Screening of Suitable Ionic Liquids as Green Solvents for Extraction of Eicosapentaenoic Acid (EPA) from Microalgae Biomass Using COSMO-RS Model
Abstract
:1. Introduction
2. Results and Discussion
Investigation of the Effect of Cations Alkyl Chain Length on EPA Extraction
3. Methodology
3.1. Computational Details of COSMO-RS
3.2. Data Collection and Screening Process
3.3. COSMO-RS Assumptions
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chowdhury, R.; Stevens, S.; Gorman, D.; Pan, A.; Warnakula, S.; Chowdhury, S.; Ward, H.; Johnson, L.; Crowe, F.; Hu, F.B.; et al. Association between fish consumption, long chain omega 3 fatty acids, and risk of cerebrovascular disease: Systematic review and meta-analysis. BMJ 2012, 345, e6698. [Google Scholar] [CrossRef] [PubMed]
- Fabian, C.J.; Kimler, B.F.; Hursting, S.D. Omega-3 fatty acids for breast cancer prevention and survivorship. Breast Cancer Res. 2015. [Google Scholar] [CrossRef] [PubMed]
- Lazo, M.; Manzano-patin, A.; Cortez-lugo, M.; Romieu, I.; Te, M.M.; Hernandez-Avila, M.; Holguin, F.; Julien, P.; Be, M.C. Omega-3 Fatty Acid Prevents Heart Rate Variability Reductions Associated with Particulate Matter. Am. J. Respir. Crit. Care Med. 2005, 132, 1534–1540. [Google Scholar]
- Marik, P.E.; Varon, J. Omega-3 dietary supplements and the risk of cardiovascular events: A systematic review. Clin. Cardiol. 2009, 32, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Rodríguez, N.; Beltrán, S.; Jaime, I.; de Diego, S.M.; Sanz, M.T.; Carballido, J.R. Production of omega-3 polyunsaturated fatty acid concentrates: A review. Innov. Food Sci. Emerg. Technol. 2010, 11, 1–12. [Google Scholar] [CrossRef]
- Ruxton, C.H.S.; Reed, S.C.; Simpson, M.J.A.; Millington, K.J. The health benefits of omega-3 polyunsaturated fatty acids: A review of the evidence. J. Hum. Nutr. Diet. 2004, 17, 449–459. [Google Scholar] [CrossRef]
- Nichols, P.D.; McManus, A.; Krail, K.; Sinclair, A.J.; Miller, M. Recent advances in omega-3: Health benefits, Sources, Products and bioavailability. Nutrients 2014, 6, 3727–3733. [Google Scholar] [CrossRef]
- Ozogul, Y.; Şimşek, A.; Balikçi, E.; Kenar, M. The effects of extraction methods on the contents of fatty acids, especially EPA and DHA in marine lipids. Int. J. Food Sci. Nutr. 2012, 63, 326–331. [Google Scholar] [CrossRef]
- Shahidi, F.; Ambigaipalan, P. Omega-3 Polyunsaturated Fatty Acids and Their Health Benefits. Annu. Rev. Food Sci. Technol. 2018, 9, 345–381. [Google Scholar] [CrossRef]
- Adarme-vega, T.C.; Lim, D.K.Y.; Timmins, M.; Vernen, F.; Li, Y.; Schenk, P.M. Microalgal biofactories: A promising approach towards sustainable omega-3 fatty acid production. Microb. Cell Fact. 2012, 11, 96. [Google Scholar] [CrossRef]
- Davidson, M.H.; Burns, J.H.; Subbaiah, P.V.; Conn, M.E.; Drennan, K.B. Marine oil capsule therapy for the treatment of hyperlipidemia. Arch. Intern. Med. 1991, 151, 1732–1740. [Google Scholar] [CrossRef] [PubMed]
- Swanson, D.; Block, R.; Mousa, S.A. Omega-3 fatty acids EPA and DHA: Health benefits throughout life. Adv. Nutr. 2012, 3, 1–7. [Google Scholar] [CrossRef]
- Conchillo, A.; Valencia, I.; Puente, A.; Astiasarán, D.A.I. Componentes Funcionales en Aceites de pescado y de Alga. 2006. Available online: http://www.redalyc.org/articulo.oa?id=309226444013 (accessed on 23 November 2005).
- Nelson, W.M. Chapter 3 Are Ionic Liquids Green Solvents? Ionic Liquids as Greener Solvents. ACS Symp. Ser. 2002, 9, 30–41. [Google Scholar]
- Berthod, A.; Ruiz-Ángel, M.J.; Carda-Broch, S. ARTICLE IN PRESS G Model Recent advances on ionic liquid uses in separation techniques. J. Chromatogr. A 2017, 1559, 1–15. [Google Scholar]
- Shiddiky, M.J.A.; Torriero, A.A.J. Application of ionic liquids in electrochemical sensing systems. Biosens. Bioelectron. 2011, 26, 1775–1787. [Google Scholar] [CrossRef]
- Keskin, S.; Kayrak-Talay, D.; Akman, U.; Hortaçsu, Ö. A review of ionic liquids towards supercritical fluid applications. J. Supercrit. Fluids 2007, 43, 150–180. [Google Scholar] [CrossRef]
- Biczak, R.; Pawłowska, B.; Bałczewski, P.; Rychter, P. The role of the anion in the toxicity of imidazolium ionic liquids. J. Hazard. Mater. 2014, 274, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Muppaneni, T.; Sun, Y.; Reddy, H.K.; Fu, J.; Lu, X.; Deng, S. Microwave-assisted extraction of lipids from microalgae using an ionic liquid solvent [BMIM][HSO4]. Fuel 2016, 178, 49–55. [Google Scholar] [CrossRef]
- Kim, Y.; Choi, Y.; Park, J.; Lee, S.; Yang, Y.; Joo, H.; Park, T.; Hwan, Y.; Hyun, S. Bioresource Technology Ionic liquid-mediated extraction of lipids from algal biomass. Bioresour. Technol. 2012, 109, 312–315. [Google Scholar] [CrossRef]
- Eckert, F.; Klamt, A.; Gmbh, C.; Kg, C.; Leverkusen, D. Fast Solvent Screening via Quantum Chemistry: COSMO-RS Approach. AIChE J. 2002, 48, 369–385. [Google Scholar] [CrossRef]
- Klamt, A. The COSMO and COSMO-RS solvation models. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2018, 8, e1338. [Google Scholar] [CrossRef]
- Klamt, A. Conductor-like Screening Model for Real Solvents: A New Approach to the Quantitative Calculation of Solvation Phenomena. J. Phys. Chem. 1995, 99, 2224–2235. [Google Scholar] [CrossRef]
- Rajabi, M.S.; Moniruzzaman, M.; Mahmood, H.; Sivapragasam, M.; Bustam, M.A. Extraction of β-carotene from organic phase using ammonium based ionic liquids. J. Mol. Liq. 2017, 227, 15–20. [Google Scholar] [CrossRef]
- Rashid, Z.; Wilfred, C.D.; Gnanasundaram, N.; Arunagiri, A.; Murugesan, T. Screening of ionic liquids as green oilfield solvents for the potential removal of asphaltene from simulated oil: COSMO-RS model approach. J. Mol. Liq. 2018, 255, 492–503. [Google Scholar] [CrossRef]
- Xing, H.; Zhang, X.; Yang, Q.; Liu, R.; Bao, Z.; Su, B.; Yang, Y.; Ren, Q. Separation of long chain fatty acids with different number of unsaturated bonds by fractional extraction: Experimental and COSMO-RS study. Food Chem. 2014, 143, 411–417. [Google Scholar] [CrossRef]
- Han, J.; Dai, C.; Yu, G.; Lei, Z. Parameterization of COSMO-RS model for ionic liquids. Green Energy Environ. 2018, 3, 247–265. [Google Scholar] [CrossRef]
- Durand, M.; Molinier, V.; Kunz, W.; Aubry, J. Classification of Organic Solvents Revisited by Using the COSMO-RS Approach. Chem. Eur. J. 2011, 5155–5164. [Google Scholar] [CrossRef]
- Zhou, T.; Qi, Z.; Sundmacher, K. Model-based method for the screening of solvents for chemical reactions. Chem. Eng. Sci. 2014, 115, 177–185. [Google Scholar] [CrossRef]
- Hassanali, A.A.; Zhong, D.; Singer, S.J. An AIMD study of CPD repair mechanism in water: Role of solvent in ring splitting. J. Phys. Chem. B 2011, 115, 3860–3871. [Google Scholar] [CrossRef]
- Jong, K.; Grisanti, L.; Hassanali, A. Hydrogen bond networks and hydrophobic effects in the amyloid β30–35 chain in water: A molecular dynamics study. J. Chem. Inf. Model. 2017, 57, 1548–1562. [Google Scholar] [CrossRef]
- Ufimtsev, I.S.; Luehr, N.; Martinez, T.J. Charge transfer and polarization in solvated proteins from ab initio molecular dynamics. J. Phys. Chem. Lett. 2011, 2, 1789–1793. [Google Scholar] [CrossRef]
- van der Vaart, A.; Merz, K.M. The role of polarization and charge transfer in the solvation of biomolecules. J. Am. Chem. Soc. 1999, 121, 9182–9190. [Google Scholar] [CrossRef]
- Ansari, N.; Dandekar, R.; Caravati, S.; Sosso, G.C.; Hassanali, A. High and low density patches in simulated liquid water. J. Chem. Phys. 2018, 149, 204507. [Google Scholar] [CrossRef] [PubMed]
- Mohan, N.; Suresh, C.H. A Molecular Electrostatic Potential Analysis of Hydrogen, Halogen, and Dihydrogen Bonds. J. Phys. Chem. A 2014, 118, 1697–1705. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Lue, B.; Thomasen, K.; Meyer, A.S.; Xu, X. Predictions of flavonoid solubility in ionic liquids by COSMO-RS: Experimental verification, structural elucidation, and solvation characterization. Green Chem. 2007, 9, 1362–1373. [Google Scholar] [CrossRef]
- Man, M.S.; Abdullah, M.A.M.; Abdullah, S.B.; Yaacob, Z. Screening Cation and Anion of Ionic Liquid for Dissolution of Silicon Dioxide using COSMO-RS. Indian J. Sci. Technol. 2017, 10. [Google Scholar] [CrossRef]
- Hawker, R.R.; Haines, R.S.; Harper, J.B. Variation of the Cation of Ionic Liquids: The Effects on Their Physicochemical Properties and Reaction Outcome. Targets Heterocycl. Syst. Prop. 2015, 18, 141–213. [Google Scholar] [CrossRef]
- Shahriari, S.; Neves, C.M.S.S.; Freire, M.G.; Coutinho, J.A.P. Role of the Hofmeister series in the formation of ionic-liquid-based aqueous biphasic systems. J. Phys. Chem. B 2012, 116, 7252–7258. [Google Scholar] [CrossRef]
- Zhang, Q.G.; Wang, N.N.; Yu, Z.W. The hydrogen bonding interactions between the ionic liquid 1-Ethyl-3-methylimidazolium ethyl sulfate and water. J. Phys. Chem. B 2010, 114, 4747–4754. [Google Scholar] [CrossRef]
- Cheong, L.; Guo, Z.; Yang, Z.; Chua, S.; Xu, X. Extraction and Enrichment of n-3 Polyunsaturated Fatty Acids and Ethyl Esters through Reversible π À π Complexation with Aromatic Rings Containing Ionic Liquids. J. Agric. Food chem. 2011, 59, 8961–8967. [Google Scholar] [CrossRef]
- Fu, H.; Li, M.; Ni, R.; Lo, Y.M. Enzymatic catalysis for sustainable production of high omega-3 triglyceride oil using imidazolium-based ionic liquids. Food Sci. Nutr. 2018, 6, 2020–2027. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Pham, P.J.; Wang, T.; Pittman, C.U., Jr.; Li, T. Selective extraction and enrichment of polyunsaturated fatty acid methyl esters from fish oil by novel π-complexing sorbents. Sep. Purif. Technol. 2009, 66, 1–8. [Google Scholar] [CrossRef]
- Li, M.; Pittman, C.U., Jr.; Li, T. Extraction of polyunsaturated fatty acid methyl esters by imidazolium-based ionic liquids containing silver tetrafluoroborate—Extraction equilibrium studies. Talanta 2009, 78, 1364–1370. [Google Scholar] [CrossRef] [PubMed]
- Orawiec, M. Activity Coe ffi cients at In fi nite Dilution of Organic Solutes and Water in Tributylethylphosphonium Diethylphosphate Using Gas—Liquid Chromatography: Thermodynamic Properties of Mixtures Containing Ionic Liquids. J. Chem. Eng. Data 2016, 61, 1793–1802. [Google Scholar]
- Heintz, A.; Sergey, P.; Verevkin, D.O. Thermodynamic Properties of Mixtures Containing Ionic Liquids. 8. Activity Coefficients at Infinite Dilution of Hydrocarbons, Alcohols, Esters, and Aldehydes in 1-Hexyl-3-methylimidazolium Bis(trifluoromethylsulfonyl) Imide Using Gas—Liquid Chromatograp. Chem. Eng 2006, 51, 434–437. [Google Scholar] [CrossRef]
- Joshua, J.; Magaret, S.; Pranesh, M.; Chellappan, K.; Cecilia, W.; Mutalib, M.I.A. Separation and Puri fi cation Technology Extractive desulfurization of model fuel oil using ester functionalized imidazolium ionic liquids. Sep. Purif. Technol. 2018, 196, 115–123. [Google Scholar]
- Sobota, M.; Vrbka, P. 1-Ethyl-3-methyl-imidazolium Nitrate. J. Phys. Chem. B 2009, 113, 4323–4332. [Google Scholar] [CrossRef]
- Marciniak, A.; Letcher, T.M. Activity Coefficients at Infinite Dilution and Physicochemical Properties for Organic Solutes and Water in the Ionic Liquid 1-Ethyl-3-methylimidazolium. J. Solut. Chem. 2014, 44, 413–430. [Google Scholar]
- Gabrić, B.; Sander, A.; Bubalo, M.C.; Macut, D. Extraction of S- and N-Compounds from the Mixture of Hydrocarbons by Ionic Liquids as Selective Solvents. Sci. World J. 2013, 2013, 512953. [Google Scholar]
- Schafer, A.; Klamt, A.; Sattel, D.; Lohrenz, J.C.W.; Eckert, F. COSMO Implementation in TURBOMOLE: Extension of an efficient quantum chemical code towards liquid systems COSMO-Implementation in TURBOMOLE: Extension of an efficient quantum chemical code towards liquid systems. Phys. Chem. Chem. Phys. 2000, 2, 2187–2193. [Google Scholar] [CrossRef]
- Klamt, A.; Jonas, V.; Bu, T.; Lohrenz, J.C.W. Le Refinement and Parametrization of COSMO-RS. J. Phys. Chem. A 1998, 5639, 5074–5085. [Google Scholar] [CrossRef]
- Lin, S.; Sandler, S.I. A Priori Phase Equilibrium Prediction from a Segment Contribution Solvation Model. Ind. Eng. Chem. Res. 2002, 41, 899–913. [Google Scholar] [CrossRef]
- Mullins, E.; Oldland, R.; Liu, Y.A.; Wang, S.; Sandler, S.I.; Chen, C.C.; Zwolak, M.; Seavey, K.C. Sigma-profile database for using COSMO-based thermodynamic methods. Ind. Eng. Chem. Res. 2006, 45, 4389–4415. [Google Scholar] [CrossRef]
- Anantharaj, R.; Banerjee, T. COSMO-RS based predictions for the desulphurization of diesel oil using ionic liquids: Effect of cation and anion combination. Fuel Process. Technol. 2011, 92, 39–52. [Google Scholar] [CrossRef]
- Zhou, T.; Chen, L.; Ye, Y.; Chen, L.; Qi, Z.; Freund, H.; Sundmacher, K. An overview of mutual solubility of ionic liquids and water predicted by COSMO-RS. Ind. Eng. Chem. Res. 2012, 51, 6256–6264. [Google Scholar] [CrossRef]
- Tumba, K.A. Infinite Dilution Activity Coefficient Measurements of Organic Solutes in Fluorinated Ionic Liquids by Gas-Liquid Chromatography and the Inert Gas Kaniki Armel Tumba. Ph.D. Thesis, Mangosuthu University, Umlazi, South Africa, 2010. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |
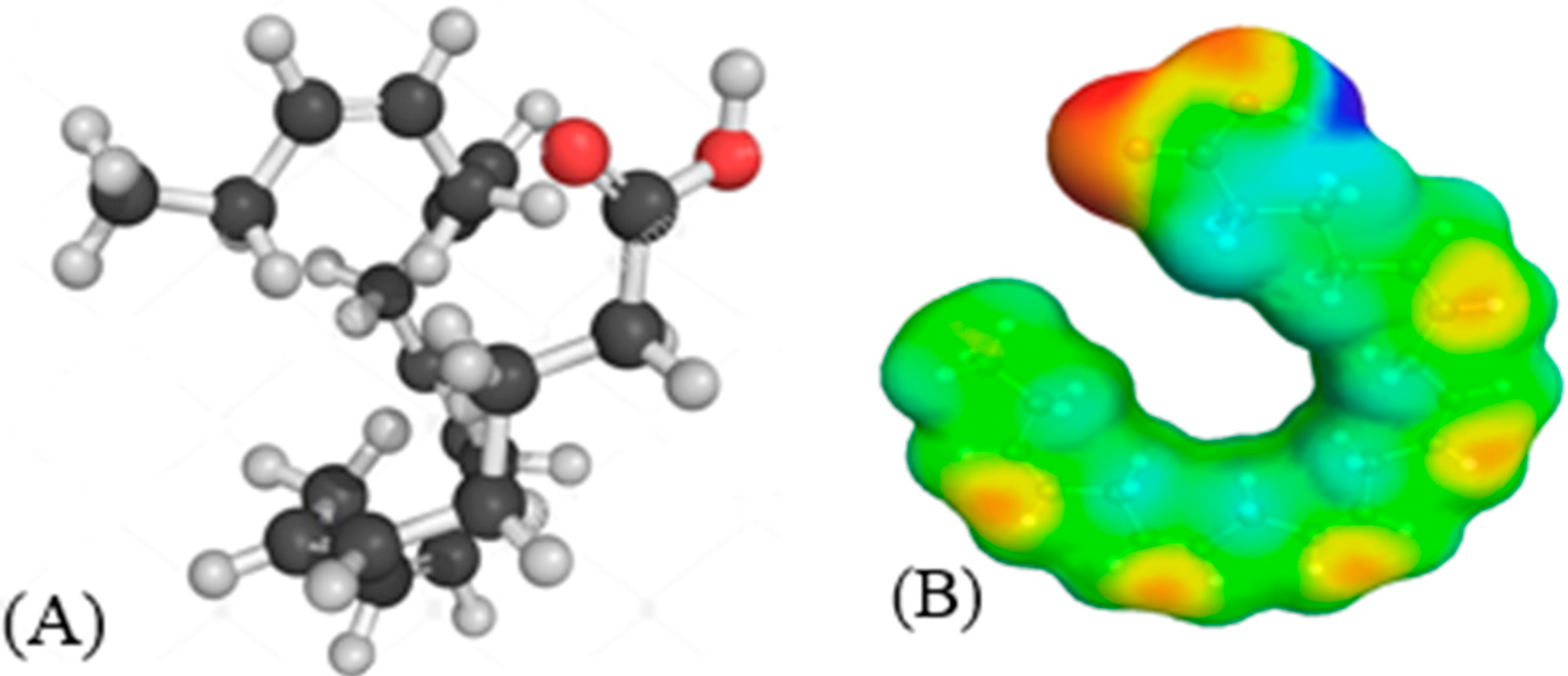
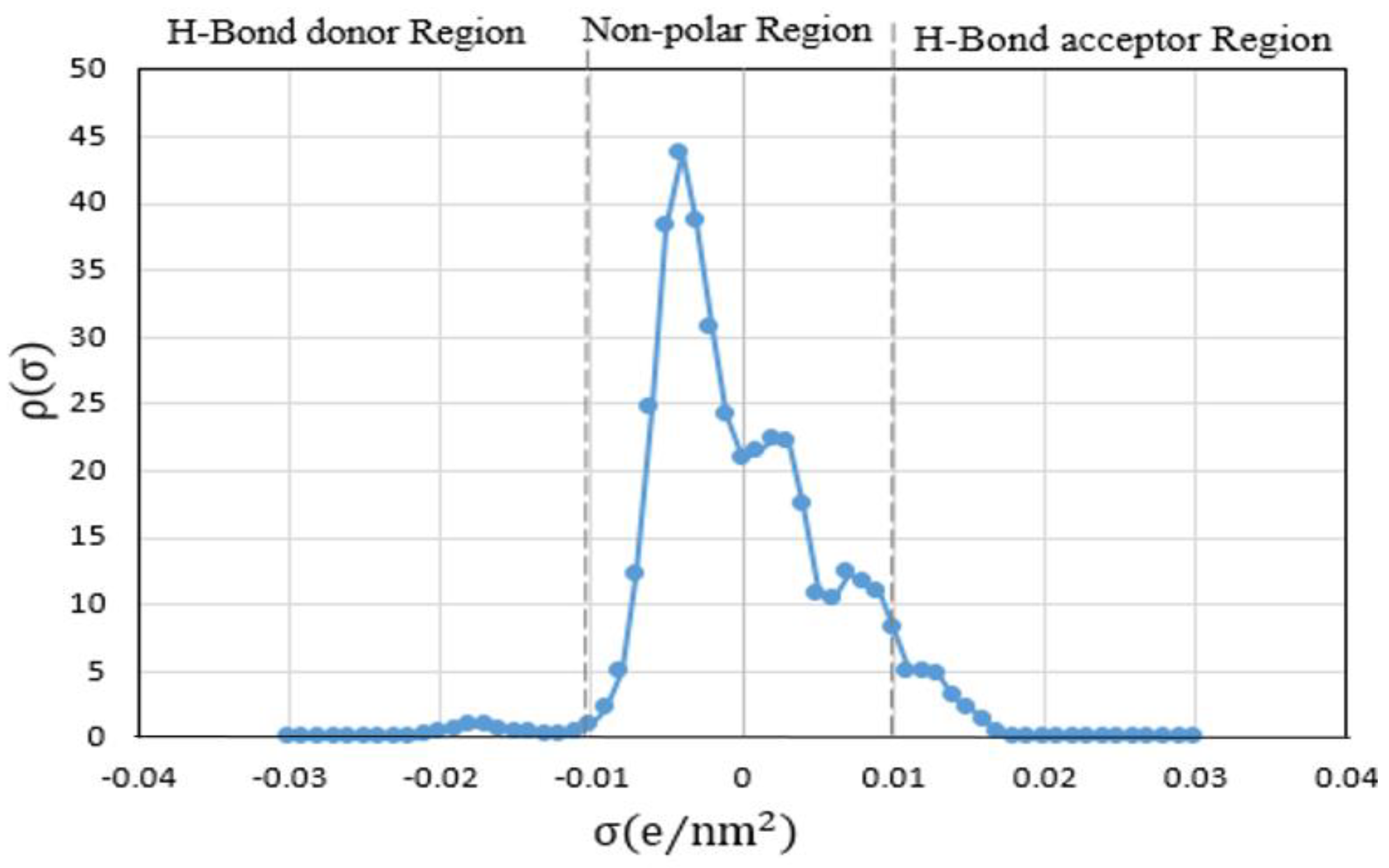
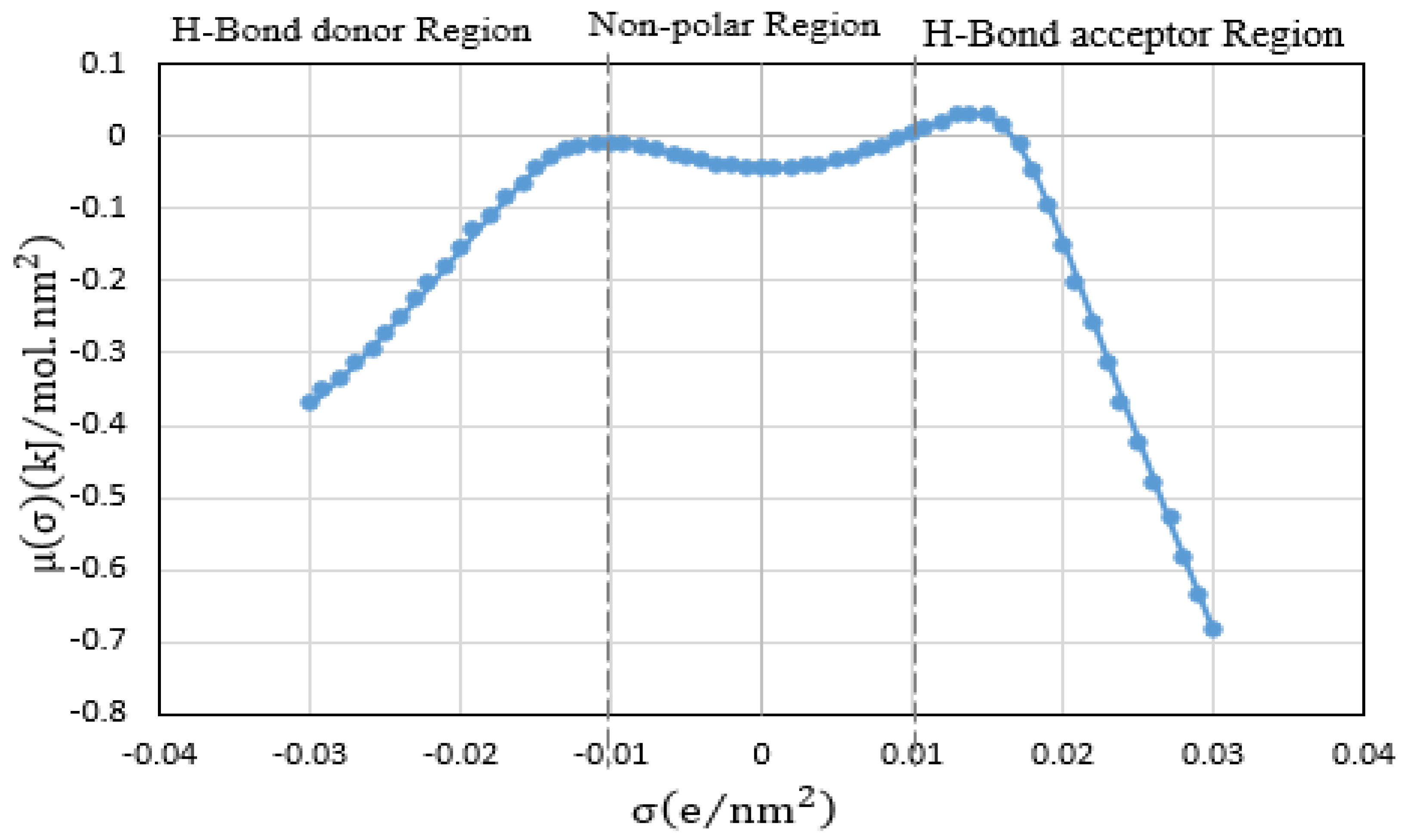

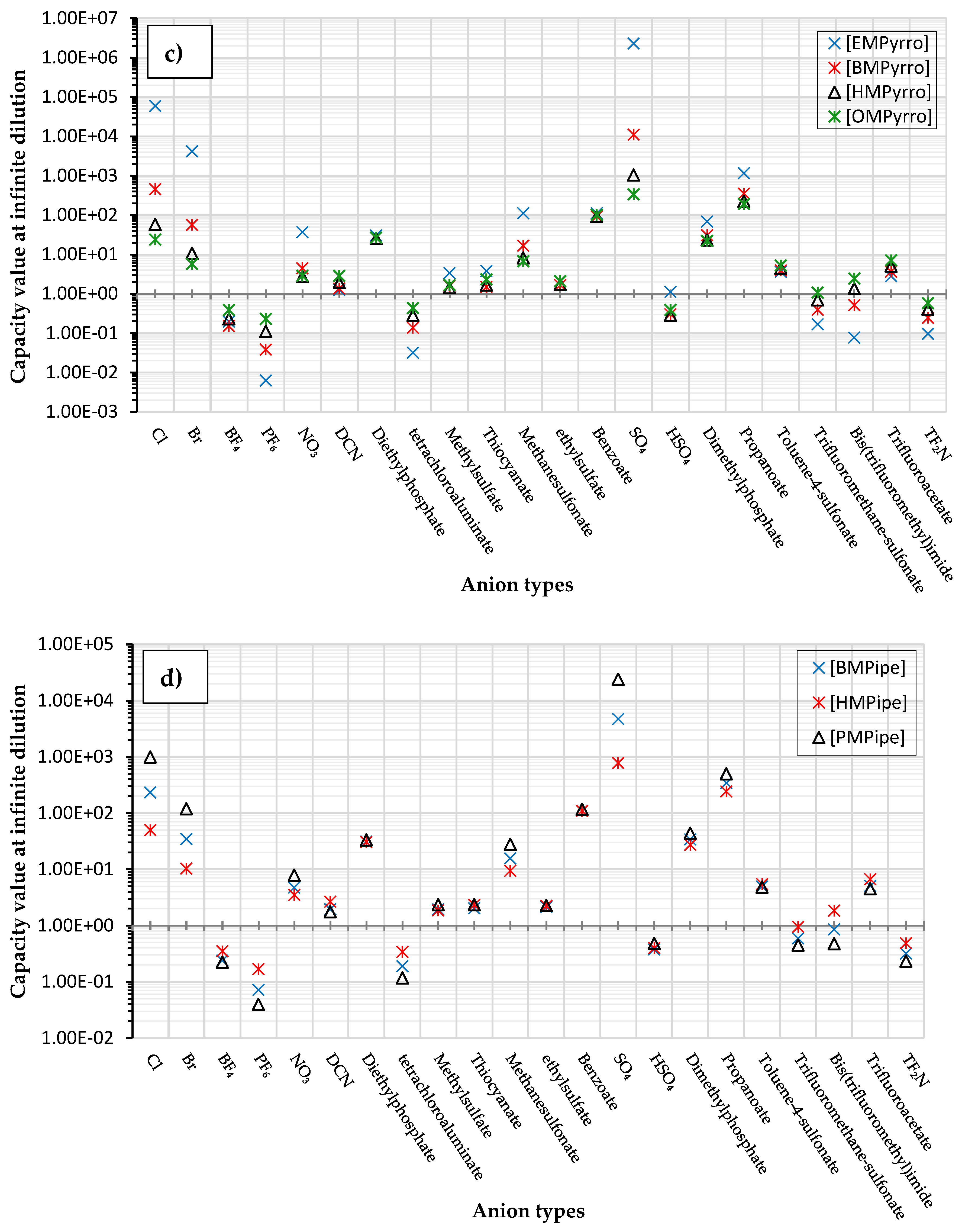


| # | Shortlisted ILs | Infinite Dilution Activity Coefficient at 298.15 K | Infinite Dilution Capacity at 298.15 K |
|---|---|---|---|
| 1 | [TMAm]SO4 | 1.00 × 10−11 | 9.97 × 1010 |
| 2 | [TMAm]Cl | 9.52 × 10−10 | 1.05 × 109 |
| 3 | [TMAm]Br | 3.61 × 10−8 | 27677690 |
| 4 | [EMPyrro]SO4 | 4.40 × 10−7 | 2273253 |
| 5 | [EMPyrro]Cl | 1.68 × 10−5 | 59473.56 |
| 6 | [EMIM]SO4 | 1.87 × 10−5 | 53360.83 |
| 7 | [MPPIP]SO4 | 4.17 × 10−5 | 23995.28 |
| 8 | [EmPyr]SO4 | 7.10 × 10−5 | 14084.39 |
| 9 | [BMPyrro]SO4 | 9.03 × 10−5 | 11073.19 |
| 10 | [BPPIP]SO4 | 2.13 × 10−4 | 4689.32 |
| 11 | [EMPyrro]Br | 2.41 × 10−4 | 4147.65 |
| 12 | [EMIM]Cl | 6.61 × 10−4 | 1513.22 |
| 13 | [TMAm]propanoate | 6.97 × 10−4 | 1434.47 |
| 14 | [TMAm]NO3 | 8.39 × 10−4 | 1191.69 |
| 15 | [EMPyrro]propanoate | 8.67 × 10−4 | 1153.16 |
| 16 | [HMPyrro]SO4 | 9.62 × 10−4 | 1039.60 |
| 17 | [MPPIP]Cl | 1.01 × 10−3 | 991.30 |
| 18 | [BMIM]SO4 | 1.28 × 10−3 | 781.77 |
| 19 | [HPPIP]SO4 | 1.28 × 10−3 | 781.45 |
| 20 | [BmPyr]SO4 | 1.37 × 10−3 | 727.47 |
| 21 | [EmPyr]Cl | 1.74 × 10−3 | 575.71 |
| Shorthand Sign | Synthetic Name | Trivial Name | Formula | Chemical Structure |
|---|---|---|---|---|
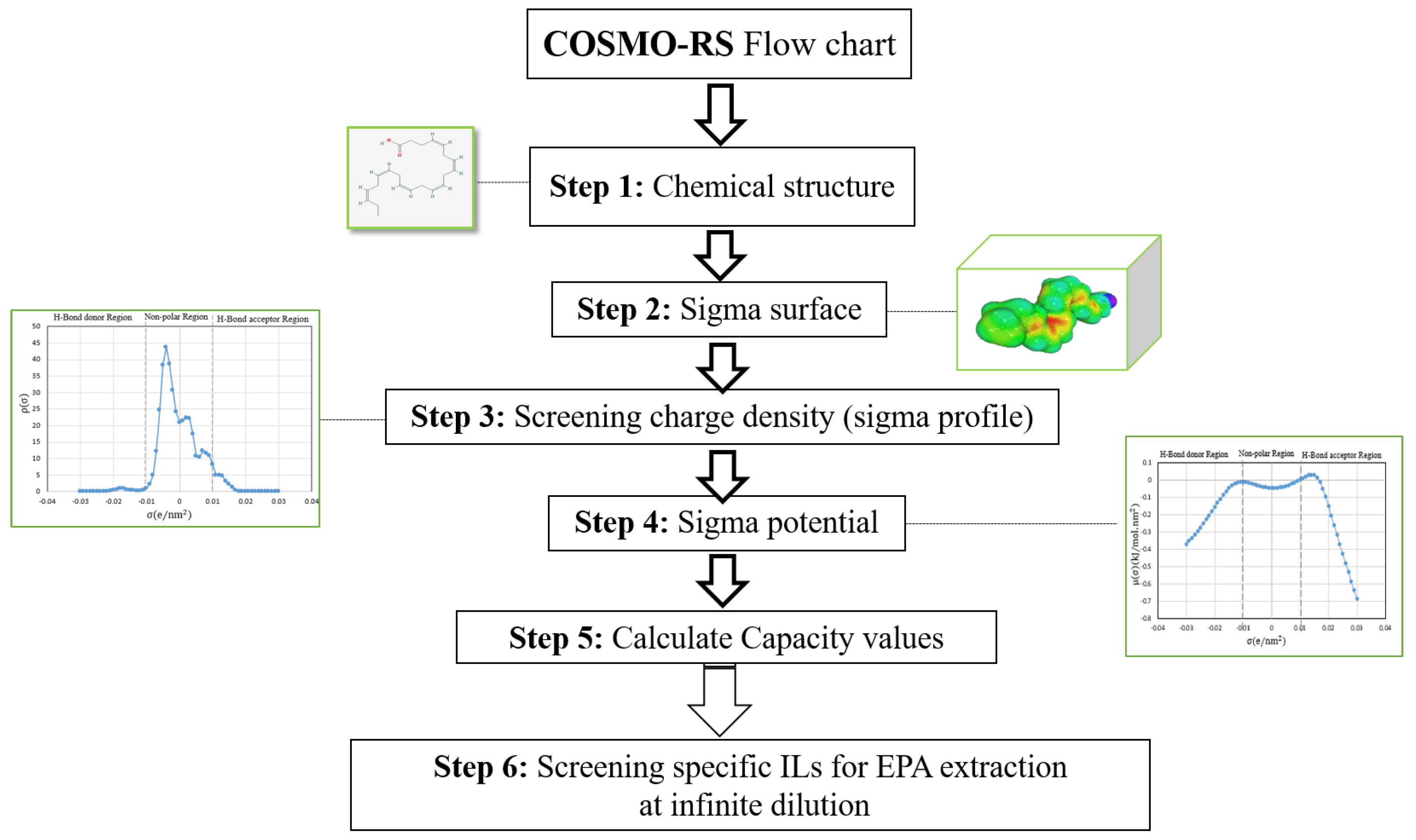
| 20:5 | Eicosapentanoic acid | EPA | C20H30O2 | 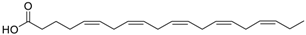 |
| No. | Anions/Abbreviations | Chemical Structure |
|---|---|---|
| 1 | Chloride [Cl] | Cl− |
| 2 | Bromide [Br] | Br− |
| 3 | Tetrafluoroborate [BF4] |  |
| 4 | Hexafluorophosphate [PF6] | 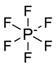 |
| 5 | Nitrate [NO3] |  |
| 6 | Dicyanamide [DCN] |  |
| 7 | Diethylphosphate [C4H10O4P] |  |
| 8 | Tetrachloro aluminate [AlCl4] |  |
| 9 | Methyl sulfate [CH4O4S] |  |
| 10 | Thiocyanate [SCN] |  |
| 11 | Methane sulfonate [C2H6O3S] |  |
| 12 | Ethyl sulfate [C2H6O4S] |  |
| 13 | Benzoate [C7H5O2] |  |
| 14 | Sulfate [SO4] | 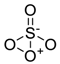 |
| 15 | Hydrogen sulphate [HSO4] |  |
| 16 | Dimethyl phosphate [C2H7O4P] |  |
| 17 | Propanoate [C3H5O2] |  |
| 18 | Toluene-4-Sulfonate [C7H7O3S] |  |
| 19 | Tri fluoro methane-Sulfonate [CF3SO3] |  |
| 20 | Bis(Trifluoromethyl)Imide [NHC2F6] |  |
| 21 | Trifluoroacetate [CF3CO2] |  |
| 22 | Bis(trifluoromethylsulfonyl) imide [TF2N] |  |
| No. | Cations/Abbreviations | Chemical Structures |
|---|---|---|
| 1 | 1-ethyl-3-methyl imidazolium [EMIM] |  |
| 2 | 1-butyl-3-methyl imidazolium [BMIM] |  |
| 3 | 1-hexyl-3-methyl imidazolium [HMIM] |  |
| 4 | 1-octyl-3-methyl imidazolium [OMIM] |  |
| 5 | 1-ethyl-3-methyl pyridinium [EMPyr] |  |
| 6 | 1-butyl-3-methyl pyridinium [BMPyr] |  |
| 7 | 1-hexyl-3-methyl pyridinium [HMPyr] |  |
| 8 | 1-octhyl-3-methyl pyridinium [OMPyr] |  |
| 9 | 1-butyl-1-methyl pyrrolidinium [BMPyrro] |  |
| 10 | 1-ethyl-1-methyl pyrrolidinium [EMPyrro] |  |
| 11 | 1-hexyl-1-methyl pyrrolidinium [HMPyrro] |  |
| 12 | 1-methyl-1-octyl pyrrolidinium [MOPyrro] |  |
| 13 | 1-butyl-1-methyl piperidinium [BMPIP] |  |
| 14 | 1-hexyl-1-methyl piperidinium [HMPIP] |  |
| 15 | 1-methyl-1-propyl piperidinium [MPPIP] |  |
| 16 | Tetra-methyl ammonium [TMAm] |  |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rezaei Motlagh, S.; Harun, R.; Awang Biak, D.R.; Hussain, S.A.; Wan Ab Karim Ghani, W.A.; Khezri, R.; Wilfred, C.D.; Elgharbawy, A.A.M. Screening of Suitable Ionic Liquids as Green Solvents for Extraction of Eicosapentaenoic Acid (EPA) from Microalgae Biomass Using COSMO-RS Model. Molecules 2019, 24, 713. https://doi.org/10.3390/molecules24040713
Rezaei Motlagh S, Harun R, Awang Biak DR, Hussain SA, Wan Ab Karim Ghani WA, Khezri R, Wilfred CD, Elgharbawy AAM. Screening of Suitable Ionic Liquids as Green Solvents for Extraction of Eicosapentaenoic Acid (EPA) from Microalgae Biomass Using COSMO-RS Model. Molecules. 2019; 24(4):713. https://doi.org/10.3390/molecules24040713
Chicago/Turabian StyleRezaei Motlagh, Shiva, Razif Harun, Dayang Radiah Awang Biak, Siti Aslina Hussain, Wan Azlina Wan Ab Karim Ghani, Ramin Khezri, Cecilia Devi Wilfred, and Amal A. M. Elgharbawy. 2019. "Screening of Suitable Ionic Liquids as Green Solvents for Extraction of Eicosapentaenoic Acid (EPA) from Microalgae Biomass Using COSMO-RS Model" Molecules 24, no. 4: 713. https://doi.org/10.3390/molecules24040713
APA StyleRezaei Motlagh, S., Harun, R., Awang Biak, D. R., Hussain, S. A., Wan Ab Karim Ghani, W. A., Khezri, R., Wilfred, C. D., & Elgharbawy, A. A. M. (2019). Screening of Suitable Ionic Liquids as Green Solvents for Extraction of Eicosapentaenoic Acid (EPA) from Microalgae Biomass Using COSMO-RS Model. Molecules, 24(4), 713. https://doi.org/10.3390/molecules24040713








