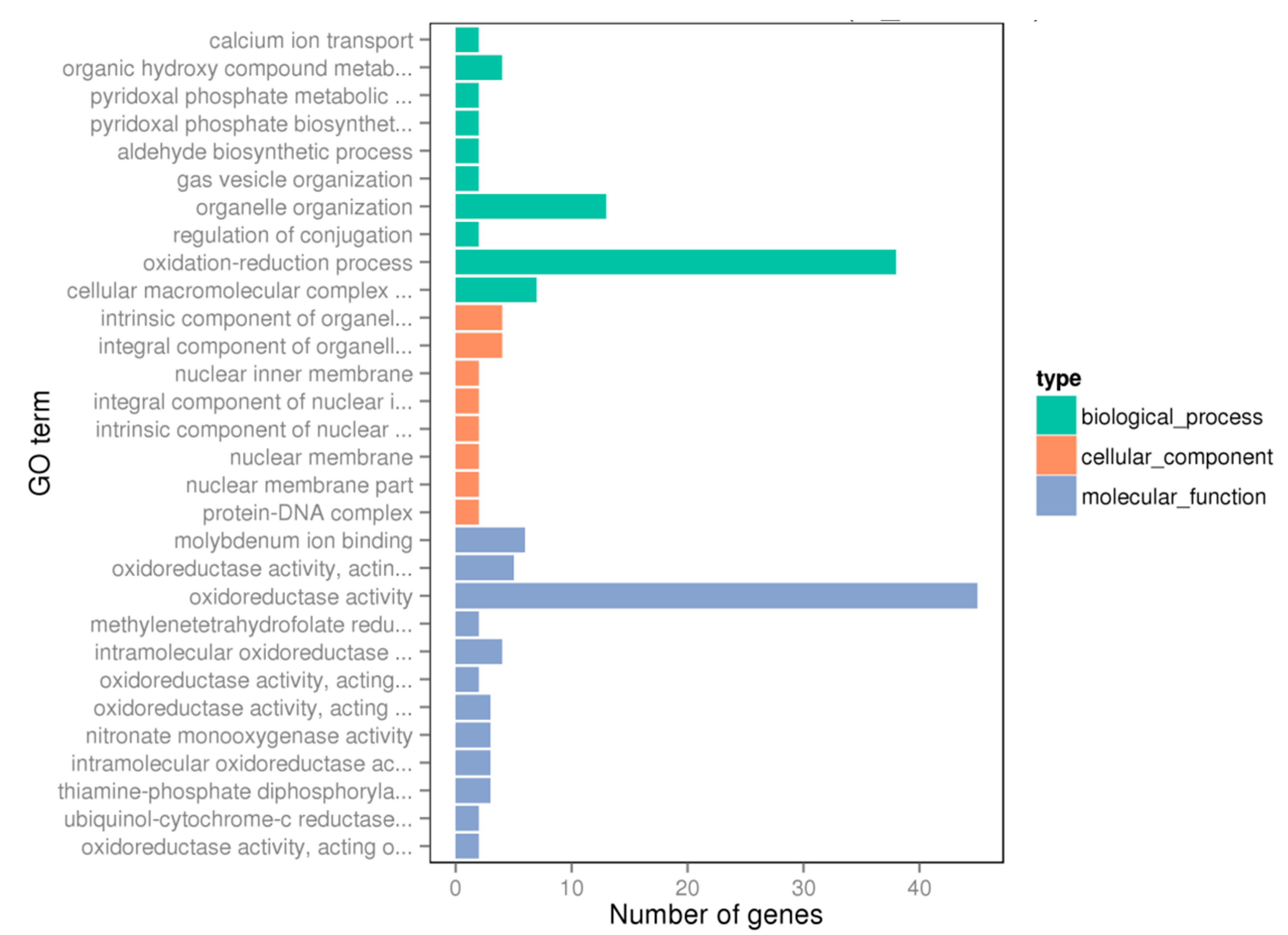
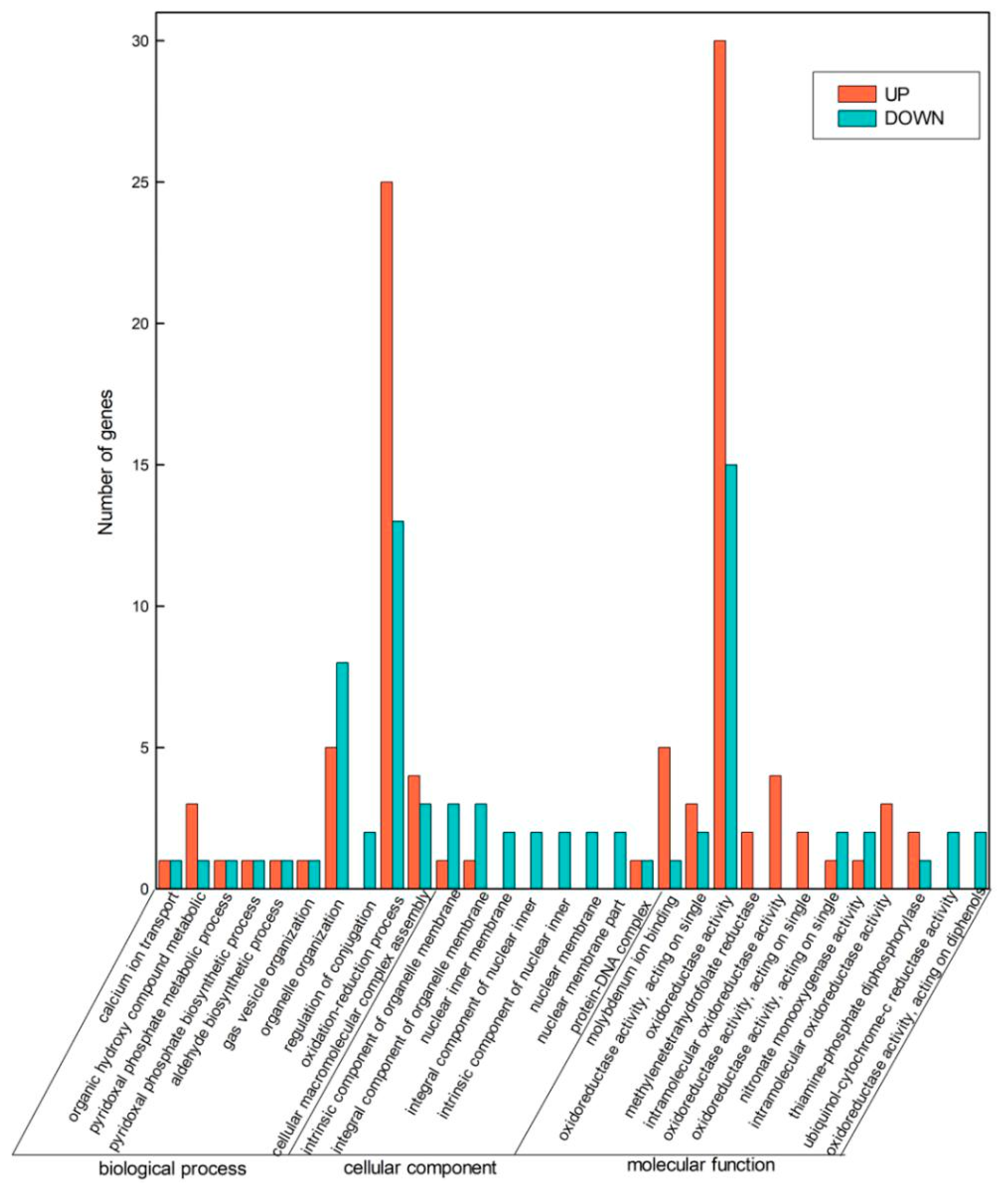
2.2. Global Changes at Transcriptome Level
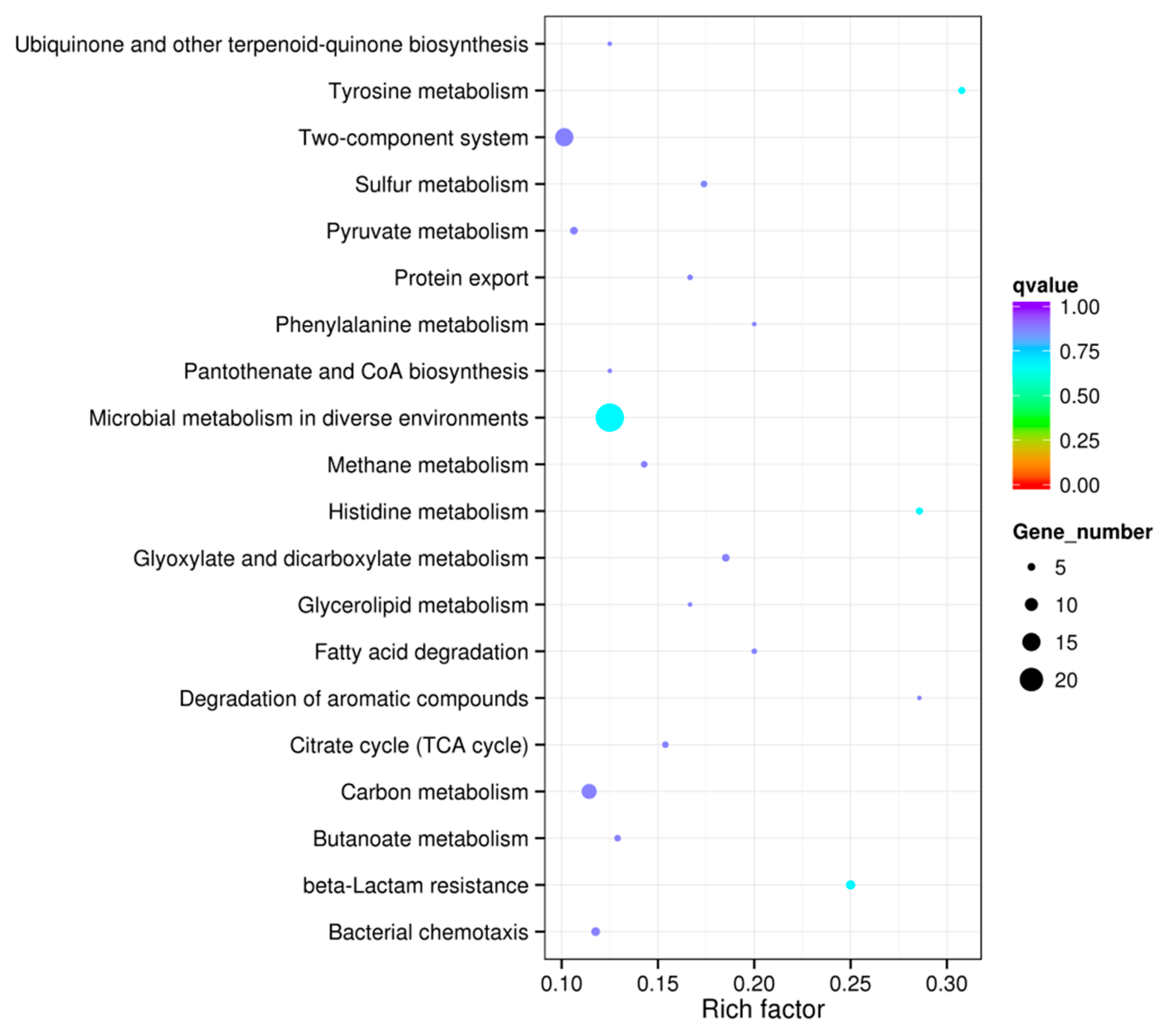
2.3. KEGG Pathway Analysis
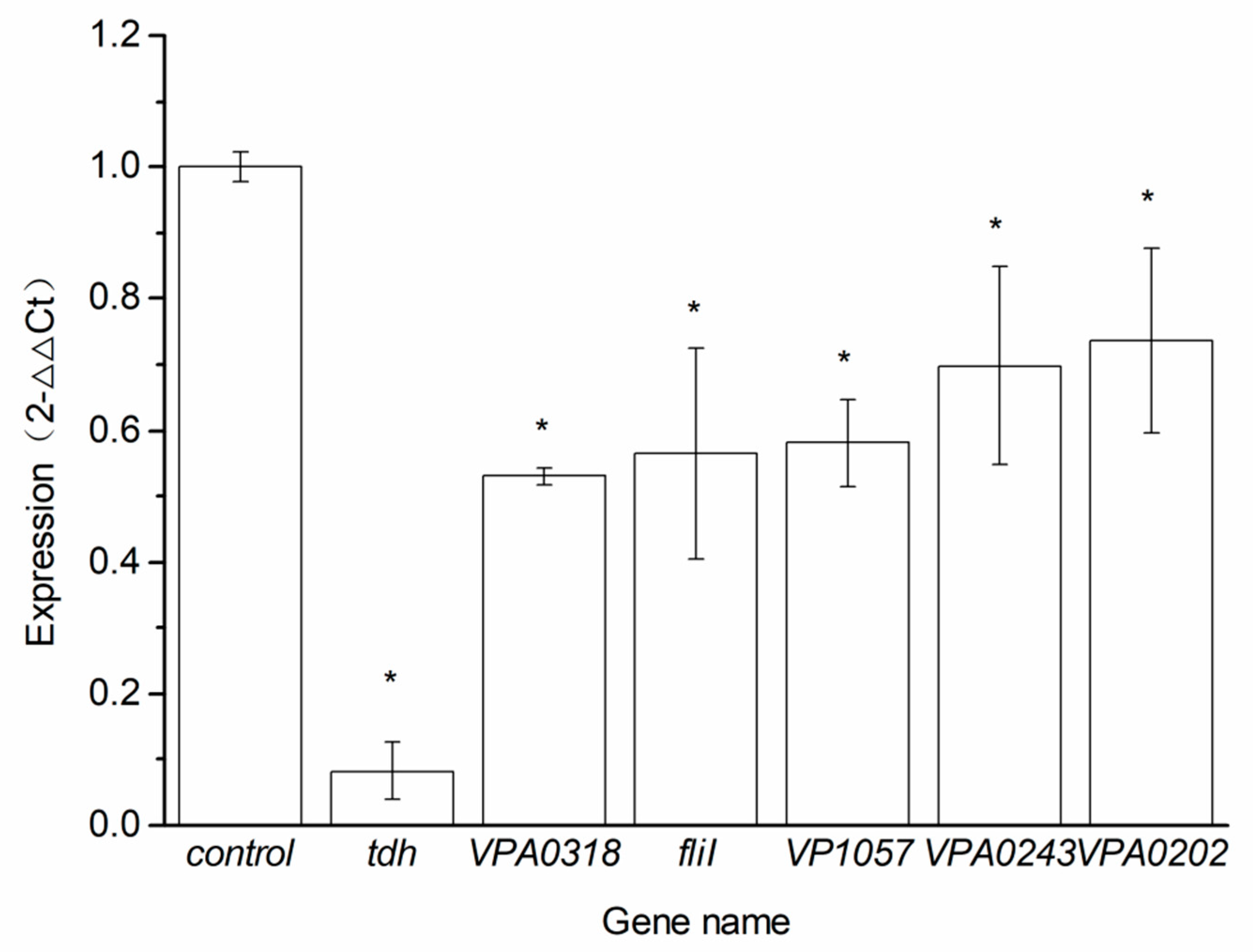
2.4. Genes Related to Virulence and Validation the RNA Sequencing Data by qRT-PCR
3. Discussion
4. Materials and Methods
4.1. Bacterial Strain and Growth Conditions
4.2. Antimicrobial Tests
4.3. RNA Extraction
4.4. Library Preparation for Strand-Specific Transcriptome Sequencing
4.5. Bioinformatic Analysis
4.6. qRT-PCR Validation of Differentially Expressed Genes
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Shen, X.; Su, Y.C. Application of grape seed extract in depuration for decontaminating Vibrio parahaemolyticus in Pacific oysters (Crassostrea gigas). Food Control 2016, 73, 601–605. [Google Scholar] [CrossRef]
- Lovell, C.R. Ecological fitness and virulence features of Vibrio parahaemolyticus in estuarine environments. Appl. Microbiol. Biotech. 2017, 101, 1781–1794. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Ma, Y.; Fu, J.; Zhao, A.; Guo, Z.; Malakar, P.K.; Pan, Y.; Zhao, Y. Effect of temperature on pathogenic and non-pathogenic Vibrio parahaemolyticus biofilm formation. Food Contr. 2016, 73, 485–491. [Google Scholar] [CrossRef]
- Su, Y.C.; Liu, C. Vibrio parahaemolyticus: A concern of seafood safety. Food Microbiol. 2007, 24, 549–558. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.Y.; Li, W.; Dong, X.; Li, M.S. Benzyl-isothiocyanate induces apoptosis and inhibits migration and invasion of hepatocellular carcinoma cells in vitro. J. Cancer 2017, 8, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Meng, X.; Li, Y.; Zhao, C.N.; Tang, G.Y.; Li, H.B. Antibacterial and antifungal activities of spices. Int. J. Mol. Sci. 2017, 18, 1283–1321. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Zhang, K.; Gao, M.; Shi, C.; Ge, C.; Qu, D.; Zhu, J.; Shi, Y.; Han, J. Inactivation of Vibrio parahaemolyticus by aqueous ozone. J. Microbiol. Biotech. 2018, 28, 1233–1246. [Google Scholar]
- Morse, M.A.; Zu, H.; Galati, A.J.; Schmidt, C.J.; Stoner, G.D. Dose-related inhibition by dietary phenethyl isothiocyanate of esophageal tumorigenesis and DNA methylation induced by N-nitrosomethylbenzylamine in rats. Cancer Lett. 1993, 72, 103–110. [Google Scholar] [CrossRef]
- Smolinska, U.; Morra, M.J.; Knudsen, G.R.; James, R.L. Isothiocyanates produced by Brassicaceae species as inhibitors of Fusarium oxysporum. Plant Disease 2003, 87, 407–412. [Google Scholar] [CrossRef]
- Dufour, V.; Stahl, M.; Baysse, C. The antibacterial properties of isothiocyanates. Microbiology 2015, 161, 229–243. [Google Scholar] [CrossRef]
- Nakano, M.; Takahashi, A.; Su, Z.; Harada, N.; Mawatari, K.; Nakaya, Y. Hfq regulates the expression of the thermostable direct hemolysin gene in Vibrio parahaemolyticus. BMC Microbiol. 2008, 8, 155. [Google Scholar] [CrossRef] [PubMed]
- Shirai, H.; Ito, H.; Hirayama, T.; Nakamoto, Y.; Nakabayashi, N.; Kumagai, K.; Nishibuchi, M. Molecular epidemiologic evidence for association of thermostable direct hemolysin (TDH) and TDH-related hemolysin of Vibrio parahaemolyticus with gastroenteritis. Infect. Immun. 1990, 58, 3568–3573. [Google Scholar] [PubMed]
- Banu, S.F.; Rubini, D.; Murugan, R.; Vadivel, V.; Gowrishankar, S.; Nithyanand, P. Exploring the antivirulent and sea food preservation efficacy of essential oil combined with DNase on Vibrio parahaemolyticus. LWT 2018, 95, 107–115. [Google Scholar] [CrossRef]
- Tan, X.J.; Qin, N.; Wu, C.Y. Transcriptome analysis of the biofilm formed by methicillinsusceptible Staphylococcus aureus. Sci. Rep. 2015, 5, 11997. [Google Scholar] [CrossRef] [PubMed]
- Pieta, L.; Escudero, F.L.G.; Jacobus, A.P. Comparative transcriptomic analysis of Listeria monocytogenes reveals upregulation of stress genes and downregulation of virulence genes in response to essential oil extracted from Baccharis psiadioides. Ann. Microbiol. 2017, 67, 479–490. [Google Scholar] [CrossRef]
- Aagesen, A.M.; Phuvasate, S.; Su, Y.C. Characterizing the adherence profiles of virulent Vibrio parahaemolyticus isolates. Microb. Ecol. 2018, 75, 152–162. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.Z.; Zhong, Y.F.; Gu, X.S. The pathogenesis, detection, and prevention of Vibrio parahaemolyticus. Front. Microbiol. 2015, 6, 144. [Google Scholar] [CrossRef] [PubMed]
- Sofrata, A.; Santangelo, E.M.; Azeem, M.; Gustafsson, A. Benzyl isothiocyanate, a major component from the roots of Salvadora persica is highly active against gram-negative bacteria. PLoS ONE 2011, 6, e23045. [Google Scholar] [CrossRef] [PubMed]
- Hong, L. Analysis of the collective food poisoning events in Shanghai from 1990 to 2000. Chinese J. Nat. Med. 2003, 5, 17–20. [Google Scholar]
- Romocastillo, M.; Andrade, A.; Espinosa, N. EscO, a functional and structural analog of the flagellar FliJ protein, is a positive regulator of EscN ATPase activity of the enteropathogenic Escherichia coli injectisome. J. Bacteriol. 2014, 196, 2227–2241. [Google Scholar] [CrossRef] [PubMed]
- Claret, L.; Calder, S.R.; Higgins, M.; Hughes, C. Oligomerization and activation of the FliI ATPase central to bacterial flagellum assembly. Mol. Microbiol. 2010, 48, 1349–1355. [Google Scholar] [CrossRef]
- Cornelis, G.R. The type III secretion injectisome. Nat. Rev. Microbiol. 2006, 4, 811–825. [Google Scholar] [CrossRef] [PubMed]
- Haiko, J.; Westerlund-Wikström, B. The role of the bacterial flagellum in adhesion and virulence. Biology 2013, 2, 1242–1267. [Google Scholar] [CrossRef] [PubMed]
- Detweiler, C.S.; Monack, D.M.; Brodsky, I.E.; Mathew, H.; Falkow, S. virK, somA and rcsC are important for systemic Salmonella enterica serovar Typhimurium infection and cationic peptide resistance. Mol. Microbiol. 2010, 48, 385–400. [Google Scholar] [CrossRef]
- Spencer, H.; Karavolos, M.H.; Bulmer, D.M.; Aldridge, P.; Chhabra, S.R.; Winzer, K.; Williams, P.; Khan, C.M.A. Genome-wide transposon mutagenesis identifies a role for host neuroendocrine stress hormones in regulating the expression of virulence genes in Salmonella. J. Bacteriol. 2010, 192, 714–724. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, R.M.; Moreira, L.M.; Ferro, J.A.; Soares, M.R.; Laia, M.L.; Varani, A.M.; de Oliveira, J.C.; Ferro, M.I. Unravelling potential virulence factor candidates in Xanthomonas citri. subsp. citri by secretome analysis. Peer J. 2016, 4, e1734. [Google Scholar] [CrossRef]
- Gabriela, T.P.; Lucia, C.D.A.; Humberto, L.M.; Ken, T.; Fernando, N.G. VirK is a periplasmic protein required for efficient secretion of plasmid-encoded toxin from enteroaggregative Escherichia coli. Infect. Immun. 2012, 80, 2276–2285. [Google Scholar]
- Abdelhamed, H.; Lu, J.; Lawrence, M.L.; Karsi, A. Involvement of tolQ and tolR genes in Edwardsiella ictaluri virulence. Microb. Pathogenesis 2016, 100, 90–94. [Google Scholar] [CrossRef]
- Heilpern, A.J.; Waldor, M.K. CTXphi infection of Vibrio cholerae requires the tolQRA gene products. J. Bacteriol. 2000, 182, 1739–1747. [Google Scholar] [CrossRef]
- Bowe, F.; Lipps, C.J.; Tsolis, R.M.; Groisman, E.; Heffron, F.; Kusters, J.G. At least four percent of the Salmonella typhimurium genome is required for fatal infection of mice. Infect. Immun. 1998, 66, 3372–3377. [Google Scholar]
- Judith, H.; Jr, R.J.; Meganm, T.; Jennifere, A.; Hshaw, W. Bacterial peptidoglycan-associated lipoprotein is released into the bloodstream in gram-negative sepsis and causes inflammation and death in mice. J. Biol. Chem. 2002, 277, 14274–14280. [Google Scholar]
- Tamayo, R.; Ryan, S.S.; Mccoy, A.J. Identification and genetic characterization of PmrA-regulated genes and genes involved in polymyxin B resistance in Salmonella enterica serovar typhimurium. Infect. Immun. 2002, 70, 6770–6778. [Google Scholar] [CrossRef] [PubMed]
- Paterson, G.K.; Northen, H.; Cone, D.B.; Willers, C.; Peters, S.E.; Maskell, D.J. Deletion of tolA in Salmonella Typhimurium generates an attenuated strain with vaccine potential. Microbiology 2009, 155, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.K.; Mccarter, L.L. ScrG, a GGDEF-EAL protein, participates in regulating swarming and sticking in Vibrio parahaemolyticus. J. Bacteriol. 2007, 189, 4094–4107. [Google Scholar] [CrossRef] [PubMed]
- Lim, B.; Beyhan, S.; Yildiz, F.H. Regulation of Vibrio polysaccharide synthesis and virulence factor production by CdgC, a GGDEF-EAL domain protein, in Vibrio cholerae. J. Bacteriol. 2007, 189, 717–729. [Google Scholar] [CrossRef] [PubMed]
- Xiong, X.P.; Wang, C.; Ye, M.Z. Differentially expressed outer membrane proteins of Vibrio alginolyticusin response to six types of antibiotics. Mar. Biotech. 2010, 12, 686–695. [Google Scholar] [CrossRef]
- KIM, M.S.; JIN, J.W.; JUNG, S.H. Genetic variations of outer membrane protein genes of Vibrio harveyi isolated in Korea and immunogenicity of OmpW in olive flounder, Paralichthys olivaceus. J. Fish. Mar. Sci. Educ. 2015, 27, 1508–1521. [Google Scholar]
- Taylor, R.K.; Manoil, C.; Mekalanos, J.J. Broad-host-range vectors for delivery of TnphoA: Use in genetic analysis of secreted virulence determinants of Vibrio cholerae. J. Bacteriol. 1989, 171, 1870–1878. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |

| Gene | Primer | Sequence (5′→3′) |
|---|---|---|
| 16S rRNA | 16S rRNA-F | TATCCTTGTTTGCCAGCGAG |
| 16S rRNA-R | CTACGACGCACTTTTTGGGA | |
| tdh | tdh-F | GGCATTTGGATGACCGAAGTA |
| tdh-R | CTGACCAATCGCAACCACTTC | |
| VPA0318 | VPA0318-F | AGGTTACTTAGCGGGTGCG |
| VPA0318-R | TTCACGGTCTTTGATGCC | |
| fliI | fliI-F | TGCGGAACCCATCAACCC |
| fliI-R | CGTCCGTCTTCGCCCAAA | |
| VP1057 | VP1057-F | CGGTTCAATCAGCCCATAC |
| VP1057-R | AACGCTTCTGCGATACCTG | |
| VPA0243 | VPA0243-F | AACGCTTCTGCGATACCTG |
| VPA0243-R | TTGCCATAGTGCGTCGTAGTCG | |
| VPA0202 | VPA0202-F | CGAAGAAGTGATGGTGGTG |
| VPA0202-R | CTCGCATTGGTGAGTTGACG |
| Sample Name | Raw Reads | Clean Reads | Clean Bases (Gb) | Error (%) | Q20 (%) | Q30 (%) | GC (%) |
|---|---|---|---|---|---|---|---|
| C_BITC1 | 11,409,316 | 10,480,246 | 1.57 | 0.02 | 97.13 | 92.51 | 48.29 |
| C_BITC2 | 12,152,830 | 11,176,478 | 1.68 | 0.02 | 96.93 | 92.11 | 48.39 |
| C_BITC3 | 11,803,196 | 11,105,878 | 1.67 | 0.02 | 97.00 | 92.28 | 48.35 |
| Q_BITC1 | 12,650,370 | 11,915,336 | 1.79 | 0.02 | 97.06 | 92.39 | 48.20 |
| Q_BITC2 | 9,990,268 | 9,170,324 | 1.38 | 0.02 | 97.05 | 92.36 | 48.01 |
| Q_BITC3 | 12,204,690 | 11,470,672 | 1.72 | 0.02 | 97.14 | 92.53 | 48.08 |
| Gene_ID | Gene Name | log2 Fold Change (Q_BITC vs. C_BITC) | Pval (Q_BITC vs. C_BITC) | Padj (Q_BITC vs. C_BITC) | Significant (Q_BITC vs. C_BITC) |
|---|---|---|---|---|---|
| VPA1509 | tdh | −0.32858 | 0.04469 | 0.62131 | DOWN |
| VPA0318 | - | −0.95983 | 8.44E−10 | 7.18E−07 | DOWN |
| VP2246 | fliI | −0.3946 | 0.029624 | 0.57503 | DOWN |
| VP1057 | - | −0.38624 | 0.049817 | 0.63785 | DOWN |
| VPA0243 | - | −0.49488 | 0.0045398 | 0.19316 | DOWN |
| VPA0202 | - | −0.4573 | 0.030909 | 0.58184 | DOWN |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, J.; Hou, H.-M.; Wu, H.-Y.; Li, K.-X.; Wang, Y.; Zhou, Q.-Q.; Zhang, G.-L. Transcriptomic Analysis of Vibrio parahaemolyticus Reveals Different Virulence Gene Expression in Response to Benzyl Isothiocyanate. Molecules 2019, 24, 761. https://doi.org/10.3390/molecules24040761
Song J, Hou H-M, Wu H-Y, Li K-X, Wang Y, Zhou Q-Q, Zhang G-L. Transcriptomic Analysis of Vibrio parahaemolyticus Reveals Different Virulence Gene Expression in Response to Benzyl Isothiocyanate. Molecules. 2019; 24(4):761. https://doi.org/10.3390/molecules24040761
Chicago/Turabian StyleSong, Jie, Hong-Man Hou, Hong-Yan Wu, Ke-Xin Li, Yan Wang, Qian-Qian Zhou, and Gong-Liang Zhang. 2019. "Transcriptomic Analysis of Vibrio parahaemolyticus Reveals Different Virulence Gene Expression in Response to Benzyl Isothiocyanate" Molecules 24, no. 4: 761. https://doi.org/10.3390/molecules24040761
APA StyleSong, J., Hou, H.-M., Wu, H.-Y., Li, K.-X., Wang, Y., Zhou, Q.-Q., & Zhang, G.-L. (2019). Transcriptomic Analysis of Vibrio parahaemolyticus Reveals Different Virulence Gene Expression in Response to Benzyl Isothiocyanate. Molecules, 24(4), 761. https://doi.org/10.3390/molecules24040761





