Hesperidin as a Neuroprotective Agent: A Review of Animal and Clinical Evidence
Abstract
1. Introduction
2. Natural Products as the Future of Neuroprotective Drugs
3. Hesperidin, Chemistry, Sources, and Therapeutic Effects
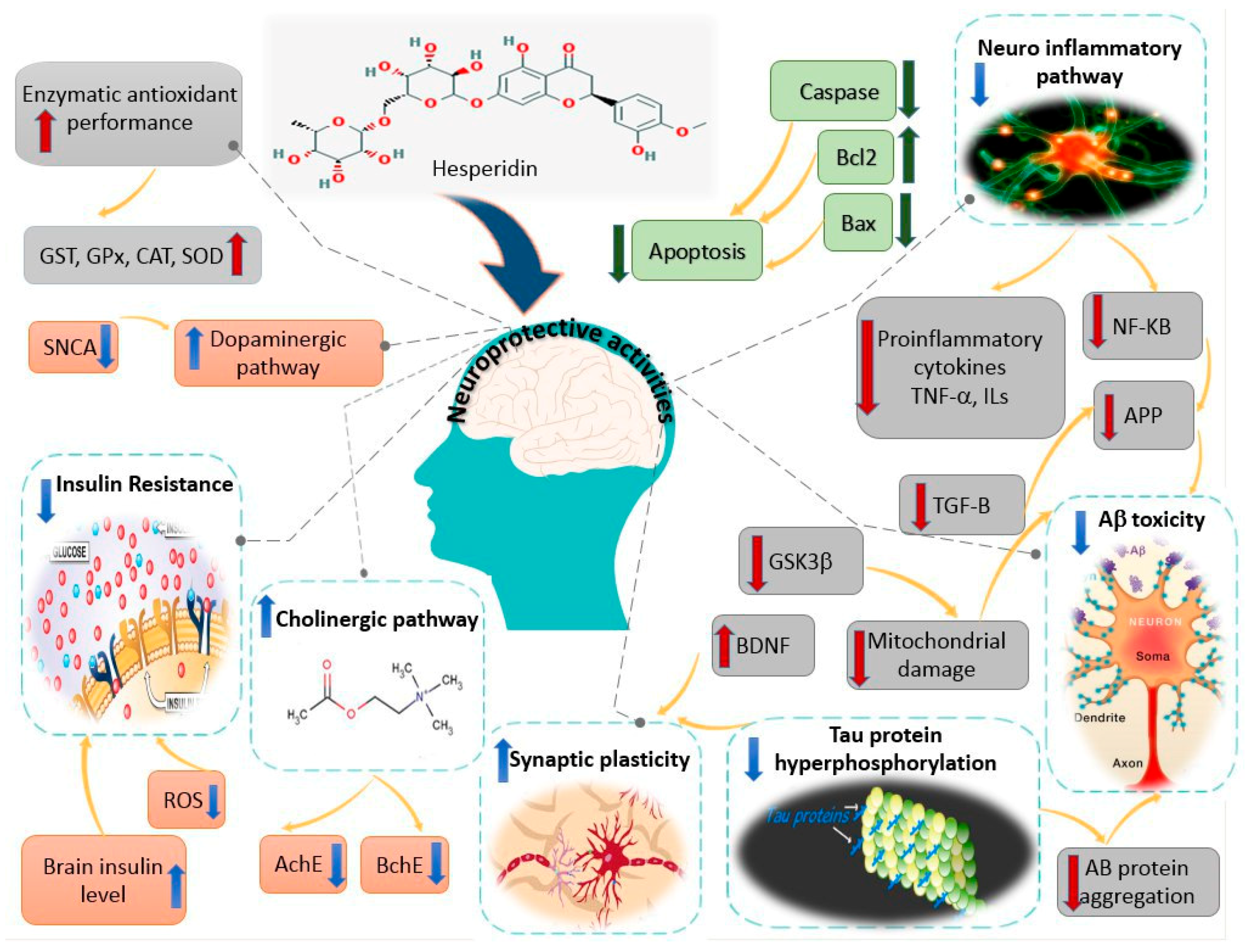
4. Hesperidin and Neuroprotection: Mechanisms of Action Based on Animal Studies (In Vivo Studies)
4.1. Parkinson’s Diseases (PD)
4.2. Dementia and Alzheimer’s Diseases
4.3. Huntington’s Disease
4.4. Multiple Sclerosis
4.5. Diabetes Mellitus Associated Neurotoxicity
5. Clinical Evidence for Neuroprotective Potential of Hesperidin (Human Studies)
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gao, H.-M.; Hong, J.-S. Why neurodegenerative diseases are progressive: Uncontrolled inflammation drives disease progression. Trend. Immunol. 2008, 29, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Uttara, B.; Singh, A.V.; Zamboni, P.; Mahajan, R. Oxidative stress and neurodegenerative diseases: A review of upstream and downstream antioxidant therapeutic options. Cur. Neuropharmacolo. 2009, 7, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Dominguez, C. Neurodegenerative Diseases; Springer Science & Business Media: Berlin, Germany, 2010; Volume 6. [Google Scholar]
- Kovacs, G.G. Concepts and classification of neurodegenerative diseases. Handb. Clin. Neurol. 2017, 145, 301–307. [Google Scholar] [PubMed]
- Lin, M.T.; Beal, M.F. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 2006, 443, 787–796. [Google Scholar] [CrossRef] [PubMed]
- Ross, C.A.; Poirier, M.A. Protein aggregation and neurodegenerative disease. Nature Med. 2004, 10, S10. [Google Scholar] [CrossRef]
- Garg, A.; Garg, S.; Zaneveld, L.; Singla, A. Chemistry and pharmacology of the citrus bioflavonoid hesperidin. Phytother. Res. 2001, 15, 655–669. [Google Scholar] [CrossRef]
- Solanki, I.; Parihar, P.; Parihar, M.S. Neurodegenerative diseases: From available treatments to prospective herbal therapy. Neurochem. Int. 2016, 95, 100–108. [Google Scholar] [CrossRef]
- Jellinger, K. Cell death mechanisms in neurodegeneration. J. Cell. Molecul. Med. 2001, 5, 1–17. [Google Scholar] [CrossRef]
- Parihar, M.; Parihar, A.; Fujita, M.; Hashimoto, M.; Ghafourifar, P. Mitochondrial association of alpha-synuclein causes oxidative stress. Cell. Mol. Life Sci. 2008, 65, 1272–1284. [Google Scholar] [CrossRef]
- Balaban, H.; Nazıroğlu, M.; Demirci, K.; Övey, İ.S. The protective role of selenium on scopolamine-induced memory impairment, oxidative stress, and apoptosis in aged rats: The involvement of TRPM2 and TRPV1 channels. Mol. Neurobiol. 2017, 54, 2852–2868. [Google Scholar] [CrossRef]
- Prakash, J.; Chouhan, S.; Yadav, S.K.; Westfall, S.; Rai, S.N.; Singh, S.P. Withania somnifera alleviates parkinsonian phenotypes by inhibiting apoptotic pathways in dopaminergic neurons. Neurochem. Res. 2014, 39, 2527–2536. [Google Scholar] [CrossRef] [PubMed]
- Rasool, M.; Malik, A.; Qureshi, M.S.; Manan, A.; Pushparaj, P.N.; Asif, M.; Qazi, M.H.; Qazi, A.M.; Kamal, M.A.; Gan, S.H. Recent updates in the treatment of neurodegenerative disorders using natural compounds. Evid. Based. Complement. Alternat. Med. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Gilgun-Sherki, Y.; Melamed, E.; Offen, D. Oxidative stress induced-neurodegenerative diseases: The need for antioxidants that penetrate the blood brain barrier. Neuropharmacology 2001, 40, 959–975. [Google Scholar] [CrossRef]
- Andersen, J.K. Oxidative stress in neurodegeneration: Cause or consequence? Nat. Med. 2004, 10, S18. [Google Scholar] [CrossRef] [PubMed]
- Bal-Price, A.; Matthias, A.; Brown, G.C. Stimulation of the NADPH oxidase in activated rat microglia removes nitric oxide but induces peroxynitrite production. J. Neurochem. 2002, 80, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Mottay, D.; Neergheen-Bhujun, V.S. Anticholinesterase and antioxidant effects of traditional herbal medicines used in the management of neurodegenerative diseases in mauritius. Arch. Med. Biomed. Res. 2015, 2, 114–130. [Google Scholar] [CrossRef]
- Furlan, V.; Konc, J.; Bren, U. Inverse Molecular Docking as a Novel Approach to Study Anticarcinogenic and Anti-Neuroinflammatory Effects of Curcumin. Molecules 2018, 23, 3351. [Google Scholar] [CrossRef]
- Hwang, S.L.; Shih, P.H.; Yen, G.C. Neuroprotective Effects of Citrus Flavonoids. J. Agric. Food Chem. 2012, 60, 877–885. [Google Scholar] [CrossRef]
- Parhiz, H.; Roohbakhsh, A.; Soltani, F.; Rezaee, R.; Iranshahi, M. Antioxidant and anti-inflammatory properties of the citrus flavonoids hesperidin and hesperetin: An updated review of their molecular mechanisms and experimental models. Phytother. Res. 2015, 29, 323–331. [Google Scholar] [CrossRef]
- Roohbakhsh, A.; Parhiz, H.; Soltani, F.; Rezaee, R.; Iranshahi, M. Neuropharmacological properties and pharmacokinetics of the citrus flavonoids hesperidin and hesperetin—A mini-review. Life Sci. 2014, 113, 1–6. [Google Scholar] [CrossRef]
- Girdhar, S.; Girdhar, A.; Verma, S.K.; Lather, V.; Pandita, D. Plant derived alkaloids in major neurodegenerative diseases: From animal models to clinical trials. J. Ayurved. Herb. Med. 2015, 1, 91–100. [Google Scholar]
- Hendriks, J.J.; de Vries, H.E.; van der Pol, S.M.; van den Berg, T.K.; van Tol, E.A.; Dijkstra, C.D. Flavonoids inhibit myelin phagocytosis by macrophages; a structure–activity relationship study. Biochem. Pharmacol. 2003, 65, 877–885. [Google Scholar] [CrossRef]
- Sternberg, Z.; Chadha, K.; Lieberman, A.; Hojnacki, D.; Drake, A.; Zamboni, P.; Rocco, P.; Grazioli, E.; Weinstock-Guttman, B.; Munschauer, F. Quercetin and interferon-β modulate immune response (s) in peripheral blood mononuclear cells isolated from multiple sclerosis patients. J. Neuroimmunol. 2008, 205, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Candiracci, M.; Piatti, E.; Dominguez-Barragán, M.A.; García-Antrás, D.; Morgado, B.; Ruano, D.; Gutiérrez, J.F.; Parrado, J.; Castaño, A. Anti-inflammatory activity of a honey flavonoid extract on lipopolysaccharide-activated N13 microglial cells. J. Agric. Food Chem. 2012, 60, 12304–12311. [Google Scholar] [CrossRef] [PubMed]
- Lau, F.C.; Bielinski, D.F.; Joseph, J.A. Inhibitory effects of blueberry extract on the production of inflammatory mediators in lipopolysaccharide-activated BV2 microglia. J. Neurosci. Res. 2007, 85, 1010–1017. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Sapkota, K.; Kim, S.; Kim, H.; Kim, S. Kaempferol acts through mitogen-activated protein kinases and protein kinase B/AKT to elicit protection in a model of neuroinflammation in BV2 microglial cells. Brit. J. Pharmacol. 2011, 164, 1008–1025. [Google Scholar]
- Li, R.; Huang, Y.G.; Fang, D.; Le, W.D. (−)-Epigallocatechin gallate inhibits lipopolysaccharide-induced microglial activation and protects against inflammation-mediated dopaminergic neuronal injury. J. Neurosci. Res. 2004, 78, 723–731. [Google Scholar] [CrossRef]
- Vafeiadou, K.; Vauzour, D.; Lee, H.Y.; Rodriguez-Mateos, A.; Williams, R.J.; Spencer, J.P. The citrus flavanone naringenin inhibits inflammatory signalling in glial cells and protects against neuroinflammatory injury. Arch. Biochem. Biophys. 2009, 484, 100–109. [Google Scholar] [CrossRef]
- Kim, H.G.; Ju, M.S.; Ha, S.K.; Lee, H.; Lee, H.; Kim, S.Y.; Oh, M.S. Acacetin protects dopaminergic cells against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced neuroinflammation in vitro and in vivo. Biol. Pharm. Bull. 2012, 35, 1287–1294. [Google Scholar] [CrossRef]
- Farooqui, T.; Farooqui, A.A. Neuroprotective Effects of Phytochemicals in Neurological Disorders; John Wiley & Sons: Hoboken, NJ, USA, 2017. [Google Scholar]
- Okello, E.J.; Leylabi, R.; McDougall, G.J. Inhibition of acetylcholinesterase by green and white tea and their simulated intestinal metabolites. Food Funct. 2012, 3, 651–661. [Google Scholar] [CrossRef]
- Jung, M.; Park, M. Acetylcholinesterase inhibition by flavonoids from Agrimonia pilosa. Molecules 2007, 12, 2130–2139. [Google Scholar] [CrossRef] [PubMed]
- Menichini, F.; Tundis, R.; Loizzo, M.R.; Bonesi, M.; Marrelli, M.; Statti, G.A.; Menichini, F.; Conforti, F. Acetylcholinesterase and butyrylcholinesterase inhibition of ethanolic extract and monoterpenes from Pimpinella anisoides V Brig.(Apiaceae). Fitoterapia 2009, 80, 297–300. [Google Scholar] [CrossRef]
- Kivrak, İ.; Duru, M.E.; Öztürk, M.; Mercan, N.; Harmandar, M.; Topçu, G. Antioxidant, anticholinesterase and antimicrobial constituents from the essential oil and ethanol extract of Salvia potentillifolia. Food Chem. 2009, 116, 470–479. [Google Scholar] [CrossRef]
- Ahmed, F.; Ghalib, R.M.; Sasikala, P.; Ahmed, K.M. Cholinesterase inhibitors from botanicals. Pharmacog. Rev. 2013, 7, 121. [Google Scholar]
- Orhan, I.; Şener, B. Sustainable Use of Various Amaryllidaceae Plants Against Alzheimer’s Disease; III WOCMAP Congress on Medicinal and Aromatic Plants-Volume 4: Targeted Screening of Medicinal and Aromatic Plants, Economics: Chiang Mai, Thailand, 2003; Volume 678, pp. 59–64. [Google Scholar]
- Jiménez-Aliaga, K.; Bermejo-Bescós, P.; Benedí, J.; Martín-Aragón, S. Quercetin and rutin exhibit antiamyloidogenic and fibril-disaggregating effects in vitro and potent antioxidant activity in APPswe cells. Life. Sci. 2011, 89, 939–945. [Google Scholar] [CrossRef] [PubMed]
- Currais, A.; Prior, M.; Dargusch, R.; Armando, A.; Ehren, J.; Schubert, D.; Quehenberger, O.; Maher, P. Modulation of p25 and inflammatory pathways by fisetin maintains cognitive function in Alzheimer’s disease transgenic mice. Aging cell 2014, 13, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.-Y.; Cheng, Y.; Yu, L.-C. Potential protection of green tea polyphenols against intracellular amyloid beta-induced toxicity on primary cultured prefrontal cortical neurons of rats. Neurosci. Let. 2012, 513, 170–173. [Google Scholar] [CrossRef] [PubMed]
- Ikan, R. Natural Products: A Laboratory Guide; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Kawaguchi, K.; Kikuchi, S.; Takayanagi, K.; Yoshikawa, T.; Kumazawa, Y. Colony stimulating factor-inducing activity of hesperidin. Planta Med. 1999, 65, 365–366. [Google Scholar] [CrossRef]
- Bhalla, N.; Dakwale, R. Chemotaxonomy of Indigofera Linn. J. Indian Bot. Soc. 1978, 57, 180–185. [Google Scholar]
- Pawloska, L. Flavonoids of B. pendula Roth and B. obscura Kot leaves. Acta. Soc. Bot. Pol. 1980, 493, 281–296. [Google Scholar]
- Kokkalou, E.; Kapetanidis, I. Flavonoids of the aerial parts of Acinos suaveolens. Pharm. Acta Helv. 1988, 636, 170–173. [Google Scholar]
- Arthur, H.R.; Hui, W.; Ma, C. 127. An examination of the rutaceae of Hong Kong. Part I. Flavonoid glycosides from Zanthoxylum species and the occurrence of optically active hesperetin. J. Chem. Soc. 1956, 632–635. [Google Scholar] [CrossRef]
- Devi, K.P.; Rajavel, T.; Nabavi, S.F.; Setzer, W.N.; Ahmadi, A.; Mansouri, K.; Nabavi, S.M. Hesperidin: A promising anticancer agent from nature. Ind. Crop Prod. 2015, 76, 582–589. [Google Scholar] [CrossRef]
- Roohbakhsh, A.; Parhiz, H.; Soltani, F.; Rezaee, R.; Iranshahi, M. Molecular mechanisms behind the biological effects of hesperidin and hesperetin for the prevention of cancer and cardiovascular diseases. Life Sci. 2015, 124 (Suppl. C), 64–74. [Google Scholar] [CrossRef]
- Bartoszewski, R.; Hering, A.; Marszałł, M.; Hajduk, J.S.; Bartoszewska, S.; Kapoor, N.; Kochan, K.; Ochocka, R. Mangiferin has an additive effect on the apoptotic properties of hesperidin in Cyclopia sp. tea extracts. PLoS ONE 2014, 9, e92128. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Kim, M.-J.; Ha, E.; Chung, J.-H. Apoptotic effect of hesperidin through caspase3 activation in human colon cancer cells, SNU-C4. Phytomedicine 2008, 15, 147–151. [Google Scholar] [CrossRef] [PubMed]
- Etcheverry, S.B.; Ferrer, E.G.; Naso, L.; Rivadeneira, J.; Salinas, V.; Williams, P.A.M. Antioxidant effects of the VO (IV) hesperidin complex and its role in cancer chemoprevention. JBIC J. Biol. Inorgan. Chem. 2008, 13, 435. [Google Scholar] [CrossRef]
- Lee, C.J.; Wilson, L.; Jordan, M.A.; Nguyen, V.; Tang, J.; Smiyun, G. Hesperidin suppressed proliferations of both Human breast cancer and androgen-dependent prostate cancer cells. Phytother. Res. 2010, 24. [Google Scholar] [CrossRef]
- Park, H.J.; Ra, J.; Han, M.Y.; Chung, J.H. Hesperidin induces apoptosis in SNU-668, human gastric cancer cells. Mol. Cell. Toxicol. 2007, 3, 31–35. [Google Scholar]
- Cincin, Z.B.; Unlu, M.; Kiran, B.; Bireller, E.S.; Baran, Y.; Cakmakoglu, B. Anti-proliferative, apoptotic and signal transduction effects of hesperidin in non-small cell lung cancer cells. Cell Oncol. 2015, 38, 195–204. [Google Scholar] [CrossRef]
- Al-Jasabi, S.; Abdullah, M. The role of antioxidant hesperidin in the attenuation of lung cancer caused by benzo [a] pyrene in Balb/c mice. World Appl. Sci. J. 2013, 22, 1106–1110. [Google Scholar]
- Julius, A.; Vedasendiyar, R.; Devakannan, A.; Rajaraman, S.; Rangasamy, B.; Saravanan, V. Effect of Hesperidin for its Anti-Proliferative Activity on Liver Cancer and Cardio Vascular Diseases. Res. J. Pharm. Technol. 2017, 10, 307–308. [Google Scholar] [CrossRef]
- Nandakumar, N.; Balasubramanian, M.P. Hesperidin protects renal and hepatic tissues against free radical-mediated oxidative stress during DMBA-induced experimental breast cancer. J. Environ. Pathol. Toxicol. Oncol. 2011, 30. [Google Scholar] [CrossRef]
- Febriansah, R.; Dyaningtyas, D.P.; Nurulita, N.A.; Meiyanto, E.; Nugroho, A.E. Hesperidin as a preventive resistance agent in MCF–7 breast cancer cells line resistance to doxorubicin. Asia. Pac. J. Trop. Biomed. 2014, 4, 228–233. [Google Scholar] [CrossRef]
- Tanaka, T.; Tanaka, T.; Tanaka, M.; Kuno, T. Cancer chemoprevention by citrus pulp and juices containing high amounts of β-cryptoxanthin and hesperidin. BioMed. Res. Int. 2011, 2012. [Google Scholar] [CrossRef] [PubMed]
- Sakata, K.; Hirose, Y.; Qiao, Z.; Tanaka, T.; Mori, H. Inhibition of inducible isoforms of cyclooxygenase and nitric oxide synthase by flavonoid hesperidin in mouse macrophage cell line. Cancer Let. 2003, 199, 139–145. [Google Scholar] [CrossRef]
- Brglez Mojzer, E.; Knez Hrnčič, M.; Škerget, M.; Knez, Ž.; Bren, U. Polyphenols: Extraction methods, antioxidative action, bioavailability and anticarcinogenic effects. Molecules 2016, 21, 901. [Google Scholar] [CrossRef]
- Bisset, N.; Houghton, P.; Hylands, P. The Medicinal Plant Industry. Medicinal Plant Industry; CRC Press: Boca Raton, FL, USA, 1991. [Google Scholar]
- Morii, S. Research for vitamin P. J. Biochem. 1939, 29, 487–501. [Google Scholar]
- Son, H.-S.; Kim, H.-S.; Ju, J.-S. Effects of rutin and hesperidin on total cholesterol concentration, transaminase and alkaline phosphatase activity in CCl4 treated rats. Appl. Biol. Chem. 1991, 34, 318–326. [Google Scholar]
- Choi, J.S.; Yokozawa, T.; Oura, H. Antihyperlipidemic effect of flavonoids from Prunus davidiana. J. Nat. Prod. 1991, 54, 218–224. [Google Scholar] [CrossRef]
- Galati, E.; Monforte, M.; Kirjavainen, S.; Forestieri, A.; Trovato, A.; Tripodo, M. Biological effects of hesperidin, a citrus flavonoid.(Note I): Antiinflammatory and analgesic activity. Farmaco (Societa Chimica Italiana: 1989) 1994, 40, 709–712. [Google Scholar]
- Morita, O.; Sasaki, H.; Sato, S. Calcium antagonists containing phenols. Pat. Jpn. Kokai Tokkyo Koho 1992, 4, 822. [Google Scholar]
- Thompson, R.; Sturtevant, M.; Bird, O. Effect of phosphorylated hesperidin and other flavonoids on fertility in mice. Science 1953, 118, 657. [Google Scholar] [CrossRef]
- Millman, N.; Rosen, F. Failure of phosphorylated hesperidin to influence fertility in rodents. Science 1953, 118, 212–213. [Google Scholar] [CrossRef]
- Filho, C.B.; Fabbro, L.D.; de Gomes, M.G.; Goes, A.T.R.; Souza, L.C.; Boeira, S.P.; Jesse, C.R. Kappa-opioid receptors mediate the antidepressant-like activity of hesperidin in the mouse forced swimming test. Eur. J. Pharmacol. 2013, 698, 286–291. [Google Scholar] [CrossRef] [PubMed]
- Thenmozhi, A.J.; Raja, T.R.W.; Janakiraman, U.; Manivasagam, T. Neuroprotective effect of hesperidin on aluminium chloride induced Alzheimer’s disease in Wistar rats. Neurochem. Res. 2015, 40, 767–776. [Google Scholar] [CrossRef]
- Cho, J. Antioxidant and neuroprotective effects of hesperidin and its aglycone hesperetin. Arch. Pharmacal. Res. 2006, 29, 699–706. [Google Scholar] [CrossRef]
- Tamilselvam, K.; Braidy, N.; Manivasagam, T.; Essa, M.M.; Prasad, N.R.; Karthikeyan, S.; Thenmozhi, A.J.; Selvaraju, S.; Guillemin, G.J. Neuroprotective effects of hesperidin, a plant flavanone, on rotenone-induced oxidative stress and apoptosis in a cellular model for Parkinson’s disease. Oxid. Med. Cell Longev. 2013, 2013. [Google Scholar] [CrossRef]
- Kumar, A.; Chaudhary, T.; Mishra, J. Minocycline modulates neuroprotective effect of hesperidin against quinolinic acid induced Huntington’s disease like symptoms in rats: Behavioral, biochemical, cellular and histological evidences. Eur. J. Pharmacol. 2013, 720, 16–28. [Google Scholar] [CrossRef]
- Meyer, O.C. Safety and security of Daflon 500 mg in venous insufficiency and in hemorrhoidal disease. Angiology 1994, 45, 579–584. [Google Scholar] [CrossRef]
- Knez Hrnčič, M.; Španinger, E.; Košir, I.J.; Knez, Ž.; Bren, U. Hop Compounds: Extraction Techniques, Chemical Analyses, Antioxidative, Antimicrobial, and Anticarcinogenic Effects. Nutrients 2019, 11, 257. [Google Scholar] [CrossRef] [PubMed]
- Antunes, M.S.; Goes, A.T.R.; Boeira, S.P.; Prigol, M.; Jesse, C.R. Protective effect of hesperidin in a model of Parkinson’s disease induced by 6-hydroxydopamine in aged mice. Nutrition 2014, 30, 1415–1422. [Google Scholar] [CrossRef] [PubMed]
- Souza, L.C.; de Gomes, M.G.; Goes, A.T.; Del Fabbro, L.; Carlos Filho, B.; Boeira, S.P.; Jesse, C.R. Evidence for the involvement of the serotonergic 5-HT 1A receptors in the antidepressant-like effect caused by hesperidin in mice. Prog. Neuro-Psychoph. 2013, 40, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Salem, H.R.A.; El-Raouf, A.; Saleh, E.M.; Shalaby, K.A. Influence of hesperidin combined with Sinemet on genetical and biochemical abnormalities in rats suffering from Parkinson’s disease. Life. Sci. J. 2012, 9, 930–945. [Google Scholar]
- Nagappan, P.; Krishnamurthy, V.; Sereen, K. Investigation of the neuroprotective effect of hesperidin on behavioural activities in 6-OHDA induced Parkinson model. Int. J. Pharm. Biol. Sci. 2014, 570–577. [Google Scholar]
- Tamilselvam, K.; Nataraj, J.; Janakiraman, U.; Manivasagam, T.; Essa, M.M. Antioxidant and anti-inflammatory potential of hesperidin against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced experimental Parkinson’s disease in mice. Int. J. Nutr. Pharm. Neurol. Dis. 2013, 3, 294. [Google Scholar]
- Nagappan, P.; Krishnamurthy, V. Anti-Parkinson Effect of Hesperidin in Combination with L-DOPA on 6-OHDA Induced Parkinsonism in Wistar Rats-A Neurochemical, Histopathological and Immunohistochemical Analysis. Int. J. Pharm. Tech. Res. 2016, 9, 266–273. [Google Scholar]
- Santos, G.; Giraldez-Alvarez, L.D.; Ávila-Rodriguez, M.; Capani, F.; Galembeck, E.; Neto, A.G.; Barreto, G.E.; Andrade, B. SUR1 Receptor Interaction with Hesperidin and Linarin Predicts Possible Mechanisms of Action of Valeriana officinalis in Parkinson. Front. Aging Neurosci. 2016, 2, 97. [Google Scholar] [CrossRef]
- Wang, D.; Liu, L.; Zhu, X.; Wu, W.; Wang, Y. Hesperidin Alleviates Cognitive Impairment, Mitochondrial Dysfunction and Oxidative Stress in a Mouse Model of Alzheimer’s Disease. Cell Mol. Neurobiol. 2014, 34, 1209–1221. [Google Scholar] [CrossRef]
- Badalzadeh, R.; Mohammadi, M.; Yousefi, B.; Farajnia, S.; Najafi, M.; Mohammadi, S. Involvement of glycogen synthase kinase-3β and oxidation status in the loss of cardioprotection by postconditioning in chronic diabetic male rats. Adv. Pharm. Bull. 2015, 5, 321. [Google Scholar] [CrossRef]
- DaRocha-Souto, B.; Coma, M.; Perez-Nievas, B.; Scotton, T.; Siao, M.; Sánchez-Ferrer, P.; Hashimoto, T.; Fan, Z.; Hudry, E.; Barroeta, I. Activation of glycogen synthase kinase-3 beta mediates β-amyloid induced neuritic damage in Alzheimer’s disease. Neurobiol. Dis. 2012, 45, 425–437. [Google Scholar] [CrossRef] [PubMed]
- Justin Thenmozhi, A.; William Raja, T.R.; Manivasagam, T.; Janakiraman, U.; Essa, M.M. Hesperidin ameliorates cognitive dysfunction, oxidative stress and apoptosis against aluminium chloride induced rat model of Alzheimer’s disease. Nutr. Neurosci. 2017, 20, 360–368. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zug, C.; Qu, H.; Schluesener, H.; Zhang, Z. Hesperidin ameliorates behavioral impairments and neuropathology of transgenic APP/PS1 mice. Behav. Brain Res. 2015, 281 (Suppl. C), 32–42. [Google Scholar] [CrossRef]
- Ghorbani, A.; Nazari, M.; Jeddi-Tehrani, M.; Zand, H. The citrus flavonoid hesperidin induces p53 and inhibits NF-κB activation in order to trigger apoptosis in NALM-6 cells: Involvement of PPARγ-dependent mechanism. Eur. J. Nutr. 2012, 51, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Gray, C.W.; Patel, A.J. Regulation of β-amyloid precursor protein isoform mRNAs by transforming growth factor-β1 and interleukin-1β in astrocytes. Mol. Brain Res. 1993, 19, 251–256. [Google Scholar] [CrossRef]
- Javed, H.; Vaibhav, K.; Ahmed, M.E.; Khan, A.; Tabassum, R.; Islam, F.; Safhi, M.M.; Islam, F. Effect of hesperidin on neurobehavioral, neuroinflammation, oxidative stress and lipid alteration in intracerebroventricular streptozotocin induced cognitive impairment in mice. J. Neurol. Sci. 2015, 348, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Hemanth Kumar, B.; Dinesh Kumar, B.; Diwan, P.V. Hesperidin, a citrus flavonoid, protects against l-methionine-induced hyperhomocysteinemia by abrogation of oxidative stress, endothelial dysfunction and neurotoxicity in Wistar rats. Pharm. Biol. 2017, 55, 146–155. [Google Scholar] [CrossRef]
- Hemanth Kumar, B.; Dinesh Kumar, B.; Diwan, P.V. Protective effects of natural dietary antioxidants fisetin and hesperidin on chronic mild hyperhomocysteinemia-induced vascular dementia in wistar rats. J. Neurol. Sci. 2017, 381, 319. [Google Scholar] [CrossRef]
- Habibyar, A.F.; Sharma, N.; Khurana, N. PASS assisted prediction and pharmacological evaluation of hesperidin against scopolamine induced amnesia in mice. Eur. J. Pharmacol. 2016, 789 (Suppl. C), 385–394. [Google Scholar] [CrossRef]
- Rosas, H.D.; Lee, S.Y.; Bender, A.C.; Zaleta, A.K.; Vangel, M.; Yu, P.; Fischl, B.; Pappu, V.; Onorato, C.; Cha, J.-H. Altered white matter microstructure in the corpus callosum in Huntington’s disease: Implications for cortical “disconnection”. Neuroimage 2010, 49, 2995–3004. [Google Scholar] [CrossRef]
- Rosas, H.; Liu, A.; Hersch, S.; Glessner, M.; Ferrante, R.; Salat, D.; van Der Kouwe, A.; Jenkins, B.; Dale, A.; Fischl, B. Regional and progressive thinning of the cortical ribbon in Huntington’s disease. Neurology 2002, 58, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Kumar, A. Protective effect of hesperidin and naringin against 3-nitropropionic acid induced Huntington’s like symptoms in rats: Possible role of nitric oxide. Behav. Brain Res. 2010, 206, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Menze, E.T.; Tadros, M.G.; Abdel-Tawab, A.M.; Khalifa, A.E. Potential neuroprotective effects of hesperidin on 3-nitropropionic acid-induced neurotoxicity in rats. Neurotoxicology 2012, 33, 1265–1275. [Google Scholar] [CrossRef] [PubMed]
- Coull, J.A.M.; Beggs, S.; Boudreau, D.; Boivin, D.; Tsuda, M.; Inoue, K.; Gravel, C.; Salter, M.W.; De Koninck, Y. BDNF from microglia causes the shift in neuronal anion gradient underlying neuropathic pain. Nature 2005, 438, 1017–1021. [Google Scholar] [CrossRef] [PubMed]
- Ontaneda, D.; Thompson, A.J.; Fox, R.J.; Cohen, J.A. Progressive multiple sclerosis: Prospects for disease therapy, repair, and restoration of function. Lancet 2017, 389, 1357–1366. [Google Scholar] [CrossRef]
- Farzaei, M.H.; Shahpiri, Z.; Bahramsoltani, R.; Najafi, F.; Rahimi, R. Efficacy and Tolerability of Phytomedicines in Multiple Sclerosis Patients: A Review. CNS Drugs 2017, 31, 867–889. [Google Scholar] [CrossRef] [PubMed]
- Minagar, A.; Shapshak, P.; Alexander, J.S. Dementia and Multiple Sclerosis: Role of Microglia and Astrocytes. Role Glia Neurotox. 2004, 263. [Google Scholar]
- Muili, K.A.; Gopalakrishnan, S.; Meyer, S.L.; Eells, J.T.; Lyons, J.-A. Amelioration of experimental autoimmune encephalomyelitis in C57BL/6 mice by photobiomodulation induced by 670 nm light. PLoS ONE 2012, 7, e30655. [Google Scholar] [CrossRef] [PubMed]
- Haghmorad, D.; Mahmoudi, M.B.; Salehipour, Z.; Jalayer, Z.; Rastin, M.; Kokhaei, P.; Mahmoudi, M. Hesperidin ameliorates immunological outcome and reduces neuroinflammation in the mouse model of multiple sclerosis. J. Neuroimmun. 2017, 302, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Ciftci, O.; Ozcan, C.; Kamisli, O.; Cetin, A.; Basak, N.; Aytac, B. Hesperidin, a citrus flavonoid, has the ameliorative effects against experimental autoimmune encephalomyelitis (EAE) in a C57BL/J6 mouse model. Neurochem. Res. 2015, 40, 1111–1120. [Google Scholar] [CrossRef] [PubMed]
- Ahtiluoto, S.; Polvikoski, T.; Peltonen, M.; Solomon, A.; Tuomilehto, J.; Winblad, B.; Sulkava, R.; Kivipelto, M. Diabetes, Alzheimer disease, and vascular dementia A population-based neuropathologic study. Neurology 2010, 75, 1195–1202. [Google Scholar] [CrossRef] [PubMed]
- Takeda, S.; Sato, N.; Uchio-Yamada, K.; Sawada, K.; Kunieda, T.; Takeuchi, D.; Kurinami, H.; Shinohara, M.; Rakugi, H.; Morishita, R. Diabetes-accelerated memory dysfunction via cerebrovascular inflammation and Aβ deposition in an Alzheimer mouse model with diabetes. Proc. Natl. Acad. Sci. USA 2010, 107, 7036–7041. [Google Scholar] [CrossRef] [PubMed]
- Craft, S. Alzheimer disease: Insulin resistance and AD--extending the translational path. Nat. Rev. Neurol. 2012, 8, 360–362. [Google Scholar] [CrossRef] [PubMed]
- Hsu, F.-L.; Liu, I.-M.; Kuo, D.-H.; Chen, W.-C.; Su, H.-C.; Cheng, J.-T. Antihyperglycemic effect of puerarin in streptozotocin-induced diabetic rats. J. Nat. Prod. 2003, 66, 788–792. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, S.S. Protective effect of hesperidin, a citrus bioflavonoid, on diabetes-induced brain damage in rats. J. Appl. Sci. Res. 2008, 4, 84. [Google Scholar]
- Khowal, S.; Mustufa, M.M.; Chaudhary, N.K.; Naqvi, S.H.; Parvez, S.; Jain, S.K.; Wajid, S. Assessment of the therapeutic potential of hesperidin and proteomic resolution of diabetes-mediated neuronal fluctuations expediting Alzheimer’s disease. RSC Adv. 2015, 5, 46965–46980. [Google Scholar] [CrossRef]
- Ashafaq, M.; Varshney, L.; Khan, M.H.A.; Salman, M.; Naseem, M.; Wajid, S.; Parvez, S. Neuromodulatory effects of hesperidin in mitigating oxidative stress in streptozotocin induced diabetes. BioMed. Res. Int. 2014, 2014. [Google Scholar] [CrossRef]
- Visnagri, A.; Kandhare, A.D.; Chakravarty, S.; Ghosh, P.; Bodhankar, S.L. Hesperidin, a flavanoglycone attenuates experimental diabetic neuropathy via modulation of cellular and biochemical marker to improve nerve functions. Pharm. Biol. 2014, 52, 814–828. [Google Scholar] [CrossRef]
- Miranda, H.; Sierralta, F.; Jorquera, V.; Poblete, P.; Prieto, J.; Noriega, V. Antinociceptive interaction of gabapentin with minocycline in murine diabetic neuropathy. Inflammopharmacology 2017, 25, 91–97. [Google Scholar] [CrossRef]
- Kakadiya, J.; Patel, D.; Shah, N. Effect of hesperidin on renal complication in experimentally induced renal damage in diabetic sprague dawley rats. J. Ecobiotech. 2010, 2, 45–50. [Google Scholar]
- Gustafson-Vickers, S.L.; Lu, V.B.; Lai, A.Y.; Todd, K.G.; Ballanyi, K.; Smith, P.A. Long-term actions of interleukin-1β on delay and tonic firing neurons in rat superficial dorsal horn and their relevance to central sensitization. Mol. Pain 2008, 4, 63. [Google Scholar] [CrossRef] [PubMed]
- Kean, R.J.; Lamport, D.J.; Dodd, G.F.; Freeman, J.E.; Williams, C.M.; Ellis, J.A.; Butler, L.T.; Spencer, J.P. Chronic consumption of flavanone-rich orange juice is associated with cognitive benefits: An 8-wk, randomized, double-blind, placebo-controlled trial in healthy older adults. Am. J. Clin. Nutr. 2015, 101, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Francis, S.; Head, K.; Morris, P.; Macdonald, I. The effect of flavanol-rich cocoa on the fMRI response to a cognitive task in healthy young people. J. Cardiovasc. Pharmacol. 2006, 47, 2152. [Google Scholar] [CrossRef]
- Lamport, D.J.; Pal, D.; Macready, A.L.; Barbosa-Boucas, S.; Fletcher, J.M.; Williams, C.M.; Spencer, J.P.; Butler, L.T. The effects of flavanone-rich citrus juice on cognitive function and cerebral blood flow: An acute, randomised, placebo-controlled cross-over trial in healthy, young adults. Br. J. Nutr. 2016, 116, 2160–2168. [Google Scholar] [CrossRef] [PubMed]
- Alharbi, M.H.; Lamport, D.J.; Dodd, G.F.; Saunders, C.; Harkness, L.; Butler, L.T.; Spencer, J.P. Flavonoid-rich orange juice is associated with acute improvements in cognitive function in healthy middle-aged males. Eur. J. Nutr. 2016, 55, 2021–2029. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Tomata, Y.; Sugiyama, K.; Sugawara, Y.; Tsuji, I. Citrus consumption and incident dementia in elderly Japanese: The Ohsaki Cohort 2006 Study. Br. J. Nutr. 2017, 117, 1174–1180. [Google Scholar] [CrossRef] [PubMed]
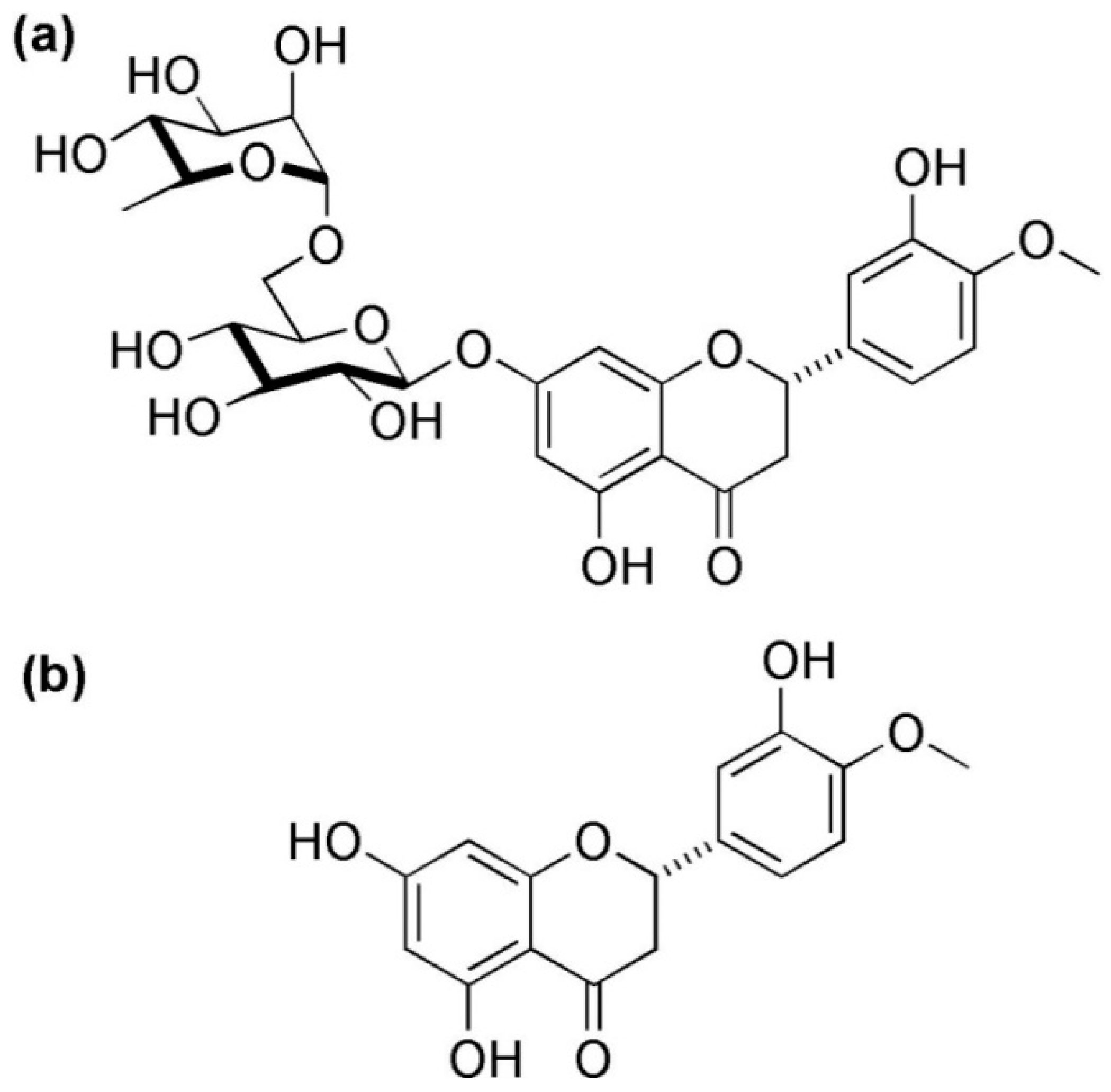
| Animal Model | Method | Mechanism of Action of Hesperidin | References |
|---|---|---|---|
| Parkinson’s Disease | 6-Hydroxydopamine (6-OHDA) induced PD in rats | ↑ GPx, GSH, TRAP and CAT activity ↑ DA, DOPAC, and HVA levels ↓ ROS and GR | [77] |
| Chlorpyrifos—induced PD in rats | ↑ CAT, GST and GSH activity ↓TG, TC, and Glucose levels ↓ MDA and SOD levels ↑ AChE | [79] | |
| 6-Hydroxydopamine (6-OHDA) induced PD in rats | ↑ DA, serotonin, epinephrine, and Norepinephrine levels | [82] | |
| Rotenone induced PD in rats | Suppression of SNCA gene expressions, and LRRK2 ↑ Parkin and PINK1 | [83] | |
| 1-Methyl-4-phenyl-1,2,3, 6-tetrahydropyridine (MPTP)-induced PD in mice | ↓ IL-1β, TNF-α, IL- 6,4,10 ↓ GFAP, iNOS, and COX-2 | [81] | |
| Alzheimer’s Disease | APP/PS1–21 mouse model of AD | Suppression of β-amyloid deposition, APP expression, microglial activity Down-regulation of TGF-β | [88] |
| APPswe/PS1dE9 transgenic mice model of AD | ↓ MDA and H2O2 level ↑ GSH, T-AOC, and mitochondrial complex I–IV activity ↓ GSK-3β | [84] | |
| AlCl3 induced rat model of AD | ↓ Bax and TBARS ↑ Bcl2 | [84] | |
| AlCl3 induced rat model of AD | ↓ AChE, APP, Aβ1–40, β and γ secretases levels | [71] | |
| Sporadic dementia of Alzheimer’s type (SDAT) | Induced by intracerebroventricularstreptozotocin (ICV-STZ) | ↓ AChE and TBARS ↓ NF-κB, COX-2, and iNOS ↑ Gangliosides levels | [91] |
| Vascular Dementia | Hyperhomocysteinemia (HHcy) induced by l-methionine in rat | ↑ CAT, GSH, and SOD ↓ AChE and MDA ↓ Serum nitrite, and serum Homocysteinemia (Hcy) | [92] |
| Amentia | Induced by scopolamine in mice | ↑ CAT and GSH ↓ AChE and TBARS | [94] |
| Huntington’s disease | Induced by 3-Nitropropionic acid (3-NP) | ↓ NO in endothelial cells ↓ (iNOS) | [96] |
| ↑ CAT ↓ MDA | [97] | ||
| Prevention of prepulse inhibition (PPI) | [98] | ||
| Induced by quinolinic acid (QA) in rats | ↓ Caspase-3 activity ↑ BDNF ↓ TNF-α | [75] | |
| Multiple Sclerosis | Induced by myelin oligodendrocyte glycoprotein (MOG) in C57BL/6 mice | ↓ IL-6, IL-17, IL-23, TNF-α, and Th17 cells transcription factor (ROR-γt) ↑ IL-10 and TGF-β | [104] |
| Induced by Experimental allergic encephalomyelitis (EAE) by MOG35-55 in C57BL/J6 mouse | ↓ IL-17, TNF-α, IL-1β ↓ Caspase-3 activity | [105] | |
| Diabetes mellitus associated neurotoxicity | Induced by streptozotocin (STZ) | ↑ GST, GR, GSH, and NP-SH ↓ LPO ↓ XO ↑Na+/K+ ATPase | [112] |
| ↑ DNA, RNA, GPX, SOD, and GSH ↓ MDA, NO, SD, ↑ Plasma glucose, Hb | [112] | ||
| Diabetic neuropathy (DN) | Induced by STZ | ↓ TNF-α IL-1β ↑ HbA1c ↑ Na+/K+ ATPase | [113] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hajialyani, M.; Hosein Farzaei, M.; Echeverría, J.; Nabavi, S.M.; Uriarte, E.; Sobarzo-Sánchez, E. Hesperidin as a Neuroprotective Agent: A Review of Animal and Clinical Evidence. Molecules 2019, 24, 648. https://doi.org/10.3390/molecules24030648
Hajialyani M, Hosein Farzaei M, Echeverría J, Nabavi SM, Uriarte E, Sobarzo-Sánchez E. Hesperidin as a Neuroprotective Agent: A Review of Animal and Clinical Evidence. Molecules. 2019; 24(3):648. https://doi.org/10.3390/molecules24030648
Chicago/Turabian StyleHajialyani, Marziyeh, Mohammad Hosein Farzaei, Javier Echeverría, Seyed Mohammad Nabavi, Eugenio Uriarte, and Eduardo Sobarzo-Sánchez. 2019. "Hesperidin as a Neuroprotective Agent: A Review of Animal and Clinical Evidence" Molecules 24, no. 3: 648. https://doi.org/10.3390/molecules24030648
APA StyleHajialyani, M., Hosein Farzaei, M., Echeverría, J., Nabavi, S. M., Uriarte, E., & Sobarzo-Sánchez, E. (2019). Hesperidin as a Neuroprotective Agent: A Review of Animal and Clinical Evidence. Molecules, 24(3), 648. https://doi.org/10.3390/molecules24030648








