Transthyretin Amyloid Fibril Disrupting Activities of Extracts and Fractions from Juglans mandshurica Maxim. var. cordiformis (Makino) Kitam.
Abstract
1. Introduction
2. Results
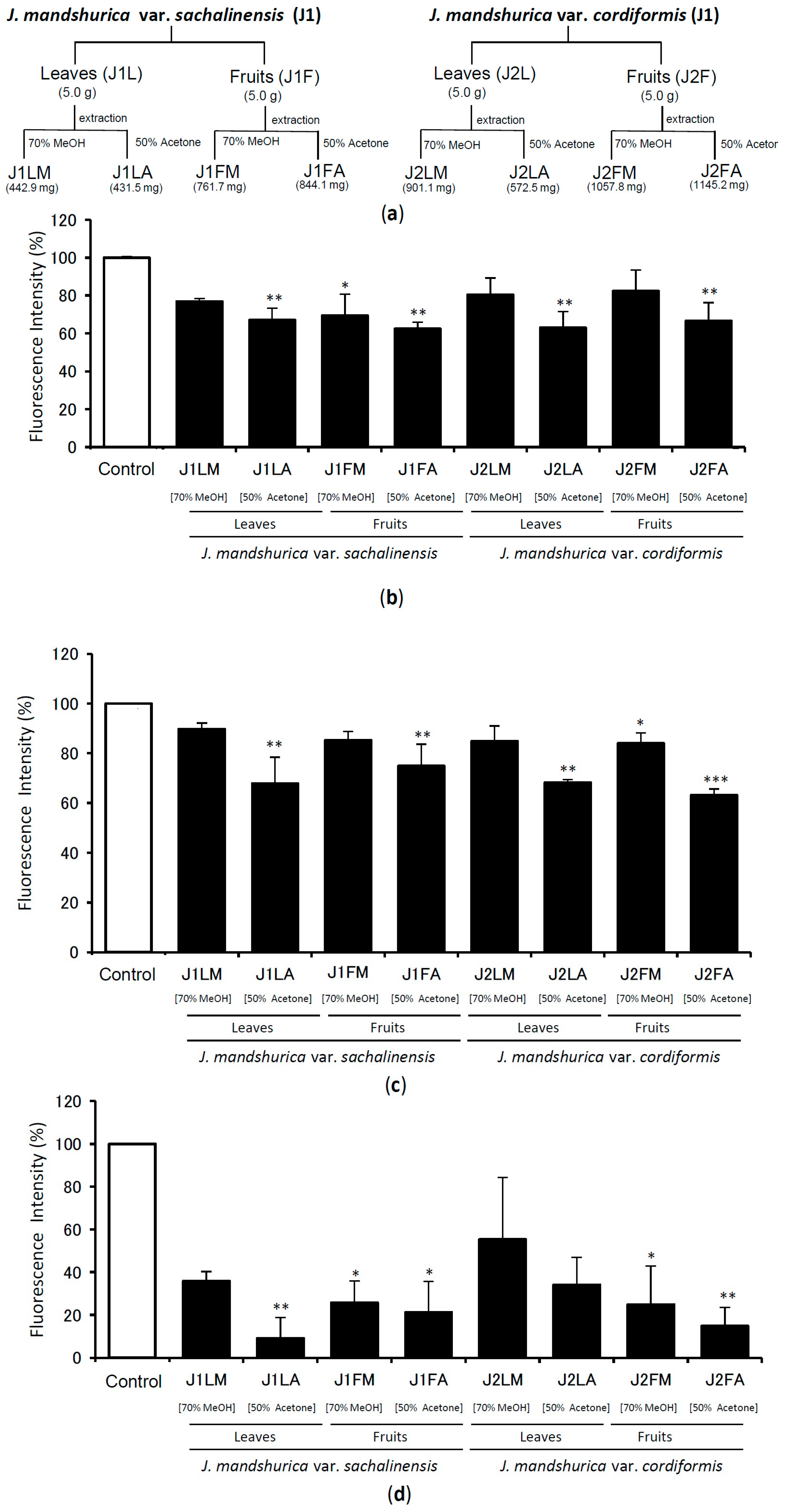
2.1. Screening of Extracts of Juglans Plants for TTR Amyloid Fibril Disrupting Activity
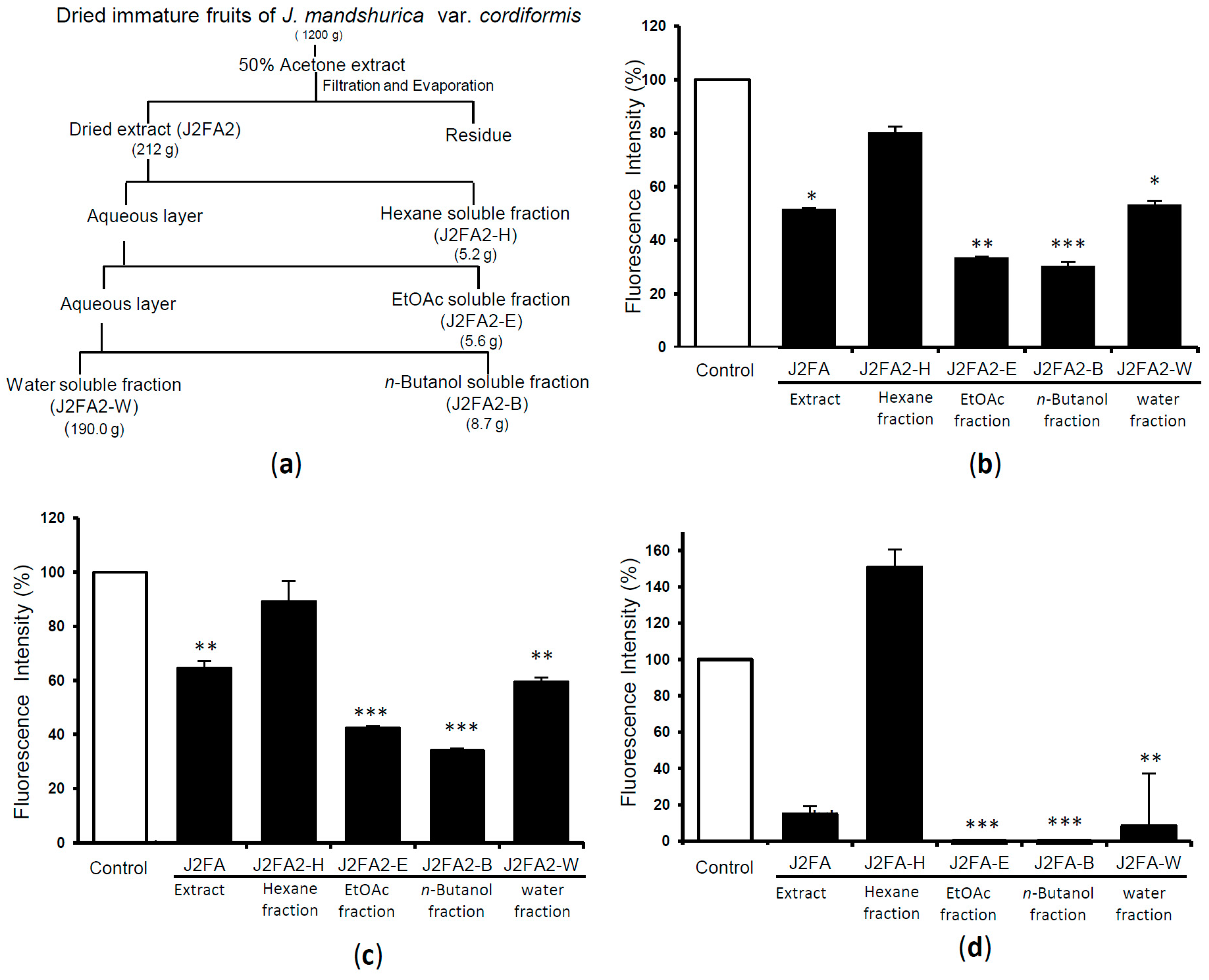
2.2. Extraction, Fractionation and Compound Isolation of Immature Fruits of J. mandshurica var. cordiformis and Their Activities
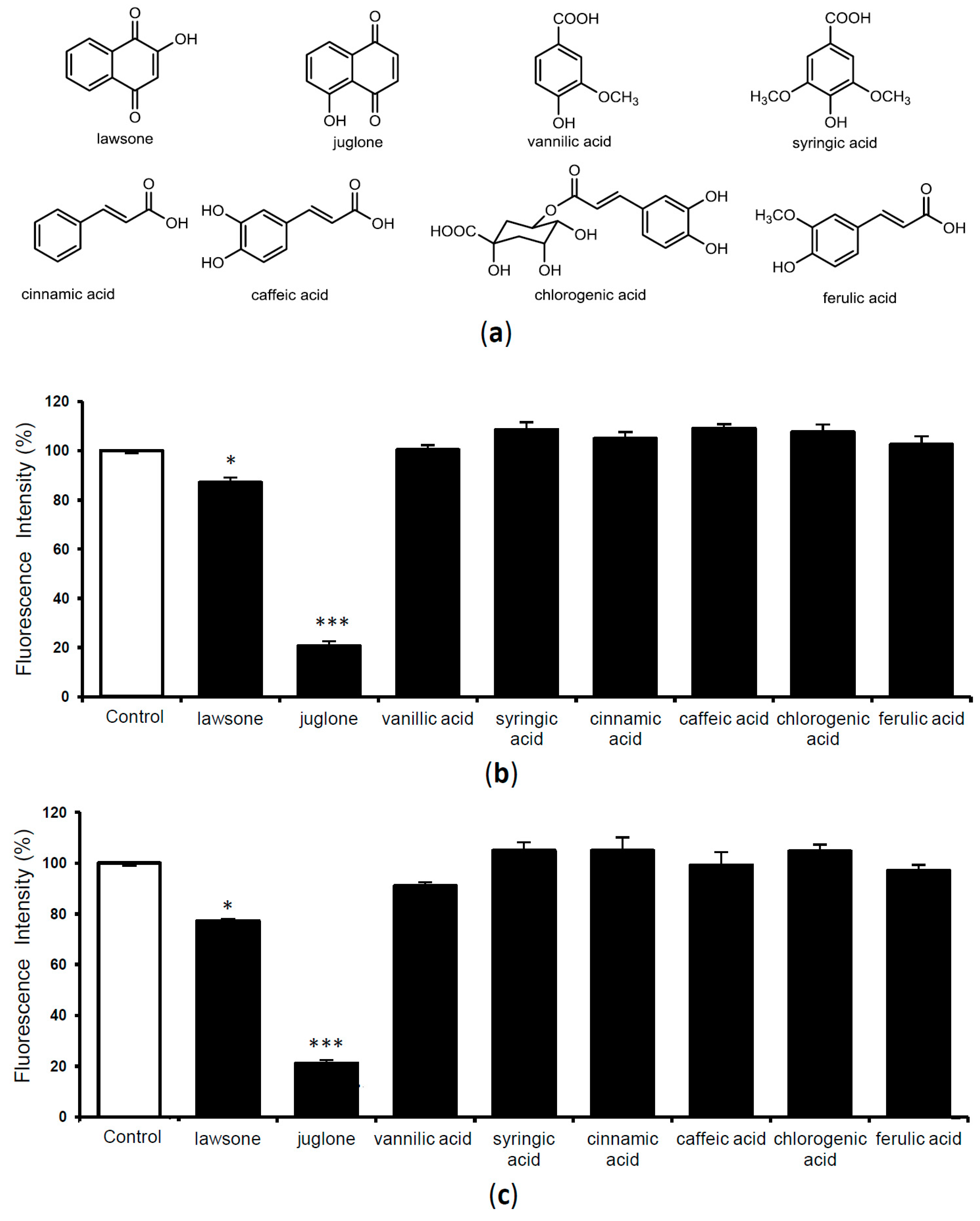
2.3. Evaluation of TTR Amyloid Fibril Disrupting Activity of Previously Isolated Compounds from Juglans Plants
2.4. Dose Dependent TTR Amyloid Fibril Disrupting Activity and Cytotoxicity Evaluation of Juglone
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Plant Materials
4.3. Extraction, Fractionation and Isolation
4.4. Thioflavin T Binding Assay
4.5. PROTEOSTAT® Protein Aggregation Assay
4.6. Cell Culture
4.7. Cell Cytotoxicity Assay Using Cytotoxicity Detection Kit Plus (LDH)
4.8. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Da Costa, G.; Ribeiro-Silva, C.; Ribeiro, R.; Gilberto, S.; Gomes, R.A.; Ferreira, A.; Mateus, É.; Barroso, E.; Coelho, A.V.; Freire, A.P.; et al. Transthyretin amyloidosis: Chaperone concentration changes and increased proteolysis in the pathway to disease. PLoS ONE 2015, 10, e0125392. [Google Scholar] [CrossRef] [PubMed]
- Quintas, A.; Vaz, D.C.; Cardoso, I.; Saraiva, M.J.M.; Brito, R.M.M. Tetramer Dissociation and Monomer Partial Unfolding Precedes Protofibril Formation in Amyloidogenic Transthyretin Variants. J. Biol. Chem. 2001, 276, 27207–27213. [Google Scholar] [CrossRef] [PubMed]
- Chan, G.G.; Koch, C.M.; Connors, L.H. Blood Proteomic Profiling in Inherited (ATTRm) and Acquired (ATTRwt) Forms of Transthyretin-Associated Cardiac Amyloidosis. J. Proteome Res. 2017, 16, 1659–1668. [Google Scholar] [CrossRef] [PubMed]
- Green, N.S.; Foss, T.R.; Kelly, J.W. Genistein, a natural product from soy, is a potent inhibitor of transthyretin amyloidosis. Proc. Natl. Acad. Sci. USA 2005, 102, 14545–14550. [Google Scholar] [CrossRef]
- Ando, Y.; Coelho, T.; Berk, J.L.; Cruz, M.W.; Ericzon, B.G.; Ikeda, S.I.; Lewis, W.D.; Obici, L.; Planté-Bordeneuve, V.; Rapezzi, C.; et al. Guideline of transthyretin-related hereditary amyloidosis for clinicians. Orphanet J. Rare Dis. 2013, 8, 31. [Google Scholar] [CrossRef] [PubMed]
- Sekijima, Y. Recent progress in the understanding and treatment of transthyretin amyloidosis. J. Clin. Pharm. Ther. 2014, 39, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Benson, M.D.; Waddington-Cruz, M.; Berk, J.L.; Polydefkis, M.; Dyck, P.J.; Wang, A.K.; Planté-Bordeneuve, V.; Barroso, F.A.; Merlini, G.; Obici, L.; et al. Inotersen Treatment for Patients with Hereditary Transthyretin Amyloidosis. N. Engl. J. Med. 2018, 379, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Adams, D.; Gonzalez-Duarte, A.; O’Riordan, W.D.; Yang, C.-C.; Ueda, M.; Kristen, A.V.; Tournev, I.; Schmidt, H.H.; Coelho, T.; Berk, J.L.; et al. Patisiran, an RNAi Therapeutic, for Hereditary Transthyretin Amyloidosis. N. Engl. J. Med. 2018, 379, 11–21. [Google Scholar] [CrossRef]
- Coelho, T.; Maia, L.F.; Da Silva, A.M.; Cruz, M.W.; Planté-Bordeneuve, V.; Suhr, O.B.; Conceiçao, I.; Schmidt, H.H.J.; Trigo, P.; Kelly, J.W.; et al. Long-term effects of tafamidis for the treatment of transthyretin familial amyloid polyneuropathy. J. Neurol. 2013, 260, 2802–2814. [Google Scholar] [CrossRef]
- Adams, D.; Buades, J.; Suhr, O.; Obici, L.; Coelho, T. Preliminary assessment of neuropathy progression in patients with hereditary ATTR amyloidosis after orthotopic liver transplantation (OLT). Orphanet J. Rare Dis. 2015, 10, P19. [Google Scholar] [CrossRef]
- Guo, L.; Giasson, B.I.; Glavis-Bloom, A.; Brewer, M.D.; Shorter, J.; Gitler, A.D.; Yang, X. A cellular system that degrades misfolded proteins and protects against neurodegeneration. Mol. Cell 2014, 55, 15–30. [Google Scholar] [CrossRef] [PubMed]
- Savelieff, M.G.; Detoma, A.S.; Derrick, J.S.; Lim, M.H. The ongoing search for small molecules to study metal-Associated amyloid-β species in alzheimers disease. Acc. Chem. Res. 2014, 47, 2475–2482. [Google Scholar] [CrossRef] [PubMed]
- Korshavn, K.J.; Jang, M.; Kwak, Y.J.; Kochi, A.; Vertuani, S.; Bhunia, A.; Manfredini, S.; Ramamoorthy, A.; Lim, M.H. Reactivity of Metal-Free and Metal-Associated Amyloid-β with Glycosylated Polyphenols and Their Esterified Derivatives. Sci. Rep. 2015, 5, 17842. [Google Scholar] [CrossRef] [PubMed]
- Okada, M.; Mitsuhashi, H.; Wada, H.; Terabayashi, S.; Kondo, K.; Murata, J.; Kikuchi, G. Newly Revised Illustrated Medicinal Plants of World; Hokuryukan Co., Ltd.: Tokyo, Japan, 2002; p. 612. [Google Scholar]
- Zhou, Y.; Yang, B.; Jiang, Y.; Liu, Z.; Liu, Y.; Wang, X.; Kuang, H. Studies on cytotoxic activity against HepG-2 cells of naphthoquinones from green walnut husks of Juglans mandshurica maxim. Molecules 2015, 20, 15572–15588. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.L.; Yu, X.F.; Qu, S.C.; Zhang, R.; Qu, X.R.; Chen, Y.P.; Ma, X.Y.; Sui, D.Y. Anti-proliferative effect of Juglone from Juglans mandshurica Maxim on human leukemia cell HL-60 by inducing apoptosis through the mitochondria-dependent pathway. Eur. J. Pharmacol. 2010, 645, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, N.; Wang, K.C.; Wegiel, J.; Malik, M.N. Walnut extract inhibits the fibrillization of amyloid beta-protein, and also defibrillizes its preformed fibrils. Curr. Alzheimer Res. 2004, 1, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Muthaiyah, B.; Essa, M.M.; Lee, M.; Chauhan, V.; Kaur, K.; Chauhan, A. Dietary supplementation of walnuts improves memory deficits and learning skills in transgenic mouse model of alzheimer’s disease. J. Alzheimer’s Dis. 2014, 42, 1397–1405. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.; Cheng, Z.; Huang, X.; Song, S. Research review in the main chemical constituents and pharmacological effects of Juglans mandshurica Maxim. Asian J. Tradit. Med. 2017, 12, 183–200. [Google Scholar]
- Babula, P.; Adam, V.; Havel, L.; Kizek, R. Noteworthy Secondary Metabolites Naphthoquinones—Their Occurrence, Pharmacological Properties and Analysis. Curr. Pharm. Anal. 2009, 5, 47–68. [Google Scholar] [CrossRef]
- Fang, L.; Ren, D.; Cui, L.; Liu, C.; Wang, J.; Liu, W.; Min, W.; Liu, J. Antifatigue, Antioxidant and Immunoregulatory Effects of Peptides Hydrolyzed from Manchurian Walnut (Juglans mandshurica Maxim.) on Mice. Grain Oil Sci. Technol. 2018, 1, 44–52. [Google Scholar]
- Selamoglu, Z. Polyphenolic compounds in human health with pharmacological properties. J. Tradit. Med. Clin. Natur. 2017, 6, e137. [Google Scholar] [CrossRef]
- Hu, F.B.; Stampfer, M.J.; Manson, J.E.; Rimm, E.B.; Colditz, G.A.; Rosner, B.A.; Speizer, F.E.; Hennekens, C.H.; Willett, W.C. Frequent nut consumption and risk of coronary heart disease in women: Prospective cohort study. BMJ 1998, 317, 1341–1345. [Google Scholar] [CrossRef]
- Liao, C.R.; Kuo, Y.H.; Ho, Y.L.; Wang, C.Y.; Yang, C.S.; Lin, C.W.; Chang, Y.S. Studies on cytotoxic constituents from the leaves of elaeagnus oldhamii maxim. In non-small cell lung cancer A549 cells. Molecules 2014, 19, 9515–9534. [Google Scholar] [CrossRef] [PubMed]
- Colaric, M.; Veberic, R.; Solar, A.; Hudina, M.; Stampar, F. Phenolic acids, syringaldehyde, and juglone in fruits of different cultivars of Juglans regia L. J. Agric. Food Chem. 2005, 53, 6390–6396. [Google Scholar] [CrossRef] [PubMed]
- Shi, A.; Huang, J.-W.; Liu, Y.; Yuan, K. Separation, antioxidant and antimicrobial activities of chemical constituents from exocarp of Juglans mandshurica Maxim. Asian J. Chem. 2013, 25, 3361. [Google Scholar] [CrossRef]
- Sekijima, Y.; Kelly, J.W.; Ikeda, S. Pathogenesis of and therapeutic strategies to ameliorate the transthyretin amyloidoses. Curr. Pharm. Des. 2008, 14, 3219–3230. [Google Scholar] [CrossRef] [PubMed]
- Connors, L.H.; Richardson, A.M.; Theberge, R.; Costello, C.E. Tabulation of transthyretin (TTR) variants as of 1/1/2000. Amyloid 2000, 7, 54–69. [Google Scholar] [CrossRef] [PubMed]
- Westermark, P.; Sletten, K.; Johansson, B.; Cornwell, G.G. Fibril in senile systemic amyloidosis is derived from normal transthyretin. Proc. Natl. Acad. Sci. USA 1990, 87, 2843–2845. [Google Scholar] [CrossRef]
- Cornwell, G.G.; Murdoch, W.L.; Kyle, R.A.; Westermark, P.; Pitkänen, P. Frequency and distribution of senile cardiovascular amyloid. A clinicopathologic correlation. Am. J. Med. 1983, 75, 618–623. [Google Scholar] [CrossRef]
- Ingbar, S.H. Observations concerning the binding of thyroid hormones by human serum prealbumin. J. Clin. Investig. 1963, 42, 143–160. [Google Scholar] [CrossRef]
- Auf’mkolk, M.; Koehrle, J.; Hesch, R.D.; Ingbar, S.H.; Cody, V. Crystal structure of phlorizin and the iodothyronine deiodinase inhibitory activity of phloretin analogues. Biochem. Pharmacol. 1986, 35, 2221–2227. [Google Scholar] [CrossRef]
- Smith, T.J.; Davis, F.B.; Deziel, M.R.; Davis, P.J.; Ramsden, D.B.; Schoenl, M. Retinoic acid inhibition of thyroxine binding to human transthyretin. BBA Gen. Subj. 1994, 1199, 76–80. [Google Scholar] [CrossRef]
- Zanotti, G.; D’acunto, M.R.; Malpeli, G.; Folli, C.; Berni, R. Crystal Structure of the Transthyretin–Retinoic-Acid Complex. Eur. J. Biochem. 1995, 234, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Miroy, G.J.; Lai, Z.; Lashuel, H.A.; Peterson, S.A.; Strang, C.; Kelly, J.W. Inhibiting transthyretin amyloid fibril formation via protein stabilization. Proc. Natl. Acad. Sci. USA 1996, 93, 15051–15056. [Google Scholar] [CrossRef] [PubMed]
- Maia, F.; Almeida, M.D.R.; Gales, L.; Kijjoa, A.; Pinto, M.M.M.; Saraiva, M.J.; Damas, A.M. The binding of xanthone derivatives to transthyretin. Biochem. Pharmacol. 2005, 70, 1861–1869. [Google Scholar] [CrossRef]
- Pullakhandam, R.; Srinivas, P.N.B.S.; Nair, M.K.; Reddy, G.B. Binding and stabilization of transthyretin by curcumin. Arch. Biochem. Biophys. 2009, 485, 115–119. [Google Scholar] [CrossRef]
- Cardoso, I. Doxycycline disrupts transthyretin amyloid: Evidence from studies in a FAP transgenic mice model. FASEB J. 2006, 20, 234–239. [Google Scholar] [CrossRef]
- Yokoyama, T.; Mizuguchi, M. Inhibition of the Amyloidogenesis of Transthyretin by Natural Products and Synthetic Compounds. Biol. Pharm. Bull. 2018, 41, 979–984. [Google Scholar] [CrossRef]
- Ercisli, S.; Esitken, A.; Turkkal, C.; Orhan, E. The allelopathic effects of juglone and walnut leaf extracts on yield, growth, chemical and PNE compositions of strawberry cv. Fern. Plant Soil Environ. 2005, 51, 283–287. [Google Scholar] [CrossRef]
- Ercisli, S.; Turkkal, C. Allelopathic effects of juglone and walnut leaf extracts on growth, fruit yield and plant tissue composition in strawberry cvs. “Camarosa” and “Sweet Charlie.” J. Hortic. Sci. Biotechnol. 2005, 80, 39–42. [Google Scholar] [CrossRef]
- Rietveld, W.J. Allelopathic effects of juglone on germination and growth of several herbaceous and woody species. J. Chem. Ecol. 1983, 9, 295–308. [Google Scholar] [CrossRef] [PubMed]
- Kiran Aithal, B.; Sunil Kumar, M.R.; Nageshwar Rao, B.; Udupa, N.; Satish Rao, B.S. Juglone, a naphthoquinone from walnut, exerts cytotoxic and genotoxic effects against cultured melanoma tumor cells. Cell Biol. Int. 2009, 33, 1039–1049. [Google Scholar] [CrossRef] [PubMed]
- Inbaraj, J.J.; Chignell, C.F. Cytotoxic Action of Juglone and Plumbagin: A Mechanistic Study Using HaCaT Keratinocytes. Chem. Res. Toxicol. 2004, 17, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.B.; Qu, Z.Y.; Zou, X. Juglone-induced apoptosis in human gastric cancer SGC-7901 cells via the mitochondrial pathway. Exp. Toxicol. Pathol. 2011, 63, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Montenegro, R.C.; Araújo, A.J.; Molina, M.T.; Filho, J.D.B.M.; Rocha, D.D.; Lopéz-Montero, E.; Goulart, M.O.F.; Bento, E.S.; Alves, A.P.N.N.; Pessoa, C.; et al. Cytotoxic activity of naphthoquinones with special emphasis on juglone and its 5-O-methyl derivative. Chem. Biol. Interact. 2010, 184, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Barathi, S.; Shailima Vardhini, R.D.; Chitra, P.; Indra Arulselvi, P. Cytotoxic effect of juglone on human peripheral blood lymphocytes. Asian J. Pharm. Clin. Res. 2013, 6, 178–186. [Google Scholar]
- Devkota, H.P.; Joshi, K.R.; Malla, K.J.; Watanabe, T.; Yahara, S. Phenolic Compounds from the Leaves and Twigs of Heynea trijuga. Shoyakugaku Zasshi (Jpn. J. Pharmacogn.) 2014, 68, 97–98. [Google Scholar]
- Devkota, H.P.; Adhikari, B.; Watanabe, T.; Yahara, S. Nonvolatile Chemical Constituents from the Leaves of Ligusticopsis wallichiana (DC.) Pimenov & Kljuykov and Their Free Radical-Scavenging Activity. J. Anal. Methods Chem. 2018, 2018, 1794650. [Google Scholar] [PubMed]
- Miyata, M.; Sato, T.; Mizuguchi, M.; Nakamura, T.; Ikemizu, S.; Nabeshima, Y.; Susuki, S.; Suwa, Y.; Morioka, H.; Ando, Y.; et al. Role of the glutamic acid 54 residue in transthyretin stability and thyroxine binding. Biochemistry 2010, 49, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Miyata, M.; Sato, T.; Kugimiya, M.; Sho, M.; Nakamura, T.; Ikemizu, S.; Chirifu, M.; Mizuguchi, M.; Nabeshima, Y.; Suwa, Y. The crystal structure of the green tea polyphenol (−)-epigallocatechin gallate—Transthyretin complex reveals a novel binding site distinct from the thyroxine binding site. Biochemistry 2010, 49, 6104–6114. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the extracts are available from the authors upon request. |


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaudhary, N.; Sasaki, R.; Shuto, T.; Watanabe, M.; Kawahara, T.; Suico, M.A.; Yokoyama, T.; Mizuguchi, M.; Kai, H.; Devkota, H.P. Transthyretin Amyloid Fibril Disrupting Activities of Extracts and Fractions from Juglans mandshurica Maxim. var. cordiformis (Makino) Kitam. Molecules 2019, 24, 500. https://doi.org/10.3390/molecules24030500
Chaudhary N, Sasaki R, Shuto T, Watanabe M, Kawahara T, Suico MA, Yokoyama T, Mizuguchi M, Kai H, Devkota HP. Transthyretin Amyloid Fibril Disrupting Activities of Extracts and Fractions from Juglans mandshurica Maxim. var. cordiformis (Makino) Kitam. Molecules. 2019; 24(3):500. https://doi.org/10.3390/molecules24030500
Chicago/Turabian StyleChaudhary, Niraj, Ryoko Sasaki, Tsuyoshi Shuto, Masato Watanabe, Teppei Kawahara, Mary Ann Suico, Takeshi Yokoyama, Mineyuki Mizuguchi, Hirofumi Kai, and Hari Prasad Devkota. 2019. "Transthyretin Amyloid Fibril Disrupting Activities of Extracts and Fractions from Juglans mandshurica Maxim. var. cordiformis (Makino) Kitam." Molecules 24, no. 3: 500. https://doi.org/10.3390/molecules24030500
APA StyleChaudhary, N., Sasaki, R., Shuto, T., Watanabe, M., Kawahara, T., Suico, M. A., Yokoyama, T., Mizuguchi, M., Kai, H., & Devkota, H. P. (2019). Transthyretin Amyloid Fibril Disrupting Activities of Extracts and Fractions from Juglans mandshurica Maxim. var. cordiformis (Makino) Kitam. Molecules, 24(3), 500. https://doi.org/10.3390/molecules24030500






